Longitudinal Point Prevalence Survey of Antimicrobial Consumption in Russian Hospitals: Results of the Global-PPS Project
Abstract
1. Introduction
2. Results
2.1. Characteristics of the Hospitals and Study Population
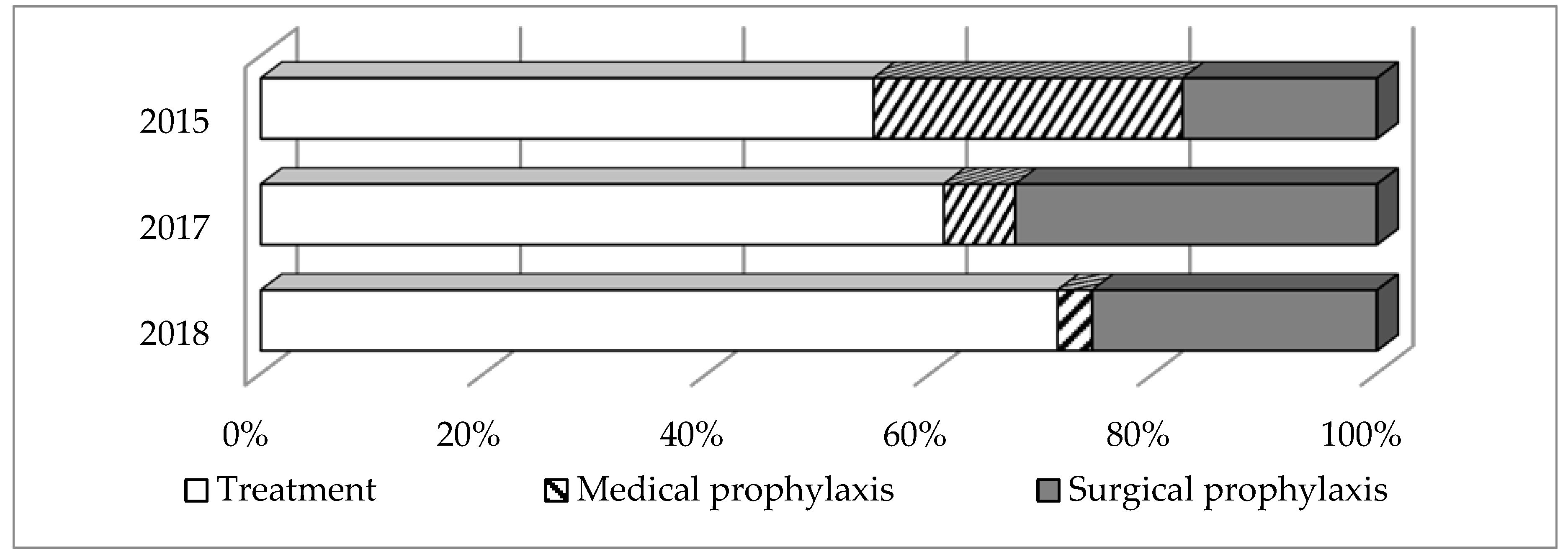
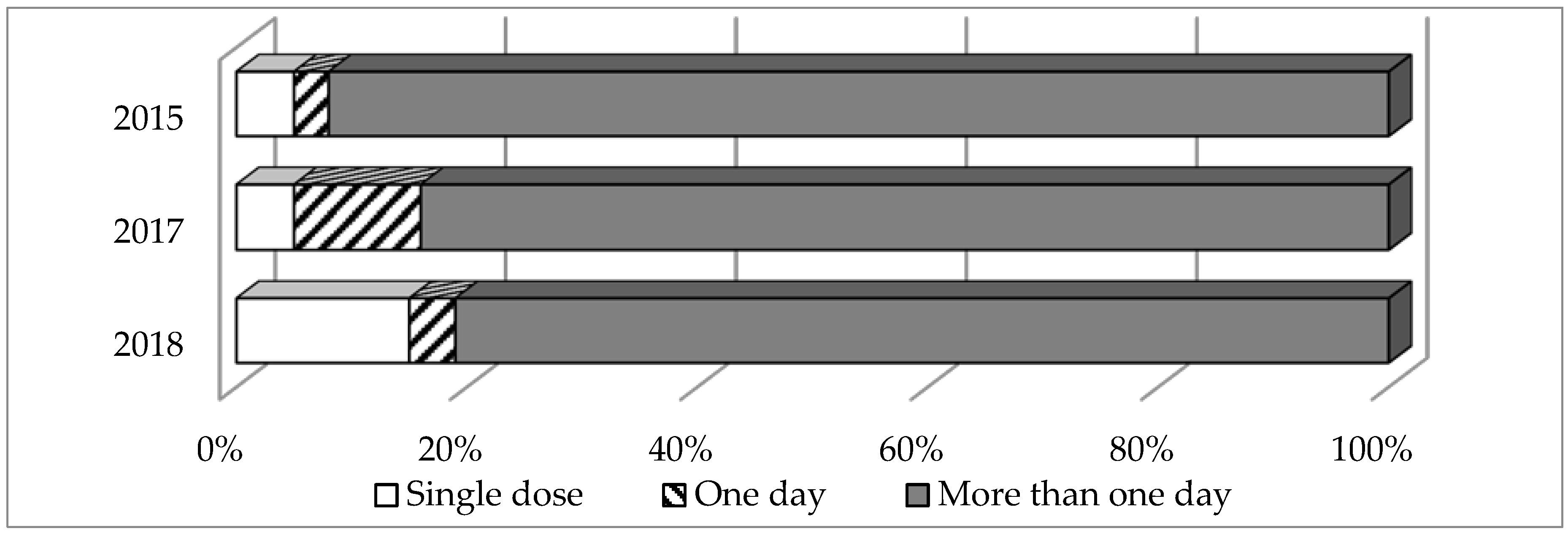
2.2. General Trends of Systemic AMD Prescribing
2.3. Key Patterns and Quality Indicators of Systemic AMD Prescribing
2.4. Quality Indicators Dynamic at the Level of Study Centres
3. Materials and Methods
- duration of perioperative prophylaxis,
- documentation of indication for prescription of antibiotic therapy,
- documentation of stop/review date,
- targeted treatment based upon microbiological result,
- treatment based upon the use of biomarker data (C-reactive protein, procalcitonin, or other).
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Maragakis, L.L.; Perencevich, E.N.; Cosgrove, S.E. Clinical and economic burden of antimicrobial resistance. Expert. Rev. Anti Infect. Ther. 2008, 6, 751–763. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, S.E. The relationship between antimicrobial resistance and patient outcomes: Mortality, length of hospital stay, and health care costs. Clin. Infect. Dis. 2006, 42, S82–S89. [Google Scholar] [CrossRef] [PubMed]
- Sukhorukova, M.V.; Edelstein, M.V.; Ivanchik, N.V.; Skleenova, E.Y.; Shajdullina, E.R.; Azyzov, I.S.; Shek, E.A.; Kuzmenkov, A.Y.; Dekhnich, A.V.; Kozlov, R.S.; et al. Antimicrobial resistance of nosocomial Enterobacterales isolates in Russia: Results of multicenter epidemiological study «MARATHON 2015–2016». Clin. Microbiol. Antimicrob. Chemother. 2019, 21, 147–159. [Google Scholar] [CrossRef]
- Edelstein, M.V.; Shek, E.A.; Sukhorukova, M.V.; Skleenova, E.Y.; Ivanchik, N.V.; Shajdullina, E.R.; Mikotina, A.V.; Kuzmenkov, A.Y.; Dekhnich, A.V.; Kozlov, R.S.; et al. Antimicrobial resistance, carbapenemase production and genotypes of nosocomial Pseudomonas aeruginosa isolates in Russia: Results of multicenter epidemiological study «MARATHON 2015–2016». Clin. Microbiol. Antimicrob. Chemother. 2019, 21, 160–170. [Google Scholar] [CrossRef]
- Shek, E.A.; Sukhorukova, M.V.; Edelstein, M.V.; Skleenova, E.Y.; Ivanchik, N.V.; Shajdullina, E.R.; Kuzmenkov, A.Y.; Dekhnich, A.V.; Kozlov, R.S.; Semyonova, N.V.; et al. Antimicrobial resistance, carbapenemase production, and genotypes of nosocomial Acinetobacter spp. isolates in Russia: Results of multicenter epidemiological study «MARATHON 2015–2016». Clin. Microbiol. Antimicrob. Chemother. 2019, 21, 171–180. [Google Scholar] [CrossRef]
- Romanov, A.V.; Dekhnich, A.V.; Sukhorukova, M.V.; Skleenova, E.Y.; Ivanchik, N.V.; Edelstein, M.V.; Kozlov, R.S.; the «MARATHON» Study Group. Antimicrobial resistance of nosocomial Staphylococcus aureus isolates in Russia: Results of multicenter epidemiological study «MARATHON»2013-2014. Clin. Microbiol. Antimicrob. Chemother. 2017, 19, 57–62. [Google Scholar]
- Lieberman, J.M. Appropriate antibiotic use and why it is important: The challenges of bacterial resistance. Pediatr. Infect. Dis. J. 2003, 22, 1143–1151. [Google Scholar] [CrossRef]
- Ansari, F.; Erntell, M.; Goossens, H.; Davey, P. The European Surveillance of Antimicrobial Consumption (ESAC) point-prevalence survey of antibacterial use in 20 European hospitals in 2006. Clin. Infect. Dis. 2009, 49, 1496–1504. [Google Scholar] [CrossRef]
- Harbarth, S.; Balkhy, H.H.; Goossens, H.; Jarlier, V.; Kluytmans, J.; Laxminarayan, R.; Saam, M.; Van Belkum, A.; Pittet, D.; for the World Healthcare-Associated Infections Resistance Forum participants. Antimicrobial resistance: One world, one fight! Antimicrob. Resist. Infect. Control. 2015, 4, 49. [Google Scholar] [CrossRef]
- Zarb, P.; Goossens, H. European Surveillance of Antimicrobial Consumption (ESAC): Value of a point-prevalence survey of antimicrobial use across Europe. Drugs 2011, 71, 745–755. [Google Scholar] [CrossRef]
- Versporten, A. Antimicrobial use and HAI rates in Adult ICU’s. In Proceedings of the International Symposium on Intensive Care and Emergency Medicine (ESICEM), Brussels, Belgium, 19–22 March 2019. [Google Scholar]
- Versporten, A.; Zarb, P.; Caniaux, I.; Gros, M.F.; Drapier, N.; Miller, M.; Jarlier, V.; Nathwani, D.; Goossens, H.; Global-PPS network. Antimicrobial Consumption and Resistance in Adult Hospital Inpatients in 53 Countries: Results of an Internet-Based Global Point Prevalence Survey. Lancet Glob. Health 2018, 6, e619–e629. [Google Scholar] [CrossRef]
- WHO Collaborating Centre for Drug Statistics Methodology. Guidelines for ATC Classification and DDD Assignment 2020; WHO Collaborating Centre for Drug Statistics Methodology: Oslo, Norway, 2019. [Google Scholar]
- Medical Informational Agency. Nosocomial Pneumonia in Adults: Russian National Guidelines, 2nd ed.; StatPearls: Moscow, Russia, 2016. [Google Scholar]
- Gelfand, B.R.; Kubishlin, V.A.; Kozlov, R.S.; Khachatrjan, N.N. (Eds.) Surgical Skin and Soft Tissue Infections: Russian National Guidelines, 2nd ed.; RASHI: Moscow, Russia, 2015. [Google Scholar]
- Savelev, V.S.; Gelfand, B.R. (Eds.) Abdominal Surgical Infection: Russian National Guidelines; BORGES Company: Moscow, Russia, 2011. [Google Scholar]
- Chuchalin, A.G.; Sinopalnikov, A.I.; Kozlov, R.S.; Avdeev, S.N.; Tyurin, I.E.; Rudnov, V.A.; Rachina, S.A.; Fesenko, O.V. Clinical guidelines on diagnosis, treatment and prevention of severe community acquired pneumonia in adults. Pulmonology 2014, 4, 13–48. [Google Scholar] [CrossRef]
- Chuchalin, A.G.; Sinopalnikov, A.I.; Kozlov, R.S.; Turin, I.E.; Rachina, S.A. Community-acquired pneumonia in adults: Clinical guidelines on diagnostics, treatment and prophylaxis. Clin. Microbiol. Antimicrob. Chemother. 2010, 12, 187–225. [Google Scholar]
- Zaitsev, A.V.; Perepanova, T.S.; Gvosdev, M.Y.; Arefeva, O.A. Urinary Tract Infections. Part 1. Guidelines №57; ABV-Press: Moscow, Russia, 2017. [Google Scholar]
- Zaitsev, A.V.; Perepanova, T.S.; Pushkar, D.Y.; Vasilyev, A.O.; Gvosdev, M.Y.; Arefeva, O.A. Urinary Tract Infections. Part 2. Guidelines №87; ABV-Press: Moscow, Russia, 2018. [Google Scholar]
- Plachouras, D.; Kärki, T.; Hansen, S.; Hopkins, S.; Lyytikäinen, O.; Moro, M.L.; Reilly, J.; Zarb, P.; Zingg, W.; Kinross, P.; et al. Antimicrobial use in European acute care hospitals: Results from the second point prevalence survey (PPS) of healthcare-associated infections and antimicrobial use, 2016 to 2017. Euro Surveill. 2018, 46, 1800393. [Google Scholar] [CrossRef] [PubMed]
- Kopczynska, M.; Sharif, B.; Unwin, H.; Lynch, J.; Forrester, A.; Zeicu, C.; Cleaver, S.; Kulikouskaya, S.; Chandy, T.; Ang, E.; et al. Real World Patterns of Antimicrobial Use and Microbiology Investigations in Patients with Sepsis outside the Critical Care Unit: Secondary Analysis of Three Nation-Wide Point Prevalence Studies. J. Clin. Med. 2019, 8, 1337. [Google Scholar] [CrossRef]
- Robertson, J.; Iwamoto, K.; Hoxha, I.; Ghazaryan, L.; Abilova, V.; Cvijanovic, A.; Pyshnik, H.; Darakhvelidze, M.; Makalkina, L.; Jakupi, A.; et al. Antimicrobial Medicines Consumption in Eastern Europe and Central Asia - An Updated Cross-National Study and Assessment of Quantitative Metrics for Policy Action. Front. Pharmacol. 2019, 9, 1156. [Google Scholar] [CrossRef]
- WHO. Global Guidelines for the Prevention of Surgical Site Infection; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization. Adopt AWaRe Homepage. Available online: https://adoptaware.org/ (accessed on 23 June 2020).
- Paterson, D.L. «Сollateral damage» from cephalosporin or quinolone antibiotic therapy. Clin. Infect. Dis. 2004, 38, S341–S345. [Google Scholar] [CrossRef]
- Saurina, G.; Quale, J.M.; Manikal, V.M.; Oydna, E.; Landman, D. Antimicrobial resistance in Enterobacteriacea in Brooklyn, NY: Epidemiology and relation to antibiotic usage patterns. J. Antimicrob. Chemother. 2000, 45, 895–898. [Google Scholar] [CrossRef][Green Version]
- Lautenbach, E.; Patel, J.B.; Bilker, W.B.; Edelstein, P.H.; Fishman, N.O. Extended spectrum beta-lactamase-producing E. coli and K. pneumoniae: Risk factors for infection and impact of resistance on outcomes. Clin. Infect. Dis. 2001, 32, 1162–1171. [Google Scholar] [CrossRef]
- Yakovlev, S.V.; Briko, N.I.; Sidorenko, S.V.; Protsenko, D.N. (Eds.) SKAT Programm for Medical Aid in Inpatients: Russian National Guidelines; Pero: Moscow, Russia, 2018. [Google Scholar]
- Gusarov, V.G.; Nesterova, E.E.; Oprishenko, I.V.; Petrova, N.V.; Zmjatin, M.N. Clinical and pharmacoeconomic results of the use of the protocol empiric antimicrobial therapy in a multidisciplinary hospital. Bull. Pirogov Med Surg. Cent. 2015, 10, 100–104. [Google Scholar]
- Rudnov, V.A.; Kolotova, G.B.; Bagin, V.A.; Nevskaya, N.N.; Belsky, D.V.; Ivanova, N.A.; Gayfutdinov, E.A. The role of antimicrobial therapy stewardship in intensive care service. Clin. Microbiol. Antimicrob. Chemother. 2018, 20, 132–140. [Google Scholar] [CrossRef]
| Characteristics | 2015 | 2017 | 2018 | Total |
|---|---|---|---|---|
| Number of hospitals, n | 7 | 7 | 7 | 21 |
| • primary hospitals | 0 | 0 | 2 | 2 |
| • secondary hospitals | 3 | 5 | 4 | 12 |
| • tertiary hospitals | 1 | 1 | 1 | 3 |
| • specialized hospitals | 0 | 1 | 0 | 1 |
| • paediatric hospitals | 3 | 0 | 0 | 3 |
| Number of beds, n | 3976 | 6359 | 5610 | 15,945 |
| Number of patients, n | 3546 | 5438 | 4611 | 13,595 |
| • adult % | 63.9 | 95.0 | 96.9 | 87.5 |
| • pediatric % | 33.6 | 3.5 | 3.1 | 11.2 |
| • neonatal % | 2.6 | 1.5 | 0.0 | 1.3 |
| Number of treated patients, n | 899 | 1255 | 1388 | 3542 |
| Number of treated patients % | 25.4 | 23.1 | 30.1 | 26.1 |
| • adult, % | 26.6 | 22.5 | 30.3 | 26.2 |
| • pediatric % | 22.5 | 26.4 | 24.5 | 23.2 |
| • neonatal % | 31.9 | 51.9 | 0 | 41.3 |
| Type of Ward | 2015 | 2017 | 2018 |
|---|---|---|---|
| Medical ward | |||
| adult | 13.8 | 15.5 | 18.0 |
| pediatric | 15.7 | 6.2 | 1.8 |
| neonatal | 18.9 | 35.9 | 0 |
| Surgical ward | |||
| adult | 30.5 | 25.6 | 38.1 |
| pediatric | 23.9 | 27.9 | 37.3 |
| ICU | |||
| adult | 68.8 | 56.6 | 59.5 |
| pediatric | 97.9 | 40.0 | 100.0 1 |
| neonatal | 88.2 | 66.7 | 0 |
| Indication | 2015 | 2017 | 2018 |
|---|---|---|---|
| Pneumonia or lower respiratory tract infection | 25.8 | 23.4 | 21.1 |
| Skin and soft tissue infection | 18.6 | 10.4 | 19.5 |
| Upper urinary tract infection | 6.6 | 11.2 | 11.4 |
| Ear, nose and throat infection | 8.5 | 11.2 | 5.9 |
| Bronchitis | 11.2 | 5.3 | 8.8 |
| Intra-abdominal infection | - | 7.6 | 7.7 |
| Bone/joint infection | 4.7 | 3.6 | 6.2 |
| Gastrointestinal infection | 8.3 | - | - |
| Obstetric/gynaecological infection | - | 3.2 | 2.5 |
| Lower urinary tract infection | 0.8 | - | 4 |
| Eye infection | 1.9 | - | - |
| Sepsis or septic shock with no clear anatomic site | 1.1 | 0.4 | - |
| Infection of the central nervous system | - | - | 1.4 |
| Antibacterials | 2015 | 2017 | 2018 |
|---|---|---|---|
| Penicillins | 9.8 | 15.0 | 11.0 |
| First-generation cephalosporins | 4.6 | 9.6 | 5.8 |
| Second-generation cephalosporins | 7.2 | 0.4 | 0.1 |
| Third-generation cephalosporins | 44.7 | 34.1 | 41.8 |
| Fourth-generation cephalosporins | 2.3 | 0.4 | 3.0 |
| Carbapenems | 4.6 | 5.9 | 4.4 |
| Quinolones | 10.2 | 15.8 | 16.5 |
| Aminoglycosides | 3.7 | 4 | 3.7 |
| Macrolides, Lincosamides, and Streptogramins | 3.0 | 2.8 | 2.9 |
| Sulfonamides and Trimethoprim | 2.2 | 1.8 | 1.5 |
| Other antibacterials | 7.5 | 9.9 | 8.7 |
| Patterns | 2015 | 2017 | 2018 |
|---|---|---|---|
| Intravenous therapy | 85.0 | 84.6 | 86.7 |
| Multiple antimicrobials per patient | 9.8 | 16.9 | 17.9 |
| Targeted therapy 1 | 14.5 | 12.1 | 15.1 |
| Treatment based on biomarker data | 19.9 | 12.1 | 17.8 |
| Compliance with the hospital antibiotic guidelines | 74.8 | 66.8 | 74.3 |
| Indication for treatment was recorded | 72.6 | 84.1 | 82.6 |
| Stop/review date documented | 40.5 | 46.5 | 61.1 |
| Quality Indicator | 2017 | 2018 |
|---|---|---|
| Compliance with the hospital antibiotic guidelines | ||
| Site #1 | 52.5 | 60.7 |
| Site #2 | 92.6 | 95.8 |
| Site #3 | 68.7 | 88.6 |
| Site #4 | 47.9 | 60.4 |
| Indication for treatment was recorded | ||
| Site #1 | 63.3 | 72.0 |
| Site #2 | 97.9 | 97.3 |
| Site #3 | 79.4 | 86.4 |
| Site #4 | 92.9 | 86.8 |
| Stop/review date documented | ||
| Site #1 | 23.5 | 25.5 |
| Site #2 | 98.5 | 97.5 |
| Site #3 | 36.5 | 42.4 |
| Site #4 | 15.4 | 9.9 |
| Treatment based on biomarker data | ||
| Site #1 | 0.5 | 13.8 |
| Site #2 | 0.0 | 14.7 |
| Site #3 | 0.9 | 0.0 |
| Site #4 | 17.8 | 19.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rachina, S.; Belkova, Y.; Kozlov, R.; Versporten, A.; Pauwels, I.; Goossens, H.; Bochanova, E.; Domanskaya, O.; Elokhina, E.; Ezhova, L.; et al. Longitudinal Point Prevalence Survey of Antimicrobial Consumption in Russian Hospitals: Results of the Global-PPS Project. Antibiotics 2020, 9, 446. https://doi.org/10.3390/antibiotics9080446
Rachina S, Belkova Y, Kozlov R, Versporten A, Pauwels I, Goossens H, Bochanova E, Domanskaya O, Elokhina E, Ezhova L, et al. Longitudinal Point Prevalence Survey of Antimicrobial Consumption in Russian Hospitals: Results of the Global-PPS Project. Antibiotics. 2020; 9(8):446. https://doi.org/10.3390/antibiotics9080446
Chicago/Turabian StyleRachina, Svetlana, Yuliya Belkova, Roman Kozlov, Ann Versporten, Ines Pauwels, Herman Goossens, Elena Bochanova, Olga Domanskaya, Elena Elokhina, Ludmila Ezhova, and et al. 2020. "Longitudinal Point Prevalence Survey of Antimicrobial Consumption in Russian Hospitals: Results of the Global-PPS Project" Antibiotics 9, no. 8: 446. https://doi.org/10.3390/antibiotics9080446
APA StyleRachina, S., Belkova, Y., Kozlov, R., Versporten, A., Pauwels, I., Goossens, H., Bochanova, E., Domanskaya, O., Elokhina, E., Ezhova, L., Mishchenko, V., Ni, O., Popov, D., Portnjagina, U., Shchetinin, E., Shegimova, V., Strezh, Y., Vityazeva, V., Zubareva, N., & Russian Global-PPS Project Study Group. (2020). Longitudinal Point Prevalence Survey of Antimicrobial Consumption in Russian Hospitals: Results of the Global-PPS Project. Antibiotics, 9(8), 446. https://doi.org/10.3390/antibiotics9080446







