Electroless Deposition of Ni-P Coatings on HNBR for Low Friction Rubber Seals
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. PVP Coating
2.3. HNBR Surface Activation and Electroless Nickel Plating
2.4. Characterization
2.4.1. Surface Functionalization
2.4.2. Ni-P Film
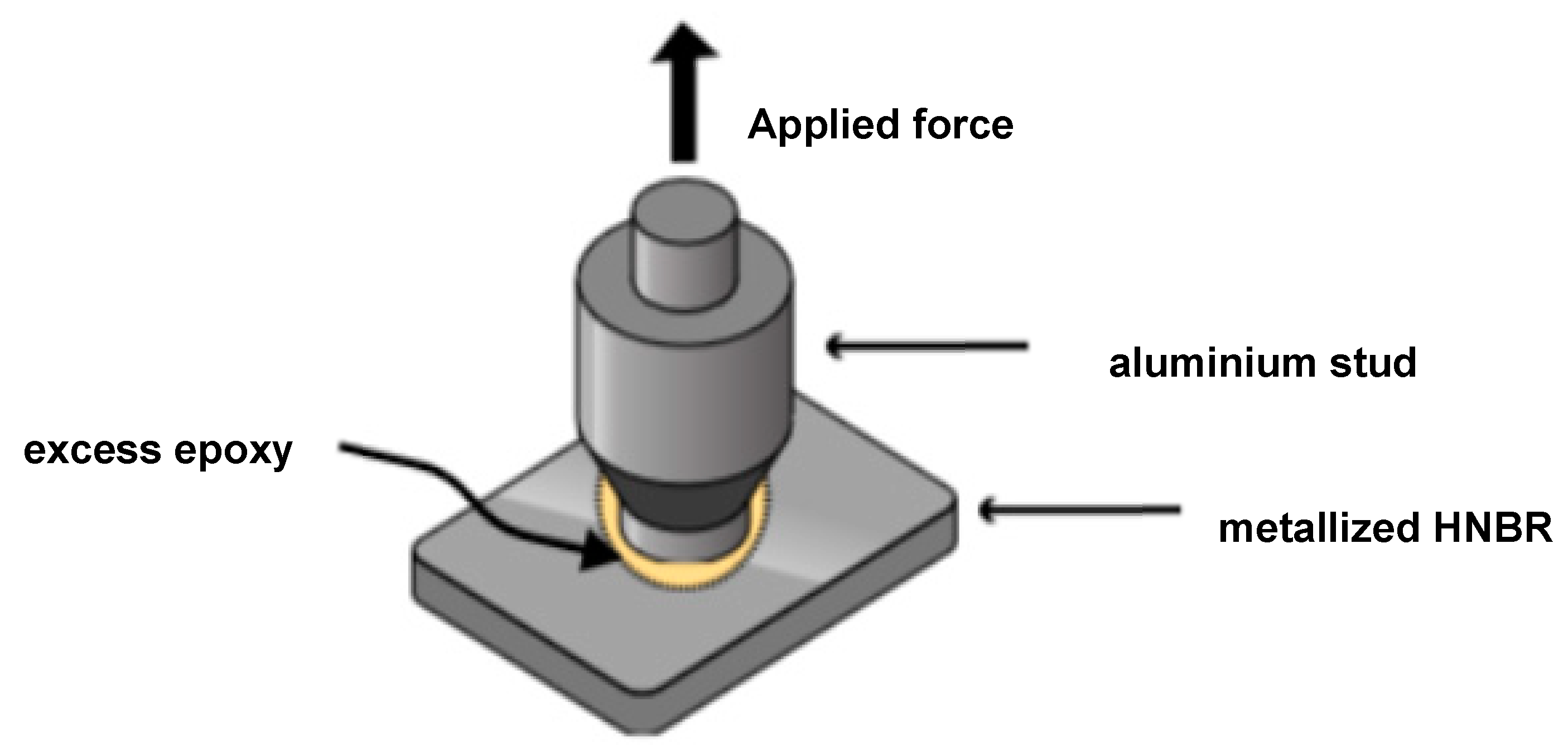
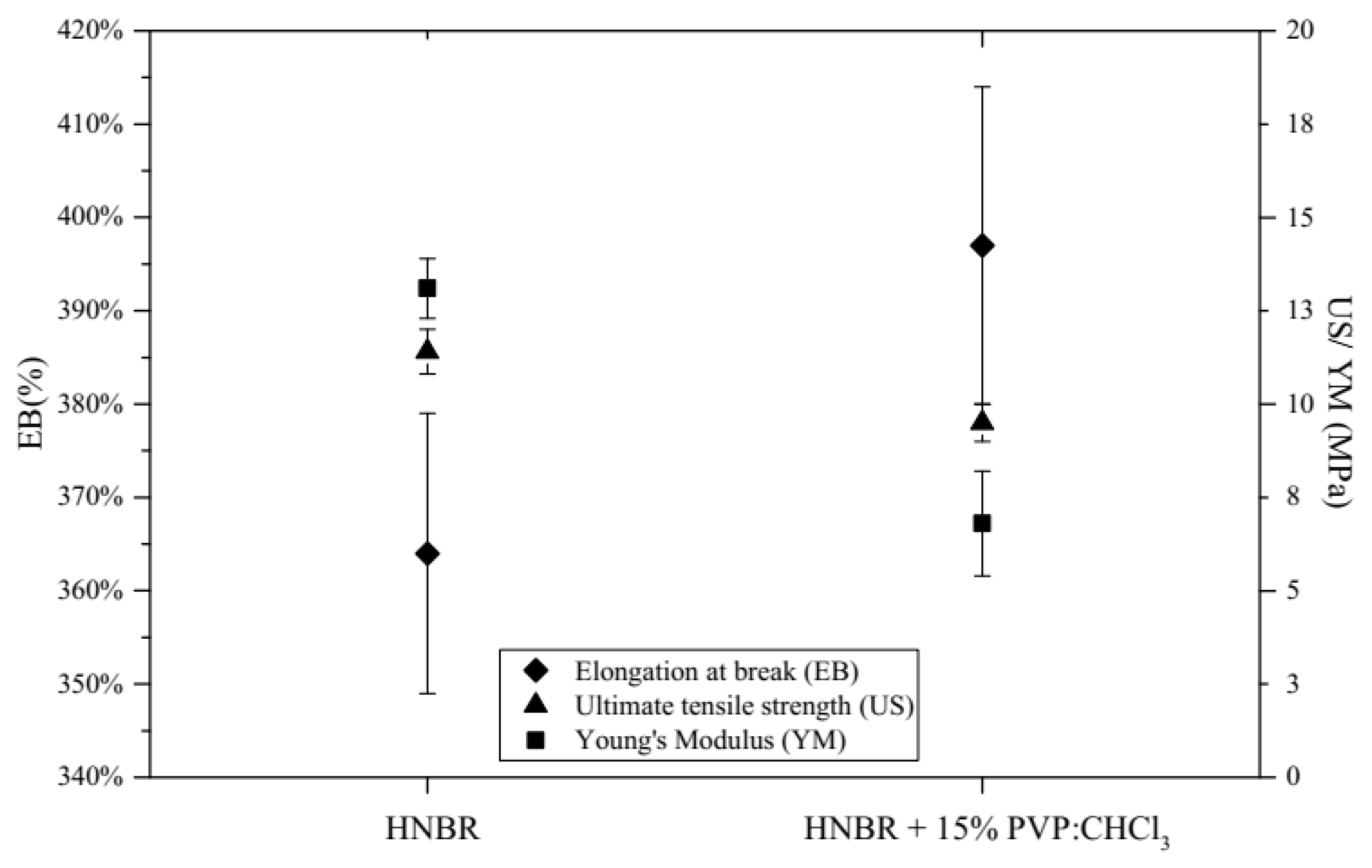
2.4.3. Mechanical Tests
2.4.4. Tribological Tests
2.4.5. Surface Conductivity
3. Results and Discussion
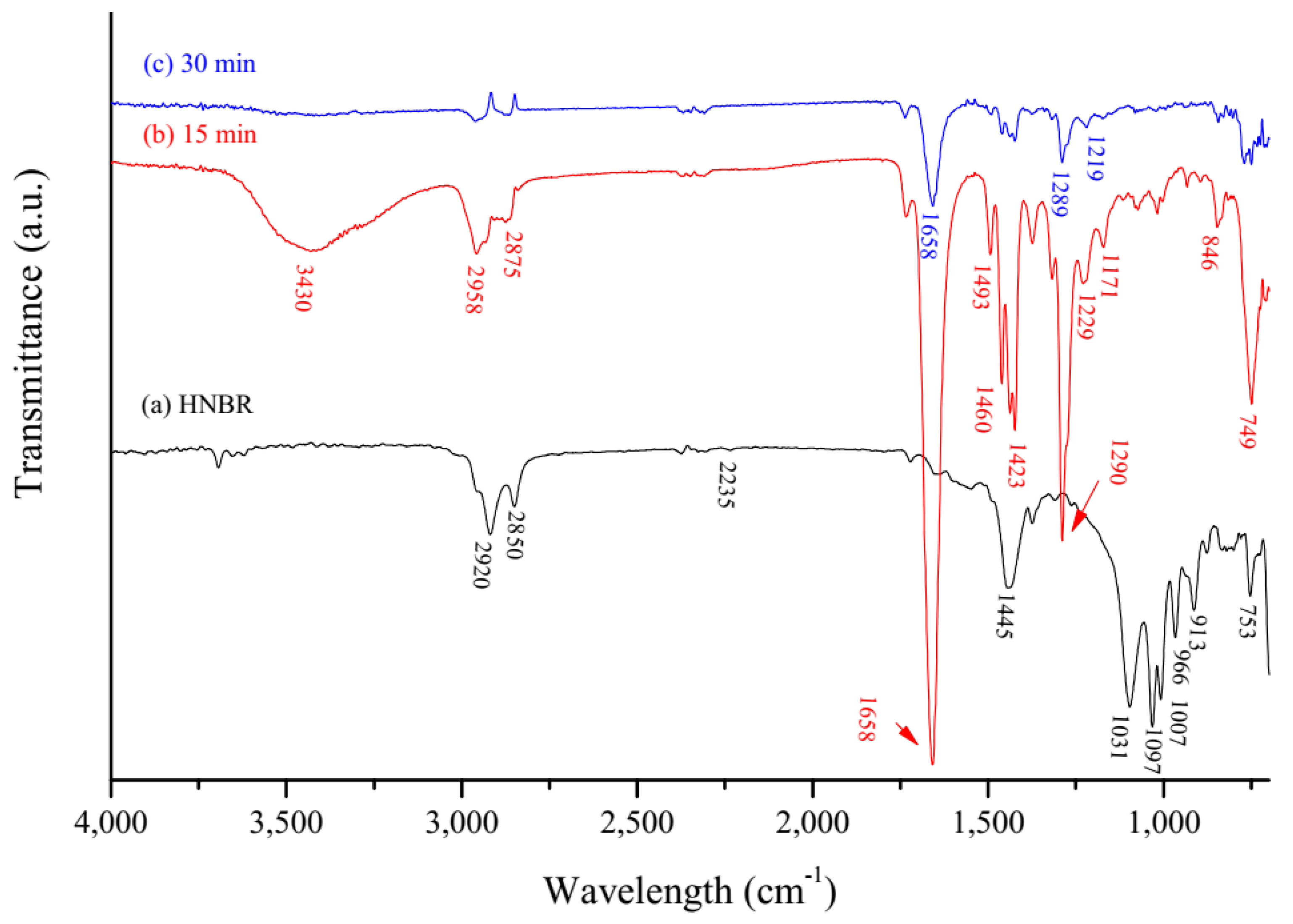
3.1. PVP Coating on the HNBR Surface
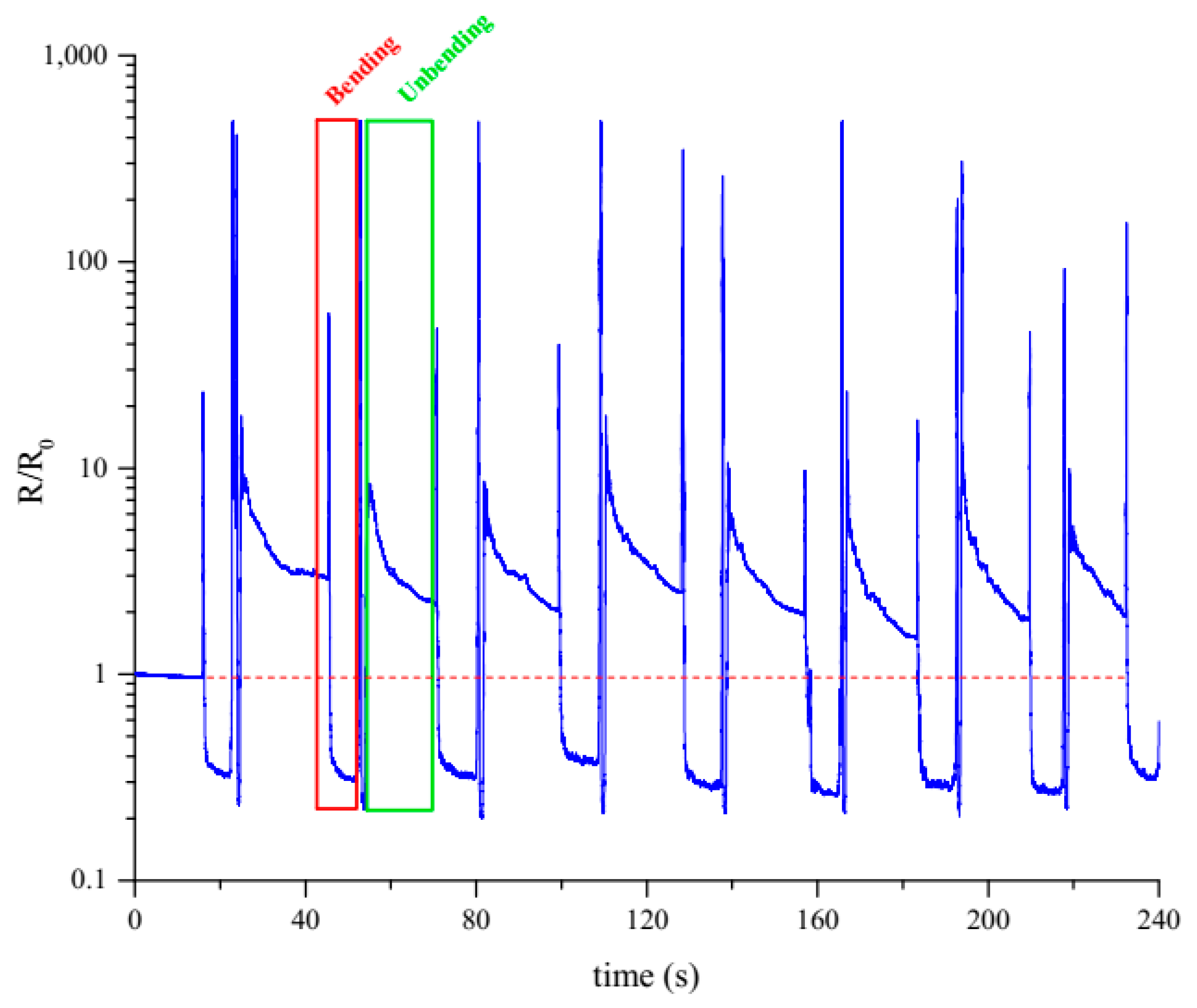
3.2. PVP Layer Stability
3.3. Formation of Catalytic Nanoparticles for ELP
3.4. Ni-P Plating in an Alkaline Bath
3.5. Ni-P Film Properties
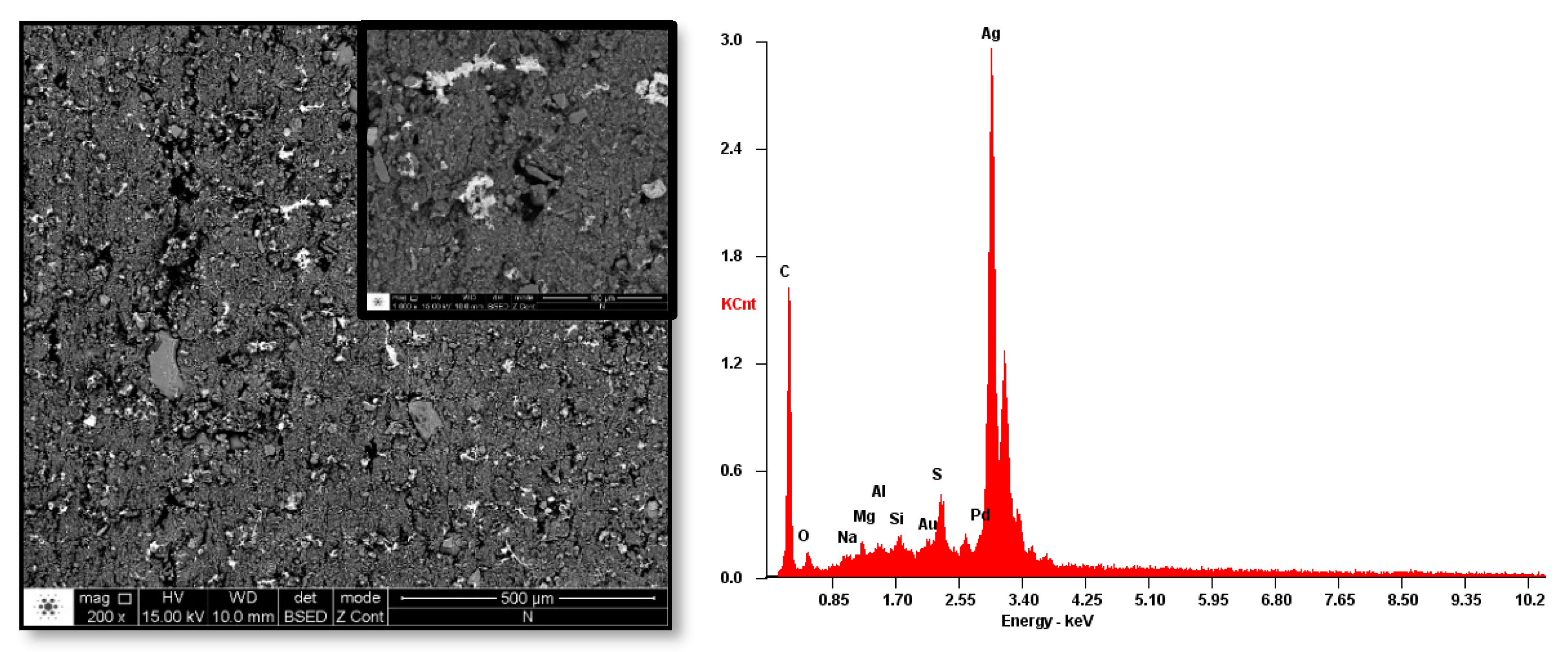
3.5.1. Morphology and Adhesion
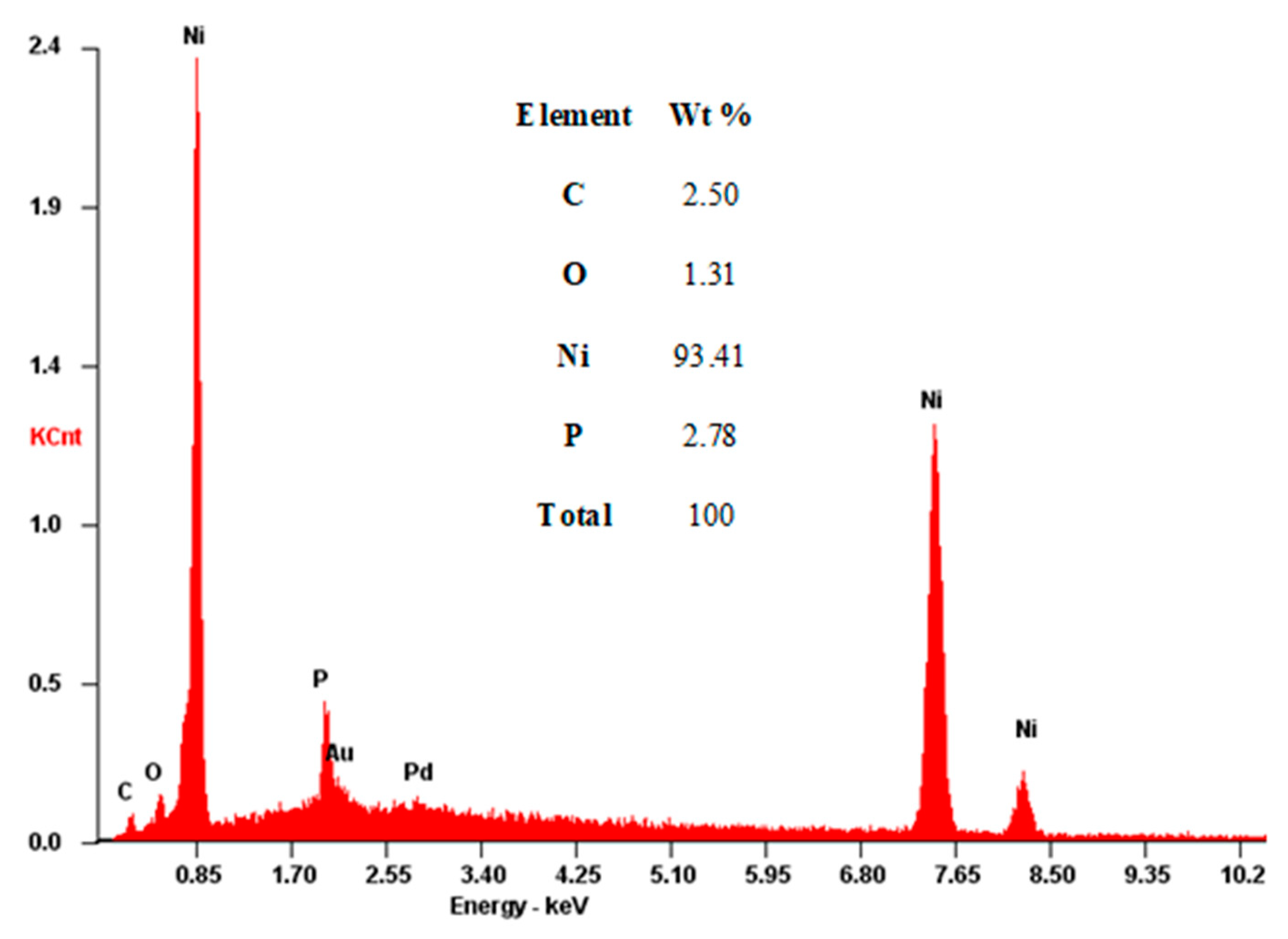
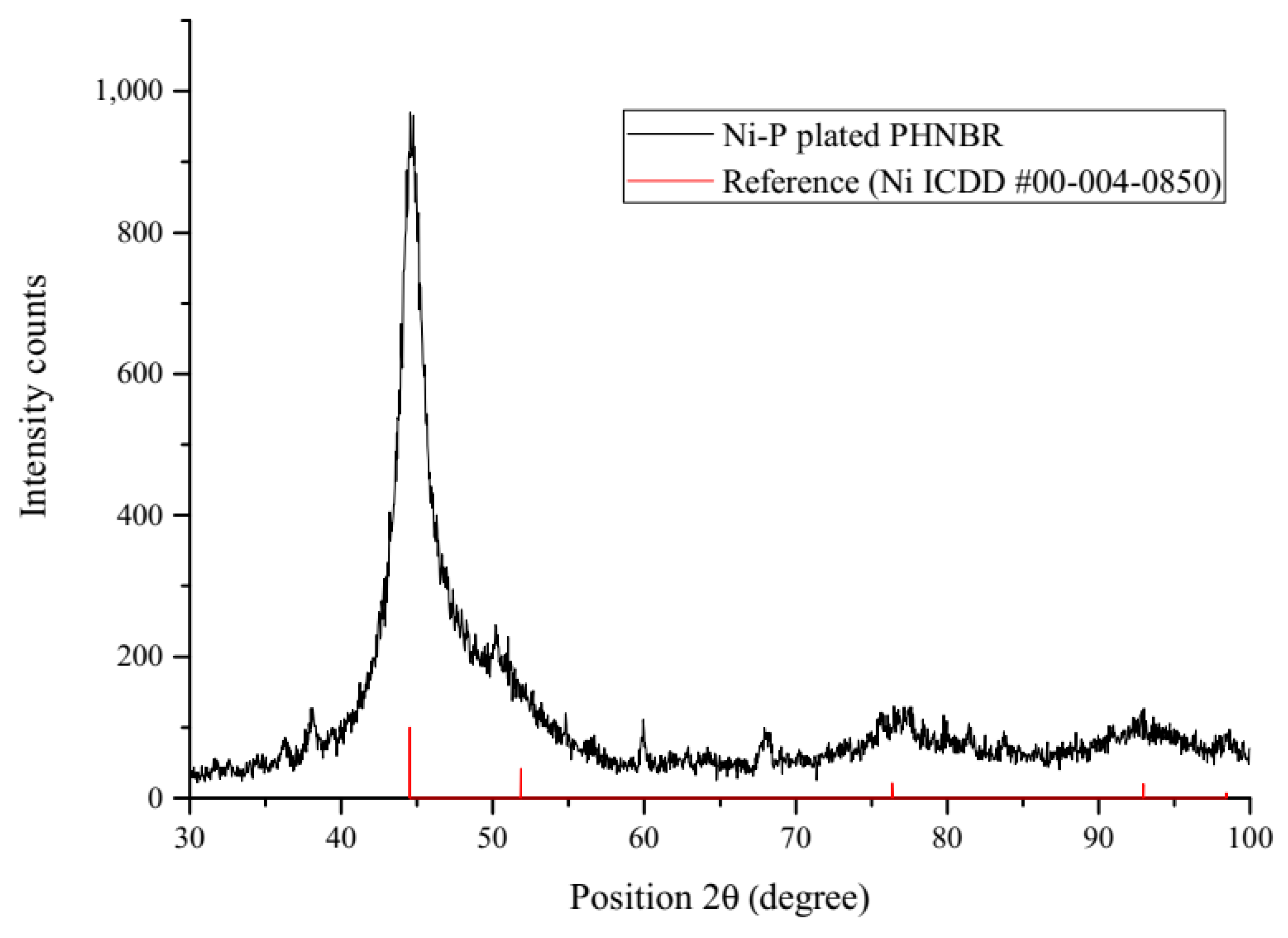
3.5.2. Composition and Structure
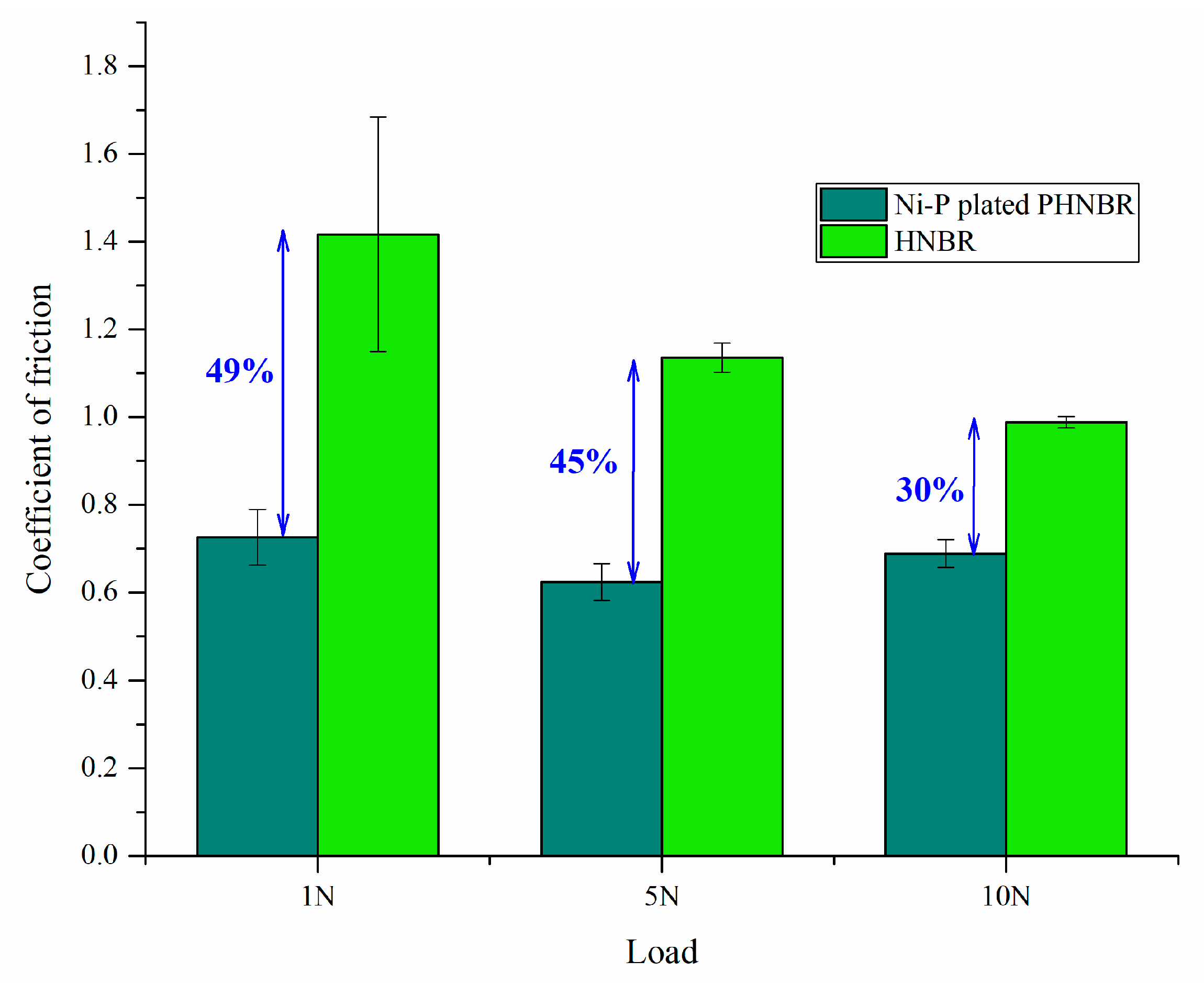
3.6. Tribological Behavior
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Affonso, L.O.A. Machinery Failure Analysis Handbook: Sustain Your Operations and Maximize Uptime; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Flitney, R.K. Seals and Sealing Handbook; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Lubwama, M.; Corcoran, B.; Sayers, K. DLC films deposited on rubber substrates: A review. Surf. Eng. 2014, 31, 1–10. [Google Scholar] [CrossRef]
- Sudagar, J.; Lian, J.; Sha, W. Electroless nickel, alloy, composite and nano coatings—A critical review. J. Alloys Compd. 2013, 571, 183–204. [Google Scholar] [CrossRef]
- Loto, C.A. Electroless Nickel Plating-A Review. Silicon 2016, 8, 177–186. [Google Scholar] [CrossRef]
- Garcia, A.; Polesel-Maris, J.; Viel, P.; Palacin, S.; Berthelot, T. Localized Ligand Induced Electroless Plating (LIEP) Process for the Fabrication of Copper Patterns onto Flexible Polymer Substrates. Adv. Funct. Mater. 2011, 21, 2096–2102. [Google Scholar] [CrossRef]
- Garcia, A.; Berthelot, T.; Viel, P.; Mesnage, A.; Jégou, P.; Nekelson, F.; Roussel, S.; Palacin, S. ABS Polymer Electroless Plating through a One-Step Poly(acrylic acid) Covalent Grafting. ACS Appl. Mater. Interfaces 2010, 2, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Berthelot, T.; Viel, P.; Polesel-Maris, J.; Palacin, S. Microscopic Study of a Ligand Induced Electroless Plating Process onto Polymers. ACS Appl. Mater. Interfaces 2010, 2, 3043–3051. [Google Scholar] [CrossRef]
- Olivera, S.; Muralidhara, H.; Venkatesh, K.; Gopalakrishna, K.; Vivek, C.S. Plating on acrylonitrile-butadiene-styrene (ABS) plastic: A review. J. Mater. Sci. 2016, 51, 3657–3674. [Google Scholar] [CrossRef]
- Du, W.; Zou, H.; Tian, M.; Zhang, L.; Wang, W. Electrically conductive acrylonitrile-butadiene rubber elastomers prepared by dopamine-induced surface functionalization and metallization. Polym. Adv. Technol. 2011, 23, 1029–1035. [Google Scholar] [CrossRef]
- Vasconcelos, B.; Vediappan, K.; Oliveira, J.; Fonseca, C. Mechanically robust silver coatings prepared by electroless plating on thermoplastic polyurethane. Appl. Surf. Sci. 2018, 443, 39–47. [Google Scholar] [CrossRef]
- Tang, X.; Wang, J.; Wang, C.; Shen, B. A novel surface activation method for Ni/Au electroless plating of acrylonitrile–butadiene–styrene. Surf. Coatings Technol. 2011, 206, 1382–1388. [Google Scholar] [CrossRef]
- Klingender, R.C. (Ed.) Handbook of Specialty Elastomers; Informa UK Limited: Abingdon, Oxfordshire, UK, 2008. [Google Scholar]
- Desai, N.P.; Hubbell, J.A. Solution technique to incorporate polyethylene oxide and other water-soluble polymers into surfaces of polymeric biomaterials. Biomaterials 1991, 12, 144–153. [Google Scholar] [CrossRef]
- Khalifa, O.R.; Sakr, E. Electroless Nickel-Phosphorus-Polymer Composite Coatings. Open Corros. J. 2009, 2, 211–215. [Google Scholar] [CrossRef]
- Perraud, S.; Vallat, M.-F.; David, M.-O.; Kuczynski, J. Network characteristics of hydrogenated nitrile butadiene rubber networks obtained by radiation crosslinking by electron beam. Polym. Degrad. Stab. 2010, 95, 1495–1501. [Google Scholar] [CrossRef]
- Wang, M.-Q.; Yan, J.; Du, S.-G.; Li, H. Electroless plating of PVC plastic through new surface modification method applying a semi-IPN hydrogel film. Appl. Surf. Sci. 2013, 277, 249–256. [Google Scholar] [CrossRef]
- Pandey, A.; Manivannan, R. Chemical reduction technique for the synthesis of nickel nanoparticles. Int. J. Eng. Res. Appl. 2015, 5, 96–100. [Google Scholar]
- Mafi, I.R.; Dehghanian, C. Comparison of the coating properties and corrosion rates in electroless Ni–P/PTFE composites prepared by different types of surfactants. Appl. Surf. Sci. 2011, 257, 8653–8658. [Google Scholar] [CrossRef]
- Kim, J.H.; Min, B.R.; Won, J.; Joo, S.H.; Kim, H.S.; Kang, Y.S. Role of Polymer Matrix in Polymer/Silver Complexes for Structure, Interactions, and Facilitated Olefin Transport. Macromolecules 2003, 36, 6183–6188. [Google Scholar] [CrossRef]
- Sivaiah, K.; Kumar, K.N.; Naresh, V.; Buddhudu, S. Structural and Optical Properties of Li+: PVP & Ag+: PVP Polymer Films. Mater. Sci. Appl. 2011, 2, 1688–1696. [Google Scholar] [CrossRef][Green Version]
- Guo, R.; Wen, J.; Gao, Y.; Li, T.; Yan, H.; Wang, H.; Niu, B.; Jiang, K. Effect of the adhesion of Ag coatings on the effectiveness and durability of antibacterial properties. J. Mater. Sci. 2018, 53, 4759–4767. [Google Scholar] [CrossRef]
- Passador, F.R.; Rodolfo, A., Jr.; Pessan, L.A. In Situ Dynamic Vulcanization of Poly (Vinyl Chloride)/Acrylonitrile-butadiene Rubber Blends. J. Macromol. Sci. Part B 2009, 48, 282–298. [Google Scholar] [CrossRef]
- Sanches, N.B.; Pedro, R.; Diniz, M.F.; Mattos, E.D.C.; Cassu, S.N.; Dutra, R.D.C.L. Infrared Spectroscopy Applied to Materials Used as Thermal Insulation and Coatings. J. Aerosp. Technol. Manag. 2013, 5, 421–430. [Google Scholar] [CrossRef]
- Lou, W.; Zhang, W.; Liu, X.; Dai, W.; Xu, D. Degradation of hydrogenated nitrile rubber (HNBR) O-rings exposed to simulated servo system conditions. Polym. Degrad. Stab. 2017, 144, 464–472. [Google Scholar] [CrossRef]
- Kim, J.-T.; Lee, D.-Y.; Oh, T.-S. Characteristics of nitrile-butadiene rubber layered silicate nanocomposites with silane coupling agent. J. Appl. Polym. Sci. 2003, 89, 2633–2640. [Google Scholar] [CrossRef]
- Peng, C.; Hou, Z.; Zhang, C.; Li, G.; Lian, H.; Cheng, Z.; Lin, J. Synthesis and luminescent properties of CaTiO3: Pr3+ microfibers prepared by electrospinning method. Opt. Express 2010, 18, 7543–7553. [Google Scholar] [CrossRef] [PubMed]
- Salih, S.I.; Jabur, A.R.; Mohammed, T. The Effect of PVP Addition on the Mechanical Properties of Ternary Polymer Blends. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Kerbala, Iraq, 26–27 March 2018; p. 012071. [Google Scholar]
- Ravindra, C.; Sarswati, M.; Sukanya, G.; Shivalila, P.; Soumya, Y.; Deepak, K. Tensile and thermal properties of poly (vinyl) pyrrolidone/vanillin incorporated polyvinyl alcohol films. Res. J. Phys. Sci. 2015, 3, 1–6. [Google Scholar]
- Krishnan, K.H.; John, S.; Srinivasan, K.N.; Praveen, J.; Ganesan, M.; Kavimani, P.M. An overall aspect of electroless Ni-P depositions-A review article. Met. Mater. Trans. A 2006, 37, 1917–1926. [Google Scholar] [CrossRef]
- Pang, H.; Bai, R.; Shao, Q.; Gao, Y.; Li, A.; Tang, Z. A novel Ag catalyzation process using swelling impregnation method for electroless Ni deposition on Kevlar® fiber. Appl. Surf. Sci. 2015, 359, 280–287. [Google Scholar] [CrossRef]
- Li, W.; Shi, G.; Lu, Y. Copper-catalyzed electroless nickel coating on poly(ethylene terephthalate) board for electromagnetic application. Int. J. Mater. Res. 2014, 105, 797–801. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, B.; Hu, L. PVP Protective Mechanism of Ultrafine Silver Powder Synthesized by Chemical Reduction Processes. J. Solid State Chem. 1996, 121, 105–110. [Google Scholar] [CrossRef]
- Mallory, G.O.; Hajdu, J.B. Electroless Plating: Fundamentals and Applications; William Andrew: Orlando, FL, USA, 1990. [Google Scholar]
- Żenkiewicz, M.; Moraczewski, K.; Rytlewski, P.; Stepczyńska, M.; Jagodziński, B. Electroless metallization of polymers. Arch. Mater. Sci. Eng. 2015, 74, 67–76. [Google Scholar]
- Shao, Q.-S.; Bai, R.-C.; Tang, Z.-Y.; Gao, Y.-F.; Sun, J.-L.; Ren, M.-S. Durable electroless Ni and Ni-P-B plating on aromatic polysulfonamide (PSA) fibers with different performances via chlorine-aided silver activation strategy. Surf. Coat. Technol. 2016, 302, 185–194. [Google Scholar] [CrossRef]
- Lu, Y.; Xue, L.; Li, F. Silver nanoparticle catalyst for electroless Ni deposition and the promotion of its adsorption onto PET substrate. Surf. Coat. Technol. 2010, 205, 519–524. [Google Scholar] [CrossRef]
- Fatema, U.K.; Gotoh, Y. Iodine-aided palladium-free catalyzation process for durable electroless nickel plating on Kevlar® fiber. Surf. Coat. Technol. 2012, 206, 3472–3478. [Google Scholar] [CrossRef]
- Hajdu, J.; Mallory, G.O. Electroless Plating: Fundamentals and Applications; American Electroplaters and Surface Finishers Society: Orlando, FL, USA, 1990; pp. 193–206. [Google Scholar]
- Watanabe, H.; Honma, H. Direct electroless nickel plating on copper circuits using DMAB as a second reducing agent. In Proceedings of the 2nd 1998 IEMT/IMC Symposium (IEEE Cat. No.98EX225), Tokyo, Japan, 15–17 April 1998; pp. 149–153. [Google Scholar]
- Meyer, J. Eigenschaften und Anwendungen von Chemisch Nickel-Dispersionsschichten. Mater. Werkst. 2008, 39, 958–962. [Google Scholar] [CrossRef]
- Han, X.; Wang, G.; He, Y.; Wang, Y.; Qiao, Y.; Zhang, L. Surface modification of ABS with Cr6+ free etching process in the electroless plating. J. Adhes. Sci. Technol. 2018, 32, 2481–2493. [Google Scholar] [CrossRef]
- Zhai, T.; Chen, J.; Gui, B.; Wang, Q.; Yang, D.A. Electroless Deposition of Ni-P on Poly (ether ether ketone)/Multi-Walled Carbon Nanotubes Composite and Improvement of the Electrical Conductivity in Direction of Thickness. J. Electrochem. Soc. 2015, 162, D613–D618. [Google Scholar] [CrossRef]
- Agarwala, R.C.; Agarwala, V. Electroless alloy/composite coatings: A review. Sadhana 2003, 28, 475–493. [Google Scholar] [CrossRef]
- Schlesinger, M. Electroless Deposition of Nickel. Mod. Electroplat. 2011, 4, 447–458. [Google Scholar] [CrossRef]
- Abrantes, L.M.; Fundo, A.; Jin, G. Influence of phosphorus content on the structure of nickel electroless deposits. J. Mater. Chem. 2001, 11, 200–203. [Google Scholar] [CrossRef][Green Version]
- Mofidi, M.; Prakash, B. Influence of counterface topography on sliding friction and wear of some elastomers under dry sliding conditions. Proc. Inst. Mech. Eng. Part J J. Eng. Tribol. 2008, 222, 667–673. [Google Scholar] [CrossRef]
- Thirumalai, S.; Hausberger, A.; Lackner, J.; Waldhauser, W.; Schwarz, T. Effect of the type of elastomeric substrate on the microstructural, surface and tribological characteristics of diamond-like carbon (DLC) coatings. Surf. Coat. Technol. 2016, 302, 244–254. [Google Scholar] [CrossRef]
- Shen, M.-X.; Dong, F.; Zhang, Z.-X.; Meng, X.-K.; Peng, X. Effect of abrasive size on friction and wear characteristics of nitrile butadiene rubber (NBR) in two-body abrasion. Tribol. Int. 2016, 103, 1–11. [Google Scholar] [CrossRef]
- Persson, B.N.J. Rubber friction: Role of the flash temperature. J. Phys. Condens. Matter 2006, 18, 7789–7823. [Google Scholar] [CrossRef] [PubMed]
- Baek, D.K.; Khonsari, M. Fretting behavior of a rubber coating: Friction characteristics of rubber debris. Wear 2006, 261, 1114–1120. [Google Scholar] [CrossRef]
- Mang, T.; Bobzin, K.; Bartels, T. Industrial Tribology: Tribosystems, Friction, Wear and Surface Engineering, Lubrication; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Tuononen, A.J. Onset of frictional sliding of rubber–glass contact under dry and lubricated conditions. Sci. Rep. 2016, 6, 27951. [Google Scholar] [CrossRef] [PubMed]
- Brizmer, V.; Kligerman, Y.; Etsion, I. Elastic–plastic spherical contact under combined normal and tangential loading in full stick. Tribol. Lett. 2006, 25, 61–70. [Google Scholar] [CrossRef]
- Martinez-Martinez, D.; De Hosson, J. On the deposition and properties of DLC protective coatings on elastomers: A critical review. Surf. Coat. Technol. 2014, 258, 677–690. [Google Scholar] [CrossRef]
- Saravanan, I.; Elayaperumal, A.; Devaraju, A.; Karthikeyan, M.; Raji, A. Wear behaviour of electroless Ni-P and Ni-P-TiO2 composite coatings on En8 steel. Mater. Today Proc. 2020, 22, 1135–1139. [Google Scholar] [CrossRef]
- Tamilarasan, T.R.; Sanjith, U.; Rajendran, R.; Rajagopal, G.; Sudagar, J. Effect of Reduced Graphene Oxide Reinforcement on the Wear Characteristics of Electroless Ni-P Coatings. J. Mater. Eng. Perform. 2018, 27, 3044–3053. [Google Scholar] [CrossRef]
- Chang, S.-H.; Chang, C.-C.; Liang, C. Electroless Ni-P plating and Heat Treatments on S45C Carbon Steel. IOP Conf. Ser. Mater. Sci. Eng. 2013, 46, 012003. [Google Scholar] [CrossRef]
- Gawne, D.; Ma, U. Wear mechanisms in electroless nickel coatings. Wear 1987, 120, 125–149. [Google Scholar] [CrossRef]
- Sahoo, P.; Das, S.K. Tribology of electroless nickel coatings—A review. Mater. Des. 2011, 32, 1760–1775. [Google Scholar] [CrossRef]
- Kanani, N. Tribological Behaviour of Electroless Nickel. Trans. IMF 1992, 70, 14–18. [Google Scholar] [CrossRef]
- Behera, A.; Sahoo, A.K. Wear behaviour of Ni based superalloy: A review. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Barman, T.K.; Sahoo, P. Normal Partitions and Hierarchical Fillings of N-Dimensional Spaces; IGI Global: Hershey, PA, USA, 2018; pp. 297–331. [Google Scholar]
- Pei, Y.T.; Eivani, A.; Zaharia, T.; Kazantzis, A.; Van De Sanden, M.R.; De Hosson, J.T.M. High throughput deposition of hydrogenated amorphous carbon coatings on rubber with expanding thermal plasma. Surf. Coatings Technol. 2014, 245, 74–83. [Google Scholar] [CrossRef]
- Lubwama, M.; Corcoran, B.; McDonnell, K.; Dowling, D.; Kirabira, J.; Sebbit, A.; Sayers, K. Flexibility and frictional behaviour of DLC and Si-DLC films deposited on nitrile rubber. Surf. Coat. Technol. 2014, 239, 84–94. [Google Scholar] [CrossRef]
- Lubwama, M.; McDonnell, K.; Kirabira, J.; Sebbit, A.; Sayers, K.; Dowling, D.; Corcoran, B. Characteristics and tribological performance of DLC and Si-DLC films deposited on nitrile rubber. Surf. Coat. Technol. 2012, 206, 4585–4593. [Google Scholar] [CrossRef]
- Aoki, Y.; Ohtake, N. Tribological properties of segment-structured diamond-like carbon films. Tribol. Int. 2004, 37, 941–947. [Google Scholar] [CrossRef]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasconcelos, B.; Serra, R.; Oliveira, J.; Fonseca, C. Electroless Deposition of Ni-P Coatings on HNBR for Low Friction Rubber Seals. Coatings 2020, 10, 1237. https://doi.org/10.3390/coatings10121237
Vasconcelos B, Serra R, Oliveira J, Fonseca C. Electroless Deposition of Ni-P Coatings on HNBR for Low Friction Rubber Seals. Coatings. 2020; 10(12):1237. https://doi.org/10.3390/coatings10121237
Chicago/Turabian StyleVasconcelos, Beatriz, Ricardo Serra, João Oliveira, and Carlos Fonseca. 2020. "Electroless Deposition of Ni-P Coatings on HNBR for Low Friction Rubber Seals" Coatings 10, no. 12: 1237. https://doi.org/10.3390/coatings10121237
APA StyleVasconcelos, B., Serra, R., Oliveira, J., & Fonseca, C. (2020). Electroless Deposition of Ni-P Coatings on HNBR for Low Friction Rubber Seals. Coatings, 10(12), 1237. https://doi.org/10.3390/coatings10121237






