Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series
Abstract
1. Introduction
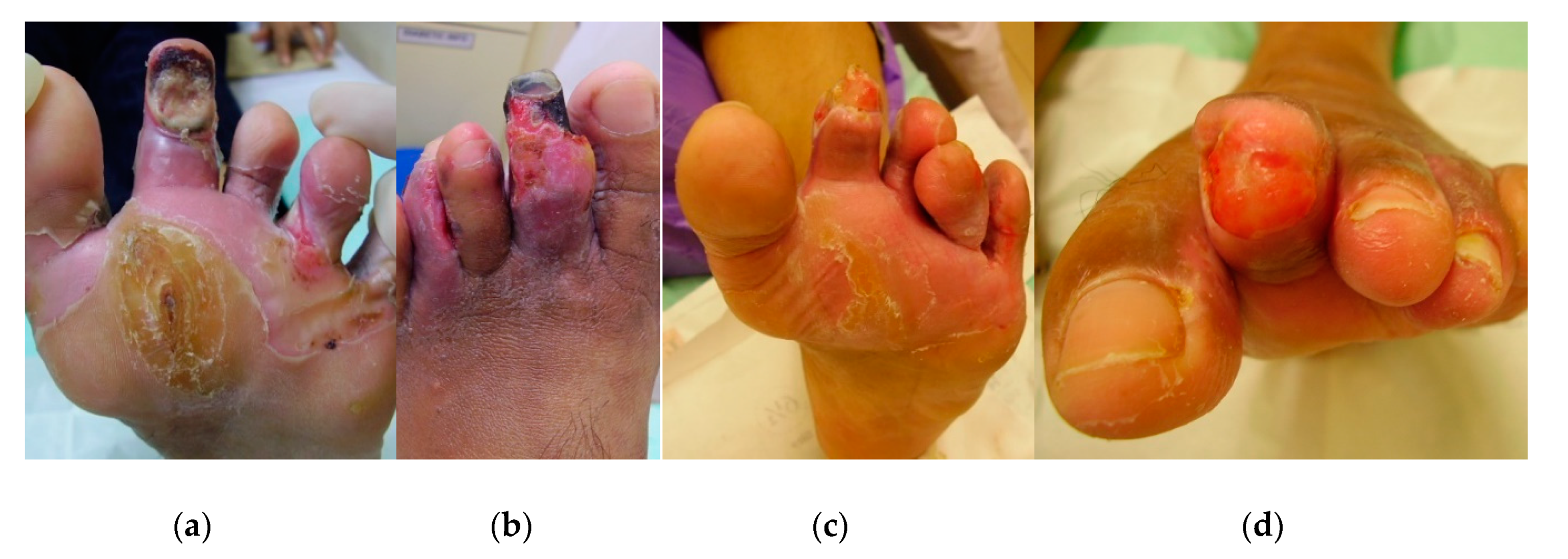
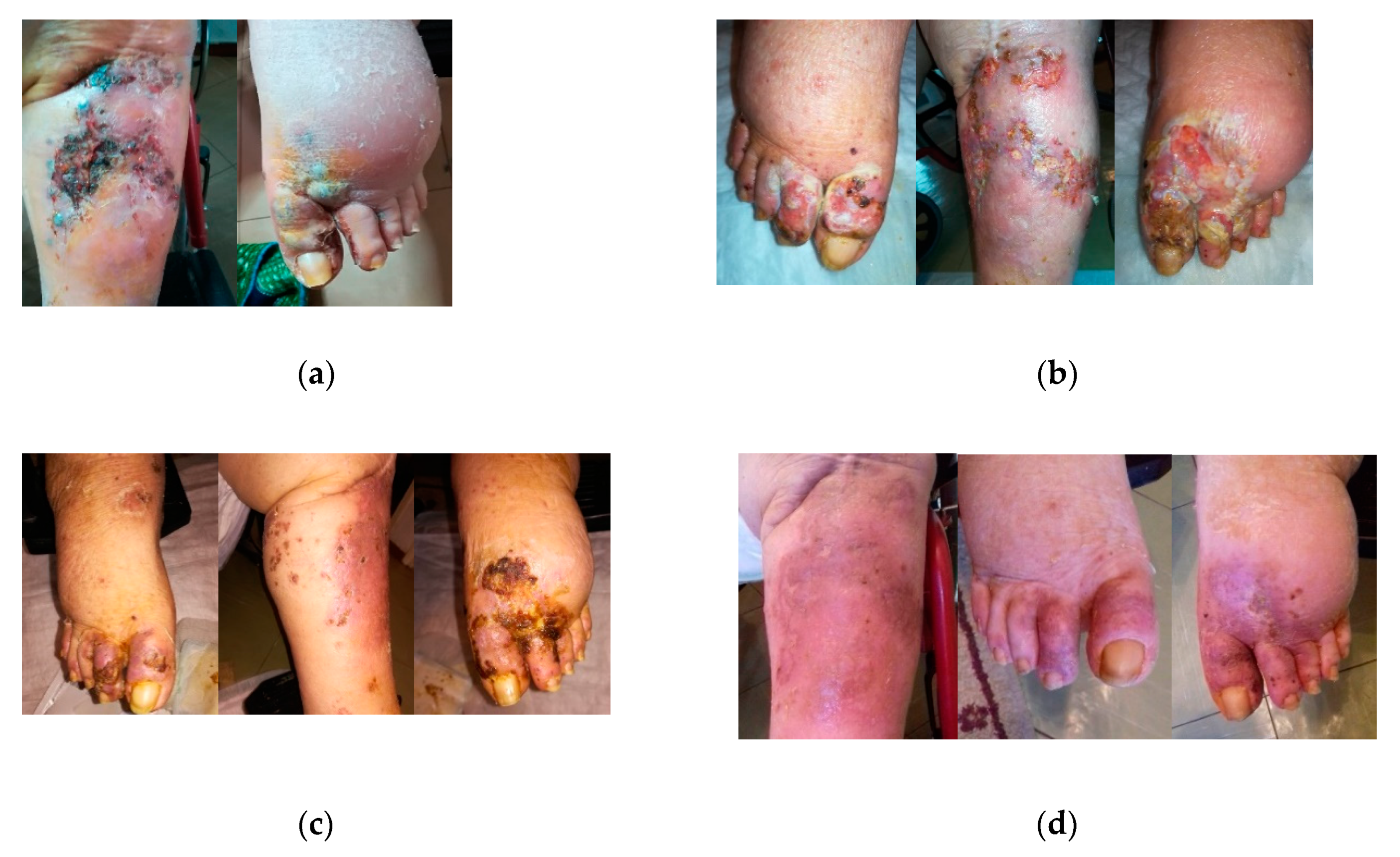
2. Results
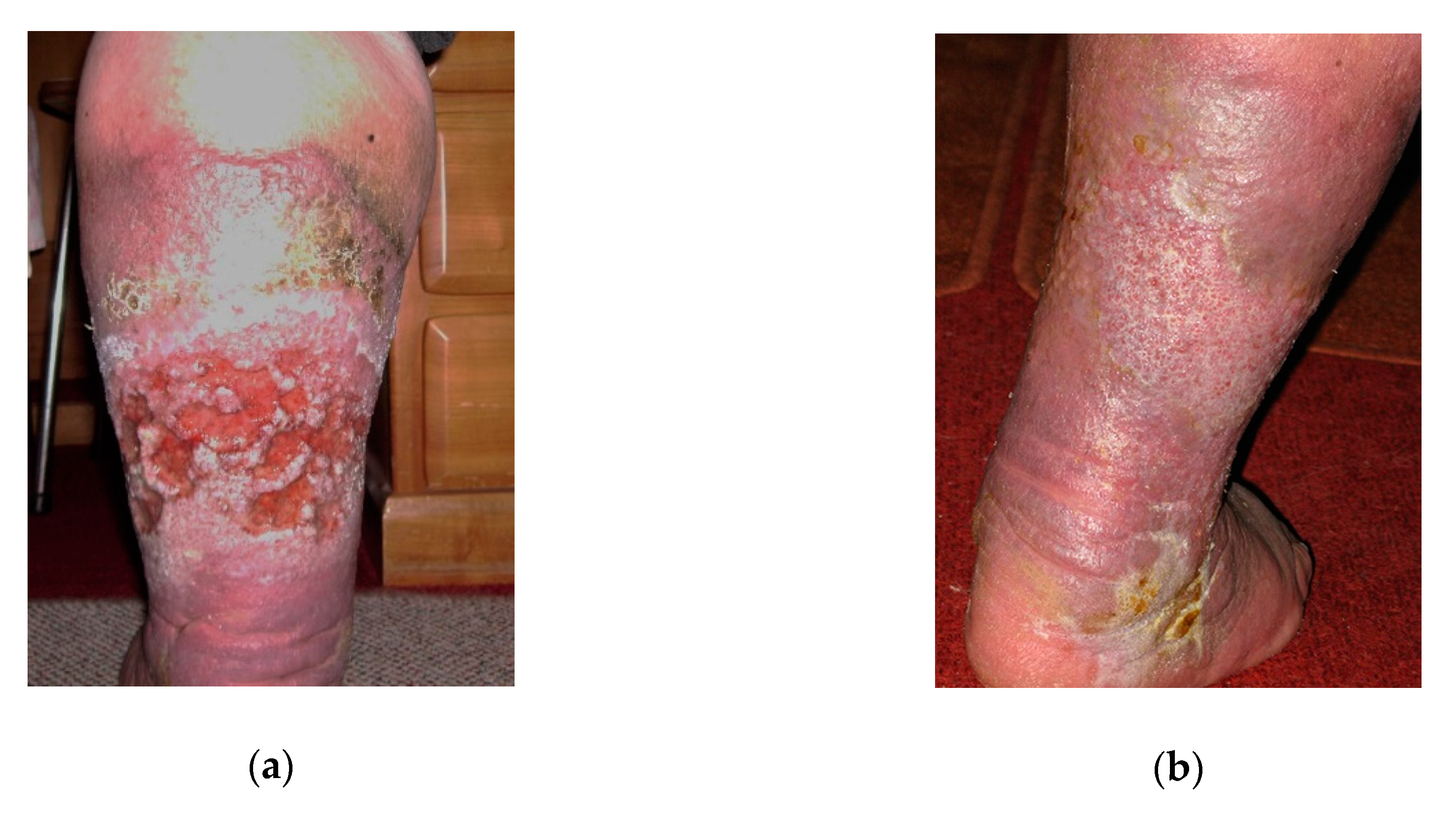
2.1. Case 1
2.2. Case 2
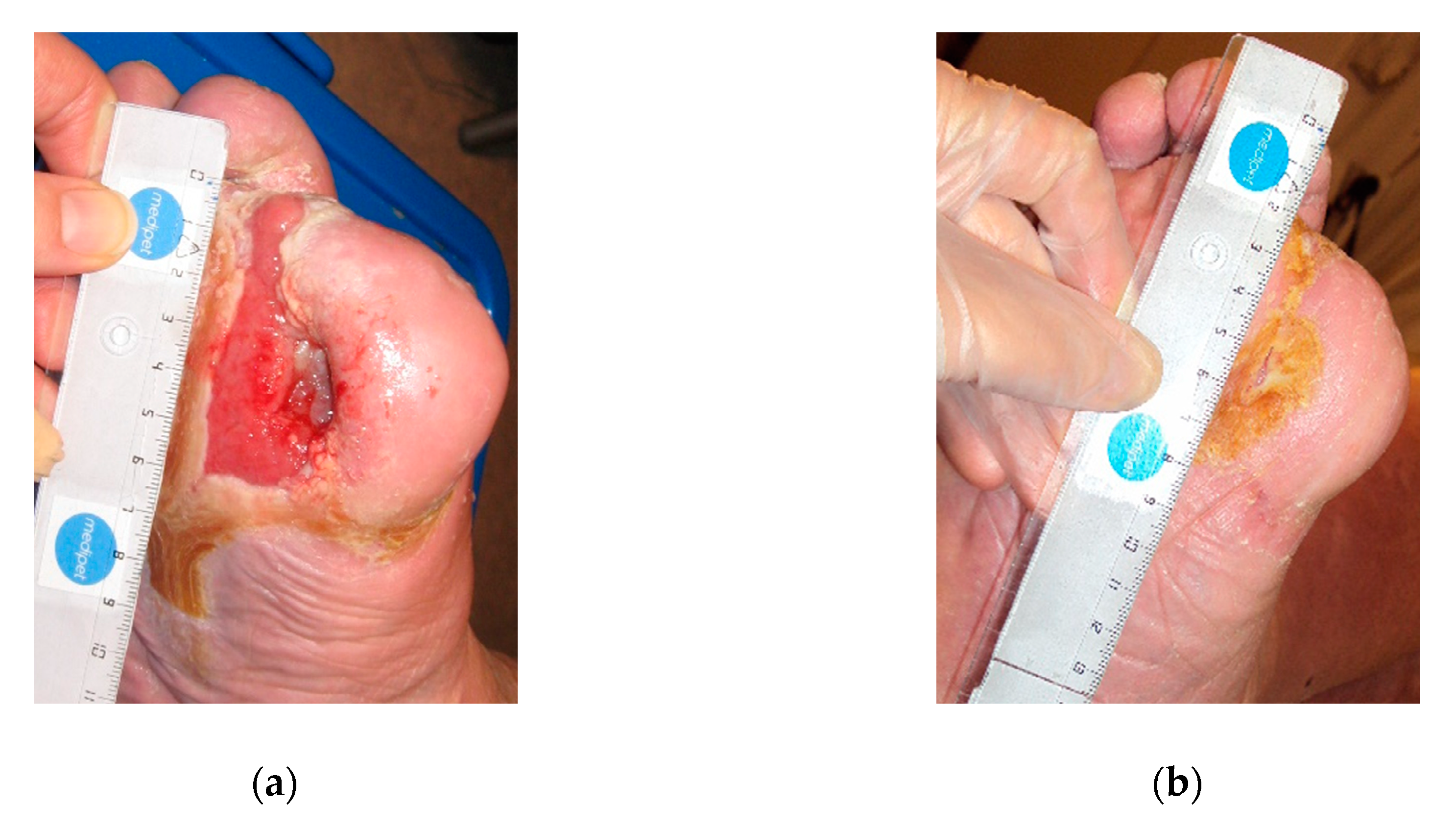
2.3. Case 3
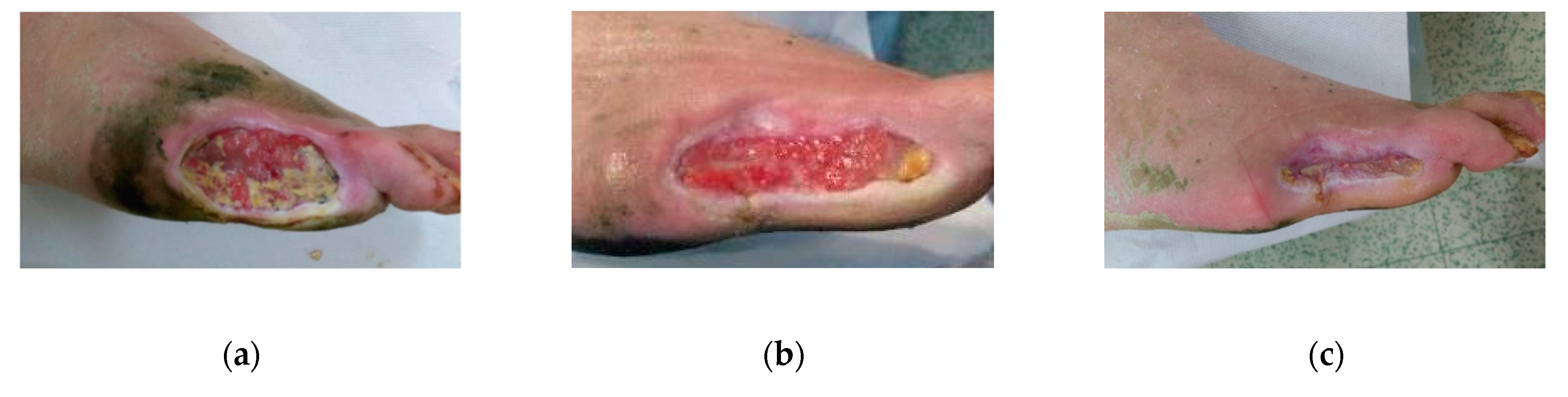
2.4. Case 4
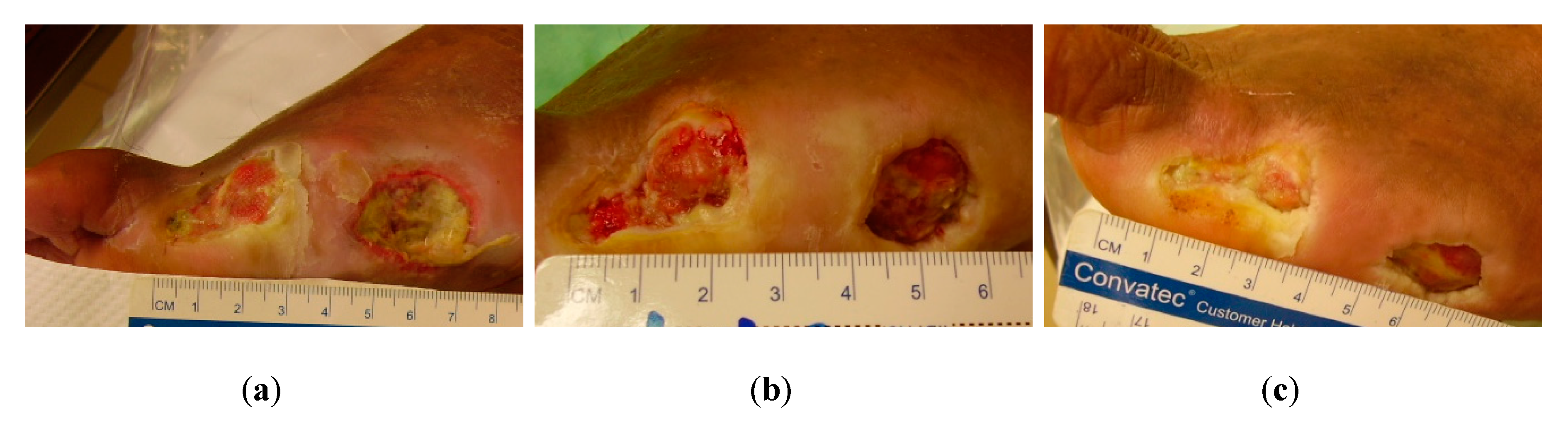
2.5. Case 5
2.6. Case 6
3. Discussion
4. Materials and Methods
4.1. Subjects and therapeutic interventions
4.2. About L-Mesitran wound care products
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Baena-Diez, J.M.; Penafiel, J.; Subirana, I.; Ramos, R.; Elosua, R.; Marin-Ibanez, A.; Guembe, M.J.; Rigo, F.; Tormo-Diaz, M.J.; Moreno-Iribas, C.; et al. Risk of Cause-Specific Death in Individuals With Diabetes: A Competing Risks Analysis. Diabetes Care 2016, 39, 1987–1995. [Google Scholar] [CrossRef] [PubMed]
- van Netten, J.J.; Bus, S.A.; Apelqvist, J.; Lipsky, B.A.; Hinchliffe, R.J.; Game, F.; Rayman, G.; Lazzarini, P.A.; Forsythe, R.O.; Peters, E.J.G.; et al. Definitions and criteria for diabetic foot disease. Diabetes Metab Res. Rev. 2020, 36 (Suppl. S1), e3268. [Google Scholar] [CrossRef] [PubMed]
- Brem, H.; Tomic-Canic, M. Cellular and molecular basis of wound healing in diabetes. J. Clin. Investig. 2007, 117, 1219–1222. [Google Scholar] [CrossRef]
- Smaropoulos, E.; Cremers, N.A.J. Treating severe wounds in pediatrics with medical grade honey: A case series. Clin. Case Rep. 2020, 8, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Molan, P.C. Re-introducing honey in the management of wounds and ulcers – theory and practice. Ostomy Wound Manage. 2002, 48, 28–40. [Google Scholar]
- Hermanns, R.; Mateescu, C.; Thrasyvoulou, A.; Tananaki, C.; Wagener, F.A.; Cremers, N.A. Defining the standards for medical grade honey. J. Apic. Res. 2020, 59, 125–135. [Google Scholar] [CrossRef]
- Postmes, T.; van den Bogaard, A.E.; Hazen, M. Honey for wounds, ulcers, and skin graft preservation. Lancet 1993, 341, 756–757. [Google Scholar] [CrossRef]
- Maddocks, S.E.; Jenkins, R.E. Honey: A sweet solution to the growing problem of antimicrobial resistance? Future Microbiol. 2013, 8, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Cremers, N.; Belas, A.; Santos Costa, S.; Couto, I.; de Rooster, H.; Pomba, C. In vitro antimicrobial efficacy of two medical grade honey formulations against common high-risk meticillin-resistant staphylococci and Pseudomonas spp. pathogens. Vet. Dermatol. 2020, 31, 90–96. [Google Scholar] [CrossRef]
- Hermanns, R.; Cremers, N.A.J.; Leeming, J.P.; van der Werf, E.T. Sweet Relief: Determining the Antimicrobial Activity of Medical Grade Honey Against Vaginal Isolates of Candida albicans. J. Fungi (Basel) 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.M.P.; Devesa, J.S.P.; Hill, P.B. In vitro efficacy of a honey-based gel against canine clinical isolates of Staphylococcus pseudintermedius and Malassezia pachydermatis. Vet. Dermatol. 2018, 29, 180-e165. [Google Scholar] [CrossRef] [PubMed]
- Saikaly, S.K.; Khachemoune, A. Honey and Wound Healing: An Update. Am. J. Clin. Dermatol. 2017, 18, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Gottrup, F. Oxygen in wound healing and infection. World J. Surg. 2004, 28, 312–315. [Google Scholar] [CrossRef]
- Rossiter, K.; Cooper, A.J.; Voegeli, D.; Lwaleed, B.A. Honey promotes angiogeneic activity in the rat aortic ring assay. J. Wound Care 2010, 19, 440, 442–446. [Google Scholar] [CrossRef]
- Winter, G.D. Formation of the scab and the rate of epithelization of superficial wounds in the skin of the young domestic pig. Nature 1962, 193, 293–294. [Google Scholar] [CrossRef]
- Lukanc, B.; Potokar, T.; Erjavec, V. Complete skin regeneration with medical honey after skin loss on the entire circumference of a leg in a cat. J. Tissue Viability 2020. [Google Scholar] [CrossRef]
- Du Toit, D.F.; Page, B.J. An in vitro evaluation of the cell toxicity of honey and silver dressings. J. Wound Care 2009, 18, 383–389. [Google Scholar] [CrossRef]
- Smaropoulos, E.; Cremers, N.A. Medical grade honey for the treatment of paediatric abdominal wounds: A case series. J. Wound Care 2020, 29, 94–99. [Google Scholar] [CrossRef]
- Kateel, R.; Adhikari, P.; Augustine, A.J.; Ullal, S. Topical honey for the treatment of diabetic foot ulcer: A systematic review. Complement. Ther. Clin. Pract. 2016, 24, 130–133. [Google Scholar] [CrossRef]
- Makhdoom, A.; Khan, M.S.; Lagahari, M.A.; Rahopoto, M.Q.; Tahir, S.M.; Siddiqui, K.A. Management of diabetic foot by natural honey. J. Ayub Med. Coll Abbottabad 2009, 21, 103–105. [Google Scholar] [PubMed]
- Surahio, A.R.; Khan, A.A.; Farooq, M.; Fatima, I. Role of honey in wound dressing in diabetic foot ulcer. J. Ayub Med. Coll Abbottabad 2014, 26, 304–306. [Google Scholar] [PubMed]
- Moghazy, A.M.; Shams, M.E.; Adly, O.A.; Abbas, A.H.; El-Badawy, M.A.; Elsakka, D.M.; Hassan, S.A.; Abdelmohsen, W.S.; Ali, O.S.; Mohamed, B.A. The clinical and cost effectiveness of bee honey dressing in the treatment of diabetic foot ulcers. Diabetes Res. Clin. Pract. 2010, 89, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Guo, M.; Zhang, N.; Wang, G. Effectiveness of honey dressing in the treatment of diabetic foot ulcers: A systematic review and meta-analysis. Complement. Ther. Clin. Pract. 2019, 34, 123–131. [Google Scholar] [CrossRef]
- Mandal, M.D.; Mandal, S. Honey: Its medicinal property and antibacterial activity. Asian Pac. J. Trop. Biomed. 2011, 1, 154–160. [Google Scholar] [CrossRef]
- Olsson, M.; Jarbrink, K.; Divakar, U.; Bajpai, R.; Upton, Z.; Schmidtchen, A.; Car, J. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair Regen. 2019, 27, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Jarbrink, K.; Ni, G.; Sonnergren, H.; Schmidtchen, A.; Pang, C.; Bajpai, R.; Car, J. The humanistic and economic burden of chronic wounds: A protocol for a systematic review. Syst. Rev. 2017, 6, 15. [Google Scholar] [CrossRef]
- Zbuchea, A. Honey, Food and Medicine: Scientific Rationale and Practical Efficiency in External Administration of Medicinal Honey for Wound Healing. J. Agric. Sci. Technol. B 2017, 7, 206–219. [Google Scholar] [CrossRef][Green Version]
- Moore, O.A.; Smith, L.A.; Campbell, F.; Seers, K.; McQuay, H.J.; Moore, R.A. Systematic review of the use of honey as a wound dressing. BMC Complement. Altern. Med. 2001, 1, 2. [Google Scholar] [CrossRef] [PubMed]
- Lusby, P.E.; Coombes, A.; Wilkinson, J.M. Honey: A potent agent for wound healing? J. Wound Ostomy Continence Nurs. 2002, 29, 295–300. [Google Scholar] [CrossRef] [PubMed]
| Case | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| t Infection | 4 weeks | 1 week | 3 weeks | 3 weeks | 3 weeks | 2 weeks |
| t Granulation | 4 weeks | 4 weeks | 3 weeks | 3 weeks | 3 weeks | 3 weeks |
| t Healing | 52 weeks | 32 weeks | 10 weeks | 12 weeks | 6 weeks | 7 weeks |
| Risk Amputation | Yes | Yes | Yes | Yes |
| Case | Initial 1° Dressing | Initial 2° Dressing | Duration and Dressing Regime | 1° Dressing Treatment Change | 2° Dressing Treatment Change | Duration and Dressing Regime |
|---|---|---|---|---|---|---|
| 1 | Soft and Net | Sterile absorptive gauzes | 4 weeks of daily changes | Soft | Sterile absorptive gauzes | 48 weeks of daily changes following weekly application |
| 2 | Ointment and Net | Sterile absorptive gauzes | 2 months of two changes per day | Soft | Sterile absorptive gauzes | 6 months of daily changes |
| 3 | Ointment and Hydro | Fixative gauzes | 7 weeks of daily changes | Soft and Tulle | Sterile absorptive gauzes | 3 weeks of daily changes |
| 4 | Ointment | Film dressing | 2 weeks of daily changes | Ointment | Alginate and film dressing | 10 weeks of daily changes |
| 5 | Ointment | Film dressing | 6 weeks of daily changes | |||
| 6 | Ointment and Net | Sterile absorptive gauzes | 3 weeks of daily changes | Soft and Tulle | Sterile absorptive gauzes | 3 weeks of daily changes |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nair, H.K.R.; Tatavilis, N.; Pospíšilová, I.; Kučerová, J.; Cremers, N.A.J. Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series. Antibiotics 2020, 9, 529. https://doi.org/10.3390/antibiotics9090529
Nair HKR, Tatavilis N, Pospíšilová I, Kučerová J, Cremers NAJ. Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series. Antibiotics. 2020; 9(9):529. https://doi.org/10.3390/antibiotics9090529
Chicago/Turabian StyleNair, Harikrishna K. R., Nektarios Tatavilis, Ivana Pospíšilová, Jana Kučerová, and Niels A. J. Cremers. 2020. "Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series" Antibiotics 9, no. 9: 529. https://doi.org/10.3390/antibiotics9090529
APA StyleNair, H. K. R., Tatavilis, N., Pospíšilová, I., Kučerová, J., & Cremers, N. A. J. (2020). Medical-Grade Honey Kills Antibiotic-Resistant Bacteria and Prevents Amputation in Diabetics with Infected Ulcers: A Prospective Case Series. Antibiotics, 9(9), 529. https://doi.org/10.3390/antibiotics9090529






