Exploration of Chemical Diversity and Antitrypanosomal Activity of Some Red Sea-Derived Actinomycetes Using the OSMAC Approach Supported by LC-MS-Based Metabolomics and Molecular Modelling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sarcophyton Glaucum Collection
2.2. Chemicals
2.3. Actinomycetes Isolation
2.4. Molecular Identification
2.5. Fermentation and Extraction
2.6. Metabolomic Profiling
2.7. In Vitro Antitrypanosomal Activity
2.8. Statistical and Multivariate Analysis
2.9. In Silico Biological Activity Predictions
2.10. Docking Experiments
2.11. Statistical Analysis
3. Results and Discussion
3.1. Identification and Phylogenetic Analysis of the Isolated Actinomycetes
3.2. In Vitro Antitrypanosomal Activity
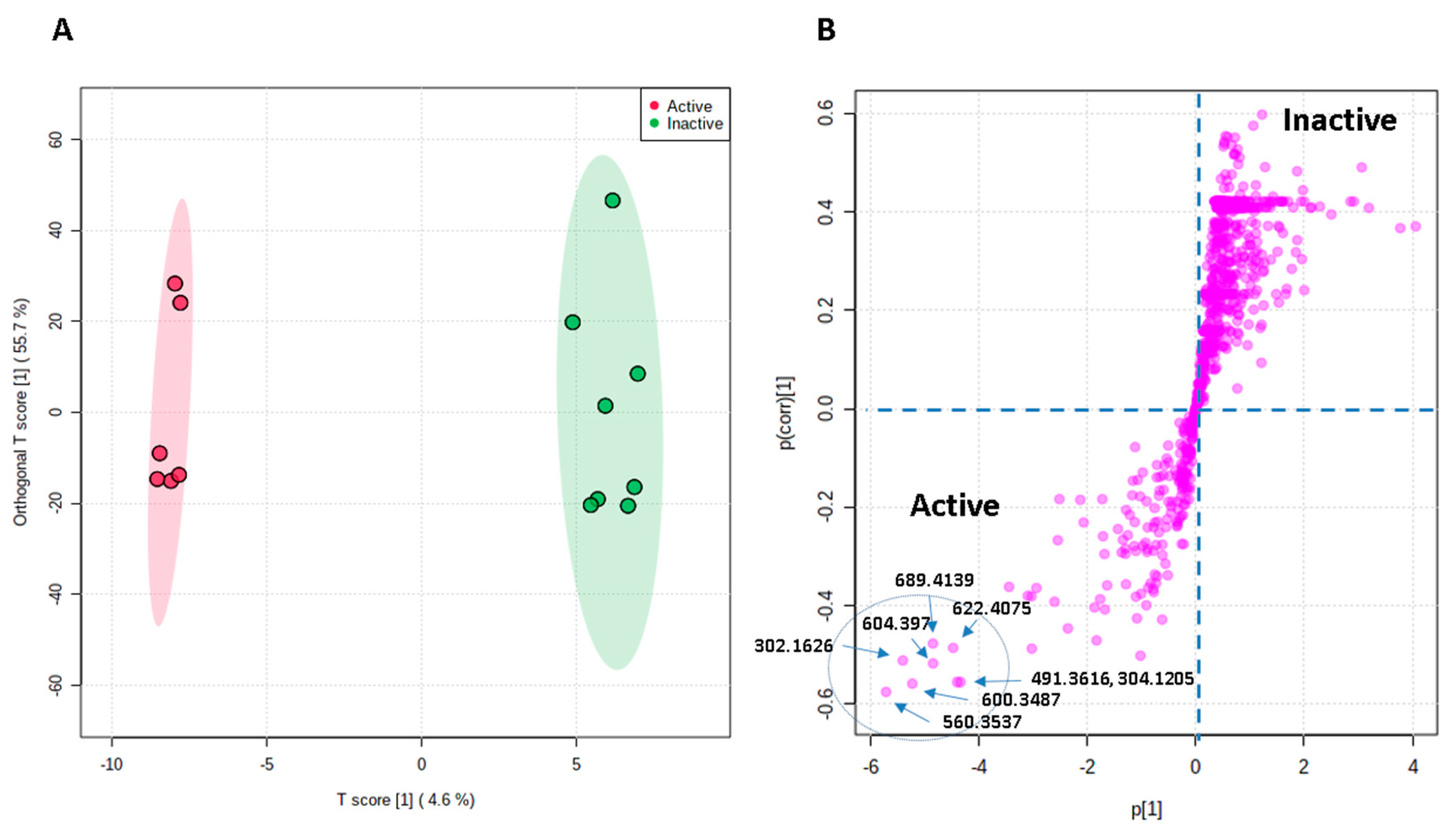
3.3. Metabolomic Profiling and Multivariate Analysis
3.4. Bioactivity–Metabolites Correlation
3.5. In Silico Predictions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hifnawy, M.S.; Fouda, M.M.; Sayed, A.M.; Mohammed, R.; Hassan, H.M.; Abouzid, S.F.; Rateb, M.E.; Keller, A.; Adamek, M.; Ziemert, N.; et al. The genus Micromonospora as a model microorganism for bioactive natural product discovery. RSC Adv. 2020, 10, 20939–20959. [Google Scholar] [CrossRef]
- Sayed, A.M.; Abdel-Wahab, N.M.; Hassan, H.M.; Abdelmohsen, U.R. Saccharopolyspora: An underexplored source for bioactive natural products. J. Appl. Microbiol. 2019, 128, 314–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Hawary, S.S.; Sayed, A.M.; Mohammed, R.; Khanfar, M.A.; Rateb, M.E.; Mohammed, T.A.; Hajjar, D.; Hassan, H.M.; Gulder, T.A.M.; Abdelmohsen, U. New Pim-1 Kinase Inhibitor from the Co-culture of Two Sponge-Associated Actinomycetes. Front. Chem. 2018, 6, 538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vicente, J.; Stewart, A.; Song, B.; Hill, R.T.; Wright, J.L. Biodiversity of Actinomycetes Associated with Caribbean Sponges and Their Potential for Natural Product Discovery. Mar. Biotechnol. 2013, 15, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Sayed, A.M.; Hassan, M.H.; Alhadrami, H.A.; Hassan, H.M.; Goodfellow, M.; Rateb, M.E. Extreme environments: Microbiology leading to specialized metabolites. J. Appl. Microbiol. 2019, 128, 630–657. [Google Scholar] [CrossRef] [Green Version]
- Shamikh, Y.I.; El Shamy, A.A.; Gaber, Y.; Abdelmohsen, U.; Madkour, M.H.; Horn, H.; Hassan, H.M.; Elmaidomy, A.H.; Alkhalifah, D.H.M.; Hozzein, W. Actinomycetes from the Red Sea Sponge Coscinoderma mathewsi: Isolation, Diversity, and Potential for Bioactive Compounds Discovery. Microorganisms 2020, 8, 783. [Google Scholar] [CrossRef]
- Abdelmohsen, U.; Bayer, K.; Hentschel, U. Diversity, abundance and natural products of marine sponge-associated actinomycetes. Nat. Prod. Rep. 2014, 31, 381–399. [Google Scholar] [CrossRef]
- Müller, R.; Wink, J. Future potential for anti-infectives from bacteria—How to exploit biodiversity and genomic potential. Int. J. Med. Microbiol. 2014, 304, 3–13. [Google Scholar] [CrossRef]
- Dalisay, D.S.; Williams, D.E.; Wang, X.L.; Centko, R.; Chen, J.; Andersen, R.J. Marine Sediment-Derived Streptomyces Bacteria from British Columbia, Canada are a Promising Microbiota Resource for the Discovery of Antimicrobial Natural Products. PLoS ONE 2013, 8, e77078. [Google Scholar] [CrossRef]
- Eltamany, E.E.; Abdelmohsen, U.; Ibrahim, A.K.; Hassanean, H.A.; Hentschel, U.; Ahmed, S.A. New antibacterial xanthone from the marine sponge-derived Micrococcus sp. EG45. Bioorg. Med. Chem. Lett. 2014, 24, 4939–4942. [Google Scholar] [CrossRef]
- Abdelmohsen, U.; Zhang, G.; Philippe, A.; Schmitz, W.; Pimentel-Elardo, S.M.; Hertlein-Amslinger, B.; Hentschel, U.; Bringmann, G. Cyclodysidins A–D, cyclic lipopeptides from the marine sponge-derived Streptomyces strain RV15. Tetrahedron Lett. 2012, 53, 23–29. [Google Scholar] [CrossRef]
- Abdelmohsen, U.; Szesny, M.; Othman, E.M.; Schirmeister, T.; Grond, S.; Stopper, H.; Hentschel, U. Antioxidant and Anti-Protease Activities of Diazepinomicin from the Sponge-Associated Micromonospora Strain RV115. Mar. Drugs 2012, 10, 2208–2221. [Google Scholar] [CrossRef] [PubMed]
- Subramani, R.; Aalbersberg, W. Marine actinomycetes: An ongoing source of novel bioactive metabolites. Microbiol. Res. 2012, 167, 571–580. [Google Scholar] [CrossRef]
- El-Hawary, S.S.; Sayed, A.M.; Mohammed, R.; Hassan, H.M.; Zaki, M.A.; Rateb, M.E.; Mohammed, T.A.; Amin, E.; Abdelmohsen, U. Epigenetic modifiers induce bioactive phenolic metabolites in the marine-derived fungus Penicillium brevicompactum. Mar. Drugs 2018, 16, 253. [Google Scholar] [CrossRef] [Green Version]
- Udwary, D.W.; Zeigler, L.; Asolkar, R.N.; Singan, V.R.; Lapidus, A.; Fenical, W.; Jensen, P.R.; Moore, B.S. Genome sequencing reveals complex secondary metabolome in the marine actinomycete Salinispora tropica. Proc. Natl. Acad. Sci. USA 2007, 104, 10376–11081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marmann, A.; Aly, A.H.; Lin, W.-H.; Wang, B.-G.; Proksch, P. Co-Cultivation—A Powerful Emerging Tool for Enhancing the Chemical Diversity of Microorganisms. Mar. Drugs 2014, 12, 1043–1065. [Google Scholar] [CrossRef] [Green Version]
- Hifnawy, M.S.; Hassan, H.M.; Mohammed, R.; Fouda, M.M.; Sayed, A.M.; Hamed, A.A.; Abouzid, S.F.; Rateb, M.E.; Alhadrami, H.A.; Abdelmohsen, U. Induction of Antibacterial Metabolites by Co-Cultivation of Two Red-Sea-Sponge-Associated Actinomycetes Micromonospora sp. UR56 and Actinokinespora sp. EG49. Mar. Drugs 2020, 18, 243. [Google Scholar] [CrossRef]
- Abdelmohsen, U.; Grkovic, T.; Balasubramanian, S.; Kamel, M.S.; Quinn, R.J.; Hentschel, U. Elicitation of secondary metabolism in actinomycetes. Biotechnol. Adv. 2015, 33, 798–811. [Google Scholar] [CrossRef] [Green Version]
- Bode, H.B.; Bethe, B.; Höfs, R.; Zeeck, A. Big Effects from Small Changes: Possible Ways to Explore Nature’s Chemical Diversity. ChemBioChem 2002, 3, 619–627. [Google Scholar] [CrossRef]
- Paranagama, P.A.; Wijeratne, E.M.K.; Gunatilaka, A.A.L. Uncovering Biosynthetic Potential of Plant-Associated Fungi: Effect of Culture Conditions on Metabolite Production by Paraphaeosphaeria quadriseptata and Chaetomium chiversii. J. Nat. Prod. 2007, 70, 1939–1945. [Google Scholar] [CrossRef]
- Wei, H.; Lin, Z.; Li, D.; Gu, Q.; Zhu, T. OSMAC (one strain many compounds) approach in the research of microbial metabolites—A review. Wei Sheng Wu Xue Bao 2010, 50, 701–709. [Google Scholar] [PubMed]
- Abdelmohsen, U.; Cheng, C.; Viegelmann, C.; Zhang, T.; Grkovic, T.; Ahmed, S.A.; Quinn, R.J.; Hentschel, U.; Edrada-Ebel, R. Dereplication strategies for targeted isolation of new antitrypanosomal actinosporins A and B from a marine sponge associated-Actinokineospora sp. EG49. Mar. Drugs 2014, 12, 1220–1244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Letunic, I.; Bork, P. Interactive Tree of Life (iTOL) v4: Recent updates and new developments. Nucleic Acids Res. 2019, 47, W256–W259. [Google Scholar] [CrossRef] [Green Version]
- Huber, W.; Koella, J.C. A comparison of three methods of estimating EC50 in studies of drug resistance of malaria parasites. Acta Trop. 1993, 55, 257–261. [Google Scholar] [CrossRef]
- Xia, J.; Psychogios, N.; Young, N.; Wishart, D.S. MetaboAnalyst: A web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009, 37, W652–W660. [Google Scholar] [CrossRef] [Green Version]
- Lagunin, A.A.; Stepanchikova, A.; Filimonov, D.A.; Poroikov, V. PASS: Prediction of activity spectra for biologically active substances. Bioinformatics 2000, 16, 747–748. [Google Scholar] [CrossRef]
- Seeliger, D.; De Groot, B.L. Ligand docking and binding site analysis with PyMOL and Autodock/Vina. J. Comput. Mol. Des. 2010, 24, 417–422. [Google Scholar] [CrossRef] [Green Version]
- Lill, M.A.; Danielson, M.L. Computer-aided drug design platform using PyMOL. J. Comput. Mol. Des. 2010, 25, 13–19. [Google Scholar] [CrossRef]
- Sayed, A.M.; Alhadrami, H.A.; El-Hawary, S.S.; Mohammed, R.; Hassan, H.M.; Rateb, M.E.; Abdelmohsen, U.; Bakeer, W. Discovery of Two Brominated Oxindole Alkaloids as Staphylococcal DNA Gyrase and Pyruvate Kinase Inhibitors via Inverse Virtual Screening. Microorganisms 2020, 8, 293. [Google Scholar] [CrossRef] [Green Version]
- Tindall, B.J.; Busse, H.-J.; Ludwig, W.; Rosselló-Móra, R.; Kämpfer, P. Notes on the characterization of prokaryote strains for taxonomic purposes. Int. J. Syst. Evol. Microbiol. 2010, 60, 249–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaewkla, O.; Thamchaipinet, A.; Franco, C.M.M. Micromonospora terminaliae sp. nov., an endophytic actinobacterium isolated from the surface-sterilized stem of the medicinal plant Terminalia mucronata. Int. J. Syst. Evol. Microbiol. 2017, 67, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Carro, L.; Pukall, R.; Spröer, C.; Kroppenstedt, R.M.; Trujillo, M.E. Micromonospora cremea sp. nov. and Micromonospora zamorensis sp. nov., isolated from the rhizosphere of Pisum sativum. Int. J. Syst. Evol. Microbiol. 2012, 62, 2971–2977. [Google Scholar] [CrossRef]
- Fang, B.; Liu, C.; Guan, X.; Song, J.; Zhao, J.; Liu, H.; Li, C.; Ning, W.; Wang, X.; Xiang, W.-S. Two new species of the genus Micromonospora: Micromonospora palomenae sp. nov. and Micromonospora harpali sp. nov. isolated from the insects. Antonie van Leeuwenhoek 2015, 108, 141–150. [Google Scholar] [CrossRef]
- Kasai, H.; Tamura, T.; Harayama, S. Intrageneric relationships among Micromonospora species deduced from gyrB-based phylogeny and DNA relatedness. Int. J. Syst. Evol. Microbiol. 2000, 50, 127–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senges, C.H.R.; Al-Dilaimi, A.; Marchbank, D.H.; Wibberg, D.; Winkler, A.; Haltli, B.; Nowrousian, M.; Kalinowski, J.; Kerr, R.G.; Bandow, J.E. The secreted metabolome of Streptomyces chartreusis and implications for bacterial chemistry. Proc. Natl. Acad. Sci. USA 2018, 115, 2490–2495. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, M.H.; Wu, Y.X.; Xu, Y.N.; Liu, P.; Yu, L.Y.; Hong, B.; Jiang, W.; Si, S.-Y. A new trichostatin analog from Streptomyces sp. CPCC 203909. J. Asian Nat. Prod. Res. 2015, 17, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Ueki, M.; Teruya, T.; Nie, L.; Usami, R.; Yoshida, M.; Osada, H. A new trichostatin derivative, trichostatin RK, from Streptomyces sp. RK98-A74. J. Antibiot. 2001, 54, 1093–1095. [Google Scholar] [CrossRef] [Green Version]
- Georgousaki, K.; Tsafantakis, N.; Gumeni, S.; Gonzalez, I.; MacKenzie, T.A.; Reyes, F.; Lambert, C.; Trougakos, I.P.; Genilloud, O.; Fokialakis, N. Screening for tyrosinase inhibitors from actinomycetes; identification of trichostatin derivatives from Streptomyces sp. CA-129531 and scale up production in bioreactor. Bioorg. Med. Chem. Lett. 2020, 30, 126952. [Google Scholar] [CrossRef]
- Kretschmer, A.; Dorgerloh, M.; Deeg, M.; Hagenmaier, H. The structures of novel insecticidal macrolides: Bafilomycins D and E, and oxohygrolidin. Agric. Biol. Chem. 1985, 49, 2509–2511. [Google Scholar]
- Fehr, T.; Sanglier, J.J.; Schuler, W.; Gschwind, L.; Ponelle, M.; Schilling, W.; Wloland, C. Antascomicins A, B, C, D and E novel FKBP12 binding compounds from a Micromonospora strain. J. Antibiot. 1996, 49, 230–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gagliardi, S.; Rees, M.; Farina, C. Chemistry and structure activity relationships of bafilomycin A1, a potent and selective inhibitor of the vacuolar H+-ATPase. Curr. Med. Chem. 1999, 6, 1197–1212. [Google Scholar] [CrossRef] [PubMed]
- Weibel, E.K.; Hadvary, P.; Hochuli, E.; Kupfer, E.; Lengsfeld, H. Lipstatin, an inhibitor of pancreatic lipase, produced by Streptomyces toxytricini. J. Antibiot. 1987, 40, 1081–1085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, P.-Y.; Wang, M.; Liu, K.; Ngai, M.H.; Sheriff, O.; Lear, M.J.; Sze, S.K.; He, C.Y.; Yao, S.Q. Parasite-Based Screening and Proteome Profiling Reveal Orlistat, an FDA-Approved Drug, as a Potential Anti Trypanosoma brucei Agent. Chem. A Eur. J. 2012, 18, 8403–8413. [Google Scholar] [CrossRef]
- Yang, L.M.; Rong-Yang, W.; McPhail, A.T.; Yokoi, T.; Lee, K.H. Neihumicin, a new cytotoxic antibiotic from Micromonospora neihuensis. J. Antibiot. 1988, 41, 488–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]



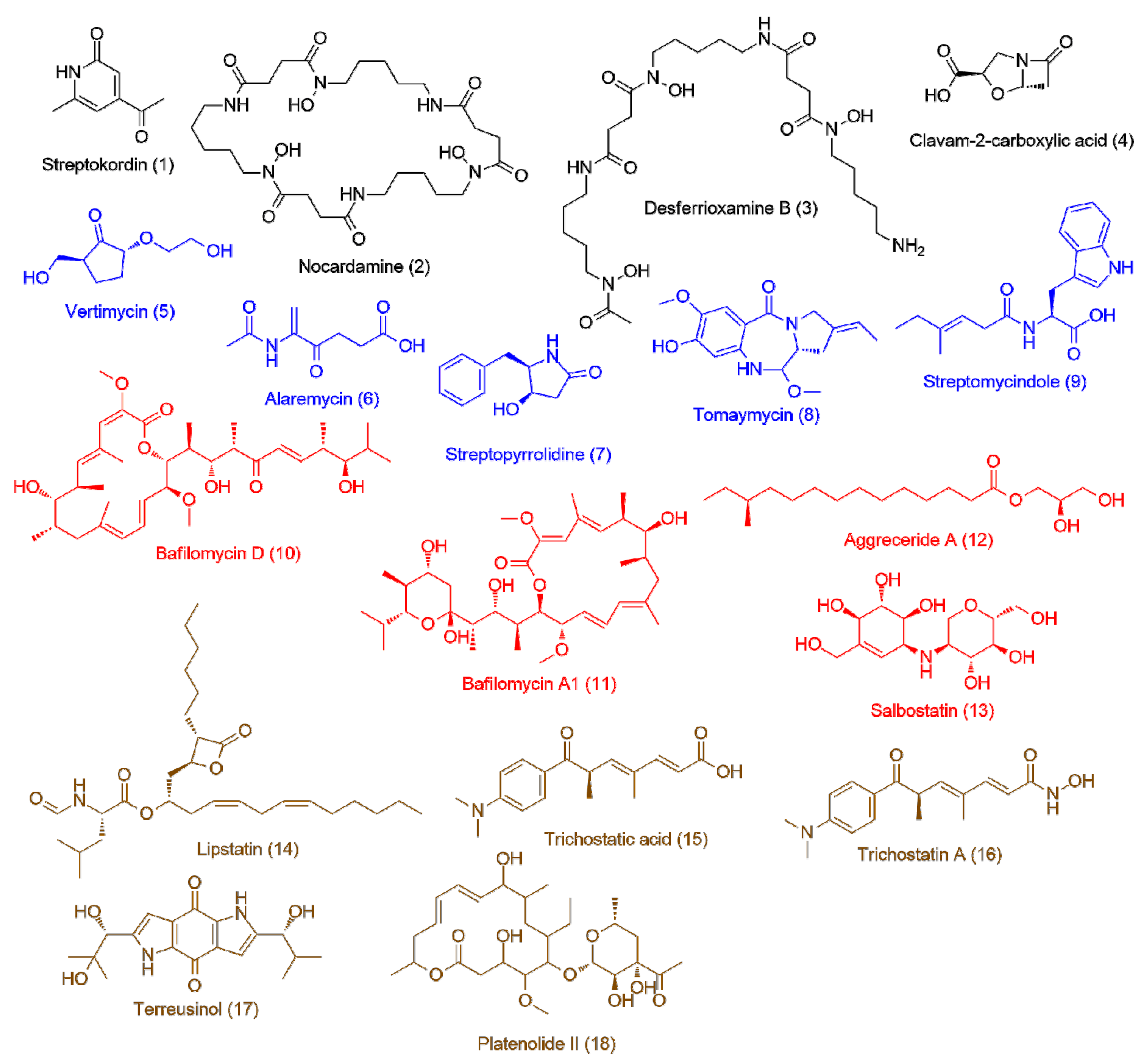
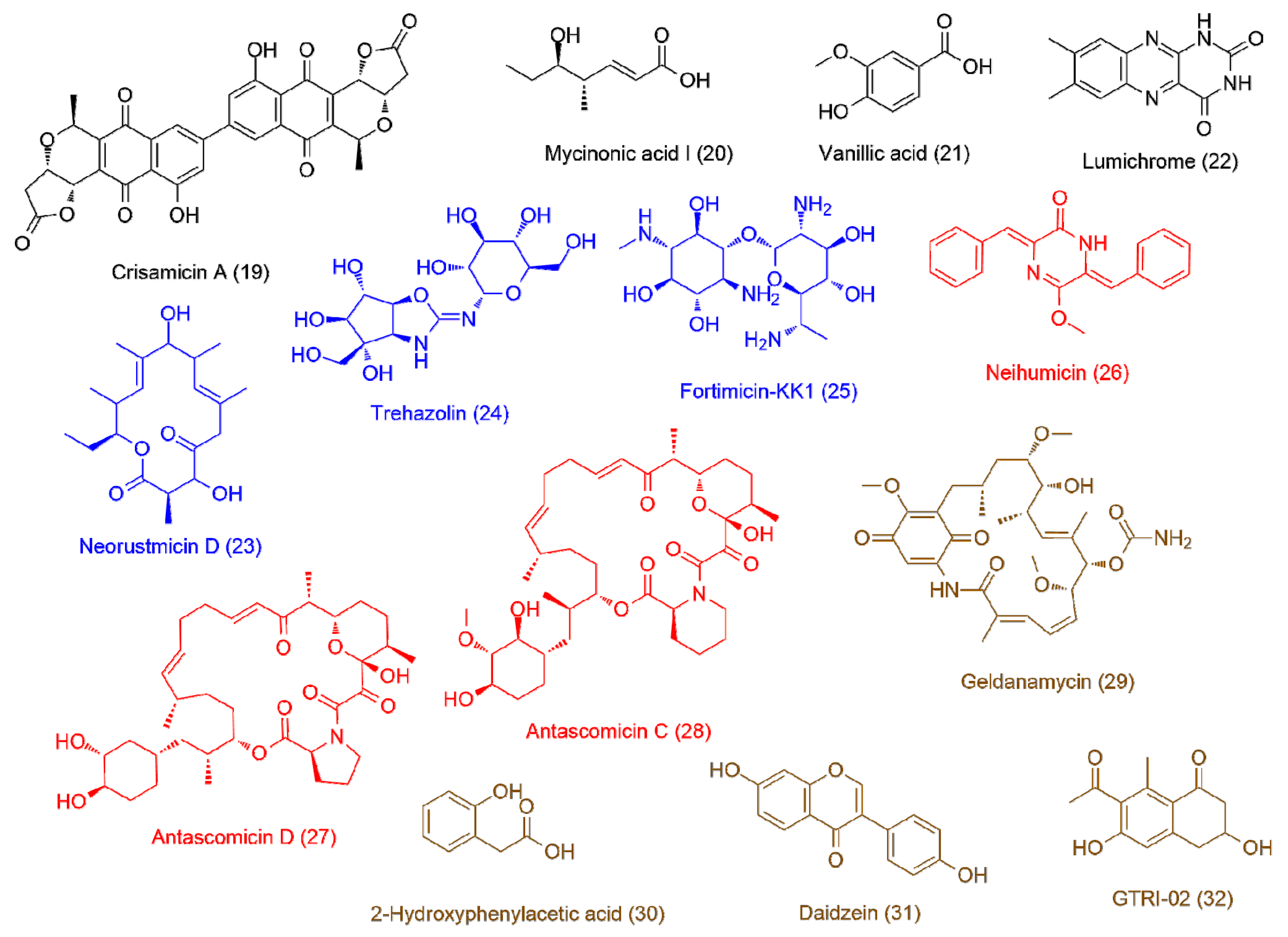
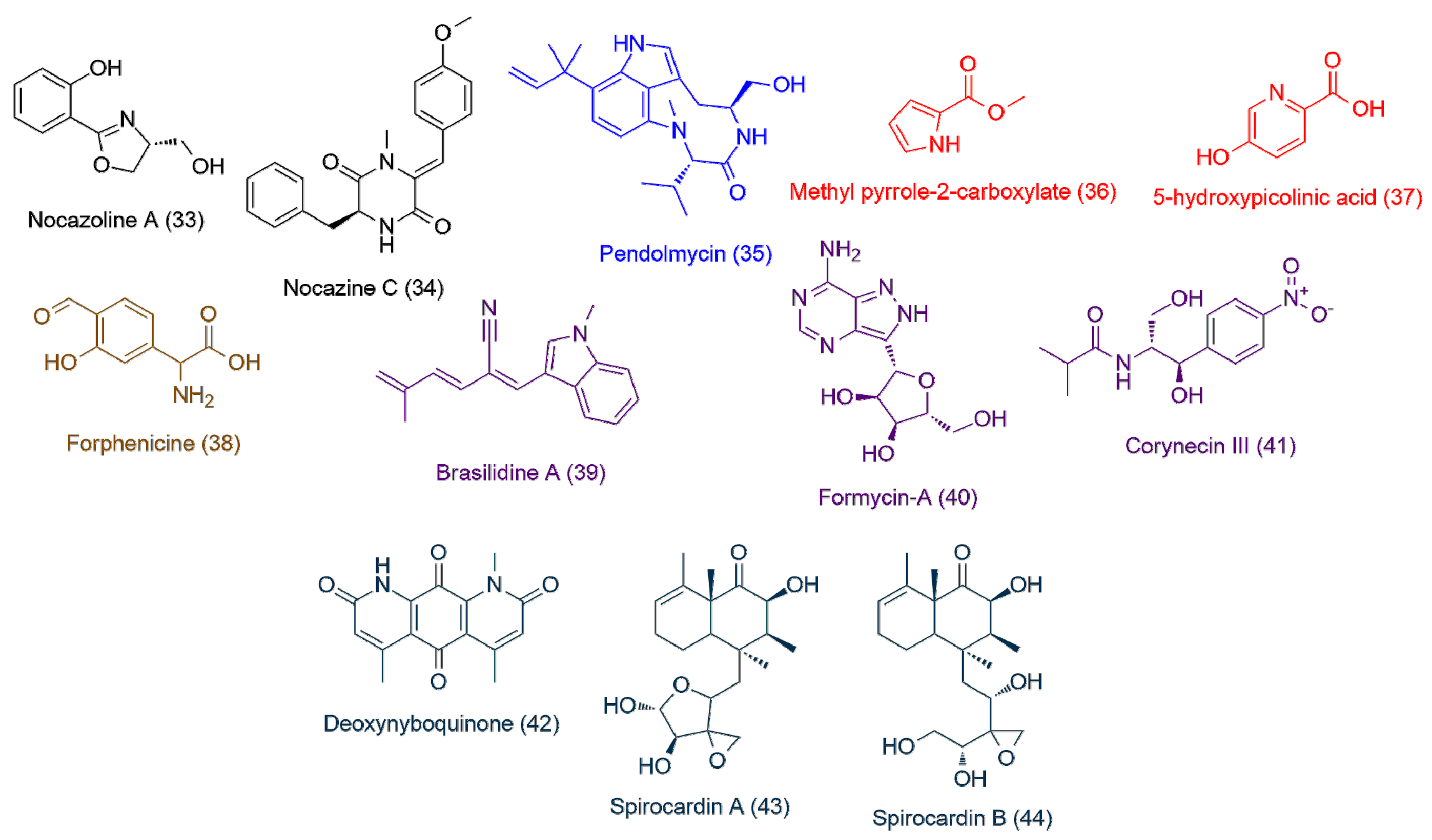



| Isolate | Accession ID | Identity [%] | Source | Ref |
|---|---|---|---|---|
| Micromonospora terminaliae DSM 101760 | CP045309.1 | 93.28 | Surface sterilized stem of Thai medicinal plant Terminalia mucronata | [32] |
| Micromonospora cremea CR30 | NR_108478.1 | 93.00 | rhizosphere of Pisum sativum | [33] |
| Micromonospora palomenae NEAU-CX1 | NR_136848.1 | 92.81 | Nymphs of stinkbug (Palomena viridissima Poda) | [34] |
| Micromonospora rosaria DSM 803 | NR_026282.1 | 92.75 | unknown | [35] |
| Culture Condition (IC50) * | No. | Retention Time | Mass | Molecular Formula | Dereplication | VIP ** Score |
|---|---|---|---|---|---|---|
| Streptomyces sp. UR23 on solid media (16.6 ± 1.8) | 1 | 2.81 | 151.0623 | C8H9NO2 | Streptokordin | 2.35 |
| 2 | 3.44 | 600.3487 | C27H48N6O9 | Nocardamine | 2.22 | |
| 3 | 8.83 | 560.3537 | C25H48N6O8 | Desferrioxamine B | 2.15 | |
| 4 | 1.81 | 157.0368 | C6H7NO4 | Clavam-2-carboxylic acid | 1.86 | |
| Streptomyces sp. UR23 in liquid media (>100) | 5 | 2.76 | 174.0881 | C8H14O4 | Vertimycin | 2.2 |
| 6 | 2.06 | 185.0681 | C8H11NO4 | Alaremycin | 1.98 | |
| 7 | 3.72 | 191.0947 | C11H13NO2 | Streptopyrrolidine | 1.91 | |
| 8 | 4.1 | 304.1408 | C16H20N2O4 | Tomaymycin | 1.85 | |
| 9 | 3.21 | 314.1629 | C18H22N2O3 | Streptomycindole | 1.82 | |
| Streptomyces sp. UR23 with Nocardia sp. UR27 on solid media (4.6 ± 1.5) | 10 | 7.81 | 604.397 | C35H56O8 | Bafilomycin D | 1.97 |
| 11 | 7.89 | 622.4075 | C35H58O9 | Bafilomycin A1 | 1.8 | |
| 12 | 4.57 | 316.2599 | C18H36O4 | Aggreceride A | 1.75 | |
| 13 | 1.86 | 321.1425 | C13H23NO8 | Salbostatin | 1.73 | |
| Streptomyces sp. UR23 with Nocardia sp. UR27 in liquid media (2.4 ± 1.1) | 14 | 11.85 | 491.3616 | C29H49NO5 | Lipstatin | 2.29 |
| 15 | 4.19 | 287.1518 | C17H21NO3 | Trichostatic acid | 1.87 | |
| 16 | 4.14 | 302.1626 | C17H22N2O3 | Trichostatin A | 1.77 | |
| 17 | 3.71 | 346.1522 | C18H22N2O5 | Terreusinol | 1.71 | |
| 18 | 3.32 | 370.2339 | C20H34O6 | Platenolide II | 1.68 | |
| Micromonospora sp. UR17 on solid media (7.8 ± 1.2) | 19 | 5.13 | 598.1115 | C32H22O12 | Crisamicin A | 2.33 |
| 20 | 3.14 | 158.0934 | C8H14O3 | Mycinonic acid I | 2.11 | |
| 21 | 2.49 | 168.0414 | C8H8O4 | Vanillic acid | 1.83 | |
| 22 | 2.13 | 242.0793 | C12H10N4O2 | Lumichrome | 1.66 | |
| Micromonospora sp. UR17 in liquid media (>100) | 23 | 3.91 | 352.2236 | C20H32O5 | Neorustmicin D | 2.22 |
| 24 | 2.55 | 366.1274 | C13H22N2O10 | Trehazolin | 1.95 | |
| 25 | 3.96 | 366.2106 | C14H30N4O7 | Fortimicin-KK1 | 1.87 | |
| Micromonospora sp. UR17 with Nocardia sp. UR27 on solid media (2.7 ± 0.7) | 26 | 3.54 | 304.1205 | C19H16N2O2 | Neihumicin | 1.77 |
| 27 | 5.89 | 645.3873 | C36H66NO9 | Antascomicin D | 1.71 | |
| 28 | 5.23 | 689.4139 | C38H59NO10 | Antascomicin C | 1.65 | |
| Micromonospora sp. UR17 with Nocardia sp. UR27 in liquid media (2.5 ± 0.9) | 29 | 4.43 | 560.2731 | C29H40N2O9 | Geldanamycin | 1.86 |
| 30 | 2.22 | 152.0467 | C8H8O3 | 2-Hydroxyphenylacetic acid | 1.82 | |
| 31 | 3.31 | 254.0573 | C15H10O4 | Daidzein | 1.73 | |
| 32 | 2.79 | 234.0888 | C13H14O4 | GTRI-02 | 1.66 | |
| Nocardiopsis sp. UR17 in solid media (>100) | 33 | 2.29 | 193.0736 | C10H11NO3 | Nocazoline A | 1.84 |
| 34 | 3.73 | 336.1473 | C20H20N2O3 | Nocazine C | 1.71 | |
| Nocardiopsis sp. UR17 on liquid media (>100) | 35 | 4.24 | 369.2418 | C22H31N3O2 | Pendolmycin | 1.64 |
| Nocardiopsis sp. UR17 with Nocardia sp. UR27 on solid media (>100) | 36 | 2.64 | 125.0471 | C6H7NO2 | Methyl pyrrole-2-carboxylate | 1.99 |
| 37 | 3.87 | 139.0272 | C6H5NO3 | 5-hydroxypicolinic acid | 1.85 | |
| Nocardiopsis sp. UR17 with Nocardia sp. UR27 in liquid media (>100) | 38 | 2.41 | 195.0524 | C9H9NO4 | Forphenicine | 1.76 |
| Nocardia sp. UR27 on solid media (>100) | 39 | 2.66 | 248.1310 | C17H16N2 | Brasilidine A | 2.13 |
| 40 | 1.72 | 267.0957 | C10H13N5O4 | Formycin-A | 1.92 | |
| 41 | 2.56 | 282.1204 | C13H18N2O5 | Corynecin III | 1.7 | |
| Nocardia sp. UR27 in liquid media (>100) | 42 | 3.86 | 284.0790 | C15H12N2O4 | Deoxynyboquinone | 1.98 |
| 43 | 3.7 | 366.2039 | C20H30O6 | Spirocardin A | 1.73 | |
| 44 | 2.92 | 368.2188 | C20H32O6 | Spirocardin B | 1.71 |
| NO. | Dereplication | Mass | Molecular Formula | Reported Activity | Probability p < 0.01 |
|---|---|---|---|---|---|
| 3 | Desferrioxamine B | 560.3537 | C25H48N6O8 | Siderophore | 2.42 × 10−6 |
| 2 | Nocardamine | 600.3487 | C27H48N6O9 | Siderophore | 3.33 × 10−5 |
| 16 | Trichostatin A | 302.1626 | C17H22N2O3 | Histone deacetylase and tyrosinase inhibitor | 1.22 × 10−5 |
| 10 | Bafilomycin D | 604.397 | C35H56O8 | Insecticidal activity | 5.43 × 10−4 |
| 28 | Antascomicin C | 689.4139 | C38H59NO10 | Antimicrobial | 2.66 × 10−4 |
| 11 | Bafilomycin A1 | 622.4075 | C35H58O9 | H+-ATPase inhibitor | 1.12 × 10−4 |
| 14 | Lipstatin | 491.3616 | C29H49NO5 | Pancreatic lipase inhibitor | 6.89 × 10−3 |
| 26 | Neihumicin | 304.1205 | C19H16N2O2 | Cytotoxic | 3.11 × 10−3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gamaleldin, N.M.; Bakeer, W.; Sayed, A.M.; Shamikh, Y.I.; El-Gendy, A.O.; Hassan, H.M.; Horn, H.; Abdelmohsen, U.R.; Hozzein, W.N. Exploration of Chemical Diversity and Antitrypanosomal Activity of Some Red Sea-Derived Actinomycetes Using the OSMAC Approach Supported by LC-MS-Based Metabolomics and Molecular Modelling. Antibiotics 2020, 9, 629. https://doi.org/10.3390/antibiotics9090629
Gamaleldin NM, Bakeer W, Sayed AM, Shamikh YI, El-Gendy AO, Hassan HM, Horn H, Abdelmohsen UR, Hozzein WN. Exploration of Chemical Diversity and Antitrypanosomal Activity of Some Red Sea-Derived Actinomycetes Using the OSMAC Approach Supported by LC-MS-Based Metabolomics and Molecular Modelling. Antibiotics. 2020; 9(9):629. https://doi.org/10.3390/antibiotics9090629
Chicago/Turabian StyleGamaleldin, Noha M., Walid Bakeer, Ahmed M. Sayed, Yara I. Shamikh, Ahmed O. El-Gendy, Hossam M. Hassan, Hannes Horn, Usama Ramadan Abdelmohsen, and Wael N. Hozzein. 2020. "Exploration of Chemical Diversity and Antitrypanosomal Activity of Some Red Sea-Derived Actinomycetes Using the OSMAC Approach Supported by LC-MS-Based Metabolomics and Molecular Modelling" Antibiotics 9, no. 9: 629. https://doi.org/10.3390/antibiotics9090629







