Tantalum Suboxide Films with Tunable Composition and Electrical Resistivity Deposited by Reactive Magnetron Sputtering
Abstract
:1. Introduction
2. Materials and Methods
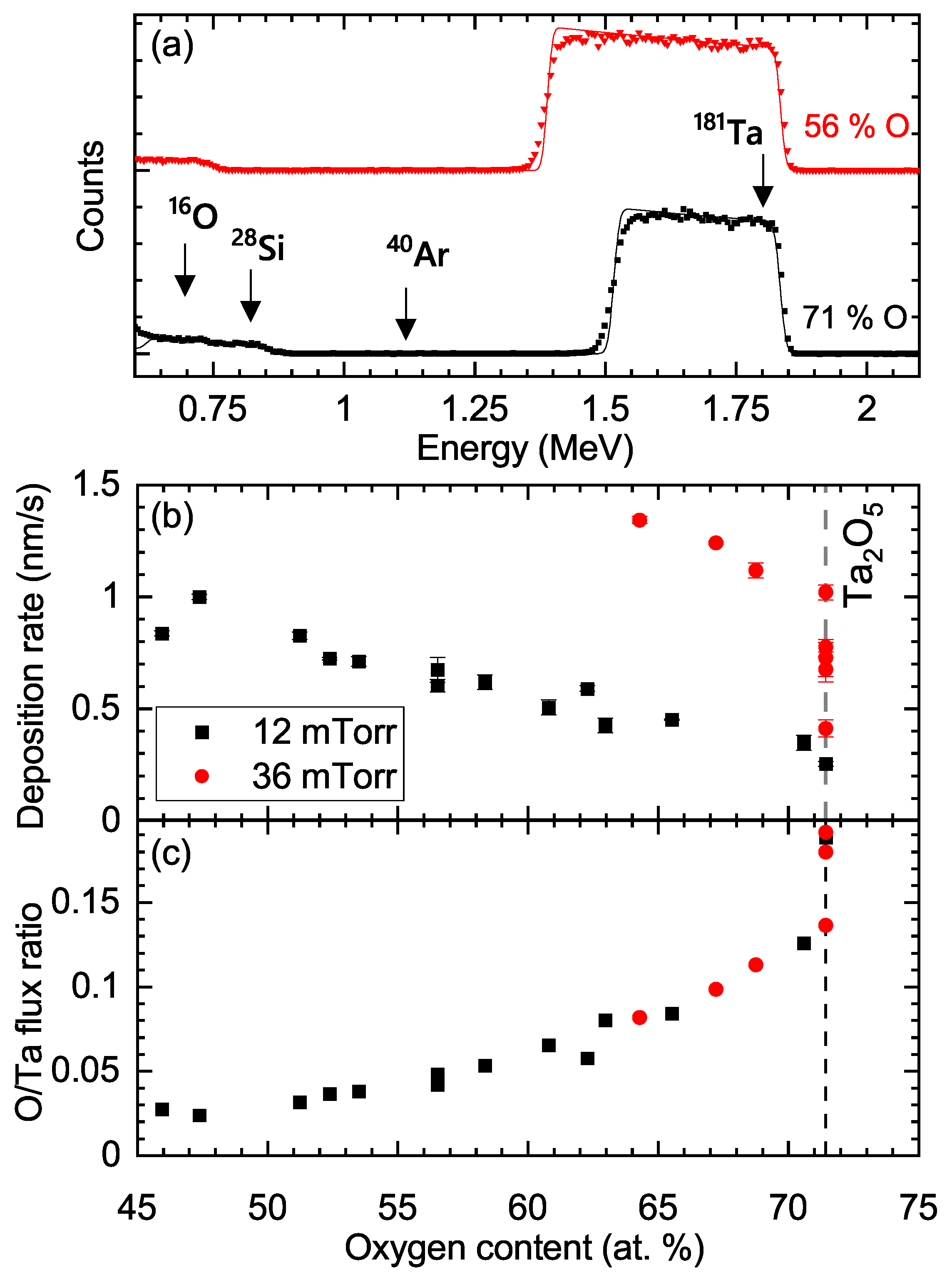
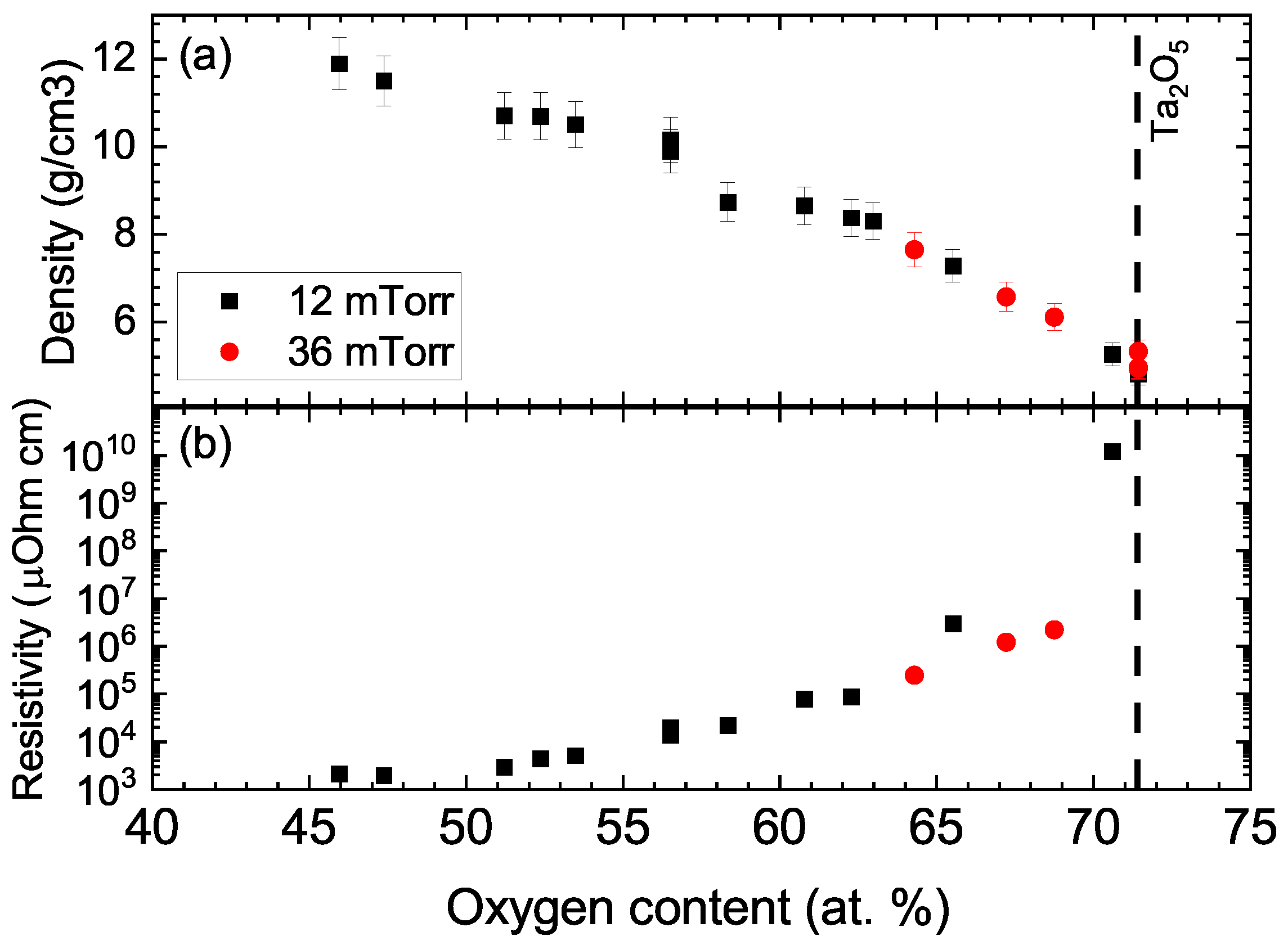
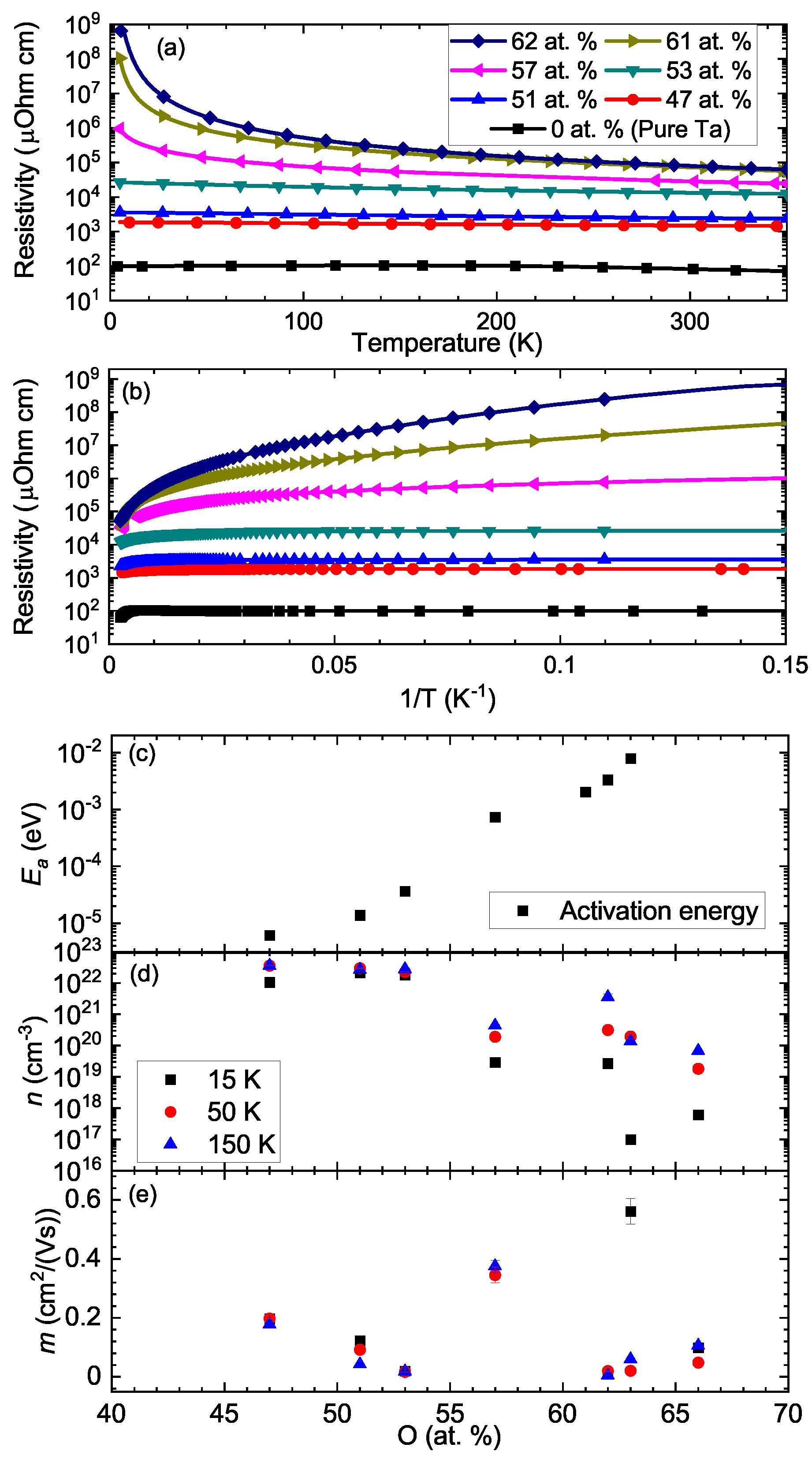
3. Results
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Chaneliere, C.; Autran, J.L.; Devine, R.A.B.; Balland, B. Tantalum pentoxide (Ta2O5) thin films for advanced dielectric applications. Mater. Sci. Eng. R 1998, 22, 269. [Google Scholar] [CrossRef]
- Wilk, G.D.; Wallace, R.M.; Anthony, J.M. High-kappa gate dielectrics: Current status and materials properties considerations. J. Appl. Phys. 2001, 89, 5243. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, J.; Gong, J. Tantalum-based semiconductors for solar water splitting. Chem. Soc. Rev. 2014, 43, 4395–4422. [Google Scholar] [CrossRef]
- Moody, J.D.; Johnson, A.; Javedani, J.; Carroll, E.; Fry, J.; Kozioziemski, B.; Kucheyev, S.O.; Logan, B.G.; Pollock, B.B.; Sio, H.; et al. Transient magnetic field diffusion considerations relevant to magnetically assisted indirect drive inertial confinement fusion. Phys. Plasmas 2020, 27, 112711. [Google Scholar] [CrossRef]
- Aji, L.B.B.; Engwall, A.M.; Bae, J.H.; Baker, A.A.; Beckham, J.L.; Shin, S.J.; Chavez, X.L.; McCall, S.K.; Moody, J.D.; Kucheyev, S.O. Sputtered Au–Ta films with tunable electrical resistivity. J. Phys. D Appl. Phys. 2021, 54, 075303. [Google Scholar]
- Engwall, A.M.; Aji, L.B.B.; Baker, A.A.; Shin, S.J.; Bae, J.H.; McCall, S.K.; Moody, J.D.; Kucheyev, S.O. Effect of substrate tilt on sputter-deposited AuTa films. Appl. Surf. Sci. 2021, 547, 149010. [Google Scholar] [CrossRef]
- Bae, J.H.; Aji, L.B.B.; Shin, S.J.; Engwall, A.M.; Nielsen, M.H.; Baker, A.A.; McCall, S.K.; Moody, J.D.; Kucheyev, S.O. Gold–tantalum alloy films deposited by high-density-plasma magnetron sputtering. J. Appl. Phys. 2021, 130, 165301. [Google Scholar] [CrossRef]
- Rosen, M.D. Fundamentals of ICF Hohlraums. In Lectures in the Scottish Universities Summer School in Physics, 2005, on High Energy Laser Matter Interactions; Jaroszynski, D.A., Bingham, R., Cairns, R.A., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 253–325. [Google Scholar]
- Perkins, L.J.; Ho, D.D.-M.; Logan, B.G.; Zimmerman, G.B.; Rhodes, M.A.; Strozzi, D.J.; Blackfield, D.T.; Hawkins, S.A. The potential of imposed magnetic fields for enhancing ignition probability and fusion energy yield in indirect-drive inertial confinement fusion. Phys. Plasmas 2017, 24, 062708. [Google Scholar] [CrossRef]
- Miura, K.; Osawa, T.; Yokota, Y.; Suzuki, T.; Hanaizumi, O. Fabrication of Tm-doped Ta2O5 thin films using a co-sputtering method. Results Phys. 2014, 4, 148. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Bai, R.; Huang, M. Optical properties of amorphous Ta2O5 thin films deposited by RF magnetron sputtering. Opt. Mater. 2019, 97, 109404. [Google Scholar] [CrossRef]
- Beckage, P.J.; Knorr, D.B.; Wu, X.M.; Lu, T.-M.; Rymaszewski, E.J. Discrete β-Ta2O5 crystallite formation in reactively sputtered amorphous thin films. J. Mat. Sci. 1998, 33, 4375. [Google Scholar] [CrossRef]
- Waterhouse, N.; Wilcox, P.S.; Willmott, D.J. Effect of oxygen on the electrical and structural properties of triode-sputtered tantalum films. J. Appl. Phys. 1971, 42, 5649. [Google Scholar] [CrossRef]
- Waterhouse, N.; Westwood, W.D. Effect of oxygen on the temperature dependence of electrical properties of reactively sputtered tantalum films. Can. J. Phys. 1971, 49, 2250. [Google Scholar] [CrossRef]
- Xu, G.; Shen, X.; Hu, Y.; Ma, P.; Cai, K. Fabrication of tantalum oxide layers onto titanium substrates for improved corrosion resistance and cytocompatibility. Surf. Coat. Tech. 2015, 272, 58. [Google Scholar] [CrossRef]
- Guoping, C.; Lingzhen, L.; Suixin, Z.; Haokang, Z. Structures and properties of a Ta2O5 thin film deposited by dc magnetron reactive sputtering in a pure O2 atmosphere. Vacuum 1990, 41, 1204. [Google Scholar] [CrossRef]
- Felten, F.; Senateur, J.P.; Labeau, M.; Yu-Zhang, K.; Abrutis, A. Deposition of Ta2O5/SiO2 multilayer films by a new process “injection mocvd”. Thin Solid Films 1997, 296, 79. [Google Scholar] [CrossRef]
- Grüger, H.; Kunath, C.; Kurth, E.; Sorge, S.; Pufe, W.; Pechstein, T. High quality rf sputtered metal oxides (Ta2O5, HfO2) and their properties after annealing. Thin Solid Films 2004, 447, 509. [Google Scholar] [CrossRef]
- Paolone, A.; Placidi, E.; Stellino, E.; Betti, M.G.; Majorana, E.; Mariani, C.; Nucara, A.; Palumbo, O.; Postorino, P.; Rago, I.; et al. Effects of the annealing of amorphous Ta2O5 coatings produced by ion beam sputtering concerning the effusion of argon and the chemical composition. J. Non Cryst. Solids 2021, 557, 120651. [Google Scholar] [CrossRef]
- Golosov, D.A. Balanced magnetic field in magnetron sputtering systems. Vacuum 2017, 139, 109. [Google Scholar] [CrossRef]
- Doolittle, L.R. Algorithms for the rapid simulation of rutherford backscattering spectra. Nucl. Instrum. Methods Phys. Res. B 1985, 9, 344. [Google Scholar] [CrossRef]
- Westwood, W.D. Sputter Deposition; AVS Education Committee: Chico, CA, USA, 2003. [Google Scholar]
- Ngaruiya, J.M.; Venkataraj, S.; Drese, R.; Kappertz, O.; Leervad-Pedersen, T.P.; Wuttig, M. Preparation and characterization of tantalum oxide films produced by reactive dc magnetron sputtering. Phys. Status Solidi A 2003, 198, 99. [Google Scholar] [CrossRef]
- Kolasinski, K.W. Surface Science: Foundations of Catalysis and Nanoscience; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Gerstenberg, D.; Calbick, C.J. Effects of nitrogen, methane, and oxygen on structure and electrical properties of thin tantalum films. J. Appl. Phys. 1964, 35, 402. [Google Scholar] [CrossRef]
- Knepper, R.; Stevens, B.; Baker, S.P. Effect of oxygen on the thermomechanical behavior of tantalum thin films during the β–α phase transformation. J. Appl. Phys. 2006, 100, 123508. [Google Scholar] [CrossRef]
- Howson, M.A.; Greig, D. Temperature dependence of conductivity arising from electron-electron interaction effects in amorphous metals. Phys. Rev. B 1984, 30, 4805. [Google Scholar] [CrossRef]
- Krikorian, E.; Sneed, R.J. Deposition of Tantalum, Tantalum Oxide, and Tantalum Nitride with Controlled Electrical Characteristics. J. Appl. Phys. 1966, 37, 3674. [Google Scholar] [CrossRef]
- Li, Y.; Sanna, S.; Norrman, K.; Christensen, D.V.; Pedersen, C.S.; Lastra, J.M.G.; Traulsen, M.L.; Esposito, V.; Pryds, N. Tuning the stoichiometry and electrical properties of tantalum oxide thin films. Appl. Surf. Sci. 2019, 470, 1071. [Google Scholar] [CrossRef]
- Zhou, Y.M.; Xie, Z.; Xiao, H.N.; Hu, P.F.; He, J. Effects of deposition parameters on tantalum films deposited by direct current magnetron sputtering. J. Vac. Sci. Technol. Vacuum Surfaces Films 2009, 27, 109. [Google Scholar] [CrossRef]
- Schwartz, N.; Reed, W.A.; Polash, P.; Read, M.H. Temperature coefficient of resistance of beta-tantalum films and mixtures with b.c.c.-tantalum. Thin Solid Films 1972, 14, 333. [Google Scholar] [CrossRef]
- Neugebauer, C.A.; Webb, M.B. Electrical conduction mechanism in ultrathin, evaporated metal films. J. Appl. Phys. 1962, 33, 74. [Google Scholar] [CrossRef]
- Neugebauer, C.A. Structural disorder phenomena in thin metal films. Phys. Thin Films 1964, 2, 1. [Google Scholar]
- Eo, M.D.; Mollmann, A.; Haddad, J.; Unlu, F.; Kulkarni, A.; Liu, M.; Tachibana, Y.; Stadler, D.; Bhardwaj, A.; Ludwig, T.; et al. Tantalum Oxide as an Efficient Alternative Electron Transporting Layer for Perovskite Solar Cells. Nanomaterials 2022, 12, 780. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baker, A.A.; Engwall, A.M.; Bayu-Aji, L.B.; Bae, J.H.; Shin, S.J.; Moody, J.D.; Kucheyev, S.O. Tantalum Suboxide Films with Tunable Composition and Electrical Resistivity Deposited by Reactive Magnetron Sputtering. Coatings 2022, 12, 917. https://doi.org/10.3390/coatings12070917
Baker AA, Engwall AM, Bayu-Aji LB, Bae JH, Shin SJ, Moody JD, Kucheyev SO. Tantalum Suboxide Films with Tunable Composition and Electrical Resistivity Deposited by Reactive Magnetron Sputtering. Coatings. 2022; 12(7):917. https://doi.org/10.3390/coatings12070917
Chicago/Turabian StyleBaker, Alexander A., Alison M. Engwall, Leonardus Bimo Bayu-Aji, John H. Bae, Swanee J. Shin, John D. Moody, and Sergei O. Kucheyev. 2022. "Tantalum Suboxide Films with Tunable Composition and Electrical Resistivity Deposited by Reactive Magnetron Sputtering" Coatings 12, no. 7: 917. https://doi.org/10.3390/coatings12070917
APA StyleBaker, A. A., Engwall, A. M., Bayu-Aji, L. B., Bae, J. H., Shin, S. J., Moody, J. D., & Kucheyev, S. O. (2022). Tantalum Suboxide Films with Tunable Composition and Electrical Resistivity Deposited by Reactive Magnetron Sputtering. Coatings, 12(7), 917. https://doi.org/10.3390/coatings12070917






