Microstructure and Properties of Hydroxyapatite Coatings Made by Aerosol Cold Spraying–Sintering Technology
Abstract
:1. Introduction
2. Materials and Methods
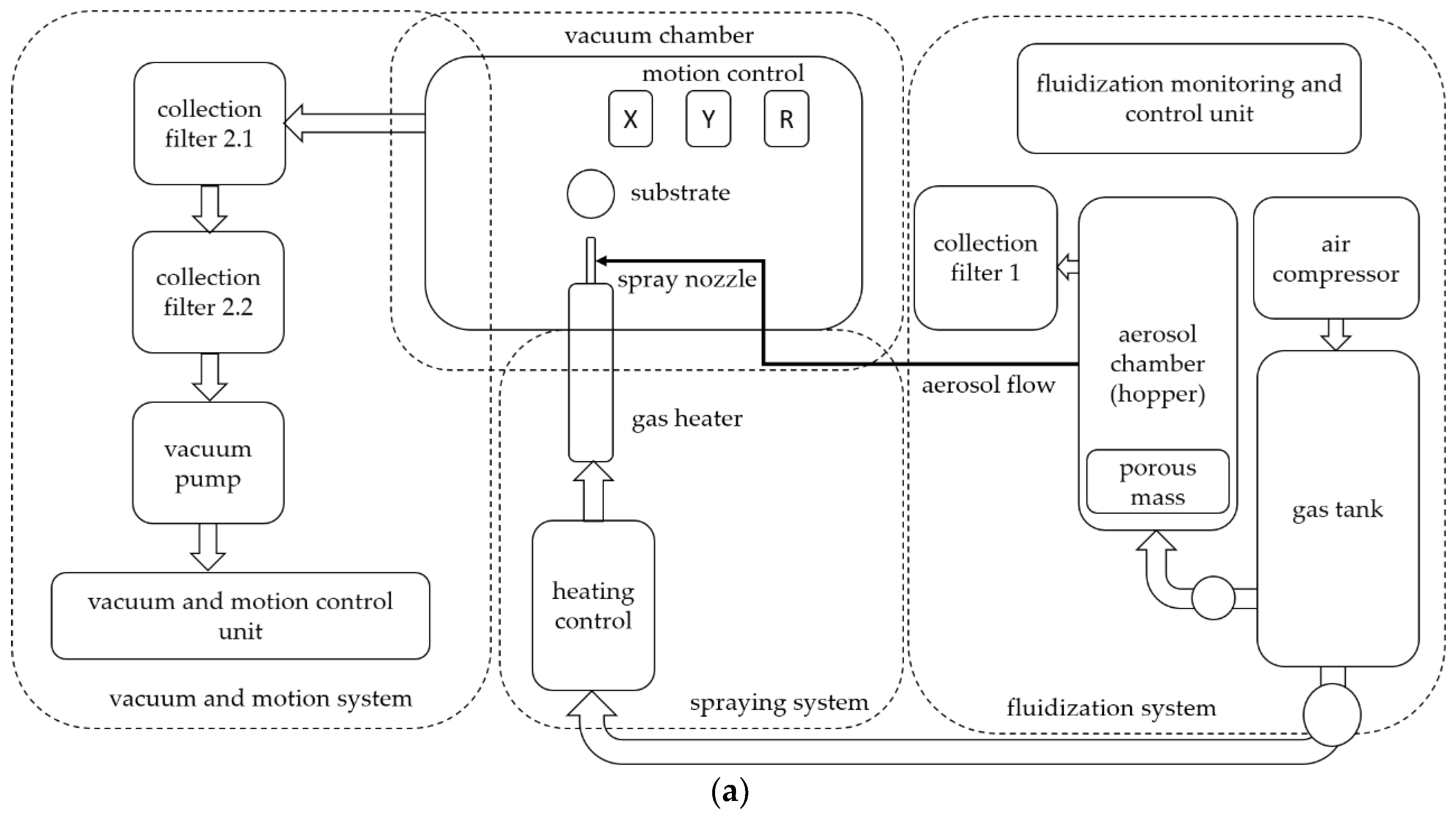
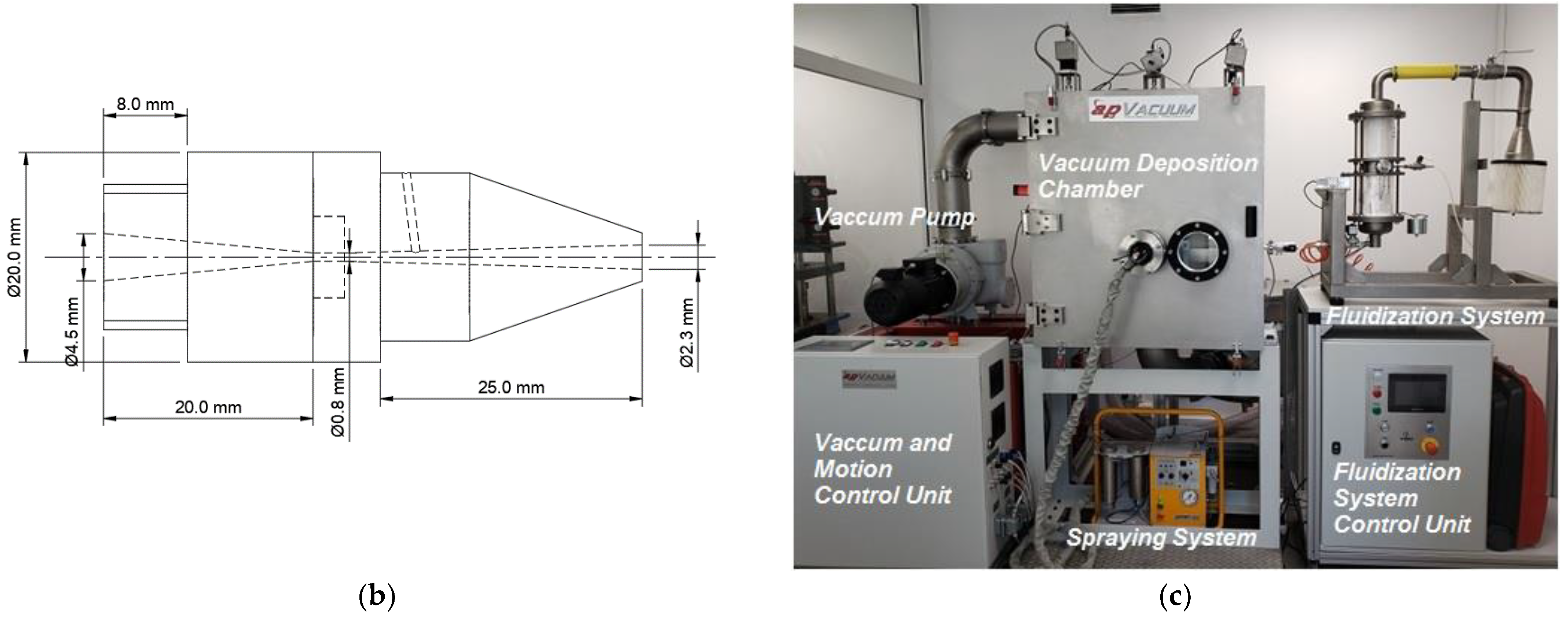
2.1. ACS Apparatus
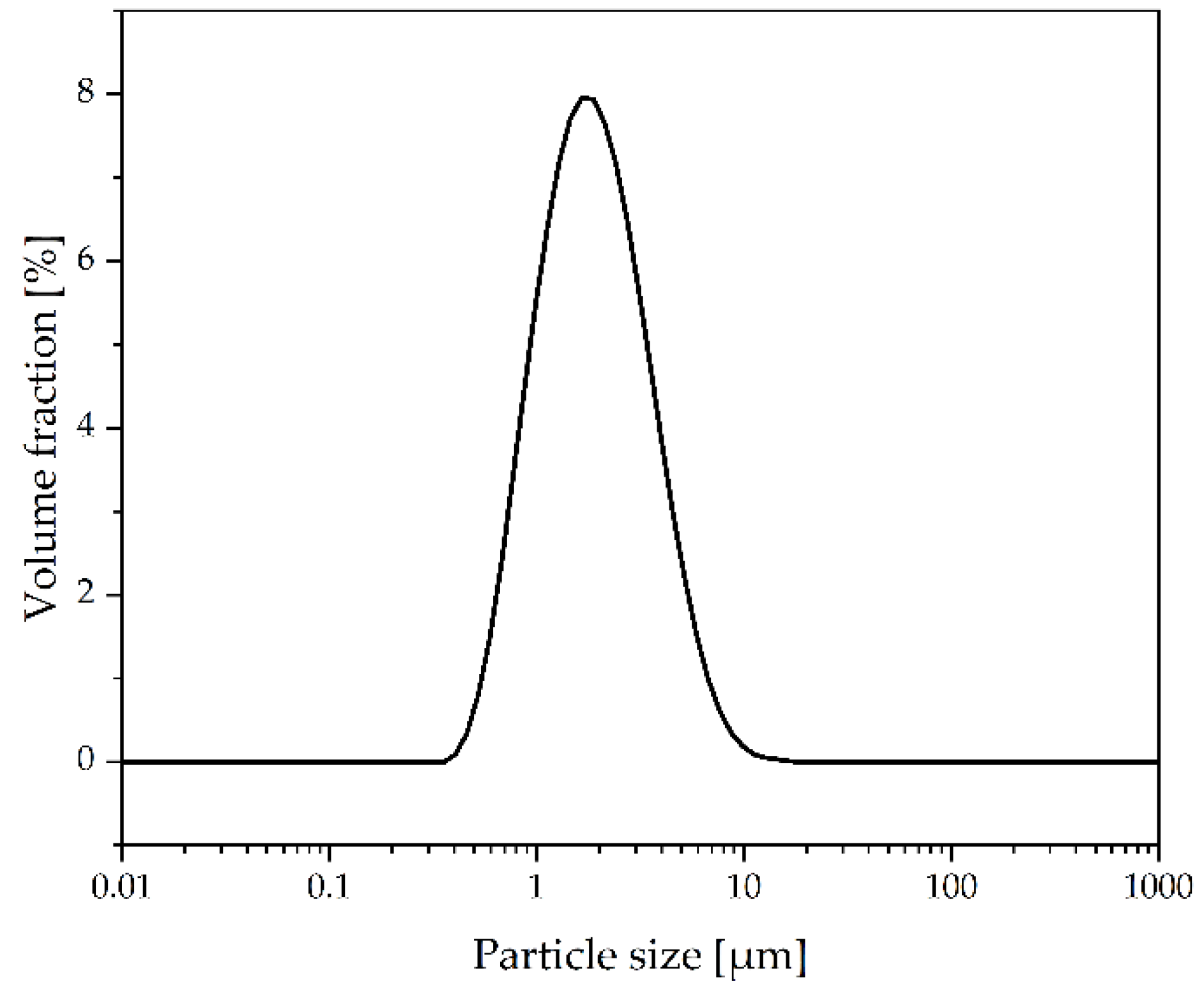
2.2. Materials and Process Parameters
2.3. Coating Sintering
2.4. Coating Structure and Property Characterization
3. Results and Discussion
3.1. Structure and Microstructure Analysis
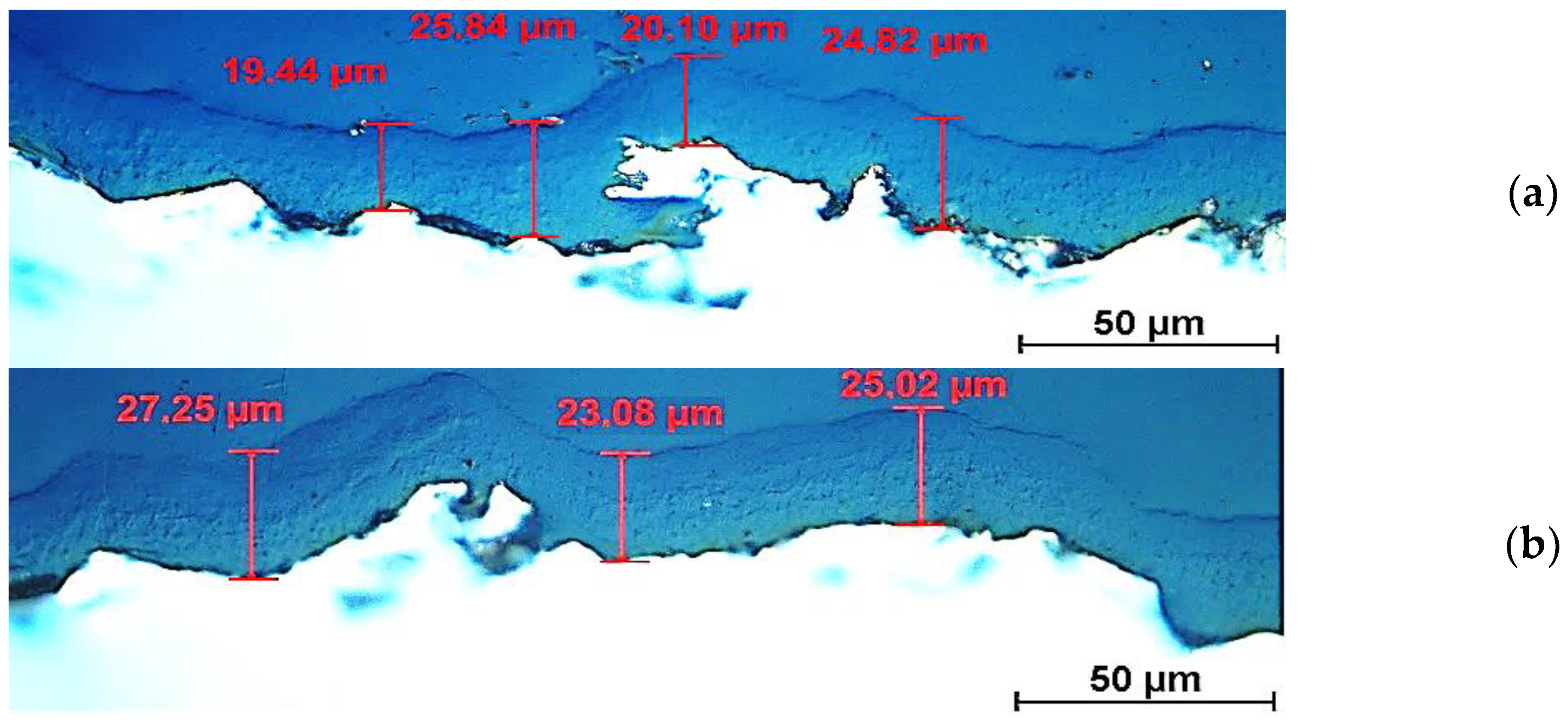
3.1.1. Cross-Sectional Coating Microstructure Observations Using Light Microscopy, Scanning and Transmission Electron Microscopy
- The crystalline structure of HA particles is preserved due to the cold spraying regime of coating structure formation, as seen on the Selected Area Electron Diffraction (SAED) micrograph (Figure 5d), crystal planes correspond to hydroxyapatite phase;
- Particle fragmentation due to high-velocity impact (Figure 5b) resulting in the presence of particle fragments of 10–20 nm size;
- Small grain size of the HA coating both in the subsurface and coating core (Figure 5b);
3.1.2. Effects of Post-Treatment Assessed by Surface Observations by Scanning Electron Microscopy and Profilometry
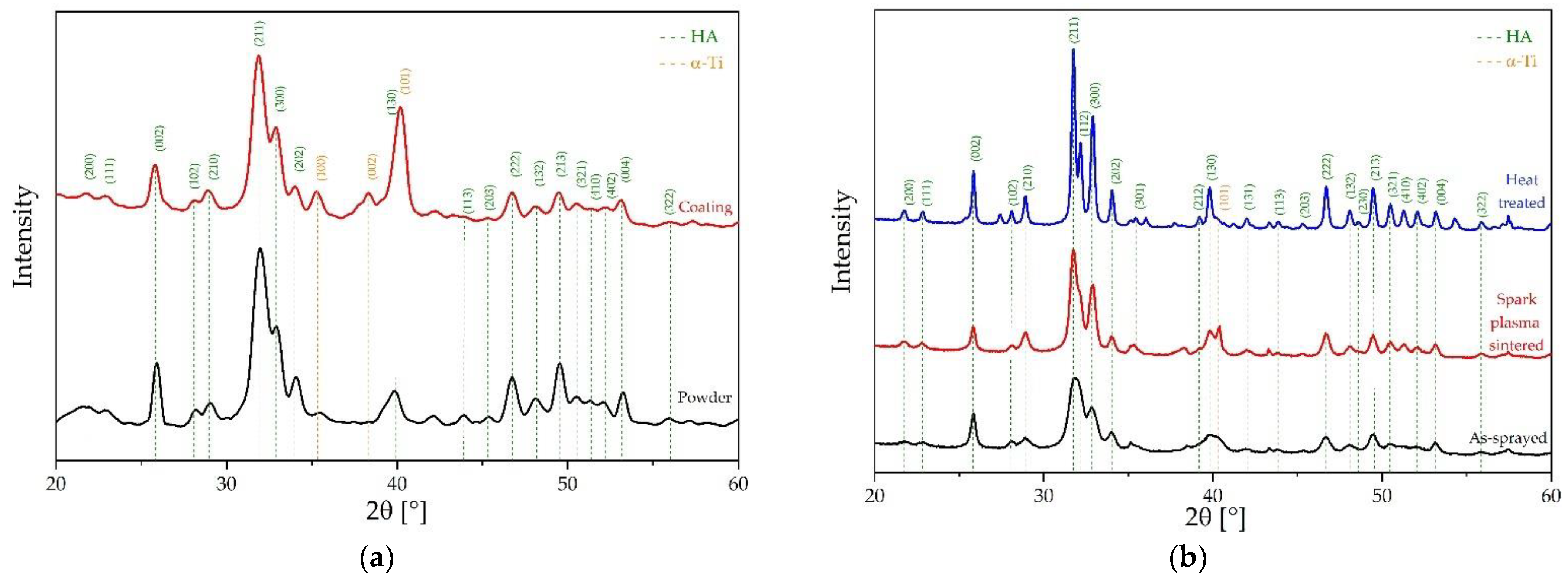
3.1.3. Structural Impact of Coating Process and Post-Treatment Analyzed Using X-ray Diffraction (XRD) Supported by Electron Dispersive Spectroscopy (EDS)
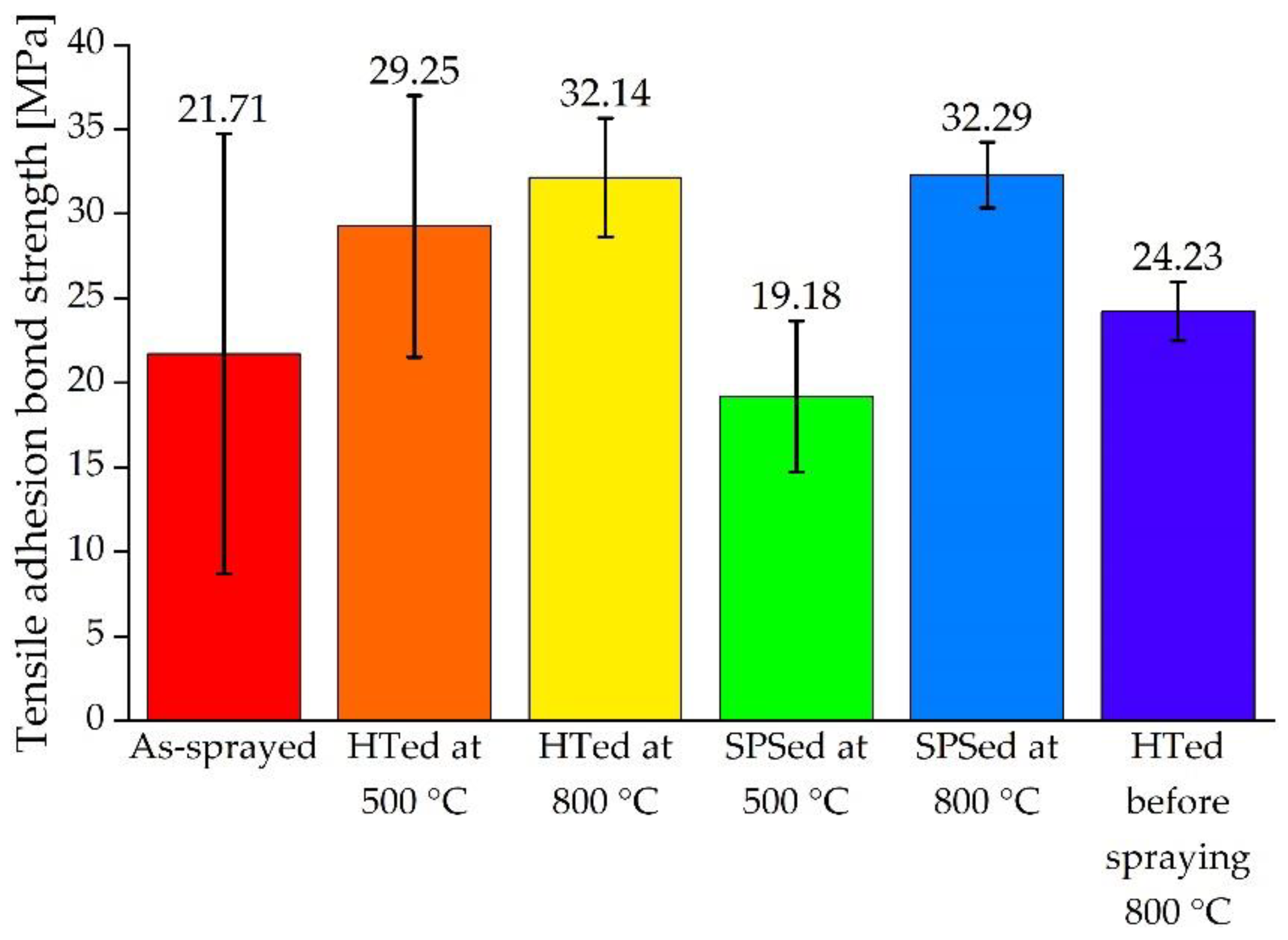
3.2. Adhesion Strength of HA Coatings
4. Conclusions
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Surmenev, R.A.; Surmeneva, M.A. A critical review of decades of research on calcium phosphate–based coatings: How far are we from their widespread clinical application? Curr. Opin. Biomed. Eng. 2019, 10, 35–44. [Google Scholar] [CrossRef]
- Heimann, R.B. Plasma-sprayed hydroxylapatite-based coatings: Chemical, mechanical, microstructural, and biomedical properties. J. Therm. Spray Technol. 2016, 25, 827–850. [Google Scholar] [CrossRef] [Green Version]
- Arcos, D.; Vallet-Regí, M. Substituted hydroxyapatite coatings of bone implants. J. Mater. Chem. B 2020, 8, 1781–1800. [Google Scholar] [CrossRef] [PubMed]
- Jaafar, A.; Hecker, C.; Árki, P.; Joseph, Y. Sol-gel derived hydroxyapatite coatings for titanium implants: A review. Bioengineering 2020, 7, 127. [Google Scholar] [CrossRef] [PubMed]
- Ritwik, A.; Saju, K.K.; Vengellur, A.; Saipriya, P.P. Development of thin-film hydroxyapatite coatings with an intermediate shellac layer produced by dip-coating process on Ti6Al4V implant materials. J. Coat. Technol. Res. 2022, 19, 597–605. [Google Scholar] [CrossRef]
- Beig, B.; Liaqat, U.; Niazi, M.F.K.; Douna, I.; Zahoor, M.; Niazi, M.B.K. Current challenges and innovative developments in hydroxyapatite-based coatings on metallic materials for bone implantation: A review. Coatings 2020, 10, 1249. [Google Scholar] [CrossRef]
- Fadli, A.; Mulya, N.; Pane, K.B.; Pratama, A. Effect of Solid Loading and Dipping Time on Microstructure and Shear Strength of Hydroxyapatite Coatings Deposited via Dip Coating Technique. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Yogyakarta, Indonesia, 4–5 August 2021. [Google Scholar]
- Surmenev, R.A.; Ivanova, A.A.; Epple, M.; Pichugin, V.F.; Surmeneva, M.A. Physical principles of radio-frequency magnetron sputter deposition of calcium-phosphate-based coating with tailored properties. Surf. Coat. Technol. 2021, 413, 127098. [Google Scholar] [CrossRef]
- Liguori, A.; Gualandi, C.; Focarete, M.L.; Biscarini, F.; Bianchi, M. The pulsed electron deposition technique for biomedical applications: A review. Coatings 2019, 10, 16. [Google Scholar] [CrossRef] [Green Version]
- Duta, L. In vivo assessment of synthetic and biological-derived calcium phosphate-based coatings fabricated by pulsed laser deposition: A review. Coatings 2021, 11, 99. [Google Scholar] [CrossRef]
- Safavi, M.S.; Walsh, F.C.; Surmeneva, M.A.; Surmenev, R.A.; Khalil-Allafi, J. Electrodeposited hydroxyapatite-based biocoatings: Recent progress and future challenges. Coatings 2021, 11, 110. [Google Scholar] [CrossRef]
- Taranu, B.-O.; Ianasi, P.; Rus, S.F.; Bucur, A.I. Simultaneous precipitation and electrodeposition of hydroxyapatite coatings at different temperatures on various metal substrates. Coatings 2022, 12, 288. [Google Scholar] [CrossRef]
- He, D.; Zhang, X.; Liu, P.; Liu, X.; Chen, X.; Ma, F.; Li, W.; Zhang, K.; Zhou, H. Effect of hydrothermal treatment temperature on the hydroxyapatite coatings deposited by electrochemical method. Surf. Coat. Technol. 2021, 406, 126656. [Google Scholar] [CrossRef]
- Ghadami, F.; Saber-Samandari, S.; Rouhi, G. The Effects of Substrates’ Materials on Hardness, Creep, and Residual Stress of the Hydroxyapatite Coating, Deposited by HVOF Method. In Proceedings of the 27th National and 5th International Iranian Conference on Biomedical Engineering (ICBME), Tehran, Iran, 26–27 November 2020; pp. 154–158. [Google Scholar]
- Tiwari, S.; Mishra, S.B. Post annealing effect on corrosion behavior, bacterial adhesion, and bioactivity of LVOF sprayed hydroxyapatite coating. Surf. Coat. Technol. 2021, 405, 126500. [Google Scholar] [CrossRef]
- Ratha, I.; Datta, P.; Balla, V.K.; Nandi, S.K.; Kundu, B. Effect of doping in hydroxyapatite as coating material on biomedical implants by plasma spraying method: A review. Ceram. Int. 2021, 47, 4426–4445. [Google Scholar] [CrossRef]
- Vilardell, A.M.; Cinca, N.; Dosta, S.; Cano, I.G.; Guilemany, J.M. Feasibility of using low pressure cold gas spray for the spraying of thick ceramic hydroxyapatite coatings. Int. J. Appl. Ceram. Technol. 2019, 16, 221–229. [Google Scholar] [CrossRef] [Green Version]
- Akedo, J. Aerosol deposition of ceramic thick films at room temperature: Densification mechanism of ceramic layers. J. Am. Ceram. Soc. 2006, 89, 1834–1839. [Google Scholar] [CrossRef]
- Hanft, D.; Exner, J.; Schubert, M.; Stöcker, T.; Fuierer, P.; Moos, R. An overview of the aerosol deposition method: Process fundamentals and new trends in materials applications. J. Ceram. Sci. Technol. 2015, 6, 147–182. [Google Scholar] [CrossRef]
- Cho, M.Y.; Lee, D.W.; Kim, I.S.; Kim, W.J.; Koo, S.M.; Lee, D.; Kim, Y.H.; Oh, J.M. Evaluation of structural and mechanical properties of aerosol-deposited bioceramic films for orthodontic brackets. Ceram. Int. 2019, 45, 6702–6711. [Google Scholar] [CrossRef]
- Yan, C.; Ma, G.; Chen, A.; Chen, Y.; Wu, J.; Wang, W.; Yang, S.; Shi, Y. Additive manufacturing of hydroxyapatite and its composite materials: A review. J. Micromechanics Mol. Phys. 2020, 5. [Google Scholar] [CrossRef]
- Han, Y.-H.; Gao, R.; Bajpai, I.; Kim, B.-N.; Yoshida, H.; Nieto, A.; Son, H.-W.; Yun, J.; Jang, B.-K.; Jhung, S.; et al. Spark plasma sintered bioceramics–from transparent hydroxyapatite to graphene nanocomposites: A review. Adv. Appl. Ceram. 2020, 119, 57–74. [Google Scholar] [CrossRef]
- Anselmi-Tamburini, U. Spark Plasma Sintering. In Encyclopedia of Materials: Technical Ceramics and Glasses; Pomeroy, M., Ed.; Elsevier: Oxford, UK, 2021; pp. 294–310. [Google Scholar]
- Mishra, T.P.; Singh, R.; Mücke, R.; Malzbender, J.; Bram, M.; Guillon, O.; Vaßen, R. Influence of process parameters on the aerosol deposition (AD) of yttria-stabilized zirconia particles. J. Therm. Spray Technol. 2021, 30, 488–502. [Google Scholar] [CrossRef]
- Leshchynsky, V.; Garbiec, D.; Sulej-Chojnacka, J.; Elseddawy, A.; Bajek, A. Aerosol Cold Spray Powder Consolidation of Hydroxyapatite Coatings and its Properties. Presented at the EuroPM 2019, Maastricht, Germany, 13–16 October 2019. [Google Scholar]
- Maev, R.G.; Leshchynsky, V. Cold Gas Dynamic Spray; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Hwang, C.; Yun, J. Flash sintering of hydroxyapatite ceramics. J. Asian Ceram. Soc. 2020, 9, 1–8. [Google Scholar] [CrossRef]
- Champion, E. Sintering of calcium phosphate bioceramics. Acta Biomater. 2012, 9, 5855–5875. [Google Scholar] [CrossRef] [PubMed]
- Exner, J.; Nazarenus, T.; Hanft, D.; Kita, J.; Moos, R. What happens during thermal post-treatment of powder aerosol deposited functional ceramic films? Explanations based on an experiment-enhanced literature survey. Adv. Mater. 2020, 32, 1908104. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Gong, Y.; Chen, X.; Zhang, B.; Zhang, H.; Jin, P.; Li, H. Room-temperature deposition of hydroxyapatite/antibiotic composite coatings by vacuum cold spraying for antibacterial applications. Surf. Coat. Technol. 2017, 330, 87–91. [Google Scholar] [CrossRef]
- Singh, A.; Singh, G.; Chawla, V. Characterization of vacuum plasma sprayed reinforced hydroxyapatite coatings on Ti–6Al–4V alloy. Trans. Indian Inst. Met. 2017, 70, 2609–2628. [Google Scholar] [CrossRef]
- Singh, S.; Prakash, C.; Singh, H. Deposition of HA-TiO2 by plasma spray on β-phase Ti-35Nb-7Ta-5Zr alloy for hip stem: Characterization, mechanical properties, corrosion, and in-vitro bioactivity. Surf. Coat. Technol. 2020, 398, 126072. [Google Scholar] [CrossRef]
- Mohammadi, M.; Tulliani, J.-M.; Montanaro, L.; Palmero, P. Gelcasting and sintering of hydroxyapatite materials: Effect of particle size and Ca/P ratio on microstructural, mechanical and biological properties. J. Eur. Ceram. Soc. 2021, 41, 7301–7310. [Google Scholar] [CrossRef]
- Sun, W.; Tan, A.W.-Y.; Wu, K.; Yin, S.; Yang, X.; Marinescu, I.; Liu, E. Post-process treatments on supersonic cold sprayed coatings: A review. Coatings 2020, 10, 123. [Google Scholar] [CrossRef] [Green Version]
- Azis, S.A.A.; Kennedy, J.; Cao, P. Effect of annealing on microstructure of hydroxyapatite coatings and their behaviours in simulated body fluid. Adv. Mater. Res. 2014, 922, 657–662. [Google Scholar] [CrossRef]
- Ito, K.; Ogawa, K. Effects of spark-plasma sintering treatment on cold-sprayed copper coatings. J. Therm. Spray Technol. 2014, 23, 104–113. [Google Scholar] [CrossRef]



| As-Coated | Pressureless Sintering | SPS | |
|---|---|---|---|
| Sample Number | Roughness Ra µm | Roughness Ra µm | Roughness Ra µm |
| 1 | 7.53 | 3.84 | 3.16 |
| 2 | 7.39 | 3.52 | 2.73 |
| 3 | 8.07 | 3.12 | 3.11 |
| Average | 7.66 | 3.49 | 3.0 |
| Element | Wt.% | At% |
|---|---|---|
| P | 25.21 | 31.68 |
| Ca | 47.94 | 46.55 |
| Ti | 26.02 | 21.14 |
| No. | State of the Coating | Average Adhesion Strength MPa | Adhesion Strength Variation MPa |
|---|---|---|---|
| 1 | As-sprayed | 21.71 | 13.02 |
| 2 | Pressureless sintered at 500 °C | 29.25 | 7.74 |
| 3 | Pressureless sintered at 800 °C | 32.14 | 3.51 |
| 4 | SPSed at 500 °C | 19.18 | 4.47 |
| 5 | SPSed at 800 °C | 32.29 | 1.94 |
| 6 | HTed of Ti alloy substrate at 800 °C before spraying | 24.23 | 1.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubicki, G.; Leshchynsky, V.; Elseddawy, A.; Wiśniewska, M.; Maev, R.G.; Jakubowicz, J.; Sulej-Chojnacka, J. Microstructure and Properties of Hydroxyapatite Coatings Made by Aerosol Cold Spraying–Sintering Technology. Coatings 2022, 12, 535. https://doi.org/10.3390/coatings12040535
Kubicki G, Leshchynsky V, Elseddawy A, Wiśniewska M, Maev RG, Jakubowicz J, Sulej-Chojnacka J. Microstructure and Properties of Hydroxyapatite Coatings Made by Aerosol Cold Spraying–Sintering Technology. Coatings. 2022; 12(4):535. https://doi.org/10.3390/coatings12040535
Chicago/Turabian StyleKubicki, Grzegorz, Volf Leshchynsky, Ahmed Elseddawy, Maria Wiśniewska, Roman G. Maev, Jarosław Jakubowicz, and Joanna Sulej-Chojnacka. 2022. "Microstructure and Properties of Hydroxyapatite Coatings Made by Aerosol Cold Spraying–Sintering Technology" Coatings 12, no. 4: 535. https://doi.org/10.3390/coatings12040535
APA StyleKubicki, G., Leshchynsky, V., Elseddawy, A., Wiśniewska, M., Maev, R. G., Jakubowicz, J., & Sulej-Chojnacka, J. (2022). Microstructure and Properties of Hydroxyapatite Coatings Made by Aerosol Cold Spraying–Sintering Technology. Coatings, 12(4), 535. https://doi.org/10.3390/coatings12040535







