Thin Benzotriazole Films for Inhibition of Carbon Steel Corrosion in Neutral Electrolytes
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
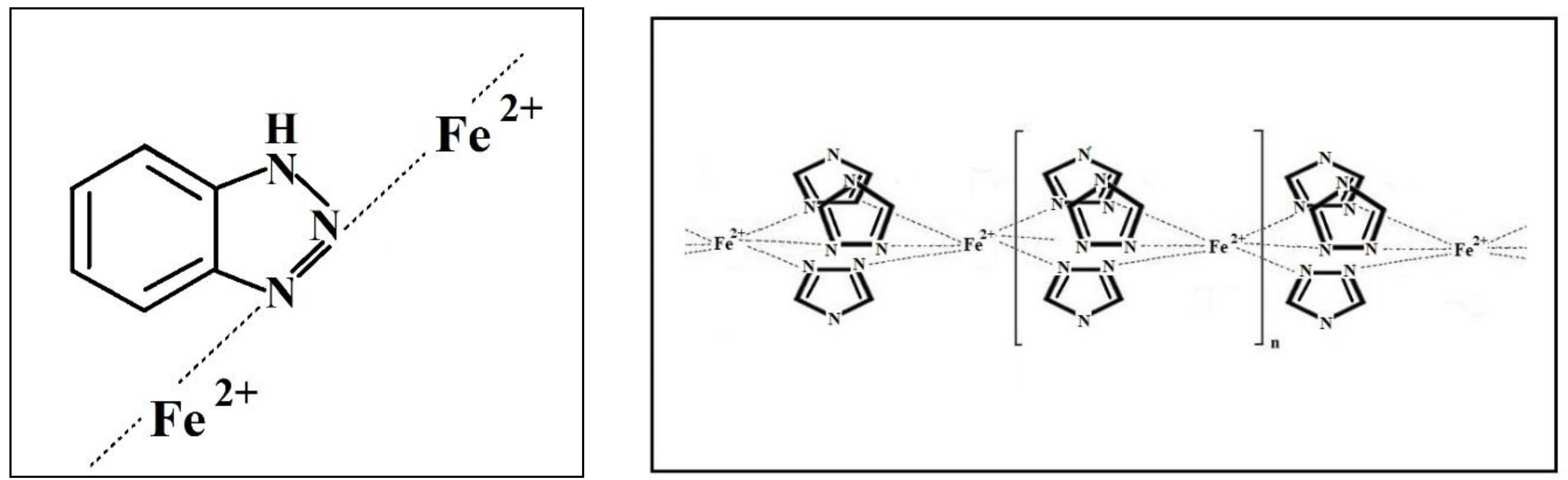
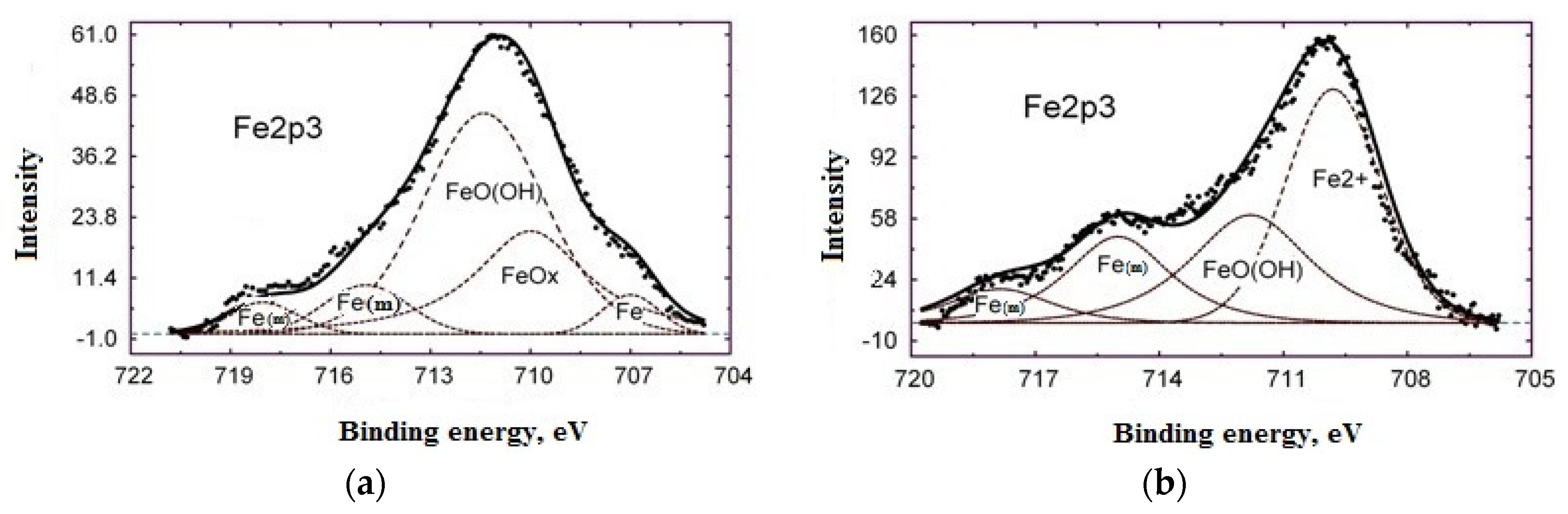
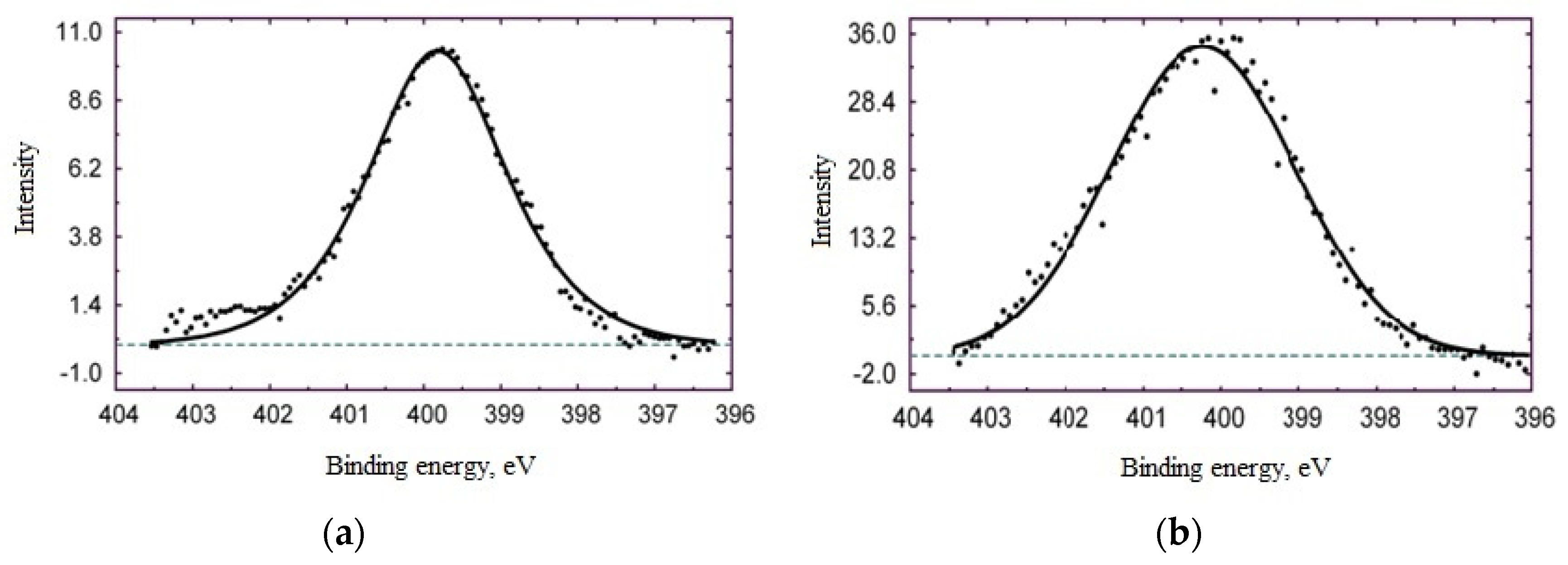
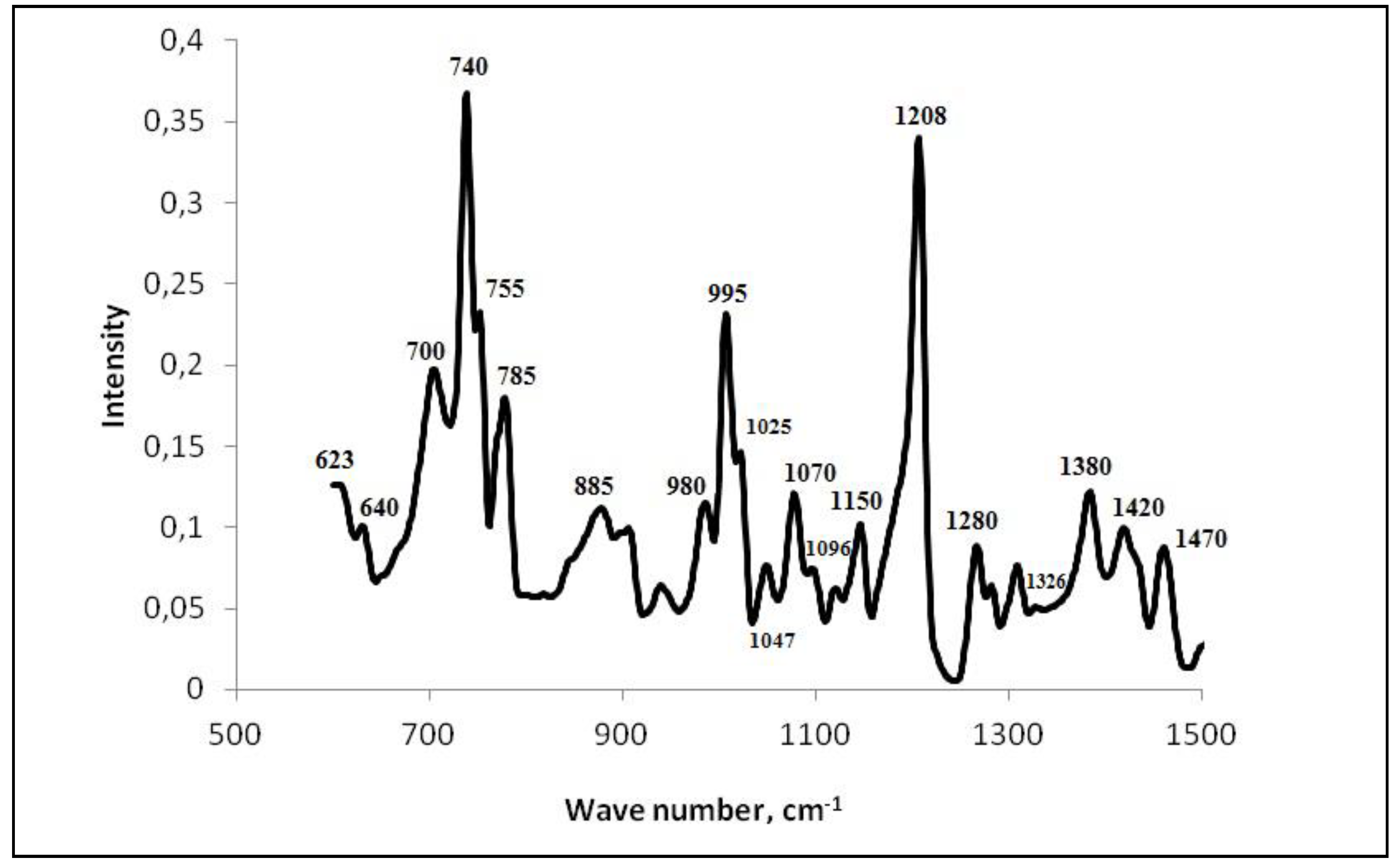
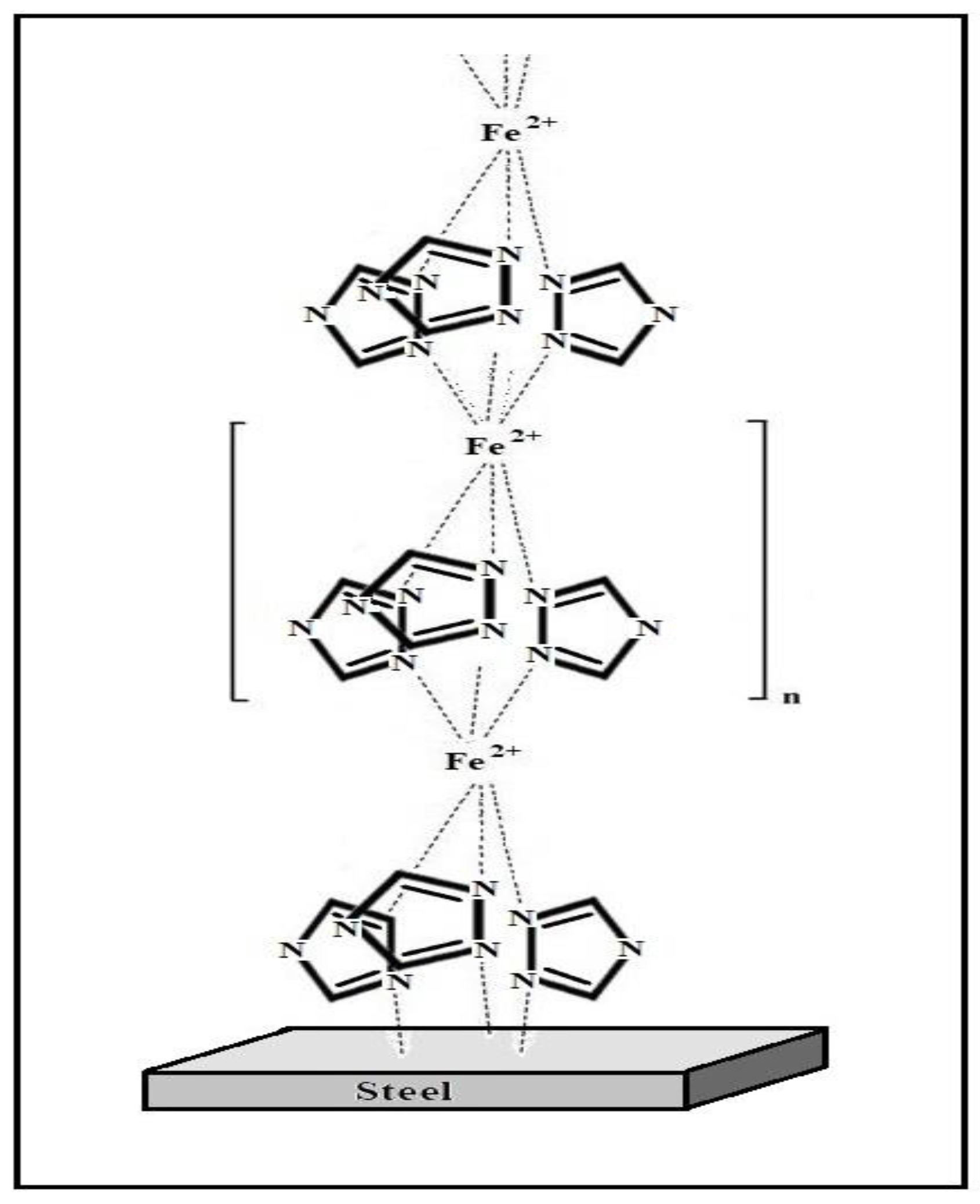
- A study on the adsorption of BTA on steel surface using XPS and IR spectroscopy showed that modification of carbon steel by an aqueous BTA solution produces a polymer-like iron-azole film with a thickness of four molecular layers (thickness 2 nm). The layer contains a bridged complex with donor-acceptor Fe-triazole bonds;
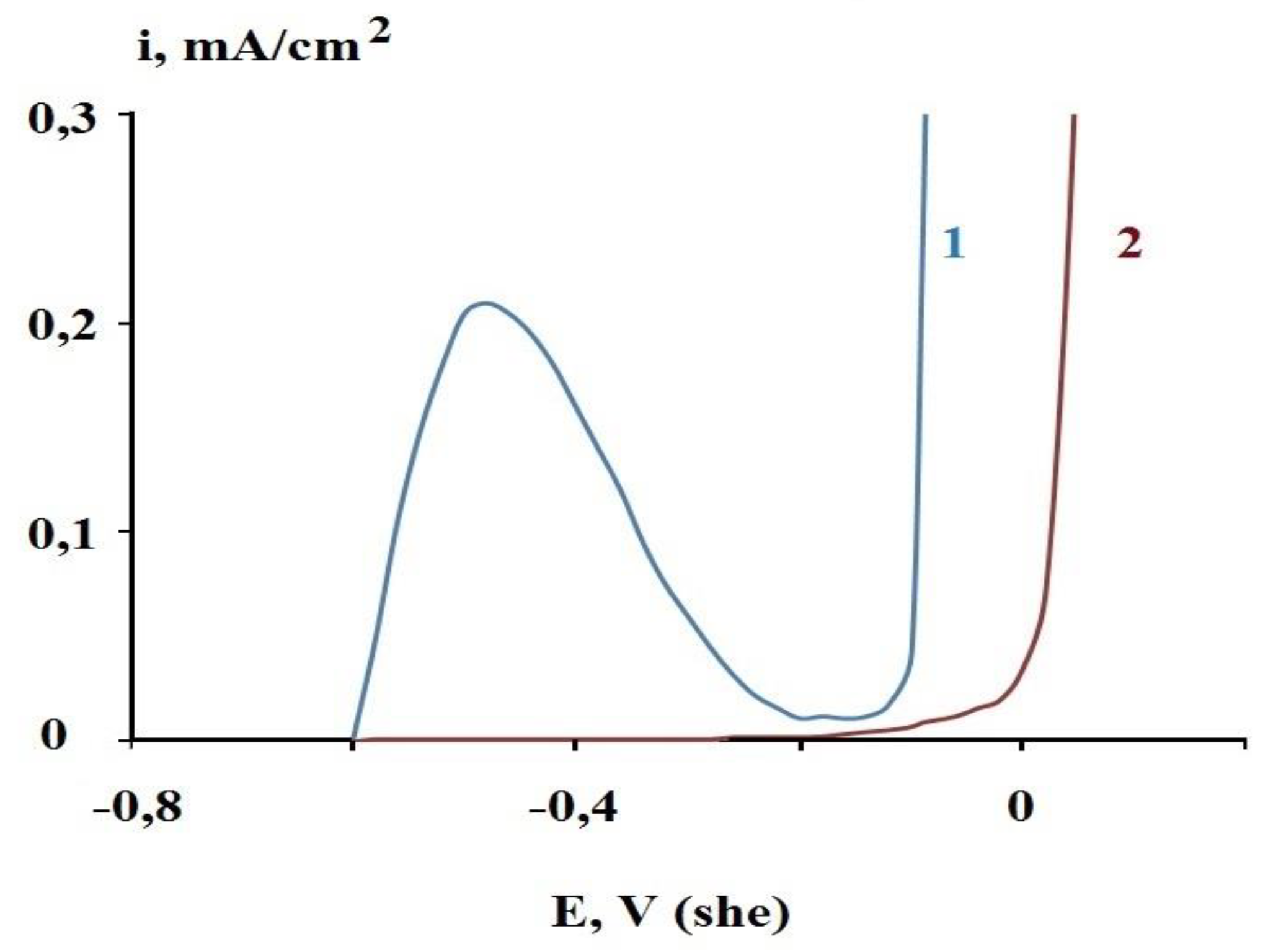
- The layer is capable of inhibiting both uniform and local corrosion of steel in the presence of chloride ions;
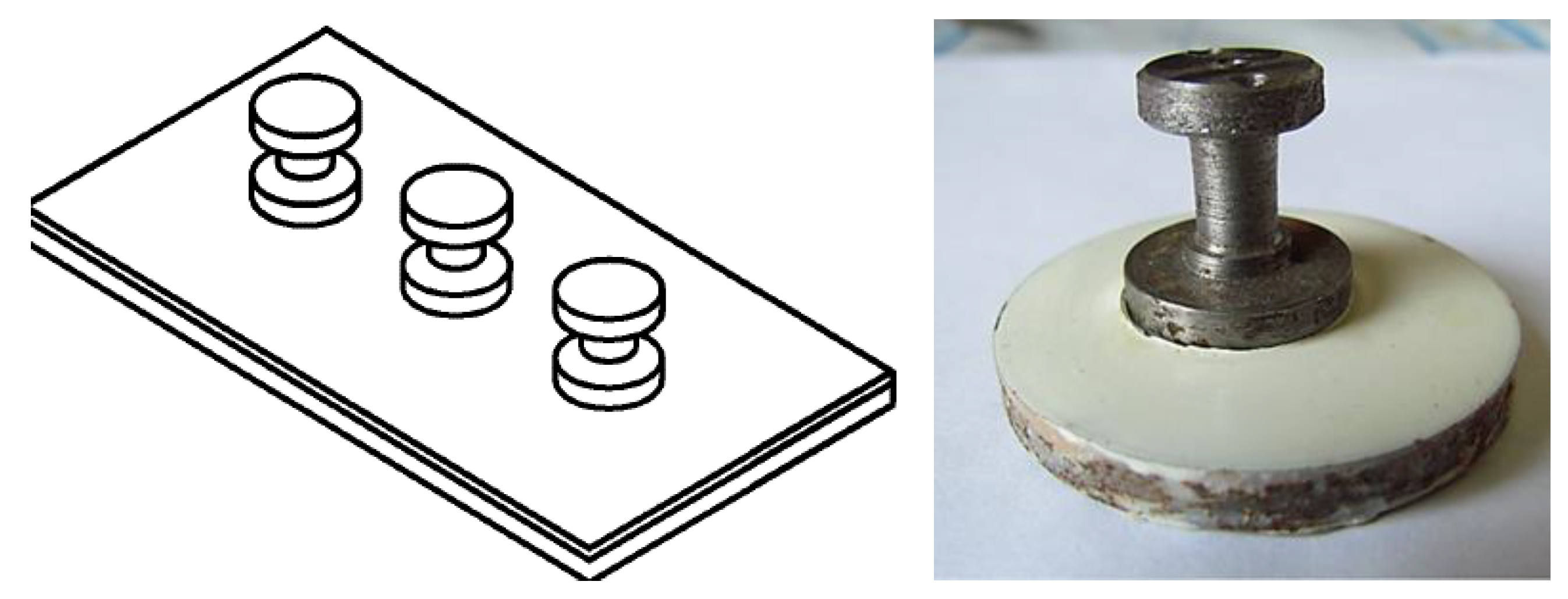
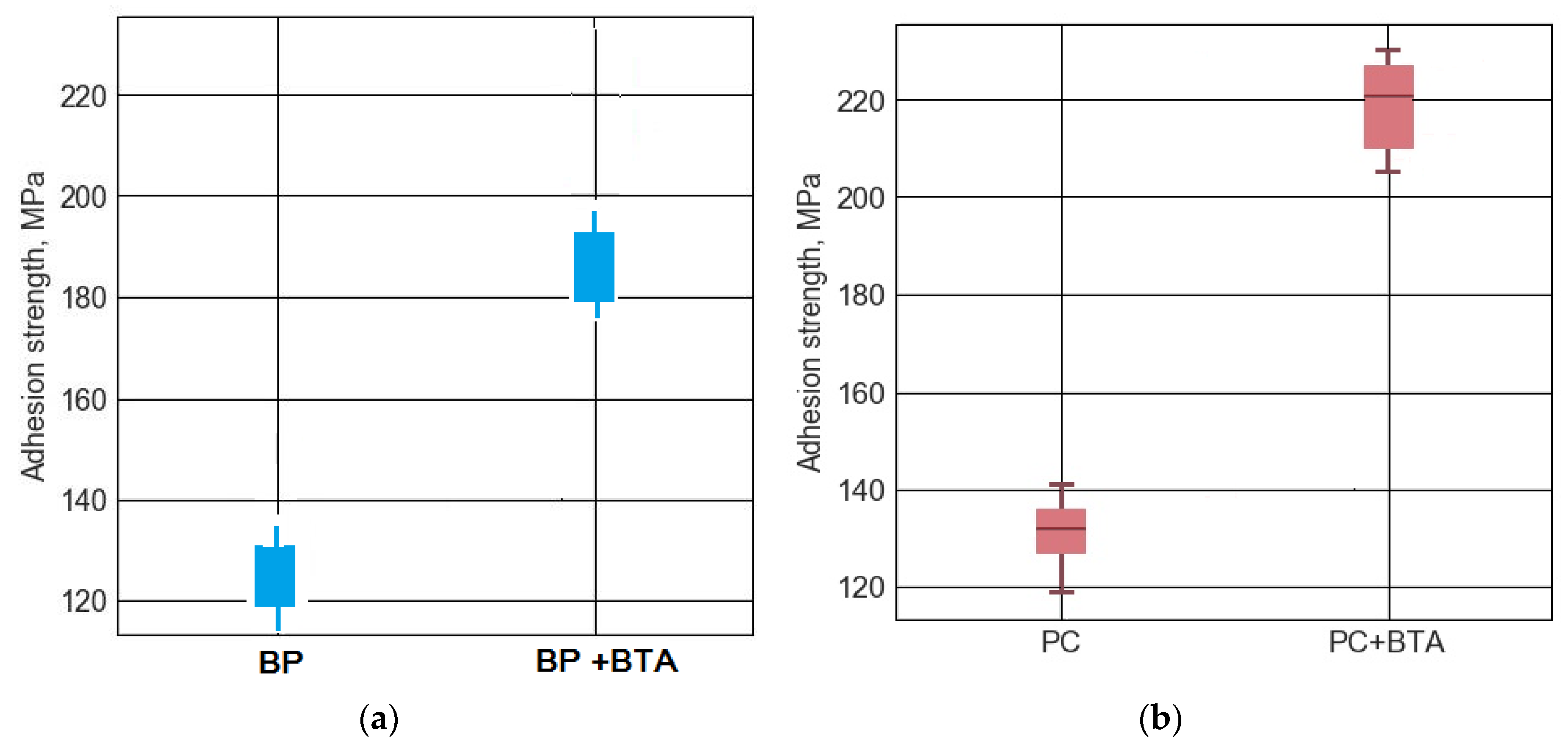
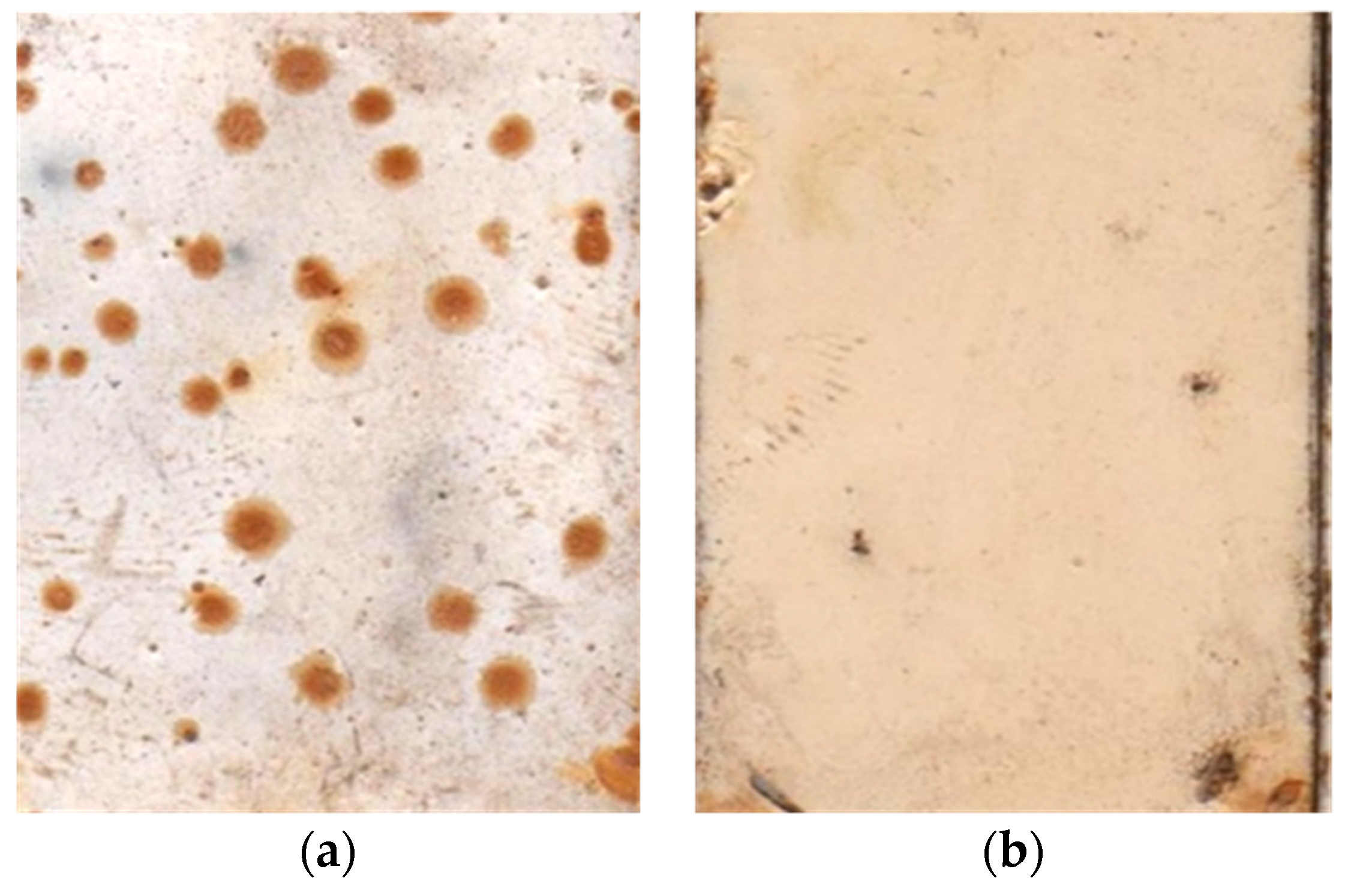
- Pretreatment of steel surface with a benzotriazole solution improves the anticorrosion characteristics such as the adhesion of the coating to the steel. The improvement of the adhesion was shown by peel out and pull off tests of the coatings after exposure in high humid air at the elevated temperature. The corrosion test shows that undercoating corrosion is also reduced by preliminary grafting to the steel surface benzotriazole layer, acting as a corrosion inhibitor and adhesion promoter.
Author Contributions
Funding
Conflicts of Interest
References
- McCafferty, E. Introduction to Corrosion Science; Springer: New York, NY, USA; Dordrecht, The Netherlands; Heidelberg, Germany; London, UK, 2010; p. 4. [Google Scholar] [CrossRef]
- Leygraf, C.; Wallinder, I.O.; Tidblad, J.; Graedel, T. Atmospheric Corrosion, 2nd ed.; John Wiley & Sons, Inc.: New Haven, CT, USA, 2016; p. 152. [Google Scholar]
- Wallinder, I.O.; Bahar, B.; Leygraf, C.; Tidblad, J. Modelling and mapping of copper runoff for Europe. J. Environ. Monit. 2007, 9, 66–73. [Google Scholar] [CrossRef]
- Sorensen, P.A.; Kiil, S.; Dam-Johansen, K.; Weinell, C.E. Anticorrosive coatings: A review. J. Coat. Technol. Res. 2009, 6, 135–176. [Google Scholar] [CrossRef]
- Tait, W.S. Corrosion Prevention and Control of Chemical Processing Equipment. In Handbook of Environmental Degradation of Materials, 2nd ed.; Kutz, M., Ed.; William Andrew Publishing: Amsterdam, The Netherlands, 2012; pp. 863–886. [Google Scholar] [CrossRef]
- Goncharova, O.A.; Luchkin, A.Y.; Kuznetsov, Y.I.; Andreev, N.N.; Andreeva, N.P.; Vesely, S.S. Octadecylamine, 1,2,3-benzotriazole and a mixture there of as chamber inhibitors of steel corrosion. Int. J. Corros. Scale Inhib. 2018, 7, 203–212. [Google Scholar] [CrossRef]
- Kuznetsov, Y.I. Triazoles as a class of multifunctional corrosion inhibitors. A review. Part I. 1,2,3-Benzotriazole and its derivatives. Copper, zinc and their alloys. Int. J. Corros. Scale Inhib. 2018, 7, 271–307. [Google Scholar] [CrossRef]
- Andreeva, N.P.; Kazanskii, L.P.; Selyaninov, I.A.; Kuznetsov, Y.I.; Ostrovskii, V.A. Adsorption of 5-Phenyltetrazole on Iron and Its Inhibition of the Dissolution of Low-Carbon Steel in a Neutral Solution. Prot. Met. Phys. Chem. Surf. 2009, 45, 806–811. [Google Scholar] [CrossRef]
- Kuznetsov, Y.I.; Kazansky, L.P. Physicochemical aspects of metal protection by azoles as corrosion inhibitors. Russ. Chem. Rev. 2008, 7, 219–232. [Google Scholar] [CrossRef]
- Abd El Haleem, S.M.; Abd El Wanees, S.; Bahgat, A. Environmental factors affecting the corrosion behaviour of reinforcing steel. VI. Benzotriazole and its derivatives as corrosion inhibitors of steel. Corros. Sci. 2014, 87, 321–333. [Google Scholar] [CrossRef]
- Kazanskii, L.P.; Selyaninov, I.A. XPES of 1,2,3_Benzotriazole Nanolayers Formed on Iron Surface. Prot. Met. Phys. Chem. Surf. 2010, 46, 797–804. [Google Scholar] [CrossRef]
- Safarmamadov, S.М.; Shoedarova, Z.A.; Mabatkadamova, K.S. Kompleksoobrazovaniye zheleza (II) i zheleza (III) S 1,2,4-triazoltiolom v srede 1 mol’/L HCL pri 273-318K. Vestnik VGU. Serija Khimia Biologia Farmazia 2017, 1, 37–42. (In Russian) [Google Scholar]
- Haasnoot, J.G. Mononuclear, oligonuclear and polynuclear metal coordination compounds with 1,2,4-triazole derivatives as ligands. Coord. Chem. Rev. 2000, 200, 131–185. [Google Scholar] [CrossRef]
- Huxel, T.; Riedel, S.; Lach, J.; Klingele, J. Iron(II) and Nickel(II) Complexes of N-Alkylimidazoles and 1-Methyl-1H-1,2,4-Triazole: X-ray Studies, Magnetic Characterization, and DFT Calculations. Z. Anorg. Allg. Chem. 2012, 638, 1–11. [Google Scholar] [CrossRef]
- Sastri, V.S. Green Corrosion Inhibitors, Theory and Practice; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; p. 169. [Google Scholar]
- Finley, K.T. Triazoles: 1,2,3; John Wiley & Sons Inc.: New York, NY, USA, 1980; p. 349. [Google Scholar]
- Kuznetsov, Y.I. Organic Inhibitors of Corrosion of Metals; Springer US: New York, NY, USA, 1996; p. 284. [Google Scholar] [CrossRef]
- Rubio, M.; Hernandez, R.; Nogales, A.; Roig, A.; Lopez, D. Structure of a spin-crossover Fe(II)-l,2,4-triazole polymer complex dispersed in an isotactic polystyrene matrix. Eur. Polym. J. 2011, 47, 52–60. [Google Scholar] [CrossRef]
- Smeets, V.; Wolf, M.; Wolny, J.A.; Schünemann, V.; Dîrtu, M.M.; Ge, J.-Y.; Moshchalkov, V.; Garcia, Y. Spin State Crossover, Vibrational, Computational, and Structural Studies of FeII 1-Isopropyl-1H-tetrazole Derivatives. Eur. J. Inorg. Chem. 2018, 3–4, 394–413. [Google Scholar] [CrossRef]
- Kelly, R.J.; Scully, J.R.; Shoesmith, D.W.; Buchheit, R.G. Electrochemical Techniques in Corrosion Science and Engineering; Marcel Dekker Inc.: New York, NY, USA; Basel, Switzerland, 2003; pp. 9–55. [Google Scholar]
- Szklarska-Smialowska, Z. Pitting and Crevice Corrosion; NACE International: Houston, TX, USA, 2005; p. 5. [Google Scholar]
- GOST 380-2005. Сталь углерoдистая oбыкнoвеннoгo качества. Common quality carbon steel. Grades. In Russian State Standard; GOST 380-2005, 2015. [Google Scholar]
- Semiletov, A.M.; Chirkunov, A.A.; Kuznetsov, Y.I.; Andreeva, N.P. Improving steel passivation with aqueous solutions of [3-(2-Aminoethylamino)propyl]trimethoxysilane. Russ. J. Phys. Chem. A 2015, 89, 1915–1922. [Google Scholar] [CrossRef]
- ASTM D610-08. Standard Practice for Evaluating Degree of Rusting on Painted Steel Surfaces; ASTM INTERNATIONAL: West Conshohocken, PA, USA, 2019. [Google Scholar] [CrossRef]
- ASTM D 3330/D 3330M—02. Standard Test Method for Peel Adhesion of Pressure-Sensitive Tape; Kenya Bureau of Standards: Nairobi, Kenya, 2018. [CrossRef]
- ISO 4624:2003. Paints and Varnishes—Pull-off Test for Adhesion; European Committee for Standardization: Brussels, Belgium, 2003. [Google Scholar]
- Matyash, I.E.; Minailova, I.A.; Mishchuk, O.N.; Serdega, В.K. Kramers-Kronig Relations in Representation of Modulation Polarimetry by an Example of Transmission Spectra of GaAs Crystal. Opt. Spectrosc. 2017, 123, 881–888. [Google Scholar] [CrossRef]
- Kazansky, L.P.; Selyaninov, I.A.; Kuznetsov, Y.I. Adsorption of 2-mercaptobenzothiazole on copper surface from phosphate. Appl. Surf. Sci. 2012, 258, 6807–6813. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Kim, S.-H.; Kim, J.-G. Effect of 1, 2, 3-Benzotriazole on the Corrosion Properties of 316L Stainless Steel in Synthetic Tap Water. Met. Mater. Int. 2015, 21, 1013–1022. [Google Scholar] [CrossRef]
- Andrikopoulos, P.C.; McCarney, K.M.; Armstrong, D.R.; Littleford, R.E.; Graham, D.; Smith, W.E. A density functional theory and resonance Raman study of a benzotriazoledye used in surface enhanced resonance Raman scattering. J. Mol. Struct. 2006, 789, 59–70. [Google Scholar] [CrossRef]
- Wang, K.; Yang, L.; Wei, W.; Zhang, L.; Chang, G. Phosphoric acid-doped poly (ether sulfone benzotriazole) for high temperature proton exchange membrane fuel cell applications. J. Membr. Sci. 2018, 549, 23–27. [Google Scholar] [CrossRef]
- Armand, F.; Badoux, C.; Bonville, P.; Ruaudel-Teixier, A.; Kahn, O. Langmuir-Blodgett Films of Spin Transition Iron (II) Metalorganic Polymers, 1. Iron (II) Complexes of Octadecyl-1,2,4-triazole. Langmuir 1995, 11, 3467–3472. [Google Scholar] [CrossRef]
- Manners, I. Synthetic Metal-Containing Polymers; WILEY-VCH: Verlag, GmbH & Co. KGaA: Weinheim, Germany, 2004; p. 91. [Google Scholar]
- Gladkikh, N.; Makarychev, Y.; Petrunin, M.; Maleeva, M.; Maksaeva, L.; Marshakov, A. Synergistic effect of silanes and azole for enhanced corrosion protection of carbon steel by polymeric coatings. Prog. Org. Coat. 2020, 138, 1–8. [Google Scholar] [CrossRef]
- Kuznetsov, Y.I. Physicochemical aspects of metal corrosion inhibition in aqueous solutions. Russ. Chem. Rev. 2004, 73, 75–87. [Google Scholar] [CrossRef]


| Mass Fraction of Chemical Elements, % | |||||||
|---|---|---|---|---|---|---|---|
| C | Mn | Si | Cr | Ni | Cu | S | P |
| 0.14–0.22 | 0.3–0.6 | <0.05 | <0.05 | <0.05 | <0.05 | <0.05 | <0.04 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrunin, M.; Maksaeva, L.; Gladkikh, N.; Makarychev, Y.; Maleeva, M.; Yurasova, T.; Nazarov, A. Thin Benzotriazole Films for Inhibition of Carbon Steel Corrosion in Neutral Electrolytes. Coatings 2020, 10, 362. https://doi.org/10.3390/coatings10040362
Petrunin M, Maksaeva L, Gladkikh N, Makarychev Y, Maleeva M, Yurasova T, Nazarov A. Thin Benzotriazole Films for Inhibition of Carbon Steel Corrosion in Neutral Electrolytes. Coatings. 2020; 10(4):362. https://doi.org/10.3390/coatings10040362
Chicago/Turabian StylePetrunin, Maxim, Liudmila Maksaeva, Natalia Gladkikh, Yuriy Makarychev, Marina Maleeva, Tatyana Yurasova, and Andrei Nazarov. 2020. "Thin Benzotriazole Films for Inhibition of Carbon Steel Corrosion in Neutral Electrolytes" Coatings 10, no. 4: 362. https://doi.org/10.3390/coatings10040362
APA StylePetrunin, M., Maksaeva, L., Gladkikh, N., Makarychev, Y., Maleeva, M., Yurasova, T., & Nazarov, A. (2020). Thin Benzotriazole Films for Inhibition of Carbon Steel Corrosion in Neutral Electrolytes. Coatings, 10(4), 362. https://doi.org/10.3390/coatings10040362






