Corrosion Protection of A36 Steel with SnO2 Nanoparticles Integrated into SiO2 Coatings
Abstract
1. Introduction
2. Materials and Methods
2.1. SnO2 Nanoparticles Preparation
2.2. SiO2 Sol-Gel Synthesis
2.3. Steel Substrate Preparation
2.4. Coatings
2.5. Characterization Methods
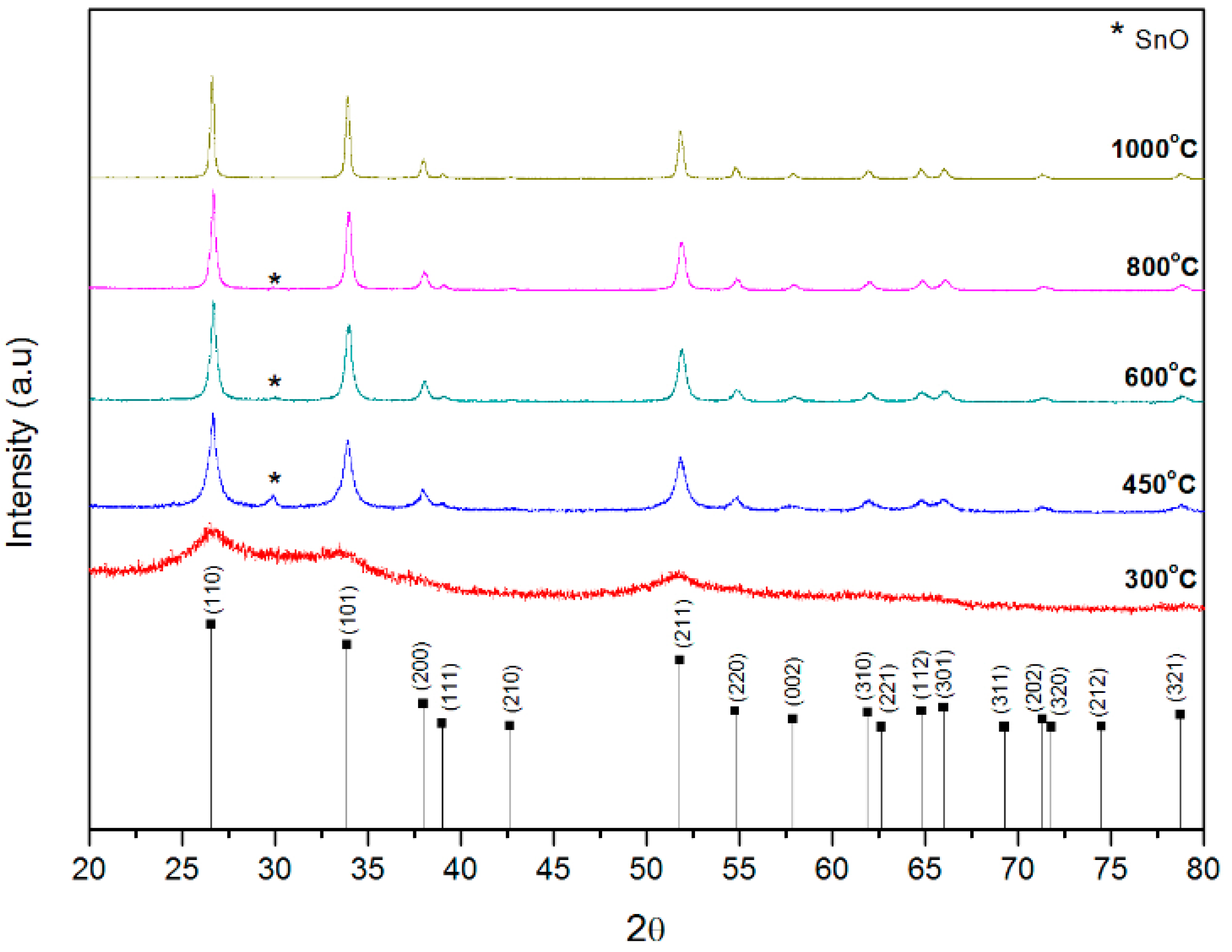
2.5.1. X-ray Diffraction and Scanning Electron Microscopy
2.5.2. Electrochemical Measurements
3. Results and Discussion
3.1. Structure of SnO2 Particles
3.2. Morphological Study of SnO2 Particles
3.3. Surface Morphology of SiO2 Coating with SnO2 Nanoparticles
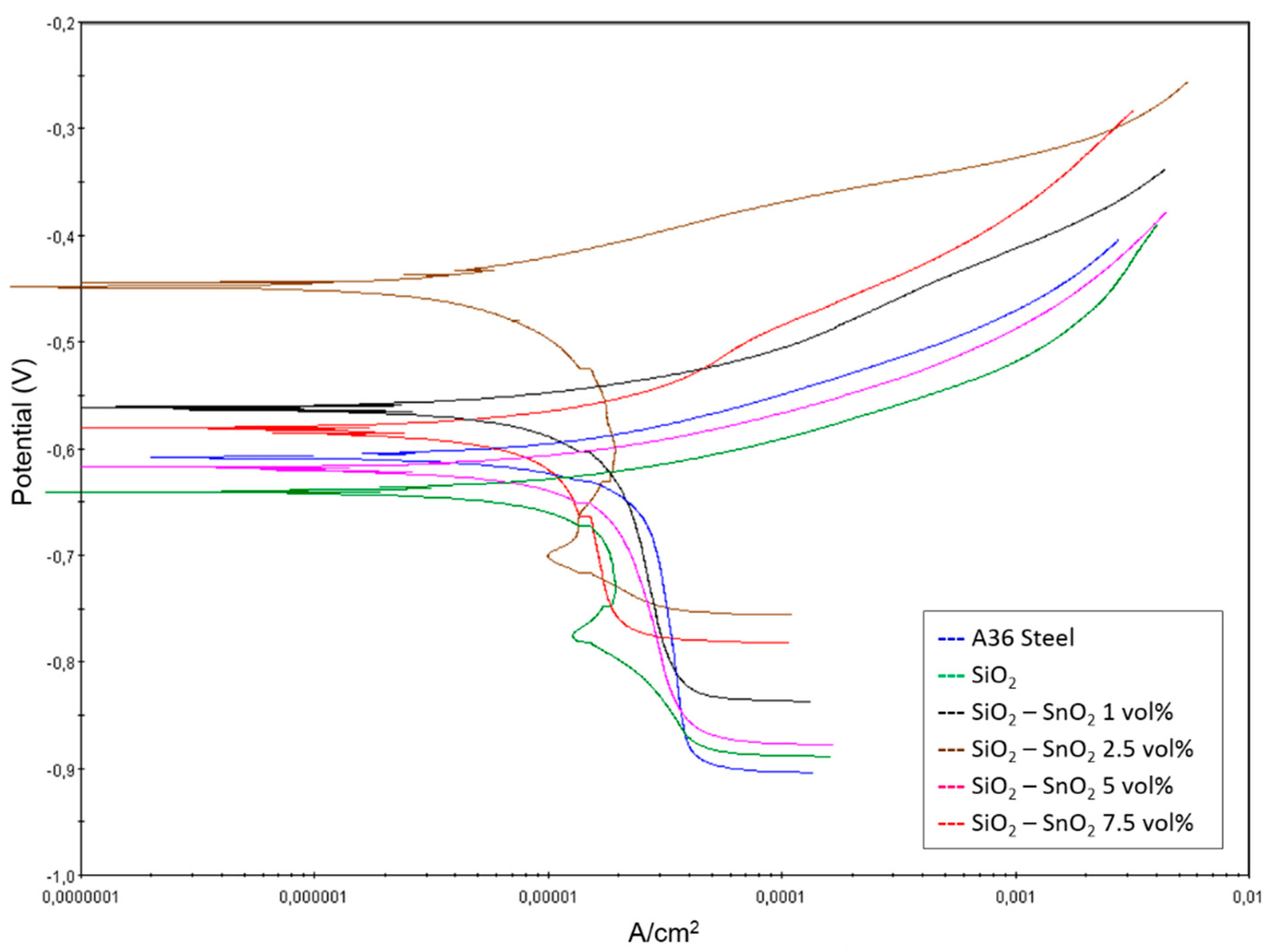
3.4. Corrosion Behaviour of SiO2 Coatings with SnO2 Nanoparticles
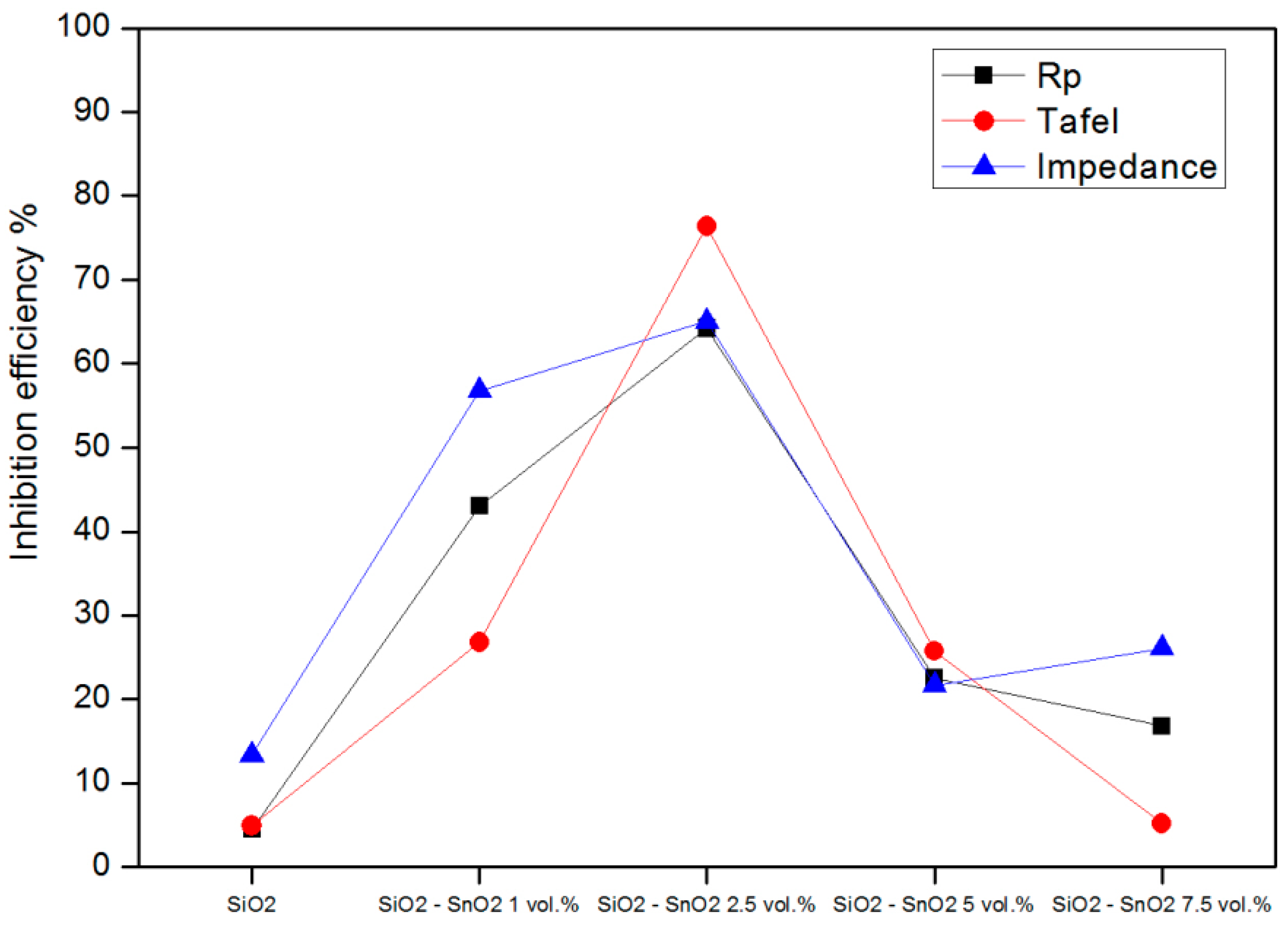
3.5. Polarization Resistance
3.6. Tafel Analysis
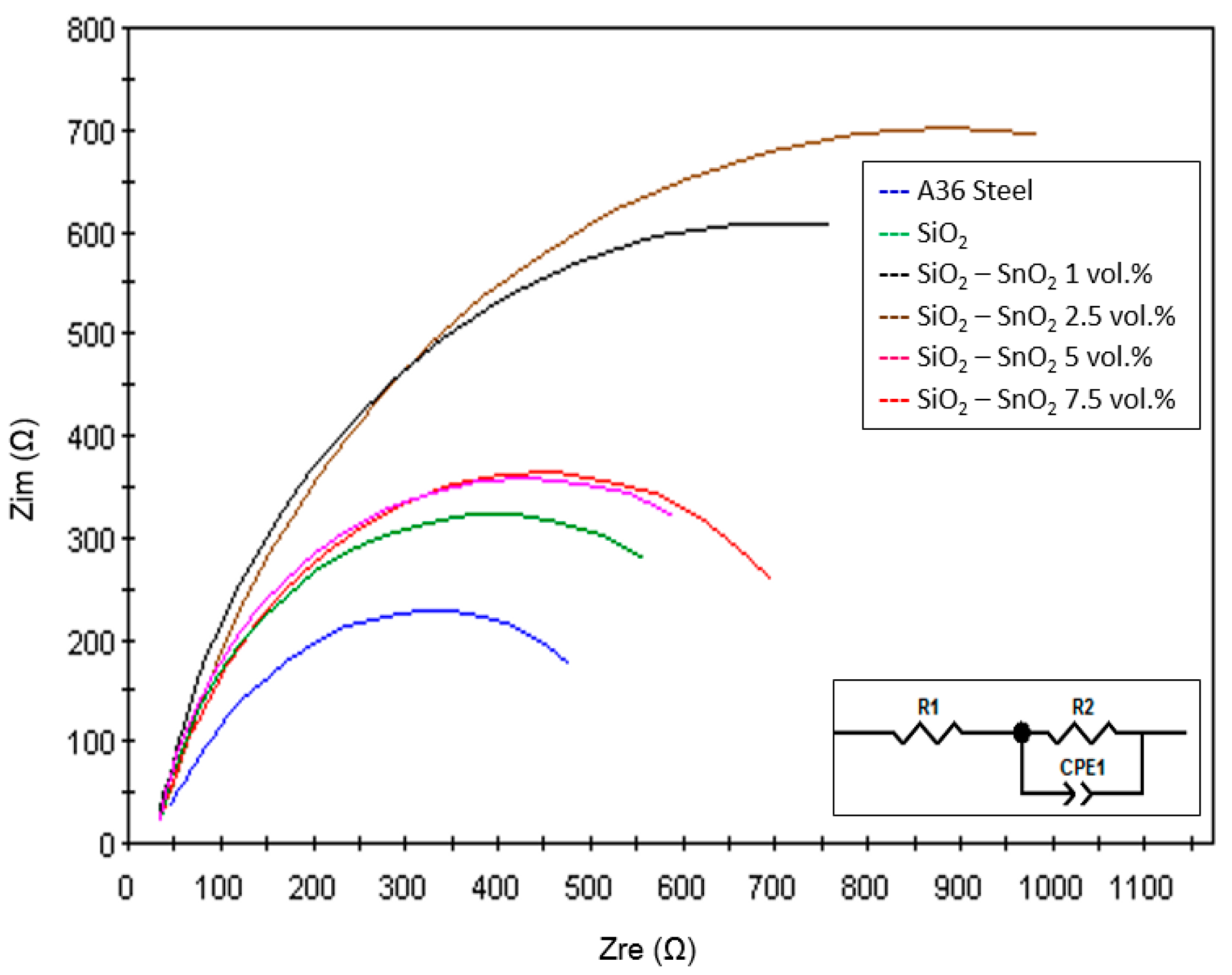
3.7. Impedance
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, M.; Jin, Z.-X.; Zhang, W.; Cao, Y.-Q.; Li, W.-M.; Wu, D.; Li, A.-D. Comparison of chemical stability and corrosion resistance of group IV metal oxide films formed by thermal and plasma-enhanced atomic layer deposition. Sci. Rep. 2019, 9, 10438–10449. [Google Scholar] [CrossRef] [PubMed]
- Gregory, O.J.; Luo, Q.; Crisman, E.E. High temperature stability of indium tin oxide thin films. Thin Solid Film 2002, 406, 286–293. [Google Scholar] [CrossRef]
- Curkovic, L.; Curkovic, H.O.; Salopek, S.; Renjo, M.M.; Segota, S. Enhancement of corrosion protection of AISI 304 stainless steel by nanostructured sol-gel TiO2 films. Corros. Sci. 2013, 77, 176–184. [Google Scholar] [CrossRef]
- Li, S.; Wang, Q.; Chen, T.; Zhou, Z.; Wang, Y.; Fu, J. Study on cerium-doped nano-TiO2 coatings for corrosion protection of 316 L stainless Steel. Nanoscale Res. Lett. 2012, 7, 1–9. [Google Scholar] [CrossRef]
- Xu, H.; Liu, W.; Cao, L.; Su, G.; Duan, R. Preparation of porous TiO2/ZnO composite film and its photocathodic protection properties for 304 stainless Steel. Appl. Surf. Sci. 2014, 301, 508–514. [Google Scholar] [CrossRef]
- Subasri, R.; Shinohara, T. The applicability of SnO2 coatings for corrosion protection of metals. Electrochem. Solid State Lett. 2004, 7, B17–B20. [Google Scholar] [CrossRef]
- Yang, L.; Qin, Z.; Pan, H.; Yun, H.; Min, Y.; Xu, Q. Corrosion protection of 304 stainless steel bipolar plates of PEMFC by coatung SnO2 film. Electrochem. Sci. 2017, 12, 10946–10957. [Google Scholar] [CrossRef]
- Atik, M.; De Lima Neto, P.; Avaca, L.A.; Aegerter, M.A.; Zarzycki, J. Protection of 316L stainless steel against corrosion by SiO2 coatings. Mater. Sci. Lett. 1994, 13, 1081–1085. [Google Scholar] [CrossRef]
- Nouri, E.; Shahmiri, M.; Rezaie, H.R.; Talayian, F. Investigation of structural evolution and electrochemical behaviour of zirconia thin films on the 316L stainless steel substrate formed via sol-gel process. Surf. Coat. Technol. 2011, 205, 5109–5115. [Google Scholar] [CrossRef]
- Norouzi, M.; Garekani, A.A. Corrosion protection by zirconia-based thin films deposited by a sol-gel spin coating method. Ceram. Int. 2014, 40, 2857–2861. [Google Scholar] [CrossRef]
- Chang, S.-H.; Chen, J.-Z.; Hsiao, S.-H.; Lin, G.-W. Nanohardness, corrosion and protein adsorption properties of CuAlO2 films deposited on 316L stainless steel for biomedical applications. Appl. Surf. Sci. 2014, 289, 455–461. [Google Scholar] [CrossRef]
- Magaña, C.R.; Ángeles, M.E.; Rodríguez, F.J. Comportamiento de películas delgadas de níquel y óxido de níquel en NaCl al 3%. Revista de Metalurgia 2006, 1, 49–55. [Google Scholar]
- Dhoke, S.K.; Khanna, A.; Sinha, T.J.M. Effect of nano-ZnO particles on the corrosion behaviorof alkyd-based waterborne coatings. Prog. Org. Coat. 2009, 64, 371–382. [Google Scholar] [CrossRef]
- Li, H.; Liang, K.; Mei, L.; Gu, S.; Wang, S. Corrosion protection of mild steel by zirconia sol-gel coatings. Mater. Sci. Lett. 2001, 20, 1081–1083. [Google Scholar] [CrossRef]
- Yu, D.; Tian, J.; Dai, J.; Wang, X. Corrosion resistance of three-layer superhydrophobic composite coating on carbon steel in seawater. Electrochim. Acta 2013, 97, 409–419. [Google Scholar] [CrossRef]
- I-Lateef, H.M.A.E.; Khalaf, M.M. Corrosion resistance of ZrO2-TiO2 nanocomposite multilayer thin films coated on carbon steel in hydrochloric acid solution. Mater. Charact. 2015, 108, 29–41. [Google Scholar] [CrossRef]
- Zheludkecich, M.L.; Salavado, I.M.; Ferreira, M.G.S. Sol-gel coatings for corrosion protection of metals. J. Mater. Chem. 2005, 15, 5099–5111. [Google Scholar] [CrossRef]
- Li, Q. Sol-gel coatings to improve the corrosion resistance of magnesium (Mg) alloys. In Corrosion Prevention of Magnesium Alloys, 1st ed.; Woodhead Publishing: Cambridge, UK, 2013; pp. 469–485. [Google Scholar]
- Wang, D.; Bierwagen, G.P. Sol-gel coatings on metals for corrosion protection. Prog. Org. Coat. 2009, 64, 327–338. [Google Scholar] [CrossRef]
- Stambolova, I.; Yordanov, S.; Lakov, L.; Vassilev, S.; Blaskov, V.; Jivov, B. Preparation of sol-gel SiO2 coatings on steel and their corrosion resistance. In National Congress on Theoretical and Applied Mechanics 2017; Institute of Mechanics: Sofia, Bulgaria, 2018. [Google Scholar]
- Montemor, M.; Pinto, R.; Ferreira, M.G.S. Chemmical composition and corrosion protection of silane films modified with CeO2 nanoparticles. Electrochim. Acta 2009, 54, 5179–5189. [Google Scholar] [CrossRef]
- Venila, R.; Kamaraj, P.; Arthanareeswari, M.; Devikala, S. Surface modification of mild steel using Ag doped SnO2 nanoparticles for corrosion inhibition. Int. J. Adv. Chem. Sci. Appl. 2014, 1, 16–20. [Google Scholar]
- Deyab, M.A.; Keera, S.T. Effect of nano- TiO2 particles size on the corrosion resistance of alkyd coating. Mater. Chem. Phys. 2014, 146, 406–411. [Google Scholar] [CrossRef]
- Shi, X.; Nguyen, T.A.; Suo, Z.; Liu, Y.; Avci, R.A. Effect of nanoparticles on the anticorrosion and mechanical properties of epoxy coating. Surf. Coat. Technol. 2009, 204, 237–245. [Google Scholar] [CrossRef]
- Zand, R.Z.; Verbeken, K.; Adriaens, A. Influence of the cerium concentration on the corrosion performance of Ce-doped silica hybrid coatings on hot dip galvanized steel substrates. Int. J. Electrochem. Sci. 2013, 8, 548–563. [Google Scholar]
- Garcia-Heras, M.; Jimenez-Morales, A.; Casal, B.; Galvan, J.C.; Radzki, S.; Villegas, M.A. Preparation and electrochemical study of cerium–silica sol–gel thin films. Alloy Compd. 2004, 380, 219–224. [Google Scholar] [CrossRef]
- Hamdy, A.S.; Butt, D.P.; Ismail, A.A. Electrochemical impedance studies of sol–gel based ceramic coatings systems in 3.5% NaCl solution. Electrochim. Acta 2007, 52, 3310–3316. [Google Scholar] [CrossRef]
- Liu, T.J.; Jin, Z.G.; Feng, L.R.; Wang, T. Conducting antimony-doped tin oxide films derived from stannous oxalate by aqueous sol-gel method. Appl. Surf. Sci. 2008, 254, 6547–6553. [Google Scholar] [CrossRef]
- Benrabah, B.; Bouaza, A.; Kadari, A.; Maaref, M.A. Impedance studies of Sb doped SnO2 thin film prepared by sol-gel process. Superlattices Microstruct. 2011, 50, 591–600. [Google Scholar] [CrossRef]
- Dong, C.; Liu, X.; Xiao, X.; Chen, G.; Wang, Y.; Djerdj, I. Combustion synthesis of porous Pt-functionalized SnO2 sheets for isopropanol gas detection with a significant enhancement in response. Mater. Chem. A 2014, 2, 20089–20095. [Google Scholar] [CrossRef]
- Andersen, S.M.; Nørgaard, C.F.; Larsen, M.J.; Skou, E. Tin Dioxide as an effective antioxidant for proton exchange membrane fuel cells. J. Power Sources 2015, 273, 158–161. [Google Scholar] [CrossRef]
- Crespo, M.D.; Meneses, E.R.; Huerta, A.T.; Murillo, A.G.; Estrada, E.M.A.; Hernández, O.C. Synthesis by Sol-gel route and characterization of ceria doped silica coatings on commercial carbon steel. Portugaliae Electrochim. Acta 2009, 3, 257–267. [Google Scholar] [CrossRef]
- Wang, H.; Huang, S.L.; Zuo, Y.J.; Zhou, T.; Zhang, L.R. Corrosion resistance of lamellar aluminium pigments coated by SiO2 by sol–gel method. Corros. Sci. 2011, 53, 161–167. [Google Scholar] [CrossRef]
- Giefers, H.; Porsch, F.; Wortmann, G. Kinetics of the Disproportionation of son. Solid State Ion. 2005, 176, 199–207. [Google Scholar] [CrossRef]
- Dai, Z.R.; Pan, Z.W.; Wang, Z.L. Growth and structure evolution of novel Tin Oxide Diskettes. J. Am. Chem. Soc. 2002, 124, 8673–8680. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Lian, Y.; Gu, M.; Yu, J.; Tang, T.B. Static and dynamic disorder in metastable phases of Tin Oxide. J. Phys. Chem. 2017, 121, 16006–16011. [Google Scholar] [CrossRef]
- Akgul, F.A.; Gumus, C.; Er, A.O.; Farha, A.H.; Akgul, G.; Ufuktepe, Y.; Liu, Z. Structural and electronic properties of SnO2. J. Alloy Compd. 2013, 579, 50–56. [Google Scholar] [CrossRef]
- Agrahari, V.; Mathpal, M.C.; Kumar, M.; Agarwal, A. Investigations of optoelctronic properties in DMS SnO2 nanoparticles. J. Alloy Compd. 2015, 622, 48–53. [Google Scholar] [CrossRef]
- Aziz, M.; Abbas, S.S.; Baharom, W.R.W. Size-controlled synthesis of SnO2 nanoparticles by sol-gel method. Mater. Lett. 2013, 91, 31–34. [Google Scholar] [CrossRef]
- Qin, L.Y.; Lian, J.S.; Jiang, Q. Effect of grain size on corrosion behavior of electrodeposited bulk nanocrystalline Ni. Trans. Nonferrous Met. Soc. China 2010, 20, 82–89. [Google Scholar] [CrossRef]
- Gollapudi, S. Grain size distribution effects on the corrosion behaviour of materials. Corros. Sci. 2012, 62, 90–94. [Google Scholar] [CrossRef]
- Afshari, V.; Dehghanian, C. Effects of grain size on the electrochemical corrosion behaviourof electrodeposited nanocrystalline Fe coatings in alkaline solution. Corros. Sci. 2009, 51, 1844–1849. [Google Scholar] [CrossRef]
- Martinez, J.A.; Esneider-Alcala, M.; Hernández, M.; Canul, M.P.; Shaji, S. Optimal parameters for synthesizing single phase spinel-type Co2SnO4 by sol–gel technique: Structure determination and microstructure evolution. J. Alloy Compd. 2013, 574, 278–282. [Google Scholar] [CrossRef]
- Wang, J.; Wu, L.K.; Zhou, J.H.; Hu, J.M.; Zhang, J.Q.; Cao, C.N. Construction of a novel painting system using electrodeposited SiO2 film as the pretreatment layer. Corros. Sci. 2013, 68, 57–65. [Google Scholar] [CrossRef]
- Rosero-Navarro, N.C.; Pellice, S.A.; Durán, A.; Aparicio, M. Effects of Ce-containing sol–gel coatings reinforced with SiO2 nanoparticles on the protection of AA2024. Corros. Sci. 2008, 50, 1283–1291. [Google Scholar] [CrossRef]
- Pradityana, A.; Shahab, A.; Noerochim, L.; Susanti, D. Inhibition of corrosion of carbon steel in 3.5% NaCl solution by Myrmecodia Pendans extract. Int. J. Corros. 2016. [Google Scholar] [CrossRef]
- Feil, F.; Furberth, W.; Schutze, M. Nanoparticle based inorganic coatings for corrosion protection of magnesium alloys. Surf. Eng. 2008, 24, 198–204. [Google Scholar] [CrossRef]



| Samples | Grain Size (nm) | % SnO |
|---|---|---|
| SnO2–450 °C | 11.5 ± 1.02 | 10 |
| SnO2–600 °C | 15.8 ± 2.02 | 5 |
| SnO2–800 °C | 18.9 ± 2.20 | 1 |
| SnO2–1000 °C | 25.7 ± 3.17 | 0 |
| SnO2 Concentration (vol %) | Ecorr (mV) | Icorr (µA·cm−2) | Rp (Ω) | IE% |
|---|---|---|---|---|
| A36 Steel (Bare) | −702.83 ± 2.09 | 24.24 ± 0.17 | 896.66 ± 6.36 | - |
| 0 | −639.52 ± 3.01 | 23.15 ± 0.36 | 939.38 ± 14.49 | 4.5 |
| 1 | −597.81 ± 0.76 | 13.82 ± 0.22 | 1572.54 ± 25.09 | 43 |
| 2.5 | −521.20 ± 8.66 | 8.68 ± 0.35 | 2507.92 ± 102.24 | 64.2 |
| 5 | −636.78 ± 0.42 | 18.79 ± 0.19 | 1156.73 ± 11.61 | 22.5 |
| 7 | −656.46 ± 2.06 | 20.16 ± 0.02 | 1078.06 ± 1.28 | 16.8 |
| SnO2 Concentration (vol %) | Ecorr (mV) | Icorr (µA cm−2) | IE% |
|---|---|---|---|
| A36 Steel (Bare) | −606.64 | 25.43 | - |
| 0 | −640.24 | 24.18 | 4.9 |
| 1 | −561.52 | 18.62 | 26.8 |
| 2.5 | −447.49 | 6.01 | 76.4 |
| 5 | −619.72 | 18.90 | 25.7 |
| 7 | −518.22 | 24.10 | 5.2 |
| SnO2 Concentration (vol %) | R1 (Ω) | R2 (Ω) | CPE1 (µF/cm2) | IE% |
|---|---|---|---|---|
| A36 Steel (bare) | 28.36 | 675.5 | 0.0012 | - |
| 0 | 29.72 | 779.9 | 0.0013 | 13.4 |
| 1 | 27.7 | 1563 | 0.0011 | 56.8 |
| 2.5 | 30.78 | 1935 | 0.0008 | 65.1 |
| 5 | 29.42 | 862.9 | 0.0013 | 21.7 |
| 7 | 29.17 | 913.9 | 0.0009 | 26.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acero-Gutiérrez, A.K.; Pérez-Flores, A.L.; Godínez-Salcedo, J.G.; Moreno-Palmerin, J.; Morales-Ramírez, Á.d.J. Corrosion Protection of A36 Steel with SnO2 Nanoparticles Integrated into SiO2 Coatings. Coatings 2020, 10, 385. https://doi.org/10.3390/coatings10040385
Acero-Gutiérrez AK, Pérez-Flores AL, Godínez-Salcedo JG, Moreno-Palmerin J, Morales-Ramírez ÁdJ. Corrosion Protection of A36 Steel with SnO2 Nanoparticles Integrated into SiO2 Coatings. Coatings. 2020; 10(4):385. https://doi.org/10.3390/coatings10040385
Chicago/Turabian StyleAcero-Gutiérrez, Ana Karen, Ana Lilia Pérez-Flores, Jesús Gilberto Godínez-Salcedo, Joel Moreno-Palmerin, and Ángel de Jesús Morales-Ramírez. 2020. "Corrosion Protection of A36 Steel with SnO2 Nanoparticles Integrated into SiO2 Coatings" Coatings 10, no. 4: 385. https://doi.org/10.3390/coatings10040385
APA StyleAcero-Gutiérrez, A. K., Pérez-Flores, A. L., Godínez-Salcedo, J. G., Moreno-Palmerin, J., & Morales-Ramírez, Á. d. J. (2020). Corrosion Protection of A36 Steel with SnO2 Nanoparticles Integrated into SiO2 Coatings. Coatings, 10(4), 385. https://doi.org/10.3390/coatings10040385






