Future Prospects for Neisseria gonorrhoeae Treatment
Abstract
1. Introduction
2. Current Treatment
3. Future Perspectives
3.1. Repurposing of Already Existing Antibiotics
3.2. New Antibiotics
3.3. Alternative Therapies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization (WHO). Global Action Plan to Control the Spread and Impact of Antimicrobial Resistance in Neisseria gonorrhoeae; WHO: Geneva, Switzerland, 2012; Available online: http://apps.who.int/iris/bitstream/10665/44863/1/9789241503501_eng.pdf (accessed on 15 May 2018).
- Bolan, G.A.; Sparling, P.F.; Wasserheit, J.N. The emerging threat of untreatable gonococcal infection. N. Engl. J. Med. 2012, 366, 485–487. [Google Scholar] [CrossRef] [PubMed]
- Hook, E.W., 3rd; Kirkcaldy, R.D. A Brief History of Evolving Diagnostics and Therapy for Gonorrhea: Lessons Learned. Clin. Infect. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Unemo, M.; Del Rio, C.; Shafer, W.M. Emerging Infections 10; Scheld, W.M., Hughes, J.M., Whitley, R.J., Eds.; American Society for Microbiology: Washington, DC, USA, 2016; Chapter 12. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Cephalosporin-Resistant Neisseria gonorrhoeae Public Health Response Plan; CDC: Atlanta, GA, USA, 2012; pp. 1–43.
- World Health Organization (WHO). Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics; WHO: Geneva, Switzerland, 2017; Available online: http://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf (accessed on 15 May 2018).
- Public Health England. UK Case of Neisseria gonorrhoeae with High-Level Resistance to Azithromycin and Resistance to Ceftriaxone Acquired Abroad; Health Protection Report; Public Health England: London, UK, 2018; Volume 12.
- Australian Government. Department of Health. Multi-Drug Resistant Gonorrhoea. 2018. Available online: http://www.health.gov.au/internet/main/publishing.nsf/Content/mr-yr18-dept-dept004.htm (accessed on 15 May 2018).
- Suay-Garcia, B.; Pérez-Gracia, M.T. Drug-Resistant Neisseria gonorrhoeae: Latest developments. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Strategies and Laboratory Methods for Strengthening Surveillance of Sexually Transmitted Infections. Available online: http://www.who.int/reproductivehealth/publications/rtis/9789241504478/en/ (accessed on 22 April 2018).
- World Health Organization (WHO). Guidelines for the Treatment of Neisseria gonorrhoeae. Available online: http://www.who.int/reproductivehealth/publications/rtis/gonorrhoea-treatment-guidelines/en/ (accessed on 22 April 2018).
- Australasia Sexual Health Alliance. Australian STI Management Guidelines for Use in Primary Care. Available online: http://www.sti.guidelines.org.au/sexually-transmissible-infections/gonorrhoea (accessed on 22 April 2018).
- Public Health Agency of Canada. Canadian Guidelines on Sexually Transmitted Infections. Available online: https://www.canada.ca/en/public-health/services/infectious-diseases/sexual-health-sexually-transmitted-infections/canadian-guidelines/sexually-transmitted-infections/canadian-guidelines-sexually-transmitted-infections-34.html (accessed on 22 April 2018).
- Bignell, C.; Unemo, M.; European STI Guidelines Editorial Board. European Guideline on the Diagnosis and Treatment of Gonorrhea in Adults. Int. J. STD AIDS 2013, 24, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Sexually Transmitted Diseases Treatment Guidelines. Available online: https://www.cdc.gov/std/tg2015/gonorrhea.htm (accessed on 22 April 2018).
- Bignell, C.; Fitzgerald, M.; Guideline Development Group; British Association for Sexual Health and HIV UK. UK national guideline for the management of gonorrhea in adults. Int. J. STD AIDS 2011, 22, 541–547. [Google Scholar] [CrossRef] [PubMed]
- The New Zealand Sexual Health Society. New Zealand Guideline for the Management of Gonorrhea, 2014, and Response to the Threat of Antimicrobial Resistance. Available online: http://www.nzshs.org/docman/guidelines/best-practice-guidelines/142-new-zealand-guideline-for-the-management-of-gonorrhoea-2014-and-response-to-the-threat-of-antimicrobial-resistance/file (accessed on 22 April 2018).
- Fifer, H.; Natarajan, U.; Jones, L.; Alexander, S.; Hughes, G.; Golparian, D.; Unemo, M. Failure of Dual Antimicrobial Therapy in Treatment of Gonorrhea. N. Engl. J. Med. 2016, 734, 2504–2506. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, K.; Chong, Y. New treatment options for infections caused by increasingly antimicrobial-resistant Neisseria gonorrhoeae. Exp. Rev. Anti-Infect. Ther. 2016, 14, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, A.; Sunniva, F.; Golparian, D.; Hamasuna, R.; Jacobsson, S.; Lindberg, M.; Jensen, J.S.; Ohnishi, M.; Unemo, M. In vitro activity and time-kill curve analysis of sitafloxacin against a global panel of antimicrobial-resistant and multidrug-resistant Neisseria gonorrhoeae isolates. APMIS 2018, 126, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Hamasuna, R.; Ohnishi, M.; Matsumoto, M.; Okumura, R.; Unemo, M.; Matsumoto, T. In vitro activity of sitafloxacin and additional newer generation Fluoroquinolones against ciprofloxacin-resistant Neisseria gonorrhoeae isolates. Microb. Drug Resist. 2018, 24, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Soge, O.O.; Salipante, S.J.; No, D.; Duffy, E.; Roberts, M.C. In Vitro Activity of Delafloxacin against Clinical Neisseria gonorrhoeae Isolates and Selection of Gonococcal Delafloxacin Resistance. Antimicrob. Agents Chemother. 2016, 60, 3106–3111. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Bala, M.; Bhargava, A.; Kakran, M.; Bhatnagar, R. In vitro efficacy of 21 dual antimicrobial combinations comprising novel and currently recommended combinations for treatment of drug resistant gonorrhoea in future era. PLoS ONE 2018, 13, e0193678. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Bala, M.; Bhargava, A.; Kakran, M.; Bhatnagar, R. In vitro synergy testing of gentamicin, an old drug suggested as future treatment option for gonorrhoea, in combination with six other antimicrobials against multidrug-resistant Neisseria gonorrhoeae strains. Sex. Transm. Dis. 2018, 45, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Alirol, E.; Wi, T.E.; Bala, M.; Bazzo, M.L.; Chen, X.S.; Deal, C.; Dillon, J.R.; Kularatne, R.; Heim, J.; Hooft van Huijsduijnen, R.; et al. Multidrug-resistant gonorrhea: A research and development roadmap to discover new medicines. PLoS Med. 2017, 14, e1002366. [Google Scholar] [CrossRef] [PubMed]
- Golparian, D.; Fernandes, P.; Ohnishi, M.; Jensen, J.S.; Unemo, M. In vitro activity of the new fluoroketolide solithromycin (CEM-101) against a large collection of clinical Neisseria gonorrhoeae isolates and international reference strains, including those with high-level antimicrobial resistance: Potential treatment option for gonorrhea? Antimicrob. Agents Chemother. 2012, 56, 2739–2742. [Google Scholar] [PubMed]
- Hook, E.W., 3rd; Golden, M.; Jamieson, B.D.; Dixon, P.B.; Harbison, H.S.; Lowens, S.; Fernandes, P. A Phase 2 Trial of Oral Solithromycin 1200 mg or 1000 mg as Single-Dose Oral Therapy for Uncomplicated Gonorrhea. Clin. Infect. Dis. 2015, 61, 1043–1048. [Google Scholar] [CrossRef] [PubMed]
- Huband, M.D.; Bradford, P.A.; Otterson, L.G.; Basarab, G.S.; Kutschke, A.C.; Giacobbe, R.A.; Patey, S.A.; Alm, R.A.; Johnstone, M.R.; Potter, M.E.; et al. In vitro antibacterial activity of AZD0914, a new spiropyrimidinetrione DNA gyrase/topoisomerase inhibitor with potent activity against Gram-positive, fastidious Gram-Negative, and atypical bacteria. Antimicrob. Agents Chemother. 2015, 59, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Unemo, M.; Ringlander, J.; Wiggins, C.; Fredlund, H.; Jacobsson, S.; Cole, M. High in vitro susceptibility to the novel spiropyrimidinetrione ETX0914 (AZD0914) among 873 contemporary clinical Neisseria gonorrhoeae isolates from 21 European countries from 2012 to 2014. Antimicrob. Agents Chemother. 2015, 59, 5220–5225. [Google Scholar] [CrossRef] [PubMed]
- Farrell, D.J.; Sader, H.S.; Rhomberg, P.R.; Scangarella-Oman, N.E.; Flamm, R.K. In vitro Activity of Gepotidacin (GSK2140944) against Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2017, 61, e02047-16. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.N.; Morris, D.H.; Avery, A.K.; Workowski, K.A.; Batteiger, B.E.; Tiffany, C.A.; Perry, C.R.; Raychaudhuri, A.; Scangarella-Oman, N.E.; Hossain, M.; et al. Gepotidacin for the Treatment of Uncomplicated Urogenital Gonorrhea: A Phase 2, Randomized, Dose-Ranging, Single-Oral Dose Evaluation. Clin. Infect. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jacobsson, S.; Paukner, S.; Golparian, D.; Jensen, J.S.; Unemo, M. In vitro activity of the novel pleuromutilin lefamulin (BC-3781) and effect of efflux pump inactivation on multidrug-resistant and extensively-drug resistant Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2017, 61, e01497. [Google Scholar] [CrossRef] [PubMed]
- Paukner, S.; Gruss, A.; Jensen, J.S. In Vitro Activity of Lefamulin against Sexually Transmitted Bacterial Pathogens. Antimicrob. Agents Chemother. 2018, 62, e02380-17. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.M.; Waidyarachchi, S.L.; Connolly, K.L.; Jerse, A.E.; Chai, W.; Lee, R.E.; Kohlhoff, S.A.; Shinabarger, D.L.; Bowlin, T.L. Aminoethyl spectinomycins as therapeutics for drug-resistant gonorrhea and chlamydial co-infections. Antimicrob. Agents Chemother. 2018, 65. [Google Scholar] [CrossRef]
- Fedarovich, A.; Djordjevic, K.A.; Swanson, S.M.; Peterson, Y.K.; Nicholas, R.A.; Davies, C. High-Throughput Screening for Novel Inhibitors of Neisseria gonorrhoeae Penicillin-Binding Protein 2. PLoS ONE 2012, 7, e44918. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Perez, J.; Hammer, L.A.; Gallagher, H.C.; de Jesus, M.; Egilmez, N.K.; Russell, M.W. Intravaginal Administration of Interleukin 12 during Genital Gonococcal Infection in Mice Induces Immunity to Heterologous Strains of Neisseria gonorrhoeae. mSphere 2018, 3, e00421. [Google Scholar] [CrossRef] [PubMed]
- Foschi, C.; Salvo, M.; Cevenini, R.; Parolin, C.; Vitali, B.; Marangoni, A. Vaginal Lactobacilli Reduce Neisseria gonorrhoeae Viability through Multiple Strategies: An in vitro Study. Front. Cell Infect. Microbiol. 2017, 7, 502. [Google Scholar] [CrossRef] [PubMed]
- Darling, E.K.; McDonald, H. A meta-analysis of the efficacy of ocular prophylactic agents used for the prevention of gonococcal and chlamydial ophtalmia neonatorum. J. Midwif. Womens Health 2010, 55, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Churchward, C.P.; Alany, R.G.; Kirk, R.S.; Walker, A.J.; Snyder, L.A.S. Prevention of Ophthalmia Neonatorum Caused by Neisseria gonorrhoeae Using a Fatty Acid-Based Formulation. MBio 2017, 8, e00534-17. [Google Scholar] [CrossRef] [PubMed]
- Connor, D.O.; Zantow, J.; Hust, M.; Bier, F.F.; von Nickisch-Rosenegk, M. Identification of Novel Immunogenic Proteins of Neisseria gonorrhoeae by Phage Display. PLoS ONE 2016, 11, e0148986. [Google Scholar] [CrossRef] [PubMed]
- Sikora, A.E.; Mills, R.H.; Weber, J.V.; Hamza, A.; Passow, B.W.; Romaine, A.; Williamson, Z.A.; Reed, R.W.; Zielke, R.A.; Korotkov, K.V. Peptide Inhibitors Targeting the Neisseria gonorrhoeae Pivotal Anaerobic Respiration Factor AniA. Antimicrob. Agents Chemother. 2017, 61, e00186-17. [Google Scholar] [CrossRef] [PubMed]
- Biedenbach, D.J.; Bouchillon, S.K.; Hackel, M.; Miller, L.A.; Scangarella-Oman, N.E.; Jakielaszek, C.; Sahm, D.F. In Vitro Activity of Gepotidacin, a Novel Triazaacenaphthylene Bacterial Topoisomerase Inhibitor, against a Broad Spectrum of Bacterial Pathogens. Antimicrob. Agents Chemother. 2016, 60, 1918–1923. [Google Scholar] [CrossRef] [PubMed]
- Churchward, C.P.; Calder, A.; Snyder, L.A.S. Mutations in Neisseria gonorrhoeae grown in sub-lethal concentrations of monocaprin do not confer resistance. PLoS ONE 2018, 13, e0195453. [Google Scholar] [CrossRef] [PubMed]
- Golkar, K.; Bagasra, O.; Pace, D.G. Bacteriophage therapy: A potential solution for the antibiotic resistance crisis. J. Infect. Dev. Ctries. 2014, 8, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 162–173. [Google Scholar] [CrossRef] [PubMed]
| WHO * [11] | Australasia [12] | Canada [13] | USA [14] | UK [15] | EU [16] | New Zealand [17] |
|---|---|---|---|---|---|---|
| Ceftriaxone 250 mg IM + Azithromycin 1 g PO Or ** Cefixime 400 mg PO + Azithromycin 1 g PO | Cetriaxone 500 mg IM + Azithromycin 1 g PO | Ceftriaxone 250 mg IM + Azithromycin 1 g PO | Ceftriaxone 250 mg IM + Azithromycin 1 g PO | Ceftriaxone 500 mg IM + Azithromycin 1 g PO | Ceftriaxone 500 mg IM + Azithromycin 1 g PO | Ceftriaxone 250 mg IM + Azithromycin 1 g PO |
| Ceftriaxone 500 mg IM + Azithromycin 2 g PO Or ** Cefixime 800 mg PO + Azithromycin 2 g PO Or ** Gentamicin 240 mg IM + Azithromycin 2 g PO Or ** Spectinomycin 2 g IM + Azithromycin 2 g PO | Cefixime 800 mg PO + Azithromycin 1 g PO Or ** Spectinomycin 2 g IM + Azithromycin 1 g PO | Cefixime 400 mg PO + Azithromycin 1 g PO | Cefixime 400 mg PO + Azithromycin 1 g PO Or ** Spectinomycin 2 g IM + Azithromycin 1 g PO Or ** Cefotaxime 500 mg IM + Azithromycin 1 g PO | Cefixime 400 mg PO + Azithromycin 2 g PO Or ** Spectinomycin 2 g IM + Azithromycin 2 g PO | Spectinomycin 2 g IM + Azithromycin 1 g PO Or ** Gentamicin 240 mg IM + Azithromycin 2 g PO |
| Future Options | Name | Action Mechanism | Structure | Reference |
|---|---|---|---|---|
| Drug repurposing | Sitafloxacin | DNA gyrase and topoisomerase IV inhibitor |  | [20,21] |
| Delafloxacin | DNA gyrase and topoisomerase IV inhibitor |  | [22] | |
| Novel dual therapies | - | - | [23,24] | |
| New antibacterial agents | Solithromycin | Protein synthesis inhibitor |  | [25,26,27] |
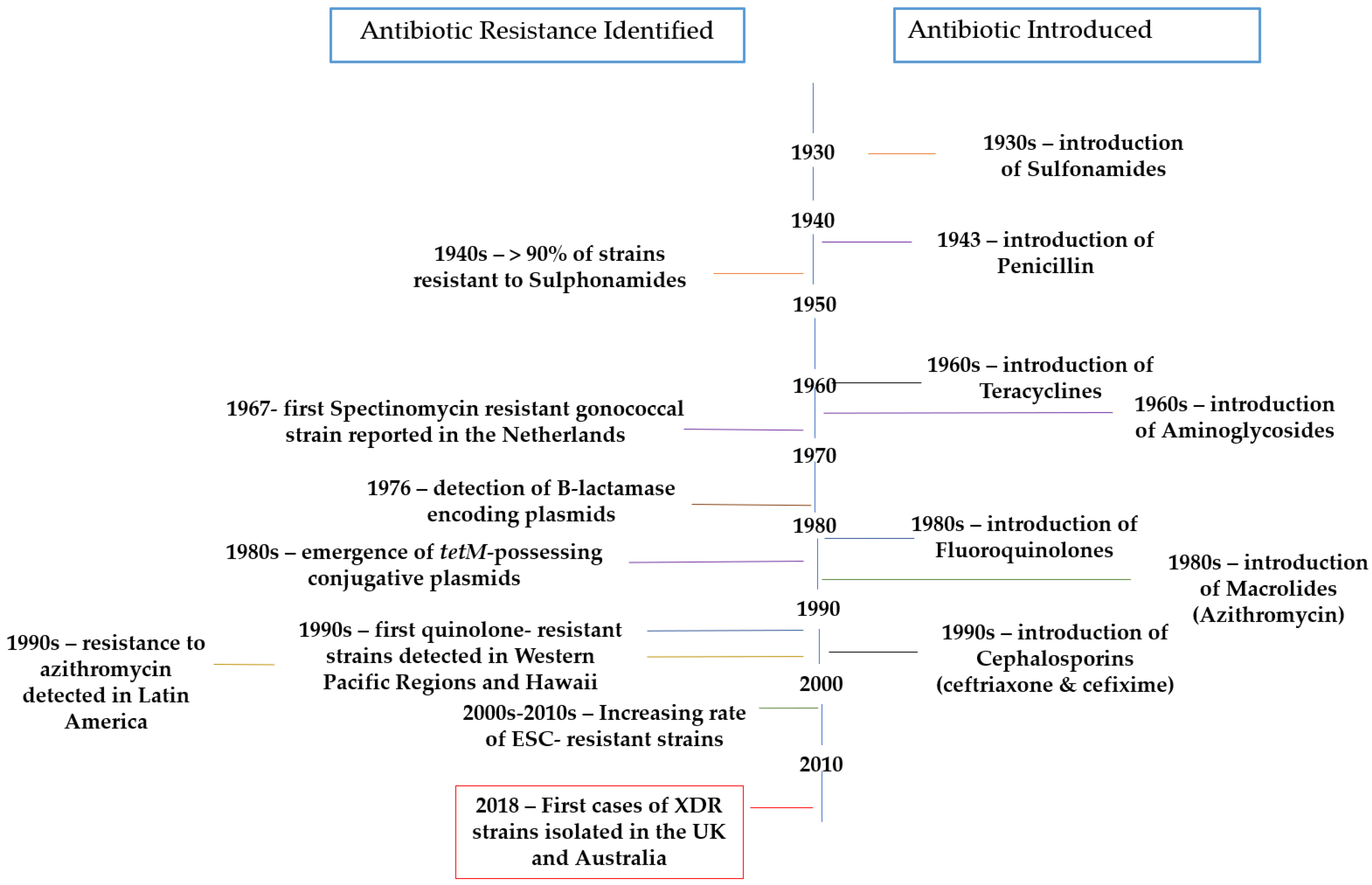
| Zoliflodacin | Spiropyrimidinetrione topoisomerase inhibitor | 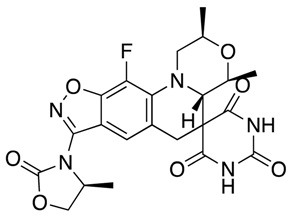 | [28,29] | |
| Gepotidacin | DNA gyrase and topoisomerase IV inhibitor | 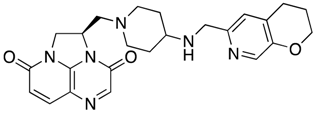 | [25,30,31] | |
| Lefamulin | Protein synthesis inhibitor |  | [32,33] | |
| Aminoethyl spectinomycins | Protein synthesis inhibitor | 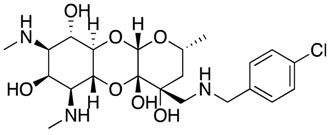 | [34] | |
| PBP2 inhibitors | Inhibition of cell wall synthesis | - | [35] | |
| Alternative therapies | IL-12 | Induction of immune response | - | [36] |
| Lactobacillus crispatus | Biosurfactant and acidic environment | - | [37] | |
| Monocaprin Myristoleic acid | Cell membrane disruption | 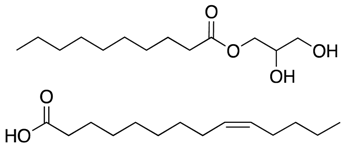 | [38,39] | |
| Bacteriophage therapy | Lysis | - | [40,41] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suay-García, B.; Pérez-Gracia, M.T. Future Prospects for Neisseria gonorrhoeae Treatment. Antibiotics 2018, 7, 49. https://doi.org/10.3390/antibiotics7020049
Suay-García B, Pérez-Gracia MT. Future Prospects for Neisseria gonorrhoeae Treatment. Antibiotics. 2018; 7(2):49. https://doi.org/10.3390/antibiotics7020049
Chicago/Turabian StyleSuay-García, Beatriz, and María Teresa Pérez-Gracia. 2018. "Future Prospects for Neisseria gonorrhoeae Treatment" Antibiotics 7, no. 2: 49. https://doi.org/10.3390/antibiotics7020049
APA StyleSuay-García, B., & Pérez-Gracia, M. T. (2018). Future Prospects for Neisseria gonorrhoeae Treatment. Antibiotics, 7(2), 49. https://doi.org/10.3390/antibiotics7020049






