Medicinal Plants and Biogenic Metal Oxide Nanoparticles: A Paradigm Shift to Treat Alzheimer’s Disease
Abstract
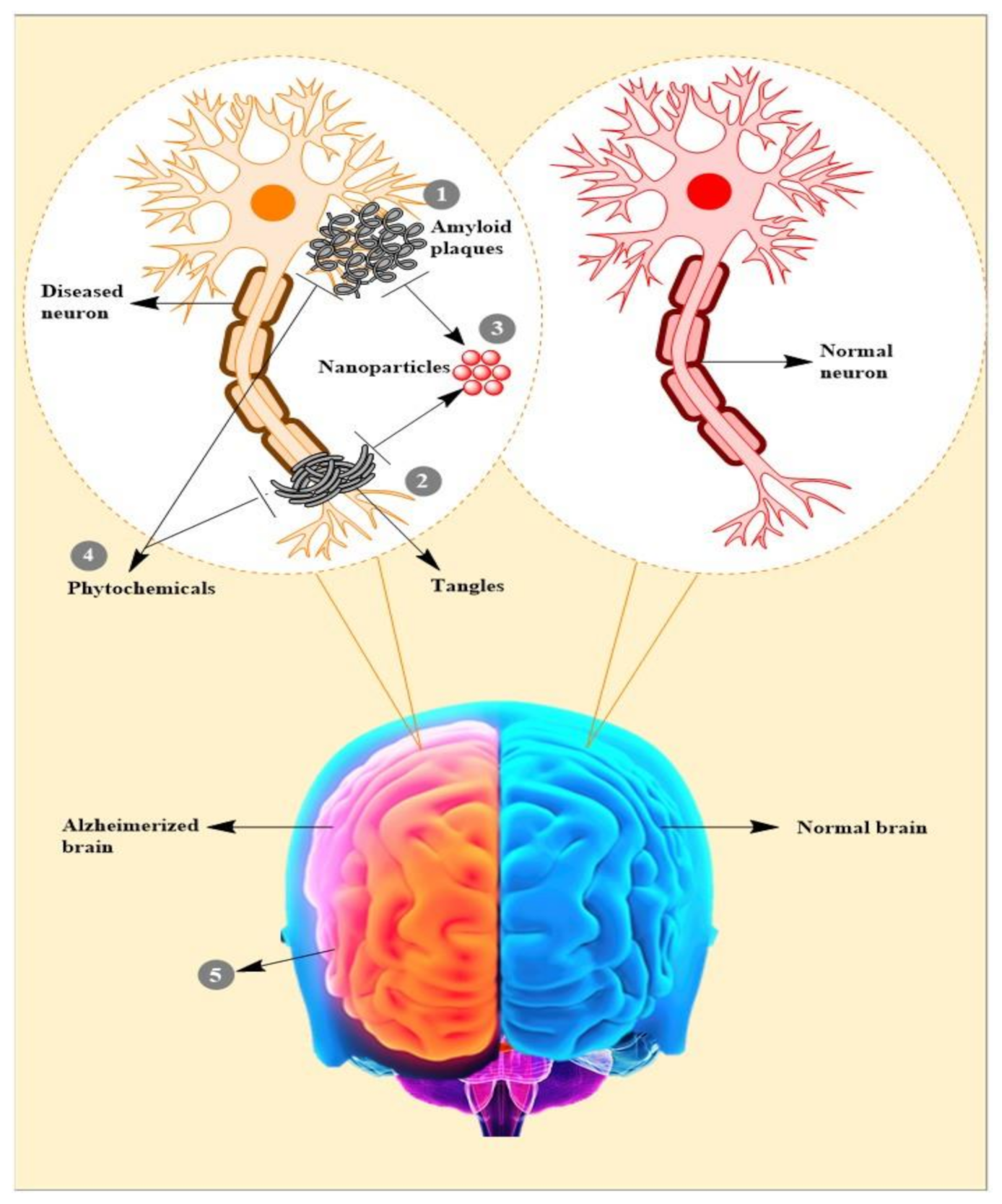
1. Introduction
1.1. Worldwide Prevalence of Alzheimer’s Disease in Aging Populations
1.2. Frequency Rate of AD
1.3. Defining Risk Factors of Alzheimer’s Disorder
1.3.1. Genetic Assumption
1.3.2. Vascular Pathway Assumption
1.3.3. Alcohol Consumption
1.3.4. High Cholesterol and Usage of Cholesterol-lowering Drugs
1.3.5. Psychosocial Assumption
1.3.6. Lethal Exposure
1.3.7. Inflammation
1.3.8. Other Factors
2. Present Therapeutic Issues and Disadvantages of Alzheimer’s Disease Therapies
3. Cholinesterase Inhibitory Activity of Different Plant Extracts
4. Biogenic Metal Nanoparticles for Treatment of Alzheimer’s Disease
5. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Akter, R.; Rahman, H.; Behl, T.; Chowdhury, M.; Rahman, A.; Manirujjaman, M.; Bulbul, I.J.; Elshenaw, S.E.; Tit, D.M.; Bungau, S. Prospective role of polyphenolic compounds in the treatment of neurodegenerative diseases. CNS Neurol. Disord. Drug Targets 2020, 20. [Google Scholar] [CrossRef]
- Bredesen, D.; Sharlin, K.; Jenkins, D.; Okuno, M.; Youngberg, W.; Cohen, S.; Stefani, A.; Brown, R.; Conger, S.; Tanio, C. Reversal of cognitive decline: 100 patients. J. Alzheimer’s Dis. Parkinsonism 2018, 8. [Google Scholar] [CrossRef]
- Budson, A.E.; Solomon, P.R. Memory Loss, Alzheimer’s Disease, and Dementia: A Practical Guide for Clinicians; Elsevier Health Sciences: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Mullane, K.; Williams, M. Alzheimer’s disease (AD) therapeutics–2: Beyond amyloid–re-defining AD and its causality to discover effective therapeutics. Biochem. Pharmacol. 2018, 158, 376–401. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Mesulam, M.-M.; Cuello, A.C.; Khachaturian, A.S.; Vergallo, A.; Farlow, M.; Snyder, P.; Giacobini, E.; Khachaturian, Z. Revisiting the cholinergic hypothesis in Alzheimer’s disease: Emerging evidence from translational and clinical research. J. Prev. Alzheimer’s Dis. 2019, 6, 2–15. [Google Scholar] [CrossRef]
- Mughal, E.U.; Javid, A.; Sadiq, A.; Murtaza, S.; Zafar, M.N.; Khan, B.A.; Sumrra, S.H.; Tahir, M.N.; Khan, K.M. Synthesis, structure-activity relationship and molecular docking studies of 3-O-flavonol glycosides as cholinesterase inhibitors. Bioorg. Med. Chem. 2018, 26, 3696–3706. [Google Scholar] [CrossRef]
- Cao, T.Q.; Ngo, Q.-M.T.; Seong, S.H.; Youn, U.J.; Kim, J.A.; Kim, J.; Kim, J.-C.; Woo, M.H.; Choi, J.S.; Min, B.S. Cholinesterase inhibitory alkaloids from the rhizomes of Coptis chinensis. Bioorg. Chem. 2018, 77, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Rafii, M.S.; Aisen, P.S. Advances in Alzheimer’s disease drug development. BMC Med. 2015, 13, 4. [Google Scholar] [CrossRef]
- Firdaus, Z.; Singh, T.D. An insight in pathophysiological mechanism of Alzheimer’s disease and its management using plant natural products. Mini Rev. Med. Chem. 2021, 21, 35–57. [Google Scholar] [CrossRef]
- Hodges, J.R. Alzheimer’s centennial legacy: Origins, landmarks and the current status of knowledge concerning cognitive aspects. Brain 2006, 129, 2811–2822. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K. Cholinesterase inhibitors as Alzheimer’s therapeutics. Mol. Med. Rep. 2019, 20, 1479–1487. [Google Scholar] [CrossRef]
- Kabir, M.T.; Uddin, M.; Begum, M.; Thangapandiyan, S.; Rahman, M.; Aleya, L.; Mathew, B.; Ahmed, M.; Barreto, G.E.; Ashraf, G.M. Cholinesterase inhibitors for Alzheimer’s disease: Multitargeting strategy based on anti-Alzheimer’s drugs repositioning. Curr. Pharm. Des. 2019, 25, 3519–3535. [Google Scholar] [CrossRef] [PubMed]
- Zaki, A.G.; El-Sayed, E.-S.R.; Abd Elkodous, M.; El-Sayyad, G.S. Microbial acetylcholinesterase inhibitors for Alzheimer’s therapy: Recent trends on extraction, detection, irradiation-assisted production improvement and nano-structured drug delivery. Appl. Microbiol. Biotechnol. 2020, 104, 4717–4735. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Jahan, S.; Alshahrani, S.; Alshehri, B.M.; Sameer, A.S.; Arafah, A.; Ahmad, A.; Rehman, M.U. Phytotherapeutic agents for neurodegenerative disorders: A neuropharmacological review. In Phytomedicine; Elsevier: Amsterdam, The Netherlands, 2021; pp. 581–620. [Google Scholar]
- Taki, M.; Hasegawa, T.; Ninoyu, Y.; Hirano, S. Efficacy of Yokukansan, a traditional Japanese herbal medicine, in patients with dizziness and irritability. Auris Nasus Larynx 2021. [Google Scholar] [CrossRef]
- Tewari, D.; Joshi, T.; Sah, A.N. Galanthus nivalis L. (snowdrop). In Naturally Occurring Chemicals Against Alzheimer’s Disease; Elsevier: Amsterdam, The Netherlands, 2021; pp. 301–315. [Google Scholar]
- Suganthy, N.; Ramkumar, V.S.; Pugazhendhi, A.; Benelli, G.; Archunan, G. Biogenic synthesis of gold nanoparticles from Terminalia arjuna bark extract: Assessment of safety aspects and neuroprotective potential via antioxidant, anticholinesterase, and antiamyloidogenic effects. Environ. Sci. Pollut. Res. 2018, 25, 10418–10433. [Google Scholar] [CrossRef]
- Mandal, S.; Debnath, K.; Jana, N.R.; Jana, N.R. Trehalose-functionalized gold nanoparticle for inhibiting intracellular protein aggregation. Langmuir 2017, 33, 13996–14003. [Google Scholar] [CrossRef] [PubMed]
- Nellore, J.; Pauline, C.; Amarnath, K. Bacopa monnieri phytochemicals mediated synthesis of platinum nanoparticles and its neurorescue effect on 1-methyl 4-phenyl 1, 2, 3, 6 tetrahydropyridine-induced experimental parkinsonism in zebrafish. J. Neurodegener. Dis. 2013, 2013. [Google Scholar] [CrossRef]
- Nazıroğlu, M.; Muhamad, S.; Pecze, L. Nanoparticles as potential clinical therapeutic agents in Alzheimer’s disease: Focus on selenium nanoparticles. Exp. Rev. Clin. Pharmacol. 2017, 10, 773–782. [Google Scholar] [CrossRef]
- Grundy, E.M.; Murphy, M. Population ageing in Europe. Oxf. Textb. Geriatr. Med. 2017, 11. [Google Scholar] [CrossRef]
- WHO. Risk Reduction of Cognitive Decline and Dementia; WHO Guidelines; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Solfrizzi, V.; Scafato, E.; Lozupone, M.; Seripa, D.; Schilardi, A.; Custodero, C.; Sardone, R.; Galluzzo, L.; Gandin, C.; Baldereschi, M. Biopsychosocial frailty and the risk of incident dementia: The Italian longitudinal study on aging. Alzheimer’s Dement. 2019, 15, 1019–1028. [Google Scholar] [CrossRef]
- Trevisan, K.; Cristina-Pereira, R.; Silva-Amaral, D.; Aversi-Ferreira, T.A. Theories of aging and the prevalence of Alzheimer’s disease. BioMed Res. Int. 2019, 2019. [Google Scholar] [CrossRef]
- Alzheimer’s Association. Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2017, 13, 325–373. [Google Scholar] [CrossRef]
- Qiu, C.; Fratiglioni, L. Epidemiology of Alzheimer’s. In Alzheimer’s Disease; Oxford University Press: New York, NY, USA, 2017. [Google Scholar]
- Lobo, A.; Launer, L.; Fratiglioni, L.; Andersen, K.; Di Carlo, A.; Breteler, M.; Copeland, J.; Dartigues, J.; Jagger, C.; Martinez-Lage, J. Prevalence of dementia and major subtypes in Europe: A collaborative study of population-based cohorts. Neurology 2000, 54. [Google Scholar]
- Kawas, C.; Gray, S.; Brookmeyer, R.; Fozard, J.; Zonderman, A. Age-specific incidence rates of Alzheimer’s disease: The Baltimore longitudinal study of aging. Neurology 2000, 54, 2072–2077. [Google Scholar] [CrossRef]
- Jorm, A.F.; Jolley, D. The incidence of dementia: A meta-analysis. Neurology 1998, 51, 728–733. [Google Scholar] [CrossRef]
- Miech, R.; Breitner, J.C.; Zandi, P.P.; Khachaturian, A.; Anthony, J.; Mayer, L. Incidence of AD may decline in the early 90s for men, later for women: The Cache county study. Neurology 2002, 58, 209–218. [Google Scholar] [CrossRef]
- Qiu, C.; Kivipelto, M.; von Strauss, E. Epidemiology of Alzheimer’s disease: Occurrence, determinants, and strategies toward intervention. Dialog. Clin. Neurosci. 2009, 11, 111. [Google Scholar]
- Chandra, V.; Pandav, R.; Dodge, H.; Johnston, J.; Belle, S.; DeKosky, S.; Ganguli, M. Incidence of Alzheimer’s disease in a rural community in India: The Indo–US study. Neurology 2001, 57, 985–989. [Google Scholar] [CrossRef]
- Kalaria, R.N.; Maestre, G.E.; Arizaga, R.; Friedland, R.P.; Galasko, D.; Hall, K.; Luchsinger, J.A.; Ogunniyi, A.; Perry, E.K.; Potocnik, F. Alzheimer’s disease and vascular dementia in developing countries: Prevalence, management, and risk factors. Lancet Neurol. 2008, 7, 812–826. [Google Scholar] [CrossRef]
- Kivipelto, M.; Mangialasche, F.; Ngandu, T. Lifestyle interventions to prevent cognitive impairment, dementia and Alzheimer disease. Nat. Rev. Neurol. 2018, 14, 653–666. [Google Scholar] [CrossRef]
- Larsson, S.C.; Traylor, M.; Malik, R.; Dichgans, M.; Burgess, S.; Markus, H.S. Modifiable pathways in Alzheimer’s disease: Mendelian randomisation analysis. BMJ 2017, 359, 5375. [Google Scholar] [CrossRef]
- Yuan, H.; Yang, W. Genetically determined serum uric acid and Alzheimer’s disease risk. J. Alzheimer’s Dis. 2018, 65, 1259–1265. [Google Scholar] [CrossRef]
- Rajabli, F.; Feliciano, B.E.; Celis, K.; Hamilton-Nelson, K.L.; Whitehead, P.L.; Adams, L.D.; Bussies, P.L.; Manrique, C.P.; Rodriguez, A.; Rodriguez, V. Ancestral origin of ApoE ε4 Alzheimer disease risk in Puerto Rican and African American populations. PLoS Gen. 2018, 14, e1007791. [Google Scholar] [CrossRef] [PubMed]
- Wajman, J.R.; Mansur, L.L.; Yassuda, M.S. Lifestyle patterns as a modifiable risk factor for late-life cognitive decline: A narrative review regarding dementia prevention. Curr. Aging Sci. 2018, 11, 90–99. [Google Scholar] [CrossRef]
- Vemuri, P.; Lesnick, T.G.; Przybelski, S.A.; Knopman, D.S.; Lowe, V.J.; Graff-Radford, J.; Roberts, R.O.; Mielke, M.M.; Machulda, M.M.; Petersen, R.C. Age, vascular health, and Alzheimer disease biomarkers in an elderly sample. Ann. Neurol. 2017, 82, 706–718. [Google Scholar] [CrossRef]
- Ricci, M.; Chiaravalloti, A.; Martorana, A.; Koch, G.; Barbagallo, G.; Schillaci, O. The role of epsilon phenotype in brain glucose consumption in Alzheimer’s disease. Ann. Nucl. Med. 2020, 34, 254–262. [Google Scholar] [CrossRef]
- Matloff, W.J.; Zhao, L.; Ning, K.; Conti, D.V.; Toga, A.W. Interaction effect of alcohol consumption and Alzheimer disease polygenic risk score on the brain cortical thickness of cognitively normal subjects. Alcohol 2020, 85, 1–12. [Google Scholar] [CrossRef]
- Topiwala, A.; Ebmeier, K.P. Effects of drinking on late-life brain and cognition. Evid. Based Ment. Health 2018, 21, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Power, M.C.; Rawlings, A.; Sharrett, A.R.; Bandeen-Roche, K.; Coresh, J.; Ballantyne, C.M.; Pokharel, Y.; Michos, E.D.; Penman, A.; Alonso, A. Association of midlife lipids with 20-year cognitive change: A cohort study. Alzheimer’s Dement. 2018, 14, 167–177. [Google Scholar] [CrossRef]
- Anstey, K.J.; Ashby-Mitchell, K.; Peters, R. Updating the evidence on the association between serum cholesterol and risk of late-life dementia: Review and meta-analysis. J. Alzheimer’s Dis. 2017, 56, 215–228. [Google Scholar] [CrossRef]
- Svensson, T.; Sawada, N.; Mimura, M.; Nozaki, S.; Shikimoto, R.; Tsugane, S. The association between midlife serum high-density lipoprotein and mild cognitive impairment and dementia after 19 years of follow-up. Transl. Psychiatry 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.A.; Bishara, D.; Perera, G.; Molokhia, M.; Rajendran, L.; Stewart, R.J. Benefits and harms of statins in people with dementia: A systematic review and meta-analysis. J. Am. Geriatr. Soc. 2020, 68, 650–658. [Google Scholar] [CrossRef]
- Bunt, C.W.; Hogan, A.J. The effect of statins on dementia and cognitive decline. Am. Fam. Physician 2017, 95, 151–152. [Google Scholar]
- Stockwell-Smith, G.; Moyle, W.; Kellett, U. The impact of early psychosocial intervention on self-efficacy of care recipient/carer dyads living with early-stage dementia—A mixed-methods study. J. Adv. Nurs. 2018, 74, 2167–2180. [Google Scholar] [CrossRef]
- Rusmaully, J.; Dugravot, A.; Moatti, J.-P.; Marmot, M.G.; Elbaz, A.; Kivimaki, M.; Sabia, S.; Singh-Manoux, A. Contribution of cognitive performance and cognitive decline to associations between socioeconomic factors and dementia: A cohort study. PLoS Med. 2017, 14, e1002334. [Google Scholar] [CrossRef] [PubMed]
- Paillard, T.; Rolland, Y.; de Souto Barreto, P. Protective effects of physical exercise in Alzheimer’s disease and Parkinson’s disease: A narrative review. J. Clin. Neurol. 2015, 11, 212. [Google Scholar] [CrossRef]
- Lauretta, G.; Ravalli, S.; Maugeri, G.; D’Agata, V.; Di Rosa, M.; Musumeci, G. The Impact of physical exercise on the hippocampus in physiological condition and ageing-related decline: Current evidence from animal and human studies. Curr. Pharm. Biotechnol. 2021, 22. [Google Scholar] [CrossRef]
- Lillo-Crespo, M.; Forner-Ruiz, M.; Riquelme-Galindo, J.; Ruiz-Fernández, D.; García-Sanjuan, S. Chess practice as a protective factor in dementia. Int. J. Environ. Res. Public Health 2019, 16, 2116. [Google Scholar] [CrossRef]
- Lafta, H.; AlMayaly, I. Potential effect of several heavy metals with Alzheimer’s disease infection. Ann. Rom. Soc. Cell Biol. 2021, 25, 3476–3484. [Google Scholar]
- Zhang, X.-Q.; Li, L.; Huo, J.-T.; Cheng, M.; Li, L.-H. Effects of repetitive transcranial magnetic stimulation on cognitive function and cholinergic activity in the rat hippocampus after vascular dementia. Neural Regen. Res. 2018, 13, 1384. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.; Schmidt, H.; Curb, J.D.; Masaki, K.; White, L.R.; Launer, L.J. Early inflammation and dementia: A 25-year follow-up of the Honolulu-Asia Aging Study. Ann. Neurol. 2002, 52, 168–174. [Google Scholar] [CrossRef]
- Szekely, C.A.; Breitner, J.C.; Fitzpatrick, A.L.; Rea, T.D.; Psaty, B.M.; Kuller, L.H.; Zandi, P.P. NSAID use and dementia risk in the Cardiovascular Health Study*: Role of APOE and NSAID type. Neurology 2008, 70, 17–24. [Google Scholar] [CrossRef]
- Arvanitakis, Z.; Grodstein, F.; Bienias, J.; Schneider, J.; Wilson, R.; Kelly, J.; Evans, D.; Bennett, D. Relation of NSAIDs to incident AD, change in cognitive function, and AD pathology. Neurology 2008, 70, 2219–2225. [Google Scholar] [CrossRef]
- Fleminger, S.; Oliver, D.; Lovestone, S.; Rabe-Hesketh, S.; Giora, A. Head injury as a risk factor for Alzheimer’s disease: The evidence 10 years on; a partial replication. J. Neurol. Neurosurg. Psychiatry 2003, 74, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Mehta, K.; Ott, A.; Kalmijn, S.; Slooter, A.; Van Duijn, C.; Hofman, A.; Breteler, M. Head trauma and risk of dementia and Alzheimer’s disease: The Rotterdam Study. Neurology 1999, 53, 1959. [Google Scholar] [CrossRef] [PubMed]
- Dal Forno, G.; Palermo, M.T.; Donohue, J.E.; Karagiozis, H.; Zonderman, A.B.; Kawas, C.H. Depressive symptoms, sex, and risk for Alzheimer’s disease. Ann. Neurol. 2005, 57, 381–387. [Google Scholar] [CrossRef]
- Sadiq, A.; Zeb, A.; Ullah, F.; Ahmad, S.; Ayaz, M.; Rashid, U.; Muhammad, N. Chemical characterization, analgesic, antioxidant, and anticholinesterase potentials of essential oils from Isodon rugosus Wall. ex. Benth. Front. Pharmacol. 2018, 9, 623. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Muhammad, S.; Shah, M.R.; Khan, A.; Rashid, U.; Farooq, U.; Ullah, F.; Sadiq, A.; Ayaz, M.; Ali, M. Neurologically potent molecules from Crataegus oxyacantha; isolation, anticholinesterase inhibition, and molecular docking. Front. Pharmacol. 2017, 8, 327. [Google Scholar] [CrossRef]
- Zohra, T.; Ovais, M.; Khalil, A.T.; Qasim, M.; Ayaz, M.; Shinwari, Z.K. Extraction optimization, total phenolic, flavonoid contents, HPLC-DAD analysis and diverse pharmacological evaluations of Dysphania ambrosioides (L.) Mosyakin & Clemants. Nat. Prod. Res. 2019, 33, 136–142. [Google Scholar]
- Ayaz, M.; Ovais, M.; Ahmad, I.; Sadiq, A.; Khalil, A.T.; Ullah, F. Biosynthesized metal nanoparticles as potential Alzheimer’s disease therapeutics. In Metal Nanoparticles for Drug Delivery and Diagnostic Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 31–42. [Google Scholar]
- López, V.; Martín, S.; Gómez-Serranillos, M.P.; Carretero, M.E.; Jäger, A.K.; Calvo, M.I. Neuroprotective and neurological properties of Melissa officinalis. Neurochem. Res. 2009, 34, 1955–1961. [Google Scholar] [CrossRef]
- Adsersen, A.; Gauguin, B.; Gudiksen, L.; Jäger, A.K. Screening of plants used in Danish folk medicine to treat memory dysfunction for acetylcholinesterase inhibitory activity. J. Ethnopharmacol. 2006, 104, 418–422. [Google Scholar] [CrossRef]
- Ferreira, A.; Proença, C.; Serralheiro, M.; Araujo, M. The in vitro screening for acetylcholinesterase inhibition and antioxidant activity of medicinal plants from Portugal. J. Ethnopharmacol. 2006, 108, 31–37. [Google Scholar] [CrossRef]
- Orhan, I.; Kartal, M.; Naz, Q.; Ejaz, A.; Yilmaz, G.; Kan, Y.; Konuklugil, B.; Şener, B.; Choudhary, M.I. Antioxidant and anticholinesterase evaluation of selected Turkish Salvia species. Food Chem. 2007, 103, 1247–1254. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Menichini, F.; Conforti, F.; Tundis, R.; Bonesi, M.; Saab, A.M.; Statti, G.A.; de Cindio, B.; Houghton, P.J.; Menichini, F. Chemical analysis, antioxidant, antiinflammatory and anticholinesterase activities of Origanum ehrenbergii boiss and Origanum syriacum L. essential oils. Food Chem. 2009, 117, 174–180. [Google Scholar] [CrossRef]
- Basavan, D.; Chalichem, N.S.; Kumar, M.K. Phytoconstituents and their possible mechanistic profile for Alzheimer’s Disease–A literature review. Curr. Drug Targets 2019, 20, 263–291. [Google Scholar] [CrossRef]
- Aazza, S.; Lyoussi, B.; Miguel, M.G. Antioxidant and antiacetylcholinesterase activities of some commercial essential oils and their major compounds. Molecules 2011, 16, 7672. [Google Scholar] [CrossRef] [PubMed]
- Okeke, B.M.; Adefegha, S.A.; Oyeleye, S.I.; Oboh, G. Effects of combined crude alkaloid-rich extracts from alligator pepper (Aframomum melegueta) and bastered melegueta (Aframomum danielli) on the enzymes crucial to erectile dysfunction—In vitro. J. Food Biochem. 2018, 42, e12550. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, B.; Yang, F.; Sun, Q.; Yang, Z.; Zhu, L. Chemical compositionand anti-acetyl cholinesterase activity of flower essential oils of artemisiaannuaat different flowering stage. IJPR 2011, 10, 265. [Google Scholar]
- Jazayeri, S.B.; Amanlou, A.; Ghanadian, N.; Pasalar, P.; Amanlou, M. A preliminary investigation of anticholinesterase activity of some Iranian medicinal plants commonly used in traditional medicine. DARU J. Pharm. Sci. 2014, 22, 1–5. [Google Scholar] [CrossRef]
- Akram, M.; Nawaz, A. Effects of medicinal plants on Alzheimer’s disease and memory deficits. Neural Regen. Res. 2017, 12, 660. [Google Scholar] [CrossRef]
- Ingkaninan, K.; Temkitthawon, P.; Chuenchom, K.; Yuyaem, T.; Thongnoi, W. Screening for acetylcholinesterase inhibitory activity in plants used in Thai traditional rejuvenating and neurotonic remedies. J. Ethnopharmacol. 2003, 89, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Kumar, V.; Houghton, P.J. Screening of Indian medicinal plants for acetylcholinesterase inhibitory activity. Phytotherapy Res. 2007, 21, 1142–1145. [Google Scholar] [CrossRef]
- Ahmed, F.; Chandra, J.; Urooj, A.; Rangappa, K. In vitro antioxidant and anticholinesterase activity of Acorus calamus and Nardostachys jatamansi rhizomes. J. Pharm. Res. 2009, 2, 830–883. [Google Scholar]
- Orhan, I.; Şener, B.; Choudhary, M.; Khalid, A. Acetylcholinesterase and butyrylcholinesterase inhibitory activity of some Turkish medicinal plants. J. Ethnopharmacol. 2004, 91, 57–60. [Google Scholar] [CrossRef]
- Zelík, P.; Lukešová, A.; Voloshko, L.N.; Štys, D.; Kopecký, J. Screening for acetylcholinesterase inhibitory activity in cyanobacteria of the genus Nostoc. J. Enzym. Inhib. Med. Chem. 2009, 24, 531–536. [Google Scholar] [CrossRef]
- Sancheti, S.; Um, B.-H.; Seo, S.-Y. 1, 2, 3, 4, 6-penta-O-galloyl-β-D-glucose: A cholinesterase inhibitor from Terminalia chebula. S. Afr. J. Bot. 2010, 76, 285–288. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Kumar, V.; Mal, M.; Houghton, P.J. Acetylcholinesterase inhibitors from plants. Phytomedicine 2007, 14, 289–300. [Google Scholar] [CrossRef]
- Vinutha, B.; Prashanth, D.; Salma, K.; Sreeja, S.; Pratiti, D.; Padmaja, R.; Radhika, S.; Amit, A.; Venkateshwarlu, K.; Deepak, M. Screening of selected Indian medicinal plants for acetylcholinesterase inhibitory activity. J. Ethnopharmacol. 2007, 109, 359–363. [Google Scholar] [CrossRef]
- Hillhouse, B.; Ming, D.S.; French, C.; Towers, G. Acetylcholine esterase inhibitors in Rhodiola rosea. Pharm. Biol. 2004, 42, 68–72. [Google Scholar] [CrossRef]
- Carradori, D.; Balducci, C.; Re, F.; Brambilla, D.; Le Droumaguet, B.; Flores, O.; Gaudin, A.; Mura, S.; Forloni, G.; Ordoñez-Gutierrez, L. Antibody-functionalized polymer nanoparticle leading to memory recovery in Alzheimer’s disease-like transgenic mouse model. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 609–618. [Google Scholar] [CrossRef]
- Brambilla, D.; Verpillot, R.; Le Droumaguet, B.; Nicolas, J.; Taverna, M.; Kóňa, J.; Lettiero, B.; Hashemi, S.H.; De Kimpe, L.; Canovi, M. PEGylated nanoparticles bind to and alter amyloid-beta peptide conformation: Toward engineering of functional nanomedicines for Alzheimer’s disease. ACS Nano 2012, 6, 5897–5908. [Google Scholar] [CrossRef]
- Feng, X.; Chen, A.; Zhang, Y.; Wang, J.; Shao, L.; Wei, L. Central nervous system toxicity of metallic nanoparticles. Int. J. Nanomed. 2015, 10, 4321. [Google Scholar]
- Yin, T.; Yang, L.; Liu, Y.; Zhou, X.; Sun, J.; Liu, J. Sialic acid (SA)-modified selenium nanoparticles coated with a high blood–brain barrier permeability peptide-B6 peptide for potential use in Alzheimer’s disease. Acta Biomater. 2015, 25, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Zohra, T.; Ovais, M.; Khalil, A.T.; Qasim, M.; Ayaz, M.; Shinwari, Z.K.; Ahmad, S.; Zahoor, M. Bio-guided profiling and HPLC-DAD finger printing of Atriplex lasiantha boiss. BMC Complement. Altern. Med. 2019, 19, 4161. [Google Scholar] [CrossRef] [PubMed]
- Barabadi, H.; Ovais, M.; Shinwari, Z.K.; Saravanan, M. Anti-cancer green bionanomaterials: Present status and future prospects. Green Chem. Lett. Rev. 2017, 10, 285–314. [Google Scholar] [CrossRef]
- Khalil, A.T.; Ayaz, M.; Ovais, M.; Wadood, A.; Ali, M.; Shinwari, Z.K.; Maaza, M. In vitro cholinesterase enzymes inhibitory potential and in silico molecular docking studies of biogenic metal oxides nanoparticles. Inorg. Nano Met. Chem. 2018, 48, 441–448. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, X.; Yu, Q.; Yang, L.; Sun, D.; Zhou, Y.; Liu, J. Epigallocatechin-3-gallate (EGCG)-stabilized selenium nanoparticles coated with Tet-1 peptide to reduce amyloid-β aggregation and cytotoxicity. ACS Appl. Mater. Interfaces 2014, 6, 8475–8487. [Google Scholar] [CrossRef]
| S. No | Name and Code of Drug | Manufacturers | Stage of Trials | Clinical Identifier Code |
|---|---|---|---|---|
| 1 | CT1812 | Cognition Therapeutics (Pittsburgh, PA, USA) | Completed | NCT03522129 |
| 2 | AZD 3293 | AstraZeneca/Lilly (Cambridge, UK) | Phase-2/3 | NCT02040987 |
| 3 | MK-1942 | Woodland Research (Rogers, AR, USA) | Phase-1 | NCT04308304 |
| 4 | PF-04360365 | Pfizer (New York, NY, USA) | Phase-2 | NCT00945672 |
| 5 | Bexarotene | Cleveland Clinic Lou (Los Vegas, NV, USA) | Phase-2 | NCT01782742 |
| 6 | Bryostatin 1 | Axiom Research/Neurotrope (New York, NY, USA) | Phase-2 | NCT04538066 |
| 7 | RO4602522 | Roche (Basel, Switzerland) | Phase-2 | NCT01677754 |
| 8 | Al-002 | Alector Inc. (South San Francisco, CA, USA) | Phase-2 | NCT04592874 |
| 9 | JNJ-54861911 | Janssen Research (New Brunswick, NJ, USA) | Phase-1 | NCT02211079 |
| 10 | PF-04360365 | Pfizer (New York, NY, USA) | Phase-1 | NCT00733642 |
| 11 | SAM-531 | Pfizer (New York, NY, USA) | Phase-1 | NCT00966966 |
| 12 | V950 | Merck Sharp & Dohme Corp (Kenilworth, NJ, USA) | Phase-1 | NCT00464334 |
| Botanical Name | Family | Common Name | Traditional Use |
|---|---|---|---|
| Acorus calamus | Araceae | Sweet flag | Insomnia, insanity, neuropathy |
| Bacopa monniera | Scrophulariaceae | Water hyssop | Epilepsy, insanity, memory loss |
| Cedrus deodera | Pinaceae | Deoder, Cedar | Insanity |
| Celastrus paniculatus | Celastraceae | Black oil tree | Anxiety, epilepsy |
| Centella asiatica | Apiaceae | Indian pennywort | Insomnia, mental retardation |
| Convolvulus pluricaulis | Convolvulaceae | Bindweed | To enhance memory and intellect |
| Coriandum sativum | Umbelliferae | Coriander | To improve vitality and memory |
| Emblica officinalis | Euphorbiaceae | Gooseberry | Anti-aging |
| Evolvulus alsinoides | Convolvulaceae | Dwarf morning glory | To improve memory |
| Glycyrrhiza glabra | Leguminosae | Licorice | To enhance memory |
| Nardostachys jatamansi | Valerianaceae | Indian spikenard | Insanity, epilepsy, insomnia |
| Nelumbo nucifera | Nelumbonaceae | Lotus | Insomnia, restlessness |
| Punica granatum | Punicaceae | Pomegranate | Anti-aging |
| Rauvolfia serpentina | Apocynaceae | Snakeroot | Insanity, epilepsy |
| Saussurea lappa | Asteraceae | Costus | To treat neuropathy |
| Terminalia chebula | Combretaceae | Chebulic myrobalan | General debility |
| Tinospora cordifolia | Menispermaceae | Tinospora | Anti-aging |
| Trigonella foenum | Fabaceae | Fenugreek | Anti-diabetic |
| Valeriana wallichii | Valerianaceae | Indian Valerian | Emotional stress |
| Withania somminifera | Solanaceae | Winter cherry | Rejuvenating nervine tonic |
| Botanical Name | Part Used | Type of Extract | AChE Inhibition and Concentration Used | BChE Inhibition and Concentration Used | Reference |
|---|---|---|---|---|---|
| Acanthus ebracteatus | Aerial part | Methanol | TLC and 96 well plate; 36.19 ± 8.00 (0.1 mg/mL) | ND | [76] |
| Andrographis paniculata | Aerial part | Hydroalcohol | 96 well plate; 50% (222.41 µg/mL) | ND | [77] |
| Acorus calamus L. | Rhizomes | Methanol | 96 well plate; 50% (791.35 µmg/L) | ND | [78] |
| Buxux sempervirens L. | Whole plant | Chloroform Methanol (1:1) | 96 well plate; 61.76 ± 0.76 (1 mg/mL) | ND | [79] |
| Carum carvi L. | Radix | Methanol | TLC and 96 well plate; 11.00 ± 0.90 (0.1 mg/mL) | ND | [80] |
| Carthamus tinctorius L. | Flower | Methanol | TLC and 96 well plate; 30.33 ± 9.22 (0.1 mg/mL) | ND | [76] |
| Capsella bursa-pastoris L. | Whole plant | Methanol | 96 well plate; 10.00 ± 2.00 (5 mg/mL) | 96 well plate; 13.00 ± 1.00 (5 mg/mL) | [81] |
| Dioscorea bulbifera L. | Whole plant | Methanol | 96 well plate; 79.00 ± 2.00 (5 mg/mL) | 96 well plate; 82.00 ± 2.00 (5 mg/mL) | [81] |
| Euonymus sachalinensis | Leaf | Methanol | 96 well plate; 10.00 ± 3.00 (5 mg/mL) | 96 well plate; 43.00 ± 1.00 (5 mg/mL) | [81] |
| Euphorbia antiquorum L. | Stem | Methanol | TLC and 96 well plate; 42.31 ± 9.10 (0.1 mg/mL) | ND | [76] |
| Hypericum undulatum | Flower | Water | UV spectrometry; 81.70 ± 3.40 (5 mg/mL) | ND | [67] |
| Lycopodium clavatum L. | Whole plant | Chloroform:Methanol (1:1) | 96 well plate; 49.85 ± 31.33 (1 mg/mL) | ND | [79] |
| Michelia champaca L. | Leaf | Methanol | TLC and 96 well plate; 38.88 ± 4.56 (0.1 mg/mL) | ND | [82] |
| Magnifera indica L. | Bark | Methanol | TLC and 96 well plate; 8.15 ± 0.77 (100 µg/mL) | ND | [83] |
| Pimpinella anisum L. | Fruit | Methanol | TLC and 96 well plate; 3.00 ± 0.10 (0.1 mg/mL) | ND | [66] |
| Paronychia argentea Lam. | Aerial parts | Water | UV spectrometry; 26.10 ± 0.82 (5 mg/mL) | ND | [67] |
| Robinia pseudoacaca L. | Whole plant | Chloroform Methanol (1:1) | 96 well plate; 26.32 ± 0.82 (1 mg/mL) | 96 well plate; 31.47 ± 0.99 (1 mg/mL) | [79] |
| Rhodiola resea L. | Root | Methanol | 96 well plate; 42.00 ± 3.20 (10 g/mL) | ND | [84] |
| Symplocos chinesis | Whole plant | Methanol | 96 well plate; 74.00 ± 2.00 (5 mg/mL) | 96 well plate; 75.00 ± 2.00 (5 mg/mL) | [81] |
| Semecarpus anacardium Linn. | Bark | Methanol | TLC and 96 well plate; 69.94 ± 0.75 (100 µg/mL) | ND | [83] |
| Terminalia bellirica | Fruit | Methanol | TLC and 96 well plate; 39.68 ± 8.15 (0.1 mg/mL) | ND | [76] |
| Terminalia chebula | Fruit | Methanol | 96 well plate; 89.00 ± 1.00 (5 mg/mL) | 96 well plate; 95.00 ± 1.00 (5 mg/mL) | [76] |
| Withania somnifera Dunal. | Root | Methanol | TLC and 96 well plate; 75.95 ± 0.16 (100 µg/mL) | ND | [83] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gul, R.; Jan, H.; Lalay, G.; Andleeb, A.; Usman, H.; Zainab, R.; Qamar, Z.; Hano, C.; Abbasi, B.H. Medicinal Plants and Biogenic Metal Oxide Nanoparticles: A Paradigm Shift to Treat Alzheimer’s Disease. Coatings 2021, 11, 717. https://doi.org/10.3390/coatings11060717
Gul R, Jan H, Lalay G, Andleeb A, Usman H, Zainab R, Qamar Z, Hano C, Abbasi BH. Medicinal Plants and Biogenic Metal Oxide Nanoparticles: A Paradigm Shift to Treat Alzheimer’s Disease. Coatings. 2021; 11(6):717. https://doi.org/10.3390/coatings11060717
Chicago/Turabian StyleGul, Roby, Hasnain Jan, Gul Lalay, Anisa Andleeb, Hazrat Usman, Rimsha Zainab, Zeeshan Qamar, Christophe Hano, and Bilal Haider Abbasi. 2021. "Medicinal Plants and Biogenic Metal Oxide Nanoparticles: A Paradigm Shift to Treat Alzheimer’s Disease" Coatings 11, no. 6: 717. https://doi.org/10.3390/coatings11060717
APA StyleGul, R., Jan, H., Lalay, G., Andleeb, A., Usman, H., Zainab, R., Qamar, Z., Hano, C., & Abbasi, B. H. (2021). Medicinal Plants and Biogenic Metal Oxide Nanoparticles: A Paradigm Shift to Treat Alzheimer’s Disease. Coatings, 11(6), 717. https://doi.org/10.3390/coatings11060717








