Comparison of the Clinical Results of Identically Designed Total Knee Prostheses with Different Surface Roughnesses
Abstract
1. Introduction
2. Materials and Methods
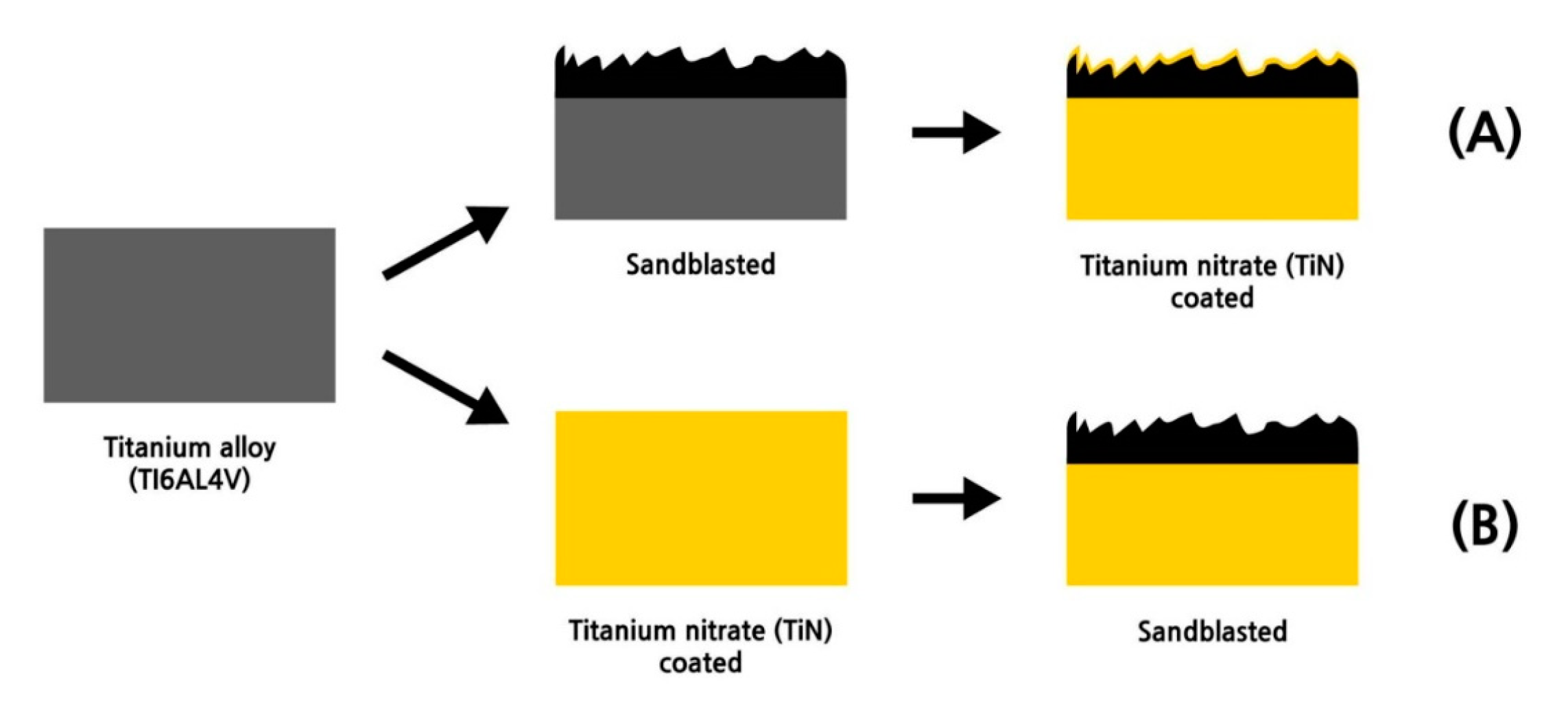
2.1. Surface Roughnesses of Prosthesis
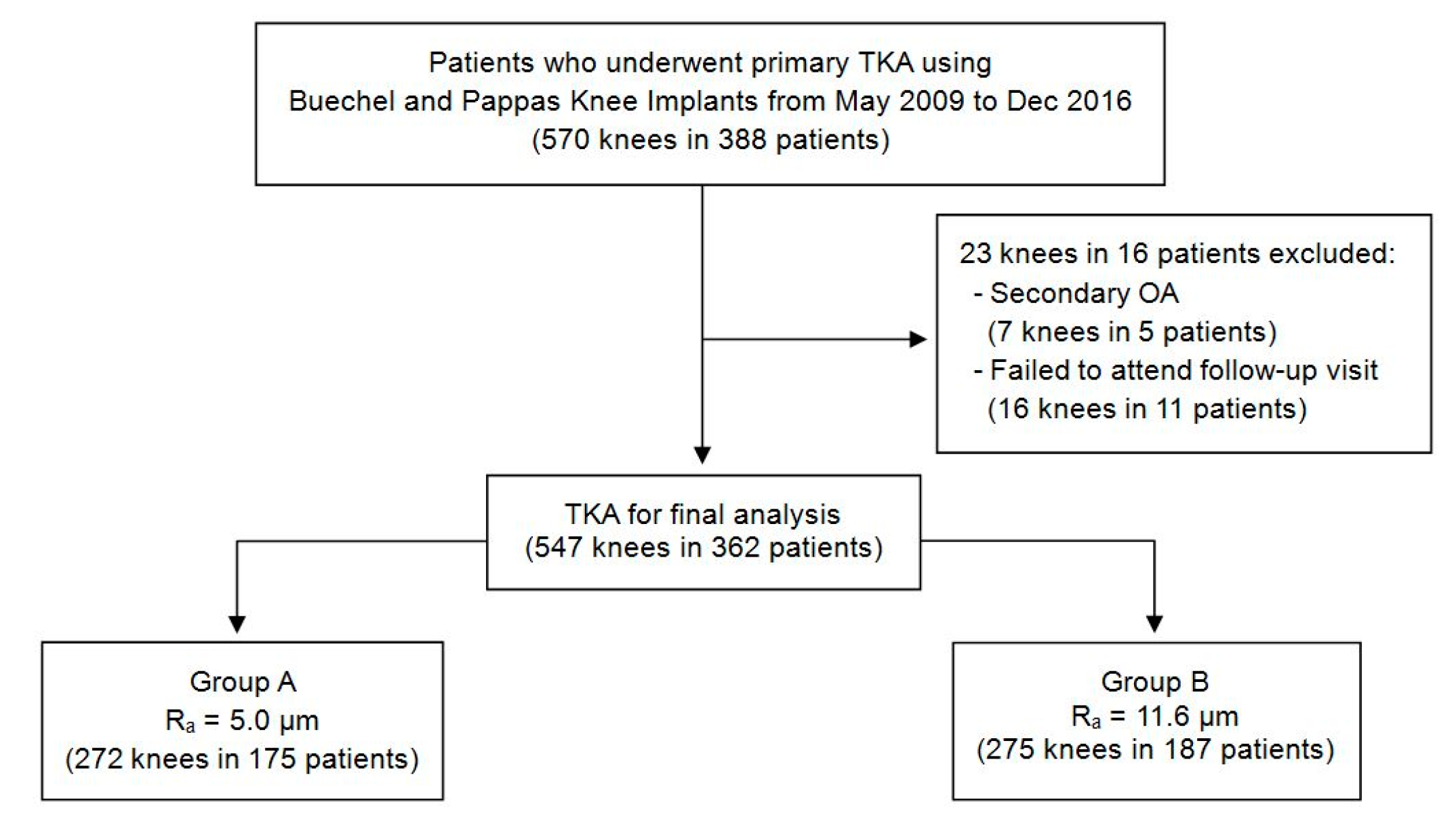
2.2. Patient Selection
2.3. Operative Procedure and Treatment after Surgery
2.4. Clinical and Radiological Assessments
2.5. Statistical Analysis
3. Results
3.1. Patients Demographics
3.2. Clinical and Radiological Outcomes
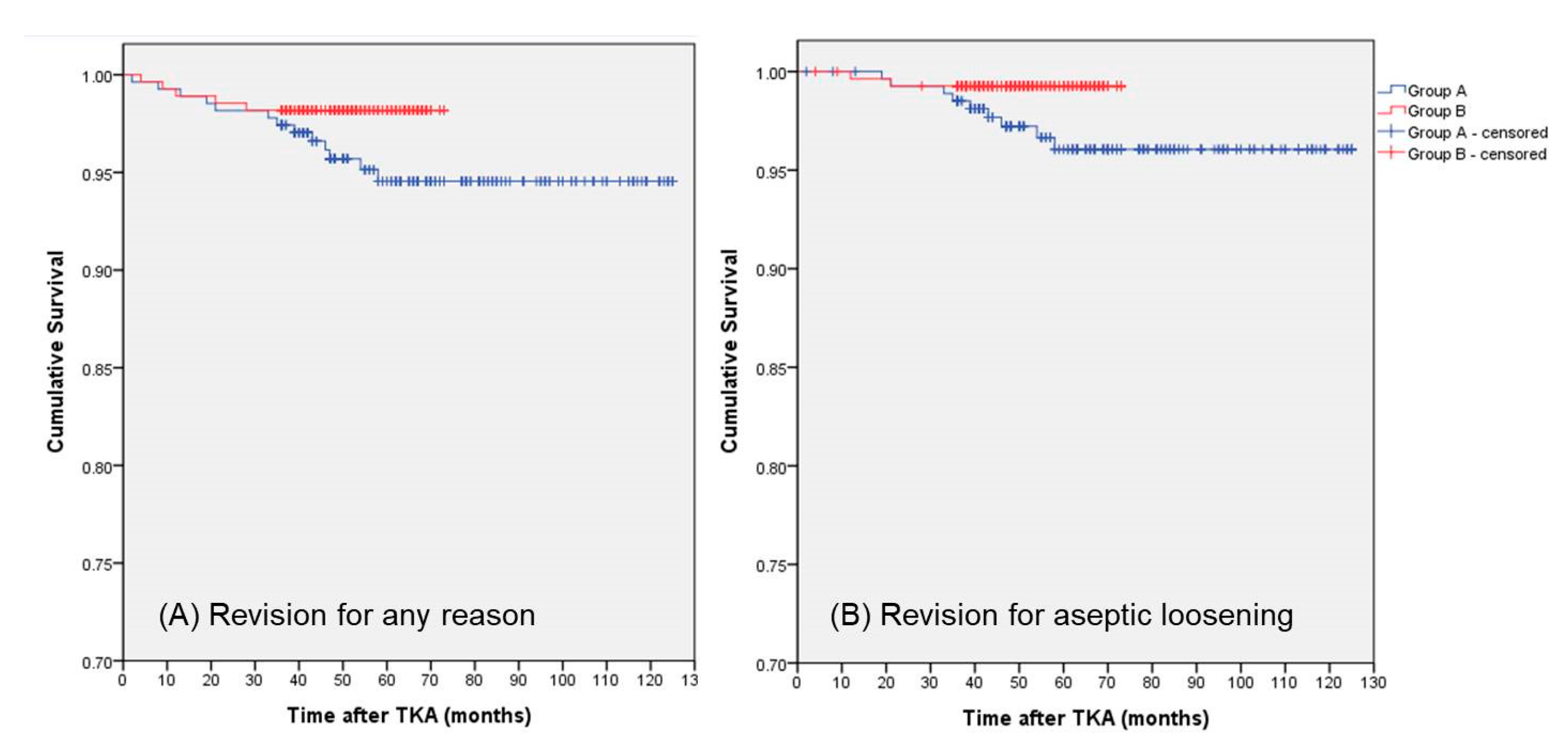
3.3. Prosthesis Survivorship
3.4. The Risk Factors of Aseptic Loosening in Multivariate Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lachiewicz, P.F.; Soileau, E.S. The rates of osteolysis and loosening associated with a modular posterior stabilized knee replacement. Results at five to fourteen years. J. Bone Joint Surg Am. 2004, 86, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, A.V., Jr.; Berasi, C.C.; Berend, K.R. Evolution of tibial fixation in total knee arthroplasty. J. Arthroplast. 2007, 22, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Jasty, M.; Maloney, W.J.; Bragdon, C.R.; O’Connor, D.O.; Haire, T.; Harris, W.H. The initiation of failure in cemented femoral components of hip arthroplasties. J. Bone Jt. Surg. 1991, 73, 551–558. [Google Scholar] [CrossRef]
- Vanlommel, J.; Luyckx, J.P.; Labey, L.; Innocenti, B.; De Corte, R.; Bellemans, J. Cementing the tibial component in total knee arthroplasty: Which technique is the best? J. Arthroplast. 2011, 26, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Pittman, G.T.; Peters, C.L.; Hines, J.L.; Bachus, K.N. Mechanical bond strength of the cement-tibial component interface in total knee arthroplasty. J. Arthroplast. 2006, 21, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Shepard, M.F.; Kabo, J.M.; Lieberman, J.R. The Frank Stinchfield Award. Influence of cement technique on the interface strength of femoral components. Clin. Orthop. Relat. Res. 2000, 381, 26–35. [Google Scholar] [CrossRef]
- Zelle, J.; Janssen, D.; Peeters, S.; Brouwer, C.; Verdonschot, N. Mixed-mode failure strength of implant–cement interface specimens with varying surface roughness. J. Biomech. 2011, 44, 780–783. [Google Scholar] [CrossRef]
- Cho, W.S. Knee Joint Arthroplasty; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Hung, J.P.; Bai, Y.W.; Hung, C.Q.; Lee, T.E. Biomechanical performance of the cemented hip stem with different surface finish. Appl. Sci. 2019, 9, 4082. [Google Scholar] [CrossRef]
- Mohler, C.G.; Callaghan, J.J.; Collis, D.K.; Johnston, R.C. Early loosening of the femoral component at the cement prosthesis interface after total hip replacement. J. Bone Jt. Surg. 1995, 77, 1315–1322. [Google Scholar] [CrossRef]
- Manley, M.T.; Stern, L.S.; Gurtowski, J. The load carrying and fatigue properites of the stem cement interface with smooth and porous coated femoral components. J. Biomed. Mater. Res. 1985, 19, 563–575. [Google Scholar] [CrossRef]
- Moon, K.H.; Hong, S.H.; Hong, T.H. Total knee replacement arthroplasty with Buechel and Pappas knee: Minimum 2-year follow-up. Clin. Orthop. Surg. 2015, 7, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Gotman, I.; Gutmanas, E.Y. Titanium nitride-based coatings on implantable medical devices. Adv. Biomater. Devices Med. 2014, 1, 53–73. [Google Scholar]
- Zimmer Biomet. Modern Cementing Technique. Available online: https://www.zimmerbiomet.com/content/dam/zimmer-biomet/medical-professionals/common/cement/modern-cementing-technique-brochure.pdf (accessed on 1 May 2020).
- Insall, J.N.; Dorr, L.D.; Scott, R.D.; Scott, W.N. Rationale of the Knee Society clinical rating system. Clin. Orthop. Relat. Res. 1989, 248, 13–14. [Google Scholar] [CrossRef]
- Gioe, T.J.; Killeen, K.K.; Grimm, K.; Mehle, S.; Scheltema, K. Why are total knee replacements revised? Analysis of early revision in a community knee implant registry. Clin. Orthop. Relat. Res. 2004, 428, 100–106. [Google Scholar] [CrossRef]
- Guha, A.R.; Debnath, U.K.; Graham, N.M. Radiolucent lines below the tibial component of a total knee replacement (TKR)—A comparison between single-and two-stage cementation techniques. Int. Orthop. 2008, 32, 453–457. [Google Scholar] [CrossRef][Green Version]
- Durig, N.; Pace, T.; Broome, B.; Osuji, O.; Harman, M.K. Clinical outcomes of tibial components with modular stems used in primary TKA. Adv. Orthop. 2014, 2014, 651279. [Google Scholar] [CrossRef]
- Lum, Z.C.; Shieh, A.K.; Dorr, L.D. Why total knees fail—A modern perspective review. World J. Orthop. 2018, 18, 60–64. [Google Scholar] [CrossRef]
- Harris, W.H. Is it advantageous to strengthen the cement–metal interface and use a collar for cemented femoral components of total hip replacement? Clin. Orthop. 1992, 285, 67–72. [Google Scholar] [CrossRef]
- Maloney, W.J.; Jasty, M.; Burke, D.W.; O’Connor, D.O.; Zalenski, E.B.; Bragdon, C.; Harris, W.H. Biomechanical and histologic investigation of emented total hip arthroplasties. A study of autopsy-retrieved femurs after in vivo cycling. Clin. Orthop. Relat. Res. 1989, 249, 129–140. [Google Scholar]
- Stauffer, R.N. Ten-year follow-up study of total hip replacement. J. Bone Joint Surg. Am. 1982, 64, 983–990. [Google Scholar] [CrossRef]
- Fornasier, V.L.; Cameron, H.U. The femoral stem/cement interface in total hip replacement. Clin. Orthop. Relat. Res. 1976, 116, 248–252. [Google Scholar] [CrossRef]
- Mikulak, S.A.; Mahoney, O.M.; dela Rosa, M.A.; Schmalzried, T.P. Loosening and osteolysis with the press-fit condylar posterior-cruciate-substituting total knee replacement. J. Bone Joint Surg. Am. 2001, 83, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Kang, S.B.; Yoon, K.S. High incidence of loosening of the femoral component in legacy posterior stabilised-flex total knee replacement. J. Bone Joint Surg. Br. 2007, 89, 1457–1461. [Google Scholar] [CrossRef] [PubMed]
- Kutzner, I.; Hallan, G.; Høl, P.J.; Furnes, O.; Gøthesen, Ø.; Figved, W.; Ellison, P. Early aseptic loosening of a mobile-bearing total knee replacement. Acta Orthop. 2018, 89, 77–83. [Google Scholar] [CrossRef]
- Van de Groes, S.; de Waal-Malefijt, M.; Verdonschot, N. Probability of mechanical loosening of the femoral component in high flexion total knee arthroplasty can be reduced by rather simple surgical techniques. Knee 2014, 21, 209–215. [Google Scholar] [CrossRef]
- Arola, D.; Stoffel, K.A.; Yang, D.T. Fatigue of the cement/bone interface: The surface texture of bone and loosening. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 76, 287–297. [Google Scholar] [CrossRef] [PubMed]

| Variable | Group A Ra = 5.0 μm (272 Knees in 175 Patients) | Group B Ra = 11.6 μm (275 Knees in 187 Patients) | p Value |
|---|---|---|---|
| Mean age at surgery (years, range) a | 68.2 ± 8.0 (45–92) | 67.6 ± 7.6 (47–93) | 0.481 |
| Gender (n, %) b | – | – | 0.494 |
| Male | 21 (12.0%) | 27 (14.4%) | |
| Female | 154 (88.0%) | 160 (85.6%) | |
| Duration of follow-up (months, range) a | 69.7 ± 25.6 (36–125) | 51.0 ± 10.6 (36–73) | 0.001 * |
| Body Mass Index (kg/m2) a | 26.1 ± 3.7 | 26.5 ± 3.9 | 0.296 |
| Bone Mineral Density (T-score) a | −1.83 ± 1.16 | −1.88 ± 1.11 | 0.768 |
| Knees (n, %) b | – | – | – |
| Bilateral | 97 (55.4%) | 88 (47.1%) | 0.111 |
| Unilateral | 78 (44.6%) | 99 (52.9%) | |
| Right | 42 (53.8%) | 53 (53.5%) | 0.967 |
| Left | 36 (46.2%) | 46 (46.5%) |
| Variable | Group A Ra = 5.0 μm (272 Knees in 175 Patients) | Group B Ra = 11.6 μm (275 Knees in 187 Patients) | p Value |
|---|---|---|---|
| Range of motion a | – | – | – |
| Preoperative flexion contracture (°) | 2.3 ± 5.0 | 2.0 ± 4.9 | 0.399 |
| Postoperative flexion contracture (°) | 0.37 ± 3.23 | 0.39 ± 1.82 | 0.950 |
| Preoperative range of motion (°) | 121.5 ± 13.0 | 119.7 ± 9.8 | 0.154 |
| Postoperative range of motion (°) | 118.1 ± 14.0 | 120.1 ± 9.1 | 0.164 |
| Outcome scores a | – | – | – |
| Preoperative clinical AKSS | 68.1 ± 8.5 | 67.1 ± 5.4 | 0.276 |
| Postoperative clinical AKSS | 95.2 ± 7.6 | 95.8 ± 7.3 | 0.515 |
| Preoperative functional AKSS | 65.7 ± 7.2 | 64.0 ± 11.5 | 0.279 |
| Postoperative functional AKSS | 83.2 ± 20.5 | 88.8 ± 17.3 | 0.023 * |
| Complications b | – | – | – |
| Superficial infection | 1 (0.4%) | 0 (0%) | 0.497 |
| Hypertrophic synovitis | 1 (0.4%) | 0 (0%) | 0.497 |
| Deep vein thrombosis | 3 (1.1%) | 4 (1.5%) | >0.999 |
| Stiffness after TKA | 8 (2.9%) | 7 (2.5%) | 0.800 |
| Revision surgery b | – | – | – |
| Any reason | 13 (4.8%) | 5 (1.8%) | 0.058 |
| Aseptic loosening | 9 (3.3%) | 2 (0.7%) | 0.035 * |
| Prosthetic joint infection | 4 (1.5%) | 2 (0.7%) | 0.448 |
| Periprosthetic fracture | 0 (0%) | 1 (0.4%) | >0.999 |
| Radiologic findings b | – | – | – |
| Femoral osteolysis | 7 (2.6%) | 2 (0.7%) | 0.105 |
| Femoral radiolucencies | 10 (5.7%) | 7 (2.5%) | 0.472 |
| Tibial osteolysis | 21 (7.7%) | 5 (1.8%) | 0.001 * |
| Tibial radiolucencies | 25 (9.2%) | 7 (2.5%) | 0.001 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, S.H.; Moon, K.H. Comparison of the Clinical Results of Identically Designed Total Knee Prostheses with Different Surface Roughnesses. Coatings 2020, 10, 486. https://doi.org/10.3390/coatings10050486
Ko SH, Moon KH. Comparison of the Clinical Results of Identically Designed Total Knee Prostheses with Different Surface Roughnesses. Coatings. 2020; 10(5):486. https://doi.org/10.3390/coatings10050486
Chicago/Turabian StyleKo, Sang Hyun, and Kyoung Ho Moon. 2020. "Comparison of the Clinical Results of Identically Designed Total Knee Prostheses with Different Surface Roughnesses" Coatings 10, no. 5: 486. https://doi.org/10.3390/coatings10050486
APA StyleKo, S. H., & Moon, K. H. (2020). Comparison of the Clinical Results of Identically Designed Total Knee Prostheses with Different Surface Roughnesses. Coatings, 10(5), 486. https://doi.org/10.3390/coatings10050486





