Enhancing Antibiotic Effect by Photodynamic: The Case of Klebsiella pneumoniae
Abstract
1. Introduction
2. Results
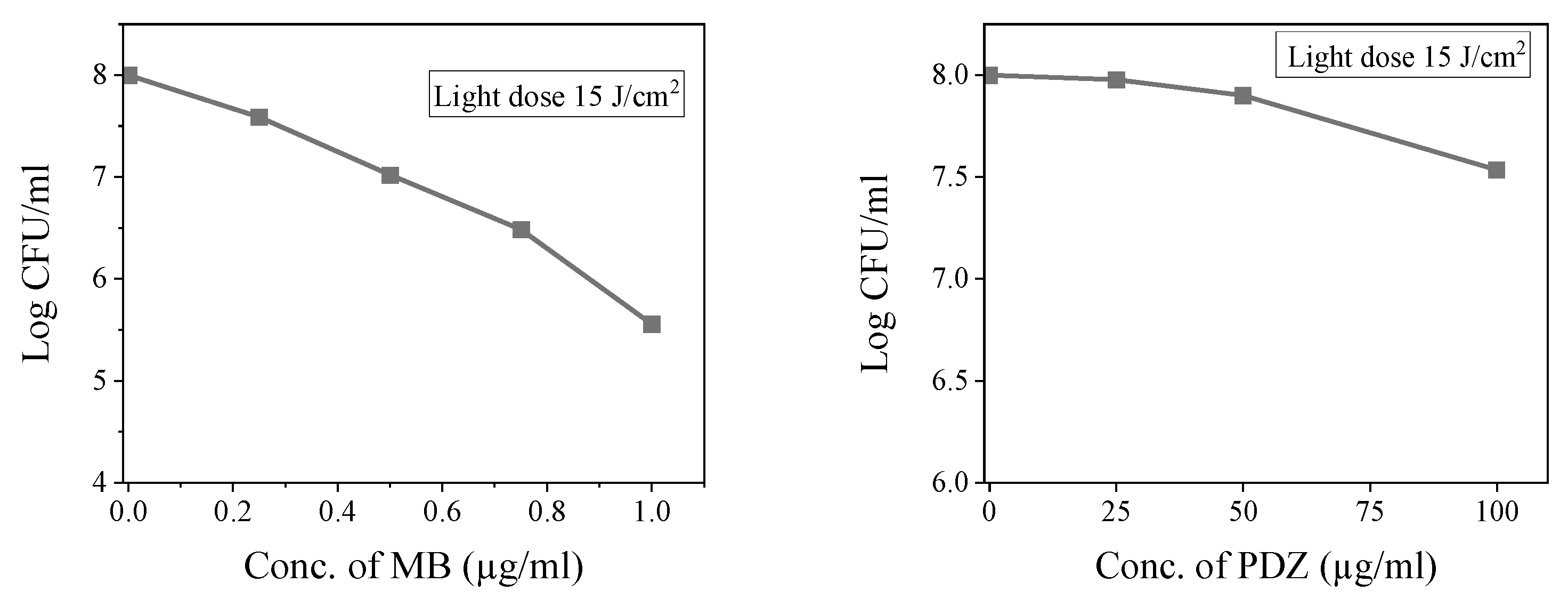
2.1. Effect of PDT Alone on Inhibition of Klebsiella pneumoniae
2.2. Minimum Inhibitory Concentrations (MIC) of Antibiotics Against K. pneumoniae
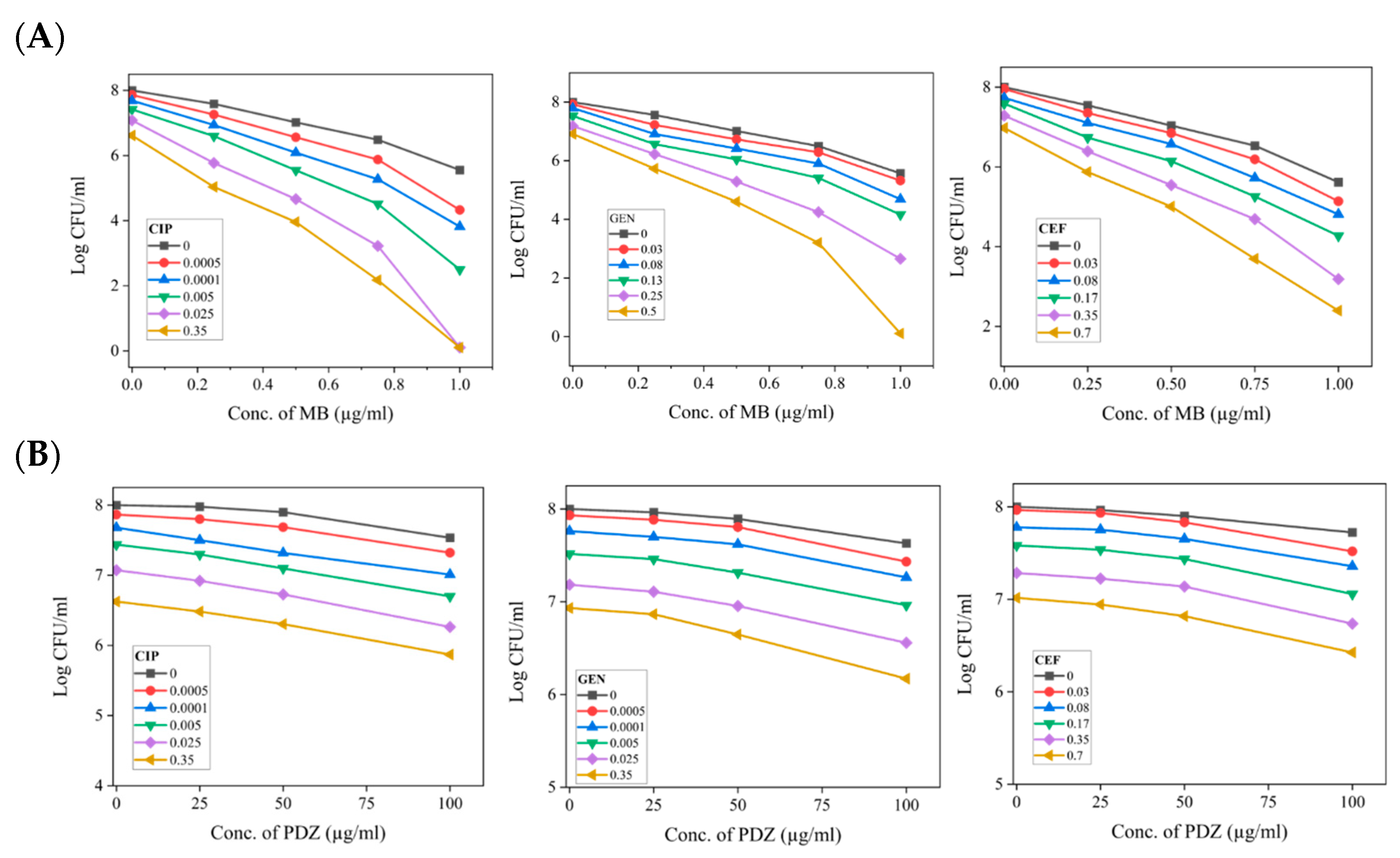
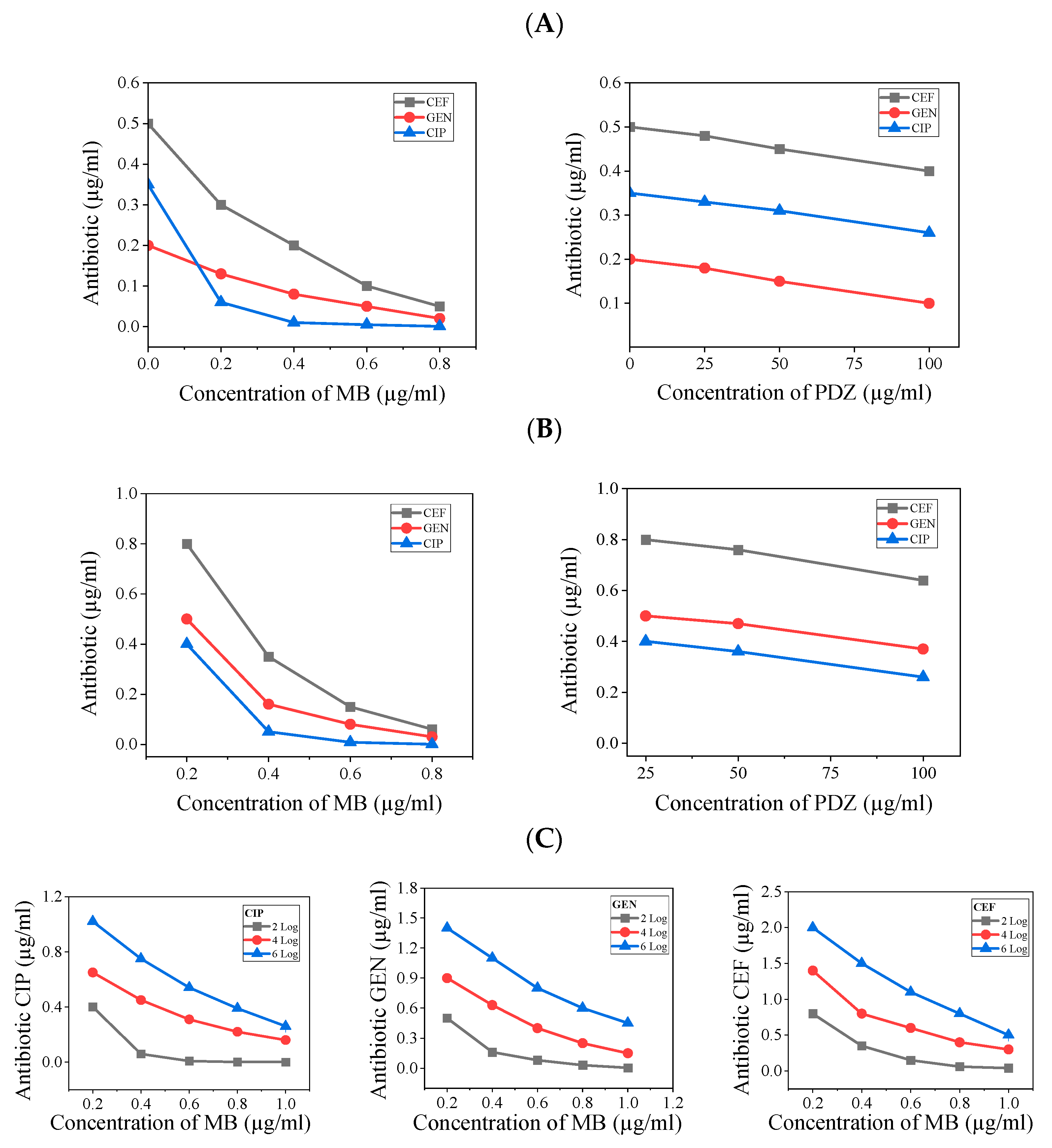
2.3. Effect of the PDT and Antibiotics Combination on K. pneumoniae
3. Discussion
3.1. PDT/Bacterial Nucleic Acid Synthesis Inhibiting Antibiotic Combinations
3.2. PDT/Bacterial Protein Synthesis Inhibiting Antibiotic Combinations
3.3. PDT/Bacterial Cell Wall Synthesis Inhibiting Antibiotic Combinations
4. Materials and Methods
4.1. Growth Media
4.2. Bacterial Strain and Culture Conditions
4.3. Antibiotic Preparation and Treatment
4.4. Photosensitizer and Light Source
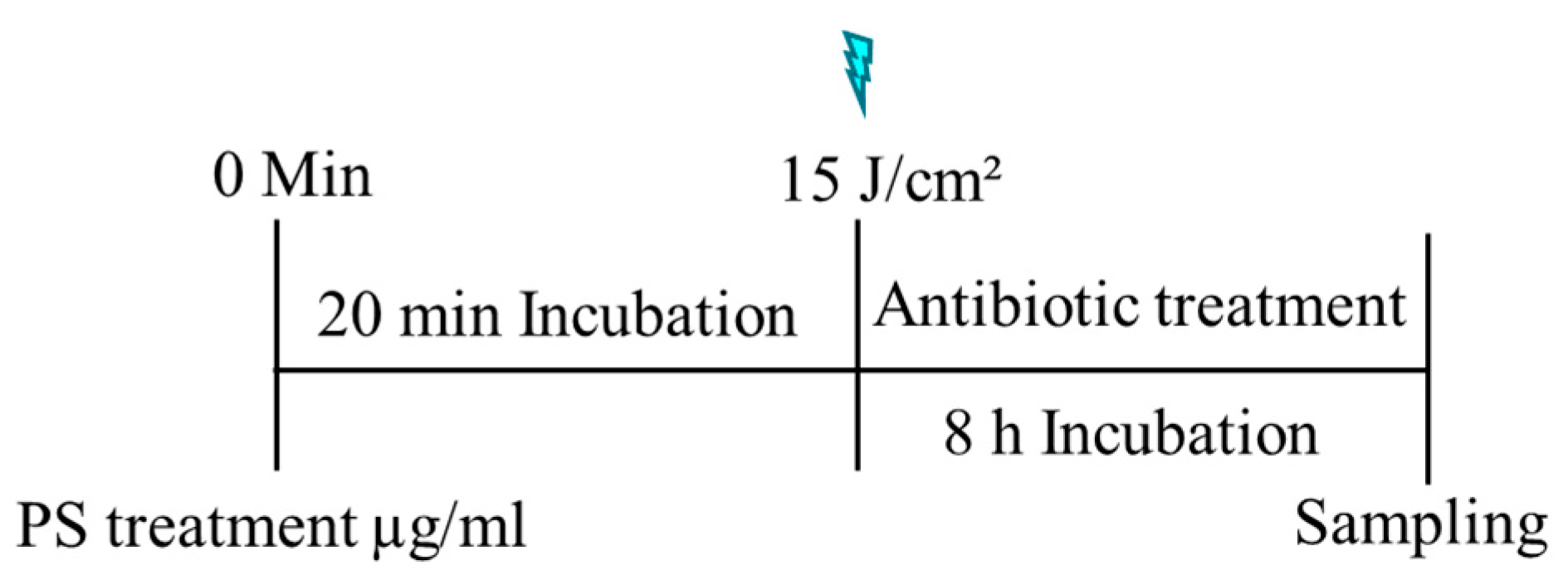
4.5. Photodynamic Inactivation Procedure
4.6. Determination of Minimum Inhibitory Concentration (MICs) of Antibiotics
4.7. Antibacterial Activity Assay
4.8. Combination Effect of Photosensitizers and Antibiotics
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chawla, K.; Piveteau, P.; Sharma, S. Klebsiella pneumoniae: A connecting link in the One Health concept. Pathog. Glob. Health 2025, 15, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Blaskovich, M.A.T.; Cooper, M.A. Antibiotics re-booted-time to kick back against drug resistance. NPJ Antimicrob. Resist. 2025, 3, 47. [Google Scholar] [CrossRef] [PubMed]
- Bravo, A.R.; Fuentealba, F.A.; González, I.A.; Palavecino, C.E. Use of Antimicrobial Photodynamic Therapy to Inactivate Multidrug-Resistant Klebsiella pneumoniae: Scoping Review. Pharmaceutics 2024, 16, 1626. [Google Scholar] [CrossRef] [PubMed]
- Dube, E. Antimicrobial Photodynamic Therapy: Self-Disinfecting Surfaces for Controlling Microbial Infections. Microorganisms 2024, 12, 1573. [Google Scholar] [CrossRef]
- Dong, J.; Tang, J.; Li, X.; Zeng, Y.; Su, X.; He, Y.; Liu, X. Latest developments in photosensitizers: Improving stability, specificity and responsiveness. Future Med. Chem. 2025, 17, 1297–1314. [Google Scholar] [CrossRef]
- Aebisher, D.; Szpara, J.; Bartusik-Aebisher, D. Advances in Medicine: Photodynamic Therapy. Int. J. Mol. Sci. 2024, 25, 8258. [Google Scholar] [CrossRef]
- Velkov, T.; Thompson, P.E.; Azad, M.A.K.; Roberts, K.D.; Bergen, P.J. History, Chemistry and Antibacterial Spectrum. Adv. Exp. Med. Biol. 2019, 1145, 15–36. [Google Scholar]
- Cardozo, A.P.M.; da Silva, D.F.T.; Fernandes, K.P.S.; Ferreira, R.C.; Lino-Dos-Santos-Franco, A.; Rodrigues, M.F.S.D.; Motta, L.J.; Cecatto, R.B. Antimicrobial photodynamic therapy with methylene blue and its derivatives in animal studies: Systematic review. Photodermatol. Photoimmunol. Photomed. 2024, 40, e12978. [Google Scholar] [CrossRef]
- Almeida, A.; Faustino, M.A.; Tomé, J.P. Photodynamic inactivation of bacteria: Finding the effective targets. Future Med. Chem. 2015, 7, 1221–1224. [Google Scholar] [CrossRef]
- Yu, H.; Huang, Z.; Wu, J.; Zhao, Z.; Hua, Y.; Yang, Y. Chlorin e6: A promising photosensitizer of anti-tumor and anti-inflammatory effects in PDT. Nanomedicine 2025, 20, 389–400. [Google Scholar] [CrossRef]
- Spesia, M.B.; Durantini, E.N. Photosensitizers combination approach to enhance photodynamic inactivation of planktonic and biofilm bacteria. Photochem. Photobiol. Sci. 2023, 22, 2433–2444. [Google Scholar] [CrossRef]
- Aroso, R.T.; Schaberle, F.A.; Arnaut, L.G.; Pereira, M.M. Photodynamic disinfection and its role in controlling infectious diseases. Photochem. Photobiol. Sci. 2021, 20, 1497–1545. [Google Scholar] [CrossRef]
- Omrani, M.A.; Tsobo, C.T.; Chalabianloo, N.; Ahmadi, F.; Abdullah, S.S.; Muanda, F.T. Fluoroquinolones and risk of nightmares: A literature review and disproportionality analysis using individual case safety reports from Food and Drug Administration Adverse Event Reporting System database. J. Psychopharmacol. 2025, 39, 2698811251344684. [Google Scholar] [CrossRef]
- Shih, M.H.; Huang, F.C. Effects of Photodynamic Therapy on Rapidly Growing Nontuberculous Mycobacteria keratitis. Invest. Ophthalmol. Vis. Sci. 2011, 52, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Beuerman, R.W.; Verma, C.S. Molecular insights into the membrane affinities of model hydrophobes. ACS Omega 2018, 3, 2498–2507. [Google Scholar] [CrossRef] [PubMed]
- Agris, P.F. Targeting Gene Transcription Prevents Antibiotic Resistance. Antibiotics 2025, 14, 345. [Google Scholar] [CrossRef] [PubMed]
- Barra, F.; Roscetto, E.; Soriano, A.A.; Vollaro, A.; Postiglione, I.; Pierantoni, G.M.; Palumbo, G.; Cataniaet, M.R. Photodynamic and antibiotic therapy in combination to fight biofilms and resistant surface bacterial infections. Int. J. Mol. Sci. 2015, 16, 20417–20430. [Google Scholar] [CrossRef]
- Reznick, Y.; Banin, E.; Lipovsky, A.; Lubart, R.; Polak, P.; Zalevsky, Z. The synergistic effect of visible light and gentamycin on Pseudomona aeruginosa microorganisms. J. Vis. Exp. 2013, 2, e4370. [Google Scholar]
- Pérez-Laguna, V.; García-Luque, I.; Ballesta, S.; Pérez-Artiaga, L.; Lampaya-Pérez, V.; Rezusta, A.; Gilaberte, Y. Photodynamic therapy using methylene blue, combined or not with gentamicin, against Staphylococcus aureus and Pseudomonas aeruginosa. Photodiagn. Photodyn. Ther. 2020, 31, 101810. [Google Scholar] [CrossRef]
- Karisik, E.; Stanojevic-Ristic, Z.; Jevtic, M.; Rasic, J.; Maric, M.; Popovic, M. Ceftriaxone-Induced Pancytopenia: A Case Report. Hematol. Rep. 2025, 17, 30. [Google Scholar] [CrossRef]
- Feng, Y.; Tonon, C.C.; Ashraf, S.; Hasan, T. Photodynamic and antibiotic therapy in combination against bacterial infections: Efficacy, determinants, mechanisms, and future perspectives. Adv. Drug Deliv. Rev. 2021, 177, 113941. [Google Scholar] [CrossRef]
- Ronqui, M.R.; Coletti, T.M.S.F.A.; de Freitas, L.M.; Miranda, E.T.; Fontana, C.R. Synergistic antimicrobial effect of photodynamic therapy and ciprofloxacin. J. Photochem. Photobiol. B 2016, 158, 122–129. [Google Scholar] [CrossRef]
- Oppezzo, O.J.; Forte Giacobone, A.F. Lethal Effect of Photodynamic Treatment on Persister Bacteria. Photochem. Photobiol. 2018, 94, 186–189. [Google Scholar] [CrossRef]
- Ghorbani, J.; Rahban, D.; Aghamiri, S.; Teymouri, A.; Bahador, A. Photosensitizers in antibacterial photodynamic therapy: An overview. Laser Ther. 2018, 27, 293–302. [Google Scholar] [CrossRef]
- Zangirolami, A.C.; Yerra, K.R.; Yakovlev, V.V.; Blanco, K.C.; Bagnato, V.S. Combined Antibiotic and Photodynamic Therapies in Pseudomonas aeruginosa: From Synergy to Antagonism. Antibiotics 2024, 13, 1111. [Google Scholar] [CrossRef]
- Willis, J.A.; Cheburkanov, V.; Chen, S.; Soares, J.M.; Kassab, G.; Blanco, K.C.; Bagnato, V.S.; de Figueiredo, P.; Yakovlev, V.V. Breaking down antibiotic resistance in methicillin-resistant Staphylococcus aureus: Combining antimicrobial photodynamic and antibiotic treatments. Proc. Natl. Acad. Sci. USA 2022, 119, e2208378119. [Google Scholar] [CrossRef]


| Bacteria | Antibiotic | MIC (µg/mL) |
|---|---|---|
| K. pneumoniae | Ciprofloxacin | 0.1875 |
| Gentamicin | 1.5 | |
| Ceftriaxone | 2.125 |
| Antibiotic | Concentrations Used (µg/mL) |
|---|---|
| Ciprofloxacin (CIP) | 0.00005, 0.0001, 0.005, 0.025, 0.35 |
| Gentamicin (GEN) | 0.03, 0.08, 0.13, 0.25, 0.5 |
| Ceftriaxone (CEF) | 0.03, 0.08, 0.17, 0.35, 0.7 |
| Photosensitizer | Concentrations Used Alone (µg/mL) | Concentrations Used in Combination with Antibiotics (µg/mL) |
|---|---|---|
| Methylene blue (MB) | 0.25, 0.5, 0.75, 1.0 | 0.2, 0.4, 0.6, 0.8 |
| Photodithazine (PDZ) | 25, 50, 100 | 25, 50, 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yerra, K.R.; Bagnato, V.S. Enhancing Antibiotic Effect by Photodynamic: The Case of Klebsiella pneumoniae. Antibiotics 2025, 14, 766. https://doi.org/10.3390/antibiotics14080766
Yerra KR, Bagnato VS. Enhancing Antibiotic Effect by Photodynamic: The Case of Klebsiella pneumoniae. Antibiotics. 2025; 14(8):766. https://doi.org/10.3390/antibiotics14080766
Chicago/Turabian StyleYerra, Koteswara Rao, and Vanderlei S. Bagnato. 2025. "Enhancing Antibiotic Effect by Photodynamic: The Case of Klebsiella pneumoniae" Antibiotics 14, no. 8: 766. https://doi.org/10.3390/antibiotics14080766
APA StyleYerra, K. R., & Bagnato, V. S. (2025). Enhancing Antibiotic Effect by Photodynamic: The Case of Klebsiella pneumoniae. Antibiotics, 14(8), 766. https://doi.org/10.3390/antibiotics14080766





