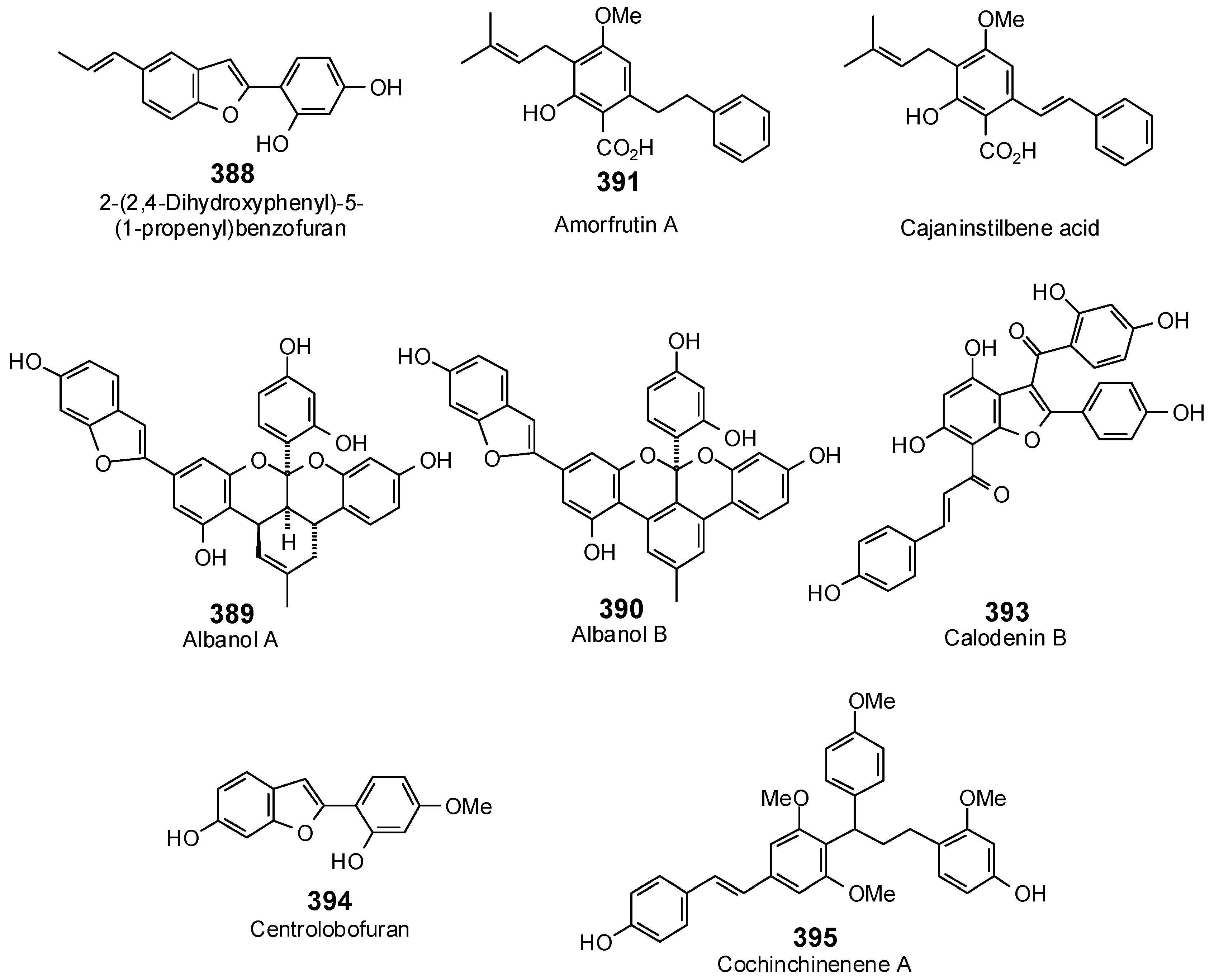
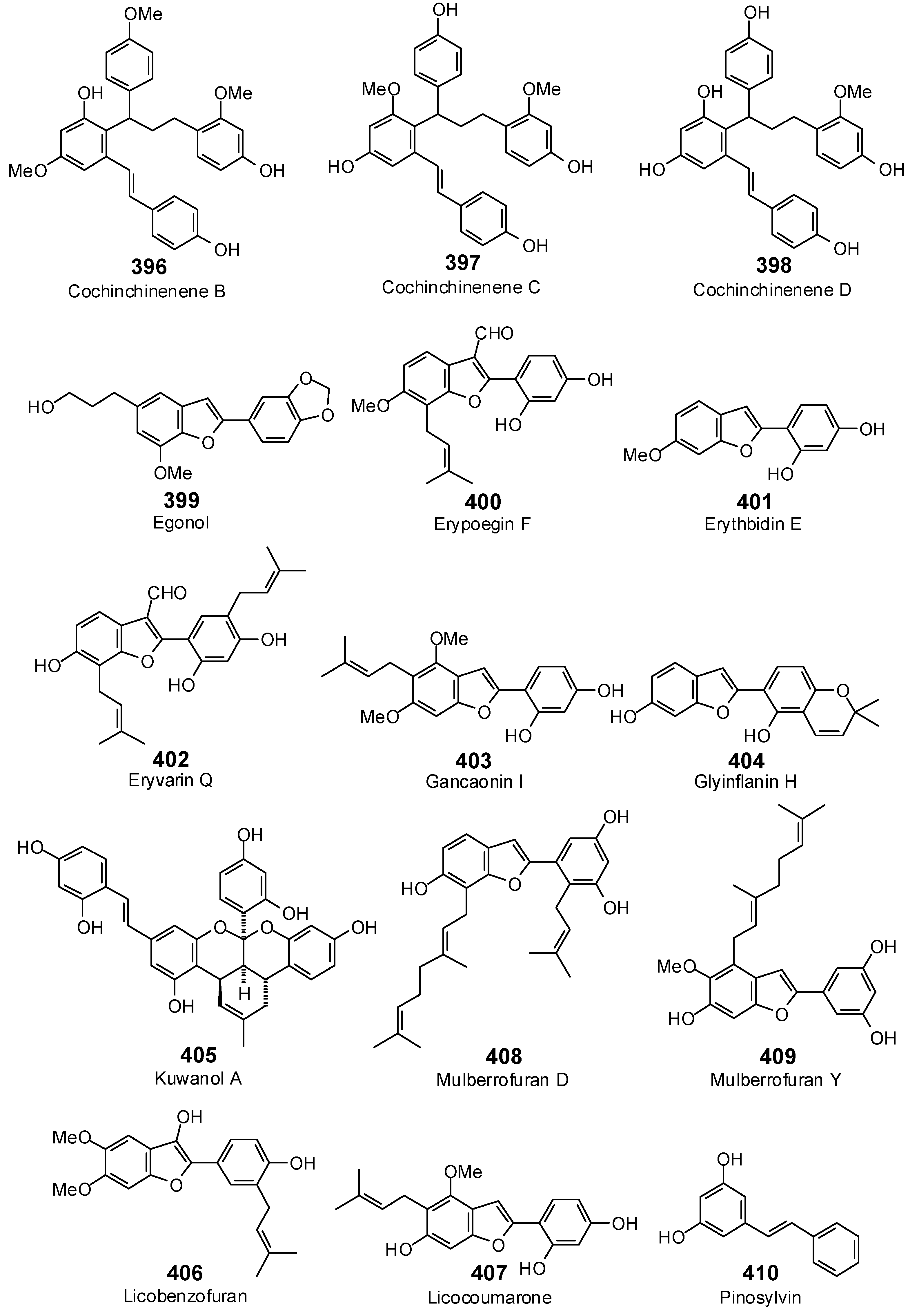
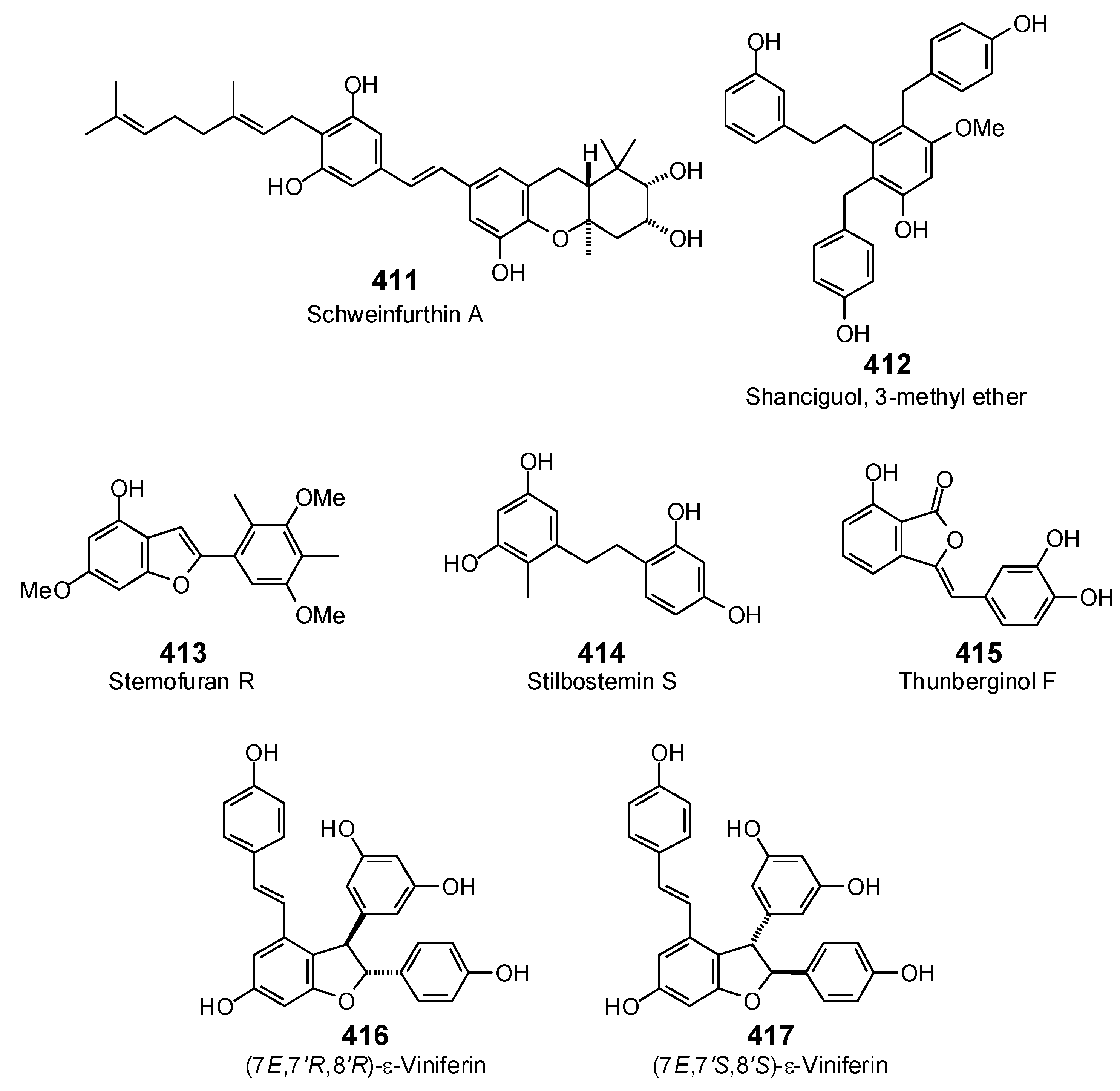
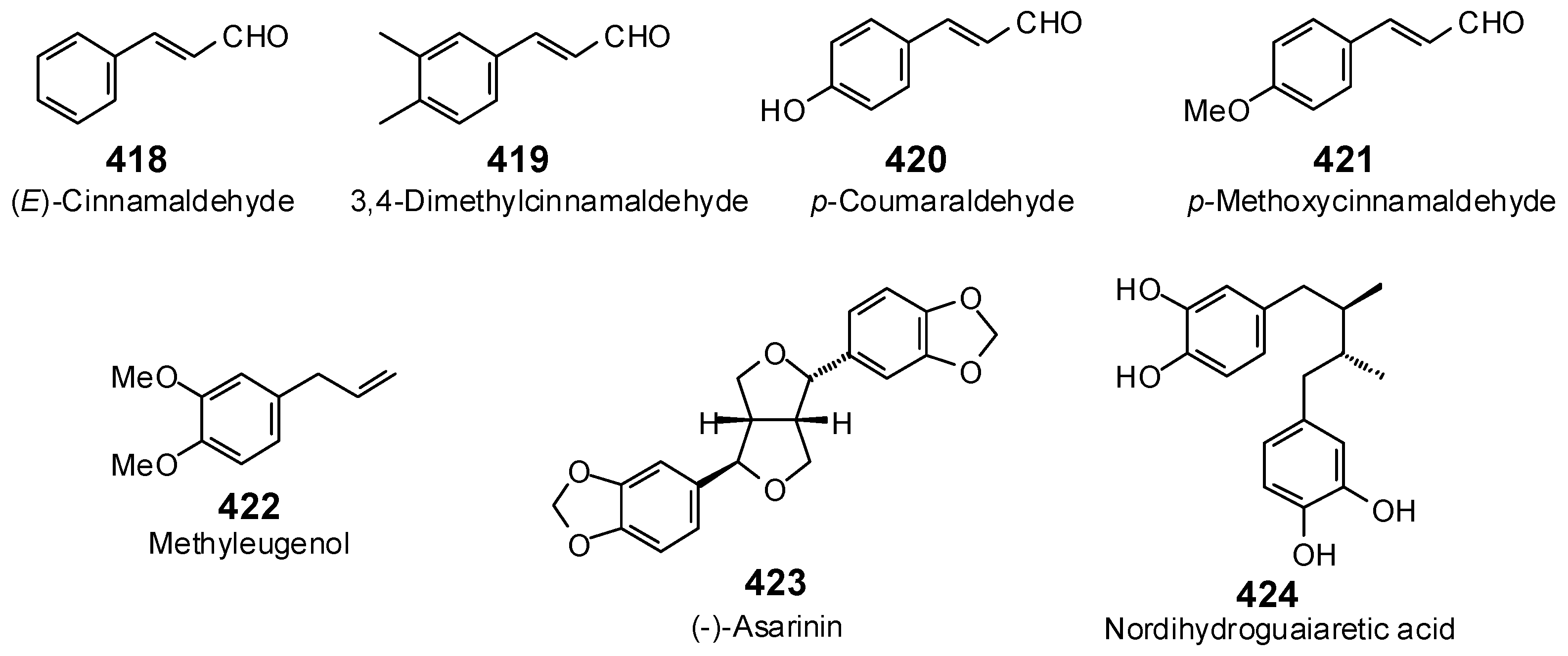
The Search for Herbal Antibiotics: An In-Silico Investigation of Antibacterial Phytochemicals
Abstract
1. Introduction
1.1. Peptide Deformylase
1.2. DNA Gyrase/Topoisomerase IV
1.3. UDP-Galactopyranose Mutase
1.4. Protein Tyrosine Phosphatase
1.5. Cytochrome P450 CYP121
1.6. NAD+-Dependent DNA Ligase
2. Computational Methods
3. Results and Discussion
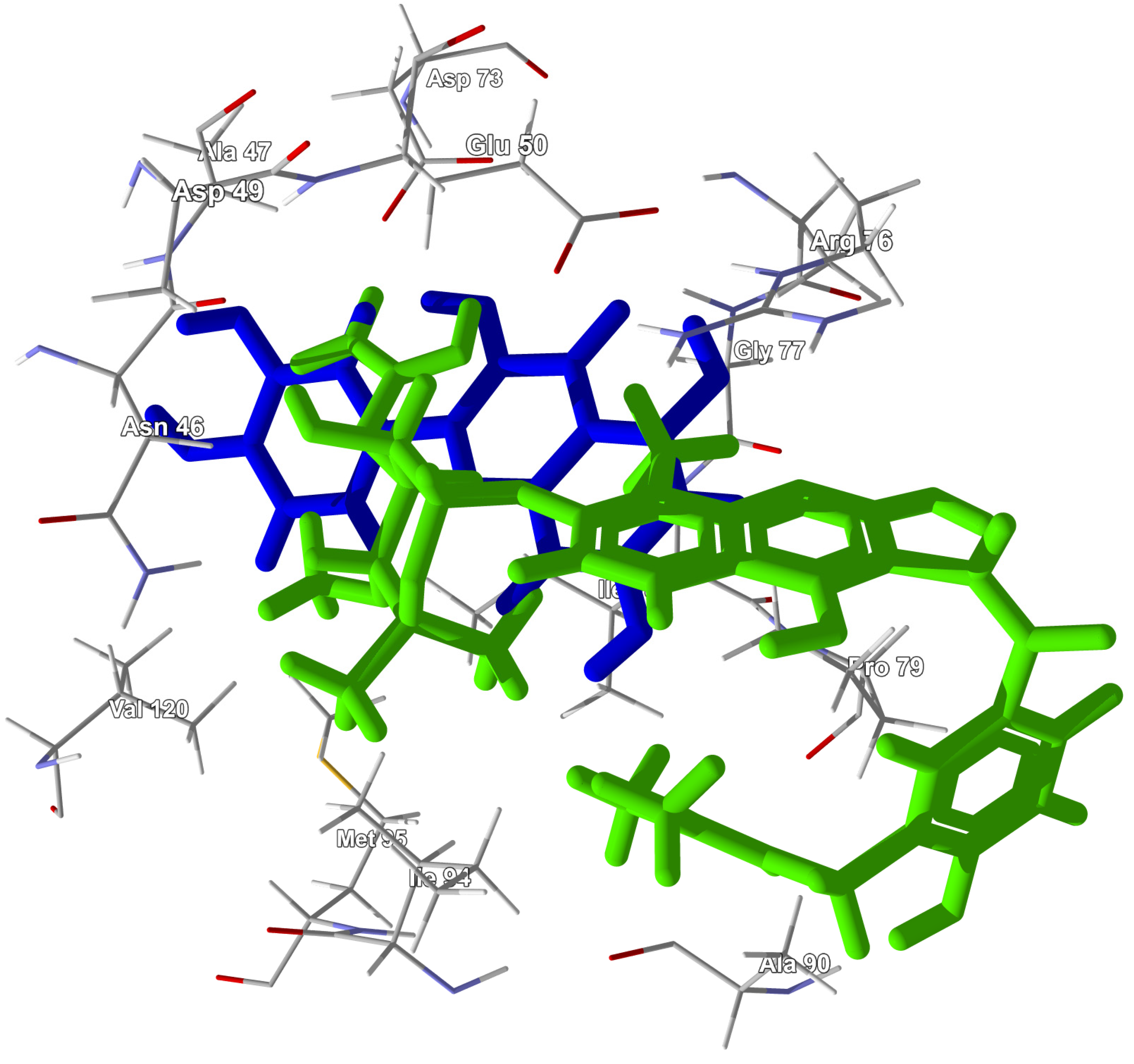
3.1. Bacterial Peptide Deformylase
3.2. Bacterial Topoisomerase IV/Gyrase B
3.3. Protein Tyrosine Phosphatase
3.4. UDP-Galactopyranose Mutase
3.5. Cytochrome P450 CYP121
3.6. NAD+-Dependent DNA Ligase
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Centers for Disease Control and Prevention (CDC). Antibiotic Resistance Threats in the United States; CDC: Atlanta, GA, USA, 2013. [Google Scholar]
- Alam, O.; Deng, T. Environmental and public health risks associated with antibiotic resistance genes (ARGs) spread in environment: A comprehensive review. Int. J. Sci. Res. Sci. Technol. 2015, 1, 128–139. [Google Scholar]
- Blair, J.M.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015, 31, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Economou, V.; Gousia, P. Agriculture and food animals as a source of antimicrobial-resistant bacteria. Infect. Drug Resist. 2015, 8, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.H.; Cohen, T.; Grad, Y.H.; Hanage, W.P.; O’Brien, T.F.; Lipsitch, M. Origin and proliferation of multiple-drug resistance in bacterial pathogens. Microbiol. Mol. Biol. Rev. 2015, 79, 101–116. [Google Scholar] [CrossRef] [PubMed]
- Van Hal, S.J.; Fowler, V.G. Is it time to replace vancomycin in the treatment of methicillin-resistant Staphylococcus aureus infections? Clin. Pract. 2013, 56, 1779–1788. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nathwani, D.; Raman, G.; Sulham, K.; Gavagham, M.; Menon, V. Clinical and economic consequences of hospital-acquired resistant and multidrug-resistant Pseudomonas aeruginosa infections: A systematic review and meta-analysis. Antimicrob. Resist. Infect. Control 2014. [Google Scholar] [CrossRef] [PubMed]
- Kim, L.; McGee, L.; Tomczyk, S.; Beall, B. Biological and epidemiological features of antibiotic-resistant Streptococcus pneumoniae in pre- and post-conjugate vaccine eras: A United States perspective. Clin. Microbiol. Rev. 2016, 29, 525–552. [Google Scholar] [CrossRef] [PubMed]
- Kempker, R.R.; Kipiani, M.; Mirtskhulava, V.; Tukvadze, N.; Magoo, M.J.; Blumberg, H.M. Acquired drug resistance in Mycobacterium tuberculosis and poor outcomes among patients with multidrug-resistant tuberculosis. Emerg. Infect. Dis. 2015, 21, 992–1001. [Google Scholar] [CrossRef] [PubMed]
- Kesavan, B.; Srividhya, K.V.; Krishnaswamy, S.; Raja, M.; Vidya, N.; Krishna Mohan, A.V. Understanding the virulence of the entero-aggregative E. coli O104:H4. Int. J. Bioinf. Res. Appl. 2015, 11, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Krüger, A.; Lucchesi, P.M.; Sanso, A.M.; Etcheverría, A.I.; Bustamante, A.V.; Burgán, J.; Fernández, D.; Leotta, G.; Friedrich, A.W.; Padola, N.L.; et al. Genetic characterization of Shiga toxin-producing Escherichia coli O26:H11 strains isolated from animal, food, and clinical samples. Front. Cell. Infect. Microbiol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gallois, C.; Hauw-Berlemont, C.; Richaud, C.; Bonacorsi, S.; Diehl, J.L.; Mainardi, J.L. Fatal necrotizing fasciitis due to necrotic toxin-producing Escherichia coli strain. New Microbes New Infect. 2015, 8, 109–112. [Google Scholar] [CrossRef] [PubMed]
- McPhillie, M.J.; Cain, R.M.; Marramore, S.; Fishwick, C.W.; Simmons, K.J. Computational methods to identify new antibacterial targets. Chem. Biol. Drug Des. 2015, 85, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Bernal, F.A.; Coy-Barrera, E. Molecular docking and multivariate analysis of xanthones as antimicrobial and antiviral agents. Molecules 2015, 20, 13165–13204. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, H.; Najafi, A.; Eslami, H.; Negahdari, B.; Moghaddam, M.M. Identification of novel bacterial DNA gyrase inhibitors: An in silico study. Res. Pharm. Sci. 2016, 11, 250–258. [Google Scholar] [PubMed]
- Ferguson, P.P.; Holloway, W.B.; Setzer, W.N.; McFeeters, H.; McFeeters, R.L. Small molecule docking supports broad and narrow spectrum potential for the inhibition of the novel antibiotic target bacterial Pth1. Antibiotics 2016. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Nazir, M.; Ali, M.S.; Hussain, H.; Lee, Y.S.; Riaz, N.; Jabbar, A. Antimicrobial natural products: An update on future antibiotic drug candidates. Nat. Prod. Rep. 2010, 27, 238–254. [Google Scholar] [CrossRef] [PubMed]
- Savoia, D. Plant-derived antimicrobial compounds: Alternatives to antibiotics. Future Microbiol. 2012, 7, 979–990. [Google Scholar] [CrossRef] [PubMed]
- Dictionary of Natural Products on DVD v.23:1; CRC Press: Boca Raton, FL, USA, 2014.
- Adams, J.M.; Capecchi, M.R. N-formylmethionyl-sRNA as the initiator of protein synthesis. Proc. Natl. Acad. Sci. USA 1966, 55, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Lucchini, G.; Bianchetti, R. Initiation of protein synthesis in isolated mitochondria and chloroplasts. Biochim. Biophys. Acta 1980, 608, 54–61. [Google Scholar] [CrossRef]
- Meinnel, T.; Mechulam, Y.; Blanquet, S. Methionine as translation start signal: A review of the enzymes of the pathway in Escherichia coli. Biochimie 1993, 75, 1061–1075. [Google Scholar] [CrossRef]
- Apfel, C.M.; Evers, S.; Hubschwerlen, C.; Pirson, W.; Page, M.G.; Keck, W. Peptide deformylase as an antibacterial drug target: Assays for detection of its inhibition in Escherichia coli cell homogenates and intact cells. Antimicrob. Agents Chemother. 2001, 45, 1053–1057. [Google Scholar] [CrossRef] [PubMed]
- Hackbarth, C.J.; Chen, D.Z.; Lewis, J.G.; Clark, K.; Mangold, J.B.; Cramer, J.A.; Margolis, P.S.; Wang, W.; Koehn, J.; Wu, C.; et al. N-Alkyl urea hydroxamic acids as a new class of peptide deformylase inhibitors with antibacterial activity. Antimicrob. Agents Chemother. 2002, 46, 2752–2764. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, B.J.; Pucci, M.J. Recent advances in bacterial topoisomerase inhibitors. Curr. Opin. Pharmacol. 2008, 8, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Pommier, Y.; Leo, E.; Zhang, H.; Marchand, C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem. Biol. 2010, 17, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Carlson, E.E.; May, J.F.; Kiessling, L.L. Chemical probes of UDP-galactopyranose mutase. Chem. Biol. 2006, 13, 825–837. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Madhurantakam, C.; Rajakumara, E.; Mazumdar, P.A.; Saha, B.; Mitra, D.; Wiker, G.H.; Sankaranarayanan, R.; Das, A.K. Crystal structure of low-molecular-weight protein tyrosine phosphatase from Mycobacterium tuberculosis at 1.9-Å resolution. J. Bacteriol. 2005, 187, 2175–2181. [Google Scholar] [CrossRef] [PubMed]
- Grundner, C.; Perrin, D.; van Huijsduijnen, R.H.; Swinnen, D.; Gonzalez, J.; Gee, C.L.; Wells, T.N.; Alber, T. Structural basis for selective inhibition of Mycobacterium tuberculosis protein tyrosine phosphatase PtpB. Structure 2007, 15, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Seward, H.E.; Roujeinikova, A.; McLean, K.J.; Munro, A.W.; Leys, D. Crystal structure of the Mycobacterium tuberculosis P450 CYP-121-fluconazole complex reveals new azol drug-P450 binding mode. J. Biol. Chem. 2006, 281, 39437–39443. [Google Scholar] [CrossRef] [PubMed]
- Belin, P.; le Du, M.H.; Fielding, A.; Lequin, O.; Jacquet, M.; Charbonnier, J.B.; Lecoq, A.; Thal, R.; Courçon, M.; Masson, C.; et al. Identification and structural basis of the reaction catalyzed by CYP121, an essential cytochrome P450 in Mycobacterium tuberculosis. Proc. Natl. Acad. Sci. USA 2009, 106, 7426–7431. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, M.E.; Coyne, A.G.; McLean, K.J.; James, G.G.; Levy, C.W.; Marino, L.B.; de Carvalho, L.P.; Chan, D.S.; Hudson, S.A.; Surade, S.; et al. Fragment-based approaches to the development of Mycobacterium tuberculosis CYP121 inhibitors. J. Med. Chem. 2016, 59, 3272–3302. [Google Scholar] [CrossRef] [PubMed]
- Mills, S.D.; Eakin, A.E.; Buurman, E.T.; Newman, J.V.; Gao, N.; Huynh, H.; Johnson, K.D.; Lahiri, S.; Shapiro, A.B.; Walkup, G.K.; et al. Novel bacterial NAD+-dependent DNA ligase inhibitors with broad-spectrum activity and antibacterial efficacy in vivo. Antimicrob. Agents Chemother. 2011, 55, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Stokes, S.S.; Huynh, H.; Gowravaram, M.; Albert, R.; Cavero-Tomas, M.; Chen, B.; Harang, J.; Loch, J.T.; Lu, M.; Mullen, G.B.; et al. Discovery of bacterial NAD+-dependent DNA ligase inhibitors: Optimization of antibacterial activity. Bioorg. Med. Chem. 2011, 21, 4556–4560. [Google Scholar] [CrossRef] [PubMed]
- Nandakumar, J.; Nair, P.A.; Shuman, S. Last stop on the road to repair: Structure of E. coli DNA ligase bound to nicked DNA-adenylate. Mol. Cell 2007, 26, 257–271. [Google Scholar] [CrossRef] [PubMed]
- Park, J.K.; Kim, K.H.; Moon, J.H.; Kim, E.E. Characterization of peptide deformylase 2 from B. cereus. J. Biochem. Mol. Biol. 2007, 6, 1050–1057. [Google Scholar] [CrossRef]
- Clements, J.M.; Beckett, R.P.; Brown, A.; Catlin, G.; Lobell, M.; Palan, S.; Thomas, W.; Whittaker, M.; Wood, S.; Salama, S.; et al. Antibiotic activity and characterization of BB-3497, a novel peptide deformylase inhibitor. Antimicrob. Agents Chemother. 2001, 45, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Guilloteau, J.P.; Mathieu, M.; Giglione, C.; Blanc, V.; Dupuy, A.; Chevrier, M.; Gil, P.; Famechon, A.; Meinnel, T.; Mikol, V. The crystal structures of four peptide deformylases bound to the antibiotic actinonin reveal two distinct types: A platform for the structure-based design of antibacterial agents. J. Mol. Biol. 2002, 320, 951–962. [Google Scholar] [CrossRef]
- Smith, K.J.; Petit, C.M.; Aubart, K.; Smyth, M.; McManus, E.; Jones, J.; Fosberry, A.; Lewis, C.; Lonetto, M.; Christensen, S.B. Structural variation and inhibitor binding in polypeptide deformylase from four different bacterial species. Protein Sci. 2003, 12, 349–360. [Google Scholar] [CrossRef] [PubMed]
- McElroy, C.A.; Amero, C.D.; Byerly, D.W.; Foster, M.P. Solution structure of peptide deformylase complexed with actinonin. 2009; to be published. [Google Scholar]
- Cheng, R.K.; Crawley, L.; Wood, M.; Barker, J.; Felicetti, B.; Whittaker, M. The structure of E. coli peptide deformylase (PDF) in complex with peptidomimetic ligand BB2827. 2009; to be published. [Google Scholar]
- Pichota, A.; Duraiswamy, J.; Yin, Z.; Keller, T.H.; Alam, J.; Liung, S.; Lee, G.; Ding, M.; Wang, G.; Chan, W.L.; et al. Peptide deformylase inhibitors of Mycobacterium tuberculosis: Synthesis, structural investigations, and biological results. Bioorg. Med. Chem. Lett. 2008, 18, 6568–6572. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.J.; Kim, H.L.; Lee, S.K.; Kim, H.W.; Kim, H.W.; Lee, J.Y.; Mikami, B.; Suh, S.W. Crystal structure of peptide deformylase from Staphylococcus aureus in complex with actinonin, a naturally occurring antibacterial agent. Proteins 2004, 57, 639–642. [Google Scholar] [CrossRef] [PubMed]
- Molteni, V.; He, X.; Nabakka, J.; Yang, K.; Kreusch, A.; Gordon, P.; Bursulaya, B.; Warner, I.; Shin, T.; Biorac, T.; et al. Identification of novel potent bicyclic peptide deformylase inhibitors. Bioorg. Med. Chem. Lett. 2004, 14, 1477–1481. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Lee, S.J.; Lee, S.K.; Yoon, H.J.; Lee, H.H.; Kim, K.K.; Lee, J.B.; Lee, B.I.; Suh, S.W. Structures of Staphylococcus aureus peptide deformylase in complex with two classes of new inhibitors. Acta Crystallogr. Sect. D 2012, 68, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Fieulaine, S.; Desmadril, M.; Meinnel, T.; Giglione, C. Understanding the highly efficient catalysis of prokaryotic peptide deformylases by shedding light on the determinants specifying the low activity of the human counterpart. Acta Crystallogr. Sect. D 2014, 70, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Holdgate, G.A.; Tunnicliffe, A.; Ward, W.H.; Weston, S.A.; Rosenbrock, G.; Barth, P.T.; Taylor, I.W.; Pauptit, R.A.; Timms, D. The entropic penalty of ordered water accounts for weaker binding of the antibiotic novobiocin to a resistant mutant of DNA gyrase: A thermodynamic and crystallographic study. Biochemistry 1997, 36, 9663–9673. [Google Scholar] [CrossRef] [PubMed]
- Bellon, S.; Parsons, J.D.; Wei, Y.; Hayakawa, K.; Swenson, L.L.; Charifson, P.S.; Lippke, J.A.; Aldape, R.; Gross, C.H. Crystal structures of Escherichia coli topoisomerase IV ParE subunit (24 and 43 kilodaltons): A single residue dictates differences in novobiocin potency against topoisomerase IV and DNA gyrase. Antimicrob. Agents Chemother. 2004, 48, 1856–1864. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Roué, M.; Spitzfaden, C.; Petrella, S.; Aubry, A.; Hann, M.; Bax, B.; Mayer, C. Mycobacterium tuberculosis DNA gyrase ATPase domain structures suggest a dissociative mechanism that explains how ATP hydrolysis is coupled to domain motion. Biochem. J. 2013, 456, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Evdokimov, A.G.; Pokross, M.; Walter, R.; Mekel, M.; Cox, B.; Li, C.; Bechard, R.; Genbauffe, F.; Andrews, R.; Diven, C.; et al. Engineering the catalytic domain of human protein tyrosine phosphatase β for structure-based drug discovery. Acta Crystallogr. Sect. D 2006, 62, 1435–1445. [Google Scholar] [CrossRef] [PubMed]
- Van Straaten, K.E.; Kuttiyatveetil, J.R.; Sevrain, C.M.; Villaume, S.A.; Jiménez-Barbero, J.; Linclau, B.; Vincent, S.P.; Sanders, D.A. Structural basis of ligand binding to UDP-galactose mutase from Mycobacterium tuberculosis using substrate and tetrafluorinated substrate analogs. J. Am. Chem. Soc. 2015, 137, 1230–1244. [Google Scholar] [CrossRef] [PubMed]
- Fonvielle, M.; le Du, M.H.; Lequin, O.; Lecoq, A.; Jacquet, M.; Thai, R.; Dubois, S.; Grach, G.; Gondry, M.; Belin, P. Substrate and reaction specificity of Mycobacterium tuberculosis cytochrome P450 CYP121. Insights from biochemical studies and crystal structures. J. Biol. Chem. 2013, 288, 17347–17359. [Google Scholar] [CrossRef] [PubMed]
- Hudson, S.A.; Surade, S.; Coyne, A.G.; McLean, K.J.; Leys, D.; Munro, A.W.; Abell, C. Overcoming the limitations of fragment merging: Rescuing a strained merged fragment series targeting Mycobacterium tuberculosis CYP 121. ChemMedChem 2013, 8, 1451–1456. [Google Scholar] [CrossRef] [PubMed]
- Surivet, J.P.; Lange, R.; Hubschwerlen, C.; Keck, W.; Specklin, J.L.; Ritz, D.; Bur, D.; Locher, H.; Seiler, P.; Strasser, D.S.; et al. Structure-guided design, synthesis and biological evaluation of novel DNA ligase inhibitors with in vitro and in vivo anti-staphylococcal activity. Bioorg. Med. Chem. Lett. 2012, 22, 6705–6711. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.K.; Tripathi, R.P.; Ramachandran, R. NAD+-dependent DNA ligase (Rv3014c) from Mycobacterium tuberculosis. Crystal structure of the adenylation domain and identification of novel inhibitors. J. Biol. Chem. 2005, 280, 30273–30281. [Google Scholar] [CrossRef] [PubMed]
- Howard, S.; Amin, N.; Benowitz, A.B.; Chiarparin, E.; Cui, H.; Deng, X.; Heightman, T.D.; Holmes, D.J.; Hopkins, A.; Huang, J.; et al. Fragment-based discovery of 6-azaindoles as inhibitors of bacterial DNA ligase. ACS Med. Chem. Lett. 2013, 4, 1208–1212. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, R.; Christensen, M.H. MolDock: A new technique for high-accuracy molecular docking. J. Med. Chem. 2006, 49, 3315–3321. [Google Scholar] [CrossRef] [PubMed]
- Halgren, T.A. Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF 94. J. Comput. Chem. 1996, 17, 490–519. [Google Scholar] [CrossRef]
- Molegro Virtual Docke, version 6.0.1; Molegro ApS: Aarhus, Denmark, 2013.
- Pan, Y.; Huang, N.; Cho, S.; MacKerell, A.D. Consideration of molecular weight during compound selection in virtual target-based database screening. J. Chem. Inf. Comput. Sci. 2003, 43, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.M.; Shen, T.W. A pharmacophore-based evolutionary approach for screening selective estrogen receptor modulators. Proteins Struct. Funct. Bioinform. 2005, 59, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Nagarsekar, A.; Xia, G.; Hayashi, J.; MacKerell, A.D. Identification of non-phosphate-containing small molecular weight inhibitors of the tyrosine kinase p56 Lck SH2 domain via in silico screening against the pY + 3 binding site. J. Med. Chem. 2004, 47, 3502–3511. [Google Scholar] [CrossRef] [PubMed]
- Hancock, C.N.; Macias, A.; Lee, E.K.; Yu, S.Y.; MacKerell, A.D.; Shapiro, P. Identification of novel extracellular signal-regulated kinase docking domain inhibitors. J. Med. Chem. 2005, 48, 4586–4595. [Google Scholar] [CrossRef] [PubMed]
- Abad-Zapatero, C.; Metz, J.T. Ligand efficiency indices as guideposts for drug discovery. Drug Discov. Today 2005, 10, 464–469. [Google Scholar] [CrossRef]
- Carta, G.; Knox, A.J.; Lloyd, D.G. Unbiasing scoring functions: A new normalization and rescoring strategy. J. Chem. Inf. Model. 2007, 47, 1564–1571. [Google Scholar] [CrossRef] [PubMed]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
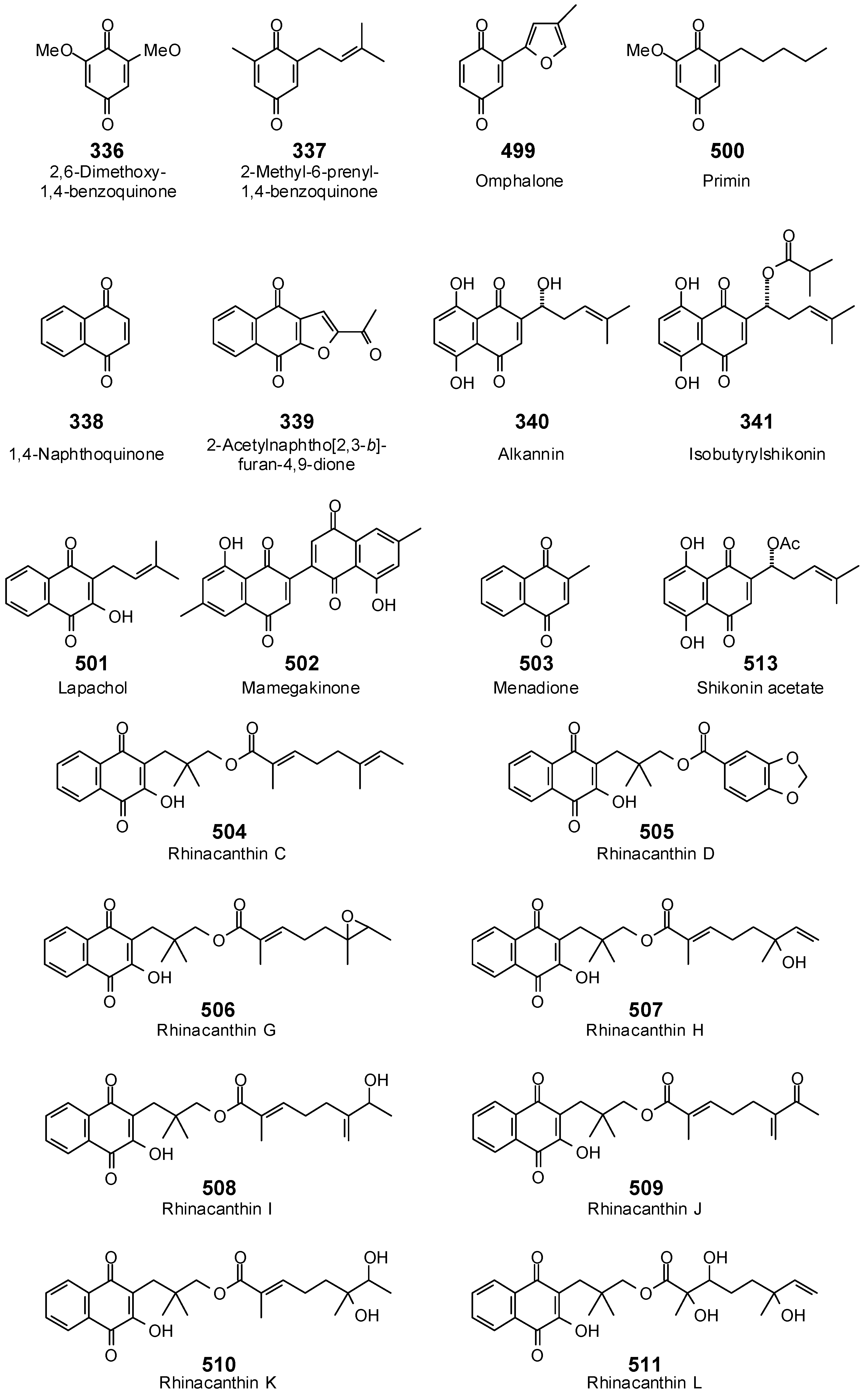
- Sheriha, G.M.; Abouamer, K.; Elshtaiwi, B.Z. An alkaloid from Haplophyllum tuberculatum. Phytochemistry 1985, 24, 884–886. [Google Scholar] [CrossRef]
- Gnan, S.O.; Sheriha, G.M. Antimicrobial activity of (+)-tuberine. J. Food Prot. 1986, 49, 340–341. [Google Scholar]
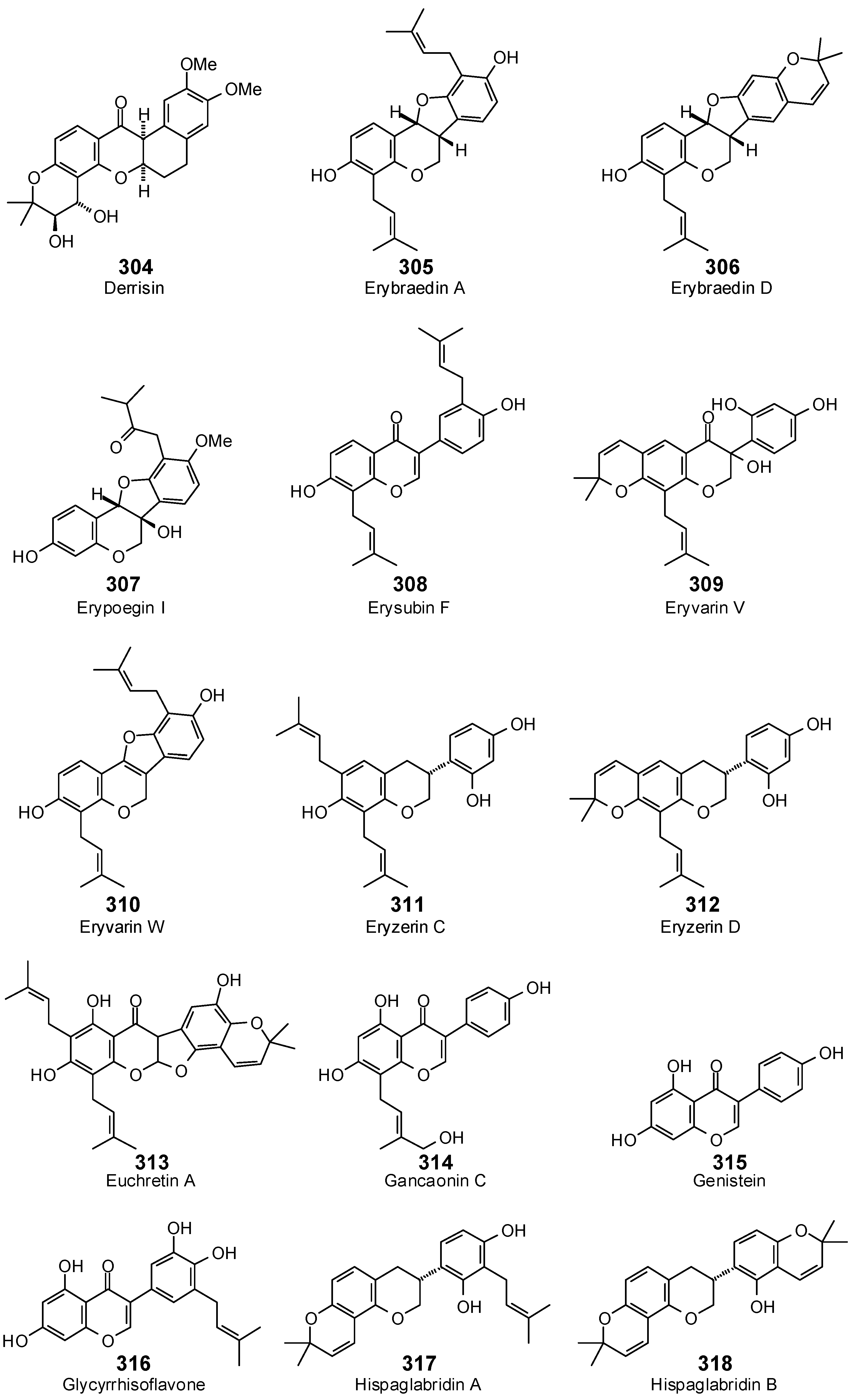
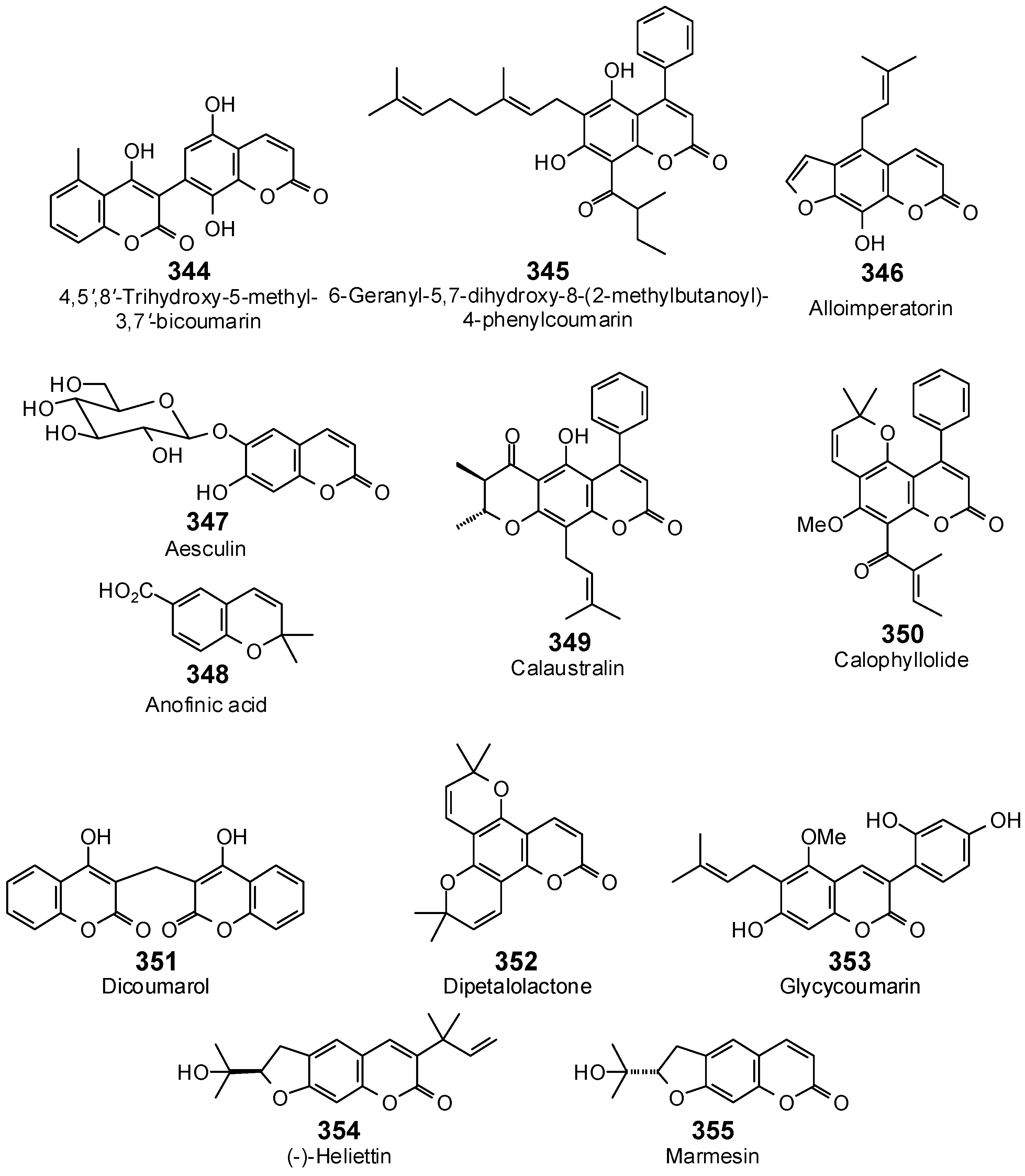
- Kuete, V.; Alibert-Franco, S.; Eyong, K.O.; Ngameni, B.; Folefoc, G.N.; Nguemeving, J.R.; Tangmouo, J.G.; Fotso, G.W.; Komguem, J.; Ouahouo, B.M.; et al. Antibacterial activity of some natural products expressing a multidrug-resistant phenotype. Int. J. Antimicrob. Agents 2011, 37, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, S.; Legault, J.; Simard, F.; Chiasson, E.; Pickette, A. New antibacterial dihydrochalcone derivatives from buds of Populus balsamifera. Tetrahedron Lett. 2013, 54, 1631–1633. [Google Scholar] [CrossRef]
- Mbaveng, A.T.; Ngameni, B.; Kuete, V.; Simo, I.K.; Ambassa, P.; Roy, R.; Bezabih, M.; Etoa, F.X.; Ngadjui, B.T.; Abegaz, B.M.; et al. Antimicrobial activity of the crude extracts and five flavonoids from the twigs of Dorstenia barteri (Moraceae). J. Ethnopharmacol. 2008, 116, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Orjala, J.; Wright, A.D.; Behrends, H.; Folkers, G.; Sticher, O.; Rüegger, H. Cytotoxic and antibacterial dihydrochalcones from Piper aduncum. J. Nat. Prod. 1994, 57, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Inamori, Y.; Baba, K.; Tsujibo, H.; Taniguchi, M.; Nakata, K.; Kozawa, M. Antibacterial activity of two chalcones, xanthoangelol and 4-hydroxyderricin, isolated from the root of Angelica keiskei Koidzumi. Chem. Pharm. Bull. 1991, 39, 1604–1605. [Google Scholar] [CrossRef] [PubMed]
- Sugamoto, K.; Matsusita, Y.; Matsui, K.; Kurogi, C.; Matsui, T. Synthesis and antibacterial activity of chalcones bearing prenyl or geranyl groups from Angelica keiskei. Tetrahedron 2011, 67, 5346–5359. [Google Scholar] [CrossRef]
- Šmejkal, K.; Chudik, S.; Klouček, P.; Marek, R.; Cvačka, J.; Urbanová, M.; Julínex, O.; Kokoška, L.; Šlapetová, T.; Holubová, P.; et al. Antibacterial C-geranylflavonoids from Poulownia tomentosa fruits. J. Nat. Prod. 2008, 71, 706–709. [Google Scholar] [CrossRef] [PubMed]
- Nanayakkara, N.P.; Burandt, C.L.; Jacob, M.R. Flavonoids with activity against methicillin-resistant Staphylococcus aureus from Dalea scandens var. paucifolia. Planta Med. 2002, 68, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Mitscher, L.A.; Gollapudi, S.R.; Khanna, I.K.; Drake, S.D.; Hanumaiah, T.; Ramaswamy, T.; Rao, K.V. Antimicrobial agents from higher plants: Activity and structural revision of flemiflavanone-D from Flemingia stricta. Phytochemistry 1985, 24, 2885–2997. [Google Scholar] [CrossRef]
- Schütz, B.A.; Wright, A.D.; Rali, T.; Sticher, O. Prenylated flavanones from leaves of Macaranga pleiostemona. Phytochemistry 1995, 40, 1273–1277. [Google Scholar] [CrossRef]
- Setzer, W.N.; Green, T.J.; Lawton, R.O.; Moriarity, D.M.; Bates, R.B.; Caldera, S.; Haber, W.A. An antibacterial vitamin E derivative from Tovomitopsis psychotriifolia. Planta Med. 1995, 61, 275–276. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.; Simmonds, M.S. Rosmarinic acid. Phytochemistry 2003, 62, 121–125. [Google Scholar] [CrossRef]
- Klančnik, A.; Piskernik, S.; Jeršek, B.; Možina, S. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. J. Microbiol. Methods 2010, 81, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Bais, H.P.; Walker, T.S.; Scheizer, H.P.; Vivanco, J.M. Root specific elicitation and antimicrobial activity of rosmarinic acid in hairy root cultures of Ocimum basilicum. Plant Physiol. Biochem. 2002, 40, 983–995. [Google Scholar] [CrossRef]
- Walker, T.S.; Bais, H.P.; Déziel, E.; Schwizer, H.P.; Rahme, L.G.; Fall, R.; Vivanco, J.M. Pseudomonas aeruginosa-plant root interactions. Pathogenicity, biofilm formation, and root exudation. Plant Physiol. 2004, 124, 320–331. [Google Scholar] [CrossRef] [PubMed]
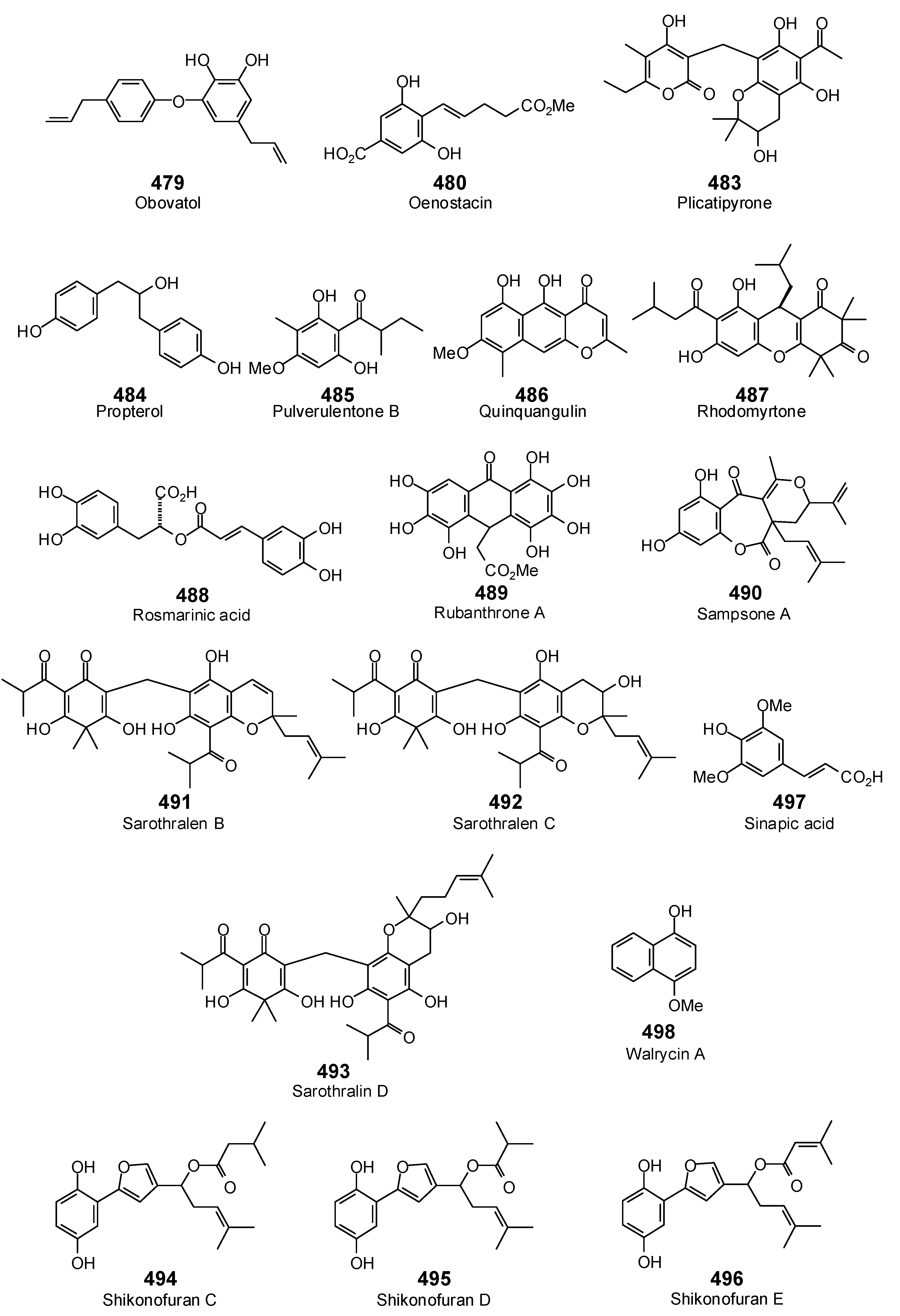
- Yoshizaki, F.; Hisamichi, S.; Kondo, Y.; Sato, Y.; Nozoe, S. Studies on shikon. III. New furylhydroquinone derivatives, shikonofurans A, B, C, D and E, from Lithospermum erythrorhizon Sieb. et Zucc. Chem. Pharm. Bull. 1982, 30, 4407–4411. [Google Scholar] [CrossRef] [Green Version]
- Puttarak, P.; Charoonratana, T.; Panichayupakaranant, P. Antimicrobial activity and stability of rhinacanthins-rich Rhinacanthus nasutus extract. Phytomedicine 2010, 17, 323–327. [Google Scholar] [CrossRef] [PubMed]
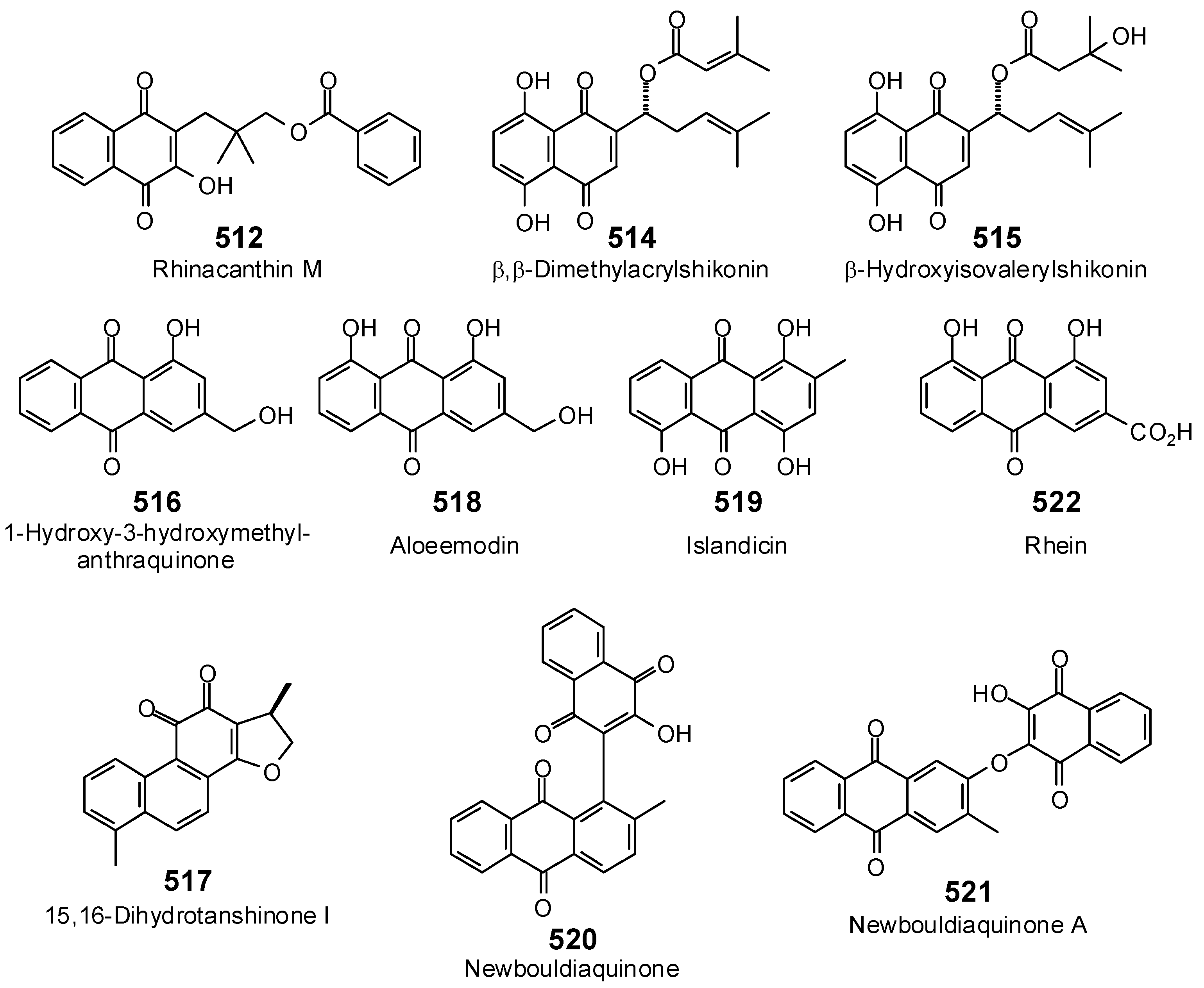
- Petit, S.; Duroc, Y.; Larue, V.; Giglione, C.; Léon, C.; Soulama, C.; Denis, A.; Dardel, F.; Meinnel, T.; Artaud, I. Structure-activity relationship analysis of the peptide deformylase inhibitor 5-bromo-1H-indole-3-acetohydroxamic acid. ChemMedChem 2009, 4, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, P.; Yu, H.; Li, J.; Wang, M.W.; Zhao, W. Anti-Helicobacter pylori and thrombin inhibitory components from Chinese dragon’s blood, Dracaena cochinchinensis. J. Nat. Prod. 2007, 70, 1570–1577. [Google Scholar] [CrossRef] [PubMed]
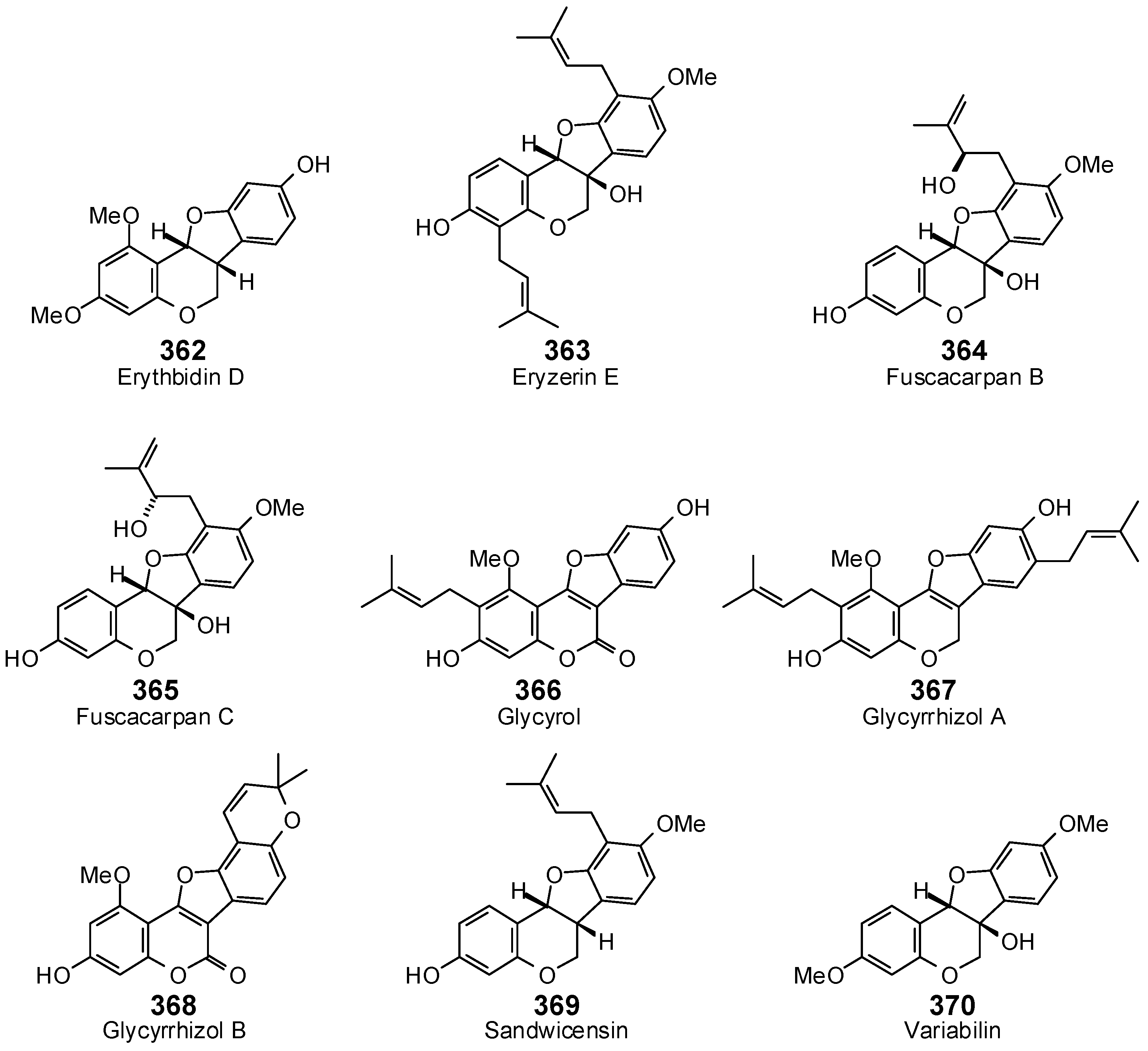
- Fukai, T.; Kaitou, K.; Terada, S. Antimicrobial activity of 2-arylbenzofurans from Morus species against methicillin-resistant Staphylococcus aureus. Fitoterapia 2005, 76, 708–711. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Hirata, M.; Etoh, H.; Sako, M.; Sato, M.; Murata, J.; Murata, H.; Darnaedi, D.; Fukai, T. Six new constituents from the roots of Erythrina variegata. Chem. Biodivers. 2004, 1, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Basri, D.F.; Xian, L.W.; Shukor, N.I.; Latip, J. Bacteriostatic antimicrobial combination: Antagonistic interaction between epsilon-viniferin and vancomycin against methicillin-resistant Staphylococcus aureus. BioMed Res. Int. 2014. [Google Scholar] [CrossRef] [PubMed]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K.; Sahu, B. Identification of natural compound inhibitors against peptide deformylase using virtual screening and molecular docking techniques. Bull. Environ. Pharmacol. Life Sci. 2015, 4, 70–80. [Google Scholar]
- Gradišar, H.; Pristovšek, P.; Plaper, A.; Jerala, R. Green tea catechins inhibit bacterial DNA gyrase by interaction with its ATP binding site. J. Med. Chem. 2007, 50, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Hossion, A.M.; Zamami, Y.; Kandahary, R.K.; Tsuchiya, T.; Ogawa, W.; Iwado, A.; Sasaki, K. Quercetin diacylglycoside analogues showing dual inhibition of DNA gyrase and topoisomerase IV as novel antibacterial agents. J. Med. Chem. 2011, 54, 3686–3703. [Google Scholar] [CrossRef] [PubMed]
- Plaper, A.; Golob, M.; Hafner, I.; Oblak, M.; Šolmajer, T.; Jerala, R. Characterization of quercetin binding site on GNA gyrase. Biochem. Biophys. Res. Commun. 2003, 306, 530–536. [Google Scholar] [CrossRef]
- Verotta, L.; Lovaglio, E.; Vidari, G.; Finzi, P.V.; Neri, M.G.; Raimondi, A.; Parapini, S.; Taramelli, D.; Riva, A.; Bombardelli, E. 4-Alkyl- and 4-phenylcoumarins from Mesua ferrea as promising multidrug resistant antibacterials. Phytochemistry 2004, 65, 2867–2879. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhou, B.; Zhang, S.; Wu, L.; Wang, Y.; Franzblau, S.G.; Zhang, Z.Y. Identification and characterization of novel inhibitors of mPTPB, an essential virulent phosphatase from Mycobacterium tuberculosis. ACS Med. Chem. Lett. 2010, 1, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Zang, X.; He, M.; Pan, S.; Xu, X. Structure-activity relationship of flavonoids on their anti-Escherichia coli activity and inhibition of DNA gyrase. J. Agric. Food Chem. 2013, 61, 8185–8190. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, S.; Zandberg, W.F.; Mohan, S.; Ko, M.; Martinez-Gutierrez, F.; Partha, S.K.; Sanders, D.A.; Av-Gay, Y.; Pinto, B.M. Antimycrobacterial activity of UDP-galactopyranose mutase inhibitors. Int. J. Antimicrob. Agents 2010, 36, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Dykhuizen, E.C.; May, J.F.; Tongpenyai, A.; Kiessling, L.L. Inhibitors of UDP-galactopyranose mutase thwart mycobacterial growth. J. Am. Chem. Soc. 2008, 130, 6706–6707. [Google Scholar] [CrossRef] [PubMed]
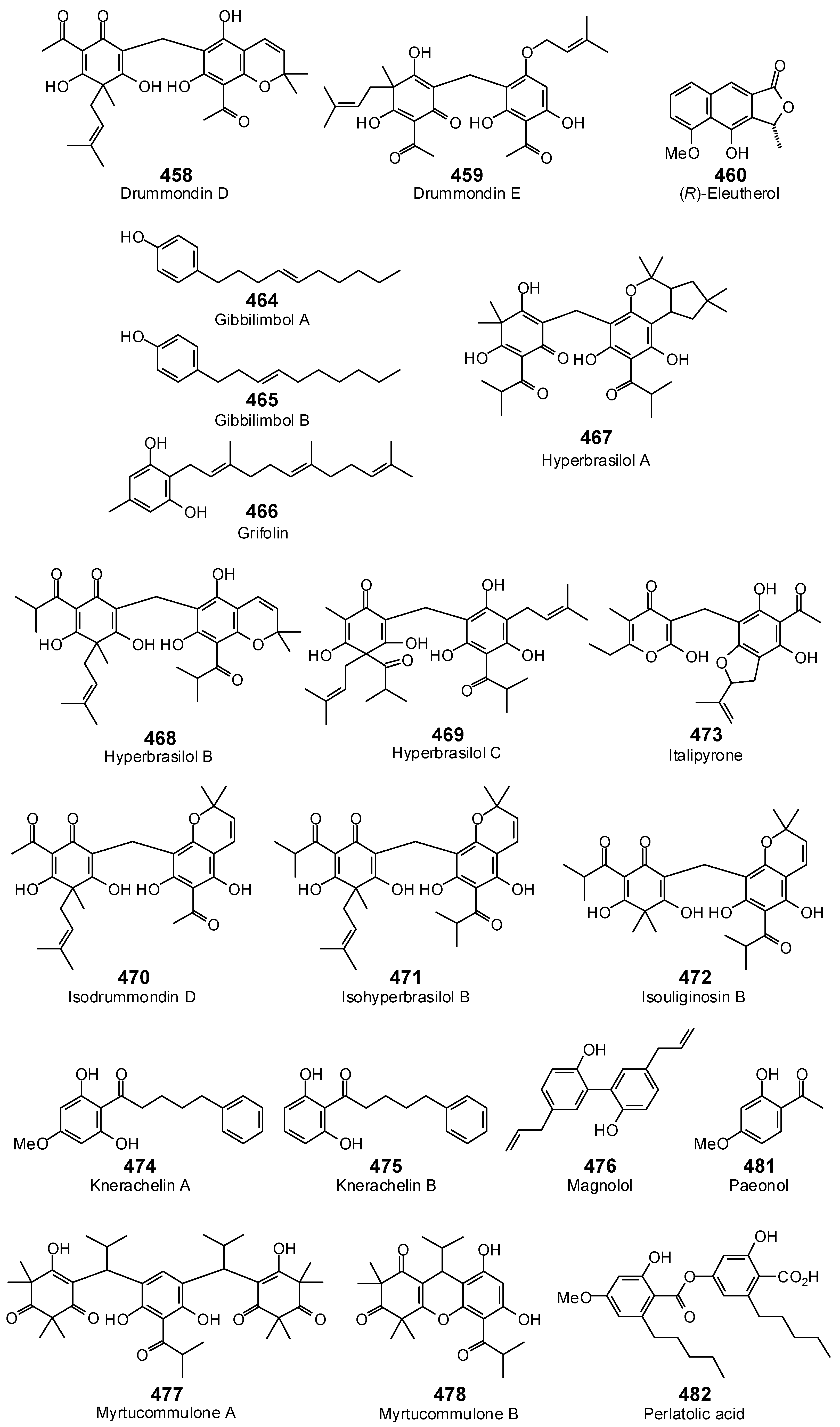
- Jayasuriya, H.; Clark, A.M.; McChesney, J.D. New antimicrobial filicinic acid derivatives from Hypericum drummondii. J. Nat. Prod. 1991, 54, 1314–1320. [Google Scholar] [CrossRef] [PubMed]
- Rocha, L.; Marston, A.; Potterat, O.; Kaplan, M.A.; Hostettmann, K. More phloroglucinols from Hypericum brasiliense. Phytochemistry 1996, 42, 185–188. [Google Scholar] [CrossRef]
- Wang, T.; Duncan, L.; Gu, W.; O’Dowd, H.; Wei, Y.; Perola, E.; Parsons, J.; Gross, C.H.; Moody, C.S.; Arends, S.J.R.; et al. Design, synthesis and biological evaluation of potent NAD+-dependent DNA ligase inhibitors as potential antibacterial agents. Part II: 4-Amino-pyrido[2,3-d]pyrimidin-5(8H)-ones. Bioorg. Med. Chem. Lett. 2012, 22, 3699–3703. [Google Scholar] [CrossRef] [PubMed]
- Siridechakorn, I.; Phakhodee, W.; Ritthiwigrom, T.; Promgool, T.; Deachathai, S.; Cheenpracha, S.; Prawat, U.; Laphookhieo, S. Antibacterial dihydrobenzopyran and xanthone derivatives from Garcinia cowa stem barks. Fitoterapia 2012, 83, 1430–1434. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.Y.; Son, K.H.; Kwon, C.S.; Kwon, G.S.; Kang, S.S. Antimicrobial and cytotoxic activity of 18 prenylated flavonoids isolated from medicinal plants: Morus alba L., Morus mongolica Schneider, Broussnetia papyrifera (L.) Vent, Sophora flavescens Ait and Echinosophora koreensis Nakai. Phytomedicine 2004, 11, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.C.; Yu, H.; Wong, C.W.; Lui, S.L.; Jolivalt, C.; Ganem-Elbaz, C.; Paris, J.M.; Morleo, B.; Litaudon, M.; Lau, C.B.; et al. Quick identification of kuraridin, a noncytotoxic anti-MRSA (methicillin-resistant Staphylococcus aureus) agent from Sophora flavescens using high-speed counter-current chromatography. J. Chromatogr. B 2012, 880, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Oh, I.; Yang, W.Y.; Chung, S.C.; Kim, T.Y.; Oh, K.B.; Shin, J. In vitro sortase A inhibitory and antimicrobial activity of flavonoids isolated from the roots of Sophora flavescens. Arch. Pharm. Res. 2011, 34, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, M.J.; King, A.B.; Graham, A.C.; Onishi, H.R.; Bartizal, K.F.; Abruzzo, G.K.; Gill, C.J.; Ramjit, H.G.; Pitzenberger, S.M.; Witherup, K.M. Antibacterial activity of lonchocarpol A. J. Nat. Prod. 1998, 61, 640–642. [Google Scholar] [CrossRef]
- Valerio, L.G. In silico toxicology for the pharmaceutical sciences. Toxocol. Appl. Pharmacol. 2009, 241, 356–370. [Google Scholar] [CrossRef] [PubMed]
- Yuriev, E.; Agostino, M.; Ramsland, P.A. Challenges and advances in computational docking: 2009 In review. J. Mol. Recognit. 2011, 24, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Yuriev, E.; Ramsland, P.A. Latest developments in molecular docking: 2010–2011 In review. J. Mol. Recognit. 2013, 26, 215–239. [Google Scholar] [CrossRef] [PubMed]




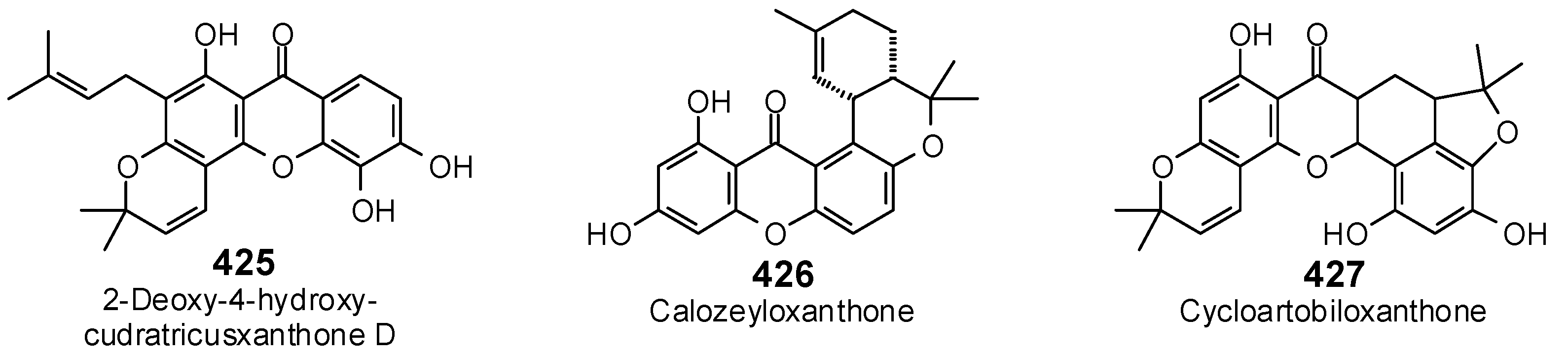
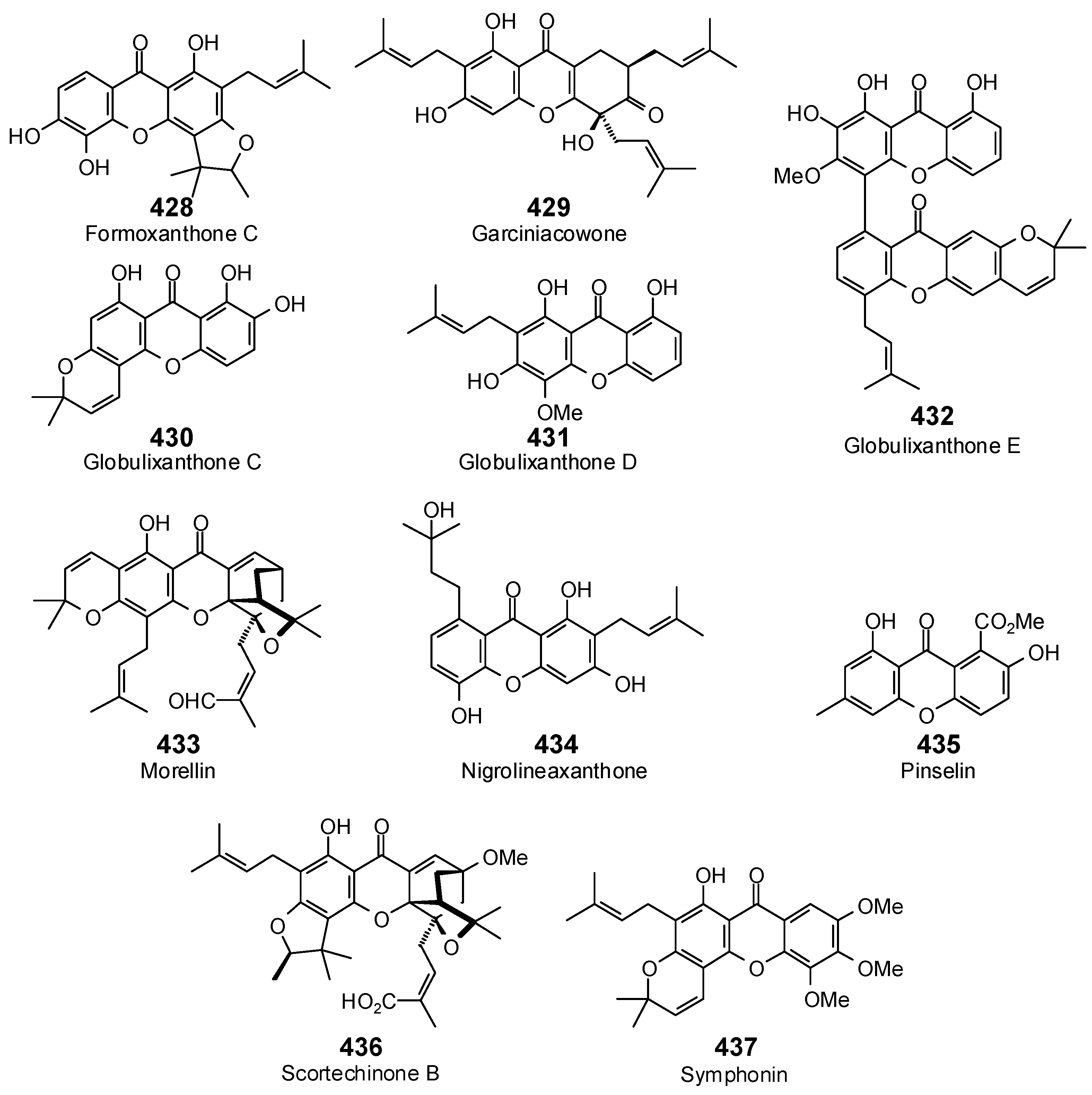
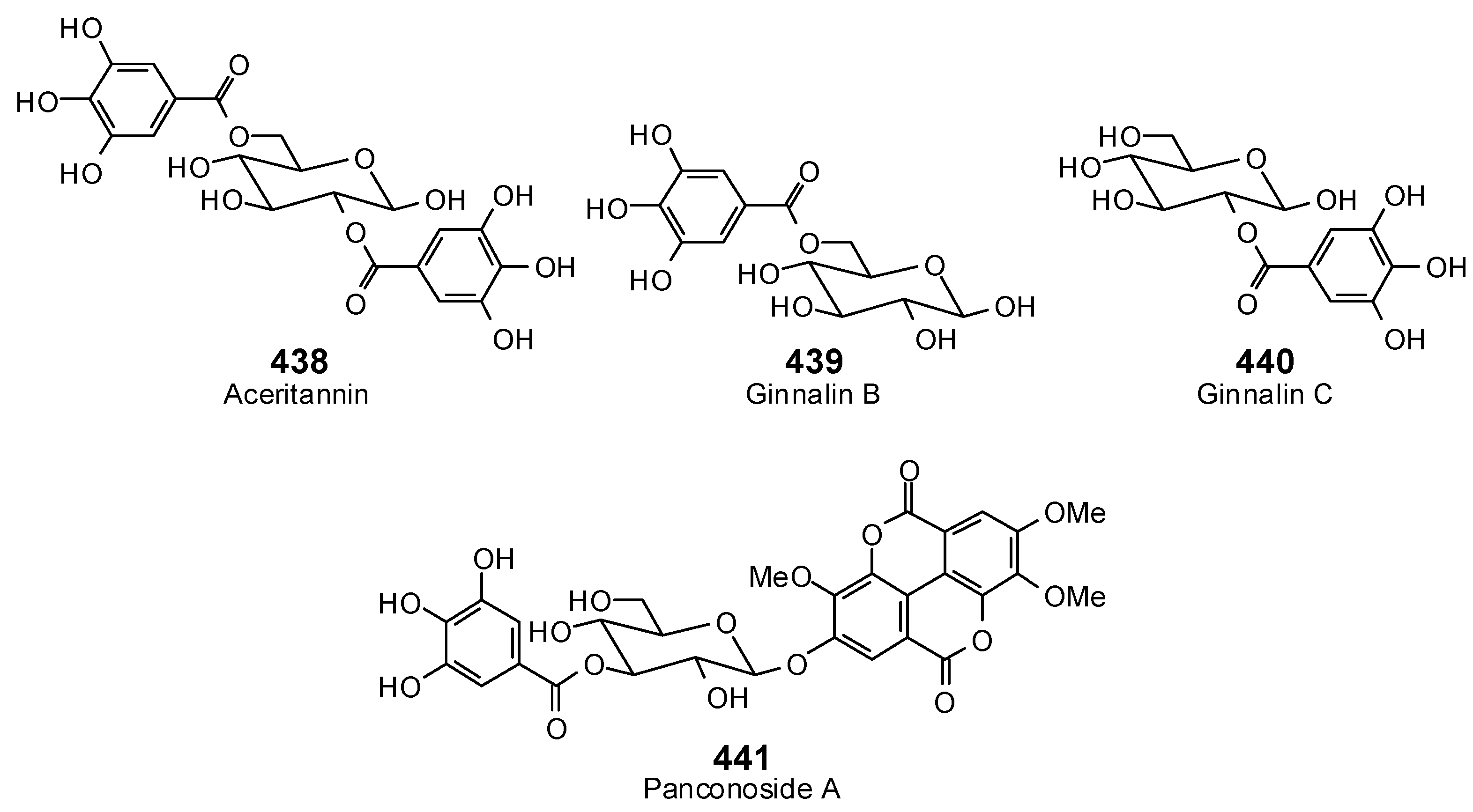
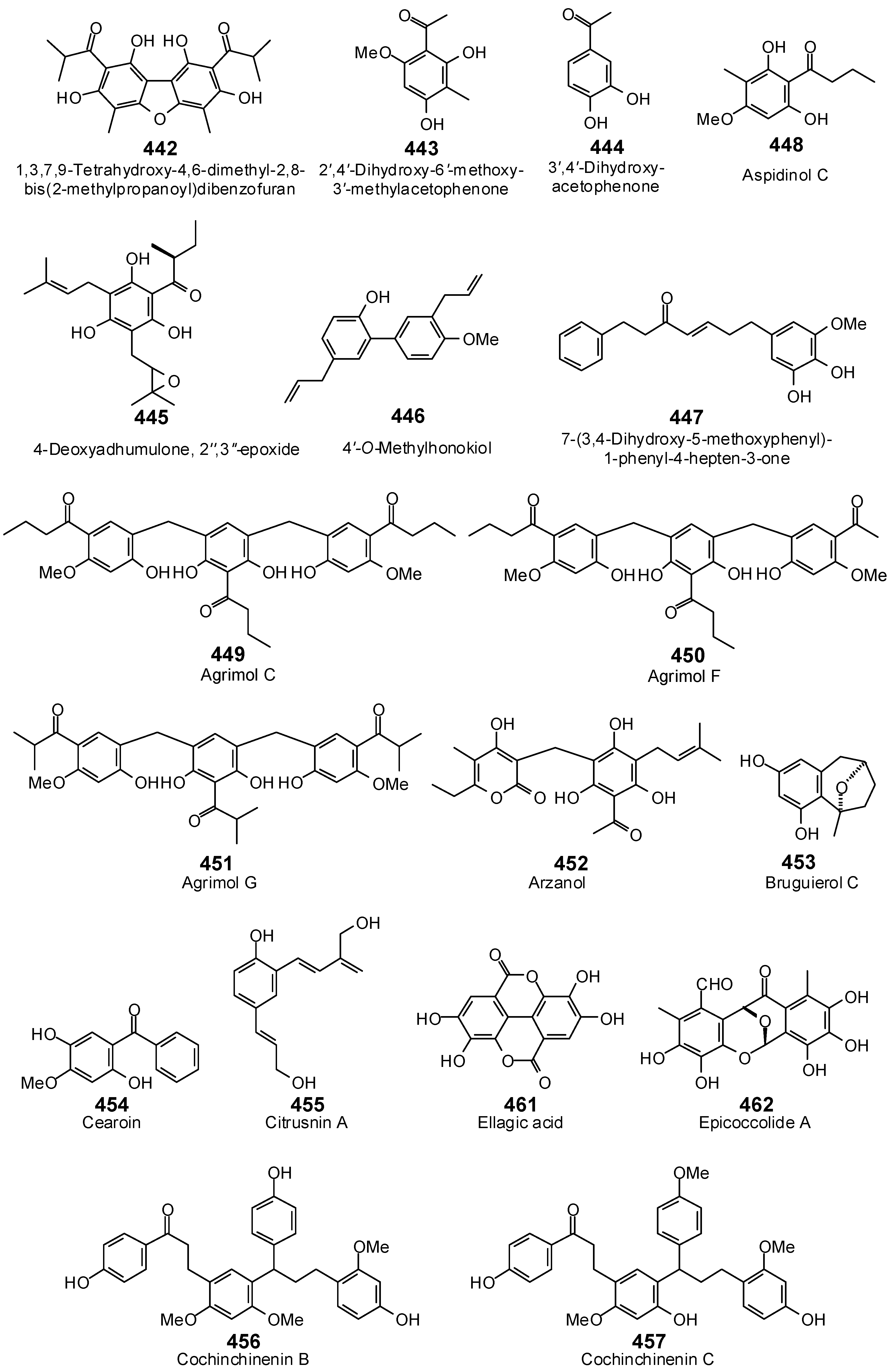

| Protein Target | PDB Code | Co-Crystallized Ligand | E (kJ/mol) | RMSD (Å) |
|---|---|---|---|---|
| BcPDF | 2OKL | Actinonin | −114.6 | 0.82 |
| EcPDF | 1G2A | Actinonin | −111.8 | 1.08 |
| 1G27 | 2-[(Formyl-hydroxy-amino)-methyl]-hexanoic acid (1-dimethylcarbamoyl-2,2-dimethyl-propyl)-amide (BB-3497) | −90.2 | 0.49 | |
| 1LRU | Actinonin | −110.0 | 1.23 | |
| 2AI8 | [Hydroxy(3-phenylpropyl)amino]methanol (SB-485345) | −56.1 | 0.71 | |
| 2KMN | Actinonin | −46.6 | 0.84 | |
| 3K6L | (2S,3R)-N4-[(1S)-1-(Dimethylcarbamoyl)-2,2-dimethylpropyl]-N1,2-dihydroxy-3-(2-methylpropyl)butanediamide (BB-2827) | −96.2 | 0.79 | |
| McPDF | 3E3U | N-[(2R)-2-{[(2S)-2-(1,3-benzoxazol-2-yl)pyrrolidin-1-yl]carbonyl}-hexyl]-N-hydroxyformamide | −99.9 | 0.55 |
| PaPDF | 1LRY | Actinonin | −97.6 | 0.71 |
| 1IX1 | Actinonin | −127.2 | 0.81 | |
| 1S17 | 2-(3,4-Dihydro-3-oxo-2H-benzo[B][1,4]thiazin-2-yl)-N-hydroxyacetamide | −92.6 | 1.10 | |
| SaPDF | 1Q1Y | Actinonin | −98.4 | 2.18 |
| 3U7K | (S)-N-(Cyclopentylmethyl)-N-(2-(hydroxyamino)-2-oxoethyl)-2-[3-(2-methoxyphenyl)ureido]-3,3-dimethylbutanamide | −120.5 | 3.12 | |
| 3U7L | (S)-N-(Cyclopentylmethyl)-2-[3-(3,5-difluorophenyl)ureido]-N-(2-(hydroxyamino)-2-oxoethyl)-3,3-dimethylbutanamide | −111.9 | 3.25 | |
| 3U7M | N-[(2R,4S)-2-butyl-4-(3-(2-fluorophenyl)ureido)-5-methyl-3-oxohexyl]-N-hydroxyformamide | −93.9 | 4.94 | |
| 3U7N | N-[(2R,4S)-2-butyl-5-methyl-4-(3-(5-methylpyridin-2-yl)ureido)-3-oxohexyl]-N-hydroxyformamide | −82.5 | 5.00 | |
| SpPDF | 2AI7 | [Hydroxy(3-phenylpropyl)amino]methanol (SB-485345) | −58.8 | 1.07 |
| 2AIA | 2-(3-Benzoylphenoxy)ethyl(hydroxyl)formamide (SB-543668) | −113.2 | 5.83 | |
| 2AIE | Hydroxy[3-(6-methylpyridin-2-yl)propyl]formamide (SB-505684) | −73.1 | 1.06 | |
| HsPDF | 4JE7 | Actinonin | −105.0 | 0.71 |
| 4JE8 | Met-Ala-Ser | −78.3 | 0.85 | |
| EcTopoIV | 1S16 | Phosphoaminophosphonic acid-adenylate ester (ADPNP) | −175.9 | 0.59 |
| EcGyrB | 1AJ6 | Novobiocin | −74.2 | 5.55 |
| MtGyrB | 3ZKB | Phosphoaminophosphonic acid-adenylate ester (ADPNP) | −158.4 | 1.04 |
| 3ZKD | Phosphoaminophosphonic acid-adenylate ester (ADPNP) | −176.2 | 0.81 | |
| MtPtp | 1U2Q | Glycerol | −29.9 | 4.43 |
| 2OZ5 | {(3-Chlorobenzyl)[(5-{[(3,3-diphenylpropyl)aminosulfonyl}-2-thienyl)methyl]-amino}(oxo)acetic acid | −148.3 | 4.92 | |
| HsPtp | 1I4H | N-(t-Butoxycarbonyl)-l-tyrosyl-N-methyl-4-(sulfoamino)-l-phenylalaninamide | −121.2 | 4.81 |
| 2I5X | (4-Ethylphenyl)sulfamic acid | −80.4 | 0.82 | |
| MtUGM | 4RPG | UDP-d-Galactopyranose | −148.3 | 0.94 |
| 4RPH | UDP-d-Galactopyranose | −162.1 | 0.34 | |
| 4RPJ | UDP | −132.0 | 0.68 | |
| 4RPK | (2R,5S)-5-[(1R)-1,2-Dihydroxyethyl]-3,3,4,4-tetrafluorotetrahydrofuran-2-yl[(2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl dihydrogen diphosphate | −154.6 | 1.89 | |
| 4RPL | [(2R,3S,4R,5R)-5-(2,4-Dioxo-3,4-dihydropyrimidin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl(2R,5S,6R)-3,3,4,4-tetrafluoro-5-hydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl dihydrogen diphosphate | −124.9 | 1.67 | |
| MtCYP121 | 3G5H | (3S,6S)-3,6-Bis(4-hydroxybenzyl)piperazine-2,5-dioneCyclo(Tyr-Tyr) | −77.6 | 4.18 |
| 4G44 | 3-(1H-1,2,4-Triazol-1-ylmethyl)aniline | −55.3 | 0.49 | |
| 4IPS | (3S,6S)-3,6-Bis(4-hydroxybenzyl)piperazin-2-one | −63.4 | 5.44 | |
| 4KTF | 4,4′-(3-Amino-1H-pyrazole-4,5-diyl)diphenol | −80.4 | 8.19 | |
| 5IBE | 4-[5-Amino-4-(3′-amino[1,1′-biphenyl]-3-yl)-1H-pyrazol-3-yl]phenol | −94.2 | 2.17 | |
| EcLigA | 2OWO | none | - | - |
| 4GLX | 2-Amino-6-bromo-7-(trifluoromethyl)-1,8-naphthyridine-3-carboxamide | −90.6 | 0.69 | |
| MtLigA | 1ZAU | Adenosine monophosphate | −77.0 | 0.80 |
| SaLigA | 4CC5 | 2-Chloranyl-6-(1H-1,2,4-triazol-3-yl)pyrazine | −52.7 | 4.57 |
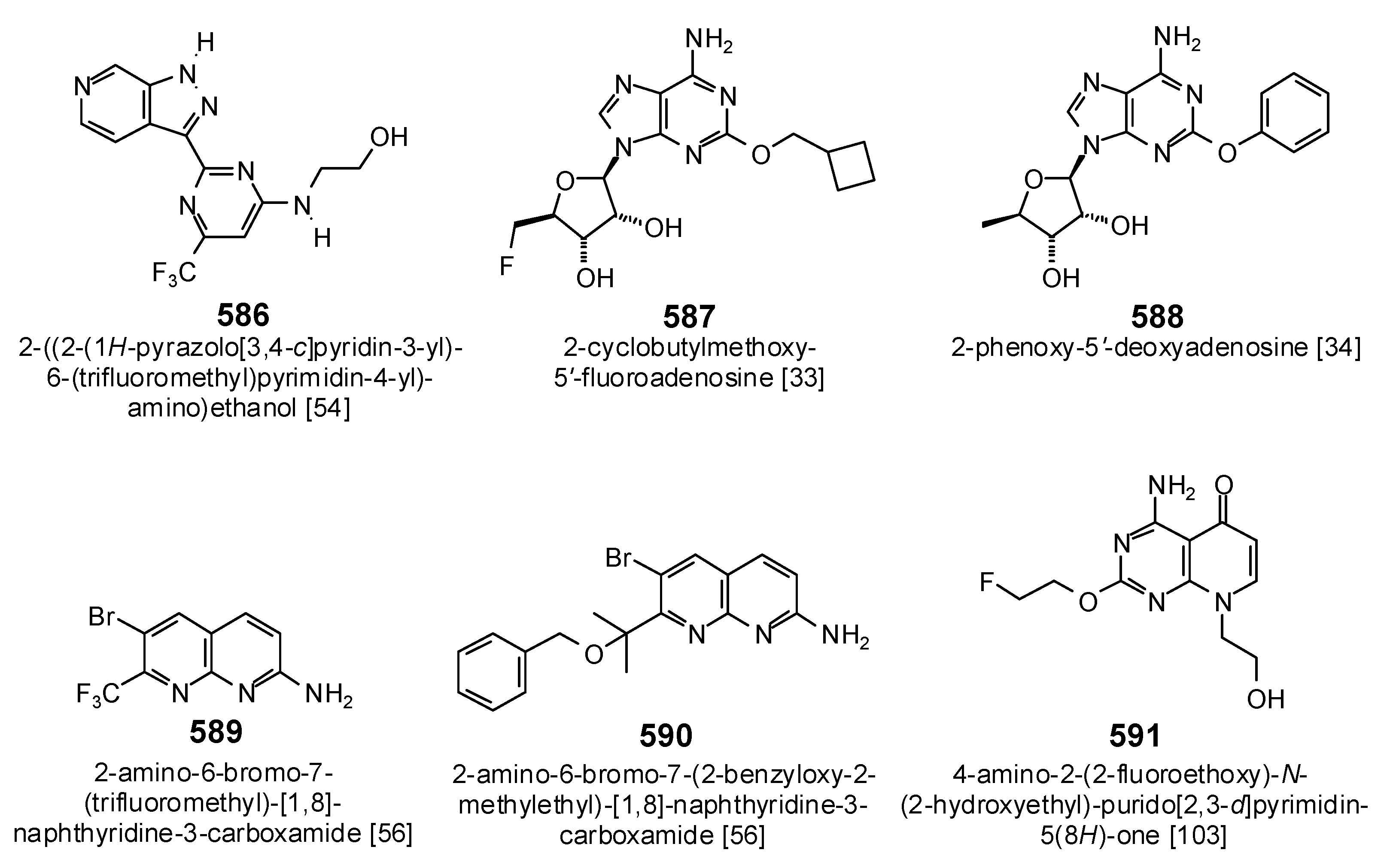
| 4CC6 | 2-{[2-(1H-Pyrazolo[3,4-c]pyridin-3-yl)-6-(trifluoromethyl)pyridin-4-yl]amino}ethanol | −110.3 | 0.65 | |
| SpLigA | 4GLW | 7-Methoxy-6-methylpteridine-2,4-diamine | −84.0 | 0.41 |
| Ligand | BcPDF | EcPDF | MtPDF | PaPDF | SaPDF | SpPDF | HsPDF | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | |
| Indole Alkaloids | ||||||||||||||
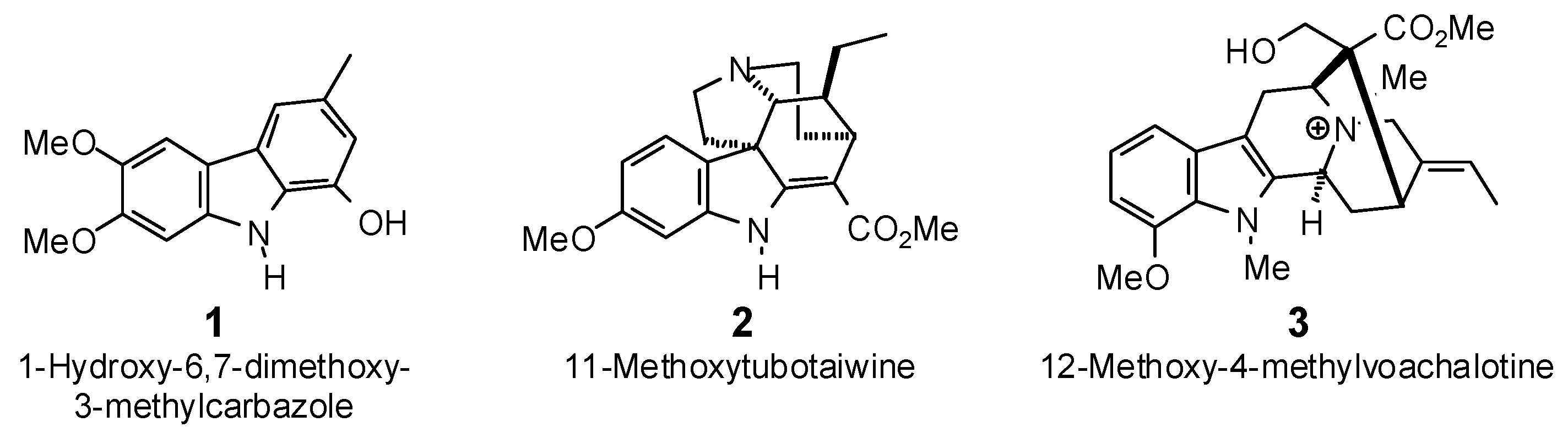
| 1-Hydroxy-6,7-dimethoxy-3-methylcarbazole (1) | −78.8 | −89.1 | −92.9 | −105.0 | −87.3 | −98.6 | −79.9 | −90.3 | −88.3 | −99.8 | −88.3 | −99.8 | −84.4 | −95.4 |
| 11-Methoxytubotaiwine (2) | −83.8 | −85.1 | −94.6 | −96.1 | −96.6 | −98.1 | −90.9 | −92.3 | −94.6 | −96.1 | −92.3 | −93.8 | −100.7 | −102.3 |
| 12-Methoxy-4-methyl-voachalotine (3) | −81.5 | −78.8 | −97.6 | −94.4 | −82.6 | −79.8 | −88.0 | −85.0 | −91.2 | −88.2 | −92.0 | −89.0 | −93.5 | −90.4 |
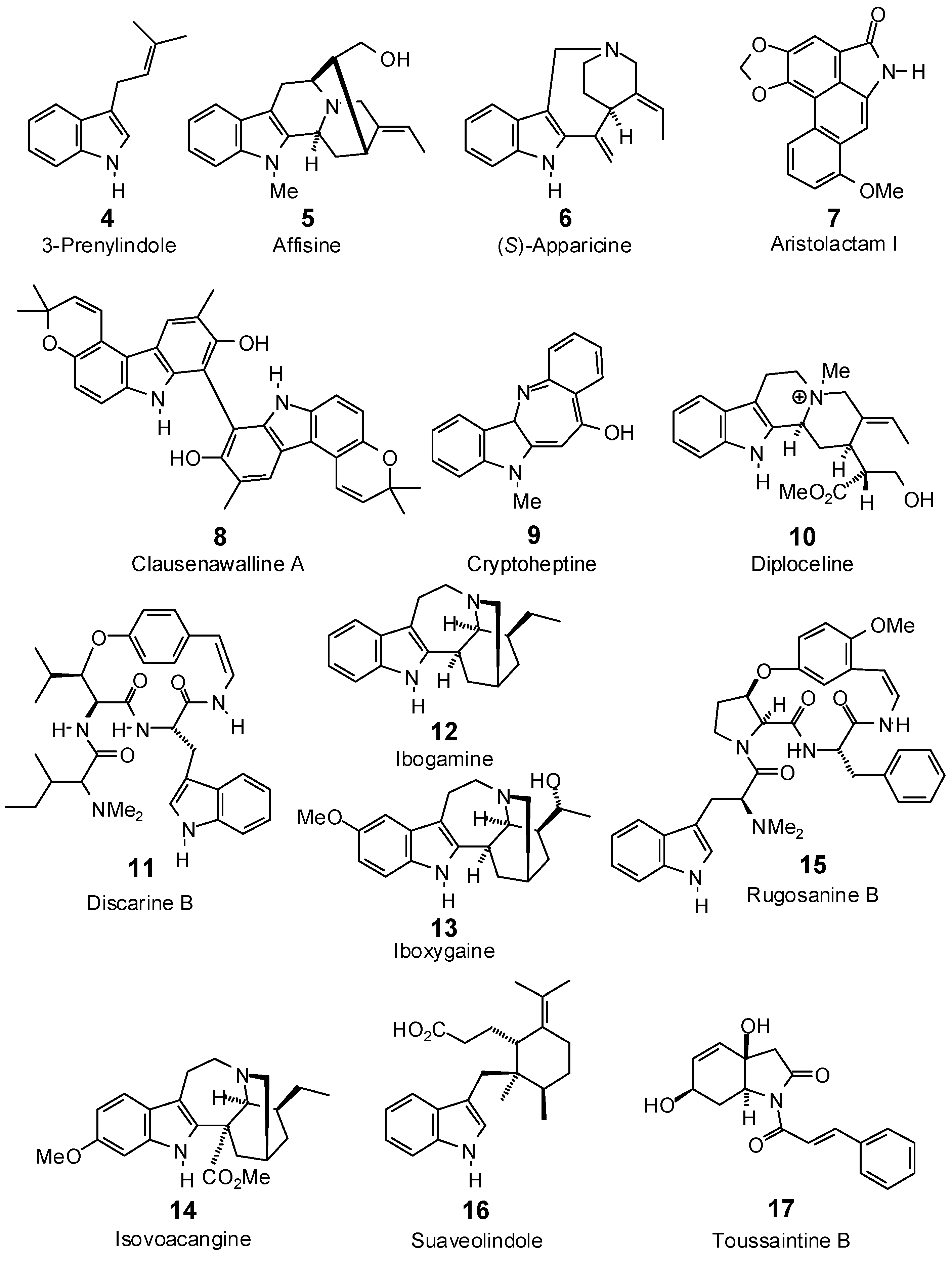
| 3-Prenylindole (4) | −80.4 | −101.4 | −83.5 | −105.3 | −84.7 | −106.8 | −84.8 | −106.9 | −82.6 | −104.1 | −79.8 | −100.7 | −87.2 | −110.0 |
| Affinisine (5) | −74.1 | −78.8 | −92.8 | −98.7 | −88.9 | −94.6 | −82.6 | −87.9 | −83.8 | −89.2 | −84.6 | −90.0 | −86.3 | −91.9 |
| Apparicine (6) | −69.2 | −77.5 | −83.9 | −94.0 | −83.4 | −93.4 | −83.3 | −93.3 | −86.2 | −96.6 | −92.7 | −103.9 | −81.8 | −91.6 |
| Aristolactam I (7) | −90.4 | −97.9 | −104.7 | −113.3 | −98.1 | −106.1 | −99.5 | −107.7 | −105.6 | −114.3 | −102.3 | −110.7 | −100.9 | −109.2 |
| Clausenawalline A (8) a | no dock | no dock | −128.8 | −112.5 | −40.1 | −35.1 | −120.0 | −104.9 | −74.0 | −64.7 | −102.2 | −89.4 | −114.5 | −100.0 |
| Cryptoheptine (9) | −77.6 | −87.2 | −96.0 | −107.9 | −79.8 | −89.6 | −91.3 | −102.5 | −95.5 | −107.2 | −98.4 | −110.5 | −86.8 | −97.5 |
| Diploceline (10) | −93.6 | −93.8 | −105.1 | −105.3 | −100.7 | −100.9 | −90.0 | −90.2 | −106.4 | −106.6 | −96.3 | −96.5 | −95.4 | −95.6 |
| Discarine B (11) | −109.8 | −95.0 | −126.2 | −109.2 | −122.3 | −105.9 | −115.8 | −100.2 | −126.0 | −109.1 | −126.7 | −109.6 | −126.0 | −109.1 |
| Ibogamine (12) | −67.3 | −74.0 | −91.1 | −100.0 | −87.8 | −96.4 | −90.5 | −99.4 | −78.9 | −86.7 | −75.4 | −82.9 | −94.6 | −104.0 |
| Iboxygaine (13) | −71.8 | −75.0 | −106.4 | −111.1 | −93.8 | −98.0 | −105.5 | −110.1 | −82.4 | −86.0 | −80.9 | −84.5 | −104.1 | −108.7 |
| Isovoacangine (14) | −90.6 | −90.9 | −100.7 | −100.9 | −89.1 | −89.4 | −94.5 | −94.8 | −83.1 | −83.3 | −96.9 | −97.2 | −89.8 | −90.1 |
| Rugosanine B (15) | −110.8 | −93.3 | −113.1 | −95.3 | −104.3 | −87.9 | −98.1 | −82.7 | −124.8 | −105.1 | −133.9 | −112.8 | −123.6 | −104.1 |
| Suaveolindole (16) | −103.0 | −104.7 | −108.1 | −109.9 | −98.5 | −100.1 | −108.1 | −109.9 | −103.2 | −105.0 | −103.8 | −105.6 | −109.0 | −110.8 |
| Toussaintine B (17) | −83.1 | −89.3 | −100.0 | −107.5 | −85.5 | −91.9 | −104.2 | −112.0 | −85.5 | −91.9 | −83.9 | −90.2 | −101.0 | −108.6 |
| Isoquinoline Alkaloids | ||||||||||||||
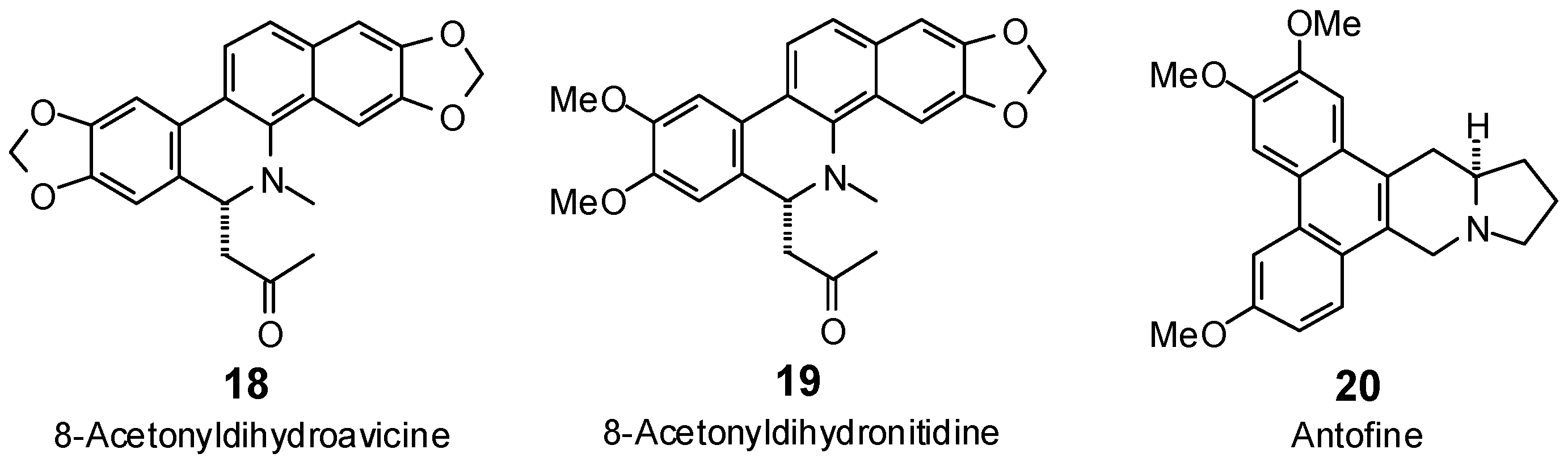
| 8-Acetonyldihydroavicine (18) | −99.8 | −98.2 | −106.8 | −105.1 | −96.4 | −94.9 | −96.6 | −95.1 | −98.6 | −97.0 | −100.0 | −98.5 | −104.4 | −102.8 |
| 8-Acetonyldihydronitidine (19) | −85.7 | −83.3 | −108.1 | −105.0 | −91.5 | −88.9 | −104.2 | −101.2 | −101.3 | −98.4 | −102.1 | −99.2 | −103.8 | −100.9 |
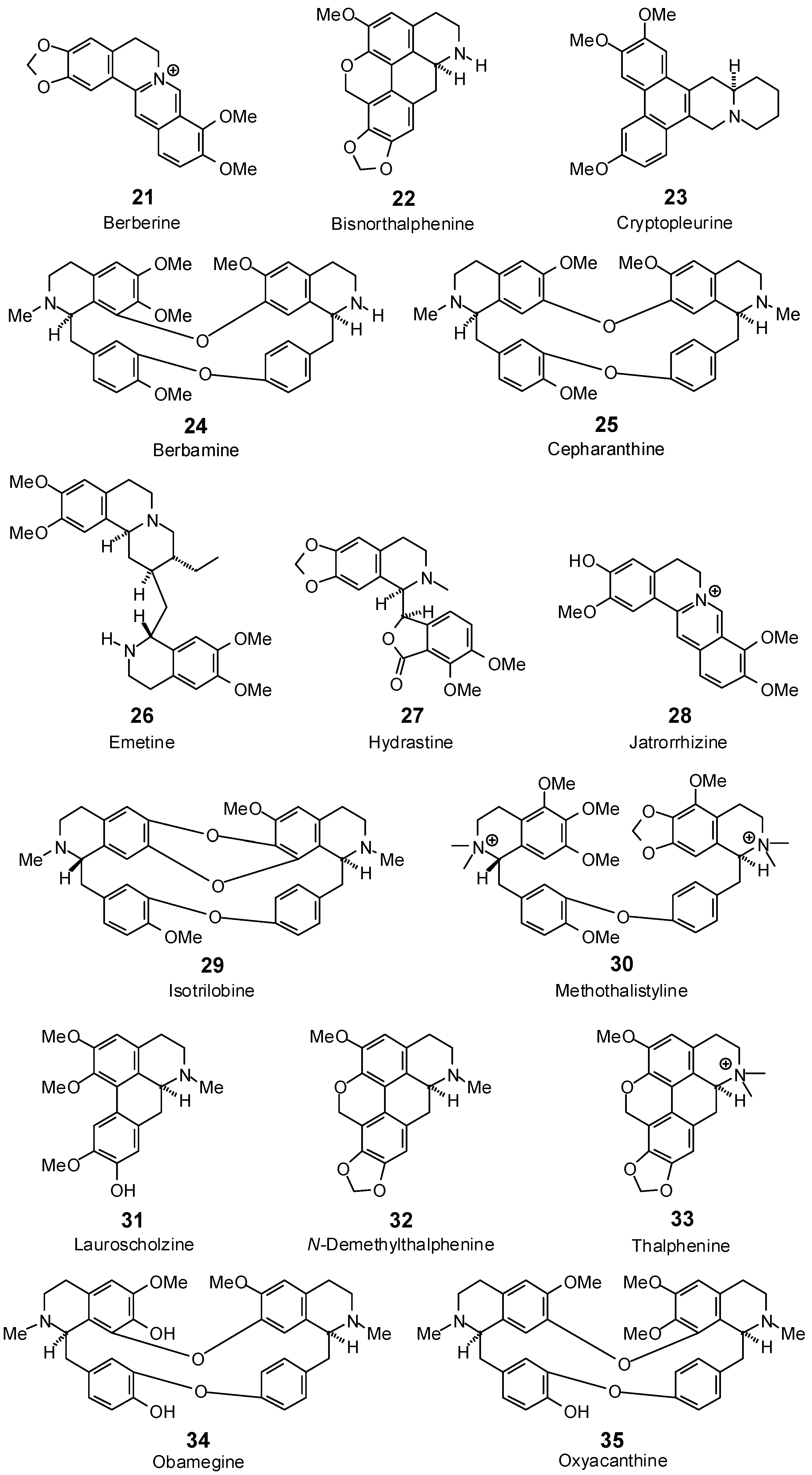
| Antofine (20) | −85.5 | −86.1 | −107.4 | −108.2 | −99.1 | −99.8 | −93.3 | −94.0 | −97.5 | −98.3 | −91.9 | −92.6 | −99.7 | −100.4 |
| Berbamine (24) | −41.7 | −35.4 | −90.1 | −76.5 | −57.2 | −48.5 | −84.3 | −71.5 | −76.7 | −65.1 | −74.4 | −63.2 | −100.3 | −85.1 |
| Berberine (21) | −84.7 | −87.6 | −103.5 | −107.0 | −91.9 | −95.0 | −88.4 | −91.4 | −88.6 | −91.6 | −87.8 | −90.7 | −87.7 | −90.7 |
| Bisnorthalphenine (22) | −95.3 | −99.8 | −101.3 | −106.1 | −104.5 | −109.5 | −97.1 | −101.7 | −98.1 | −102.8 | −99.7 | −104.4 | −96.5 | −101.0 |
| Cepharanthine (25) | −86.7 | −73.7 | −92.5 | −78.5 | −74.4 | −63.2 | −93.4 | −79.3 | −114.5 | −97.2 | −121.5 | −103.2 | −96.8 | −82.2 |
| Cryptopleurine (23) | −80.6 | −80.2 | −105.6 | −105.1 | −98.9 | −98.4 | −98.5 | −98.0 | −96.1 | −95.6 | −89.7 | −89.2 | −99.9 | −99.4 |
| Emetine (26) | −111.2 | −102.0 | −119.4 | −109.6 | −117.4 | −107.8 | −120.8 | −110.9 | −120.2 | −110.3 | −109.9 | −100.9 | −110.9 | −101.8 |
| Hydrastine (27) | −80.2 | −79.4 | −112.6 | −111.4 | −101.6 | −100.5 | −94.6 | −93.6 | −97.9 | −96.9 | −102.0 | −101.0 | −109.5 | −108.4 |
| Isotrilobine (29) | −47.8 | −41.3 | −103.3 | −89.3 | −100.3 | −86.6 | −86.3 | −74.5 | −99.3 | −85.7 | −87.6 | −75.6 | −102.4 | −88.4 |
| Jatrorrhizine (28) | −86.4 | −89.1 | −101.5 | −104.7 | −95.1 | −98.1 | −92.0 | −94.9 | −93.2 | −96.2 | −92.3 | −95.2 | −94.7 | −97.7 |
| Lauroscholtzine (31) | −71.5 | −73.6 | −95.5 | −98.2 | −93.0 | −95.6 | −91.1 | −93.7 | −98.4 | −101.2 | −93.5 | −96.2 | −90.7 | −93.3 |
| Methothalistyline (30) | −76.6 | −61.6 | −112.1 | −90.2 | −104.5 | −84.1 | −115.7 | −93.1 | −108.1 | −87.0 | −127.9 | −102.9 | −132.7 | −106.8 |
| N-Demethylthalphenine (32) | −100.4 | −103.7 | −98.7 | −101.9 | −107.6 | −111.1 | −102.2 | −105.6 | −102.1 | −105.4 | −98.0 | −101.2 | −93.5 | −96.6 |
| Obamegine (34) | no dock | no dock | −83.1 | −71.1 | −19.4 | −16.6 | −67.9 | −58.0 | −83.2 | −71.1 | −83.7 | −71.6 | −102.0 | −87.2 |
| Oxyacanthine (35) | −39.7 | −33.7 | −93.7 | −79.5 | 11.1 | 9.4 | −85.3 | −72.4 | −69.8 | −59.2 | −85.8 | −72.8 | −94.1 | −79.8 |
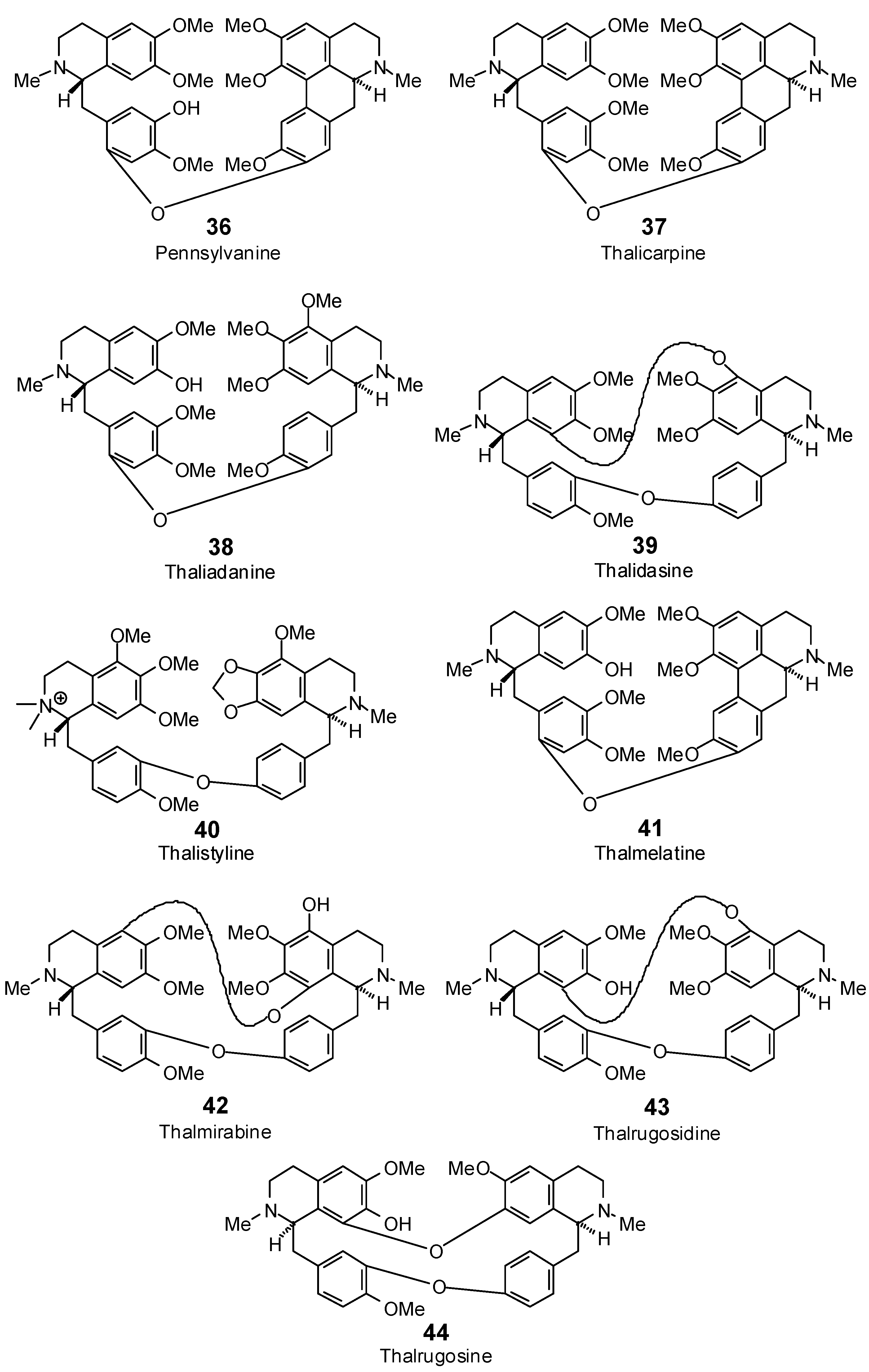
| Pennsylvanine (36) | −102.4 | −83.6 | −131.0 | −106.9 | −113.2 | −92.4 | −108.7 | −88.7 | −126.2 | −103.1 | −129.9 | −106.1 | −137.8 | −112.5 |
| Thaliadanine (38) | −91.0 | −73.2 | −131.2 | −105.5 | −89.9 | −72.3 | −108.5 | −87.2 | −123.6 | −99.4 | −124.3 | −100.0 | −124.7 | −100.3 |
| Thalicarpine (37) | −87.8 | −71.2 | −128.3 | −104.0 | −105.2 | −85.3 | −105.9 | −85.9 | −130.1 | −105.5 | −115.1 | −93.3 | −114.6 | −93.0 |
| Thalidasine (39) | −64.5 | −53.4 | −82.5 | −68.4 | −67.3 | −55.8 | −92.8 | −76.9 | −82.9 | −68.7 | −103.5 | −85.8 | −102.2 | −84.7 |
| Thalistyline (40) | −84.1 | −68.2 | −119.3 | −96.7 | −96.0 | −77.8 | −111.9 | −90.7 | −118.2 | −95.8 | −117.8 | −95.5 | −120.6 | −97.8 |
| Thalmelatine (41) | −87.7 | −71.6 | −130.3 | −106.4 | −102.8 | −83.9 | −116.5 | −95.1 | −124.1 | −101.3 | −130.5 | −106.5 | −145.0 | −118.4 |
| Thalmirabine (42) | −76.5 | −62.9 | −85.1 | −69.9 | −39.2 | −32.2 | −73.6 | −60.5 | −111.1 | −91.3 | −100.2 | −82.4 | −97.4 | −80.1 |
| Thalphenine (33) | −88.5 | −90.1 | −102.6 | −104.5 | −91.2 | −92.8 | −94.6 | −96.3 | −93.8 | −95.5 | −91.6 | −93.2 | −93.0 | −94.6 |
| Thalrugosidine (43) | no dock | no dock | −90.7 | −75.7 | −42.4 | −35.4 | −89.4 | −74.6 | −97.2 | −81.1 | −106.9 | −89.2 | −96.1 | −80.2 |
| Thalrugosine (44) | no dock | no dock | −85.7 | −72.7 | −25.2 | −21.4 | −69.3 | −58.8 | −83.6 | −70.9 | −97.6 | −82.8 | −104.2 | −88.4 |
| Piperidine, Pyrrole, Pyrrolizidine, Quinoline, and Steroidal Alkaloids | ||||||||||||||
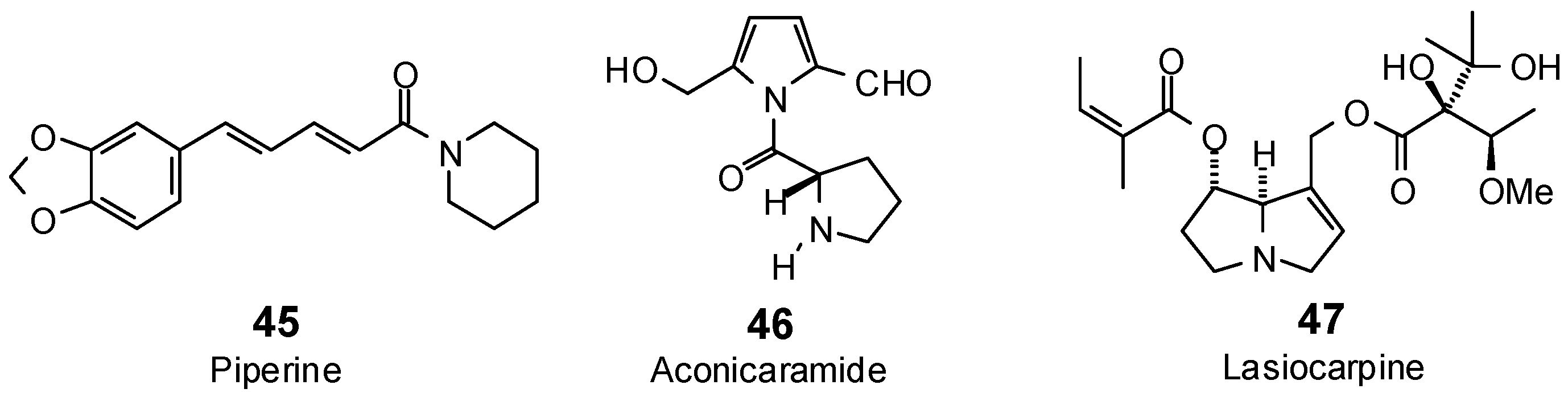
| Aconicaramide (46) | −86.0 | −102.0 | −89.6 | −106.3 | −80.8 | −96.0 | −89.1 | −105.8 | −89.7 | −106.5 | −85.4 | −101.4 | −89.5 | −106.2 |
| Lasiocarpine (47) | −82.9 | −80.2 | −121.9 | −117.8 | −104.0 | −100.5 | −106.1 | −102.6 | −117.3 | −113.4 | −108.5 | −104.9 | −119.5 | −115.6 |
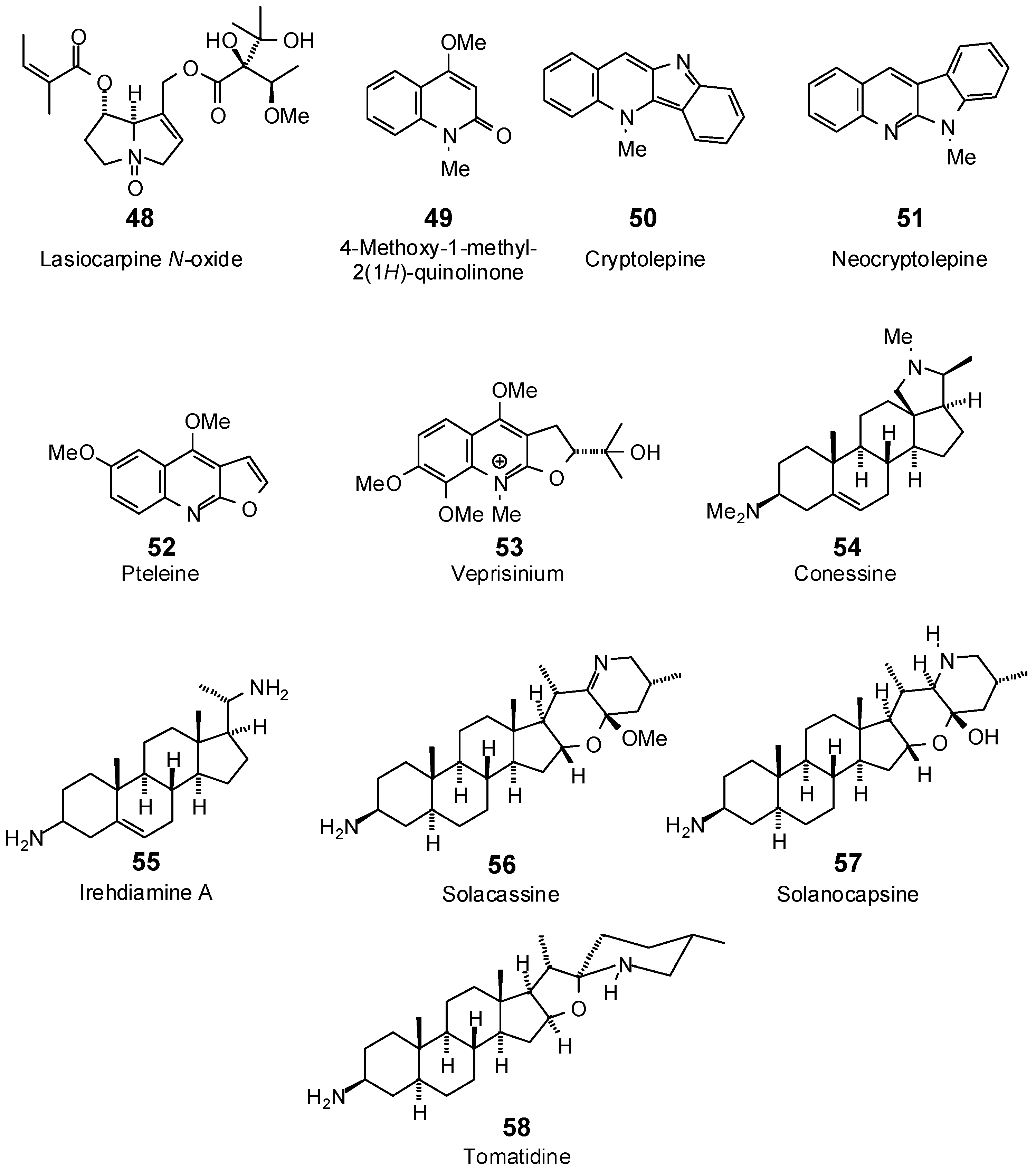
| Lasiocarpine N-oxide (48) | −94.5 | −90.2 | −119.1 | −113.6 | −110.0 | −105.0 | −104.5 | −99.8 | −106.8 | −102.0 | −108.2 | −103.2 | −117.7 | −112.4 |
| Piperine (45) | −88.8 | −97.0 | −107.3 | −117.2 | −89.5 | −97.7 | −104.9 | −114.5 | −95.0 | −103.8 | −94.8 | −103.5 | −104.3 | −113.9 |
| 4-Methoxy-1-methyl-2(1H)-quinolinone (49) | −70.1 | −87.8 | −73.5 | −92.0 | −68.7 | −86.0 | −72.6 | −90.9 | −72.3 | −90.6 | −73.1 | −91.5 | −72.1 | −90.3 |
| Cryptolepine (50) | −74.4 | −87.0 | −85.3 | −99.8 | −79.1 | −92.5 | −82.8 | −96.9 | −86.8 | −101.6 | −88.4 | −103.4 | −80.9 | −94.6 |
| Neocryptolepine (51) | −71.2 | −83.2 | −83.5 | −97.7 | −77.5 | −90.6 | −79.9 | −93.5 | −87.6 | −102.5 | −87.7 | −102.6 | −80.4 | −94.1 |
| Pteleine (52) | −84.5 | −99.2 | −90.0 | −105.8 | −83.4 | −98.0 | −89.3 | −104.9 | −88.9 | −104.4 | −88.2 | −103.6 | −85.6 | −100.6 |
| Veprisinium (53) | −89.8 | −93.0 | −99.2 | −102.8 | −101.7 | −105.4 | −100.0 | −103.6 | −95.2 | −98.6 | −94.3 | −97.7 | −96.3 | −99.8 |
| Conessine (54) | −70.4 | −71.4 | −103.5 | −104.9 | −83.6 | −84.8 | −74.2 | −75.3 | −88.6 | −89.8 | −95.1 | −96.4 | −81.9 | −83.0 |
| Irehdiamine A (55) | −84.2 | −88.9 | −89.8 | −94.8 | −98.5 | −104.0 | −89.4 | −94.3 | −91.2 | −96.2 | −87.0 | −91.8 | −85.4 | −90.0 |
| Solacassine (56) | −75.6 | −71.3 | −92.6 | −87.4 | −83.1 | −78.4 | −86.5 | −81.6 | −89.6 | −84.6 | −92.4 | −87.2 | −86.7 | −81.8 |
| Solanocapsine (57) | −75.3 | −71.7 | −94.2 | −89.6 | −88.8 | −84.6 | −74.3 | −70.7 | −87.6 | −83.4 | −85.1 | −81.0 | −97.4 | −92.7 |
| Tomatidine (58) | −71.2 | −68.6 | −94.8 | −91.4 | −65.7 | −63.3 | −88.3 | −85.1 | −90.7 | −87.4 | −87.1 | −83.9 | −98.0 | −94.5 |
| Miscellaneous Alkaloids | ||||||||||||||
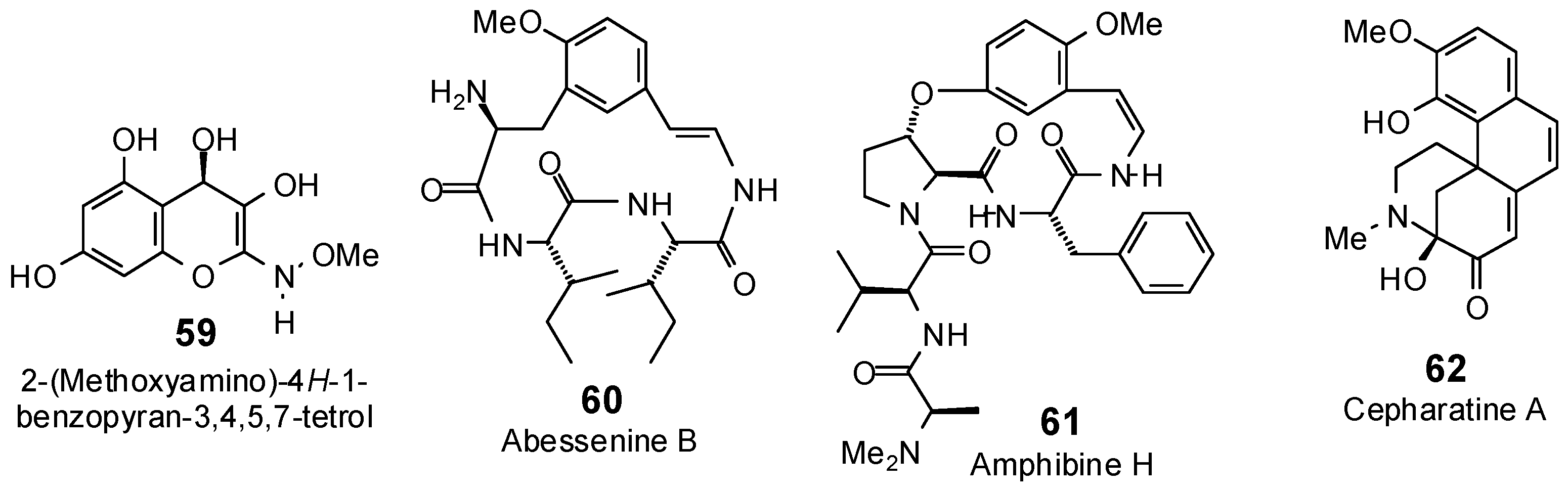
| 2-(Methoxyamino)-4H-1-benzo-pyran-3,4,5,7-tetrol (59) | −84.7 | −97.8 | −87.8 | −101.4 | −80.5 | −93.0 | −89.2 | −103.0 | −85.9 | −99.2 | −89.3 | −103.1 | −84.6 | −97.7 |
| Abyssenine C (60) | −78.7 | −74.2 | −102.5 | −96.6 | −111.3 | −104.8 | −106.6 | −100.4 | −103.1 | −97.1 | −101.2 | −95.4 | −103.7 | −97.6 |
| Amphibine H (61) | −64.2 | −54.6 | −115.2 | −97.9 | −123.8 | −105.2 | −118.6 | −100.8 | −119.9 | −101.9 | −117.1 | −99.5 | −121.1 | −102.9 |
| Cepharatine A (62) | −81.4 | −86.2 | −97.4 | −103.1 | −78.9 | −83.5 | −92.4 | −97.8 | −95.7 | −101.3 | −94.5 | −100.1 | −90.3 | −95.6 |
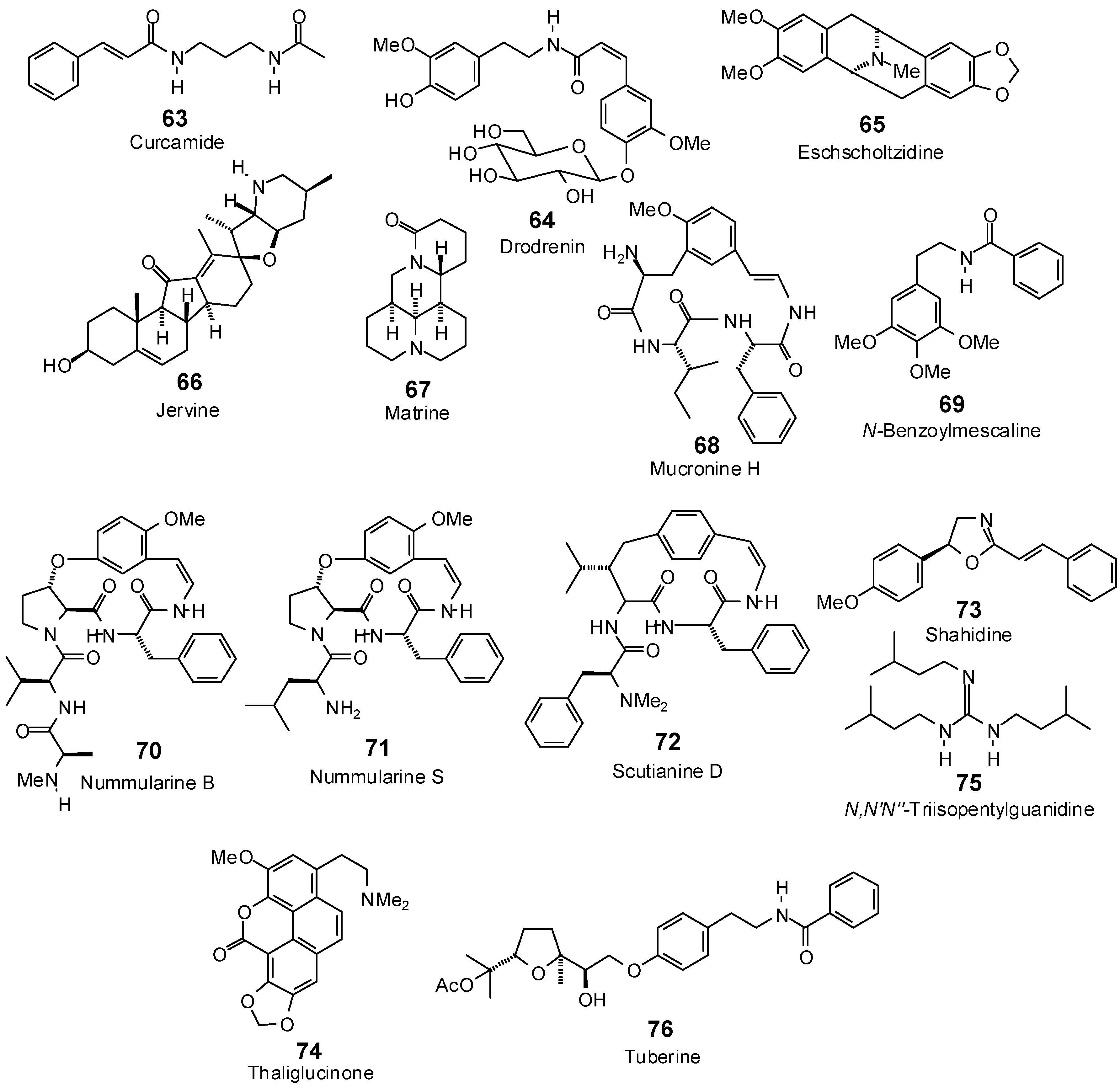
| Curcamide (63) | −93.8 | −107.5 | −98.8 | −113.3 | −93.0 | −106.6 | −100.5 | −115.2 | −97.4 | −111.8 | −95.5 | −109.5 | −102.7 | −117.8 |
| Drodrenin (64) | −113.2 | −102.1 | −148.6 | −134.1 | −123.6 | −111.6 | −135.5 | −122.3 | −130.6 | −117.9 | −135.6 | −122.3 | −132.2 | −119.4 |
| Eschscholtzidine (65) | −79.9 | −82.3 | −104.0 | −107.2 | −94.8 | −97.7 | −92.3 | −95.2 | −94.0 | −96.9 | −91.4 | −94.3 | −99.1 | −102.1 |
| Jervine (66) | −71.6 | −68.5 | −96.4 | −92.1 | −83.5 | −79.8 | −92.5 | −88.4 | −92.3 | −88.3 | −100.9 | −96.4 | −97.7 | −93.4 |
| Matrine (67) | −80.2 | −91.7 | −91.0 | −104.1 | −83.6 | −95.7 | −90.8 | −103.9 | −85.9 | −98.3 | −87.7 | −100.3 | −85.0 | −97.3 |
| Mucronine H (68) | −101.8 | −93.6 | −109.2 | −100.4 | −111.0 | −102.0 | −103.5 | −95.1 | −109.0 | −100.2 | −114.6 | −105.3 | −124.4 | −114.3 |
| N-Benzoylmescaline (69) | −101.3 | −107.0 | −109.7 | −115.9 | −100.9 | −106.6 | −112.4 | −118.7 | −103.4 | −109.2 | −99.0 | −104.6 | −109.9 | −116.1 |
| Nummularine B (70) | −107.3 | −91.9 | −126.2 | −108.1 | −127.8 | −109.5 | −108.7 | −93.1 | −129.4 | −110.8 | −122.9 | −105.2 | −119.4 | −102.2 |
| Nummularine S (71) | −84.2 | −75.3 | −120.7 | −107.8 | −116.1 | −103.8 | −112.8 | −100.8 | −124.0 | −110.8 | −119.0 | −106.4 | −130.6 | −116.7 |
| Scutianine B (72) | −90.1 | −78.2 | −128.5 | −111.5 | −103.5 | −89.8 | −124.6 | −108.1 | −118.8 | −103.1 | −118.3 | −102.6 | −126.8 | −110.0 |
| Shahidine (73) | −85.2 | −93.7 | −105.5 | −116.1 | −88.1 | −96.9 | −98.5 | −108.4 | −93.8 | −103.1 | −95.6 | −105.2 | −101.8 | −112.0 |
| Thaliglucinone (74) | −92.9 | −93.4 | −116.0 | −116.6 | −106.1 | −106.7 | −119.4 | −120.1 | −104.9 | −105.5 | −106.9 | −107.5 | −103.3 | −103.8 |
| Triisopenylguanidine (75) | −91.4 | −101.7 | −107.6 | −119.8 | −95.6 | −106.4 | −98.4 | −109.6 | −97.2 | −108.2 | −92.1 | −102.6 | −101.4 | −112.9 |
| Tuberine (76) | −92.7 | −85.7 | −136.7 | −126.5 | −103.7 | −95.9 | −105.6 | −97.7 | −121.9 | −112.7 | −121.1 | −112.0 | −121.7 | −112.6 |
| Monoterpenoids | ||||||||||||||
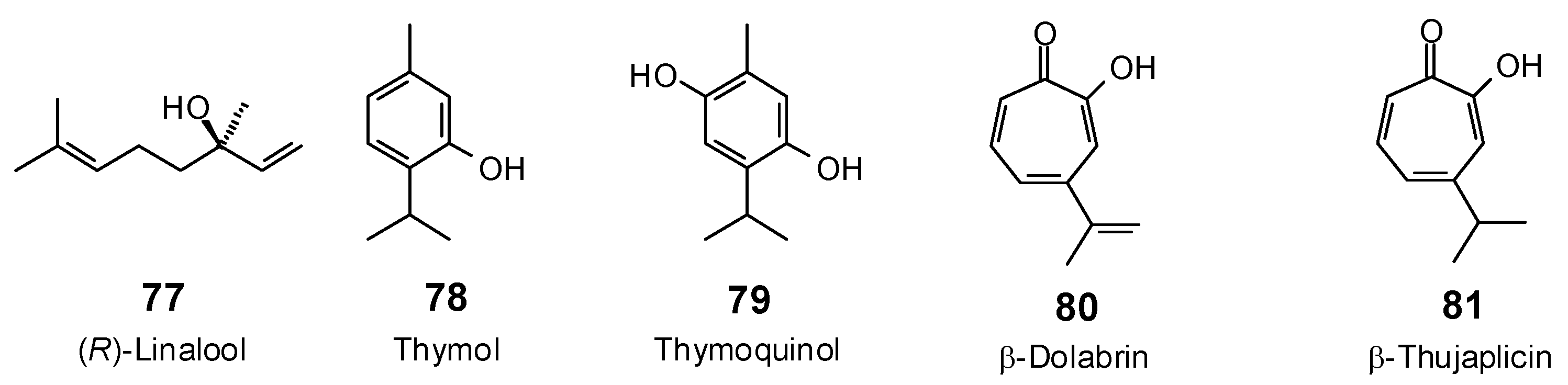
| Linalool (77) | −78.7 | −105.6 | −81.4 | −109.1 | −75.9 | −101.7 | −80.0 | −107.3 | −77.0 | −103.2 | −77.9 | −104.4 | −80.4 | −107.8 |
| Thymol (78) | −68.6 | −92.7 | −71.0 | −96.1 | −65.8 | −89.0 | −67.5 | −91.3 | −69.5 | −94.1 | −69.0 | −93.3 | −68.6 | −92.8 |
| Thymoquinol (79) | −73.7 | −96.4 | −76.2 | −99.6 | −70.6 | −92.4 | −75.7 | −98.9 | −76.9 | −100.5 | −75.7 | −99.0 | −73.8 | −96.5 |
| β-Dolabrin (80) | −76.9 | −101.3 | −76.9 | −101.4 | −74.2 | −97.8 | −77.3 | −102.0 | −77.9 | −102.7 | −77.7 | −102.4 | −75.0 | −98.9 |
| β-Thujaplicin (81) | −74.9 | −98.4 | −75.8 | −99.5 | −73.4 | −96.3 | −75.6 | −99.3 | −76.2 | −100.1 | −76.9 | −100.9 | −74.7 | −98.1 |
| Sesquiterpenoids | ||||||||||||||
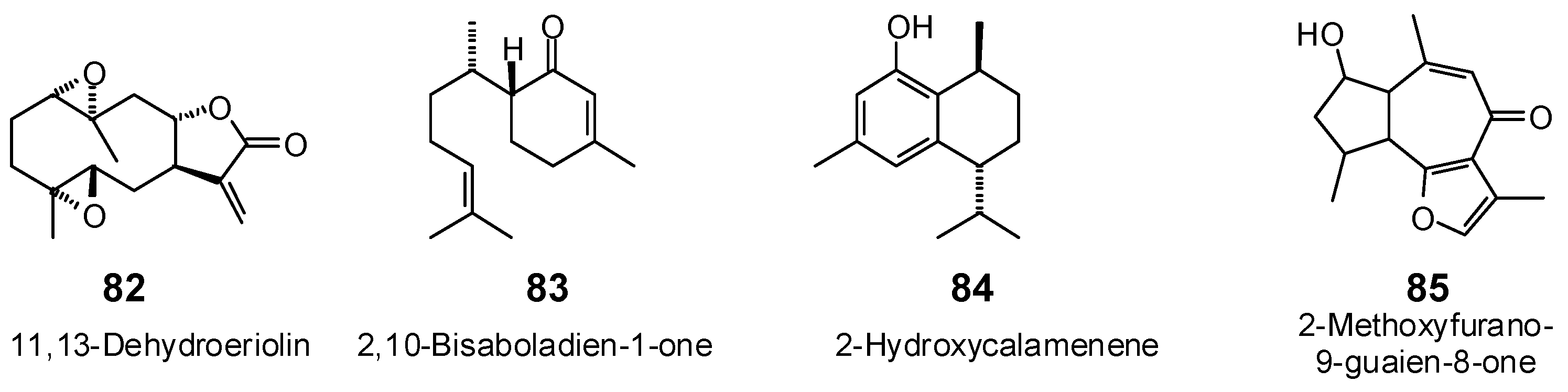
| 11,13-Dehydroeriolin (82) | −85.2 | −95.4 | −100.0 | −112.0 | −89.1 | −99.8 | −101.1 | −113.2 | −93.0 | −104.2 | −86.9 | −97.4 | −84.6 | −94.8 |
| 2,10-Bisaboladien-1-one (83) | −84.5 | −100.6 | −91.0 | −108.3 | −94.6 | −112.6 | −86.0 | −102.3 | −93.0 | −110.7 | −89.4 | −106.4 | −86.2 | −102.7 |
| 2-Hydroxycalamenene (84) | −81.1 | −96.8 | −85.0 | −101.5 | −81.5 | −97.3 | −84.5 | −100.9 | −83.6 | −99.8 | −82.8 | −98.9 | −78.0 | −93.1 |
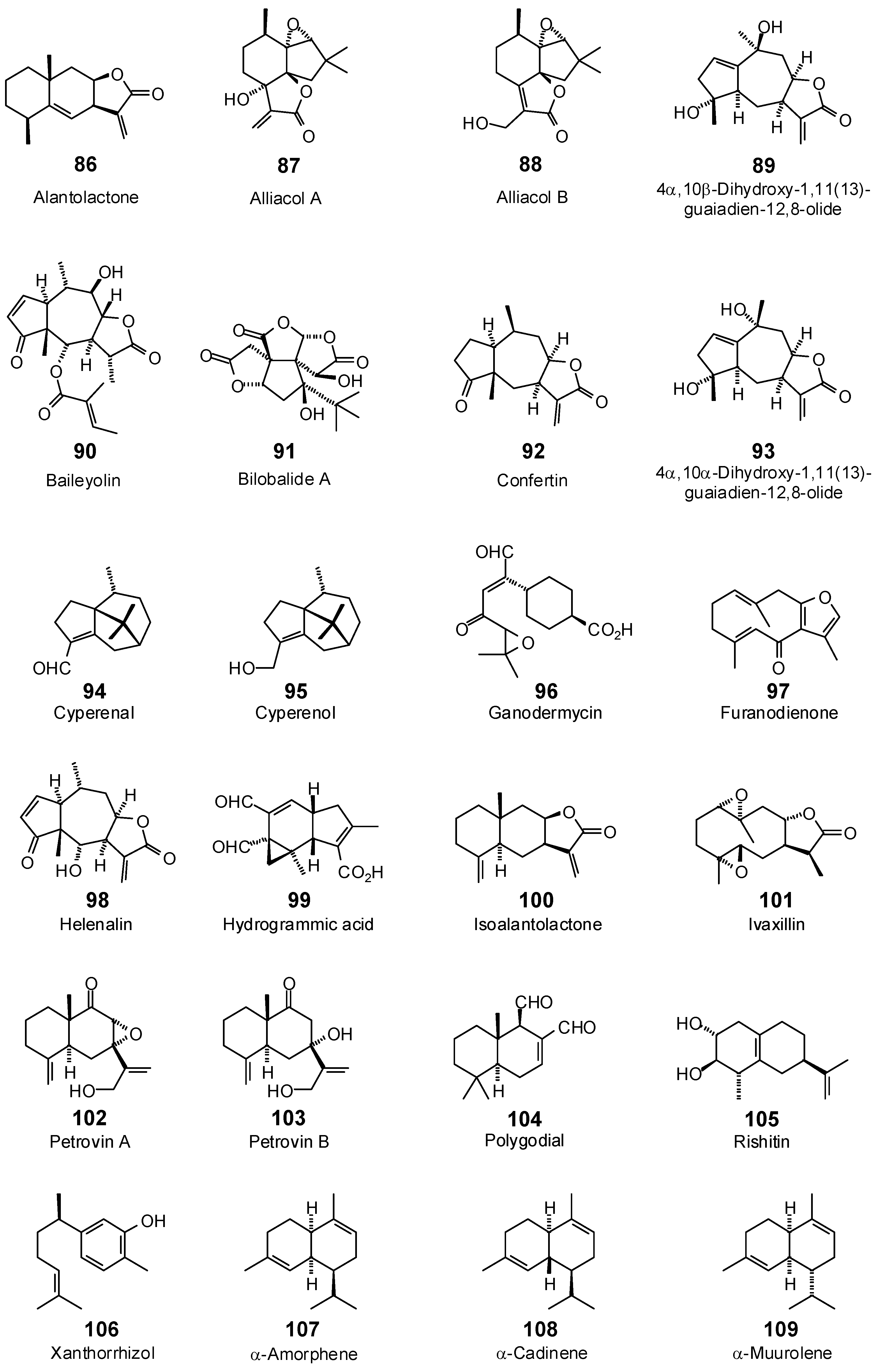
| 2-Methoxyfurano-9-guaien-8-one (85) | −91.7 | −103.3 | −100.3 | −112.9 | −93.2 | −104.9 | −102.4 | −115.3 | −102.9 | −115.8 | −101.3 | −114.1 | −95.4 | −107.5 |
| 4α,10α-Dihydroxy-1,11(13)-guaiadien-12,8-olide (93) | −89.8 | −100.6 | −93.3 | −104.5 | −87.2 | −97.7 | −97.1 | −108.7 | −91.3 | −102.3 | −90.0 | −100.8 | −96.6 | −108.2 |
| 4α,10β-Dihydroxy-1,11(13)-guaiadien-12,8-olide (89) | −94.0 | −105.3 | −99.8 | −111.8 | −104.1 | −116.6 | −98.7 | −110.6 | −102.0 | −114.3 | −93.3 | −104.6 | −96.7 | −108.3 |
| Alantolactone (86) | −78.9 | −92.2 | −80.7 | −94.4 | −77.3 | −90.4 | −82.9 | −97.0 | −86.2 | −100.8 | −83.2 | −97.3 | −78.2 | −91.4 |
| Alliacol A (87) | −45.9 | −51.5 | −75.8 | −85.0 | −67.2 | −75.3 | −77.4 | −86.7 | −73.1 | −81.9 | −72.7 | −81.4 | −77.0 | −86.3 |
| Alliacol B (88) | −78.5 | −87.9 | −82.2 | −92.0 | −90.5 | −101.4 | −81.8 | −91.6 | −85.3 | −95.5 | −80.3 | −89.9 | −81.7 | −91.6 |
| Artemisinic acid (113) | −84.4 | −98.4 | −85.0 | −99.2 | −86.2 | −100.5 | −83.8 | −97.7 | −88.3 | −103.0 | −86.0 | −100.3 | −81.4 | −94.9 |
| Baileyolin (90) | −98.8 | −99.6 | −103.1 | −104.0 | −111.4 | −112.3 | −105.2 | −106.1 | −113.3 | −114.3 | −108.2 | −109.1 | −103.2 | −104.0 |
| Bilobalide A (91) | −78.0 | −81.4 | −93.0 | −97.1 | −95.6 | −99.9 | −82.5 | −86.2 | −98.6 | −103.0 | −102.0 | −106.5 | −95.8 | −100.0 |
| Confertin (92) | −83.4 | −95.4 | −90.9 | −104.0 | −91.4 | −104.6 | −90.5 | −103.5 | −89.2 | −102.0 | −87.4 | −100.0 | −87.5 | −100.1 |
| Cyperenal (94) | −46.2 | −55.2 | −65.6 | −78.3 | −70.6 | −84.3 | −60.6 | −72.4 | −63.2 | −75.4 | −67.6 | −80.7 | −60.9 | −72.7 |
| Cyperenol (95) | −36.1 | −43.0 | −65.5 | −77.9 | −70.6 | −84.0 | −57.8 | −68.8 | −62.6 | −74.5 | −64.7 | −77.1 | −57.1 | −68.0 |
| Furanodienone (97) | −84.3 | −98.9 | −88.4 | −103.7 | −87.1 | −102.1 | −94.7 | −111.1 | −89.0 | −104.4 | −88.5 | −103.8 | −87.5 | −102.6 |
| Ganodermycin (96) | −111.8 | −122.8 | −109.5 | −120.3 | −115.7 | −127.1 | −109.3 | −120.1 | −113.6 | −124.8 | −110.9 | −121.8 | −105.4 | −115.7 |
| Helenalin (98) | −69.8 | −78.4 | −81.9 | −92.0 | −82.8 | −93.0 | −80.0 | −89.8 | −84.2 | −94.6 | −85.8 | −96.4 | −86.5 | −97.2 |
| Hydrogrammic acid (99) | −83.0 | −93.5 | −91.4 | −103.0 | −86.8 | −97.7 | −88.5 | −99.6 | −88.9 | −100.1 | −87.8 | −98.8 | −83.7 | −94.3 |
| Isoalantolactone (100) | −79.3 | −92.8 | −84.7 | −99.0 | −76.8 | −89.8 | −87.9 | −102.9 | −85.1 | −99.5 | −85.4 | −99.9 | −78.0 | −91.2 |
| Ivaxillin (101) | −81.1 | −90.6 | −98.4 | −110.0 | −90.3 | −100.9 | −100.6 | −112.4 | −93.6 | −104.6 | −90.4 | −101.0 | −82.8 | −92.6 |
| Petrovin A (102) | −84.1 | −96.2 | −88.9 | −101.7 | −84.5 | −96.6 | −90.1 | −103.0 | −88.5 | −101.2 | −86.0 | −98.3 | −86.2 | −98.7 |
| Petrovin B (103) | −84.8 | −96.8 | −87.5 | −99.9 | −80.5 | −91.9 | −89.9 | −102.6 | −86.6 | −98.8 | −84.7 | −96.6 | −85.9 | −98.0 |
| Polygodial (104) | −79.0 | −92.1 | −84.1 | −98.1 | −88.1 | −102.7 | −82.8 | −96.5 | −88.8 | −103.6 | −86.1 | −100.4 | −74.8 | −87.2 |
| Rishitin (105) | −75.0 | −89.0 | −80.0 | −94.9 | −77.0 | −91.4 | −81.6 | −96.8 | −79.3 | −94.1 | −80.8 | −95.9 | −81.0 | −96.1 |
| Xanthorrhizol (106) | −86.1 | −102.8 | −90.9 | −108.5 | −91.2 | −108.9 | −90.1 | −107.5 | −92.3 | −110.3 | −88.6 | −105.8 | −91.7 | −109.4 |
| α-Amorphene (107) | −75.5 | −92.2 | −79.5 | −97.1 | −80.2 | −97.9 | −77.9 | −95.1 | −78.1 | −95.3 | −73.8 | −90.1 | −72.0 | −87.9 |
| α-Cadinene (108) | −73.3 | −89.5 | −80.6 | −98.4 | −79.8 | −97.4 | −80.4 | −98.1 | −79.6 | −97.2 | −77.6 | −94.8 | −77.8 | −94.9 |
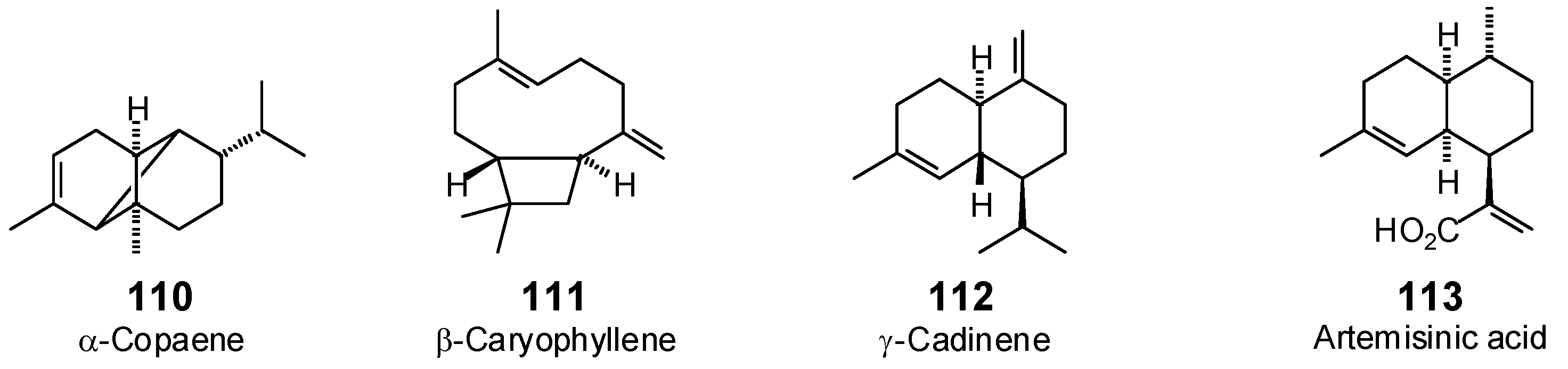
| α-Copaene (110) | −66.9 | −83.6 | −65.6 | −81.9 | −67.3 | −84.1 | −66.7 | −83.3 | −73.2 | −91.5 | −68.6 | −85.7 | −68.0 | −85.0 |
| α-Muurolene (109) | −76.9 | −93.9 | −79.1 | −96.6 | −75.9 | −92.7 | −78.2 | −95.4 | −78.7 | −96.1 | −77.3 | −94.3 | −79.0 | −96.4 |
| γ-Cadinene (112) | −77.2 | −94.2 | −81.4 | −99.4 | −77.5 | −94.6 | −81.9 | −99.9 | −81.4 | −99.4 | −79.7 | −97.3 | −79.7 | −97.3 |
| Diterpenoids | ||||||||||||||
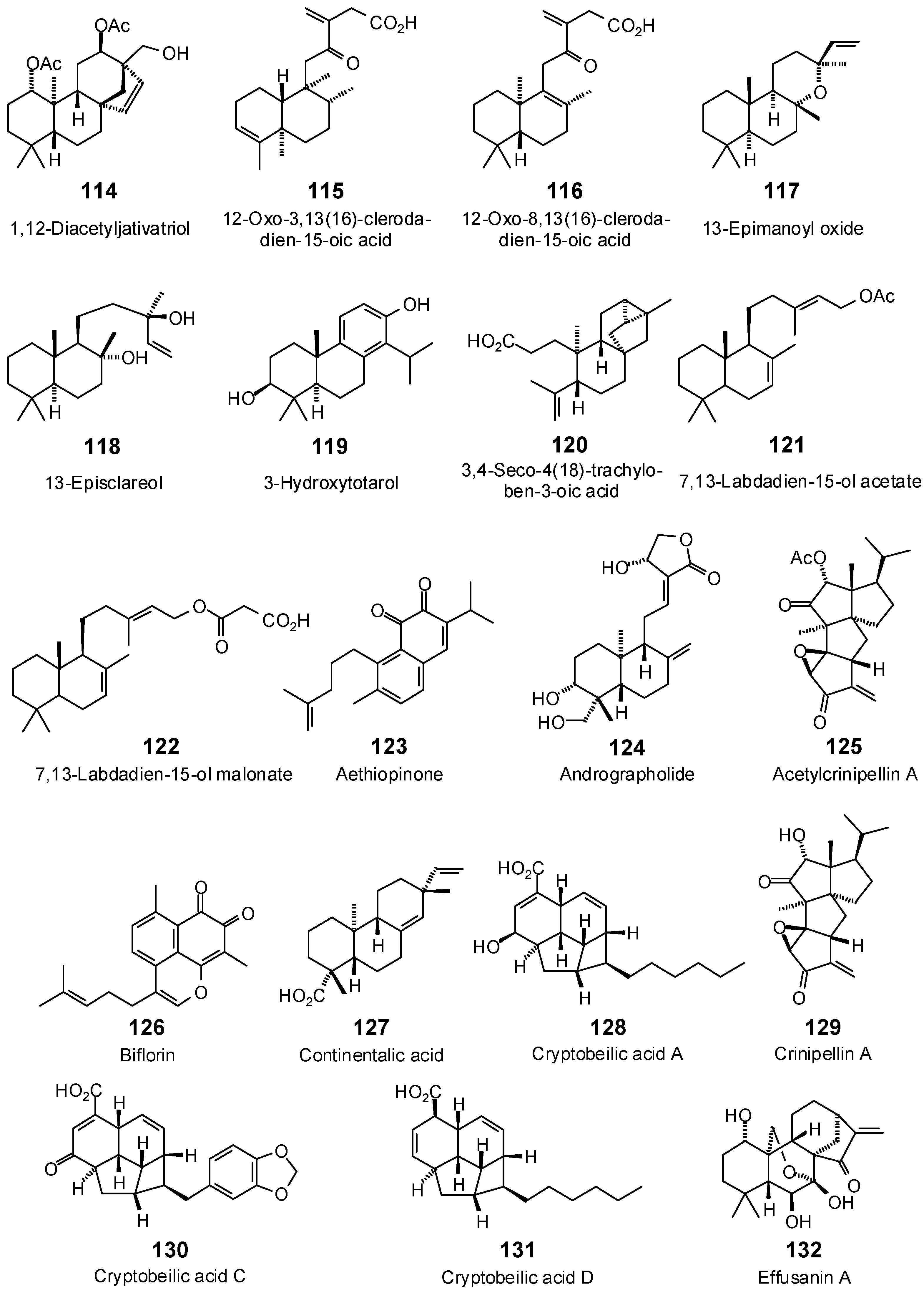
| 1,12-Diacetyljativatriol (114) | −96.9 | −94.2 | −107.0 | −104.0 | −107.1 | −104.1 | −98.6 | −95.9 | −101.6 | −98.8 | −102.1 | −99.3 | −102.4 | −99.5 |
| 12-Oxo-3,13(16)-clerodadien-15-oic acid (115) | −104.5 | −110.0 | −102.9 | −108.3 | −98.3 | −103.5 | −101.8 | −107.1 | −109.2 | −114.9 | −96.9 | −102.0 | −100.8 | −106.1 |
| 12-Oxo-8,13(16)-clerodadien-15-oic acid (116) | −93.4 | −98.3 | −106.7 | −112.3 | −102.3 | −107.8 | −99.8 | −105.1 | −106.6 | −112.3 | −107.9 | −113.6 | −95.3 | −100.3 |
| 13-Epimanoyl oxide (117) | −58.8 | −63.8 | −81.6 | −88.6 | −86.1 | −93.5 | −81.0 | −88.0 | −78.6 | −85.3 | −78.8 | −85.6 | −76.7 | −83.3 |
| 13-Episclareol (118) | −88.1 | −93.8 | −101.5 | −108.0 | −103.0 | −109.6 | −99.6 | −106.0 | −99.1 | −105.5 | −94.6 | −100.7 | −95.7 | −101.8 |
| 3,4-Seco-4(18)-trachyloben-3-oic acid (120) | −99.3 | −106.4 | −99.0 | −106.1 | −99.6 | −106.7 | −96.3 | −103.2 | −99.3 | −106.4 | −96.5 | −103.3 | −94.9 | −101.6 |
| 3-Hydroxytotarol (119) | −82.6 | −88.5 | −87.0 | −93.2 | −83.0 | −88.9 | −89.1 | −95.4 | −86.2 | −92.3 | −81.7 | −87.5 | −84.8 | −90.8 |
| 7,13-Labdadien-15-ol acetate (121) | −76.8 | −79.7 | −108.6 | −112.7 | −105.3 | −109.3 | −106.9 | −110.9 | −105.1 | −109.0 | −102.0 | −105.8 | −104.1 | −108.1 |
| 7,13-Labdadien-15-ol malonate (122) | −99.8 | −99.4 | −121.2 | −120.6 | −111.4 | −110.9 | −115.9 | −115.4 | −115.9 | −115.4 | −122.7 | −122.2 | −105.3 | −104.8 |
| Acetylcrinipellin A (125) | −90.1 | −90.1 | −100.1 | −100.0 | −96.4 | −96.4 | −91.8 | −91.7 | −97.7 | −97.7 | −88.5 | −88.4 | −99.4 | −99.4 |
| Aethiopinone (123) | −89.4 | −96.4 | −104.4 | −112.6 | −92.8 | −100.1 | −105.6 | −113.9 | −92.5 | −99.7 | −94.5 | −102.0 | −100.0 | −107.8 |
| Andrographolide (124) | −89.7 | −91.5 | −102.9 | −104.9 | −101.3 | −103.3 | −102.6 | −104.6 | −106.9 | −109.0 | −104.6 | −106.6 | −99.9 | −101.9 |
| Biflorin (126) | −92.0 | −99.4 | −102.1 | −110.4 | −93.6 | −101.1 | −101.0 | −109.2 | −102.0 | −110.2 | −100.3 | −108.4 | −95.5 | −103.3 |
| Continentalic acid (127) | −74.3 | −79.6 | −91.4 | −97.9 | −76.7 | −82.2 | −80.7 | −86.4 | −82.5 | −88.4 | −81.9 | −87.7 | −78.7 | −84.3 |
| Crinipellin A (129) | −79.7 | −82.9 | −96.3 | −100.1 | −99.9 | −103.9 | −82.0 | −85.3 | −92.6 | −96.3 | −84.8 | −88.2 | −94.9 | −98.7 |
| Cryptobeilic acid A (128) | −89.8 | −94.8 | −116.8 | −123.2 | −106.8 | −112.7 | −112.3 | −118.5 | −103.4 | −109.1 | −102.9 | −108.6 | −108.4 | −114.4 |
| Cryptobeilic acid C (130) | −95.2 | −95.8 | −124.6 | −125.5 | −105.0 | −105.7 | −122.0 | −122.8 | −116.1 | −116.9 | −125.2 | −126.0 | −113.5 | −114.2 |
| Cryptobeilic acid D (131) | −92.2 | −98.9 | −103.5 | −111.1 | −100.5 | −107.8 | −96.4 | −103.5 | −98.0 | −105.2 | −100.5 | −107.9 | −98.6 | −105.8 |
| Effusanin A (132) | −65.1 | −66.5 | −83.0 | −84.8 | −87.7 | −89.6 | −77.7 | −79.4 | −89.6 | −91.6 | −74.6 | −76.2 | −87.4 | −89.3 |
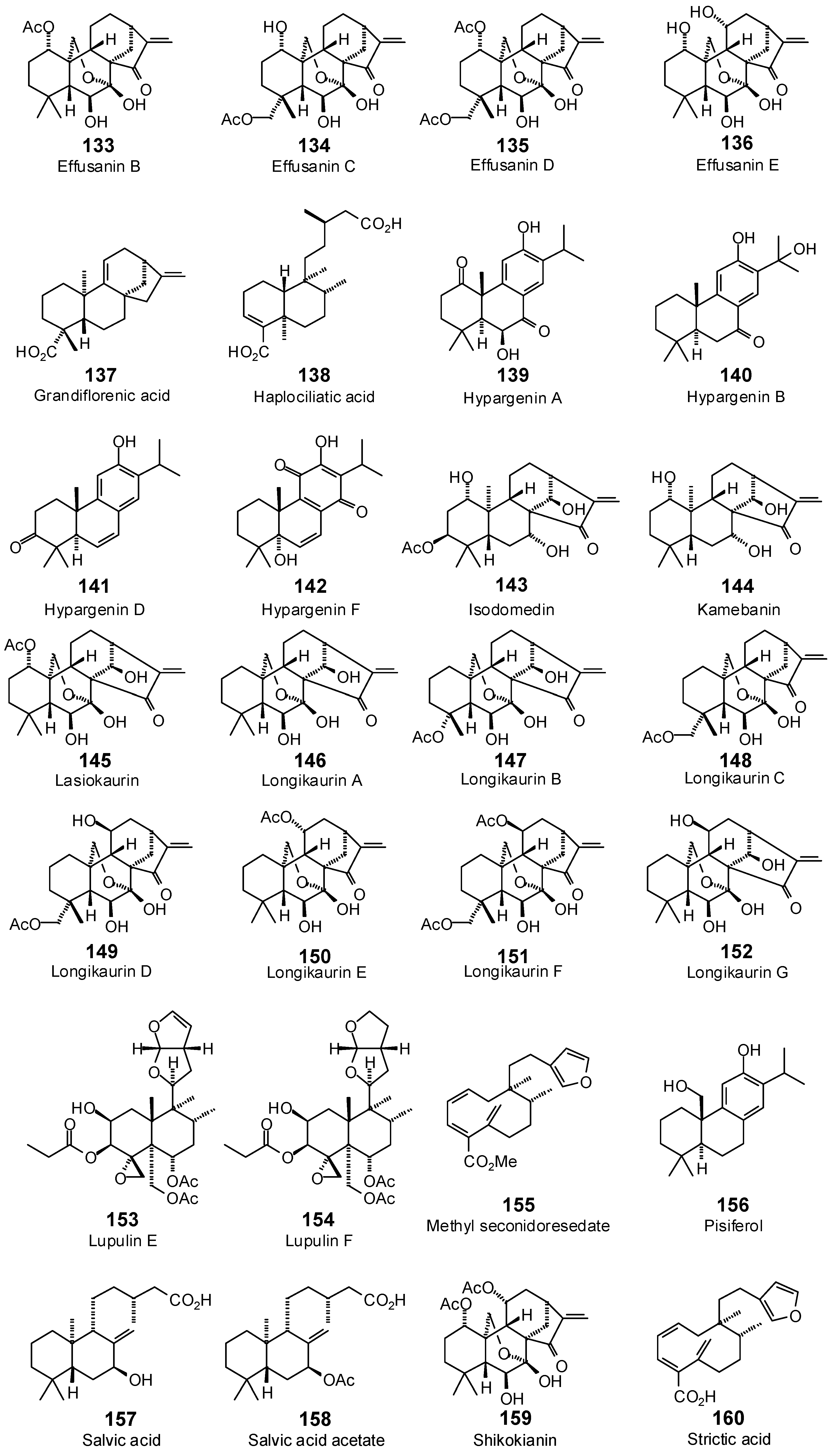
| Effusanin B (133) | −80.7 | −79.4 | −94.3 | −92.8 | −99.9 | −98.3 | −91.1 | −89.7 | −100.9 | −99.2 | −95.0 | −93.5 | −89.5 | −88.0 |
| Effusanin C (134) | −71.5 | −69.4 | −86.0 | −83.5 | −91.7 | −89.0 | −82.0 | −79.6 | −95.4 | −92.6 | −91.1 | −88.4 | −95.0 | −92.2 |
| Effusanin D (135) | −74.6 | −70.0 | −101.8 | −95.6 | −100.7 | −94.6 | −99.8 | −93.8 | −105.0 | −98.6 | −101.4 | −95.2 | −96.9 | −91.0 |
| Effusanin E (136) | −73.5 | −74.0 | −86.6 | −87.2 | −87.2 | −87.8 | −79.1 | −79.6 | −92.2 | −92.8 | −85.4 | −85.9 | −89.6 | −90.2 |
| Grandiflorenic acid (137) | −65.3 | −70.1 | −81.1 | −87.1 | −75.8 | −81.4 | −59.4 | −63.8 | −70.6 | −75.8 | −74.9 | −80.4 | −70.7 | −75.9 |
| Haplociliatic acid (138) | −92.3 | −95.4 | −104.2 | −107.8 | −102.0 | −105.4 | −95.0 | −98.2 | −103.9 | −107.4 | −109.6 | −113.3 | −100.6 | −103.9 |
| Hypargenin A (139) | −83.8 | −87.1 | −95.7 | −99.6 | −91.5 | −95.2 | −89.4 | −93.0 | −97.2 | −101.1 | −94.1 | −97.9 | −88.8 | −92.4 |
| Hypargenin B (140) | −81.8 | −86.3 | −90.3 | −95.3 | −81.9 | −86.4 | −84.3 | −88.9 | −94.4 | −99.6 | −87.4 | −92.2 | −83.5 | −88.1 |
| Hypargenin D (141) | −72.3 | −77.8 | −89.2 | −96.0 | −82.0 | −88.2 | −79.5 | −85.6 | −85.3 | −91.8 | −82.5 | −88.7 | −82.5 | −88.8 |
| Hypargenin F (142) | −71.0 | −73.9 | −86.1 | −89.5 | −72.4 | −75.3 | −79.9 | −83.1 | −79.8 | −83.0 | −83.3 | −86.6 | −73.2 | −76.1 |
| Isodomedin (143) | −66.4 | −65.2 | −94.5 | −92.8 | −95.8 | −94.1 | −78.1 | −76.7 | −98.8 | −97.1 | −98.2 | −96.4 | −76.7 | −75.3 |
| Kamebanin (144) | −72.7 | −75.3 | −95.5 | −98.9 | −81.2 | −84.1 | −74.3 | −77.0 | −101.5 | −105.2 | −79.3 | −82.2 | −74.2 | −76.9 |
| Lasiokaurin (145) | −82.0 | −79.5 | −101.0 | −97.9 | −100.5 | −97.4 | −84.3 | −81.7 | −99.4 | −96.3 | −91.4 | −88.6 | −70.6 | −68.4 |
| Longikaurin A (146) | −67.8 | −69.3 | −79.4 | −81.1 | −90.4 | −92.3 | −76.1 | −77.7 | −89.0 | −91.0 | −81.6 | −83.3 | −70.7 | −72.2 |
| Longikaurin B (147) | −63.0 | −61.1 | −83.9 | −81.5 | −93.2 | −90.4 | −78.1 | −75.8 | −94.0 | −91.3 | −89.0 | −86.4 | −69.9 | −67.8 |
| Longikaurin C (148) | −27.9 | −27.4 | −80.2 | −78.9 | −88.1 | −86.7 | −79.2 | −78.0 | −91.0 | −89.5 | −90.2 | −88.7 | −89.2 | −87.7 |
| Longikaurin D (149) | −51.3 | −49.8 | −84.3 | −81.8 | −96.0 | −93.2 | −84.9 | −82.4 | −95.8 | −93.0 | −91.7 | −89.0 | −96.8 | −94.0 |
| Longikaurin E (150) | −75.8 | −77.4 | −84.0 | −85.8 | −85.9 | −87.8 | −82.1 | −83.9 | −88.9 | −90.9 | −79.3 | −81.0 | −84.4 | −86.3 |
| Longikaurin F (151) | −88.2 | −82.9 | −98.6 | −92.7 | −111.3 | −104.6 | −98.8 | −92.8 | −114.1 | −107.1 | −102.9 | −96.7 | −107.7 | −101.1 |
| Longikaurin G (152) | −74.6 | −75.1 | −81.2 | −81.7 | −96.0 | −96.6 | −80.8 | −81.3 | −91.5 | −92.1 | −87.1 | −87.6 | −75.8 | −76.3 |
| Lupulin E (153) | −97.3 | −86.8 | −117.1 | −104.5 | −112.5 | −100.4 | −108.4 | −96.7 | −99.1 | −88.5 | −102.7 | −91.7 | −119.9 | −107.0 |
| Lupulin F (154) | −87.2 | −77.8 | −116.1 | −103.5 | −115.2 | −102.7 | −99.8 | −88.9 | −103.0 | −91.8 | −101.8 | −90.7 | −116.3 | −103.7 |
| Methyl seconidoresedate (155) | −89.3 | −93.0 | −106.5 | −110.9 | −97.2 | −101.3 | −109.3 | −113.9 | −97.5 | −101.6 | −101.4 | −105.7 | −99.7 | −103.9 |
| Pisiferol (156) | −70.5 | −75.5 | −86.2 | −92.4 | −86.5 | −92.6 | −75.9 | −81.3 | −93.3 | −100.0 | −88.0 | −94.2 | −82.1 | −87.9 |
| Salvic acid (157) | −85.1 | −89.2 | −101.6 | −106.5 | −89.6 | −93.9 | −105.0 | −110.1 | −97.3 | −102.0 | −95.9 | −100.5 | −97.3 | −102.0 |
| Salvic acid acetate (158) | −92.3 | −92.9 | −114.4 | −115.1 | −91.3 | −91.8 | −104.1 | −104.7 | −103.3 | −104.0 | −102.8 | −103.5 | −103.8 | −104.5 |
| Shikokianin (159) | −63.1 | −59.2 | −104.1 | −97.8 | −93.7 | −88.0 | −93.9 | −88.2 | −93.0 | −87.3 | −97.0 | −91.1 | −84.8 | −79.7 |
| Strictic acid (160) | −88.5 | −93.6 | −103.1 | −109.0 | −94.5 | −99.9 | −106.8 | −112.9 | −97.2 | −102.8 | −98.6 | −104.3 | −96.5 | −102.0 |
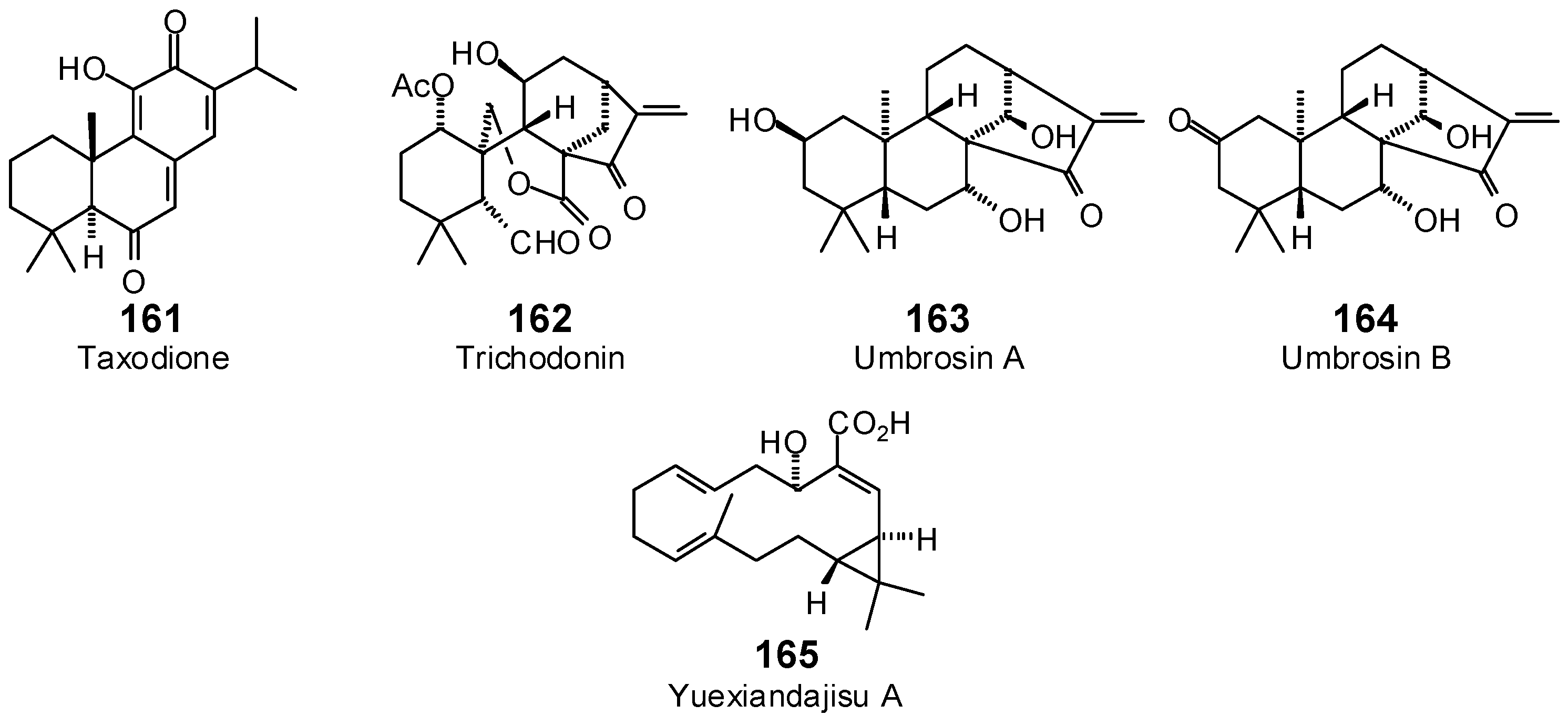
| Taxodione (161) | −69.8 | −73.8 | −92.1 | −97.4 | −87.3 | −92.3 | −76.9 | −81.3 | −92.9 | −98.2 | −90.8 | −96.0 | −74.2 | −78.4 |
| Trichodonin (162) | −80.6 | −78.3 | −91.6 | −89.1 | −67.4 | −65.5 | −82.0 | −79.7 | −80.1 | −77.9 | −91.9 | −89.3 | −84.1 | −81.7 |
| Umbrosin A (163) | −74.7 | −77.4 | −93.3 | −96.6 | −83.8 | −86.8 | −78.7 | −81.5 | −95.2 | −98.7 | −85.8 | −88.8 | −76.2 | −78.9 |
| Umbrosin B (164) | −76.3 | −79.2 | −88.8 | −92.2 | −86.7 | −90.0 | −78.3 | −81.2 | −86.6 | −89.9 | −86.6 | −89.9 | −73.4 | −76.2 |
| Yuexiandajisu A (165) | −104.3 | −109.8 | −114.0 | −120.0 | −101.1 | −106.5 | −113.3 | −119.3 | −109.8 | −115.6 | −98.4 | −103.6 | −106.3 | −111.9 |
| Triterpenoids | ||||||||||||||
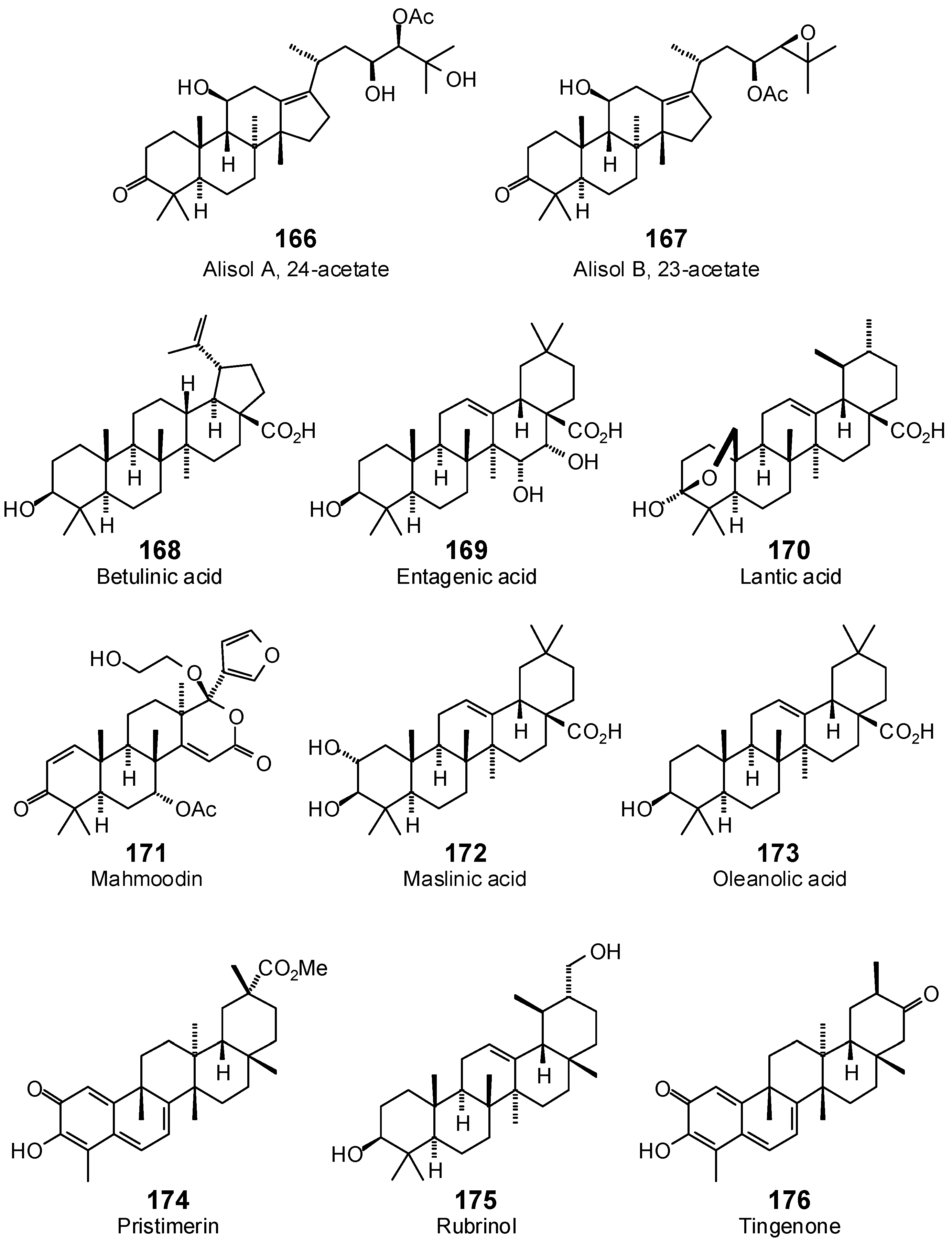
| Alisol A 24-acetate (166) | −88.7 | −78.6 | −96.5 | −85.6 | −104.7 | −92.9 | −90.7 | −80.5 | −105.4 | −93.5 | −100.4 | −89.1 | −103.2 | −91.6 |
| Alisol B 23-acetate (167) | −63.6 | −57.0 | −112.6 | −101.0 | −107.2 | −96.2 | −81.1 | −72.8 | −111.0 | −99.6 | −98.1 | −88.0 | −113.2 | −101.6 |
| Betulinic acid (168) | −98.3 | −91.8 | −91.4 | −85.3 | −113.0 | −105.5 | −88.8 | −82.9 | −105.7 | −98.7 | −94.3 | −88.0 | −102.3 | −95.5 |
| Entagenic acid (169) | −56.4 | −51.5 | −101.0 | −92.2 | −86.3 | −78.8 | −69.3 | −63.3 | −85.1 | −77.7 | −79.1 | −72.2 | −87.7 | −80.0 |
| Lantic acid (170) | −38.1 | −35.2 | −79.1 | −73.1 | −82.2 | −76.0 | −74.4 | −68.8 | −80.1 | −74.0 | −83.1 | −76.8 | −88.8 | −82.1 |
| Mahmoodin (171) | −81.3 | −72.4 | −97.6 | −86.9 | −84.8 | −75.5 | −87.8 | −78.1 | −86.6 | −77.1 | −76.8 | −68.3 | −99.8 | −88.9 |
| Maslinic acid (172) | −43.6 | −40.3 | −98.9 | −91.3 | −87.0 | −80.3 | −80.1 | −73.9 | −84.5 | −78.0 | −82.6 | −76.3 | −92.1 | −85.0 |
| Oleanolic acid (173) | −70.3 | −65.7 | −95.2 | −88.9 | −81.8 | −76.4 | −77.9 | −72.7 | −86.4 | −80.7 | −82.8 | −77.3 | −88.4 | −82.6 |
| Pristimerin (174) | −81.9 | −76.0 | −103.5 | −96.1 | −98.1 | −91.0 | −89.9 | −83.5 | −96.0 | −89.1 | −88.3 | −81.9 | −102.8 | −95.5 |
| Rubrinol (175) | −70.8 | −66.8 | −88.0 | −83.0 | −82.0 | −77.4 | −79.6 | −75.1 | −87.1 | −82.2 | −86.6 | −81.7 | −95.0 | −89.6 |
| Tingenone (176) | −71.6 | −68.7 | −92.0 | −88.2 | −83.9 | −80.5 | −80.6 | −77.3 | −83.2 | −79.8 | −87.3 | −83.8 | −94.1 | −90.3 |
| Chalcones | ||||||||||||||
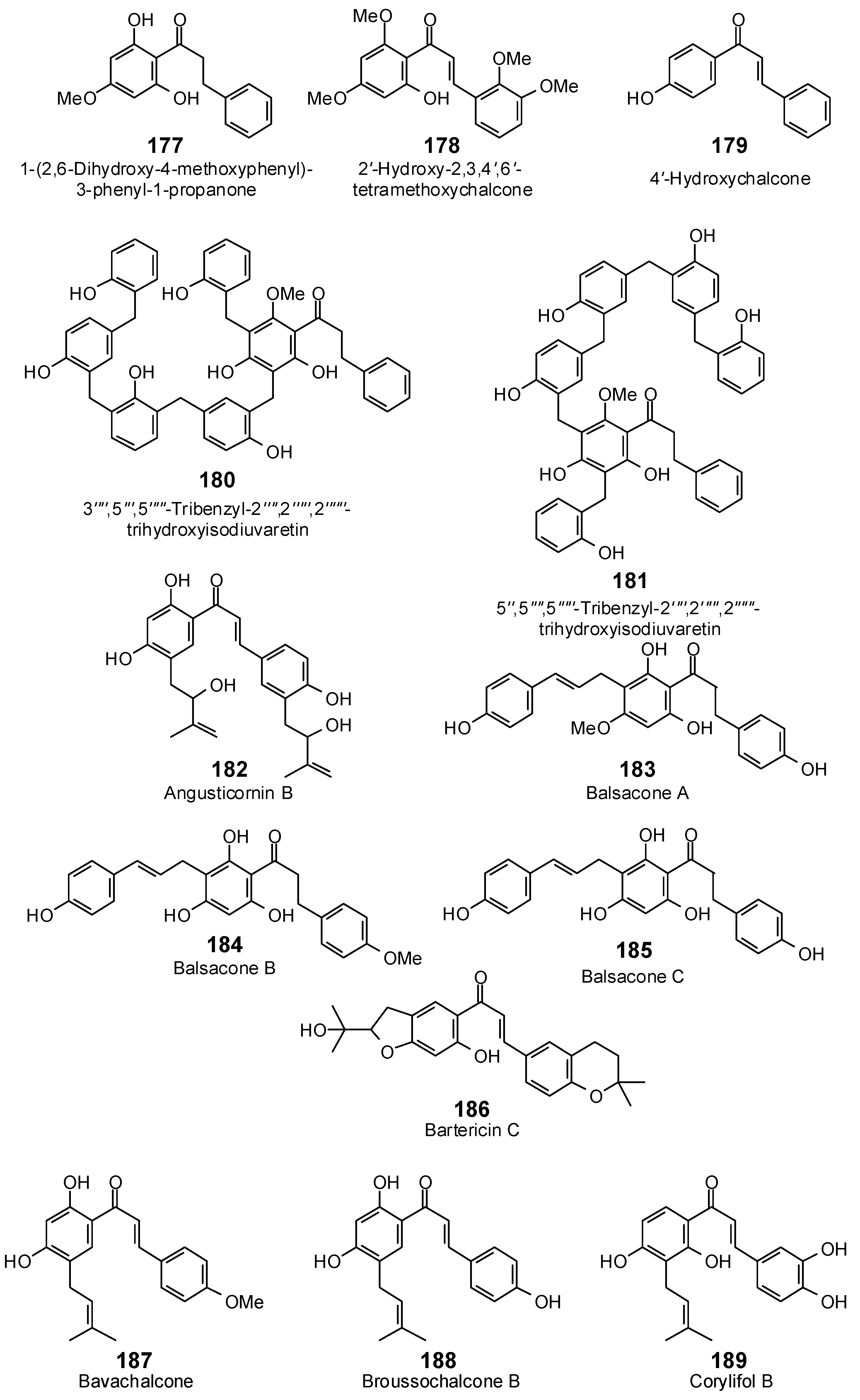
| 1-(2,6-Dihydroxy-4-methoxyphenyl)-3-phenyl-1-propanone (177) | −92.7 | −102.8 | −107.4 | −119.1 | −93.8 | −104.1 | −101.8 | −112.9 | −100.6 | −111.6 | −98.9 | −109.7 | −105.3 | −116.8 |
| 2′-Hydroxy-2,3,4′,6′-tetramethoxychalcone (178) | −91.9 | −94.2 | −110.7 | −113.6 | −92.8 | −95.2 | −108.6 | −111.4 | −103.6 | −106.3 | −99.7 | −102.2 | −117.5 | −120.5 |
| 3′′′′,5′′′,5′′′′′-Tribenzyl-2′′′′,2′′′′′,2′′′′′′-trihydroxyisodiuvaretin (180) | −41.6 | −32.2 | −114.0 | −88.2 | −141.1 | −109.1 | −129.7 | −100.3 | −147.7 | −114.2 | −126.3 | −97.7 | −145.6 | −112.6 |
| 4′-Hydroxychalcone (179) | −78.9 | −93.3 | −94.2 | −111.5 | −81.1 | −96.0 | −86.5 | −102.3 | −90.3 | −106.9 | −87.3 | −103.4 | −89.4 | −105.9 |
| 5″,5′′′′,5′′′′′-Tribenzyl-2′′′′,2′′′′′,2′′′′′′-trihydroxyisodiuvaretin (181) | −100.9 | −78.0 | −114.7 | −88.7 | −152.4 | −117.9 | −164.0 | −126.9 | −145.6 | −112.6 | −136.0 | −105.2 | −156.9 | −121.4 |
| Angusticornin B (182) | −117.5 | −112.4 | −143.5 | −137.2 | −134.4 | −128.6 | −134.7 | −128.8 | −129.3 | −123.7 | −131.4 | −125.7 | −126.7 | −121.2 |
| Balsacone A (183) | −106.9 | −102.6 | −127.5 | −122.4 | −122.3 | −117.4 | −122.4 | −117.5 | −127.6 | −122.4 | −121.9 | −117.0 | −124.1 | −119.1 |
| Balsacone B (184) | −109.2 | −104.8 | −129.0 | −123.8 | −123.0 | −118.0 | −124.7 | −119.7 | −128.3 | −123.2 | −132.4 | −127.0 | −119.9 | −115.1 |
| Balsacone C (185) | −109.0 | −107.3 | −124.2 | −122.1 | −121.4 | −119.5 | −120.0 | −118.1 | −127.4 | −125.3 | −132.3 | −130.1 | −122.4 | −120.4 |
| Bartericin C (186) | −75.7 | −73.3 | −123.3 | −119.4 | −109.6 | −106.2 | −107.7 | −104.3 | −102.2 | −99.0 | −105.6 | −102.3 | −112.7 | −109.2 |
| Bavachalcone (187) | −105.6 | −109.0 | −121.5 | −125.3 | −114.0 | −117.6 | −120.0 | −123.9 | −116.0 | −119.7 | −112.3 | −115.9 | −116.9 | −120.6 |
| Broussochalcone B (188) | −102.3 | −107.1 | −117.5 | −122.9 | −109.7 | −114.8 | −119.4 | −124.9 | −103.2 | −108.0 | −109.7 | −114.8 | −114.5 | −119.8 |
| Corylifol B (189) | −106.2 | −109.3 | −121.0 | −124.6 | −112.9 | −116.2 | −123.3 | −127.0 | −113.7 | −117.1 | −117.7 | −121.2 | −119.7 | −123.3 |
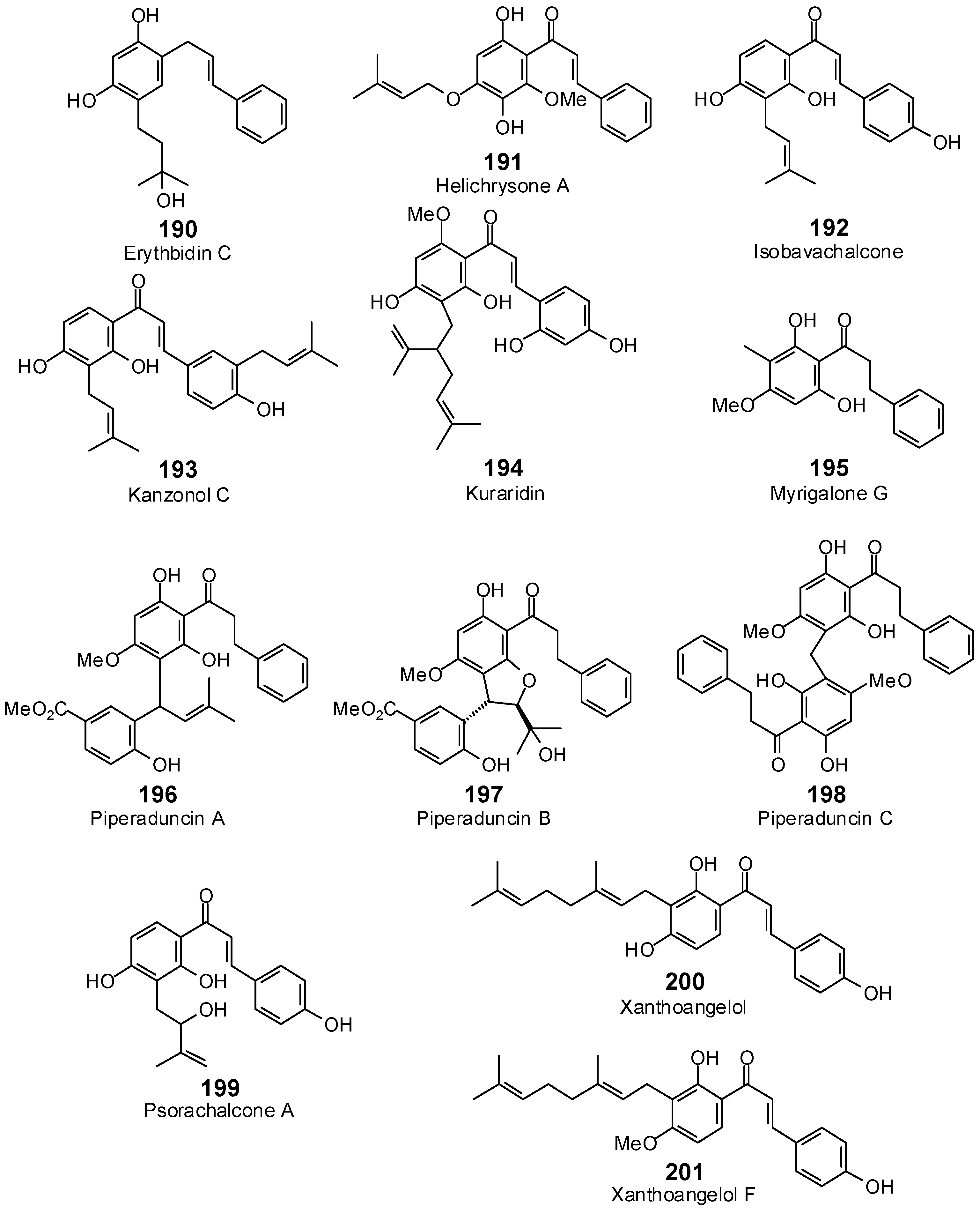
| Erythbidin C (190) | −98.5 | −104.4 | −119.5 | −126.6 | −103.3 | −109.5 | −115.5 | −122.4 | −108.7 | −115.1 | −113.0 | −119.7 | −112.2 | −118.9 |
| Helichrysone A (191) | −91.6 | −93.1 | −117.8 | −119.7 | −99.4 | −101.0 | −112.4 | −114.2 | −109.2 | −110.9 | −102.7 | −104.3 | −104.9 | −106.6 |
| Isobavachalcone (192) | −104.3 | −109.2 | −117.8 | −123.2 | −111.5 | −116.7 | −114.0 | −119.3 | −112.4 | −117.7 | −113.6 | −118.9 | −115.8 | −121.2 |
| Kanzonol C (193) | −110.3 | −108.4 | −133.3 | −130.9 | −125.2 | −123.0 | −128.4 | −126.1 | −130.1 | −127.8 | −135.9 | −133.4 | −131.8 | −129.4 |
| Kuraridin (194) | −117.2 | −110.9 | −124.0 | −117.3 | −105.7 | −100.0 | −127.9 | −121.0 | −121.1 | −114.6 | −111.5 | −105.6 | −136.6 | −129.2 |
| Myrigalone G (195) | −89.4 | −97.5 | −105.7 | −115.3 | −96.9 | −105.7 | −105.5 | −115.0 | −104.4 | −113.9 | −99.7 | −108.8 | −104.9 | −114.5 |
| Piperaduncin A (196) | −113.3 | −103.3 | −141.0 | −128.5 | −121.0 | −110.3 | −130.8 | −119.2 | −132.2 | −120.5 | −120.8 | −110.1 | −131.7 | −120.1 |
| Piperaduncin B (197) | −117.9 | −106.3 | −139.3 | −125.6 | −112.0 | −101.0 | −141.8 | −127.9 | −144.3 | −130.2 | −134.3 | −121.2 | −133.6 | −120.5 |
| Piperaduncin C (198) | −110.2 | −96.3 | −155.9 | −136.2 | −104.1 | −91.0 | −149.2 | −130.4 | −127.2 | −111.2 | −131.8 | −115.2 | −149.4 | −130.6 |
| Psorachalcone A (199) | −102.2 | −105.2 | −118.9 | −122.4 | −107.2 | −110.4 | −116.8 | −120.3 | −112.0 | −115.3 | −114.5 | −117.9 | −116.7 | −120.2 |
| Xanthoangelol (200) | −117.6 | −115.4 | −135.2 | −132.8 | −131.9 | −129.5 | −131.3 | −129.0 | −132.1 | −129.7 | −130.6 | −128.3 | −132.6 | −130.2 |
| Xanthoangelol F (201) | −115.9 | −112.5 | −129.8 | −126.0 | −135.6 | −131.6 | −127.0 | −123.3 | −126.2 | −122.5 | −126.5 | −122.8 | −127.5 | −123.7 |
| Flavonoids | ||||||||||||||
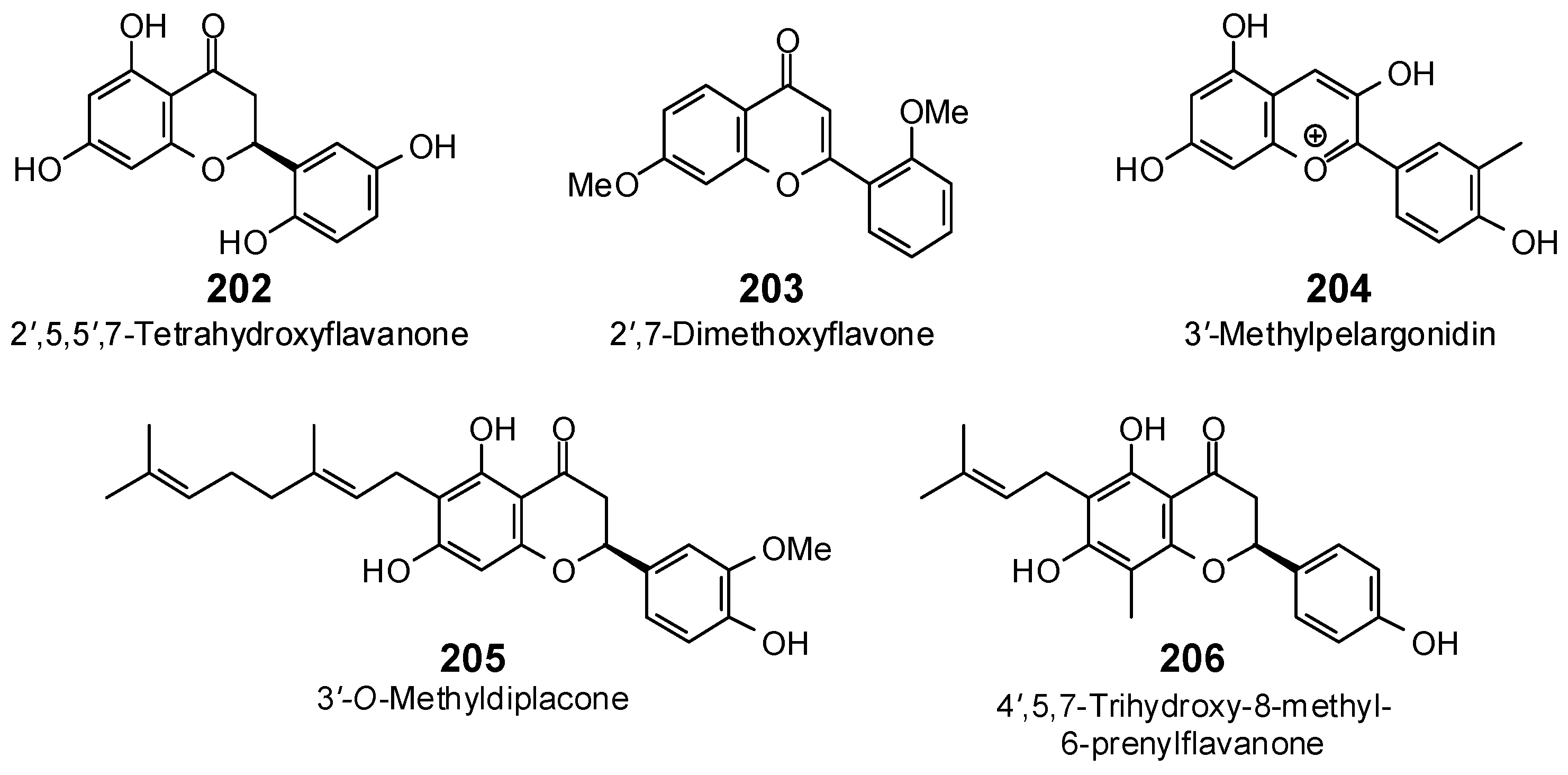
| 2′,5,5′,7-Tetrahydroxyflavanone (202) | −81.2 | −88.4 | −95.9 | −104.4 | −84.1 | −91.5 | −95.4 | −103.9 | −87.4 | −95.2 | −87.7 | −95.5 | −88.2 | −96.0 |
| 2′,7-Dimethoxyflavone (203) | −84.2 | −92.3 | −94.7 | −103.8 | −87.5 | −95.9 | −95.8 | −105.0 | −98.8 | −108.3 | −95.9 | −105.1 | −94.8 | −103.8 |
| 3′′′′-(2-Hydroxybenzyl)-isouvarinol (218) | −77.8 | −63.6 | −130.5 | −106.6 | −109.5 | −89.5 | −133.1 | −108.8 | −123.5 | −100.9 | −147.9 | −120.9 | −149.8 | −122.4 |
| 3′′′′-(2-Hydroxybenzyl)uvarinol (217) | −81.3 | −66.5 | −129.1 | −105.5 | −135.5 | −110.7 | −141.3 | −115.5 | −117.5 | −96.0 | −131.1 | −107.2 | −153.7 | −125.6 |
| 3′-Methylpelargonidin (204) | −90.7 | −99.1 | −98.9 | −108.0 | −87.4 | −95.5 | −101.5 | −110.8 | −92.0 | −100.5 | −98.0 | −107.1 | −92.4 | −100.9 |
| 3′-O-Methyldiplacone (205) | −67.4 | −63.8 | −133.5 | −126.4 | −120.3 | −113.9 | −122.7 | −116.1 | −111.4 | −105.5 | −109.3 | −103.4 | −128.9 | −122.0 |
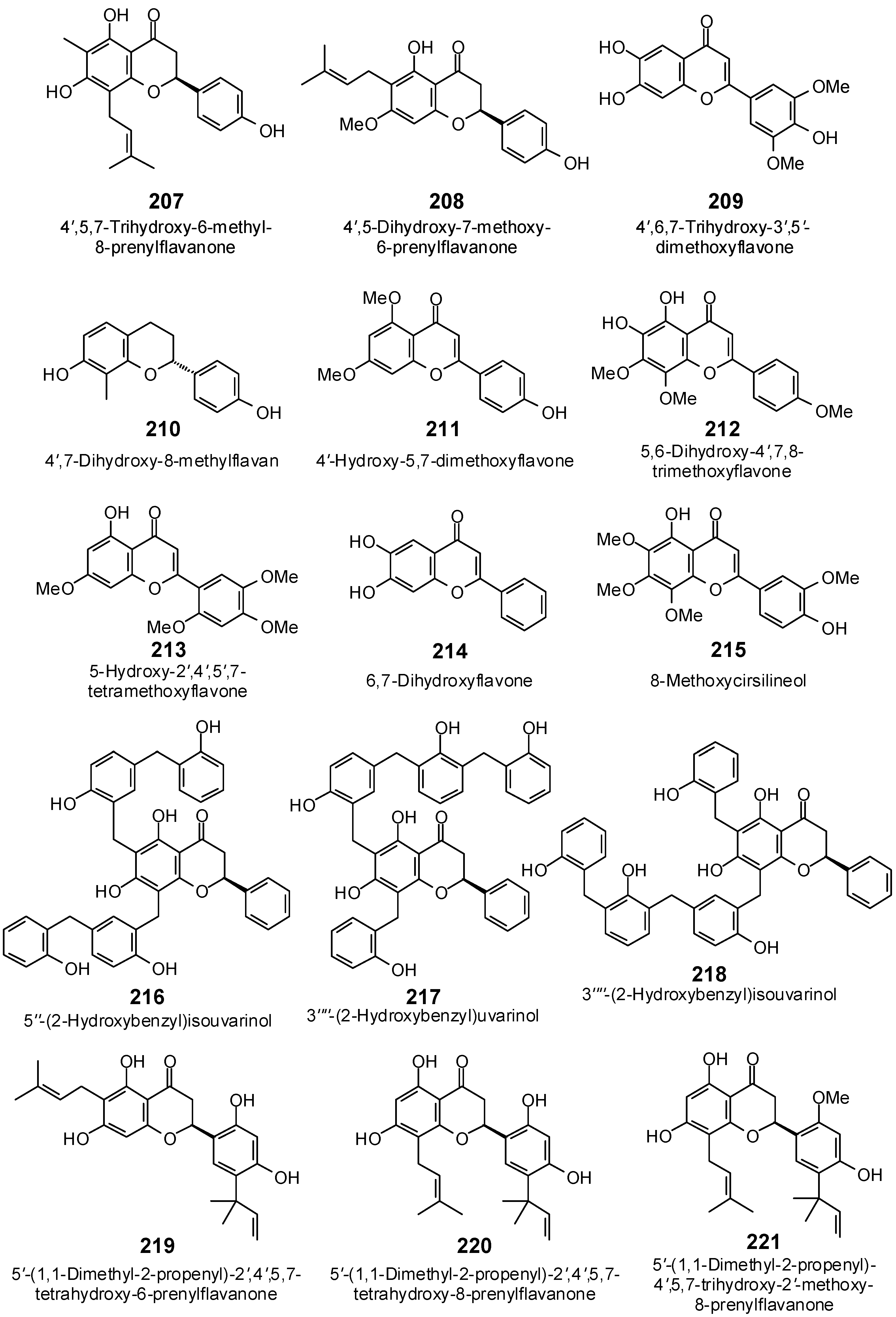
| 4′,5,7-Trihydroxy-6-methyl-8-prenylflavanone (207) | −87.9 | −89.3 | −102.2 | −103.8 | −107.1 | −108.8 | −99.8 | −101.4 | −98.9 | −100.5 | −94.2 | −95.7 | −105.2 | −106.9 |
| 4′,5,7-Trihydroxy-8-methyl-6-prenylflavanone (206) | −84.8 | −86.2 | −106.0 | −107.7 | −94.0 | −95.5 | −105.6 | −107.3 | −96.9 | −98.5 | −88.8 | −90.2 | −105.6 | −107.3 |
| 4′,5-Dihydroxy-7-methoxy-6-prenylflavanone (208) | −92.3 | −93.8 | −111.3 | −113.1 | −96.5 | −98.0 | −112.1 | −113.9 | −96.6 | −98.2 | −91.5 | −93.0 | −106.4 | −108.1 |
| 4′,6,7-Trihydroxy-3′,5′-dimethoxyflavone (209) | −90.8 | −94.5 | −109.8 | −114.2 | −99.9 | −103.9 | −104.2 | −108.4 | −97.9 | −101.9 | −97.9 | −101.8 | −102.6 | −106.7 |
| 4′,7-Dihydroxy-8-methylflavan (210) | −80.2 | −90.7 | −87.3 | −98.8 | −80.9 | −91.5 | −87.8 | −99.4 | −81.8 | −92.6 | −85.8 | −97.1 | −86.7 | −98.1 |
| 4′-Hydroxy-5,7-dimethoxy-flavone (211) | −83.6 | −90.0 | −98.6 | −106.1 | −96.0 | −103.3 | −91.5 | −98.4 | −88.5 | −95.3 | −84.9 | −91.4 | −91.7 | −98.7 |
| 5″-(2-Hydroxybenzyl)-isouvarinol (216) | −113.5 | −92.8 | −143.5 | −117.3 | −132.9 | −108.6 | −137.9 | −112.7 | −153.3 | −125.3 | −134.9 | −110.2 | −156.6 | −128.0 |
| 5′-(1,1-Dimethyl-2-propenyl)-2′,4′,5,7-tetrahydroxy-6-prenyl-flavanone (219) | −102.7 | −98.3 | −122.9 | −117.6 | −101.9 | −97.5 | −132.2 | −126.5 | −111.6 | −106.8 | −99.5 | −95.2 | −117.4 | −112.3 |
| 5′-(1,1-Dimethyl-2-propenyl)-2′,4′,5,7-tetrahydroxy-8-prenylflavanone (220) | −106.9 | −102.3 | −131.3 | −125.6 | −112.7 | −107.8 | −128.4 | −122.8 | −119.4 | −114.2 | −115.9 | −110.8 | −124.1 | −118.7 |
| 5′-(1,1-Dimethyl-2-propenyl)-4′,5,7-trihydroxy-2′-methoxy-8-prenylflavanone (221) | −103.2 | −97.6 | −132.8 | −125.7 | −122.3 | −115.7 | −128.4 | −121.6 | −111.4 | −105.4 | −106.7 | −101.0 | −124.5 | −117.8 |
| 5,6-Dihydroxy-4′,7,8-trimethoxy-flavone (212) | −95.2 | −97.6 | −110.1 | −113.0 | −96.1 | −98.6 | −101.2 | −103.8 | −100.9 | −103.5 | −102.4 | −105.0 | −103.2 | −105.8 |
| 5-Hydroxy-2′,4′,5′,7-Tetra-methoxyflavone (213) | −99.4 | −100.6 | −110.2 | −111.5 | −105.0 | −106.3 | −112.5 | −113.9 | −95.6 | −96.8 | −102.3 | −103.5 | −104.1 | −105.4 |
| 6,7-Dihydroxyflavone (214) | −76.1 | −86.3 | −87.5 | −99.3 | −80.8 | −91.7 | −92.0 | −104.4 | −88.6 | −100.6 | −85.4 | −97.0 | −87.9 | −99.8 |
| 8-Methoxycirsilineol (215) | −93.3 | −93.0 | −117.9 | −117.6 | −103.7 | −103.5 | −118.0 | −117.7 | −99.6 | −99.4 | −96.4 | −96.2 | −107.6 | −107.3 |
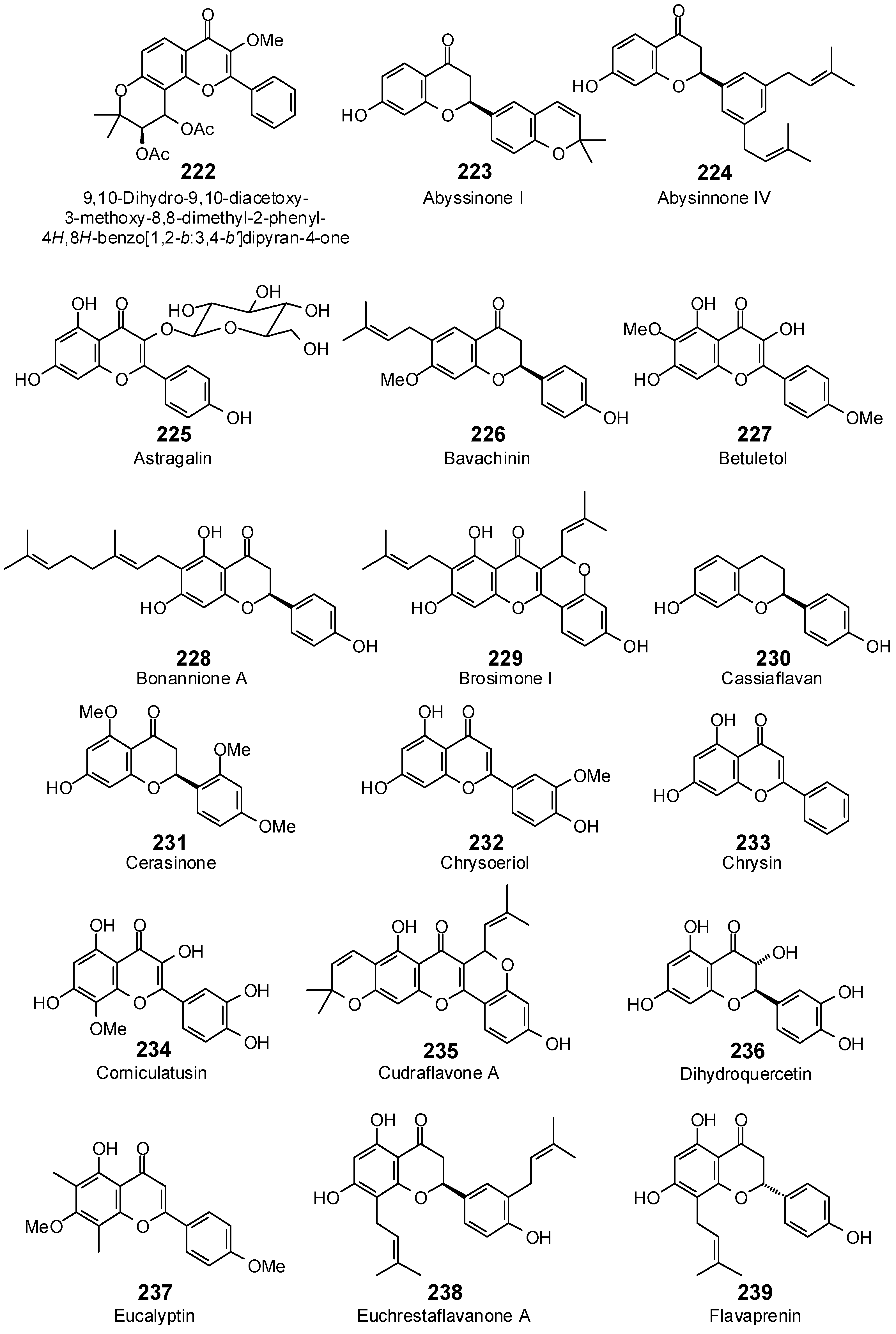
| 9,10-Dihydro-9,10-diacetoxy-3-methoxy-8,8-dimethyl-2-phenyl-4H,8H-benzo[1,2-b:3,4-b′]-dipyran-4-one (222) | −97.9 | −91.7 | −102.6 | −96.1 | −101.4 | −95.0 | −100.5 | −94.2 | −99.9 | −93.6 | −97.2 | −91.0 | −98.6 | −92.4 |
| Abyssinone I (223) | −85.5 | −89.6 | −108.9 | −114.2 | −87.8 | −92.0 | −98.8 | −103.6 | −86.9 | −91.1 | −87.4 | −91.7 | −101.1 | −106.0 |
| Abyssinone IV (224) | −98.6 | −96.8 | −125.9 | −123.6 | −107.4 | −105.5 | −117.3 | −115.2 | −113.0 | −111.0 | −116.1 | −114.1 | −120.3 | −118.2 |
| Astragalin (225) | −100.1 | −94.1 | −117.0 | −109.9 | −95.4 | −89.6 | −112.8 | −106.0 | −118.5 | −111.3 | −105.7 | −99.3 | −116.3 | −109.3 |
| Bavachinin (226) | −97.3 | −100.4 | −111.8 | −115.4 | −103.4 | −106.7 | −116.3 | −120.0 | −101.5 | −104.8 | −101.5 | −104.7 | −112.9 | −116.5 |
| Betuletol (227) | −82.6 | −85.9 | −102.6 | −106.7 | −82.1 | −85.4 | −98.4 | −102.4 | −87.1 | −90.6 | −90.9 | −94.5 | −103.1 | −107.3 |
| Bonannione A (228) | −81.3 | −78.8 | −129.0 | −125.0 | −121.6 | −117.8 | −120.4 | −116.7 | −112.2 | −108.7 | −110.6 | −107.2 | −116.7 | −113.1 |
| Brosimone I (229) | −90.4 | −86.7 | −114.0 | −109.4 | −100.1 | −96.0 | −110.2 | −105.7 | −100.8 | −96.8 | −97.9 | −94.0 | −102.0 | −97.9 |
| Cassiaflavan (230) | −76.1 | −87.7 | −85.7 | −98.9 | −75.4 | −87.0 | −84.3 | −97.2 | −76.6 | −88.3 | −82.1 | −94.7 | −83.3 | −96.1 |
| Cerasinone (231) | −83.4 | −86.7 | −101.3 | −105.4 | −91.2 | −94.8 | −106.2 | −110.4 | −88.8 | −92.3 | −95.3 | −99.2 | −99.4 | −103.4 |
| Chrysin (233) | −79.0 | −89.6 | −85.7 | −97.3 | −81.5 | −92.5 | −86.6 | −98.2 | −82.0 | −93.1 | −86.4 | −98.1 | −85.6 | −97.2 |
| Chrysoeriol (232) | −86.0 | −92.4 | −101.0 | −108.5 | −92.1 | −98.9 | −101.8 | −109.3 | −90.1 | −96.7 | −97.6 | −104.8 | −98.7 | −106.0 |
| Corniculatusin (234) | −95.1 | −98.8 | −105.0 | −109.0 | −103.0 | −106.9 | −109.1 | −113.3 | −103.6 | −107.6 | −97.5 | −101.2 | −100.0 | −103.8 |
| Cudraflavone A (235) | −85.8 | −82.4 | −108.6 | −104.4 | −97.2 | −93.5 | −100.2 | −96.3 | −96.7 | −93.0 | −85.7 | −82.4 | −100.4 | −96.5 |
| Dihydroquercetin (236) | −84.1 | −89.9 | −98.0 | −104.8 | −75.8 | −81.1 | −98.5 | −105.3 | −85.4 | −91.3 | −88.2 | −94.3 | −87.9 | −93.9 |
| Eucalyptin (237) | −87.4 | −91.3 | −98.6 | −103.0 | −90.4 | −94.4 | −95.8 | −100.0 | −93.4 | −97.5 | −94.0 | −98.2 | −101.9 | −106.4 |
| Euchrestaflavanone A (238) | −114.5 | −111.0 | −126.3 | −122.4 | −118.2 | −114.6 | −130.4 | −126.3 | −121.4 | −117.6 | −128.0 | −124.1 | −126.9 | −123.0 |
| Flavaprenin (239) | −99.0 | −101.9 | −101.7 | −104.7 | −108.9 | −112.2 | −107.6 | −110.8 | −103.2 | −106.3 | −104.9 | −108.0 | −112.7 | −116.1 |
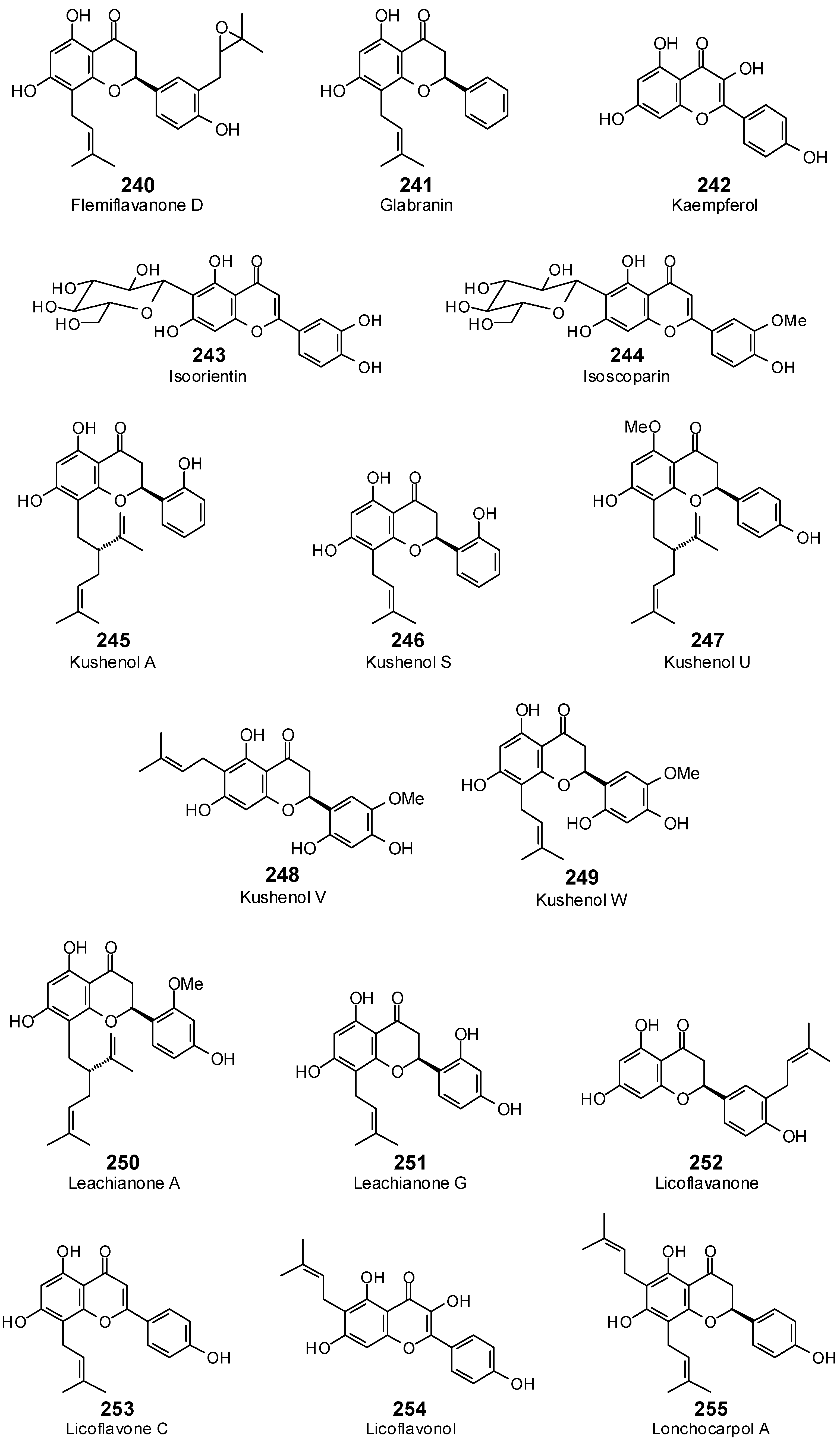
| Flemiflavanone D (240) | −104.6 | −100.0 | −131.3 | −125.6 | −118.9 | −113.7 | −120.0 | −114.8 | −117.0 | −111.9 | −111.1 | −106.3 | −129.3 | −123.7 |
| Glabranin (241) | −94.8 | −99.2 | −102.6 | −107.4 | −99.2 | −103.8 | −103.5 | −108.3 | −101.9 | −106.6 | −102.7 | −107.5 | −107.0 | −112.0 |
| Isoorientin (243) | −81.8 | −76.8 | −111.7 | −104.9 | −103.4 | −97.1 | −107.0 | −100.5 | −101.9 | −95.7 | −102.1 | −95.9 | −117.2 | −110.1 |
| Isoscoparin (244) | −78.4 | −72.9 | −113.6 | −105.7 | −113.4 | −105.5 | −115.2 | −107.1 | −108.9 | −101.2 | −106.5 | −99.0 | −123.0 | −114.4 |
| Kaempferol (242) | −83.1 | −90.7 | −94.9 | −103.6 | −86.6 | −94.5 | −90.3 | −98.5 | −84.1 | −91.8 | −86.4 | −94.3 | −89.4 | −97.5 |
| Kushenol A (245) | −105.3 | −102.1 | −120.0 | −116.3 | −113.1 | −109.6 | −117.5 | −113.9 | −116.8 | −113.2 | −115.6 | −112.0 | −110.7 | −107.3 |
| Kushenol S (246) | −94.7 | −97.6 | −103.2 | −106.3 | −100.8 | −103.8 | −105.3 | −108.4 | −100.4 | −103.4 | −105.1 | −108.2 | −104.7 | −107.8 |
| Kushenol U (247) | −85.9 | −82.3 | −121.5 | −116.5 | −104.7 | −100.3 | −113.4 | −108.7 | −112.9 | −108.2 | −105.5 | −101.0 | −112.1 | −107.4 |
| Kushenol V (248) | −99.4 | −96.9 | −116.3 | −113.4 | −98.2 | −95.8 | −126.9 | −123.7 | −104.5 | −101.9 | −103.8 | −101.2 | −113.7 | −110.9 |
| Kushenol W (249) | −102.6 | −101.3 | −120.6 | −119.0 | −109.9 | −108.5 | −120.6 | −119.1 | −111.3 | −109.8 | −114.7 | −113.2 | −114.4 | −112.9 |
| Leachianone A (250) | −108.8 | −102.9 | −121.2 | −114.7 | −119.4 | −113.0 | −121.1 | −114.6 | −117.1 | −110.9 | −114.7 | −108.6 | −124.2 | −117.6 |
| Leachianone G (251) | −99.4 | −100.8 | −104.5 | −106.0 | −111.5 | −113.1 | −106.6 | −108.1 | −103.9 | −105.4 | −105.3 | −106.8 | −109.7 | −111.3 |
| Licoflavanone (252) | −94.2 | −97.0 | −111.9 | −115.2 | −107.2 | −110.4 | −111.1 | −114.4 | −99.1 | −102.0 | −106.2 | −109.4 | −107.3 | −110.5 |
| Licoflavone C (253) | −100.9 | −104.1 | −105.3 | −108.7 | −109.0 | −112.5 | −106.3 | −109.7 | −105.5 | −108.9 | −107.5 | −110.9 | −109.4 | −112.9 |
| Licoflavonol (254) | −85.6 | −86.9 | −113.1 | −114.9 | −96.7 | −98.3 | −113.6 | −115.5 | −98.0 | −99.6 | −95.6 | −97.2 | −104.9 | −106.6 |
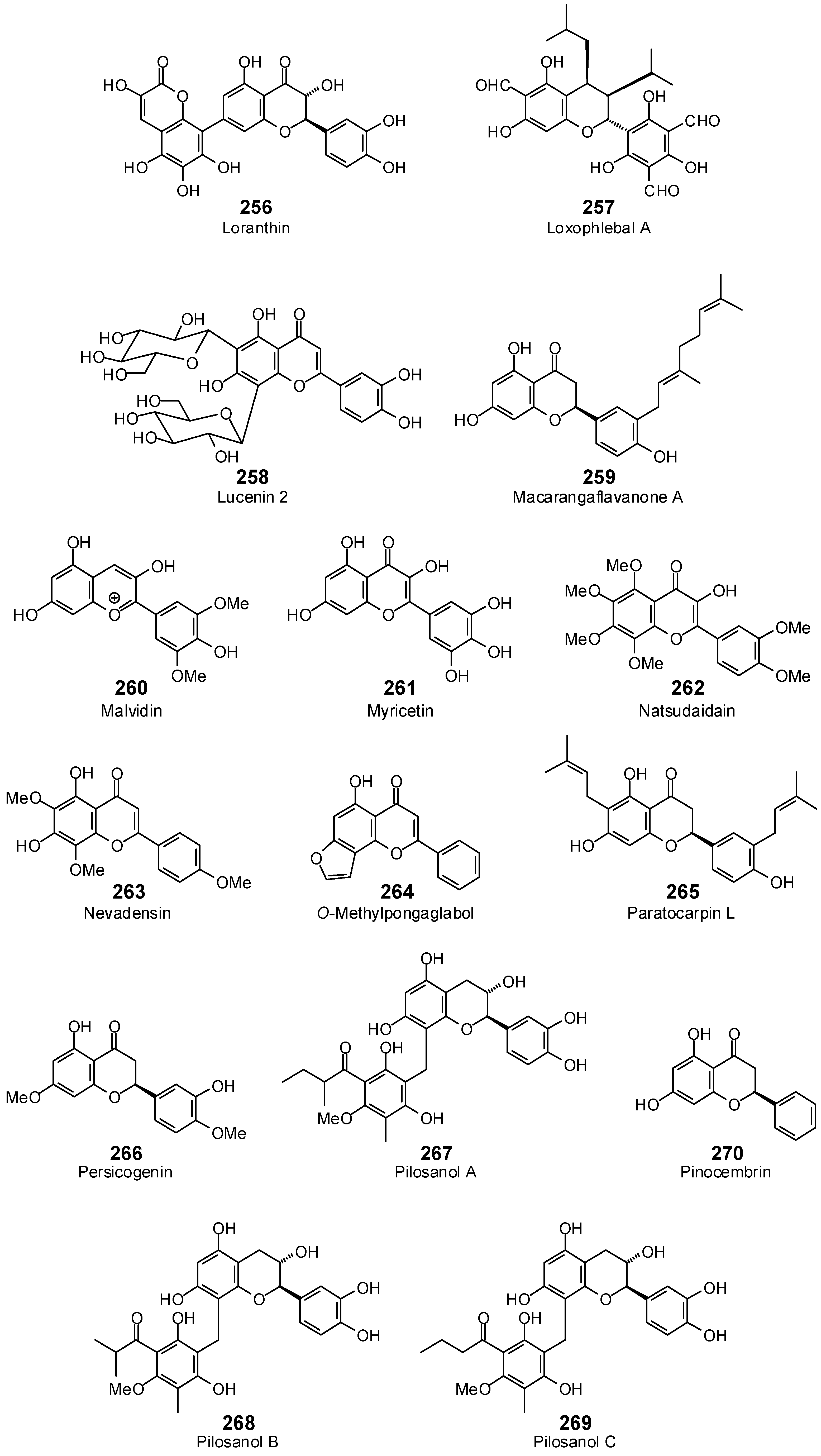
| Lonchocarpol A (255) | −93.8 | −90.9 | −110.7 | −107.3 | −113.9 | −110.3 | −112.0 | −108.5 | −116.1 | −112.5 | −111.9 | −108.5 | −111.5 | −108.0 |
| Loranthin (256) | −106.0 | −96.3 | −122.8 | −111.6 | −113.8 | −103.4 | −114.0 | −103.5 | −119.4 | −108.4 | −122.4 | −111.2 | −117.2 | −106.4 |
| Loxophlebal A (257) | −99.3 | −91.7 | −119.1 | −110.0 | −100.1 | −92.4 | −112.6 | −104.0 | −103.0 | −95.1 | −98.8 | −91.2 | −129.2 | −119.3 |
| Lucenin 2 (258) | −100.5 | −85.2 | −131.5 | −111.4 | −116.3 | −98.6 | −120.8 | −102.4 | −118.1 | −100.1 | −101.5 | −86.0 | −138.6 | −117.4 |
| Macarangaflavanone A (259) | −105.6 | −102.3 | −127.0 | −123.1 | −118.4 | −114.7 | −123.0 | −119.2 | −117.4 | −113.8 | −130.7 | −126.6 | −113.7 | −110.1 |
| Malvidin (260) | −89.6 | −93.1 | −108.4 | −112.7 | −99.8 | −103.7 | −111.6 | −115.9 | −97.7 | −101.6 | −95.1 | −98.8 | −98.7 | −102.6 |
| Myricetin (261) | −86.3 | −90.9 | −105.2 | −110.8 | −95.8 | −100.9 | −103.0 | −108.4 | −92.8 | −97.7 | −88.0 | −92.7 | −98.8 | −104.0 |
| Natsudaidain (262) | −78.2 | −75.2 | −118.9 | −114.3 | −102.9 | −99.0 | −105.1 | −101.0 | −99.1 | −95.3 | −102.4 | −98.4 | −108.6 | −104.4 |
| Nevadensin (263) | −85.9 | −88.1 | −106.9 | −109.6 | −96.8 | −99.3 | −110.0 | −112.9 | −94.8 | −97.2 | −98.3 | −100.9 | −106.8 | −109.6 |
| O-Methylpongaglabol (264) | −93.3 | −101.0 | −97.6 | −105.8 | −102.5 | −111.0 | −100.2 | −108.5 | −96.8 | −104.9 | −95.7 | −103.6 | −99.3 | −107.6 |
| Paratocarpin L (265) | −107.6 | −104.2 | −127.2 | −123.2 | −119.5 | −115.8 | −128.1 | −124.1 | −117.8 | −114.1 | −106.1 | −102.8 | −118.8 | −115.1 |
| Persicogenin (266) | −89.7 | −94.6 | −103.1 | −108.8 | −92.5 | −97.7 | −98.8 | −104.3 | −95.2 | −100.5 | −94.0 | −99.2 | −100.2 | −105.7 |
| Pilosanol A (267) | −114.7 | −101.2 | −124.2 | −109.7 | −112.9 | −99.7 | −124.4 | −109.8 | −111.9 | −98.8 | −131.6 | −116.2 | −129.0 | −113.9 |
| Pilosanol B (268) | −111.7 | −99.5 | −129.2 | −115.0 | −97.9 | −87.1 | −124.4 | −110.7 | −118.2 | −105.3 | −124.3 | −110.6 | −127.4 | −113.4 |
| Pilosanol C (269) | −115.8 | −103.1 | −132.5 | −118.0 | −108.4 | −96.5 | −123.4 | −109.9 | −121.1 | −107.8 | −125.4 | −111.7 | −130.0 | −115.7 |
| Pinocembrin (270) | −76.4 | −86.5 | −86.2 | −97.6 | −79.4 | −89.9 | −84.6 | −95.8 | −79.0 | −89.4 | −84.1 | −95.2 | −83.4 | −94.4 |
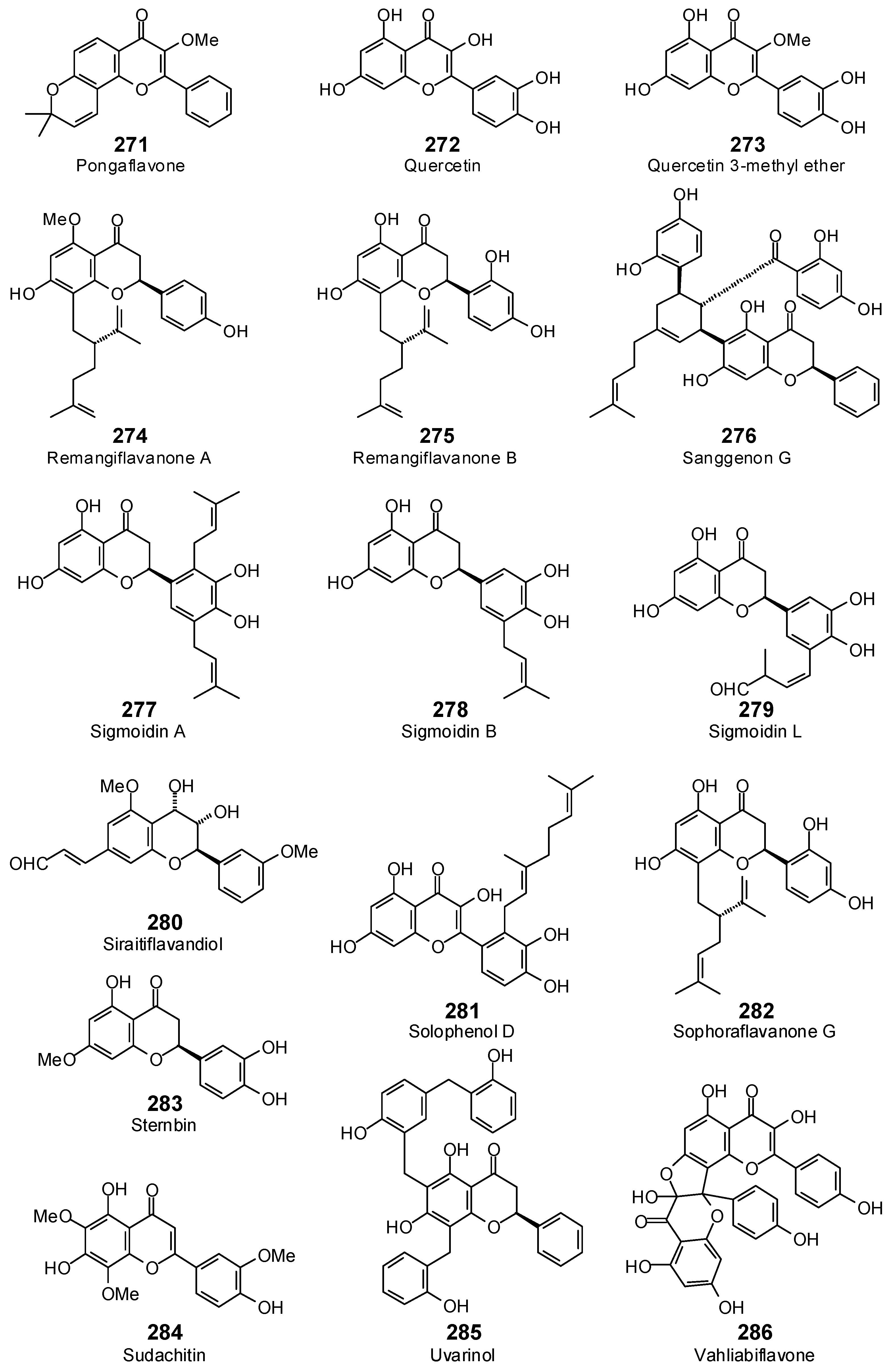
| Pongaflavone (271) | −84.1 | −87.1 | −99.0 | −102.6 | −92.2 | −95.5 | −92.1 | −95.4 | −102.6 | −106.3 | −93.0 | −96.3 | −83.3 | −86.3 |
| Quercetin (272) | −83.6 | −89.6 | −100.0 | −107.2 | −92.3 | −98.9 | −95.5 | −102.3 | −89.1 | −95.5 | −88.0 | −94.3 | −94.4 | −101.2 |
| Quercetin 3-methyl ether (273) | −91.9 | −96.9 | −99.5 | −105.0 | −91.9 | −97.0 | −97.2 | −102.6 | −89.4 | −94.3 | −93.4 | −98.5 | −95.3 | −100.6 |
| Remangiflavanone A (274) | −101.5 | −97.3 | −124.9 | −119.7 | −113.6 | −108.9 | −114.2 | −109.4 | −113.5 | −108.7 | −103.6 | −99.2 | −114.4 | −109.6 |
| Remangiflavanone B (275) | −110.0 | −105.2 | −117.4 | −112.3 | −119.1 | −113.9 | −121.4 | −116.2 | −119.2 | −114.0 | −118.9 | −113.7 | −117.4 | −112.3 |
| Sanggenon G (276) | −35.5 | −28.8 | −127.7 | −103.7 | −108.3 | −87.9 | −124.4 | −101.0 | −129.3 | −105.0 | −131.1 | −106.5 | −178.3 | −144.7 |
| Sigmoidin A (277) | −108.2 | −103.5 | −126.6 | −121.1 | −117.3 | −112.3 | −126.1 | −120.7 | −119.8 | −114.6 | −114.1 | −109.2 | −128.5 | −122.9 |
| Sigmoidin B (278) | −98.1 | −99.5 | −117.3 | −118.9 | −111.4 | −112.9 | −114.3 | −115.9 | −102.2 | −103.6 | −100.5 | −101.9 | −112.4 | −114.0 |
| Sigmoidin L (279) | −106.5 | −106.6 | −119.4 | −119.5 | −117.4 | −117.5 | −119.7 | −119.8 | −108.0 | −108.1 | −106.9 | −107.0 | −115.3 | −115.4 |
| Siraitiflavandiol (280) | −96.6 | −98.0 | −113.0 | −114.6 | −101.8 | −103.2 | −102.4 | −103.8 | −97.6 | −99.0 | −97.4 | −98.8 | −114.1 | −115.8 |
| Solophenol D (281) | −108.3 | −102.5 | −127.4 | −120.5 | −119.4 | −113.0 | −136.5 | −129.1 | −122.7 | −116.1 | −122.9 | −116.3 | −123.8 | −117.1 |
| Sophoraflavanone G (282) | −102.9 | −98.5 | −128.2 | −122.6 | −115.7 | −110.7 | −120.5 | −115.3 | −116.5 | −111.5 | −116.6 | −111.6 | −116.4 | −111.3 |
| Sternbin (283) | −86.1 | −92.3 | −102.8 | −110.1 | −87.5 | −93.7 | −98.5 | −105.5 | −92.2 | −98.8 | −94.8 | −101.6 | −97.0 | −103.9 |
| Sudachitin (284) | −93.4 | −94.4 | −118.3 | −119.5 | −102.5 | −103.5 | −114.8 | −116.0 | −96.1 | −97.1 | −97.5 | −98.5 | −114.0 | −115.2 |
| Uvarinol (285) | −97.2 | −84.1 | −135.9 | −117.6 | −110.7 | −95.8 | −124.7 | −107.8 | −135.1 | −116.8 | −125.2 | −108.3 | −143.8 | −124.4 |
| Vahliabiflavone (286) | −95.7 | −82.9 | −104.3 | −90.4 | −106.0 | −91.9 | −104.9 | −91.0 | −119.0 | −103.2 | −106.5 | −92.3 | −110.4 | −95.7 |
| Vitexin (287) | −111.9 | −106.4 | −117.8 | −112.0 | −113.8 | −108.2 | −120.2 | −114.2 | −118.1 | −112.3 | −115.5 | −109.8 | −123.0 | −117.0 |
| Wogonin (288) | −87.6 | −95.8 | −94.1 | −102.8 | −91.1 | −99.6 | −96.2 | −105.2 | −91.0 | −99.5 | −93.4 | −102.2 | −94.0 | −102.8 |
| Isoflavonoids | ||||||||||||||
| 2″,3″-Epoxybolusanthol B (289) | −104.6 | −104.6 | −113.5 | −113.4 | −103.6 | −103.5 | −112.9 | −112.8 | −108.7 | −108.7 | −101.5 | −101.4 | −116.8 | −116.7 |
| 3′,5,7-Trihydroxy-4′-methoxy-5′,6-diprenylisoflavanone (290) | −87.5 | −82.8 | −124.9 | −118.2 | −107.4 | −101.6 | −124.0 | −117.4 | −112.6 | −106.6 | −118.5 | −112.1 | −130.5 | −123.5 |
| 4″-Hydroxydiphysolone (292) | −98.9 | −98.8 | −114.3 | −114.2 | −100.9 | −100.8 | −94.6 | −94.6 | −108.7 | −108.6 | −99.0 | −99.0 | −106.9 | −106.8 |
| 5,7-Dihydroxy-2′-methoxy-3′,4′-methylenedioxyisoflavanone (293) | −83.4 | −86.7 | −96.7 | −100.6 | −86.3 | −89.7 | −93.0 | −96.7 | −93.9 | −97.7 | −89.1 | −92.7 | −99.6 | −103.6 |
| 6a-Hydroxyphaseollin (291) | −76.2 | −78.7 | −101.8 | −105.0 | −91.8 | −94.7 | −92.1 | −95.0 | −84.4 | −87.0 | −82.2 | −84.8 | −103.1 | −106.4 |
| Amorphaquinone (294) | −80.8 | −82.7 | −99.5 | −101.9 | −89.0 | −91.1 | −98.9 | −101.3 | −89.5 | −91.6 | −90.5 | −92.7 | −93.1 | −95.3 |
| Asphodelin A (295) | −84.4 | −93.9 | −94.0 | −104.5 | −82.7 | −91.9 | −95.6 | −106.3 | −86.7 | −96.5 | −93.4 | −103.9 | −91.5 | −101.7 |
| Bidwillon A (296) | −55.7 | −54.0 | −127.1 | −123.2 | −110.0 | −106.6 | −108.6 | −105.2 | −113.7 | −110.1 | −105.4 | −102.2 | −120.3 | −116.6 |
| Bolucarpan A (297) | −82.8 | −83.0 | −97.4 | −97.7 | −92.9 | −93.2 | −89.8 | −90.0 | −89.4 | −89.7 | −102.1 | −102.4 | −92.6 | −92.9 |
| Bolucarpan B (298) | −77.9 | −78.3 | −101.1 | −101.6 | −91.9 | −92.4 | −85.8 | −86.3 | −90.3 | −90.7 | −104.1 | −104.6 | −96.0 | −96.5 |
| Bolucarpan D (299) | −77.9 | −80.5 | −97.4 | −100.7 | −89.2 | −92.2 | −80.6 | −83.3 | −86.7 | −89.7 | −100.3 | −103.6 | −91.1 | −94.2 |
| Bolusanthol B (300) | −98.2 | −99.6 | −112.0 | −113.6 | −101.1 | −102.5 | −108.3 | −109.8 | −107.2 | −108.8 | −100.2 | −101.6 | −112.5 | −114.1 |
| Cajanol (301) | −81.1 | −85.6 | −93.8 | −98.9 | −86.1 | −90.9 | −87.2 | −92.1 | −87.0 | −91.8 | −85.9 | −90.6 | −91.8 | −96.9 |
| Chandalone (302) | −63.0 | −61.2 | −122.2 | −118.8 | −113.3 | −110.2 | −116.7 | −113.5 | −109.3 | −106.2 | −109.8 | −106.8 | −122.2 | −118.8 |
| Dalversinol A (303) | −108.9 | −104.2 | −118.9 | −113.8 | −104.5 | −100.0 | −113.8 | −108.9 | −111.8 | −106.9 | −110.1 | −105.4 | −114.0 | −109.1 |
| Derrisin (304) | −88.8 | −84.7 | −97.1 | −92.6 | −81.8 | −78.0 | −82.9 | −79.1 | −82.4 | −78.6 | −86.6 | −82.6 | −87.0 | −83.0 |
| Erybraedin A (305) | −91.4 | −89.8 | −120.1 | −117.9 | −100.8 | −99.0 | −110.0 | −108.1 | −100.3 | −98.5 | −101.8 | −100.0 | −116.9 | −114.8 |
| Erybraedin D (306) | −97.9 | −96.3 | −106.6 | −104.9 | −102.5 | −100.8 | −107.0 | −105.2 | −96.4 | −94.8 | −99.7 | −98.0 | −106.0 | −104.2 |
| Erypoegin I (307) | −101.8 | −101.9 | −111.1 | −111.2 | −106.9 | −107.0 | −108.7 | −108.9 | −117.0 | −117.2 | −100.6 | −100.8 | −112.1 | −112.2 |
| Erysubin F (308) | −119.6 | −117.7 | −124.6 | −122.5 | −124.4 | −122.4 | −115.0 | −113.1 | −116.6 | −114.7 | −126.4 | −124.3 | −129.8 | −127.7 |
| Eryvarin V (309) | −91.8 | −88.0 | −115.0 | −110.1 | −96.8 | −92.7 | −114.8 | −110.0 | −104.3 | −99.9 | −98.7 | −94.6 | −99.2 | −95.0 |
| Eryvarin W (310) | −109.5 | −107.8 | −122.5 | −120.5 | −118.8 | −116.9 | −115.8 | −113.9 | −122.4 | −120.4 | −115.4 | −113.5 | −120.0 | −118.0 |
| Eryzerin C (311) | −75.0 | −73.6 | −122.3 | −119.9 | −108.0 | −105.9 | −110.1 | −108.0 | −113.5 | −111.3 | −106.6 | −104.5 | −120.3 | −117.9 |
| Eryzerin D (312) | −76.6 | −75.2 | −112.1 | −110.1 | −96.4 | −94.6 | −108.3 | −106.3 | −104.7 | −102.9 | −103.4 | −101.6 | −103.9 | −102.0 |
| Euchretin A (313) | −25.3 | −22.9 | −114.5 | −103.6 | −115.6 | −104.5 | −99.2 | −89.7 | −115.4 | −104.3 | −114.4 | −103.5 | −122.2 | −110.5 |
| Gancaonin C (314) | −105.9 | −107.6 | −116.7 | −118.6 | −104.0 | −105.7 | −113.7 | −115.5 | −108.7 | −110.5 | −108.4 | −110.2 | −115.2 | −117.1 |
| Genistein (315) | −81.3 | −90.4 | −92.5 | −102.9 | −78.6 | −87.4 | −86.0 | −95.6 | −75.6 | −84.1 | −82.5 | −91.7 | −87.7 | −97.6 |
| Glycyrrhisoflavone (316) | −98.3 | −99.9 | −113.9 | −115.8 | −110.6 | −112.4 | −109.4 | −111.1 | −102.1 | −103.7 | −105.9 | −107.6 | −116.2 | −118.1 |
| Hispaglabridin A (317) | −38.2 | −37.5 | −117.1 | −115.0 | −107.8 | −105.8 | −111.6 | −109.6 | −93.7 | −92.0 | −106.3 | −104.4 | −118.1 | −115.9 |
| Hispaglabridin B (318) | −71.6 | −70.4 | −113.3 | −111.4 | −91.8 | −90.3 | −111.1 | −109.3 | −79.1 | −77.8 | −88.5 | −87.0 | −97.2 | −95.6 |
| Hydroxycristacarpone (319) | −96.7 | −96.8 | −112.7 | −112.8 | −108.2 | −108.4 | −100.5 | −100.6 | −113.0 | −113.2 | −110.3 | −110.4 | −100.3 | −100.4 |
| Isoneorautenol (320) | −82.2 | −86.2 | −96.1 | −100.8 | −83.2 | −87.2 | −80.1 | −84.0 | −83.3 | −87.4 | −87.9 | −92.1 | −86.5 | −90.7 |
| Lachnoisoflavone A (321) | −65.6 | −66.8 | −118.5 | −120.6 | −107.3 | −109.2 | −93.5 | −95.1 | −105.3 | −107.2 | −105.5 | −107.4 | −98.4 | −100.2 |
| Licoisoflavanone (322) | −76.9 | −78.2 | −105.5 | −107.2 | −91.8 | −93.2 | −103.0 | −104.7 | −87.4 | −88.8 | −84.6 | −85.9 | −100.7 | −102.3 |
| Licoricidin (323) | −106.3 | −101.7 | −122.2 | −116.9 | −114.1 | −109.1 | −125.1 | −119.7 | −114.8 | −109.8 | −114.5 | −109.6 | −117.0 | −111.9 |
| Lupinalbin C (324) | −79.3 | −79.5 | −104.0 | −104.3 | −94.1 | −94.3 | −90.1 | −90.4 | −94.3 | −94.5 | −92.5 | −92.7 | −97.1 | −97.4 |
| Mucronulatol (326) | −83.9 | −89.9 | −98.8 | −105.9 | −90.1 | −96.5 | −96.0 | −102.9 | −80.9 | −86.7 | −86.8 | −93.0 | −94.0 | −100.7 |
| Neomillinol (325) | −81.1 | −84.7 | −106.1 | −110.8 | −96.8 | −101.1 | −94.9 | −99.1 | −93.6 | −97.7 | −85.8 | −89.6 | −101.5 | −106.0 |
| Pendulone (327) | −77.9 | −82.2 | −98.2 | −103.6 | −85.9 | −90.6 | −94.6 | −99.8 | −81.4 | −85.9 | −84.3 | −89.0 | −92.1 | −97.2 |
| Phyllanone B (328) | −96.4 | −91.2 | −121.7 | −115.1 | −115.3 | −109.2 | −116.5 | −110.3 | −116.0 | −109.8 | −117.0 | −110.7 | −122.4 | −115.9 |
| Shinpterocarpin (329) | −73.3 | −76.8 | −88.0 | −92.3 | −84.4 | −88.5 | −82.2 | −86.2 | −85.0 | −89.2 | −82.0 | −86.0 | −85.9 | −90.1 |
| Neoflavonoids | ||||||||||||||
| Inophyllum A (330) | −98.8 | −96.0 | −109.9 | −106.9 | −100.1 | −97.3 | −102.0 | −99.2 | −102.2 | −99.4 | −97.5 | −94.8 | −105.0 | −102.1 |
| Inophyllum C (331) | −95.4 | −92.9 | −104.1 | −101.4 | −92.0 | −89.6 | −103.4 | −100.7 | −102.7 | −100.0 | −92.6 | −90.1 | −105.8 | −103.0 |
| Mammea A/BA (332) | −107.7 | −104.5 | −123.2 | −119.6 | −118.6 | −115.1 | −120.5 | −116.9 | −119.6 | −116.1 | −110.5 | −107.2 | −125.6 | −121.9 |
| Mammea A/BB (333) | −101.4 | −98.4 | −120.8 | −117.3 | −114.7 | −111.3 | −112.3 | −109.0 | −117.6 | −114.2 | −107.2 | −104.0 | −121.8 | −118.3 |
| Mesuol (334) | −92.5 | −90.8 | −118.7 | −116.6 | −114.1 | −112.0 | −114.2 | −112.2 | −107.6 | −105.6 | −102.7 | −100.8 | −112.0 | −110.0 |
| Pterocarpans | ||||||||||||||
| 1-Methoxyphaseollidin (356) | −92.1 | −93.6 | −105.6 | −107.3 | −109.5 | −111.2 | −112.3 | −114.1 | −105.1 | −106.8 | −99.0 | −100.5 | −104.9 | −106.6 |
| Aracarpene 1 (357) | −83.2 | −89.3 | −95.0 | −102.0 | −88.1 | −94.6 | −85.7 | −92.0 | −97.9 | −105.1 | −88.3 | −94.8 | −89.7 | −96.3 |
| Aracarpene 2 (358) | −81.3 | −87.3 | −98.1 | −105.3 | −90.1 | −96.7 | −88.6 | −95.2 | −94.9 | −101.9 | −86.0 | −92.3 | −91.3 | −98.1 |
| Calopocarpin (360) | −96.7 | −101.2 | −103.8 | −108.7 | −108.1 | −113.2 | −103.3 | −108.1 | −103.8 | −108.7 | −103.0 | −107.8 | −100.8 | −105.5 |
| Cristacarpin (359) | −82.4 | −83.7 | −107.2 | −108.9 | −107.9 | −109.7 | −102.8 | −104.5 | −108.1 | −109.8 | −95.7 | −97.2 | −102.3 | −104.0 |
| Erythbidin D (362) | −84.8 | −91.1 | −92.4 | −99.2 | −92.7 | −99.5 | −94.1 | −101.1 | −90.4 | −97.1 | −85.8 | −92.1 | −91.5 | −98.2 |
| Eryzerin E (363) | −100.9 | −96.7 | −122.4 | −117.2 | −103.3 | −98.9 | −111.1 | −106.4 | −113.7 | −108.9 | −114.5 | −109.7 | −120.6 | −115.6 |
| Fuscacarpan B (364) | −86.2 | −86.3 | −111.4 | −111.6 | −99.6 | −99.7 | −98.3 | −98.4 | −102.5 | −102.7 | −96.5 | −96.6 | −105.8 | −105.9 |
| Fuscacarpan C (365) | −84.7 | −84.8 | −112.7 | −112.8 | −100.9 | −101.0 | −101.4 | −101.5 | −113.2 | −113.4 | −98.0 | −98.1 | −102.9 | −103.0 |
| Glycyrol (366) | −90.2 | −90.6 | −105.5 | −106.0 | −105.9 | −106.4 | −102.2 | −102.7 | −103.6 | −104.1 | −101.9 | −102.4 | −94.9 | −95.4 |
| Glycyrrhizol A (367) | −90.8 | −87.1 | −120.0 | −115.2 | −120.9 | −116.0 | −109.4 | −105.0 | −109.5 | −105.1 | −106.8 | −102.5 | −117.0 | −112.3 |
| Glycyrrhizol B (368) | −88.3 | −90.1 | −104.6 | −106.7 | −98.7 | −100.7 | −94.4 | −96.3 | −103.6 | −105.6 | −100.1 | −102.1 | −98.0 | −99.9 |
| Sandwicensin (369) | −83.2 | −85.9 | −105.3 | −108.6 | −104.1 | −107.4 | −107.0 | −110.4 | −103.4 | −106.7 | −96.3 | −99.3 | −98.9 | −102.1 |
| Variabilin (370) | −77.0 | −82.7 | −93.1 | −99.9 | −94.7 | −101.7 | −84.6 | −90.8 | −98.1 | −105.3 | −89.2 | −95.8 | −93.7 | −100.6 |
| ent-Sophoracarpan A (361) | −79.2 | −85.0 | −96.6 | −103.7 | −90.0 | −96.7 | −97.6 | −104.8 | −92.3 | −99.1 | −92.8 | −99.6 | −95.4 | −102.5 |
| Chromones | ||||||||||||||
| 3-(3-Hydroxy-4-methoxy-benzylidene)-6,7-dimethoxy-4-chromanone (371) | −78.1 | −80.2 | −113.4 | −116.6 | −97.5 | −100.2 | −99.4 | −102.2 | −94.3 | −96.9 | −85.2 | −87.5 | −97.7 | −100.5 |
| 4′,5,7-Trihydroxy-6,8-dimethyl-homoisoflavanone (372) | −83.5 | −88.3 | −105.0 | −111.0 | −89.2 | −94.4 | −98.5 | −104.2 | −86.6 | −91.6 | −86.7 | −91.7 | −98.2 | −103.9 |
| 4′,5,7-Trihydroxy-6-methyl-homoisoflavanone (373) | −79.0 | −84.8 | −102.7 | −110.3 | −86.7 | −93.1 | −98.7 | −105.9 | −84.3 | −90.5 | −95.3 | −102.3 | −91.9 | −98.7 |
| 7-O-Methylbonducellin (374) | −75.5 | −81.4 | −102.1 | −110.2 | −84.0 | −90.6 | −92.8 | −100.1 | −76.9 | −83.0 | −83.5 | −90.0 | −89.5 | −96.5 |
| 8-Methylophiopogonanone B (375) | −86.3 | −90.0 | −108.3 | −112.9 | −87.7 | −91.4 | −103.9 | −108.3 | −94.9 | −98.9 | −92.6 | −96.5 | −104.1 | −108.4 |
| Bonducellin (376) | −75.3 | −82.5 | −95.5 | −104.7 | −83.3 | −91.2 | −90.0 | −98.7 | −79.6 | −87.3 | −80.3 | −88.0 | −90.8 | −99.6 |
| Odoratumone A (377) | −89.3 | −91.6 | −114.3 | −117.2 | −97.6 | −100.2 | −110.5 | −113.3 | −98.4 | −101.0 | −97.6 | −100.1 | −110.7 | −113.6 |
| Sappanone A 3′,4′-methylene ether (378) | −81.8 | −88.2 | −102.1 | −110.1 | −92.3 | −99.5 | −94.4 | −101.8 | −86.4 | −93.2 | −83.6 | −90.2 | −94.5 | −101.9 |
| Sappanone A 4′-methyl ether (379) | −79.9 | −86.0 | −101.4 | −109.1 | −81.4 | −87.6 | −94.1 | −101.2 | −87.7 | −94.4 | −82.4 | −88.7 | −97.0 | −104.4 |
| Sappanone A trimethyl ether (380) | −82.4 | −86.1 | −111.8 | −116.7 | −89.0 | −92.9 | −107.5 | −112.3 | −90.2 | −94.2 | −89.7 | −93.6 | −94.8 | −99.0 |
| Condensed Tannins | ||||||||||||||
| GB 1 (381) | −121.0 | −105.6 | −118.3 | −103.3 | −100.9 | −88.1 | −127.4 | −111.2 | −145.4 | −127.0 | −123.4 | −107.8 | −116.1 | −101.4 |
| Proanthocyanidin A1 (382) | −95.2 | −82.3 | −126.7 | −109.4 | −129.2 | −111.6 | −105.9 | −91.5 | −116.3 | −100.5 | −122.7 | −106.0 | −117.6 | −101.6 |
| Proanthocyanidin A2 (383) | −94.3 | −81.5 | −109.1 | −94.3 | −130.1 | −112.4 | −109.4 | −94.5 | −110.2 | −95.2 | −121.5 | −105.0 | −113.9 | −98.4 |
| Procyanidin B4 (384) | −97.8 | −84.4 | −118.8 | −102.5 | −98.7 | −85.1 | −115.7 | −99.8 | −123.1 | −106.2 | −112.6 | −97.2 | −113.5 | −97.9 |
| Procyanidin B5 (385) | 23.0 | 19.9 | −114.3 | −98.7 | −111.8 | −96.5 | −131.1 | −113.1 | −131.3 | −113.3 | −115.6 | −99.7 | −146.4 | −126.3 |
| Procyanidin B6 (386) | −68.6 | −59.2 | −123.6 | −106.6 | −101.3 | −87.4 | −111.5 | −96.2 | −135.0 | −116.5 | −121.6 | −104.9 | −126.1 | −108.8 |
| Teatannin (387) | −96.6 | −91.1 | −131.8 | −124.4 | −103.4 | −97.6 | −118.4 | −111.8 | −107.6 | −101.6 | −104.7 | −98.8 | −124.7 | −117.7 |
| Coumarins | ||||||||||||||
| 4,5′,8′-Trihydroxy-5-methyl-3,7′-bicoumarin (344) | −97.3 | −99.0 | −104.3 | −106.2 | −89.6 | −91.2 | −103.4 | −105.3 | −95.2 | −96.9 | −92.9 | −94.6 | −104.1 | −106.0 |
| 6-Geranyl-5,7-dihydroxy-8(2-methylbutanoyl)-4-phenyl-coumarin (345) | −112.6 | −103.8 | −127.7 | −117.7 | −115.8 | −106.8 | −128.6 | −118.5 | −122.3 | −112.7 | −121.3 | −111.8 | −132.2 | −121.9 |
| (–)-Heliettin (354) | −70.0 | −89.5 | −72.8 | −93.1 | −66.9 | −85.5 | −73.5 | −93.9 | −70.0 | −89.4 | −71.3 | −91.1 | −70.4 | −89.9 |
| Aesculin (347) | −99.8 | −102.7 | −111.0 | −114.4 | −104.2 | −107.3 | −103.4 | −106.5 | −100.9 | −103.9 | −97.8 | −100.7 | −102.1 | −105.1 |
| Alloimperatorin (346) | −95.2 | −105.8 | −100.7 | −112.0 | −99.5 | −110.7 | −101.6 | −112.9 | −97.1 | −107.9 | −97.7 | −108.6 | −98.4 | −109.4 |
| Anofinic acid (348) | −75.4 | −92.0 | −80.1 | −97.8 | −77.9 | −95.1 | −78.2 | −95.4 | −77.7 | −94.9 | −78.1 | −95.3 | −83.4 | −101.8 |
| Calaustralin (349) | −90.9 | −88.4 | −111.9 | −108.8 | −111.2 | −108.1 | −107.3 | −104.3 | −100.5 | −97.8 | −97.5 | −94.7 | −114.3 | −111.1 |
| Calophyllolide (350) | −94.3 | −90.8 | −120.9 | −116.4 | −111.0 | −106.9 | −111.4 | −107.3 | −113.9 | −109.6 | −114.6 | −110.4 | −101.2 | −97.4 |
| Dicoumarol (351) | −95.2 | −98.4 | −101.6 | −105.1 | −80.7 | −83.5 | −96.8 | −100.1 | −93.5 | −96.6 | −90.3 | −93.4 | −99.6 | −103.0 |
| Dipetalolactone (352) | −84.9 | −90.1 | −95.2 | −101.1 | −101.3 | −107.6 | −96.3 | −102.2 | −89.8 | −95.3 | −90.7 | −96.3 | −91.2 | −96.9 |
| Glycycoumarin (353) | −89.1 | −89.4 | −113.1 | −113.5 | −106.0 | −106.3 | −102.7 | −103.0 | −100.7 | −101.0 | −95.5 | −95.8 | −100.3 | −100.6 |
| Marmesin (355) | −69.8 | −80.0 | −87.2 | −100.0 | −80.1 | −91.9 | −84.0 | −96.3 | −81.7 | −93.8 | −83.8 | −96.1 | −77.9 | −89.3 |
| Stilbenoids | ||||||||||||||
| 2-(2,4-Dihydroxyphenyl-5-(1-propenyl)benzofuran (388) | −82.8 | −92.6 | −96.0 | −107.3 | −90.3 | −100.9 | −97.5 | −109.0 | −85.2 | −95.2 | −88.9 | −99.3 | −94.9 | −106.0 |
| Albanol A (389) | −79.4 | −69.2 | −116.3 | −101.3 | −101.2 | −88.1 | −125.2 | −109.1 | −111.8 | −97.4 | −106.7 | −92.9 | −147.1 | −128.1 |
| Albanol B (390) | no dock | no dock | −138.1 | −120.5 | −23.4 | −20.4 | −109.7 | −95.8 | −106.7 | −93.2 | −113.0 | −98.7 | −114.7 | −100.2 |
| Amorfrutin A (391) | −100.4 | −103.3 | −121.8 | −125.4 | −103.6 | −106.7 | −117.0 | −120.5 | −113.0 | −116.3 | −109.3 | −112.5 | −118.5 | −122.0 |
| Cajaninstilbene acid (392) | −97.3 | −100.3 | −116.3 | −120.0 | −99.0 | −102.1 | −112.7 | −116.3 | −97.3 | −100.4 | −97.9 | −101.0 | −118.9 | −122.6 |
| Calodenin B (393) | −114.8 | −102.4 | −148.6 | −132.5 | −135.7 | −121.0 | −150.0 | −133.7 | −157.6 | −140.6 | −143.5 | −127.9 | −147.0 | −131.1 |
| Centrolobofuran (394) | −82.1 | −93.0 | −95.3 | −107.9 | −83.6 | −94.7 | −93.4 | −105.8 | −85.8 | −97.1 | −84.0 | −95.0 | −90.6 | −102.6 |
| Cochinchinenene A (395) | −84.4 | −74.5 | −113.7 | −100.3 | −102.5 | −90.5 | −107.9 | −95.2 | −134.6 | −118.8 | −119.9 | −105.8 | −135.8 | −119.9 |
| Cochinchinenene B (396) | −101.2 | −90.9 | −145.2 | −130.5 | −101.1 | −90.9 | −124.5 | −111.9 | −130.4 | −117.2 | −119.8 | −107.6 | −156.8 | −140.8 |
| Cochinchinenene C (397) | −114.8 | −104.1 | −135.8 | −123.1 | −133.2 | −120.7 | −117.1 | −106.2 | −133.7 | −121.2 | −116.3 | −105.4 | −138.2 | −125.3 |
| Cochinchinenene D (398) | −127.5 | −116.7 | −140.3 | −128.4 | −107.9 | −98.7 | −121.1 | −110.9 | −124.0 | −113.5 | −121.1 | −110.8 | −145.0 | −132.8 |
| Egonol (399) | −108.1 | −112.9 | −120.1 | −125.4 | −103.9 | −108.5 | −114.9 | −119.9 | −104.1 | −108.7 | −105.0 | −109.6 | −119.2 | −124.5 |
| Erypoegin F (400) | −104.3 | −106.2 | −122.4 | −124.6 | −120.3 | −122.4 | −121.9 | −124.1 | −115.1 | −117.2 | −114.8 | −116.9 | −117.1 | −119.2 |
| Erythbidin E (401) | −53.5 | −60.6 | −97.7 | −110.6 | −86.9 | −98.3 | −91.0 | −103.0 | −87.0 | −98.5 | −85.5 | −96.8 | −91.8 | −103.9 |
| Eryvarin Q (402) | −119.5 | −116.0 | −140.8 | −136.7 | −126.3 | −122.6 | −143.0 | −138.8 | −130.9 | −127.1 | −131.6 | −127.7 | −133.9 | −130.0 |
| Gancaonin I (403) | −108.9 | −110.6 | −120.0 | −121.9 | −103.7 | −105.4 | −120.9 | −122.9 | −110.5 | −112.2 | −108.4 | −110.2 | −120.8 | −122.7 |
| Glyinflanin H (404) | −90.5 | −96.3 | −101.8 | −108.4 | −88.3 | −94.0 | −102.3 | −108.9 | −86.5 | −92.0 | −88.9 | −94.6 | −90.9 | −96.7 |
| Kuwanol A (405) | −74.5 | −64.8 | −111.1 | −96.7 | −101.9 | −88.7 | −129.6 | −112.8 | −119.2 | −103.7 | −116.2 | −101.1 | −155.8 | −135.6 |
| Licobenzofuran (406) | −106.3 | −108.0 | −117.4 | −119.2 | −119.7 | −121.6 | −112.6 | −114.4 | −109.1 | −110.8 | −112.5 | −114.3 | −113.3 | −115.1 |
| Licocoumarone (407) | −102.0 | −105.0 | −120.5 | −124.1 | −98.7 | −101.7 | −120.7 | −124.3 | −104.4 | −107.5 | −98.6 | −101.5 | −118.4 | −122.0 |
| Mulberrofuran D (408) | −122.0 | −114.8 | −145.5 | −136.9 | −120.8 | −113.6 | −136.9 | −128.8 | −129.7 | −122.0 | −129.5 | −121.8 | −134.3 | −126.3 |
| Mulberrofuran Y (409) | −114.6 | −111.0 | −134.3 | −130.1 | −113.6 | −110.0 | −128.8 | −124.8 | −126.8 | −122.9 | −123.2 | −119.4 | −137.9 | −133.6 |
| Pinosylvin (410) | −61.5 | −74.1 | −88.4 | −106.6 | −77.5 | −93.4 | −78.8 | −95.0 | −74.9 | −90.3 | −76.6 | −92.3 | −80.0 | −96.4 |
| Schweinfurthin A (411) | −30.4 | −26.7 | −82.8 | −72.7 | −114.6 | −100.7 | −113.9 | −100.1 | −116.6 | −102.4 | −120.7 | −106.0 | −123.6 | −108.5 |
| Shanciguol 3-methyl ether (412) | −107.2 | −101.1 | −125.1 | −118.0 | −112.9 | −106.5 | −119.7 | −113.0 | −122.2 | −115.3 | −116.9 | −110.3 | −130.9 | −123.5 |
| Stemofuran R (413) | −92.6 | −96.5 | −110.7 | −115.3 | −95.4 | −99.4 | −106.3 | −110.8 | −98.9 | −103.0 | −100.8 | −105.0 | −106.3 | −110.8 |
| Stilbostemin S (414) | −92.1 | −100.3 | −107.7 | −117.2 | −93.5 | −101.8 | −102.0 | −111.0 | −101.8 | −110.8 | −101.6 | −110.6 | −97.8 | −106.4 |
| Thunberginol F (415) | −85.5 | −95.1 | −99.1 | −110.2 | −85.7 | −95.3 | −96.7 | −107.6 | −89.3 | −99.3 | −86.8 | −96.6 | −96.0 | −106.8 |
| (7E,7′R,8′R)-ε-Viniferin (416) | −101.7 | −95.1 | −134.0 | −125.3 | −118.1 | −110.4 | −110.8 | −103.6 | −114.7 | −107.3 | −115.6 | −108.1 | −131.7 | −123.2 |
| (7E,7′S,8′S)-ε-Viniferin (417) | −103.2 | −96.5 | −134.3 | −125.6 | −112.6 | −105.3 | −111.0 | −103.8 | −119.1 | −111.3 | −118.8 | −111.1 | −126.9 | −118.7 |
| Phenylpropanoids and Lignans | ||||||||||||||
| (E)-Cinnamaldehyde (418) | −69.5 | −98.1 | −70.6 | −99.6 | −66.4 | −93.7 | −71.3 | −100.6 | −64.3 | −90.7 | −66.8 | −94.2 | −68.2 | −96.3 |
| 3,4-Dimethylcinnamaldehyde (419) | −77.2 | −102.2 | −81.6 | −108.0 | −72.2 | −95.5 | −77.2 | −102.3 | −77.1 | −102.0 | −78.7 | −104.2 | −79.4 | −105.1 |
| Methyleugenol (422) | −79.9 | −102.1 | −81.8 | −104.5 | −74.8 | −95.6 | −78.1 | −99.8 | −79.6 | −101.7 | −80.4 | −102.7 | −79.8 | −101.9 |
| p-Coumaraldehyde (420) | −73.8 | −100.3 | −75.9 | −103.2 | −67.9 | −92.3 | −72.8 | −99.0 | −69.8 | −94.8 | −72.5 | −98.6 | −74.3 | −101.0 |
| p-Methoxycinnamaldehyde (421) | −79.0 | −104.2 | −82.1 | −108.2 | −71.7 | −94.5 | −78.5 | −103.5 | −74.1 | −97.7 | −76.3 | −100.6 | −81.3 | −107.2 |
| (–)-Asarinin (423) | −98.1 | −99.6 | −120.0 | −121.9 | −96.7 | −98.2 | −106.8 | −108.5 | −109.4 | −111.1 | −110.6 | −112.4 | −117.3 | −119.2 |
| Nordihydroguaiaretic acid (424) | −88.6 | −94.9 | −106.0 | −113.6 | −103.9 | −111.3 | −100.1 | −107.3 | −98.0 | −105.0 | −93.5 | −100.1 | −104.6 | −112.1 |
| Xanthones | ||||||||||||||
| 2-Deoxy-4-Hydroxycudratricus-xanthone D (425) | −92.1 | −90.3 | −103.2 | −101.2 | −110.9 | −108.8 | −108.2 | −106.1 | −97.7 | −95.8 | −96.2 | −94.3 | −96.4 | −94.5 |
| Calozeyloxanthone (426) | −86.8 | −86.3 | −88.3 | −87.8 | −83.8 | −83.3 | −86.3 | −85.8 | −92.6 | −92.1 | −99.1 | −98.5 | −92.1 | −91.6 |
| Cycloartobiloxanthone (427) | −89.3 | −84.8 | −99.1 | −94.0 | −101.4 | −96.3 | −103.8 | −98.6 | −98.4 | −93.5 | −96.6 | −91.7 | −94.4 | −89.6 |
| Formoxanthone C (428) | −103.0 | −100.8 | −101.9 | −99.8 | −107.6 | −105.3 | −105.2 | −103.0 | −108.6 | −106.3 | −104.4 | −102.2 | −107.6 | −105.3 |
| Garciniacowone (429) | −71.0 | −65.8 | −124.5 | −115.4 | −110.4 | −102.3 | −106.7 | −98.9 | −123.0 | −114.0 | −115.0 | −106.6 | −119.9 | −111.1 |
| Globulixanthone C (430) | −81.5 | −85.1 | −102.8 | −107.3 | −90.9 | −95.0 | −94.4 | −98.6 | −94.5 | −98.7 | −90.6 | −94.6 | −89.5 | −93.5 |
| Globulixanthone D (431) | −90.9 | −93.4 | −109.6 | −112.6 | −88.5 | −90.9 | −99.0 | −101.7 | −98.4 | −101.2 | −98.9 | −101.7 | −99.6 | −102.3 |
| Globulixanthone E (432) | −68.0 | −57.3 | −114.7 | −96.8 | −110.9 | −93.6 | −89.7 | −75.7 | −104.9 | −88.6 | −110.4 | −93.2 | −124.0 | −104.6 |
| Morellin (433) | −88.3 | −77.7 | −92.7 | −81.6 | −102.7 | −90.4 | −98.8 | −87.0 | −107.7 | −94.8 | −107.7 | −94.8 | −97.7 | −86.0 |
| Nigrolineaxanthone N (434) | −98.5 | −96.2 | −105.1 | −102.7 | −99.2 | −97.0 | −103.5 | −101.1 | −120.9 | −118.1 | −106.5 | −104.1 | −99.0 | −96.7 |
| Pinselin (435) | −94.0 | −100.9 | −91.2 | −97.9 | −95.4 | −102.4 | −99.2 | −106.5 | −102.7 | −110.2 | −96.4 | −103.5 | −98.3 | −105.6 |
| Scortechinone B (436) | −95.9 | −82.1 | −123.2 | −105.5 | −112.5 | −96.3 | −85.9 | −73.5 | −113.0 | −96.7 | −102.3 | −87.6 | −107.4 | −91.9 |
| Symphonin (437) | −97.5 | −91.3 | −107.7 | −100.9 | −109.3 | −102.4 | −103.9 | −97.3 | −106.0 | −99.2 | −103.0 | −96.5 | −94.7 | −88.7 |
| Hydrolyzable Tannins | ||||||||||||||
| 1,2,3,4,6-Pentagalloylglucose (335) | −115.6 | −84.8 | −178.0 | −130.6 | −148.1 | −108.7 | −161.0 | −118.1 | −157.5 | −115.6 | −151.6 | −111.3 | −184.7 | −135.5 |
| Aceritannin (438) | −112.5 | −104.1 | −133.9 | −123.9 | −133.0 | −123.1 | −146.6 | −135.7 | −128.6 | −119.0 | −139.2 | −128.9 | −121.8 | −112.8 |
| Ginnalin B (439) | −92.2 | −97.3 | −101.3 | −106.9 | −102.7 | −108.4 | −104.0 | −109.7 | −100.2 | −105.7 | −99.4 | −104.9 | −106.3 | −112.1 |
| Ginnalin C (440) | −88.4 | −93.3 | −106.6 | −112.5 | −94.6 | −99.8 | −110.4 | −116.5 | −100.8 | −106.4 | −94.1 | −99.3 | −106.0 | −111.9 |
| Panconoside A (441) | −79.7 | −65.9 | −137.5 | −113.6 | −118.4 | −97.8 | −124.5 | −102.9 | −119.3 | −98.6 | −112.5 | −93.0 | −134.2 | −110.9 |
| Miscellaneous Phenolics | ||||||||||||||
| 1,3,7,9-Tetrahydroxy-4,6-dimethyl-2,8-bis(2-methyl-propanoyl)-dibenzofuran (442) | −93.7 | −91.4 | −119.9 | −117.0 | −104.7 | −102.1 | −106.2 | −103.6 | −102.4 | −99.8 | −100.3 | −97.9 | −108.3 | −105.7 |
| 2′,4′-Dihydroxy-6′-methoxy-3′-methylacetophenone (443) | −76.2 | −94.3 | −76.7 | −94.9 | −75.5 | −93.4 | −77.6 | −96.0 | −74.5 | −92.2 | −75.7 | −93.7 | −74.7 | −92.4 |
| 3′,4′-Dihydroxyacetophenone (444) | −68.6 | −92.4 | −71.7 | −96.6 | −67.3 | −90.6 | −70.5 | −95.0 | −70.3 | −94.7 | −70.2 | −94.5 | −68.1 | −91.8 |
| 4′-O-Methylhonokiol (446) | −95.2 | −104.6 | −107.7 | −118.3 | −100.8 | −110.7 | −107.5 | −118.1 | −97.7 | −107.3 | −97.0 | −106.6 | −100.2 | −110.1 |
| 4-Deoxyadhumulone 2″,3″-epoxide (445) | −75.3 | −75.9 | −114.6 | −115.5 | −97.6 | −98.4 | −104.9 | −105.8 | −103.4 | −104.2 | −98.7 | −99.5 | −109.0 | −109.9 |
| 7-(3,4-Dihydroxy-5-methoxy-phenyl)-1-phenyl-4-hepten-3-one (447) | −103.6 | −108.2 | −124.2 | −129.7 | −104.9 | −109.5 | −121.5 | −126.9 | −111.0 | −115.9 | −110.6 | −115.5 | −117.6 | −122.8 |
| Agrimol C (449) | −102.3 | −84.1 | −136.6 | −112.3 | −104.0 | −85.5 | −127.8 | −105.1 | −126.9 | −104.4 | −126.4 | −103.9 | −119.5 | −98.3 |
| Agrimol F (450) | −107.7 | −89.9 | −127.3 | −106.1 | −106.0 | −88.4 | −121.5 | −101.3 | −126.6 | −105.6 | −122.2 | −101.9 | −125.8 | −104.9 |
| Agrimol G (451) | −85.0 | −69.9 | −128.4 | −105.5 | −104.6 | −86.0 | −122.2 | −100.5 | −132.6 | −109.0 | −124.6 | −102.4 | −134.9 | −110.9 |
| Arzanol (452) | −96.8 | −94.3 | −118.6 | −115.5 | −89.1 | −86.8 | −118.2 | −115.1 | −102.1 | −99.4 | −108.1 | −105.3 | −110.5 | −107.6 |
| Aspidinol C (448) | −82.6 | −97.7 | −92.9 | −109.9 | −85.4 | −101.1 | −89.8 | −106.3 | −89.2 | −105.5 | −93.9 | −111.1 | −90.1 | −106.7 |
| Bruguierol C (453) | −80.3 | −97.7 | −76.2 | −92.7 | −76.7 | −93.4 | −80.0 | −97.3 | −78.7 | −95.7 | −80.0 | −97.4 | −76.4 | −93.0 |
| Cearoin (454) | −87.2 | −100.3 | −91.7 | −105.4 | −90.1 | −103.6 | −88.6 | −101.9 | −94.3 | −108.5 | −89.2 | −102.6 | −87.1 | −100.1 |
| Citrusnin A (455) | −99.2 | −116.1 | −101.3 | −118.5 | −92.4 | −108.1 | −101.4 | −118.6 | −99.2 | −116.0 | −100.4 | −117.5 | −101.7 | −119.0 |
| Cochinchinenin B (456) | −126.9 | −111.9 | −140.4 | −123.8 | −123.6 | −109.0 | −132.2 | −116.6 | −127.4 | −112.3 | −139.8 | −123.2 | −139.4 | −122.8 |
| Cochinchinenin C (457) | −127.2 | −112.2 | −132.9 | −117.1 | −123.3 | −108.7 | −121.7 | −107.3 | −132.0 | −116.3 | −130.5 | −115.0 | −146.7 | −129.3 |
| Drummondin D (458) | −87.0 | −79.0 | −125.2 | −113.7 | −112.3 | −102.0 | −121.0 | −109.9 | −112.3 | −101.9 | −113.2 | −102.8 | −120.9 | −109.7 |
| Drummondin E (459) | −112.1 | −101.7 | −129.4 | −117.4 | −107.8 | −97.7 | −125.8 | −114.1 | −117.4 | −106.5 | −122.4 | −111.0 | −126.4 | −114.6 |
| Eleutherol (460) | −86.3 | −99.2 | −94.1 | −108.3 | −87.5 | −100.7 | −94.4 | −108.5 | −90.6 | −104.2 | −90.8 | −104.5 | −90.6 | −104.2 |
| Ellagicacid (461) | −91.6 | −98.1 | −98.4 | −105.4 | −98.3 | −105.3 | −98.7 | −105.7 | −96.3 | −103.1 | −98.4 | −105.5 | −96.1 | −103.0 |
| Epicoccolide A (462) | −81.3 | −81.1 | −91.8 | −91.6 | −91.7 | −91.5 | −84.0 | −83.8 | −92.5 | −92.3 | −90.3 | −90.1 | −93.6 | −93.4 |
| Gibbilimbol A (464) | −86.3 | −100.9 | −102.8 | −120.2 | −91.4 | −106.8 | −91.9 | −107.5 | −96.1 | −112.4 | −91.3 | −106.8 | −92.8 | −108.5 |
| Gibbilimbol B (465) | −89.6 | −104.8 | −101.1 | −118.3 | −95.2 | −111.3 | −96.8 | −113.3 | −95.5 | −111.7 | −95.2 | −111.3 | −94.8 | −110.8 |
| Grifolin (466) | −101.2 | −105.5 | −124.0 | −129.2 | −106.3 | −110.7 | −119.4 | −124.4 | −109.3 | −113.9 | −120.1 | −125.2 | −116.4 | −121.3 |
| Hyperbrasilol A (467) | −18.6 | −16.2 | −131.4 | −114.0 | −113.4 | −98.4 | −127.0 | −110.2 | −115.4 | −100.1 | −87.5 | −76.0 | −123.3 | −107.0 |
| Hyperbrasilol B (468) | −104.7 | −91.7 | −133.0 | −116.5 | −108.6 | −95.1 | −125.6 | −110.1 | −114.7 | −100.5 | −111.4 | −97.6 | −127.3 | −111.5 |
| Hyperbrasilol C (469) | −110.4 | −96.6 | −131.6 | −115.1 | −108.6 | −95.1 | −115.9 | −101.4 | −117.1 | −102.5 | −111.8 | −97.9 | −120.6 | −105.5 |
| Isodrummondin D (470) | −105.0 | −95.4 | −122.9 | −111.6 | −108.6 | −98.6 | −113.1 | −102.7 | −116.4 | −105.7 | −115.6 | −105.0 | −120.1 | −109.0 |
| Isohyperbrasilol B (471) | −106.2 | −93.0 | −122.9 | −107.6 | −111.7 | −97.9 | −118.6 | −103.9 | −123.7 | −108.4 | −113.7 | −99.6 | −133.8 | −117.2 |
| Isouliginosin B (472) | −104.7 | −94.9 | −119.5 | −108.4 | −114.8 | −104.1 | −119.5 | −108.4 | −117.7 | −106.8 | −105.4 | −95.6 | −125.0 | −113.3 |
| Italipyrone (473) | −104.2 | −101.6 | −120.5 | −117.5 | −105.5 | −102.9 | −115.0 | −112.2 | −108.1 | −105.4 | −105.4 | −102.8 | −112.2 | −109.5 |
| Knerachelin A (474) | −100.6 | −108.0 | −115.2 | −123.7 | −103.7 | −111.3 | −108.7 | −116.7 | −101.5 | −109.0 | −107.3 | −115.1 | −108.7 | −116.7 |
| Knerachelin B (475) | −94.7 | −105.3 | −110.2 | −122.6 | −98.8 | −109.9 | −100.0 | −111.2 | −98.5 | −109.5 | −102.4 | −113.8 | −102.7 | −114.2 |
| Magnolol (476) | −95.7 | −106.9 | −108.5 | −121.3 | −93.0 | −103.9 | −110.6 | −123.6 | −100.3 | −112.1 | −99.1 | −110.7 | −102.0 | −114.0 |
| Myrtucommulone A (477) | −84.9 | −69.8 | −115.2 | −94.7 | −90.7 | −74.6 | −100.5 | −82.6 | −121.8 | −100.2 | −102.6 | −84.4 | −126.7 | −104.1 |
| Myrtucommulone B (478) | −103.5 | −99.8 | −99.8 | −96.2 | −98.5 | −94.9 | −111.2 | −107.2 | −103.3 | −99.6 | −98.7 | −95.2 | −98.5 | −94.9 |
| Obovatol (479) | −89.7 | −98.3 | −105.7 | −115.8 | −99.3 | −108.8 | −104.8 | −114.8 | −96.6 | −105.9 | −91.2 | −99.9 | −97.5 | −106.9 |
| Oenostacin (480) | −95.4 | −106.6 | −104.8 | −117.2 | −94.8 | −105.9 | −107.6 | −120.2 | −105.2 | −117.6 | −101.7 | −113.7 | −104.0 | −116.3 |
| Paeonol (481) | −75.7 | −99.0 | −77.8 | −101.8 | −71.1 | −93.0 | −75.4 | −98.6 | −76.6 | −100.1 | −76.9 | −100.5 | −74.7 | −97.6 |
| Perlatolic acid (482) | −106.6 | −100.4 | −129.6 | −122.1 | −116.4 | −109.7 | −125.7 | −118.4 | −127.9 | −120.5 | −132.0 | −124.3 | −126.8 | −119.4 |
| Plicatipyrone (483) | −81.3 | −78.2 | −106.0 | −101.9 | −96.2 | −92.5 | −101.4 | −97.4 | −103.8 | −99.8 | −98.5 | −94.7 | −105.4 | −101.3 |
| Propterol (484) | −85.2 | −98.0 | −98.1 | −112.8 | −91.6 | −105.3 | −87.6 | −100.8 | −93.5 | −107.5 | −93.0 | −107.0 | −91.5 | −105.2 |
| Pulverulentone B (485) | −80.3 | −93.1 | −92.6 | −107.3 | −88.6 | −102.8 | −91.1 | −105.7 | −94.1 | −109.2 | −95.8 | −111.1 | −90.5 | −104.9 |
| Quinquangulin (486) | −78.6 | −85.7 | −86.3 | −94.1 | −83.1 | −90.7 | −81.4 | −88.8 | −86.6 | −94.4 | −86.7 | −94.6 | −84.3 | −91.9 |
| Rhodomyrtone (487) | −94.0 | −88.7 | −111.4 | −105.1 | −82.5 | −77.9 | −100.9 | −95.2 | −110.7 | −104.4 | −107.0 | −101.0 | −109.5 | −103.3 |
| Rosmarinic acid (488) | −99.0 | −100.0 | −129.8 | −131.2 | −111.5 | −112.7 | −129.9 | −131.3 | −107.3 | −108.4 | −106.3 | −107.4 | −119.8 | −121.0 |
| Rubanthrone A (489) | −102.1 | −101.5 | −105.3 | −104.6 | −108.4 | −107.8 | −101.3 | −100.7 | −107.5 | −106.9 | −105.8 | −105.2 | −105.1 | −104.5 |
| Sampsone A (490) | −81.4 | −80.5 | −98.2 | −97.1 | −98.5 | −97.4 | −99.2 | −98.1 | −97.5 | −96.4 | −91.8 | −90.8 | −101.1 | −99.9 |
| Sarothralen B (491) | −98.2 | −85.3 | −140.1 | −121.7 | −106.3 | −92.3 | −128.7 | −111.9 | −132.2 | −114.9 | −122.5 | −106.5 | −118.2 | −102.7 |
| Sarothralen C (492) | −93.5 | −80.4 | −161.4 | −138.8 | −71.2 | −61.2 | −127.8 | −109.9 | −122.5 | −105.3 | −127.7 | −109.8 | −128.3 | −110.3 |
| Sarothralen D (493) | −106.3 | −91.4 | −146.7 | −126.1 | −110.6 | −95.1 | −121.5 | −104.5 | −135.8 | −116.8 | −120.5 | −103.6 | −130.0 | −111.7 |
| Shikonofuran C (494) | −98.2 | −99.3 | −130.8 | −132.4 | −105.6 | −106.9 | −121.1 | −122.6 | −111.4 | −112.8 | −108.0 | −109.3 | −125.1 | −126.6 |
| Shikonofuran D (495) | −101.0 | −103.6 | −125.6 | −128.8 | −102.0 | −104.6 | −120.8 | −123.9 | −109.4 | −112.2 | −103.9 | −106.6 | −118.8 | −121.9 |
| Shikonofuran E (496) | −101.2 | −102.6 | −130.8 | −132.7 | −112.7 | −114.3 | −124.0 | −125.8 | −108.1 | −109.6 | −105.6 | −107.1 | −123.7 | −125.5 |
| Sinapic acid (497) | −83.4 | −98.7 | −89.8 | −106.2 | −84.9 | −100.5 | −87.2 | −103.2 | −84.3 | −99.8 | −84.6 | −100.1 | −87.3 | −103.3 |
| Walrycin A (498) | −68.1 | −87.7 | −73.6 | −94.8 | −66.9 | −86.1 | −73.9 | −95.1 | −71.4 | −91.9 | −71.5 | −92.1 | −70.3 | −90.5 |
| Quinones | ||||||||||||||
| 2,6-Dimethoxy-1,4-benzo-quinone (336) | −66.9 | −87.2 | −69.3 | −90.2 | −67.6 | −88.1 | −69.2 | −90.2 | −68.0 | −88.5 | −66.9 | −87.1 | −68.3 | −89.0 |
| 2-Methyl-6-prenyl-1,4-benzo-quinone (337) | −80.4 | −100.6 | −83.4 | −104.3 | −78.6 | −98.3 | −84.0 | −105.0 | −82.9 | −103.6 | −81.0 | −101.3 | −85.9 | −107.4 |
| Omphalone (499) | −88.5 | −111.1 | −90.1 | −113.0 | −84.6 | −106.1 | −91.2 | −114.4 | −85.7 | −107.5 | −87.2 | −109.5 | −90.0 | −113.0 |
| Primin (500) | −82.1 | −99.6 | −91.4 | −110.9 | −80.5 | −97.6 | −88.5 | −107.3 | −88.3 | −107.1 | −87.2 | −105.7 | −93.6 | −113.5 |
| 1,4-Naphthoquinone (338) | −63.6 | −84.5 | −67.6 | −89.9 | −61.4 | −81.6 | −68.0 | −90.4 | −64.1 | −85.3 | −65.4 | −86.9 | −65.4 | −87.0 |
| 2-Acetylnaphtho[2,3-b]furan-4,9-dione (339) | −83.4 | −96.5 | −90.3 | −104.5 | −83.7 | −96.8 | −91.8 | −106.2 | −89.4 | −103.4 | −89.3 | −103.3 | −89.2 | −103.2 |
| Alkannin (340) | −88.7 | −96.6 | −104.5 | −113.8 | −93.5 | −101.8 | −104.8 | −114.1 | −95.9 | −104.3 | −92.8 | −101.0 | −99.1 | −107.8 |
| Isobutyrylshikonin (341) | −98.6 | −99.8 | −112.4 | −113.7 | −106.2 | −107.5 | −118.7 | −120.2 | −105.8 | −107.1 | −101.8 | −103.0 | −113.3 | −114.6 |
| Lapachol (501) | −83.9 | −96.8 | −91.8 | −105.9 | −84.5 | −97.5 | −91.8 | −105.8 | −88.8 | −102.4 | −85.4 | −98.5 | −92.6 | −106.8 |
| Mamegakinone (502) | −92.8 | −92.6 | −106.7 | −106.5 | −91.3 | −91.1 | −105.7 | −105.4 | −92.4 | −92.2 | −87.2 | −87.0 | −104.1 | −103.8 |
| Menadione (503) | −69.9 | −90.4 | −73.0 | −94.4 | −65.5 | −84.7 | −72.6 | −93.8 | −70.9 | −91.7 | −72.2 | −93.3 | −69.5 | −89.8 |
| Rhinacanthin C (504) | −114.9 | −111.1 | −128.5 | −124.3 | −112.3 | −108.7 | −122.9 | −118.9 | −114.2 | −110.4 | −114.3 | −110.6 | −116.1 | −112.3 |
| Rhinacanthin D (505) | −100.4 | −97.3 | −128.7 | −124.7 | −112.8 | −109.3 | −118.6 | −114.9 | −118.4 | −114.7 | −125.1 | −121.2 | −122.1 | −118.3 |
| Rhinacanthin G (506) | −115.2 | −110.0 | −136.0 | −129.9 | −116.2 | −111.0 | −123.7 | −118.2 | −122.9 | −117.4 | −123.3 | −117.7 | −118.1 | −112.8 |
| Rhinacanthin H (507) | −96.7 | −92.4 | −131.2 | −125.3 | −98.6 | −94.2 | −121.2 | −115.8 | −117.8 | −112.6 | −119.6 | −114.3 | −124.3 | −118.8 |
| Rhinacanthin I (508) | −116.4 | −111.2 | −130.9 | −125.0 | −109.9 | −105.0 | −124.2 | −118.6 | −128.3 | −122.5 | −121.9 | −116.5 | −133.2 | −127.2 |
| Rhinacanthin J (509) | −124.5 | −119.1 | −126.5 | −121.0 | −112.4 | −107.6 | −127.9 | −122.3 | −121.1 | −115.9 | −124.6 | −119.2 | −119.5 | −114.3 |
| Rhinacanthin K (510) | −98.9 | −93.2 | −130.3 | −122.8 | −121.4 | −114.3 | −117.3 | −110.5 | −123.0 | −115.9 | −118.7 | −111.8 | −119.1 | −112.2 |
| Rhinacanthin L (511) | −105.6 | −98.3 | −134.8 | −125.5 | −110.6 | −103.0 | −118.2 | −110.1 | −118.3 | −110.1 | −116.8 | −108.8 | −120.0 | −111.7 |
| Rhinacanthin M (512) | −110.0 | −110.8 | −117.2 | −118.0 | −99.6 | −100.3 | −110.8 | −111.5 | −105.3 | −106.0 | −105.4 | −106.1 | −115.7 | −116.4 |
| Shikonin acetate (513) | −95.0 | −98.9 | −109.3 | −113.7 | −101.2 | −105.3 | −114.2 | −118.8 | −103.7 | −107.9 | −98.8 | −102.8 | −108.8 | −113.2 |
| β,β-Dimethylacrylshikonin (514) | −100.5 | −100.6 | −121.3 | −121.5 | −107.0 | −107.1 | −125.7 | −125.9 | −109.9 | −110.0 | −107.7 | −107.8 | −121.4 | −121.5 |
| β-Hydroxyisovaleryshikonin (515) | −103.2 | −101.7 | −126.2 | −124.3 | −106.6 | −105.0 | −120.7 | −118.9 | −110.3 | −108.7 | −105.1 | −103.5 | −120.0 | −118.3 |
| 1-Hydroxy-3-hydroxymethyl-anthraquinone (516) | −84.8 | −96.2 | −93.8 | −106.4 | −85.2 | −96.7 | −92.1 | −104.5 | −96.1 | −109.0 | −93.7 | −106.3 | −88.5 | −100.4 |
| Aloeemodin (518) | −85.6 | −95.2 | −94.6 | −105.2 | −88.3 | −98.2 | −97.4 | −108.3 | −96.3 | −107.1 | −95.7 | −106.4 | −86.9 | −96.7 |
| Islandicin (519) | −77.5 | −86.2 | −91.0 | −101.2 | −84.9 | −94.4 | −92.9 | −103.3 | −90.8 | −101.0 | −93.8 | −104.3 | −79.3 | −88.2 |
| Newbouldiaquinone (521) | −82.2 | −80.6 | −101.6 | −99.6 | −99.3 | −97.4 | −93.5 | −91.7 | −93.8 | −92.0 | −93.9 | −92.1 | −95.0 | −93.2 |
| Newbouldiaquinone A (520) | −105.6 | −102.2 | −112.2 | −108.5 | −92.5 | −89.5 | −105.8 | −102.3 | −96.7 | −93.5 | −104.1 | −100.7 | −114.3 | −110.6 |
| Rhein (522) | −90.4 | −98.9 | −100.6 | −110.0 | −88.7 | −97.0 | −100.2 | −109.6 | −96.1 | −105.1 | −99.4 | −108.7 | −90.6 | −99.0 |
| 15,16-Dihydrotanshinone I (517) | −81.1 | −89.4 | −90.7 | −99.9 | −83.8 | −92.3 | −89.0 | −98.0 | −86.3 | −95.0 | −85.4 | −94.0 | −86.3 | −95.0 |
| Acetylene, Glucoside, and Other Miscellaneous Phytochemicals | ||||||||||||||
| 1,7-Diphenyl-4-(2-phenylethyl)-1-heptene-3,5-dione (530) | −99.6 | −98.6 | −127.4 | −126.1 | −115.0 | −113.9 | −114.4 | −113.3 | −127.2 | −126.0 | −114.1 | −113.0 | −130.7 | −129.5 |
| 1,7-Diphenyl-5-hepten-3-one (531) | −96.6 | −108.2 | −114.5 | −128.2 | −98.4 | −110.3 | −107.9 | −120.9 | −100.1 | −112.1 | −95.1 | −106.6 | −108.0 | −120.9 |
| 3′-Demothexycyclocurcumin (532) | −96.6 | −99.7 | −112.7 | −116.3 | −97.1 | −100.2 | −103.9 | −107.2 | −113.7 | −117.3 | −113.3 | −116.9 | −110.4 | −114.0 |
| 5,7-Dihydroxyphthalide (533) | −68.6 | −89.7 | −71.2 | −93.1 | −66.0 | −86.4 | −70.8 | −92.6 | −70.4 | −92.1 | −69.7 | −91.1 | −72.9 | −95.3 |
| 6-Methyl-4,5-dithia-2-octene (534) | −65.9 | −86.8 | −65.5 | −86.4 | −63.6 | −83.8 | −65.0 | −85.7 | −64.6 | −85.1 | −64.8 | −85.4 | −65.9 | −86.8 |
| 7-Epiclusianone (535) | −100.2 | −90.6 | −126.3 | −114.2 | −115.1 | −104.1 | −125.9 | −113.9 | −113.0 | −102.1 | −111.1 | −100.5 | −127.8 | −115.6 |
| Allamandin (536) | −82.3 | −87.6 | −94.9 | −101.0 | −96.5 | −102.7 | −86.3 | −91.9 | −99.6 | −106.0 | −93.4 | −99.4 | −88.8 | −94.5 |
| Allicin (537) | −65.3 | −86.1 | −65.4 | −86.3 | −62.6 | −82.6 | −64.1 | −84.5 | −64.8 | −85.5 | −62.9 | −83.0 | −66.7 | −87.9 |
| Amadannulen (538) | −92.0 | −91.6 | −112.5 | −112.1 | −98.4 | −97.9 | −111.5 | −111.0 | −114.0 | −113.5 | −108.4 | −108.0 | −111.7 | −111.2 |
| Anemonin (539) | −72.0 | −89.7 | −73.9 | −92.0 | −70.4 | −87.7 | −76.7 | −95.6 | −78.2 | −97.4 | −77.0 | −95.9 | −81.8 | −102.0 |
| Antibiotic CZ 34 (540) | −68.6 | −81.7 | −84.8 | −100.9 | −89.3 | −106.3 | −80.7 | −96.1 | −82.0 | −97.7 | −78.6 | −93.6 | −82.5 | −98.2 |
| Argutone (541) | −75.4 | −95.0 | −78.9 | −99.4 | −72.2 | −90.9 | −74.9 | −94.4 | −74.0 | −93.2 | −74.6 | −93.9 | −76.3 | −96.1 |
| Bakuchiol (542) | −87.3 | −98.8 | −106.6 | −120.7 | −92.0 | −104.1 | −98.3 | −111.3 | −99.6 | −112.7 | −91.8 | −103.9 | −97.5 | −110.4 |
| Brasiliensophyllic acid A (543) | −112.9 | −98.4 | −139.3 | −121.5 | −123.7 | −107.8 | −130.4 | −113.7 | −118.5 | −103.3 | −118.9 | −103.7 | −128.9 | −112.4 |
| Brasiliensophyllic acid C (544) | −114.6 | −99.1 | −130.8 | −113.1 | −121.7 | −105.3 | −137.5 | −118.9 | −122.0 | −105.5 | −127.2 | −110.0 | −126.8 | −109.6 |
| Centrolobin (545) | −96.7 | −102.5 | −106.5 | −112.8 | −102.0 | −108.1 | −103.4 | −109.6 | −108.1 | −114.6 | −102.6 | −108.8 | −101.9 | −108.0 |
| Chamone I (546) | −102.5 | −88.8 | −125.8 | −109.0 | −110.1 | −95.4 | −117.3 | −101.7 | −113.2 | −98.1 | −115.6 | −100.2 | −133.6 | −115.8 |
| Chamone II (547) | −98.9 | −85.8 | −132.4 | −114.9 | −115.7 | −100.4 | −115.7 | −100.4 | −116.8 | −101.3 | −110.5 | −95.9 | −130.6 | −113.3 |
| Champanone A (548) | −84.1 | −95.2 | −102.1 | −115.5 | −85.6 | −96.9 | −99.5 | −112.7 | −93.2 | −105.5 | −92.8 | −105.0 | −101.7 | −115.2 |
| Dhelwangin (549) | −87.3 | −103.4 | −89.4 | −105.8 | −83.4 | −98.7 | −86.6 | −102.5 | −90.7 | −107.4 | −88.9 | −105.2 | −89.5 | −105.9 |
| Garcinoic acid (550) | −108.9 | −104.0 | −134.6 | −128.5 | −113.4 | −108.3 | −129.4 | −123.6 | −135.3 | −129.2 | −127.3 | −121.5 | −132.0 | −126.0 |
| Ginkgolide A (551) | −69.1 | −66.9 | −100.9 | −97.8 | −90.7 | −87.9 | −74.5 | −72.2 | −97.6 | −94.6 | −96.3 | −93.3 | −90.3 | −87.5 |
| Guttiferone E (552) | −112.9 | −96.1 | −160.7 | −136.8 | −120.6 | −102.6 | −129.5 | −110.3 | −130.2 | −110.8 | −129.9 | −110.6 | −135.0 | −114.9 |
| Helipyrone B (553) | −89.4 | −95.4 | −99.0 | −105.5 | −89.2 | −95.2 | −99.3 | −105.9 | −92.5 | −98.6 | −89.7 | −95.7 | −91.1 | −97.1 |
| Helipyrone C (554) | −84.6 | −91.7 | −94.2 | −102.1 | −84.4 | −91.4 | −94.5 | −102.4 | −88.7 | −96.1 | −88.9 | −96.3 | −88.7 | −96.1 |
| Ialibinone A (555) | −87.2 | −89.4 | −97.5 | −100.0 | −96.2 | −98.7 | −94.8 | −97.2 | −99.2 | −101.7 | −97.9 | −100.4 | −101.4 | −104.0 |
| Ialibinone B (556) | −81.9 | −84.0 | −96.7 | −99.2 | −85.0 | −87.2 | −88.1 | −90.4 | −94.3 | −96.8 | −91.7 | −94.1 | −95.5 | −97.9 |
| Ialibinone C (557) | −86.9 | −88.0 | −99.4 | −100.6 | −98.4 | −99.6 | −96.9 | −98.1 | −103.1 | −104.3 | −97.1 | −98.2 | −100.4 | −101.6 |
| Ialibinone D (558) | −84.0 | −85.0 | −93.3 | −94.4 | −83.5 | −84.5 | −93.5 | −94.7 | −93.5 | −94.6 | −98.7 | −99.9 | −100.7 | −102.0 |
| Isobrasiliensophyllic acid A (559) | −99.1 | −86.4 | −120.1 | −104.7 | −122.1 | −106.5 | −122.0 | −106.4 | −102.3 | −89.2 | −119.5 | −104.2 | −110.8 | −96.6 |
| Moskachan C (560) | −87.4 | −103.8 | −90.9 | −107.9 | −87.2 | −103.4 | −92.3 | −109.6 | −94.8 | −112.5 | −91.3 | −108.4 | −91.3 | −108.4 |
| Nimbolide (561) | −107.4 | −99.5 | −123.1 | −114.1 | −95.1 | −88.2 | −113.7 | −105.4 | −125.7 | −116.6 | −119.8 | −111.0 | −111.7 | −103.5 |
| Pectinolide H (562) | −98.6 | −109.9 | −103.0 | −114.8 | −98.8 | −110.1 | −106.4 | −118.6 | −108.0 | −120.3 | −102.7 | −114.5 | −106.3 | −118.5 |
| Propolone A (563) | −98.9 | −89.5 | −127.3 | −115.1 | −108.3 | −97.9 | −106.5 | −96.3 | −115.3 | −104.3 | −102.1 | −92.3 | −120.4 | −108.8 |
| Sellovicine B (564) | −82.3 | −95.9 | −93.2 | −108.6 | −84.1 | −98.1 | −92.2 | −107.5 | −86.0 | −100.3 | −89.2 | −104.0 | −87.1 | −101.6 |
| Simonin A (565) | −84.4 | −81.2 | −109.6 | −105.6 | −104.3 | −100.4 | −105.6 | −101.6 | −117.0 | −112.7 | −106.3 | −102.4 | −111.2 | −107.1 |
| Tenulin (566) | −83.0 | −88.5 | −89.7 | −95.7 | −81.9 | −87.3 | −89.9 | −95.9 | −96.6 | −103.0 | −91.1 | −97.2 | −93.2 | −99.4 |
| Atractylodin (522) | −66.5 | −84.3 | −86.2 | −109.3 | −76.8 | −97.4 | −78.0 | −98.9 | −76.6 | −97.2 | −78.8 | −100.0 | −84.0 | −106.5 |
| Atractylodinol (523) | −75.1 | −92.6 | −90.3 | −111.4 | −83.6 | −103.1 | −82.7 | −101.9 | −81.2 | −100.1 | −87.5 | −107.8 | −89.7 | −110.6 |
| Capillene (342) | −65.5 | −87.8 | −71.6 | −96.0 | −69.3 | −92.9 | −70.8 | −94.9 | −72.6 | −97.4 | −74.9 | −100.4 | −72.1 | −96.7 |
| Peniophorin A (524) | −101.4 | −111.1 | −106.8 | −117.1 | −96.7 | −105.9 | −103.7 | −113.6 | −111.0 | −121.6 | −109.1 | −119.5 | −107.7 | −118.0 |
| Peniophorin B (525) | −84.8 | −102.2 | −93.2 | −112.3 | −88.4 | −106.6 | −94.5 | −113.9 | −87.2 | −105.1 | −85.5 | −103.0 | −89.8 | −108.3 |
| Thiarubrin A (526) | −70.3 | −82.7 | −85.5 | −100.6 | −77.6 | −91.2 | −76.4 | −89.8 | −73.7 | −86.7 | −77.2 | −90.8 | −74.7 | −87.9 |
| Arbutin (527) | −90.0 | −99.8 | −92.0 | −102.0 | −91.6 | −101.7 | −94.8 | −105.2 | −91.9 | −101.9 | −93.8 | −104.1 | −88.0 | −97.6 |
| Aucubin (528) | −99.3 | −101.7 | −104.7 | −107.2 | −103.7 | −106.2 | −111.0 | −113.6 | −101.2 | −103.6 | −94.7 | −97.0 | −103.6 | −106.1 |
| Diospyrodin (529) | −88.6 | −93.7 | −97.9 | −103.5 | −97.4 | −103.1 | −96.3 | −101.8 | −95.0 | −100.5 | −90.4 | −95.6 | −96.6 | −102.2 |
| Synthetic Inhibitors | ||||||||||||||
| 06-1467 [23] (567) | −111.0 | −114.0 | −122.7 | −125.9 | −113.7 | −116.8 | −129.8 | −133.2 | −107.4 | −110.3 | −109.5 | −112.4 | - | - |
| 64-1811 [23] (569) | −84.3 | −99.3 | −86.3 | −101.7 | −80.8 | −95.2 | −84.5 | −99.6 | −83.5 | −98.4 | −84.5 | −99.5 | - | - |
| 66-6976 [23] (568) | −91.1 | −101.5 | −101.4 | −112.9 | −92.8 | −103.3 | −91.1 | −101.4 | −99.1 | −110.4 | −97.6 | −108.7 | - | - |
| N-Hydroxy-2-(5-methylsulfanyl-1H-indol-3-yl)acetamide [86] (570) | −89.4 | −104.0 | −101.6 | −118.2 | −80.5 | −93.7 | −91.9 | −106.8 | −95.7 | −111.3 | −88.9 | −103.4 | - | - |
| 2-(1-Benzyl-5-bromoindol-3-yl)-N-hydroxyacetamide [86] (572) | −105.8 | −107.0 | −112.6 | −113.9 | −106.8 | −108.0 | −106.9 | −108.2 | −104.4 | −105.6 | −104.9 | −106.1 | - | - |
| 2-(3-Benzyl-5-bromoindol-1-yl)-N-hydroxyacetamide [86] (571) | −101.2 | −102.3 | −117.5 | −118.9 | −94.5 | −95.6 | −110.2 | −111.4 | −101.7 | −102.9 | −103.4 | −104.5 | - | - |
| Ligand | EcTopoIV | EcGyrB | MtGyrB | MtPtp | HsPtp | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | |
| Indole Alkaloids | ||||||||||
| 1-Hydroxy-6,7-dimethoxy-3-methylcarbazole (1) | −96.8 | −109.5 | −86.9 | −98.2 | −94.7 | −107.0 | −82.8 | −93.7 | −86.9 | −98.2 |
| 11-Methoxytubotaiwine (2) | −75.7 | −76.9 | −95.3 | −96.8 | −89.7 | −91.2 | −92.5 | −94.0 | −79.2 | −80.5 |
| 12-Methoxy-4-methylvoachalotine (3) | −94.0 | −90.9 | −76.4 | −73.8 | −80.1 | −77.5 | −98.9 | −95.6 | −38.4 | −37.1 |
| 3-Prenylindole (4) | −83.9 | −105.9 | −81.5 | −102.8 | −89.2 | −112.5 | −75.1 | −94.7 | −78.6 | −99.1 |
| Affinisine (5) | −102.0 | −108.5 | −76.4 | −81.3 | −103.2 | −109.8 | −82.9 | −88.2 | −74.1 | −78.8 |
| Apparicine (6) | −89.6 | −100.3 | −80.7 | −90.4 | −84.7 | −94.9 | −83.6 | −93.6 | −70.4 | −78.9 |
| Aristolactam I (7) | −103.7 | −112.2 | −93.6 | −101.3 | −100.5 | −108.8 | −93.8 | −101.5 | −88.4 | −95.6 |
| Clausenawalline A (8) a | −145.2 | −126.9 | −86.8 | −75.9 | −114.5 | −100.1 | −103.2 | −90.2 | −60.2 | −52.6 |
| Cryptoheptine (9) | −88.8 | −99.7 | −72.6 | −81.5 | −91.4 | −102.6 | −75.6 | −84.9 | −92.6 | −104.1 |
| Diploceline (10) | −105.2 | −105.4 | −82.8 | −83.0 | −97.7 | −97.9 | −90.2 | −90.4 | −50.8 | −50.9 |
| Discarine B (11) | −49.3 | −42.6 | −103.6 | −89.7 | −118.8 | −102.8 | −130.1 | −112.6 | −44.8 | −38.8 |
| Ibogamine (12) | −85.6 | −94.0 | −76.6 | −84.1 | −85.8 | −94.2 | −78.7 | −86.5 | −83.1 | −91.2 |
| Iboxygaine (13) | −91.8 | −95.9 | −76.2 | −79.6 | −94.5 | −98.7 | −81.4 | −85.0 | −83.7 | −87.4 |
| Isovoacangine (14) | −98.8 | −99.1 | −87.3 | −87.5 | −94.6 | −94.9 | −94.2 | −94.4 | −81.2 | −81.4 |
| Rugosanine B (15) | −118.2 | −99.6 | −93.1 | −78.4 | −64.4 | −54.3 | −137.2 | −115.5 | −91.8 | −77.3 |
| Suaveolindole (16) | −111.9 | −113.8 | −101.6 | −103.3 | −106.5 | −108.3 | −98.8 | −100.5 | −99.0 | −100.6 |
| Toussaintine B (17) | −99.8 | −107.2 | −85.1 | −91.4 | −99.9 | −107.4 | −87.5 | −94.1 | −85.4 | −91.8 |
| Isoquinoline Alkaloids | ||||||||||
| 8-Acetonyldihydroavicine (18) | −117.3 | −113.9 | −102.1 | −99.2 | −115.4 | −112.1 | −111.7 | −108.5 | −89.6 | −87.0 |
| 8-Acetonyldihydronitidine (19) | −114.9 | −113.1 | −88.0 | −86.7 | −113.8 | −112.0 | −95.3 | −93.8 | −86.9 | −85.6 |
| Antofine (20) | −116.6 | −117.4 | −95.8 | −96.5 | −114.3 | −115.2 | −87.6 | −88.3 | −83.5 | −84.1 |
| Berbamine (24) | no dock | no dock | −59.9 | −50.9 | no dock | no dock | −106.8 | −90.6 | −57.1 | −48.5 |
| Berberine (21) | −117.4 | −121.4 | −98.5 | −101.9 | −109.1 | −112.8 | −83.6 | −86.4 | −44.3 | −45.8 |
| Bisnorthalphenine (22) | −103.0 | −107.9 | −84.2 | −88.2 | −102.3 | −107.2 | −85.5 | −89.6 | −72.6 | −76.1 |
| Cepharanthine (25) | no dock | no dock | −91.9 | −78.1 | no dock | no dock | −130.5 | −110.8 | −62.9 | −53.4 |
| Cryptopleurine (23) | −118.7 | −118.1 | −88.5 | −88.1 | −113.6 | −113.0 | −86.4 | −86.0 | −85.3 | −84.8 |
| Emetine (26) | −127.8 | −117.3 | −92.3 | −84.7 | −119.6 | −109.8 | −106.9 | −98.1 | −75.6 | −69.4 |
| Hydrastine (27) | −127.0 | −125.7 | −97.5 | −96.5 | −124.8 | −123.5 | −96.8 | −95.8 | −76.1 | −75.3 |
| Isotrilobine (29) | −77.7 | −67.1 | −72.7 | −62.8 | −45.7 | −39.5 | −121.6 | −105.0 | −56.9 | −49.1 |
| Jatrorrhizine (28) | −110.6 | −114.1 | −97.1 | −100.1 | −107.4 | −110.8 | −79.2 | −81.7 | −60.3 | −62.2 |
| Lauroscholtzine (31) | −104.5 | −107.5 | −80.3 | −82.6 | −106.8 | −109.9 | −83.0 | −85.4 | −66.5 | −68.4 |
| Methothalistyline (30) | −17.3 | −13.9 | −70.6 | −56.8 | no dock | no dock | −123.1 | −99.1 | −50.7 | −40.8 |
| N-Demethylthalphenine (32) | −105.1 | −108.6 | −83.2 | −85.9 | −102.2 | −105.6 | −80.5 | −83.2 | −42.3 | −43.7 |
| Obamegine (34) | no dock | no dock | −60.6 | −51.8 | no dock | no dock | −105.8 | −90.5 | −59.9 | −51.2 |
| Oxyacanthine (35) | −42.8 | −36.3 | −65.6 | −55.7 | no dock | no dock | −110.2 | −93.5 | −54.6 | −46.3 |
| Pennsylvanine (36) | −145.7 | −119.0 | −113.8 | −92.9 | no dock | no dock | −116.7 | −95.3 | −81.9 | −66.8 |
| Thaliadanine (38) | −127.9 | −102.9 | −116.0 | −93.2 | −57.1 | −45.9 | −131.9 | −106.0 | −82.4 | −66.3 |
| Thalicarpine (37) | −141.3 | −114.6 | −89.6 | −72.7 | −14.4 | −11.6 | −131.6 | −106.8 | −45.1 | −36.6 |
| Thalidasine (39) | no dock | no dock | −73.1 | −60.6 | no dock | no dock | −98.4 | −81.5 | −47.3 | −39.2 |
| Thalistyline (40) | −74.8 | −60.6 | −98.4 | −79.7 | no dock | no dock | −114.6 | −92.9 | −77.3 | −62.6 |
| Thalmelatine (41) | −142.7 | −116.5 | −97.6 | −79.7 | −45.6 | −37.3 | −110.6 | −90.3 | −91.4 | −74.6 |
| Thalmirabine (42) | no dock | no dock | −93.9 | −77.2 | no dock | no dock | −97.7 | −80.3 | −48.5 | −39.9 |
| Thalphenine (33) | −105.2 | −107.1 | −80.6 | −82.1 | −100.3 | −102.1 | −83.6 | −85.1 | −48.2 | −49.1 |
| Thalrugosidine (43) | no dock | no dock | −84.6 | −70.6 | no dock | no dock | −92.2 | −77.0 | −58.8 | −49.1 |
| Thalrugosine (44) | no dock | no dock | −57.5 | −48.7 | no dock | no dock | −102.8 | −87.2 | −59.7 | −50.7 |
| Piperidine, Pyrrole, Pyrrolizidine, Quinoline, and Steroidal Alkaloids | ||||||||||
| Aconicaramide (46) | −98.8 | −117.3 | −83.8 | −99.5 | −101.1 | −120.1 | −78.1 | −92.7 | −91.8 | −108.9 |
| Lasiocarpine (47) | −117.0 | −113.1 | −92.3 | −89.2 | −121.7 | −117.6 | −105.2 | −101.7 | −87.4 | −84.5 |
| Lasiocarpine N-oxide (48) | −120.3 | −114.8 | −79.4 | −75.8 | −123.5 | −117.9 | −102.5 | −97.9 | −71.1 | −67.9 |
| Piperine (45) | −109.9 | −120.0 | −93.3 | −101.9 | −100.9 | −110.2 | −86.5 | −94.5 | −89.3 | −97.6 |
| 4-Methoxy-1-methyl-2(1H)-quinolinone (49) | −76.4 | −95.7 | −65.4 | −81.9 | −77.4 | −97.0 | −61.3 | −76.8 | −64.6 | −80.9 |
| Cryptolepine (50) | −91.9 | −107.4 | −79.4 | −92.8 | −83.7 | −97.9 | −77.5 | −90.7 | −81.9 | −95.8 |
| Neocryptolepine (51) | −90.7 | −106.1 | −80.9 | −94.6 | −83.6 | −97.8 | −68.2 | −79.8 | −81.0 | −94.7 |
| Pteleine (52) | −98.4 | −115.6 | −82.2 | −96.6 | −93.1 | −109.4 | −73.4 | −86.3 | −79.9 | −93.9 |
| Veprisinium (53) | −101.5 | −105.2 | −56.8 | −58.9 | −97.3 | −100.8 | −81.9 | −84.8 | −74.0 | −76.7 |
| Conessine (54) | −100.0 | −101.4 | −70.6 | −71.5 | −90.6 | −91.9 | −84.6 | −85.8 | −58.1 | −58.9 |
| Irehdiamine A (55) | −102.9 | −108.6 | −86.4 | −91.2 | −99.9 | −105.4 | −78.9 | −83.3 | −25.3 | −26.7 |
| Solacassine (56) | −89.4 | −84.3 | −28.5 | −26.9 | −65.6 | −61.9 | −87.3 | −82.4 | −83.2 | −78.5 |
| Solanocapsine (57) | −95.8 | −91.2 | −74.5 | −70.9 | −85.6 | −81.5 | −83.0 | −79.1 | −52.8 | −50.3 |
| Tomatidine (58) | −101.5 | −97.8 | −70.7 | −68.1 | −89.2 | −86.0 | −63.6 | −61.3 | −61.9 | −59.7 |
| Miscellaneous Alkaloids | ||||||||||
| 2-(Methoxyamino)-4H-1-benzopyran-3,4,5,7-tetrol (59) | −87.0 | −100.5 | −75.4 | −87.0 | −89.0 | −102.8 | −86.9 | −100.4 | −86.0 | −99.4 |
| Abyssenine C (60) | −126.9 | −119.6 | −93.9 | −88.5 | −99.7 | −93.9 | −106.0 | −99.8 | −64.9 | −61.2 |
| Amphibine H (61) | −95.6 | −81.2 | −66.1 | −56.2 | −66.7 | −56.7 | −132.3 | −112.4 | −99.4 | −84.5 |
| Cepharatine A (62) | −88.6 | −93.8 | −78.0 | −82.6 | −89.8 | −95.0 | −75.9 | −80.3 | −68.5 | −72.5 |
| Curcamide (63) | −107.4 | −123.2 | −99.4 | −114.0 | −107.0 | −122.8 | −88.9 | −102.0 | −89.9 | −103.2 |
| Drodrenin (64) | −154.5 | −139.4 | −128.6 | −116.1 | −156.5 | −141.2 | −120.7 | −109.0 | −102.5 | −92.5 |
| Eschscholtzidine (65) | −117.3 | −120.9 | −81.7 | −84.2 | −108.4 | −111.7 | −89.9 | −92.7 | −83.8 | −86.4 |
| Jervine (66) | −88.0 | −84.1 | −95.1 | −90.9 | −49.8 | −47.6 | −90.7 | −86.7 | −31.3 | −29.9 |
| Matrine (67) | −87.0 | −99.5 | −76.3 | −87.3 | −81.9 | −93.7 | −73.8 | −84.4 | −75.9 | −86.8 |
| Mucronine H (68) | −99.1 | −91.1 | −114.9 | −105.6 | −98.2 | −90.3 | −108.2 | −99.5 | −61.7 | −56.7 |
| N-Benzoylmescaline (69) | −120.9 | −127.7 | −107.5 | −113.6 | −122.1 | −129.0 | −94.6 | −99.9 | −85.5 | −90.4 |
| Nummularine B (70) | −83.6 | −71.6 | −123.6 | −105.9 | −62.9 | −53.9 | −133.1 | −114.0 | −103.8 | −88.9 |
| Nummularine S (71) | −116.4 | −104.1 | −111.3 | −99.4 | −139.9 | −125.1 | −109.6 | −97.9 | −95.8 | −85.6 |
| Scutianine B (72) | −133.8 | −116.1 | −103.3 | −89.6 | −55.9 | −48.5 | −130.4 | −113.2 | −96.9 | −84.1 |
| Shahidine (73) | −115.2 | −126.7 | −94.7 | −104.2 | −119.6 | −131.6 | −87.7 | −96.5 | −88.2 | −97.0 |
| Thaliglucinone (74) | −118.3 | −119.0 | −116.5 | −117.2 | −119.9 | −120.5 | −98.3 | −98.9 | −83.1 | −83.6 |
| Triisopenylguanidine (75) | −109.6 | −122.0 | −100.4 | −111.7 | −105.1 | −117.0 | −89.1 | −99.2 | −91.9 | −102.3 |
| Tuberine (76) | −126.0 | −116.6 | −112.6 | −104.1 | −114.3 | −105.8 | −109.6 | −101.4 | −80.2 | −74.2 |
| Monoterpenoids | ||||||||||
| Linalool (77) | −75.4 | −101.1 | −70.3 | −94.3 | −74.4 | −99.8 | −66.2 | −88.7 | −77.1 | −103.3 |
| Thymol (78) | −70.3 | −95.1 | −64.1 | −86.7 | −69.5 | −94.0 | −58.9 | −79.7 | −65.0 | −88.0 |
| Thymoquinol (79) | −75.1 | −98.2 | −66.1 | −86.5 | −73.9 | −96.7 | −67.7 | −88.6 | −67.1 | −87.7 |
| β-Dolabrin (80) | −74.8 | −98.7 | −66.0 | −87.0 | −81.8 | −107.9 | −70.5 | −92.9 | −78.1 | −103.0 |
| β-Thujaplicin (81) | −80.3 | −105.5 | −63.2 | −82.9 | −81.2 | −106.6 | −72.0 | −94.5 | −76.9 | −101.0 |
| Sesquiterpenoids | ||||||||||
| 11,13-Dehydroeriolin (82) | −83.6 | −93.7 | −72.6 | −81.3 | −88.5 | −99.1 | −79.4 | −89.0 | −70.7 | −79.2 |
| 2,10-Bisaboladien-1-one (83) | −87.3 | −104.0 | −91.8 | −109.3 | −89.1 | −106.1 | −78.7 | −93.6 | −78.0 | −92.8 |
| 2-Hydroxycalamenene (84) | −73.5 | −87.8 | −68.8 | −82.2 | −68.3 | −81.6 | −71.6 | −85.5 | −64.3 | −76.7 |
| 2-Methoxyfurano-9-guaien-8-one (85) | −92.6 | −104.2 | −86.5 | −97.4 | −88.3 | −99.4 | −82.9 | −93.3 | −79.4 | −89.5 |
| 4α,10α-Dihydroxy-1,11(13)guaiadien-12,8-olide (93) | −86.6 | −97.1 | −79.9 | −89.6 | −86.1 | −96.5 | −80.0 | −89.6 | −63.9 | −71.6 |
| 4α,10β-Dihydroxy-1,11(13)guaiadien-12,8-olide (89) | −42.4 | −47.5 | −79.9 | −89.5 | −75.9 | −85.0 | −84.0 | −94.1 | −67.5 | −75.6 |
| Alantolactone (86) | −80.0 | −93.6 | −67.5 | −78.9 | −80.6 | −94.3 | −71.5 | −83.6 | −54.3 | −63.6 |
| Alliacol A (87) | −76.6 | −85.8 | −72.3 | −81.0 | −79.6 | −89.2 | −73.9 | −82.8 | −56.6 | −63.4 |
| Alliacol B (88) | −81.2 | −91.0 | −73.9 | −82.8 | −86.6 | −97.0 | −78.0 | −87.4 | −55.4 | −62.0 |
| Artemisinic acid (113) | −100.5 | −117.1 | −75.6 | −88.1 | −105.8 | −123.4 | −79.4 | −92.6 | −76.5 | −89.2 |
| Baileyolin (90) | −104.9 | −105.8 | −96.9 | −97.7 | −90.2 | −90.9 | −84.1 | −84.9 | −86.1 | −86.9 |
| Bilobalide A (91) | −89.9 | −93.9 | −83.7 | −87.4 | −82.1 | −85.7 | −87.6 | −91.5 | −62.7 | −65.5 |
| Confertin (92) | −89.3 | −102.1 | −83.6 | −95.7 | −96.2 | −110.0 | −76.3 | −87.3 | −68.3 | −78.1 |
| Cyperenal (94) | −59.6 | −71.2 | −61.1 | −72.9 | −64.6 | −77.1 | −63.5 | −75.8 | −45.2 | −54.0 |
| Cyperenol (95) | −62.2 | −74.1 | −61.8 | −73.5 | −63.5 | −75.6 | −65.3 | −77.7 | −47.9 | −57.0 |
| Furanodienone (97) | −86.5 | −101.4 | −80.0 | −93.9 | −85.0 | −99.7 | −81.0 | −95.0 | −67.6 | −79.2 |
| Ganodermycin (96) | −109.3 | −120.1 | −93.1 | −102.3 | −111.4 | −122.4 | −106.3 | −116.7 | −104.1 | −114.4 |
| Helenalin (98) | −83.6 | −93.9 | −75.7 | −85.0 | −87.3 | −98.0 | −76.9 | −86.4 | −67.5 | −75.8 |
| Hydrogrammic acid (99) | −93.0 | −104.7 | −77.9 | −87.7 | −85.0 | −95.8 | −88.9 | −100.1 | −51.5 | −58.0 |
| Isoalantolactone (100) | −73.3 | −85.7 | −62.6 | −73.3 | −78.9 | −92.3 | −70.7 | −82.7 | −53.3 | −62.3 |
| Ivaxillin (101) | −83.2 | −93.0 | −77.4 | −86.5 | −88.7 | −99.2 | −79.9 | −89.3 | −53.1 | −59.3 |
| Petrovin A (102) | −78.8 | −90.1 | −78.0 | −89.3 | −76.8 | −87.9 | −79.6 | −91.0 | −54.1 | −61.9 |
| Petrovin B (103) | −81.7 | −93.3 | −81.4 | −92.9 | −96.3 | −109.8 | −74.4 | −84.9 | −82.9 | −94.6 |
| Polygodial (104) | −83.2 | −97.0 | −79.9 | −93.2 | −84.0 | −98.0 | −73.7 | −86.0 | −22.2 | −25.9 |
| Rishitin (105) | −81.4 | −96.6 | −72.7 | −86.3 | −82.1 | −97.5 | −76.5 | −90.8 | −83.0 | −98.5 |
| Xanthorrhizol (106) | −85.4 | −102.0 | −87.4 | −104.4 | −85.6 | −102.2 | −81.7 | −97.6 | −82.4 | −98.3 |
| α-Amorphene (107) | −85.8 | −104.8 | −66.2 | −80.8 | −82.0 | −100.1 | −66.2 | −80.9 | −50.6 | −61.8 |
| α-Cadinene (108) | −74.0 | −90.4 | −68.0 | −83.0 | −68.2 | −83.2 | −66.4 | −81.1 | −76.0 | −92.8 |
| α-Copaene (110) | −70.2 | −87.7 | −59.9 | −74.9 | −70.3 | −87.8 | −65.8 | −82.2 | −52.9 | −66.1 |
| α-Muurolene (109) | −77.1 | −94.1 | −69.5 | −84.9 | −71.0 | −86.7 | −71.7 | −87.6 | −65.5 | −80.0 |
| γ-Cadinene (112) | −77.7 | −94.8 | −67.1 | −82.0 | −80.6 | −98.4 | −63.8 | −77.9 | −59.0 | −72.1 |
| Diterpenoids | ||||||||||
| 1,12-Diacetyljativatriol (114) | −76.8 | −74.6 | −82.2 | −79.9 | −64.7 | −62.9 | −98.0 | −95.3 | −36.1 | −35.1 |
| 12-Oxo-3,13(16)-clerodadien-15-oic acid (115) | −97.1 | −102.2 | −104.6 | −110.1 | −103.2 | −108.7 | −93.7 | −98.7 | −86.1 | −90.6 |
| 12-Oxo-8,13(16)-clerodadien-15-oic acid (116) | −108.4 | −114.1 | −99.0 | −104.2 | −104.1 | −109.6 | −98.8 | −104.1 | −82.1 | −86.4 |
| 13-Epimanoyl oxide (117) | −71.9 | −78.0 | −67.1 | −72.8 | −81.1 | −88.0 | −72.9 | −79.1 | −43.0 | −46.7 |
| 13-Episclareol (118) | −92.2 | −98.1 | −94.2 | −100.2 | −87.7 | −93.3 | −87.4 | −93.0 | −61.2 | −65.1 |
| 3,4-Seco-4(18)-trachyloben-3-oic acid (120) | −87.4 | −93.7 | −75.1 | −80.5 | −86.4 | −92.5 | −79.6 | −85.3 | −66.5 | −71.3 |
| 3-Hydroxytotarol (119) | −90.5 | −96.9 | −75.9 | −81.3 | −87.6 | −93.9 | −81.5 | −87.3 | −73.6 | −78.8 |
| 7,13-Labdadien-15-ol acetate (121) | −106.6 | −110.6 | −94.9 | −98.4 | −111.9 | −116.1 | −97.3 | −101.0 | −86.2 | −89.5 |
| 7,13-Labdadien-15-ol malonate (122) | −122.6 | −122.1 | −88.7 | −88.4 | −115.6 | −115.1 | −106.2 | −105.8 | −82.4 | −82.1 |
| Acetylcrinipellin A (125) | −105.4 | −105.4 | −72.1 | −72.0 | −74.1 | −74.1 | −96.4 | −96.3 | −50.4 | −50.4 |
| Aethiopinone (123) | −101.0 | −108.9 | −96.9 | −104.5 | −106.0 | −114.3 | −82.1 | −88.6 | −86.2 | −92.9 |
| Andrographolide (124) | −100.6 | −102.6 | −90.9 | −92.7 | −110.8 | −113.0 | −98.4 | −100.3 | −97.9 | −99.9 |
| Biflorin (126) | −104.5 | −113.0 | −91.7 | −99.1 | −100.8 | −108.9 | −89.2 | −96.4 | −91.2 | −98.6 |
| Continentalic acid (127) | −77.1 | −82.6 | −53.9 | −57.7 | −85.3 | −91.4 | −81.8 | −87.6 | no dock | no dock |
| Crinipellin A (129) | −82.2 | −85.5 | −81.9 | −85.1 | −93.9 | −97.6 | −93.5 | −97.2 | −69.9 | −72.7 |
| Cryptobeilic acid A (128) | −118.0 | −124.5 | −98.9 | −104.4 | −114.2 | −120.5 | −97.6 | −103.0 | −65.6 | −69.3 |
| Cryptobeilic acid C (130) | −129.4 | −130.3 | −108.7 | −109.5 | −128.7 | −129.5 | −99.4 | −100.1 | −62.3 | −62.8 |
| Cryptobeilic acid D (131) | −120.3 | −129.2 | −100.7 | −108.1 | −122.0 | −130.9 | −99.2 | −106.5 | −89.2 | −95.8 |
| Effusanin A (132) | −49.0 | −50.1 | −73.9 | −75.6 | −47.5 | −48.5 | −80.5 | −82.2 | −39.7 | −40.5 |
| Effusanin B (133) | −73.0 | −71.8 | −58.5 | −57.6 | −40.4 | −39.8 | −84.6 | −83.2 | no dock | no dock |
| Effusanin C (134) | −64.2 | −62.4 | −77.6 | −75.3 | −33.6 | −32.6 | −95.8 | −93.0 | −51.2 | −49.7 |
| Effusanin D (135) | −75.2 | −70.6 | −73.7 | −69.3 | −63.7 | −59.8 | −101.7 | −95.5 | −51.9 | −48.7 |
| Effusanin E (136) | −46.4 | −46.7 | −69.0 | −69.5 | −31.3 | −31.5 | −78.0 | −78.5 | −18.3 | −18.4 |
| Grandiflorenic acid (137) | −60.0 | −64.4 | −59.6 | −64.0 | −73.7 | −79.2 | −73.4 | −78.8 | −35.3 | −37.8 |
| Haplociliatic acid (138) | −99.3 | −102.7 | −94.1 | −97.2 | −105.1 | −108.6 | −110.8 | −114.5 | −92.2 | −95.3 |
| Hypargenin A (139) | −84.2 | −87.6 | −89.5 | −93.1 | −84.5 | −87.9 | −80.0 | −83.2 | −75.5 | −78.5 |
| Hypargenin B (140) | −82.2 | −86.7 | −85.9 | −90.7 | −88.6 | −93.4 | −82.6 | −87.2 | −91.9 | −96.9 |
| Hypargenin D (141) | −91.4 | −98.4 | −94.0 | −101.2 | −91.0 | −97.9 | −77.5 | −83.3 | −26.9 | −29.0 |
| Hypargenin F (142) | −96.9 | −100.7 | −73.0 | −76.0 | −87.2 | −90.7 | −76.0 | −79.0 | −27.4 | −28.5 |
| Isodomedin (143) | −70.3 | −69.1 | −64.6 | −63.5 | −80.5 | −79.0 | −93.9 | −92.2 | no dock | no dock |
| Kamebanin (144) | −65.8 | −68.1 | −67.3 | −69.7 | −60.0 | −62.1 | −79.4 | −82.2 | no dock | no dock |
| Lasiokaurin (145) | −63.1 | −61.1 | −73.1 | −70.8 | −52.7 | −51.0 | −86.0 | −83.4 | no dock | no dock |
| Longikaurin A (146) | −56.8 | −58.1 | −56.4 | −57.6 | −61.1 | −62.4 | −76.1 | −77.8 | −36.8 | −37.6 |
| Longikaurin B (147) | −71.1 | −69.0 | −72.4 | −70.3 | −21.4 | −20.7 | −94.1 | −91.3 | −57.5 | −55.8 |
| Longikaurin C (148) | −67.0 | −65.9 | −76.5 | −75.3 | −22.4 | −22.0 | −96.8 | −95.2 | −51.2 | −50.4 |
| Longikaurin D (149) | −76.1 | −73.8 | −77.1 | −74.8 | −51.8 | −50.3 | −98.5 | −95.6 | −58.0 | −56.3 |
| Longikaurin E (150) | −61.6 | −62.9 | −62.6 | −63.9 | −41.2 | −42.1 | −77.2 | −78.9 | −53.5 | −54.6 |
| Longikaurin F (151) | −83.9 | −78.8 | −88.5 | −83.1 | −72.8 | −68.4 | −106.9 | −100.4 | −73.6 | −69.1 |
| Longikaurin G (152) | −64.3 | −64.8 | −49.3 | −49.6 | −27.9 | −28.1 | −76.3 | −76.8 | −17.5 | −17.6 |
| Lupulin E (153) | −104.1 | −92.9 | −64.9 | −58.0 | −94.7 | −84.5 | −117.7 | −105.1 | −83.1 | −74.2 |
| Lupulin F (154) | −75.2 | −67.0 | −75.4 | −67.2 | −93.3 | −83.2 | −117.1 | −104.4 | −71.0 | −63.3 |
| Methyl seconidoresedate (155) | −103.9 | −108.2 | −94.7 | −98.7 | −107.1 | −111.6 | −96.7 | −100.8 | −82.8 | −86.3 |
| Pisiferol (156) | −60.9 | −65.3 | −93.6 | −100.2 | −84.6 | −90.6 | −76.3 | −81.7 | −78.5 | −84.0 |
| Salvic acid (157) | −90.6 | −94.9 | −84.1 | −88.1 | −99.2 | −104.0 | −93.3 | −97.9 | −90.2 | −94.6 |
| Salvic acid acetate (158) | −107.8 | −108.5 | −85.4 | −86.0 | −113.4 | −114.2 | −94.9 | −95.6 | −87.9 | −88.4 |
| Shikokianin (159) | −72.3 | −67.9 | −68.7 | −64.5 | −12.7 | −11.9 | −95.0 | −89.2 | −15.9 | −15.0 |
| Strictic acid (160) | −85.4 | −90.3 | −75.1 | −79.4 | −99.8 | −105.6 | −108.1 | −114.3 | −67.3 | −71.2 |
| Taxodione (161) | −93.6 | −99.0 | −92.8 | −98.1 | −90.6 | −95.8 | −82.5 | −87.3 | −40.1 | −42.4 |
| Trichodonin (162) | −11.3 | −11.0 | −75.1 | −73.0 | −36.5 | −35.5 | −90.5 | −88.0 | −60.6 | −59.0 |
| Umbrosin A (163) | −57.6 | −59.6 | −68.5 | −71.0 | −59.6 | −61.7 | −77.0 | −79.7 | no dock | no dock |
| Umbrosin B (164) | −69.2 | −71.8 | −42.6 | −44.2 | −69.1 | −71.7 | −78.2 | −81.1 | no dock | no dock |
| Yuexiandajisu A (165) | −82.8 | −87.2 | −73.8 | −77.8 | −65.3 | −68.7 | −90.4 | −95.2 | −52.3 | −55.1 |
| Triterpenoids | ||||||||||
| Alisol A 24-acetate (166) | −102.3 | −90.7 | −77.4 | −68.6 | −45.9 | −40.7 | −101.7 | −90.2 | −49.3 | −43.7 |
| Alisol B 23-acetate (167) | −82.2 | −73.8 | −102.3 | −91.8 | −80.5 | −72.2 | −117.7 | −105.6 | 19.5 | 17.5 |
| Betulinic acid (168) | −26.4 | −24.7 | −75.5 | −70.5 | −34.3 | −32.1 | −101.3 | −94.5 | −25.6 | −23.9 |
| Entagenic acid (169) | no dock | no dock | no dock | no dock | no dock | no dock | −99.2 | −90.6 | 33.3 | 30.4 |
| Lantic acid (170) | −21.3 | −19.7 | −76.7 | −70.9 | no dock | no dock | −89.7 | −82.9 | 16.4 | 15.2 |
| Mahmoodin (171) | no dock | no dock | −73.9 | −65.8 | no dock | no dock | −125.8 | −112.0 | −10.2 | −9.1 |
| Maslinic acid (172) | −59.5 | −54.9 | −63.1 | −58.2 | no dock | no dock | −105.2 | −97.1 | −32.2 | −29.7 |
| Oleanolic acid (173) | −60.7 | −56.7 | −79.9 | −74.6 | no dock | no dock | −100.3 | −93.6 | −14.1 | −13.2 |
| Pristimerin (174) | −35.5 | −33.0 | −81.1 | −75.3 | −25.4 | −23.6 | −82.3 | −76.4 | −54.2 | −50.3 |
| Rubrinol (175) | no dock | no dock | −84.3 | −79.5 | no dock | no dock | −99.2 | −93.5 | −28.9 | −27.2 |
| Tingenone (176) | −37.6 | −36.0 | −68.2 | −65.5 | −24.1 | −23.1 | −90.7 | −87.1 | −57.6 | −55.2 |
| Chalcones | ||||||||||
| 1-(2,6-Dihydroxy-4-methoxyphenyl)-3-phenyl-1-propanone (177) | −112.2 | −124.4 | −97.3 | −107.9 | −112.3 | −124.6 | −82.8 | −91.9 | −87.7 | −97.3 |
| 2′-Hydroxy-2,3,4′,6′-tetramethoxychalcone (178) | −127.2 | −130.4 | −92.7 | −95.0 | −127.3 | −130.6 | −91.9 | −94.3 | −84.8 | −87.0 |
| 3′′′′,5′′′,5′′′′′-Tribenzyl-2′′′′,2′′′′′,2′′′′′′-trihydroxyisodiuvaretin (180) | −84.3 | −65.2 | −120.6 | −93.3 | no dock | no dock | −127.4 | −98.5 | −105.3 | −81.5 |
| 4′-Hydroxychalcone (179) | −92.5 | −109.5 | −85.8 | −101.6 | −92.5 | −109.4 | −82.7 | −97.9 | −87.9 | −104.0 |
| 5″,5′′′′,5′′′′′-Tribenzyl-2′′′′,2′′′′′,2′′′′′′-trihydroxyisodiuvaretin (181) | −52.4 | −40.5 | −128.6 | −99.5 | no dock | no dock | −159.7 | −123.5 | −116.6 | −90.2 |
| Angusticornin B (182) | −154.9 | −148.2 | −79.5 | −76.0 | −151.8 | −145.2 | −127.3 | −121.8 | −97.1 | −92.9 |
| Balsacone A (183) | −144.0 | −138.2 | −107.4 | −103.1 | −143.1 | −137.3 | −113.8 | −109.3 | −107.6 | −103.3 |
| Balsacone B (184) | −139.9 | −134.3 | −113.4 | −108.8 | −144.0 | −138.2 | −116.0 | −111.4 | −107.8 | −103.5 |
| Balsacone C (185) | −132.9 | −130.7 | −107.2 | −105.5 | −133.6 | −131.4 | −112.5 | −110.7 | −109.0 | −107.3 |
| Bartericin C (186) | −118.5 | −114.8 | −66.2 | −64.1 | −104.8 | −101.5 | −112.8 | −109.3 | −68.4 | −66.3 |
| Bavachalcone (187) | −137.5 | −141.9 | −101.8 | −105.1 | −136.4 | −140.8 | −103.1 | −106.4 | −92.8 | −95.8 |
| Broussochalcone B (188) | −127.2 | −133.1 | −107.7 | −112.7 | −130.5 | −136.5 | −103.6 | −108.4 | −105.3 | −110.2 |
| Corylifol B (189) | −138.2 | −142.3 | −113.7 | −117.0 | −140.0 | −144.2 | −107.5 | −110.7 | −107.2 | −110.4 |
| Erythbidin C (190) | −122.4 | −129.7 | −96.7 | −102.5 | −121.2 | −128.4 | −102.6 | −108.7 | −102.7 | −108.9 |
| Helichrysone A (191) | −124.8 | −126.8 | −109.9 | −111.7 | −125.4 | −127.4 | −102.7 | −104.3 | −98.7 | −100.3 |
| Isobavachalcone (192) | −127.7 | −133.7 | −110.6 | −115.7 | −133.8 | −140.0 | −100.7 | −105.3 | −103.3 | −108.1 |
| Kanzonol C (193) | −151.6 | −148.8 | −119.0 | −116.9 | −159.8 | −156.9 | −111.8 | −109.8 | −88.1 | −86.5 |
| Kuraridin (194) | −148.2 | −140.3 | −122.1 | −115.5 | −145.9 | −138.1 | −121.3 | −114.8 | −104.2 | −98.7 |
| Myrigalone G (195) | −111.4 | −121.5 | −98.3 | −107.3 | −115.0 | −125.5 | −91.9 | −100.3 | −88.3 | −96.3 |
| Piperaduncin A (196) | −137.5 | −125.3 | −113.2 | −103.2 | −134.3 | −122.5 | −119.7 | −109.1 | −82.7 | −75.4 |
| Piperaduncin B (197) | −134.6 | −121.4 | −124.5 | −112.3 | −152.4 | −137.5 | −122.3 | −110.3 | −102.4 | −92.4 |
| Piperaduncin C (198) | −134.1 | −117.2 | −130.8 | −114.3 | −112.3 | −98.1 | −128.3 | −112.1 | −78.9 | −68.9 |
| Psorachalcone A (199) | −130.1 | −134.0 | −112.2 | −115.6 | −137.6 | −141.7 | −96.9 | −99.8 | −99.3 | −102.2 |
| Xanthoangelol (200) | −143.1 | −140.5 | −113.3 | −111.2 | −140.1 | −137.6 | −110.2 | −108.2 | −93.1 | −91.4 |
| Xanthoangelol F (201) | −147.0 | −142.7 | −123.4 | −119.7 | −133.1 | −129.2 | −116.5 | −113.1 | −101.2 | −98.3 |
| Flavonoids | ||||||||||
| 2′,5,5′,7-Tetrahydroxyflavanone (202) | −102.5 | −111.6 | −87.5 | −95.2 | −96.1 | −104.6 | −86.1 | −93.7 | −91.4 | −99.5 |
| 2′,7-Dimethoxyflavone (203) | −105.9 | −116.1 | −90.8 | −99.5 | −103.5 | −113.5 | −79.5 | −87.1 | −81.0 | −88.8 |
| 3′′′′-(2-Hydroxybenzyl)isouvarinol (218) | −158.3 | −129.4 | −157.9 | −129.0 | −115.6 | −94.4 | −130.8 | −106.9 | −126.0 | −103.0 |
| 3′′′′-(2-Hydroxybenzyl)uvarinol (217) | −155.5 | −127.1 | −153.1 | −125.1 | −125.1 | −102.2 | −152.8 | −124.9 | −93.3 | −76.3 |
| 3′-Methylpelargonidin (204) | −97.6 | −106.6 | −88.0 | −96.1 | −94.6 | −103.3 | −87.6 | −95.7 | −90.2 | −98.6 |
| 3′-O-Methyldiplacone (205) | −134.5 | −127.3 | −107.4 | −101.7 | −130.8 | −123.8 | −115.3 | −109.1 | −104.7 | −99.1 |
| 4′,5,7-Trihydroxy-6-methyl-8-prenylflavanone (207) | −116.4 | −118.2 | −101.1 | −102.7 | −110.0 | −111.8 | −90.9 | −92.4 | −104.2 | −105.9 |
| 4′,5,7-Trihydroxy-8-methyl-6-prenylflavanone (206) | −110.4 | −112.2 | −110.1 | −111.8 | −110.4 | −112.1 | −94.5 | −96.0 | −105.6 | −107.3 |
| 4',5-Dihydroxy-7-methoxy-6-prenylflavanone (208) | −126.1 | −128.2 | −109.3 | −111.1 | −120.3 | −122.3 | −96.3 | −97.8 | −100.4 | −102.0 |
| 4′,6,7-Trihydroxy-3′,5′-dimethoxyflavone (209) | −115.1 | −119.7 | −109.5 | −113.9 | −110.4 | −114.8 | −96.2 | −100.1 | −99.7 | −103.7 |
| 4′,7-Dihydroxy-8-methylflavan (210) | −88.8 | −100.6 | −82.8 | −93.7 | −83.5 | −94.5 | −77.6 | −87.9 | −85.2 | −96.4 |
| 4′-Hydroxy-5,7-dimethoxyflavone (211) | −104.1 | −112.0 | −84.7 | −91.2 | −109.3 | −117.6 | −88.6 | −95.3 | −94.1 | −101.2 |
| 5″-(2-Hydroxybenzyl)isouvarinol (216) | −184.9 | −151.1 | −136.0 | −111.2 | −140.6 | −114.9 | −137.1 | −112.0 | −143.1 | −116.9 |
| 5′-(1,1-Dimethyl-2-propenyl)-2′,4′,5,7-tetrahydroxy-6-prenylflavanone (219) | −128.0 | −122.4 | −106.4 | −101.8 | −116.3 | −111.3 | −98.5 | −94.2 | −68.6 | −65.6 |
| 5′-(1,1-Dimethyl-2-propenyl)-2′,4′,5,7-tetrahydroxy-8-prenylflavanone (220) | −127.0 | −121.5 | −96.0 | −91.8 | −114.5 | −109.5 | −99.4 | −95.1 | −84.4 | −80.8 |
| 5′-(1,1-Dimethyl-2-propenyl)-4′,5,7-trihydroxy-2′-methoxy-8-prenylflavanone (221) | −114.9 | −108.8 | −75.8 | −71.7 | −124.0 | −117.3 | −96.2 | −91.0 | −96.9 | −91.7 |
| 5,6-Dihydroxy-4′,7,8-trimethoxyflavone (212) | −108.6 | −111.4 | −92.7 | −95.1 | −109.6 | −112.5 | −74.3 | −76.2 | −33.0 | −33.8 |
| 5-Hydroxy-2′,4′,5′,7-Tetramethoxyflavone (213) | −115.1 | −116.5 | −103.7 | −104.9 | −123.3 | −124.8 | −91.7 | −92.8 | −56.8 | −57.5 |
| 6,7-Dihydroxyflavone (214) | −98.1 | −111.3 | −90.2 | −102.4 | −89.5 | −101.6 | −88.1 | −100.0 | −94.5 | −107.3 |
| 8-Methoxycirsilineol (215) | −124.1 | −123.8 | −111.2 | −110.9 | −111.1 | −110.8 | −88.7 | −88.5 | −99.3 | −99.1 |
| 9,10-Dihydro-9,10-diacetoxy-3-methoxy-8,8-dimethyl-2-phenyl-4H,8H-benzo[1,2-b:3,4-b′]dipyran-4-one (222) | −81.8 | −76.6 | −73.8 | −69.1 | −76.4 | −71.5 | −113.3 | −106.1 | −75.8 | −71.0 |
| Abyssinone I (223) | −105.9 | −111.0 | −71.8 | −75.3 | −100.1 | −104.9 | −86.2 | −90.4 | −95.7 | −100.4 |
| Abyssinone IV (224) | −139.9 | −137.4 | −96.2 | −94.5 | −135.9 | −133.4 | −93.4 | −91.7 | −117.7 | −115.6 |
| Astragalin (225) | −136.8 | −128.5 | −103.9 | −97.6 | −127.6 | −119.8 | −103.7 | −97.5 | −107.4 | −100.9 |
| Bavachinin (226) | −119.3 | −123.1 | −111.3 | −114.9 | −118.8 | −122.5 | −96.5 | −99.5 | −100.8 | −104.0 |
| Betuletol (227) | −110.9 | −115.3 | −94.6 | −98.4 | −104.6 | −108.8 | −83.3 | −86.6 | −79.5 | −82.7 |
| Bonannione A (228) | −130.5 | −126.4 | −96.3 | −93.3 | −131.5 | −127.5 | −108.1 | −104.7 | −107.2 | −103.9 |
| Brosimone I (229) | −120.0 | −115.1 | −105.0 | −100.7 | −128.0 | −122.8 | −97.2 | −93.3 | −110.4 | −106.0 |
| Cassiaflavan (230) | −85.4 | −98.4 | −80.9 | −93.3 | −86.0 | −99.2 | −82.0 | −94.6 | −85.6 | −98.7 |
| Cerasinone (231) | −108.4 | −112.7 | −88.3 | −91.8 | −111.6 | −116.1 | −85.6 | −89.1 | −77.3 | −80.4 |
| Chrysin (233) | −97.1 | −110.2 | −87.1 | −98.9 | −89.5 | −101.6 | −84.9 | −96.3 | −88.6 | −100.5 |
| Chrysoeriol (232) | −105.9 | −113.7 | −99.9 | −107.2 | −105.7 | −113.5 | −95.2 | −102.2 | −93.4 | −100.3 |
| Corniculatusin (234) | −109.2 | −113.3 | −100.2 | −104.1 | −102.3 | −106.2 | −93.0 | −96.5 | −103.3 | −107.2 |
| Cudraflavone A (235) | −109.6 | −105.4 | −100.2 | −96.3 | −109.4 | −105.1 | −93.8 | −90.1 | −102.9 | −98.9 |
| Dihydroquercetin (236) | −103.3 | −110.4 | −96.7 | −103.4 | −96.3 | −103.0 | −94.5 | −101.0 | −98.0 | −104.7 |
| Eucalyptin (237) | −109.3 | −114.1 | −91.8 | −95.9 | −102.6 | −107.2 | −75.3 | −78.6 | −82.7 | −86.4 |
| Euchrestaflavanone A (238) | −137.0 | −132.7 | −112.6 | −109.1 | −137.5 | −133.3 | −93.2 | −90.3 | −50.9 | −49.3 |
| Flavaprenin (239) | −115.7 | −119.2 | −99.5 | −102.5 | −112.5 | −115.9 | −95.0 | −97.8 | −105.3 | −108.4 |
| Flemiflavanone D (240) | −128.9 | −123.3 | −97.0 | −92.8 | −132.8 | −127.0 | −108.0 | −103.3 | −86.7 | −83.0 |
| Glabranin (241) | −116.1 | −121.5 | −95.5 | −99.9 | −108.9 | −113.9 | −84.3 | −88.2 | −97.1 | −101.6 |
| Isoorientin (243) | −126.3 | −118.7 | −118.7 | −111.5 | −124.4 | −116.8 | −103.0 | −96.7 | −109.4 | −102.7 |
| Isoscoparin (244) | −117.1 | −108.9 | −125.0 | −116.2 | −121.4 | −112.8 | −98.4 | −91.5 | −103.4 | −96.1 |
| Kaempferol (242) | −93.2 | −101.7 | −90.1 | −98.3 | −92.5 | −100.9 | −87.0 | −94.9 | −95.8 | −104.5 |
| Kushenol A (245) | −141.0 | −136.6 | −109.0 | −105.6 | −139.7 | −135.4 | −92.6 | −89.7 | −100.7 | −97.6 |
| Kushenol S (246) | −118.5 | −122.1 | −95.5 | −98.4 | −112.5 | −115.8 | −84.8 | −87.3 | −95.4 | −98.3 |
| Kushenol U (247) | −136.4 | −130.7 | −114.1 | −109.3 | −142.9 | −136.9 | −105.5 | −101.1 | −105.0 | −100.6 |
| Kushenol V (248) | −126.9 | −123.8 | −113.5 | −110.7 | −123.6 | −120.5 | −91.5 | −89.3 | −98.0 | −95.6 |
| Kushenol W (249) | −126.7 | −125.1 | −105.8 | −104.4 | −119.8 | −118.3 | −90.8 | −89.7 | −99.2 | −97.9 |
| Leachianone A (250) | −142.5 | −134.9 | −100.9 | −95.5 | −140.0 | −132.5 | −102.6 | −97.1 | −105.2 | −99.6 |
| Leachianone G (251) | −115.9 | −117.6 | −100.5 | −101.9 | −120.3 | −122.0 | −92.2 | −93.5 | −106.0 | −107.5 |
| Licoflavanone (252) | −116.0 | −119.5 | −96.0 | −98.9 | −121.6 | −125.2 | −100.7 | −103.7 | −100.1 | −103.1 |
| Licoflavone C (253) | −115.0 | −118.7 | −100.0 | −103.2 | −117.5 | −121.3 | −91.8 | −94.7 | −108.5 | −112.0 |
| Licoflavonol (254) | −118.6 | −120.5 | −109.3 | −111.1 | −118.6 | −120.5 | −99.0 | −100.6 | −105.3 | −107.0 |
| Lonchocarpol A (255) | −121.9 | −118.1 | −117.9 | −114.2 | −123.5 | −119.6 | −102.6 | −99.4 | −118.0 | −114.3 |
| Loranthin (256) | −127.5 | −115.8 | −118.2 | −107.3 | −128.5 | −116.7 | −114.6 | −104.1 | −109.2 | −99.2 |
| Loxophlebal A (257) | −128.6 | −118.7 | −104.0 | −96.0 | −119.5 | −110.3 | −104.6 | −96.5 | −92.0 | −84.9 |
| Lucenin 2 (258) | −86.0 | −72.9 | −103.5 | −87.7 | −108.6 | −92.0 | −134.2 | −113.8 | −123.9 | −105.0 |
| Macarangaflavanone A (259) | −138.2 | −133.9 | −104.3 | −101.0 | −135.6 | −131.4 | −107.5 | −104.2 | −81.3 | −78.7 |
| Malvidin (260) | −113.5 | −117.9 | −96.3 | −100.0 | −107.1 | −111.2 | −102.2 | −106.2 | −92.5 | −96.2 |
| Myricetin (261) | −103.3 | −108.8 | −99.3 | −104.5 | −104.1 | −109.6 | −97.9 | −103.1 | −99.2 | −104.5 |
| Natsudaidain (262) | −123.4 | −118.6 | −112.1 | −107.8 | −118.4 | −113.8 | −90.4 | −86.9 | −45.0 | −43.2 |
| Nevadensin (263) | −119.8 | −122.9 | −96.6 | −99.1 | −113.7 | −116.6 | −85.7 | −87.9 | −67.8 | −69.6 |
| O-Methylpongaglabol (264) | −102.3 | −110.8 | −89.5 | −97.0 | −99.6 | −107.9 | −91.1 | −98.7 | −93.7 | −101.6 |
| Paratocarpin L (265) | −132.3 | −128.2 | −111.8 | −108.3 | −134.4 | −130.2 | −102.9 | −99.7 | −105.8 | −102.6 |
| Persicogenin (266) | −107.9 | −113.8 | −91.9 | −96.9 | −110.9 | −117.0 | −74.9 | −79.0 | −90.3 | −95.2 |
| Pilosanol A (267) | −136.9 | −120.8 | −118.8 | −104.8 | −128.4 | −113.3 | −114.8 | −101.4 | −98.6 | −87.0 |
| Pilosanol B (268) | −139.4 | −124.1 | −114.6 | −102.1 | −114.3 | −101.8 | −111.3 | −99.1 | −93.6 | −83.4 |
| Pilosanol C (269) | −140.8 | −125.3 | −123.9 | −110.3 | −126.6 | −112.7 | −116.7 | −103.9 | −108.7 | −96.8 |
| Pinocembrin (270) | −94.5 | −107.0 | −83.1 | −94.1 | −88.3 | −99.9 | −82.7 | −93.6 | −84.7 | −95.9 |
| Pongaflavone (271) | −99.7 | −103.3 | −78.6 | −81.4 | −103.1 | −106.8 | −89.6 | −92.8 | −101.2 | −104.9 |
| Quercetin (272) | −102.9 | −110.3 | −94.6 | −101.4 | −96.7 | −103.6 | −93.8 | −100.5 | −99.8 | −106.9 |
| Quercetin 3-methyl ether (273) | −100.4 | −106.0 | −91.7 | −96.7 | −96.5 | −101.8 | −94.0 | −99.2 | −104.6 | −110.4 |
| Remangiflavanone A (274) | −133.0 | −127.4 | −112.4 | −107.6 | −133.1 | −127.5 | −101.1 | −96.9 | −106.7 | −102.2 |
| Remangiflavanone B (275) | −136.4 | −130.5 | −109.2 | −104.4 | −142.3 | −136.2 | −105.7 | −101.2 | −104.0 | −99.5 |
| Sanggenon G (276) | −107.6 | −87.3 | −105.6 | −85.7 | −118.6 | −96.3 | −129.7 | −105.3 | −97.5 | −79.2 |
| Sigmoidin A (277) | −134.3 | −128.5 | −119.0 | −113.8 | −134.4 | −128.6 | −111.0 | −106.2 | −103.3 | −98.8 |
| Sigmoidin B (278) | −120.5 | −122.2 | −97.5 | −98.9 | −120.3 | −122.0 | −106.6 | −108.1 | −78.8 | −79.9 |
| Sigmoidin L (279) | −120.7 | −120.8 | −99.4 | −99.5 | −121.3 | −121.4 | −109.3 | −109.5 | −89.0 | −89.1 |
| Siraitiflavandiol (280) | −118.4 | −120.1 | −105.6 | −107.0 | −117.0 | −118.6 | −113.1 | −114.7 | −96.9 | −98.3 |
| Solophenol D (281) | −145.5 | −137.7 | −118.7 | −112.3 | −141.7 | −134.1 | −115.3 | −109.1 | −85.0 | −80.4 |
| Sophoraflavanone G (282) | −137.3 | −131.3 | −110.3 | −105.5 | −144.7 | −138.4 | −99.3 | −95.0 | −106.7 | −102.1 |
| Sternbin (283) | −101.5 | −108.8 | −93.7 | −100.4 | −106.8 | −114.4 | −95.3 | −102.1 | −96.1 | −103.0 |
| Sudachitin (284) | −120.7 | −122.0 | −112.6 | −113.8 | −118.2 | −119.5 | −83.4 | −84.3 | −101.0 | −102.0 |
| Uvarinol (285) | −153.4 | −132.7 | −122.7 | −106.1 | −149.9 | −129.6 | −119.7 | −103.6 | −135.1 | −116.9 |
| Vahliabiflavone (286) | no dock | no dock | −87.6 | −75.9 | no dock | no dock | −115.6 | −100.2 | −57.4 | −49.8 |
| Vitexin (287) | −129.6 | −123.2 | −97.8 | −93.0 | −129.8 | −123.4 | −99.0 | −94.1 | −114.9 | −109.2 |
| Wogonin (288) | −103.2 | −112.8 | −90.7 | −99.2 | −96.9 | −106.0 | −87.5 | −95.7 | −91.5 | −100.0 |
| Isoflavonoids | ||||||||||
| 2″,3″-Epoxybolusanthol B (289) | −126.2 | −126.1 | −94.8 | −94.7 | −123.8 | −123.7 | −98.9 | −98.9 | −95.6 | −95.6 |
| 3′,5,7-Trihydroxy-4′-methoxy-5′,6-diprenylisoflavanone (290) | −131.8 | −124.7 | −111.5 | −105.5 | −126.4 | −119.7 | −109.5 | −103.6 | −102.4 | −96.9 |
| 4″-Hydroxydiphysolone (292) | −122.8 | −122.7 | −104.0 | −103.9 | −120.3 | −120.2 | −98.7 | −98.6 | −100.7 | −100.6 |
| 5,7-Dihydroxy-2′-methoxy-3′,4′-methylenedioxyisoflavanone (293) | −119.3 | −124.1 | −91.5 | −95.2 | −116.5 | −121.2 | −77.2 | −80.3 | −79.8 | −83.0 |
| 6a-Hydroxyphaseollin (291) | −103.5 | −106.7 | −86.7 | −89.4 | −92.0 | −95.0 | −91.2 | −94.1 | −83.2 | −85.8 |
| Amorphaquinone (294) | −114.3 | −117.0 | −82.7 | −84.7 | −112.4 | −115.1 | −83.7 | −85.7 | −83.1 | −85.1 |
| Asphodelin A (295) | −90.7 | −100.9 | −92.6 | −103.0 | −95.0 | −105.7 | −81.1 | −90.2 | −86.2 | −95.9 |
| Bidwillon A (296) | −125.7 | −121.8 | −110.5 | −107.0 | −126.3 | −122.4 | −103.4 | −100.2 | −111.6 | −108.1 |
| Bolucarpan A (297) | −110.0 | −110.4 | −96.3 | −96.5 | −101.1 | −101.4 | −73.2 | −73.4 | −71.7 | −71.9 |
| Bolucarpan B (298) | −106.3 | −106.8 | −97.9 | −98.3 | −96.6 | −97.1 | −77.0 | −77.3 | −72.4 | −72.8 |
| Bolucarpan D (299) | −87.5 | −90.4 | −91.6 | −94.7 | −79.7 | −82.4 | −76.7 | −79.3 | −63.6 | −65.8 |
| Bolusanthol B (300) | −124.5 | −126.3 | −99.8 | −101.2 | −124.6 | −126.3 | −102.3 | −103.7 | −102.4 | −103.8 |
| Cajanol (301) | −109.2 | −115.2 | −88.8 | −93.7 | −104.5 | −110.3 | −85.5 | −90.3 | −79.4 | −83.7 |
| Chandalone (302) | −118.7 | −115.4 | −104.3 | −101.4 | −119.5 | −116.2 | −100.4 | −97.7 | −63.3 | −61.6 |
| Dalversinol A (303) | −123.2 | −117.9 | −92.8 | −88.8 | −123.0 | −117.7 | −102.2 | −97.8 | −94.5 | −90.4 |
| Derrisin (304) | −87.0 | −82.9 | −73.4 | −70.0 | −63.7 | −60.8 | −78.5 | −74.8 | −18.9 | −18.1 |
| Erybraedin A (305) | −125.4 | −123.1 | −98.2 | −96.5 | −123.2 | −121.0 | −104.0 | −102.1 | −89.0 | −87.4 |
| Erybraedin D (306) | −129.2 | −127.1 | −94.8 | −93.3 | −126.3 | −124.3 | −92.7 | −91.1 | −80.0 | −78.7 |
| Erypoegin I (307) | −106.4 | −106.5 | −93.4 | −93.5 | −117.6 | −117.7 | −104.0 | −104.2 | −76.8 | −76.9 |
| Erysubin F (308) | −139.5 | −137.2 | −114.2 | −112.3 | −132.7 | −130.6 | −113.7 | −111.8 | −79.4 | −78.1 |
| Eryvarin V (309) | −104.7 | −100.3 | −99.6 | −95.5 | −96.9 | −92.8 | −107.9 | −103.4 | −86.3 | −82.7 |
| Eryvarin W (310) | −118.1 | −116.2 | −104.3 | −102.6 | −115.0 | −113.1 | −107.6 | −105.9 | −75.3 | −74.1 |
| Eryzerin C (311) | −128.1 | −125.6 | −104.9 | −102.8 | −127.7 | −125.1 | −104.6 | −102.5 | −114.6 | −112.4 |
| Eryzerin D (312) | −122.6 | −120.4 | −99.5 | −97.7 | −124.7 | −122.4 | −99.3 | −97.5 | −98.9 | −97.1 |
| Euchretin A (313) | −128.3 | −116.1 | −92.3 | −83.5 | −130.0 | −117.6 | −113.0 | −102.2 | −77.2 | −69.9 |
| Gancaonin C (314) | −113.0 | −114.8 | −97.2 | −98.7 | −115.8 | −117.6 | −102.6 | −104.3 | −107.4 | −109.2 |
| Genistein (315) | −95.8 | −106.5 | −87.7 | −97.5 | −96.8 | −107.6 | −84.2 | −93.6 | −93.6 | −104.1 |
| Glycyrrhisoflavone (316) | −121.6 | −123.6 | −98.9 | −100.5 | −122.2 | −124.1 | −100.7 | −102.3 | −95.8 | −97.3 |
| Hispaglabridin A (317) | −122.9 | −120.7 | −101.0 | −99.2 | −122.6 | −120.4 | −94.1 | −92.4 | −95.5 | −93.8 |
| Hispaglabridin B (318) | −112.8 | −111.0 | −31.1 | −30.6 | −107.5 | −105.8 | −84.8 | −83.4 | −61.9 | −60.9 |
| Hydroxycristacarpone (319) | −96.2 | −96.3 | −87.8 | −87.9 | −91.8 | −91.9 | −91.6 | −91.7 | −63.6 | −63.7 |
| Isoneorautenol (320) | −109.2 | −114.5 | −75.7 | −79.4 | −114.0 | −119.6 | −81.1 | −85.0 | −72.2 | −75.7 |
| Lachnoisoflavone A (321) | −112.5 | −114.5 | −98.9 | −100.6 | −116.3 | −118.3 | −94.8 | −96.5 | −97.4 | −99.1 |
| Licoisoflavanone (322) | −110.9 | −112.7 | −64.0 | −65.1 | −98.6 | −100.1 | −92.6 | −94.1 | −83.0 | −84.4 |
| Licoricidin (323) | −136.7 | −130.7 | −108.7 | −104.0 | −138.7 | −132.7 | −108.4 | −103.7 | −107.1 | −102.4 |
| Lupinalbin C (324) | −113.1 | −113.4 | −104.1 | −104.4 | −110.9 | −111.2 | −92.0 | −92.3 | −93.9 | −94.2 |
| Mucronulatol (326) | −99.6 | −106.7 | −80.0 | −85.8 | −101.4 | −108.6 | −86.3 | −92.5 | −92.6 | −99.2 |
| Neomillinol (325) | −103.3 | −107.9 | −96.9 | −101.1 | −113.5 | −118.5 | −93.8 | −98.0 | −93.6 | −97.8 |
| Pendulone (327) | −107.6 | −113.6 | −79.4 | −83.8 | −105.6 | −111.4 | −92.9 | −98.1 | −74.3 | −78.4 |
| Phyllanone B (328) | −138.9 | −131.4 | −98.8 | −93.5 | −139.5 | −132.0 | −112.8 | −106.8 | −101.9 | −96.5 |
| Shinpterocarpin (329) | −97.7 | −102.5 | −81.2 | −85.1 | −95.2 | −99.8 | −95.3 | −99.9 | −79.1 | −82.9 |
| Neoflavonoids | ||||||||||
| Inophyllum A (330) | −103.7 | −100.8 | −82.6 | −80.3 | −101.8 | −99.0 | −90.4 | −87.9 | −77.2 | −75.1 |
| Inophyllum C (331) | −111.3 | −108.4 | −97.1 | −94.6 | −107.6 | −104.8 | −87.4 | −85.2 | −71.3 | −69.5 |
| Mammea A/BA (332) | −127.9 | −124.1 | −110.5 | −107.2 | −117.2 | −113.8 | −100.8 | −97.8 | −112.0 | −108.7 |
| Mammea A/BB (333) | −123.4 | −119.8 | −101.6 | −98.6 | −118.6 | −115.1 | −95.0 | −92.2 | −104.6 | −101.6 |
| Mesuol (334) | −124.3 | −122.1 | −108.0 | −106.1 | −122.2 | −120.0 | −101.2 | −99.4 | −106.9 | −105.0 |
| Pterocarpans | ||||||||||
| 1-Methoxyphaseollidin (356) | −117.4 | −119.3 | −97.6 | −99.2 | −118.8 | −120.7 | −97.6 | −99.2 | −68.5 | −69.6 |
| Aracarpene 1 (357) | −105.0 | −112.7 | −84.6 | −90.9 | −105.1 | −112.8 | −87.7 | −94.2 | −89.8 | −96.5 |
| Aracarpene 2 (358) | −102.9 | −110.5 | −94.1 | −101.1 | −108.8 | −116.9 | −78.7 | −84.5 | −85.6 | −91.9 |
| Calopocarpin (360) | −111.9 | −117.1 | −103.9 | −108.7 | −113.2 | −118.5 | −102.7 | −107.4 | −99.3 | −103.9 |
| Cristacarpin (359) | −121.4 | −123.4 | −91.5 | −92.9 | −119.0 | −120.9 | −98.3 | −99.8 | −88.5 | −89.9 |
| Erythbidin D (362) | −103.0 | −110.6 | −93.5 | −100.4 | −112.8 | −121.1 | −88.4 | −95.0 | −80.6 | −86.6 |
| Eryzerin E (363) | −140.8 | −135.0 | −96.5 | −92.4 | −129.6 | −124.2 | −99.4 | −95.2 | −102.8 | −98.5 |
| Fuscacarpan B (364) | −116.4 | −116.6 | −88.4 | −88.6 | −118.6 | −118.7 | −99.2 | −99.3 | −88.9 | −89.0 |
| Fuscacarpan C (365) | −116.5 | −116.7 | −90.6 | −90.7 | −111.3 | −111.4 | −99.8 | −99.9 | −86.8 | −86.9 |
| Glycyrol (366) | −122.3 | −122.9 | −102.1 | −102.6 | −119.2 | −119.7 | −88.8 | −89.2 | −102.0 | −102.4 |
| Glycyrrhizol A (367) | −135.5 | −130.0 | −54.2 | −52.0 | −124.6 | −119.6 | −100.1 | −96.1 | −87.0 | −83.5 |
| Glycyrrhizol B (368) | −110.1 | −112.3 | −80.8 | −82.4 | −117.7 | −120.1 | −86.6 | −88.4 | −73.5 | −75.0 |
| Sandwicensin (369) | −115.6 | −119.3 | −103.1 | −106.3 | −116.4 | −120.1 | −94.8 | −97.8 | −98.5 | −101.6 |
| Variabilin (370) | −103.9 | −111.5 | −85.6 | −91.9 | −107.1 | −115.0 | −83.4 | −89.5 | −73.5 | −78.9 |
| ent-Sophoracarpan A (361) | −106.2 | −114.0 | −76.3 | −82.0 | −99.0 | −106.2 | −89.4 | −96.0 | −82.8 | −88.9 |
| Chromones | ||||||||||
| 3-(3-Hydroxy-4-methoxybenzylidene)-6,7-dimethoxy-4-chromanone (371) | −123.1 | −126.5 | −98.9 | −101.6 | −116.1 | −119.3 | −86.7 | −89.1 | −75.0 | −77.1 |
| 4′,5,7-Trihydroxy-6,8-dimethylhomoisoflavanone (372) | −102.9 | −108.9 | −93.5 | −98.8 | −97.9 | −103.6 | −83.4 | −88.2 | −84.3 | −89.1 |
| 4′,5,7-Trihydroxy-6-methylhomoisoflavanone (373) | −99.8 | −107.1 | −93.5 | −100.4 | −104.1 | −111.8 | −86.2 | −92.6 | −82.2 | −88.2 |
| 7-O-Methylbonducellin (374) | −108.5 | −117.0 | −94.7 | −102.1 | −108.5 | −117.0 | −83.2 | −89.8 | −77.7 | −83.8 |
| 8-Methylophiopogonanone B (375) | −113.2 | −118.0 | −91.8 | −95.7 | −110.5 | −115.1 | −82.9 | −86.4 | −77.3 | −80.6 |
| Bonducellin (376) | −102.6 | −112.4 | −88.3 | −96.8 | −104.4 | −114.4 | −94.8 | −103.9 | −85.8 | −94.1 |
| Odoratumone A (377) | −117.1 | −120.2 | −95.2 | −97.7 | −117.5 | −120.6 | −88.2 | −90.5 | −74.8 | −76.8 |
| Sappanone A 3′,4′-methylene ether (378) | −110.6 | −119.3 | −96.3 | −103.9 | −109.0 | −117.6 | −96.1 | −103.6 | −90.8 | −97.9 |
| Sappanone A 4′-methyl ether (379) | −108.5 | −116.7 | −95.2 | −102.4 | −108.3 | −116.6 | −96.6 | −104.0 | −90.5 | −97.4 |
| Sappanone A trimethyl ether (380) | −123.0 | −128.4 | −103.8 | −108.4 | −123.7 | −129.1 | −87.5 | −91.4 | −85.4 | −89.2 |
| Condensed Tannins | ||||||||||
| GB 1 (381) | no dock | no dock | −35.1 | −30.7 | −37.2 | −32.5 | −103.7 | −90.5 | −79.9 | −69.8 |
| Proanthocyanidin A1 (382) | −80.6 | −69.6 | −104.7 | −90.5 | −41.3 | −35.7 | −111.7 | −96.5 | −96.1 | −83.0 |
| Proanthocyanidin A2 (383) | −70.8 | −61.1 | −94.8 | −81.9 | −21.3 | −18.4 | −107.4 | −92.8 | −104.1 | −89.9 |
| Procyanidin B4 (384) | −66.7 | −57.6 | −97.2 | −83.9 | no dock | no dock | −119.8 | −103.3 | −65.5 | −56.5 |
| Procyanidin B5 (385) | −68.4 | −59.1 | −61.6 | −53.1 | −41.8 | −36.0 | −115.2 | −99.4 | −88.3 | −76.2 |
| Procyanidin B6 (386) | −74.6 | −64.4 | no dock | no dock | −47.5 | −41.0 | −119.6 | −103.2 | −104.0 | −89.7 |
| Teatannin (387) | −139.2 | −131.4 | −62.8 | −59.3 | −143.4 | −135.3 | −116.0 | −109.5 | −108.3 | −102.2 |
| Coumarins | ||||||||||
| 4,5′,8′-Trihydroxy-5-methyl-3,7′-bicoumarin (344) | −109.2 | −111.2 | −104.3 | −106.2 | −112.1 | −114.1 | −94.5 | −96.2 | −93.1 | −94.7 |
| 6-Geranyl-5,7-dihydroxy-8(2-methylbutanoyl)-4-phenylcoumarin (345) | −154.6 | −142.5 | −112.3 | −103.6 | −166.5 | −153.4 | −113.8 | −104.9 | −120.1 | −110.7 |
| (–)-Heliettin (354) | −79.0 | −100.9 | −66.0 | −84.4 | −79.6 | −101.7 | −81.8 | −104.6 | −77.4 | −98.9 |
| Aesculin (347) | −115.1 | −118.5 | −108.7 | −112.0 | −115.1 | −118.5 | −89.2 | −91.9 | −93.3 | −96.1 |
| Alloimperatorin (346) | −103.5 | −115.1 | −95.0 | −105.7 | −103.4 | −115.0 | −93.1 | −103.6 | −95.9 | −106.6 |
| Anofinic acid (348) | −86.1 | −105.1 | −75.2 | −91.8 | −90.1 | −110.0 | −75.7 | −92.5 | −88.4 | −108.0 |
| Calaustralin (349) | −125.0 | −121.5 | −95.8 | −93.2 | −120.0 | −116.6 | −92.8 | −90.2 | −101.5 | −98.7 |
| Calophyllolide (350) | −112.3 | −108.1 | −85.2 | −82.0 | −114.2 | −110.0 | −99.2 | −95.5 | −12.5 | −12.0 |
| Dicoumarol (351) | −114.7 | −118.6 | −80.0 | −82.7 | −115.5 | −119.4 | −88.1 | −91.1 | −85.1 | −88.0 |
| Dipetalolactone (352) | −100.0 | −106.2 | −83.5 | −88.7 | −89.6 | −95.2 | −84.0 | −89.2 | −75.1 | −79.8 |
| Glycycoumarin (353) | −116.4 | −116.7 | −110.0 | −110.3 | −124.0 | −124.3 | −103.9 | −104.2 | −101.6 | −101.9 |
| Marmesin (355) | −96.2 | −110.3 | −81.6 | −93.6 | −96.9 | −111.1 | −94.6 | −108.5 | −93.4 | −107.1 |
| Stilbenoids | ||||||||||
| 2-(2,4-Dihydroxyphenyl-5-(1-propenyl)benzofurans (388) | −106.3 | −118.8 | −98.6 | −110.2 | −111.2 | −124.3 | −90.3 | −100.9 | −94.9 | −106.1 |
| Albanol A (389) | no dock | no dock | −85.8 | −74.8 | no dock | no dock | −117.8 | −102.6 | −97.4 | −84.8 |
| Albanol B (390) | −124.6 | −108.8 | −99.3 | −86.7 | −77.3 | −67.5 | −114.8 | −100.2 | −87.8 | −76.7 |
| Amorfrutin A (391) | −121.4 | −125.0 | −111.8 | −115.1 | −117.7 | −121.2 | −100.0 | −103.0 | −97.4 | −100.3 |
| Cajaninstilbene acid (392) | −122.8 | −126.7 | −117.7 | −121.4 | −117.5 | −121.2 | −96.0 | −99.0 | −96.4 | −99.4 |
| Calodenin B (393) | −160.9 | −143.4 | −143.2 | −127.7 | −143.4 | −127.8 | −138.4 | −123.4 | −124.1 | −110.6 |
| Centrolobofuran (394) | −95.5 | −108.1 | −100.9 | −114.2 | −100.1 | −113.4 | −89.5 | −101.4 | −92.8 | −105.0 |
| Cochinchinenene A (395) | −134.0 | −118.3 | −87.8 | −77.5 | −97.1 | −85.7 | −124.2 | −109.6 | −99.0 | −87.3 |
| Cochinchinenene B (396) | −147.2 | −132.2 | −118.2 | −106.2 | −151.4 | −136.0 | −126.2 | −113.3 | −98.9 | −88.9 |
| Cochinchinenene C (397) | −142.5 | −129.2 | −104.2 | −94.5 | −139.6 | −126.5 | −115.1 | −104.4 | −90.8 | −82.3 |
| Cochinchinenene D (398) | −140.2 | −128.3 | −98.1 | −89.8 | −134.9 | −123.5 | −107.7 | −98.6 | −109.3 | −100.1 |
| Egonol (399) | −126.3 | −131.8 | −111.4 | −116.3 | −123.3 | −128.8 | −108.9 | −113.8 | −97.6 | −102.0 |
| Erypoegin F (400) | −128.1 | −130.4 | −121.7 | −123.9 | −133.4 | −135.8 | −97.8 | −99.6 | −114.0 | −116.0 |
| Erythbidin E (401) | −99.8 | −112.9 | −94.8 | −107.3 | −99.3 | −112.4 | −89.2 | −101.0 | −92.2 | −104.3 |
| Eryvarin Q (402) | −143.2 | −139.0 | −121.3 | −117.7 | −134.3 | −130.4 | −108.5 | −105.4 | −65.7 | −63.8 |
| Gancaonin I (403) | −125.3 | −127.3 | −105.9 | −107.6 | −124.1 | −126.1 | −102.0 | −103.6 | −104.0 | −105.7 |
| Glyinflanin H (404) | −110.0 | −117.0 | −86.0 | −91.5 | −106.9 | −113.7 | −93.1 | −99.0 | −92.1 | −98.1 |
| Kuwanol A (405) | no dock | no dock | −94.5 | −82.2 | −33.8 | −29.4 | −91.3 | −79.4 | −97.8 | −85.0 |
| Licobenzofuran (406) | −134.2 | −136.3 | −111.7 | −113.5 | −129.4 | −131.5 | −114.2 | −116.1 | −86.1 | −87.5 |
| Licocoumarone (407) | −122.8 | −126.4 | −102.4 | −105.5 | −121.4 | −125.0 | −100.6 | −103.6 | −103.9 | −107.0 |
| Mulberrofuran D (408) | −150.0 | −141.1 | −137.9 | −129.8 | −151.2 | −142.2 | −118.1 | −111.1 | −101.6 | −95.5 |
| Mulberrofuran Y (409) | −153.9 | −149.2 | −127.2 | −123.2 | −146.2 | −141.7 | −124.5 | −120.7 | −115.6 | −112.0 |
| Pinosylvin (410) | −86.0 | −103.7 | −83.5 | −100.6 | −84.6 | −102.0 | −81.6 | −98.3 | −88.2 | −106.3 |
| Schweinfurthin A (411) | −48.3 | −42.4 | no dock | no dock | no dock | no dock | −133.1 | −116.9 | −76.9 | −67.5 |
| Shanciguol 3-methyl ether (412) | −130.9 | −123.5 | −113.4 | −107.0 | −123.4 | −116.4 | −112.2 | −105.9 | −102.5 | −96.7 |
| Stemofuran R (413) | −117.3 | −122.2 | −90.5 | −94.3 | −115.9 | −120.8 | −84.2 | −87.7 | −79.7 | −83.1 |
| Stilbostemin S (414) | −100.6 | −109.5 | −100.4 | −109.3 | −106.4 | −115.8 | −89.5 | −97.4 | −80.6 | −87.7 |
| Thunberginol F (415) | −96.1 | −106.9 | −95.4 | −106.1 | −101.3 | −112.6 | −96.9 | −107.8 | −98.8 | −109.8 |
| (7E,7′R,8′R)-ε-Viniferin (416) | −121.2 | −113.4 | −113.6 | −106.3 | −108.8 | −101.8 | −108.8 | −101.7 | −109.2 | −102.1 |
| (7E,7′S,8′S)-ε-Viniferin (417) | −133.0 | −124.4 | −95.4 | −89.3 | −127.6 | −119.3 | −111.9 | −104.6 | −114.7 | −107.2 |
| Phenylpropanoids and Lignans | ||||||||||
| (E)-Cinnamaldehyde (418) | −72.2 | −101.8 | −60.9 | −86.0 | −73.9 | −104.3 | −64.7 | −91.3 | −65.9 | −93.0 |
| 3,4-Dimethylcinnamaldehyde (419) | −79.6 | −105.4 | −70.3 | −93.1 | −82.3 | −109.0 | −69.9 | −92.6 | −73.9 | −97.8 |
| Methyleugenol (422) | −79.9 | −102.1 | −73.4 | −93.7 | −82.3 | −105.2 | −70.5 | −90.1 | −75.4 | −96.3 |
| p-Coumaraldehyde (420) | −76.9 | −104.4 | −65.4 | −88.9 | −77.5 | −105.3 | −69.2 | −94.1 | −73.4 | −99.8 |
| p-Methoxycinnamaldehyde (421) | −76.9 | −101.4 | −72.3 | −95.3 | −80.3 | −105.8 | −69.4 | −91.5 | −73.4 | −96.8 |
| (−)-Asarinin (423) | −137.5 | −139.7 | −103.8 | −105.4 | −134.7 | −136.8 | −101.8 | −103.5 | −98.2 | −99.8 |
| Nordihydroguaiaretic acid (424) | −112.3 | −120.3 | −94.7 | −101.5 | −110.9 | −118.8 | −97.6 | −104.5 | −95.1 | −101.8 |
| Xanthones | ||||||||||
| 2-Deoxy-4-Hydroxycudratricusxanthone D (425) | −126.3 | −123.8 | −93.9 | −92.1 | −112.0 | −109.8 | −92.4 | −90.6 | −96.8 | −94.9 |
| Calozeyloxanthone (426) | −81.7 | −81.2 | −87.9 | −87.4 | −99.7 | −99.1 | −80.5 | −80.0 | −72.2 | −71.7 |
| Cycloartobiloxanthone (427) | −106.3 | −100.9 | −47.1 | −44.7 | −107.5 | −102.0 | −94.5 | −89.7 | −67.6 | −64.2 |
| Formoxanthone C (428) | −118.9 | −116.4 | −103.8 | −101.6 | −122.4 | −119.8 | −101.7 | −99.5 | −102.5 | −100.4 |
| Garciniacowone (429) | −131.0 | −121.5 | −85.4 | −79.2 | −110.7 | −102.6 | −115.1 | −106.7 | −103.2 | −95.6 |
| Globulixanthone C (430) | −100.1 | −104.5 | −77.5 | −80.9 | −103.0 | −107.6 | −79.4 | −83.0 | −90.1 | −94.1 |
| Globulixanthone D (431) | −107.6 | −110.6 | −98.8 | −101.5 | −109.6 | −112.6 | −84.6 | −87.0 | −94.7 | −97.3 |
| Globulixanthone E (432) | −61.5 | −51.9 | −96.8 | −81.7 | −68.4 | −57.7 | −107.1 | −90.4 | −91.6 | −77.3 |
| Morellin (433) | no dock | no dock | no dock | no dock | no dock | no dock | −115.0 | −101.2 | −48.8 | −42.9 |
| Nigrolineaxanthone N (434) | −128.2 | −125.3 | −104.0 | −101.6 | −126.4 | −123.5 | −100.6 | −98.3 | −106.0 | −103.6 |
| Pinselin (435) | −98.0 | −105.2 | −87.3 | −93.8 | −95.7 | −102.8 | −78.1 | −83.8 | −83.0 | −89.1 |
| Scortechinone B (436) | no dock | no dock | −65.1 | −55.7 | no dock | no dock | −125.7 | −107.6 | −104.2 | −89.2 |
| Symphonin (437) | −134.9 | −126.4 | −112.2 | −105.0 | −125.9 | −118.0 | −96.7 | −90.6 | −93.1 | −87.2 |
| Hydrolyzable Tannins | ||||||||||
| 1,2,3,4,6-Pentagalloylglucose (335) | −65.6 | −48.1 | −137.4 | −100.9 | −16.9 | −12.4 | −170.4 | −125.0 | −130.1 | −95.4 |
| Aceritannin (438) | −151.6 | −140.3 | −116.8 | −108.1 | −158.1 | −146.4 | −118.4 | −109.6 | −118.4 | −109.6 |
| Ginnalin B (439) | −108.1 | −114.1 | −99.8 | −105.3 | −113.7 | −120.0 | −97.2 | −102.6 | −100.9 | −106.5 |
| Ginnalin C (440) | −105.5 | −111.3 | −99.0 | −104.4 | −105.8 | −111.7 | −94.3 | −99.5 | −104.0 | −109.7 |
| Panconoside A (441) | −141.1 | −116.6 | −118.4 | −97.9 | −99.7 | −82.4 | −129.1 | −106.7 | −85.0 | −70.3 |
| Miscellaneous Phenolics | ||||||||||
| 1,3,7,9-Tetrahydroxy-4,6-dimethyl-2,8-bis(2-methyl-propanoyl)-dibenzofuran (442) | −114.8 | −112.0 | −89.7 | −87.5 | −110.1 | −107.4 | −97.0 | −94.7 | −58.8 | −57.4 |
| 2′,4′-Dihydroxy-6′-methoxy-3′-methylacetophenone (443) | −85.3 | −105.5 | −69.2 | −85.6 | −83.8 | −103.7 | −69.5 | −85.9 | −70.6 | −87.3 |
| 3′,4′-Dihydroxyacetophenone (444) | −71.3 | −96.0 | −63.9 | −86.1 | −72.2 | −97.2 | −74.8 | −100.7 | −73.4 | −98.9 |
| 4′-O-Methylhonokiol (446) | −109.1 | −119.9 | −99.7 | −109.6 | −108.3 | −119.0 | −85.9 | −94.4 | −94.2 | −103.5 |
| 4-Deoxyadhumulone 2″,3″-epoxide (445) | −115.3 | −116.3 | −98.6 | −99.4 | −111.3 | −112.3 | −93.3 | −94.0 | −104.3 | −105.1 |
| 7-(3,4-Dihydroxy-5-methoxyphenyl)-1-phenyl-4-hepten-3-one (447) | −123.7 | −129.2 | −100.1 | −104.5 | −128.8 | −134.5 | −99.2 | −103.6 | −100.6 | −105.0 |
| Agrimol C (449) | −120.0 | −98.6 | −101.9 | −83.8 | −115.8 | −95.2 | −129.7 | −106.7 | −84.1 | −69.1 |
| Agrimol F (450) | −144.8 | −120.7 | −113.8 | −94.9 | −123.0 | −102.5 | −121.3 | −101.2 | −98.9 | −82.5 |
| Agrimol G (451) | −153.5 | −126.2 | −95.4 | −78.4 | −109.1 | −89.7 | −123.7 | −101.7 | −95.6 | −78.6 |
| Arzanol (452) | −133.2 | −129.7 | −94.5 | −92.0 | −132.9 | −129.4 | −106.5 | −103.7 | −113.5 | −110.5 |
| Aspidinol C (448) | −89.1 | −105.5 | −76.8 | −90.9 | −92.4 | −109.3 | −69.0 | −81.7 | −79.4 | −93.9 |
| Bruguierol C (453) | −81.4 | −99.1 | −69.5 | −84.6 | −81.2 | −98.8 | −81.3 | −98.9 | −76.9 | −93.6 |
| Cearoin (454) | −97.6 | −112.3 | −89.7 | −103.2 | −98.3 | −113.0 | −79.1 | −91.0 | −87.8 | −101.0 |
| Citrusnin A (455) | −102.4 | −119.7 | −89.3 | −104.4 | −104.3 | −122.0 | −87.5 | −102.4 | −93.3 | −109.2 |
| Cochinchinenin B (456) | −155.4 | −137.0 | −125.2 | −110.3 | −140.5 | −123.9 | −145.8 | −128.6 | −112.3 | −99.0 |
| Cochinchinenin C (457) | −155.7 | −137.3 | −117.3 | −103.4 | −143.2 | −126.3 | −135.2 | −119.2 | −106.1 | −93.5 |
| Drummondin D (458) | −112.1 | −101.8 | −102.5 | −93.0 | −89.6 | −81.4 | −118.0 | −107.2 | −91.9 | −83.4 |
| Drummondin E (459) | −147.2 | −133.5 | −117.7 | −106.8 | −112.3 | −101.8 | −116.4 | −105.5 | −99.6 | −90.3 |
| Eleutherol (460) | −90.3 | −103.9 | −83.8 | −96.4 | −84.7 | −97.4 | −76.4 | −87.9 | −63.0 | −72.4 |
| Ellagicacid (461) | −103.2 | −110.6 | −91.7 | −98.3 | −100.7 | −107.9 | −85.7 | −91.8 | −74.6 | −80.0 |
| Epicoccolide A (462) | −105.3 | −105.1 | −94.6 | −94.3 | −111.8 | −111.6 | −88.6 | −88.4 | −45.9 | −45.8 |
| Gibbilimbol A (464) | −99.1 | −115.9 | −87.7 | −102.6 | −102.4 | −119.8 | −85.1 | −99.5 | −84.9 | −99.3 |
| Gibbilimbol B (465) | −95.8 | −112.0 | −87.8 | −102.6 | −102.3 | −119.7 | −82.8 | −96.8 | −87.4 | −102.2 |
| Grifolin (466) | −127.7 | −133.1 | −98.6 | −102.7 | −130.6 | −136.0 | −106.6 | −111.1 | −90.8 | −94.7 |
| Hyperbrasilol A (467) | −126.0 | −109.4 | −97.7 | −84.8 | −128.1 | −111.1 | −113.9 | −98.9 | −99.3 | −86.2 |
| Hyperbrasilol B (468) | −129.5 | −113.4 | −74.5 | −65.2 | −95.0 | −83.3 | −105.4 | −92.4 | −97.0 | −85.0 |
| Hyperbrasilol C (469) | −97.7 | −85.5 | −122.7 | −107.4 | −121.5 | −106.3 | −110.2 | −96.5 | −108.5 | −94.9 |
| Isodrummondin D (470) | −117.5 | −106.7 | −91.0 | −82.6 | −106.3 | −96.5 | −108.8 | −98.8 | −88.1 | −80.0 |
| Isohyperbrasilol B (471) | −129.2 | −113.2 | −110.1 | −96.4 | −119.5 | −104.7 | −116.1 | −101.7 | −83.9 | −73.5 |
| Isouliginosin B (472) | −117.3 | −106.4 | −110.7 | −100.3 | −122.5 | −111.1 | −118.8 | −107.8 | −84.6 | −76.7 |
| Italipyrone (473) | −126.0 | −122.9 | −100.0 | −97.6 | −130.7 | −127.5 | −112.3 | −109.6 | −91.9 | −89.6 |
| Knerachelin A (474) | −122.6 | −131.6 | −108.9 | −117.0 | −121.2 | −130.1 | −94.5 | −101.5 | −92.3 | −99.0 |
| Knerachelin B (475) | −110.8 | −123.2 | −99.8 | −111.0 | −111.1 | −123.5 | −84.2 | −93.6 | −90.8 | −101.0 |
| Magnolol (476) | −101.7 | −113.7 | −92.5 | −103.3 | −103.1 | −115.2 | −87.0 | −97.2 | −79.1 | −88.4 |
| Myrtucommulone A (477) | −94.6 | −77.7 | −82.4 | −67.8 | no dock | no dock | −129.0 | −106.1 | −106.7 | −87.7 |
| Myrtucommulone B (478) | −42.0 | −40.5 | −77.0 | −74.2 | −49.4 | −47.6 | −74.2 | −71.6 | −76.5 | −73.8 |
| Obovatol (479) | −111.1 | −121.8 | −87.1 | −95.5 | −109.0 | −119.4 | −93.3 | −102.3 | −85.5 | −93.7 |
| Oenostacin (480) | −112.5 | −125.7 | −101.7 | −113.6 | −110.6 | −123.6 | −99.4 | −111.1 | −106.0 | −118.4 |
| Paeonol (481) | −80.1 | −104.8 | −65.8 | −86.1 | −78.0 | −102.0 | −74.2 | −97.1 | −76.4 | −99.9 |
| Perlatolic acid (482) | −144.5 | −136.1 | −112.3 | −105.8 | −140.9 | −132.7 | −121.1 | −114.1 | −109.0 | −102.7 |
| Plicatipyrone (483) | −114.3 | −109.9 | −93.0 | −89.4 | −120.6 | −116.0 | −101.0 | −97.1 | −79.5 | −76.4 |
| Propterol (484) | −92.7 | −106.6 | −85.4 | −98.2 | −96.4 | −110.9 | −83.7 | −96.2 | −87.8 | −101.0 |
| Pulverulentone B (485) | −99.5 | −115.3 | −80.5 | −93.3 | −100.2 | −116.2 | −73.4 | −85.1 | −79.1 | −91.7 |
| Quinquangulin (486) | −103.2 | −112.6 | −93.4 | −101.9 | −102.6 | −111.9 | −78.3 | −85.4 | −80.9 | −88.3 |
| Rhodomyrtone (487) | −75.2 | −71.0 | −94.6 | −89.3 | −39.1 | −36.9 | −100.6 | −94.9 | −96.9 | −91.4 |
| Rosmarinic acid (488) | −126.0 | −127.3 | −112.1 | −113.3 | −136.8 | −138.2 | −107.2 | −108.3 | −106.5 | −107.6 |
| Rubanthrone A (489) | −113.5 | −112.8 | −96.5 | −96.0 | −114.8 | −114.2 | −99.7 | −99.1 | −88.0 | −87.5 |
| Sampsone A (490) | −102.4 | −101.3 | −102.1 | −100.9 | −71.2 | −70.4 | −89.3 | −88.3 | −67.7 | −67.0 |
| Sarothralen B (491) | −40.1 | −34.8 | −99.2 | −86.2 | −96.1 | −83.5 | −99.6 | −86.6 | −70.4 | −61.2 |
| Sarothralen C (492) | −54.5 | −46.8 | −31.4 | −27.0 | −115.6 | −99.4 | −115.3 | −99.1 | −61.0 | −52.4 |
| Sarothralen D (493) | −127.3 | −109.5 | −130.9 | −112.6 | −119.5 | −102.7 | −117.1 | −100.7 | −104.1 | −89.5 |
| Shikonofuran C (494) | −133.7 | −135.3 | −103.4 | −104.7 | −123.6 | −125.1 | −102.4 | −103.7 | −109.1 | −110.4 |
| Shikonofuran D (495) | −126.6 | −129.9 | −110.3 | −113.1 | −126.9 | −130.1 | −103.4 | −106.1 | −105.3 | −108.0 |
| Shikonofuran E (496) | −126.6 | −128.4 | −113.5 | −115.1 | −134.9 | −136.8 | −105.2 | −106.7 | −109.5 | −111.1 |
| Sinapic acid (497) | −100.7 | −119.2 | −83.6 | −98.9 | −106.4 | −125.9 | −89.9 | −106.4 | −92.4 | −109.4 |
| Walrycin A (498) | −75.6 | −97.4 | −66.0 | −85.0 | −75.8 | −97.5 | −64.8 | −83.4 | −71.6 | −92.2 |
| Quinones | ||||||||||
| 2,6-Dimethoxy-1,4-benzoquinone (336) | −77.5 | −101.0 | −66.2 | −86.3 | −80.9 | −105.4 | −61.5 | −80.1 | −63.7 | −82.9 |
| 2-Methyl-6-prenyl-1,4-benzoquinone (337) | −86.8 | −108.5 | −80.2 | −100.3 | −90.5 | −113.1 | −81.8 | −102.2 | −78.9 | −98.7 |
| Omphalone (499) | −91.7 | −115.1 | −79.2 | −99.3 | −87.4 | −109.7 | −78.2 | −98.2 | −81.9 | −102.7 |
| Primin (500) | −88.9 | −107.9 | −89.0 | −108.0 | −92.3 | −112.0 | −79.0 | −95.8 | −78.6 | −95.4 |
| 1,4-Naphthoquinone (338) | −71.2 | −94.6 | −64.6 | −85.9 | −69.4 | −92.2 | −57.8 | −76.8 | −66.9 | −88.9 |
| 2-Acetylnaphtho[2,3-b]furan-4,9-dione (339) | −98.4 | −113.8 | −91.9 | −106.3 | −97.3 | −112.6 | −82.4 | −95.3 | −93.1 | −107.7 |
| Alkannin (340) | −103.1 | −112.2 | −94.3 | −102.6 | −97.0 | −105.5 | −88.4 | −96.2 | −60.7 | −66.1 |
| Isobutyrylshikonin (341) | −122.6 | −124.1 | −101.9 | −103.2 | −115.6 | −117.0 | −90.3 | −91.4 | −74.4 | −75.3 |
| Lapachol (501) | −95.5 | −110.1 | −90.4 | −104.3 | −91.7 | −105.8 | −75.2 | −86.7 | −86.9 | −100.3 |
| Mamegakinone (502) | −112.2 | −111.9 | −89.4 | −89.2 | −111.0 | −110.7 | −87.8 | −87.6 | −83.8 | −83.6 |
| Menadione (503) | −75.5 | −97.6 | −70.0 | −90.4 | −72.4 | −93.6 | −61.6 | −79.6 | −70.6 | −91.3 |
| Rhinacanthin C (504) | −145.5 | −140.7 | −99.2 | −96.0 | −143.5 | −138.8 | −104.8 | −101.4 | −97.3 | −94.1 |
| Rhinacanthin D (505) | −145.7 | −141.2 | −98.3 | −95.2 | −151.8 | −147.1 | −104.8 | −101.6 | −107.6 | −104.2 |
| Rhinacanthin G (506) | −142.4 | −136.0 | −111.1 | −106.2 | −147.4 | −140.8 | −102.9 | −98.3 | −95.8 | −91.5 |
| Rhinacanthin H (507) | −155.9 | −148.9 | −107.4 | −102.6 | −140.9 | −134.6 | −104.7 | −100.0 | −101.1 | −96.6 |
| Rhinacanthin I (508) | −142.1 | −135.7 | −119.4 | −114.1 | −146.1 | −139.6 | −104.6 | −99.9 | −108.7 | −103.9 |
| Rhinacanthin J (509) | −137.0 | −131.1 | −98.0 | −93.7 | −153.2 | −146.5 | −110.6 | −105.8 | −111.7 | −106.9 |
| Rhinacanthin K (510) | −145.0 | −136.6 | −108.8 | −102.5 | −142.7 | −134.4 | −113.5 | −106.9 | −104.8 | −98.7 |
| Rhinacanthin L (511) | −146.7 | −136.6 | −102.7 | −95.6 | −146.1 | −136.0 | −110.6 | −103.0 | −97.9 | −91.2 |
| Rhinacanthin M (512) | −133.5 | −134.4 | −110.1 | −110.8 | −131.2 | −132.1 | −94.2 | −94.8 | −92.3 | −93.0 |
| Shikonin acetate (513) | −110.7 | −115.2 | −91.7 | −95.3 | −109.2 | −113.6 | −99.7 | −103.7 | −89.0 | −92.5 |
| β,β-Dimethylacrylshikonin (514) | −125.9 | −126.0 | −106.0 | −106.2 | −122.4 | −122.6 | −99.5 | −99.7 | −77.4 | −77.5 |
| β-Hydroxyisovaleryshikonin (515) | −123.2 | −121.4 | −104.1 | −102.6 | −124.3 | −122.5 | −101.6 | −100.1 | −99.3 | −97.9 |
| 1-Hydroxy-3-hydroxymethylanthraquinone (516) | −95.5 | −108.4 | −84.0 | −95.3 | −92.7 | −105.2 | −79.7 | −90.5 | −84.5 | −95.9 |
| Aloeemodin (518) | −96.3 | −107.1 | −85.3 | −94.9 | −96.2 | −107.0 | −80.0 | −89.0 | −85.6 | −95.2 |
| Islandicin (519) | −91.5 | −101.7 | −80.1 | −89.1 | −87.7 | −97.6 | −75.2 | −83.6 | −70.8 | −78.8 |
| Newbouldiaquinone (521) | −95.3 | −93.4 | −93.4 | −91.6 | −96.9 | −95.0 | −93.8 | −92.0 | −86.7 | −85.0 |
| Newbouldiaquinone A (520) | −132.6 | −128.3 | −88.9 | −86.0 | −133.8 | −129.5 | −106.6 | −103.1 | −94.9 | −91.9 |
| Rhein (522) | −101.3 | −110.8 | −91.0 | −99.5 | −101.3 | −110.8 | −80.8 | −88.4 | −95.8 | −104.8 |
| 15,16-Dihydrotanshinone I (517) | −96.1 | −105.8 | −80.7 | −88.9 | −93.2 | −102.6 | −74.5 | −82.1 | −72.5 | −79.8 |
| Acetylene, Glucoside, and Other Miscellaneous Phytochemicals | ||||||||||
| 1,7-Diphenyl-4-(2-phenylethyl)-1-heptene-3,5-dione (530) | −138.6 | −137.3 | −108.3 | −107.3 | −135.2 | −133.9 | −115.7 | −114.6 | −103.7 | −102.8 |
| 1,7-Diphenyl-5-hepten-3-one (531) | −107.5 | −120.4 | −100.8 | −112.9 | −110.4 | −123.7 | −88.4 | −99.0 | −87.3 | −97.8 |
| 3′-Demothexycyclocurcumin (532) | −122.3 | −126.2 | −91.0 | −93.9 | −121.0 | −124.9 | −112.7 | −116.2 | −99.0 | −102.2 |
| 5,7-Dihydroxyphthalide (533) | −74.6 | −97.6 | −64.7 | −84.6 | −75.0 | −98.1 | −72.3 | −94.5 | −73.2 | −95.7 |
| 6-Methyl-4,5-dithia-2-octene (534) | −66.5 | −87.6 | −60.1 | −79.2 | −67.5 | −89.0 | −57.5 | −75.8 | −61.2 | −80.7 |
| 7-Epiclusianone (535) | −105.3 | −95.2 | −102.8 | −92.9 | −101.1 | −91.4 | −105.1 | −95.1 | −97.4 | −88.1 |
| Allamandin (536) | −103.4 | −110.1 | −101.6 | −108.1 | −99.3 | −105.7 | −95.2 | −101.3 | −81.8 | −87.1 |
| Allicin (537) | −72.4 | −95.4 | −61.9 | −81.6 | −69.8 | −92.0 | −59.7 | −78.6 | −65.3 | −86.0 |
| Amadannulen (538) | −110.1 | −109.6 | −102.9 | −102.5 | −82.5 | −82.1 | −99.7 | −99.3 | −110.5 | −110.0 |
| Anemonin (539) | −86.9 | −108.3 | −65.0 | −81.0 | −90.4 | −112.7 | −80.5 | −100.3 | −41.5 | −51.7 |
| Antibiotic CZ 34 (540) | −85.8 | −102.2 | −83.0 | −98.8 | −87.1 | −103.7 | −79.5 | −94.6 | −80.2 | −95.4 |
| Argutone (541) | −83.6 | −105.3 | −68.8 | −86.6 | −81.2 | −102.3 | −77.0 | −97.0 | −78.7 | −99.1 |
| Bakuchiol (542) | −103.0 | −116.6 | −90.8 | −102.8 | −103.6 | −117.2 | −89.6 | −101.4 | −94.5 | −107.0 |
| Brasiliensophyllic acid A (543) | −101.8 | −88.7 | −101.7 | −88.7 | −93.0 | −81.1 | −121.4 | −105.8 | −72.6 | −63.3 |
| Brasiliensophyllic acid C (544) | −110.4 | −95.4 | −108.3 | −93.6 | −96.8 | −83.7 | −129.6 | −112.1 | −105.9 | −91.6 |
| Centrolobin (545) | −116.5 | −123.4 | −94.4 | −100.0 | −119.7 | −126.8 | −88.9 | −94.2 | −93.5 | −99.0 |
| Chamone I (546) | −102.3 | −88.6 | −107.6 | −93.3 | −109.9 | −95.3 | −111.1 | −96.3 | −97.7 | −84.7 |
| Chamone II (547) | −109.3 | −94.9 | −104.3 | −90.5 | no dock | no dock | −108.4 | −94.1 | −66.3 | −57.5 |
| Champanone A (548) | −105.8 | −119.8 | −91.8 | −104.0 | −105.5 | −119.5 | −86.2 | −97.5 | −91.8 | −103.9 |
| Dhelwangin (549) | −94.5 | −111.9 | −94.5 | −111.8 | −100.7 | −119.2 | −78.0 | −92.3 | −82.8 | −98.0 |
| Garcinoic acid (550) | −148.8 | −142.1 | −106.8 | −102.0 | −151.0 | −144.2 | −131.6 | −125.7 | −103.9 | −99.3 |
| Ginkgolide A (551) | −48.2 | −46.7 | −91.8 | −88.9 | −51.4 | −49.8 | −96.8 | −93.8 | −36.7 | −35.6 |
| Guttiferone E (552) | −90.7 | −77.2 | −119.6 | −101.8 | −116.8 | −99.4 | −136.4 | −116.1 | −102.5 | −87.3 |
| Helipyrone B (553) | −102.4 | −109.2 | −94.8 | −101.1 | −104.1 | −111.0 | −78.7 | −84.0 | −77.0 | −82.1 |
| Helipyrone C (554) | −97.0 | −105.1 | −91.3 | −98.9 | −98.0 | −106.1 | −73.2 | −79.3 | −64.0 | −69.3 |
| Ialibinone A (555) | −86.5 | −88.8 | −84.5 | −86.7 | −85.0 | −87.2 | −87.0 | −89.2 | −77.9 | −79.9 |
| Ialibinone B (556) | −56.5 | −57.9 | −81.3 | −83.4 | −30.3 | −31.0 | −87.6 | −89.9 | −70.1 | −71.9 |
| Ialibinone C (557) | −93.8 | −94.9 | −89.0 | −90.1 | −94.0 | −95.1 | −94.9 | −96.0 | −87.7 | −88.7 |
| Ialibinone D (558) | −38.3 | −38.8 | −91.8 | −92.9 | −81.6 | −82.6 | −86.5 | −87.6 | −79.8 | −80.8 |
| Isobrasiliensophyllic acid A (559) | −97.3 | −84.8 | −103.3 | −90.0 | −96.0 | −83.7 | −131.0 | −114.2 | −83.2 | −72.6 |
| Moskachan C (560) | −92.1 | −109.3 | −90.5 | −107.5 | −95.0 | −112.8 | −86.1 | −102.2 | −83.4 | −99.0 |
| Nimbolide (561) | −37.6 | −34.9 | −77.1 | −71.4 | −67.9 | −63.0 | −107.4 | −99.5 | −82.6 | −76.6 |
| Pectinolide H (562) | −112.0 | −124.8 | −98.6 | −109.9 | −117.0 | −130.4 | −93.2 | −103.9 | −97.7 | −108.9 |
| Propolone A (563) | −107.8 | −97.5 | −100.7 | −91.1 | −70.1 | −63.4 | −108.5 | −98.1 | −78.0 | −70.6 |
| Sellovicine B (564) | −98.7 | −115.1 | −93.4 | −108.9 | −98.2 | −114.5 | −79.9 | −93.2 | −88.5 | −103.2 |
| Simonin A (565) | −109.7 | −105.6 | −94.3 | −90.8 | −102.8 | −99.0 | −113.8 | −109.5 | −85.5 | −82.3 |
| Tenulin (566) | −102.9 | −109.7 | −74.4 | −79.4 | −101.6 | −108.3 | −82.0 | −87.5 | −66.7 | −71.1 |
| Atractylodin (522) | −93.5 | −118.6 | −86.5 | −109.6 | −92.2 | −116.9 | −75.8 | −96.1 | −80.7 | −102.4 |
| Atractylodinol (523) | −100.8 | −124.3 | −91.0 | −112.2 | −97.4 | −120.1 | −84.5 | −104.2 | −85.4 | −105.3 |
| Capillene (342) | −71.8 | −96.3 | −68.5 | −91.8 | −70.1 | −94.0 | −65.1 | −87.2 | −71.6 | −96.0 |
| Peniophorin A (524) | −116.7 | −127.9 | −97.5 | −106.9 | −112.5 | −123.3 | −95.5 | −104.7 | −89.5 | −98.1 |
| Peniophorin B (525) | −97.9 | −118.0 | −89.8 | −108.3 | −104.4 | −125.8 | −86.9 | −104.8 | −88.6 | −106.8 |
| Thiarubrin A (526) | −93.0 | −109.4 | −92.0 | −108.2 | −90.0 | −105.8 | −73.2 | −86.1 | −75.1 | −88.4 |
| Arbutin (527) | −93.4 | −103.6 | −86.0 | −95.4 | −89.1 | −98.9 | −87.1 | −96.6 | −90.3 | −100.2 |
| Aucubin (528) | −105.2 | −107.7 | −98.2 | −100.5 | −110.1 | −112.7 | −93.1 | −95.3 | −79.9 | −81.8 |
| Diospyrodin (529) | −122.2 | −129.2 | −101.7 | −107.5 | −105.4 | −111.5 | −97.3 | −102.9 | −96.2 | −101.8 |
| Known/Synthetic Inhibitors | ||||||||||
| (−)-Epicatechin | −95.4 | −103.6 | −90.6 | −98.3 | −93.5 | −101.6 | - | - | - | - |
| (−)-Epicatechin gallate | −140.4 | −132.5 | −87.6 | −82.6 | −143.4 | −135.3 | - | - | - | - |
| (−)-Epigallocatechin | −95.9 | −102.3 | −91.8 | −97.9 | −98.5 | −105.0 | - | - | - | - |
| (−)-Epigallocatechin 3-gallate | −141.3 | −131.8 | −90.8 | −84.7 | −142.9 | −133.3 | - | - | - | - |
| Norfloxacin | −123.6 | −130.0 | −94.0 | −98.9 | −112.3 | −118.2 | - | - | - | - |
| Novobiocin | −119.7 | −101.2 | −114.2 | −96.6 | −120.7 | −102.1 | - | - | - | - |
| Quercetin (272) | −102.9 | −110.3 | −94.1 | −100.8 | −96.8 | −103.7 | - | - | - | - |
| 3461-2296 [97] (573) | - | - | - | - | - | - | −99.3 | −102.5 | - | - |
| 4236-0754 [97] (574) | - | - | - | - | - | - | −102.1 | −101.3 | - | - |
| 5591-1074 [97] (575) | - | - | - | - | - | - | −115.1 | −110.7 | - | - |
| C609-0168 [97] (576) | - | - | - | - | - | - | −104.2 | −98.4 | - | - |
| C609-0177 [97] (577) | - | - | - | - | - | - | −110.9 | −100.0 | - | - |
| C609-0383 [97] (578) | - | - | - | - | - | - | −128.2 | −117.5 | - | - |
| Ligand | MtUGM | MtCYP121 | EcLigA | MtLigA | SaLigA | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | Edock | DSnorm | |
| Indole Alkaloids | ||||||||||
| 1-Hydroxy-6,7-dimethoxy-3-methylcarbazole (1) | −80.2 | −90.7 | −93.5 | −105.7 | −91.7 | −103.7 | −84.7 | −95.7 | −99.7 | −112.7 |
| 11-Methoxytubotaiwine (2) | −82.4 | −83.7 | −91.6 | −93.0 | −113.5 | −115.3 | −86.8 | −88.2 | −88.9 | −90.3 |
| 12-Methoxy-4-methylvoachalotine (3) | −98.3 | −95.0 | −91.6 | −88.6 | −103.3 | −99.8 | −68.4 | −66.1 | −88.4 | −85.5 |
| 3-Prenylindole (4) | −77.8 | −98.1 | −84.1 | −106.1 | −78.8 | −99.4 | −82.0 | −103.4 | −86.8 | −109.5 |
| Affinisine (5) | −84.8 | −90.2 | −83.4 | −88.8 | −97.0 | −103.2 | −66.4 | −70.7 | −89.9 | −95.7 |
| Apparicine (6) | −82.4 | −92.3 | −78.8 | −88.2 | −87.2 | −97.7 | −78.1 | −87.5 | −82.9 | −92.8 |
| Aristolactam I (7) | −95.0 | −102.8 | −98.9 | −107.1 | −101.9 | −110.3 | −91.4 | −98.9 | −107.1 | −115.9 |
| Clausenawalline A a (8) | no dock | no dock | −110.2 | −96.3 | −103.7 | −90.7 | −64.2 | −56.1 | −90.4 | −79.0 |
| Cryptoheptine (9) | −85.2 | −95.7 | −82.6 | −92.8 | −88.4 | −99.2 | −67.3 | −75.6 | −95.6 | −107.3 |
| Diploceline (10) | −82.0 | −82.1 | −97.4 | −97.6 | −112.5 | −112.8 | −96.5 | −96.7 | −54.6 | −54.7 |
| Discarine B (11) | −91.2 | −78.9 | −138.3 | −119.7 | −55.6 | −48.1 | −89.1 | −77.1 | −116.2 | −100.5 |
| Ibogamine (12) | −84.5 | −92.8 | −83.5 | −91.7 | −88.9 | −97.6 | −87.7 | −96.4 | −87.5 | −96.1 |
| Iboxygaine (13) | −101.4 | −105.8 | −96.1 | −100.3 | −104.7 | −109.3 | −98.0 | −102.3 | −103.3 | −107.9 |
| Isovoacangine (14) | −94.8 | −95.0 | −96.3 | −96.6 | −106.1 | −106.4 | −82.0 | −82.3 | −88.9 | −89.2 |
| Rugosanine B (15) | −45.3 | −38.2 | −139.7 | −117.7 | −128.5 | −108.2 | −111.9 | −94.2 | −123.4 | −104.0 |
| Suaveolindole (16) | −103.3 | −105.1 | −102.6 | −104.4 | −109.3 | −111.2 | −99.0 | −100.7 | −98.4 | −100.1 |
| Toussaintine B (17) | −92.9 | −99.8 | −87.6 | −94.2 | −97.4 | −104.7 | −94.5 | −101.6 | −108.3 | −116.4 |
| Isoquinoline Alkaloids | ||||||||||
| 8-Acetonyldihydroavicine (18) | −103.4 | −100.4 | −101.8 | −98.8 | −108.6 | −105.5 | −99.1 | −96.3 | −101.6 | −98.7 |
| 8-Acetonyldihydronitidine (19) | −96.2 | −94.7 | −102.1 | −100.5 | −116.2 | −114.5 | −90.8 | −89.4 | −99.6 | −98.0 |
| Antofine (20) | −67.2 | −67.7 | −103.0 | −103.7 | −112.8 | −113.7 | −96.1 | −96.9 | −116.8 | −117.7 |
| Berbamine (24) | no dock | no dock | −48.9 | −41.5 | −80.8 | −68.6 | −58.9 | −50.0 | −64.0 | −54.3 |
| Berberine (21) | −100.6 | −104.0 | −96.1 | −99.4 | −95.5 | −98.8 | −97.8 | −101.1 | −99.0 | −102.3 |
| Bisnorthalphenine (22) | −84.6 | −88.6 | −92.6 | −97.0 | −105.0 | −110.0 | −87.9 | −92.1 | −105.9 | −111.0 |
| Cepharanthine (25) | no dock | no dock | −107.2 | −91.0 | no dock | no dock | −84.3 | −71.6 | −80.0 | −68.0 |
| Cryptopleurine (23) | −71.2 | −70.8 | −101.3 | −100.8 | −109.7 | −109.2 | −91.6 | −91.1 | −116.1 | −115.5 |
| Emetine (26) | −99.7 | −91.5 | −111.8 | −102.7 | −115.3 | −105.8 | −97.0 | −89.0 | −78.6 | −72.1 |
| Hydrastine (27) | −108.1 | −107.0 | −108.0 | −106.9 | −113.9 | −112.7 | −108.4 | −107.2 | −106.8 | −105.7 |
| Isotrilobine (29) | no dock | no dock | −140.7 | −121.6 | −75.4 | −65.1 | −66.2 | −57.2 | −61.9 | −53.5 |
| Jatrorrhizine (28) | −91.8 | −94.8 | −98.8 | −101.9 | −93.9 | −96.9 | −94.0 | −97.0 | −100.2 | −103.4 |
| Lauroscholtzine (31) | −90.5 | −93.1 | −92.2 | −94.8 | −102.7 | −105.7 | −87.3 | −89.9 | −95.3 | −98.1 |
| Methothalistyline (30) | −99.6 | −80.1 | −141.2 | −113.7 | −110.4 | −88.8 | −96.0 | −77.3 | −113.7 | −91.5 |
| N-Demethylthalphenine (32) | −83.8 | −86.6 | −84.4 | −87.1 | −106.8 | −110.3 | −93.1 | −96.2 | −106.7 | −110.2 |
| Obamegine (34) | no dock | no dock | −105.4 | −90.1 | −41.8 | −35.8 | −82.4 | −70.4 | −72.2 | −61.7 |
| Oxyacanthine (35) | no dock | no dock | −125.4 | −106.4 | −85.8 | −72.8 | −58.8 | −49.9 | −88.1 | −74.7 |
| Pennsylvanine (36) | −76.5 | −62.4 | −134.2 | −109.6 | −140.8 | −114.9 | −101.2 | −82.6 | −124.9 | −102.0 |
| Thaliadanine (38) | −98.9 | −79.5 | −132.1 | −106.2 | −155.7 | −125.2 | −103.6 | −83.3 | −139.6 | −112.2 |
| Thalicarpine (37) | −75.0 | −60.8 | −110.1 | −89.3 | −119.0 | −96.5 | −96.2 | −78.0 | −124.3 | −100.8 |
| Thalidasine (39) | no dock | no dock | −103.1 | −85.4 | no dock | no dock | −64.0 | −53.0 | −96.6 | −80.1 |
| Thalistyline (40) | −58.8 | −47.7 | −159.7 | −129.4 | −145.7 | −118.1 | −101.9 | −82.6 | −116.1 | −94.1 |
| Thalmelatine (41) | −44.0 | −36.0 | −130.7 | −106.7 | −134.7 | −109.9 | −102.3 | −83.6 | −115.7 | −94.5 |
| Thalmirabine (42) | no dock | no dock | −20.7 | −17.0 | no dock | no dock | −101.7 | −83.6 | −70.2 | −57.7 |
| Thalphenine (33) | −86.5 | −88.0 | −94.8 | −96.5 | −110.8 | −112.8 | −86.7 | −88.3 | −113.7 | −115.8 |
| Thalrugosidine (43) | no dock | no dock | −96.5 | −80.6 | no dock | no dock | −78.1 | −65.2 | −93.4 | −78.0 |
| Thalrugosine (44) | no dock | no dock | −119.6 | −101.5 | −47.1 | −40.0 | −89.9 | −76.3 | −73.9 | −62.7 |
| Piperidine, Pyrrole, Pyrrolizidine, Quinoline, and Steroidal Alkaloids | ||||||||||
| Aconicaramide (46) | −79.7 | −94.6 | −83.7 | −99.4 | −84.2 | −99.9 | −83.0 | −98.6 | −95.6 | −113.4 |
| Lasiocarpine (47) | −119.3 | −115.3 | −123.0 | −118.9 | −119.0 | −115.0 | −95.6 | −92.4 | −118.7 | −114.8 |
| Lasiocarpine N-oxide (48) | −122.3 | −116.7 | −106.5 | −101.7 | −122.5 | −116.9 | −89.5 | −85.4 | −114.0 | −108.8 |
| Piperine (45) | −94.7 | −103.4 | −105.3 | −115.0 | −100.4 | −109.7 | −99.8 | −109.0 | −105.8 | −115.5 |
| 4-Methoxy-1-methyl-2(1H)-quinolinone (49) | −61.8 | −77.4 | −75.7 | −94.8 | −70.9 | −88.8 | −61.3 | −76.8 | −75.2 | −94.1 |
| Cryptolepine (50) | −70.3 | −82.2 | −84.6 | −99.0 | −89.9 | −105.1 | −73.6 | −86.1 | −85.1 | −99.5 |
| Neocryptolepine (51) | −73.3 | −85.7 | −84.7 | −99.0 | −84.0 | −98.2 | −72.4 | −84.6 | −87.5 | −102.3 |
| Pteleine (52) | −75.0 | −88.1 | −76.8 | −90.3 | −81.8 | −96.1 | −78.3 | −92.0 | −88.1 | −103.5 |
| Veprisinium (53) | −93.4 | −96.7 | −79.4 | −82.2 | −102.1 | −105.7 | −82.7 | −85.6 | −92.4 | −95.8 |
| Conessine (54) | −94.3 | −95.6 | −92.2 | −93.5 | −88.0 | −89.3 | −76.3 | −77.4 | −78.5 | −79.6 |
| Irehdiamine A (55) | −87.0 | −91.8 | −90.3 | −95.2 | −84.5 | −89.1 | −79.0 | −83.4 | −82.6 | −87.1 |
| Solacassine (56) | −59.2 | −55.9 | −88.1 | −83.1 | −105.2 | −99.2 | −73.6 | −69.4 | −71.9 | −67.8 |
| Solanocapsine (57) | −72.0 | −68.6 | −100.0 | −95.2 | −104.4 | −99.4 | −83.9 | −79.9 | −83.6 | −79.6 |
| Tomatidine (58) | −63.1 | −60.9 | −86.8 | −83.7 | −94.0 | −90.6 | −84.5 | −81.5 | −85.2 | −82.2 |
| Miscellaneous Alkaloids | ||||||||||
| 2-(Methoxyamino)-4H-1-benzopyran-3,4,5,7-tetrol (59) | −86.1 | −99.4 | −91.0 | −105.1 | −88.4 | −102.1 | −84.3 | −97.4 | −92.5 | −106.8 |
| Abyssenine C (60) | −86.7 | −81.7 | −108.1 | −101.8 | −112.6 | −106.0 | −89.2 | −84.0 | −98.3 | −92.6 |
| Amphibine H (61) | −13.3 | −11.3 | −146.3 | −124.3 | −99.4 | −84.5 | −99.4 | −84.5 | −70.2 | −59.7 |
| Cepharatine A (62) | −87.5 | −92.7 | −85.6 | −90.6 | −93.4 | −98.9 | −70.2 | −74.3 | −95.2 | −100.7 |
| Curcamide (63) | −94.1 | −107.9 | −97.6 | −111.9 | −97.1 | −111.4 | −101.0 | −115.8 | −108.6 | −124.6 |
| Drodrenin (64) | −148.0 | −133.6 | −133.2 | −120.2 | −144.1 | −130.1 | −138.5 | −125.0 | −158.0 | −142.6 |
| Eschscholtzidine (65) | −88.2 | −90.9 | −91.0 | −93.8 | −97.0 | −100.0 | −86.0 | −88.6 | −94.4 | −97.3 |
| Jervine (66) | −32.2 | −30.8 | −87.8 | −84.0 | −80.0 | −76.4 | −94.9 | −90.7 | −89.5 | −85.5 |
| Matrine (67) | −75.9 | −86.9 | −65.2 | −74.6 | −86.2 | −98.6 | −71.9 | −82.3 | −89.5 | −102.4 |
| Mucronine H (68) | −69.0 | −63.4 | −118.4 | −108.8 | −126.1 | −115.9 | −95.6 | −87.9 | −119.6 | −109.9 |
| N-Benzoylmescaline (69) | −97.8 | −103.3 | −100.3 | −106.0 | −104.6 | −110.5 | −97.8 | −103.3 | −115.5 | −122.0 |
| Nummularine B (70) | no dock | no dock | −148.7 | −127.4 | −135.6 | −116.2 | −101.4 | −86.9 | −81.5 | −69.8 |
| Nummularine S (71) | −37.6 | −33.6 | −122.1 | −109.2 | −131.4 | −117.4 | −109.9 | −98.2 | −114.2 | −102.1 |
| Scutianine B (72) | −20.2 | −17.5 | −127.6 | −110.8 | −86.5 | −75.1 | −95.8 | −83.1 | −93.0 | −80.7 |
| Shahidine (73) | −94.7 | −104.2 | −104.7 | −115.2 | −104.5 | −114.9 | −109.1 | −120.0 | −116.1 | −127.7 |
| Thaliglucinone (74) | −96.8 | −97.3 | −106.1 | −106.7 | −125.1 | −125.8 | −96.4 | −97.0 | −123.8 | −124.5 |
| Triisopenylguanidine (75) | −94.6 | −105.4 | −91.8 | −102.2 | −98.7 | −109.8 | −103.8 | −115.5 | −107.9 | −120.1 |
| Tuberine (76) | −133.6 | −123.6 | −124.6 | −115.3 | −124.3 | −115.0 | −122.7 | −113.5 | −126.7 | −117.2 |
| Monoterpenoids | ||||||||||
| Linalool (77) | −64.8 | −86.9 | −66.7 | −89.4 | −64.7 | −86.7 | −72.4 | −97.0 | −73.5 | −98.5 |
| Thymol (78) | −60.7 | −82.1 | −60.7 | −82.0 | −63.7 | −86.2 | −63.3 | −85.7 | −72.7 | −98.3 |
| Thymoquinol (79) | −65.8 | −86.1 | −66.8 | −87.4 | −70.0 | −91.6 | −65.9 | −86.2 | −78.1 | −102.1 |
| β-Dolabrin (80) | −70.7 | −93.2 | −73.0 | −96.2 | −73.8 | −97.4 | −73.3 | −96.6 | −76.0 | −100.2 |
| β-Thujaplicin (81) | −70.5 | −92.6 | −71.8 | −94.3 | −71.7 | −94.1 | −73.4 | −96.4 | −75.1 | −98.6 |
| Sesquiterpenoids | ||||||||||
| 11,13-Dehydroeriolin (82) | −91.9 | −103.0 | −77.5 | −86.9 | −82.3 | −92.2 | −73.4 | −82.3 | −81.8 | −91.6 |
| 2,10-Bisaboladien-1-one (83) | −80.7 | −96.1 | −84.0 | −100.0 | −84.1 | −100.1 | −87.4 | −104.1 | −93.1 | −110.9 |
| 2-Hydroxycalamenene (84) | −71.5 | −85.4 | −67.2 | −80.2 | −76.4 | −91.2 | −66.6 | −79.5 | −74.3 | −88.7 |
| 2-Methoxyfurano-9-guaien-8-one (85) | −85.7 | −96.5 | −71.7 | −80.8 | −94.1 | −106.0 | −83.7 | −94.3 | −90.2 | −101.6 |
| 4α,10α-Dihydroxy-1,11(13)guaiadien-12,8-olide (93) | −89.3 | −100.1 | −80.6 | −90.3 | −83.3 | −93.3 | −79.6 | −89.1 | −82.7 | −92.6 |
| 4α,10β-Dihydroxy-1,11(13)guaiadien-12,8-olide (89) | −91.6 | −102.7 | −78.9 | −88.4 | −85.9 | −96.2 | −77.3 | −86.5 | −81.0 | −90.8 |
| Alantolactone (86) | −73.4 | −85.9 | −65.7 | −76.8 | −73.9 | −86.4 | −55.5 | −65.0 | −46.6 | −54.5 |
| Alliacol A (87) | −76.5 | −85.8 | −70.3 | −78.8 | −78.1 | −87.5 | −68.3 | −76.6 | −67.6 | −75.8 |
| Alliacol B (88) | −76.7 | −85.9 | −74.6 | −83.6 | −81.0 | −90.8 | −67.0 | −75.1 | −73.5 | −82.4 |
| Artemisinic acid (113) | −78.3 | −91.3 | −71.3 | −83.1 | −76.3 | −88.9 | −76.2 | −88.8 | −75.1 | −87.6 |
| Baileyolin (90) | −99.8 | −100.6 | −99.9 | −100.7 | −105.9 | −106.8 | −96.8 | −97.7 | −88.2 | −88.9 |
| Bilobalide A (91) | −90.1 | −94.1 | −84.9 | −88.7 | −93.5 | −97.6 | −59.4 | −62.0 | −67.4 | −70.3 |
| Confertin (92) | −88.6 | −101.4 | −70.8 | −81.0 | −79.5 | −91.0 | −60.7 | −69.4 | −74.3 | −85.0 |
| Cyperenal (94) | −70.5 | −84.2 | −56.1 | −67.0 | −65.5 | −78.2 | −52.8 | −63.0 | −48.1 | −57.5 |
| Cyperenol (95) | −71.6 | −85.3 | −59.7 | −71.0 | −68.2 | −81.1 | −55.3 | −65.8 | −45.0 | −53.6 |
| Furanodienone (97) | −88.7 | −104.0 | −77.1 | −90.5 | −84.2 | −98.8 | −67.9 | −79.6 | −73.5 | −86.2 |
| Ganodermycin (96) | −101.0 | −110.9 | −90.2 | −99.1 | −97.0 | −106.6 | −97.4 | −107.0 | −111.3 | −122.3 |
| Helenalin (98) | −90.4 | −101.6 | −70.3 | −78.9 | −84.6 | −95.0 | −67.2 | −75.5 | −54.8 | −61.5 |
| Hydrogrammic acid (99) | −87.2 | −98.2 | −80.1 | −90.2 | −88.2 | −99.3 | −83.5 | −94.0 | −83.5 | −94.0 |
| Isoalantolactone (100) | −78.2 | −91.4 | −65.3 | −76.4 | −74.7 | −87.4 | −54.7 | −64.0 | −70.6 | −82.6 |
| Ivaxillin (101) | −88.1 | −98.4 | −76.0 | −84.9 | −80.6 | −90.1 | −72.5 | −81.1 | −77.1 | −86.1 |
| Petrovin A (102) | −87.4 | −100.0 | −70.5 | −80.6 | −71.9 | −82.3 | −74.8 | −85.6 | −66.6 | −76.2 |
| Petrovin B (103) | −81.9 | −93.4 | −78.5 | −89.5 | −73.2 | −83.5 | −76.1 | −86.8 | −56.3 | −64.3 |
| Polygodial (104) | −78.2 | −91.2 | −67.9 | −79.2 | −76.6 | −89.4 | −67.8 | −79.0 | −73.3 | −85.5 |
| Rishitin (105) | −72.7 | −86.3 | −79.0 | −93.8 | −87.2 | −103.5 | −78.5 | −93.1 | −86.6 | −102.8 |
| Xanthorrhizol (106) | −82.0 | −97.9 | −82.2 | −98.1 | −82.1 | −98.0 | −87.1 | −104.0 | −92.7 | −110.7 |
| α-Amorphene (107) | −67.1 | −81.9 | −64.6 | −78.8 | −68.5 | −83.6 | −71.7 | −87.5 | −62.8 | −76.7 |
| α-Cadinene (108) | −68.9 | −84.2 | −72.5 | −88.5 | −73.1 | −89.2 | −66.9 | −81.6 | −73.3 | −89.5 |
| α-Copaene (110) | −61.0 | −76.3 | −56.4 | −70.5 | −56.3 | −70.4 | −56.4 | −70.5 | −54.4 | −68.0 |
| α-Muurolene (109) | −68.1 | −83.1 | −72.2 | −88.2 | −68.8 | −84.0 | −66.3 | −80.9 | −68.9 | −84.2 |
| γ-Cadinene (112) | −70.6 | −86.2 | −69.5 | −84.8 | −74.0 | −90.3 | −72.1 | −88.0 | −71.3 | −87.1 |
| Diterpenoids | ||||||||||
| 1,12-Diacetyljativatriol (114) | −82.4 | −80.1 | −92.1 | −89.5 | −112.9 | −109.7 | −82.5 | −80.2 | −85.1 | −82.8 |
| 12-Oxo-3,13(16)-clerodadien-15-oic acid (115) | −99.6 | −104.9 | −87.3 | −91.9 | −97.4 | −102.6 | −88.6 | −93.3 | −89.0 | −93.7 |
| 12-Oxo-8,13(16)-clerodadien-15-oic acid (116) | −98.8 | −104.0 | −90.9 | −95.7 | −95.2 | −100.2 | −94.8 | −99.8 | −89.6 | −94.3 |
| 13-Epimanoyl oxide (117) | −79.6 | −86.4 | −68.1 | −74.0 | −73.9 | −80.2 | −59.1 | −64.1 | −55.8 | −60.6 |
| 13-Episclareol (118) | −91.9 | −97.8 | −80.1 | −85.3 | −90.2 | −96.0 | −84.7 | −90.1 | −39.4 | −41.9 |
| 3,4-Seco-4(18)-trachyloben-3-oic acid (120) | −88.2 | −94.5 | −88.4 | −94.6 | −80.7 | −86.4 | −86.1 | −92.3 | −80.0 | −85.7 |
| 3-Hydroxytotarol (119) | −78.8 | −84.4 | −79.9 | −85.5 | −85.9 | −92.0 | −64.1 | −68.7 | −81.2 | −87.0 |
| 7,13-Labdadien-15-ol acetate (121) | −100.2 | −104.0 | −99.5 | −103.3 | −105.7 | −109.7 | −95.8 | −99.4 | −101.8 | −105.7 |
| 7,13-Labdadien-15-ol malonate (122) | −117.9 | −117.4 | −109.5 | −109.1 | −120.5 | −120.0 | −113.1 | −112.6 | −96.1 | −95.6 |
| Acetylcrinipellin A (125) | −103.7 | −103.6 | −92.8 | −92.7 | −107.7 | −107.7 | −78.2 | −78.2 | −64.8 | −64.8 |
| Aethiopinone (123) | −94.6 | −102.0 | −87.8 | −94.7 | −108.2 | −116.7 | −93.2 | −100.6 | −109.1 | −117.6 |
| Andrographolide (124) | −103.2 | −105.3 | −95.2 | −97.0 | −104.8 | −106.9 | −97.1 | −99.0 | −99.2 | −101.2 |
| Biflorin (126) | −94.4 | −102.0 | −93.1 | −100.7 | −107.5 | −116.2 | −90.6 | −97.9 | −107.8 | −116.5 |
| Continentalic acid (127) | −78.1 | −83.6 | −75.9 | −81.3 | −85.1 | −91.1 | −61.4 | −65.8 | −84.7 | −90.7 |
| Crinipellin A (129) | −108.9 | −113.3 | −84.7 | −88.0 | −98.4 | −102.4 | −71.3 | −74.1 | −73.5 | −76.4 |
| Cryptobeilic acid A (128) | −103.4 | −109.1 | −98.0 | −103.4 | −103.1 | −108.8 | −97.9 | −103.3 | −118.1 | −124.6 |
| Cryptobeilic acid C (130) | −107.1 | −107.8 | −114.2 | −114.9 | −117.9 | −118.7 | −102.6 | −103.3 | −120.3 | −121.0 |
| Cryptobeilic acid D (131) | −98.3 | −105.5 | −99.7 | −107.0 | −103.1 | −110.7 | −102.5 | −110.0 | −107.0 | −114.8 |
| Effusanin A (132) | −78.1 | −79.8 | −73.1 | −74.7 | −81.6 | −83.4 | −70.9 | −72.5 | −43.9 | −44.9 |
| Effusanin B (133) | −75.8 | −74.5 | −78.2 | −76.9 | −92.7 | −91.2 | −89.9 | −88.4 | −65.7 | −64.6 |
| Effusanin C (134) | −95.9 | −93.1 | −82.0 | −79.6 | −96.1 | −93.2 | −44.8 | −43.5 | −61.5 | −59.7 |
| Effusanin D (135) | −88.4 | −83.0 | −96.4 | −90.5 | −109.0 | −102.3 | −72.8 | −68.4 | −71.0 | −66.7 |
| Effusanin E (136) | −81.2 | −81.7 | −72.7 | −73.2 | −83.7 | −84.3 | −55.0 | −55.4 | −42.0 | −42.2 |
| Grandiflorenic acid (137) | −76.5 | −82.1 | −77.4 | −83.1 | −77.2 | −82.8 | −73.2 | −78.6 | −49.8 | −53.5 |
| Haplociliatic acid (138) | −99.7 | −103.1 | −95.2 | −98.5 | −105.8 | −109.3 | −88.8 | −91.8 | −85.9 | −88.8 |
| Hypargenin A (139) | −82.5 | −85.8 | −83.6 | −87.0 | −94.0 | −97.7 | −77.1 | −80.2 | −74.1 | −77.1 |
| Hypargenin B (140) | −84.1 | −88.7 | −81.6 | −86.1 | −88.3 | −93.2 | −75.2 | −79.3 | −84.8 | −89.4 |
| Hypargenin D (141) | −79.9 | −86.0 | −82.1 | −88.3 | −88.6 | −95.3 | −76.0 | −81.7 | −88.5 | −95.2 |
| Hypargenin F (142) | −88.3 | −91.8 | −86.1 | −89.5 | −87.4 | −90.9 | −70.0 | −72.8 | −77.2 | −80.3 |
| Isodomedin (143) | −78.2 | −76.8 | −84.8 | −83.3 | −76.2 | −74.8 | −75.0 | −73.7 | −89.1 | −87.5 |
| Kamebanin (144) | −91.3 | −94.5 | −74.0 | −76.7 | −79.4 | −82.2 | −45.6 | −47.2 | −51.5 | −53.3 |
| Lasiokaurin (145) | −81.6 | −79.1 | −84.7 | −82.1 | −93.6 | −90.7 | −86.7 | −84.0 | −52.1 | −50.5 |
| Longikaurin A (146) | −83.1 | −84.9 | −71.8 | −73.4 | −78.1 | −79.8 | −66.7 | −68.2 | −62.9 | −64.2 |
| Longikaurin B (147) | −90.7 | −88.1 | −89.9 | −87.3 | −94.5 | −91.7 | −81.4 | −79.0 | −46.1 | −44.7 |
| Longikaurin C (148) | −80.8 | −79.5 | −87.8 | −86.4 | −96.6 | −95.0 | −51.6 | −50.8 | −72.9 | −71.7 |
| Longikaurin D (149) | −85.3 | −82.8 | −88.0 | −85.4 | −100.5 | −97.6 | −80.9 | −78.6 | −55.0 | −53.4 |
| Longikaurin E (150) | −83.4 | −85.2 | −72.7 | −74.3 | −81.0 | −82.8 | −64.5 | −65.9 | −33.4 | −34.1 |
| Longikaurin F (151) | −95.6 | −89.8 | −92.7 | −87.1 | −103.8 | −97.5 | −91.1 | −85.6 | −82.0 | −77.1 |
| Longikaurin G (152) | −86.7 | −87.3 | −75.6 | −76.1 | −81.9 | −82.5 | −71.8 | −72.3 | −62.5 | −62.9 |
| Lupulin E (153) | −103.9 | −92.7 | −111.5 | −99.5 | −124.8 | −111.4 | −89.3 | −79.7 | −91.8 | −82.0 |
| Lupulin F (154) | −44.3 | −39.5 | −108.0 | −96.3 | −124.3 | −110.8 | −77.5 | −69.1 | −91.1 | −81.2 |
| Methyl seconidoresedate (155) | −110.4 | −115.0 | −103.5 | −107.8 | −110.7 | −115.3 | −92.0 | −95.8 | −88.2 | −91.9 |
| Pisiferol (156) | −83.1 | −89.0 | −80.9 | −86.6 | −85.7 | −91.7 | −81.2 | −87.0 | −82.2 | −88.1 |
| Salvic acid (157) | −97.4 | −102.1 | −88.6 | −92.9 | −97.3 | −102.0 | −95.9 | −100.6 | −86.0 | −90.2 |
| Salvic acid acetate (158) | −96.2 | −96.9 | −94.6 | −95.2 | −109.2 | −109.9 | −99.3 | −100.0 | −96.0 | −96.7 |
| Shikokianin (159) | −59.4 | −55.8 | −86.5 | −81.2 | −101.4 | −95.2 | −83.1 | −78.0 | −57.1 | −53.6 |
| Strictic acid (160) | −104.7 | −110.7 | −98.2 | −103.9 | −108.4 | −114.6 | −89.8 | −94.9 | −88.5 | −93.5 |
| Taxodione (161) | −85.4 | −90.3 | −81.4 | −86.0 | −98.6 | −104.3 | −80.0 | −84.6 | −91.9 | −97.2 |
| Trichodonin (162) | −89.3 | −86.8 | −92.5 | −89.9 | −94.2 | −91.6 | −72.4 | −70.4 | −60.4 | −58.7 |
| Umbrosin A (163) | −85.9 | −88.9 | −76.1 | −78.8 | −81.7 | −84.6 | −55.3 | −57.3 | −61.1 | −63.2 |
| Umbrosin B (164) | −89.7 | −93.1 | −75.2 | −78.0 | −81.6 | −84.7 | −42.6 | −44.3 | −44.2 | −45.8 |
| Yuexiandajisu A (165) | −75.2 | −79.2 | −86.1 | −90.6 | −96.9 | −102.1 | −88.0 | −92.6 | −77.6 | −81.7 |
| Triterpenoids | ||||||||||
| Alisol A 24-acetate (166) | −95.7 | −84.9 | −109.9 | −97.5 | −123.5 | −109.5 | −91.2 | −80.9 | −111.1 | −98.5 |
| Alisol B 23-acetate (167) | −79.4 | −71.2 | −112.2 | −100.7 | −114.5 | −102.7 | −99.8 | −89.5 | −107.9 | −96.8 |
| Betulinic acid (168) | −65.7 | −61.3 | −94.9 | −88.6 | −97.4 | −90.9 | no dock | no dock | −41.3 | −38.6 |
| Entagenic acid (169) | −24.8 | −22.6 | −96.6 | −88.2 | −82.1 | −74.9 | −61.4 | −56.1 | −38.4 | −35.1 |
| Lantic acid (170) | −31.6 | −29.2 | −83.6 | −77.3 | −92.6 | −85.6 | −57.1 | −52.8 | −35.8 | −33.1 |
| Mahmoodin (171) | −51.0 | −45.4 | −116.5 | −103.7 | −93.6 | −83.4 | −79.9 | −71.2 | −76.3 | −67.9 |
| Maslinic acid (172) | −54.7 | −50.5 | −94.9 | −87.6 | −83.1 | −76.7 | −64.1 | −59.2 | −67.9 | −62.7 |
| Oleanolic acid (173) | −29.6 | −27.7 | −92.0 | −85.9 | −75.2 | −70.2 | −34.9 | −32.6 | −59.6 | −55.7 |
| Pristimerin (174) | −72.9 | −67.7 | −104.7 | −97.2 | −79.8 | −74.1 | −36.1 | −33.5 | −78.1 | −72.5 |
| Rubrinol (175) | −36.0 | −34.0 | −86.7 | −81.8 | −94.3 | −89.0 | −23.0 | −21.7 | −37.9 | −35.7 |
| Tingenone (176) | −50.0 | −48.0 | −89.1 | −85.5 | −84.2 | −80.8 | −21.4 | −20.6 | −78.8 | −75.7 |
| Chalcones | ||||||||||
| 1-(2,6-Dihydroxy-4-methoxyphenyl)-3-phenyl-1-propanone (177) | −89.0 | −98.7 | −85.2 | −94.5 | −95.8 | −106.3 | −96.7 | −107.3 | −111.7 | −123.9 |
| 2′-Hydroxy-2,3,4′,6′-tetramethoxychalcone (178) | −111.5 | −114.4 | −107.3 | −110.0 | −123.4 | −126.6 | −92.2 | −94.6 | −128.4 | −131.7 |
| 3′′′′,5′′′,5′′′′′-Tribenzyl-2′′′′,2′′′′′,2′′′′′′-trihydroxyisodiuvaretin (180) | −122.3 | −94.6 | −158.0 | −122.3 | −70.8 | −54.7 | −124.0 | −95.9 | −129.4 | −100.1 |
| 4'-Hydroxychalcone (179) | −80.9 | −95.7 | −87.4 | −103.4 | −88.6 | −104.8 | −90.7 | −107.3 | −99.2 | −117.5 |
| 5″,5′′′′,5′′′′′-Tribenzyl-2′′′′,2′′′′′,2′′′′′′-trihydroxyisodiuvaretin (181) | −166.5 | −128.8 | −163.7 | −126.6 | −150.9 | −116.8 | −78.4 | −60.6 | −193.6 | −149.8 |
| Angusticornin B (182) | −133.8 | −128.0 | −147.1 | −140.7 | −149.2 | −142.7 | −127.8 | −122.3 | −150.4 | −143.8 |
| Balsacone A (183) | −126.9 | −121.8 | −138.3 | −132.7 | −140.6 | −134.9 | −123.4 | −118.5 | −157.2 | −150.9 |
| Balsacone B (184) | −128.3 | −123.1 | −144.8 | −138.9 | −143.3 | −137.6 | −122.8 | −117.8 | −155.1 | −148.9 |
| Balsacone C (185) | −126.1 | −124.1 | −132.9 | −130.7 | −134.9 | −132.7 | −114.8 | −113.0 | −143.7 | −141.4 |
| Bartericin C (186) | −111.4 | −107.9 | −113.1 | −109.6 | −119.2 | −115.5 | −106.9 | −103.6 | −114.2 | −110.7 |
| Bavachalcone (187) | −117.0 | −120.7 | −115.7 | −119.4 | −122.7 | −126.6 | −120.0 | −123.8 | −135.9 | −140.2 |
| Broussochalcone B (188) | −111.7 | −116.9 | −119.9 | −125.5 | −120.7 | −126.3 | −122.2 | −127.9 | −128.5 | −134.4 |
| Corylifol B (189) | −114.0 | −117.4 | −115.5 | −118.9 | −125.8 | −129.6 | −124.0 | −127.7 | −130.9 | −134.8 |
| Erythbidin C (190) | −108.0 | −114.5 | −104.4 | −110.7 | −111.8 | −118.5 | −109.7 | −116.2 | −119.5 | −126.6 |
| Helichrysone A (191) | −119.4 | −121.3 | −113.1 | −114.9 | −114.7 | −116.5 | −119.3 | −121.2 | −130.6 | −132.7 |
| Isobavachalcone (192) | −113.7 | −119.0 | −111.7 | −116.9 | −118.6 | −124.1 | −121.3 | −126.9 | −124.8 | −130.6 |
| Kanzonol C (193) | −132.3 | −129.9 | −136.2 | −133.8 | −140.2 | −137.7 | −135.3 | −132.8 | −144.8 | −142.2 |
| Kuraridin (194) | −132.0 | −125.0 | −141.7 | −134.1 | −148.8 | −140.8 | −129.7 | −122.7 | −150.7 | −142.6 |
| Myrigalone G (195) | −91.9 | −100.2 | −91.9 | −100.3 | −100.4 | −109.5 | −90.9 | −99.2 | −109.9 | −119.9 |
| Piperaduncin A (196) | −140.3 | −127.9 | −130.4 | −118.8 | −146.9 | −133.9 | −78.6 | −71.7 | −137.3 | −125.2 |
| Piperaduncin B (197) | −136.1 | −122.7 | −136.6 | −123.2 | −137.8 | −124.3 | −113.4 | −102.3 | −138.1 | −124.5 |
| Piperaduncin C (198) | −128.9 | −112.6 | −150.2 | −131.3 | −157.3 | −137.5 | −106.9 | −93.4 | −125.1 | −109.3 |
| Psorachalcone A (199) | −112.7 | −116.1 | −117.2 | −120.7 | −121.4 | −125.0 | −109.4 | −112.7 | −125.7 | −129.4 |
| Xanthoangelol (200) | −132.8 | −130.4 | −130.8 | −128.4 | −137.8 | −135.3 | −135.9 | −133.4 | −153.0 | −150.3 |
| Xanthoangelol F (201) | −132.6 | −128.7 | −122.4 | −118.8 | −143.9 | −139.7 | −114.5 | −111.2 | −142.7 | −138.5 |
| Flavonoids | ||||||||||
| 2′,5,5′,7-Tetrahydroxyflavanone (202) | −89.2 | −97.1 | −94.0 | −102.3 | −99.1 | −107.9 | −86.6 | −94.3 | −101.8 | −110.8 |
| 2′,7-Dimethoxyflavone (203) | −86.1 | −94.4 | −94.0 | −103.0 | −95.3 | −104.4 | −94.8 | −103.9 | −100.0 | −109.7 |
| 3′′′′-(2-Hydroxybenzyl)isouvarinol (218) | −151.2 | −123.6 | −179.4 | −146.7 | −143.0 | −116.9 | −130.3 | −106.5 | −143.3 | −117.2 |
| 3′′′′-(2-Hydroxybenzyl)uvarinol (217) | −152.4 | −124.6 | −157.7 | −128.9 | −148.8 | −121.6 | −135.0 | −110.4 | −159.7 | −130.6 |
| 3′-Methylpelargonidin (204) | −90.7 | −99.1 | −99.7 | −108.9 | −98.5 | −107.6 | −88.3 | −96.5 | −103.4 | −113.0 |
| 3′-O-Methyldiplacone (205) | −136.8 | −129.5 | −131.4 | −124.3 | −139.0 | −131.5 | −122.3 | −115.8 | −140.4 | −132.9 |
| 4′,5,7-Trihydroxy-6-methyl-8-prenylflavanone (207) | −102.0 | −103.6 | −97.2 | −98.7 | −109.0 | −110.7 | −100.4 | −102.0 | −121.6 | −123.5 |
| 4′,5,7-Trihydroxy-8-methyl-6-prenylflavanone (206) | −109.4 | −111.1 | −98.9 | −100.5 | −113.4 | −115.2 | −95.2 | −96.7 | −119.2 | −121.1 |
| 4′,5-Dihydroxy-7-methoxy-6-prenylflavanone (208) | −111.2 | −113.0 | −104.8 | −106.5 | −112.4 | −114.2 | −101.1 | −102.7 | −118.5 | −120.4 |
| 4′,6,7-Trihydroxy-3′,5′-dimethoxyflavone (209) | −103.7 | −107.8 | −108.4 | −112.7 | −108.5 | −112.9 | −100.8 | −104.8 | −110.7 | −115.1 |
| 4′,7-Dihydroxy-8-methylflavan (210) | −81.4 | −92.2 | −84.4 | −95.5 | −89.1 | −100.9 | −83.8 | −94.8 | −99.0 | −112.0 |
| 4′-Hydroxy-5,7-dimethoxyflavone (211) | −93.2 | −100.3 | −101.0 | −108.7 | −100.1 | −107.7 | −95.9 | −103.2 | −104.9 | −112.9 |
| 5″-(2-Hydroxybenzyl)isouvarinol (216) | −127.0 | −103.8 | −161.5 | −132.0 | −174.9 | −142.9 | −109.8 | −89.7 | −164.2 | −134.2 |
| 5′-(1,1-Dimethyl-2-propenyl)-2′,4′,5,7-tetrahydroxy-6-prenylflavanone (219) | −126.8 | −121.3 | −117.1 | −112.1 | −115.9 | −110.8 | −115.3 | −110.3 | −136.9 | −131.0 |
| 5′-(1,1-Dimethyl-2-propenyl)-2′,4′,5,7-tetrahydroxy-8-prenylflavanone (220) | −116.8 | −111.7 | −114.4 | −109.4 | −122.1 | −116.8 | −96.8 | −92.6 | −120.9 | −115.7 |
| 5′-(1,1-Dimethyl-2-propenyl)-4′,5,7-trihydroxy-2′-methoxy-8-prenylflavanone (221) | −125.1 | −118.4 | −109.1 | −103.2 | −122.9 | −116.3 | −101.4 | −96.0 | −115.4 | −109.2 |
| 5,6-Dihydroxy-4′,7,8-trimethoxyflavone (212) | −97.6 | −100.1 | −95.6 | −98.1 | −111.0 | −113.9 | −98.9 | −101.4 | −112.6 | −115.5 |
| 5-Hydroxy-2′,4′,5′,7-Tetramethoxyflavone (213) | −108.2 | −109.5 | −111.3 | −112.7 | −112.5 | −113.9 | −103.4 | −104.6 | −109.8 | −111.1 |
| 6,7-Dihydroxyflavone (214) | −80.2 | −91.0 | −91.6 | −103.9 | −94.2 | −106.9 | −86.1 | −97.7 | −98.0 | −111.2 |
| 8-Methoxycirsilineol (215) | −107.5 | −107.2 | −98.5 | −98.3 | −115.2 | −114.9 | −98.1 | −97.9 | −111.5 | −111.2 |
| 9,10-Dihydro-9,10-diacetoxy-3-methoxy-8,8-dimethyl-2-phenyl-4H,8H-benzo[1,2-b:3,4-b′]dipyran-4-one (222) | −84.0 | −78.7 | −102.4 | −95.9 | −118.3 | −110.8 | −93.6 | −87.6 | −100.3 | −93.9 |
| Abyssinone I (223) | −101.6 | −106.5 | −106.0 | −111.1 | −105.4 | −110.5 | −105.2 | −110.4 | −101.4 | −106.3 |
| Abyssinone IV (224) | −116.2 | −114.1 | −128.5 | −126.2 | −128.3 | −126.0 | −117.6 | −115.5 | −135.9 | −133.5 |
| Astragalin (225) | −118.8 | −111.6 | −114.5 | −107.6 | −143.0 | −134.3 | −118.3 | −111.2 | −136.1 | −127.8 |
| Bavachinin (226) | −106.7 | −110.1 | −104.6 | −107.9 | −112.1 | −115.7 | −94.8 | −97.8 | −117.6 | −121.3 |
| Betuletol (227) | −94.4 | −98.2 | −106.6 | −110.8 | −111.8 | −116.3 | −97.8 | −101.7 | −118.6 | −123.4 |
| Bonannione A (228) | −119.7 | −116.0 | −125.3 | −121.4 | −130.6 | −126.5 | −123.2 | −119.4 | −132.1 | −128.0 |
| Brosimone I (229) | −104.1 | −99.9 | −116.1 | −111.4 | −117.4 | −112.7 | −97.0 | −93.1 | −146.3 | −140.4 |
| Cassiaflavan (230) | −78.8 | −90.9 | −83.3 | −96.1 | −84.0 | −96.9 | −80.7 | −93.0 | −90.9 | −104.8 |
| Cerasinone (231) | −97.3 | −101.2 | −103.0 | −107.1 | −112.0 | −116.5 | −98.2 | −102.1 | −116.2 | −120.9 |
| Chrysin (233) | −82.6 | −93.8 | −91.7 | −104.1 | −94.7 | −107.4 | −86.3 | −97.9 | −95.1 | −107.9 |
| Chrysoeriol (232) | −95.7 | −102.8 | −100.3 | −107.7 | −109.1 | −117.2 | −97.8 | −105.0 | −106.7 | −114.6 |
| Corniculatusin (234) | −103.0 | −106.9 | −98.9 | −102.7 | −114.6 | −119.0 | −96.3 | −100.0 | −113.5 | −117.8 |
| Cudraflavone A (235) | −103.0 | −99.0 | −109.7 | −105.5 | −107.3 | −103.2 | −90.8 | −87.3 | −110.1 | −105.8 |
| Dihydroquercetin (236) | −99.2 | −106.1 | −98.4 | −105.2 | −103.5 | −110.7 | −92.6 | −98.9 | −103.6 | −110.8 |
| Eucalyptin (237) | −92.2 | −96.3 | −93.7 | −97.9 | −100.8 | −105.3 | −95.3 | −99.5 | −110.5 | −115.4 |
| Euchrestaflavanone A (238) | −123.0 | −119.2 | −115.6 | −112.0 | −139.0 | −134.7 | −115.0 | −111.4 | −123.5 | −119.7 |
| Flavaprenin (239) | −104.9 | −108.0 | −95.7 | −98.6 | −114.3 | −117.7 | −101.9 | −104.9 | −116.5 | −120.0 |
| Flemiflavanone D (240) | −129.1 | −123.5 | −117.3 | −112.2 | −139.5 | −133.5 | −112.4 | −107.5 | −126.6 | −121.2 |
| Glabranin (241) | −100.5 | −105.2 | −91.5 | −95.7 | −114.4 | −119.7 | −94.1 | −98.4 | −109.6 | −114.7 |
| Isoorientin (243) | −97.3 | −91.4 | −125.8 | −118.2 | −124.7 | −117.2 | −101.7 | −95.6 | −129.4 | −121.6 |
| Isoscoparin (244) | −103.1 | −95.9 | −134.5 | −125.0 | −125.6 | −116.8 | −98.8 | −91.8 | −127.7 | −118.8 |
| Kaempferol (242) | −94.3 | −102.8 | −97.6 | −106.5 | −101.8 | −111.1 | −87.0 | −95.0 | −101.9 | −111.1 |
| Kushenol A (245) | −104.3 | −101.1 | −112.1 | −108.6 | −127.3 | −123.4 | −108.5 | −105.2 | −122.3 | −118.5 |
| Kushenol S (246) | −105.3 | −108.5 | −99.3 | −102.3 | −114.3 | −117.7 | −96.0 | −98.8 | −109.2 | −112.5 |
| Kushenol U (247) | −105.2 | −100.8 | −113.5 | −108.8 | −123.2 | −118.0 | −116.2 | −111.4 | −136.7 | −130.9 |
| Kushenol V (248) | −127.2 | −124.1 | −119.1 | −116.2 | −126.1 | −123.1 | −56.4 | −55.0 | −115.9 | −113.1 |
| Kushenol W (249) | −115.2 | −113.7 | −105.6 | −104.2 | −121.5 | −119.9 | −107.3 | −105.9 | −120.3 | −118.7 |
| Leachianone A (250) | −107.2 | −101.4 | −116.6 | −110.4 | −131.1 | −124.0 | −112.3 | −106.3 | −128.0 | −121.1 |
| Leachianone G (251) | −106.5 | −108.0 | −106.8 | −108.3 | −116.6 | −118.3 | −101.6 | −103.0 | −118.4 | −120.1 |
| Licoflavanone (252) | −110.8 | −114.1 | −115.6 | −119.0 | −117.8 | −121.3 | −110.4 | −113.7 | −114.5 | −117.9 |
| Licoflavone C (253) | −107.5 | −110.9 | −97.1 | −100.2 | −117.2 | −121.0 | −104.9 | −108.2 | −118.0 | −121.7 |
| Licoflavonol (254) | −110.0 | −111.7 | −109.2 | −110.9 | −119.7 | −121.7 | −98.1 | −99.7 | −125.9 | −127.9 |
| Lonchocarpol A (255) | −126.3 | −122.4 | −116.9 | −113.3 | −127.3 | −123.4 | −107.7 | −104.4 | −134.0 | −129.9 |
| Loranthin (256) | −135.7 | −123.2 | −143.8 | −130.6 | −128.2 | −116.4 | −111.7 | −101.5 | −132.5 | −120.3 |
| Loxophlebal A (257) | −121.0 | −111.7 | −107.6 | −99.3 | −120.3 | −111.0 | −95.7 | −88.3 | −96.3 | −88.9 |
| Lucenin 2 (258) | −96.6 | −81.8 | −132.8 | −112.6 | −153.2 | −129.8 | −133.3 | −113.0 | −143.1 | −121.3 |
| Macarangaflavanone A (259) | −131.3 | −127.2 | −134.3 | −130.1 | −136.6 | −132.3 | −121.9 | −118.2 | −137.5 | −133.2 |
| Malvidin (260) | −104.1 | −108.2 | −106.4 | −110.6 | −106.6 | −110.7 | −92.8 | −96.4 | −107.8 | −112.0 |
| Myricetin (261) | −100.0 | −105.3 | −101.0 | −106.4 | −103.9 | −109.4 | −97.0 | −102.1 | −104.0 | −109.5 |
| Natsudaidain (262) | −110.5 | −106.2 | −95.6 | −91.9 | −118.7 | −114.1 | −88.0 | −84.6 | −116.6 | −112.1 |
| Nevadensin (263) | −95.0 | −97.4 | −99.8 | −102.4 | −110.0 | −112.8 | −98.7 | −101.3 | −118.4 | −121.4 |
| O-Methylpongaglabol (264) | −93.6 | −101.4 | −95.6 | −103.5 | −104.4 | −113.1 | −90.0 | −97.5 | −101.2 | −109.7 |
| Paratocarpin L (265) | −132.0 | −127.9 | −131.3 | −127.2 | −128.6 | −124.6 | −118.4 | −114.7 | −139.7 | −135.3 |
| Persicogenin (266) | −98.0 | −103.4 | −98.6 | −104.0 | −103.2 | −108.9 | −97.2 | −102.6 | −108.3 | −114.3 |
| Pilosanol A (267) | −123.9 | −109.4 | −127.5 | −112.6 | −147.3 | −130.0 | −101.4 | −89.5 | −118.5 | −104.6 |
| Pilosanol B (268) | −120.6 | −107.4 | −120.9 | −107.6 | −143.6 | −127.9 | −100.7 | −89.7 | −133.9 | −119.2 |
| Pilosanol C (269) | −116.3 | −103.6 | −121.9 | −108.6 | −141.3 | −125.8 | −106.8 | −95.1 | −143.1 | −127.4 |
| Pinocembrin (270) | −80.9 | −91.6 | −91.6 | −103.6 | −93.1 | −105.3 | −79.9 | −90.5 | −94.1 | −106.5 |
| Pongaflavone (271) | −98.7 | −102.2 | −85.2 | −88.2 | −95.9 | −99.3 | −84.8 | −87.9 | −94.0 | −97.4 |
| Quercetin (272) | −97.5 | −104.4 | −98.2 | −105.2 | −106.8 | −114.5 | −93.6 | −100.2 | −106.4 | −114.0 |
| Quercetin 3-methyl ether (273) | −102.0 | −107.6 | −95.2 | −100.4 | −108.0 | −114.0 | −98.6 | −104.0 | −107.9 | −113.8 |
| Remangiflavanone A (274) | −103.2 | −98.9 | −115.7 | −110.9 | −130.9 | −125.4 | −108.9 | −104.4 | −132.9 | −127.3 |
| Remangiflavanone B (275) | −106.4 | −101.8 | −115.6 | −110.6 | −125.8 | −120.4 | −120.7 | −115.5 | −128.9 | −123.3 |
| Sanggenon G (276) | no dock | no dock | −174.6 | −141.8 | −126.9 | −103.0 | −124.0 | −100.6 | −170.1 | −138.1 |
| Sigmoidin A (277) | −116.3 | −111.3 | −120.4 | −115.2 | −125.9 | −120.5 | −121.9 | −116.6 | −135.0 | −129.2 |
| Sigmoidin B (278) | −114.3 | −115.9 | −121.1 | −122.8 | −117.3 | −119.0 | −115.0 | −116.6 | −120.2 | −121.9 |
| Sigmoidin L (279) | −114.4 | −114.5 | −117.4 | −117.5 | −126.1 | −126.3 | −115.8 | −116.0 | −116.5 | −116.6 |
| Siraitiflavandiol (280) | −105.8 | −107.3 | −99.4 | −100.8 | −110.9 | −112.4 | −109.9 | −111.4 | −114.0 | −115.6 |
| Solophenol D (281) | −131.5 | −124.5 | −127.8 | −121.0 | −143.8 | −136.1 | −127.1 | −120.2 | −139.4 | −132.0 |
| Sophoraflavanone G (282) | −104.0 | −99.5 | −117.4 | −112.3 | −127.9 | −122.3 | −116.2 | −111.2 | −127.6 | −122.1 |
| Sternbin (283) | −92.5 | −99.1 | −97.8 | −104.8 | −98.6 | −105.6 | −91.9 | −98.5 | −99.1 | −106.1 |
| Sudachitin (284) | −108.2 | −109.3 | −98.7 | −99.7 | −116.4 | −117.6 | −102.3 | −103.4 | −116.1 | −117.3 |
| Uvarinol (285) | −131.4 | −113.6 | −135.7 | −117.3 | −164.7 | −142.5 | −124.4 | −107.6 | −148.9 | −128.7 |
| Vahliabiflavone (286) | −70.2 | −60.8 | −111.4 | −96.6 | −77.8 | −67.5 | −95.0 | −82.4 | −83.5 | −72.4 |
| Vitexin (287) | −95.2 | −90.5 | −114.4 | −108.8 | −138.3 | −131.5 | −116.5 | −110.7 | −123.8 | −117.7 |
| Wogonin (288) | −88.2 | −96.4 | −87.6 | −95.8 | −100.6 | −110.0 | −87.6 | −95.8 | −100.5 | −109.9 |
| Isoflavonoids | ||||||||||
| 2″,3″-Epoxybolusanthol B (289) | −101.0 | −100.9 | −104.3 | −104.2 | −114.6 | −114.5 | −110.1 | −110.0 | −111.1 | −111.1 |
| 3′,5,7-Trihydroxy-4′-methoxy-5′,6-diprenylisoflavanone (290) | −128.1 | −121.3 | −122.6 | −116.0 | −128.6 | −121.7 | −121.9 | −115.4 | −134.3 | −127.1 |
| 4″-Hydroxydiphysolone (292) | −118.0 | −118.0 | −107.9 | −107.9 | −121.9 | −121.8 | −93.8 | −93.7 | −121.7 | −121.6 |
| 5,7-Dihydroxy-2′-methoxy-3′,4′-methylenedioxyisoflavanone (293) | −98.4 | −102.4 | −98.4 | −102.4 | −102.7 | −106.8 | −99.0 | −103.0 | −115.0 | −119.6 |
| 6a-Hydroxyphaseollin (291) | −96.4 | −99.5 | −90.7 | −93.6 | −90.3 | −93.2 | −93.7 | −96.6 | −91.6 | −94.5 |
| Amorphaquinone (294) | −99.4 | −101.8 | −89.3 | −91.4 | −100.6 | −103.0 | −69.6 | −71.3 | −106.4 | −108.9 |
| Asphodelin A (295) | −87.0 | −96.8 | −86.4 | −96.1 | −90.3 | −100.5 | −83.0 | −92.3 | −91.9 | −102.2 |
| Bidwillon A (296) | −115.7 | −112.1 | −128.3 | −124.3 | −122.4 | −118.6 | −120.6 | −116.9 | −125.8 | −121.9 |
| Bolucarpan A (297) | −93.9 | −94.2 | −87.2 | −87.5 | −97.9 | −98.2 | −79.5 | −79.8 | −61.3 | −61.5 |
| Bolucarpan B (298) | −98.9 | −99.4 | −88.0 | −88.4 | −98.3 | −98.7 | −79.3 | −79.6 | −76.3 | −76.7 |
| Bolucarpan D (299) | −88.2 | −91.2 | −85.6 | −88.5 | −91.7 | −94.8 | −67.2 | −69.4 | −71.1 | −73.5 |
| Bolusanthol B (300) | −97.2 | −98.6 | −101.4 | −102.8 | −112.0 | −113.5 | −108.2 | −109.7 | −111.7 | −113.2 |
| Cajanol (301) | −91.9 | −97.0 | −87.6 | −92.4 | −97.6 | −103.0 | −101.5 | −107.1 | −107.1 | −113.0 |
| Chandalone (302) | −119.9 | −116.6 | −124.1 | −120.7 | −124.5 | −121.1 | −113.8 | −110.6 | −130.9 | −127.3 |
| Dalversinol A (303) | −120.3 | −115.1 | −111.4 | −106.6 | −123.2 | −117.9 | −79.6 | −76.2 | −118.2 | −113.1 |
| Derrisin (304) | −81.0 | −77.2 | −88.8 | −84.7 | −98.8 | −94.2 | −66.7 | −63.6 | −82.4 | −78.6 |
| Erybraedin A (305) | −109.4 | −107.4 | −104.4 | −102.5 | −116.5 | −114.4 | −74.9 | −73.6 | −109.3 | −107.3 |
| Erybraedin D (306) | −98.5 | −96.9 | −99.9 | −98.3 | −105.7 | −104.0 | −73.3 | −72.1 | −97.4 | −95.9 |
| Erypoegin I (307) | −108.7 | −108.8 | −95.7 | −95.8 | −110.1 | −110.2 | −95.9 | −96.0 | −103.0 | −103.2 |
| Erysubin F (308) | −123.6 | −121.6 | −135.4 | −133.2 | −126.5 | −124.4 | −125.2 | −123.2 | −128.9 | −126.8 |
| Eryvarin V (309) | −105.6 | −101.2 | −101.7 | −97.4 | −110.4 | −105.7 | −110.2 | −105.6 | −125.3 | −120.0 |
| Eryvarin W (310) | −120.3 | −118.3 | −115.2 | −113.4 | −128.6 | −126.5 | −106.3 | −104.6 | −118.6 | −116.7 |
| Eryzerin C (311) | −115.2 | −113.0 | −120.4 | −118.0 | −117.2 | −114.9 | −112.0 | −109.8 | −131.7 | −129.1 |
| Eryzerin D (312) | −106.5 | −104.6 | −116.4 | −114.3 | −113.9 | −111.8 | −98.4 | −96.7 | −120.7 | −118.5 |
| Euchretin A (313) | −42.4 | −38.3 | −127.9 | −115.7 | −144.2 | −130.4 | −117.4 | −106.2 | −141.5 | −127.9 |
| Gancaonin C (314) | −108.6 | −110.3 | −107.5 | −109.2 | −109.9 | −111.7 | −106.4 | −108.1 | −116.1 | −117.9 |
| Genistein (315) | −83.6 | −93.0 | −87.7 | −97.5 | −93.6 | −104.1 | −78.7 | −87.6 | −100.1 | −111.3 |
| Glycyrrhisoflavone (316) | −113.9 | −115.7 | −116.0 | −117.8 | −113.6 | −115.4 | −108.8 | −110.6 | −124.3 | −126.3 |
| Hispaglabridin A (317) | −103.5 | −101.6 | −115.5 | −113.4 | −116.1 | −114.0 | −108.9 | −106.9 | −116.0 | −113.9 |
| Hispaglabridin B (318) | −12.3 | −12.1 | −88.8 | −87.4 | −103.6 | −101.9 | −100.8 | −99.1 | −107.7 | −105.9 |
| Hydroxycristacarpone (319) | −101.4 | −101.6 | −95.9 | −96.0 | −109.3 | −109.4 | −89.6 | −89.7 | −103.1 | −103.2 |
| Isoneorautenol (320) | −86.4 | −90.6 | −86.8 | −91.0 | −96.5 | −101.2 | −55.5 | −58.2 | −109.7 | −115.1 |
| Lachnoisoflavone A (321) | −103.1 | −104.9 | −101.4 | −103.2 | −110.5 | −112.4 | −99.6 | −101.4 | −123.9 | −126.1 |
| Licoisoflavanone (322) | −95.8 | −97.4 | −96.5 | −98.1 | −114.9 | −116.7 | −104.0 | −105.6 | −100.2 | −101.8 |
| Licoricidin (323) | −119.0 | −113.9 | −126.1 | −120.6 | −135.0 | −129.1 | −118.8 | −113.6 | −127.4 | −121.9 |
| Lupinalbin C (324) | −96.8 | −97.1 | −105.3 | −105.6 | −104.4 | −104.7 | −89.3 | −89.5 | −119.0 | −119.3 |
| Mucronulatol (326) | −93.3 | −99.9 | −91.9 | −98.4 | −98.5 | −105.5 | −94.5 | −101.3 | −106.1 | −113.7 |
| Neomillinol (325) | −95.6 | −99.9 | −98.4 | −102.8 | −101.7 | −106.2 | −99.6 | −104.0 | −110.3 | −115.2 |
| Pendulone (327) | −94.6 | −99.8 | −88.5 | −93.4 | −98.8 | −104.2 | −85.9 | −90.7 | −101.5 | −107.1 |
| Phyllanone B (328) | −128.0 | −121.2 | −116.4 | −110.2 | −130.5 | −123.5 | −102.9 | −97.4 | −128.9 | −122.0 |
| Shinpterocarpin (329) | −92.0 | −96.4 | −95.6 | −100.2 | −98.6 | −103.4 | −80.0 | −83.9 | −100.8 | −105.7 |
| Neoflavonoids | ||||||||||
| Inophyllum A (330) | −83.4 | −81.1 | −89.4 | −86.9 | −98.2 | −95.5 | −65.5 | −63.6 | −95.6 | −93.0 |
| Inophyllum C (331) | −69.8 | −68.0 | −90.5 | −88.1 | −111.3 | −108.4 | −82.8 | −80.7 | −92.6 | −90.1 |
| Mammea A/BA (332) | −111.4 | −108.1 | −102.7 | −99.6 | −125.5 | −121.8 | −114.4 | −111.1 | −126.0 | −122.3 |
| Mammea A/BB (333) | −120.0 | −116.5 | −104.3 | −101.3 | −116.1 | −112.7 | −115.5 | −112.1 | −124.5 | −120.9 |
| Mesuol (334) | −104.8 | −102.9 | −104.6 | −102.7 | −123.7 | −121.5 | −109.3 | −107.4 | −130.8 | −128.4 |
| Pterocarpans | ||||||||||
| 1-Methoxyphaseollidin (356) | −99.1 | −100.7 | −101.6 | −103.2 | −119.3 | −121.3 | −111.5 | −113.3 | −117.9 | −119.8 |
| Aracarpene 1 (357) | −87.3 | −93.8 | −95.1 | −102.1 | −105.2 | −113.0 | −82.9 | −89.1 | −102.9 | −110.5 |
| Aracarpene 2 (358) | −88.0 | −94.5 | −96.0 | −103.1 | −106.6 | −114.4 | −88.3 | −94.8 | −103.1 | −110.7 |
| Calopocarpin (360) | −99.5 | −104.1 | −104.3 | −109.2 | −101.6 | −106.3 | −104.8 | −109.7 | −110.9 | −116.1 |
| Cristacarpin (359) | −109.1 | −110.9 | −101.9 | −103.5 | −109.6 | −111.3 | −100.9 | −102.5 | −108.3 | −110.0 |
| Erythbidin D (362) | −94.8 | −101.7 | −95.5 | −102.5 | −95.5 | −102.5 | −91.2 | −97.9 | −104.1 | −111.8 |
| Eryzerin E (363) | −107.5 | −103.0 | −121.3 | −116.2 | −125.3 | −120.0 | −118.2 | −113.3 | −120.5 | −115.4 |
| Fuscacarpan B (364) | −100.7 | −100.8 | −101.0 | −101.2 | −105.8 | −105.9 | −101.5 | −101.6 | −101.5 | −101.6 |
| Fuscacarpan C (365) | −98.5 | −98.6 | −102.0 | −102.1 | −109.7 | −109.9 | −97.3 | −97.4 | −102.5 | −102.7 |
| Glycyrol (366) | −108.1 | −108.6 | −110.1 | −110.6 | −119.8 | −120.4 | −94.6 | −95.1 | −129.1 | −129.8 |
| Glycyrrhizol A (367) | −125.4 | −120.4 | −127.8 | −122.6 | −127.4 | −122.3 | −89.6 | −85.9 | −123.9 | −118.9 |
| Glycyrrhizol B (368) | −96.7 | −98.6 | −99.8 | −101.8 | −104.4 | −106.4 | −82.4 | −84.1 | −93.4 | −95.3 |
| Sandwicensin (369) | −97.6 | −100.7 | −103.9 | −107.2 | −110.9 | −114.4 | −97.1 | −100.2 | −112.0 | −115.6 |
| Variabilin (370) | −89.5 | −96.1 | −91.6 | −98.4 | −92.2 | −99.0 | −96.5 | −103.6 | −103.6 | −111.2 |
| ent-Sophoracarpan A (361) | −85.4 | −91.7 | −82.4 | −88.5 | −89.5 | −96.1 | −78.0 | −83.7 | −101.3 | −108.7 |
| Chromones | ||||||||||
| 3-(3-Hydroxy-4-methoxybenzylidene)-6,7-dimethoxy-4-chromanone (371) | −96.1 | −98.8 | −100.2 | −102.9 | −113.2 | −116.4 | −94.0 | −96.6 | −106.8 | −109.8 |
| 4′,5,7-Trihydroxy-6,8-dimethylhomoisoflavanone (372) | −91.4 | −96.6 | −94.3 | −99.7 | −102.7 | −108.6 | −74.9 | −79.2 | −117.4 | −124.1 |
| 4′,5,7-Trihydroxy-6-methylhomoisoflavanone (373) | −87.9 | −94.4 | −98.8 | −106.1 | −101.1 | −108.6 | −83.4 | −89.5 | −114.3 | −122.7 |
| 7-O-Methylbonducellin (374) | −89.2 | −96.2 | −92.6 | −99.8 | −99.0 | −106.8 | −90.1 | −97.2 | −99.6 | −107.4 |
| 8-Methylophiopogonanone B (375) | −95.4 | −99.5 | −95.4 | −99.5 | −103.5 | −107.8 | −89.3 | −93.1 | −123.3 | −128.5 |
| Bonducellin (376) | −86.9 | −95.3 | −92.9 | −101.8 | −101.9 | −111.7 | −85.9 | −94.1 | −98.8 | −108.3 |
| Odoratumone A (377) | −98.7 | −101.3 | −104.2 | −106.8 | −105.6 | −108.3 | −92.4 | −94.7 | −124.9 | −128.1 |
| Sappanone A 3′,4′-methylene ether (378) | −94.0 | −101.4 | −97.7 | −105.3 | −107.7 | −116.1 | −90.7 | −97.9 | −104.7 | −112.9 |
| Sappanone A 4′-methyl ether (379) | −90.9 | −97.9 | −95.3 | −102.6 | −104.9 | −112.9 | −88.9 | −95.7 | −96.4 | −103.7 |
| Sappanone A trimethyl ether (380) | −92.4 | −96.5 | −101.6 | −106.1 | −110.3 | −115.2 | −78.7 | −82.2 | −102.3 | −106.8 |
| Condensed Tannins | ||||||||||
| GB 1 (381) | −76.3 | −66.6 | −123.2 | −107.6 | −107.4 | −93.8 | −92.9 | −81.1 | −91.9 | −80.2 |
| Proanthocyanidin A1 (382) | −40.8 | −35.2 | −110.1 | −95.1 | −92.8 | −80.2 | −97.6 | −84.3 | −121.2 | −104.7 |
| Proanthocyanidin A2 (383) | −12.3 | −10.6 | −105.7 | −91.3 | −122.7 | −106.0 | −89.7 | −77.5 | −123.7 | −106.9 |
| Procyanidin B4 (384) | −111.1 | −95.9 | −145.7 | −125.8 | −120.6 | −104.0 | −120.5 | −104.0 | −99.6 | −86.0 |
| Procyanidin B5 (385) | −77.8 | −67.1 | −142.5 | −123.0 | −153.0 | −132.1 | −74.1 | −63.9 | −128.4 | −110.8 |
| Procyanidin B6 (386) | −41.1 | −35.5 | −148.1 | −127.8 | −131.9 | −113.8 | −89.6 | −77.4 | −161.6 | −139.5 |
| Teatannin (387) | −119.7 | −112.9 | −113.6 | −107.2 | −133.0 | −125.5 | −115.2 | −108.7 | −145.4 | −137.2 |
| Coumarins | ||||||||||
| 4,5′,8′-Trihydroxy-5-methyl-3,7′-bicoumarin (344) | −106.7 | −108.6 | −103.0 | −104.9 | −107.6 | −109.6 | −94.2 | −95.9 | −105.2 | −107.0 |
| 6-Geranyl-5,7-dihydroxy-8(2-methylbutanoyl)-4-phenylcoumarin (345) | −139.0 | −128.2 | −124.6 | −114.8 | −141.0 | −130.0 | −125.9 | −116.0 | −149.2 | −137.6 |
| (–)-Heliettin (354) | −71.0 | −90.7 | −73.4 | −93.8 | −74.5 | −95.1 | −67.3 | −86.0 | −75.4 | −96.3 |
| Aesculin (347) | −104.4 | −107.6 | −99.4 | −102.4 | −111.8 | −115.1 | −99.0 | −102.0 | −121.1 | −124.7 |
| Alloimperatorin (346) | −97.9 | −108.9 | −92.2 | −102.6 | −102.0 | −113.4 | −84.0 | −93.5 | −101.6 | −113.0 |
| Anofinic acid (348) | −71.4 | −87.2 | −71.4 | −87.1 | −80.1 | −97.7 | −82.0 | −100.1 | −79.9 | −97.6 |
| Calaustralin (349) | −115.4 | −112.2 | −104.9 | −101.9 | −121.1 | −117.8 | −101.9 | −99.1 | −109.5 | −106.5 |
| Calophyllolide (350) | −102.1 | −98.3 | −102.8 | −98.9 | −111.2 | −107.1 | −102.5 | −98.7 | −108.8 | −104.8 |
| Dicoumarol (351) | −95.1 | −98.3 | −93.1 | −96.3 | −102.9 | −106.4 | −95.2 | −98.4 | −118.7 | −122.8 |
| Dipetalolactone (352) | −84.1 | −89.3 | −80.4 | −85.4 | −105.9 | −112.4 | −86.3 | −91.7 | −104.6 | −111.1 |
| Glycycoumarin (353) | −107.2 | −107.5 | −112.6 | −112.9 | −118.7 | −119.1 | −106.0 | −106.4 | −122.7 | −123.1 |
| Marmesin (355) | −83.0 | −95.2 | −83.0 | −95.2 | −86.7 | −99.4 | −82.4 | −94.6 | −93.6 | −107.4 |
| Stilbenoids | ||||||||||
| 2-(2,4-Dihydroxyphenyl-5-(1-propenyl)benzofurans (388) | −94.3 | −105.3 | −95.4 | −106.6 | −102.1 | −114.1 | −95.9 | −107.2 | −104.4 | −116.7 |
| Albanol A (389) | no dock | no dock | −142.0 | −123.7 | no dock | no dock | −100.1 | −87.2 | −136.8 | −119.2 |
| Albanol B (390) | no dock | no dock | −106.6 | −93.0 | −75.9 | −66.3 | −110.2 | −96.2 | −135.2 | −118.0 |
| Amorfrutin A (391) | −114.0 | −117.4 | −105.8 | −109.0 | −113.2 | −116.6 | −97.0 | −99.9 | −126.9 | −130.7 |
| Cajaninstilbene acid (392) | −117.7 | −121.4 | −107.9 | −111.3 | −115.8 | −119.4 | −53.3 | −55.0 | −113.5 | −117.1 |
| Calodenin B (393) | −159.1 | −141.9 | −155.9 | −139.0 | −158.9 | −141.6 | −143.8 | −128.2 | −165.3 | −147.4 |
| Centrolobofuran (394) | −87.2 | −98.7 | −92.9 | −105.1 | −94.2 | −106.6 | −95.3 | −107.9 | −106.0 | −120.0 |
| Cochinchinenene A (395) | −112.8 | −99.6 | −143.0 | −126.2 | −123.9 | −109.3 | −112.9 | −99.7 | −137.4 | −121.3 |
| Cochinchinenene B (396) | −122.7 | −110.2 | −142.7 | −128.2 | −131.4 | −118.1 | −127.9 | −114.9 | −136.1 | −122.3 |
| Cochinchinenene C (397) | −110.4 | −100.1 | −137.3 | −124.5 | −137.0 | −124.2 | −104.5 | −94.8 | −127.7 | −115.8 |
| Cochinchinenene D (398) | −119.6 | −109.5 | −140.6 | −128.7 | −135.7 | −124.2 | −122.6 | −112.2 | −127.6 | −116.8 |
| Egonol (399) | −114.9 | −120.0 | −116.0 | −121.2 | −116.8 | −122.0 | −107.2 | −112.0 | −120.4 | −125.8 |
| Erypoegin F (400) | −117.7 | −119.8 | −115.7 | −117.8 | −126.7 | −128.9 | −115.8 | −117.9 | −118.2 | −120.3 |
| Erythbidin E (401) | −89.9 | −101.8 | −95.4 | −108.0 | −98.2 | −111.2 | −92.7 | −104.9 | −103.3 | −117.0 |
| Eryvarin Q (402) | −137.9 | −133.9 | −122.7 | −119.1 | −135.8 | −131.8 | −136.1 | −132.1 | −140.3 | −136.2 |
| Gancaonin I (403) | −112.2 | −114.0 | −112.2 | −114.0 | −117.2 | −119.0 | −111.0 | −112.7 | −118.3 | −120.2 |
| Glyinflanin H (404) | −98.9 | −105.3 | −97.7 | −104.0 | −101.5 | −108.1 | −103.0 | −109.6 | −110.3 | −117.4 |
| Kuwanol A (405) | no dock | no dock | −149.0 | −129.6 | no dock | no dock | −48.6 | −42.3 | −142.3 | −123.8 |
| Licobenzofuran (406) | −115.9 | −117.8 | −128.7 | −130.8 | −119.7 | −121.6 | −119.7 | −121.6 | −123.9 | −125.9 |
| Licocoumarone (407) | −110.3 | −113.6 | −110.1 | −113.4 | −117.1 | −120.6 | −104.5 | −107.6 | −116.9 | −120.4 |
| Mulberrofuran D (408) | −135.0 | −127.0 | −142.6 | −134.1 | −153.3 | −144.2 | −126.6 | −119.1 | −148.9 | −140.1 |
| Mulberrofuran Y (409) | −121.9 | −118.1 | −129.1 | −125.1 | −143.6 | −139.1 | −130.2 | −126.2 | −134.1 | −129.9 |
| Pinosylvin (410) | −76.0 | −91.6 | −81.6 | −98.3 | −84.8 | −102.2 | −87.5 | −105.5 | −86.5 | −104.2 |
| Schweinfurthin A (411) | −84.9 | −74.6 | −139.0 | −122.1 | −123.4 | −108.3 | −133.1 | −116.9 | −134.6 | −118.2 |
| Shanciguol 3-methyl ether (412) | −123.9 | −116.9 | −130.4 | −123.0 | −133.7 | −126.1 | −121.4 | −114.5 | −129.6 | −122.3 |
| Stemofuran R (413) | −104.6 | −109.0 | −105.1 | −109.5 | −104.8 | −109.2 | −110.5 | −115.2 | −109.0 | −113.6 |
| Stilbostemin S (414) | −98.5 | −107.2 | −100.0 | −108.9 | −105.7 | −115.1 | −89.9 | −97.8 | −107.4 | −116.9 |
| Thunberginol F (415) | −90.2 | −100.4 | −94.5 | −105.1 | −100.3 | −111.6 | −98.7 | −109.7 | −111.0 | −123.4 |
| (7E,7′R,8′R)-ε-Viniferin (416) | −104.3 | −97.5 | −134.4 | −125.7 | −89.7 | −83.9 | −111.9 | −104.7 | −121.5 | −113.6 |
| (7E,7′S,8′S)-ε-Viniferin (417) | −106.1 | −99.3 | −134.6 | −125.9 | −134.2 | −125.5 | −116.2 | −108.7 | −128.6 | −120.3 |
| Phenylpropanoids and Lignans | ||||||||||
| (E)-Cinnamaldehyde (418) | −59.6 | −84.2 | −64.5 | −91.0 | −69.6 | −98.2 | −62.4 | −88.0 | −70.9 | −100.0 |
| 3,4-Dimethylcinnamaldehyde (419) | −68.2 | −90.2 | −73.0 | −96.6 | −75.0 | −99.3 | −72.9 | −96.5 | −82.0 | −108.6 |
| Methyleugenol (422) | −66.2 | −84.6 | −77.6 | −99.2 | −70.8 | −90.4 | −74.8 | −95.5 | −85.3 | −109.0 |
| p-Coumaraldehyde (420) | −66.1 | −89.9 | −71.4 | −97.1 | −66.4 | −90.2 | −67.0 | −91.1 | −74.8 | −101.6 |
| p-Methoxycinnamaldehyde (421) | −69.2 | −91.2 | −69.1 | −91.1 | −81.1 | −106.9 | −72.8 | −96.0 | −83.5 | −110.1 |
| (−)-Asarinin (423) | −107.5 | −109.2 | −111.5 | −113.3 | −114.5 | −116.3 | −115.6 | −117.4 | −111.4 | −113.2 |
| Nordihydroguaiaretic acid (424) | −107.9 | −115.6 | −104.4 | −111.8 | −103.3 | −110.6 | −98.0 | −105.0 | −115.6 | −123.9 |
| Xanthones | ||||||||||
| 2-Deoxy-4-Hydroxycudratricusxanthone D (425) | −101.5 | −99.5 | −106.9 | −104.8 | −111.1 | −109.0 | −99.0 | −97.1 | −112.2 | −110.0 |
| Calozeyloxanthone (426) | −95.4 | −94.9 | −88.9 | −88.4 | −101.4 | −100.8 | −100.6 | −100.0 | −81.5 | −81.0 |
| Cycloartobiloxanthone (427) | −122.5 | −116.3 | −101.7 | −96.6 | −100.2 | −95.1 | −83.5 | −79.3 | −100.7 | −95.6 |
| Formoxanthone C (428) | −102.4 | −100.2 | −100.5 | −98.3 | −121.3 | −118.8 | −101.7 | −99.5 | −112.7 | −110.3 |
| Garciniacowone (429) | −108.3 | −100.4 | −135.4 | −125.5 | −139.0 | −128.9 | −109.6 | −101.6 | −146.0 | −135.3 |
| Globulixanthone C (430) | −92.0 | −96.1 | −91.1 | −95.1 | −95.4 | −99.7 | −82.4 | −86.1 | −101.4 | −105.9 |
| Globulixanthone D (431) | −104.1 | −107.0 | −106.5 | −109.4 | −116.1 | −119.3 | −91.0 | −93.5 | −114.5 | −117.7 |
| Globulixanthone E (432) | −11.6 | −9.7 | −116.0 | −97.9 | −140.1 | −118.2 | −69.0 | −58.2 | −111.3 | −93.9 |
| Morellin (433) | −104.3 | −91.9 | −127.6 | −112.3 | −118.7 | −104.5 | −78.8 | −69.4 | −93.4 | −82.3 |
| Nigrolineaxanthone N (434) | −110.2 | −107.6 | −120.6 | −117.9 | −125.2 | −122.4 | −104.8 | −102.4 | −133.5 | −130.4 |
| Pinselin (435) | −86.6 | −93.0 | −92.7 | −99.6 | −94.3 | −101.3 | −80.8 | −86.7 | −101.7 | −109.2 |
| Scortechinone B (436) | −20.9 | −17.9 | −138.5 | −118.6 | −85.9 | −73.5 | −120.0 | −102.7 | −112.6 | −96.4 |
| Symphonin (437) | −110.8 | −103.7 | −105.9 | −99.2 | −120.7 | −113.0 | −101.8 | −95.3 | −110.4 | −103.4 |
| Hydrolyzable Tannins | ||||||||||
| 1,2,3,4,6-Pentagalloylglucose (335) | −74.0 | −54.3 | −194.4 | −142.7 | −122.4 | −89.8 | −98.8 | −72.5 | −175.1 | −128.5 |
| Aceritannin (438) | −142.1 | −131.6 | −142.0 | −131.5 | −146.2 | −135.3 | −133.5 | −123.6 | −138.3 | −128.1 |
| Ginnalin B (439) | −103.7 | −109.4 | −104.5 | −110.2 | −113.9 | −120.2 | −107.1 | −113.1 | −119.6 | −126.2 |
| Ginnalin C (440) | −104.5 | −110.3 | −107.2 | −113.1 | −107.1 | −113.0 | −101.3 | −106.9 | −117.4 | −123.9 |
| Panconoside A (441) | −99.9 | −82.6 | −140.4 | −116.0 | −166.3 | −137.5 | −98.5 | −81.4 | −164.5 | −136.0 |
| Miscellaneous Phenolics | ||||||||||
| 1,3,7,9-Tetrahydroxy-4,6-dimethyl-2,8-bis(2-methyl-propanoyl)dibenzofuran (442) | −111.5 | −108.8 | −111.0 | −108.3 | −109.4 | −106.8 | −104.8 | −102.3 | −128.2 | −125.0 |
| 2′,4′-Dihydroxy-6′-methoxy-3′-methylacetophenone (443) | −69.8 | −86.3 | −70.2 | −86.8 | −72.3 | −89.4 | −67.4 | −83.4 | −77.1 | −95.4 |
| 3′,4′-Dihydroxyacetophenone (444) | −62.5 | −84.1 | −62.0 | −83.5 | −72.1 | −97.1 | −64.4 | −86.8 | −72.7 | −97.9 |
| 4′-O-Methylhonokiol (446) | −103.6 | −113.8 | −105.3 | −115.7 | −101.5 | −111.5 | −102.9 | −113.0 | −113.7 | −124.9 |
| 4-Deoxyadhumulone 2″,3″-epoxide (445) | −101.5 | −102.4 | −100.9 | −101.7 | −115.7 | −116.6 | −105.4 | −106.3 | −125.9 | −127.0 |
| 7-(3,4-Dihydroxy-5-methoxyphenyl)-1-phenyl-4-hepten-3-one (447) | −115.0 | −120.1 | −112.9 | −117.9 | −113.5 | −118.5 | −109.6 | −114.5 | −122.4 | −127.8 |
| Agrimol C (449) | −108.6 | −89.3 | −130.9 | −107.6 | −128.1 | −105.3 | −101.8 | −83.7 | −164.9 | −135.6 |
| Agrimol F (450) | −105.7 | −88.2 | −133.1 | −111.0 | −146.5 | −122.2 | −91.4 | −76.2 | −152.2 | −126.9 |
| Agrimol G (451) | −16.9 | −13.9 | −146.7 | −120.6 | −146.0 | −120.0 | −101.2 | −83.2 | −153.9 | −126.5 |
| Arzanol (452) | −110.9 | −108.0 | −122.3 | −119.1 | −125.7 | −122.4 | −112.6 | −109.7 | −133.3 | −129.9 |
| Aspidinol C (448) | −81.8 | −96.8 | −78.0 | −92.3 | −79.0 | −93.5 | −77.5 | −91.7 | −87.3 | −103.3 |
| Bruguierol C (453) | −76.4 | −93.0 | −74.1 | −90.1 | −71.2 | −86.7 | −67.5 | −82.2 | −73.2 | −89.0 |
| Cearoin (454) | −82.7 | −95.1 | −89.1 | −102.5 | −86.1 | −99.1 | −84.4 | −97.1 | −107.1 | −123.1 |
| Citrusnin A (455) | −90.1 | −105.4 | −99.2 | −116.1 | −89.1 | −104.2 | −100.0 | −117.0 | −107.2 | −125.4 |
| Cochinchinenin B (456) | −128.3 | −113.1 | −153.5 | −135.3 | −150.1 | −132.3 | −120.0 | −105.8 | −151.3 | −133.4 |
| Cochinchinenin C (457) | −133.8 | −118.0 | −155.2 | −136.8 | −167.6 | −147.8 | −85.7 | −75.5 | −151.0 | −133.1 |
| Drummondin D (458) | −134.4 | −122.0 | −117.8 | −106.9 | −129.1 | −117.2 | −99.6 | −90.4 | −107.1 | −97.3 |
| Drummondin E (459) | −138.3 | −125.4 | −128.9 | −116.9 | −136.5 | −123.8 | −96.9 | −87.8 | −128.4 | −116.5 |
| Eleutherol (460) | −79.3 | −91.2 | −89.8 | −103.3 | −89.0 | −102.4 | −74.5 | −85.7 | −91.3 | −105.0 |
| Ellagicacid (461) | −95.0 | −101.7 | −82.8 | −88.7 | −109.3 | −117.1 | −84.2 | −90.2 | −103.6 | −111.0 |
| Epicoccolide A (462) | −87.2 | −87.0 | −87.0 | −86.8 | −107.4 | −107.2 | −73.9 | −73.7 | −99.3 | −99.1 |
| Gibbilimbol A (464) | −93.0 | −108.7 | −91.6 | −107.1 | −95.0 | −111.1 | −95.1 | −111.2 | −105.8 | −123.7 |
| Gibbilimbol B (465) | −88.3 | −103.3 | −95.2 | −111.3 | −93.4 | −109.2 | −92.8 | −108.6 | −100.8 | −117.9 |
| Grifolin (466) | −112.5 | −117.2 | −115.3 | −120.2 | −118.4 | −123.4 | −100.7 | −104.9 | −128.3 | −133.7 |
| Hyperbrasilol A (467) | −87.1 | −75.6 | −137.0 | −118.9 | −121.2 | −105.1 | −79.6 | −69.1 | −88.8 | −77.0 |
| Hyperbrasilol B (468) | −129.4 | −113.4 | −127.0 | −111.3 | −139.0 | −121.8 | −97.3 | −85.3 | −115.4 | −101.1 |
| Hyperbrasilol C (469) | −149.3 | −130.6 | −129.2 | −113.1 | −147.7 | −129.3 | −77.3 | −67.6 | −143.4 | −125.5 |
| Isodrummondin D (470) | −107.9 | −98.0 | −125.7 | −114.2 | −142.8 | −129.7 | −77.9 | −70.7 | −117.5 | −106.7 |
| Isohyperbrasilol B (471) | −97.3 | −85.2 | −126.6 | −110.9 | −134.5 | −117.8 | −99.3 | −87.0 | −128.4 | −112.5 |
| Isouliginosin B (472) | −119.3 | −108.2 | −123.7 | −112.1 | −118.7 | −107.7 | −99.4 | −90.1 | −121.2 | −109.9 |
| Italipyrone (473) | −115.0 | −112.1 | −107.7 | −105.0 | −125.3 | −122.2 | −107.7 | −105.0 | −129.2 | −126.0 |
| Knerachelin A (474) | −104.9 | −112.6 | −96.8 | −104.0 | −106.5 | −114.3 | −103.3 | −111.0 | −123.9 | −133.0 |
| Knerachelin B (475) | −93.2 | −103.6 | −95.1 | −105.7 | −101.1 | −112.5 | −95.2 | −105.9 | −109.5 | −121.8 |
| Magnolol (476) | −97.3 | −108.8 | −102.5 | −114.6 | −97.0 | −108.4 | −99.1 | −110.8 | −107.3 | −119.9 |
| Myrtucommulone A (477) | −13.8 | −11.4 | −122.2 | −100.5 | −36.0 | −29.6 | −72.1 | −59.2 | no dock | no dock |
| Myrtucommulone B (478) | −68.2 | −65.8 | −96.1 | −92.6 | −106.3 | −102.5 | −77.3 | −74.6 | −85.1 | −82.1 |
| Obovatol (479) | −95.5 | −104.7 | −104.6 | −114.7 | −101.7 | −111.5 | −95.4 | −104.6 | −110.7 | −121.3 |
| Oenostacin (480) | −96.0 | −107.3 | −98.4 | −110.0 | −106.2 | −118.7 | −103.6 | −115.8 | −113.1 | −126.4 |
| Paeonol (481) | −62.1 | −81.2 | −66.5 | −86.9 | −72.6 | −95.0 | −69.1 | −90.4 | −83.7 | −109.5 |
| Perlatolic acid (482) | −137.2 | −129.3 | −127.1 | −119.8 | −145.4 | −136.9 | −119.9 | −113.0 | −145.6 | −137.1 |
| Plicatipyrone (483) | −106.6 | −102.5 | −104.3 | −100.3 | −118.2 | −113.7 | −100.8 | −96.9 | −130.9 | −125.8 |
| Propterol (484) | −89.5 | −102.9 | −91.0 | −104.6 | −91.9 | −105.7 | −89.7 | −103.2 | −101.7 | −117.0 |
| Pulverulentone B (485) | −80.0 | −92.8 | −83.0 | −96.3 | −82.9 | −96.1 | −68.2 | −79.0 | −93.5 | −108.4 |
| Quinquangulin (486) | −88.3 | −96.3 | −87.1 | −95.0 | −91.4 | −99.7 | −71.6 | −78.1 | −97.1 | −105.9 |
| Rhodomyrtone (487) | −94.4 | −89.1 | −105.1 | −99.2 | −96.8 | −91.3 | −84.8 | −80.0 | −91.4 | −86.2 |
| Rosmarinic acid (488) | −123.1 | −124.4 | −114.5 | −115.7 | −123.6 | −124.9 | −120.4 | −121.6 | −125.9 | −127.3 |
| Rubanthrone A (489) | −100.2 | −99.6 | −96.2 | −95.6 | −115.9 | −115.2 | −94.3 | −93.7 | −100.1 | −99.5 |
| Sampsone A (490) | −103.0 | −101.9 | −96.6 | −95.6 | −104.4 | −103.2 | −85.9 | −84.9 | −87.7 | −86.7 |
| Sarothralen B (491) | −131.8 | −114.5 | −137.7 | −119.7 | −145.5 | −126.4 | −115.6 | −100.5 | −129.2 | −112.2 |
| Sarothralen C (492) | −130.3 | −112.0 | −126.7 | −108.9 | −132.4 | −113.9 | −46.3 | −39.8 | −112.5 | −96.7 |
| Sarothralen D (493) | −104.1 | −89.5 | −142.6 | −122.6 | −128.7 | −110.7 | −110.6 | −95.1 | −135.2 | −116.2 |
| Shikonofuran C (494) | −118.9 | −120.3 | −120.8 | −122.3 | −120.1 | −121.5 | −116.3 | −117.7 | −128.2 | −129.7 |
| Shikonofuran D (495) | −115.6 | −118.6 | −119.1 | −122.1 | −122.3 | −125.4 | −106.0 | −108.8 | −130.0 | −133.3 |
| Shikonofuran E (496) | −123.0 | −124.7 | −122.9 | −124.6 | −125.9 | −127.6 | −104.1 | −105.6 | −132.3 | −134.1 |
| Sinapic acid (497) | −77.3 | −91.5 | −90.1 | −106.6 | −84.8 | −100.3 | −88.8 | −105.1 | −86.5 | −102.3 |
| Walrycin A (498) | −58.6 | −75.5 | −71.0 | −91.4 | −73.8 | −95.0 | −66.7 | −85.9 | −71.5 | −92.1 |
| Quinones | ||||||||||
| 2,6-Dimethoxy-1,4-benzoquinone (336) | −60.2 | −78.5 | −70.2 | −91.5 | −70.7 | −92.1 | −67.4 | −87.8 | −73.1 | −95.2 |
| 2-Methyl-6-prenyl-1,4-benzoquinone (337) | −71.8 | −89.7 | −84.2 | −105.3 | −77.4 | −96.7 | −83.6 | −104.5 | −86.9 | −108.6 |
| Omphalone (499) | −73.3 | −92.0 | −79.3 | −99.4 | −84.8 | −106.4 | −79.5 | −99.7 | −86.6 | −108.7 |
| Primin (500) | −75.0 | −91.0 | −83.3 | −101.0 | −84.7 | −102.8 | −84.5 | −102.5 | −91.1 | −110.5 |
| 1,4-Naphthoquinone (338) | −56.7 | −75.4 | −65.3 | −86.8 | −67.6 | −89.9 | −61.1 | −81.3 | −69.8 | −92.8 |
| 2-Acetylnaphtho[2,3-b]furan-4,9-dione (339) | −78.6 | −90.9 | −90.9 | −105.2 | −89.5 | −103.5 | −85.9 | −99.3 | −95.6 | −110.5 |
| Alkannin (340) | −94.7 | −103.1 | −95.5 | −104.0 | −103.8 | −113.0 | −84.1 | −91.6 | −107.0 | −116.5 |
| Isobutyrylshikonin (341) | −108.3 | −109.6 | −99.7 | −100.9 | −116.8 | −118.3 | −82.1 | −83.1 | −120.5 | −122.0 |
| Lapachol (501) | −86.1 | −99.3 | −87.0 | −100.3 | −89.4 | −103.1 | −88.6 | −102.2 | −101.5 | −117.1 |
| Mamegakinone (502) | −86.8 | −86.6 | −92.1 | −91.8 | −103.5 | −103.2 | −82.5 | −82.3 | −93.5 | −93.3 |
| Menadione (503) | −61.8 | −79.9 | −71.4 | −92.3 | −71.9 | −92.9 | −64.0 | −82.7 | −73.0 | −94.4 |
| Rhinacanthin C (504) | −131.5 | −127.2 | −124.0 | −119.9 | −130.8 | −126.5 | −109.5 | −105.9 | −131.8 | −127.5 |
| Rhinacanthin D (505) | −126.1 | −122.2 | −122.4 | −118.6 | −127.4 | −123.5 | −104.4 | −101.2 | −122.5 | −118.7 |
| Rhinacanthin G (506) | −130.7 | −124.8 | −119.6 | −114.2 | −130.9 | −125.1 | −109.1 | −104.2 | −130.4 | −124.6 |
| Rhinacanthin H (507) | −129.7 | −123.9 | −128.1 | −122.3 | −127.5 | −121.8 | −108.5 | −103.7 | −128.1 | −122.4 |
| Rhinacanthin I (508) | −130.7 | −124.8 | −123.1 | −117.6 | −133.8 | −127.8 | −118.0 | −112.7 | −138.1 | −131.9 |
| Rhinacanthin J (509) | −127.4 | −121.9 | −120.2 | −115.0 | −128.5 | −122.9 | −107.8 | −103.2 | −135.3 | −129.4 |
| Rhinacanthin K (510) | −134.8 | −127.0 | −117.4 | −110.6 | −129.1 | −121.6 | −96.9 | −91.3 | −142.2 | −134.0 |
| Rhinacanthin L (511) | −132.7 | −123.5 | −125.6 | −117.0 | −129.4 | −120.5 | −106.6 | −99.2 | −115.2 | −107.2 |
| Rhinacanthin M (512) | −118.0 | −118.8 | −104.0 | −104.7 | −113.1 | −113.8 | −100.7 | −101.4 | −128.3 | −129.1 |
| Shikonin acetate (513) | −103.6 | −107.7 | −104.0 | −108.1 | −112.4 | −116.9 | −81.5 | −84.8 | −118.5 | −123.2 |
| β,β-Dimethylacrylshikonin (514) | −121.4 | −121.6 | −113.3 | −113.4 | −125.9 | −126.1 | −91.0 | −91.1 | −123.3 | −123.4 |
| β-Hydroxyisovaleryshikonin (515) | −119.2 | −117.4 | −113.2 | −111.6 | −128.3 | −126.5 | −95.9 | −94.5 | −124.2 | −122.4 |
| 1-Hydroxy-3-hydroxymethylanthraquinone (516) | −79.2 | −89.9 | −81.6 | −92.6 | −89.9 | −102.0 | −74.5 | −84.5 | −93.7 | −106.3 |
| Aloeemodin (518) | −85.2 | −94.8 | −86.5 | −96.2 | −95.6 | −106.3 | −75.5 | −83.9 | −96.6 | −107.4 |
| Islandicin (519) | −83.7 | −93.1 | −85.9 | −95.5 | −93.2 | −103.6 | −73.1 | −81.3 | −92.9 | −103.3 |
| Newbouldiaquinone (521) | −108.2 | −106.1 | −97.6 | −95.7 | −110.9 | −108.7 | −84.1 | −82.5 | −74.5 | −73.0 |
| Newbouldiaquinone A (520) | −104.8 | −101.4 | −113.2 | −109.5 | −120.0 | −116.1 | −82.8 | −80.1 | −140.9 | −136.3 |
| Rhein (522) | −86.4 | −94.5 | −89.6 | −98.0 | −98.6 | −107.8 | −77.4 | −84.7 | −100.0 | −109.4 |
| 15,16-Dihydrotanshinone I (517) | −79.0 | −87.0 | −84.7 | −93.3 | −97.5 | −107.3 | −71.4 | −78.6 | −93.1 | −102.6 |
| Acetylene, Glucoside, and Other Miscellaneous Phytochemicals | ||||||||||
| 1,7-Diphenyl-4-(2-phenylethyl)-1-heptene-3,5-dione (530) | −126.7 | −125.5 | −113.3 | −112.3 | −129.5 | −128.2 | −117.8 | −116.7 | −139.8 | −138.5 |
| 1,7-Diphenyl-5-hepten-3-one (531) | −102.0 | −114.3 | −94.3 | −105.6 | −97.9 | −109.6 | −96.6 | −108.2 | −108.7 | −121.8 |
| 3'-Demothexycyclocurcumin (532) | −109.1 | −112.6 | −109.9 | −113.4 | −117.9 | −121.7 | −95.8 | −98.9 | −117.4 | −121.2 |
| 5,7-Dihydroxyphthalide (533) | −62.8 | −82.1 | −69.7 | −91.1 | −72.8 | −95.2 | −65.9 | −86.2 | −71.7 | −93.8 |
| 6-Methyl-4,5-dithia-2-octene (534) | −58.5 | −77.1 | −65.8 | −86.7 | −60.1 | −79.1 | −65.1 | −85.7 | −67.2 | −88.6 |
| 7-Epiclusianone (535) | −92.4 | −83.5 | −116.5 | −105.4 | −113.6 | −102.8 | −107.3 | −97.0 | −88.4 | −79.9 |
| Allamandin (536) | −94.5 | −100.6 | −95.4 | −101.5 | −97.3 | −103.5 | −86.9 | −92.5 | −91.7 | −97.6 |
| Allicin (537) | −56.7 | −74.8 | −63.3 | −83.4 | −57.0 | −75.1 | −59.1 | −77.8 | −66.2 | −87.3 |
| Amadannulen (538) | −107.7 | −107.3 | −111.7 | −111.2 | −123.0 | −122.5 | −105.4 | −105.0 | −113.7 | −113.2 |
| Anemonin (539) | −71.7 | −89.3 | −76.0 | −94.7 | −70.8 | −88.3 | −64.7 | −80.6 | −65.9 | −82.1 |
| Antibiotic CZ 34 (540) | −83.7 | −99.7 | −75.6 | −90.0 | −77.9 | −92.8 | −85.9 | −102.2 | −84.8 | −101.0 |
| Argutone (541) | −70.1 | −88.3 | −74.7 | −94.1 | −76.8 | −96.7 | −76.6 | −96.5 | −79.7 | −100.4 |
| Bakuchiol (542) | −94.0 | −106.3 | −89.6 | −101.4 | −100.5 | −113.7 | −96.7 | −109.4 | −104.0 | −117.7 |
| Brasiliensophyllic acid A (543) | −110.8 | −96.6 | −124.8 | −108.8 | −127.0 | −110.8 | −94.8 | −82.7 | −122.3 | −106.6 |
| Brasiliensophyllic acid C (544) | −109.1 | −94.4 | −140.6 | −121.6 | −119.0 | −102.9 | −101.5 | −87.8 | −95.4 | −82.5 |
| Centrolobin (545) | −102.6 | −108.8 | −103.0 | −109.2 | −110.6 | −117.2 | −106.3 | −112.7 | −116.5 | −123.5 |
| Chamone I (546) | −107.6 | −93.3 | −125.2 | −108.5 | −106.2 | −92.1 | −106.2 | −92.1 | −103.3 | −89.5 |
| Chamone II (547) | −68.0 | −59.0 | −130.2 | −113.0 | −109.2 | −94.8 | −80.5 | −69.9 | −97.2 | −84.3 |
| Champanone A (548) | −87.4 | −98.9 | −93.3 | −105.6 | −95.2 | −107.8 | −97.3 | −110.1 | −103.7 | −117.4 |
| Dhelwangin (549) | −84.1 | −99.5 | −82.9 | −98.1 | −82.9 | −98.1 | −85.1 | −100.7 | −95.1 | −112.6 |
| Garcinoic acid (550) | −130.3 | −124.5 | −128.9 | −123.1 | −141.3 | −134.9 | −107.3 | −102.5 | −147.6 | −141.0 |
| Ginkgolide A (551) | −94.7 | −91.8 | −101.4 | −98.3 | −94.5 | −91.5 | −73.9 | −71.6 | −73.9 | −71.6 |
| Guttiferone E (552) | −119.4 | −101.6 | −139.9 | −119.0 | −117.5 | −100.0 | −109.1 | −92.8 | −97.4 | −82.9 |
| Helipyrone B (553) | −97.6 | −104.1 | −81.6 | −87.1 | −97.7 | −104.2 | −92.2 | −98.3 | −110.0 | −117.3 |
| Helipyrone C (554) | −90.2 | −97.8 | −78.8 | −85.4 | −92.2 | −99.9 | −84.7 | −91.8 | −105.0 | −113.7 |
| Ialibinone A (555) | −86.4 | −88.6 | −90.7 | −93.0 | −96.1 | −98.6 | −85.7 | −87.9 | −80.7 | −82.8 |
| Ialibinone B (556) | −82.0 | −84.1 | −85.4 | −87.6 | −96.0 | −98.5 | −81.4 | −83.5 | −78.5 | −80.5 |
| Ialibinone C (557) | −85.8 | −86.8 | −94.3 | −95.4 | −100.6 | −101.8 | −89.2 | −90.3 | −86.9 | −88.0 |
| Ialibinone D (558) | −87.2 | −88.3 | −94.0 | −95.2 | −100.4 | −101.6 | −84.3 | −85.3 | −82.1 | −83.1 |
| Isobrasiliensophyllic acid A (559) | −109.8 | −95.7 | −130.4 | −113.7 | −123.7 | −107.9 | −87.2 | −76.1 | −102.2 | −89.1 |
| Moskachan C (560) | −82.1 | −97.5 | −88.7 | −105.3 | −87.5 | −103.9 | −89.0 | −105.6 | −98.3 | −116.6 |
| Nimbolide (561) | −94.3 | −87.5 | −111.7 | −103.5 | −126.4 | −117.2 | −95.0 | −88.0 | −82.3 | −76.3 |
| Pectinolide H (562) | −98.3 | −109.5 | −89.0 | −99.2 | −98.5 | −109.9 | −102.4 | −114.1 | −109.9 | −122.5 |
| Propolone A (563) | −61.3 | −55.4 | −111.8 | −101.1 | −94.9 | −85.8 | −104.3 | −94.3 | −91.0 | −82.3 |
| Sellovicine B (564) | −84.5 | −98.5 | −86.5 | −100.9 | −85.2 | −99.3 | −86.9 | −101.4 | −99.8 | −116.4 |
| Simonin A (565) | −128.9 | −124.1 | −106.9 | −103.0 | −116.1 | −111.8 | −102.6 | −98.8 | −105.0 | −101.1 |
| Tenulin (566) | −87.5 | −93.3 | −82.7 | −88.2 | −85.7 | −91.4 | −74.8 | −79.7 | −76.7 | −81.8 |
| Atractylodin (522) | −78.7 | −99.9 | −83.4 | −105.7 | −80.7 | −102.3 | −83.6 | −106.0 | −87.2 | −110.6 |
| Atractylodinol (523) | −87.6 | −108.1 | −91.0 | −112.2 | −85.5 | −105.4 | −88.0 | −108.5 | −94.9 | −117.0 |
| Capillene (342) | −67.4 | −90.4 | −74.2 | −99.4 | −69.6 | −93.3 | −72.6 | −97.3 | −79.5 | −106.6 |
| Peniophorin A (524) | −99.3 | −108.8 | −104.0 | −114.0 | −98.8 | −108.3 | −103.7 | −113.7 | −117.1 | −128.4 |
| Peniophorin B (525) | −80.7 | −97.2 | −88.8 | −107.0 | −87.4 | −105.4 | −83.9 | −101.1 | −95.6 | −115.3 |
| Thiarubrin A (526) | −76.3 | −89.8 | −87.1 | −102.5 | −80.3 | −94.5 | −80.2 | −94.4 | −87.1 | −102.4 |
| Arbutin (527) | −90.2 | −100.0 | −92.1 | −102.1 | −95.5 | −105.9 | −88.1 | −97.7 | −98.7 | −109.5 |
| Aucubin (528) | −108.5 | −111.1 | −93.7 | −96.0 | −105.7 | −108.2 | −92.5 | −94.7 | −110.9 | −113.6 |
| Diospyrodin (529) | −108.2 | −114.4 | −91.4 | −96.7 | −94.4 | −99.8 | −89.7 | −94.8 | −116.6 | −123.3 |
| Synthetic Inhibitors | ||||||||||
| (4-chlorophenyl)-[1-(4-chlorophenyl)-3-hydroxy-5-methyl-1H-pyrazol-4-yl]-methanone [99] (579) | −103.7 | −106.1 | ||||||||
| 3-(4-iodophenyl)-2-[4-(3,4-dichlorophenyl)-thiazol-2-ylamino]-propionic acid [99] (580) | −112.3 | −100.5 | ||||||||
| 3-phenyl-2-[5-(3-chlorobenzylidene)-2-thioxo-4-thiazolidinone]-propionic acid [100] (581) | −120.5 | −117.2 | ||||||||
| 4-[3-amino-4-(4-hydroxyphenyl)-1H-pyrazol-5-yl]benzene-1,3-diol [53] (582) | −92.3 | −101.1 | ||||||||
| 4,4′-{3-[(4-hydroxyphenyl)amino]-1H-pyrazole-4,5-diyl}diphenol [53] (583) | −119.2 | −120.5 | ||||||||
| 4-(3′-Amino-[1,1′-biphenyl]-3-yl)-1H-pyrazol-5-amine [32] (584) | −92.1 | −105.1 | ||||||||
| 4-(5-Amino-4-(3'-amino-[1,1′-biphenyl]-3-yl)-1H-pyrazol-3-yl)-phenol [32] (585) | −124.2 | −127.6 | ||||||||
| Chloroquine | −101.2 | −106.4 | −93.5 | −98.3 | −101.8 | −107.0 | ||||
| Doxorubicin | −119.6 | −105.4 | −103.8 | −91.5 | −144.2 | −127.0 | ||||
| 2-((2-(1H-pyrazolo[3,4-c]pyridin-3-yl)-6-(trifluoromethyl)pyrimidin-4-yl)-amino)-ethanol [56] (586) | −113.0 | −118.2 | −105.2 | −110.1 | −123.4 | −129.2 | ||||
| 2-cyclobutylmethoxy-5′-fluoroadenosine [33] (587) | −119.4 | −121.4 | −115.8 | −117.8 | −132.3 | −134.5 | ||||
| 2-phenoxy-5′-deoxyadenosine [34] (588) | −118.4 | −121.6 | −112.9 | −115.9 | −120.9 | −124.2 | ||||
| 2-amino-6-bromo-7-(trifluoromethyl)-[1,8]-naphthyridine-3-carboxamide [54] (589) | −99.6 | −103.1 | −78.3 | −81.0 | −90.1 | −93.3 | ||||
| 2-amino-6-bromo-7-(2-benzyloxy-2-methylethyl)-[1,8]-naphthyridine-3-carboxamide [54] (590) | −122.1 | −117.7 | −91.1 | −87.8 | −117.4 | −113.1 | ||||
| 4-amino-2-(2-fluoroethoxy)-N-(2-hydroxyethyl)-purido[2,3-d]pyrimidin-5(8H)-one [103] (591) | −98.3 | −109.6 | −81.3 | −90.6 | −93.5 | −104.3 | ||||
| 4-amino-2-(2-ethylcyclohexoxy)-N-(2-hydroxycyclopentyl)-purido[2,3-d]-pyrimidin-5(8H)-one [103] (592) | −120.6 | −122.1 | −97.0 | −98.2 | −115.7 | −117.1 | ||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Snow Setzer, M.; Sharifi-Rad, J.; Setzer, W.N. The Search for Herbal Antibiotics: An In-Silico Investigation of Antibacterial Phytochemicals. Antibiotics 2016, 5, 30. https://doi.org/10.3390/antibiotics5030030
Snow Setzer M, Sharifi-Rad J, Setzer WN. The Search for Herbal Antibiotics: An In-Silico Investigation of Antibacterial Phytochemicals. Antibiotics. 2016; 5(3):30. https://doi.org/10.3390/antibiotics5030030
Chicago/Turabian StyleSnow Setzer, Mary, Javad Sharifi-Rad, and William N. Setzer. 2016. "The Search for Herbal Antibiotics: An In-Silico Investigation of Antibacterial Phytochemicals" Antibiotics 5, no. 3: 30. https://doi.org/10.3390/antibiotics5030030
APA StyleSnow Setzer, M., Sharifi-Rad, J., & Setzer, W. N. (2016). The Search for Herbal Antibiotics: An In-Silico Investigation of Antibacterial Phytochemicals. Antibiotics, 5(3), 30. https://doi.org/10.3390/antibiotics5030030