Activity of Bacteriophage and Complex Tannins against Biofilm-Forming Shiga Toxin-Producing Escherichia coli from Canada and South Africa
Abstract
1. Introduction
2. Results
2.1. Experiment 1
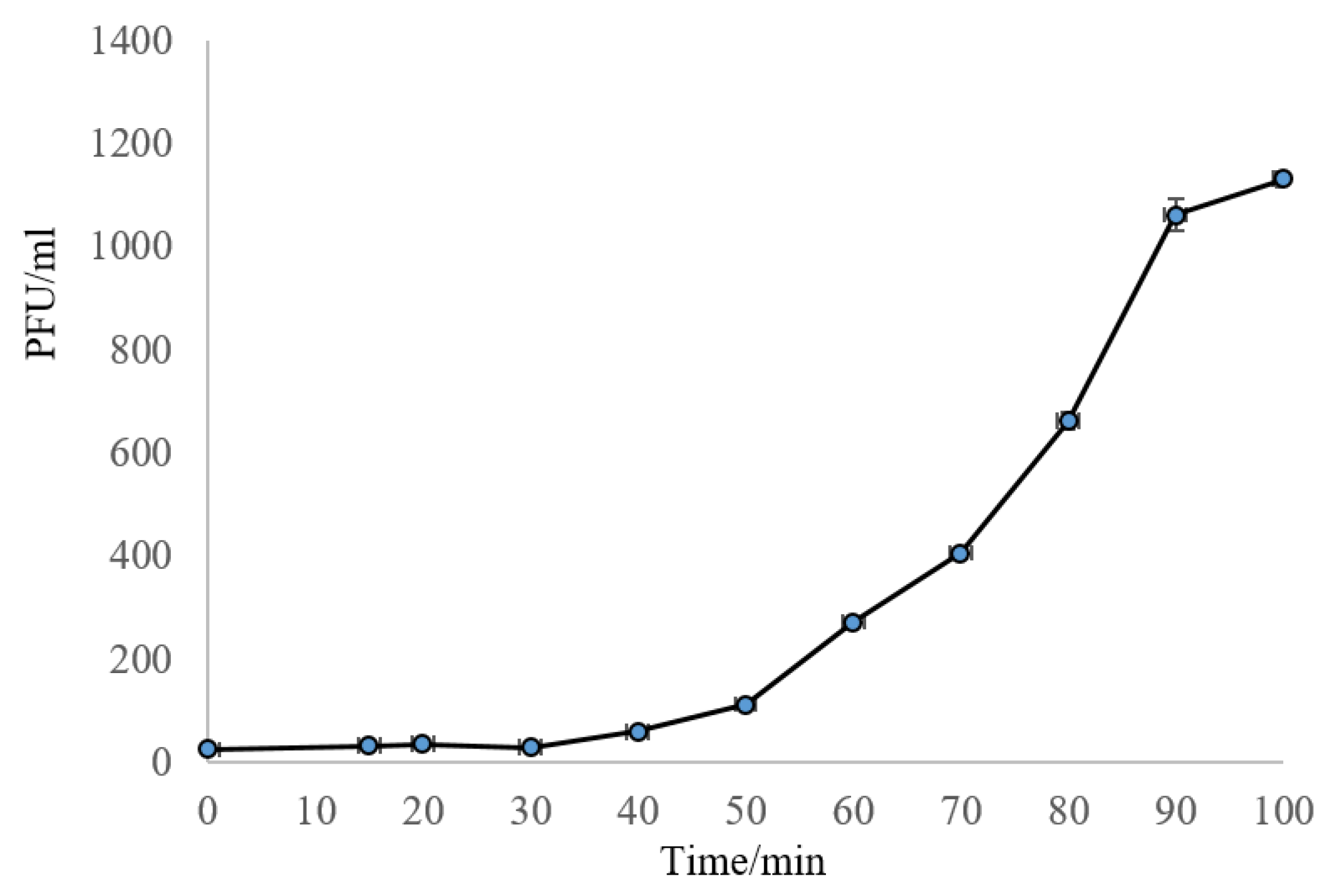
2.1.1. One-Step Growth Curve, and Susceptibility of O154:H10 and O113:H21 to Phage SA21RB
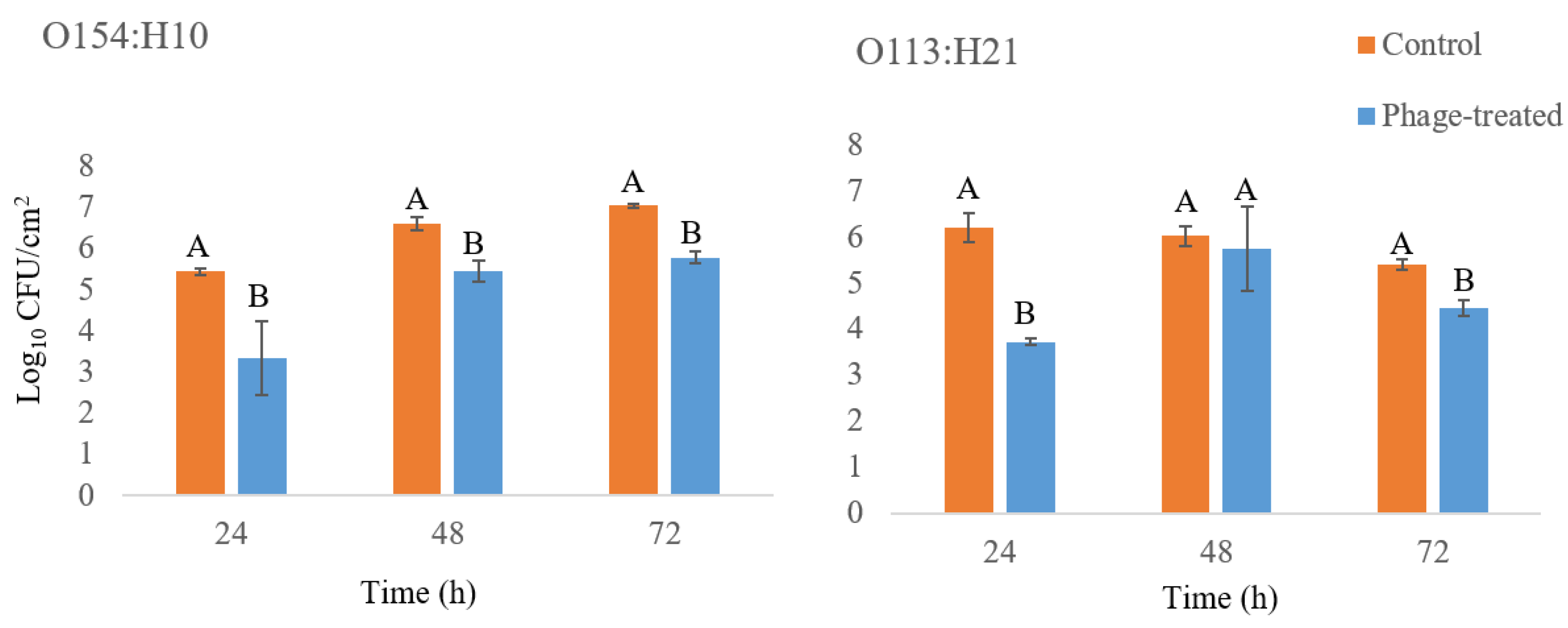
2.1.2. Treatment of Biofilms with Bacteriophage SA21RB Plate Count and Scanning Electron Microscopy (SEM)
2.2. Experiment 2
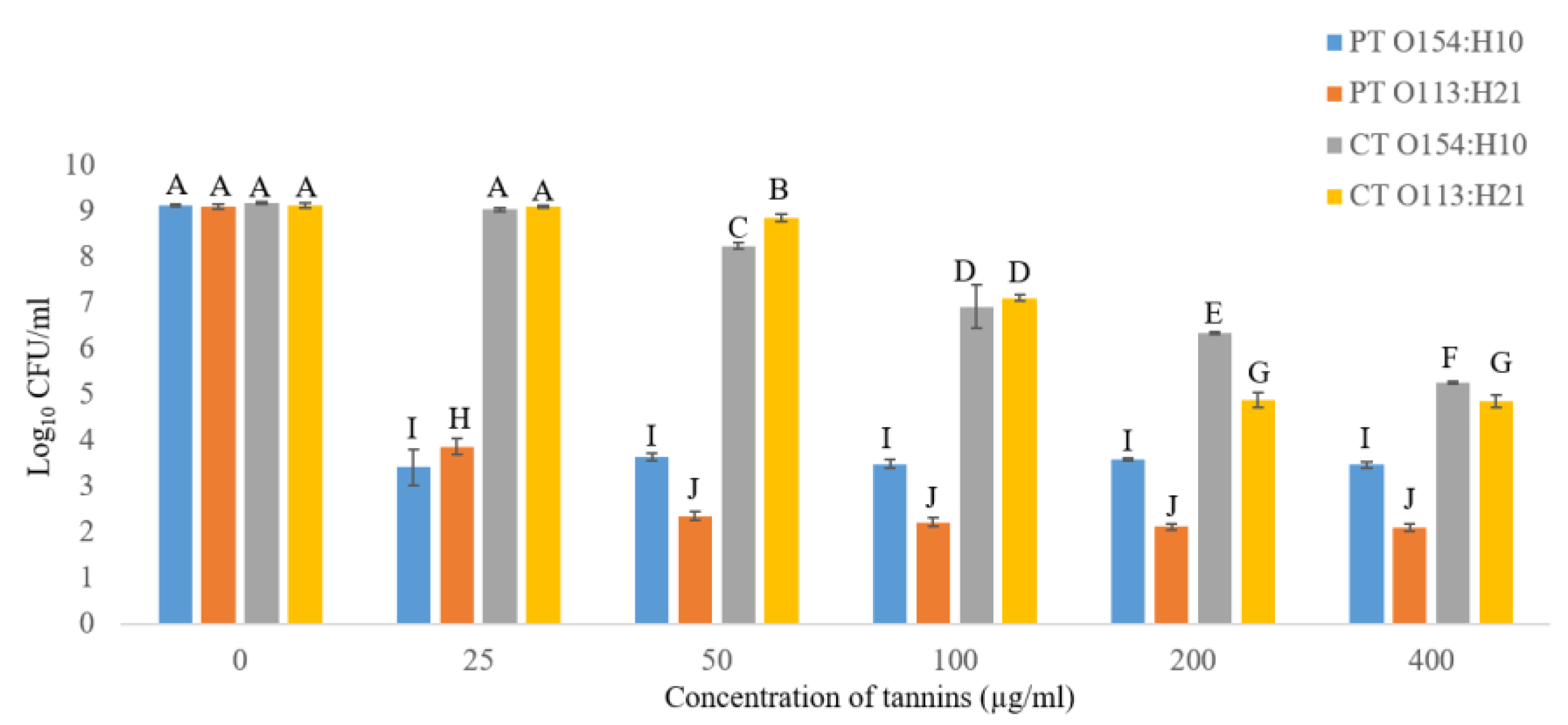
2.2.1. Effects of Condensed Tannins (CT) and Phlorotannins (PT) on Growth of O154:H10 and O113:H21
2.2.2. Anti-Biofilm Activity of Phlorotannins
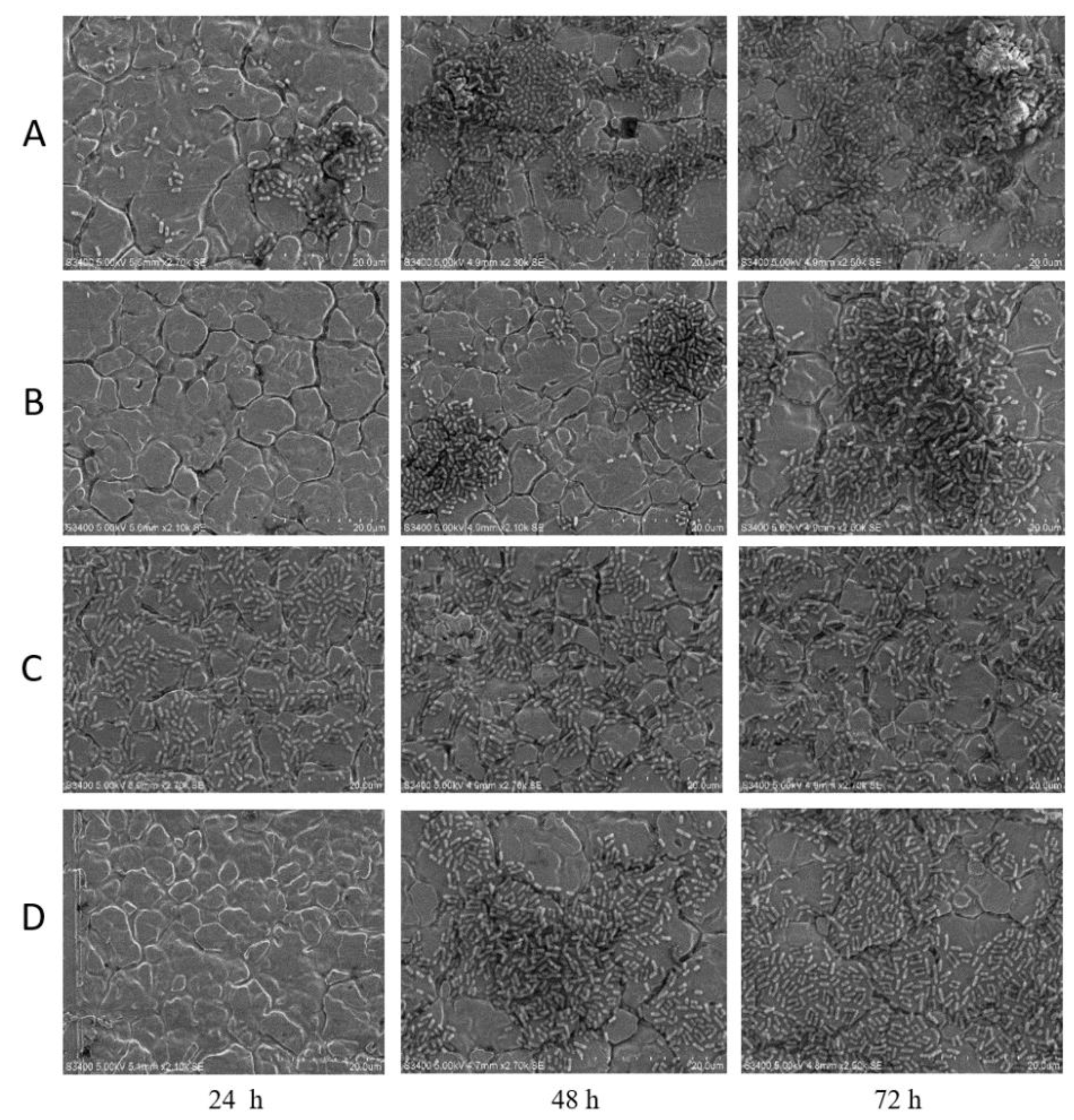
2.2.3. SEM Analysis
2.3. Experiment 3
Sensitivity of Phage SA21RB to Phlorotannins
3. Discussion
3.1. Biofilm Removal Using Phage SA21BR
3.2. Biofilm Prevention Using Phlorotannins
3.3. Inactivation of Phage SA21RB by Phlorotannins
4. Materials and Methods
4.1. Origin of Strains and Preparation
4.2. Experiment 1
4.2.1. Bacteriophage Preparation and Titration
4.2.2. One-Step Growth Curve
4.2.3. Microplate Phage Virulence Assay
4.2.4. Biofilm Removal Using Phage SA21RB
4.3. Experiment 2
4.3.1. Isolation of Condensed Tannins and Phlorotannins
4.3.2. Effects of Condensed Tannins (CT) and Phlorotannins (PT) on Growth of O154:H10 and O113:H21 E. coli
4.3.3. Anti-Biofilm Assay of Phlorotannins on Stainless Coupons
4.3.4. Enumeration of the Planktonic and Adhered Bacterial Cells after Anti-Biofilm Assay
4.4. Scanning Electron Microscopy (SEM) Analysis
4.5. Experiment 3
Virucidal Action of Phlorotannins against Phage SA21RB
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hussain, M.A.; Dawson, C.O. Economic impact of food safety outbreaks on food businesses. Foods 2013, 2, 585–589. [Google Scholar] [CrossRef]
- Beutin, L.; Martin, A. Outbreak of Shiga toxin–producing Escherichia coli (STEC) O104:H4 infection in Germany causes a paradigm shift with regard to human pathogenicity of STEC strains. J. Food Prot. 2012, 75, 408–418. [Google Scholar] [CrossRef]
- Mikhail, A.; Jenkins, C.; Dallman, T.; Inns, T.; Douglas, A.; Martín, A.; Fox, A.; Cleary, P.; Elson, R.; Hawker, J. An outbreak of Shiga toxin-producing Escherichia coli O157:H7 associated with contaminated salad leaves: Epidemiological, genomic and food trace back investigations. Epidemiol. Infect. 2018, 146, 187–196. [Google Scholar] [CrossRef]
- Wilson, D.; Dolan, G.; Aird, H.; Sorrell, S.; Dallman, T.J.; Jenkins, C.; Robertson, L.; Gorton, R. Farm-to-fork investigation of an outbreak of Shiga toxin-producing. Escherichia coli O157. Microb. Genom. 2018, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Karmali, M.A.; Mascarenhas, M.; Shen, S.; Ziebell, K.; Johnson, S.; Reid-Smith, R.; Isaac-Renton, J.; Clark, C.; Rahn, K.; Kaper, J.B. Association of genomic O island 122 of Escherichia coli EDL 933 with verocytotoxin-producing Escherichia coli seropathotypes that are linked to epidemic and/or serious disease. J. Clin. Microbiol. 2003, 41, 4930–4940. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.C.; Delannoy, S.; Lacher, D.W.; dos Santos, L.F.; Beutin, L.; Fach, P.; Rivas, M.; Hartland, E.L.; Paton, A.W.; Guth, B.E. Genetic diversity and virulence potential of Shiga toxin-producing Escherichia coli O113:H21 strains isolated from clinical, environmental, and food sources. Appl. Environ. Microbiol. 2014, 80, 4757–4763. [Google Scholar] [CrossRef] [PubMed]
- Bumunang, E.W.; McAllister, T.A.; King, R.; Ortega Polo, R.; Stanford, K.; Zaheer, R.; Niu, Y.D.; Ateba, C.N. Characterization of Non-O157 Escherichia coli from Cattle Faecal Samples in the North-West Province of South Africa. Microorganisms 2019, 7, 272. [Google Scholar] [CrossRef] [PubMed]
- Bumunang, E.W.; Ateba, C.; Stanford, K.; McAllister, T.A.; Niu, Y.D. Biofilm formation by South-African Shiga toxigenic non-O157 Escherichia coli on stainless steel coupons. Can. J. Microbiol. 2020, 66, 328–336. [Google Scholar] [CrossRef]
- Wang, R. Biofilms and meat safety: A mini-review. J. Food Prot. 2018, 82, 120–127. [Google Scholar] [CrossRef]
- Galie, S.; García-Gutiérrez, C.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Biofilms in the food industry: Health aspects and control methods. Front. Microbiol. 2018, 9, 1–18. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Wingender, J.; Szewzyk, U.; Steinberg, P.; Rice, S.A.; Kjelleberg, S. Biofilms: An emergent form of bacterial life. Nat. Rev. Microbiol. 2016, 14, 563–575. [Google Scholar] [CrossRef]
- Lahiri, D.; Dash, S.; Dutta, R.; Nag, M. Elucidating the effect of anti-biofilm activity of bioactive compounds extracted from plants. J. Biosci. 2019, 44, 1–19. [Google Scholar] [CrossRef]
- Mulcahy, H.; Charron-Mazenod, L.; Lewenza, S. Extracellular DNA chelates cations and induces antibiotic resistance in Pseudomonas aeruginosa biofilms. PLoS Pathog. 2008, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.W.; Mah, T.-F. Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol. Rev. 2017, 41, 276–301. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.; Allan, R.N.; Howlin, R.P.; Stoodley, P.; Hall-Stoodley, L. Targeting microbial biofilms: Current and prospective therapeutic strategies. Nat. Rev. Microbiol. 2017, 15, 740–755. [Google Scholar] [CrossRef] [PubMed]
- Chmielewski, R.; Frank, J. Biofilm formation and control in food processing facilities. Compr. Rev. Food Sci. 2003, 2, 22–32. [Google Scholar] [CrossRef]
- Bayoumi, M.A.; Kamal, R.M.; El Aal, S.F.A.; Awad, E.I. Assessment of a regulatory sanitization process in Egyptian dairy plants in regard to the adherence of some food-borne pathogens and their biofilms. Int. J. Food Microbiol. 2012, 158, 225–231. [Google Scholar] [CrossRef]
- Singh, S.; Singh, S.K.; Chowdhury, I.; Singh, R. Understanding the mechanism of bacterial biofilms resistance to antimicrobial agents. Open Microbiol. J. 2017, 11, 53–62. [Google Scholar] [CrossRef]
- Falowo, A.B.; Akimoladun, O.F. Veterinary Drug Residues in Meat and Meat Products: Occurrence, Detection and Implications: Veterinary Pharmaceuticals. IntechOpen 2019, 1–18. [Google Scholar]
- Joerger, R. Alternatives to antibiotics: Bacteriocins, antimicrobial peptides and bacteriophages. Poult Sci. 2003, 82, 640–647. [Google Scholar] [CrossRef]
- Hagens, S.; Loessner, M.J. Bacteriophage for biocontrol of foodborne pathogens: Calculations and considerations. Curr. Pharm. Biotechnol. 2010, 11, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Drulis-Kawa, Z.; Majkowska-Skrobek, G.; Maciejewska, B.; Delattre, A.-S.; Lavigne, R. Learning from bacteriophages-advantages and limitations of phage and phage-encoded protein applications. Curr. Protein Pept. Sci. 2012, 13, 699–722. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.K.; Abedon, S.T. Bacteriophages and their enzymes in biofilm control. Curr. Pharm. Des. 2015, 21, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Ryu, J.H.; Beuchat, L. Inactivation of Escherichia coli O157:H7 in biofilm on stainless steel by treatment with an alkaline cleaner and a bacteriophage. J. Appl. Microbiol. 2005, 99, 449–459. [Google Scholar] [CrossRef]
- Chibeu, A.; Lingohr, E.J.; Masson, L.; Manges, A.; Harel, J.; Ackermann, H.-W.; Kropinski, A.M.; Boerlin, P. Bacteriophages with the ability to degrade uropathogenic Escherichia coli biofilms. Viruses 2012, 4, 471–487. [Google Scholar] [CrossRef]
- Arachchi, G.J.G.; Cridge, A.G.; Dias-Wanigasekera, B.M.; Cruz, C.D.; McIntyre, L.; Liu, R.; Flint, S.H.; Mutukumira, A.N. Effectiveness of phages in the decontamination of Listeria monocytogenes adhered to clean stainless steel, stainless steel coated with fish protein, and as a biofilm. J. Ind. Microbiol. 2013, 40, 1105–1116. [Google Scholar]
- Abedon, S. Ecology of anti-biofilm agents II: Bacteriophage exploitation and biocontrol of biofilm bacteria. Pharmaceuticals 2015, 8, 559–589. [Google Scholar] [CrossRef]
- Gutiérrez, D.; Rodríguez-Rubio, L.; Martínez, B.; Rodríguez, A.; García, P. Bacteriophages as weapons against bacterial biofilms in the food industry. Front. Microbiol. 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Slobodníková, L.; Fialová, S.; Rendeková, K.; Kováč, J.; Mučaji, P. Antibiofilm activity of plant polyphenols. Molecules 2016, 21, 1717. [Google Scholar] [CrossRef]
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.-F.; Alarcon, E.I. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, 1–36. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, Z.; Bach, S.; McAllister, T. Sensitivity of Escherichia coli to seaweed (Ascophyllum nodosum) phlorotannins and terrestrial tannins. Asian-Australas. J. Anim. Sci. 2009, 22, 238–245. [Google Scholar] [CrossRef]
- Olajuyigbe, O.; Afolayan, A. In vitro antibacterial activities of crude aqueous and ethanolic extracts of the stem bark of Acacia mearnsii De Wild. Afr. J. Pharm. 2011, 5, 1234–1240. [Google Scholar] [CrossRef]
- Wang, Y.; Jin, L.; Ominski, K.H.; He, M.; Xu, Z.; Krause, D.O.; Acharya, S.N.; Wittenberg, K.M.; Liu, X.L.; Stanford, K.; et al. Screening of condensed tannins from Canadian prairie forages for anti-Escherichia coli O157:H7 with an emphasis on purple prairie clover (Dalea purpurea Vent). J. Food Prot. 2013, 76, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Koczan, J.M.; Lenneman, B.R.; McGrath, M.J.; Sundin, G.W. Cell surface attachment structures contribute to biofilm formation and xylem colonization by Erwinia amylovora. Appl. Environ. Microbiol. 2011, 77, 7031–7039. [Google Scholar] [CrossRef]
- Trentin, D.S.; Silva, D.B.; Amaral, M.W.; Zimmer, K.R.; Silva, M.V.; Lopes, N.P.; Giordani, R.B.; Macedo, A.J. Tannins possessing bacteriostatic effect impair Pseudomonas aeruginosa adhesion and biofilm formation. PLoS ONE 2013, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.J.; Yu, H.H.; Kim, Y.J.; Lee, N.-K.; Paik, H.-D. Anti-Biofilm Activity of Grapefruit Seed Extract against Staphylococcus aureus and Escherichia coli. J. Microbiol. Biotechnol. 2019, 29, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Bennick, A. Interaction of plant polyphenols with salivary proteins. Crit. Rev. Oral Biol. Med. 2002, 13, 184–196. [Google Scholar] [CrossRef]
- Paengkoum, P.; Phonmun, T.; Liang, J.; Huang, X.; Tan, H.; Jahromi, M. Molecular Weight, Protein Binding Affinity and Methane Mitigation of Condensed Tannins from Mangosteen-peel (Garcinia mangostana L). Asian-Australas. J. Anim. Sci. 2015, 28, 1442–1448. [Google Scholar] [CrossRef]
- Bumunang, E.W.; McAllister, T.A.; Stanford, K.; Anany, H.; Niu, Y.D.; Ateba, C.N. Characterization of Non-O157 STEC Infecting Bacteriophages Isolated from Cattle Faeces in North-West South Africa. Microorganisms 2019, 7, 615. [Google Scholar] [CrossRef]
- Kokare, C.; Chakraborty, S.; Khopade, A.; Mahadik, K.R. Biofilm: Importance and applications. Indian J. Biotechnol. 2009, 8, 159–168. [Google Scholar]
- Davies, D. Understanding biofilm resistance to antibacterial agents. Nat. Rev. Drug Discov. 2003, 2, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Gopal, N.; Hill, C.; Ross, P.R.; Beresford, T.P.; Fenelon, M.A.; Cotter, P.D. The prevalence and control of Bacillus and related spore-forming bacteria in the dairy industry. Front. Microbiol. 2015, 6, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Holmfeldt, K.; Middelboe, M.; Nybroe, O.; Riemann, L. Large variabilities in host strain susceptibility and phage host range govern interactions between lytic marine phages and their Flavobacterium hosts. Appl. Environ. Microbiol. 2007, 73, 6730–6739. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.D.; Johnson, R.P.; Xu, Y.; McAllister, T.A.; Sharma, R.; Louie, M.; Stanford, K. Host range and lytic capability of four bacteriophages against bovine and clinical human isolates of Shiga toxin-producing Escherichia coli O157:H. J. Appl. Microbiol. 2009, 107, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Stone, E.; Campbell, K.; Grant, I.; McAuliffe, O. Understanding and exploiting phage–Host interactions. Viruses 2019, 11, 567. [Google Scholar] [CrossRef]
- Sadekuzzaman, M.; Yang, S.; Mizan, M.F.R.; Ha, S.D. Reduction of Escherichia coli O157:H7 in biofilms using bacteriophage BPECO. J. Food Sci. 2017, 82, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Soni, K.A.; Nannapaneni, R. Removal of Listeria monocytogenes biofilms with bacteriophage P100. J. Food Prot. 2010, 73, 1519–1524. [Google Scholar] [CrossRef]
- Sillankorva, S.; Neubauer, P.; Azeredo, J. Pseudomonas fluorescens biofilms subjected to phage phiIBB-PF7A. BMC Biotechnol. 2008, 8, 1–12. [Google Scholar] [CrossRef]
- Sillankorva, S.; Oliveira, R.; Vieira, M.J.; Sutherland, I.; Azeredo, J. Pseudomonas fluorescens infection by bacteriophage ΦS1: The influence of temperature, host growth phase and media. FEMS Microbiol. Lett. 2004, 241, 13–20. [Google Scholar] [CrossRef]
- Sillankorva, S.; Neubauer, P.; Azeredo, J. Phage control of dual species biofilms of Pseudomonas fluorescens and Staphylococcus lentus. Biofouling 2010, 26, 567–575. [Google Scholar] [CrossRef]
- Sutherland, I.W.; Hughes, K.A.; Skillman, L.C.; Tait, K. The interaction of phage and biofilms. FEMS Microbiol. Lett. 2004, 232, 1–6. [Google Scholar] [CrossRef]
- Łoś, M.; Golec, P.; Łoś, J.M.; Węglewska-Jurkiewicz, A.; Czyż, A.; Węgrzyn, A.; Węgrzyn, G.; Neubauer, P. Effective inhibition of lytic development of bacteriophages λ, P1 and T4 by starvation of their host, Escherichia coli. BMC Biotechnol. 2007, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Abedon, S.T. Bacteriophage exploitation of bacterial biofilms: Phage preference for less mature targets? FEMS Microbiol. Lett. 2016, 363, 1–5. [Google Scholar] [CrossRef]
- Harper, D.R.; Parracho, H.M.; Walker, J.; Sharp, R.; Hughes, G.; Werthén, M.; Lehman, S.; Morales, S. Bacteriophages and biofilms. Antibiotics 2014, 3, 270–284. [Google Scholar] [CrossRef]
- Yan, J.; Mao, J.; Xie, J. Bacteriophage polysaccharide depolymerases and biomedical applications. BioDrugs 2014, 28, 265–274. [Google Scholar] [CrossRef]
- Nagayama, K.; Iwamura, Y.; Shibata, T.; Hirayama, I.; Nakamura, T. Bactericidal activity of phlorotannins from the brown alga Ecklonia kurome. J. Antimicrob. Chemother. 2002, 50, 889–893. [Google Scholar] [CrossRef]
- Pérez, M.J.; Falqué, E.; Domínguez, H. Antimicrobial action of compounds from marine seaweed. Mar. Drugs 2016, 14, 1–38. [Google Scholar]
- Okuda, T.; Ito, H. Tannins of constant structure in medicinal and food plants—Hydrolyzable tannins and polyphenols related to tannins. Molecules 2011, 16, 2191–2217. [Google Scholar] [CrossRef]
- Da Silva Trentin, D.; Giordani, R.B.; Zimmer, K.R.; Da Silva, A.G.; Da Silva, M.V.; dos Santos Correia, M.T.; Baumvol, I.J.R.; Macedo, A.J. Potential of medicinal plants from the Brazilian semi-arid region (Caatinga) against Staphylococcus epidermidis planktonic and biofilm lifestyles. J. Ethnopharmacol. 2011, 137, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Hu, W.; Tian, Z.; Yuan, D.; Yi, G.; Zhou, Y.; Cheng, Q.; Zhu, J.; Li, M. Developing natural products as potential anti-biofilm agents. Chin. Med. 2019, 14, 1–14. [Google Scholar] [CrossRef]
- Moraes, J.O.; Cruz, E.A.; Souza, E.G.; Oliveira, T.C.; Alvarenga, V.O.; Peña, W.E.; Sant’Ana, A.S.; Magnani, M. Predicting adhesion and biofilm formation boundaries on stainless steel surfaces by five Salmonella enterica strains belonging to different serovars as a function of pH, temperature and NaCl concentration. Int. J. Food Microbiol. 2018, 281, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Makkar, H.; Becker, K. Effect of pH, temperature, and time on inactivation of tannins and possible implications in detannification studies. J. Agric. Food Chem. 1996, 44, 1291–1295. [Google Scholar] [CrossRef]
- Ford, L.; Theodoridou, K.; Sheldrake, G.N.; Walsh, P.J. A critical review of analytical methods used for the chemical characterisation and quantification of phlorotannin compounds in brown seaweeds. Phytochem. Anal. 2019, 30, 587–599. [Google Scholar] [CrossRef] [PubMed]
- Imbs, T.; Zvyagintseva, T. Phlorotannins are polyphenolic metabolites of brown algae. Russ. J. Mar. Biol. 2018, 44, 263–273. [Google Scholar] [CrossRef]
- Davey, M.E.; O’toole, G.A. Microbial biofilms: From ecology to molecular genetics. Microbiol. Mol. Biol. Rev. 2000, 64, 847–867. [Google Scholar] [CrossRef]
- McAllister, T.A.; Martinez, T.; Bae, H.D.; Muir, A.D.; Yanke, L.J.; Jones, G.A. Characterization of condensed tannins purified from legume forages: Chromophore production, protein precipitation, and inhibitory effects on cellulose digestion. J. Chem. Ecol. 2005, 31, 2049–2068. [Google Scholar] [CrossRef]
- Li, Y.; Iwaasa, A.; Wang, Y.; Jin, L.; Han, G.; Zhao, M. Condensed tannins concentration of selected prairie legume forages as affected by phenological stages during two consecutive growth seasons in western Canada. Can. J. Plant Sci. 2014, 94, 817–826. [Google Scholar] [CrossRef]
- Vilhelmova-Ilieva, N.; Galabov, A.S.; Mileva, M. Tannins as Antiviral Agents, Tannins-Structural Properties, Biological Properties and Current Knowledge. IntechOpen 2019, 1–14. [Google Scholar]
- Kulikov, E.E.; Golomidova, A.K.; Letarova, M.A.; Kostryukova, E.S.; Zelenin, A.S.; Prokhorov, N.S.; Letarov, A.V. Genomic sequencing and biological characteristics of a novel Escherichia coli bacteriophage 9 g, a putative representative of a new Siphoviridae genus. Viruses 2014, 6, 5077–5092. [Google Scholar] [CrossRef]
- Theisen, L.L.; Erdelmeier, C.A.; Spoden, G.A.; Boukhallouk, F.; Sausy, A.; Florin, L.; Muller, C.P. Tannins from Hamamelis virginiana bark extract: Characterization and improvement of the antiviral efficacy against influenza A virus and human papillomavirus. PLoS ONE 2014, 9, e88062. [Google Scholar] [CrossRef]
- Liu, H.; Meng, R.; Wang, J.; Niu, Y.D.; Li, J.; Stanford, K.; McAllister, T. Inactivation of Escherichia coli O157 bacteriophages by using a mixture of ferrous sulfate and tea extract. J. Food Prot. 2015, 78, 2220–2226. [Google Scholar] [CrossRef] [PubMed]
- De Siqueira, R.; Dodd, C.; Rees, C. Evaluation of the natural virucidal activity of teas for use in the phage amplification assay. Int. J. Food Microbiol. 2006, 111, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Stanford, K.; McAllister, T.A.; Johnson, R.P.; Chen, J.; Hou, H.; Zhang, G.; Niu, Y.D. Biofilm Formation, Virulence Gene Profiles, and Antimicrobial Resistance of Nine Serogroups of Non-O157 Shiga Toxin-Producing Escherichia coli. Foodborne Pathog. Dis. 2016, 13, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Bumunang, E.W.; Stanford, K.; Bie, X.; Niu, Y.D.; McAllister, T.A. Biofilm Formation by Shiga Toxin-Producing Escherichia coli on Stainless Steel Coupons as Affected by Temperature and Incubation Time. Microorganisms 2019, 7, 95. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.D.; Stanford, K.; McAllister, T.A.; Ackermann, H.-W. Characterization of 4 T1-like lytic bacteriophages that lyse Shiga-toxin Escherichia coli O157:H7. Can. J. Microbiol. 2012, 58, 923–927. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Russell, D. Molecular Cloning: A Laboratory Manual, 3rd ed.; Cold Spring Harbor: New York, NY, USA, 2001. [Google Scholar]
- Ellis, E.L.; Delbrück, M. The growth of bacteriophage. J. Gen. Physiol. 1939, 22, 365–384. [Google Scholar] [CrossRef]
- Arnold, J.; Bailey, G. Surface finishes on stainless steel reduce bacterial attachment and early biofilm formation: Scanning electron and atomic force microscopy study. Poult. Sci. 2000, 79, 1839–1845. [Google Scholar] [CrossRef]


| Phage Titer (Plaque-Forming Units per Milliliter (PFU/mL)) | O154:H10 | O113:H21 |
|---|---|---|
| 1 × 1013 | C | C |
| 1 × 1012 | SC | C |
| 1 × 1011 | T | C |
| 1 × 1010 | T | SC |
| 1 × 109 | T | T |
| Dilution | Phage Titer (PFU/mL) | |
|---|---|---|
| Phage Only | Phage-PT | |
| 1 | TNTC | 1.5 × 103 |
| 8 | TNTC | 0 |
| 9 | TNTC | 0 |
| 10 | 8.7 × 1012 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bumunang, E.W.; Ateba, C.N.; Stanford, K.; Niu, Y.D.; Wang, Y.; McAllister, T.A. Activity of Bacteriophage and Complex Tannins against Biofilm-Forming Shiga Toxin-Producing Escherichia coli from Canada and South Africa. Antibiotics 2020, 9, 257. https://doi.org/10.3390/antibiotics9050257
Bumunang EW, Ateba CN, Stanford K, Niu YD, Wang Y, McAllister TA. Activity of Bacteriophage and Complex Tannins against Biofilm-Forming Shiga Toxin-Producing Escherichia coli from Canada and South Africa. Antibiotics. 2020; 9(5):257. https://doi.org/10.3390/antibiotics9050257
Chicago/Turabian StyleBumunang, Emmanuel W., Collins N. Ateba, Kim Stanford, Yan D. Niu, Y. Wang, and Tim A. McAllister. 2020. "Activity of Bacteriophage and Complex Tannins against Biofilm-Forming Shiga Toxin-Producing Escherichia coli from Canada and South Africa" Antibiotics 9, no. 5: 257. https://doi.org/10.3390/antibiotics9050257
APA StyleBumunang, E. W., Ateba, C. N., Stanford, K., Niu, Y. D., Wang, Y., & McAllister, T. A. (2020). Activity of Bacteriophage and Complex Tannins against Biofilm-Forming Shiga Toxin-Producing Escherichia coli from Canada and South Africa. Antibiotics, 9(5), 257. https://doi.org/10.3390/antibiotics9050257







