Investigating the Synergic Effects of WS2 and ECAP on Degradation Behavior of AZ91 Magnesium Alloy
Abstract
1. Introduction
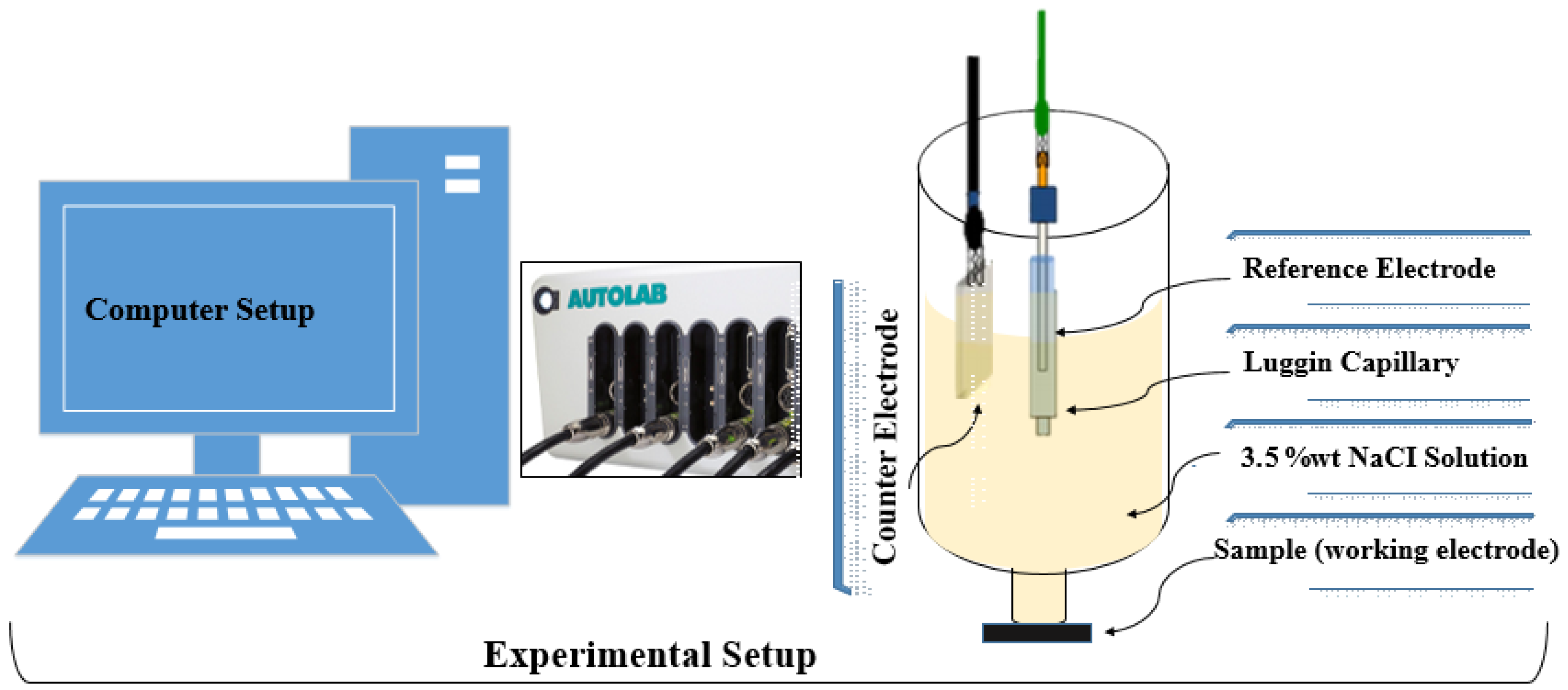
2. Materials and Method
3. Results and Discussion
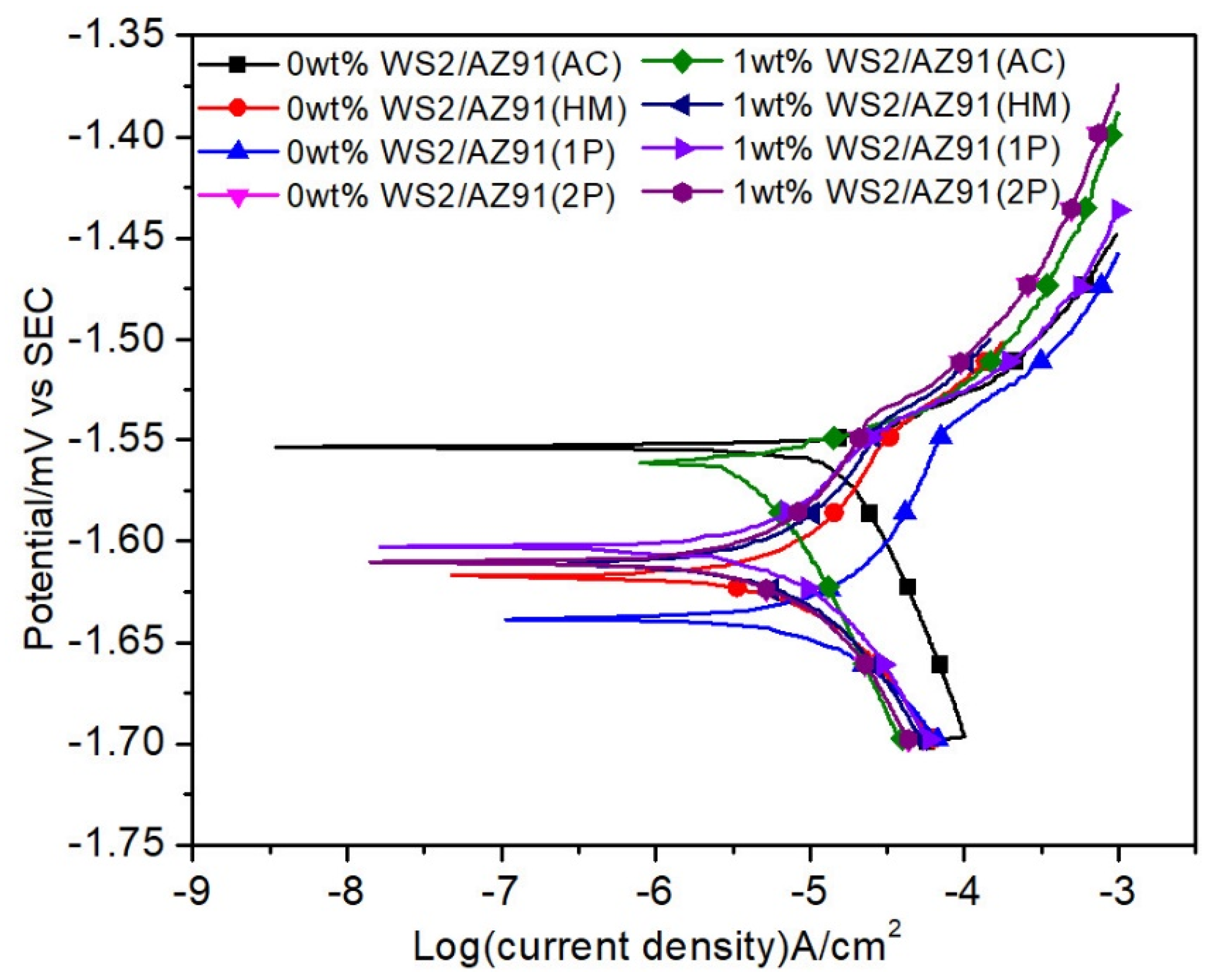
3.1. Potentiodynamic Polarization
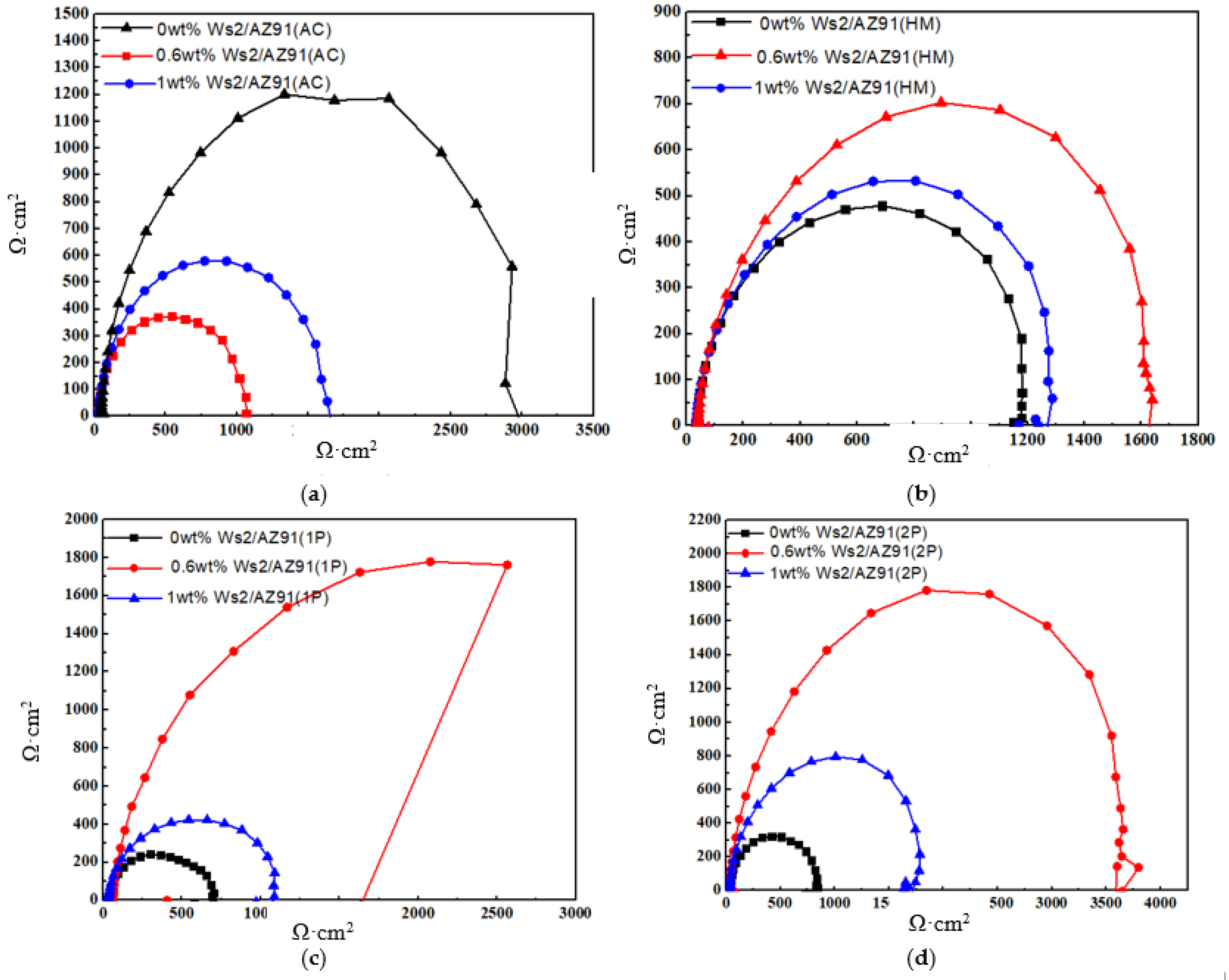
3.2. Electrochemical Impedance Spectroscopy
3.3. Microstructural Characterization
4. Conclusions
- The monolithic AZ91 exhibits a maximum corrosion resistance and degradation rate (4.5120 m·a−1) in as-cast conditions.
- Two-pass ECAPed 1 wt% WS2/AZ91 has a corrosion potential of (−1.6101 mV) and a corrosion rate of (4.5951 m·a−1); however, a minimum corrosion potential (−1.63849 mV) and maximum degradation rate (4.613 m·a−1) was recorded for one-pass ECAPed 0 wt% WS2/AZ91.
- All composites exhibit similar Tafel curves, which indicates that similar corrosion behavior has taken place in all samples.
- The degradability of AZ91 is increased in all conditions under the synergic effect of WS2 and severe plastic deformation.
- The passive layers of oxide and hydroxide film were established on the corroded surface of all composites
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sundaresan, R.; Froes, F.H. Mechanical Alloying in the Titanium-Magnesium System. Key Eng. Mater. 1991, 29–31, 199–206. [Google Scholar] [CrossRef]
- Rashad, M.; Pan, F.; Liu, Y.; Chen, X.; Lin, H.; Pan, R.; Asif, M.; She, J. High Temperature Formability of Graphene Nanoplatelets-AZ31 Composites Fabricated by Stir-Casting Method. J. Magnes. Alloy. 2016, 4, 270–277. [Google Scholar] [CrossRef]
- Mediaswanti, K.; Wen, C.; Ivanova, E.P.; Berndt, C.C.; Malherbe, F.; Thi, V.; Pham, V.T.H.; Wang, J. A Review on Bioactive Porous Metallic Biomaterials. J. Biomim. Biomater. Tissue Eng. 2013, 18, 1–8. [Google Scholar] [CrossRef]
- Wang, X.J.; Hu, X.S.; Liu, W.Q.; Du, J.F.; Wu, K.; Huang, Y.D.; Zheng, M.Y. Ageing Behavior of As-Cast SiCp/AZ91 Mg Matrix Composites. Mater. Sci. Eng. A 2017, 682, 491–500. [Google Scholar] [CrossRef]
- Yeganeh, M.; Mohammadi, N. Superhydrophobic Surface of Mg Alloys: A Review. J. Magnes. Alloy. 2018, 6, 59–70. [Google Scholar] [CrossRef]
- Luo, A.A. Magnesium Casting Technology for Structural Applications. J. Magnes. Alloy. 2013, 1, 2–22. [Google Scholar] [CrossRef]
- Kumar, D.; Thakur, L. Recent Studies on the Fabrication of Magnesium Based Metal Matrix Nano-Composites by Using Ultrasonic Stir Casting Technique—A Review. Mater. Sci. Forum 2019, 969, 889–894. [Google Scholar] [CrossRef]
- Huang, S.-J.; Abbas, A. Effects of Tungsten Disulfide on Microstructure and Mechanical Properties of AZ91 Magnesium Alloy Manufactured by Stir Casting. J. Alloys Compd. 2020, 817, 153321. [Google Scholar] [CrossRef]
- Abbas, A.; Huang, S.-J. Qualitative and Quantitative Investigation of As-Cast and Aged CNT/AZ31 Metal Matrix Composites. JOM 2020, 72, 2272–2282. [Google Scholar] [CrossRef]
- Abbas, A.; Huang, S.-J. ECAP Effects on Microstructure and Mechanical Behavior of Annealed WS2/AZ91 Metal Matrix Composite. J. Alloys Compd. 2020, 835, 155466. [Google Scholar] [CrossRef]
- Yu, W.; Wang, X.; Zhao, H.; Ding, C.; Huang, Z.; Zhai, H.; Guo, Z.; Xiong, S. Microstructure, Mechanical Properties and Fracture Mechanism of Ti2AlC Reinforced AZ91D Composites Fabricated by Stir Casting. J. Alloys Compd. 2017, 702, 199–208. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, W.; Li, B.; Ren, H.; Qi, Y.; Zhao, D. Effects of Milling Duration on Electrochemical Hydrogen Storage Behavior of As-Milled Mg–Ce–Ni–Al-Based Alloys for Use in Ni-Metal Hydride Batteries. J. Phys. Chem. Solids 2019, 133, 178–186. [Google Scholar] [CrossRef]
- Kim, W.J.; Jeong, H.G.; Jeong, H.T. Achieving High Strength and High Ductility in Magnesium Alloys Using Severe Plastic Deformation Combined with Low-Temperature Aging. Scr. Mater. 2009, 61, 1040–1043. [Google Scholar] [CrossRef]
- Bin, B.S.J.; Tan, Y.T.; Fong, K.S.; Tan, M.J. Effect of Severe Plastic Deformation and Post-Annealing on the Mechanical Properties and Bio-Corrosion Rate of AZ31 Magnesium Alloy. Procedia Eng. 2017, 207, 1475–1480. [Google Scholar] [CrossRef]
- Ralston, K.D.; Birbilis, N. Effect of Grain Size on Corrosion: A Review. Corrosion 2010, 66, 075005–075013. [Google Scholar] [CrossRef]
- Huot, J. Nanocrystalline Metal Hydrides Obtained by Severe Plastic Deformations. Metals 2012, 2, 22–40. [Google Scholar] [CrossRef]
- Hamu, G.B.; Eliezer, D.; Wagner, L. The Relation between Severe Plastic Deformation Microstructure and Corrosion Behavior of AZ31 Magnesium Alloy. J. Alloys Compd. 2009, 468, 222–229. [Google Scholar] [CrossRef]
- Abbas, A.; Huang, S.-J. Investigation of Severe Plastic Deformation Effects on Microstructure and Mechanical Properties of WS2/AZ91 Magnesium Metal Matrix Composites. Mater. Sci. Eng. A 2020, 780, 139211. [Google Scholar] [CrossRef]
- Mehrian, S.S.M.; Rahsepar, M.; Khodabakhshi, F.; Gerlich, A.P. Effects of Friction Stir Processing on the Microstructure, Mechanical and Corrosion Behaviors of an Aluminum-Magnesium Alloy. Surf. Coat. Technol. 2021, 405, 126647. [Google Scholar] [CrossRef]
- Liu, H.; Yan, Y.; Wu, X.; Fang, H.; Chu, X.; Huang, J.; Zhang, J.; Song, J.; Yu, K. Effects of Al and Sn on Microstructure, Corrosion Behavior and Electrochemical Performance of Mg–Al-Based Anodes for Magnesium-Air Batteries. J. Alloys Compd. 2021, 859, 157755. [Google Scholar] [CrossRef]
- Liu, X.; Shan, D.; Song, Y.; Han, E. Effects of Heat Treatment on Corrosion Behaviors of Mg-3Zn Magnesium Alloy. Trans. Nonferrous Met. Soc. China 2010, 20, 1345–1350. [Google Scholar] [CrossRef]
- Abbas, A.; Rajagopal, V.; Huang, S.-J. Magnesium Metal Matrix Composites and Their Applications. In Magnesium Alloys; IntechOpen: London, UK, 2021. [Google Scholar]
- Li, Z.; Peng, Z.; Qiu, Y.; Qi, K.; Chen, Z.; Guo, X. Study on Heat Treatment to Improve the Microstructure and Corrosion Behavior of ZK60 Magnesium Alloy. J. Mater. Res. Technol. 2020, 9, 11201–11219. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, G.; Zeng, S.; Zhang, S.; Long, F.; Shi, Q. The Corrosion Behavior of Mg-9Al-XRE Magnesium Alloys Modified by Friction Stir Processing. J. Alloys Compd. 2021, 851, 156835. [Google Scholar] [CrossRef]
- Esmaily, M.; Svensson, J.E.; Fajardo, S.; Birbilis, N.; Frankel, G.S.; Virtanen, S.; Arrabal, R.; Thomas, S.; Johansson, L.G. Fundamentals and Advances in Magnesium Alloy Corrosion. Prog. Mater. Sci. 2017, 89, 92–193. [Google Scholar] [CrossRef]
- Song, Y.; Han, E.-H.; Dong, K.; Shan, D.; Yim, C.D.; You, B.S. Effect of Hydrogen on the Corrosion Behavior of the Mg–XZn Alloys. J. Magnes. Alloy. 2014, 2, 208–213. [Google Scholar] [CrossRef]
- Xu, F.; Luo, L.; Xiong, L.; Liu, Y. Microstructure and Corrosion Behavior of ALD Al2O3 Film on AZ31 Magnesium Alloy with Different Surface Roughness. J. Magnes. Alloy. 2020, 8, 480–492. [Google Scholar] [CrossRef]
- García-Rodríguez, S.; Torres, B.; Pulido-González, N.; Otero, E.; Rams, J. Corrosion Behavior of 316L Stainless Steel Coatings on ZE41 Magnesium Alloy in Chloride Environments. Surf. Coat. Technol. 2019, 378, 124994. [Google Scholar] [CrossRef]
- Mahdavian, M.; Attar, M.M. Another Approach in Analysis of Paint Coatings with EIS Measurement: Phase Angle at High Frequencies. Corros. Sci. 2006, 48, 4152–4157. [Google Scholar] [CrossRef]
- Xu, Z.; Eduok, U.; Tiamiyu, A.A.; Szpunar, J. Anodic Dissolution Pattern of Magnesium Alloy in Different Media: Effects of Solution Treatment on Its Microstructure and Corrosion Behaviour. Eng. Fail. Anal. 2020, 107, 104234. [Google Scholar] [CrossRef]
- Vaughan, M.W.; Karayan, A.I.; Srivastava, A.; Mansoor, B.; Seitz, J.M.; Eifler, R.; Karaman, I.; Castaneda, H.; Maier, H.J. The Effects of Severe Plastic Deformation on the Mechanical and Corrosion Characteristics of a Bioresorbable Mg-ZKQX6000 Alloy. Mater. Sci. Eng. C 2020, 115, 111130. [Google Scholar] [CrossRef]
- Jamesh, M.I.; Wu, G.; Zhao, Y.; Jin, W.; McKenzie, D.R.; Bilek, M.M.M.; Chu, P.K. Effects of Zirconium and Nitrogen Plasma Immersion Ion Implantation on the Electrochemical Corrosion Behavior of Mg–Y–RE Alloy in Simulated Body Fluid and Cell Culture Medium. Corros. Sci. 2014, 86, 239–251. [Google Scholar] [CrossRef]
- Sajjad, U.; Abbas, A.; Sadeghianjahromi, A.; Abbas, N.; Liaw, J.-S.; Wang, C.-C. Enhancing Corrosion Resistance of Al 5050 Alloy Based on Surface Roughness and Its Fabrication Methods; an Experimental Investigation. J. Mater. Res. Technol. 2021, 11, 1859–1867. [Google Scholar] [CrossRef]
- Yang, Y.; Xiong, X.; Chen, J.; Peng, X.; Chen, D.; Pan, F. Research Advances in Magnesium and Magnesium Alloys Worldwide in 2020. J. Magnes. Alloy. 2021, 9, 705–747. [Google Scholar] [CrossRef]
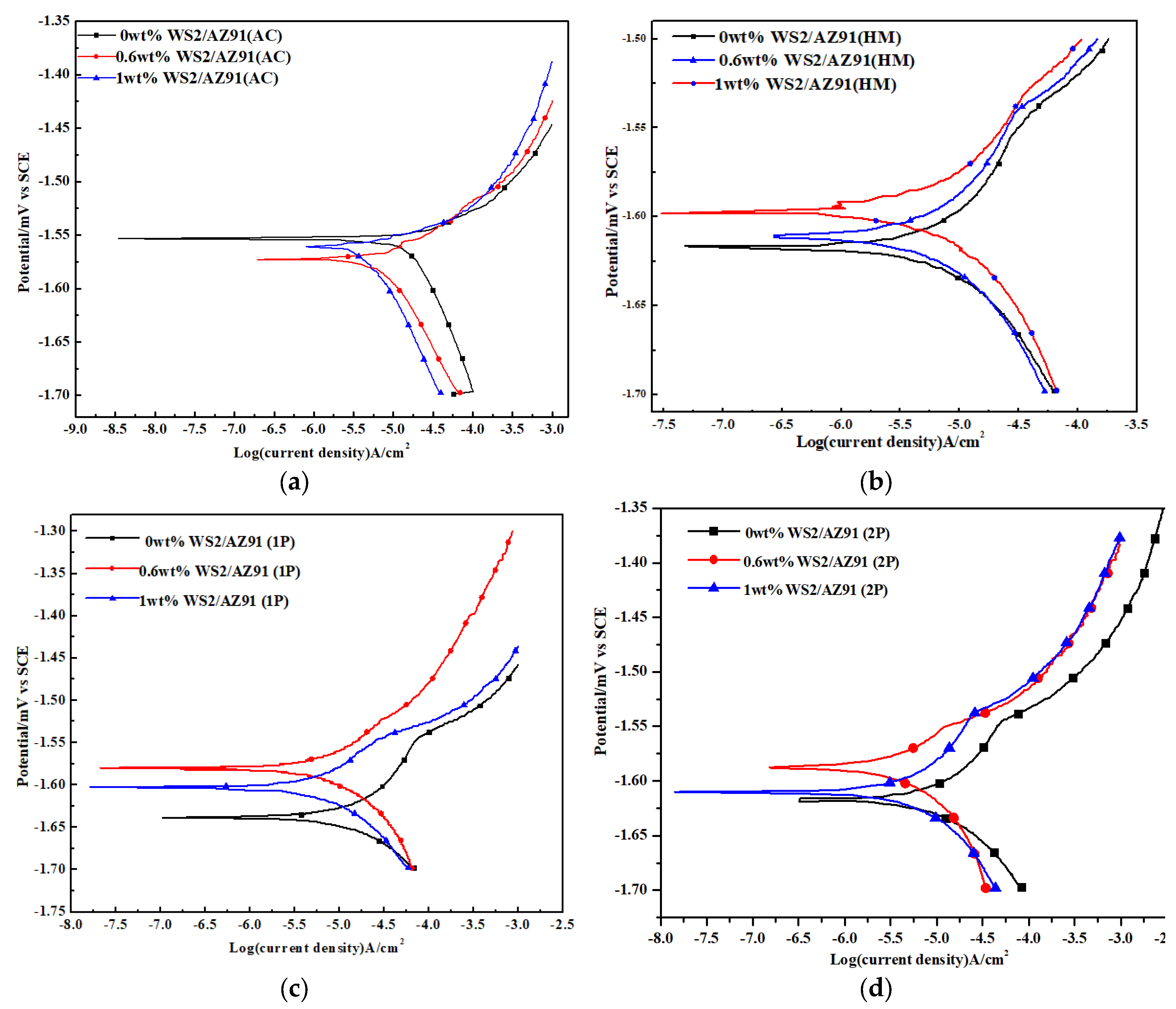

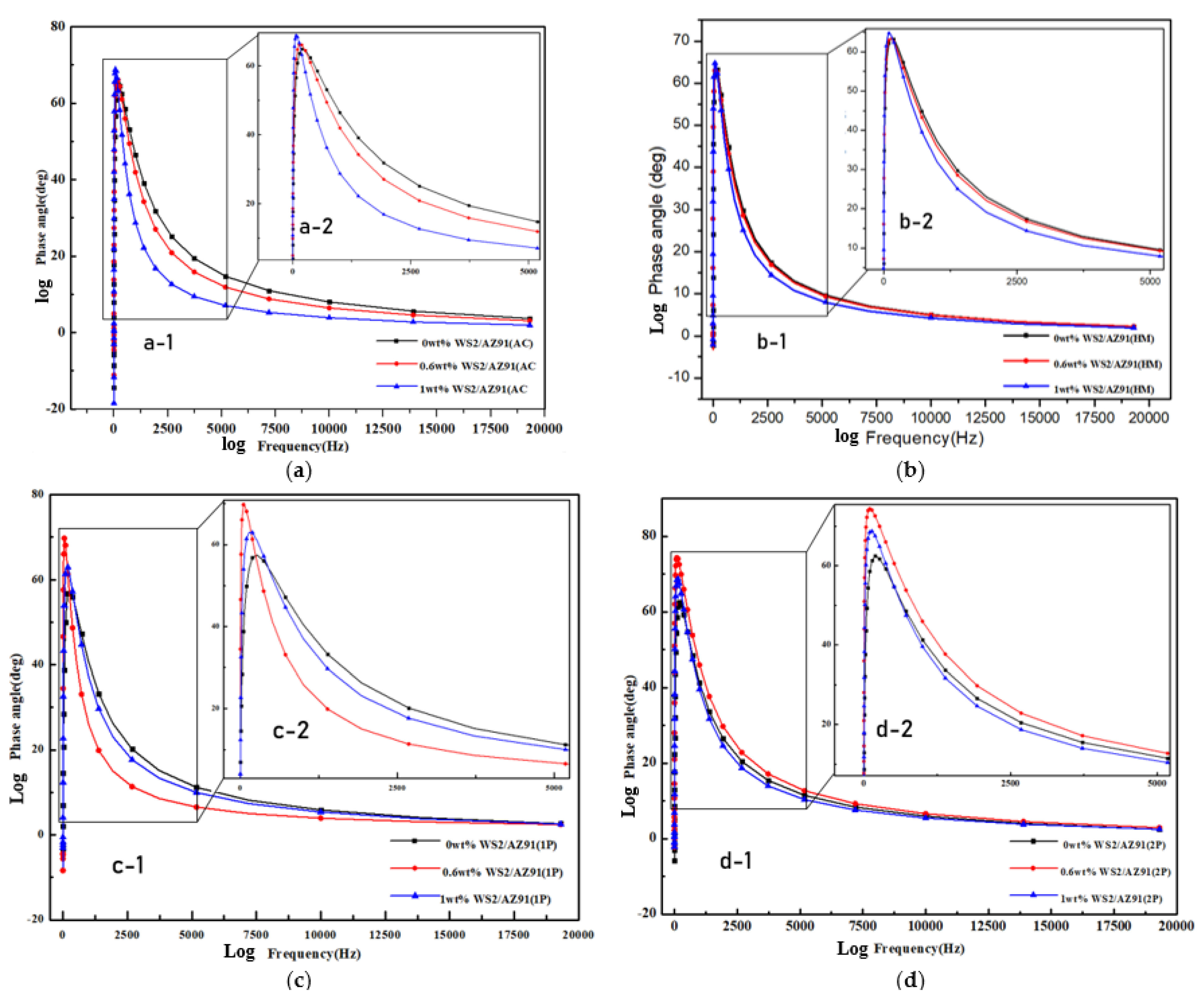
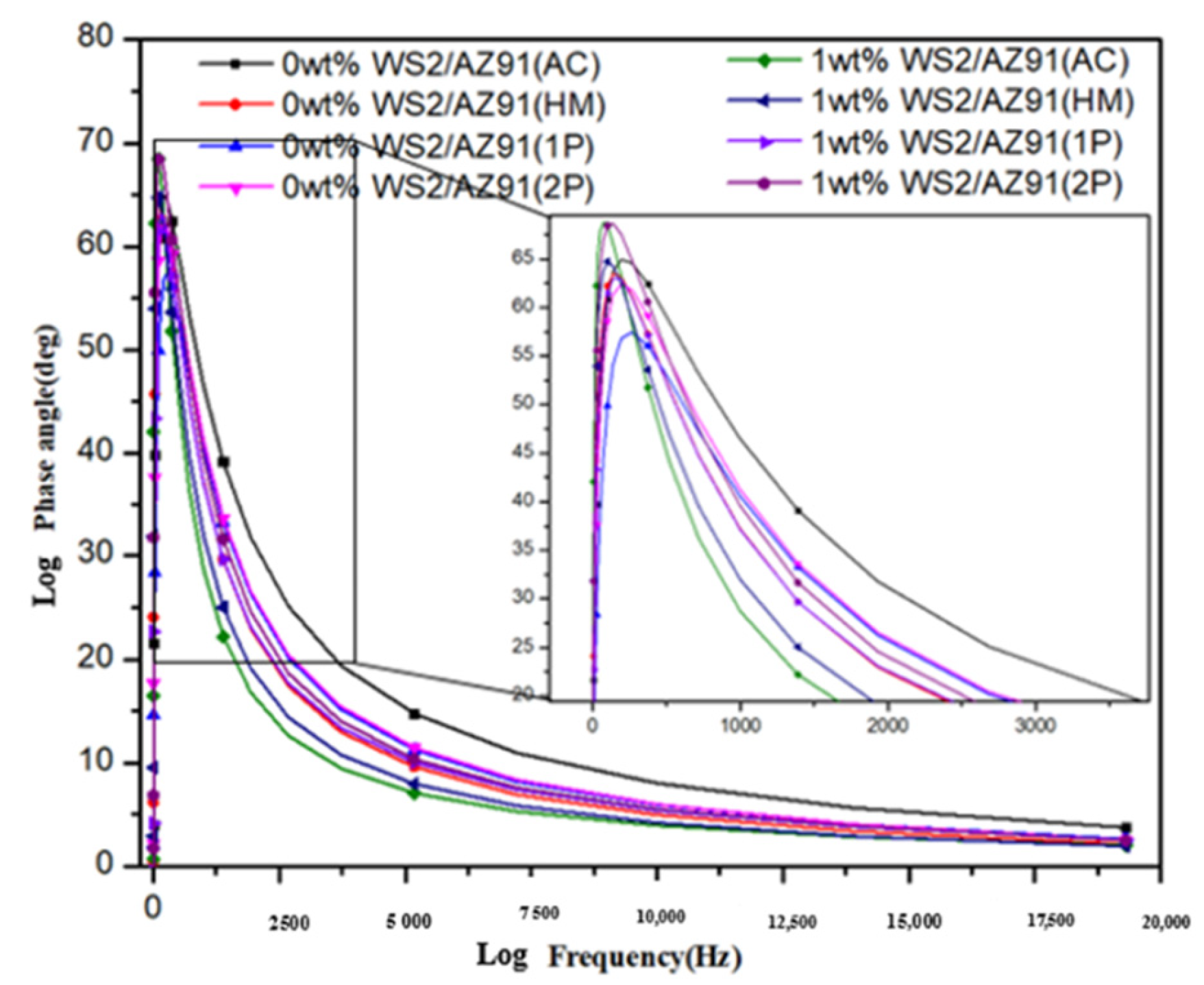

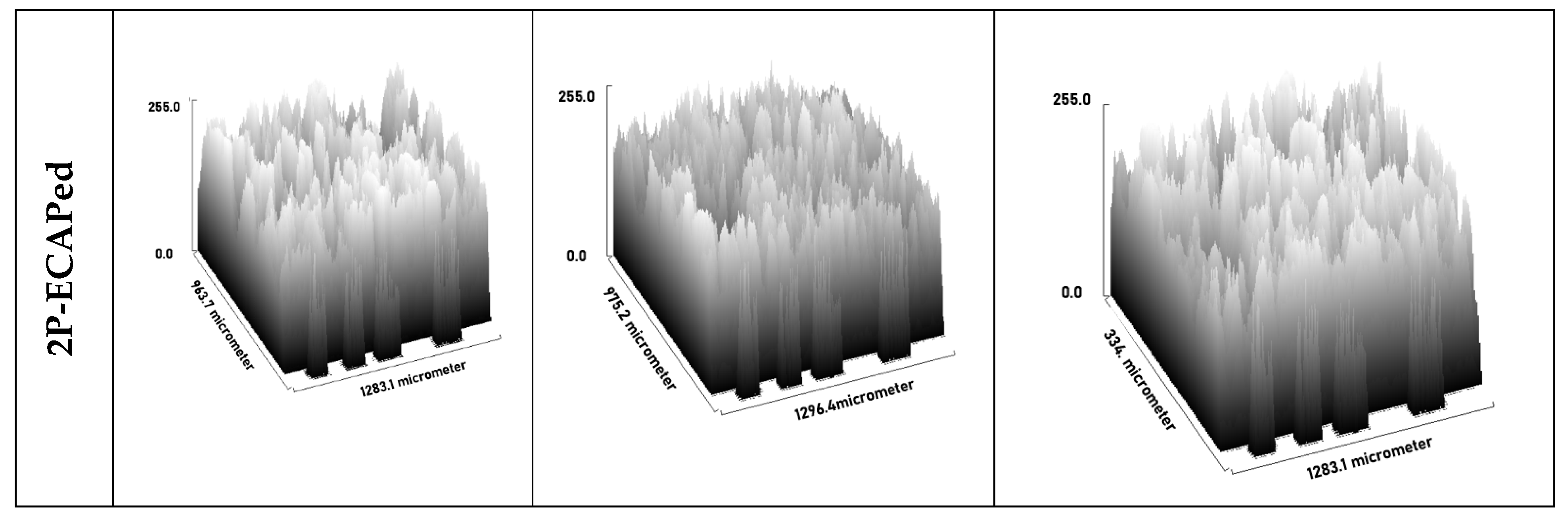
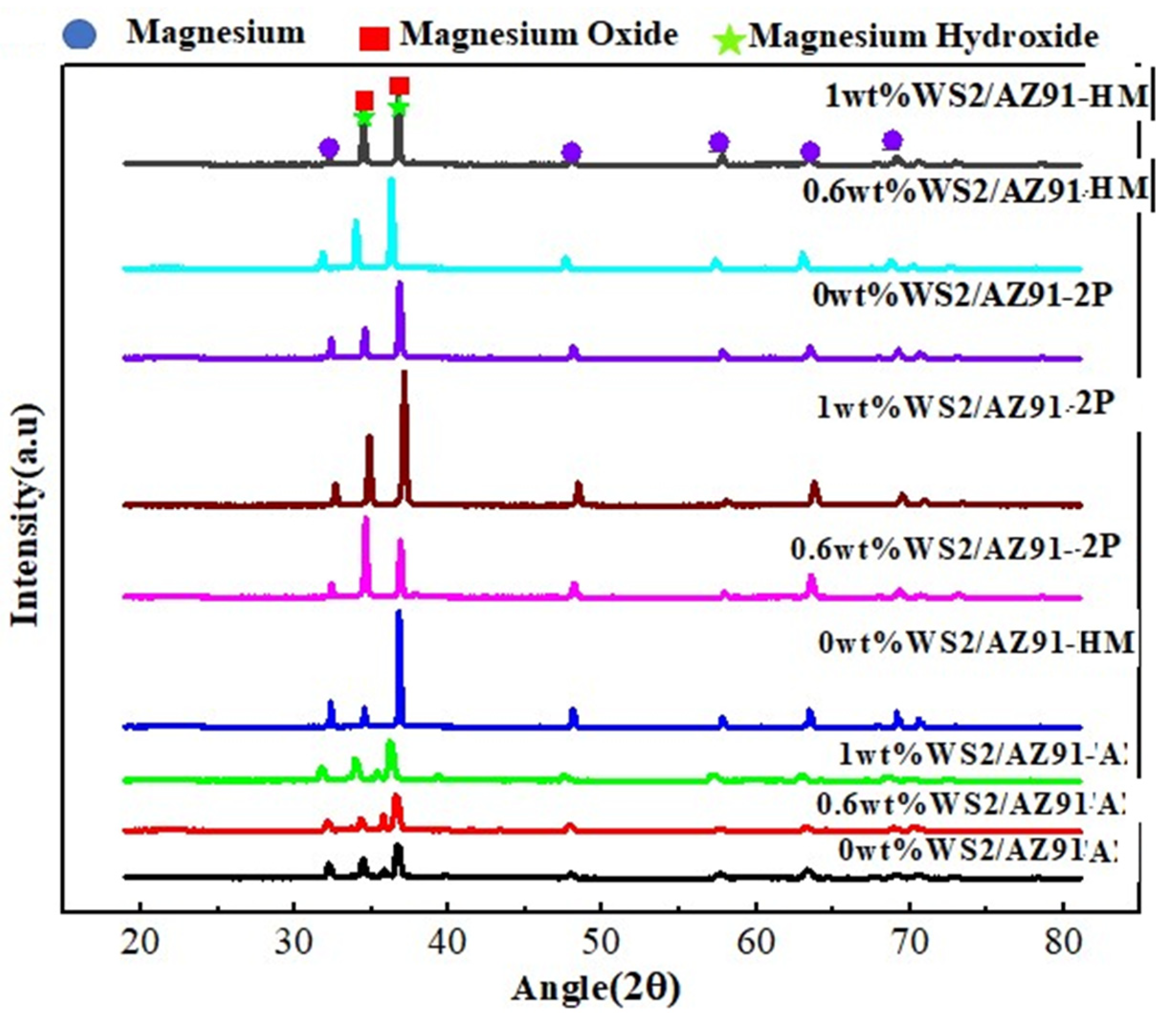
| Composite | Condition | Ecorr mV) | Icorr (µA·cm−2) | R (m·a−1) | Βc (mV·dec−1) | Βa (mV·dec−1) | Ra (µm) |
|---|---|---|---|---|---|---|---|
| 0 wt% WS2/AZ91 | as-cast | −1.55334 ± 0.00011 | 1.232 ± 0.001 | 4.09955 ± 0.0005 | 0.4272 ± 0.0002 | 0.6321 ± 0.0002 | 33.90 ± 0.1 |
| 0.6 wt% WS2/AZ91 | −1.57288 ± 0.00011 | 1. 954 ± 0.001 | 4.16262 ± 0.0005 | 0.4384 ± 0.0002 | 0.5935 ± 0.0002 | 34.55 ± 0.1 | |
| 1 wt% WS2/AZ91 | −1.56097 ± 0.00011 | 1.286 ± 0.001 | 4.1446 ± 0.0005 | 0.4394 ± 0.0002 | 0.5934 ± 0.0002 | 36.31 ± 0.1 | |
| 0 wt% WS2/AZ91 | Homogenized | −1.61652 ± 0.00008 | 4.430 ± 0.001 | 4.51401 ± 0.0005 | 0.4284 ± 0.0001 | 0.5493 ± 0.0001 | 36.95 ± 0.2 |
| 0.6 wt% WS2/AZ91 | −1.59821 ± 0.00008 | 3.110 ± 0.001 | 4.45995 ± 0.0005 | 0.4566 ± 0.0001 | 0.6487 ± 0.0001 | 40.63 ± 0.2 | |
| 1 wt% WS2/AZ91 | −1.61163 ± 0.00008 | 4.295 ± 0.001 | 4.5120 ±0.0005 | 0.5548 ± 0.0001 | 0.7432 ± 0.0001 | 41.84 ± 0.2 | |
| 0 wt% WS2/AZ91 | 1-pass ECAPed | −1.63849 ± 0.00012 | 5.80 ± 0.002 | 4.613 ± 0.0003 | 0.1779 ± 0.0002 | 0.3543 ± 0.0002 | 38.43 ± 0.1 |
| 0.6 wt% WS2/AZ91 | −1.5799 ± 0.00012 | 3.237 ± 0.002 | 4.3248 ± 0.0003 | 0.3267 ± 0.0002 | 0.6633 ± 0.0002 | 41.25 ± 0.1 | |
| 1 wt% WS2/AZ91 | −1.60278 ± 0.00012 | 3.103 ± 0.002 | 4.5043 ± 0.0003 | 0.1890 ± 0.0002 | 0.4317 ± 0.0002 | 43.23 ± 0.1 | |
| 0 wt% WS2/AZ91 | 2-pass ECAPed | −1.61865 ± 0.00012 | 3.132 ± 0.002 | 4.52302 ± 0.0002 | 0.2126 ± 0.0003 | 0.6249 ± 0.0003 | 42.28 ± 0.1 |
| 0.6 wt% WS2/AZ91 | −1.58752 ± 0.00011 | 2.219 ± 0.002 | 4.43292 ± 0.0002 | 0.2033 ± 0.0003 | 0.7585 ± 0.0003 | 42.59 ± 0.1 | |
| 1 wt% WS2/AZ91 | −1.61011 ± 0.00011 | 3.153 ± 0.002 | 4.5951 ± 0.0002 | 0.1995 ± 0.0003 | 0.5409 ± 0.0003 | 47.37 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbas, A.; Huang, S.-J. Investigating the Synergic Effects of WS2 and ECAP on Degradation Behavior of AZ91 Magnesium Alloy. Coatings 2022, 12, 1710. https://doi.org/10.3390/coatings12111710
Abbas A, Huang S-J. Investigating the Synergic Effects of WS2 and ECAP on Degradation Behavior of AZ91 Magnesium Alloy. Coatings. 2022; 12(11):1710. https://doi.org/10.3390/coatings12111710
Chicago/Turabian StyleAbbas, Aqeel, and Song-Jeng Huang. 2022. "Investigating the Synergic Effects of WS2 and ECAP on Degradation Behavior of AZ91 Magnesium Alloy" Coatings 12, no. 11: 1710. https://doi.org/10.3390/coatings12111710
APA StyleAbbas, A., & Huang, S.-J. (2022). Investigating the Synergic Effects of WS2 and ECAP on Degradation Behavior of AZ91 Magnesium Alloy. Coatings, 12(11), 1710. https://doi.org/10.3390/coatings12111710


.jpg)




