Revisiting Tetra-p-Sulphonated Porphyrin as Antimicrobial Photodynamic Therapy Agent
Abstract
1. Introduction
2. Historical Considerations of aPDT
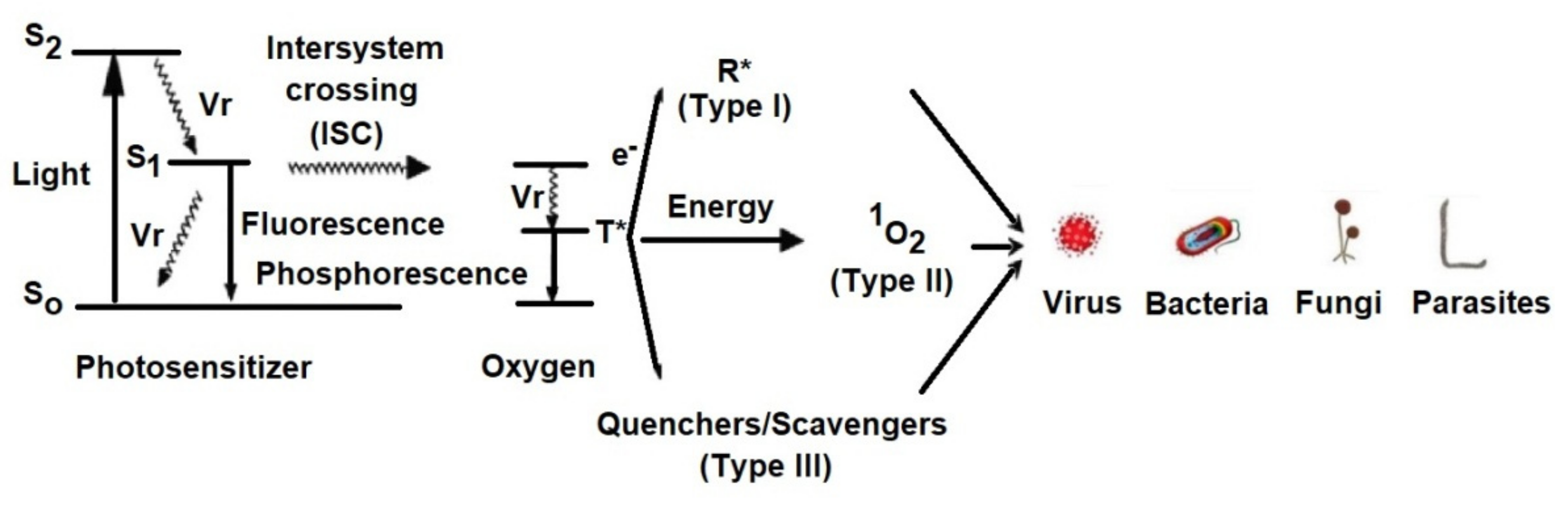
3. Mechanism of aPDT
- Light excites the ground state photosensitizer to an excited singlet state.
- The formation of triplets of excited sensitizer molecule (intersystem crossing).
- However, this state is short lived and can decay to the ground state by radiative or non-radiative transition directly emitting light as fluorescence.
- From this excited state, the photosensitive substance can then return to the ground state by phosphorescence.
- The triplet excited state of the photosensitizer is able to react with oxygen in its triplet state, generating singlet oxygen (type II reaction) or initiating free radical chain reactions with superoxide and hydrogen peroxide ions as well as hydroxyl radicals (type I reaction).
3.1. Type I Mechanism
- hydrogen transfer: 3S * + RH → SH + R˙
- electron transfer: 3S * + RH → S− + RH+
- formation of hydrogen dioxide
- formation of superoxide anion
3.2. Type II Mechanism
- intermolecular exchange
- cellular oxidation
4. Light Sources
5. Photosensitizers Used for Photodynamic Inactivation of Microorganisms
- hydrophobic photosensitizers without peripheral substituents with electric charge and being slightly soluble in water or alcohol (phthalocyanines and naphthalocyanines, hematoporphyrin, hematoporphyrin derivative (HpD), porfimer sodium, and porphyrin precursors)
- hydrophilic photosensitizers that contain three or more peripheral substituents with electric charge and have a high solubility in water at physiological pH.
- amphiphilic photosensitizers that contain one or two peripheral substituents with electric charges, soluble in water or alcohol, at physiological pH. In their structure, there are always two regions, one hydrophobic (represented by porphyrin with electrically charged groups) and another hydrophilic [60].
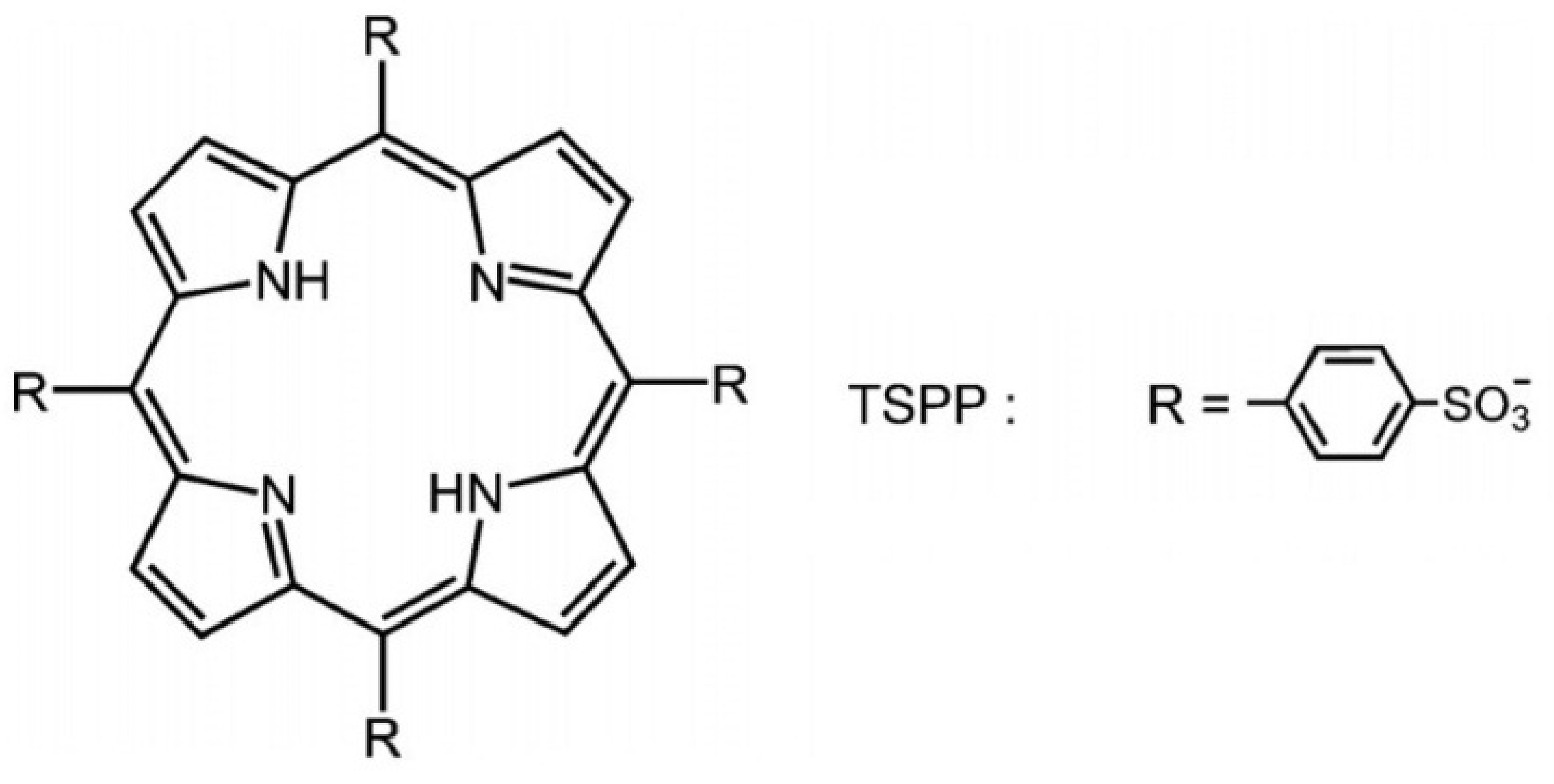
6. Anionic Photosensitizers as Anti-Viral Agent for aPDT
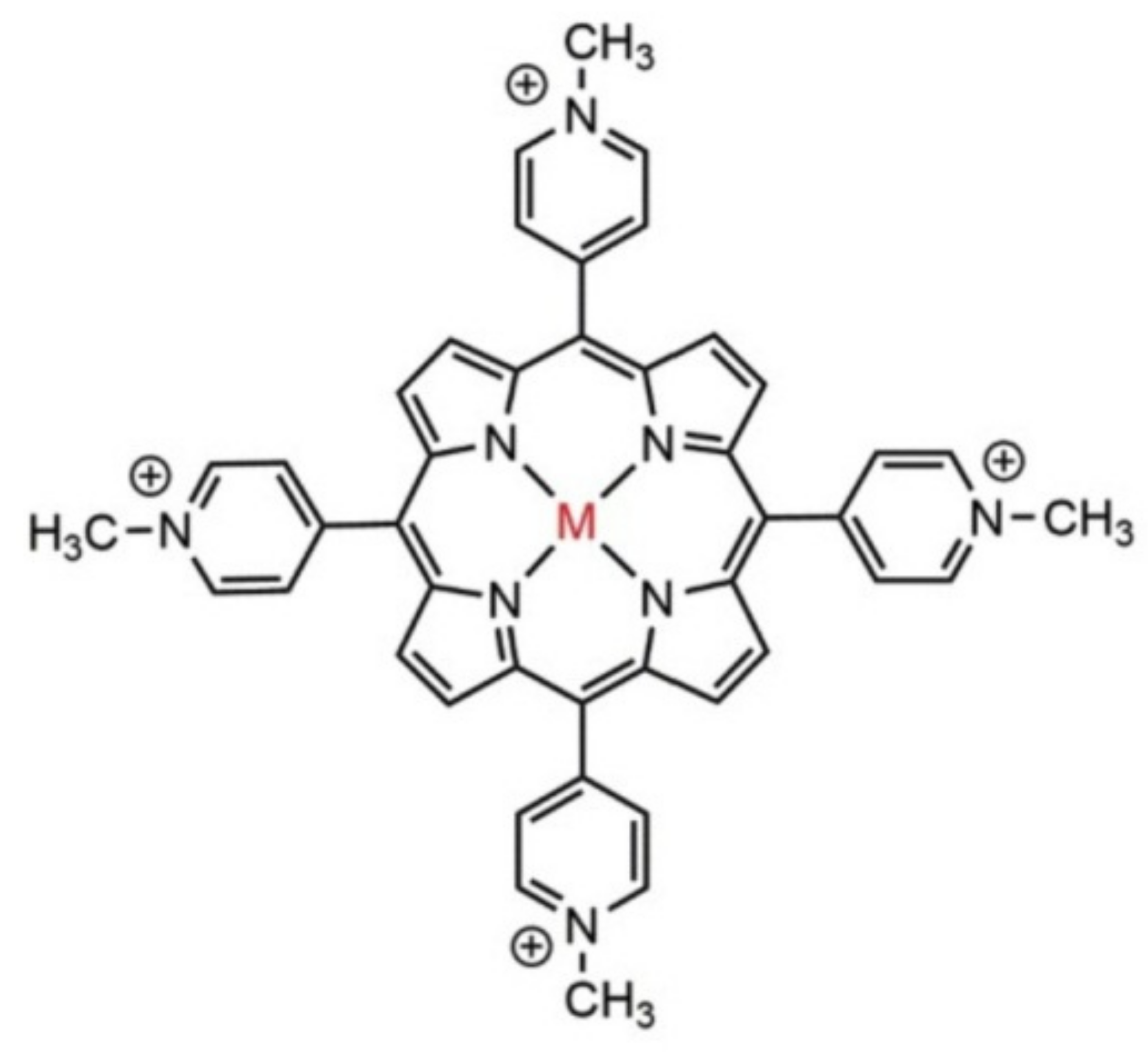
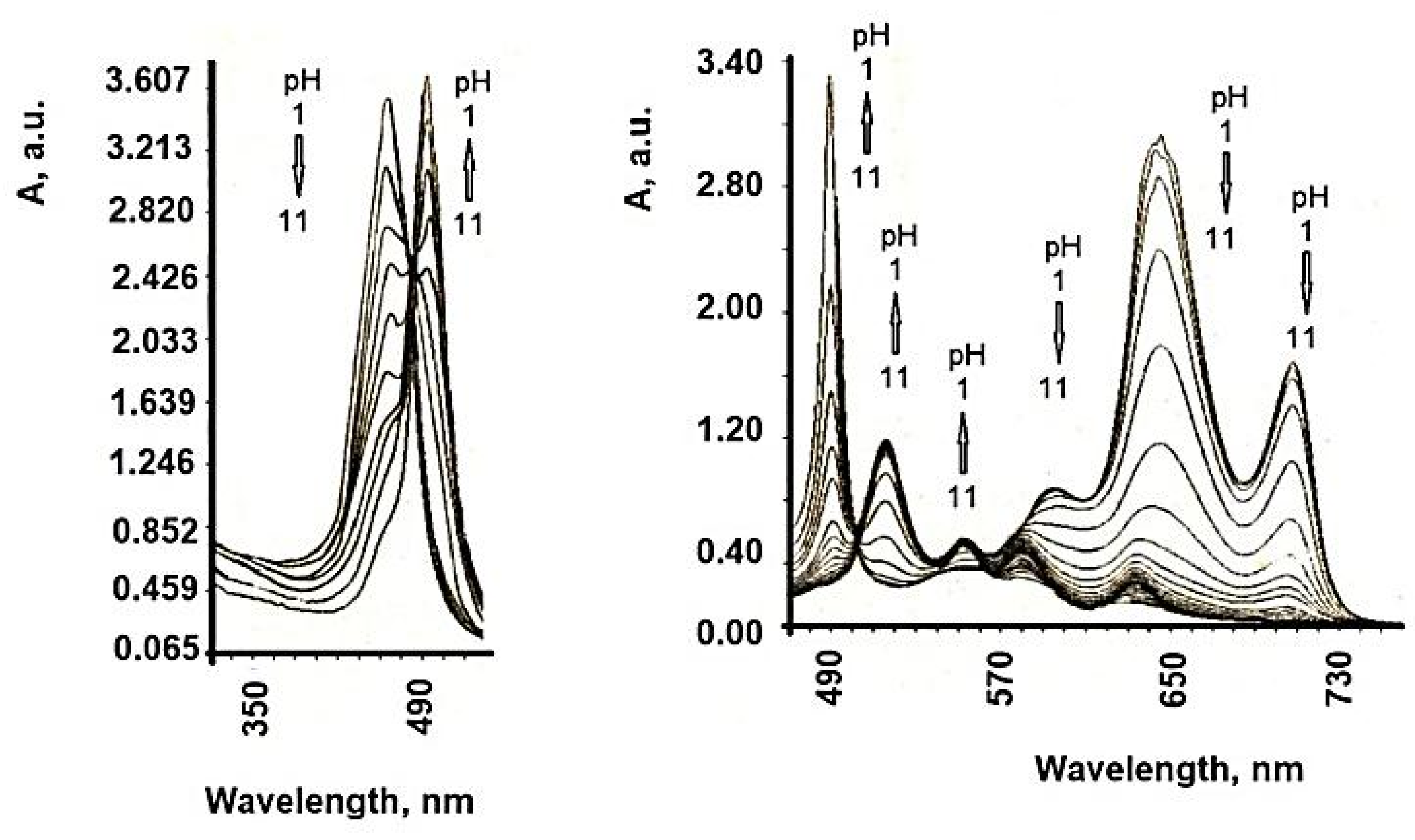
7. Influence of Dicationic (J-Aggregates) TSPP form on aPDT
8. Extension of Studies to SARS-CoV-2 (COVID-19)
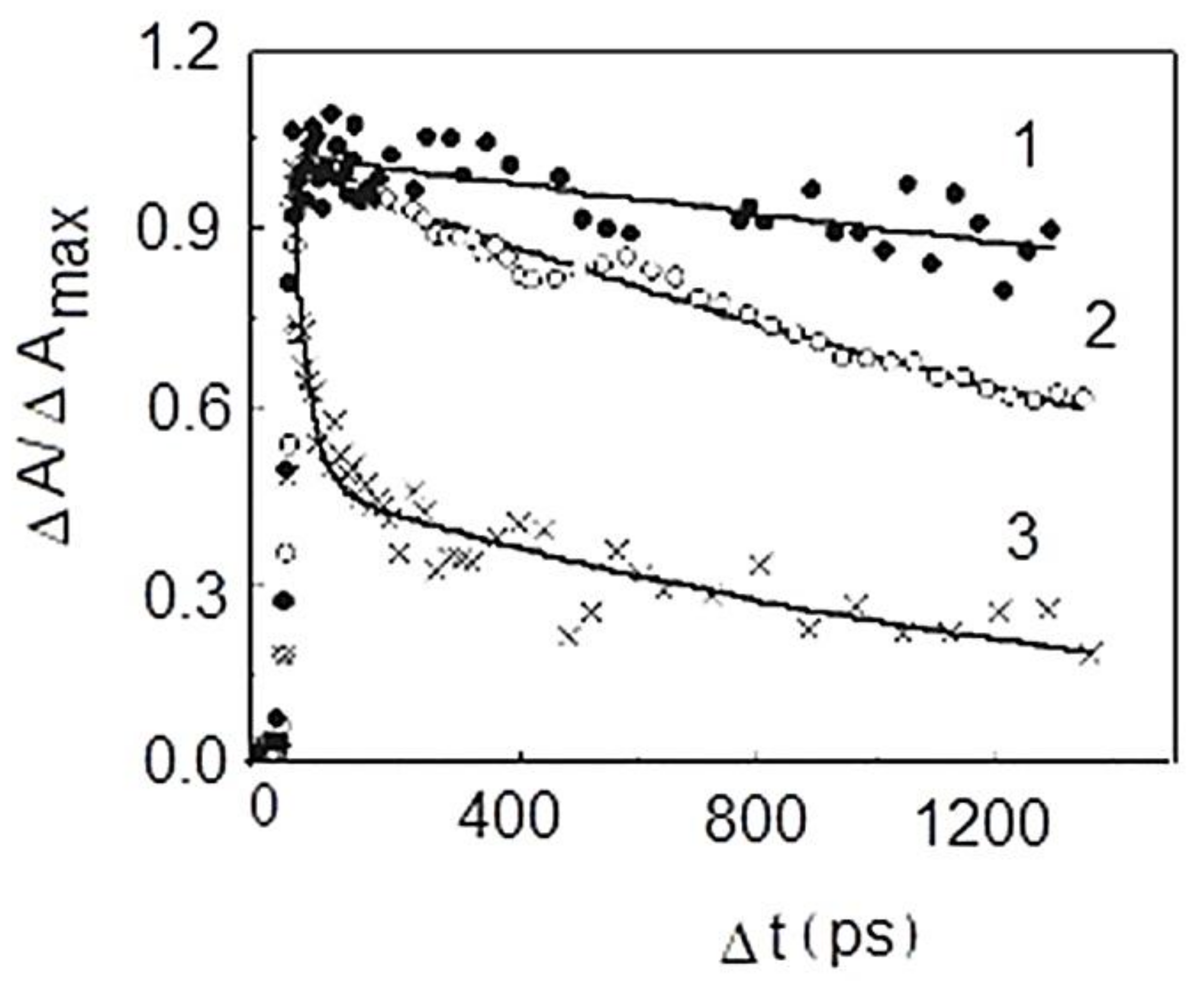
9. Photophysical and Photochemical Properties of PS
10. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Nicholas, J.L. Photo(chemo)therapy: General principles. Clin. Dermatol. 1997, 15, 745–752. [Google Scholar]
- Ion, R.M. The photodynamic therapy of cancer-a photosensitisation or a photocatalytic process? Progr. Catal. 1997, 1, 55–62. [Google Scholar]
- Evensen, J.F.; Moan, J.; Winkelman, J.W. Toxic and phototoxic effects of tetraphenylporphine sulphonate and haematoporphyrin derivative in vitro. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1987, 51, 477–491. [Google Scholar] [CrossRef]
- Qian, P.; Evensen, J.F.; Rimington, C.; Moan, J. A comparison of different photosensitizing dyes with respect to uptake C3H-tumors and tissues of mice. Cancer Lett. 1987, 36, 1–10. [Google Scholar] [CrossRef]
- Boda, D.; Neagu, M.; Constantin, C.; Diaconeasa, A.; Ianosi, S.; Ion, R.M.; Amalinei, C.; Stanoiu, B.; Crauciuc, E.; Toma, O. New photosensitizers versus aminolevulinic acid (ala) in experimental photodynamic therapy of actinic keratosis—A case report. Ann. Alexandru Ioan Cuza Univ. Sect. Genet. Mol. Biol. 2009, 3, 62–69. [Google Scholar]
- Winkelman, J.W.; Collins, G.H. Neurotoxicity of tetraphenylporphinesulfonate TPPS4 and its relation to photodynamic therapy. Photochem. Photobiol. 1987, 46, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Streleckova, E.; Kodetova, D.; Pouckova, P.; Zadinova, M.; Lukas, E.; Rokyta, R.; Jirsa, M. Meso-tetra-(4-sulfonatophenyl)-porphine of low neurotoxicity. SB Lek. 1995, 96, 7–13. [Google Scholar] [PubMed]
- Huang, L.; El-Hussein, A.; Xuan, W.; Hamblin, M.R. Potentiation by potassium iodide reveals that the anionic porphyrin TPPS4 is a surprisingly effective photosensitizer for antimicrobial photodynamic inactivation. J. Photochem. Photobiol. B Biol. 2018, 178, 277–286. [Google Scholar] [CrossRef]
- Nunn, J.F. Ancient Egyptian Medicine; University of Oklahoma Press: Norman, OK, USA, 1996. [Google Scholar]
- Dougherty, T.; Henderson, J.; Schwartz, B.W.; Winkelman, J.W.; Lipson, R.L. Historical Perspective, in Photodynamic Therapy; Henderson, B.W., Dougherty, T.J., Eds.; Marcel Dekker: New York, NY, USA, 1992; pp. 1–15. [Google Scholar]
- Daniell, M.D.; Hill, J.S. A history of photodynamic therapy. Aust. N. Z. J. Surg. 1991, 61, 340–348. [Google Scholar] [CrossRef]
- Raab, O. Ueber die Wirkung fluorescirender Stoffe auf Iufusorien. Z. Biol. 1900, 39, 524. [Google Scholar]
- Yamamoto, N. Photodynamic inactivation of bacteriophage and its inhibition. J. Bacteriol. 1958, 75, 443–448. [Google Scholar] [CrossRef]
- Felber, T.D.; Smith, E.B.; Knox, J.M. Photodynamic inactivation of herpes simplex. JAMA 1973, 22, 289–295. [Google Scholar] [CrossRef]
- Wallis, C.; Melnick, J.L. Photodynamic inactivation of animal viruses: A review. Photochem. Photobiol. 1965, 4, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Sperandio, F.F.; Huang, Y.-Y.; Hamblin, M.R. Antimicrobial photodynamic therapy to kill gram-negative bacteria. Recent Pat. Anti-Infect. Drug Discov. 2013, 8, 108–120. [Google Scholar] [CrossRef]
- Simon, M.I.; Vunakis, V. The photodynamic reaction of methylene blue with DNA. J. Mol. Biol. 1962, 4, 488–493. [Google Scholar] [CrossRef]
- Embleton, M.L.; Nair, S.P.; Heywood, W.; Menon, D.C.; Cookson, B.C.; Wilson, M. Development of a novel targeting system for lathal photosensitization of antibiotic-resistant strains of Staphylococcus aureus. Antimicrob. Agents Chemother. 2005, 49, 3690–3696. [Google Scholar] [CrossRef]
- Sahu, K.; Sharma, M.; Bansal, H.; Dube, A.; Gupta, P.K. Topical photodynamic treatment with poly-l-lysine–chlorin p6 conjugate improves wound healing by reducing hyperinflammatory response in Pseudomonas aeruginosa-infected wounds of mice. Lasers Med. Sci. 2012, 28, 465–471. [Google Scholar] [CrossRef]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy—What we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef] [PubMed]
- Sobotta, L.; Skupin-Mrugalska, P.; Mielcarek, J.; Goslinski, T.; Balzarini, J. Photosensitizers mediated photodynamic inactivation against virus particles. Mini-Rev. Med. Chem. 2015, 15, 503–521. [Google Scholar] [CrossRef]
- Janouskova, O.; Rakusan, J.; Karaskova, M.; Holada, K. Photodynamic inactivation of prions by disulfonated hydroxyaluminium phthalocyanine. J. Gen. Virol. 2012, 93, 2512–2517. [Google Scholar] [CrossRef][Green Version]
- Strasfeld, L.; Chou, S. Antiviral drug resistance: Mechanisms and clinical implications. Infect. Dis. Clin. N. Am. 2010, 24, 413–437. [Google Scholar] [CrossRef]
- Ion, R.M.; Corobea, M.C. Porphyrin models as sensitizers in photodynamic inactivation of Herpes Simplex Virus. In Proceedings of the IV International Conference on Antimicrobial Research—ICAR 2016, Torremolinos-Malaga, Spain, 29 June–1 July 2016. [Google Scholar]
- Wiehe, A.; O’Brien, J.M.; Senge, M.O. Trends and targets in antiviral phototherapy. Photochem. Photobiol. Sci. 2019, 18, 2565–2612. [Google Scholar] [CrossRef]
- Li, W.-Y.; Xu, J.-G.; He, X.-W. Characterization of the binding of methylene blue to DNA by spectroscopic methods. Anal. Lett. 2000, 33, 2453–2464. [Google Scholar] [CrossRef]
- Baptista, M.S.; Cadet, J.; Di Mascio, P.; Ghogare, A.A.; Greer, A.; Hamblin, M.R.; Lorente, C.; Nunez, S.C.; Ribeiro, M.S.; Thomas, A.H.; et al. Type I and type II photosensitized oxidation reactions: Guidelines and mechanistic pathways. Photochem. Photobiol. 2017, 93, 912–919. [Google Scholar] [CrossRef]
- Zhang, B.; Zheng, L.; Huang, Y.; Mo, Q.; Wang, X.; Qian, K. Detection of nucleic acid lesions during photochemical inac-tivation of RNA viruses by treatment with methylene blue and light using real-time PCR. Photochem. Photobiol. 2011, 87, 365–369. [Google Scholar] [CrossRef]
- Banerjee, I.; Douaisi, M.P.; Mondal, D.; Kane, R.S. Light-activated nanotube–porphyrin conjugates as effective antiviral agents. Nanotechnology 2012, 23, 105101. [Google Scholar] [CrossRef]
- Morikawa, K.; Suda, G.; Sakamoto, N. Viral life cycle of hepatitis B virus: Host factors and druggable targets. Hepatol. Res. 2016, 46, 871–877. [Google Scholar] [CrossRef]
- De Clercq, E.; Li, G. Approved antiviral drugs over the past 50 years. Clin. Microbiol. Rev. 2016, 29, 695–747. [Google Scholar] [CrossRef]
- Lenard, J.; Vanderoef, R. Photoinactivation of influenza virus fusion and infectivity by rose Bengal. Photochem. Photobiol. 1993, 58, 527–531. [Google Scholar] [CrossRef]
- Monjo, A.L.-A.; Pringle, E.S.; Thornbury, M.; Duguay, B.A.; Monro, S.M.A.; Hetu, M.; Knight, D.; Cameron, C.G.; McFarland, S.A.; McCormick, C. Photodynamic activation of herpes simplex viruses. Viruses 2018, 10, 532. [Google Scholar] [CrossRef]
- Belanger, J.M.; Raviv, Y.; Viard, M.; de la Cruz, J.M.; Nagashima, K.; Blumenthal, R. Characterization of the effects of ar-yl-azido compounds and UVA irradiation on the viral proteins and infectivity of human immunodeficiency virus type 1. Photochem. Photobiol. 2010, 86, 1099–1108. [Google Scholar] [CrossRef]
- Warfield, K.L.; Swenson, D.L.; Olinger, G.G.; Kalina, W.V.; Viard, M.; Aitichou, M.; Chi, X.; Ibrahim, S.; Blumenthal, R.; Raviv, Y.; et al. Ebola virus inactivation with preservation of antigenic and structural integrity by a photoinducible alkylating agent. J. Infect. Dis. 2007, 196, S276–S283. [Google Scholar] [CrossRef]
- Raviv, Y.; Blumenthal, R.; Tompkins, S.M.; Humberd, J.; Hogan, R.J.; Viard, M. Hydrophobic inactivation of influenza viruses confers preservation of viral structure with enhanced immunogenicity. J. Virol. 2008, 82, 4612–4619. [Google Scholar] [CrossRef]
- Dairou, J.; Vever-Bizet, C.; Brault, D. Interaction of sulfonated anionic porphyrins with HIV glycoprotein gp120: Photo-damages revealed by inhibition of antibody binding to V3 and C5 domains. Antiviral Res. 2004, 61, 37–47. [Google Scholar] [CrossRef]
- Tavares, A.; Carvalho, C.M.B.; Faustino, M.A.; Neves, M.G.P.M.S.; Tomé, J.P.C.; Tomé, A.C.; Cavaleiro, J.A.S.; Cunha, Â.; Gomes, N.C.M.; Alves, E.; et al. Antimicrobial photodynamic therapy: Study of bacterial recovery viability and potential development of resistance after treatment. Mar. Drugs 2010, 8, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.; Cunha, A.; Faustino, M.A.F.; Tomé, A.C.; Neves, M.G.P.M.S. Porphyrins as antimibrobial photosensitizing agents. In Photodynamic Inactivation of Microbial Pathogens: Medical and Environmental Applications; Hamblin, M.R., Jori, G., Eds.; Royal Society of Chemistry: Cambridge, UK, 2011; pp. 83–160. [Google Scholar]
- Vardevanyan, P.O.; Antonyan, A.P.; Parsadanyan, M.A.; Shahinyan, M.A.; Ham-bardzumyan, L.A. Mechanisms for binding between methylene blue and DNA. J. Appl. Spectrosc. 2013, 80, 595–599. [Google Scholar] [CrossRef]
- Fekrazad, R. Photobiomodulation and antiviral photodynamic therapy as a possible novel approach in COVID-19 management. PhotobiomodulationPhotomed. Laser Surg. 2020, 38, 255–257. [Google Scholar] [CrossRef]
- Almeida, A.; Faustino, M.A.F.; Neves, M.G.P.M.S. Antimicrobial photodynamic therapy in the control of COVID-19. Antibiotics 2020, 9, 320. [Google Scholar] [CrossRef]
- Bond, P. Ethnicity and the relationship between covid-19 and the herpes simplex viruses. Med. Hypotheses 2021, 146, 110447. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I.; et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- Mesquita, M.Q.; Dias, C.; Neves, M.G.P.M.S.; Almeida, A.; Faustino, M.A.F. Revisiting current photoactive materials for an-timicrobial photodynamic therapy. Molecules 2018, 23, 2424. [Google Scholar] [CrossRef]
- Foote, C.S. Mechanisms of photosensitized oxidation. Science 1968, 162, 963–970. [Google Scholar] [CrossRef]
- Ion, R.M. Near-Infrared Dyes for High Technology Applications; Daehne, S., Resch-Genger, U., Wolfbeis, O., Eds.; NATO ASI SERIES; Kluwer Academic Publishers: Dordrecht, The Netherlands; Boston, MA, USA; London, UK, 1998; Volume 3/52, pp. 87–114. ISBN 0-7923-5101-0. [Google Scholar]
- Kato, H.; Komagoe, K.; Inoue, T.; Masuda, K.; Katsu, T. Structure–activity relationship of porphyrin-induced photoinactivation with membrane function in bacteria and erythrocytes. Photochem. Photobiol. Sci. 2018, 17, 954–963. [Google Scholar] [CrossRef]
- Wainwright, M. Photodynamic antimicrobial chemotherapy (PACT). J. Antimicrob. Chemother. 1998, 42, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Bertoloni, G.; Lauro, F.M.; Cortella, G.; Merchat, M. Photosensitizing activity of hematoporphyrin on Staphylococcus aureus cells. Biochim. Biophys. Acta Gen. Subj. 2000, 1475, 169–174. [Google Scholar] [CrossRef]
- Ion, R.M.; Planner, A.; Wiktorowicz, K.; Frackowiak, D. Incorporation of various porphyrins into human blood cells measured using the flow-cytometry, the absorption and emission spectroscopy. Acta Biochim. Pol. 1998, 45, 833–842. [Google Scholar] [CrossRef]
- Salva, K.A. Photodynamic therapy: Unapproved uses, dosages, or indications. Clin. Derm. 2002, 20, 571–581. [Google Scholar] [CrossRef]
- Juzeniene, A.; Juzenas, P.; Ma, L.-W.; Iani, V.; Moan, J. Effectiveness of different light sources for 5-aminolevulinic acid photodynamic therapy. Lasers Med. Sci. 2004, 19, 139–149. [Google Scholar] [CrossRef]
- Koshi, E.; Mohan, A.; Rajesh, S.; Philip, K. Antimicrobial photodynamic therapy: An overview. J. Indian Soc. Periodontol. 2011, 15, 323–327. [Google Scholar] [CrossRef]
- Kashef, N.; Huang, Y.-Y.; Hamblin, M.R. Advances in antimicrobial photodynamic inactivation at the nanoscale. Nanophotonics 2017, 6, 853–879. [Google Scholar] [CrossRef]
- Hu, X.; Huang, Y.-Y.; Wang, Y.; Wang, X.; Hamblin, M.R. Antimicrobial photodynamic therapy to control clinically relevant biofilm infections. Front. Microbiol. 2018, 9, 1299. [Google Scholar] [CrossRef]
- Kuo, W.-S.; Chang, C.-Y.; Chen, H.-H.; Hsu, C.-L.L.; Wang, J.-Y.; Kao, H.-F.; Chou, L.C.-S.; Chen, Y.-C.; Chen, S.-J.; Chang, W.-T.; et al. Two-photon photoexcited photodynamic therapy and contrast agent with antimicrobial graphene quantum dots. ACS Appl. Mater. Interfaces 2016, 8, 30467–30474. [Google Scholar] [CrossRef]
- Wozniak, A.; Grinholc, M. Combined antimicrobial activity of photodynamic inactivation and antimicrobials–state of the art. Front. Microbiol. 2018, 9, 930. [Google Scholar] [CrossRef] [PubMed]
- Bartolomeu, M.; Coimbra, S.; Cunha, A.; Neves, M.G.P.M.S.; Cavaleiro, J.A.S.; Faustino, M.A.F.; Almeida, A. Indirect and direct damage to genomic DNA induced by 5,10,15-tris(1-methylpyridinium-4-yl)-20-(pentafluorophenyl)porphyrin upon photody-namic action. J. Porph. Phthal. 2016, 20, 331–336. [Google Scholar] [CrossRef]
- Wainwright, M.; McLean, A. Rational design of phenothiazinium derivatives and photoantimicrobial drug discovery. Dyes Pigments 2017, 136, 590–600. [Google Scholar] [CrossRef]
- Yao, T.-T.; Wang, J.; Xue, Y.-F.; Yu, W.-J.; Gao, Q.; Ferreira, L.; Ren, K.-F.; Ji, J. A photodynamic antibacterial spray-coating based on the host–guest immobilization of the photosensitizer methylene blue. J. Mater. Chem. B 2019, 7, 5089–5095. [Google Scholar] [CrossRef]
- Friedman, L.I.; Skripchenko, A.; Wagner, S.J. Photodynamic Inactivation of Pathogens in Blood by Phenothiazines and Oxygen. Patent WO/2001/049328, 28 December 2000. [Google Scholar]
- Ion, R.-M.; Boda, D. Porphyrin—Based supramolecular nanotubes generated by aggregation processes. Rev. Chim. 2008, 59, 205–207. [Google Scholar] [CrossRef]
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagen. 2017, 58, 235–263. [Google Scholar] [CrossRef]
- Snipes, W.; Keller, G.; Woog, J.; Vickroy, T.; Deering, R.; Keith, A. Inactivation of lipid-containing viruses by hydrophobic photosensitizers and near-UV radiation. Photochem. Photobiol. 1979, 29, 780–785. [Google Scholar] [CrossRef]
- Le Gall, T.; Lemercier, G.; Chevreux, S.; Tucking, K.-S.; Ravel, J.; Thetiot, F.; Jonas, U.; Schönherr, H.; Montier, T. Ruthenium (II) polypyridyl complexes as photosensitizers for antibacterial photodynamic therapy: A structure-activity study on clinical bac-terial strains. ChemMedChem 2018, 13, 2229–2239. [Google Scholar] [CrossRef]
- Minnock, A.; Vernon, D.I.; Schofield, J.; Griffiths, J.; Parish, J.H.; Brown, S.B. Mechanism of uptake of a cationic water-soluble pyridinium zinc phthalocyanine across the outer membrane of Escherichia coli. Antimicrob. Agents Chemo-ther. 2000, 44, 522–527. [Google Scholar] [CrossRef]
- Hancock, R.E.W. Alterations in outer membrane permeability. Annu. Rev. Microbiol. 1984, 38, 237–264. [Google Scholar] [CrossRef]
- Hancock, R.E. The bacterial outer membrane as a drug barrier. Trends Microbiol. 1997, 5, 37–42. [Google Scholar] [CrossRef]
- Hancock, R.E.; Farmer, S.W. Mechanism of uptake of deglucoteicoplanin amide derivatives across outer membranes of Escherichia coli and Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 1993, 37, 453–456. [Google Scholar] [CrossRef]
- Vieira, C.; Gomes, A.T.; Mesquita, M.Q.; Moura, N.M.M.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Almeida, A. An insight into the potentiation effect of potassium iodide on aPDT e_cacy. Front. Microbiol. 2018, 9, 2665–2670. [Google Scholar] [CrossRef]
- Vieira, C.; Santos, A.; Mesquita, M.Q.; Gomes, A.T.P.C.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Almeida, A. Advances in aPDT based on the combination of a porphyrinic formulation with potassium iodide: Effectiveness on bacteria and fungi plankton-ic/biofilm forms and viruses. J. Porph. Phthal. 2019, 23, 534–545. [Google Scholar] [CrossRef]
- Costa, L.; Tomé, J.P.C.; Neves, M.D.G.P.M.S.; Tomé, A.C.; Cavaleiro, J.A.S.; Cunha, A.; Faustino, M.A.F.; Almeida, A. Sus-ceptibility of non-enveloped DNA- and RNA-type viruses to photodynamic inactivation. Photochem. Photobiol. Sci. 2012, 11, 1520–1530. [Google Scholar] [CrossRef]
- Wu, J.J.; Li, N.; Li, K.A.; Liu, F. J-aggregates of diprotonated tetrakis(4- sulfonatophenyl)porphyrin induced by ionic liquid 1-butyl-3-methylimidazolium tetrafluoroborate. J. Phys. Chem. B 2008, 112, 8134–8138. [Google Scholar] [CrossRef]
- Kemnitz, K.; Sakaguchi, T. Water-soluble porphyrin monomer-dimer systems: Fluorescence dynamics and thermodynamic properties. Chem. Phys. Lett. 1992, 196, 497–502. [Google Scholar] [CrossRef]
- Ribo, J.M.; Crusats, J.; Farrera, J.-A.; Valero, M.L. Aggregation in water solutions of tetrasodium diprotonated meso-tetrakis (4-sulfonatophenyl) porphyrin. J. Chem. Soc. Chem. Comm. 1974, 6, 681–690. [Google Scholar] [CrossRef]
- Hattori, S.; Ishii, K. Magneto-chiral dichroism of aromatic conjugated systems. Opt. Mater. Express 2014, 4, 2423–2432. [Google Scholar] [CrossRef]
- Farjtabar, A.; Gharib, F.; Farajtabar, A. Solvent effect on protonation constants of 5, 10, 15, 20-tetrakis(4-sulfonatophenyl)porphyrin in different aqueous solutions of methanol and ethanol. J. Solut. Chem. 2010, 39, 231–244. [Google Scholar] [CrossRef]
- Kobayashi, T. (Ed.) J-Aggregates; World Scientific Publishing: Singapore, 1996. [Google Scholar]
- Corsini, A.; Herrmann, O. Aggregation of meso-tetra-(p-sulphonatophenyl) porphine and its Cu(II) and Zn (II) complexes in aqeous solution. Talanta 1986, 33, 335–339. [Google Scholar] [CrossRef]
- Cunderlikova, B.; Bjørklund, E.G.; Pettersen, E.O.; Moan, J. pH-dependent spectral properties of HpIX, TPPS2a, mTHPP and mTHPC. Photochem. Photobiol. 2001, 74, 246–252. [Google Scholar] [CrossRef]
- Kadish, K.M.; Maiya, G.B.; Araullo, C.; Guillard, R. Micellar effects on the aggregation of tetraanionic porphyrins. Spectroscopic characterization of free-base meso-tetrakis(4-sulfonatophenyl)porphyrin, (TPPS)H2, and (TPPS)M (M = Zn(II), Cu(II), V02+) in aqueous micellar media. Inorg. Chem. 1989, 28, 2125–2131. [Google Scholar] [CrossRef]
- Kano, K.; Takei, M.; Hashimoto, S. Cationic porphyrins in water. 1H NMR and fluorescence studies on dimer and molecular complex formation. J. Phys. Chem. 1990, 94, 181–187. [Google Scholar] [CrossRef]
- Kano, K.; Tanaka, N.; Minamizono, H.; Kawakita, Y. Tetraarylporphyrins as probes for studying mechanism of inclusion-complex formation of cyclodextrins. Effect of microscopic environment on inclusion of ionic guests. Chem. Lett. 1996, 25, 925–926. [Google Scholar] [CrossRef]
- Valanciunaite, J.; Poderys, V.; Bagdonas, S.; Rotomskis, R.; Selskis, A. Protein induced formation of porphyrin (TPPS4) nanostructures. J. Phys. Conf. Ser. 2007, 61, 1207–1211. [Google Scholar] [CrossRef]
- Aggarwal, L.P.F.; Borissevitch, I.E. On the dynamics of the TPPS4 aggregation in aqueous solutions: Successive formation of H and J aggregates. Spectrochim. Acta Part A Mol. Biomol. Spectr. 2006, 63, 227–233. [Google Scholar] [CrossRef]
- Faraon, V.; Ion, R.-M.; Pop, S.-F.; Van-Staden, R.; Van-Staden, J.-F. Porphyrins as molecular nanomaterials. In Proceedings of the SPIE 7821, Advanced Topics in Optoelectronics, Microelectronics, and Nanotechnologies, V, 78212H, Constanta, Romania, 4 December 2010. [Google Scholar] [CrossRef]
- Jori, G.; Coppellotti, O. Inactivation of pathogenic microorganisms by photodynamic techniques: Mechanistic aspects and perspective applications. Anti-Infect. Agents Med. Chem. 2007, 6, 913–931. [Google Scholar] [CrossRef]
- Gottfried, V.; Kimmel, S. Temperature effects on photosensitized processes. J. Photochem. Photobiol. B Biol. 1991, 8, 419–430. [Google Scholar] [CrossRef]
- Kochevar, I.E.; Bouvier, J.; Lynch, M.; Lin, C.W.; Chi-Wei, L. Influence of dye and protein location on photosensitization of the plasma membrane. Biochim. Biophys. Acta Biomembr. 1994, 1196, 172–180. [Google Scholar] [CrossRef]
- Lytle, C.; Carney, P.; Felten, R.; Bushar, H.; Straight, R. Inactivation and mutagenesis of herpes virus by photodynamic treatment with therapeutic dyes. Photochem. Photobiol. 1989, 50, 367–371. [Google Scholar] [CrossRef]
- Ion, R.M.; Safta, I.; Natile, G. Photodynamic Inactivation of Herpes Simplex Viruses with Porphyrin Derivatives. Available online: http://www.photobiology.com/photobiology2000/rodica1/index.htm (accessed on 24 February 2021).
- Smetana, Z.; Ben-Hur, E.; Mendelson, E.; Salzberg, S.; Wagner, P.; Malik, Z. Herpes simplex virus proteins are damaged following photodynamic inactivation with phthalocyanines. J Photochem Photobiol B. 1998, 15, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Gradova, M.A.; Kuryakov, V.N.; Lobanov, A.V. The role of the counterions in self-assembly of j-aggregates from meso-aryl substituted porphyrin diacids in aqueous solutions. Macroheterocycles 2015, 8, 244–251. [Google Scholar] [CrossRef]
- Conrado, P.C.V.; Sakita, K.M.; Arita, G.S.; Galinari, C.B.; Gonçalves, R.S.; Lopes, L.D.G.; Lonardoni, M.V.C.; Teixeira, J.J.V.; Bonfim-Mendonça, P.S.; Kioshima, E.S. A systematic review of photodynamic therapy as an antiviral treatment: Potential guidance for dealing with SARS-CoV-2. Photodiagnosis Photodyn Ther. 2021, 34, 102221. [Google Scholar] [CrossRef] [PubMed]
- Frackowiak, D.; Planner, A.; Ion, R.M.; Wiktorowicz, K. Incorporation of dyes in resting and stimulated leukocytes. In Synthesis, Properties and Applications of Near-Infrared Dyes in High Technology Fields; NATO ASI Series; Daehne, S., Ed.; Springer: Dordrecht, The Netherlands, 1998. [Google Scholar]
- Afrasiabi, S.; Pourhajibagher, M.; Raoofian, R.; Tabarzad, M.; Bahador, A. Therapeutic applications of nucleic acid aptamers in microbial infections. J. Biomed. Sci. 2020, 27, 6–13. [Google Scholar] [CrossRef]
- Heidary, F.; Gharebaghi, R. Ivermectin: A systematic review from antiviral effects to COVID-19 complementary regimen. J. Antibiot. 2020, 73, 593–602. [Google Scholar] [CrossRef]
- Mettenleiter, T.C.; Klupp, B.G.; Granzow, H. Herpes Virus assembly: A tale of two membranes. Curr. Opin. Microbiol. 2006, 9, 423–429. [Google Scholar] [CrossRef]
- Kipshidze, N.; Yeo, N.; Kipshidze, N. Photodynamic therapy for COVID-19. Nat. Photonics 2020, 14, 651–652. [Google Scholar] [CrossRef]
- Kharkwal, G.B.; Sharma, S.K.; Huang, Y.Y.; Dai, T.; Hamblin, M.R. Lasers in surgery and medicine. Lasers Surg. Med. 2011, 43, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Majiya, H.; Adeyemi, O.O.; Stonehouse, N.J.; Millner, P. Photodynamic inactivation of bacteriophage MS2: The A-protein is the target of virus inactivation. J. Photochem. Photobiol. B Biol. 2018, 178, 404–411. [Google Scholar] [CrossRef]
- Bojadzic, D.; Alcazar, O.; Buchwald, P. Methylene Blue Inhibits the SARS-CoV-2 Spike–ACE2 Protein-Protein Interaction—A Mechanism that can Contribute to its Antiviral Activity Against COVID-19. Frontiers in Pharmacology. 2021, 11, 600372. [Google Scholar] [CrossRef]
- Baltazar, L.M.; Eray, A.; Santos, D.A.; Cisalpino, P.S.; Friedman, A.J.; Nosanchuk, J.D. Antimicrobial photodynamic therapy: An effective alternative approach to control fungal infections. Front. Microbiol. 2015, 6, 202. [Google Scholar] [CrossRef]
- Svyatchenko, V.A.; Nikonov, S.D.; Mayorov, A.P.; Gelfond, M.L.; Loktev, V.B. Antiviral photodynamic therapy: Inactivation and inhibition of SARS-CoV-2 in vitro using methylene blue and radahlorin. Photodiagnosis Photodyn. Ther. 2020, 33, 102112. [Google Scholar] [CrossRef]
- Engelhardt, V.; Krammer, B.; Plaetzer, K. Antibacterial photodynamic therapy using water-soluble formulations of hy-pericin or mTHPC is effective in inactivation of Staphylococcus aureus. Photochem. Photobiol. Sci. 2010, 9, 365–369. [Google Scholar] [CrossRef]
- Ogilby, R. Singlet oxygen: There is indeed something new under the sun. Chem. Soc. Rev. 2020, 39, 3181–3209. [Google Scholar] [CrossRef]
- Ericson, M.B.; Grapengiesser, S.; Gudmundson, F.; Wennberg, A.-M.; Larkö, O.; Moan, J.; Rosén, A. A spectroscopic study of the photobleaching of protoporphyrin IX in solution. Lasers Med. Sci. 2003, 18, 56–62. [Google Scholar] [CrossRef]
- Demidova, T.N.; Hamblin, M.R. Photodynamic therapy targeted to pathogens. Int. J. Immunopathol. Pharmacol. 2004, 17, 245–254. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, J.M.; Gaffney, D.K.; Wang, T.P.; Sieber, F. Merocyanine 540-sensitized photoinactivation of enveloped viruses in blood products: Site and mechanism of phototoxicity. Blood 1992, 80, 277–285. [Google Scholar] [CrossRef]
- Narayana Rao, D.; Venugopal Rao, S.; Aranda, F.J.; Rao, D.V.G.L.H.; Nakashima, M.; Akkara, J.A. Ultrafast relaxation times of matalloporphyrins by time-resolved degenerate four-wave mixing with incoherent light. J. Opt. Soc. Am. B 1997, 14, 2710–2715. [Google Scholar] [CrossRef]
- Rodriguez, J.; Kirmaier, C.; Holten, D. Optical properties of metalloporphyrin excited states. J. Am. Chem. Soc. 1989, 111, 6500–6506. [Google Scholar] [CrossRef]
- Olejarz, B.; Bursa, B.; Szyperska, I.; Ion, R.-M.; Dudkowiak, A. Spectral properties and deactivation processes of anionic porphyrin coupled with TiO2 nanostructure. Int. J. Thermophys. 2009, 31, 163–171. [Google Scholar] [CrossRef]
- Chow, Y.F.A.; Dolphin, D.; Paine, J.P.; McGarvey, D.; Pottier, R.; Truscott, T.G. The excited states of covalently linked dimeric porphyrins: I: The excited singlet states. J. Photochem. Photobiol. B Biol. 1988, 2, 253–263. [Google Scholar] [CrossRef]
- Akins, D.L.; Özelik Zhu, V.S.H.-R.; Guo, C. Absorption and Raman scattering by aggregated meso-Tetrakis(p-sulfonatophenyl)porphine. J. Phys. Chem. 1996, 100, 14390–14396. [Google Scholar] [CrossRef]
- Kalyanasundaram, K.; Neumann-Spallart, M. Photophysical and redox properties of water-soluble porphyrins in aqueous media. J. Phys. Chem. 1982, 86, 5163–5169. [Google Scholar] [CrossRef]
- Karns, G.A.; Gallagher, W.A.; Elliott, W.B. Dimerization constants of water-soluble porphyrins in aqueouse alkali. Bioorg. Chem. 1979, 8, 69–81. [Google Scholar] [CrossRef]
- Luciano, M.; Brückner, C. Modifications of Porphyrins and Hydroporphyrins for Their Solubilization in Aqueous Media. Molecules 2017, 22, 980. [Google Scholar] [CrossRef]
- Tran-Thi, T.H.; Lipskier, J.F.; Maillard, P.; Momenteau, M.; Lopez-Castillo, J.L.; Jay-Gerin, J.-P. Effect of the exciton Coupling on the optical and photophysical properties of face-to-face porphyrin dimer and trimer. A treatment including the solvent stabilization effect. J. Phys. Chem. 1992, 96, 1073–1082. [Google Scholar] [CrossRef]
- Almeida, M.A.; Cavaleiro, J.A.S.; Rocha, J.; Carvalho, C.M.B.; Costa, L.A.S.; Alves, E.S.C.F.; Cunha, M.A.S.D.A.; Tomé, J.P.C.; Faustino, M.A.F.; Neves, M.G.P.M.S.; et al. Nanomagnet-Porphyrin Hybrid Materials: Synthesis and Water Disinfection Application. Portuguese Patent No. PT 103828, 21 September 2009. [Google Scholar]
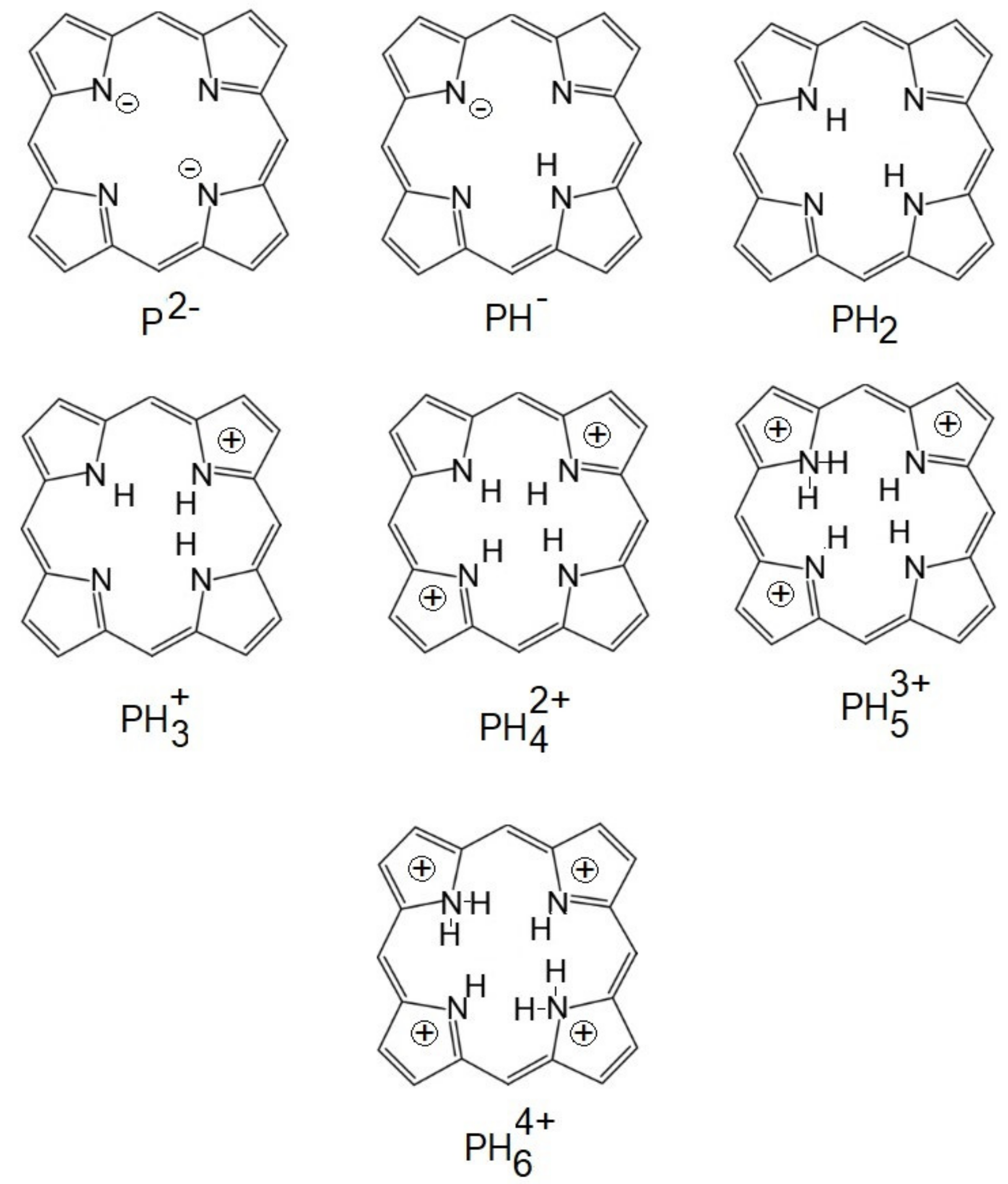

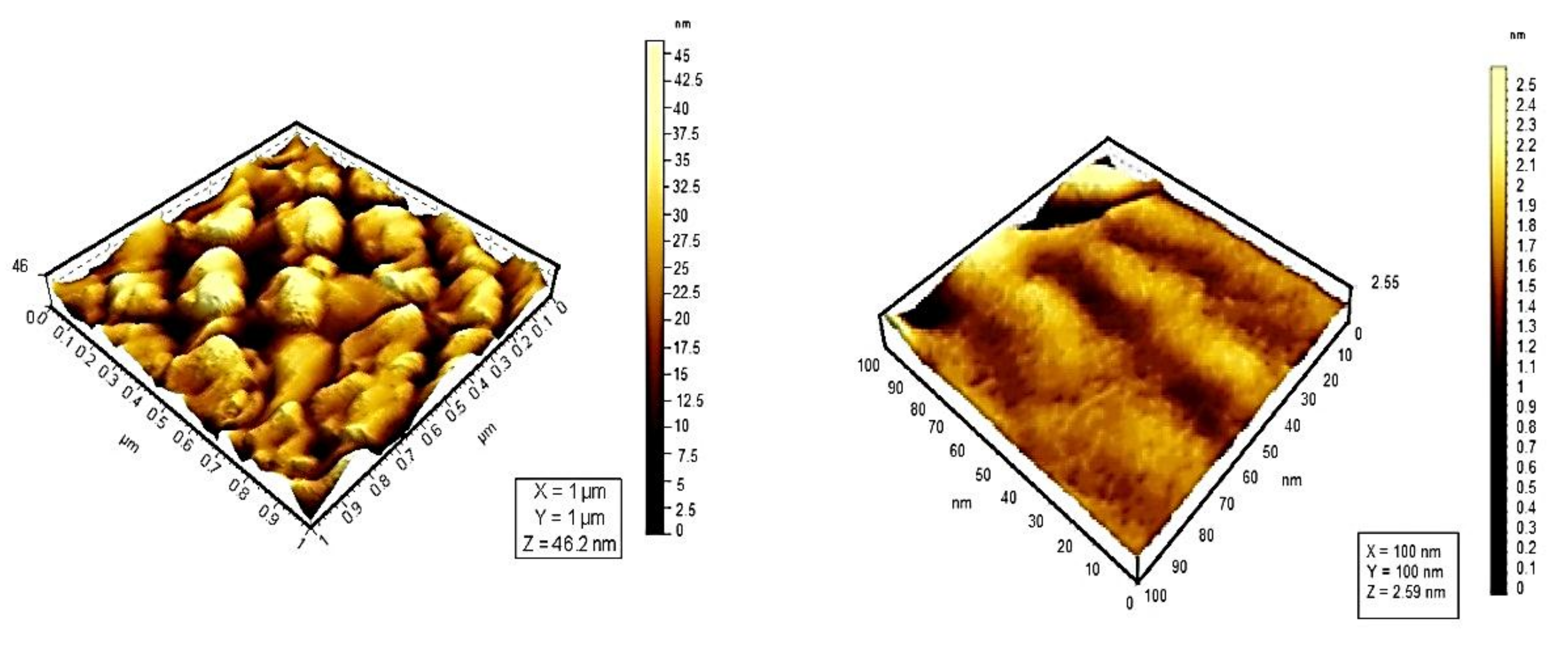

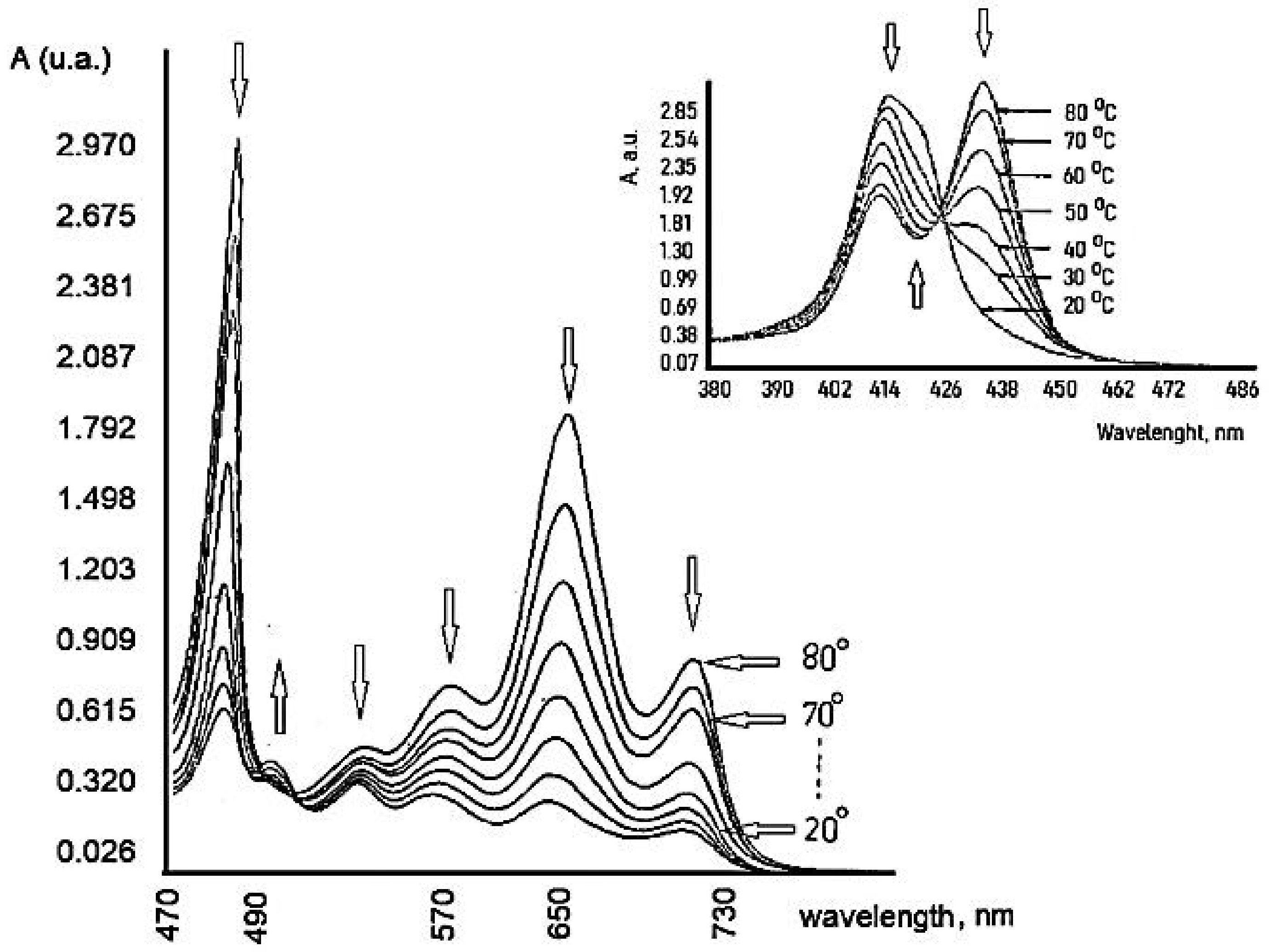
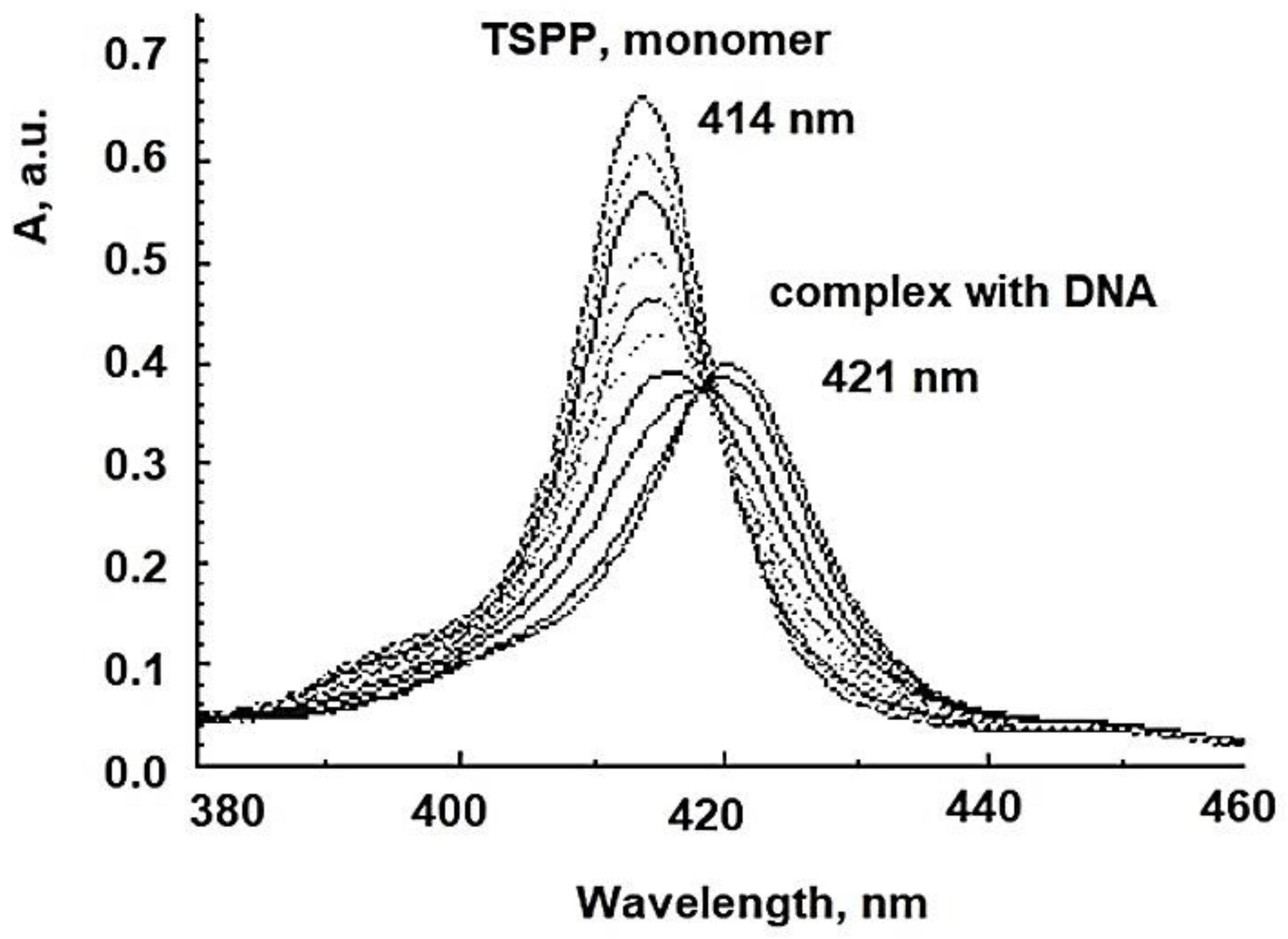

| Photosensitizer | λabs (nm) |
|---|---|
| Psoralen | 300–380 |
| Acridine | 400–500 |
| Cyanine | 500–600 |
| Porphyrin | 600–650 |
| Perylenequinonoid | 600–650 |
| Phenothiazinium (methylene blue, toluidine blue O) | 620–660 |
| Phthalocyanine | 660–700 |
| Crystal violet | 550–610 |
| Rose Bengal | 450–650 |
| Neutral Red | 460–550 |
| Congo Red | 400–560 |
| Riboflavin | 300–600 |
| Eosin B | 514–544 |
| TSPP Form | Absorption Bands (nm) |
|---|---|
| neutral | 412; 515; 551; 579; 633 |
| dication | 433; 550; 594; 644 |
| J-aggregate | 422; 490; 707 |
| H-aggregate | 401; 517; 552; 593; 650 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ion, R.-M. Revisiting Tetra-p-Sulphonated Porphyrin as Antimicrobial Photodynamic Therapy Agent. Coatings 2021, 11, 393. https://doi.org/10.3390/coatings11040393
Ion R-M. Revisiting Tetra-p-Sulphonated Porphyrin as Antimicrobial Photodynamic Therapy Agent. Coatings. 2021; 11(4):393. https://doi.org/10.3390/coatings11040393
Chicago/Turabian StyleIon, Rodica-Mariana. 2021. "Revisiting Tetra-p-Sulphonated Porphyrin as Antimicrobial Photodynamic Therapy Agent" Coatings 11, no. 4: 393. https://doi.org/10.3390/coatings11040393
APA StyleIon, R.-M. (2021). Revisiting Tetra-p-Sulphonated Porphyrin as Antimicrobial Photodynamic Therapy Agent. Coatings, 11(4), 393. https://doi.org/10.3390/coatings11040393






