Antifungal Effect of A Chimeric Peptide Hn-Mc against Pathogenic Fungal Strains
Abstract
1. Introduction
2. Results and Discussion
2.1. Antifungal Activity of Peptides against Pathogenic Fungi
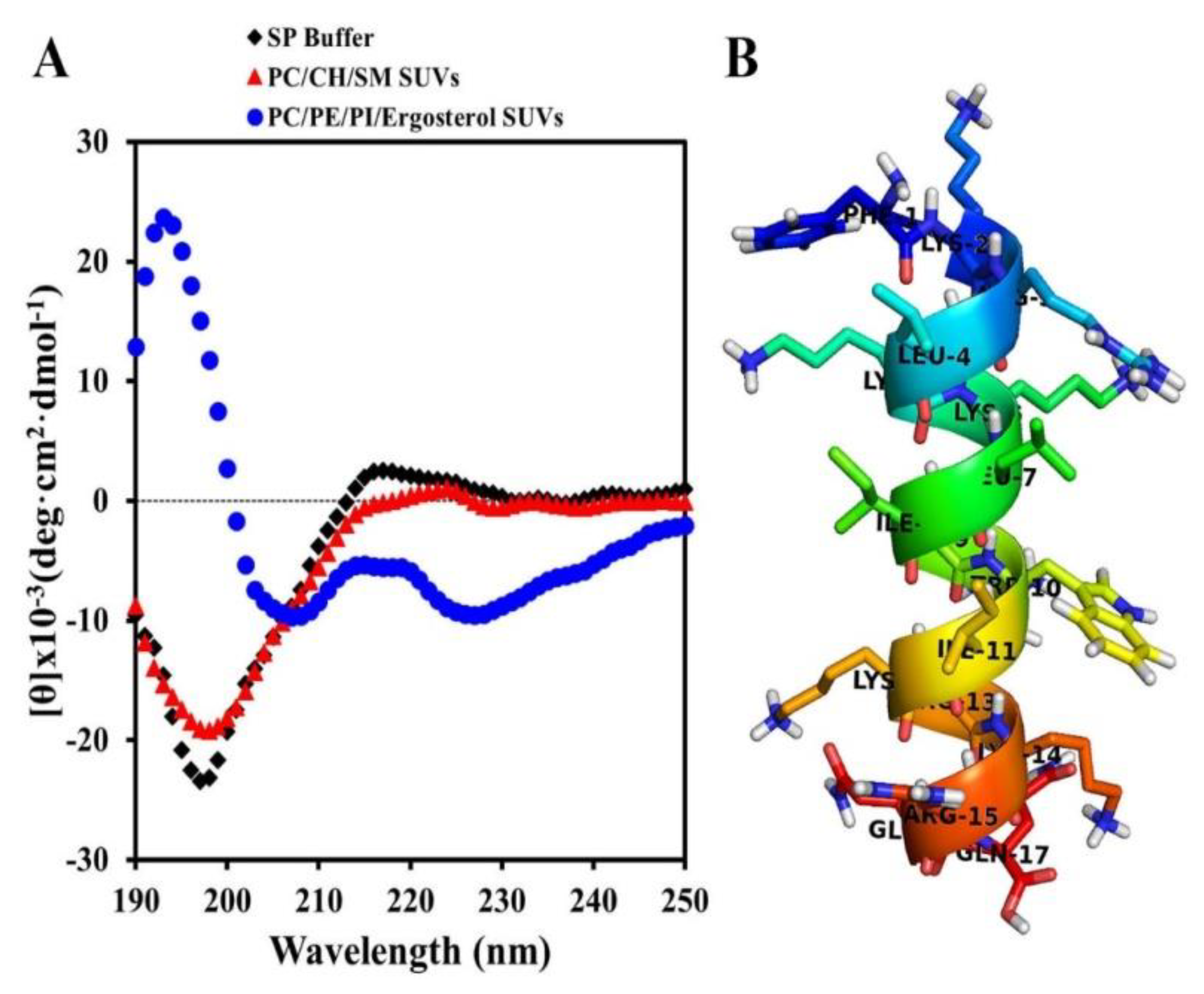
2.2. Secondary Structures of Hn-Mc in Various Environments
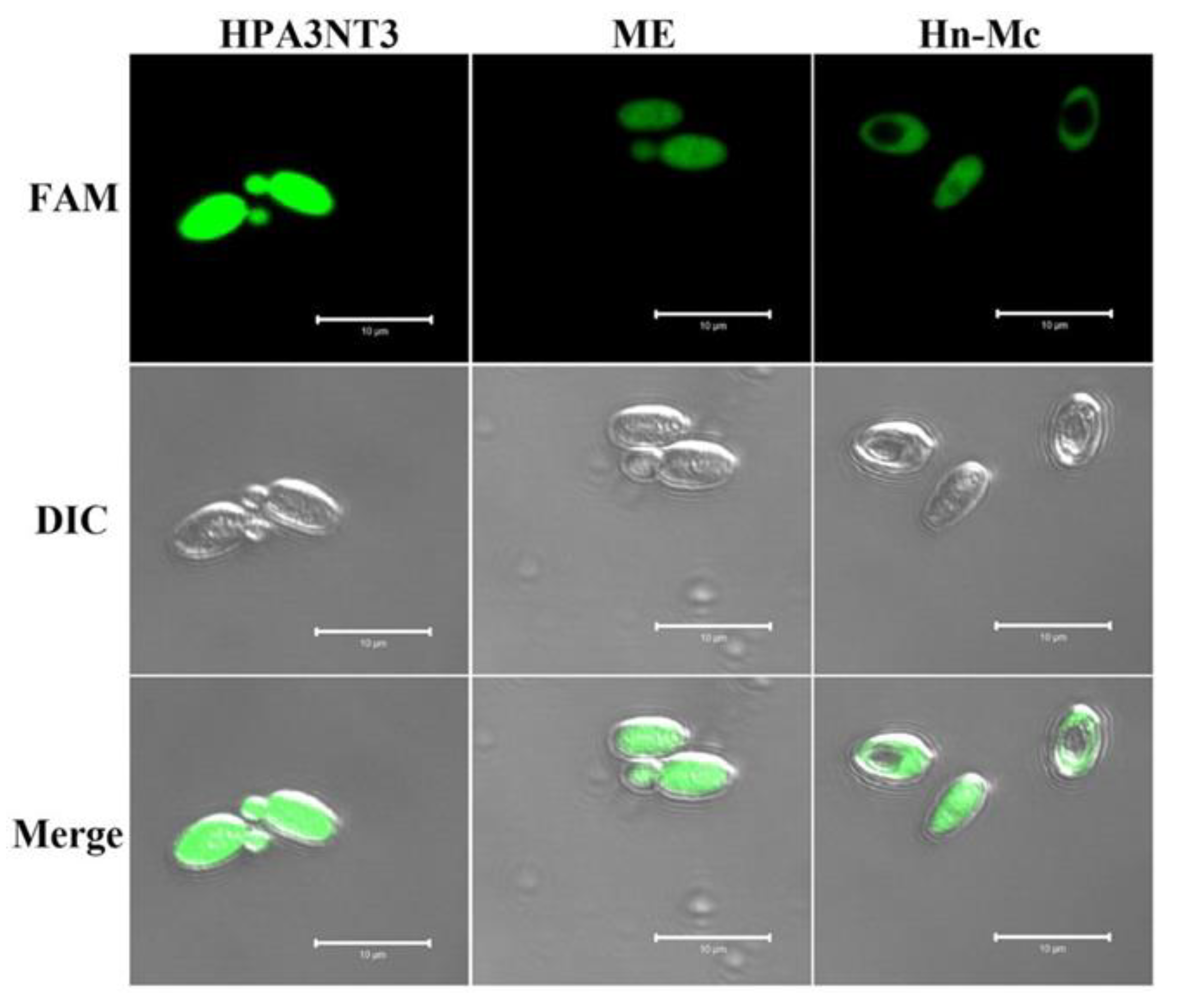
2.3. Intracellular Localization of FAM-Labeled Peptides in Fungal Cells
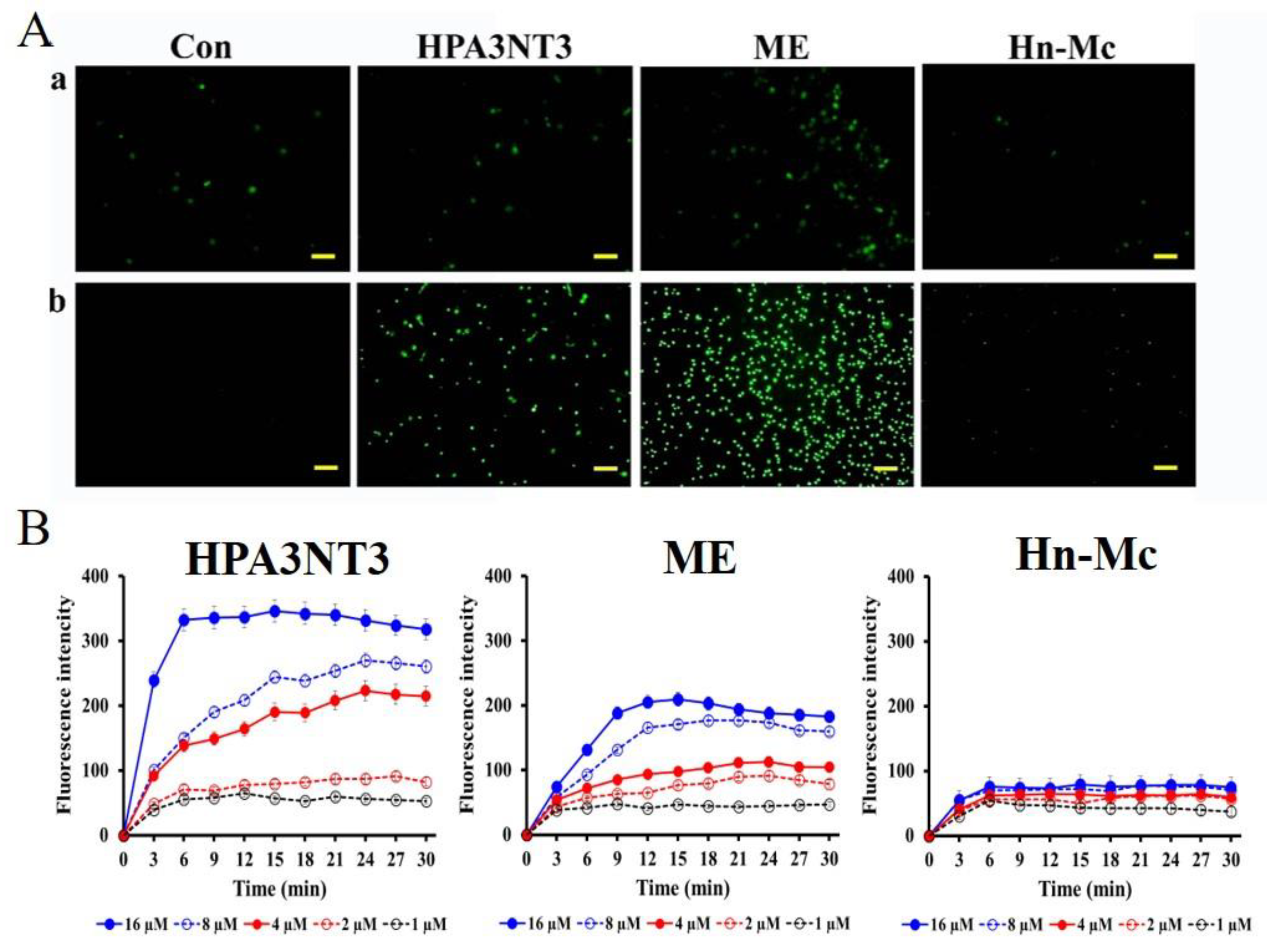
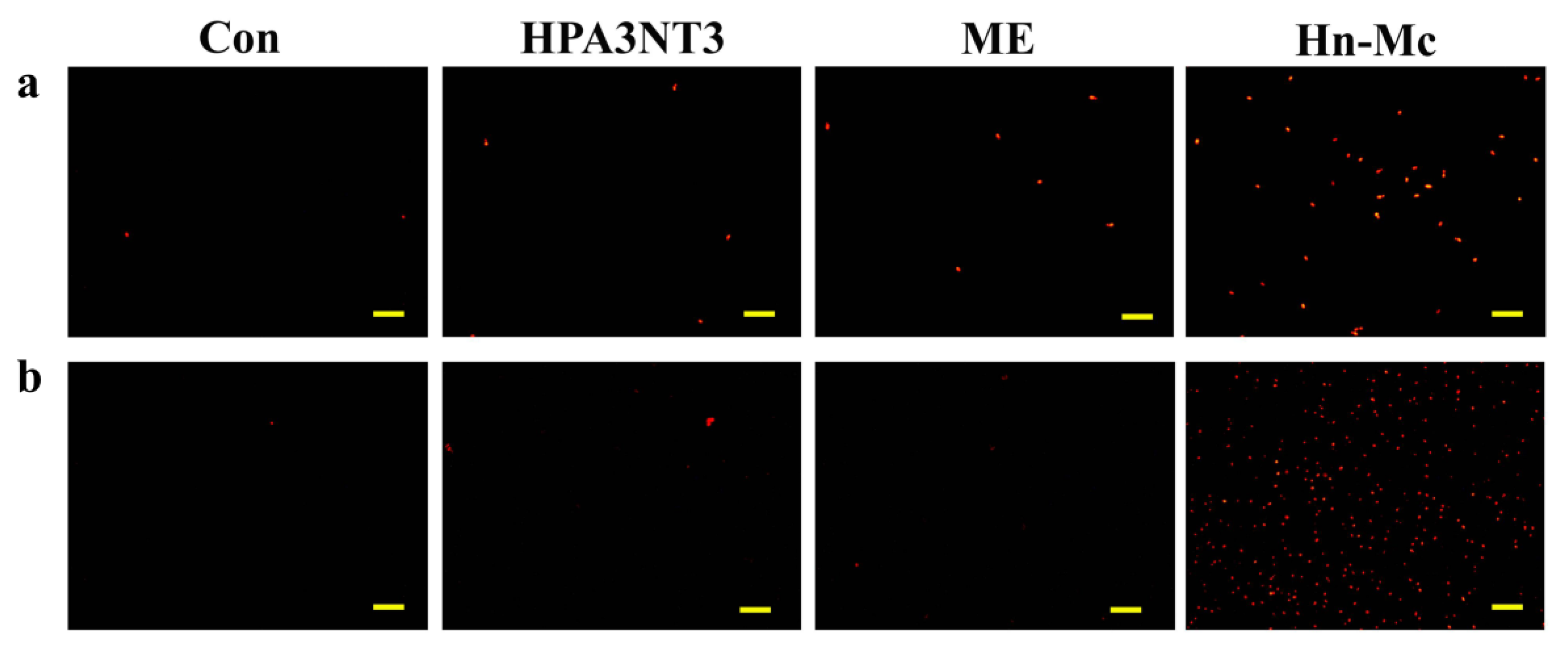
2.4. Fungal Membrane Permeability of Peptides
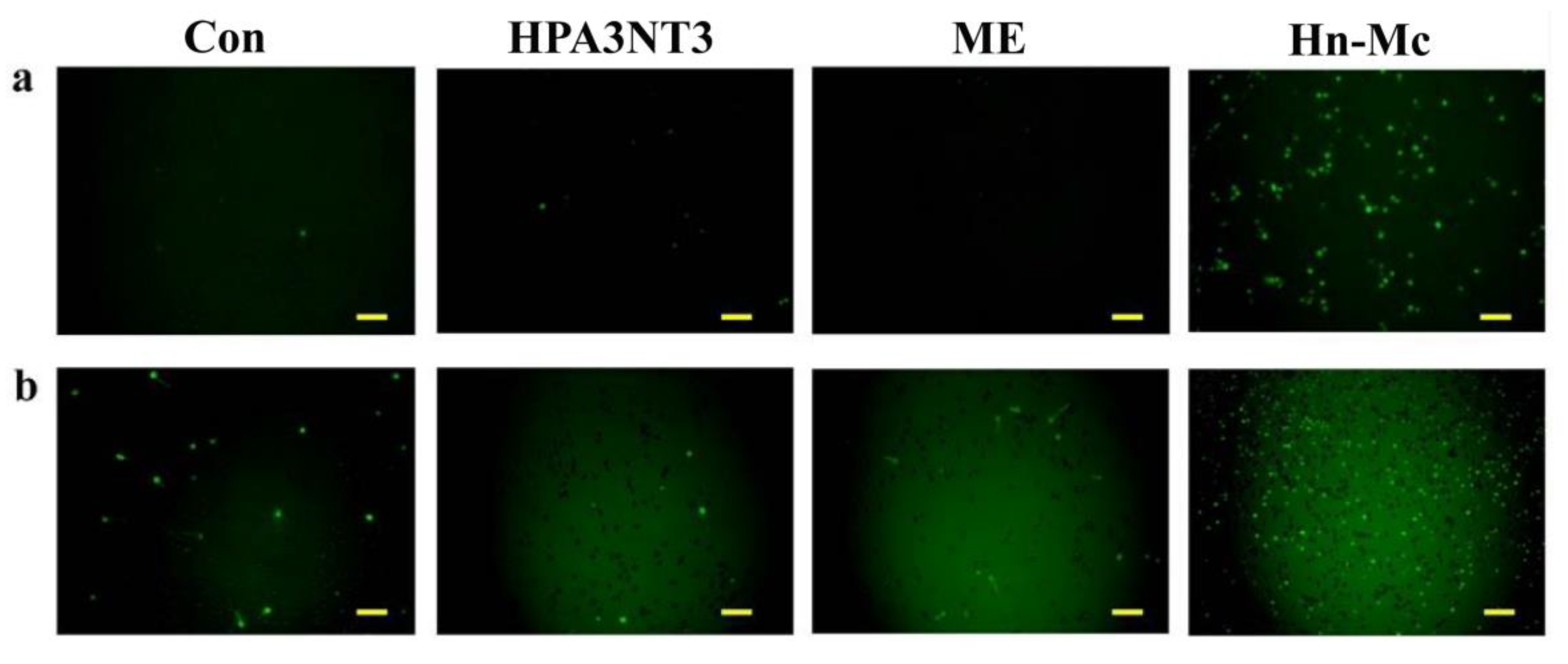
2.5. Intracellular ROS Production by Peptides
2.6. Time-Dependent Morphological Alterations in C. tropicalis with Hn-Mc
3. Materials and Methods
3.1. Materials
3.2. Fungal Cells
3.3. Peptide Synthesis
3.4. Antifungal Assay
3.5. Circular Dichroism (CD)
3.6. Prediction of Secondary Structure
3.7. SYTOX-Green Uptake
3.8. Confocal Laser Scanning Microscopy (CLSM)
3.9. Determination of Intercellular ROS Levels
3.10. Scanning Electron Microscopy (SEM)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Drew, R.H.; Townsend, M.L.; Pound, M.W.; Johnson, S.W.; Perfect, J.R. Recent advances in the treatment of life-threatening, invasive fungal infections. Expert Opin. Pharmacother. 2013, 14, 2361–2374. [Google Scholar] [CrossRef]
- Mavor, A.L.; Thewes, S.; Hube, B. Systemic fungal infections caused by Candida species: Epidemiology, infection process and virulence attributes. Curr. Drug Targets 2005, 6, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Denning, D.W.; Pleuvry, A.; Cole, D.C. Global burden of AMPA in adults asthma and its complication chronic pulmonary aspergillosis in adults. Med. Mycol. 2013, 5, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Taveira, G.B.; Mello, É.O.; Carvalho, A.O.; Regente, M.; Pinedo, M.; de La Canal, L.; Rodrigues, R.; Gomes, V.M. Antimicrobial activity and mechanism of action of a thionin-like peptide from Capsicum annuum fruits and combinatorial treatment with fluconazole against Fusarium solani. Biopolymers 2017, 108. [Google Scholar] [CrossRef]
- Arnold, T.M.; Dotson, E.; Sarosi, G.A.; Hage, C.A. Traditional and emerging antifungal therapies. Proc. Am. Thorac. Soc. 2010, 7, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Rex, J.H.; Rinaldi, M.G.; Pfaller, M.A. Resistance of Candida species to fluconazole. Antimicrob. Agents Cemother. 1995, 39, 1–8. [Google Scholar] [CrossRef]
- Liu, T.T.; Lee, R.E.B.; Baker, K.S.; Lee, R.E.; Wei, L.; Hamayouni, R.; Roger, P.D. Genome-wide expression profiling of the response to azole, polyene, echinocandin, and pyrimidine antifungal agents in Candida albicans. Antimicrob. Agents Chemother. 2005, 49, 2226–2236. [Google Scholar] [CrossRef] [PubMed]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef]
- Demain, A.L.; Sanchez, S. Microbial drug discovery: 80 years of progress. J. Antibiot. 2009, 62, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Jenssen, H.; Hamill, P.; Hancock, R.E. Peptide antimicrobial agent. Clini. Microbiol. Rev. 2006, 19, 491–511. [Google Scholar] [CrossRef]
- Aoki, W.; Veda, M. Characterization of antimicrobial peptides toward the development of novel antibiotics. Pharmaceuticals (Basel) 2013, 6, 1055–1081. [Google Scholar] [CrossRef]
- Kim, Y.M.; Kim, N.H.; Lee, J.W.; Jang, J.S.; Park, Y.H.; Park, S.C.; Jang, M.K. Novel chimeric peptide with enhanced cell specificity and anti-inflammatory activity. Biochem. Biophys. Res. Commun. 2015, 463, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Ramsdale, M. Programmed cell death in pathogenic fungi. Biochim. Biophys. Acta 2008, 1783, 1369–1380. [Google Scholar] [CrossRef] [PubMed]
- Herker, E.; Jungwirth, H.; Lehmann, K.A.; Maldener, C.; Fröhlich, K.U.; Wissing, S.; Büttner, S.; Fehr, M.; Sigrist, S.; Madeo, F. Chronological aging leads to apoptosis in yeast. J. Cell Biol. 2004, 164, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Munoz, A.J.; Wanichthanarak, K.; Meza, E.; Petranovic, D. Systems biology of yeast cell death. FEMS Yeast Res. 2012, 12, 249–265. [Google Scholar] [CrossRef]
- Park, S.C.; Kim, J.Y.; Shin, S.O.; Jeong, C.Y.; Kim, M.H.; Shin, S.Y.; Cheong, G.W.; Park, Y.; Hahm, K.S. Investigation of toroidal pore and oligomerization by melittin using trasnsmission electron microscopy. Biochem. Biophys. Res. Commun. 2006, 343, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Dantas Ada, S.; Day, A.; Ikeh, M.; Kos, I.; Achan, B.; Quinn, J. Oxidative stress responses in the human fungal pathogen, Candida albicans. Biomolecules 2015, 5, 142–165. [Google Scholar] [CrossRef]
- Scherz-Shouval, R.; Elazar, Z. ROS, mitochondria and the regulation of autophagy. Trend Cell Biol. 2007, 7, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Park, S.C.; Cheong, M.S.; Kim, E.J.; Kim, J.H.; Chi, Y.H.; Jang, M.K. Antifungal effect of Arabidopsis SGT1 proteins via mitochondrial reactive oxygen species. J. Agric. Food Chem. 2017, 65, 8340–8347. [Google Scholar] [CrossRef]
- Lee, H.; Hwang, J.S.; Lee, D.G. Scolopendin, an antimicrobial peptide from centipede, attenuates mitochondrial functions and triggers apoptosis in Candida albicans. Biochem. J. 2017, 474, 635–645. [Google Scholar] [CrossRef]
- Park, S.C.; Kim, J.Y.; Jeong, C.; Yoo, S.; Hahm, K.S.; Park, Y. A plausible mode of action of pseudin-2, an antimicrobial peptide from Pseudis Paradoxa. Biochim, Biophys. Acta 2011, 1808, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Park, S.C.; Kim, M.H.; Hossain, M.A.; Shin, S.Y.; Kim, Y.; Stella, L.; Wade, J.D.; Park, Y.; Hahm, K.S. Amphipathic alpha-helical peptide, HP (2-20), and its analogues derived from Helicobacter pylori: Pore formation mechanism in various lipid compositions. Biochim. Biophys. Acta 2008, 1778, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Maupetit, J.; Derreumaux, P.; Tufféry, P. Improved PEP-FOLD Approach for Peptide and Miniprotein Structure Prediction. J. Chem. Theory Comput. 2014, 10, 4745–4758. [Google Scholar] [CrossRef]
- Mangoni, M.L.; Papo, N.; Barra, D.; Simmaco, M.; Bozzi, A.; Di Giulio, A.; Rinaldi, A.C. Effects of the antimicrobial peptide temporin L on cell morphology, membrane permeability and viability of Escherichia coli. Biocchem. J. 2004, 380, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.D.; Morrell, J.L.; Hamer, J.E. Identification and characterization of Aspergillus nidulans mutants detective in cytokinesis. Genetics 1994, 136, 517–532. [Google Scholar]
- Park, S.C.; Jung, Y.J.; Lee, Y.; Kim, I.R.; Seol, M.A.; Kim, E.J.; Jang, M.J.; Lee, J.R. Functional characterization of the Arabidopsis universal stress protein AtUSP with an antifungal activity. Biochem. Biophys. Res. Commun. 2017, 486, 923–929. [Google Scholar] [CrossRef]
- Wang, H.; Chen, Y.; Zhai, N.; Chen, X.; Gan, F.; Li, H.; Huang, K. Ochratoxin A-induced apoptosis of EPEC-J2 cells through ROS-mediated mitochondrial permeability transition pore opening pathway. J. Agric. Food Chem. 2017, 65, 10630–10637. [Google Scholar] [CrossRef]

| Fungal Strains | MIC (μM) | ||
|---|---|---|---|
| HPA3NT3 | ME | Hn-Mc | |
| Yeast | |||
| C. albicans | 16 | 8–16 | 16 |
| C. krusei | 8 | 4 | 8 |
| C. parapsilosis | 16 | 16 | 16 |
| C. tropicalis | 4 | 4 | 4 |
| T. beigellii | 1 | 2 | 1 |
| Mold | |||
| T. rubrum | 2 | 2 | 1–2 |
| F. moniliforme | 2 | 2 | 1–2 |
| F. solani | 2 | 2 | 1 |
| F. oxysporum | 2 | 2 | 1–2 |
| A. flavus | 2–4 | 16 | 2–4 |
| A. fumigatus | 2 | 8 | 2–4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.-Y.; Park, S.-C.; Noh, G.; Kim, H.; Yoo, S.-H.; Kim, I.R.; Lee, J.R.; Jang, M.-K. Antifungal Effect of A Chimeric Peptide Hn-Mc against Pathogenic Fungal Strains. Antibiotics 2020, 9, 454. https://doi.org/10.3390/antibiotics9080454
Kim J-Y, Park S-C, Noh G, Kim H, Yoo S-H, Kim IR, Lee JR, Jang M-K. Antifungal Effect of A Chimeric Peptide Hn-Mc against Pathogenic Fungal Strains. Antibiotics. 2020; 9(8):454. https://doi.org/10.3390/antibiotics9080454
Chicago/Turabian StyleKim, Jin-Young, Seong-Cheol Park, Gwangbok Noh, Heabin Kim, Su-Hyang Yoo, Il Ryong Kim, Jung Ro Lee, and Mi-Kyeong Jang. 2020. "Antifungal Effect of A Chimeric Peptide Hn-Mc against Pathogenic Fungal Strains" Antibiotics 9, no. 8: 454. https://doi.org/10.3390/antibiotics9080454
APA StyleKim, J.-Y., Park, S.-C., Noh, G., Kim, H., Yoo, S.-H., Kim, I. R., Lee, J. R., & Jang, M.-K. (2020). Antifungal Effect of A Chimeric Peptide Hn-Mc against Pathogenic Fungal Strains. Antibiotics, 9(8), 454. https://doi.org/10.3390/antibiotics9080454






