Sulfur and Oxygen Effects on High-Si Ductile Iron Casting Skin Formation
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Chemical Composition
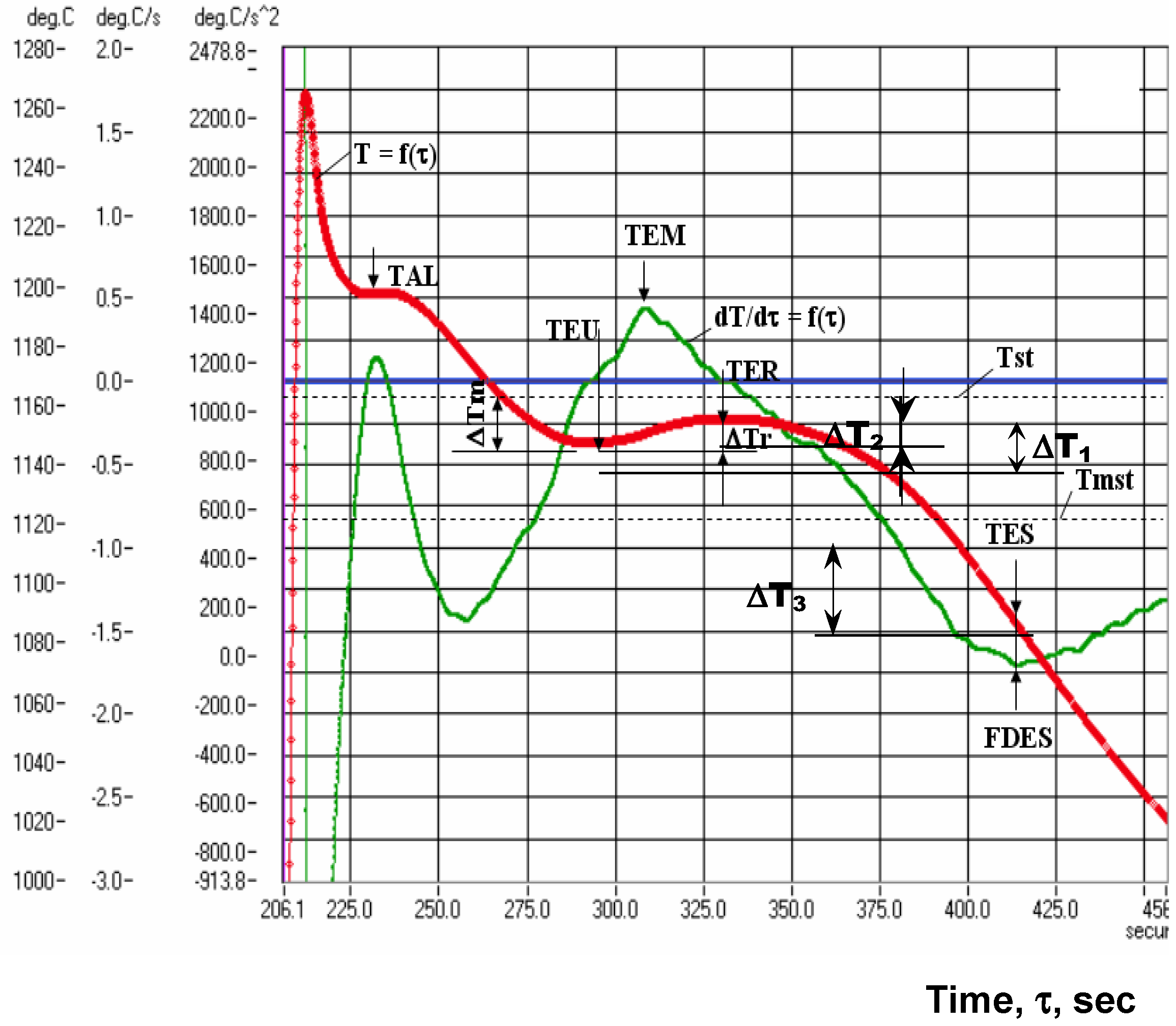
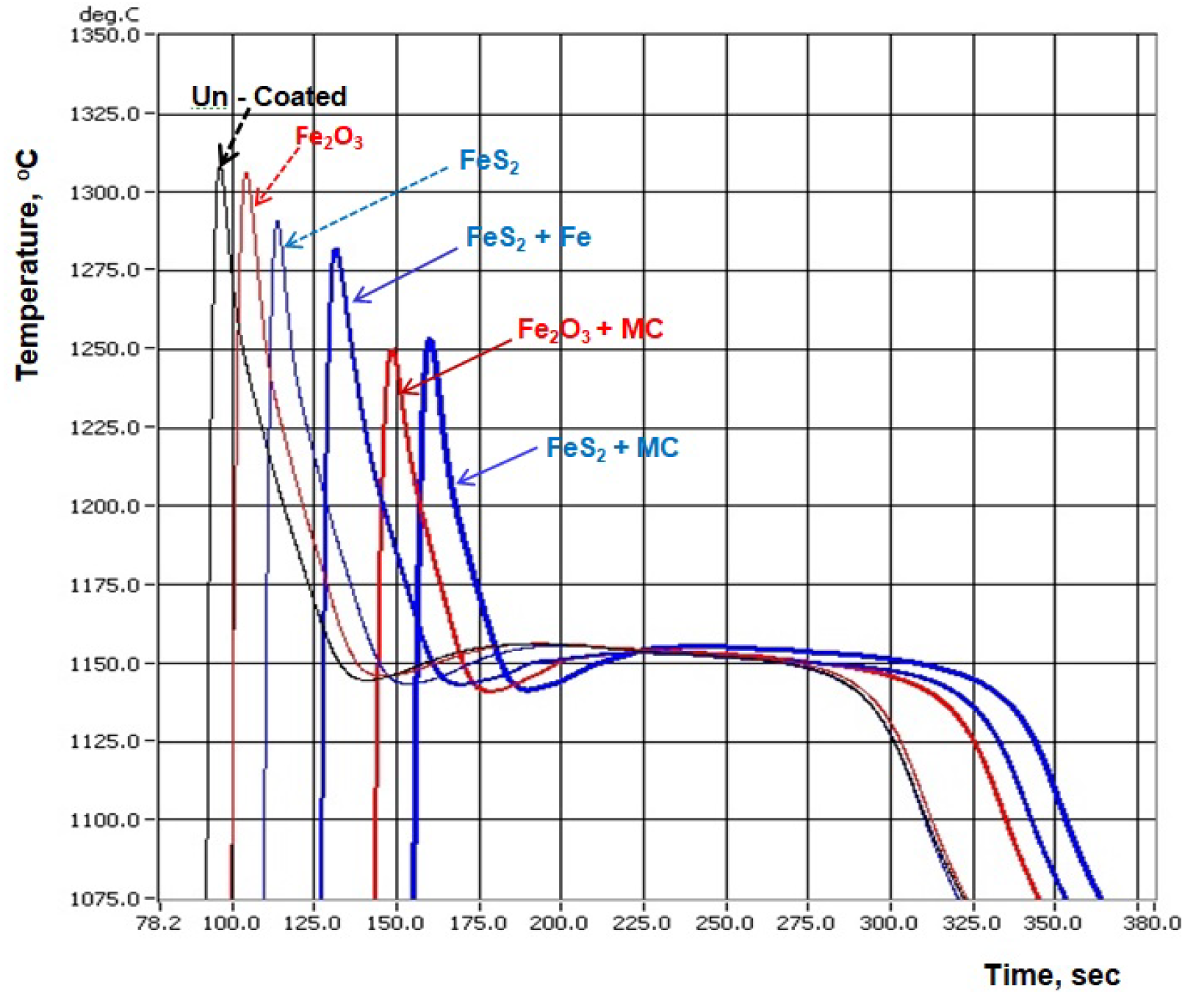
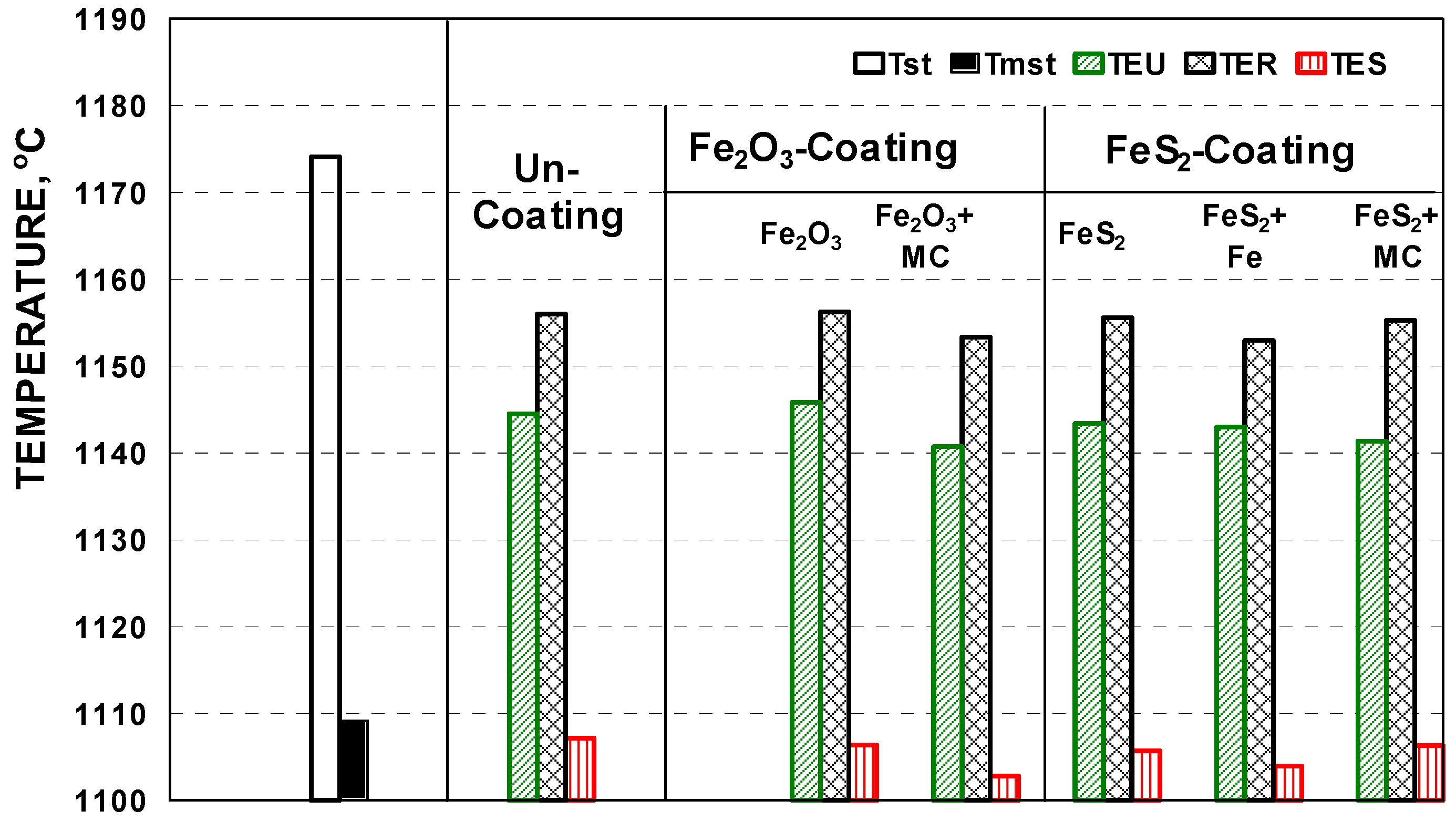
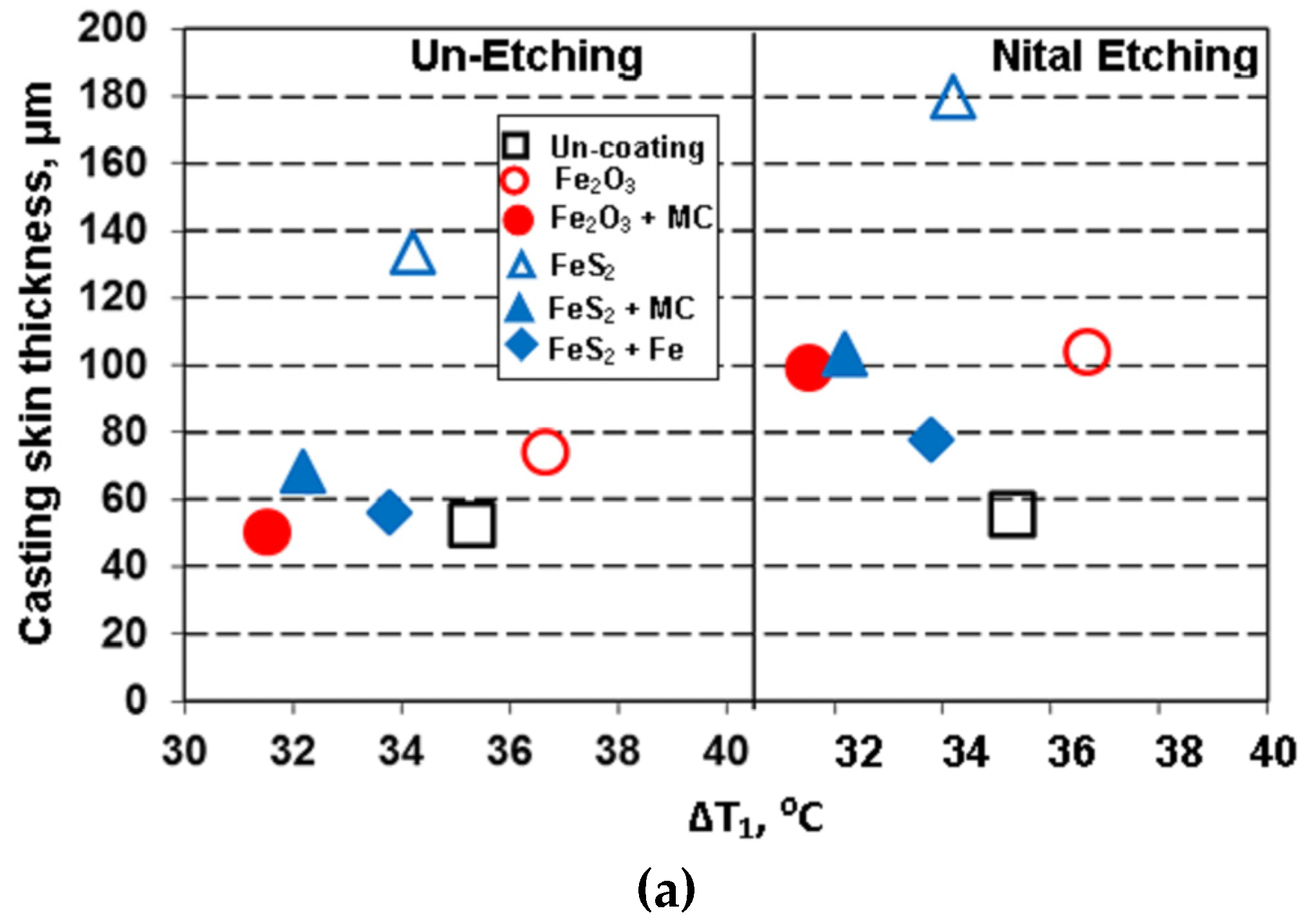
3.2. Thermal (Cooling Curves) Analysis
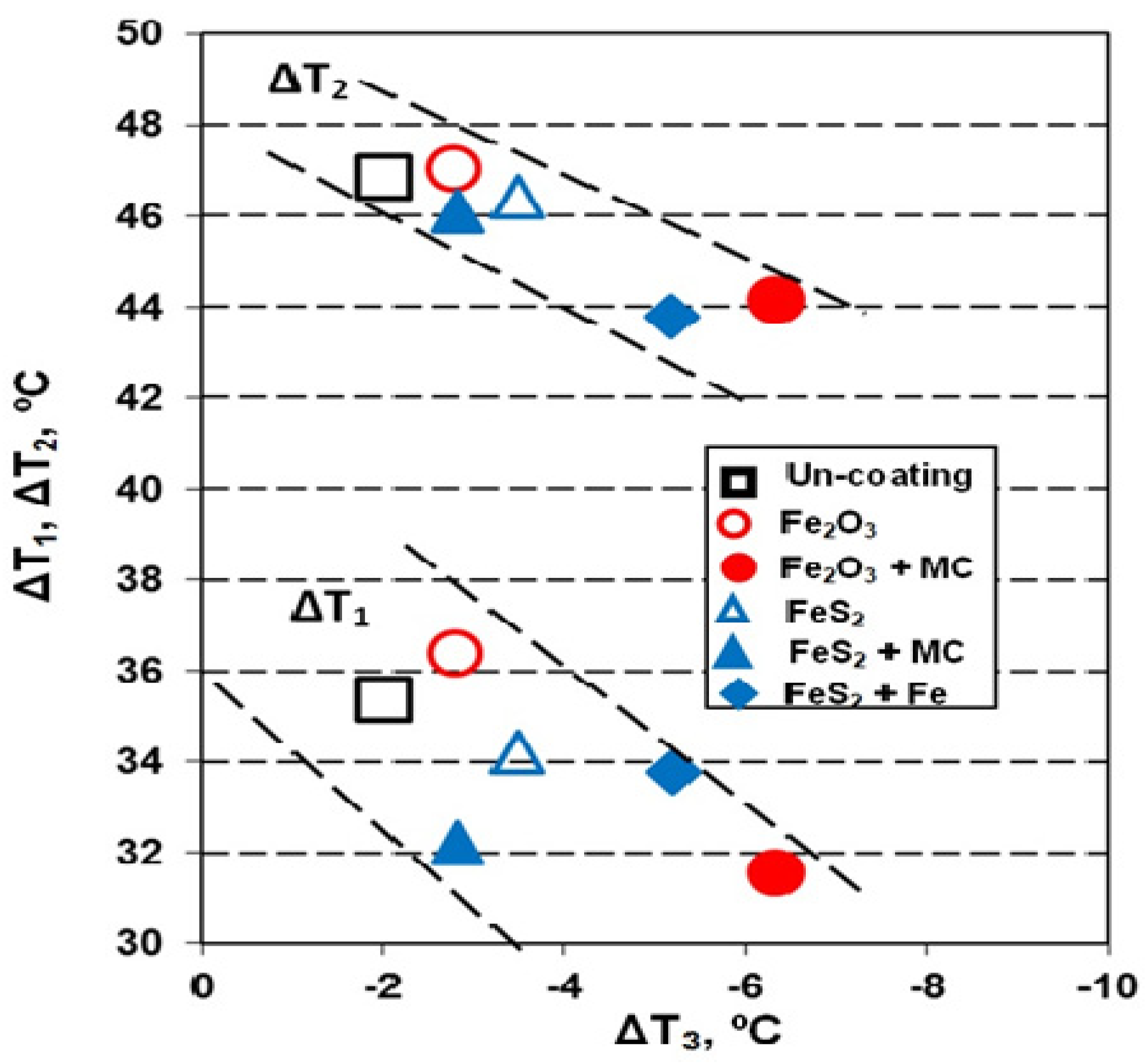
- ΔT1 < 0 and ΔT2 < 0: Only carbides, not graphite formation (white cast iron);
- ΔT1 < 0 and ΔT2 > 0: Carbides at the beginning and graphite at the end of eutectic reaction (mottled cast iron);
- ΔT1 > 0 and ΔT2 > 0: Only graphite formation (graphitic cast iron);
- ΔT3 < 0 usually, as TES < Tmst for most of the commercial cast irons, solidified in industrial conditions (the lower the TES (more negative), the higher the incidence of inter-cells events formation).
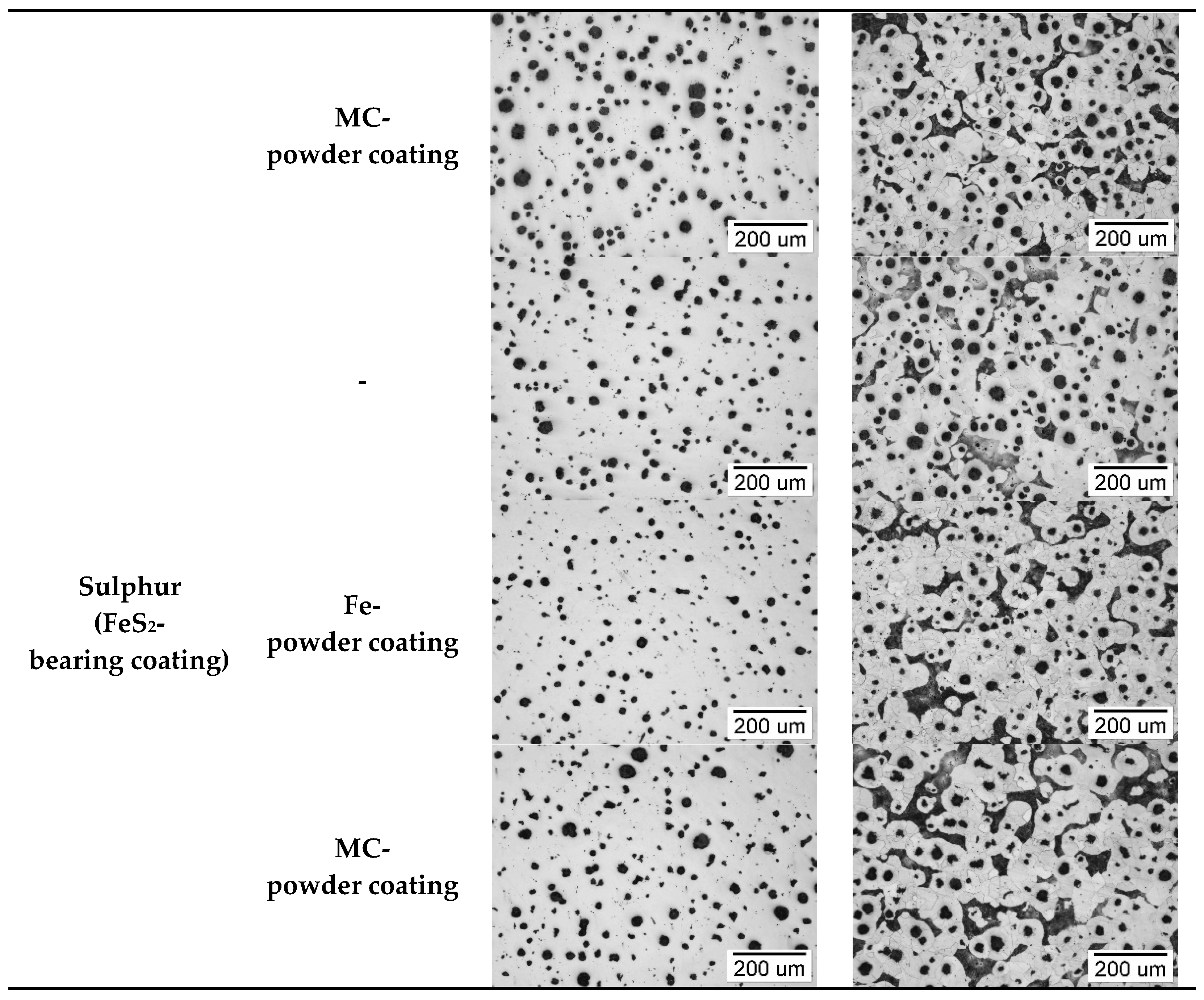
3.3. Structure Analysis
3.3.1. Casting Body
- ANG is the area of particles classified as nodules (RSF = 0.625 − 1.0);
- AIG—area of particles classified as intermediates (RSF = 0.525 − 0.625);
- Atot—area of all graphite particles;
- RSF = 4A/(π lm2);
- A—area of the graphite particle in question;
- lm—maximum axis length of the graphite particle in question (maximum distance between two points on the graphite particle perimeter).
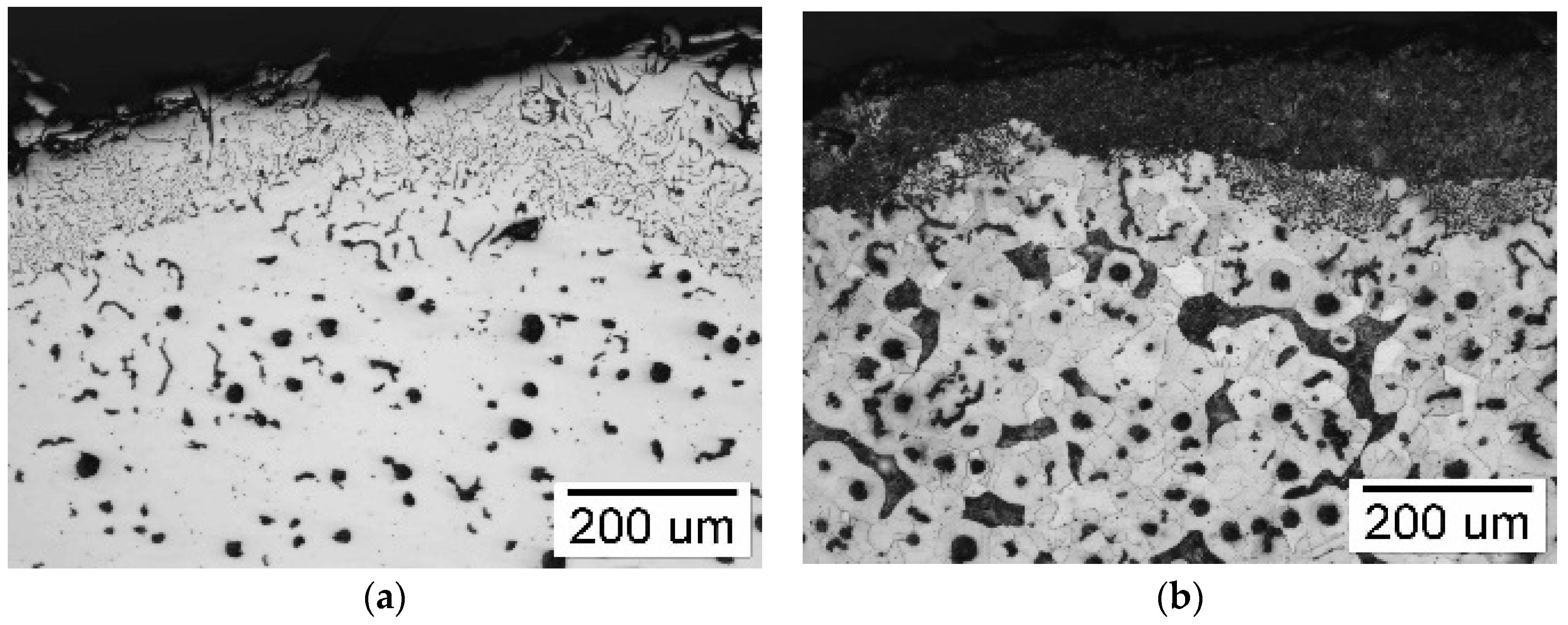
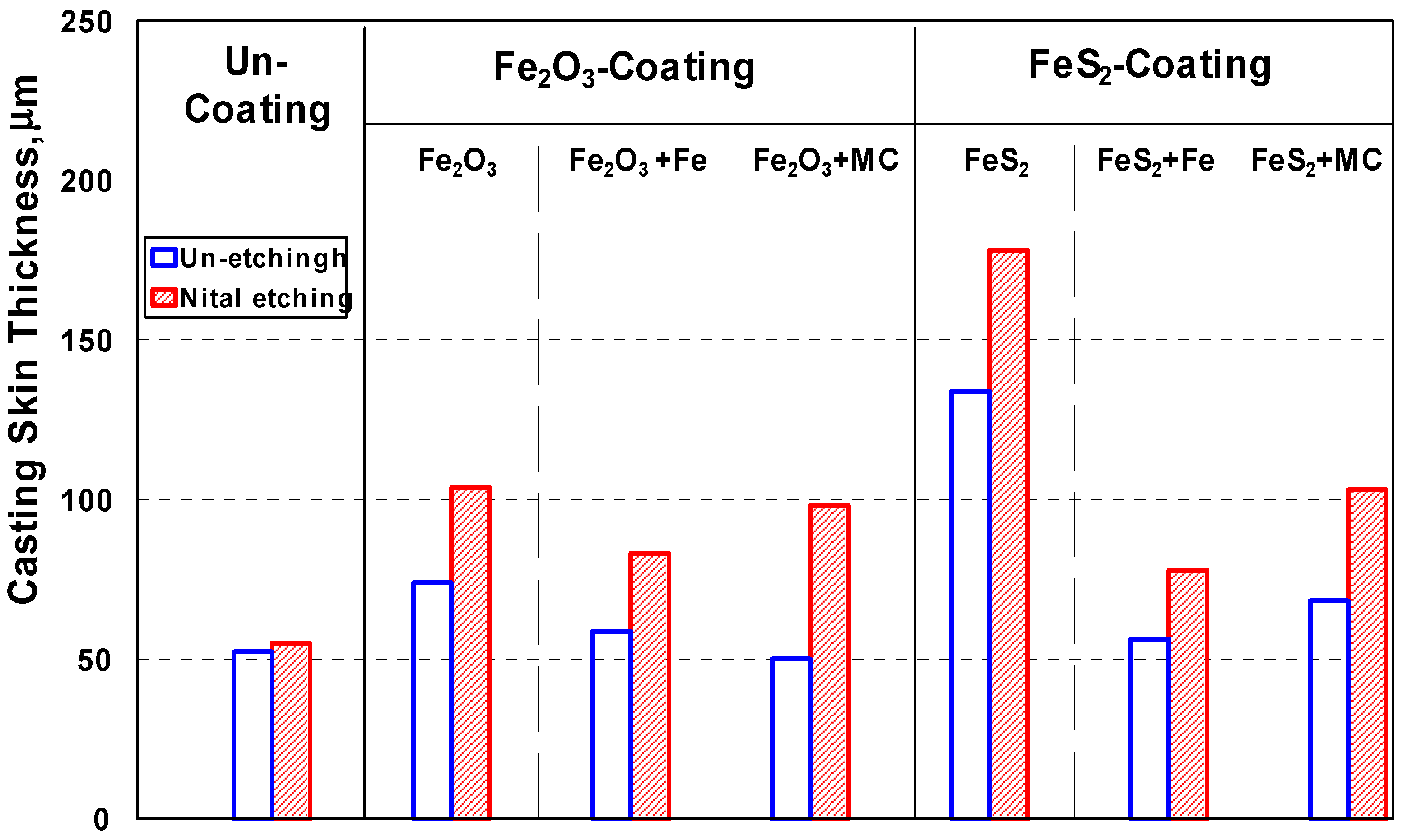
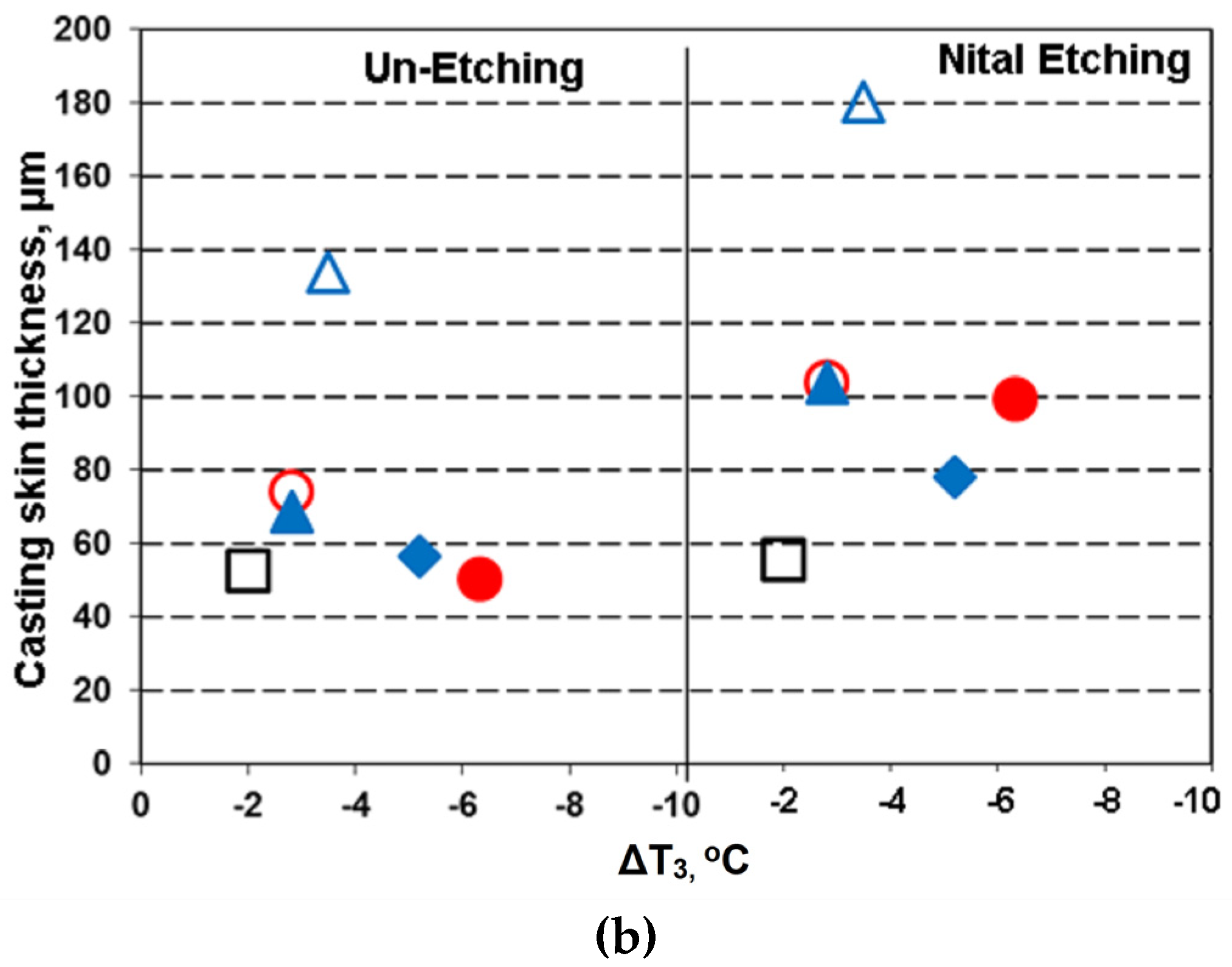
3.3.2. Surface Layer of Castings
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Boonmee, S.; Stefanescu, D.M. Occurrence and effect of casting skin in compacted graphite iron. Int. J. Cast. Met. Res. 2016, 29, 47–54. [Google Scholar] [CrossRef]
- Baier, J.; Koppen, M. Incidence and Avoidance of Defects Attributable to Molding Sands. In Manual of Casting Defects; IKO: Erbsloh, Marl, Germany, 1994; pp. 32–35. [Google Scholar]
- Marti, F.; Karsay, S.I. Localized flake graphite structure as a result of a reaction between molten ductile iron and some components of the mould. AFS Trans. 1979, 87, 221–226. [Google Scholar]
- Xiaogan, H. Nodular Iron Surface Deterioration Due to PTSA in Resin. AFS Trans. 1992, 100, 9–15. [Google Scholar]
- Bauer, W. Research on the Surface Graphite Generation in the Ductile Iron Casting in PTSA-Furan Resin Moulds. Giesserei Praxis 1982, 11, 175–183. [Google Scholar]
- Boonmee, S.; Stefanescu, D.M. Casting skin management in compacted graphite iron. Part II: Mechanism of casting skin formation. AFS Trans. 2013, 121, 449–459. [Google Scholar]
- Quing, J.; Lekakh, S.; Richards, V. No-bake S-containing mold-DI metal interactions: Consequences and potential applications. AFS Trans. 2013, 121, 13–20. [Google Scholar]
- Ivan, N.; Chisamera, M.; Riposan, I. Mg-bearing coating of resin sand—PTSA moulds to control graphite degeneration in the surface layer of ductile iron castings. Mater. Sci. Technol. 2012, 28, 1246–1253. [Google Scholar] [CrossRef]
- Ivan, N.; Chisamera, M.; Riposan, I. Influence of magnesium content and coating type on graphite degeneration in surface layer of iron castings in resin sand—PTSA moulds. ISIJ Int. 2012, 52, 1848–1855. [Google Scholar] [CrossRef][Green Version]
- Ivan, N.; Chisamera, M.; Riposan, I. Mold coatings to reduce graphite degeneration in the surface layer of ductile Iron castings. Int. J. Metalcasting 2012, 6, 61–69. [Google Scholar] [CrossRef]
- Ivan, N.; Chisamera, M.; Riposan, I. Graphite degeneration in the surface layer of ductile iron castings. Int. J. Cast Met. Res. 2013, 26, 138–142. [Google Scholar] [CrossRef]
- Ivan, N.; Chisamera, M.; Riposan, I.; Stan, S. Control of graphite degeneration in the surface layer of Mg-treated iron castings in resin sand—P-Toluol Sulphonic Acid (PTSA) molds. AFS Trans. 2013, 121, 379–390. [Google Scholar]
- Chisamera, M.; Ivan, N.; Riposan, I.; Stan, S. Iron casting skin management in no-bake mould—Effects of magnesium residual level and mould coating. China Foundry 2015, 12, 222–230. [Google Scholar]
- QuiK-Cup® QuiK-Lab® E Thermal Analysis of Cast Iron. Available online: https://www.heraeus.com/media/media/hen/doc_hen/measurement_instruments/quik-cup.pdf (accessed on 20 June 2020).
- Anca, D. Research on the Loss Modifying Effect in the Superficial Layer Phenomenon of the Iron Castings. Ph.D. Thesis, POLITEHNICA University of Bucharest, Bucharest, Romania, 2014. [Google Scholar]
- Thielemann, T. Zur Wirkung van Spurenelementen in Gusseisen mit Kugelgraphit. Giessereitechnik 1970, 16, 16–24. [Google Scholar]
- Riposan, I.; Chisamera, M.; Stan, S.; White, D. Role of Residual Aluminium in Ductile Iron Solidification. AFS Trans. 2007, 115, 423–433. [Google Scholar]
- Sillen, R.V. Optimizing inoculation practice by means of thermal analysis. In Proceedings of the AFS International Inoculation Conference, Rosemont, IL, USA, 6–8 April 1998. [Google Scholar]
- Sillen, R.V. Novacast Technologies. 2006. Available online: www.novacast.se (accessed on 20 June 2020).
- Leca, L.; Prisecaru, I. Thermophysical and Thermodynamical Properties; Editura Tehnica: Bucharest, Romania, 1994. [Google Scholar]
- Riposan, I.; Skaland, T. Modification and inoculation of castiron. In Cast Iron Science and Technology Handbook; Stefanescu, D.M., Ed.; American Society of Materials: Cleveland, OH, USA, 2017; pp. 160–176. [Google Scholar]
- Anca, D.; Chisamera, M.; Stan, S.; Riposan, I. Graphite degeneration in High Si, Mg-treated iron castings–Sulphur and oxygen addition effects. Int. J. Metalcasting 2019. [Google Scholar] [CrossRef]
- Anca, D.; Chisamera, M.; Stan, S.; Riposan, I. Graphite degeneration in the superficial layer of high Si-ductile iron casting as influence of inoculation and protective coating against sulphur diffusion into the iron melt. J. Mater. Res. Technol. 2019, 8, 5160–5170. [Google Scholar] [CrossRef]



| Treatment Alloy | Si | Ca | Al | TRE* | Mg | Ba | Fe | Addition Amount (wt.%) | Addition Technique | |
|---|---|---|---|---|---|---|---|---|---|---|
| Role | Type | |||||||||
| Si alloying | FeSi90 | Min. 90 | Max. 1.0 | Max. 0.5 | - | - | - | Max. 10 | 2.0 | Furnace |
| Nodulizer | FeSiCaMgRE | 46 | 1.87 | 0.85 | 1.1 | 8.2 | - | bal | 2.0 | Tundish - Cover |
| Inoculant | FeSiCaBaAl | 75 | 1.0 | 1.1 | - | - | 1.0 | bal | 0.8 | Pouring Ladle |
| Chemical Composition, wt.% | Carbon Equivalent * % | Antinodulizing Factor ** | Pearlitic Factor *** | |||||
|---|---|---|---|---|---|---|---|---|
| C | Si | Mn | P | S | Mg | CE | K | Px |
| 3.65 | 3.15 | 0.1 | 0.013 | 0.004 | 0.049 | 4.6 | 0.72 | −0.22 |
| Material | Thermal Diffusivity (W s1/2 /m2 K) | Specific Heat (J/kg K) | Thermal Conductivity (W/m K) | Density (kg/m3) |
|---|---|---|---|---|
| Ceramic cup | 1487 | 1280 | 1.08 | 1600 |
| S-bearing coating | 3585 | 547 | 5 | 4700 |
| O-bearing coating | 2044 | 650 | 1.26 | 5100 |
| Fe-powder-bearing coating | 16,221 | 450 | 74.4 | 7870 |
| Carbonic-material-bearing coating | 14,410 | 710 | 129 | 2267 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anca, D.; Chisamera, M.; Stan, S.; Stan, I.; Riposan, I. Sulfur and Oxygen Effects on High-Si Ductile Iron Casting Skin Formation. Coatings 2020, 10, 618. https://doi.org/10.3390/coatings10070618
Anca D, Chisamera M, Stan S, Stan I, Riposan I. Sulfur and Oxygen Effects on High-Si Ductile Iron Casting Skin Formation. Coatings. 2020; 10(7):618. https://doi.org/10.3390/coatings10070618
Chicago/Turabian StyleAnca, Denisa, Mihai Chisamera, Stelian Stan, Iuliana Stan, and Iulian Riposan. 2020. "Sulfur and Oxygen Effects on High-Si Ductile Iron Casting Skin Formation" Coatings 10, no. 7: 618. https://doi.org/10.3390/coatings10070618
APA StyleAnca, D., Chisamera, M., Stan, S., Stan, I., & Riposan, I. (2020). Sulfur and Oxygen Effects on High-Si Ductile Iron Casting Skin Formation. Coatings, 10(7), 618. https://doi.org/10.3390/coatings10070618




