Comparison of Heavy Metals Removal from Aqueous Solution by Moringa oleifera Leaves and Seeds
Abstract
1. Introduction
2. Materials and Methods
2.1. Biosorbents
2.2. Preparation of Metallic Ions Solutions
2.3. Characterization Techniques
2.4. Point of Zero Charge (pHPZC)
2.5. Sorption Experiments
3. Results and Discussions
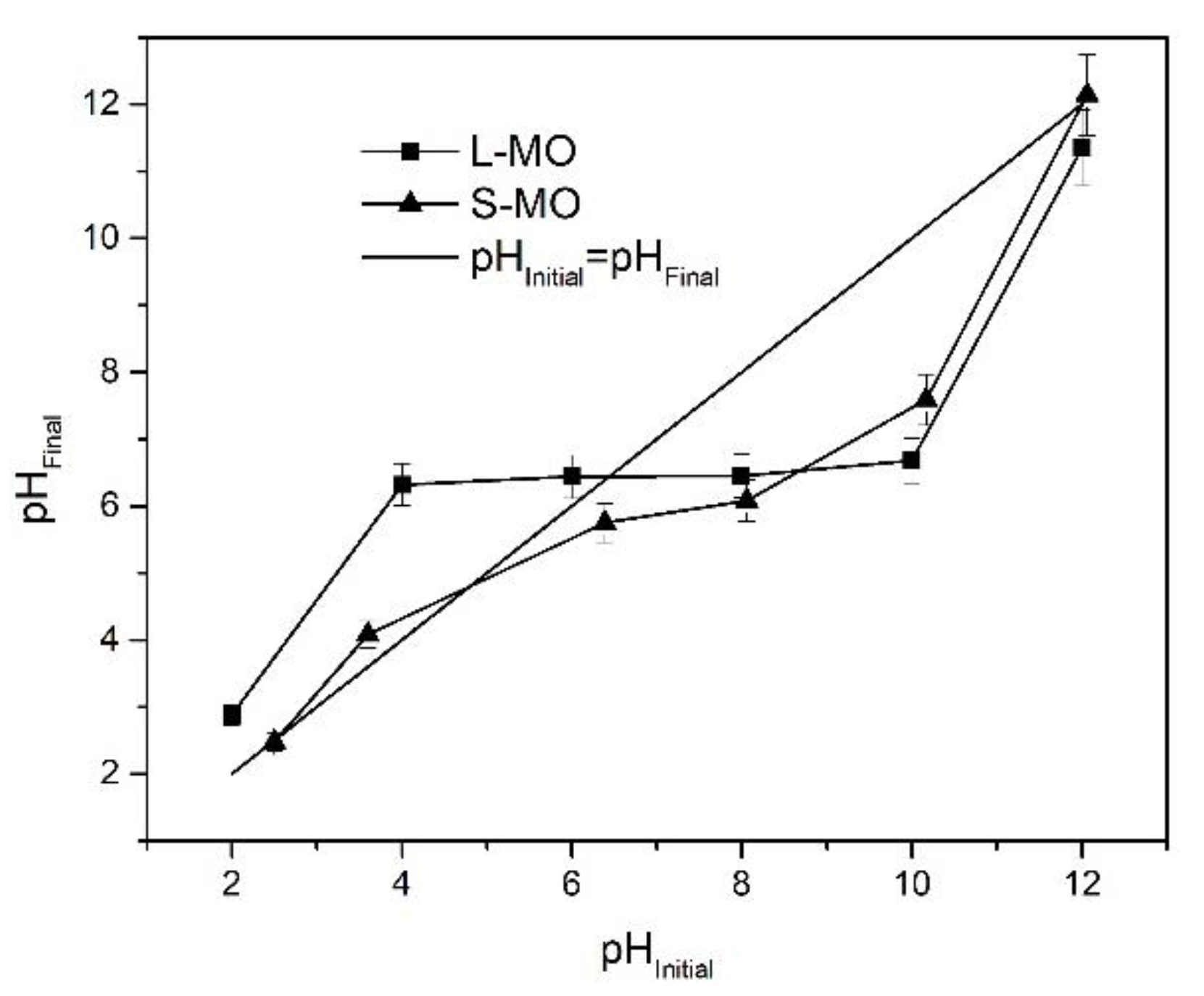
3.1. Point of Zero Charge (pHPZC)
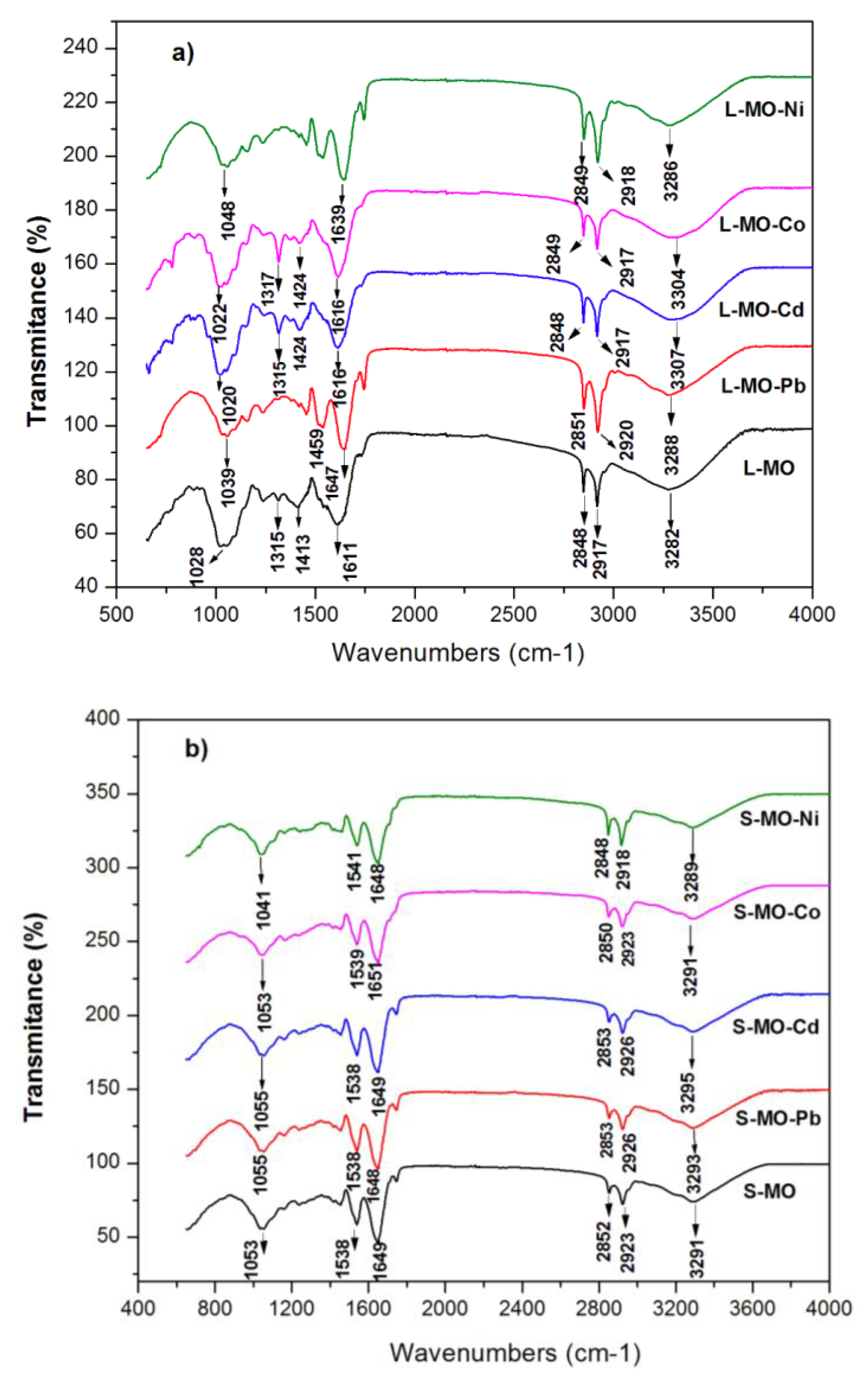
3.2. FTIR Spectra
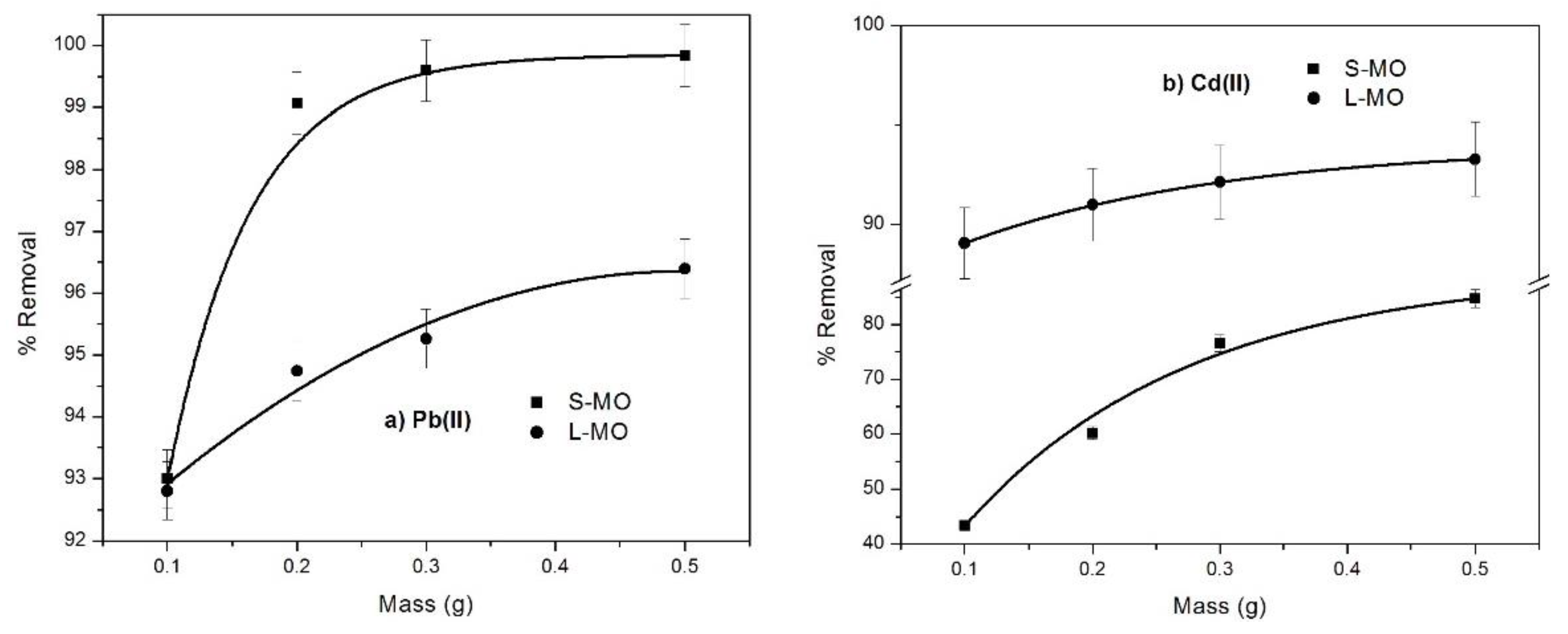
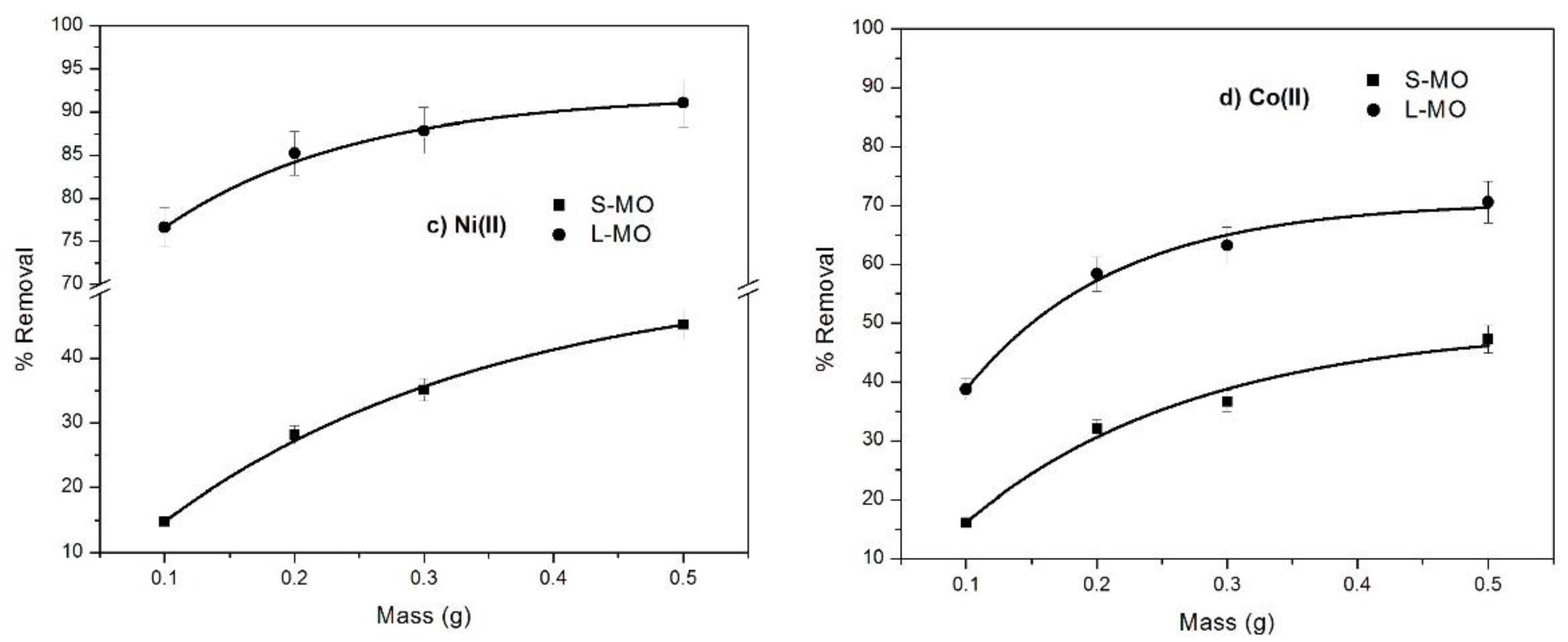
3.3. Effect of the Dosage
3.4. Kinetic
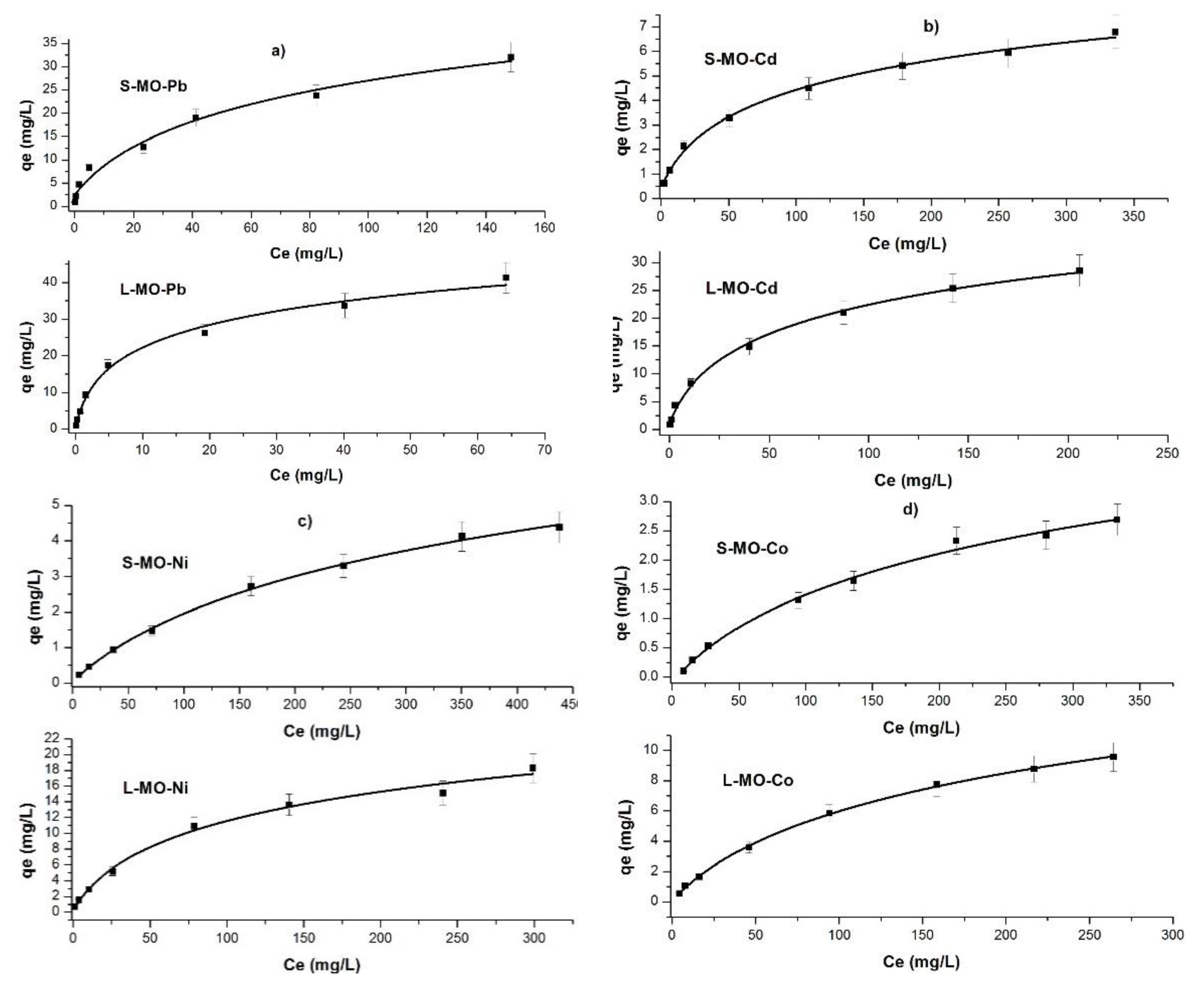
3.5. Isotherms
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, Y.; Lei, J.; Zhang, Y.; Zhu, J.; Lu, Y.; Wu, X.; Fang, H. Determining discharge characteristics and limits of heavy metals and metalloids for wastewater treatment plants (WWTPs) in China based on statistical methods. Water 2018, 10, 1248. [Google Scholar] [CrossRef]
- Obasi, P.N.; Akudinobi, B.B. Potential health risk and levels of heavy metals in water resources of lead–zinc mining communities of Abakaliki, southeast Nigeria. Appl. Water Sci. 2020, 10, 1–23. [Google Scholar] [CrossRef]
- Correa, M.L.; Velasquez, J.A.; Quintana, G.C. Uncommon Crop Residues as Ni (II) and Cd (II) Biosorbents. Ind. Eng. Chem. Res. 2012, 51, 12456–12462. [Google Scholar] [CrossRef]
- Marín, A.P.; Aguilar, M.I.; Meseguer, V.F.; Ortuno, J.F.; Sáez, J.; Lloréns, M. Biosorption of chromium (III) by orange (Citrus cinensis) waste: Batch and continuous studies. Chem. Eng. J. 2009, 155, 199–206. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K.; Witek-Krowiak, A. State of the art for the biosorption process—A Review. Appl. Biochem. Biotechnol. 2013, 170, 1389–1416. [Google Scholar] [CrossRef]
- Basso, M.C.; Cerrella, E.G.; Cukierman, A.L. Lignocellulosic materials as potential biosorbents of trace toxic metals from wastewater. Ind. Eng. Chem. Res. 2002, 41, 3580–3585. [Google Scholar] [CrossRef]
- Ayangbenro, A.S.; Babalola, O.O. A new strategy for heavy metal polluted environments: A review of microbial biosorbents. Int. J. Environ. Res. Public Health 2017, 14, 94. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef]
- Gadd, G.M. Biosorption: Critical review of scientific rationale, environmental importance and significance for pollution treatment. J. Chem. Technol. Biotechnol. 2009, 84, 13–28. [Google Scholar] [CrossRef]
- Ngah, W.W.; Hanafiah, M.M. Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: A review. Bioresour. Technol. 2008, 99, 3935–3948. [Google Scholar] [CrossRef]
- Gutha, Y.; Munagapati, V.S.; Alla, S.R.; Abburi, K. Biosorptive removal of Ni (II) from aqueous solution by caesalpinia bonducella seed powder biosorptive removal of Ni (II) from aqueous solution by Caesalpinia bonducella seed powder. Sep. Sci. Technol. 2011, 46, 2291–2297. [Google Scholar] [CrossRef]
- Fiorentin, L.D.; Trigueros, D.E.; Módenes, A.N.; Espinoza-Quiñones, F.R.; Pereira, N.C.; Barros, S.T.; Santos, O.A. Biosorption of reactive blue 5G dye onto drying orange bagasse in batch system: Kinetic and equilibrium modeling. Chem. Eng. J. 2010, 163, 68–77. [Google Scholar] [CrossRef]
- Rao, R.A.K.; Rehman, F. Use of Polyalthia longifolia Seeds (Seeds of Indian Mast Tree) as Adsorbent for the Removal of Cd (II) from Aqueous Solution Use of Polyalthia longifolia Seeds (Seeds of Indian Mast Tree) as Adsorbent for the Removal of Cd (II) from Aqueous Soluti. J. Dispers. Sci. Technol. 2012, 33, 37–41. [Google Scholar] [CrossRef]
- Jain, C.K.; Malik, D.S.; Yadav, A.K. Applicability of plant based biosorbents in the removal of heavy metals: A review. Environ. Process. 2016, 3, 495–523. [Google Scholar] [CrossRef]
- Asgarzadeh, S.; Rostamian, R.; Faez, E.; Maleki, A.; Daraei, H. Biosorption of Pb (II), Cu (II), and Ni (II) ions onto novel lowcost P. eldarica leaves-based biosorbent: Isotherm, kinetics, and operational parameters investigation. Desalin. Water Treat. 2015, 57, 14544–14551. [Google Scholar] [CrossRef]
- Meseguer, V.F.; Ortuño, J.F.; Aguilar, M.I.; Pinzón-Bedoya, M.L.; Lloréns, M.; Sáez, J.; Pérez-Marín, A.B. Biosorption of cadmium (II) from aqueous solutions by natural and modified non-living leaves of Posidonia oceanica. Environ. Sci. Pollut. Res. 2016, 32, 24032–24046. [Google Scholar] [CrossRef]
- Ounsadi, H.; Khalidi, A.; Abdennouri, M.; Barka, N. Biosorption potential of Diplotaxis harra and Glebionis coronaria L. biomasses for the removal of Cd(II) and Co(II) from aqueous solutions. J. Environ. Chem. Eng. 2015, 3, 822–830. [Google Scholar] [CrossRef]
- Hanif, M.A.; Nadeem, R.; Bhatti, H.N.; Ahmad, N.R.; Ansari, T.M. Ni(II) biosorption by Cassia fistula (Golden Shower) biomass. J. Hazard. Mater. 2007, 139, 345–355. [Google Scholar] [CrossRef]
- Long, J.; Huang, X.; Fan, X.; Peng, Y.; Xia, J. Effective adsorption of nickel (II) with Ulva lactuca dried biomass: Isotherms, kinetics and mechanisms Jianyou Long, Xiaona Huang, Xiaoli Fan, Yan Peng and Jianrong Xia. Water Sci. Technol. 2018, 78, 156–164. [Google Scholar] [CrossRef]
- Aksu, Z. Application of biosorption for the removal of organic pollutants: A review. Process Biochem. 2005, 40, 997–1026. [Google Scholar] [CrossRef]
- Wang, M.X.; Zhang, Q.L.; Yao, S.J. A novel biosorbent formed of marine-derived Penicillium janthinellum mycelial pellets for removing dyes from dye-containing wastewater. Chem. Eng. J. 2015, 259, 837–844. [Google Scholar] [CrossRef]
- Zamzow, M.J.; Eichbaum, B.R.; Sandgren, K.R.; Shanks, D.E. Removal of heavy metals and other cations from wastewater using zeolites. Sep. Sci. Technol. 1990, 25, 1555–1569. [Google Scholar] [CrossRef]
- Ghebremichael, K.; Gebremedhin, N.; Amy, G. Performance of Moringa oliefera as a biosorbent for chromium removal. Water Sci. Technol. 2010, 62, 1106–1111. [Google Scholar] [CrossRef]
- Pandey, V.N.; Chauhan, V.; Pandey, V.S.; Upadhyaya, P.P.; Kopp, O.R. Moringa oleifera Lam: A Biofunctional Edible Plant from India, Phytochemistry and Medicinal Properties. J. Plant Stud. 2019, 8, 10–19. [Google Scholar] [CrossRef][Green Version]
- Sajidu, S.M.; Henry, E.M.T.; Kwamdera, G.; Mataka, L. Removal of lead, iron and cadmium ions by means of polyelectrolytes of the Moringa Oleifera whole seed kernel. WIT Trans. Ecol. Environ. 2005, 80, 251–258. [Google Scholar] [CrossRef]
- Kumari, P.; Sharma, P.; Srivastava, S.; Srivastava, M.M. Biosorption studies on shelled Moringa oleifera Lamarck seed powder: Removal and recovery of arsenic from aqueous system. Int. J. Miner. Process. 2006, 78, 131–139. [Google Scholar] [CrossRef]
- Imran, M.; Anwar, K.; Akram, M.; Shah, G.M.; Ahmad, I.; Samad Shah, N.; Khan, Z.U.H.; Rashid, M.I.; Akhtar, M.N.; Ahmad, S.; et al. Biosorption of Pb (II) from contaminated water onto Moringa oleifera biomass: Kinetics and equilibrium studies. Int. J. Phytoremed. 2019, 21, 777–789. [Google Scholar] [CrossRef]
- Bhatti, H.N.; Mumtaz, B.; Hanif, M.A.; Nadeem, R. Removal of Zn (II) ions from aqueous solution using Moringa oleifera Lam. (horseradish tree) biomass. Process Biochem. 2007, 42, 547–553. [Google Scholar] [CrossRef]
- Reddy, D.H.K.; Ramana, D.K.V.; Seshaiah, K.; Reddy, A.V.R. Biosorption of Ni (II) from aqueous phase by Moringa oleifera bark, a low cost biosorbent. Desalination 2011, 268, 150–157. [Google Scholar] [CrossRef]
- Matouq, M.; Jildeh, N.; Qtaishat, M.; Hindiyeh, M.; Al Syouf, M.Q. The adsorption kinetics and modeling for heavy metals removal from wastewater by Moringa pods. J. Environ. Chem. Eng. 2015, 3, 775–784. [Google Scholar] [CrossRef]
- Abatal, M.; Olguin, M.T. Comparative adsorption behavior between phenol and p-nitrophenol by Na- and HDTMA-clinoptilolite-rich tuff. Environ. Earth Sci. 2013, 69. [Google Scholar] [CrossRef]
- Peng, S.; Hao, K.; Han, F.; Tang, Z.; Niu, B.; Zhang, X.; Wang, Z.; Hong, S. Enhanced removal of bisphenol-AF onto chitosan-modified zeolite by sodium cholate in aqueous solutions. Carbohydr. Polym. 2015, 130, 364–371. [Google Scholar] [CrossRef]
- Araújo, C.S.; Alves, V.N.; Rezende, H.C.; Almeida, I.L.; De Assuncao, R.; Tarley, C.R.; Segatelli, M.G.; Coelho, N.M.M. Characterization and use of Moringa oleifera seeds as biosorbent for removing metal ions from aqueous effluents. Water Sci. Technol. 2010, 62, 2198–2203. [Google Scholar] [CrossRef]
- Elumalai, K.; Velmurugan, S.; Ravi, S.; Kathiravan, V.; Ashokkumar, S. Green synthesis of Zinc oxide nanoparticles using Moringa oleifera leaf extract and evaluation of its antimicrobial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 143, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Reddy, D.H.K.; Seshaiah, K.; Reddy, A.V.R.; Rao, M.M.; Wang, M.C. Biosorption of Pb2+ from aqueous solutions by Moringa oleifera bark: Equilibrium and kinetic studies. J. Hazard. Mater. 2010, 174, 831–838. [Google Scholar] [CrossRef]
- Kumar, Y.P.; King, P.; Prasad, V.S.R.K. Equilibrium and kinetic studies for the biosorption system of copper (II) ion from aqueous solution using Tectona grandis L.f. leaves powder. J. Hazard. Mater. 2006, 137, 1211–1217. [Google Scholar] [CrossRef]
- Rubeš, M.; Koudelková, E.; De Oliveira Ramos, F.S.; Trachta, M.; Bludský, O.; Bulánek, R. Experimental and theoretical study of propene adsorption on K-FER zeolites: New evidence of bridged complex formation. J. Phys. Chem. C 2018, 122, 6128–6136. [Google Scholar] [CrossRef]
- Bennett, R.; Mellon, F.; Perkins, L.; Centre, J.I.; Kroon, P.A. Profiling glucosinolates and phenolics in vegetative and reproductive tissues of the multi-purpose trees Moringa oleifera L. (Horseradish Tree) and Moringa profiling glucosinolates and phenolics in vegetative and reproductive tissues of the multi-purpo. J. Agric. Food Chem. 2003, 51, 3546–3553. [Google Scholar] [CrossRef]
- Cong, N.; Chen, S.; Hsu, H.; Li, C. Separation of three divalent cations (Cu2+, Co2+ and Ni2+) by NF membranes from pHs 3 to 5. DES 2013, 328, 51–57. [Google Scholar] [CrossRef]
- Puigdomenech, I. Hydra/Medusa Chemical Equilibrium Database and Plotting Software; KTH Royal Institute of Technology: Stockholm, Sweden, 2004. [Google Scholar]
- Munagapati, V.S.; Yarramuthi, V.; Nadavala, S.K.; Alla, S.R.; Abburi, K. Biosorption of Cu(II), Cd(II) and Pb(II) by Acacia leucocephala bark powder: Kinetics, equilibrium and thermodynamics. Chem. Eng. J. 2010, 157, 357–365. [Google Scholar] [CrossRef]
- Tofan, L.; Teodosiu, C.; Paduraru, C.; Wenkert, R. Cobalt (II) removal from aqueous solutions by natural hemp fibers: Batch and fixed-bed column studies. Appl. Surf. Sci. 2013, 285, 33–39. [Google Scholar] [CrossRef]

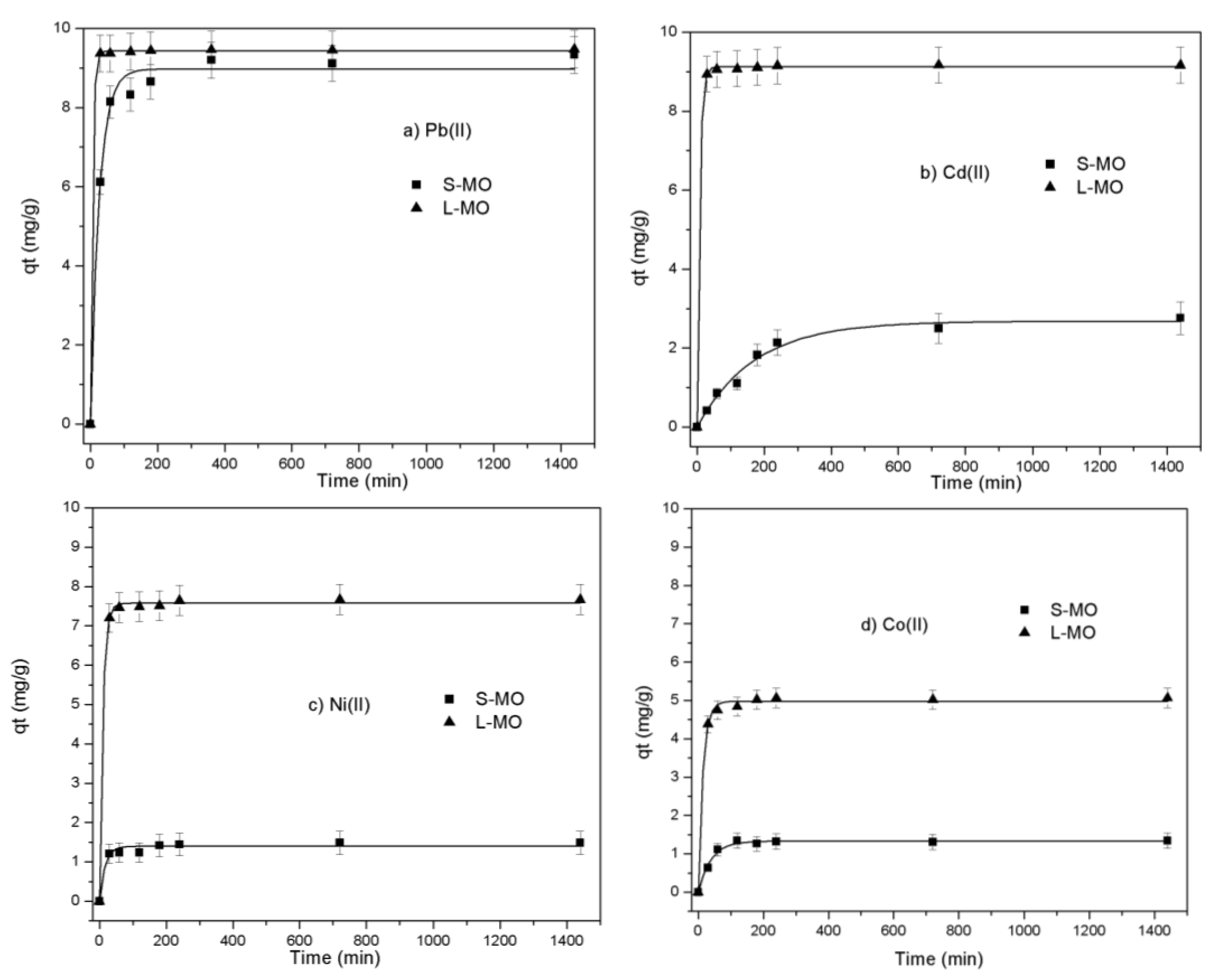
| Metallic Specie | Biosorbent | Kinetic Models | ||||||
|---|---|---|---|---|---|---|---|---|
| Pseudo-Second Order | Pseudo-First Order | |||||||
| - | - | qe,Exp | qe,Cal | K2/(10−2) | R2 | qe,Cal | K1/(10−2) | R2 |
| Pb(II) | L-MO | 9.48 | 9.48 | 12.02 | 1.0 | 0.41 | 0.02 | 0.637 |
| - | S-MO | 9.34 | 9.41 | 0.75 | 0.999 | 1.38 | 0.22 | 0.780 |
| Cd(II) | L-MO | 9.17 | 9.17 | 12.0 | 1.0 | 0.12 | 0.22 | 0.793 |
| - | S-MO | 2.76 | 3.10 | 0.19 | 0.997 | 2.09 | 0.14 | 0.898 |
| Ni(II) | L-MO | 7.68 | 7.68 | 5.28 | 1.0 | 0.35 | 0.47 | 0.919 |
| - | S-MO | 1.48 | 1.50 | 5.08 | 0.999 | 0.30 | 0.43 | 0.922 |
| Co(II) | L-MO | 5.02 | 5.07 | 4.93 | 1.0 | 0.27 | 0.37 | 0.408 |
| S-MO | 1.34 | 1.35 | 4.78 | 0.999 | 0.13 | 0.23 | 0.145 | |
| Isotherm Models | Parameters | Pb | Cd | Ni | Co | ||||
|---|---|---|---|---|---|---|---|---|---|
| L-MO | S-MO | L-MO | S-MO | L-MO | S-MO | L-MO | S-MO | ||
| Freundlich | n | 1.77 | 2.08 | 1.83 | 2.12 | 1.69 | 1.43 | 1.47 | 1.30 |
| 1/n | 0.56 | 0.48 | 0.54 | 0.47 | 0.59 | 0.70 | 0.68 | 0.77 | |
| KF | 5.01 | 3.11 | 1.85 | 0.47 | 0.69 | 0.07 | 0.24 | 0.03 | |
| Langmuir | R2 | 0.95 | 0.98 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 |
| Qm | 49.50 | 13.29 | 16.13 | 4.97 | 10.16 | 3.61 | 10.94 | 5.80 | |
| KL (10−2) | 15.89 | 55.01 | 13.89 | 5.60 | 5.71 | 1.12 | 1.23 | 0.33 | |
| R2 | 0.99 | 0.99 | 0.99 | 0.98 | 0.99 | 0.99 | 0.99 | 0.99 | |
| Metal Ion | Biosorbent | Qm (mg g−1) | Reference |
|---|---|---|---|
| Pb(II) | Pinus eldarica leaves | 40.0 | [16] |
| Prosopis juliflora | 40.32 | [15] | |
| Strychnos potatorum L | 16.42 | [15] | |
| Modified Aspergillus niger | 32.6 | [9] | |
| Moringa oilefera bark | 34.6 | [36] | |
| Moringa oilefera leaves | 45.83 | [28] | |
| Moringa oilefera leaves | 49.50 | In this study | |
| Moringa oilefera seeds | 13.29 | In this study | |
| Modified Moringa oleifera leaves | 171.37 | [42] | |
| Cd(II) | Polyalthia longifolia seeds | 10.56 | [14] |
| Raw and modified Posidonia oceanica | 0.45–0.98 | [17] | |
| Moringa oilefera leaves | 16.13 | In this study | |
| Moringa oilefera seeds | 4.97 | In this study | |
| Co(II) | Diplotaxis harra | 33.02 | [18] |
| Glebionis coronaria L | 24.52 | [18] | |
| Natural hemp fibers | 7.8 | [43] | |
| Moringa oilefera leaves | 10.94 | In this study | |
| Moringa oilefera seeds | 5.80 | In this study | |
| Ni(II) | Modified Aspergillus niger | 0.96 | [9] |
| Leaves Cassia fistula | 145.29 | [19] | |
| Pods bark Cassia fistula | 188.40 | [19] | |
| Pinus eldarica leaves | 33.0 | [16] | |
| Ulva lactuca | 38.28 | [20] | |
| Modified Moringa oleifera leaves | 163.88 | [42] | |
| Modified Moringa oleifera bark | 26.84 | [30] | |
| Moringa pods | 5.53 | [31] | |
| Moringa oilefera leaves | 10.16 | In this study | |
| Moringa oilefera seeds | 3.61 | In this study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abatal, M.; Olguin, M.T.; Anastopoulos, I.; Giannakoudakis, D.A.; Lima, E.C.; Vargas, J.; Aguilar, C. Comparison of Heavy Metals Removal from Aqueous Solution by Moringa oleifera Leaves and Seeds. Coatings 2021, 11, 508. https://doi.org/10.3390/coatings11050508
Abatal M, Olguin MT, Anastopoulos I, Giannakoudakis DA, Lima EC, Vargas J, Aguilar C. Comparison of Heavy Metals Removal from Aqueous Solution by Moringa oleifera Leaves and Seeds. Coatings. 2021; 11(5):508. https://doi.org/10.3390/coatings11050508
Chicago/Turabian StyleAbatal, Mohamed, M. T. Olguin, Ioannis Anastopoulos, Dimitrios A. Giannakoudakis, Eder Claudio Lima, Joel Vargas, and Claudia Aguilar. 2021. "Comparison of Heavy Metals Removal from Aqueous Solution by Moringa oleifera Leaves and Seeds" Coatings 11, no. 5: 508. https://doi.org/10.3390/coatings11050508
APA StyleAbatal, M., Olguin, M. T., Anastopoulos, I., Giannakoudakis, D. A., Lima, E. C., Vargas, J., & Aguilar, C. (2021). Comparison of Heavy Metals Removal from Aqueous Solution by Moringa oleifera Leaves and Seeds. Coatings, 11(5), 508. https://doi.org/10.3390/coatings11050508










