Biomimetic Coatings Obtained by Combinatorial Laser Technologies
Abstract
1. Biomimetic Materials
2. Biomimetic Coatings: In Vitro Testing Strategies and Clinical Trials
2.1. In Vitro Testing of Inorganic Coatings for Tissue Engineering
2.2. In Vitro Testing of Organic Coatings for Tissue Engineering
2.3. In Vitro Testing of Bio-Coatings for Cancer Research
2.4. In Vivo Clinical Trials for Regenerative Medicine
3. Biomimetic Coatings: Advantages and Drawbacks of Processing Technologies
3.1. Biomimetic Processing Technologies
3.2. Bio-Coating Adhesion Issues
4. Biomimetic Laser Processing
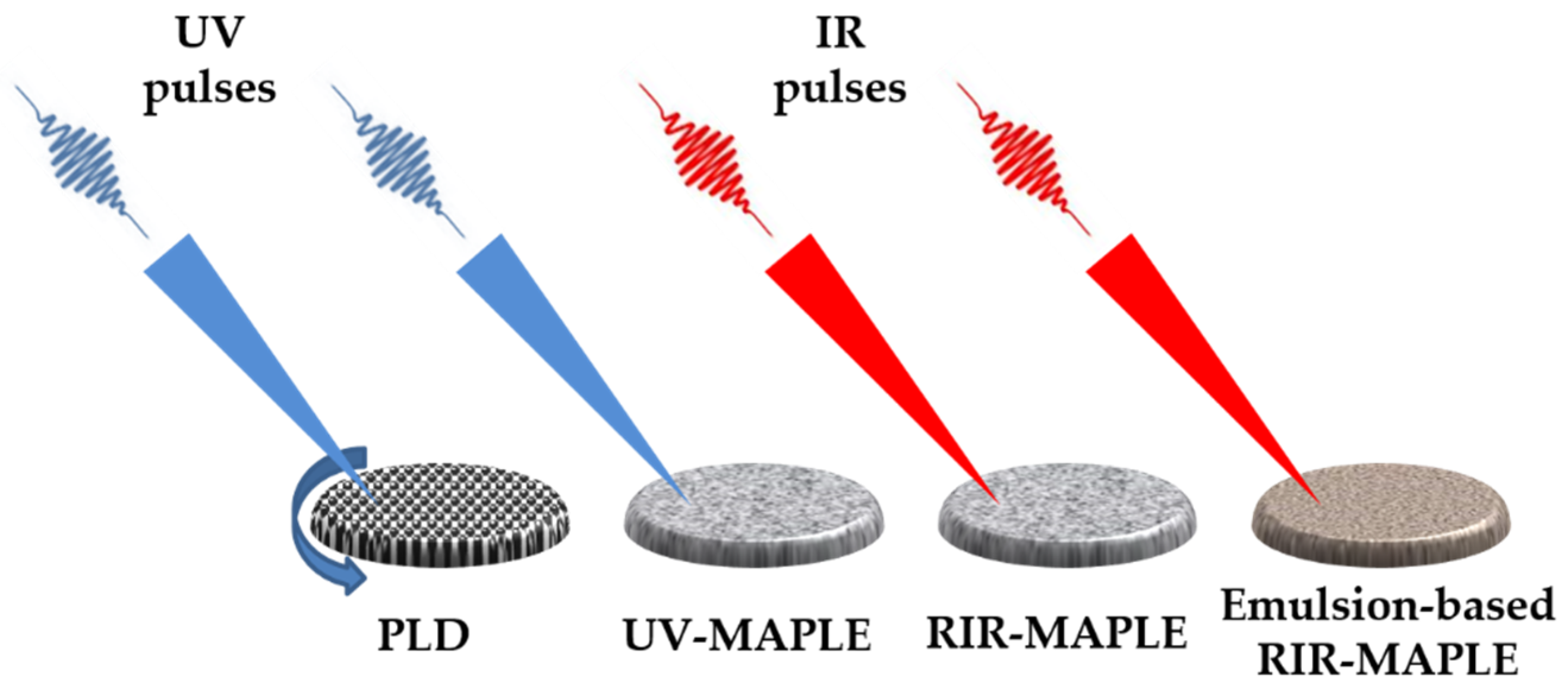
4.1. Pulsed Laser Deposition
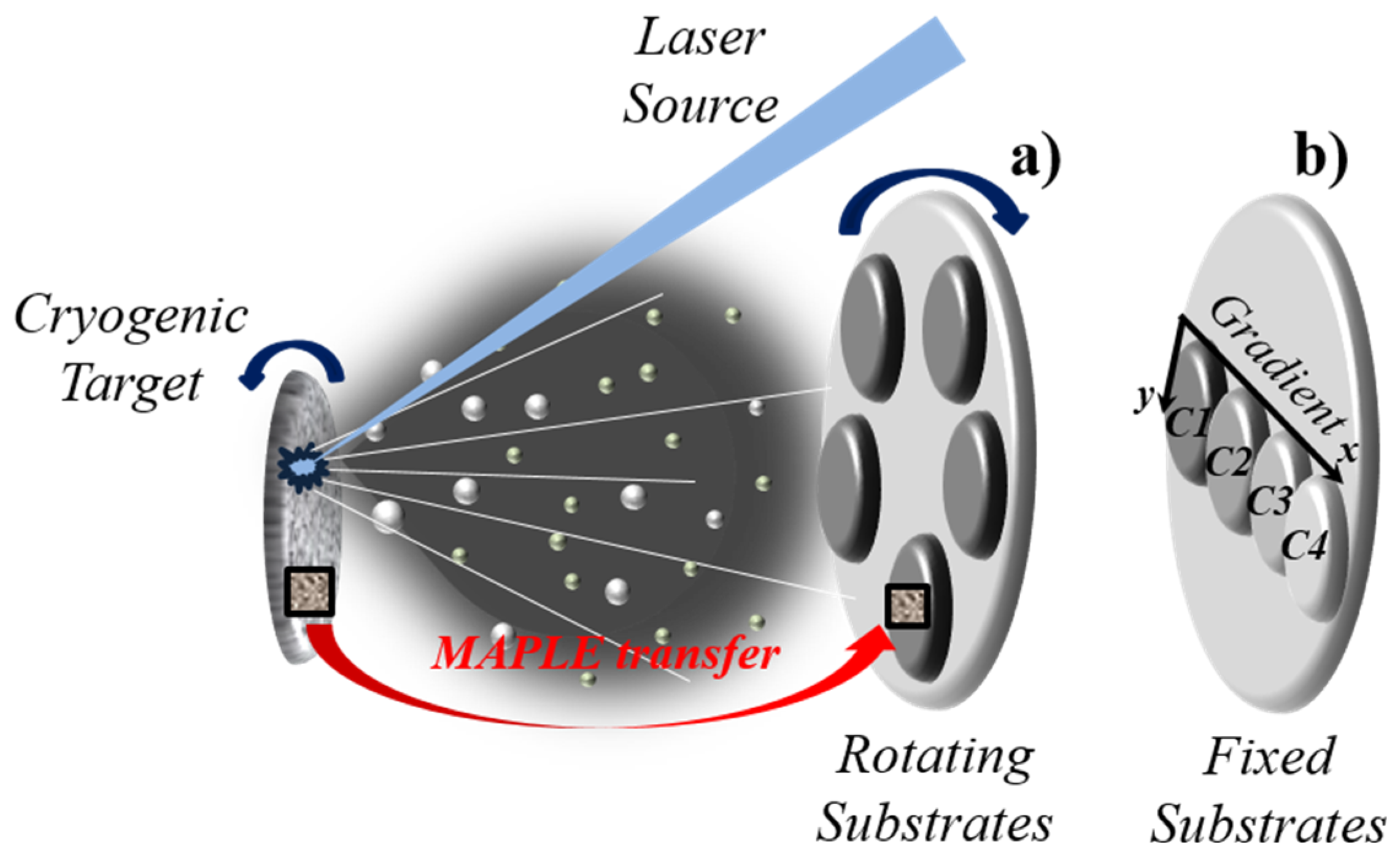
4.2. Matrix Assisted Pulsed Laser Evaporation
5. Bio-Coatings with Multilayer Configurations and Gradient of Composition by Laser Deposition
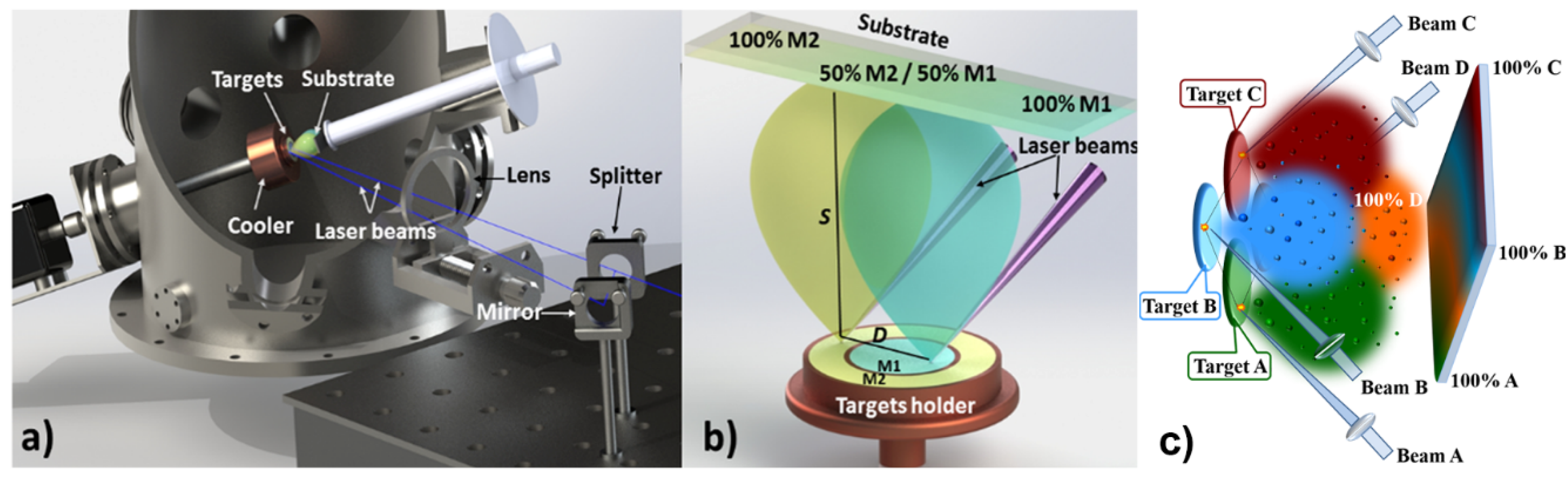
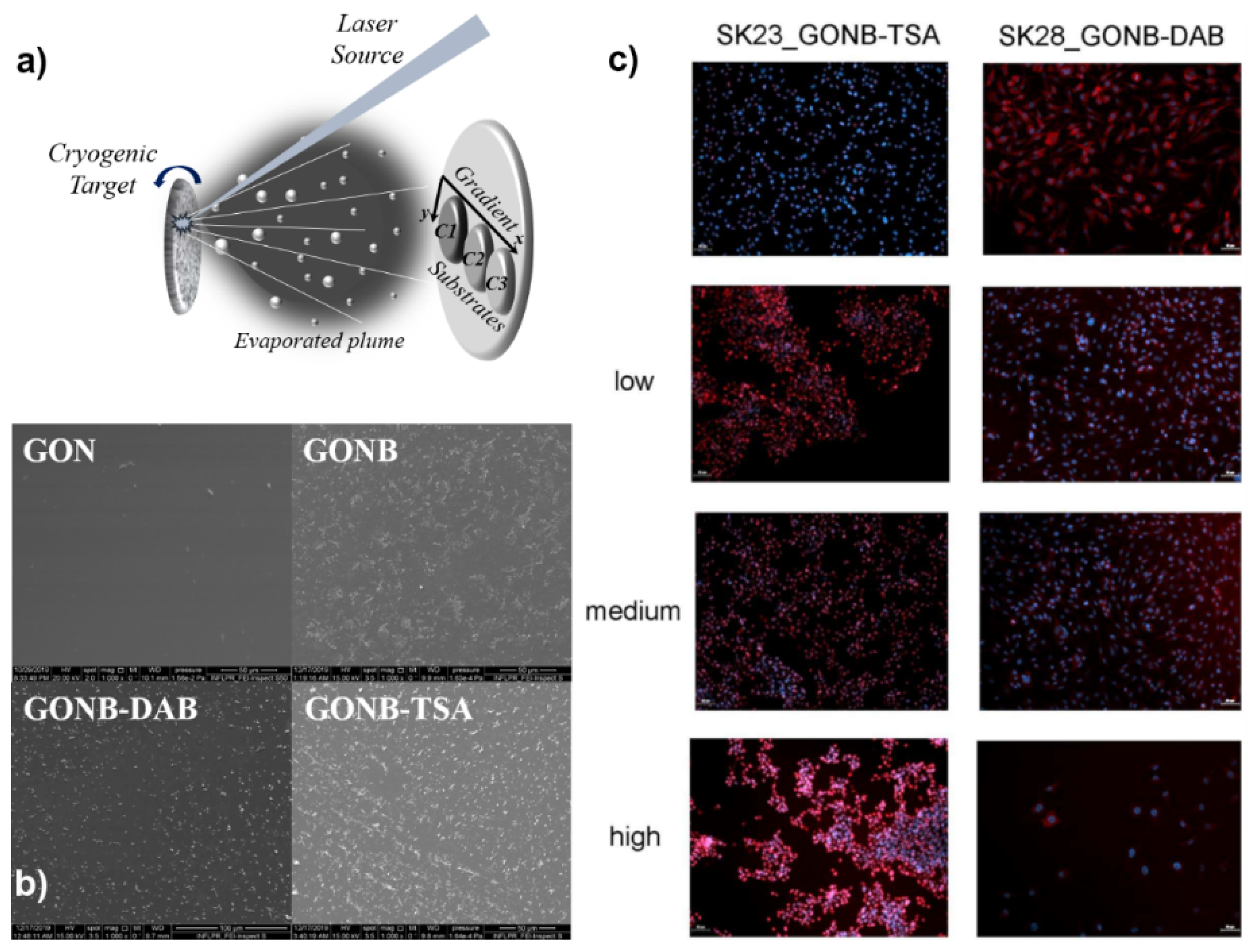
5.1. Combinatorial Laser Technologies
5.2. Ion-Doped Inorganic Bio-Coatings Obtained by Combinatorial-Matrix Assisted Pulsed Laser Evaporation
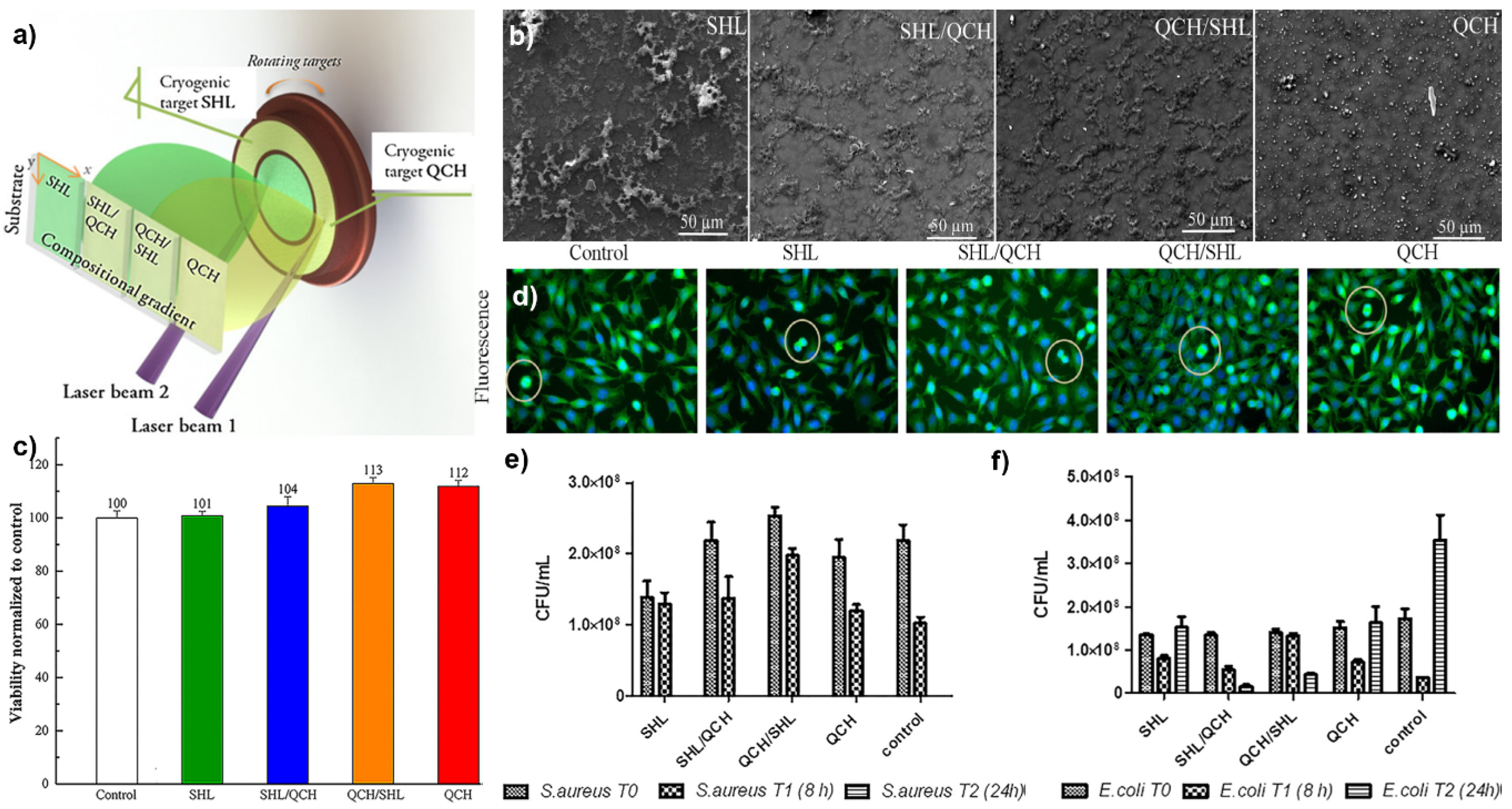
5.3. Multi-Functional Organic Bio-Coatings Obtained by C-MAPLE
5.4. Hybrid Bio-Coatings by MAPLE for Anti-Tumor Drug Delivery to Cancer Cells
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Schummer, J. Multidisciplinarity, interdisciplinarity, and patterns of research collaboration in nanoscience and nanotechnology. Scientometrics 2004, 59, 425–465. [Google Scholar] [CrossRef]
- Zhang, F. Grand challenges for nanoscience and nanotechnology in energy and health. Front. Chem. 2017, 5, 80. [Google Scholar] [CrossRef] [PubMed]
- Patil, A.; Mishra, V.; Thakur, S.; Riyaz, B.; Kaur, A.; Khursheed, R.; Patil, K.; Sathe, B. Nanotechnology derived nanotools in biomedical perspectives: An update. Curr. Nanosci. 2019, 15, 137–146. [Google Scholar] [CrossRef]
- Hench, L.L.; Polak, J.M. Third-generation biomedical materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef] [PubMed]
- Ning, C.; Zhou, L.; Tan, G. Fourth-generation biomedical materials. Mater. Today 2016, 19, 2–3. [Google Scholar] [CrossRef]
- Correia, S.I.; Pereira, H.; Silva-Correia, J.; Van Dijk, C.; Espregueira-Mendes, J.; Oliveira, J.M.; Reis, R. Current concepts: Tissue engineering and regenerative medicine applications in the ankle joint. J. R. Soc. Interface 2014, 11, 20130784. [Google Scholar] [CrossRef]
- Sima, F.; Axente, E.; Ristoscu, C.; Gallet, O.; Anselme, K.; Mihailescu, I. Bioresponsive surfaces and interfaces fabricated by innovative laser approaches. Adv. Mater. Interfaces 2016, 427–462. [Google Scholar]
- Negut, I.; Grumezescu, V.; Sima, L.E.; Axente, E. Recent advances of graphene family nanomaterials for nanomedicine. In Fullerens, Graphenes and Nanotubes; Elsevier: Amsterdam, The Netherlands, 2018; pp. 413–455. [Google Scholar]
- Cohen, D.J.; Nelson, W.J.; Maharbiz, M.M. Galvanotactic control of collective cell migration in epithelial monolayers. Nat. Mater. 2014, 13, 409–417. [Google Scholar] [CrossRef]
- Drevelle, O.; Faucheux, N. Biomimetic materials for controlling bone cell responses. Front. Biosci. 2013, 5, 369–395. [Google Scholar] [CrossRef][Green Version]
- Asri, R.; Harun, W.; Samykano, M.; Lah, N.; Ghani, S.; Tarlochan, F.; Raza, M. Corrosion and surface modification on biocompatible metals: A review. Mater. Sci. Eng. C 2017, 77, 1261–1274. [Google Scholar] [CrossRef]
- Sridhar, T.; Mudali, U.K.; Subbaiyan, M. Preparation and characterisation of electrophoretically deposited hydroxyapatite coatings on type 316l stainless steel. Corros. Sci. 2003, 45, 237–252. [Google Scholar] [CrossRef]
- Robin, A.; Silva, G.; Rosa, J.L. Corrosion behavior of ha-316l ss biocomposites in aqueous solutions. Mater. Res. 2013, 16, 1254–1259. [Google Scholar] [CrossRef]
- Graziani, G.; Bianchi, M.; Sassoni, E.; Russo, A.; Marcacci, M. Ion-substituted calcium phosphate coatings deposited by plasma-assisted techniques: A review. Mater. Sci. Eng. C 2017, 74, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Zhang, J.; Zhang, C.; Barbieri, D.; Yuan, H.; Moroni, L.; Feng, G. The role of calcium phosphate surface structure in osteogenesis and the mechanism involved. Acta Biomater. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Dalbay, M.T.; Luo, X.; Vrij, E.; Barbieri, D.; Moroni, L.; de Bruijn, J.D.; van Blitterswijk, C.A.; Chapple, J.P.; Knight, M.M. Topography of calcium phosphate ceramics regulates primary cilia length and tgf receptor recruitment associated with osteogenesis. Acta Biomater. 2017, 57, 487–497. [Google Scholar] [CrossRef]
- Othman, Z.; Fernandes, H.; Groot, A.J.; Luider, T.M.; Alcinesio, A.; de Melo Pereira, D.; Guttenplan, A.P.; Yuan, H.; Habibovic, P. The role of enpp1/pc-1 in osteoinduction by calcium phosphate ceramics. Biomaterials 2019, 210, 12–24. [Google Scholar] [CrossRef]
- Bohner, M.; Miron, R.J. A proposed mechanism for material-induced heterotopic ossification. Mater. Today 2019, 22, 132–141. [Google Scholar] [CrossRef]
- Leach, J.K.; Whitehead, J. Materials-directed differentiation of mesenchymal stem cells for tissue engineering and regeneration. Acs Biomater. Sci. Eng. 2017, 4, 1115–1127. [Google Scholar] [CrossRef]
- Yeung, W.K.; Reilly, G.C.; Matthews, A.; Yerokhin, A. In vitro biological response of plasma electrolytically oxidized and plasma-sprayed hydroxyapatite coatings on ti–6al–4v alloy. J. Biomed. Mater. Res. Part B Appl. Biomater. 2013, 101, 939–949. [Google Scholar] [CrossRef]
- Ke, D.; Vu, A.A.; Bandyopadhyay, A.; Bose, S. Compositionally graded doped hydroxyapatite coating on titanium using laser and plasma spray deposition for bone implants. Acta Biomater. 2019, 84, 414–423. [Google Scholar] [CrossRef]
- Bose, S.; Fielding, G.; Tarafder, S.; Bandyopadhyay, A. Trace element doping in calcium phosphate ceramics to understand osteogenesis and angiogenesis. Trends Biotechnol. 2013, 31. [Google Scholar] [CrossRef] [PubMed]
- Capuccini, C.; Torricelli, P.; Sima, F.; Boanini, E.; Ristoscu, C.; Bracci, B.; Socol, G.; Fini, M.; Mihailescu, I.; Bigi, A. Strontium-substituted hydroxyapatite coatings synthesized by pulsed-laser deposition: In vitro osteoblast and osteoclast response. Acta Biomater. 2008, 4, 1885–1893. [Google Scholar] [CrossRef]
- Boanini, E.; Torricelli, P.; Fini, M.; Sima, F.; Serban, N.; Mihailescu, I.N.; Bigi, A. Magnesium and strontium doped octacalcium phosphate thin films by matrix assisted pulsed laser evaporation. J. Inorg. Biochem. 2012, 107, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Boanini, E.; Torricelli, P.; Sima, F.; Axente, E.; Fini, M.; Mihailescu, I.N.; Bigi, A. Strontium and zoledronate hydroxyapatites graded composite coatings for bone prostheses. J. Colloid. Interface Sci. 2015, 448, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Roy, M.; Bandyopadhyay, A.; Bose, S. Induction plasma sprayed sr and mg doped nano hydroxyapatite coatings on ti for bone implant. J. Biomed. Mater. Res. Part B Appl. Biomater. 2011, 99, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.-S.; Zhou, W.-S.; He, X.-W.; Liu, W.; Bai, B.-L.; Zhou, Q.; Huang, Z.-L.; Tu, K.-K.; Li, H.; Sun, T. A comparative study of zinc, magnesium, strontium-incorporated hydroxyapatite-coated titanium implants for osseointegration of osteopenic rats. Mater. Sci. Eng. C 2016, 62, 226–232. [Google Scholar] [CrossRef]
- Landi, E.; Logroscino, G.; Proietti, L.; Tampieri, A.; Sandri, M.; Sprio, S. Biomimetic mg-substituted hydroxyapatite: From synthesis to in vivo behaviour. J. Mater. Sci. Mater. Med. 2008, 19, 239–247. [Google Scholar] [CrossRef]
- Huiskes, R.; Weinans, H.; Van Rietbergen, B. The relationship between stress shielding and bone resorption around total hip stems and the effects of flexible materials. Clin. Orthop. Relat. Res. 1992, 124–134. [Google Scholar] [CrossRef]
- Scarisoreanu, N.; Craciun, F.; Ion, V.; Birjega, R.; Bercea, A.; Dinca, V.; Dinescu, M.; Sima, L.; Icriverzi, M.; Roseanu, A. Lead-free piezoelectric (ba, ca)(zr, ti) o3 thin films for biocompatible and flexible devices. ACS Appl. Mater. Interfaces 2017, 9, 266–278. [Google Scholar] [CrossRef]
- Jannasch, M.; Gaetzner, S.; Weigel, T.; Walles, H.; Schmitz, T.; Hansmann, J. A comparative multi-parametric in vitro model identifies the power of test conditions to predict the fibrotic tendency of a biomaterial. Sci. Rep. 2017, 7, 1–17. [Google Scholar] [CrossRef]
- Sima, L.E. Extracellular signals for guiding mesenchymal stem cells osteogenic fate. Curr. Stem Cell Res. Ther. 2017, 12, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, A.; Pelka, S.; Gray, D.S.; Moghe, P.V. Engineering lineage potency and plasticity of stem cells using epigenetic molecules. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Dennis, S.C.; Berkland, C.J.; Bonewald, L.F.; Detamore, M.S. Endochondral ossification for enhancing bone regeneration: Converging native extracellular matrix biomaterials and developmental engineering in vivo. Tissue Eng. Part B Rev. 2015, 21, 247–266. [Google Scholar] [CrossRef]
- Thompson, E.M.; Matsiko, A.; Farrell, E.; Kelly, D.J.; O’Brien, F.J. Recapitulating endochondral ossification: A promising route to in vivo bone regeneration. J. Tiss. Eng. Regen. Med. 2015, 9, 889–902. [Google Scholar] [CrossRef] [PubMed]
- Petersen, A.; Princ, A.; Korus, G.; Ellinghaus, A.; Leemhuis, H.; Herrera, A.; Klaumünzer, A.; Schreivogel, S.; Woloszyk, A.; Schmidt-Bleek, K. A biomaterial with a channel-like pore architecture induces endochondral healing of bone defects. Nat. Commun. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Mikael, P.E.; Golebiowska, A.A.; Xin, X.; Rowe, D.W.; Nukavarapu, S.P. Evaluation of an engineered hybrid matrix for bone regeneration via endochondral ossification. Ann. Biomed. Eng. 2020, 48, 992–1005. [Google Scholar] [CrossRef]
- Timashev, P.; Kuznetsova, D.; Koroleva, A.; Prodanets, N.; Deiwick, A.; Piskun, Y.; Bardakova, K.; Dzhoyashvili, N.; Kostjuk, S.; Zagaynova, E. Novel biodegradable star-shaped polylactide scaffolds for bone regeneration fabricated by two-photon polymerization. Nanomedicine 2016, 11, 1041–1053. [Google Scholar] [CrossRef]
- Won, J.-E.; Yun, Y.-R.; Jang, J.-H.; Yang, S.-H.; Kim, J.-H.; Chrzanowski, W.; Wall, I.B.; Knowles, J.C.; Kim, H.-W. Multifunctional and stable bone mimic proteinaceous matrix for bone tissue engineering. Biomaterials 2015, 56, 46–57. [Google Scholar] [CrossRef]
- Polini, A.; Pisignano, D.; Parodi, M.; Quarto, R.; Scaglione, S. Osteoinduction of human mesenchymal stem cells by bioactive composite scaffolds without supplemental osteogenic growth factors. PLoS ONE 2011, 6, e26211. [Google Scholar] [CrossRef]
- Arafat, M.T.; Lam, C.X.; Ekaputra, A.K.; Wong, S.Y.; Li, X.; Gibson, I. Biomimetic composite coating on rapid prototyped scaffolds for bone tissue engineering. Acta Biomater. 2011, 7, 809–820. [Google Scholar] [CrossRef]
- Ciapetti, G.; Granchi, D.; Devescovi, V.; Baglio, S.R.; Leonardi, E.; Martini, D.; Jurado, M.J.; Olalde, B.; Armentano, I.; Kenny, J.M. Enhancing osteoconduction of plla-based nanocomposite scaffolds for bone regeneration using different biomimetic signals to mscs. Int. J. Mol. Sci. 2012, 13, 2439–2458. [Google Scholar] [CrossRef] [PubMed]
- Sima, F.; Axente, E.; Iordache, I.; Luculescu, C.; Gallet, O.; Anselme, K.; Mihailescu, I. Combinatorial matrix assisted pulsed laser evaporation of a biodegradable polymer and fibronectin for protein immobilization and controlled release. Appl. Surf. Sci. 2014, 306, 75–79. [Google Scholar] [CrossRef]
- Vinson, B.T.; Phamduy, T.B.; Shipman, J.; Riggs, B.; Strong, A.L.; Sklare, S.C.; Murfee, W.L.; Burow, M.E.; Bunnell, B.A.; Huang, Y. Laser direct-write based fabrication of a spatially-defined, biomimetic construct as a potential model for breast cancer cell invasion into adipose tissue. Biofabrication 2017, 9, 025013. [Google Scholar] [CrossRef] [PubMed]
- Coon, M.E.; Stephan, S.B.; Gupta, V.; Kealey, C.P.; Stephan, M.T. Nitinol thin films functionalized with car-t cells for the treatment of solid tumours. Nat. Biomed. Eng. 2020, 4, 195–206. [Google Scholar] [CrossRef]
- Tobin, E.J. Recent coating developments for combination devices in orthopedic and dental applications: A literature review. Adv. Drug Deliv. Rev. 2017, 112, 88–100. [Google Scholar] [CrossRef] [PubMed]
- Nazar, H. Developing biomaterials for tissue engineering and regenerative medicine. Pharm. J. 2013, 291, 223. [Google Scholar]
- Aguado, B.A.; Grim, J.C.; Rosales, A.M.; Watson-Capps, J.J.; Anseth, K.S. Engineering precision biomaterials for personalized medicine. Sci. Transl. Med. 2018, 10, eaam8645. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Zhang, L.; Yang, L.; Zhu, F.; Ding, M.; Lin, F.; Wang, Z.; Li, Y. “Click” chemistry in polymeric scaffolds: Bioactive materials for tissue engineering. J. Control. Release 2018, 273, 160–179. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A. A critical review of decades of research on calcium-phosphate-based coatings: How far are we from their widespread clinical application? Curr. Opin. Biomed. Eng. 2019. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Ivanova, A.A. Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis–a review. Acta Biomater. 2014, 10, 557–579. [Google Scholar] [CrossRef]
- Van Hoof, M.; Wigren, S.; Ivarsson Blechert, J.; Molin, M.; Andersson, H.; Mateijsen, D.; Bom, S.J.; Calmels, M.; van der Rijt, A.J.; Flynn, M.C. A multinational cost-consequence analysis of a bone conduction hearing implant system—a randomized trial of a conventional vs. A less invasive treatment with new abutment technology. Front. Neurol. 2020, 11, 106. [Google Scholar] [CrossRef] [PubMed]
- Wikesjö, U.M.; Susin, C.; Qahash, M.; Polimeni, G.; Leknes, K.N.; Shanaman, R.H.; Prasad, H.S.; Rohrer, M.D.; Hall, J. The critical-size supraalveolar peri-implant defect model: Characteristics and use. J. Clin. Periodontol. 2006, 33, 846–854. [Google Scholar] [CrossRef] [PubMed]
- León, B.; Jansen, J.A. Thin Calcium Phosphate Coatings for Medical Implants. Available online: http://public.eblib.com/EBLPublic/PublicView.do?ptiID=450789 (accessed on 9 May 2020).
- Popescu-Pelin, G.; Sima, F.; Sima, L.; Mihailescu, C.; Luculescu, C.; Iordache, I.; Socol, M.; Socol, G.; Mihailescu, I. Hydroxyapatite thin films grown by pulsed laser deposition and matrix assisted pulsed laser evaporation: Comparative study. Appl. Surf. Sci. 2017, 418, 580–588. [Google Scholar] [CrossRef]
- Shaikh, S.; Kedia, S.; Majumdar, A.G.; Subramanian, M.; Sinha, S. 45s5 bioactive glass coating on ti6al4v alloy using pulsed laser deposition technique. Mater. Res. Express 2020, 6, 125428. [Google Scholar] [CrossRef]
- Socol, G.; Socol, M.; Sima, L.; Petrescu, S.; Enculescu, M.; Sima, F.; Miroiu, M.; Popescu-Pelin, G.; Stefan, N.; Cristescu, R. Combinatorial pulsed laser deposition of ag-containing calcium phosphate coatings. Dig. J. Nanomat. Biostruct. 2012, 7, 563–576. [Google Scholar]
- Mihailescu, I.N.; Bociaga, D.; Socol, G.; Stan, G.E.; Chifiriuc, M.-C.; Bleotu, C.; Husanu, M.A.; Popescu-Pelin, G.; Duta, L.; Luculescu, C.R. Fabrication of antimicrobial silver-doped carbon structures by combinatorial pulsed laser deposition. Int. J. Pharm. 2016, 515, 592–606. [Google Scholar] [CrossRef]
- Icriverzi, M.; Rusen, L.; Brajnicov, S.; Bonciu, A.; Dinescu, M.; Cimpean, A.; Evans, R.W.; Dinca, V.; Roseanu, A. Macrophage in vitro response on hybrid coatings obtained by matrix assisted pulsed laser evaporation. Coatings 2019, 9, 236. [Google Scholar] [CrossRef]
- Negut, I.; Floroian, L.; Ristoscu, C.; Mihailescu, C.N.; Mirza Rosca, J.C.; Tozar, T.; Badea, M.; Grumezescu, V.; Hapenciuc, C.; Mihailescu, I.N. Functional bioglass—biopolymer double nanostructure for natural antimicrobial drug extracts delivery. Nanomaterials 2020, 10, 385. [Google Scholar] [CrossRef]
- Visan, A.; Stan, G.E.; Ristoscu, C.; Popescu-Pelin, G.; Sopronyi, M.; Besleaga, C.; Luculescu, C.; Chifiriuc, M.C.; Hussien, M.; Marsan, O. Combinatorial maple deposition of antimicrobial orthopedic maps fabricated from chitosan and biomimetic apatite powders. Int. J. Pharm. 2016, 511, 505–515. [Google Scholar] [CrossRef]
- Axente, E.; Ristoscu, C.; Bigi, A.; Sima, F.; Mihailescu, I.N. Combinatorial laser synthesis of biomaterial thin films: Selection and processing for medical applications. In Advances in the Application of Lasers in Materials Science; Springer: Berlin, Germany, 2018; pp. 309–338. [Google Scholar]
- Axente, E.; Sima, F. Biomimetic nanostructures with compositional gradient grown by combinatorial matrix-assisted pulsed laser evaporation for tissue engineering. Curr. Med. Chem. 2020, 27, 903–918. [Google Scholar] [CrossRef]
- Mihailescu, N.; Haskoylu, M.E.; Ristoscu, C.; Bostan, M.S.; Sopronyi, M.; Eroğlu, M.S.; Chifiriuc, M.C.; Mustaciosu, C.C.; Axente, E.; Oner, E.T. Gradient multifunctional biopolymer thin film assemblies synthesized by combinatorial maple. Appl. Surf. Sci. 2019, 466, 628–636. [Google Scholar] [CrossRef]
- Sima, L.E.; Chiritoiu, G.; Negut, I.; Grumezescu, V.; Orobeti, S.; Munteanu, C.V.; Sima, F.; Axente, E. Functionalized graphene oxide thin films for anti-tumor drug delivery to melanoma cells. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Hakobyan, D.; Kerouredan, O.; Remy, M.; Dusserre, N.; Medina, C.; Devillard, R.; Fricain, J.-C.; Oliveira, H. Laser-assisted bioprinting for bone repair. In 3d Bioprinting; Springer: Berlin, Germany, 2020; pp. 135–144. [Google Scholar]
- Papavlu, A.P.; Dinca, V.; Dinescu, M. Laser printing of proteins and biomaterials. Laser Print. Funct. Mater. 3d Microfabr. Electron. Biomed. 2018. [Google Scholar]
- Keriquel, V.; Oliveira, H.; Rémy, M.; Ziane, S.; Delmond, S.; Rousseau, B.; Rey, S.; Catros, S.; Amédée, J.; Guillemot, F. In situ printing of mesenchymal stromal cells, by laser-assisted bioprinting, for in vivo bone regeneration applications. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Pichugin, V.F.; Surmenev, R.A.; Shesterikov, E.V.; Ryabtseva, M.; Eshenko, E.; Tverdokhlebov, S.I.; Prymak, O.; Epple, M. The preparation of calcium phosphate coatings on titanium and nickel–titanium by rf-magnetron-sputtered deposition: Composition, structure and micromechanical properties. Surf. Coat. Technol. 2008, 202, 3913–3920. [Google Scholar] [CrossRef]
- Fedotkin, A.; Bolbasov, E.; Kozelskaya, A.; Dubinenko, G.; Shesterikov, E.; Ashrafov, A.; Tverdokhlebov, S. Calcium phosphate coating deposition by radio frequency magnetron sputtering in the various inert gases: The pilot study. Mater. Chem. Phys. 2019, 235, 121735. [Google Scholar] [CrossRef]
- Stan, G.E.; Pasuk, I.; Husanu, M.A.; Enculescu, I.; Pina, S.; Lemos, A.F.; Tulyaganov, D.U.; El Mabrouk, K.; Ferreira, J.M. Highly adherent bioactive glass thin films synthetized by magnetron sputtering at low temperature. J. Mater. Sci. Mater. Med. 2011, 22, 2693–2710. [Google Scholar] [CrossRef] [PubMed]
- Sima, L.E.; Stan, G.E.; Morosanu, C.O.; Melinescu, A.; Ianculescu, A.; Melinte, R.; Neamtu, J.; Petrescu, S.M. Differentiation of mesenchymal stem cells onto highly adherent radio frequency-sputtered carbonated hydroxylapatite thin films. J. Biomed. Mater. Res. Part A 2010, 95, 1203–1214. [Google Scholar] [CrossRef] [PubMed]
- Popa, A.; Stan, G.; Besleaga, C.; Ion, L.; Maraloiu, V.; Tulyaganov, D.; Ferreira, J. Submicrometer hollow bioglass cones deposited by radio frequency magnetron sputtering: Formation mechanism, properties, and prospective biomedical applications. ACS Appl. Mater. Interfaces 2016, 8, 4357–4367. [Google Scholar] [CrossRef] [PubMed]
- Liguori, A.; Gualandi, C.; Focarete, M.L.; Biscarini, F.; Bianchi, M. The pulsed electron deposition technique for biomedical applications: A review. Coatings 2020, 10, 16. [Google Scholar] [CrossRef]
- Unabia, R.B.; Bonebeau, S.; Candidato, R.T., Jr.; Pawłowski, L. Preliminary study on copper-doped hydroxyapatite coatings obtained using solution precursor plasma spray process. Surf. Coat. Technol. 2018, 353, 370–377. [Google Scholar] [CrossRef]
- Vilardell, A.; Cinca, N.; Garcia-Giralt, N.; Dosta, S.; Cano, I.; Nogués, X.; Guilemany, J. In-vitro comparison of hydroxyapatite coatings obtained by cold spray and conventional thermal spray technologies. Mater. Sci. Eng. C 2020, 107, 110306. [Google Scholar] [CrossRef] [PubMed]
- Henao, J.; Poblano-Salas, C.; Monsalve, M.; Corona-Castuera, J.; Barceinas-Sanchez, O. Bio-active glass coatings manufactured by thermal spray: A status report. J. Mater. Res. Technol. 2019. [Google Scholar] [CrossRef]
- Alkekhia, D.; Hammond, P.T.; Shukla, A. Layer-by-layer biomaterials for drug delivery. Annu. Rev. Biomed. Eng. 2020, 22. [Google Scholar] [CrossRef] [PubMed]
- Visan, A.I.; Popescu-Pelin, G.; Gherasim, O.; Mihailescu, A.; Socol, M.; Zgura, I.; Chiritoiu, M.; Elena Sima, L.; Antohe, F.; Ivan, L. Long-term evaluation of dip-coated pcl-blend-peg coatings in simulated conditions. Polymers 2020, 12, 717. [Google Scholar] [CrossRef] [PubMed]
- Brigaud, I.; Agniel, R.; Leroy-Dudal, J.; Kellouche, S.; Ponche, A.; Bouceba, T.; Mihailescu, N.; Sopronyi, M.; Viguier, E.; Ristoscu, C. Synergistic effects of bmp-2, bmp-6 or bmp-7 with human plasma fibronectin onto hydroxyapatite coatings: A comparative study. Acta Biomater. 2017, 55, 481–492. [Google Scholar] [CrossRef]
- Banta, R.A.; Collins, T.W.; Curley, R.; O’Connell, J.; Young, P.W.; Holmes, J.D.; Flynn, E.J. Regulated phase separation in nanopatterned protein-polysaccharide thin films by spin coating. Colloids Surf. B Biointerfaces 2020, 190, 110967. [Google Scholar] [CrossRef]
- Ishikawa, K.; Garskaite, E.; Kareiva, A. Sol–gel synthesis of calcium phosphate-based biomaterials—A review of environmentally benign, simple, and effective synthesis routes. J. Sol Gel Sci. Technol. 2020, 1–22. [Google Scholar] [CrossRef]
- Ballarre, J.; Aydemir, T.; Liverani, L.; Roether, J.; Goldmann, W.; Boccaccini, A. Versatile bioactive and antibacterial coating system based on silica, gentamicin, and chitosan: Improving early stage performance of titanium implants. Surf. Coat. Technol. 2020, 381, 125138. [Google Scholar] [CrossRef]
- Boccaccini, A.R.; Keim, S.; Ma, R.; Li, Y.; Zhitomirsky, I. Electrophoretic deposition of biomaterials. J. R. Soc. Interface R. Soc. 2010, 7 (Suppl. 5), S581–S613. [Google Scholar] [CrossRef]
- Singh, S.; Singh, G.; Bala, N. Electrophoretic deposition of hydroxyapatite-iron oxide-chitosan composite coatings on ti-13nb-13zr alloy for biomedical applications. Thin Solid Film. 2020, 697, 137801. [Google Scholar] [CrossRef]
- Lu, Y.-P.; Li, M.-S.; Li, S.-T.; Wang, Z.-G.; Zhu, R.-F. Plasma-sprayed hydroxyapatite+ titania composite bond coat for hydroxyapatite coating on titanium substrate. Biomaterials 2004, 25, 4393–4403. [Google Scholar] [CrossRef] [PubMed]
- Grubova, I.Y.; Surmeneva, M.A.; Ivanova, A.A.; Kravchuk, K.; Prymak, O.; Epple, M.; Buck, V.; Surmenev, R.A. The effect of patterned titanium substrates on the properties of silver-doped hydroxyapatite coatings. Surf. Coat. Technol. 2015, 276, 595–601. [Google Scholar] [CrossRef]
- Nelea, V.; Ristoscu, C.; Chiritescu, C.; Ghica, C.; Mihailescu, I.; Pelletier, H.; Mille, P.; Cornet, A. Pulsed laser deposition of hydroxyapatite thin films on ti-5al-2.5 fe substrates with and without buffer layers. Appl. Surf. Sci. 2000, 168, 127–131. [Google Scholar] [CrossRef]
- Nelea, V.; Morosanu, C.; Iliescu, M.; Mihailescu, I. Hydroxyapatite thin films grown by pulsed laser deposition and radio-frequency magnetron sputtering: Comparative study. Appl. Surf. Sci. 2004, 228, 346–356. [Google Scholar] [CrossRef]
- Duta, L.; Popescu, A.C. Current status on pulsed laser deposition of coatings from animal-origin calcium phosphate sources. Coatings 2019, 9, 335. [Google Scholar] [CrossRef]
- Garcia-Sanz, F.; Mayor, M.; Arias, J.; Pou, J.; Leon, B.; Perez-Amor, M. Hydroxyapatite coatings: A comparative study between plasma-spray and pulsed laser deposition techniques. J. Mater. Sci. Mater. Med. 1997, 8, 861–865. [Google Scholar] [CrossRef]
- Mohseni, E.; Zalnezhad, E.; Bushroa, A.R. Comparative investigation on the adhesion of hydroxyapatite coating on ti–6al–4v implant: A review paper. Int. J. Adhes. Adhes. 2014, 48, 238–257. [Google Scholar] [CrossRef]
- Smith, J.R.; Lamprou, D.A. Polymer coatings for biomedical applications: A review. Trans. Imf. 2014, 92, 9–19. [Google Scholar] [CrossRef]
- Hallab, N.J.; Bundy, K.J.; O’Connor, K.; Moses, R.L.; Jacobs, J.J. Evaluation of metallic and polymeric biomaterial surface energy and surface roughness characteristics for directed cell adhesion. Tissue Eng. 2001, 7, 55–71. [Google Scholar] [CrossRef]
- Mihailescu, I.N.; Bigi, A.; Gyorgy, E.; Ristoscu, C.; Sima, F.; Oner, E.T. Biomaterial thin films by soft pulsed laser technologies for biomedical applications. In Lasers in Materials Science; Springer: Berlin, Germany, 2014; pp. 271–294. [Google Scholar]
- Sima, F.; Davidson, P.M.; Dentzer, J.; Gadiou, R.; Pauthe, E.; Gallet, O.; Mihailescu, I.N.; Anselme, K. Inorganic–organic thin implant coatings deposited by lasers. ACS Appl. Mater. Interfaces 2015, 7, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Axente, E.; Sima, F.; Ristoscu, C.; Mihailescu, N.; Mihailescu, I.N. Biopolymer thin films synthesized by advanced pulsed laser techniques. Recent Adv. Biopolym. 2016, 73. [Google Scholar]
- Stiff-Roberts, A.D.; Ge, W. Organic/hybrid thin films deposited by matrix-assisted pulsed laser evaporation (maple). Appl. Phys. Rev. 2017, 4, 041303. [Google Scholar] [CrossRef]
- Schmidt, V.; Belegratis, M.R. Introduction and scope of the book. In Laser Technology in Biomimetics; Springer: Berlin, Germany, 2013; pp. 1–12. [Google Scholar]
- Craciun, D.; Craciun, V. Reactive pulsed laser deposition of tin. Appl. Surf. Sci. 1992, 54, 75–77. [Google Scholar] [CrossRef]
- Lunney, J.G. Pulsed laser deposition of metal and metal multilayer films. Appl. Surf. Sci. 1995, 86, 79–85. [Google Scholar] [CrossRef]
- Caricato, A.P.; Luches, A. Applications of the matrix-assisted pulsed laser evaporation method for the deposition of organic, biological and nanoparticle thin films: A review. Appl. Phys. A 2011, 105, 565–582. [Google Scholar] [CrossRef]
- Sima, F.; Mutlu, E.C.; Eroglu, M.S.; Sima, L.E.; Serban, N.; Ristoscu, C.; Petrescu, S.M.; Oner, E.T.; Mihailescu, I.N. Levan nanostructured thin films by maple assembling. Biomacromolecules 2011, 12, 2251–2256. [Google Scholar] [CrossRef]
- Motoc, M.; Axente, E.; Popescu, C.; Sima, L.; Petrescu, S.; Mihailescu, I.; Gyorgy, E. Active protein and calcium hydroxyapatite bilayers grown by laser techniques for therapeutic applications. J. Biomed. Mater. Res. Part A 2013, 101, 2706–2711. [Google Scholar] [CrossRef]
- Dinca, V.; Florian, P.E.; Sima, L.E.; Rusen, L.; Constantinescu, C.; Evans, R.W.; Dinescu, M.; Roseanu, A. Maple-based method to obtain biodegradable hybrid polymeric thin films with embedded antitumoral agents. Biomed. Microdev. 2014, 16, 11–21. [Google Scholar] [CrossRef]
- Sima, F.; Davidson, P.; Pauthe, E.; Sima, L.; Gallet, O.; Mihailescu, I.; Anselme, K. Fibronectin layers by matrix-assisted pulsed laser evaporation from saline buffer-based cryogenic targets. Acta Biomater. 2011, 7, 3780–3788. [Google Scholar] [CrossRef]
- Sima, F.; Davidson, P.; Pauthe, E.; Gallet, O.; Anselme, K.; Mihailescu, I. Thin films of vitronectin transferred by maple. Appl. Phys. A 2011, 105, 611–617. [Google Scholar] [CrossRef]
- McGill, R.A.; Chrisey, D.B. Method of Producing a Film Coating by Matrix Assisted Pulsed Laser Deposition. U.S. Patent 6,025,036, 15 February 2000. [Google Scholar]
- Jelinek, M.; Kocourek, T.; Remsa, J.; Cristescu, R.; Mihailescu, I.; Chrisey, D. Maple applications in studying organic thin films. Laser Phys. 2007, 17, 66–70. [Google Scholar] [CrossRef]
- Califano, V.; Bloisi, F.; Vicari, L.R.; Colombi, P.; Bontempi, E.; Depero, L.E. Maple deposition of biomaterial multilayers. Appl. Surf. Sci. 2008, 254, 7143–7148. [Google Scholar] [CrossRef]
- Stamatin, L.; Cristescu, R.; Socol, G.; Moldovan, A.; Mihaiescu, D.; Stamatin, I.; Mihailescu, I.; Chrisey, D. Laser deposition of fibrinogen blood proteins thin films by matrix assisted pulsed laser evaporation. Appl. Surf. Sci. 2005, 248, 422–427. [Google Scholar] [CrossRef]
- Guha, S.; Adil, D.; Ukah, N.; Gupta, R.; Ghosh, K. Maple-deposited polymer films for improved organic device performance. Appl. Phys. A 2011, 105, 547–554. [Google Scholar] [CrossRef]
- Axente, E.; Sopronyi, M.; Ghimbeu, C.M.; Nita, C.; Airoudj, A.; Schrodj, G.; Sima, F. Matrix-assisted pulsed laser evaporation: A novel approach to design mesoporous carbon films. Carbon 2017, 122, 484–495. [Google Scholar] [CrossRef]
- Del Pino, Á.P.; Ramadan, M.A.; Lebière, P.G.; Ivan, R.; Logofatu, C.; Yousef, I.; György, E. Fabrication of graphene-based electrochemical capacitors through reactive inverse matrix assisted pulsed laser evaporation. Appl. Surf. Sci. 2019, 484, 245–256. [Google Scholar] [CrossRef]
- Del Pino, Á.P.; López, M.R.; Ramadan, M.A.; Lebière, P.G.; Logofatu, C.; Martínez-Rovira, I.; Yousef, I.; György, E. Enhancement of the supercapacitive properties of laser deposited graphene-based electrodes through carbon nanotube loading and nitrogen doping. Phys. Chem. Chem. Phys. 2019, 21, 25175–25186. [Google Scholar] [CrossRef]
- Palla-Papavlu, A.; Constantinescu, C.; Dinca, V.; Matei, A.; Moldovan, A.; Mitu, B.; Dinescu, M. Polyisobutylene thin films obtained by matrix assisted pulsed laser evaporation for sensors applications. Sens. Lett. 2010, 8, 502–506. [Google Scholar] [CrossRef]
- Palla-Papavlu, A.; Dinca, V.; Dinescu, M.; Di Pietrantonio, F.; Cannatà, D.; Benetti, M.; Verona, E. Matrix-assisted pulsed laser evaporation of chemoselective polymers. Appl. Phys. A 2011, 105, 651–659. [Google Scholar] [CrossRef]
- Dinca, V.; Viespe, C.; Brajnicov, S.; Constantinoiu, I.; Moldovan, A.; Bonciu, A.; Toader, C.N.; Ginghina, R.E.; Grigoriu, N.; Dinescu, M. Maple assembled acetylcholinesterase–polyethylenimine hybrid and multilayered interfaces for toxic gases detection. Sensors 2018, 18, 4265. [Google Scholar] [CrossRef] [PubMed]
- Caricato, A.P.; Ge, W.; Stiff-Roberts, A.D. Uv-and rir-maple: Fundamentals and applications. In Advances in the Application of Lasers in Materials Science; Springer: Berlin/Heidelberg, Germany, 2018; pp. 275–308. [Google Scholar]
- Smausz, T.; Megyeri, G.; Kékesi, R.; Vass, C.; György, E.; Sima, F.; Mihailescu, I.N.; Hopp, B. Comparative study on pulsed laser deposition and matrix assisted pulsed laser evaporation of urease thin films. Thin Solid Film 2009, 517, 4299–4302. [Google Scholar] [CrossRef]
- György, E.; Sima, F.; Mihailescu, I.; Smausz, T.; Megyeri, G.; Kékesi, R.; Hopp, B.; Zdrentu, L.; Petrescu, S. Immobilization of urease by laser techniques: Synthesis and application to urea biosensors. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2009, 89, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Predoi, D.; Ciobanu, C.S.; Radu, M.; Costache, M.; Dinischiotu, A.; Popescu, C.; Axente, E.; Mihailescu, I.; Gyorgy, E. Hybrid dextran-iron oxide thin films deposited by laser techniques for biomedical applications. Mater. Sci. Eng. C 2012, 32, 296–302. [Google Scholar] [CrossRef]
- Axente, E.; Sima, F.; Sima, L.E.; Erginer, M.; Eroglu, M.S.; Serban, N.; Ristoscu, C.; Petrescu, S.M.; Oner, E.T.; Mihailescu, I.N. Combinatorial maple gradient thin film assemblies signalling to human osteoblasts. Biofabrication 2014, 6, 035010. [Google Scholar] [CrossRef] [PubMed]
- Miroiu, F.; Socol, G.; Visan, A.; Stefan, N.; Craciun, D.; Craciun, V.; Dorcioman, G.; Mihailescu, I.; Sima, L.; Petrescu, S. Composite biocompatible hydroxyapatite–silk fibroin coatings for medical implants obtained by matrix assisted pulsed laser evaporation. Mater. Sci. Eng. B 2010, 169, 151–158. [Google Scholar] [CrossRef]
- Rașoga, O.; Sima, L.; Chirițoiu, M.; Popescu-Pelin, G.; Fufă, O.; Grumezescu, V.; Socol, M.; Stănculescu, A.; Zgură, I.; Socol, G. Biocomposite coatings based on poly (3-hydroxybutyrate-co-3-hydroxyvalerate)/calcium phosphates obtained by maple for bone tissue engineering. Appl. Surf. Sci. 2017, 417, 204–212. [Google Scholar] [CrossRef]
- Cristescu, R.; Popescu, C.; Socol, G.; Iordache, I.; Mihailescu, I.; Mihaiescu, D.; Grumezescu, A.; Balan, A.; Stamatin, I.; Chifiriuc, C. Magnetic core/shell nanoparticle thin films deposited by maple: Investigation by chemical, morphological and in vitro biological assays. Appl. Surf. Sci. 2012, 258, 9250–9255. [Google Scholar] [CrossRef]
- Grumezescu, A.M.; Cristescu, R.; Chifiriuc, M.; Dorcioman, G.; Socol, G.; Mihailescu, I.; Mihaiescu, D.E.; Ficai, A.; Vasile, O.; Enculescu, M. Fabrication of magnetite-based core–shell coated nanoparticles with antibacterial properties. Biofabrication 2015, 7, 015014. [Google Scholar] [CrossRef]
- Dinca, V.; Liu, Q.; Brajnicov, S.; Bonciu, A.; Vlad, A.; Dinu, C.Z. Composites formed from tungsten trioxide and graphene oxide for the next generation of electrochromic interfaces. Compos. Commun. 2020, 17, 115–122. [Google Scholar] [CrossRef]
- Greer, J.A. Design challenges for matrix assisted pulsed laser evaporation and infrared resonant laser evaporation equipment. Appl. Phys. A 2011, 105, 661–671. [Google Scholar] [CrossRef]
- Bubb, D.; O’Malley, S.; Antonacci, C.; Simonson, D.; McGill, R. Matrix-assisted laser deposition of a sorbent oligomer using an infrared laser. J. Appl. Phys. 2004, 95, 2175–2177. [Google Scholar] [CrossRef]
- Pate, R.; McCormick, R.; Chen, L.; Zhou, W.; Stiff-Roberts, A.D. Rir-maple deposition of conjugated polymers for application to optoelectronic devices. Appl. Phys. A 2011, 105, 555–563. [Google Scholar] [CrossRef]
- Piqué, A. The matrix-assisted pulsed laser evaporation (maple) process: Origins and future directions. Appl. Phys. A 2011, 105, 517–528. [Google Scholar] [CrossRef]
- Zhigilei, L.V.; Leveugle, E.; Garrison, B.J.; Yingling, Y.G.; Zeifman, M.I. Computer simulations of laser ablation of molecular substrates. Chem. Rev. 2003, 103, 321–348. [Google Scholar] [CrossRef] [PubMed]
- Leveugle, E.; Zhigilei, L.V. Molecular dynamics simulation study of the ejection and transport of polymer molecules in matrix-assisted pulsed laser evaporation. J. Appl. Phys. 2007, 102, 074914. [Google Scholar] [CrossRef]
- Tabetah, M.; Matei, A.; Constantinescu, C.; Mortensen, N.P.; Dinescu, M.; Schou, J.; Zhigilei, L.V. The minimum amount of “matrix” needed for matrix-assisted pulsed laser deposition of biomolecules. J. Phys. Chem. B 2014, 118, 13290–13299. [Google Scholar] [CrossRef]
- Yingling, Y.G.; Garrison, B.J. Coarse-grained chemical reaction model. J. Phys. Chem. B 2004, 108, 1815–1821. [Google Scholar] [CrossRef]
- Torres, R.D.; Johnson, S.L.; Haglund, R.F.; Hwang, J.; Burn, P.L.; Holloway, P.H. Mechanisms of resonant infrared matrix-assisted pulsed laser evaporation. Crit. Rev. Solid State Mater. Sci. 2011, 36, 16–45. [Google Scholar] [CrossRef]
- Sima, F.; Axente, E.; Sima, L.E.; Tuyel, U.; Eroglu, M.S.; Serban, N.; Ristoscu, C.; Petrescu, S.M.; Oner, E.T.; Mihailescu, I.N. Combinatorial matrix-assisted pulsed laser evaporation: Single-step synthesis of biopolymer compositional gradient thin film assemblies. Appl. Phys. Lett. 2012, 101, 233705. [Google Scholar] [CrossRef]
- Takeuchi, I. Combinatorial pulsed laser deposition. In Pulsed Laser Deposition of Thin Films: Applications-Led Growth of Functional Materials; John Wiley Sons Inc.: Hoboken, NJ, USA, 2007; pp. 161–175. [Google Scholar]
- Boanini, E.; Torricelli, P.; Sima, F.; Axente, E.; Fini, M.; Mihailescu, I.N.; Bigi, A. Gradient coatings of strontium hydroxyapatite/zinc β-tricalcium phosphate as a tool to modulate osteoblast/osteoclast response. J. Inorg. Biochem. 2018, 183, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xing, H.; Zhao, B.; Wang, Y.; Zhang, X.; Ren, Y.; Yan, N.; Gao, T.; Li, J.; Zhang, L.; Wang, H. Rapid construction of fe–co–ni composition-phase map by combinatorial materials chip approach. ACS Comb. Sci. 2018, 20, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Motemani, Y.; Khare, C.; Savan, A.; Hans, M.; Paulsen, A.; Frenzel, J.; Somsen, C.; Mücklich, F.; Eggeler, G.; Ludwig, A. Nanostructured ti–ta thin films synthesized by combinatorial glancing angle sputter deposition. Nanotechnology 2016, 27, 495604. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Sask, K.; Brash, J.; Zhitomirsky, I. Surface modifications of nitinol for biomedical applications. Colloids Surf. B Biointerfaces 2008, 67, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Hoogenboom, R.; Meier, M.A.; Schubert, U.S. Combinatorial methods, automated synthesis and high-throughput screening in polymer research: Past and present. Macromol. Rapid Commun. 2003, 24, 15–32. [Google Scholar] [CrossRef]
- Meredith, J.C.; Smith, A.P.; Karim, A.; Amis, E.J. Combinatorial materials science for polymer thin-film dewetting. Macromolecules 2000, 33, 9747–9756. [Google Scholar] [CrossRef]
- Boanini, E.; Gazzano, M.; Bigi, A. Ionic substitutions in calcium phosphates synthesized at low temperature. Acta Biomater. 2010, 6, 1882–1894. [Google Scholar] [CrossRef]
- Arcos, D.; Vallet-Regí, M. Substituted hydroxyapatite coatings of bone implants. J. Mater. Chem. B 2020, 8, 1781–1800. [Google Scholar] [CrossRef]
- Marie, P. Strontium ranelate: A novel mode of action optimizing bone formation and resorption. Osteoporos. Int. 2005, 16, S7–S10. [Google Scholar] [CrossRef]
- Gallacher, S.; Dixon, T. Impact of treatments for postmenopausal osteoporosis (bisphosphonates, parathyroid hormone, strontium ranelate, and denosumab) on bone quality: A systematic review. Calcif. Tissue Int. 2010, 87, 469–484. [Google Scholar] [CrossRef]
- Lynch, R.J. Zinc in the mouth, its interactions with dental enamel and possible effects on caries; a review of the literature. Int. Dent. J. 2011, 61, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Bostan, M.S.; Senol, M.; Cig, T.; Peker, I.; Goren, A.C.; Ozturk, T.; Eroglu, M.S. Controlled release of 5-aminosalicylicacid from chitosan based ph and temperature sensitive hydrogels. Int. J. Biol. Macromol. 2013, 52, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Chou, J.; Hao, J.; Hatoyama, H.; Ben-Nissan, B.; Milthorpe, B.; Otsuka, M. Effect of biomimetic zinc-containing tricalcium phosphate (zn–tcp) on the growth and osteogenic differentiation of mesenchymal stem cells. J. Tissue Eng. Regen. Med. 2015, 9, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Öner, E.T.; Hernández, L.; Combie, J. Review of levan polysaccharide: From a century of past experiences to future prospects. Biotechnol. Adv. 2016, 34, 827–844. [Google Scholar] [CrossRef]
- Berg, A.; Oner, E.T.; Combie, J.; Schneider, B.; Ellinger, R.; Weisser, J.; Wyrwa, R.; Schnabelrauch, M. Formation of new, cytocompatible hydrogels based on photochemically crosslinkable levan methacrylates. Int. J. Biol. Macromol. 2018, 107, 2312–2319. [Google Scholar] [CrossRef]
- Demirci, T.; Hasköylü, M.E.; Eroğlu, M.S.; Hemberger, J.; Öner, E.T. Levan-based hydrogels for controlled release of amphotericin b for dermal local antifungal therapy of candidiasis. Eur. J. Pharm. Sci. 2020, 145, 105255. [Google Scholar] [CrossRef]
- De Siqueira, E.C.; de Souza Rebouças, J.; Pinheiro, I.O.; Formiga, F.R. Levan-based nanostructured systems: An overview. Int. J. Pharm. 2020, 580, 119242. [Google Scholar] [CrossRef]
- Erginer, M.; Akcay, A.; Coskunkan, B.; Morova, T.; Rende, D.; Bucak, S.; Baysal, N.; Ozisik, R.; Eroglu, M.S.; Agirbasli, M. Sulfated levan from halomonas smyrnensis as a bioactive, heparin-mimetic glycan for cardiac tissue engineering applications. Carbohydr. Polym. 2016, 149, 289–296. [Google Scholar] [CrossRef]
- Gomes, T.D.; Caridade, S.G.; Sousa, M.P.; Azevedo, S.; Kandur, M.Y.; Öner, E.T.; Alves, N.M.; Mano, J.F. Adhesive free-standing multilayer films containing sulfated levan for biomedical applications. Acta Biomater. 2018, 69, 183–195. [Google Scholar] [CrossRef]
- Francesko, A.; Tzanov, T. Chitin, chitosan and derivatives for wound healing and tissue engineering. In Biofunctionalization of Polymers and their Applications; Springer: Berlin, Germany, 2010; pp. 1–27. [Google Scholar]
- Elieh-Ali-Komi, D.; Hamblin, M.R. Chitin and chitosan: Production and application of versatile biomedical nanomaterials. Int. J. Adv. Res. 2016, 4, 411. [Google Scholar]
- Poth, N.; Seiffart, V.; Gross, G.; Menzel, H.; Dempwolf, W. Biodegradable chitosan nanoparticle coatings on titanium for the delivery of bmp-2. Biomolecules 2015, 5, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Ferreira, J.; Kannan, S. Mechanically stable antimicrobial chitosan–pva–silver nanocomposite coatings deposited on titanium implants. Carbohydr. Polym. 2015, 121, 37–48. [Google Scholar] [CrossRef]
- Bernkop-Schnürch, A.; Dünnhaupt, S. Chitosan-based drug delivery systems. Eur. J. Pharm. Biopharm. 2012, 81, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, Y.; Liang, R.; An, X.; Wang, K.; Shen, G.; Tu, Y.; Zhu, J.; Tao, J. Recent advances in targeted nanoparticles drug delivery to melanoma. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 769–794. [Google Scholar] [CrossRef] [PubMed]
- Guadagnini, R.; Halamoda Kenzaoui, B.; Walker, L.; Pojana, G.; Magdolenova, Z.; Bilanicova, D.; Saunders, M.; Juillerat-Jeanneret, L.; Marcomini, A.; Huk, A. Toxicity screenings of nanomaterials: Challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology 2015, 9, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W. Mutations of the braf gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef]
- Huang, T.; Karsy, M.; Zhuge, J.; Zhong, M.; Liu, D. B-raf and the inhibitors: From bench to bedside. J. Hematol. Oncol. 2013, 6, 30. [Google Scholar] [CrossRef]
- Maertens, O.; Kuzmickas, R.; Manchester, H.E.; Emerson, C.E.; Gavin, A.G.; Guild, C.J.; Wong, T.C.; De Raedt, T.; Bowman-Colin, C.; Hatchi, E. Mapk pathway suppression unmasks latent DNA repair defects and confers a chemical synthetic vulnerability in braf-, nras-, and nf1-mutant melanomas. Cancer Discov. 2019, 9, 526–545. [Google Scholar] [CrossRef]

| Deposition Method | Advantages | Drawbacks | Bio-Coating Materials/Application |
|---|---|---|---|
| Laser Techniques | |||
| Pulsed laser deposition (PLD) | Stoichiometric and adherent coatings, morphology control, easy to obtain multilayered thin films, good versatility of experimental design, thickness control. | Limited to inorganic coatings, small area covering (few cm), high costs. Micrometer-sized droplets and particulates on surfaces. | Inorganic coatings: HA [54,55], and Bioglass (BG) [56]/Implant devices. |
| Combinatorial-Pulsed laser deposition (C-PLD) | Preserve the properties of PLD, synthesis of combinatorial libraries of thin films, controlled doping of coatings, cover larger substrates (glass slide). | Limited to inorganic coatings, high costs. | Inorganic coatings: Ag-doped HA [57], Ag-doped Carbon [58]/Model surfaces. |
| Matrix assisted pulsed laser evaporation (MAPLE) | Applied to both organic and inorganic coatings, multilayers and multistructures, nanoparticulate films, thickness control. | Generation of micrometer-sized droplets and particulates on surfaces, small covering areas. | Inorganic, organic, hybrid coatings: HA [55], HA/Lactoferrin/polyethylene glycol-polycaprolactone copolymer [59], hybrid BG-biopolymer [60]/Implant devices, drug delivery. |
| Combinatorial-Matrix assisted pulsed laser evaporation (C-MAPLE) | Preserve the properties of MAPLE, single-step synthesis of combinatorial bio-coatings, suitable for organic, inorganic and composites, cover larger substrates (glass slide). | Generation of micrometer-sized droplets and particulates on surfaces, high costs. | Inorganic, organic, hybrid coatings: Chitosan/ bio-mimetic apatite [61], CaPs, biopolymers [62,63] Levan and Chitosan blends [64], Graphene Oxide, BSA protein, drugs [65]/Implant devices, drug delivery, model surfaces. |
| Laser-induced forward transfer (LIFT) | Micro-patterns with high spatial resolution, size and separation distance between structures, easy control. | Limited to patterns, difficulties for large area micro-fabrication, difficult to control the height of the patterns. | Inorganic, organic, hybrid micropatterns: collagen and nanoHA [66], proteins and biomaterials [67], mesenchymal stromal cells [68]/Implant devices, drug delivery, model surfaces. |
| Non-Laser Techniques | |||
| Radio frequency magnetron sputtering (RF-MS) | Stoichiometric transfer, uniform dense coatings, good versatility of experimental design. | Limited to inorganic coatings, amorphous coatings, rather expensive. | Inorganic coatings: CaP [69,70], BG [71,72,73]/Implant devices. |
| Pulsed electron deposition (PED) | Stoichiometric and adherent thin films, low cost compared to other PVD techniques. | Limited to inorganic coatings, small area covering. | Inorganic coatings: HA, CaP, biogenic CaP, BG [74]/Implant devices. |
| Plasma spray (PS) | Simplest, operates at low costs. | Use for inorganic coatings only, poor coating adhesion, weak bonding strength at ceramic-metal interface. | Inorganic coatings: HA [75,76], and BG [77]/Implant devices. |
| Layer-by-layer (LBL) | Good thickness control, viscoelasticity/bioactivity, coat multiple substrates of all scales. | Poor layers adhesion to substrates, difficult to create multilayers due to solvent issues, difficult to generate gradient coatings. | Organic coatings: biomaterials for drug delivery [78]/Drug delivery, model surfaces. |
| Langmuir-Blodgett dip coating | Possibility to assemble monolayers, high spatial coverage. | Limited to very thin films, typically used for organic coatings only. | Organic coatings: PCL-Blend-PEG [79]/Drug delivery, model surfaces. |
| Adsorption on surface | Simple, rapid and inexpensive, useful for organic bio-coatings. | Poor adhesion on substrates, poor uniformity, difficult to create multi-layer assembling. | Organic coatings: BMP-2, BMP-6, BMP-7 with fibronectin onto HA coatings [80]/Implant devices, drug delivery. |
| Spin coating | Simple and inexpensive, uniform coatings, accurate thickness control, high spatial coverage. | Use for 2D surfaces only, typically used for organic coatings, solvent issue in case of multilayers, poor adherence. | Organic coatings: protein-polysaccharide thin films [81]/Implant devices, drug delivery. |
| Sol-gel (SG) | Available for both organic and inorganic coatings, simple operation, high versatility. | Poor adhesion to substrates, difficult to create multilayers due to solvent issues, difficult to generate gradient coatings. | Inorganic, organic, hybrid coatings: HA, BG [82], Gentamicin/Chitosan/BG composite [83]/Implant devices, drug delivery. |
| Electrophoretic deposition (EPD) | Useful for both organic and inorganic coatings, simple processing setup. | Difficult to create multilayers due to solvent issues, poor coating adhesion. | Inorganic, organic, hybrid coatings: HA–iron oxide–chitosan composite [84,85]/Implant devices, drug delivery. |
| Cells | Malignancy Phenotype | Mutation Status | Pigmentation |
|---|---|---|---|
| MNT-1 | Primary melanoma | BRAF V600E | Highly pigmented |
| SKmel28 | Primary melanoma | BRAF V600E | Amelanotic |
| MelJuSo | Primary melanoma | BRAF wt; N-Ras Q61K | Amelanotic |
| A375 | Metastatic melanoma | BRAF V600E | Amelanotic |
| SKmel23 | Metastatic melanoma | BRAF wt | Pigmented |
| NHEM | Normal primary melanocytes | - | Pigmented |
| HDF | Normal dermal fibroblasts | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Axente, E.; Elena Sima, L.; Sima, F. Biomimetic Coatings Obtained by Combinatorial Laser Technologies. Coatings 2020, 10, 463. https://doi.org/10.3390/coatings10050463
Axente E, Elena Sima L, Sima F. Biomimetic Coatings Obtained by Combinatorial Laser Technologies. Coatings. 2020; 10(5):463. https://doi.org/10.3390/coatings10050463
Chicago/Turabian StyleAxente, Emanuel, Livia Elena Sima, and Felix Sima. 2020. "Biomimetic Coatings Obtained by Combinatorial Laser Technologies" Coatings 10, no. 5: 463. https://doi.org/10.3390/coatings10050463
APA StyleAxente, E., Elena Sima, L., & Sima, F. (2020). Biomimetic Coatings Obtained by Combinatorial Laser Technologies. Coatings, 10(5), 463. https://doi.org/10.3390/coatings10050463







