Thioridazine: A Non-Antibiotic Drug Highly Effective, in Combination with First Line Anti-Tuberculosis Drugs, against Any Form of Antibiotic Resistance of Mycobacterium tuberculosis Due to Its Multi-Mechanisms of Action
Abstract
:1. Introduction
2. Mycobacterium tuberculosis and Phenothiazines: Chlorpromazine, Thioridazine, in Vitro Activities
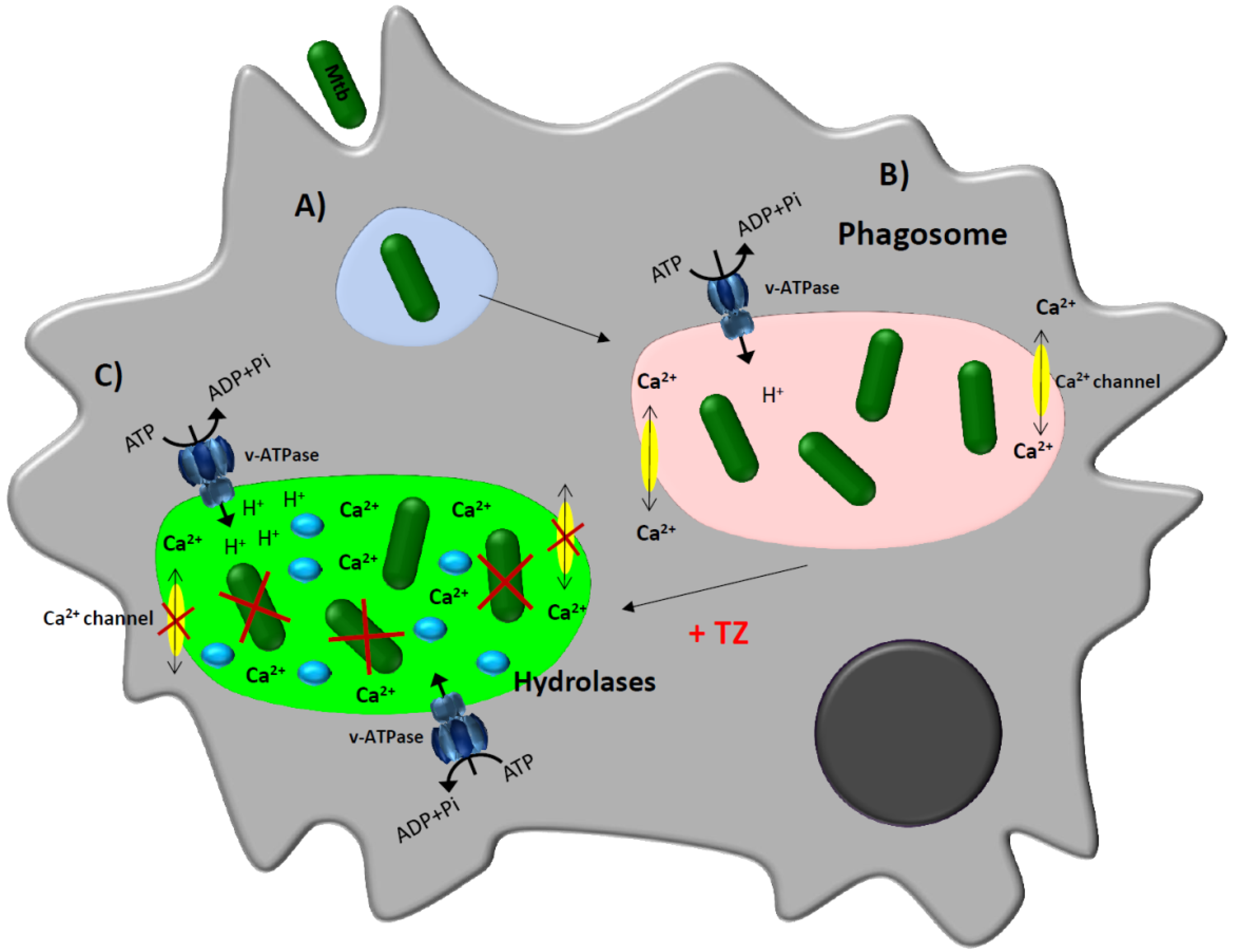
3. Thioridazine and Its Effect on Intracellular Mycobacterium tuberculosis
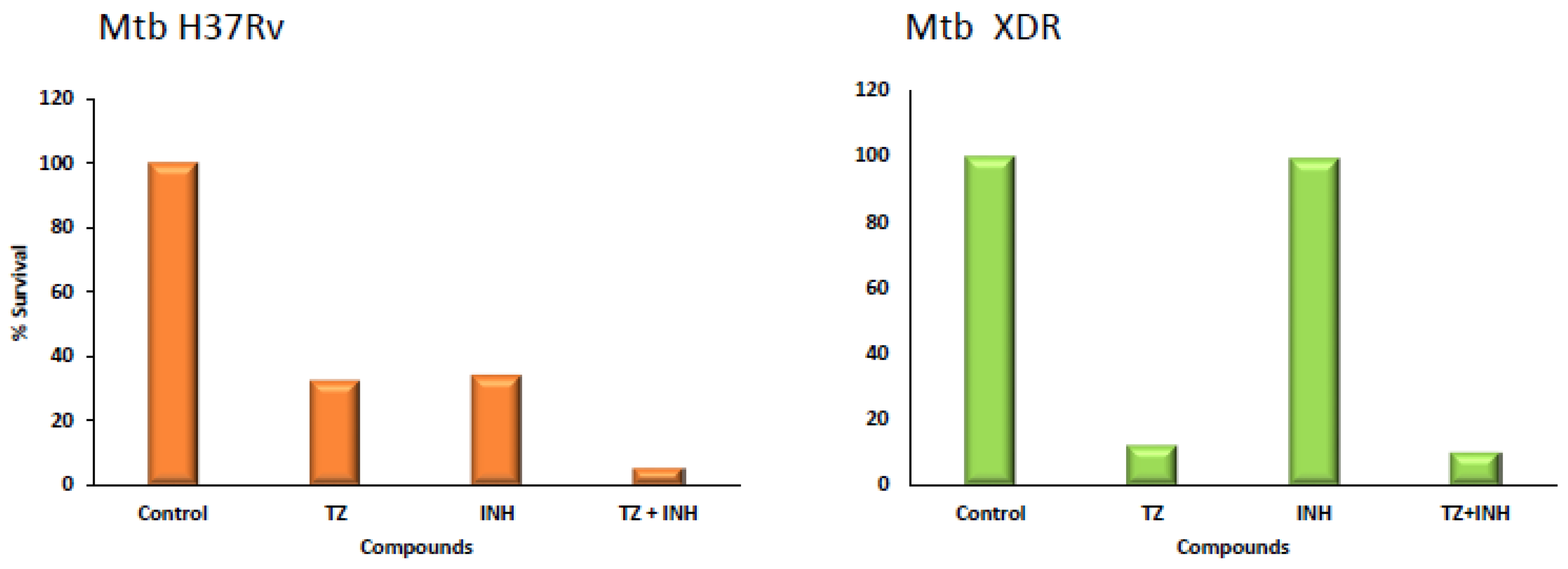
4. Mono and Combinational Therapy with TZ: The Mouse and the Human
5. Important Considerations for Therapy of MDR/XDR Mtb Patients with TZ in Combination with Antibiotics to Which the Infecting Organism Is Resistant
6. Costs Associated with the Care of an MDR-TB Patient
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Munsiff, S.S.; Nivin, B.; Sacajiu, G.; Mathema, B.; Bifani, P.; Kreiswirth, B.N. Persistence of a highly resistant strain of tuberculosis in New York City during 1990–1999. J. Infect. Dis. 2003, 188, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Matteelli, A.; Roggi, A.; Carvalho, A.C. Extensively drug-resistant tuberculosis: Epidemiology and management. Clin. Epidemiol. 2014, 6, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Parida, S.K.; Axelsson-Robertson, R.; Rao, M.V.; Singh, N.; Master, I.; Lutckii, A.; Keshavjee, S.; Andersson, J.; Zumla, A.; Maeurer, M. Totally drug-resistant tuberculosis and adjunct therapies. J. Intern. Med. 2015, 277, 388–405. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Tuberculosis Report 2015; World Health Organization: Geneva, Switzerland, 2015; Available online: http://www.who.int/tb/publications/global_report/en/ (accessed on 3 October 2016).
- World Health Organization. Key Bottlenecks in M/XDR-TB Control and Patient Care; World Health Organization: Geneva, Switzerland, 2009; Available online: http://www.who.int/tb/challenges/mdr/bottlenecks/en/ (accessed on 3 October 2016).
- Agyeman, A.A.; Ofori-Asenso, R. Efficacy and safety profile of linezolid in the treatment of multidrug-resistant (MDR) and extensively drug-resistant (XDR) tuberculosis: A systematic review and meta-analysis. Ann. Clin. Microbiol. Antimicrob. 2016, 15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Falagas, M.E.; Vardakas, K.Z.; Wang, R.; Qin, R.; Wang, J.; Liu, Y. Systematic review and meta-analysis of the efficacy and safety of therapy with linezolid containing regimens in the treatment of multidrug-resistant and extensively drug-resistant tuberculosis. J. Thorac. Dis. 2015, 7, 603–615. [Google Scholar] [PubMed]
- Olaru, I.D.; von Groote-Bidlingmaier, F.; Heyckendorf, J.; Yew, W.W.; Lange, C.; Chang, K.C. Novel drugs against tuberculosis: A clinician’s perspective. Eur. Respir. J. 2015, 45, 1119–1131. [Google Scholar] [CrossRef] [PubMed]
- Ohlow, M.J.; Moosmann, B. Phenothiazine: The seven lives of pharmacology’s first lead structure. Drug Discov. Today 2011, 16, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Healy, D. Explorations in a New World. In The Creation of Psychopharmacology; Harvard University Press: Cambridge, CA, USA, 2004. [Google Scholar]
- Gonzalez-Gonzalez, E. Use of largactil in pulmonary tuberculosis. Rev. Esp. Tuberc. 1958, 27, 134–136. [Google Scholar] [PubMed]
- Molnár, J.; Béládi, I.; Földes, I. Studies on antituberculotic action of some phenothiazine derivatives in vitro. Zentralbl. Bakteriol. Orig. A 1977, 239, 521–526. [Google Scholar] [PubMed]
- Kristiansen, J.E.; Vergmann, B. The antibacterial effect of selected phenothiazines and thioxanthenes on slow-growing mycobacteria. Acta Pathol. Microbiol. Immunol. Scand. B 1986, 94, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Crowle, A.J.; Douvas, G.S.; May, M.H. Chlorpromazine: A drug potentially useful for treating mycobacterial infections. Chemotherapy 1992, 38, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Gardos, G.; Cole, J.O. Maintenance antipsychotic therapy: Is the cure worse than the disease? Am. J. Psychiatry 1976, 133, 32–36. [Google Scholar] [PubMed]
- Amaral, L.; Kristiansen, J.E.; Abebe, L.S.; Millett, W. Inhibition of the respiration of multi-drug resistant clinical isolates of Mycobacterium tuberculosis by thioridazine: Potential use for initial therapy of freshly diagnosed tuberculosis. J. Antimicrob. Chemother. 1996, 38, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Kristiansen, J.E.; Viveiros, M.; Atouguia, J. Activity of phenothiazines against antibiotic-resistant Mycobacterium tuberculosis: A review supporting further studies that may elucidate the potential use of thioridazine as anti-tuberculosis therapy. J. Antimicrob. Chemother. 2001, 47, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Ängeby, K.; Juréen, P.; Chryssanthou, E.; Schön, T. Tentative susceptibility testing breakpoint for the neuroleptic drug thioridazine, a treatment option for multi-and extensively drug resistant tuberculosis. Int. J. Mycobacteriol. 2012, 1, 177–179. [Google Scholar] [CrossRef] [PubMed]
- Musuka, S.; Srivastava, S.; Dona, C.W.S.; Meek, C.; Leff, R.; Pasipanodya, J.; Gumbo, T. Thioridazine pharmacokinetic-pharmacodynamic parameters “Wobble” during treatment of tuberculosis: A theoretical basis for shorter-duration curative monotherapy with congeners. Antimicrob. Agents Chemother. 2013, 57, 5870–5877. [Google Scholar] [CrossRef] [PubMed]
- Vesenbeckh, S.; Krieger, D.; Bettermann, G.; Schönfeld, N.; Bauer, T.T.; Rüssmann, H.; Mauch, H. Neuroleptic drugs in the treatment of tuberculosis: Minimal inhibitory concentrations of different phenothiazines against Mycobacterium tuberculosis. Tuberculosis 2016, 98, 27–29. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zhang, J.; Li, C.; Guo, Q.; Jiang, Y.; Wei, J.; Qiu, Y.; Zhao, X.; Zhao, L.L.; Lu, J.; et al. Antimycobacterial activity of five efflux pump inhibitors against Mycobacterium tuberculosis clinical isolates. J. Antibiot. 2016, 69, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Coelho, T.; Machado, D.; Couto, I.; Maschmann, R.; Ramos, D.; von Groll, A.; Rossetti, M.L.; Silva, P.A.; Viveiros, M. Enhancement of antibiotic activity by efflux inhibitors against multidrug resistant Mycobacterium tuberculosis clinical isolates from Brazil. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Pieroni, M.; Machado, D.; Azzali, E.; Santos Costa, S.; Couto, I.; Costantino, G.; Viveiros, M. Rational design and synthesis of thioridazine analogues as enhancers of the antituberculosis therapy. J. Med. Chem. 2015, 58, 5842–5853. [Google Scholar] [CrossRef] [PubMed]
- Ordway, D.; Viveiros, M.; Leandro, C.; Bettencourt, R.; Almeida, J.; Martins, M.; Kristiansen, J.E.; Molnar, J.; Amaral, L. Clinical concentrations of thioridazine kill intracellular multidrug-resistant Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2003, 47, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.; Viveiros, M.; Amaral, L. Inhibitors of Ca2+ and K+ transport enhance intracellular killing of M. tuberculosis by non-killing macrophages. In Vivo 2008, 22, 69–75. [Google Scholar] [PubMed]
- Machado, D.; Pires, D.; Perdigão, J.; Couto, I.; Portugal, I.; Martins, M.; Amaral, L.; Anes, E.; Viveiros, M. Ion channel blockers as antimicrobial agents, efflux inhibitors, and enhancers of macrophage killing activity against drug resistant Mycobacterium tuberculosis. PLoS ONE 2016, 11, e0149326. [Google Scholar] [CrossRef] [PubMed]
- De Knegt, G.J.; ten Kate, M.T.; van Soolingen, D.; Aarnoutse, R.; Boeree, M.J.; Bakker-Woudenberg, I.A.; de Steenwinkel, J.E. Enhancement of in vitro activity of tuberculosis drugs by addition of thioridazine is not reflected by improved in vivo efficacy. Tuberculosis 2014, 94, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Van Ingen, J. The broad-spectrum antimycobacterial activities of phenothiazines, in vitro: Somewhere in all of this there may be patentable potentials. Recent Pat. Antiinfect. Drug Discov. 2011, 6, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.; Schelz, Z.; Martins, A.; Molnar, J.; Hajös, G.; Riedl, Z.; Viveiros, M.; Yalcin, I.; Aki-Sener, E.; Amaral, L. In vitro and ex vivo activity of thioridazine derivatives against Mycobacterium tuberculosis. Int. J. Antimicrob. Agents 2007, 29, 338–340. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Kristiansen, J.; Lorian, V. Synergic effect of chlorpromazine on the activity of some antibiotics. J. Antimicrob. Chemother. 1992, 30, 556–558. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Lorian, V. Effects of chlorpromazine on the cell envelope proteins of Escherichia coli. Antimicrob. Agents Chemother. 1991, 35, 1923–1924. [Google Scholar] [CrossRef] [PubMed]
- Viveiros, M.; Amaral, L. Enhancement of antibiotic activity against poly-drug resistant Mycobacterium tuberculosis by phenothiazines. Int. J. Antimicrob. Agents 2001, 17, 225–228. [Google Scholar] [CrossRef]
- Viveiros, M.; Jesus, A.; Brito, M.; Leandro, C.; Martins, M.; Ordway, D.; Molnar, A.M.; Molnar, J.; Amaral, L. Inducement and reversal of tetracycline resistance in Escherichia coli K-12 and expression of proton gradient-dependent multidrug efflux pump genes. Antimicrob. Agents Chemother. 2005, 49, 3578–3582. [Google Scholar] [CrossRef] [PubMed]
- Viveiros, M.; Portugal, I.; Bettencourt, R.; Victor, T.C.; Jordaan, A.M.; Leandro, C.; Ordway, D.; Amaral, L. Isoniazid-induced transient high-level resistance in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2002, 46, 2804–2810. [Google Scholar] [CrossRef] [PubMed]
- Machado, D.; Couto, I.; Perdigão, J.; Rodrigues, L.; Portugal, I.; Baptista, P.; Veigas, B.; Amaral, L.; Viveiros, M. Contribution of efflux to the emergence of isoniazid and multidrug resistance in Mycobacterium tuberculosis. PLoS ONE 2012, 7, e34538. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.; Machado, D.; Couto, I.; Amaral, L.; Viveiros, M. Contribution of efflux activity to isoniazid resistance in the Mycobacterium tuberculosis complex. Infect. Genet. Evol. 2012, 12, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.K.; Mazumdar, K.; Dastidar, S.G.; Karakousis, P.C.; Amaral, L. New patentable use of an old neuroleptic compound thioridazine to combat tuberculosis: A gene regulation perspective. Recent Pat. Antiinfect. Drug Discov. 2011, 6, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.K.; Mehra, S.; Kaushal, D. A Mycobacterium tuberculosis sigma factor network responds to cell-envelope damage by the promising anti-mycobacterial thioridazine. PLoS ONE 2010, 5, e10069. [Google Scholar] [CrossRef] [PubMed]
- De Keijzer, J.; Mulder, A.; de Haas, P.E.; de Ru, A.H.; Heerkens, E.M.; Amaral, L.; van Soolingen, D.; van Veelen, P.A. Thioridazine alters the cell-envelope permeability of Mycobacterium tuberculosis. J. Proteom. Res. 2016, 15, 1776–1786. [Google Scholar] [CrossRef] [PubMed]
- Yano, T.; Li, L.S.; Weinstein, E.; Teh, J.S.; Rubin, H. Steady-state kinetics and inhibitory action of antitubercular phenothiazines on Mycobacterium tuberculosis type-II NADH-menaquinone oxidoreductase (NDH-2). J. Biol. Chem. 2006, 281, 11456–11463. [Google Scholar] [CrossRef] [PubMed]
- Teh, J.S.; Yano, T.; Rubin, H. Type II NADH: Menaquinone oxidoreductase of Mycobacterium tuberculosis. Infect. Disord. Drug Targets. 2007, 7, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Singh, A. Phenothiazines as anti-tubercular agents: Mechanistic insights and clinical implications. Expert Opin. Invest. Drugs 2011, 20, 1665–1676. [Google Scholar] [CrossRef] [PubMed]
- Sohaskey, C.D. Nitrate enhances the survival of Mycobacterium tuberculosis during inhibition of respiration. J. Bacteriol. 2008, 190, 2981–2986. [Google Scholar] [CrossRef] [PubMed]
- Sohaskey, C. Latent tuberculosis: Is there a role for thioridazine? Recent Pat. Antiinfect. Drug Discov. 2011, 6, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Daniel, W.A.; Wojcikowski, J. Contribution of lysosomal trapping to the total tissue uptake of psychotropic drugs. Pharmacol. Toxicol. 1997, 80, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Daniel, W.A.; Wojcikowski, J. The role of lysosomes in the cellular distribution of thioridazine and potential drug interactions. Toxicol. Appl. Pharmacol. 1999, 158, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Leonard, A.; Viveiros, M.; Kristiansen, J.E. “Non-Antibiotics”: Alternative therapy for the management of MDRTB and MRSA in economically disadvantaged Countries. Curr. Drug Targets 2006, 7, 887–891. [Google Scholar]
- Reeves, E.P.; Lu, H.; Jacobs, H.L.; Messina, C.G.; Bolsover, S.; Gabella, G.; Potma, E.O.; Warley, A.; Roes, J.; Segal, A.W. Killing activity of neutrophils is mediated through activation of proteases by K+ flux. Nature 2002, 416, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Ahluwalia, J.; Tinker, A.; Clapp, L.H.; Duchen, M.R.; Abramov, A.Y.; Pope, S.; Nobles, M.; Segal, A.W. The large-conductance Ca2+-activated K+ channel is essential for innate immunity. Nature 2004, 427, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Viveiros, M.; Martins, M.; Rodrigues, L.; Machado, D.; Couto, I.; Ainsa, J.; Amaral, L. Inhibitors of mycobacterial efflux pumps as potential boosters for anti-tubercular drugs. Expert Rev. Anti. Infect. Ther. 2012, 10, 983–998. [Google Scholar] [CrossRef] [PubMed]
- Torrelles, J.B.; Schlesinger, L.S. Diversity in Mycobacterium tuberculosis mannosylated cell wall determinants impacts adaptation to the host. Tuberculosis 2010, 90, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Józefowski, S.; Sobota, A.; Kwiatkowska, K. How Mycobacterium tuberculosis subverts host immune responses. Bioessays 2008, 30, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Yoshikai, Y. Immunological protection against mycobacterium tuberculosis infection. Crit. Rev. Immunol. 2006, 26, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Wall, A.A.; Condon, N.D.; Yeo, J.C.; Hamilton, N.A.; Stow, J.L. Dynamic imaging of the recycling endosomal network in macrophages. Methods Cell Biol. 2015, 130, 1–18. [Google Scholar] [PubMed]
- Van der Wel, N.; Hava, D.; Houben, D.; Fluitsma, D.; van Zon, M.; Pierson, J.; Brenner, M.; Peters, P.J.M. Tuberculosis and M. leprae translocate from the phagolysosome to the cytosol in myeloid cells. Cell 2007, 129, 1287–1298. [Google Scholar] [CrossRef] [PubMed]
- Ganley, I.G. Autophagosome maturation and lysosomal fusion. Essays Biochem. 2013, 55, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Harvey, W.R. Voltage coupling of primary H+ V-ATPases to secondary Na+- or K+-dependent transporters. J. Exp. Biol. 2009, 212, 1620–1629. [Google Scholar] [CrossRef] [PubMed]
- Csonka, Á.; Spengler, G.; Martins, A.; Ocsovszki, I.; Christensen, J.B.; Hendricks, O.; Kristiansen, J.E.; Amaral, L.; Molnar, J. Effect of thioridazine stereoisomers on the drug accumulation of mouse lymphoma and human prostate cancer cell lines in vitro. In Vivo 2013, 27, 815–820. [Google Scholar] [PubMed]
- Spengler, G.; Molnar, J.; Viveiros, M.; Amaral, L. Thioridazine induces apoptosis of multidrug-resistant mouse lymphoma cells transfected with the human ABCB1 and inhibits the expression of P-glycoprotein. Anticancer Res. 2011, 31, 4201–4205. [Google Scholar] [PubMed]
- Spengler, G.; Takács, D.; Horváth, A.; Riedl, Z.; Hajós, G.; Amaral, L.; Molnár, J. Multidrug resistance reversing activity of newly developed phenothiazines on P-glycoprotein (ABCB1)-related resistance of mouse T-lymphoma cells. Anticancer Res. 2014, 34, 1737–1741. [Google Scholar] [PubMed]
- Amaral, L.; Martins, M.; Viveiros, M. Enhanced killing of intracellular multidrug-resistant Mycobacterium tuberculosis by compounds that affect the activity of efflux pumps. J. Antimicrob. Chemother. 2007, 59, 1237–1246. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.N.; Takaki, K.; Connolly, L.E.; Wiedenhoft, H.; Winglee, K.; Humbert, O.; Edelstein, P.H.; Cosma, C.L.; Ramakrishnan, L. Drug tolerance in replicating mycobacteria mediated by a macrophage-induced efflux mechanism. Cell 2011, 145, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Tyagi, S.; Almeida, D.V.; Maiga, M.C.; Ammerman, N.C.; Bishai, W.R. Acceleration of tuberculosis treatment by adjunctive therapy with verapamil as an efflux inhibitor. Am. J. Respir. Crit. Care Med. 2013, 188, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Molnar, J. Mechanisms by which thioridazine in combination with antibiotics cures extensively drug-resistant infections of pulmonary tuberculosis. In Vivo 2014, 28, 267–271. [Google Scholar] [PubMed]
- Amaral, L.; Martins, A.; Spengler, G.; Hunyadi, A.; Molnar, J. The mechanism by which the phenothiazine thioridazine contributes to cure problematic drug-resistant forms of pulmonary tuberculosis: Recent patents for “new use”. Recent Pat. Antiinfect. Drug Discov. 2013, 8, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Udwadia, Z.; Abbate, E.; van Soolingen, D. The added effect of thioridazine in the treatment of drug-resistant tuberculosis. Int. J. Tuberc. Lung Dis. 2012, 16, 1706–1708. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Molnar, J. Why and how thioridazine in combination with antibiotics to which the infective strain is resistant will cure totally drug-resistant tuberculosis. Expert Rev. Anti. Infect. Ther. 2012, 10, 869–873. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L. Thioridazine: An old neuroleptic effective against totally drug resistant tuberculosis. Acta Med. Port. 2012, 25, 118–121. [Google Scholar] [PubMed]
- Martins, M. Targeting the human macrophage with combinations of drugs and inhibitors of Ca2+ and K+ transport to enhance the killing of intracellular multi-drug resistant Mycobacterium tuberculosis (MDR-TB)—A novel, patentable approach to limit the emergence of XDR-TB. Recent Pat. Antiinfect. Drug Discov. 2011, 6, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Martins, A.; Molnar, J.; Kristiansen, J.E.; Martins, M.; Viveiros, M.; Rodrigues, L.; Spengler, G.; Couto, I.; Ramos, J.; et al. Phenothiazines, bacterial efflux pumps and targeting the macrophage for enhanced killing of intracellular XDRTB. In Vivo 2010, 24, 409–424. [Google Scholar] [PubMed]
- Gupta, S.; Salam, N.; Srivastava, V.; Singla, R.; Behera, D.; Khayyam, K.U.; Korde, R.; Malhotra, P.; Saxena, R.; Natarajan, K. Voltage gated calcium channels negatively regulate protective immunity to Mycobacterium tuberculosis. PLoS ONE 2009, 4, e5305. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.; Viveiros, M.; Kristiansen, J.E.; Molnar, J.; Amaral, L. The curative activity of thioridazine on mice infected with Mycobacterium tuberculosis. In Vivo 2007, 21, 771–775. [Google Scholar] [PubMed]
- Dutta, N.K.; Pinn, M.L.; Karakousis, P.C. Sterilizing activity of thioridazine in combination with the first-line regimen against acute murine tuberculosis. Antimicrob. Agents Chemother. 2014, 58, 5567–5569. [Google Scholar] [CrossRef] [PubMed]
- Dutta, N.K.; Pinn, M.L.; Karakousis, P.C. Reduced emergence of isoniazid resistance with concurrent use of thioridazine against acute murine tuberculosis. Antimicrob. Agents Chemother. 2014, 58, 4048–4053. [Google Scholar] [CrossRef] [PubMed]
- Van Soolingen, D.; Hernandez-Pando, R.; Orozco, H.; Aguilar, D.; Magis-Escurra, C.; Amaral, L.; van Ingen, J.; Boeree, M.J. The antipsychotic thioridazine shows promising therapeutic activity in a mouse model of multidrug-resistant tuberculosis. PLoS ONE 2010, 5, e12640. [Google Scholar] [CrossRef] [PubMed]
- Abbate, E.; Vescovo, M.; Natiello, M.; Cufré, M.; García, A.; Gonzalez Montaner, P.; Ambroggi, M.; Ritacco, V.; van Soolingen, D. Successful alternative treatment of extensively drug-resistant tuberculosis in Argentina with a combination of linezolid, moxifloxacin and thioridazine. J. Antimicrob. Chemother. 2012, 67, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Udwadia, Z.F.; Sen, T.; Pinto, L.M. Safety and efficacy of thioridazine as salvage therapy in Indian patients with XDR-TB. Recent Pat. Antiinfect. Drug Discov. 2011, 6, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Martins, A.; Spengler, G.; Molnar, J. Efflux pumps of Gram-negative bacteria: What they do, how they do it, with what and how to deal with them. Front. Pharmacol. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Hunyadi, A.; Amaral, L. Mechanisms of resistance in bacteria: An evolutionary approach. Open Microbiol. J. 2013, 7, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Spengler, G.; Rodrigues, L.; Martins, A.; Martins, M.; McCusker, M.; Cerca, P.; Machado, L.; Costa, S.S.; Ntokou, E.; Couto, I.; et al. Genetic response of Salmonella enterica serotype Enteritidis to thioridazine rendering the organism resistant to the agent. Int. J. Antimicrob. Agents. 2012, 39, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Walter, N.D.; Dolganov, G.M.; Garcia, B.J.; Worodria, W.; Andama, A.; Musisi, E.; Ayakaka, I.; Van, T.T.; Voskuil, M.I.; de Jong, B.C.; et al. Transcriptional adaptation of drug-tolerant Mycobacterium tuberculosis during treatment of human tuberculosis. J. Infect. Dis. 2015, 212, 990–998. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; Cerca, P.; Spengler, G.; Machado, L.; Martins, A.; Couto, I.; Viveiros, M.; Fanning, S.; Pagès, J.M. Ethidium bromide efflux by Salmonella: Modulation by metabolic energy, pH, ions and phenothiazines. Int. J. Antimicrob. Agents 2011, 38, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Machado, L.; Costa, S.; Cerca, P.; Spengler, G.; Viveiros, M.; Amaral, L. Role of calcium in the efflux system of Escherichia coli. Int. J. Antimicrob. Agents 2011, 37, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Black, P.A.; Warren, R.M.; Louw, G.E.; van Helden, P.D.; Victor, T.C.; Kana, B.D. Energy metabolism and drug efflux in Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2014, 58, 2491–2503. [Google Scholar] [CrossRef] [PubMed]
- Schmalstieg, A.M.; Srivastava, S.; Belkaya, S.; Deshpande, D.; Meek, C.; Leff, R.; van Oers, N.S.; Gumbo, T. The antibiotic resistance arrow of time: Efflux pump induction is a general first step in the evolution of mycobacterial drug resistance. Antimicrob. Agents Chemother. 2012, 56, 4806–4815. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Spengler, G.; Martins, M.; Rodrigues, L.; Viveiros, M.; Davin-Regli, A.; Chevalier, J.; Couto, I.; Pagès, J.M.; Amaral, L. Physiological characterisation of the efflux pump system of antibiotic-susceptible and multidrug-resistant Enterobacter aerogenes. Int. J. Antimicrob. Agents 2010, 36, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Iversen, C.; Rodrigues, L.; Spengler, G.; Ramos, J.; Kern, W.V.; Couto, I.; Viveiros, M.; Fanning, S.; Pages, J.M.; et al. An AcrAB-mediated multidrug-resistant phenotype is maintained following restoration of wild-type activities by efflux pump genes and their regulators. Int. J. Antimicrob. Agents 2009, 34, 602–604. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Spengler, G.; Rodrigues, L.; Viveiros, M.; Ramos, J.; Martins, M.; Couto, I.; Fanning, S.; Pagès, J.M.; Bolla, J.M.; et al. pH Modulation of efflux pump activity of multi-drug resistant Escherichia coli: Protection during its passage and eventual colonization of the colon. PLoS ONE 2009, 4, e6656. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Couto, I.; Aagaard, L.; Martins, M.; Viveiros, M.; Kristiansen, J.E.; Amaral, L. Prolonged exposure of methicillin-resistant Staphylococcus aureus (MRSA) COL strain to increasing concentrations of oxacillin results in a multidrug-resistant phenotype. Int. J. Antimicrob. Agents 2007, 29, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Viveiros, M.; Dupont, M.; Rodrigues, L.; Couto, I.; Davin-Regli, A.; Martins, M.; Pagès, J.M.; Amaral, L. Antibiotic stress, genetic response and altered permeability of E. coli. PLoS ONE 2007, 2, e365. [Google Scholar] [CrossRef] [PubMed]
- Amaral, L.; van Soolingen, D. A novel advanced laboratory diagnosis to guide tuberculosis drug therapy. Recent Pat. Antiinfect. Drug Discov. 2015, 10, 71–73. [Google Scholar] [CrossRef] [PubMed]
- Beach, S.R.; Celano, C.M.; Noseworthy, P.A.; Januzzi, J.L.; Huffman, J.C. QTc prolongation, torsades de pointes, and psychotropic medications. Psychosomatics 2013, 54, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Thanacoody, R.H.; Daly, A.K.; Reilly, J.G.; Ferrier, I.N.; Thomas, S.H. Factors affecting drug concentrations and QT interval during thioridazine therapy. Clin. Pharmacol. Ther. 2007, 82, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Thanacoody, R.H. Thioridazine: The good and the bad. Recent Pat. Antiinfect. Drug Discov. 2011, 6, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.S.; Koh, W.J. Synthetic investigational new drugs for the treatment of tuberculosis. Expert Opin. Invest. Drugs 2016, 25, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Marks, S.M.; Hirsch-Moverman, Y.; Salcedo, K.; Graviss, E.A.; Oh, P.; Seaworth, B.; Flood, J.; Armstrong, L.; Armitige, L.; Mase, S. TB epidemiologic studies consortium. Characteristics and costs of multidrug-resistant tuberculosis in-patient care in the United States, 2005–2007. Int. J. Tuberc. Lung Dis. 2016, 20, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Shah, N.S.; Pratt, R.; Armstrong, L.; Robison, V.; Castro, K.G.; Cegielski, J.P. Extensively drug-resistant tuberculosis in the United States, 1993–2007. JAMA 2008, 300, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- Raviglione, M.; Sulis, G. Tuberculosis 2015: Burden, challenges and strategy for control and elimination. Infect. Dis Rep. 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Sotgiu, G.; Centis, R.; D’ambrosio, L.; Migliori, G.B. Tuberculosis treatment and drug regimens. Cold Spring Harb. Perspect. Med. 2015, 5, a017822. [Google Scholar] [CrossRef] [PubMed]
- Wasserman, S.; Meintjes, G.; Maartens, G. Linezolid in the treatment of drug-resistant tuberculosis: The challenge of its narrow therapeutic index. Expert Rev. Anti. Infect. Ther. 2016, 14, 901–915. [Google Scholar] [CrossRef] [PubMed]
- Parumasivam, T.; Chan, J.G.; Pang, A.; Quan, D.H.; Triccas, J.A.; Britton, W.J.; Chan, H.K. In vitro evaluation of novel inhalable dry powders consisting of thioridazine and rifapentine for rapid tuberculosis treatment. Eur. J. Pharm. Biopharm. 2016, 107, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Vibe, C.B.; Fenaroli, F.; Pires, D.; Wilson, S.R.; Bogoeva, V.; Kalluru, R.; Speth, M.; Anes, E.; Griffiths, G.; Hildahl, J. Thioridazine in PLGA nanoparticles reduces toxicity and improves rifampicin therapy against mycobacterial infection in zebrafish. Nanotoxicology 2016, 10, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Winters, N.; Butler-Laporte, G.; Menzies, D. Efficacy and safety of World Health Organization group 5 drugs for multidrug-resistant tuberculosis treatment. Eur. Respir. J. 2015, 46, 1461–1470. [Google Scholar] [CrossRef] [PubMed]
| Culture Systems | TZ | References |
|---|---|---|
| MICs | ||
| MB7H10 | 8–10 μg/mL | [18,19,20] |
| Broth microdilution | 8–15 μg/mL | [21,22,23] |
| Bactec TB-460 | 15 μg/mL | [24,25] |
| Bactec MGIT 960 | 15 μg/mL | [26] |
| Cytotoxicity * | ||
| IC50 | 13.78 μM | [23] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amaral, L.; Viveiros, M. Thioridazine: A Non-Antibiotic Drug Highly Effective, in Combination with First Line Anti-Tuberculosis Drugs, against Any Form of Antibiotic Resistance of Mycobacterium tuberculosis Due to Its Multi-Mechanisms of Action. Antibiotics 2017, 6, 3. https://doi.org/10.3390/antibiotics6010003
Amaral L, Viveiros M. Thioridazine: A Non-Antibiotic Drug Highly Effective, in Combination with First Line Anti-Tuberculosis Drugs, against Any Form of Antibiotic Resistance of Mycobacterium tuberculosis Due to Its Multi-Mechanisms of Action. Antibiotics. 2017; 6(1):3. https://doi.org/10.3390/antibiotics6010003
Chicago/Turabian StyleAmaral, Leonard, and Miguel Viveiros. 2017. "Thioridazine: A Non-Antibiotic Drug Highly Effective, in Combination with First Line Anti-Tuberculosis Drugs, against Any Form of Antibiotic Resistance of Mycobacterium tuberculosis Due to Its Multi-Mechanisms of Action" Antibiotics 6, no. 1: 3. https://doi.org/10.3390/antibiotics6010003
APA StyleAmaral, L., & Viveiros, M. (2017). Thioridazine: A Non-Antibiotic Drug Highly Effective, in Combination with First Line Anti-Tuberculosis Drugs, against Any Form of Antibiotic Resistance of Mycobacterium tuberculosis Due to Its Multi-Mechanisms of Action. Antibiotics, 6(1), 3. https://doi.org/10.3390/antibiotics6010003






