Complex Regulatory Networks Governing Production of the Glycopeptide A40926
Abstract
1. The Glycopeptides
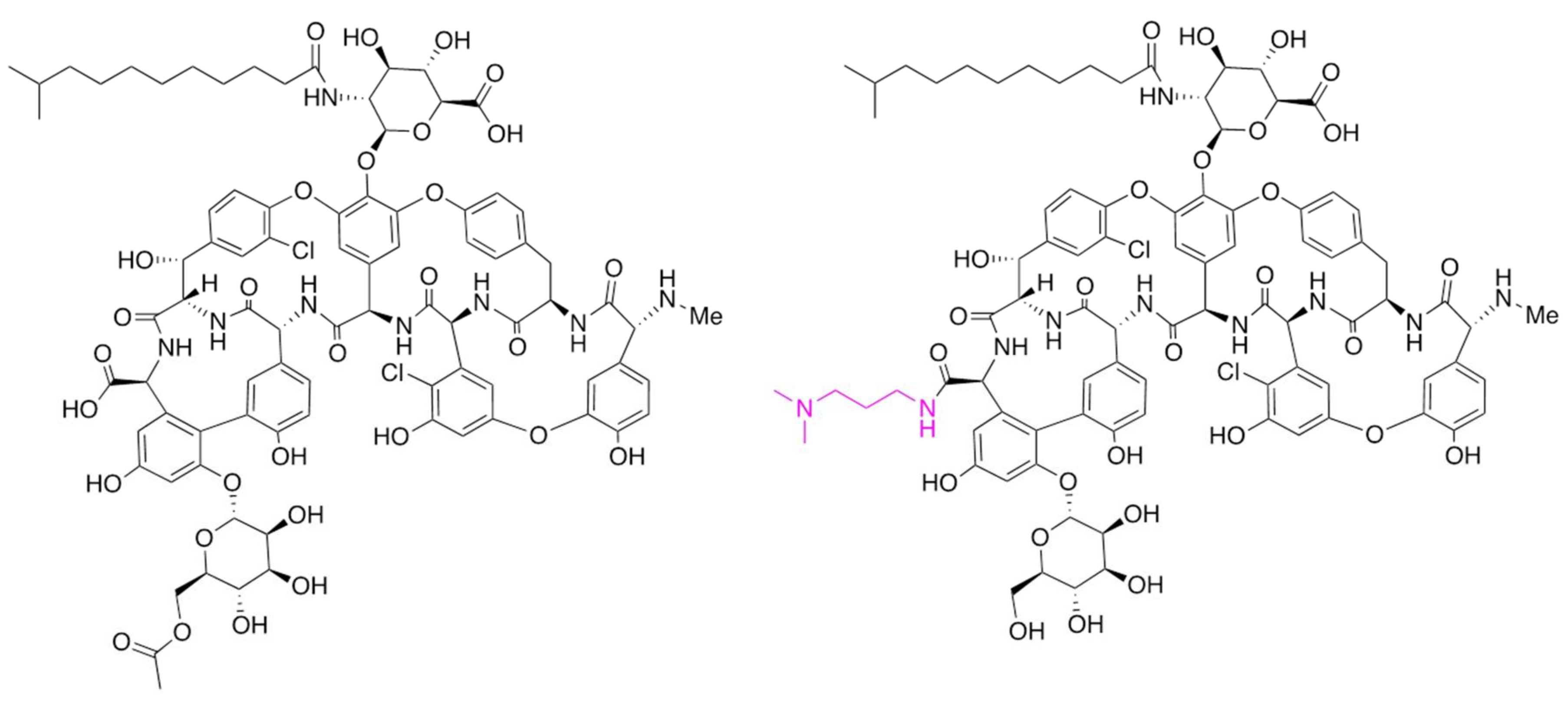
2. Development of Dalbavancin
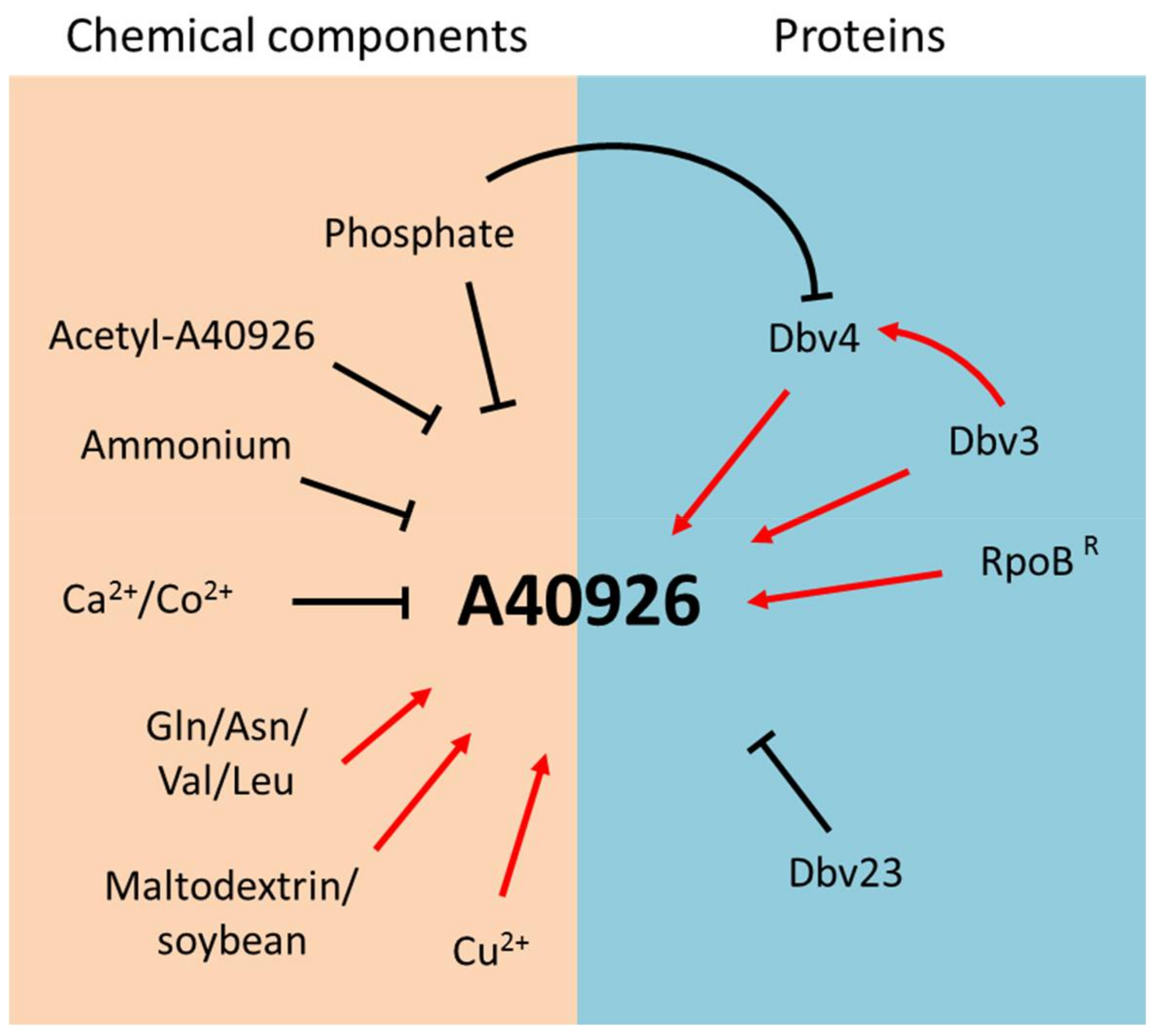
3. Improvement of A40926 Production
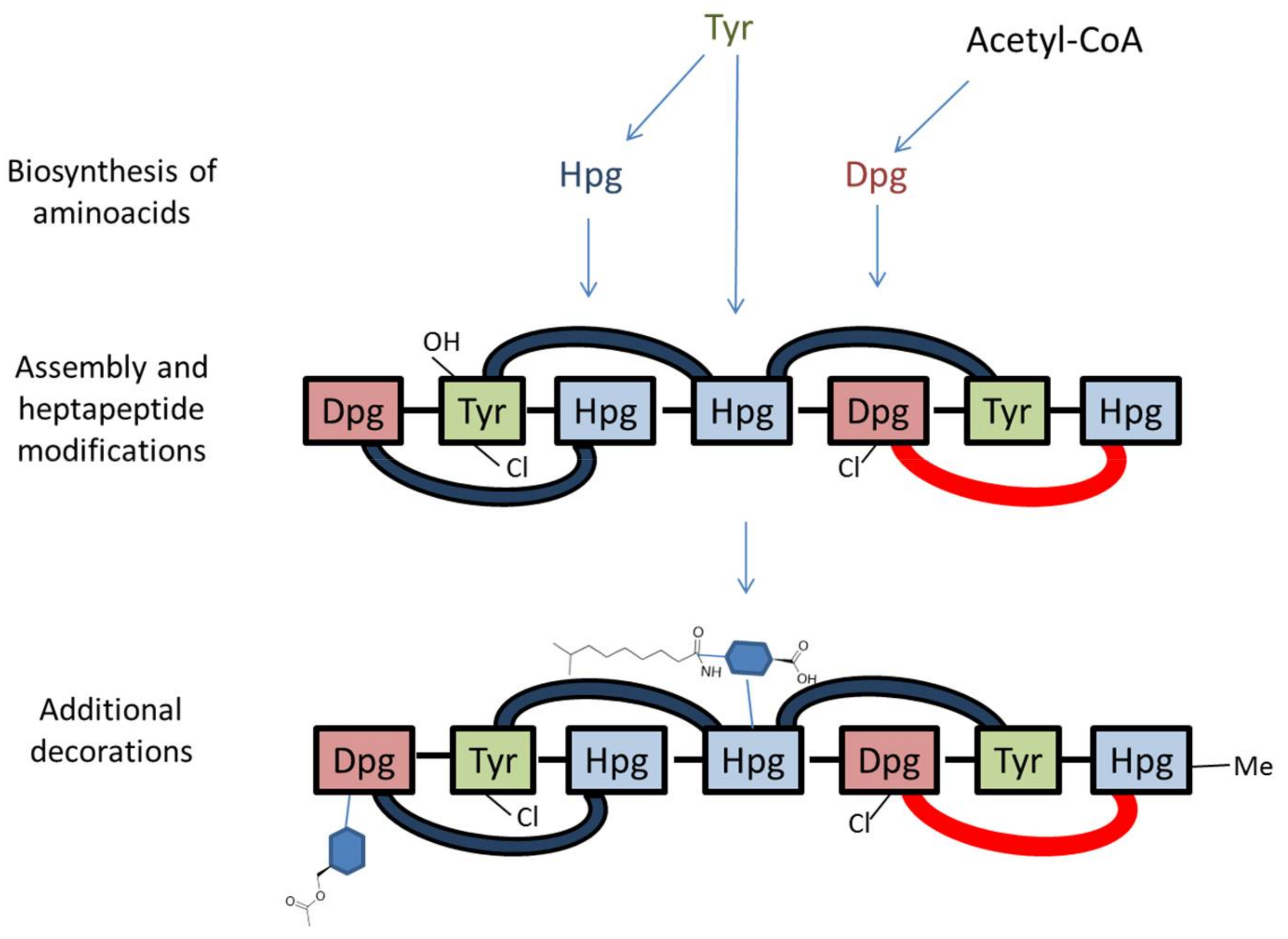
4. The dbv Gene Cluster: Main Features
5. Cluster-Specific Regulatory Genes
6. Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kahne, D.; Leimkuhler, C.; Lu, W.; Walsh, C. Glycopeptide and Lipoglycopeptide Antibiotics. Chem. Rev. 2005, 105, 425–448. [Google Scholar] [CrossRef] [PubMed]
- Van Bambeke, F. Lipoglycopeptide antibacterial agents in gram-positive infections: A comparative review. Drugs 2015, 75, 2073–2095. [Google Scholar] [CrossRef] [PubMed]
- Klinker, K.P.; Borgert, S.J. Beyond vancomycin: The tail of the lipoglycopeptides. Clin. Ther. 2015, 37, 2619–2636. [Google Scholar] [CrossRef] [PubMed]
- Tailhades, J.; Schoppet, M.; Greule, A.; Peschke, M.; Brieke, C.; Cryle, M.J. A route to diastereomerically pure phenylglycine thioester peptides: Crucial intermediates for investigating glycopeptide antibiotic biosynthesis. Chem. Commun. (Camb.) 2018, 54, 2146–2149. [Google Scholar] [CrossRef] [PubMed]
- Kittila, T.; Kittel, C.; Tailhades, J.; Butz, D.; Schoppet, M.; Buttner, A.; Goode, R.J.A.; Schittenhelm, R.B.; van Pee, K.H.; Sussmuth, R.D.; et al. Halogenation of glycopeptide antibiotics occurs at the amino acid level during non-ribosomal peptide synthesis. Chem. Sci. 2017, 8, 5992–6004. [Google Scholar] [CrossRef] [PubMed]
- Peschke, M.; Brieke, C.; Goode, R.J.A.; Schittenhelm, R.B.; Cryle, M.J. Chlorinated glycopeptide antibiotic peptide precursors improve cytochrome P450-catalyzed cyclization cascade efficiency. Biochemistry 2017, 56, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Thaker, M.N.; Wang, W.; Spanogiannopoulos, P.; Waglechner, N.; King, A.M.; Medina, R.; Wright, G.D. Identifying producers of antibacterial compounds by screening for antibiotic resistance. Nat. Biotechnol. 2013, 31, 922–927. [Google Scholar] [CrossRef] [PubMed]
- Yim, G.; Kalan, L.; Koteva, K.; Thaker, M.N.; Waglechner, N.; Tang, I.; Wright, G.D. Harnessing the synthetic capabilities of glycopeptide antibiotic tailoring enzymes: Characterization of the UK-68,597 biosynthetic cluster. Chembiochem 2014, 15, 2613–2623. [Google Scholar] [CrossRef] [PubMed]
- Yim, G.; Thaker, M.N.; Koteva, K.; Wright, G. Glycopeptide antibiotic biosynthesis. J. Antibiot. (Tokyo) 2014, 67, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Stegmann, E.; Frasch, H.J.; Wohlleben, W. Glycopeptide biosynthesis in the context of basic cellular functions. Curr. Opin. Microbiol. 2010, 13, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Malabarba, A.; Goldstein, B.P. Origin, structure, and activity in vitro and in vivo of dalbavancin. J. Antimicrob. Chemother. 2005, 55, ii15–ii20. [Google Scholar] [CrossRef] [PubMed]
- Zerilli, L.F.; Edwards, D.M.; Borghi, A.; Gallo, G.G.; Selva, E.; Denaro, M.; Lancini, G.C. Determination of the acyl moieties of the antibiotic complex A40926 and their relation with the membrane lipids of the producer strain. Rapid Commun. Mass Spectrom. 1992, 6, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Maffioli, S.I.; Cruz, J.C.S.; Monciardini, P.; Sosio, M.; Donadio, S. Advancing cell wall inhibitors towards clinical applications. J. Ind. Microbiol. Biotechnol. 2016, 43, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Dalmastri, C.; Gastaldo, L.; Marcone, G.L.; Binda, E.; Congiu, T.; Marinelli, F. Classification of Nonomuraea sp. ATCC 39727, an actinomycete that produces the glycopeptide antibiotic A40926, as Nonomuraea gerenzanensis sp. nov. Int. J. Syst. Evol. Microbiol. 2016, 66, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsson, N.; Bruheim, P.; Nielsen, J. Glucose metabolism in the antibiotic producing actinomycete Nonomuraea sp. ATCC 39727. Biotechnol. Bioeng. 2004, 88, 652–663. [Google Scholar] [CrossRef] [PubMed]
- Technikova-Dobrova, Z.; Damiano, F.; Tredici, S.M.; Vigliotta, G.; di Summa, R.; Palese, L.; Abbrescia, A.; Labonia, N.; Gnoni, G.V.; Alifano, P. Design of mineral medium for growth of Actinomadura sp. ATCC 39727, producer of the glycopeptide A40926: Effects of calcium ions and nitrogen sources. Appl. Microbiol. Biotechnol. 2004, 65, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Jovetic, S.; Feroggio, M.; Marinelli, F.; Lancini, G. Factors influencing cell fatty acid composition and A40926 antibiotic complex production in Nonomuraea sp. ATCC 39727. J. Ind. Microbiol. Biotechnol. 2008, 35, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Beltrametti, F.; Jovetic, S.; Feroggio, M.; Gastaldo, L.; Selva, E.; Marinelli, F. Valine influences production and complex composition of glycopeptide antibiotic A40926 in fermentations of Nonomuraea sp. ATCC 39727. J. Antibiot. (Tokyo) 2004, 57, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Vigliotta, G.; Tredici, S.M.; Damiano, F.; Montinaro, M.R.; Pulimeno, R.; di Summa, R.; Massardo, D.R.; Gnoni, G.V.; Alifano, P. Natural merodiploidy involving duplicated rpob alleles affects secondary metabolism in a producer actinomycete. Mol. Microbiol. 2005, 55, 396–412. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Xu, T.; Zhang, G.; Zhao, J.; Gao, Z.; Zhang, C. High-yield production of lipoglycopeptide antibiotic A40926 using a mutant strain Nonomuraea sp. Dp-13 in optimized medium. Prep. Biochem. Biotechnol. 2016, 46, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Sosio, M.; Stinchi, S.; Beltrametti, F.; Lazzarini, A.; Donadio, S. The gene cluster for the biosynthesis of the glycopeptide antibiotic A40926 by Nonomuraea species. Chem. Biol. 2003, 10, 541–549. [Google Scholar] [CrossRef]
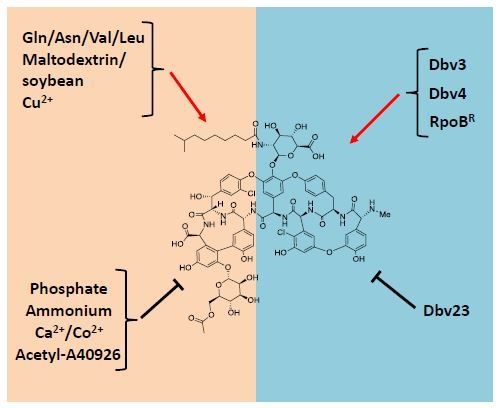
- Alduina, R.; Lo Piccolo, L.; D’Alia, D.; Ferraro, C.; Gunnarsson, N.; Donadio, S.; Puglia, A.M. Phosphate-controlled regulator for the biosynthesis of the dalbavancin precursor A40926. J. Bacteriol. 2007, 189, 8120–8129. [Google Scholar] [CrossRef] [PubMed]
- Lo Grasso, L.; Maffioli, S.; Sosio, M.; Bibb, M.; Puglia, A.M.; Alduina, R. Two master switch regulators trigger A40926 biosynthesis in Nonomuraea sp. Strain ATCC 39727. J. Bacterial. 2015, 197, 2536–2544. [Google Scholar] [CrossRef] [PubMed]
- Stinchi, S.; Carrano, L.; Lazzarini, A.; Feroggio, M.; Grigoletto, A.; Sosio, M.; Donadio, S. A derivative of the glycopeptide A40926 produced by inactivation of the beta-hydroxylase gene in Nonomuraea sp. ATCC 39727. FEMS Microbiol. Lett. 2006, 256, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Brunzelle, J.S.; Nair, S.K. Crystal structures of lipoglycopeptide antibiotic deacetylases: Implications for the biosynthesis of A40926 and teicoplanin. Chem. Biol. 2008, 15, 533–545. [Google Scholar] [CrossRef] [PubMed]
- Kruger, R.G.; Lu, W.; Oberthur, M.; Tao, J.; Kahne, D.; Walsh, C.T. Tailoring of glycopeptide scaffolds by the acyltransferases from the teicoplanin and A-40,926 biosynthetic operons. Chem. Biol. 2005, 12, 131–140. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, Y.S.; Ho, J.Y.; Huang, C.C.; Lyu, S.Y.; Lee, C.Y.; Huang, Y.T.; Wu, C.J.; Chan, H.C.; Huang, C.J.; Hsu, N.S.; et al. A unique flavin mononucleotide-linked primary alcohol oxidase for glycopeptide A40926 maturation. J. Am. Chem. Soc. 2007, 129, 13384–13385. [Google Scholar] [CrossRef] [PubMed]
- Sosio, M.; Canavesi, A.; Stinchi, S.; Donadio, S. Improved production of A40926 by Nonomuraea sp. through deletion of a pathway-specific acetyltransferase. Appl. Microbiol. Biotechnol. 2010, 87, 1633–1638. [Google Scholar] [CrossRef] [PubMed]
- Sosio, M.; Donadio, S. Understanding and manipulating glycopeptide pathways: The example of the dalbavancin precursor A40926. J. Ind. Microbiol. Biotechnol. 2006, 33, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Arthur, M.; Molinas, C.; Courvalin, P. The VanS-VanR two-component regulatory system controls synthesis of depsipeptide peptidoglycan precursors in Enterococcus faecium BM4147. J. Bacteriol. 1992, 174, 2582–2591. [Google Scholar] [CrossRef] [PubMed]
- Arthur, M.; Reynolds, P.; Courvalin, P. Glycopeptide resistance in enterococci. Trends Microbiol. 1996, 4, 401–407. [Google Scholar] [CrossRef]
- Arthur, M.; Depardieu, F.; Courvalin, P. Regulated interactions between partner and non-partner sensors and response regulators that control glycopeptide resistance gene expression in enterococci. Microbiology 1999, 145, 1849–1858. [Google Scholar] [CrossRef] [PubMed]
- Stegmann, E.; Frasch, H.J.; Kilian, R.; Pozzi, R. Self-resistance mechanisms of actinomycetes producing lipid II-targeting antibiotics. Int. J. Med. Microbiol. 2015, 305, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Frasch, H.J.; Kalan, L.; Kilian, R.; Martin, T.; Wright, G.D.; Stegmann, E. Alternative pathway to a glycopeptide-resistant cell wall in the balhimycin producer Amycolatopsis balhimycina. ACS Infect. Dis. 2015, 1, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Marcone, G.L.; Beltrametti, F.; Binda, E.; Carrano, L.; Foulston, L.; Hesketh, A.; Bibb, M.; Marinelli, F. Novel mechanism of glycopeptide resistance in the A40926 producer Nonomuraea sp. ATCC 39727. Antimicrob. Agents Chemother. 2010, 54, 2465–2472. [Google Scholar] [CrossRef] [PubMed]
- Hugonnet, J.E.; Haddache, N.; Veckerle, C.; Dubost, L.; Marie, A.; Shikura, N.; Mainardi, J.L.; Rice, L.B.; Arthur, M. Peptidoglycan cross-linking in glycopeptide-resistant actinomycetales. Antimicrob. Agents Chemother. 2014, 58, 1749–1756. [Google Scholar] [CrossRef] [PubMed]
- Menges, R.; Muth, G.; Wohlleben, W.; Stegmann, E. The ABC transporter Tba of Amycolatopsis balhimycina is required for efficient export of the glycopeptide antibiotic balhimycin. Appl. Microbiol. Biotechnol. 2007, 77, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Donadio, S.; Sosio, M.; Stegmann, E.; Weber, T.; Wohlleben, W. Comparative analysis and insights into the evolution of gene clusters for glycopeptide antibiotic biosynthesis. Mol. Genet. Genom. 2005, 274, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Shawky, R.M.; Puk, O.; Wietzorrek, A.; Pelzer, S.; Takano, E.; Wohlleben, W.; Stegmann, E. The border sequence of the balhimycin biosynthesis gene cluster from Amycolatopsis balhimycina contains bbr, encoding a strR-like pathway-specific regulator. J. Mol. Microbiol. Biotechnol. 2007, 13, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.F. Phosphate control of the biosynthesis of antibiotics and other secondary metabolites is mediated by the PhoR-PhoP system: An unfinished story. J. Bacteriol. 2004, 186, 5197–5201. [Google Scholar] [CrossRef] [PubMed]
- Sola-Landa, A.; Moura, R.S.; Martin, J.F. The two-component PhoR-PhoP system controls both primary metabolism and secondary metabolite biosynthesis in Streptomyces lividans. Proc. Natl. Acad. Sci. USA 2003, 100, 6133–6138. [Google Scholar] [CrossRef] [PubMed]
- Sola-Landa, A.; Rodriguez-Garcia, A.; Franco-Dominguez, E.; Martin, J.F. Binding of PhoP to promoters of phosphate-regulated genes in Streptomyces coelicolor: Identification of pho boxes. Mol. Microbiol. 2005, 56, 1373–1385. [Google Scholar] [CrossRef] [PubMed]
- Horbal, L.; Kobylyanskyy, A.; Truman, A.W.; Zaburranyi, N.; Ostash, B.; Luzhetskyy, A.; Marinelli, F.; Fedorenko, V. The pathway-specific regulatory genes, tei15* and tei16*, are the master switches of teicoplanin production in Actinoplanes teichomyceticus. Appl. Microbiol. Biotechnol. 2014, 98, 9295–9309. [Google Scholar] [CrossRef] [PubMed]
- D’Argenio, V.; Petrillo, M.; Pasanisi, D.; Pagliarulo, C.; Colicchio, R.; Tala, A.; de Biase, M.S.; Zanfardino, M.; Scolamiero, E.; Pagliuca, C.; et al. The complete 12 Mb genome and transcriptome of Nonomuraea gerenzanensis with new insights into its duplicated “magic” RNA polymerase. Sci. Rep. 2016, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Alduina, R.; Giardina, A.; Gallo, G.; Renzone, G.; Ferraro, C.; Contino, A.; Scaloni, A.; Donadio, S.; Puglia, A.M. Expression in Streptomyces lividans of Nonomuraea genes cloned in an artificial chromosome. Appl. Microbiol. Biotechnol. 2005, 68, 656–662. [Google Scholar] [CrossRef] [PubMed]
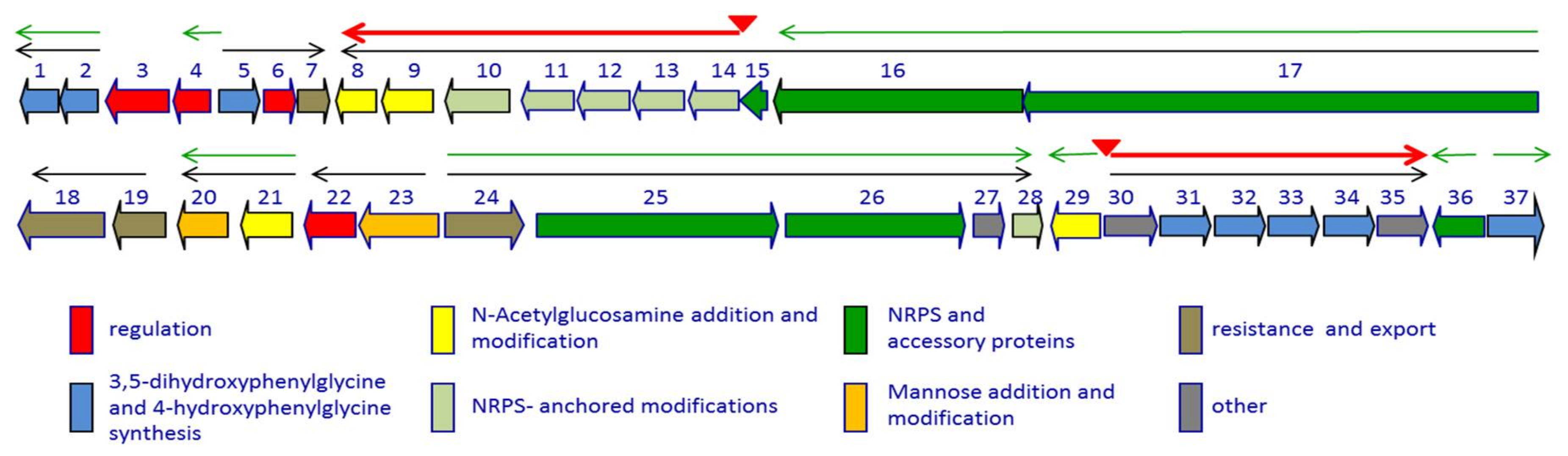
| Transcriptional Unit | Function(s) |
|---|---|
| dbv1-dbv2 | Hpg biosynthesis |
| dbv3 | Regulation |
| dbv4 | Regulation |
| dbv5-dbv7 | Hpg biosynthesis; regulation; resistance |
| dbv8-dbv14 | N-Acylation; Halogenation; glycosylation; cross-links |
| dbv15-dbv17 | NRPS |
| dbv18-dbv19 | Export |
| dbv20-dbv21 | Mannose addition; N-sugar deacetylation |
| dbv22-dbv23 | Regulation; mannose O-acetylation |
| dbv24-dbv28 | Export; NRPS; Tyr β-hydroxylation |
| dbv29 | N-sugar oxidation |
| dbv30-dbv35 | Dpg biosynthesis |
| dbv36 | NRPS accessory protein |
| dbv37 | Hpg and Dpg biosynthesis |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alduina, R.; Sosio, M.; Donadio, S. Complex Regulatory Networks Governing Production of the Glycopeptide A40926. Antibiotics 2018, 7, 30. https://doi.org/10.3390/antibiotics7020030
Alduina R, Sosio M, Donadio S. Complex Regulatory Networks Governing Production of the Glycopeptide A40926. Antibiotics. 2018; 7(2):30. https://doi.org/10.3390/antibiotics7020030
Chicago/Turabian StyleAlduina, Rosa, Margherita Sosio, and Stefano Donadio. 2018. "Complex Regulatory Networks Governing Production of the Glycopeptide A40926" Antibiotics 7, no. 2: 30. https://doi.org/10.3390/antibiotics7020030
APA StyleAlduina, R., Sosio, M., & Donadio, S. (2018). Complex Regulatory Networks Governing Production of the Glycopeptide A40926. Antibiotics, 7(2), 30. https://doi.org/10.3390/antibiotics7020030