Streptomyces-Derived Bioactive Pigments: Ecofriendly Source of Bioactive Compounds
Abstract
:1. Introduction
2. Results
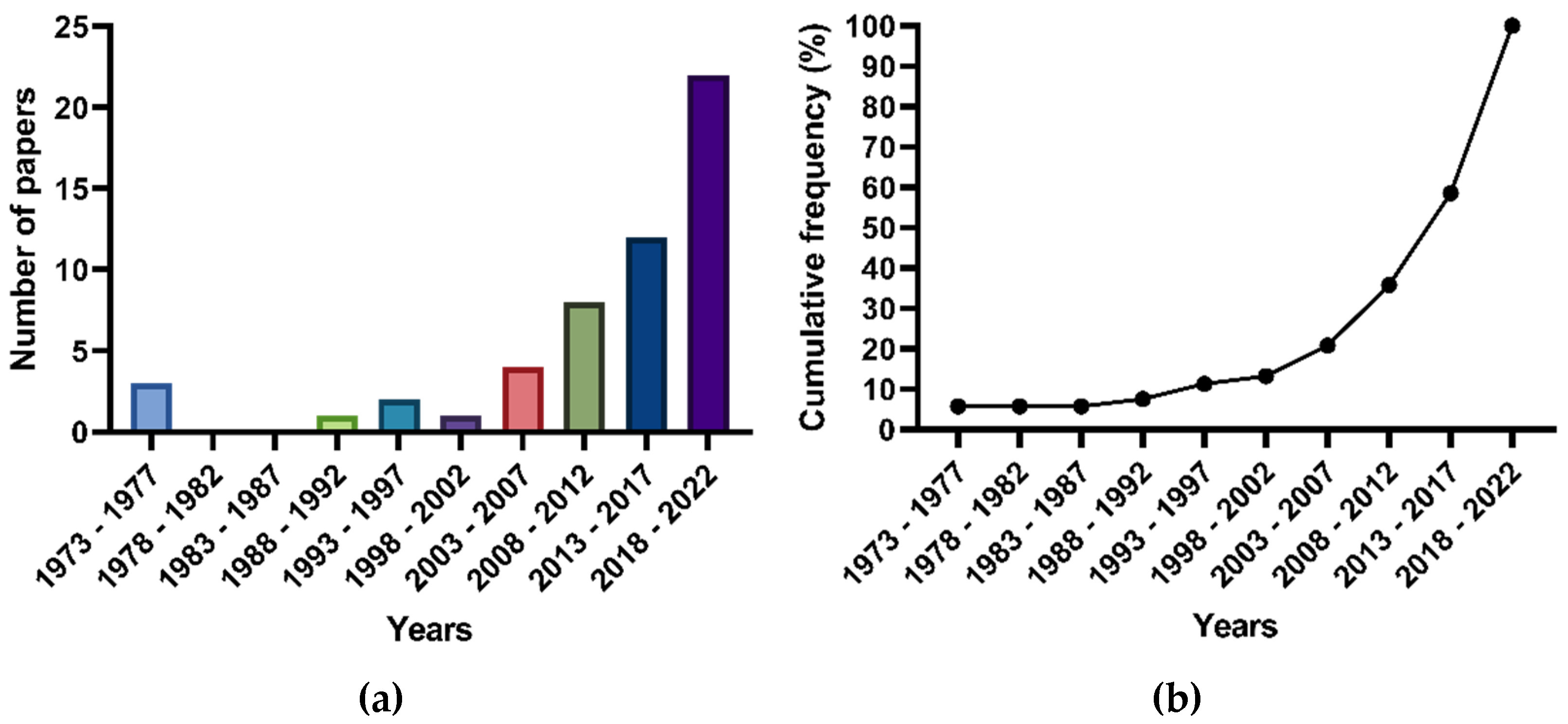
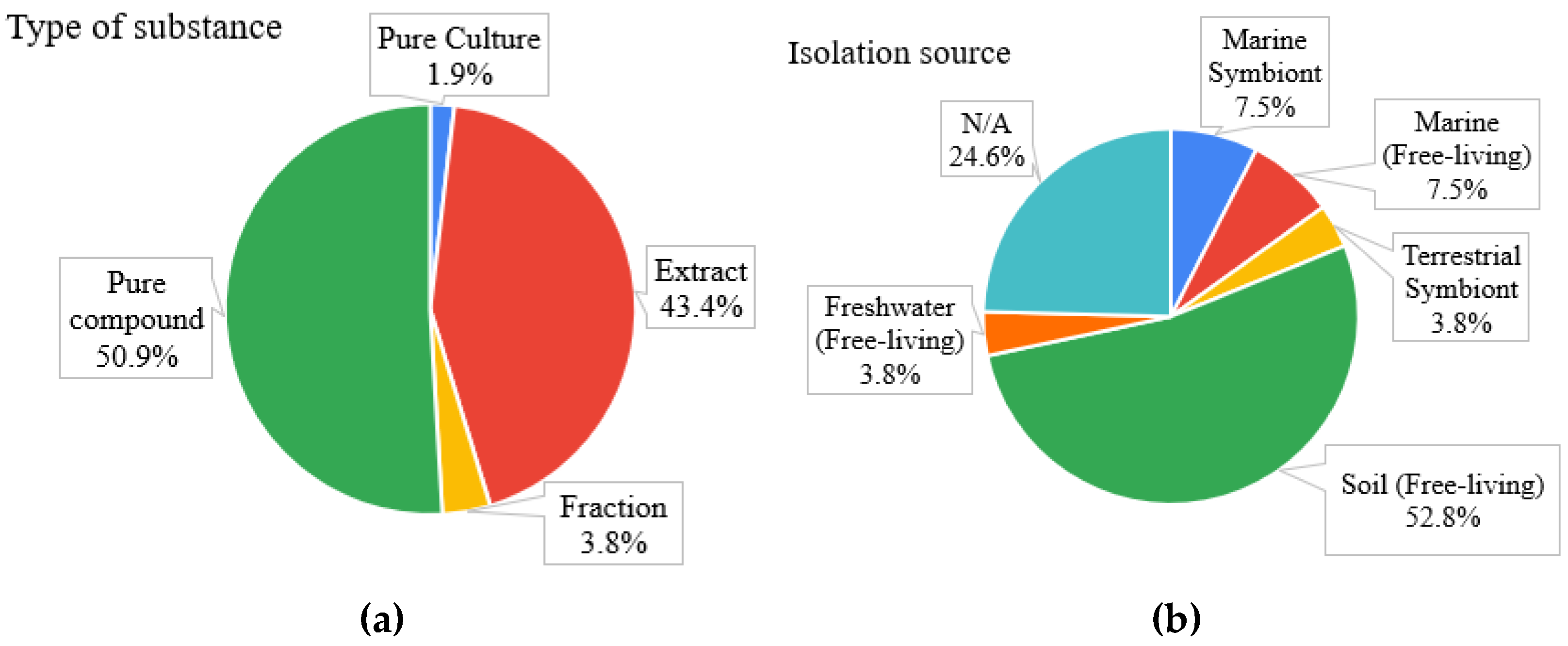
2.1. General Findings
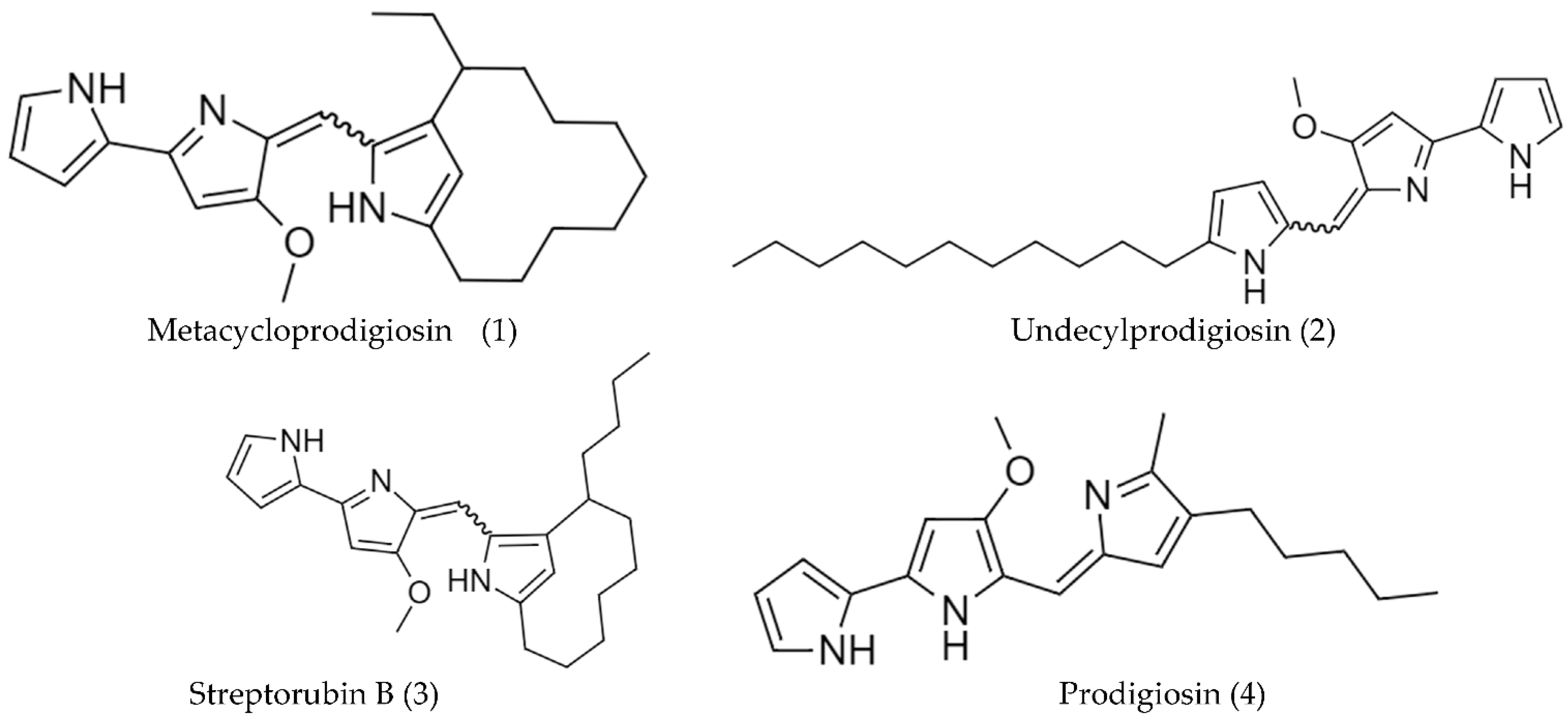
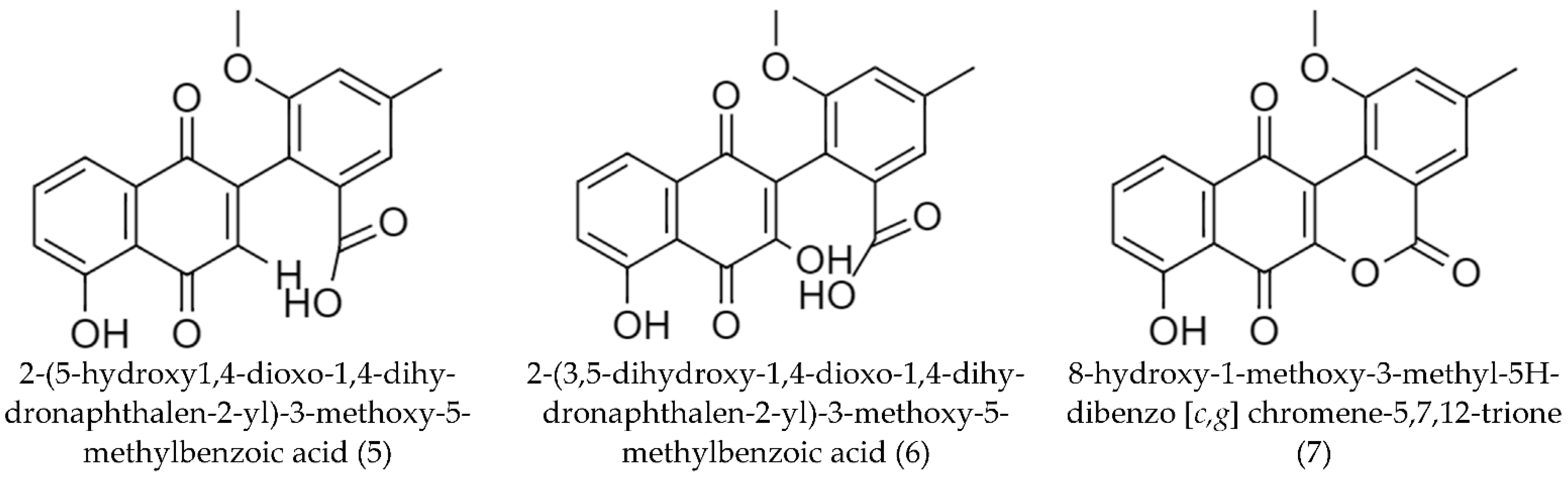
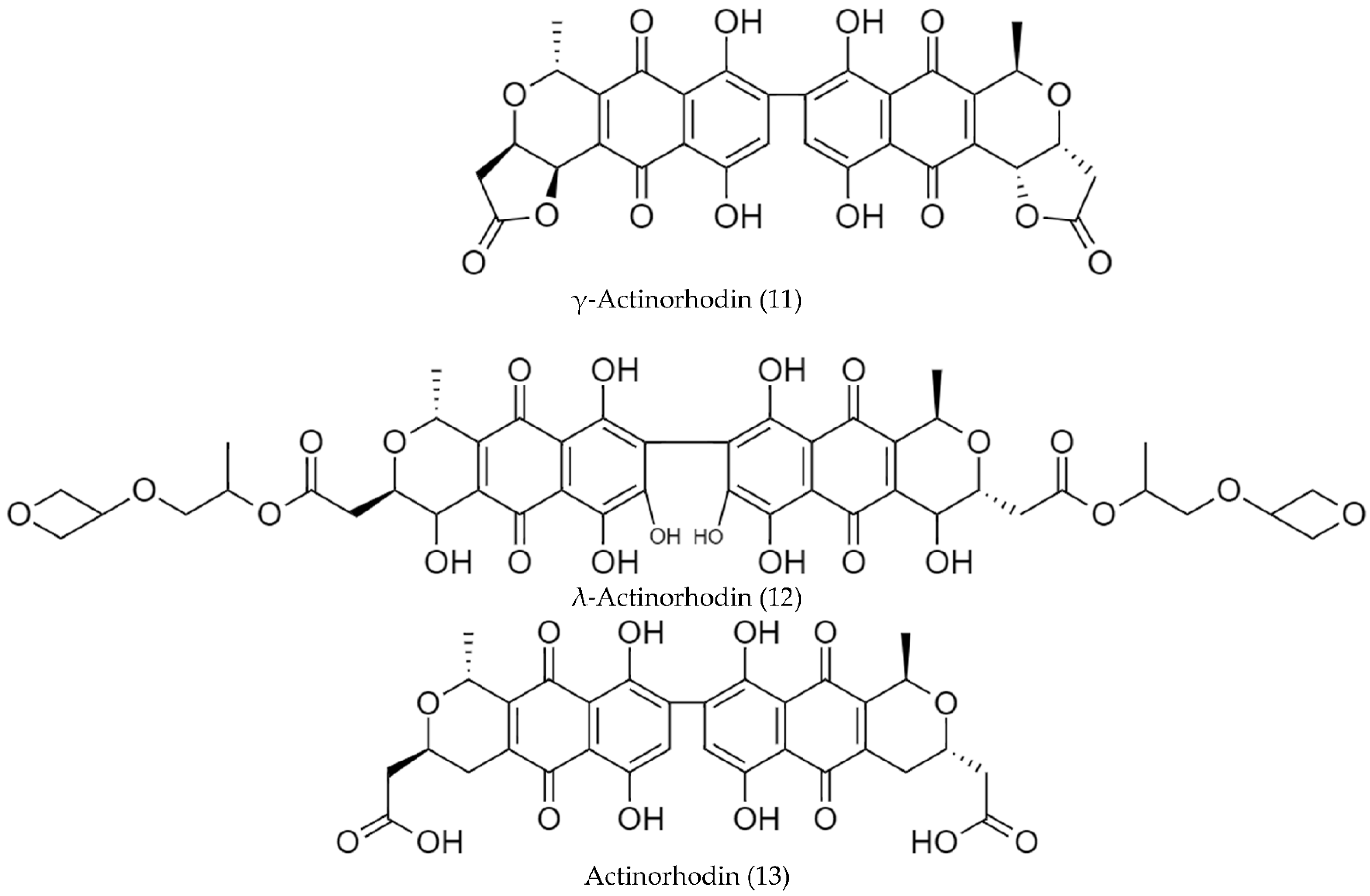
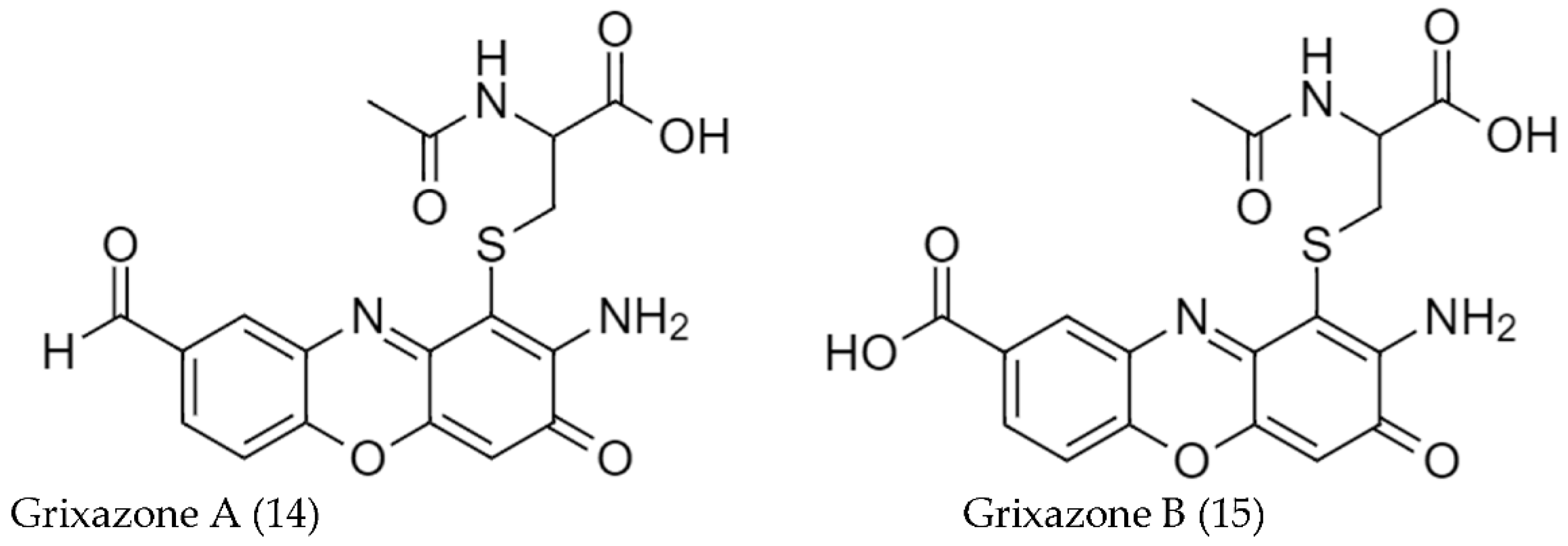
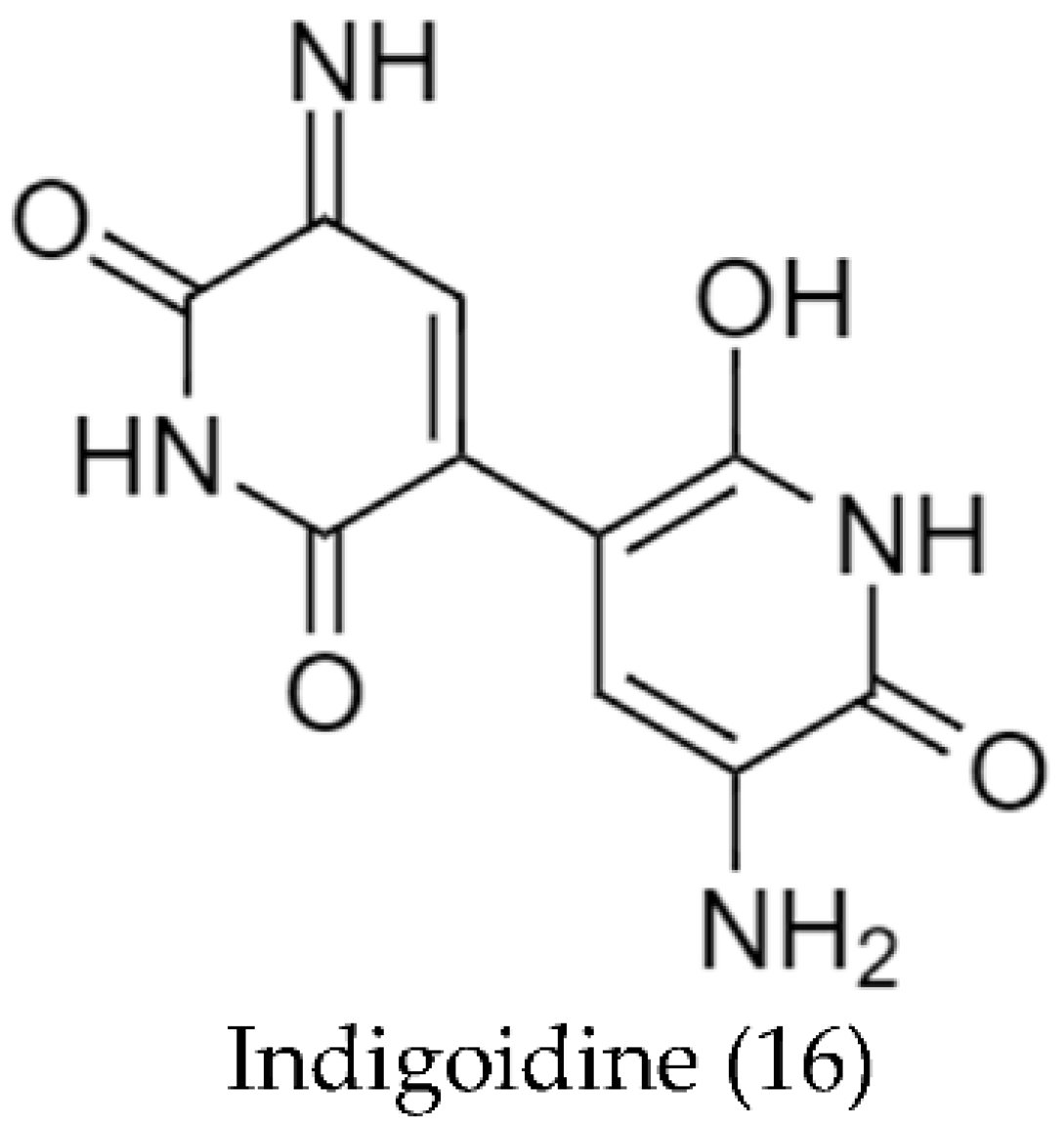
2.2. Biosynthetic Pathways and Structure of Streptomyces Pigments
2.3. Pigment Purification
2.4. Stability Tests of the Pigments
2.5. Optimization of the Pigment’s Production
| Streptomyces Strain | Type/Color of Pigment | Yield Reported (mg/L) | Optimized Variable | Optimization Result | Ref. |
|---|---|---|---|---|---|
| Streptomyces sp. MVCS13 | Melanin | 239 | Temperature | 50 °C | [66] |
| pH | 7.4 | ||||
| L-Tyrosine | 0.75 g/L | ||||
| Asparagine | 1.5 g/L | ||||
| MgSO4 | 0.25 g/L | ||||
| NaCl | 0.75 g/L | ||||
| FeSO4 | 0.015 g/L | ||||
| Trace salt solution | 1.5 mL/L | ||||
| Streptomyces sp. F1 Streptomyces sp. F2 Streptomyces sp. F3 | Melanin | 21,130 | Carbon Source | Starch 1% w/v | [60] |
| Nitrogen source | Soyabean 0.2% w/v | ||||
| Salinity | 15 ppt | ||||
| Temperature | 35 °C | ||||
| pH | 7 | ||||
| Incubation time | 168 h | ||||
| Cheaper source | Sugarcane waste | ||||
| Streptomyces sp. PM4 | Red pigment | 1874 | Carbon Source | Maltose (4.06 g/L) | [61] |
| Nitrogen source | Peptone (7.34 g/L) | ||||
| Yeast extract (4.34 g/L) | |||||
| Tyrosine (2.89 g/L) | |||||
| Streptomyces sp. AQBWWS1 | Carotenoid | N/A 1 | Carbon Source | Glucose | [71] |
| Xylose | |||||
| Amino acids | Cystine | ||||
| Tryptophan | |||||
| NaCl Concentration | 2.50% | ||||
| Streptomyces sp. D25 | Yellow pigment | 1225 | Carbon Source | Glucose | [62] |
| Fructose | |||||
| Nitrogen source | Malt Extract | ||||
| pH | 7, 9, 11 | ||||
| Temperature | 30 °C, 40 °C | ||||
| NaCl Concentration | 1%–5% | ||||
| Streptomyces sp. S45 | Pinkish-brown pigment | N/A | Carbon Source | Glucose | [63] |
| Rhamnose | |||||
| Nitrogen source | Soybean meal | ||||
| Minerals | CaCl2 | ||||
| pH | 7 | ||||
| Temperature | 30 °C | ||||
| Streptomyces glaucescens NEAE-H | Melanin | 350 | Incubation period | 6 days | [40] |
| Nitrogen source | Protease-peptone (5 g/L) | ||||
| Ferric ammonium citrate (0.5 g/L) | |||||
| Streptomyces sp. ZL- 24 | Melanin | 138 | NiCl2 | 3.05 Mm | [94] |
| FeSO4 | 1.33 g/L | ||||
| Soy peptone | 20.31 g/L | ||||
| pH | 7 | ||||
| Temperature | 30 °C | ||||
| Inoculation size | 3% (v/v) | ||||
| Incubation period | 5 days | ||||
| Streptomyces sp. LS-1 | May be actinorhodin-related compounds. | N/A 1 | Carbon Source | Glucose | [97] |
| Nitrogen source | KNO3 | ||||
| Streptomyces canaries M8 | Carotenoid | N/A | NaCl Concentration | >10% | [105] |
| Streptomyces sp. Ac-1 | Yellow pigment | N/A 2 | Agitation | 100 rpm | [65] |
| NaCl Concentration | 2% | ||||
| pH | 5 | ||||
| Streptomyces sp. Ac-2 | Yellow pigment | N/A 2 | Agitation | Steady state | [65] |
| NaCl Concentration | 4% | ||||
| pH | 9 | ||||
| Streptomyces sp. | Red pigment | N/A | Temperature | 37 °C | [68] |
| pH (Solid media) | 10.5 or 7 | ||||
| pH (Broth culture) | 7 |
2.6. Bioactivity Results
2.6.1. Antimicrobial Activity
2.6.2. Antioxidant Activity
2.6.3. Cytotoxic Activity
2.7. Applications of Streptomyces Pigments
Streptomyces Pigments with Antibiofilm/Antifouling Potential
2.8. Future Perspectives
3. Materials and Methods
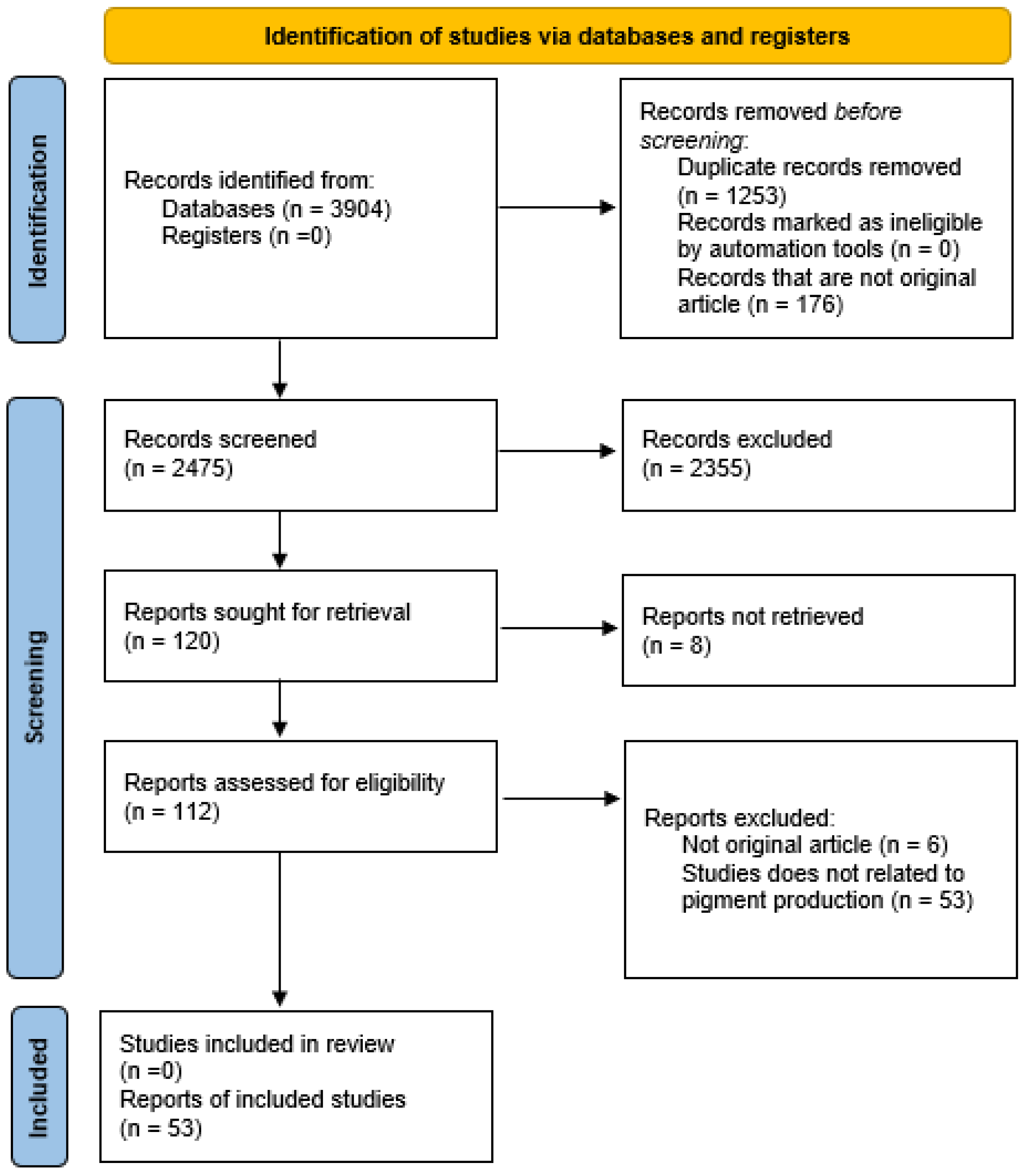
3.1. Databases and Search Strategy
3.2. Selection Procedure
3.3. Data Collection and Tabulation
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Venil, C.K.; Zakaria, Z.A.; Ahmada, W.A. Bacterial Pigments and Their Applications. Process Biochem. 2013, 48, 1065–1079. [Google Scholar] [CrossRef]
- Zollinger, H. Color Chemistry. Synthesis, Properties and Applications of Organic Dyes and Pigments. 3rd Revised Edition. Angew. Chem. Int. Ed. 2004, 43, 5291–5292. [Google Scholar] [CrossRef]
- Lomax, S.Q.; Learner, T. A Review of the Classes, Structures and Methods of Analysis of Synthetic Organic Pigments. J. Am. Inst. Conserv. 2006, 45, 107–125. [Google Scholar] [CrossRef]
- Nicolaou, K.C. Organic Synthesis: The Art and Science of Replicating the Molecules of Living Nature and Creating Others like Them in the Laboratory. Proc. R. Soc. A Math. Phys. Eng. Sci. 2014, 470, 20130690. [Google Scholar] [CrossRef]
- Deraniyagala, H. Textile Colour Waste and Sustainability. In Colour Design; Elsevier: Amsterdam, The Netherlands, 2017; pp. 653–669. [Google Scholar]
- Slama, H.B.; Bouket, A.C.; Pourhassan, Z.; Alenezi, F.N.; Silini, A.; Cherif-Silini, H.; Oszako, T.; Luptakova, L.; Golińska, P.; Belbahri, L. Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods. Appl. Sci. 2021, 11, 6255. [Google Scholar] [CrossRef]
- Soni, I.; Kumar, P.; Sharma, S.; Kudur Jayaprakash, G. A Short Review on Electrochemical Sensing of Commercial Dyes in Real Samples Using Carbon Paste Electrodes. Electrochem 2021, 2, 274–294. [Google Scholar] [CrossRef]
- Jamee, R.; Siddique, R. Biodegradation of Synthetic Dyes of Textile Effluent by Microorganisms: An Environmentally and Economically Sustainable Approach. Eur. J. Microbiol. Immunol. 2019, 9, 114–118. [Google Scholar] [CrossRef]
- Sajda, S. Affat Classifications, Advantages, Disadvantages, Toxicity Effects of Natural and Synthetic Dyes: A Review. Univ. Thi-Qar J. Sci. 2021, 8, 130–135. [Google Scholar]
- Zahra, N. Effect of Natural and Synthetic Dyes on Human Health. Int. Res. J. Biol. Sci. 2017, 6, 23–29. [Google Scholar]
- Sharma, V.; McKone, H.T.; Markow, P.G. A Global Perspective on the History, Use, and Identification of Synthetic Food Dyes. J. Chem. Educ. 2011, 88, 24–28. [Google Scholar] [CrossRef]
- Kobylewski, S.; Jacobson, M.F. Food Dyes A Rainbow of Risks, 1st ed.; Center for Science in the Public Interest: Washington, DC, USA, 2010. [Google Scholar]
- Bakkiyaraj, D.; Pandian, S.T.K. In Vitro and in Vivo Antibiofilm Activity of a Coral Associated Actinomycete against Drug Resistant Staphylococcus aureus Biofilms. Biofouling 2010, 26, 711–717. [Google Scholar] [CrossRef]
- Adeel, S.; Abrar, S.; Kiran, S.; Farooq, T.; Gulzar, T.; Jamal, M. Sustainable Application of Natural Dyes in Cosmetic Industry. In Handbook of Renewable Materials for Coloration and Finishing; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 189–211. [Google Scholar]
- Valle-Vega, P.; Lucas-Florentino, B. Toxicología de Alimentos; Instituto Nacional de Salud Pública, Centro Nacional de Salud Ambiental: Cuernavaca, México, 2000; ISBN 92 75 37004 4. [Google Scholar]
- Kumar, S.; Kumar, V.; Ambika, A.A.A.; Nag, D.; Kumar, V.; Darnal, S.; Thakur, V.; Patial, V.; Singh, D. Microbial Pigments: Learning from Himalayan Perspective to Industrial Applications. J. Ind. Microbiol. Biotechnol. 2022, 49, kuac017. [Google Scholar] [CrossRef]
- Fakruddin, M. Biosurfactant: Production and Application. J. Pet. Environ. Biotechnol. 2012, 3, 124. [Google Scholar] [CrossRef] [Green Version]
- Pailliè-Jiménez, M.E.; Stincone, P.; Brandelli, A. Natural Pigments of Microbial Origin. Front. Sustain. Food Syst. 2020, 4, 590439. [Google Scholar] [CrossRef]
- Sen, T.; Barrow, C.J.; Deshmukh, S.K. Microbial Pigments in the Food Industry—Challenges and the Way Forward. Front. Nutr. 2019, 6, 7. [Google Scholar] [CrossRef] [Green Version]
- Omae, I. General Aspects of Tin-Free Antifouling Paints. Chem. Rev. 2003, 103, 3431–3448. [Google Scholar] [CrossRef]
- Tuli, H.S.; Chaudhary, P.; Beniwal, V.; Sharma, A.K. Microbial Pigments as Natural Color Sources: Current Trends and Future Perspectives. J. Food Sci. Technol. 2015, 52, 4669–4678. [Google Scholar] [CrossRef] [Green Version]
- Malik, K.; Tokkas, J.; Goyal, S. Microbial Pigments: A Review. Int. J. Microb. Resour. Technol. 2012, 1, 361–365. [Google Scholar]
- Narsing Rao, M.P.; Xiao, M.; Li, W. Fungal and Bacterial Pigments: Secondary Metabolites with Wide Applications. Front. Microbiol. 2017, 8, 1113. [Google Scholar] [CrossRef] [Green Version]
- Morales-Oyervides, L.; Oliveira, J.; Sousa-Gallagher, M.; Méndez-Zavala, A.; Montañez, J.C. Assessment of the Dyeing Properties of the Pigments Produced by Talaromyces spp. J. Fungi 2017, 3, 38. [Google Scholar] [CrossRef] [Green Version]
- Paul, T.; Bandyopadhyay, T.K.; Mondal, A.; Tiwari, O.N.; Muthuraj, M.; Bhunia, B. A Comprehensive Review on Recent Trends in Production, Purification, and Applications of Prodigiosin. Biomass Convers. Biorefinery 2020, 12, 1409–1431. [Google Scholar] [CrossRef]
- Nithyanand, P.; Thenmozhi, R.; Rathna, J.; Pandian, S.K. Inhibition of Streptococcus pyogenes Biofilm Formation by Coral-Associated Actinomycetes. Curr. Microbiol. 2010, 60, 454–460. [Google Scholar] [CrossRef]
- Page, K.; Wilson, M.; Parkin, I.P. Antimicrobial Surfaces and Their Potential in Reducing the Role of the Inanimate Environment in the Incidence of Hospital-Acquired Infections. J. Mater. Chem. 2009, 19, 3819–3831. [Google Scholar] [CrossRef]
- Müller, M.M.; Hausmann, R. Regulatory and Metabolic Network of Rhamnolipid Biosynthesis: Traditional and Advanced Engineering towards Biotechnological Production. Appl. Microbiol. Biotechnol. 2011, 91, 251–264. [Google Scholar] [CrossRef]
- Jurić, S.; Jurić, M.; Król-Kilińska, Ż.; Vlahoviček-Kahlina, K.; Vinceković, M.; Dragović-Uzelac, V.; Donsì, F. Sources, Stability, Encapsulation and Application of Natural Pigments in Foods. Food Rev. Int. 2020, 38, 1735–1790. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. Natural Food Pigments and Colorants. Curr. Opin. Food Sci. 2016, 7, 20–26. [Google Scholar] [CrossRef]
- Venil, C.K.; Dufossé, L.; Renuka Devi, P. Bacterial Pigments: Sustainable Compounds With Market Potential for Pharma and Food Industry. Front. Sustain. Food Syst. 2020, 4, 100. [Google Scholar] [CrossRef]
- Jacobson, G.; Wasileski, J. Production of Food Colorants by Fermentation. In Bioprocess Production of Flavor, Fragrance, and Color Ingredients; Gabelman, A., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1997; pp. 205–237. [Google Scholar]
- Kim, M.-J.; Lim, J.-W.; Seo, J.-H.; Jung, H.-K. Hybrid Optimization Strategy Using Response Surface Methodology and Genetic Algorithm for Reducing Cogging Torque of SPM. J. Electr. Eng. Technol. 2011, 6, 202–207. [Google Scholar] [CrossRef] [Green Version]
- Soliev, A.B.; Hosokawa, K.; Enomoto, K. Bioactive Pigments from Marine Bacteria: Applications and Physiological Roles. Evid. -Based Complement. Altern. Med. 2011, 2011, 670349. [Google Scholar] [CrossRef] [Green Version]
- Ramesh, C.; Prasastha, V.R.; Venkatachalam, M.; Dufossé, L. Natural Substrates and Culture Conditions to Produce Pigments from Potential Microbes in Submerged Fermentation. Fermentation 2022, 8, 460. [Google Scholar] [CrossRef]
- Suryawanshi, R.K.; Patil, C.D.; Borase, H.P.; Narkhede, C.P.; Stevenson, A.; Hallsworth, J.E.; Patil, S.V. Towards an Understanding of Bacterial Metabolites Prodigiosin and Violacein and Their Potential for Use in Commercial Sunscreens. Int. J. Cosmet. Sci. 2015, 37, 98–107. [Google Scholar] [CrossRef]
- Choksi, J.; Vora, J.; Shrivastava, N. Bioactive Pigments from Isolated Bacteria and Its Antibacterial, Antioxidant and Sun Protective Application Useful for Cosmetic Products. Indian J. Microbiol. 2020, 60, 379–382. [Google Scholar] [CrossRef]
- Silva, T.R.; Tavares, R.S.N.; Canela-Garayoa, R.; Eras, J.; Rodrigues, M.V.N.; Neri-Numa, I.A.; Pastore, G.M.; Rosa, L.H.; Schultz, J.A.A.; Debonsi, H.M.; et al. Chemical Characterization and Biotechnological Applicability of Pigments Isolated from Antarctic Bacteria. Mar. Biotechnol. 2019, 21, 416–429. [Google Scholar] [CrossRef]
- Tendulkar, P.A.; Surve, M.A.; Deodhar, M.A. Sun Protective Formulation From Carotenoids Extracted From Thermotolerant Genera Synechocystis pevalekii. Int. J. Pharm. Sci. Res. 2018, 9, 3223–3234. [Google Scholar] [CrossRef]
- El-Naggar, N.E.-A.; El-Ewasy, S.M. Bioproduction, Characterization, Anticancer and Antioxidant Activities of Extracellular Melanin Pigment Produced by Newly Isolated Microbial Cell Factories Streptomyces glaucescens NEAE-H. Sci. Rep. 2017, 7, 42129. [Google Scholar] [CrossRef]
- Alihosseini, F.; Ju, K.-S.; Lango, J.; Hammock, B.D.; Sun, G. Antibacterial Colorants: Characterization of Prodiginines and Their Applications on Textile Materials. Biotechnol. Prog. 2008, 24, 742–747. [Google Scholar] [CrossRef] [Green Version]
- Yusof, N.Z. Isolation and Applications of Red Pigments from Serratia marcescens. Ph.D. Thesis, Universiti Teknologi Malaysia, Johor, Malaysia, 2008. [Google Scholar]
- Shirata, A.; Tsukamoto, T.; Yasui, H.; Hata, T.; Hayasaka, S.; Kojima, A.; Kato, H. Isolation of Bacteria Producing Bluish-Purple Pigment and Use for Dyeing. Jpn. Agric. Res. Q 2000, 34, 131–140. [Google Scholar]
- Metwally, R.A.; El Sikaily, A.; El-Sersy, N.A.; Ghozlan, H.A.; Sabry, S.A. Antimicrobial Activity of Textile Fabrics Dyed with Prodigiosin Pigment Extracted from Marine Serratia rubidaea RAM_Alex Bacteria. Egypt. J. Aquat. Res. 2021, 47, 301–305. [Google Scholar] [CrossRef]
- Tran-Ly, A.N.; Reyes, C.; Schwarze, F.; Ribera, J. Microbial Production of Melanin and Its Various Applications. World J. Microbiol. Biotechnol. 2020, 36, 170. [Google Scholar] [CrossRef]
- Dharmaraj, S. Marine Streptomyces as a Novel Source of Bioactive Substances. World J. Microbiol. Biotechnol. 2010, 26, 2123–2139. [Google Scholar] [CrossRef]
- Bull, A.T. (Ed.) Microbial Diversity and Bioprospecting; ASM Press: Washington, DC, USA, 2003; ISBN 9781683672173. [Google Scholar]
- Berdi, J. Bioactive Microbial Metabolites. J. Antibiot. Antibiot. 2005, 58, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Braña, A.F.; Fiedler, H.-P.; Nava, H.; González, V.; Vizcaíno, A.S.; Molina, A.; Acuña, J.L.; García, L.A.; Blanco, G. Two Streptomyces Species Producing Antibiotic, Antitumor, and Anti-Inflammatory Compounds Are Widespread among Intertidal Macroalgae and Deep-Sea Coral Reef Invertebrates from the Central Cantabrian Sea. Microb. Ecol. 2015, 69, 512–524. [Google Scholar] [CrossRef]
- Harir, M.; Bendif, H.; Bellahcene, M.; Fortas, Z.; Pogni, R. Streptomyces Secondary Metabolites. In Basic Biology and Applications of Actinobacteria; IntechOpen: London, UK, 2018; pp. 99–121. [Google Scholar]
- Siddharth, S.; Vittal, R.R. Evaluation of Antimicrobial, Enzyme Inhibitory, Antioxidant and Cytotoxic Activities of Partially Purified Volatile Metabolites of Marine Streptomyces sp.S2A. Microorganisms 2018, 6, 72. [Google Scholar] [CrossRef] [Green Version]
- Azman, A.S.; Mawang, C.I.; Abubakar, S. Bacterial Pigments: The Bioactivities and as an Alternative for Therapeutic Applications. Nat. Prod. Commun. 2018, 13, 1747–1754. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Grand View Research. Dyes & Pigments Market Size, Share & Trends Analysis Report by Product (Dyes, Pigments), by Application, by Regions, and Segment Forecasts, 2022–2030; Grand View Research: San Francisco, CA, USA, 2020. [Google Scholar]
- Manivasagan, P.; Venkatesan, J.; Sivakumar, K.; Kim, S.-K. Actinobacterial Melanins: Current Status and Perspective for the Future. World J. Microbiol. Biotechnol. 2013, 29, 1737–1750. [Google Scholar] [CrossRef]
- Pathom-aree, W.; Stach, J.E.M.; Ward, A.C.; Horikoshi, K.; Bull, A.T.; Goodfellow, M. Diversity of Actinomycetes Isolated from Challenger Deep Sediment (10,898 m) from the Mariana Trench. Extremophiles 2006, 10, 181–189. [Google Scholar] [CrossRef]
- Been Hen, N.; Ken-ichiro, S.; Hai Meng, T. Microbial Diversity in Asia Technology and Prospects; World Scientific Publishing Company: Toh Tuck Link, Singapore, 2001; ISBN 9810243081. [Google Scholar]
- UN Environment Programme World Conservation Monitoring Centre. The State of Biodiversity in Asia and the Pacific: A Mid-Term Review of Progress towards the Aichi Biodiversity Targets; UNEP-WCMC: Cambridge, UK, 2016; ISBN 9789280735116. [Google Scholar]
- Gautham, S.A.; Shobha, K.S.; Onkarappa, R.; Prashith, K.T.R. Isolation, Characterisation and Antimicrobial Potential of Streptomyces Species from Western Ghats of Karnataka, India. Res. J. Pharm. Technol. 2012, 5, 233–238. [Google Scholar]
- Vasanthabharathi, V.; Lakshminarayanan, R.; Jayalakshmi, S. Melanin Production from Marine Streptomyces. Afr. J. Biotechnol. 2011, 10, 11224–11234. [Google Scholar] [CrossRef] [Green Version]
- Karuppiah, V.; Aarthi, C.; Ramohan; Sivakumar, K.; Kannan, L. Statistical Optimization and Anticancer Activity of a Red Pigment Isolated from Streptomyces sp. PM4. Asian Pac. J. Trop. Biomed. 2013, 3, 650–656. [Google Scholar] [CrossRef] [Green Version]
- Manikkam, R.; Venugopal, G.; Ramasamy, B.; Kumar, V. Effect of Critical Medium Components and Culture Conditions on Antitubercular Pigment Production from Novel Streptomyces sp. D25 Isolated from Thar Desert, Rajasthan. J. Appl. Pharm. Sci. 2015, 5, 15–19. [Google Scholar] [CrossRef] [Green Version]
- Vaishnavi, M.; Manigundan, K.; Smalia, T.; Nandhini, S.U.; Gopikrishnan, V.; Kumar, A.; Hanna, L.E.; Radhakrishnan, M.; Aruni, W. Antibacterial and Anti-HIV Activity of Extracellular Pigment from Streptomyces Sp. S45 Isolated from Sabarimala Forest Soil, India. Indian J. Exp. Biol. 2020, 58, 861–868. [Google Scholar] [CrossRef]
- Gerber, N.N.; Lechevalier, M.P. Prodiginine (Prodigiosin-like) Pigments from Streptomyces and Other Aerobic Actinomycetes. Can. J. Microbiol. 1976, 22, 658–667. [Google Scholar] [CrossRef]
- Naligama, K.; Weerasinghe, K.; Halmillawewa, A. Characterization of Bioactive Actinomycetes Isolated from Kadolkele Mangrove Sediments, Sri Lanka. Pol. J. Microbiol. 2022, 71, 191–204. [Google Scholar] [CrossRef]
- Sivaperumal, P.; Kamala, K.; Rajaram, R.; Mishra, S.S. Melanin from Marine Streptomyces sp. (MVCS13) with Potential Effect against Ornamental Fish Pathogens of Carassius Auratus (Linnaeus, 1758). Biocatal. Agric. Biotechnol. 2014, 3, 134–141. [Google Scholar] [CrossRef]
- Li, C.; Ji, C.; Tang, B. Purification, Characterisation and Biological Activity of Melanin from Streptomyces sp. FEMS Microbiol. Lett. 2018, 365, fny077. [Google Scholar] [CrossRef] [Green Version]
- Kamble Geetanjali, R.; Gireesh Babu, K.; Hiremath Shivaprakash, V.; Hiremath Murigendra, B. In Vitro Antimicrobial and Anti-Proliferative Activity of Crude Methanolic Extract of Pigment from Streptomycetes spp. on HT-1080 Fibro Sarcoma Cell Line. Res. J. Biotechnol. 2022, 17, 64–69. [Google Scholar] [CrossRef]
- Chen, W.; Ye, K.; Zhu, X.; Zhang, H.; Si, R.; Chen, J.; Chen, Z.; Song, K.; Yu, Z.; Han, B. Actinomycin X2, an Antimicrobial Depsipeptide from Marine-Derived Streptomyces cyaneofuscatus Applied as a Good Natural Dye for Silk Fabric. Mar. Drugs 2021, 20, 16. [Google Scholar] [CrossRef]
- Wibowo, J.T.; Kellermann, M.Y.; Petersen, L.E.; Alfiansah, Y.R.; Lattyak, C.; Schupp, P.J. Characterization of an Insoluble and Soluble Form of Melanin Produced by Streptomyces cavourensis SV 21, a Sea Cucumber Associated Bacterium. Mar. Drugs 2022, 20, 54. [Google Scholar] [CrossRef]
- Dharmaraj, S.; Ashokkumar, B.; Dhevendaran, K. Food-Grade Pigments from Streptomyces Sp. Isolated from the Marine Sponge Callyspongia diffusa. Food Res. Int. 2009, 42, 487–492. [Google Scholar] [CrossRef]
- King, R.R.; Lawrence, C.H.; Calhoun, L.A. Isolation and Identification of Pigments Generated in Vitro by Streptomyces acidiscabies. J. Agric. Food Chem. 1996, 44, 2849–2851. [Google Scholar] [CrossRef]
- Azimi, S.; Baserisalehi, M.; Bahador, N. Evaluation of Antimicrobial Pigment Produced by Streptomyces coeruleorubidus. Nat. Environ. Pollut. Technol. 2014, 13, 641–644. [Google Scholar]
- Soundari, A.P.G.; Mani, V.M.; Bose, V.S.C.; Jabastin, J.; Priyadarisini, V.B. A Preliminary Assessment of Yellow Pigment from Streptomyces parvulus C5-5Y. J. Pure Appl. Microbiol. 2017, 11, 197–203. [Google Scholar] [CrossRef]
- Selvameenal, L.; Radhakrishnan, M.; Balagurunathan, R. Antibiotic Pigment from Desert Soil Actinomycetes; Biological Activity, Purification and Chemical Screening. Indian J. Pharm. Sci. 2009, 71, 499–504. [Google Scholar] [CrossRef] [Green Version]
- Zhu, H.-H.; Guo, J.; Yao, Q.; Yang, S.-Z.; Deng, M.-R.; Le Phuong, T.B.; Hanh, V.T.; Ryan, M.J. Streptomyces vietnamensis sp. Nov., a Streptomycete with Violet Blue Diffusible Pigment Isolated from Soil in Vietnam. Int. J. Syst. Evol. Microbiol. 2007, 57, 1770–1774. [Google Scholar] [CrossRef] [Green Version]
- Meng-xi, L.I.; Hui-bin, H.; Jie-yun, L.; Jing-xiao, C.A.O.; Zhen-wang, Z. Antibacterial Performance of a Streptomyces spectabilis Strain Producing Metacycloprodigiosin. Curr. Microbiol. 2021, 78, 2569–2576. [Google Scholar] [CrossRef]
- Al-Ghamdi, S.A.; Jastaniah, S.D.; Amasha, R.H. Isolation and Screening of Actinomycetes from Umm Jirsan Cave, Saudi Arabia for Their Antibacterial Activity. Biosci. Biotechnol. Res. Commun. 2021, 14, 351–356. [Google Scholar] [CrossRef]
- Manikkam, R.; Ponnuswamy, S.; Joseph, J.; Kumar, V. Antitubercular Activity of the Pigment from Forest Soil Streptomyces sp. SFA5. Bangladesh J. Pharmacol. 2016, 11, 138–140. [Google Scholar] [CrossRef] [Green Version]
- Machushynets, N.V.; Elsayed, S.S.; Du, C.; Siegler, M.A.; de la Cruz, M.; Genilloud, O.; Hankemeier, T.; Van, W.P. Discovery of Actinomycin L, a New Member of the Actinomycin Family of Antibiotics. Sci. Rep. 2022, 12, 2813. [Google Scholar] [CrossRef]
- Singh, N.; Naik, B.; Kumar, V.; Kumar, A.; Kumar, V.; Gupta, S. Actinobacterial Assisted Synthesis of Nanoparticles and Its Iological Activity. J. Microbiol. Biotechnol. Food Sci. 2021, 10, 604–608. [Google Scholar] [CrossRef]
- Zhu, Y.; Shang, X.; Yang, L.; Zheng, S.; Liu, K.; Li, X. Purification, Identification and Properties of a New Blue Pigment Produced from Streptomyces sp. A1013Y. Food Chem. 2020, 308, 125600. [Google Scholar] [CrossRef]
- Kramar, A.D.; Ilic-Tomic, T.R.; Lađarević, J.M.; Nikodinovic-Runic, J.B.; Kostic, M.M. Halochromic Cellulose Textile Obtained via Dyeing with Biocolorant Isolated from Streptomyces sp. Strain NP4. Cellulose 2021, 28, 8771–8784. [Google Scholar] [CrossRef]
- Prashanthi, K.; Suryan, S.; Varalakshmi, K.N. In Vitro Anticancer Property of Yellow Pigment from Streptomyces griseoaurantiacus JUACT 01. Braz. Arch. Biol. Technol. 2015, 58, 869–876. [Google Scholar] [CrossRef]
- Saengkhae, C.; Srivibool, R.; Watanadilok, R.; Enomoto, K. Partially Purified Pigment Extract from Streptomyces A 16-1 Induces Apoptosis of Human Carcinoma of Nasopharynx Cell (KB Cells) via the Mitochondrial and Caspase-3 Pathway. Walailak J. Sci. Technol. 2017, 14, 51–63. [Google Scholar] [CrossRef]
- Zhang, H.; Zhan, J.; Su, K.; Zhang, Y. A Kind of Potential Food Additive Produced by Streptomyces coelicolor: Characteristics of Blue Pigment and Identification of a Novel Compound, λ-Actinorhodin. Food Chem. 2006, 95, 186–192. [Google Scholar] [CrossRef]
- Abdelfattah, M.S.; Toume, K.; Arai, M.A.; Masu, H.; Ishibashi, M. Katorazone, a New Yellow Pigment with a 2-Azaquinone-Phenylhydrazone Structure Produced by Streptomyces sp. IFM 11299. Tetrahedron Lett. 2012, 53, 3346–3348. [Google Scholar] [CrossRef]
- Stankovic, N.; Radulovic, V.; Petkovic, M.; Vuckovic, I.; Jadranin, M.; Vasiljevic, B.; Nikodinovic-Runic, J. Streptomyces sp. JS520 Produces Exceptionally High Quantities of Undecylprodigiosin with Antibacterial, Antioxidative, and UV-Protective Properties. Appl. Microbiol. Biotechnol. 2012, 96, 1217–1231. [Google Scholar] [CrossRef]
- Abraham, J.; Chauhan, R. Profiling of Red Pigment Produced by Streptomyces sp. JAR6 and Its Bioactivity. 3 Biotech 2018, 8, 22. [Google Scholar] [CrossRef]
- Bayram, S.; Dengiz, C.; Gerçek, Y.C.; Cetin, I.; Topcul, M.R. Bioproduction, Structure Elucidation and in Vitro Antiproliferative Effect of Eumelanin Pigment from Streptomyces parvus BSB49. Arch. Microbiol. 2020, 202, 2401–2409. [Google Scholar] [CrossRef]
- Sajjad, W.; Ahmad, S.; Aziz, I.; Azam, S.S.; Hasan, F.; Shah, A.A. Antiproliferative, Antioxidant and Binding Mechanism Analysis of Prodigiosin from Newly Isolated Radio-Resistant Streptomyces sp. Strain WMA-LM31. Mol. Biol. Rep. 2018, 45, 1787–1798. [Google Scholar] [CrossRef]
- Mohanasrinivasan, V.; SriramKalyan, P.; Nandi, I.; Subathradevi, C.; Selvarajan, E.; Suganthi, V.; Naine, J.S. Fermentative Production of Extracellular Pigment from Streptomyces coelicolor MSIS1. Res. J. Biotechnol. 2013, 8, 30–40. [Google Scholar]
- Vijayabharathi, R.; Bruheim, P.; Andreassen, T.; Raja, D.S.; Devi, P.B.; Sathyabama, S.; Priyadarisini, V.B. Assessment of Resistomycin, as an Anticancer Compound Isolated and Characterized from Streptomyces aurantiacus AAA5. J. Microbiol. 2011, 49, 920–926. [Google Scholar] [CrossRef]
- Wang, L.; Li, Y.; Li, Y. Metal Ions Driven Production, Characterization and Bioactivity of Extracellular Melanin from Streptomyces sp. ZL-24. Int. J. Biol. Macromol. 2019, 123, 521–530. [Google Scholar] [CrossRef]
- Novakova, R.; Odnogova, Z.; Kutas, P.; Feckova, L.; Kormanec, J. Identification and Characterization of an Indigoidine-like Gene for a Blue Pigment Biosynthesis in Streptomyces aureofaciens CCM 3239. Folia Microbiol. 2010, 55, 119–125. [Google Scholar] [CrossRef]
- Kramar, A.; Ilic-Tomic, T.; Petkovic, M.; Radulović, N.; Kostic, M.; Jocic, D.; Nikodinovic-Runic, J. Crude Bacterial Extracts of Two New Streptomyces sp. Isolates as Bio-Colorants for Textile Dyeing. World J. Microbiol. Biotechnol. 2014, 30, 2231–2240. [Google Scholar] [CrossRef]
- Lu, L.; Cui, H.L.; Chen, Y.N.; Yuan, S. Isolation and Identification of Streptomyces Sp. and Assay of Its Exocellular Water-Soluble Blue Pigments. Folia Microbiol. 2002, 47, 493–498. [Google Scholar] [CrossRef]
- Ohnishi, Y.; Furusho, Y.; Higashi, T.; Chun, H.K.; Furihata, K.; Sakuda, S.; Horinouchi, S. Structures of Grixazone A and B, A-Factor-Dependent Yellow Pigments Produced under Phosphate Depletion by Streptomyces Griseus. J. Antibiot. 2004, 57, 218–223. [Google Scholar] [CrossRef]
- Ramesh, C.; Vinithkumar, N.V.; Kirubagaran, R.; Venil, C.K.; Dufossé, L. Applications of Prodigiosin Extracted from Marine Red Pigmented Bacteria Zooshikella sp. and Actinomycete Streptomyces sp. Microorganisms 2020, 8, 556. [Google Scholar] [CrossRef]
- Prasad, R.; Sasikala, V.; Vetrivel, K.S.; Dharmalingam, K. A Novel Extracellular Protein of Streptomyces peucetius Binds to Daunorubicin but Does Not Inhibit the Bioactivity of the Antibiotic. Biochem. Biophys. Res. Commun. 2003, 311, 460–464. [Google Scholar] [CrossRef]
- Victor, T.M.M.; Ndlovu, T.M.; Filho, M.; Pessela, B.C.; Bull, S.; Ward, A.C. Production and Evaluation of Two Antibiotics of Streptomyces coelicolor A3(2), Prodigiosin and Actinorhodin under Solid State Fermentation, Using Micro-Porous Culture. Chem. Eng. Process. Process Intensif. 2022, 170, 108685. [Google Scholar] [CrossRef]
- Ahn, S.-Y.; Jang, S.; Sudheer, P.D.V.N.; Choi, K.-Y. Microbial Production of Melanin Pigments from Caffeic Acid and L-Tyrosine Using Streptomyces glaucescens and FCS-ECH-Expressing Escherichia coli. Int. J. Mol. Sci. 2021, 22, 2413. [Google Scholar] [CrossRef]
- Hobbs, G.; Frazer, C.M.; Gardner, D.C.J.; Flett, F.; Oliver, S.G. Pigmented Antibiotic Production by Streptomyces coelicolor A3(2): Kinetics and the Influence of Nutrients. J. Gen. Microbiol. 1990, 136, 2291–2296. [Google Scholar] [CrossRef] [Green Version]
- Bystrykh, L.V.; Fernández-Moreno, M.A.; Herrema, J.K.; Malpartida, F.; Hopwood, D.A.; Dijkhuizen, L. Production of Actinorhodin-Related “Blue Pigments” by Streptomyces coelicolor A3(2). J. Bacteriol. 1996, 178, 2238–2244. [Google Scholar] [CrossRef] [Green Version]
- Sakr, A.A.; Ali, M.F.; Ghaly, M.F.; Abdel-Haliem, M.-S.F. Discoloration of Ancient Egyptian Mural Paintings by Streptomyces Strains and Methods of Its Removal. Int. J. Conserv. Sci. 2012, 3, 249–258. [Google Scholar]
- Wasserman, H.H.; Rodgers, G.C.; Keith, D.D. Undecyclprodigiosin. Tetrahedron 1976, 32, 1851–1854. [Google Scholar] [CrossRef]
- Bayram, S. Production, Purification, and Characterization of Streptomyces sp. Strain MPPS2 Extracellular Pyomelanin Pigment. Arch. Microbiol. 2021, 203, 4419–4426. [Google Scholar] [CrossRef]
- Scribner, H.E.; Tang, T.; Bradley, S.G. Production of a Sporulation Pigment by Streptomyces venezuelae. Appl. Microbiol. 1973, 25, 873–879. [Google Scholar] [CrossRef] [Green Version]
- Finger, M.; Sentek, F.; Hartmann, L.; Palacio-Barrera, A.M.; Schlembach, I.; Rosenbaum, M.A.; Büchs, J. Insights into Streptomyces coelicolor A3(2) Growth and Pigment Formation with High-Throughput Online Monitoring. Eng. Life Sci. 2022, 1–10. [Google Scholar] [CrossRef]
- Bayram, S. A Comparative Characterization Study between Fungal and Bacterial Eumelanin Pigments. Indian J. Microbiol. 2022, 62, 393–400. [Google Scholar] [CrossRef]
- Darshan, N.; Manonmani, H.K. Prodigiosin and Its Potential Applications. J. Food Sci. Technol. 2015, 52, 5393–5407. [Google Scholar] [CrossRef] [Green Version]
- López López, L.I.; Nery Flores, D.S.; Silva Belmares, S.Y.; Sáenz Galindo, A. Naphthoquinones: Biological Properties and Synthesis of Lawsone and Derivatives-a Structured Review. Rev. La Fac. Química Farm. 2014, 21, 248–258. [Google Scholar]
- Richardson, M.; Khosla, C. 1.18-Structure, Function, and Engineering of Bacterial Aromatic Polyketide Synthases. In Comprehensive Natural Products Chemistry; Barton, S.D., Nakanishi, K., Meth-Cohn, O., Eds.; Pergamon: Oxford, UK, 1999; pp. 473–494. ISBN 978-0-08-091283-7. [Google Scholar]
- Mak, S.; Nodwell, J.R. Actinorhodin Is a Redox-Active Antibiotic with a Complex Mode of Action against Gram-Positive Cells. Mol. Microbiol. 2017, 106, 597–613. [Google Scholar] [CrossRef]
- Suzuki, H.; Furusho, Y.; Higashi, T.; Ohnishi, Y.; Horinouchi, S. A Novel O-Aminophenol Oxidase Responsible for Formation of the Phenoxazinone Chromophore of Grixazone*. J. Biol. Chem. 2006, 281, 824–833. [Google Scholar] [CrossRef] [Green Version]
- Wink, J.; Fatemeh, M.; Javad, H. Biology and Biotechnology of Actinobacteria; Wink, J., Mohammadipanah, F., Hamedi, J., Eds.; Springer International Publishing: Cham, Switzerland, 2017; ISBN 978-3-319-60338-4. [Google Scholar]
- Han, Y.; Heijden, R.; Verpoorte, R. Biosynthesis of Anthraquinones in Cell Cultures of the Rubiaceae. Plant Cell Tissue Organ Cult. 2001, 67, 201–220. [Google Scholar] [CrossRef]
- Jakobi, K.; Hertweck, C. A Gene Cluster Encoding Resistomycin Biosynthesis in Streptomyces resistomycificus; Exploring Polyketide Cyclization beyond Linear and Angucyclic Patterns. J. Am. Chem. Soc. 2004, 126, 2298–2299. [Google Scholar] [CrossRef]
- Delgado-Vargas, F.; Jiménez, A.R.; Paredes-López, O.; Francis, F.J. Natural Pigments: Carotenoids, Anthocyanins, and Betalains-Characteristics, Biosynthesis, Processing, and Stability; Taylor & Francis: London, UK, 2000; Volume 40, ISBN 1040869009. [Google Scholar]
- Lorquin, F.; Ziarelli, F.; Amouric, A.; Di Giorgio, C.; Robin, M.; Piccerelle, P.; Lorquin, J. Production and Properties of Non-Cytotoxic Pyomelanin by Laccase and Comparison to Bacterial and Synthetic Pigments. Sci. Rep. 2021, 11, 8538. [Google Scholar] [CrossRef]
- Marques, L.G.; Freire, J.T. Analysis of Freeze-Drying of Tropical Fruits. Dry. Technol. 2005, 23, 2169–2184. [Google Scholar] [CrossRef]
- Silver, L.L. Challenges of Antibacterial Discovery. Clin. Microbiol. Rev. 2011, 24, 71–109. [Google Scholar] [CrossRef] [Green Version]
- Wright, G.D. Solving the Antibiotic Crisis. ACS Infect. Dis. 2015, 1, 80–84. [Google Scholar] [CrossRef]
- Barka, E.A.; Vatsa, P.; Sanchez, L.; Gaveau-Vaillant, N.; Jacquard, C.; Meier-Kolthoff, J.P.; Klenk, H.-P.; Clément, C.; Ouhdouch, Y.; van Wezel, G.P. Taxonomy, Physiology, and Natural Products of Actinobacteria. Microbiol. Mol. Biol. Rev. 2016, 80, 1–43. [Google Scholar] [CrossRef] [Green Version]
- Sakai-Kawada, F.E.; Ip, C.G.; Hagiwara, K.A.; Awaya, J.D. Biosynthesis and Bioactivity of Prodiginine Analogs in Marine Bacteria, Pseudoalteromonas: A Mini Review. Front. Microbiol. 2019, 10, 1715. [Google Scholar] [CrossRef] [Green Version]
- Davies, K.J.A. Oxidative Stress: The Paradox of Aerobic Life. Biochem. Soc. Symp. 1995, 61, 1–31. [Google Scholar] [CrossRef]
- Halliwell, B. Antioxidants in Human Health and Disease. Annu. Rev. Nutr. 1996, 16, 33–50. [Google Scholar] [CrossRef]
- Namiki, M. Antioxidants/Antimutagens in Food. Crit. Rev. Food Sci. Nutr. 1990, 29, 273–300. [Google Scholar] [CrossRef]
- Kim, D.-O.; Lee, K.W.; Lee, H.J.; Lee, C.Y. Vitamin C Equivalent Antioxidant Capacity (VCEAC) of Phenolic Phytochemicals. J. Agric. Food Chem. 2002, 50, 3713–3717. [Google Scholar] [CrossRef]
- Floegel, A.; Kim, D.-O.; Chung, S.-J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH Assays to Measure Antioxidant Capacity in Popular Antioxidant-Rich US Foods. J. Food Compos. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Arnao, M.B. Some Methodological Problems in the Determination of Antioxidant Activity Using Chromogen Radicals: A Practical Case. Trends Food Sci. Technol. 2000, 11, 419–421. [Google Scholar] [CrossRef]
- Cordero, R.J.B.; Vij, R.; Casadevall, A. Microbial Melanins for Radioprotection and Bioremediation. Microb. Biotechnol. 2017, 10, 1186–1190. [Google Scholar] [CrossRef] [Green Version]
- Barros, L.; Ferreira, M.-J.; Queirós, B.; Ferreira, I.C.F.R.; Baptista, P. Total Phenols, Ascorbic Acid, β-Carotene and Lycopene in Portuguese Wild Edible Mushrooms and Their Antioxidant Activities. Food Chem. 2007, 103, 413–419. [Google Scholar] [CrossRef]
- Lin, X.; Wen, Y.; Li, M.; Chen, Z.; Guo, J.; Song, Y.; Li, J. A New Strain of Streptomyces avermitilis Produces High Yield of Oligomycin A with Potent Anti-Tumor Activity on Human Cancer Cell Lines in Vitro. Appl. Microbiol. Biotechnol. 2009, 81, 839–845. [Google Scholar] [CrossRef]
- Aykul, S.; Martinez-Hackert, E. Determination of Half-Maximal Inhibitory Concentration Using Biosensor-Based Protein Interaction Analysis. Anal. Biochem. 2016, 508, 97–103. [Google Scholar] [CrossRef] [Green Version]
- Kazakova, O.B.; Giniyatullina, G.V.; Mustafin, A.G.; Babkov, D.A.; Sokolova, E.V.; Spasov, A.A. Evaluation of Cytotoxicity and A-glucosidase Inhibitory Activity of Amide and Polyamino-derivatives of Lupane Triterpenoids. Molecules 2020, 25, 4833. [Google Scholar] [CrossRef]
- Zhang, M.; Aguilera, D.; Das, C.; Vasquez, H.; Zage, P.; Gopalakrishnan, V.; Wolff, J. Measuring Cytotoxicity: A New Perspective on LC50. Anticancer Res. 2007, 27, 35–38. [Google Scholar]
- Han, Z.; Zhao, X.; Zhang, E.; Ma, J.; Zhang, H.; Li, J.; Xie, W.; Li, X. Resistomycin Induced Apoptosis and Cycle Arrest in Human Hepatocellular Carcinoma Cells by Activating P38 MAPK Pathway In Vitro and In Vivo. Pharmaceuticals 2021, 14, 958. [Google Scholar] [CrossRef]
- Thenmozhi, R.; Nithyanand, P.; Rathna, J.; Karutha Pandian, S. Antibiofilm Activity of Coral-Associated Bacteria against Different Clinical M Serotypes of Streptococcus pyogenes. FEMS Immunol. Med. Microbiol. 2009, 57, 284–294. [Google Scholar] [CrossRef] [Green Version]
- Sauer, K.; Thatcher, E.; Northey, R.; Gutierrez, A.A. Neutral Super-Oxidised Solutions Are Effective in Killing P. aeruginosa Biofilms. Biofouling 2009, 25, 45–54. [Google Scholar] [CrossRef]
- National Research Council (US). Bacterial Biofilms and Biofouling: Translational Research in Marine Biotechnology. In Opportunities for Environmental Applications of Marine Biotechnology: Proceedings of the October 5-6, 1999, Workshop; Board on Biology, National Research Council (US), Ocean Studies Board; National Academies Press (US): Washington, DC, USA, 2000. [Google Scholar]
- Pereira, F.; Almeida, J.R.; Paulino, M.; Grilo, I.R.; Macedo, H.; Cunha, I.; Sobral, R.G.; Vasconcelos, V.; Gaudêncio, S.P. Antifouling Napyradiomycins from Marine-Derived Actinomycetes Streptomyces Aculeolatus. Mar. Drugs 2020, 18, 63. [Google Scholar] [CrossRef] [Green Version]
- Callow, J.A.; Callow, M.E. Trends in the Development of Environmentally Friendly Fouling-Resistant Marine Coatings. Nat. Commun. 2011, 2, 244. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Huang, Y.; Sheng, Y.; Su, P.; Qiu, Y.; Ke, C.; Feng, D. Antifouling Activity towards Mussel by Small-Molecule Compounds from a Strain of Vibrio Alginolyticus Bacterium Associated with Sea Anemone haliplanella sp. J. Microbiol. Biotechnol. 2017, 27, 460–470. [Google Scholar] [CrossRef] [Green Version]
- Gopikrishnan, V.; Radhakrishnan, M.; Shanmugasundaram, T.; Ramakodi, M.P.; Balagurunathan, R. Isolation, Characterization and Identification of Antibiofouling Metabolite from Mangrove Derived Streptomyces sampsonii PM33. Sci. Rep. 2019, 9, 12975. [Google Scholar] [CrossRef]
- Radhakrishnan, M.; Gopikrishnan, V.; Vijayalakshmi, G.; Kumar, V. In Vitro Antioxidant Activity and Antimicrobial Activity against Biofilm Forming Bacteria by the Pigment from Desert Soil Streptomyces sp. D25. J. Appl. Pharm. Sci. 2016, 6, 148–150. [Google Scholar]
- Deveoğlu, O.; Karadağ, R. A Review on the Flavonoids—A Dye Source. Int. J. Adv. Eng. Pure Sci. 2019, 3, 188–200. [Google Scholar] [CrossRef] [Green Version]
- DH, S.; MA, H. Antibiofilm Activity of Streptomyces toxytricini Fz94 against Candida albicans ATCC 10231. Microb. Biosyst. 2017, 2, 26–39. [Google Scholar] [CrossRef]
- Gopikrishnan, V.; Radhakrishnan, M.; Shanmugasundaram, T.; Pazhanimurugan, R.; Balagurunathan, R. Antibiofouling Potential of Quercetin Compound from Marine-Derived Actinobacterium, Streptomyces fradiae PE7 and Its Characterization. Environ. Sci. Pollut. Res. 2016, 23, 13832–13842. [Google Scholar] [CrossRef]





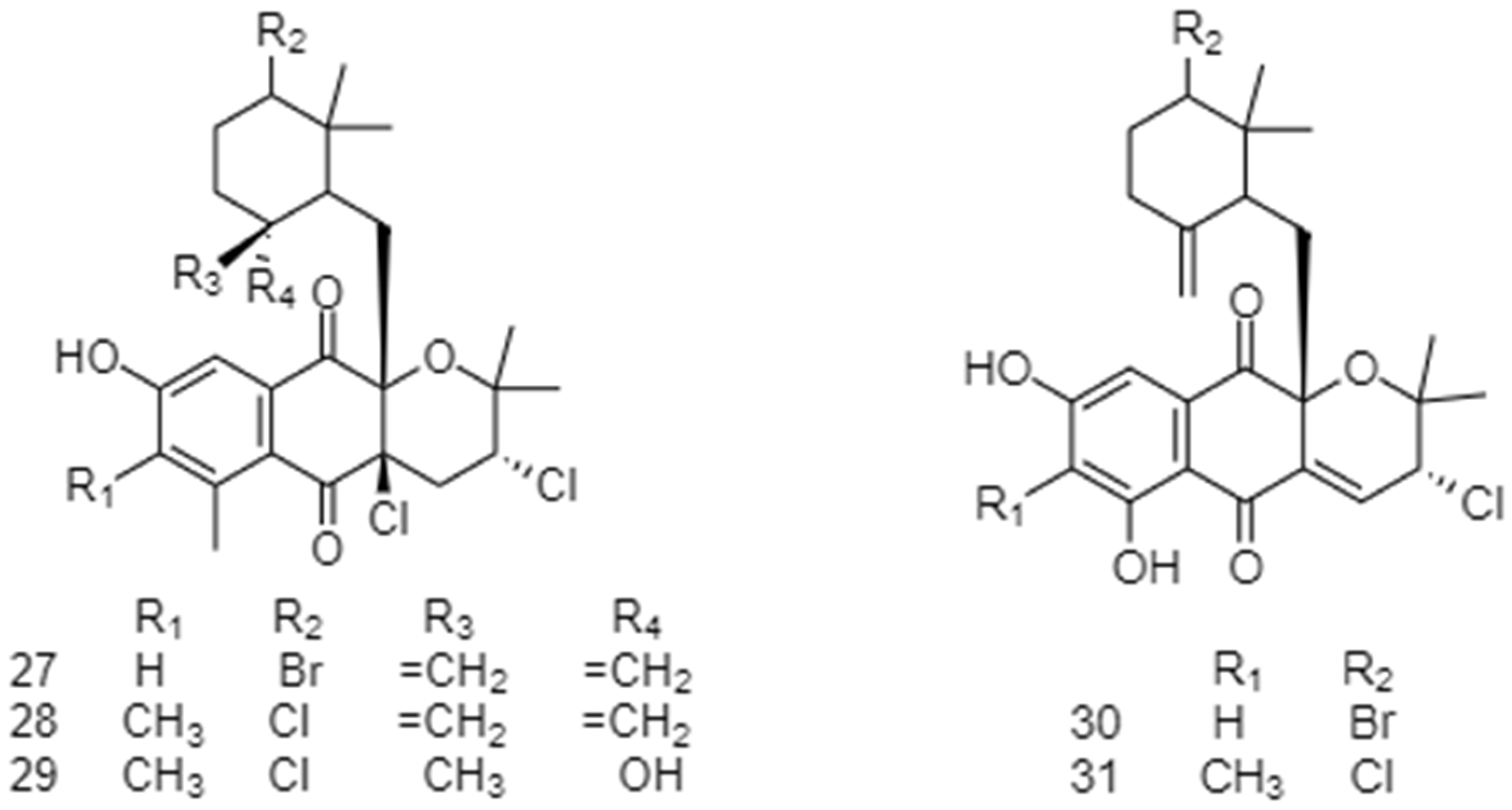
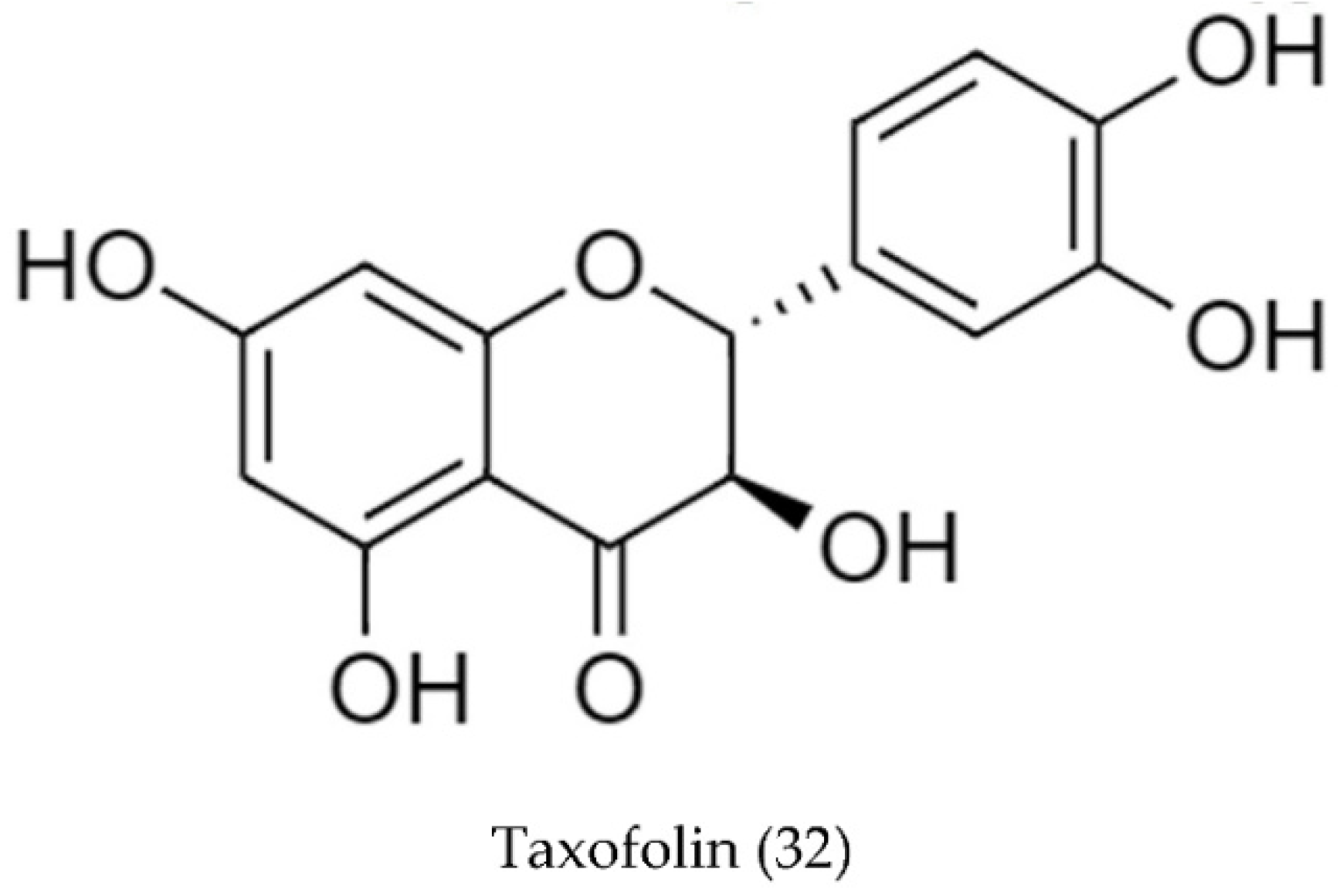

| Type of Source | Bioactivity | No. Strains | No. Compounds | Ref. |
|---|---|---|---|---|
| Freshwater (Free-living) | Antimicrobial | 6 | 3 | [64,65] |
| Marine (Free-living) | Antimicrobial | 4 | 1 | [60,66] |
| Antioxidant | 1 | 1 | [67] | |
| Multiple 1 | 1 | N/A 2 | [68] | |
| Marine Symbiont | Antimicrobial | 1 | N/A 2 | [69] |
| Cytotoxic | 1 | N/A 2 | [61] | |
| Multiple 1 | 1 | N/A 2 | [70] | |
| N/A 3 | 1 | N/A 2 | [71] | |
| Terrestrial Symbiont | N/A 3 | 1 | 3 | [72] |
| Soil (Free-living) | Antimicrobial | 11 | 3 | [62,63,73,74,75,76,77,78,79,80,81] |
| Antioxidant | 1 | 1 | [82] | |
| Cytotoxic | 5 | 2 | [83,84,85,86,87] | |
| Multiple 1 | 8 | 6 | [40,88,89,90,91,92,93,94] | |
| N/A 3 | 4 | 1 | [95,96,97] | |
| N/A 1 | Antimicrobial | 4 | 6 | [98,99,100,101] |
| Antioxidant | 1 | 1 | [102] | |
| N/A 3 | 15 | 7 | [103,104,105,106,107,108,109,110] |
| Streptomyces Strain | Type/Color of Pigment | Yield Reported (mg/L) | Ref. |
|---|---|---|---|
| Streptomyces sp. MVCS13 | Melanin | 239 | [66] |
| Streptomyces sp. F1 | Melanin | 21130 | [60] |
| Streptomyces sp. F2 | |||
| Streptomyces sp. F3 | |||
| Streptomyces sp. | Melanin | 1460 | [67] |
| Streptomyces glaucescens NEAE-H | Melanin | 350 | [40] |
| Streptomyces sp. ZL-24 | Melanin | 59 to 138 | [94] |
| Streptomyces glaucescens KCTC988 | Melanin | 125 1 | [102] |
| Streptomyces cavourensis SV 21 | Melanin | 670 | [70] |
| Streptomyces parvus BSB49 | Eumelanin | 160 to 240 | [90] |
| Streptomyces sp. CWW6 | Streptorubrin A (prodiginine pigment) | 20 | [64] |
| Streptomyces sp. WMA-LM31 | Prodigiosin (4) | 30 | [91] |
| Streptomyces sp. JS520 | Red pigment | <1 to 139 1 | [88] |
| Streptomyces sp. PM4 | Red pigment | 1874 | [61] |
| Streptomyces acidiscabies | Naphthoquinone derivatives: Bright yellow compound (5) 2 | 63 | [72] |
| Orange compound (6) 3 | 28 | ||
| Compound (7) 4 | 2 | ||
| Streptomyces sp. D25 | Yellow pigment | 175 to 1225 5 | [62] |
| Streptomyces griseoaurantiacus JUACT 01 | Yellow pigment | 4300 | [84] |
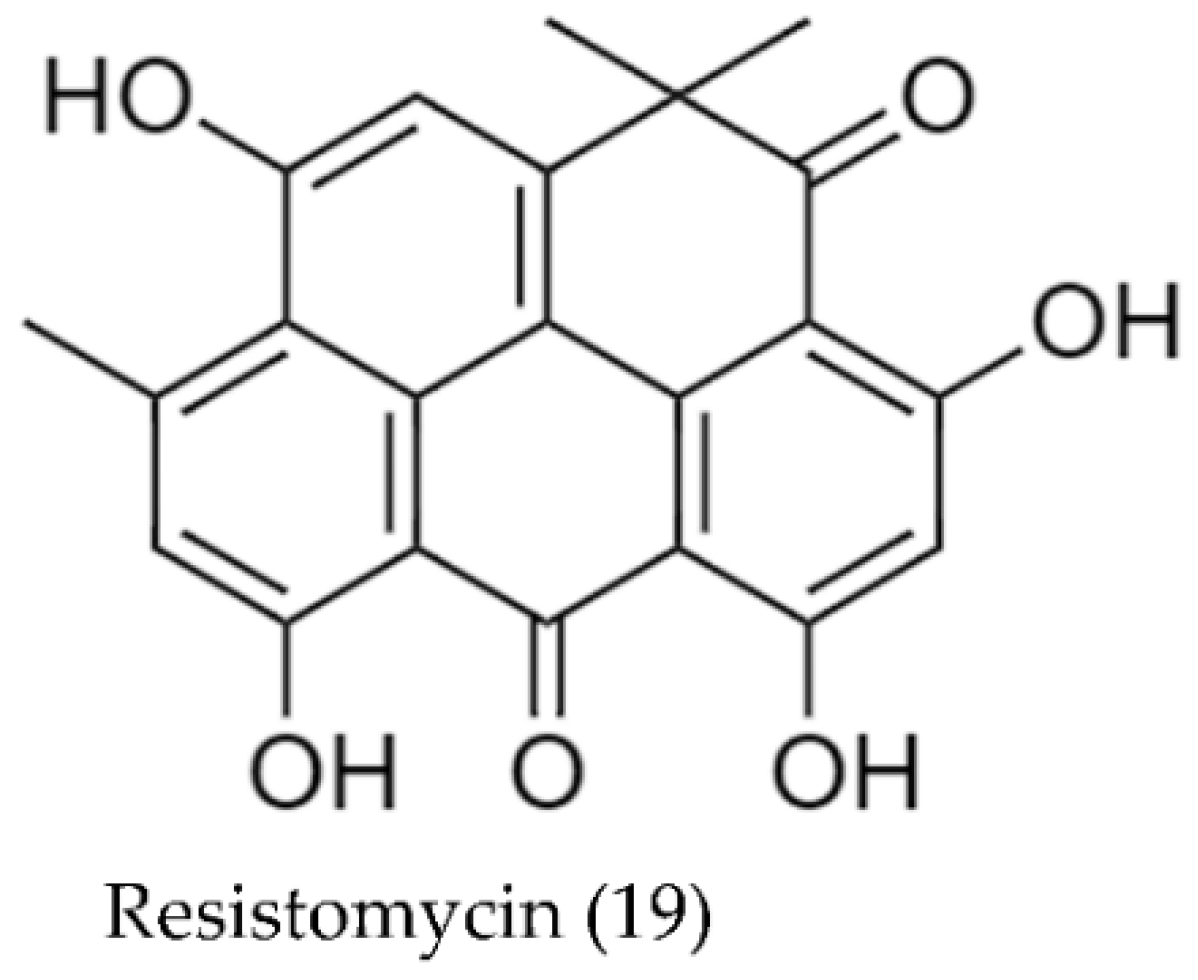
| Streptomyces aurantiacus AAA5 | Resistomycin (19) (yellow compound) | 52 | [93] |
| S. griseus IFO13350 w | Grixazone A (14) | 5 | [98] |
| Grixazone B (14) | 2 | ||
| Streptomyces sp. A1013Y | Blue pigment | 2 | [82] |
| S. coelicolor 100 | Blue pigment | 3000 | [86] |
| Streptomyces coelicolor MSIS1 | Red or blue, depending on conditions | 5030 (shake flasks) 9000 (bioreactor) | [92] |
| Streptomyces coelescens ATCC 19830 (NP2) | Deep blue | 3000 to 4000 | [96] |
| Streptomyces anthocyanicus ATCC 19821 (NP4) | Deep red | 3000 to 4000 | [96] |
| Streptomyces Strain | Type/Color of Pigment | Stability Results | Ref. |
|---|---|---|---|
| Streptomyces vietnamensis sp. nov. GIMV4.0001 | Violet–blue pigment | pH-sensitive | [76] |
| Stable at high temperature | |||
| Stable under UV light | |||
| Streptomyces spectabilis L20190601 | Metacycloprodigiosin (1) | pH-sensitive, red at pH 3.0 and yellow or orange at pH 9.0 | [77] |
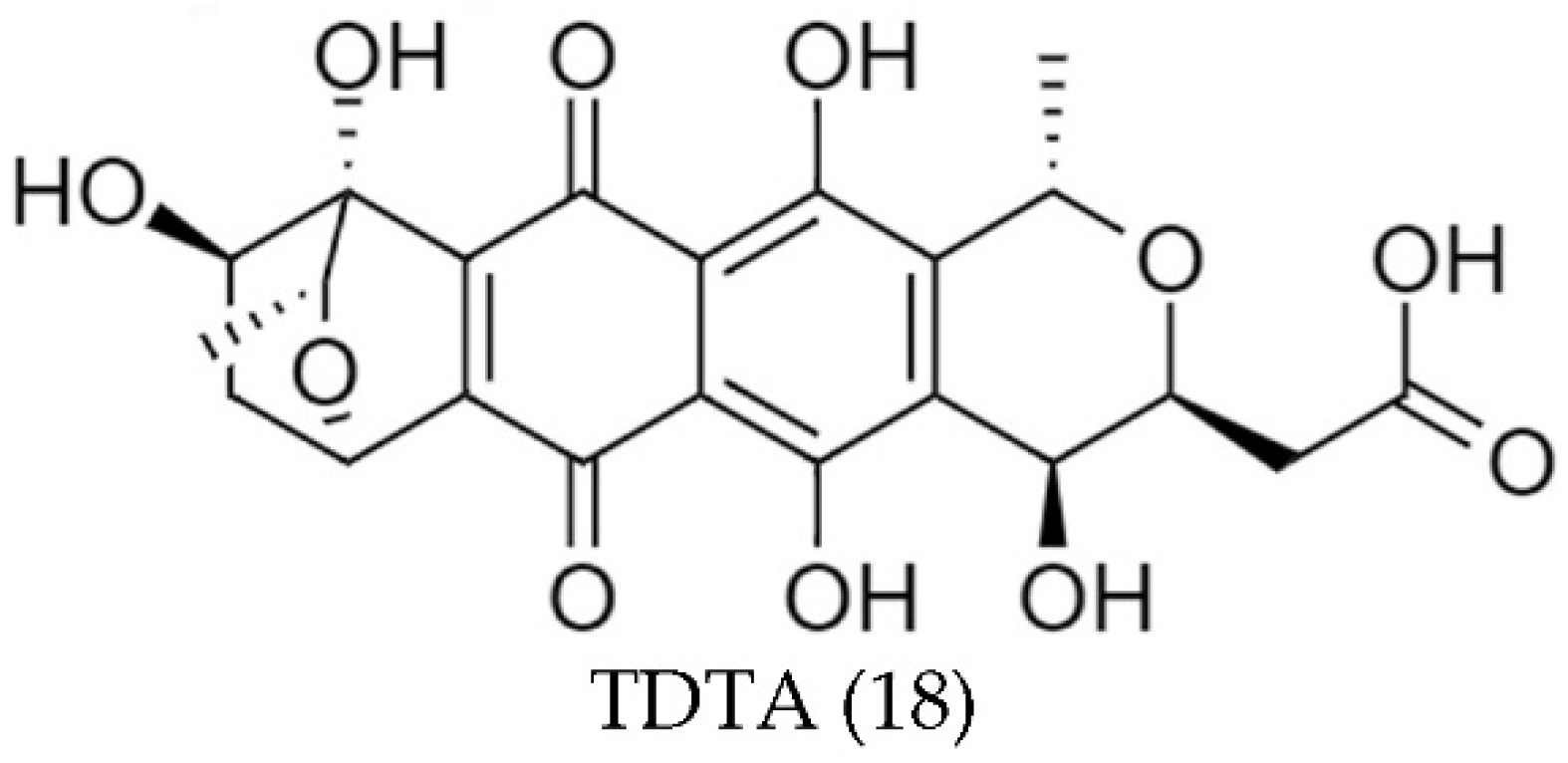
| Streptomyces sp. A1013Y | TDTA (18) | Stable in a wide range of pHs | [82] |
| Thermo-stable | |||
| Good stability with indoor incandescent and ultraviolet lights, but unstable with sunlight | |||
| Stable with most metal ions and vitamins except Fe3+, Cu2+, and Al3+ | |||
| Streptomyces coelicolor 100 | λ-Actinorhodin (12) | pH-sensitive; red at pH < 7, amaranth at pH 7–8, blue at pH > 8 | [86] |
| Photo-stable | |||
| Thermo-stable | |||
| Resistant to oxidants and reducers under acid conditions and to reducers under alkaline conditions. | |||
| Stable with food additives | |||
| Stable with most metal ions except Fe2+ and Pb2+ | |||
| Streptomyces coelicolor MSIS1 | May be one of actinorhodinic acid. | pH-sensitive, red at pH < 7, amaranth at pH 7–8, blue at pH > 8 | [92] |
| Streptomyces coelescens ATCC 19830 (NP2) | Deep blue | pH-sensitive | [96] |
| Streptomyces anthocyanicus ATCC 19821 (NP4) | Deep red | pH-sensitive | [96] |
| Streptomyces sp. LS-1 | May be actinorhodin-related compounds. | Sensitive to low pH | [97] |
| Relatively photo-stable | |||
| Relatively thermo-stable | |||
| Streptomyces parvullus M4 | Red | pH-Stable | [105] |
| Streptomyces coelicolor M6 | Red | pH-Stable | [105] |
| Streptomyces cyaneofuscatus | Actinomycin X2 (10) | Excellent thermal stability | [69] |
| Acid and alkali resistance |
| Streptomyces Strains | Source | Positive Antimicrobial Tests | Inhibition Zone (mm) | Ref. |
| Streptomyces sp. F1 | Pure colonies | E. coli, Lactobacillus vulgaris, Proteus mirabilus, Vibrio cholera, S. aureus, S. typhi, S. paratyphi, and K. oxytoca | N/A | [60] |
| Streptomyces sp. F2 | ||||
| Streptomyces sp. F3 | ||||
| Streptomyces coeruleorubidus NBRC 12844 | Extract | S. aureus ATCC 1112, B. cereus ATCC1015, P. aeruginosa ATCC 1074, C. freundii ATCC 8090, K. pneumoniae ATCC 1053, and S. marcescens ATCC 14756 | N/A | [73] |
| Streptomyces sp. D10 | Fraction | MRSA | 15 | [75] |
| VRSA | 20 | |||
| E. coli | 15 | |||
| Klebsiella sp. | 10 | |||
| Streptomyces sp. D25 | Extract | MRSA | 22 1 | [62] |
| Streptomyces sp. SAG-85 | Extract | MRSA | 23 2 | [78] |
| S. marcescens | 47 2 | |||
| Streptomyces sp. ZL-24 | Compound 3 (Melanin) | P. aeruginosa ATCC 9027 | 26–32 | [94] |
| E. coli ATCC 8379 | 21–29 | |||
| S. aureus ATCC6538 | 15–30 | |||
| Mycobacterium smegmatis ATCC 10231 | 17–36 | |||
| Streptomyces coelicolor A3(2) | Extract | B. subtilis, S. aureus, E. coli, and Pseudomonas fluorescens | N/A | [101] |
| Streptomyces cyaneofuscatus | Compound (Actinomycin X2 (10)) | S. aureus ATCC 6538 | 20 | [69] |
| Streptomyces sp. Ac-2 | Extract | P. aeruginosa ATCC 27853 | 18 2 | |
| S. aureus ATCC 25923 | 22 2 | [65] | ||
| E. coli ATCC 25922 | 21 2 | |||
| Streptomyces sp. | Extract 4 | S. aureus MTCC 3160 | 4 | [68] |
| B. subtilis MTCC 736 | 10 | |||
| E. coli MTCC 1554 | 6 | |||
| Vibrio cholera MTCC 3906 | 5 | |||
| Streptomyces sp. NS-05 | Extract 5 | E. coli MTCC 739 | 5 2 | [81] |
| Proteus vulgaris MTCC 6380 | 8 2 |
| Streptomyces Strain (Source/Compound Name) | Positive Antimicrobial Tests | MIC (μg/mL) | Ref. |
|---|---|---|---|
| S. spectabilis L20190601 1 (Metacycloprodigiosin (1)) | Staphylococcus aureus | <1 | [77] |
| Bacillus subtilis | <1 | ||
| Escherichia coli | 4 | ||
| Streptococcus pyogenes | <1 | ||
| Pseudomonas aeruginosa | <1 | ||
| Bacillus typhi | 1 | ||
| Candida albicans | 2 | ||
| Trichophyton rubrum | 64 | ||
| Streptomyces sp. JS520 1 (Undecylprodigiosin (2)) | Micrococcus luteus ATCC 379 | 50 | [88] |
| Bacillus subtilis ATCC 6633 | 50 | ||
| Candida albicans ATCC 10231 | 100 | ||
| Candida albicans ATCC 10259 | 200 | ||
| Streptomyces sp. JAR6 1 (Undecylprodigiosin (2)) | Salmonella sp. | 150 | [89] |
| Bacillus subtilis | 50 | ||
| Proteus mirabilis | 80 | ||
| Shigella sp. | 100 | ||
| Escherichia coli | 170 | ||
| Enterococcus sp. | 120 | ||
| Klebsiella pneumoniae | 180 | ||
| Streptomyces sp. MVCS13 (Melanin) | Bacillus sp. FPO1 | 23 2 | [66] |
| Aeromonas sp. FPO2 | 27 2 | ||
| Citrobacter sp. FPO3 | 21 2 | ||
| Edwardsiella sp. FPO4 | 20 2 | ||
| Vibrio sp. FPO5 | 18 2 | ||
| Aeromonas sp. FPO6 | 22 2 | ||
| Streptomyces aurantiacus AAA5 (Resistomycin (19)) | S. epidermis | 8 2 | [93] |
| Enterococcus faecalis | 5 2 | ||
| Bacillus subtilis | 25 2 | ||
| Staphylococcus aureus | 13 2 | ||
| Klebsiella pneumoniae | 16 2 | ||
| Shigella sp. | 45 2 | ||
| Proteus vulgaris | 70 2 | ||
| Escherichia coli | 42 2 | ||
| Pseudomonas aeruginosa | 34 2 | ||
| Salmonella typhii | 15 2 | ||
| Streptomyces sp. MBT27 1 (Actinomycins L1 (8)) 3 | Staphylococcus aureus MB5393 | 4–8 | [80] |
| Staphylococcus aureus ATCC29213 | 2–4 | ||
| Vancomycin-sensitive Enterococcus faecium | 4–8 | ||
| Vancomycin-resistant Enterococcus faecium | 4–8 | ||
| S. epidermidis | 4–8 | ||
| Escherichia coli ATCC25922 | >128 | ||
| Klebsiella pneumoniae ATCC700603 | >128 | ||
| Streptomyces sp. MBT27 1 (Actinomycins L2 (9)) 3 | Staphylococcus aureus MB5393 | 8–16 | |
| Vancomycin-sensitive Enterococcus faecium | 8–16 | [80] | |
| Vancomycin-resistant Enterococcus faecium | 8–16 | ||
| Streptomyces parvulus C5-5Y (Fraction F5) | S. aureus | 125 | [74] |
| S. epidermidis | 125 | ||
| Enterococcus faecalis | 250 | ||
| E. coli | 375 | ||
| Pseudomonas sp. | 125 | ||
| K. pneumoniae | 125 | ||
| S. typhi | 125 | ||
| Proteus vulgaris | 500 | ||
| Shigella sp. | 125 | ||
| Streptococcus mutans | 125 | ||
| Streptomyces sp. S45 (Fraction) | S. aureus ATCC 29213 | 2 | [63] |
| Streptomyces sp. BSE6.1 (Prodigiosin (4)) | S. aureus MTCC1430 | 400 | [99] |
| Streptomyces Strain (Source) | Antioxidant Method Evaluated | IC50 (μg/mL) | Equivalence to Vitamin C (μg) | Ref. |
|---|---|---|---|---|
| Streptomyces sp. A1013Y (TDTA (18)) | DPPH | 41 | <1 | [82] |
| ABTS | 14 | 1 | ||
| Streptomyces glaucescens KCTC988 (Melanin) | ABTS | 25,080 | N/A | [102] |
| ABTS (In presence of copper ions) | 7890 | N/A |
| Streptomyces Strain (Source) | Antioxidant Method Evaluated | Concentration Evaluated (μg/mL) | Percentage of Activity | Ref. |
|---|---|---|---|---|
| Streptomyces sp. (Melanin) | Hydroxyl radical scavenging activity | 500 | 70% | [67] |
| Streptomyces glaucescens NEAE-H (Melanin) | ABTS | 100 | 57% | [40] |
| Streptomyces parvus BSB49 (Eumelanin) | DPPH | 250 | 88% | [90] |
| ABTS | 250 | 75% | ||
| Streptomyces sp. WMA-LM31 (Prodigiosin) | DPPH | 10 | 60% | [91] |
| Lipid peroxidation inhibition assay | 10 | 25% | ||
| In vitro protein oxidation inhibition assay | 10 | 55% | ||
| Streptomyces sp. ZL-24 (Melanin) | DPPH | 5 | 65% | [94] |
| Hydroxyl radical scavenging activity | 50 | 96% | ||
| Superoxide scavenging activity | 10 | 43% | ||
| 50 | 60% |
| Streptomyces Strain (Source) | Antioxidant Method Evaluated | Results of Antioxidant Activity | Ref. |
|---|---|---|---|
| Streptomyces sp. (Melanin) | Superoxide radical scavenging activity | Moderate scavenger of superoxide radical in vitro and exhibited a strong dose–effect relationship. | [67] |
| Reducing power assay | Antioxidant activity of melanin might be due to redox reactions. | ||
| Streptomyces sp. JS520 (Undecylprodigiosin (2)) | Ferric thiocyanate method | Undecylprodigiosin (2) did not perform as well as commercially available antioxidant α-tocopherol; however, it was effective in delaying lipid peroxidation. | [88] |
| Hydrogen peroxide disc-diffusion assay | Undecylprodigiosin (2) acted as a scavenger of H2O2 that is released through the process of peroxidation. | ||
| Streptomyces sp. JAR6 (Undecylprodigiosin (2)) | DPPH | Strain JAR6 was able to reduce compounds to pale yellow hydrazine as a DPPH radical. | [89] |
| Streptomyces coelicolor MSIS1 (Extract) | Reducing Power Assay | The pigment had positive results for all the concentrations: 10 mg/mL, 50 mg/mL, and 100 mg/mL. | [92] |
| Streptomyces cavourensis SV 21 (Melanin) | DPPH | Acid-treated forms of melanin showed much stronger radical scavenging ability than the intact melanin derivatives. | [70] |
| Hydroxyl radical scavenging activity | Rapid oxidation and bleaching of the melanin pigment and thus its capacity to scavenge H2O2 out of the environment. | ||
| Streptomyces sp. (Extract) | Free radical scavenging activity | The pigment showed increasing free radical scavenging activity and total antioxidant activity with increased concentrations. | [68] |
| Ferric Reducing Antioxidant Power | |||
| Hydroxyl Radical Scavenging Activity |
| Streptomyces Strain | Pigment | Concentration (μg/mL) | Cell Line | Cell Density (Cells/Well) | Time | IC50 (μg/mL) | Ref. |
|---|---|---|---|---|---|---|---|
| Streptomyces sp. PM4 | Red pigment | 10, 20, 30, 40, 50 | HT1080 | 2 × 104 | 24 h | 18.5 | [61] |
| Hep2 | 15.3 | ||||||
| HeLa | 9.6 | ||||||
| MCF7 | 8.5 | ||||||
| Streptomyces griseoaurantiacus JUACT 01 | Yellow Pigment | 2.5, 5, 10, 20 | HeLa | N/A | 24 h | 5.31 | [84] |
| 48 h | 2 | ||||||
| 72 h | 1.8 | ||||||
| HepG2 | 24 h | 26.33 | |||||
| 48 h | 1.75 | ||||||
| 72 h | 1.41 | ||||||
| Human lymphocytes | 24 h, 48 h, 72 h | Any cytotoxicity | |||||
| Streptomyces sp. A 16-1 | Red pigment (Fr 5, Fr6, and Fr7) | 0–8 | KB cells | 5 × 104 | 48 h | 0.04 ± 0.005 (Fr 5) | [85] |
| 0.20 ± 0.02 (Fr 6) | |||||||
| 0.55 ± 0.05 (Fr 7) | |||||||
| PBMCs | Low Cytotoxicity | ||||||
| Streptomyces sp. JAR6 | Red pigment (Undecylprodigiosin (2)) | 18.75, 37.5, 75, 150, 300 | HeLa | 1 × 104 | 48 h | 145 | [89] |
| Streptomyces glaucescens NEAE-H | Melanin | 1.56, 3.125, 6.25, 12.5, 25, 50, 100 | HFB4 | 1 × 104 | 24 h | 16.34 ± 1.31 | [40] |
| WI-38 | 37.05 ± 2.40 | ||||||
| WISH | 48.07 ± 2.76 | ||||||
| Streptomyces sp. WMA-LM31 | Prodigiosin (4) | 5, 10, 15, 20 | HepG2 | 1 × 104 | 24 h | 12.66 | [91] |
| HeLa | 14.83 | ||||||
| Streptomyces parvus BSB49 | Eumelanin | 2.72 × 106–1.09 × 107 1 | HeLa | 3 × 104 | 24 h | 5.45 × 106 1 | [90] |
| Streptomyces sp. NP4 | Prodigiosin (4) | N/A 2 | HaCat | 1 × 104 | 48 h | No significant cytotoxic effect | [83] |
| MRC-5 | |||||||
| Streptomyces sp. | N/A | N/A | HT-1080 | N/A | N/A | 202.13 | [68] |
| HeLa | 253.86 |
| Streptomyces Strain | Pigment Evaluated | Concentration (μg/mL) | Cell Line | Time of Treatment | GI50 (μg/mL) | LC50 (μg/mL) | Ref. |
|---|---|---|---|---|---|---|---|
| Streptomyces aurantiacus AAA5 | Resistomycin (19) | 5, 0.5, 0.05, 0.005, 0.0005 | HepG2 | N/A | 5 × 10−3 | 1 × 10−2 | [93] |
| HeLa | 6 × 10−3 | 1 × 10−2 |
| Streptomyces Strain | Type of Assay | N° | Doses (mg/kg) | General Results | LD50 (mg/kg) | Ref. |
|---|---|---|---|---|---|---|
| S. coelicolor 100 (λ-Actinorhodin (12)) | Mouse acute toxicity trial | First Assay | 1500 and 15,000 | Mouse death resulted from taking an overdose pigment once by oral gavages. | >15,000 | [86] |
| Second Assay | 0, 464, 1000, 2155, 4633, 10,000, and 15,000 | Nontoxic substance |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarmiento-Tovar, A.A.; Silva, L.; Sánchez-Suárez, J.; Diaz, L. Streptomyces-Derived Bioactive Pigments: Ecofriendly Source of Bioactive Compounds. Coatings 2022, 12, 1858. https://doi.org/10.3390/coatings12121858
Sarmiento-Tovar AA, Silva L, Sánchez-Suárez J, Diaz L. Streptomyces-Derived Bioactive Pigments: Ecofriendly Source of Bioactive Compounds. Coatings. 2022; 12(12):1858. https://doi.org/10.3390/coatings12121858
Chicago/Turabian StyleSarmiento-Tovar, Aixa A., Laura Silva, Jeysson Sánchez-Suárez, and Luis Diaz. 2022. "Streptomyces-Derived Bioactive Pigments: Ecofriendly Source of Bioactive Compounds" Coatings 12, no. 12: 1858. https://doi.org/10.3390/coatings12121858







