Characterization and In Vitro Studies of Low Reflective Magnetite (Fe3O4) Thin Film on Stainless Steel 420A Developed by Chemical Method
Abstract
1. Introduction
2. Methodology
2.1. Sample Preparation and Coating Procedure
2.2. Taguchi Design of Experiments
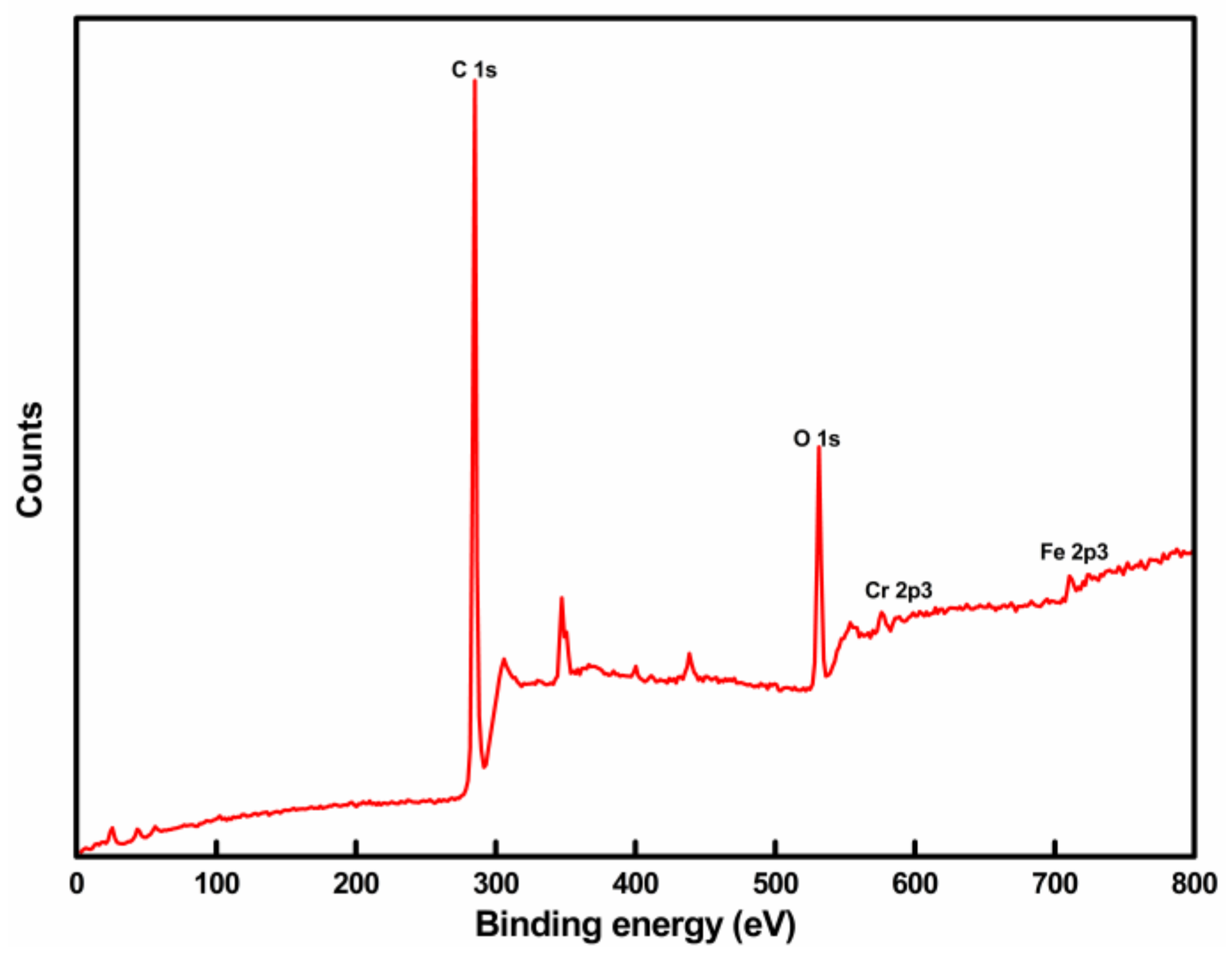
2.3. X-ray Photoelectron Spectroscopy (XPS)
2.4. Cytotoxity Assessment
2.5. Corrosion Analysis
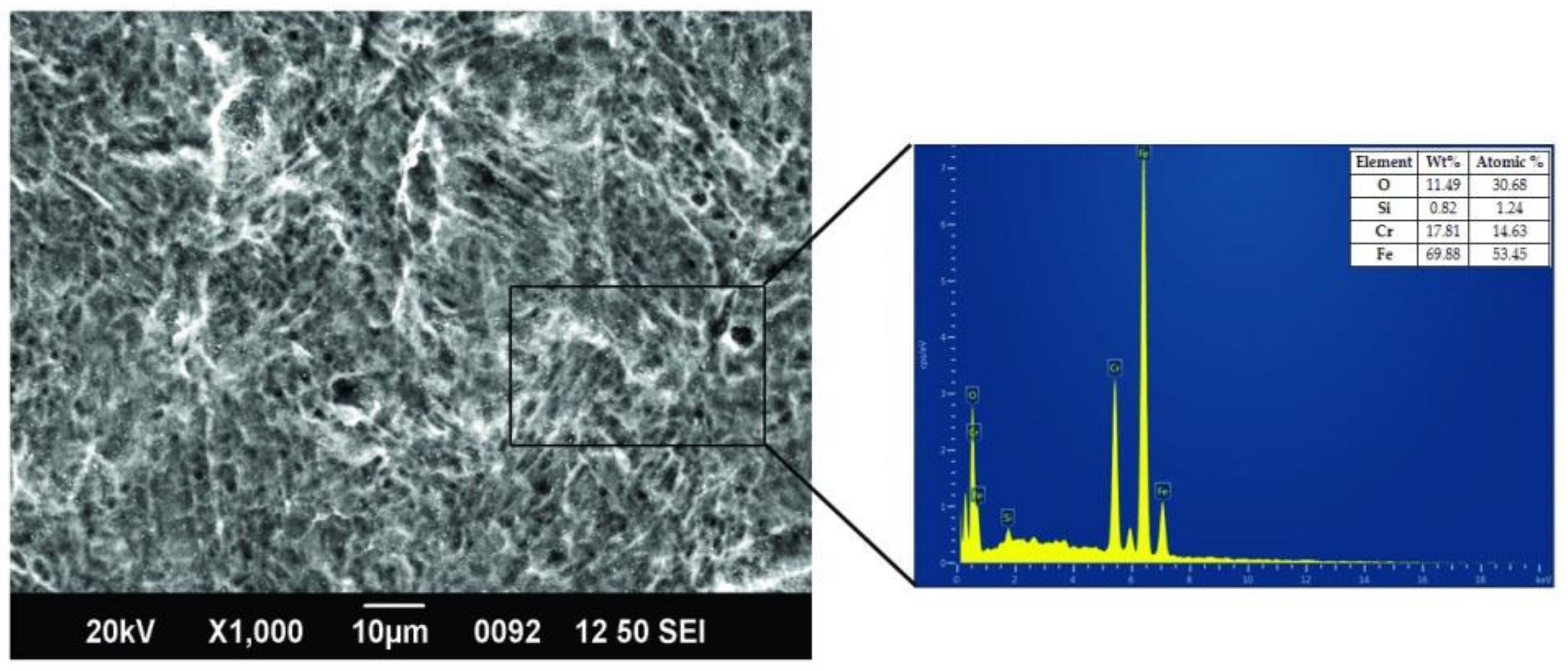
2.6. Repeated Sterilization and Morphology Studies
3. Results and Discussion
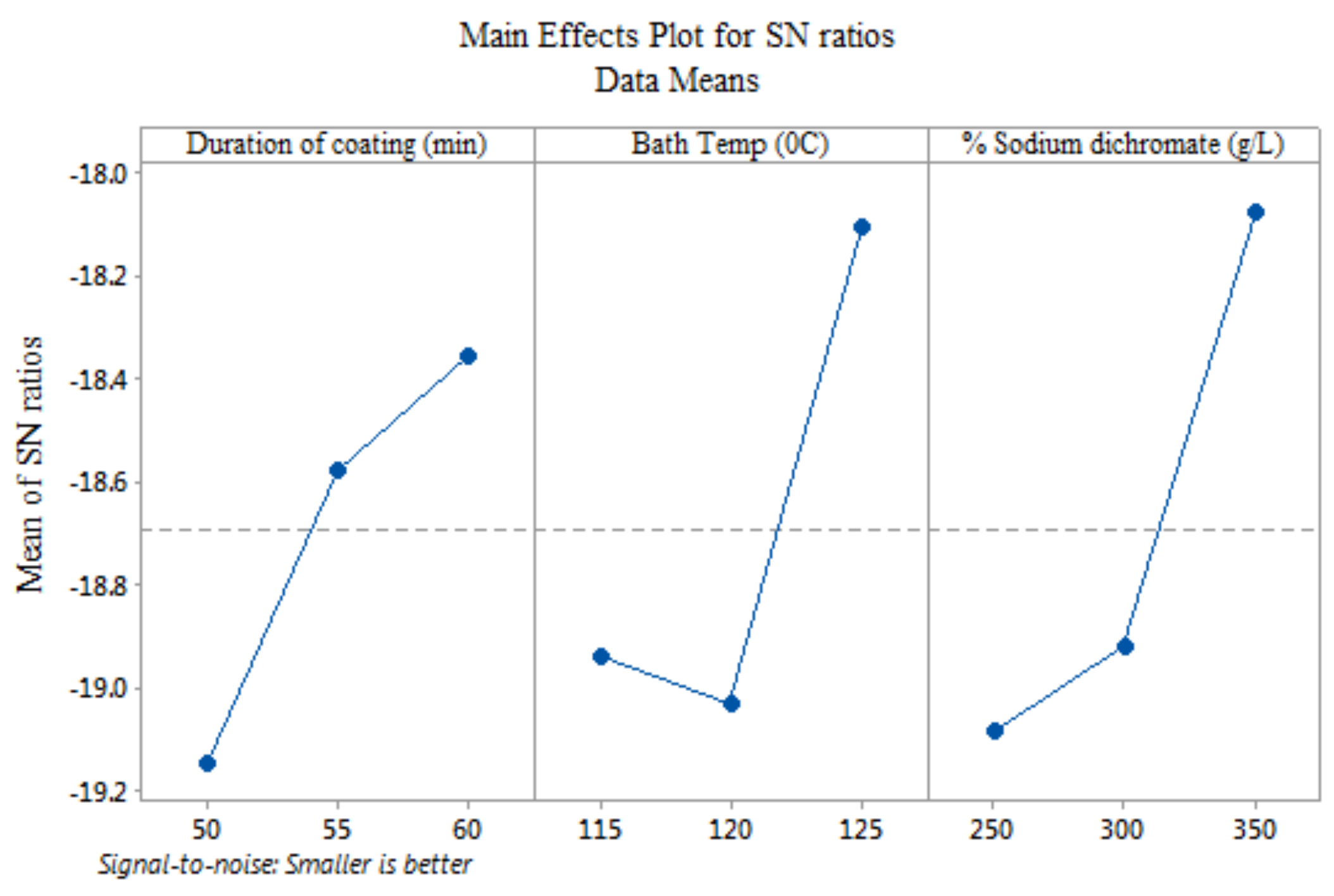
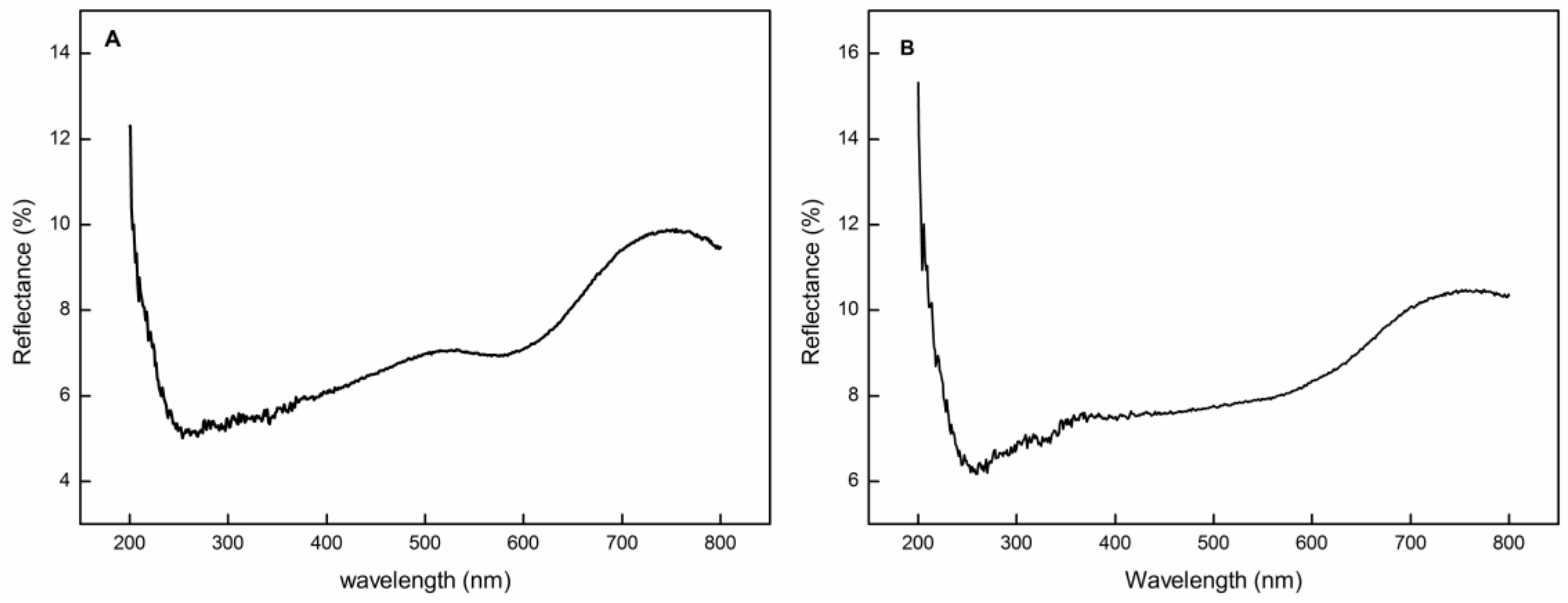
3.1. Process Parameter Optimization
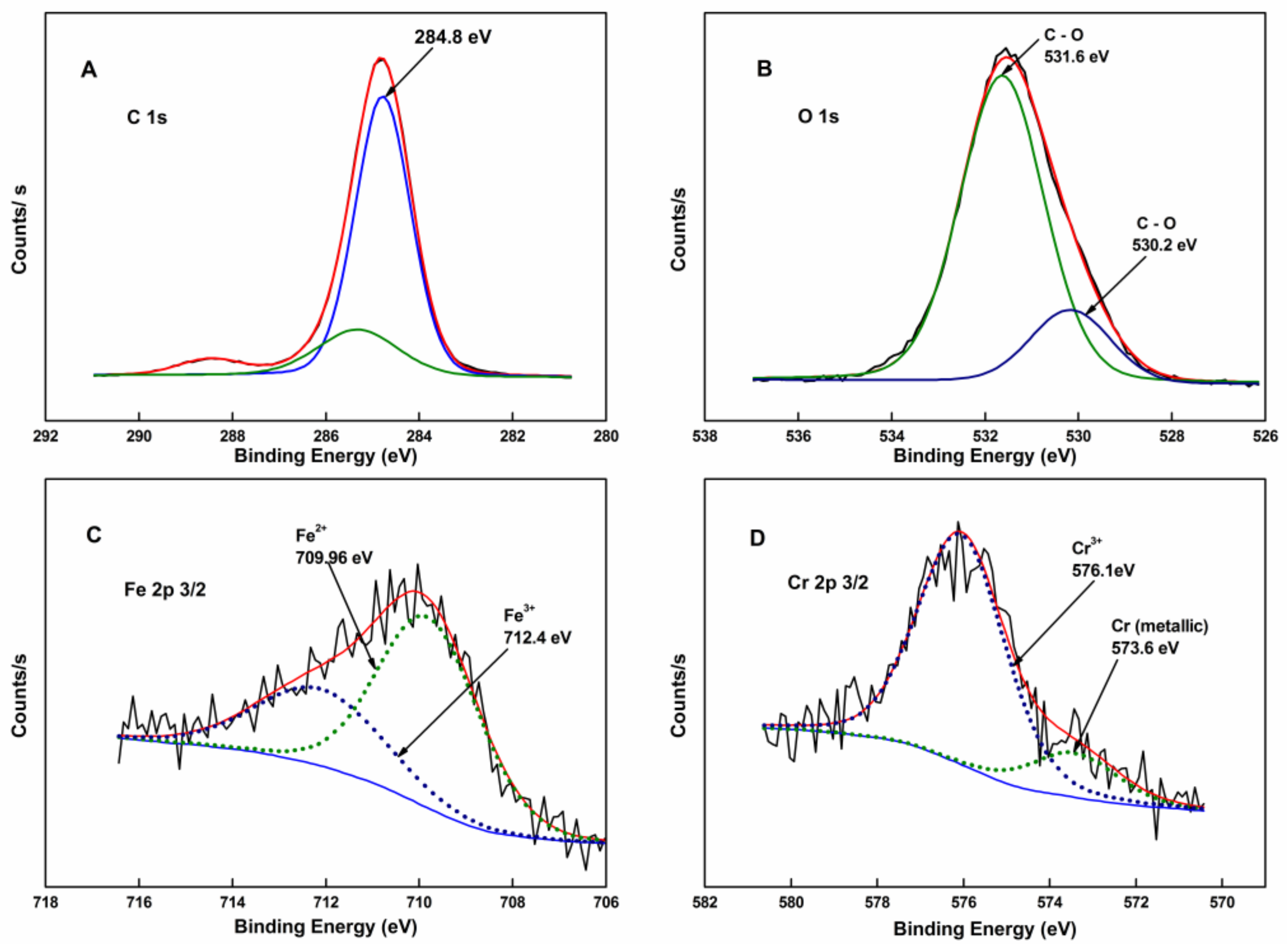
3.2. XPS Analysis of Coating
3.3. In Vitro Cytotoxicity Study
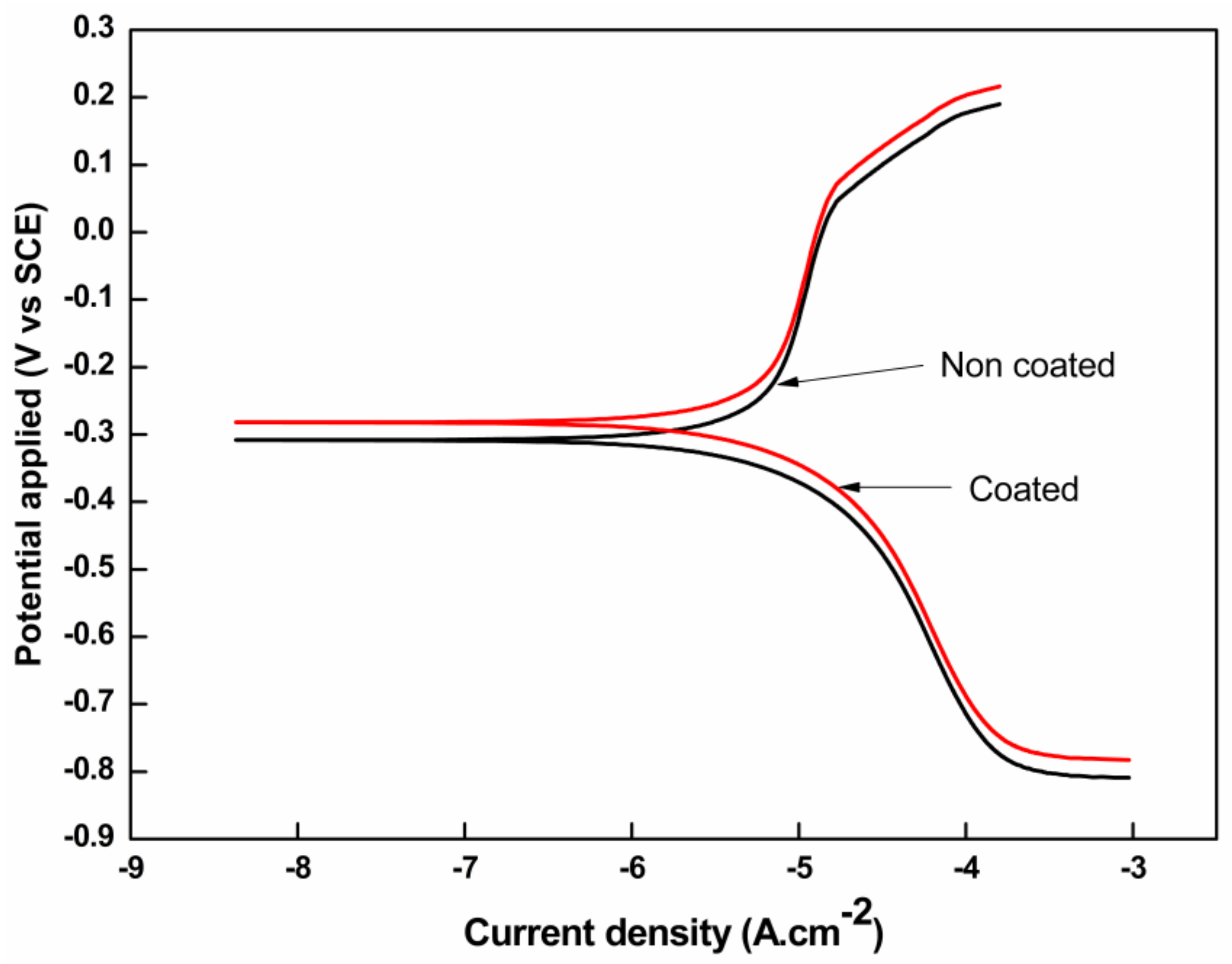
3.4. Potentiodynamic Polarization and Salt Spray Studies
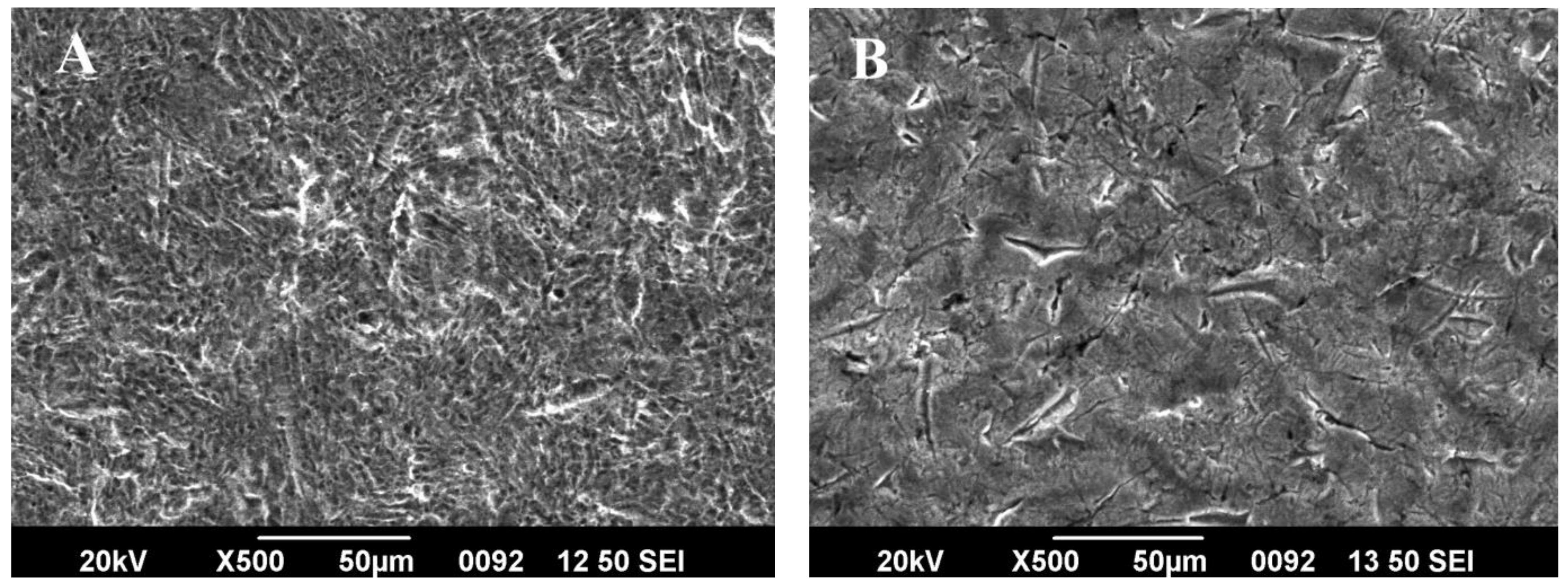
3.5. Effect of Repeated Sterilization on Coating
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability and Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| SS | Stainless steel |
| XPS | X-ray photoelectron spectroscopy |
| R | Average percentage reflection |
| AISI | American iron and steel institute |
| DLC | Diamond-Like Carbon |
| CVD | Chemical vapor deposition |
| PVD | Physical vapor deposition |
| wt.% | Weight percentage |
| D | Duration of coating |
| T | Bath temperature |
| C | wt% of sodium dichromate |
| S/N | Signal-to-noise |
| OA | Orthogonal Array |
| DF | Degrees of freedom |
| SS | Sum of squares |
| MS | Mean square |
| F | Fisher’s ratio |
| P | Probability value |
| MEM | Minimum essential medium |
| FBS | Fetal bovine serum |
| UHMWPE | Ultra-high molecular weight polyethylene |
| SCE | Saturated calomel electrode |
| PDP | Potentiodynamic polarization |
| SBF | Simulated body fluid |
| OCP | Open circuit potential |
| Icorr | Corrosion current |
| Ecorr | Corrosion potential |
| Rp | Polarization resistance |
| βa | Anodic Tafel slope |
| βc | Cathodic Tafel slope |
| CR | Corrosion rate |
References
- Festas, A.; Ramos, A.; Davim, J.P. Medical devices biomaterials—A review. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 2019, 234, 218–228. [Google Scholar] [CrossRef]
- Chauhan, L.R.; Singh, M.; Bajpai, J.K.; Misra, K.; Agarwal, A. Development of chemical conversion coating for blackening of a grade of stainless steel useful for fabrication of optical devices. J. Surf. Sci. Technol. 2017, 32, 99. [Google Scholar] [CrossRef]
- Ali, S.; Rani, A.M.A.; Baig, Z.; Ahmed, S.W.; Hussain, G.; Subramaniam, K.; Hastuty, S.; Rao, T.V. Biocompatibility and corrosion resistance of metallic biomaterials. Corros. Rev. 2020, 38, 381–402. [Google Scholar] [CrossRef]
- Zhang, L.-C.; Chen, L.-Y.; Wang, L. Surface modification of titanium and titanium alloys: Technologies, developments, and future interests. Adv. Eng. Mater. 2020, 1–16. [Google Scholar] [CrossRef]
- Lim, J.J.; Erdman, A.G. A review of mechanism used in laparoscopic surgical instruments. Mech. Mach. Theory 2003, 38, 1133–1147. [Google Scholar] [CrossRef]
- Hollstein, F.; Louda, P. Bio-compatible low reflective coatings for surgical tools using reactive d.c.-magnetron sputtering and arc evaporation—a comparison regarding steam sterilization resistance and nickel diffusion. Surf. Coat. Technol. 1999, 120–121, 672–681. [Google Scholar] [CrossRef]
- Reghuraj, A.; Saju, K. Black oxide conversion coating on metals: A review of coating techniques and adaptation for SAE 420A surgical grade stainless steel. Mater. Today Proc. 2017, 4, 9534–9541. [Google Scholar] [CrossRef]
- Ooi, S.W.; Yan, P.; Vegter, R.H. Black oxide coating and its effectiveness on prevention of hydrogen uptake. Mater. Sci. Technol. 2018, 35, 12–25. [Google Scholar] [CrossRef]
- Lira-Cantú, M.; Sabio, A.M.; Brustenga, A.; Gomez-Romero, P. Electrochemical deposition of black nickel solar absorber coatings on stainless steel AISI316L for thermal solar cells. Sol. Energy Mater. Sol. Cells 2005, 87, 685–694. [Google Scholar] [CrossRef]
- Nagode, A.; Klančnik, G.; Schwarczova, H.; Kosec, B.; Gojić, M.; Kosec, L. Analyses of defects on the surface of hot plates for an electric stove. Eng. Fail. Anal. 2012, 23, 82–89. [Google Scholar] [CrossRef]
- Lebbai, M.; Kim, J.-K.; Szeto, W.K.; Yuen, M.M.F.; Tong, P. Optimization of black oxide coating thickness as an adhesion promoter for copper substrate in plastic integrated-circuit packages. J. Electron. Mater. 2003, 32, 558–563. [Google Scholar] [CrossRef]
- Kim, J.-K.; Woo, R.S.; Hung, P.Y.; Lebbai, M. Adhesion performance of black oxide coated copper substrates: Effects of moisture sensitivity test. Surf. Coat. Technol. 2006, 201, 320–328. [Google Scholar] [CrossRef]
- Zaffora, A.; Di Franco, F.; Virtù, D.; Pavia, F.C.; Ghersi, G.; Virtanen, S.; Santamaria, M. Tuning of the Mg alloy AZ31 anodizing process for biodegradable implants. ACS Appl. Mater. Interfaces 2021, 13, 12866–12876. [Google Scholar] [CrossRef]
- Sarkar, S.; Mukherjee, A.; Baranwal, R.K.; De, J.; Biswas, C.; Majumdar, G. Prediction and parametric optimization of surface roughness of electroless Ni-Co-P coating using Box-Behnken design. J. Mech. Behav. Mater. 2019, 28, 153–161. [Google Scholar] [CrossRef]
- Kaneko, M.; Hiratsuka, M.; Alanazi, A.; Nakamori, H.; Namiki, K.; Hirakuri, K. Surface reformation of medical devices with DLC coating. Materials 2021, 14, 376. [Google Scholar] [CrossRef]
- Sharma, A.; Rani, R.U.; Mayanna, S. Thermal studies on electrodeposited black oxide coating on magnesium alloys. Thermochim. Acta 2001, 376, 67–75. [Google Scholar] [CrossRef]
- Tepe, B.; Gunay, B. Evaluation of pre-treatment processes for HRS (hot rolled steel) in powder coating. Prog. Org. Coat. 2008, 62, 134–144. [Google Scholar] [CrossRef]
- Li, G.; Niu, L.; Lian, J.; Jiang, Z. A black phosphate coating for C1008 steel. Surf. Coat. Technol. 2004, 176, 215–221. [Google Scholar] [CrossRef]
- Zhao, M.; Wu, S.; Luo, J.; Fukuda, Y.; Nakae, H. A chromium-free conversion coating of magnesium alloy by a phosphate–permanganate solution. Surf. Coat. Technol. 2006, 200, 5407–5412. [Google Scholar] [CrossRef]
- Mohsin, I.; He, K.; Li, Z.; Zhang, F.; Du, R. Optimization of the polishing efficiency and torque by using taguchi method and ANOVA in robotic polishing. Appl. Sci. 2020, 10, 824. [Google Scholar] [CrossRef]
- Zalnezhad, E.; Sarhan, A.A.D.; Hamdi, M. Optimizing the PVD TiN thin film coating’s parameters on aerospace AL7075-T6 alloy for higher coating hardness and adhesion with better tribological properties of the coating surface. Int. J. Adv. Manuf. Technol. 2012, 64, 281–290. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, R.; Jaiswal, R.; Kumar, A. Optimization of process parameters of electron beam welded Fe49Co2V alloys. Int. J. Eng. Trans. B Appl. 2020, 33, 870–876. [Google Scholar] [CrossRef]
- Liu, X.; Rodeheaver, D.P.; White, J.C.; Wright, A.M.; Walker, L.M.; Zhang, F.; Shannon, S. A comparison of in vitro cytotoxicity assays in medical device regulatory studies. Regul. Toxicol. Pharmacol. 2018, 97, 24–32. [Google Scholar] [CrossRef]
- Vidal, M.N.P.; Granjeiro, J.M. Cytotoxicity tests for evaluating medical devices: An alert for the development of biotechnology health products. J. Biomed. Sci. Eng. 2017, 10, 431–443. [Google Scholar] [CrossRef]
- Farzam, M.; Baghery, P.; Dezfully, H.R.M. Corrosion study of steel API 5A, 5L and AISI 1080, 1020 in drill-mud environment of Iranian hydrocarbon fields. Int. Sch. Res. Not. 2011, 2011, 681535. [Google Scholar] [CrossRef]
- Zaludin, M.A.F.; Jamal, Z.A.Z.; Derman, M.N.; Kasmuin, M.Z. Fabrication of calcium phosphate coating on pure magnesium substrate via simple chemical conversion coating: Surface properties and corrosion performance evaluations. J. Mater. Res. Technol. 2019, 8, 981–987. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.; Crow, N.; Nieto, A. Effect of surface roughness on pitting corrosion of AZ31 Mg Alloy. Metals 2020, 10, 651. [Google Scholar] [CrossRef]
- Vannozzi, L.; Catalano, E.; Telkhozhayeva, M.; Teblum, E.; Yarmolenko, A.; Avraham, E.S.; Konar, R.; Nessim, G.D.; Ricotti, L. Graphene oxide and reduced graphene oxide nanoflakes coated with glycol chitosan, propylene glycol alginate, and polydopamine: Characterization and cytotoxicity in human chondrocytes. Nanomaterials 2021, 11, 2105. [Google Scholar] [CrossRef] [PubMed]
- Ramazanov, S.; Sobola, D.; Orudzhev, F.; Knápek, A.; Polčák, J.; Potoček, M.; Kaspar, P.; Dallaev, R. Surface modification and enhancement of ferromagnetism in BiFeO3 nanofilms deposited on HOPG. Nanomaterials 2020, 10, 1990. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- Grosvenor, A.; Kobe, B.A.; Biesinger, M.C.; McIntyre, N.S. Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf. Interface Anal. 2004, 36, 1564–1574. [Google Scholar] [CrossRef]
- Liu, R.; Conradie, J.; Erasmus, E. Comparison of X-ray photoelectron spectroscopy multiplet splitting of Cr 2p peaks from chromium tris(β-diketonates) with chemical effects. J. Electron Spectrosc. Relat. Phenom. 2016, 206, 46–51. [Google Scholar] [CrossRef]
- Faghani, G.; Rabiee, S.M.; Nourouzi, S.; Elmkhah, H. Corrosion behavior of TiN/CrN nanoscale multi-layered coating in Ringer’s solution. Int. J. Eng. 2020, 33, 329–336. [Google Scholar] [CrossRef]
- Hanoz, D.; Settimi, A.G.; Dabalà, M. Characterization of black coating on Fe360 steel obtained with immersion in aqueous solutions. Surf. Interfaces 2021, 26, 101317. [Google Scholar] [CrossRef]



| Input Parameter | Symbol | Levels | ||
|---|---|---|---|---|
| A | B | C | ||
| Coating Time (min) | D | 50 | 55 | 60 |
| Bath temp (°C) | T | 115 ± 2 | 120 ± 2 | 125 ± 2 |
| Wt.% of sodium dichromate (g/L) | C | 250 | 300 | 350 |
| Sample Code | OA | Avg. Percentage Reflectance | S/N Ratio | ||
|---|---|---|---|---|---|
| D | T | C | R (%) | R (dB) | |
| I | 1 | 1 | 1 | 9.75 | −19.7801 |
| II | 1 | 2 | 2 | 9.64 | −19.6815 |
| III | 1 | 3 | 3 | 7.93 | −17.9855 |
| IV | 2 | 1 | 2 | 9.01 | −19.0945 |
| V | 2 | 2 | 3 | 8.21 | −18.2869 |
| VI | 2 | 3 | 1 | 8.27 | −18.3501 |
| VII | 3 | 1 | 3 | 7.9 | −17.9525 |
| VIII | 3 | 2 | 1 | 9.04 | −19.1234 |
| IX | 3 | 3 | 2 | 7.93 | −17.9855 |
| Level | D | T | C |
|---|---|---|---|
| 1 | −19.15 | −18.94 | −19.08 |
| 2 | −18.58 | −19.03 | −18.92 |
| 3 | −18.35 | −18.11 | −18.07 |
| Delta | 0.8 | 0.92 | 1.01 |
| Rank | 3 | 2 | 1 |
| Output Characteristics | Preliminary Parameter Combination | Optimal Combination of Parameter | |
|---|---|---|---|
| Predicted Values | Experimental Values | ||
| Level | D1-T1-C1 | D3-T3-C3 | D3-T3-C3 |
| Avg. reflectance (%) | 9.75 | D3-T3-C3 | 7.546 |
| S/N ratio (dB) | −19.7801 | −17.1491 | −17.5631 |
| S/N ratio enhancements (dB) | 2.217 | - | - |
| Percentage decrement in R | 29.21% | - | - |
| Parameters | DF | SS | MS | F | P | % Contribution |
|---|---|---|---|---|---|---|
| Coating time (D) | 2 | 1.08179 | 0.54088 | 224.33 | 0.004 | 24.55 |
| Bath temp (T) | 2 | 1.56349 | 0.78174 | 324.23 | 0.003 | 35.49 |
| wt.% of Na2Cr2O7 (C) | 2 | 1.75582 | 0.87991 | 364.11 | 0.003 | 39.85 |
| Error | 2 | 0.00482 | 0.00241 | - | - | - |
| Total | 8 | 4.40589 | - | - | - | - |
| Coating Characteristics | Optimal Parameter Combination | Predicted Value | Experimental Value |
|---|---|---|---|
| Reflectance (R) | D3-T3-C3 (60 min, 125 °C, 350 g/L) | 7.296% | 7.546% |
| Sample | Corrosion Parameters | |||||
|---|---|---|---|---|---|---|
| Ecorr (mv) | Icorr (mA/cm2) | βa (mV·decade−1) | βc (mV·decade−1) | Rp (Ω Cm2) | Corrosion Rate (mm/y) | |
| SS 420A Coated Sample | −283.57 | 0.0239 | −910.88 | 392.74 | 8855.7 | 0.310 |
| SS 420A Non coated (Control Sample) | −308.51 | 0.0351 | −863.36 | 379.16 | 8029.1 | 0.413 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aruvathottil Rajan, R.; Saju, K.K.; Aravindakshan, R. Characterization and In Vitro Studies of Low Reflective Magnetite (Fe3O4) Thin Film on Stainless Steel 420A Developed by Chemical Method. Coatings 2021, 11, 1145. https://doi.org/10.3390/coatings11091145
Aruvathottil Rajan R, Saju KK, Aravindakshan R. Characterization and In Vitro Studies of Low Reflective Magnetite (Fe3O4) Thin Film on Stainless Steel 420A Developed by Chemical Method. Coatings. 2021; 11(9):1145. https://doi.org/10.3390/coatings11091145
Chicago/Turabian StyleAruvathottil Rajan, Reghuraj, Kaiprappady Kunchu Saju, and Ritwik Aravindakshan. 2021. "Characterization and In Vitro Studies of Low Reflective Magnetite (Fe3O4) Thin Film on Stainless Steel 420A Developed by Chemical Method" Coatings 11, no. 9: 1145. https://doi.org/10.3390/coatings11091145
APA StyleAruvathottil Rajan, R., Saju, K. K., & Aravindakshan, R. (2021). Characterization and In Vitro Studies of Low Reflective Magnetite (Fe3O4) Thin Film on Stainless Steel 420A Developed by Chemical Method. Coatings, 11(9), 1145. https://doi.org/10.3390/coatings11091145





