1. Introduction
Material surface friction and wear [
1] and corrosion are the two main reasons for the failure of mechanical parts [
1,
2]. In the current environment of rapid development of aerospace, automobile, and high-end equipment manufacturing industries, the requirements for wear resistance and corrosion resistance of mechanical parts in various fields are constantly increasing [
3,
4,
5]. Sabyasachi Ghosh et al. reported that phase-separated PEDOT:PSS ornamented with reduced graphene oxide (rGO) nanosheets, deposited on the merino wool/nylon (W−N) composite textile. This developed fabric could perform as a new potentially scalable single product in intelligent smart garments, portable next-generation electronics, and the growing threat of EM pollution. They also reported a route to fabricate rGO/Ag decorated cotton fabric having high electrical conductivity and superior electromagnetic shielding efficiency [
6,
7]. Shivam Tiwari et al. reported that the fluorescent nitrogen-doped carbon dots which have been produced by a bottom-up wet-chemical method employing citric acid as a precursor could be used for specific applications in different research fields such as sensors, solar cells, biomedicine, and catalysis [
8]. The use of electrodeposition technology [
9] to deposit enhanced coatings on metal substrates has been widely used because of its high efficiency and economy. Among them, nickel nanocomposite coatings [
10] are particularly widely used because of their high wear resistance and corrosion resistance.
As a new type of zero-dimensional carbon-based material [
11,
12], graphene quantum dots have a size of less than 100 nm; due to its size, it has obvious quantum confinement effect and sideband effect, which also makes graphene quantum dots have more emerging physicochemical properties (such as chemical inertness, excellent water solubility). Deposition of neutral particles (such as nitrides, carbides, etc.,) in the coatings can improve the grain growth process and effectively improve the mechanical and chemical properties of the coating [
13]. Incorporating graphene quantum dots with excellent properties as a second phase additive into the nickel coating can effectively improve its mechanical properties [
14] and corrosion resistance. Electrodeposition technology has the problem that the second phase additives are easy to agglomerate and affect the composite coatings. The study found that the supercritical fluid technology [
15] can make the electrodeposition solution form an excellent emulsification system [
16] through reasonable system temperature, pressure, and suitable surfactant. The emulsification system enables the electrodeposition system to have better mass transfer performance and can effectively separate the second additive, thereby improving the agglomeration of the second phase additive. Direct current (DC) electrodeposition is easy to generate pores, and the coatings have high residual stress. The study found that pulse electrodeposition [
17] compared with DC electrodeposition, the prepared coatings have the advantages of low residual stress [
18], smooth surface, and tight bonding between the coatings and the base metals. The pulse duty cycle controls the on-off time of the current and has a great influence on the cathode overpotential. A suitable pulse duty cycle can significantly improve the quality of coatings.
In previous studies, it was found that the properties of the coatings prepared by pulse electrodeposition was better than that of the coating prepared by DC electrodeposition. For example, in terms of microhardness, under the same conditions, pure Ni coatings prepared with better parameters are used. The microhardness of pure Ni coatings prepared by pulse electrodeposition is about 100 HV higher than that of pure Ni coatings prepared by DC electrodeposition. Moreover, it was found that supercritical technology has a significant effect on the dispersion of graphene quantum dots. In this work, graphene quantum dots were used as the second phase additive, and the Ni-based composite coating of graphene quantum dots was prepared by pulsed electrodeposition technology with the assistance of supercritical CO2 fluid. Through preliminary research, it is preliminarily determined that the optimal current parameters are current density 6 A/dm2; frequency 2000 Hz; plating time 60 min; optimal supercritical condition parameters are pressure 10 MPa and temperature 50 ℃. By comparing the microstructure, mechanical properties, and corrosion resistance of coatings prepared with different pulse duty cycles, a more suitable pulse duty cycle was explored. To further help with the research and development of metal matrix nanocomposite coatings, they were enhanced by adding second phase additives by pulse electrodeposition as a processing method.
2. Materials and Methods
2.1. Sample Preparation
The preparation formula of graphene quantum dots(GQDs) is shown in
Table 1. Add citric acid (C
6H
8O
7·H
2O) and urea (CH
4N
2O) to 5 mL of deionized water (H
2O), stir until clear, then place in a polytetrafluoroethylene reactor, seal the reactor, and place it in an oven at 180 °C for 8 h. After the heat preservation, take out the reacted liquid, pour it into alcohol, put it into a centrifuge for centrifugation, wash the separated graphene quantum dots with alcohol, dry them, put them in a sealed bottle, and store them in a dry and dark place for future use.
The supercritical CO
2-assisted electrodeposition device is shown in
Figure 1. The temperature measurement device, temperature control device, cooling system, and air pressure pump, respectively, meet the temperature and pressure requirements for supercritical electrodeposition work. In this experiment, the anode and cathode materials were 20 mm × 20 mm copper sheet and 25 mm × 25 mm nickel plate with a purity of 99.9%, and the distance between the cathode and anode was 20 mm. After rust removal, oil removal, oxide film removal, sandpaper grinding (1200–5000 mesh), and polishing for copper sheets, after rust removal, oil removal, and oxide film removal for nickel plates, use insulating glue to fix the copper sheet and the nickel plate on the working cathode and anode respectively, and ensure that the cathode and anode were well energized. The Chunlin ultrasonic cleaner and EMS-12 split magnetic stirrer were used to fully stir the plating solution and fully disperse the graphene quantum dots in the plating solution. TMN surfactant was added to the plating solution before plating. The formula of the bath and the process parameters of the coatings (Ni-GQDs- I, Ni-GQDs- II, Ni-GQDs- III, Ni-GQDs- IV and Ni) prepared by different preparation processes are shown in
Table 2 and
Table 3, respectively.
2.2. Analysis and Characterization
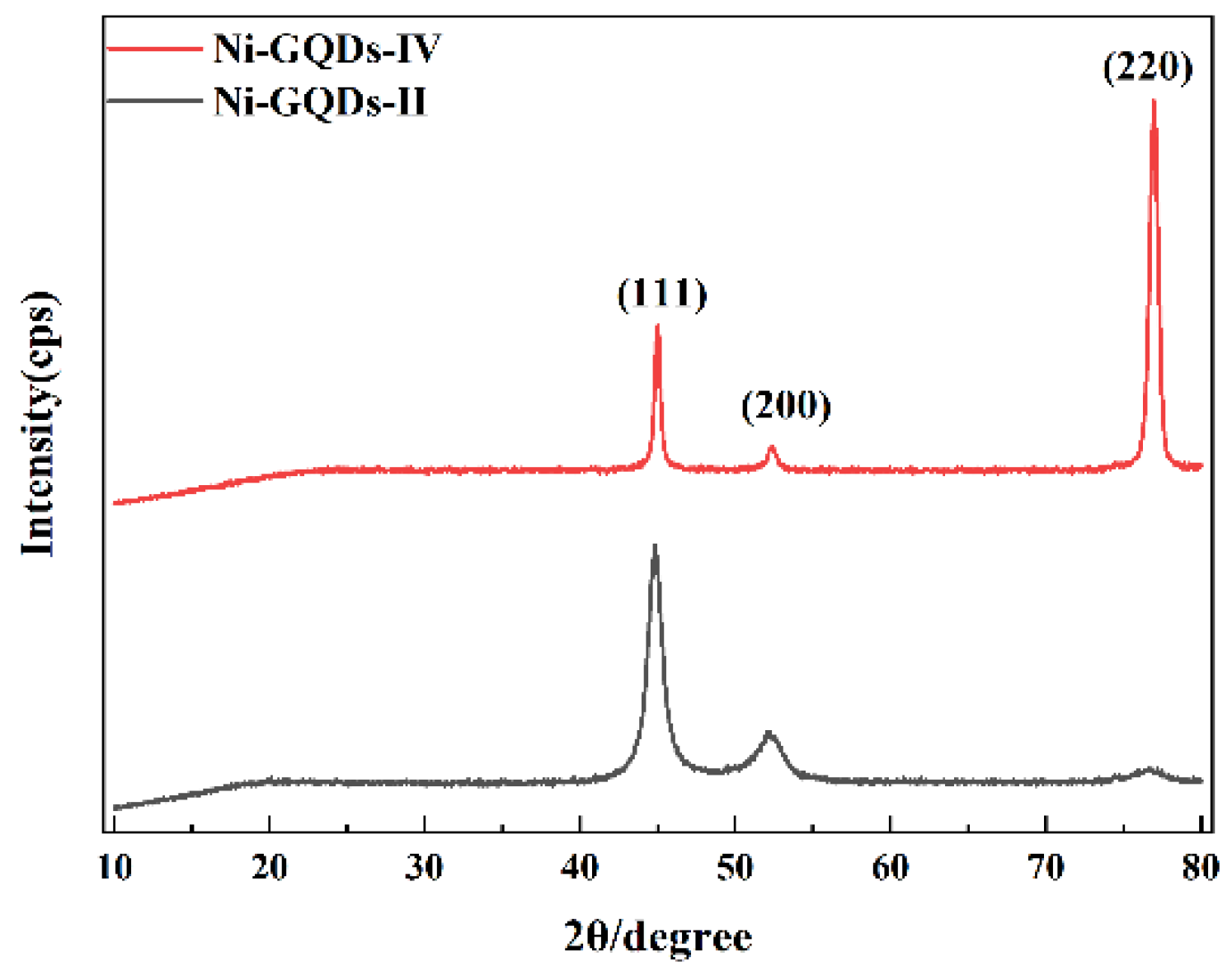
After the samples were ion-thinned by Gatan 691 ion-thinning instrument, the state of graphene quantum dots in the coating was observed by Talos F200× G2 high-resolution transmission electron microscope (Talos, Beijing, China), and the elements of the coating were analyzed by SDD super-X energy spectrometer (EDAX, Beijing, China). The microscopic morphology of the coating and the coating after immersion corrosion were observed by Sigma-500 scanning electron microscope (Zeiss, Oberkochen, Germany). The carbon content of the coating was analyzed by Like CS844 carbon and sulfur analyzer (LECO, Laboratory Equipment Corporaton, Saint Joseph, MI, USA). The chemical structure of the coating was determined by HR Evolution Raman spectrometer (HORIBA, Kyoto, Japan). The selected laser was He-Ne, the wavelength was 633 nm, the spot size was 1 μm, and the scanning range was 1000–3000 cm−1. The nickel crystal preferred orientation of the coating was tested by X’PERT POWDER X-Ray diffractometer (Malvern, Shanghai, China)target was used, the scanning range was 20°–80°, and the step size was 0.0131303°.
HXD-1000TMS/LCD digital microhardness tester (Yongheng Optical Instrument Manufacturing Co., Ltd, Shanghai, China) was used to test the hardness of the coating, and each sample selected 5 points at different positions for testing, and the average value of these 5 points is the hardness value of the sample. The Nanovea TRB friction (Nanovea, Irvine, CA, USA) and wear testing machine was used to test the wear resistance of the coating. The steel ball with a diameter of 6 mm was used as the abrasive piece. The speed was 120 r/min, the load was 5 N, and the test time was 10 min. The Nanovea PS50 optical profiler (Nanovea, Irvine, CA, USA) was used to scan the surface topography of the wear scar. The scanning area was 2 mm × 2 mm, the step size was 5 μm, and the scanning rate was 3.33 mm/s. The Nyquist spectrum, Tafel polarization curve, and Bode spectrum were tested on the CHI760E electrochemical workstation. The coatings were immersed in a 3.5 g/L NaCl solution for 120 h at normal temperature and pressure, rinsed with alcohol after soaking, and then observed by a scanning electron microscope for the microscopic morphology of the coatings.