Ultra-Short Pulsed Laser Deposition of Oxides, Borides and Carbides of Transition Elements
Abstract
1. Introduction
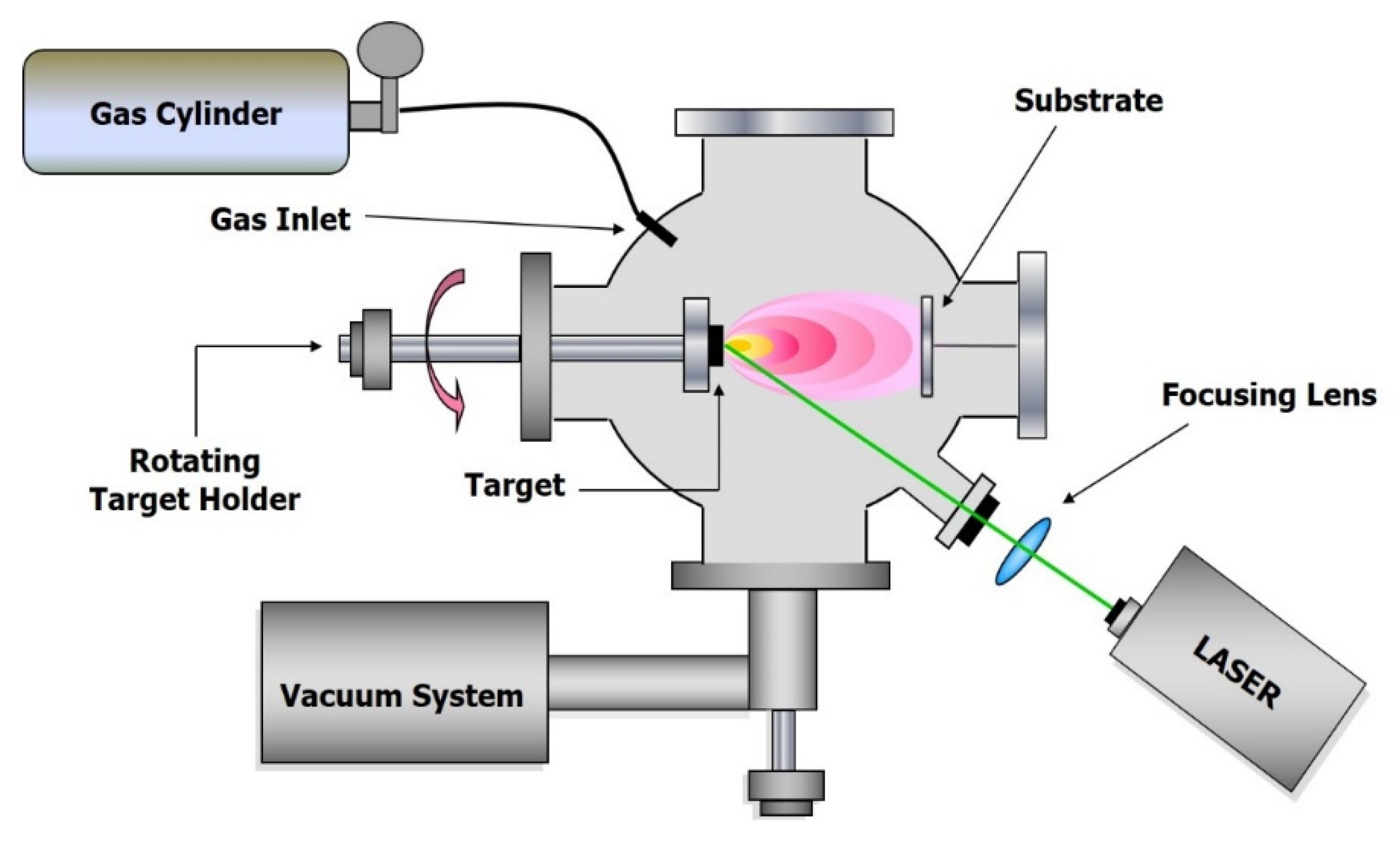
2. Ultra-Short Pulsed Laser Ablation
3. Oxides
3.1. Zinc Oxide
3.2. Titanium Dioxide
3.3. Other oxides
3.3.1. Vanadium Oxide
3.3.2. Indium Tin Oxide
4. Borides
4.1. Rhenium Boride
4.2. Zirconium Boride
4.3. Ruthenium Boride
4.4. Rhodium and Iridium Borides
4.5. Tungsten Borides
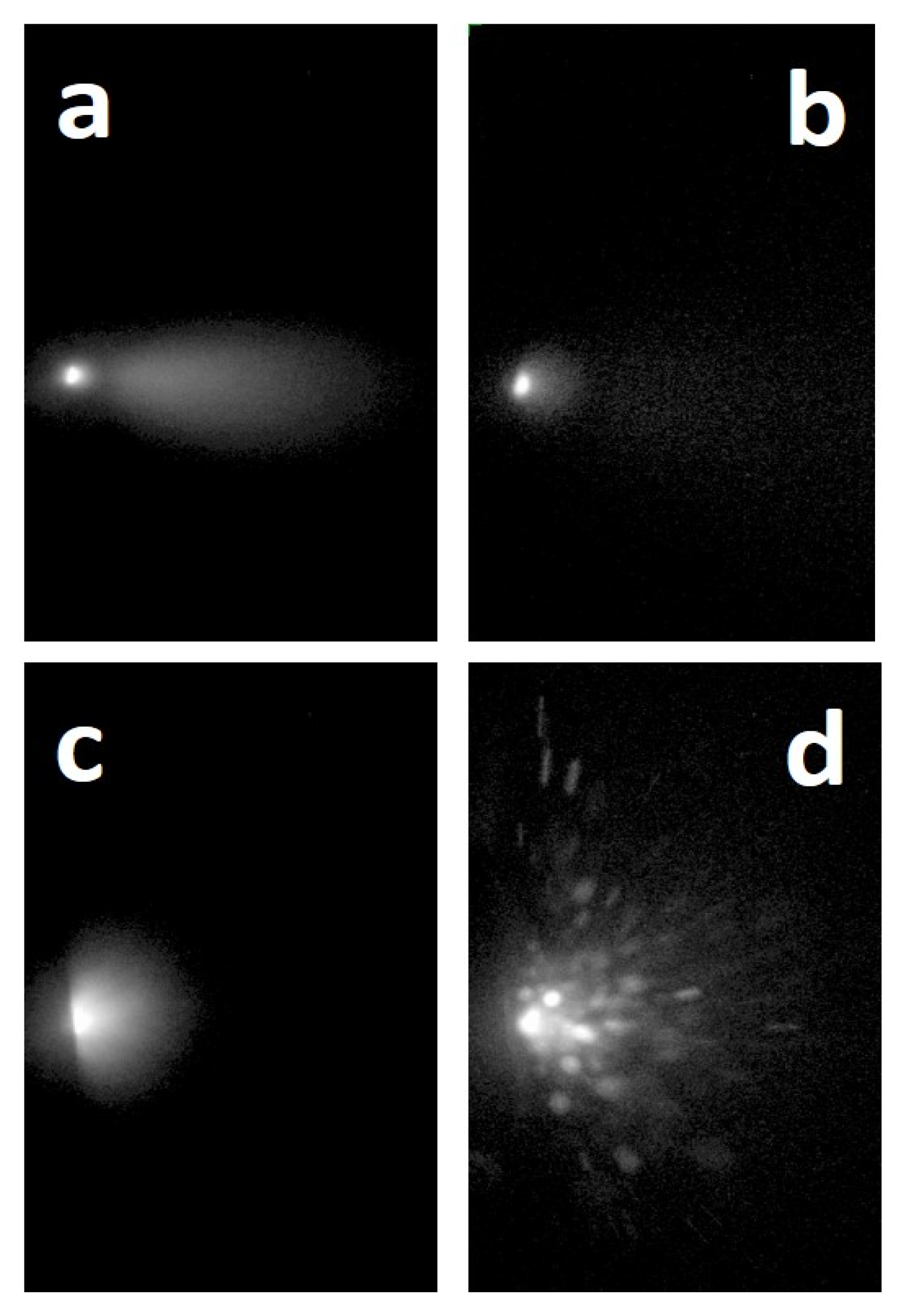
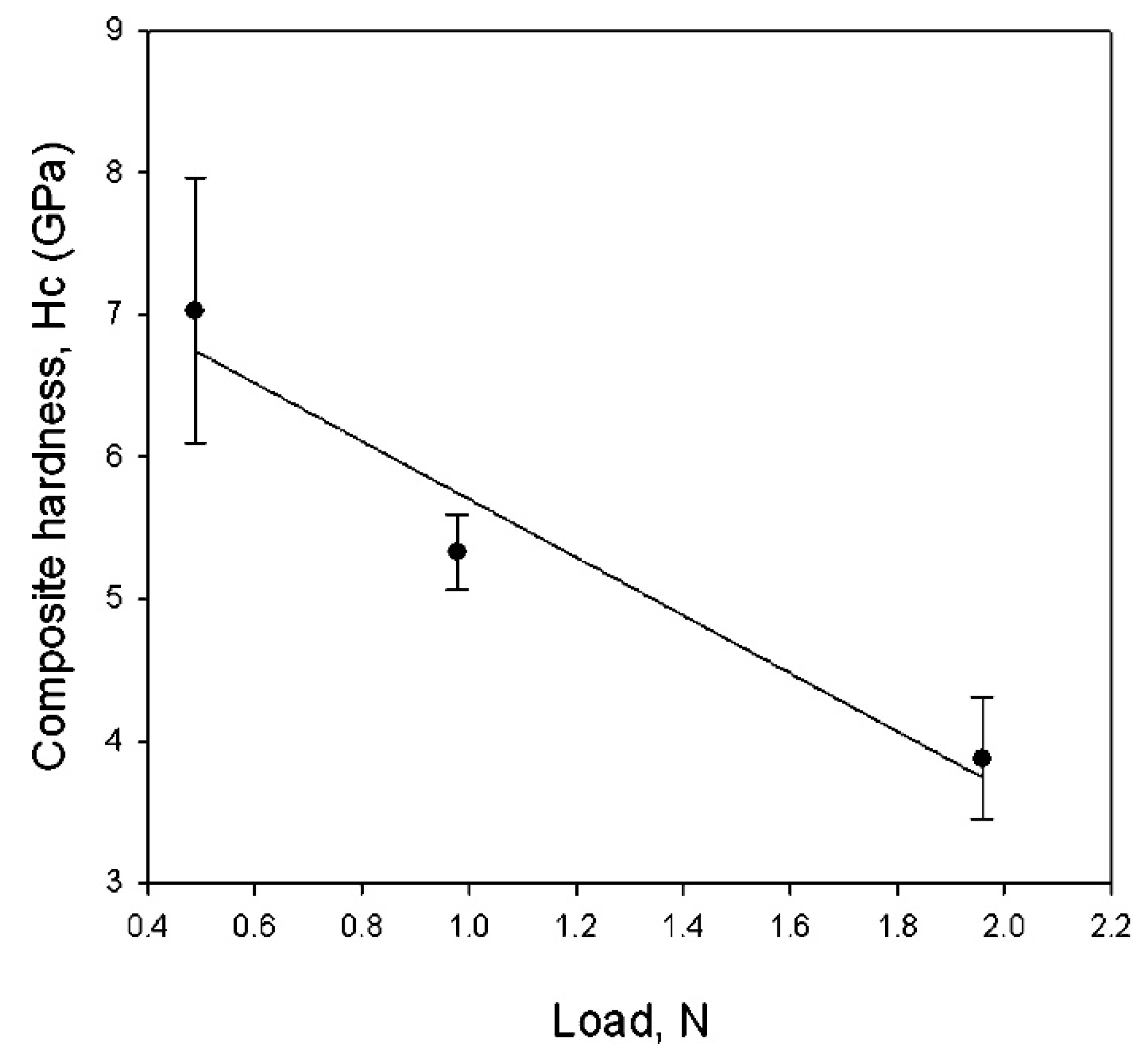
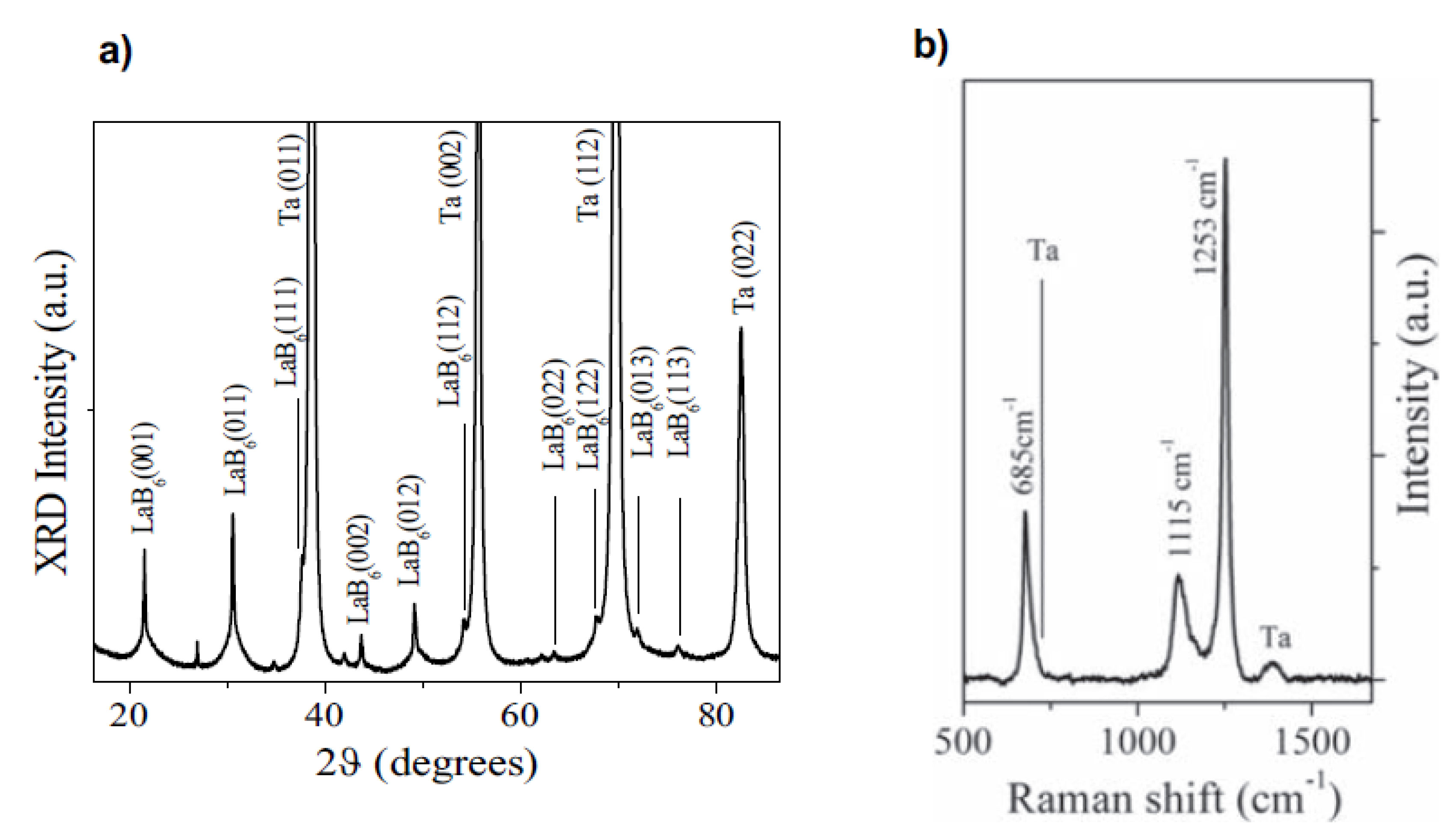
4.6. Lanthanum Boride
4.7. Chromium Boride
5. Carbides
5.1. Titanium Carbide
5.2. Zirconium and Hafnium Carbides
5.3. Vanadium and Niobium Carbides
5.4. Tantalum Carbide
5.5. Chromium Carbide
5.6. Molybdenum Carbide
5.7. Tungsten Carbide
6. Conclusions and Outlook
Funding
Conflicts of Interest
References
- Margrave, J.L.; Toth, L.E. (Eds.) Transition Metal Carbides and Nitrides; Academic Press: New York, NY, USA, 1971. [Google Scholar]
- McLeod, A.D.; Haggerty, J.S.; Sadoway, D.R. Electrical resistivities of monocrystalline and polycrystalline TiB2. J. Am. Ceram. Soc. 1984, 67, 705–708. [Google Scholar] [CrossRef]
- Wang, X.; Martin, P.J.; Kinder, T.J. Characteristics of TiB2 films prepared by ion beam sputtering. Surf. Coat. Technol. 1996, 78, 37–41. [Google Scholar] [CrossRef]
- Thomas, S.C.; Ren, X.M.; Gottesfeld, S.; Zelenay, P. Direct methanol fuel cells: Progress in cell performance and cathode research. Electrochim. Acta 2002, 47, 3741–3748. [Google Scholar] [CrossRef]
- Piegari, A.; Flory, F. (Eds.) Optical Thin Films and Coatings—From Materials to Applications, 2nd ed.; Woodhead Publishing: Cambridge, UK, 2018. [Google Scholar]
- Hogmark, S.; Jacobson, S.; Larsson, M. Design and evaluation of tribological coatings. Wear 2000, 246, 20–33. [Google Scholar] [CrossRef]
- Seshan, K.; Schepis, D. (Eds.) Handbook of Thin Film Deposition, 4th ed.; Elsevier: Oxford, UK, 2018. [Google Scholar]
- Martin, P.M. (Ed.) Handbook of Deposition Technologies for Films and Coatings, 3rd ed.; Elsevier: Oxford, UK, 2010. [Google Scholar]
- Eason, R. (Ed.) Pulsed Laser Deposition of Thin Films, Applications-Led Growth of Functional Materials; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Chrisey, D.B.; Hubler, G.K. (Eds.) Pulsed Laser Deposition of Thin Films; Wiley: New York, NY, USA, 1994. [Google Scholar]
- Karnati, P.; Haque, A.; Taufique, M.F.N.; Ghosh, K. A Systematic Study on the Structural and Optical Properties of Vertically Aligned Zinc Oxide Nanorods Grown by High Pressure Assisted Pulsed Laser Deposition Technique. Nanomaterials 2018, 8, 62. [Google Scholar] [CrossRef]
- Osiac, M.; Cioatera, N.; Jigau, M. Structural, Morphological, and Optical Properties of Iron Doped WO3 Thin Film Prepared by Pulsed Laser Deposition. Coatings 2020, 10, 412. [Google Scholar] [CrossRef]
- Zhang, Y.; Russo, R.E.; Mao, S.S. Femtosecond laser assisted growth of ZnO nanowires. Appl. Phys. Lett. 2005, 87, 133115. [Google Scholar] [CrossRef]
- Grigoriev, S.N.; Fominskii, V.Y.; Romanov, R.I.; Gnedovets, A.G. Tribological properties of gradient Mo-Se-Ni-C thin films obtained by pulsed laser deposition in standard and shadow mask configurations. Thin Solid Films 2014, 556, 35–43. [Google Scholar] [CrossRef]
- Fominskii, V.Y.; Grigoriev, S.N.; Gnedovets, A.G.; Romanov, R.I. Specific Features of Ion Initiated Processes during Pulsed Laser Deposition of MoSe2 Coatings in Pulsed Electric Fields. Tech. Phys. Lett. 2012, 38, 683–686. [Google Scholar] [CrossRef]
- Balling, P.; Schou, J. Femtosecond-laser ablation dynamics of dielectrics: Basics and applications for thin films. Rep. Prog. Phys. 2013, 76, 036502. [Google Scholar] [CrossRef]
- Miller, J.C.; Haglund, R.F. (Eds.) Laser Ablation and Desorption; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- Geohegan, D.B. Diagnostics and Characteristics of Laser-Produced Plasmas. In Pulsed Laser Deposition of Thin Films, 1st ed.; Chrisey, D.B., Hubler, G.K., Eds.; Wiley: New York, NY, USA, 1994; pp. 115–165. [Google Scholar]
- Bäuerle, D. Laser Processing and Chemistry; Springer: Berlin, Germany, 2011. [Google Scholar]
- Chen, L.C. Particulates Generated by Pulsed Laser Ablation. In Pulsed Laser Deposition of Thin Films, 1st ed.; Chrisey, D.B., Hubler, G.K., Eds.; Wiley: New York, USA, 1994; pp. 167–198. [Google Scholar]
- Ossi, P.M. Cluster Synthesis and Cluster-Assembled Film Deposition in Nanosecond Pulsed Laser Ablation. In Laser-Surface Interactions for New Materials Production; Miotello, A., Ossi, P.M., Eds.; Springer: Berlin, Germany, 2010; pp. 99–124. [Google Scholar]
- Stafe, M.; Marcu, A.; Puscas, N.N. Pulsed Laser Ablation of Solids; Springer: Heidelberg, Germany, 2014. [Google Scholar]
- Keller, U. Ultrafast solid-state lasers. In Landolt-Börnstein. Laser Physics and Applications. Subvolume B: Laser Systems. Part I; Herziger, G., Weber, H., Proprawe, R., Eds.; Springer: Heidelberg, Germany, 2007; pp. 33–170. [Google Scholar]
- Keller, U. Ultrafast solid-state laser oscillators: A success story for the last 20 years with no end in sight. Appl. Phys. B 2010, 100, 15–28. [Google Scholar] [CrossRef]
- Mayer, A.S.; Phillips, C.R.; Keller, U. Watt-level 10-gigahertz solid-state laser enabled by self-defocusing nonlinearities in an aperiodically poled crystal. Nat. Commun. 2017, 8, 1673. [Google Scholar] [CrossRef] [PubMed]
- Saltarelli, F.; Diebold, A.; Graumann, I.J.; Phillips, C.R.; Keller, U. Self-phase modulation cancellation in a high-power ultrafast thin-disk laser oscillator. Optica 2018, 5, 1603–1606. [Google Scholar] [CrossRef]
- Albert, O.; Roger, S.; Glinec, Y.; Loulergue, J.C.; Etchepare, J.; Boulmer-Leborgne, C.; Perriere, J.; Millon, A. Time-resolved spectroscopy measurements of a titanium plasma induced by nanosecond and femtosecond lasers. Appl. Phys. A 2003, 76, 319–323. [Google Scholar] [CrossRef]
- Amoruso, S.; Ausanio, G.; Bruzzese, R.; Vitiello, M.; Wang, X. Femtosecond laser pulse irradiation of solid targets as a general route to nanoparticle formation in a vacuum. Phys. Rev. B 2005, 71, 033406. [Google Scholar] [CrossRef]
- Teghil, R.; D’Alessio, L.; De Bonis, A.; Galasso, A.; Villani, P.; Santagata, A. Femtosecond pulsed laser ablation and deposition of titanium carbide. Thin Solid Films 2006, 515, 1411–1418. [Google Scholar] [CrossRef]
- Boulmer-Leborgne, C.; Benzerga, R.; Perrière, J. Nanoparticle Formation by Femtosecond Laser Ablation. In Laser-Surface Interactions for New Materials Production; Miotello, A., Ossi, P.M., Eds.; Springer: Heidelberg, Germany, 2010; pp. 125–140. [Google Scholar]
- De Bonis, A.; Santagata, A.; Rau, J.V.; Latini, A.; Mori, T.; Medici, L.; Teghil, R. Two-phase zirconium boride thin film obtained by ultra-short pulsed laser ablation of a ZrB12 target. App. Surf. Sci. 2013, 283, 715–721. [Google Scholar] [CrossRef]
- De Bonis, A.; Galasso, A.; Latini, A.; Rau, J.V.; Santagata, A.; Curcio, M.; Teghil, R. Femtosecond Pulsed Laser Deposition of Chromium Diboride-Rich Thin Films. Coatings 2019, 9, 777. [Google Scholar] [CrossRef]
- Perez, D.; Lewis, L.J. Molecular-dynamics study of ablation of solids under femtosecond laser pulses. Phys. Rev. B 2003, 67, 184102. [Google Scholar] [CrossRef]
- Lewis, J.L.; Perez, D. Laser ablation with short and ultrashort laser pulses: Basic mechanisms from molecular-dynamics simulations. Appl. Surf. Sci. 2009, 255, 5101–5106. [Google Scholar] [CrossRef]
- Besner, S.; Meunier, M. Laser Synthesis of Nanomaterials. In Laser Precision Fabrication; Sugioka, K., Meunier, M., Piqué, A., Eds.; Springer: Berlin, Germany, 2010; pp. 163–187. [Google Scholar]
- Banks, P.S.; Dinh, L.; Stuart, B.C.; Feit, M.D.; Komashko, A.M.; Rubenchik, A.M.; Perry, M.D.; McLean, W. Short-pulse laser deposition of diamond-like carbon thin films. Appl. Phys A 1999, 69, S347–S353. [Google Scholar] [CrossRef]
- Teghil, R.; D’Alessio, L.; Santagata, A.; Zaccagnino, M.; Ferro, D.; Sordelet, D.J. Picosecond and femtosecond pulsed laser ablation and deposition of quasicrystals. Appl. Surf. Sci. 2003, 210, 307–317. [Google Scholar] [CrossRef]
- Ossi, P.M.; Dinescu, M. Creating Nanostructures with Lasers. In Laser Processing of Materials; Schaaf, P., Ed.; Springer: Berlin, Germany, 2010; pp. 146–151. [Google Scholar]
- O’Connell, G.; Donnelly, T.; Lunney, J.G. Nanoparticle plume dynamics in femtosecond laser ablation of gold. Appl. Phys. A 2014, 117, 289–293. [Google Scholar] [CrossRef]
- Tsakiris, N.; Anoop, K.K.; Ausanio, G.; Gill-Comeau, M.; Bruzzese, R.; Amoruso, S.; Lewis, J. Ultrashort laser ablation of bulk copper targets: Dynamics and size distribution of the generated nanoparticles. J. Appl. Phys. 2014, 115, 243301. [Google Scholar] [CrossRef]
- Povarnitsyn, M.E.; Itina, T.E.; Sentis, M.; Khishchenko, K.V.; Levashov, P.R. Material decomposition mechanisms in femtosecond laser interactions with metals. Phys. Rev. B 2007, 75, 235414. [Google Scholar] [CrossRef]
- Kobayashi, T.; Matsuo, Y. Study on the carbon fragment anions produced by femtosecond laser ablation of solid C60. J. Chem. Phys. 2011, 134, 064320. [Google Scholar] [CrossRef]
- Teghil, R.; D’Alessio, L.; De Bonis, A.; Galasso, A.; Ibris, N.; Salvi, A.M.; Santagata, A.; Villani, P. Nanoparticles and thin film formation in ultrashort pulsed laser deposition of vanadium oxide. J. Phys. Chem. A 2009, 113, 14969–14974. [Google Scholar] [CrossRef]
- Schou, J.; Amoruso, S.; Lunney, J.G. Plume dynamics. In Laser Ablation and its Applications; Phypps, C., Ed.; Springer: New York, NY, USA, 2007; pp. 67–95. [Google Scholar]
- Trelenberg, T.W.; Dinh, L.N.; Saw, C.K.; Stuart, B.C.; Balooch, M. Femtosecond pulsed laser ablation of GaAs. Appl. Surf. Sci. 2004, 221, 364–369. [Google Scholar] [CrossRef]
- Trelenberg, T.W.; Dinh, L.N.; Stuart, B.C.; Balooch, M. Femtosecond pulsed laser ablation of metal alloy and semiconductor targets. Appl. Surf. Sci. 2004, 229, 268–274. [Google Scholar] [CrossRef]
- Teghil, R.; De Bonis, A.; Galasso, A.; Santagata, A.; Villani, P.; Sordelet, D.J. Role and importance of nanoparticles in femtosecond pulsed laser ablation deposition of Al-Cu-Fe quasicrystal. Chem. Phys. Lett. 2007, 438, 85–88. [Google Scholar] [CrossRef]
- Teghil, R.; D’Alessio, L.; De Bonis, A.; Ferro, D.; Galasso, A.; Lanza, G.; Santagata, A.; Villani, P.; Sordelet, D.J. Ultra-short pulse laser ablation of Al70Cu20Fe10 alloy: Nanoparticles generation and thin films deposition. Thin Solid Film 2009, 517, 1880–1886. [Google Scholar] [CrossRef]
- Henley, S.J.; Carey, J.D.; Silva, S.R.P.; Fuge, G.M.; Ashfold, M.N.R.; Anglos, D. Dynamics of confined plumes during the short and ultra-short pulsed laser ablation of graphite. Phys. Rev. B 2005, 72, 205413. [Google Scholar] [CrossRef]
- Al-Shboul, K.F.; Harilal, S.S.; Hassanein, A. Spatio-temporal mapping of ablated species in ultrafast laser-produced graphite plasmas. Appl. Phys. Lett. 2012, 100, 221106. [Google Scholar] [CrossRef]
- Kumar, R.; Kumar, G.; Umar, A. Pulse Laser Deposited Nanostructured ZnO Thin Films: A Review. J. Nanosci. Nanotechol. 2014, 14, 1911–1930. [Google Scholar] [CrossRef]
- Özgür, Ü.; Alivov, Y.I.; Liu, C.; Teke, A.; Reshchikov, M.A.; Doğan, S.; Avrutin, V.; Cho, S.-J.; Morkoç, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 041301. [Google Scholar] [CrossRef]
- Okoshi, M.; Higashikawa, K.; Hanabusa, M. Pulsed laser deposition of ZnO thin films using a femtosecond laser. Appl. Surf. Sci. 2000, 154/155, 424–427. [Google Scholar] [CrossRef]
- Millon, E.; Albert, O.; Loulergue, J.C.; Etchepare, J.; Hulin, D.; Seiler, W.; Perriere, J. Growth of heteroepitaxial ZnO thin films by femtosecond pulsed-laser deposition. J. Appl. Phys. 2000, 88, 6937–6939. [Google Scholar] [CrossRef]
- Perriere, J.; Millon, E.; Seiler, W.; Boulmer-Leborgne, C.; Craciun, V.; Albert, O.; Loulergue, J.C.; Etchepare, J. Comparison between ZnO films grown by femtosecond and nanosecond laser ablation. J. Appl. Phys. 2002, 91, 690–696. [Google Scholar] [CrossRef]
- Okoshi, M.; Higashikawa, K.; Hanabusa, M. Wavelength dependence of femtosecond pulsed laser deposition of zinc oxide films. Japan. J. Appl. Phys. 2001, 40, 1287–1288. [Google Scholar] [CrossRef]
- Perrière, J.S.; Millon, E.; Craciun, V. ZnO and ZnO-related compounds. In Pulsed Laser Deposition of Thin Films, Applications-Led Growth of Functional Materials; Eason, R., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2006; pp. 261–289. [Google Scholar]
- Klini, A.; Manousaki, A.; Anglos, D.; Fotakis, C. Growth of ZnO thin films by ultraviolet pulsed-laser ablation: Study of plume dynamics. J. Appl. Phys. 2005, 98, 123301. [Google Scholar] [CrossRef]
- Lansiart, L.; Millon, E.; Perrière, J.; Mathias, J.; Petit, A.; Seiler, W.; Boulmer-Leborgne, C. Structural properties of ZnO films grown by picosecond pulsed-laser deposition. Appl. Surf. Sci. 2012, 258, 9112–9115. [Google Scholar] [CrossRef]
- De Mesa, J.A.; Amo, A.M.; Miranda, J.J.C.; Salazar, H.O.; Sarmago, R.V.; Garcia, W.O. Effects of Deposition Pressure and Target-Substrate Distance on Growth of ZnO by Femtosecond Pulsed Laser Deposition. J. Laser Micro Nanoen 2016, 11, 21–24. [Google Scholar] [CrossRef][Green Version]
- Hashmi, J.Z.; Siraj, K.; Latif, A.; Murray, M.; Jose, G. Study of deposition parameters for the fabrication of ZnO thin films using femtosecond laser. Appl. Phys. A 2016, 122, 763. [Google Scholar] [CrossRef]
- Kokaj, J.; Shuaib, A.; Makdisi, Y.; Nair, R.; Mathew, J. Femtosecond laser based deposition of nanoparticles on a thin film and its characterization. Kuwait J. Sci. 2018, 45, 37–45. [Google Scholar]
- Hashmi, J.Z.; Siraj, K.; Latif, A.; Naseem, S.; Murray, M.; Jose, G. The role of samarium incorporated structural defects in ZnO thin films prepared by femtosecond pulsed laser deposition. J. Alloy. Compd. 2019, 800, 191–197. [Google Scholar] [CrossRef]
- Sanz, M.; Walczak, M.; de Nalda, R.; Oujja, M.; Marco, J.F.; Rodriguez, J.; Izquierdo, J.G.; Banares, L.; Castillejo, M. Femtosecond pulsed laser deposition of nanostructured TiO2 films. Appl. Surf. Sci. 2009, 255, 5206–5210. [Google Scholar] [CrossRef]
- Sanz, M.; Castillejo, M.; Amoruso, S.; Ausanio, G.; Bruzzese, R.; Wang, X. Ultra-fast laser ablation and deposition of TiO2. Appl. Phys. A 2010, 101, 639–644. [Google Scholar] [CrossRef]
- Gamez, F.; Plaza-Reyes, A.; Hurtado, P.; Guillen, E.; Anta, J.A.; Martınez-Haya, B.; Perez, S.; Sanz, M.; Castillejo, M.; Izquierdo, J.G.; et al. Nanoparticle TiO2 films prepared by pulsed laser deposition: Laser desorption and cationization of model adsorbates. J. Phys. Chem. C 2010, 114, 17409–17415. [Google Scholar] [CrossRef]
- Cavaliere, E.; Ferrini, G.; Pingue, P.; Gavioli, L. Fractal TiO2 Nanostructures by Nonthermal Laser Ablation at Ambient Pressure. J. Phys. Chem. C 2013, 117, 23305–23312. [Google Scholar] [CrossRef]
- Celardo, G.L.; Archetti, D.; Ferrini, G.; Gavioli, L.; Pingue, P.; Cavaliere, E. Evidence of diffusive fractal aggregation of TiO2 nanoparticles by femtosecond laser ablation at ambient conditions. Mater. Res. Express 2017, 4, 015013. [Google Scholar] [CrossRef]
- Cavaliere, E.; Benetti, G.; Celardo, G.L.; Archetti, D.; Pingue, P.; Ferrini, G.; Gavioli, L. Aggregation and fractal formation of Au and TiO2 nanostructures obtained by fs-pulsed laser deposition: Experiment and simulation. J. Nanopart. Res. 2017, 19, 311. [Google Scholar] [CrossRef]
- Gao, B.; Zhang, S.; Ju, X.; Lin, Y.; Wang, X. Femtosecond pulsed laser deposition of nanostructured TiO2 films in atmosphere. AIP Adv. 2017, 7, 095206. [Google Scholar] [CrossRef]
- Amoruso, S.; Tuzi, S.; Pallotti, D.K.; Aruta, C.; Bruzzese, R.; Chiarella, F.; Fittipaldi, R.; Lettieri, S.; Maddalena, P.; Sambri, A.; et al. Structural characterization of nanoparticles-assembled titanium dioxide films produced by ultrafast laser ablation and deposition in background oxygen. Appl. Surf. Sci. 2013, 270, 307–311. [Google Scholar] [CrossRef]
- Pallotti, D.K.; Orabona, E.; Amoruso, S.; Aruta, C.; Bruzzese, R.; Chiarella, F.; Tuzi, S.; Maddalena, P.; Lettieri, S. Multi-band photoluminescence in TiO2 nanoparticles-assembled films produced by femtosecond pulsed laser deposition. J. Appl. Phys. 2013, 114, 043503. [Google Scholar] [CrossRef]
- Pallotti, D.; Orabona, E.; Amoruso, S.; Maddalena, P.; Lettieri, S. Modulation of mixed-phase titania photoluminescence by oxygen adsorption. Appl. Phys Lett. 2014, 105, 031903. [Google Scholar] [CrossRef]
- Ni, X.; Sang, L.; Zhang, H.; Anoop, K.; Amoruso, S.; Wang, X.; Fittipaldi, R.; Li, T.; Hu, M.; Xu, L. Femtosecond laser deposition of TiO2 nanoparticle-assembled films with embedded CdS nanoparticles. Optoelectron. Lett. 2014, 10, 43–46. [Google Scholar] [CrossRef][Green Version]
- Sang, L.; Zhang, H.; Ni, X.; Anoop, K.K.; Fittipaldi, R.; Wang, X.; Amoruso, S. Hydrogen-evolving photoanode of TiO2 nanoparticles film deposited by a femtosecond laser. Int. J. Hydrogen Energy 2015, 40, 779–785. [Google Scholar] [CrossRef]
- Teghil, R.; Ferro, D.; Galasso, A.; Giardini, A.; Marotta, V.; Parisi, G.P.; Santagata, A.; Villani, P. Femtosecond pulsed laser deposition of nanostructured ITO thin films. Mater. Sci. Eng. C 2007, 27, 1034–1037. [Google Scholar] [CrossRef]
- Eisa, W.H.; Khafagi, M.G.; Shabaka, A.A.; Talaat, H.; Abd El-Karim, A.M. Femtosecond pulsed laser induced growth of highly transparent indium-tin-oxide thin films: Effect of deposition temperature and oxygen partial pressure. Optics 2015, 126, 3789–3794. [Google Scholar] [CrossRef]
- Latini, A.; Rau, J.V.; Ferro, D.; Teghil, R.; Rossi Albertini, V.; Barinov, S.M. Superhard Rhenium Diboride Films: Preparation and Characterization. Chem. Mater. 2008, 20, 4507–4511. [Google Scholar] [CrossRef]
- Chung, H.Y.; Weinberger, M.B.; Levine, J.B.; Kavner, A.; Yang, J.M.; Tolbert, S.H.; Kaner, R.B. Synthesis of ultra-incompressible superhard rhenium diboride at ambient pressure. Science 2007, 316, 436–439. [Google Scholar] [CrossRef]
- ASM. Handbook Alloy Phase Diagrams; ASM International: Materials Park, OH, USA, 1992; Volume 3. [Google Scholar]
- Rau, J.V.; Ferro, D.; Falcone, M.B.; Generosi, A.; Rossi Albertini, V.; Latini, A.; Teghil, R.; Barinov, S.M. Hardness of zirconium diboride films deposited on titanium substrates. Mater. Chem. Phys. 2008, 112, 504–509. [Google Scholar] [CrossRef]
- Tamura, H.; Konoue, M.; Sawaoka, A.B. Zirconium boride and tantalum carbide coatings sprayed by electrothermal explosion of powders. J. Therm. Spray. Technol. 1997, 6, 463–468. [Google Scholar] [CrossRef]
- De Bonis, A.; Teghil, R.; Rau, J.V.; Galasso, A.; Orlando, S.; Santagata, A. Characterization of gaseous phase and nanoparticles produced in ultra-short pulsed laser ablation of transition metal borides. Appl. Surf. Sci. 2011, 257, 5315–5318. [Google Scholar] [CrossRef]
- Chung, H.; Weinberger, M.B.; Yang, J.; Tolbert, S.H.; Kaner, R.B. Correlation between hardness and elastic moduli of the ultraincompressible transition metal diborides RuB2, OsB2, and ReB2. Appl. Phys. Lett. 2008, 92, 261904. [Google Scholar] [CrossRef]
- Rau, J.V.; Latini, A.; Generosi, A.; Rossi Albertini, V.; Ferro, D.; Teghil, R.; Barinov, S.M. Deposition and characterization of superhard biphasic ruthenium boride films. Acta Mater. 2009, 57, 673–681. [Google Scholar] [CrossRef]
- Weinberger, M.B.; Levine, J.B.; Chung, H.; Cumberland, R.W.; Rasool, H.I.; Yang, J.; Kaner, R.B.; Tolbert, S.H. Incompressibility and Hardness of Solid Solution Transition Metal Diborides: Os1−xRuxB2. Chem. Mater. 2009, 21, 1915–1921. [Google Scholar] [CrossRef]
- Latini, A.; Rau, J.V.; Teghil, R.; Generosi, A.; Rossi Albertini, V. Superhard Properties of Rhodium and Iridium Boride Films. ACS Appl. Mater. Interfaces 2010, 2, 581–587. [Google Scholar] [CrossRef]
- Samsonov, G.V.; Kosenko, V.A.; Rud’, B.M.; Sidorova, V.G. Preparation and properties of the compound IrB1.1. Inorg. Mater. 1972, 8, 671–672. [Google Scholar]
- Zeiringer, I.; Cheng, X.; Chen, X.; Bauer, E.; Giester, G.; Rogl, P.F. Crystal structures and constitution of the binary system iridium-boron. Sci. China Mater. 2015, 58, 649–668. [Google Scholar] [CrossRef]
- Rau, J.V.; Latini, A.; Teghil, R.; De Bonis, A.; Fosca, A.; Caminiti, R.; Rossi Albertini, V. Superhard Tungsten Tetraboride Films Prepared by Pulsed Laser Deposition Method. ACS Appl. Mater. Interfaces 2011, 3, 3738–3743. [Google Scholar] [CrossRef]
- Schlesinger, M.E.; Liao, P.K.; Spear, K.E. The B-La (Boron-Lanthanum) System. J. Phase Equilib. 1999, 20, 73–78. [Google Scholar] [CrossRef]
- Lafferty, J.M. Boride Cathodes. J. Appl. Phys. 1951, 22, 299–309. [Google Scholar] [CrossRef]
- Bellucci, A.; Mastellone, M.; Orlando, S.; Girolami, M.; Generosi, A.; Paci, B.; Soltani, P.; Mezzi, A.; Kaciulis, S.; Polini, R.; et al. Lanthanum (oxy)boride thin films for thermionic emission applications. Appl. Surf. Sci. 2019, 479, 296–302. [Google Scholar] [CrossRef]
- Liao, P.K.; Spear, K.E. The B-Cr (boron-chromium) system. Bull. Alloy Phase Diagr. 1986, 7, 232–237. [Google Scholar] [CrossRef]
- Pierson, H.O. Handbook of Refractory Carbides and Nitrides; Noyes Publications: Westwood, CA, USA, 1996. [Google Scholar]
- Krajewski, A.; D’Alessio, L.; De Maria, G. Physico-chemical and thermophysical properties of cubic binary carbides. Cryst. Res. Technol. 1998, 33, 341–374. [Google Scholar] [CrossRef]
- Teghil, R.; D’Alessio, L.; De Bonis, A.; Galasso, A.; Villani, P.; Zaccagnino, M.; Santagata, A.; Ferro, D. Femtosecond pulsed laser ablation of group 4 carbides. Appl. Surf. Sci. 2005, 247, 51–56. [Google Scholar] [CrossRef]
- Ferro, D.; Rau, J.V.; Rossi Albertini, V.; Generosi, A.; Teghil, R.; Barinov, S.M. Pulsed laser deposited hard TiC, ZrC, HfC and TaC films on titanium: Hardness and an energy-dispersive X-ray diffraction study. Surf. Coat. Technol. 2008, 202, 1455–1461. [Google Scholar] [CrossRef]
- Andrievski, R.A.; Spivak, I.I. Strength of Refractory Compounds and Related Materials; Metallurgy: Tcheljabinsk, Russia, 1989. [Google Scholar]
- Teghil, R.; De Bonis, A.; Galasso, A.; Villani, P.; Santagata, A.; Ferro, D.; Barinov, S.M. Nanostructured thin films obtained by ultra-short pulse laser deposition of vanadium carbide. Appl. Surf. Sci. 2009, 255, 5220–5223. [Google Scholar] [CrossRef]
- Sansone, M.; De Bonis, A.; Santagata, A.; Rau, J.V.; Galasso, A.; Teghil, R. Pulsed laser ablation and deposition of niobium carbide. Appl. Surf. Sci. 2016, 374, 112–116. [Google Scholar] [CrossRef]
- Duhalde, S.; Colaco, R.; Audebert, F.; Perrone, A.; Zocco, A. Deposition of NbC thin films by pulsed laser ablation. Appl. Phys. A 1999, 69, S569–S571. [Google Scholar] [CrossRef]
- Teghil, R.; De Bonis, A.; Galasso, A.; Villani, P.; Santagata, A. Femtosecond pulsed laser ablation deposition of tantalum carbide. Appl. Surf. Sci. 2007, 254, 1220–1223. [Google Scholar] [CrossRef]
- Wallace, T.C.; Butt, D.P. Review of diffusion and vaporization of Group 4 and 5 transition metal carbides. In Chemistry of Transition Metal Carbides and Nitrides; Oyama, S.T., Ed.; Blackie Academic and Professional: Glasgow, UK, 1996; pp. 53–90. [Google Scholar]
- Avatbé, R.G. The thermodynamic stability of monocarbides of transition metals from sub groups IV–VI. Powder Metall. Met. Ceram. 1965, 4, 122–128. [Google Scholar]
- Jubair, M.; Tanveer Karim, A.M.M.; Nuruzzaman, M.; Zilani, M.A.K. Comparison of structural, mechanical and optical properties of tantalum hemicarbide with tantalum monocarbide: Ab initio calculations. J. Phys. Commun. 2019, 3, 055017. [Google Scholar] [CrossRef]
- Hackett, K.; Verhoef, S.; Cutler, R.A.; Shetty, D.K. Phase Constitution and Mechanical Properties of Carbides in the Ta–C System. J. Am. Ceram. Soc. 2009, 92, 2404–2407. [Google Scholar] [CrossRef]
- Doi, K.; Hiraishi, S.; Kawasaki, H.; Suda, Y. Preparation of Crystalline Chromium Carbide Thin Films Synthesized By Pulsed Nd:YAG Laser Deposition. Mater. Res. Soc. Symp. 2000, 617, J7.8. [Google Scholar] [CrossRef]
- Teghil, R.; Santagata, A.; De Bonis, A.; Galasso, A.; Villani, P. Chromium carbide thin films deposited by ultra-short pulse laser deposition. Appl. Surf. Sci. 2009, 255, 7729–7733. [Google Scholar] [CrossRef]
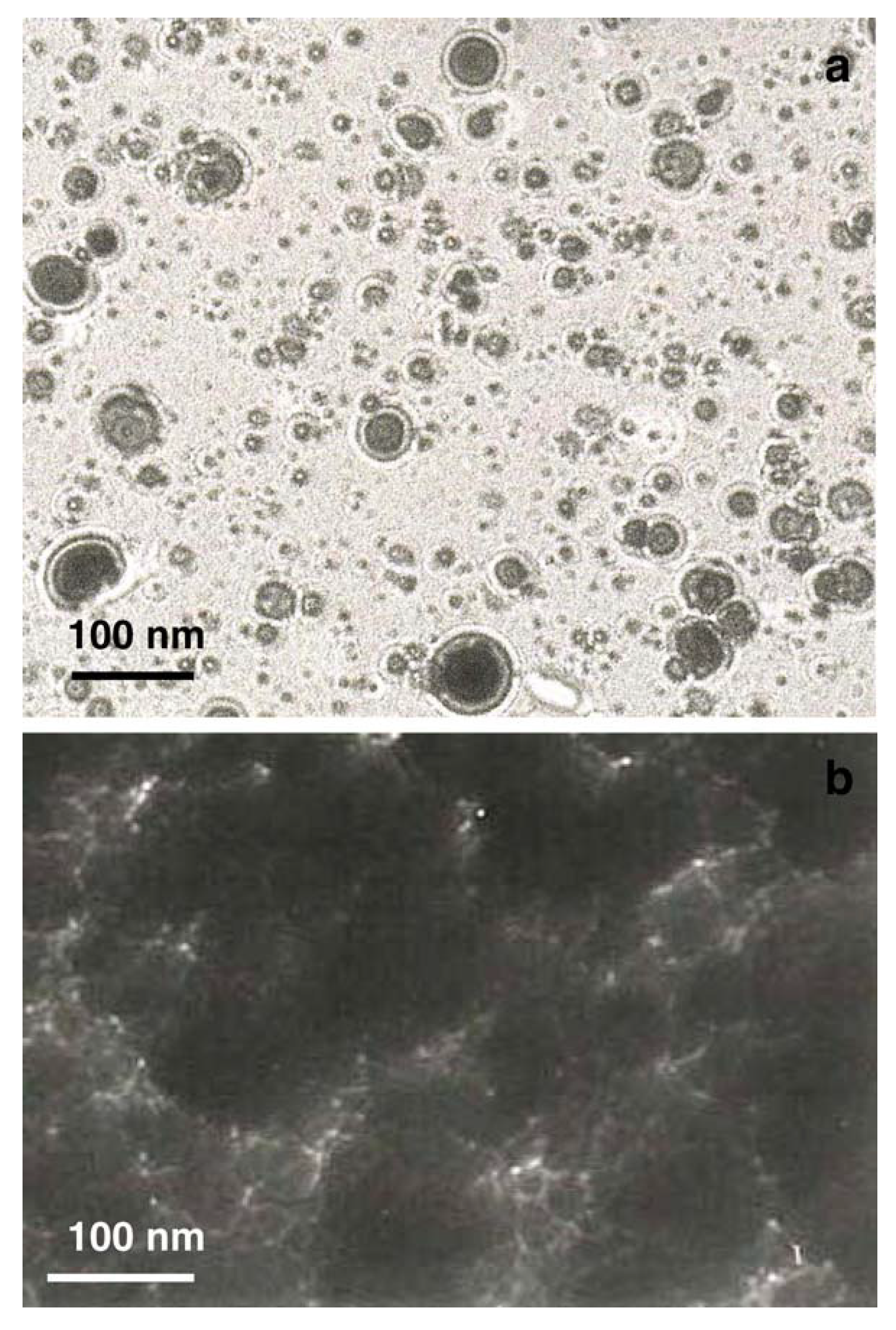
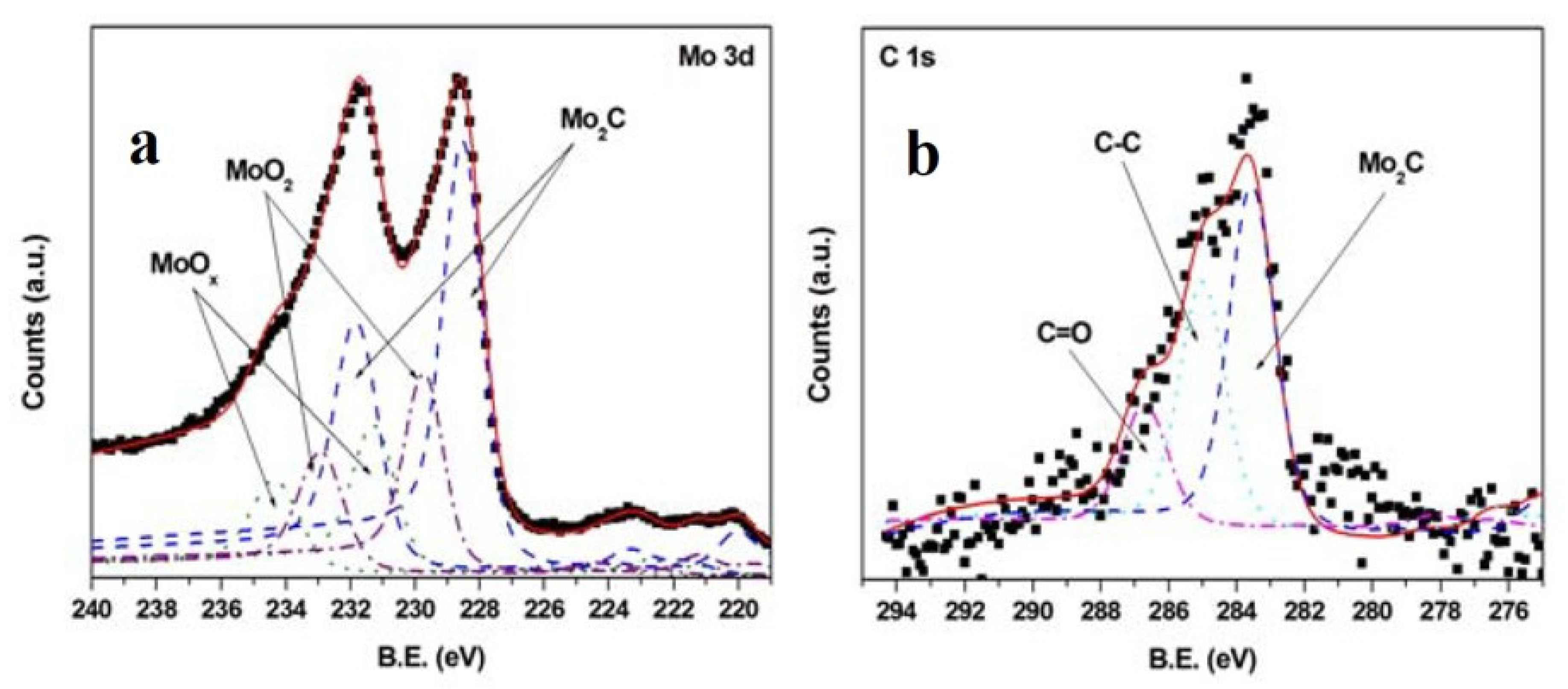
- De Bonis, A.; Santagata, A.; Sansone, M.; Rau, J.V.; Mori, T.; Teghil, R. Femtosecond pulsed laser ablation of molybdenum carbide: Nanoparticles and thin film characteristics. Appl. Surf. Sci. 2013, 278, 321–324. [Google Scholar] [CrossRef]
- Teghil, R.; De Bonis, A.; Galasso, A.; Sansone, M.; Rau, J.V.; Santagata, A. Nanostructured molybdenum carbide thin films obtained by femtosecond pulsed laser deposition. Phys. Status Solidi C 2012, 9, 2370–2373. [Google Scholar] [CrossRef]
- De Bonis, A.; Teghil, R.; Santagata, A.; Galasso, A.; Rau, J.V. Thin films deposited by femtosecond pulsed laser ablation of tungsten carbide. Appl. Surf. Sci. 2012, 258, 9198–9201. [Google Scholar] [CrossRef]
- Cappelli, E.; Bellucci, A.; Orlando, S.; Trucchi, D.M.; Mezzi, A.; Valentini, V. PLD deposition of tungsten carbide contact for diamond photodiodes. Influence of process conditions on electronic and chemical aspects. Appl. Surf. Sci. 2013, 278, 111–116. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Bonis, A.; Teghil, R. Ultra-Short Pulsed Laser Deposition of Oxides, Borides and Carbides of Transition Elements. Coatings 2020, 10, 501. https://doi.org/10.3390/coatings10050501
De Bonis A, Teghil R. Ultra-Short Pulsed Laser Deposition of Oxides, Borides and Carbides of Transition Elements. Coatings. 2020; 10(5):501. https://doi.org/10.3390/coatings10050501
Chicago/Turabian StyleDe Bonis, Angela, and Roberto Teghil. 2020. "Ultra-Short Pulsed Laser Deposition of Oxides, Borides and Carbides of Transition Elements" Coatings 10, no. 5: 501. https://doi.org/10.3390/coatings10050501
APA StyleDe Bonis, A., & Teghil, R. (2020). Ultra-Short Pulsed Laser Deposition of Oxides, Borides and Carbides of Transition Elements. Coatings, 10(5), 501. https://doi.org/10.3390/coatings10050501





