Antimicrobial Action Mechanisms of Natural Compounds Isolated from Endophytic Microorganisms
Abstract
1. Introduction
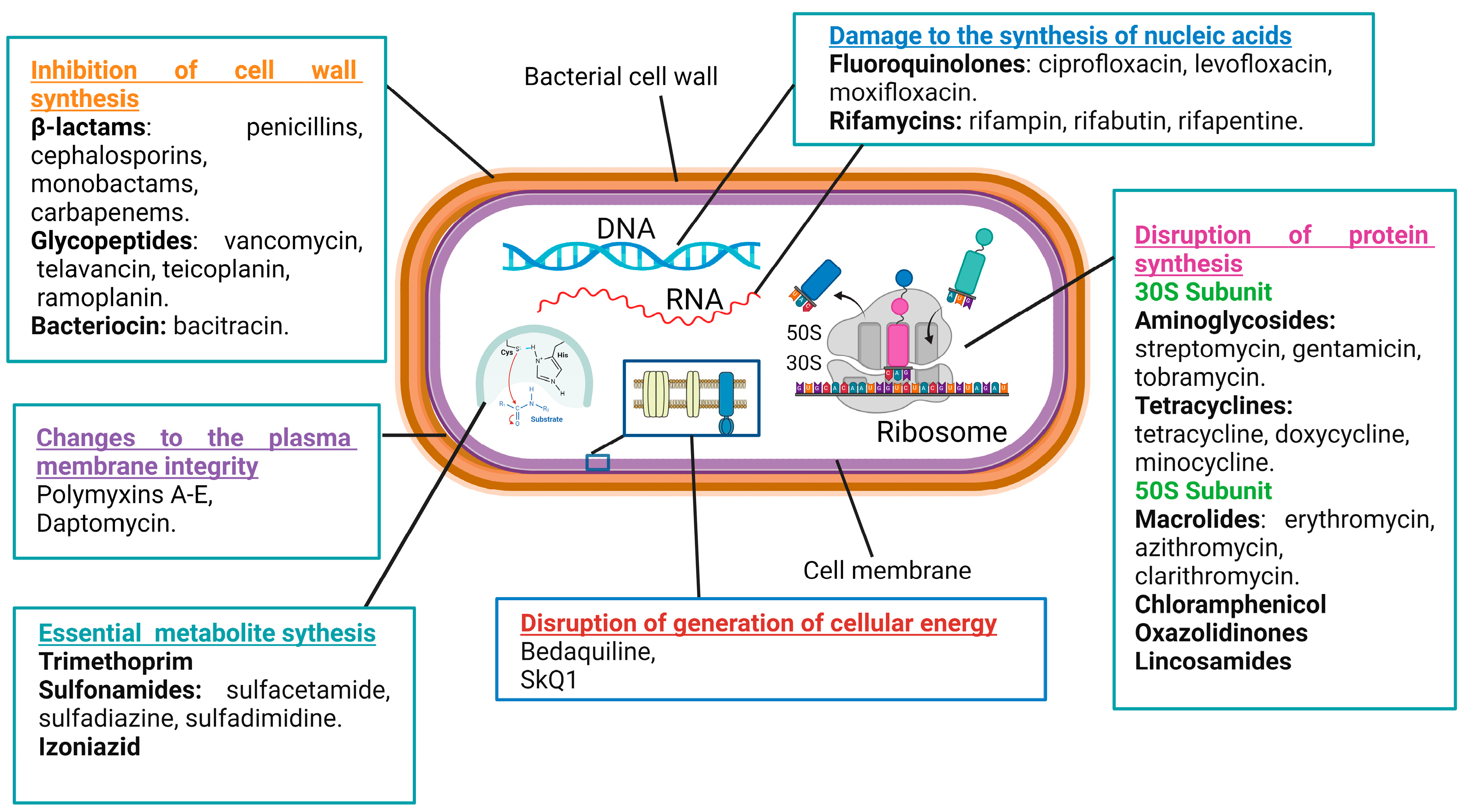
2. Mechanisms of Action of Antimicrobial Drugs
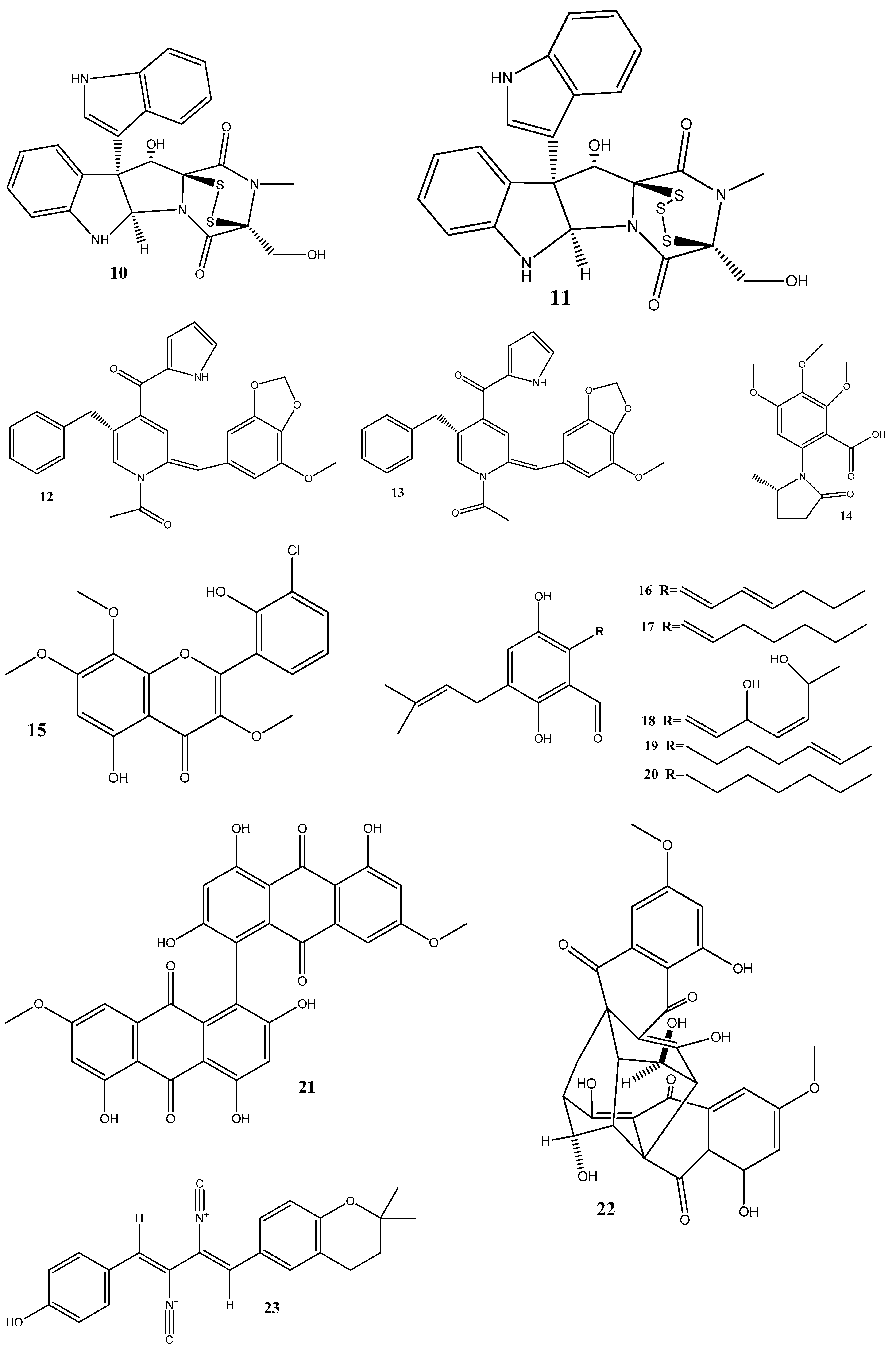
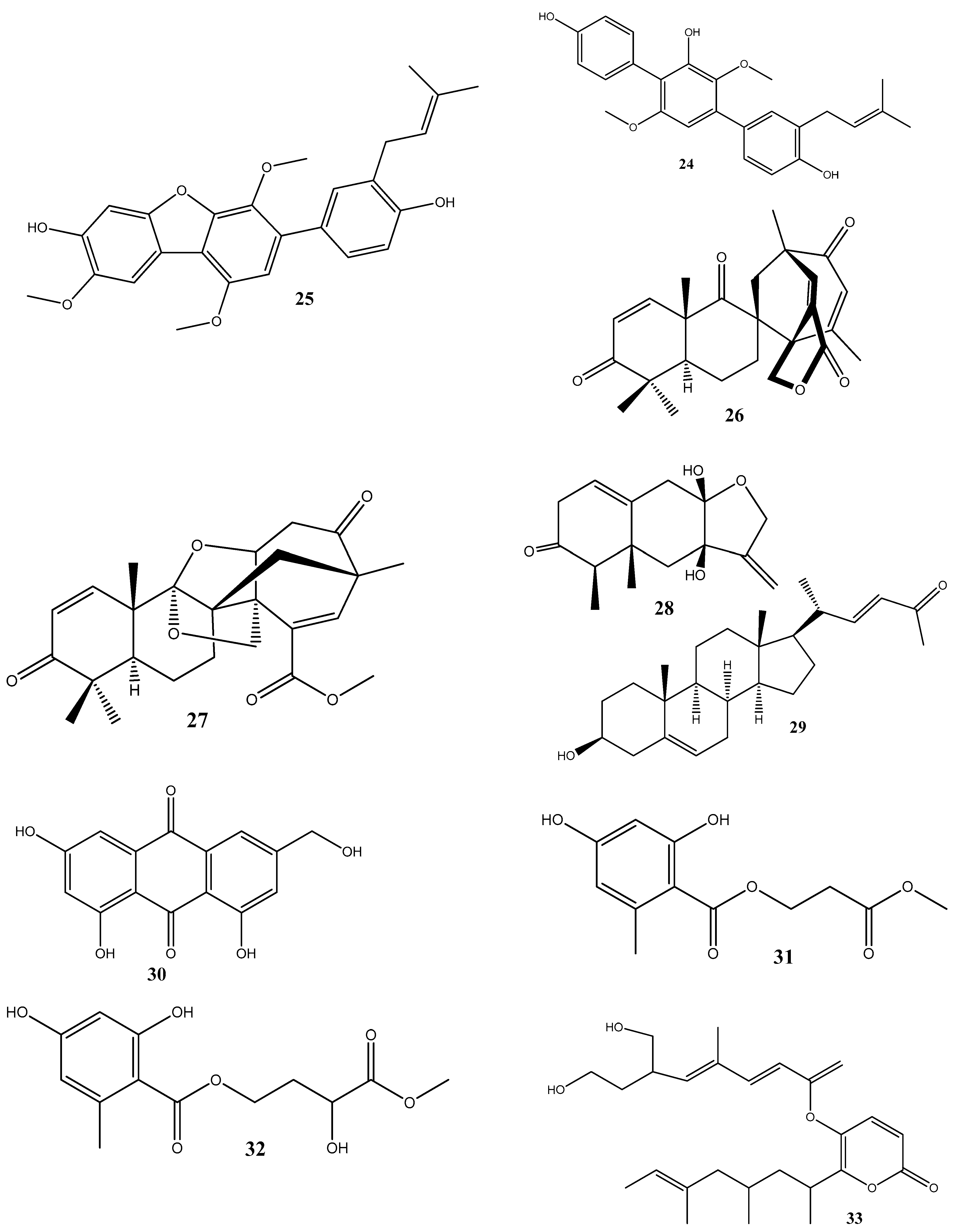
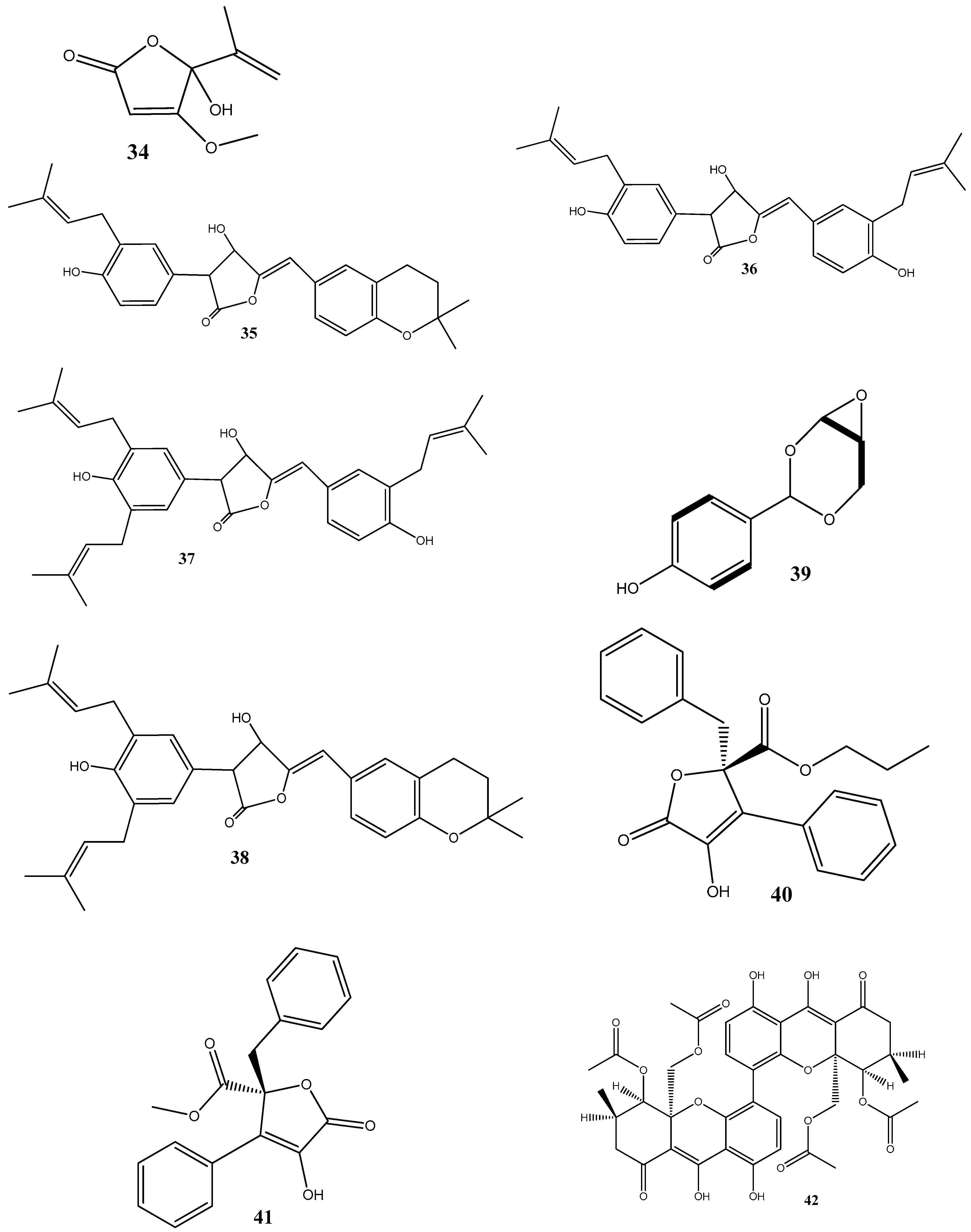
3. Antimicrobial Effects and Mechanisms of Action of Natural Compounds Isolated from Endophytes
4. Effects of Natural Compounds Obtained from Endophytes on Antibiotic-Resistant Bacteria
5. Biosynthetic Gene Clusters of Secondary Metabolites of Endophytic Fungi
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Baker, R.E.; Mahmud, A.S.; Miller, I.F.; Rajeev, M.; Rasambainarivo, F.; Rice, B.L.; Takahashi, S.; Tatem, A.J.; Wagner, C.E.; Wang, L.-F.; et al. Infectious Disease in an Era of Global Change. Nat. Rev. Microbiol. 2022, 20, 193–205. [Google Scholar] [CrossRef]
- Bloom, D.E.; Cadarette, D. Infectious Disease Threats in the Twenty-First Century: Strengthening the Global Response. Front. Immunol. 2019, 10, 549. [Google Scholar] [CrossRef]
- Zhang, X.-X.; Jin, Y.-Z.; Lu, Y.-H.; Huang, L.-L.; Wu, C.-X.; Lv, S.; Chen, Z.; Xiang, H.; Zhou, X.-N. Infectious Disease Control: From Health Security Strengthening to Health Systems Improvement at Global Level. Glob. Health Res. Policy 2023, 8, 38. [Google Scholar] [CrossRef]
- Murray, C.J.L.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Robles Aguilar, G.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Oneill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. In London: Review on Antimicrobial Resistance; Government of the United Kingdom: London, UK, 2016. [Google Scholar]
- Baltz, R.H. Antimicrobials from Actinomycetes: Back to the Future. Microbe 2007, 2, 125–131. [Google Scholar]
- Baltz, R.H. Renaissance in Antibacterial Discovery from Actinomycetes. Curr. Opin. Pharmacol. 2008, 8, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Lavore, F.; Maity, S.; Derks, M.G.N.; Jones, C.R.; Vermeulen, B.J.A.; Melcrová, A.; Morris, M.A.; Becker, L.M.; Wang, X.; et al. Teixobactin Kills Bacteria by a Two-Pronged Attack on the Cell Envelope. Nature 2022, 608, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Stokes, J.M.; Yang, K.; Swanson, K.; Jin, W.; Cubillos-Ruiz, A.; Donghia, N.M.; MacNair, C.R.; French, S.; Carfrae, L.A.; Bloom-Ackermann, Z.; et al. A Deep Learning Approach to Antibiotic Discovery. Cell 2020, 180, 688–702.e13. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Meyer, K.J.; Iinishi, A.; Favre-Godal, Q.; Green, R.; Manuse, S.; Caboni, M.; Mori, M.; Niles, S.; Ghiglieri, M.; et al. A New Antibiotic Selectively Kills Gram-Negative Pathogens. Nature 2019, 576, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Terekhov, S.S.; Smirnov, I.V.; Malakhova, M.V.; Samoilov, A.E.; Manolov, A.I.; Nazarov, A.S.; Danilov, D.V.; Dubiley, S.A.; Osterman, I.A.; Rubtsova, M.P.; et al. Ultrahigh-Throughput Functional Profiling of Microbiota Communities. Proc. Natl. Acad. Sci. USA 2018, 115, 9551–9556. [Google Scholar] [CrossRef] [PubMed]
- Gouda, S.; Das, G.; Sen, S.K.; Shin, H.-S.; Patra, J.K. Endophytes: A Treasure House of Bioactive Compounds of Medicinal Importance. Front. Microbiol. 2016, 7, 1538. [Google Scholar] [CrossRef]
- Tiwari, P.; Bae, H. Endophytic Fungi: Key Insights, Emerging Prospects, and Challenges in Natural Product Drug Discovery. Microorganisms 2022, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Kumar, A. Secondary Metabolites from Endophytic Fungi: Production, Methods of Analysis, and Diverse Pharmaceutical Potential. Symbiosis 2023, 90, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Meshram, V.; Gupta, M.; Goyal, S.; Qureshi, K.A.; Jaremko, M.; Shukla, K.K. Fungal Endophytes: Microfactories of Novel Bioactive Compounds with Therapeutic Interventions; A Comprehensive Review on the Biotechnological Developments in the Field of Fungal Endophytic Biology over the Last Decade. Biomolecules 2023, 13, 1038. [Google Scholar] [CrossRef] [PubMed]
- Eshboev, F.; Egamberdieva, D. Medicinal Plant Associated Endophytic Fungi: Metabolites and Bioactivity; Endophytes, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2024. [Google Scholar]
- Chow, C.; Padda, K.P.; Puri, A.; Chanway, C.P. An Archaic Approach to a Modern Issue: Endophytic Archaea for Sustainable Agriculture. Curr. Microbiol. 2022, 79, 322. [Google Scholar] [CrossRef] [PubMed]
- Kandel, S.; Joubert, P.; Doty, S. Bacterial Endophyte Colonization and Distribution within Plants. Microorganisms 2017, 5, 77. [Google Scholar] [CrossRef] [PubMed]
- Rutkowska, N.; Drożdżyński, P.; Ryngajłło, M.; Marchut-Mikołajczyk, O. Plants as the Extended Phenotype of Endophytes—The Actual Source of Bioactive Compounds. Int. J. Mol. Sci. 2023, 24, 10096. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.; Kaur, T.; Chhabra, I.; Panja, A.; Paul, S.; Kumar, V.; Malik, T. Endophytic Fungi: Hidden Treasure Chest of Antimicrobial Metabolites Interrelationship of Endophytes and Metabolites. Front. Microbiol. 2023, 14, 1227830. [Google Scholar] [CrossRef]
- Digra, S.; Nonzom, S. An Insight into Endophytic Antimicrobial Compounds: An Updated Analysis. Plant Biotechnol. Rep. 2023, 17, 427–457. [Google Scholar] [CrossRef]
- Damavandi, M.S.; Shojaei, H.; Esfahani, B.N. The Anticancer and Antibacterial Potential of Bioactive Secondary Metabolites Derived from Bacterial Endophytes in Association with Artemisia Absinthium. Sci. Rep. 2023, 13, 18473. [Google Scholar] [CrossRef]
- Sharma, M.; Bharti, S.; Goswami, A.; Mallubhotla, S. Diversity, Antimicrobial, Antioxidant, and Anticancer Activity of Culturable Fungal Endophyte Communities in Cordia Dichotoma. Molecules 2023, 28, 6926. [Google Scholar] [CrossRef]
- Yadav, G.; Meena, M. Bioprospecting of Endophytes in Medicinal Plants of Thar Desert: An Attractive Resource for Biopharmaceuticals. Biotechnol. Rep. 2021, 30, e00629. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.; Mamadalieva, N.Z.; Ali, I.; Elizbit; Green, I.R.; Wang, D.; Zou, L.; Simal-Gandara, J.; Cao, H.; Xiao, J. Fungal Glycosides: Structure and Biological Function. Trends Food Sci. Technol. 2021, 110, 611–651. [Google Scholar] [CrossRef]
- Hussain, H.; Elizbit; Ali, I.; Mamadalieva, N.Z.; Abbas, G.; Ali, M.; Zaman, G.; Khan, A.; Hassan, U.; Green, I.R. Fruitful Decade of Phoma Secondary Metabolites from 2011 to 2020: Chemistry, Chemical Diversity, and Biological Activities. In Phoma: Diversity, Taxonomy, Bioactivities, and Nanotechnology; Springer International Publishing: Cham, Switzerland, 2022; pp. 183–203. ISBN 9783030812171. [Google Scholar]
- Deshmukh, S.K.; Dufossé, L.; Chhipa, H.; Saxena, S.; Mahajan, G.B.; Gupta, M.K. Fungal Endophytes: A Potential Source of Antibacterial Compounds. J. Fungi 2022, 8, 164. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Singh, D.K.; Kharwar, R.N.; White, J.F.; Gond, S.K. Fungal Endophytes as Efficient Sources of Plant-Derived Bioactive Compounds and Their Prospective Applications in Natural Product Drug Discovery: Insights, Avenues, and Challenges. Microorganisms 2021, 9, 197. [Google Scholar] [CrossRef] [PubMed]
- Kousar, R.; Naeem, M.; Jamaludin, M.I.; Arshad, A.; Shamsuri, A.N.; Ansari, N.; Akhtar, S.; Hazafa, A.; Uddin, J.; Khan, A.; et al. Exploring the Anticancer Activities of Novel Bioactive Compounds Derived from Endophytic Fungi: Mechanisms of Action, Current Challenges and Future Perspectives. Am. J. Cancer Res. 2022, 12, 2897–2919. [Google Scholar] [PubMed]
- Hussain, H.; Ali, I.; Elizbit; Hussain, W.; Mamadalieva, N.Z.; Hussain, A.; Ali, M.; Ahmed, I.; Ullah, I.; Green, I.R. Synthetic Studies towards Fungal Glycosides: An Overview. Curr. Org. Chem. 2020, 24, 2865–2901. [Google Scholar] [CrossRef]
- Hur, J.Y.; Jeong, E.; Kim, Y.C.; Lee, S.R. Strategies for Natural Product Discovery by Unlocking Cryptic Biosynthetic Gene Clusters in Fungi. Separations 2023, 10, 333. [Google Scholar] [CrossRef]
- Albarano, L.; Esposito, R.; Ruocco, N.; Costantini, M. Genome Mining as New Challenge in Natural Products Discovery. Mar. Drugs 2020, 18, 199. [Google Scholar] [CrossRef]
- Grijseels, S.; Pohl, C.; Nielsen, J.C.; Wasil, Z.; Nygård, Y.; Nielsen, J.; Frisvad, J.C.; Nielsen, K.F.; Workman, M.; Larsen, T.O.; et al. Identification of the Decumbenone Biosynthetic Gene Cluster in Penicillium Decumbens and the Importance for Production of Calbistrin. Fungal Biol. Biotechnol. 2018, 5, 18. [Google Scholar] [CrossRef]
- Nah, H.-J.; Pyeon, H.-R.; Kang, S.-H.; Choi, S.-S.; Kim, E.-S. Cloning and Heterologous Expression of a Large-Sized Natural Product Biosynthetic Gene Cluster in Streptomyces Species. Front. Microbiol. 2017, 8, 394. [Google Scholar] [CrossRef] [PubMed]
- Sulheim, S.; Fossheim, F.A.; Wentzel, A.; Almaas, E. Automatic Reconstruction of Metabolic Pathways from Identified Biosynthetic Gene Clusters. BMC Bioinform. 2021, 22, 81. [Google Scholar] [CrossRef]
- Singh, S.P.; Qureshi, A.; Hassan, W. Mechanisms of Action by Antimicrobial Agents: A Review. Mcgill J. Med. 2021, 1–10. [Google Scholar] [CrossRef]
- Pancu, D.F.; Scurtu, A.; Macasoi, I.G.; Marti, D.; Mioc, M.; Soica, C.; Coricovac, D.; Horhat, D.; Poenaru, M.; Dehelean, C. Antibiotics: Conventional Therapy and Natural Compounds with Antibacterial Activity—A Pharmaco-Toxicological Screening. Antibiotics 2021, 10, 401. [Google Scholar] [CrossRef] [PubMed]
- Hards, K.; Cook, G.M. Targeting Bacterial Energetics to Produce New Antimicrobials. Drug Resist. Updat. 2018, 36, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, P.; Yarlagadda, V.; Ghosh, C.; Haldar, J. A Review on Cell Wall Synthesis Inhibitors with an Emphasis on Glycopeptide Antibiotics. Medchemcomm 2017, 8, 516–533. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, M.K. Antibiotics That Inhibit Cell Wall Synthesis. In Chemistry of Antibiotics and Related Drugs; Springer International Publishing: Cham, Switzerland, 2016; pp. 49–94. ISBN 9783319407449. [Google Scholar]
- Nguyen, R.; Khanna, N.R.; Safadi, A.O. Bacitracin Topical; StatPearls: Treasure Island, FL, USA, 2023. [Google Scholar]
- Nourbakhsh, F.; Lotfalizadeh, M.; Badpeyma, M.; Shakeri, A.; Soheili, V. From Plants to Antimicrobials: Natural Products against Bacterial Membranes. Phytother. Res. 2022, 36, 33–52. [Google Scholar] [CrossRef]
- Epand, R.M.; Walker, C.; Epand, R.F.; Magarvey, N.A. Molecular Mechanisms of Membrane Targeting Antibiotics. Biochim. Biophys. Acta Biomembr. 2016, 1858, 980–987. [Google Scholar] [CrossRef]
- Maddison, J.E.; Watson, A.D.J.; Elliott, J. Antibacterial Drugs. In Small Animal Clinical Pharmacology; Elsevier: Amsterdam, The Netherlands, 2008; pp. 148–185. [Google Scholar]
- Parker, N.; Schneegurt, M.; Tu, A.-H.T.; Lister, P.; Forster, B.M. Microbiology; OpenStax: Houston, TX, USA, 2016. [Google Scholar]
- Nazarov, P.A.; Osterman, I.A.; Tokarchuk, A.V.; Karakozova, M.V.; Korshunova, G.A.; Lyamzaev, K.G.; Skulachev, M.V.; Kotova, E.A.; Skulachev, V.P.; Antonenko, Y.N. Mitochondria-Targeted Antioxidants as Highly Effective Antibiotics. Sci. Rep. 2017, 7, 1394. [Google Scholar] [CrossRef]
- Cambau, E.; Guillard, T. Antimicrobials That Affect the Synthesis and Conformation of Nucleic Acids. Rev. Sci. Tech. 2012, 31, 77–87. [Google Scholar] [CrossRef]
- Santos, J.A.; Lamers, M.H. Novel Antibiotics Targeting Bacterial Replicative DNA Polymerases. Antibiotics 2020, 9, 776. [Google Scholar] [CrossRef]
- Pham, T.D.M.; Ziora, Z.M.; Blaskovich, M.A.T. Quinolone Antibiotics. Medchemcomm 2019, 10, 1719–1739. [Google Scholar] [CrossRef]
- Tunitskaya, V.L.; Khomutov, A.R.; Kochetkov, S.N.; Kotovskaya, S.K.; Charushin, V.N. Inhibition of DNA Gyrase by Levofloxacin and Related Fluorine-Containing Heterocyclic Compounds. Acta Naturae 2011, 3, 94–99. [Google Scholar] [CrossRef]
- Hooper, D.C.; Jacoby, G.A. Topoisomerase Inhibitors: Fluoroquinolone Mechanisms of Action and Resistance. Cold Spring Harb. Perspect. Med. 2016, 6, a025320. [Google Scholar] [CrossRef] [PubMed]
- Bush, N.G.; Diez-Santos, I.; Abbott, L.R.; Maxwell, A. Quinolones: Mechanism, Lethality and Their Contributions to Antibiotic Resistance. Molecules 2020, 25, 5662. [Google Scholar] [CrossRef] [PubMed]
- Maslow, M.J.; Celhay, C. 27—Rifamycins. Mand. Douglas Bennett’s Princ. Pract. Infect. Diseases 2015, 1, 339–349.e3. [Google Scholar] [CrossRef]
- Rothstein, D.M. Rifamycins, Alone and in Combination. Cold Spring Harb. Perspect. Med. 2016, 6, a027011. [Google Scholar] [CrossRef]
- Mosaei, H.; Zenkin, N. Inhibition of RNA Polymerase by Rifampicin and Rifamycin-like Molecules. EcoSal Plus 2020, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Fief, C.A.; Hoang, K.G.; Phipps, S.D.; Wallace, J.L.; Deweese, J.E. Examining the Impact of Antimicrobial Fluoroquinolones on Human DNA Topoisomerase IIα and IIβ. ACS Omega 2019, 4, 4049–4055. [Google Scholar] [CrossRef] [PubMed]
- Arenz, S.; Wilson, D.N. Bacterial Protein Synthesis as a Target for Antibiotic Inhibition. Cold Spring Harb. Perspect. Med. 2016, 6, a025361. [Google Scholar] [CrossRef]
- McCoy, L.S.; Xie, Y.; Tor, Y. Antibiotics That Target Protein Synthesis. Wiley Interdiscip. Rev. RNA 2011, 2, 209–232. [Google Scholar] [CrossRef]
- Osterman, I.; Bogdanov, A.; Dontsova, O.; Sergiev, P. Techniques for Screening Translation Inhibitors. Antibiotics 2016, 5, 22. [Google Scholar] [CrossRef]
- Krause, K.M.; Serio, A.W.; Kane, T.R.; Connolly, L.E. Aminoglycosides: An Overview. Cold Spring Harb. Perspect. Med. 2016, 6, a027029. [Google Scholar] [CrossRef]
- Becker, B.; Cooper, M.A. Aminoglycoside Antibiotics in the 21st Century. ACS Chem. Biol. 2013, 8, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Chopra, I.; Roberts, M. Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef] [PubMed]
- Ashfaq, M.; Hashmi, M.Z.; Mumtaz, A.; Javed, D.; Ain, N.U.; Shifaqat, S.; Rehman, M.S.U. Environmental Risk Assessment of Antibiotics and AMR/ARGs. Antibiot. Antimicrob. Resist. Genes Environ. 2020, 1, 331–349. [Google Scholar] [CrossRef]
- Myers, A.G.; Clark, R.B. Discovery of Macrolide Antibiotics Effective against Multi-Drug Resistant Gram-Negative Pathogens. Acc. Chem. Res. 2021, 54, 1635–1645. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, T.; Dormoi, J.; Madamet, M.; Pradines, B. Macrolides and Associated Antibiotics Based on Similar Mechanism of Action like Lincosamides in Malaria. Malar. J. 2016, 15, 85. [Google Scholar] [CrossRef] [PubMed]
- Capasso, C.; Supuran, C.T. Sulfa and Trimethoprim-like Drugs—Antimetabolites Acting as Carbonic Anhydrase, Dihydropteroate Synthase and Dihydrofolate Reductase Inhibitors. J. Enzym. Inhib. Med. Chem. 2014, 29, 379–387. [Google Scholar] [CrossRef]
- AlRabiah, H.; Allwood, J.W.; Correa, E.; Xu, Y.; Goodacre, R. pH Plays a Role in the Mode of Action of Trimethoprim on Escherichia Coli. PLoS ONE 2018, 13, e0200272. [Google Scholar] [CrossRef]
- Drozdowska, D. Trimethoprim and Its Derivatives. In Comprehensive Pharmacology; Elsevier: Amsterdam, The Netherlands, 2022; pp. 271–294. [Google Scholar]
- Ovung, A.; Bhattacharyya, J. Sulfonamide Drugs: Structure, Antibacterial Property, Toxicity, and Biophysical Interactions. Biophys. Rev. 2021, 13, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Zessel, K.; Mohring, S.; Hamscher, G.; Kietzmann, M.; Stahl, J. Biocompatibility and Antibacterial Activity of Photolytic Products of Sulfonamides. Chemosphere 2014, 100, 167–174. [Google Scholar] [CrossRef]
- Lin, L.-B.; Gao, Y.-Q.; Han, R.; Xiao, J.; Wang, Y.-M.; Zhang, Q.; Zhai, Y.-J.; Han, W.-B.; Li, W.-L.; Gao, J.-M. Alkylated Salicylaldehydes and Prenylated Indole Alkaloids from the Endolichenic Fungus Aspergillus chevalieri and Their Bioactivities. J. Agric. Food Chem. 2021, 69, 6524–6534. [Google Scholar] [CrossRef] [PubMed]
- Pratiwi, R.H.; Hidayat, I.; Hanafi, M.; Mangunwardoyo, W. Antibacterial Compound Produced by Pseudomonas Aeruginosa Strain UICC B-40, an Endophytic Bacterium Isolated from Neesia Altissima. J. Microbiol. 2017, 55, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Hou, S.-Y.; Zhang, T.-Y.; Wu, Y.-Y.; Zhang, M.-Y.; Yan, X.-M.; Xia, M.-Y.; Zhang, Y.-X. Cytotoxic and Antimicrobial Indole Alkaloids from an Endophytic Fungus Chaetomium sp. SYP-F7950 of Panax notoginseng. RSC Adv. 2019, 9, 28754–28763. [Google Scholar] [CrossRef] [PubMed]
- Arora, P.; Wani, Z.A.; Nalli, Y.; Ali, A.; Riyaz-Ul-Hassan, S. Antimicrobial Potential of Thiodiketopiperazine Derivatives Produced by Phoma sp., an Endophyte of Glycyrrhiza glabra Linn. Microb. Ecol. 2016, 72, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Wu, Y.-Y.; Zhang, T.-Y.; Zhang, M.-Y.; Peng, F.; Lin, B.; Zhang, Y.-X. New Antimicrobial Compounds Produced by Endophytic Penicillium janthinellum Isolated from Panax notoginseng as Potential Inhibitors of FtsZ. Fitoterapia 2018, 131, 35–43. [Google Scholar] [CrossRef]
- Rehberg, N.; Akone, H.S.; Ioerger, T.R.; Erlenkamp, G.; Daletos, G.; Gohlke, H.; Proksch, P.; Kalscheuer, R. Chlorflavonin Targets Acetohydroxyacid Synthase Catalytic Subunit IlvB1 for Synergistic Killing of Mycobacterium Tuberculosis. ACS Infect. Dis. 2018, 4, 123–134. [Google Scholar] [CrossRef]
- Fathallah, N.; Raafat, M.M.; Issa, M.Y.; Abdel-Aziz, M.M.; Bishr, M.; Abdelkawy, M.A.; Salama, O. Bio-Guided Fractionation of Prenylated Benzaldehyde Derivatives as Potent Antimicrobial and Antibiofilm from Ammi Majus L. Fruits-Associated Aspergillus amstelodami. Molecules 2019, 24, 4118. [Google Scholar] [CrossRef]
- Oktavia, L.; Krishna, V.S.; Rekha, E.M.; Fathoni, A.; Sriram, D.; Agusta, A. Anti-Mycobacterial Activity of Two Natural Bisanthraquinones:(+)-1, 10-Bislunatin and (+)-2, 20-Epicytoskyrin A. In IOP Conference Series: Earth and Environmental Science; IOP Publ. Ltd.: Tokyo, Japan, 2020. [Google Scholar]
- Zhang, W.; Wei, W.; Shi, J.; Chen, C.; Zhao, G.; Jiao, R.; Tan, R. Natural Phenolic Metabolites from Endophytic Aspergillus sp. IFB-YXS Antimicrob Activity. Bioorg. Med. Chem. Lett. 2015, 25, 2698–2701. [Google Scholar] [CrossRef]
- He, Y.; Hu, Z.; Sun, W.; Li, Q.; Li, X.-N.; Zhu, H.; Huang, J.; Liu, J.; Wang, J.; Xue, Y.; et al. Spiroaspertrione A, a Bridged Spirocyclic Meroterpenoid, as a Potent Potentiator of Oxacillin against Methicillin-Resistant Staphylococcus aureus from Aspergillus sp. TJ23. J. Org. Chem. 2017, 82, 3125–3131. [Google Scholar] [CrossRef]
- Qiao, Y.; Zhang, X.; He, Y.; Sun, W.; Feng, W.; Liu, J.; Hu, Z.; Xu, Q.; Zhu, H.; Zhang, J.; et al. Aspermerodione, a Novel Fungal Metabolite with an Unusual 2,6-Dioxabicyclo[2.2.1]Heptane Skeleton, as an Inhibitor of Penicillin-Binding Protein 2a. Sci. Rep. 2018, 8, 5454. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.-B.; Xiao, J.; Zhang, Q.; Han, R.; Xu, B.; Yang, S.-X.; Han, W.-B.; Tang, J.-J.; Gao, J.-M. Eremophilane Sesquiterpenoids with Antibacterial and Anti-Inflammatory Activities from the Endophytic Fungus Septoria rudbeckiae. J. Agric. Food Chem. 2021, 69, 11878–11889. [Google Scholar] [CrossRef]
- Graf, T.N.; Kao, D.; Rivera-Chávez, J.; Gallagher, J.M.; Raja, H.A.; Oberlies, N.H. Drug Leads from Endophytic Fungi: Lessons Learned via Scaled Production. Planta Med. 2020, 86, 988–996. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-Y.; Zhang, T.-Y.; Zhang, M.-Y.; Cheng, J.; Zhang, Y.-X. An Endophytic Fungi of Ginkgo biloba L. Produces Antimicrobial Metabolites as Potential Inhibitors of FtsZ of Staphylococcus aureus. Fitoterapia 2018, 128, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Eshboev, F.; Karakozova, M.; Abdurakhmanov, J.; Bobakulov, K.; Dolimov, K.; Abdurashidov, A.; Baymirzaev, A.; Makhnyov, A.; Terenteva, E.; Sasmakov, S.; et al. Antimicrobial and Cytotoxic Activities of the Secondary Metabolites of Endophytic Fungi Isolated from the Medicinal Plant Hyssopus Officinalis. Antibiotics 2023, 12, 1201. [Google Scholar] [CrossRef] [PubMed]
- Machado, F.P.; Kumla, D.; Pereira, J.A.; Sousa, E.; Dethoup, T.; Freitas-Silva, J.; Costa, P.M.; Mistry, S.; Silva, A.M.S.; Kijjoa, A. Prenylated Phenylbutyrolactones from Cultures of a Marine Sponge-Associated Fungus Aspergillus flavipes KUFA1152. Phytochemistry 2021, 185, 112709. [Google Scholar] [CrossRef] [PubMed]
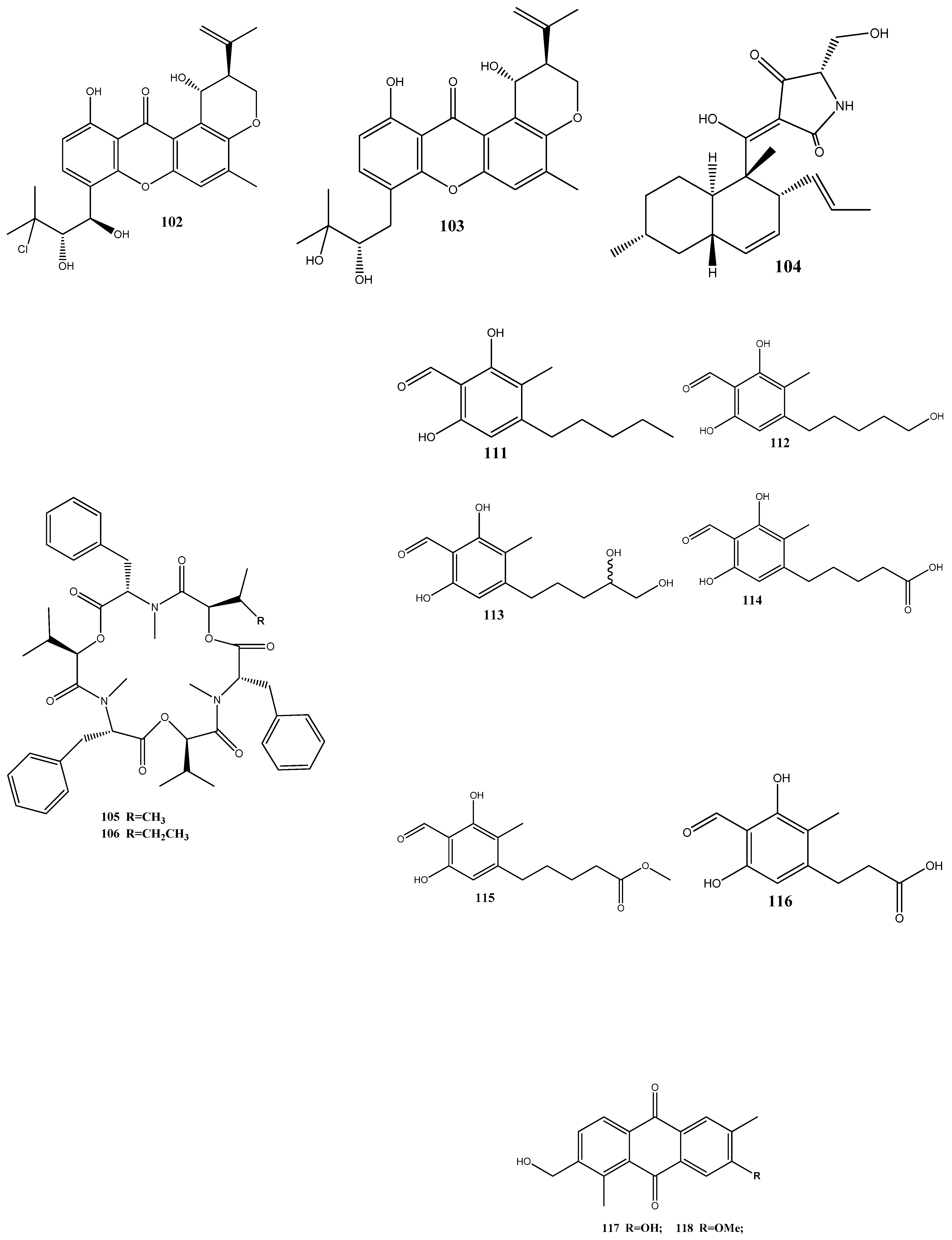
- Subban, K.; Subramani, R.; Johnpaul, M. A Novel Antibacterial and Antifungal Phenolic Compound from the Endophytic fungus Pestalotiopsis mangiferae. Nat. Prod. Res. 2013, 27, 1445–1449. [Google Scholar] [CrossRef]
- Bai, Z.-Q.; Lin, X.; Wang, Y.; Wang, J.; Zhou, X.; Yang, B.; Liu, J.; Yang, X.; Wang, Y.; Liu, Y. New Phenyl Derivatives from Endophytic Fungus Aspergillus flavipes AIL8 Derived of Mangrove Plant Acanthus ilicifolius. Fitoterapia 2014, 95, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Ramos, G.d.C.; Silva-Silva, J.V.; Watanabe, L.A.; Siqueira, J.E.d.S.; Almeida-Souza, F.; Calabrese, K.S.; Marinho, A.M.d.R.; Marinho, P.S.B.; Oliveira, A.S.d. Phomoxanthone A, Compound of Endophytic Fungi Paecilomyces sp. And Its Potential Antimicrobial and Antiparasitic. Antibiotics 2022, 11, 1332. [Google Scholar] [CrossRef]
- Wei, L.; Zhang, Q.; Xie, A.; Xiao, Y.; Guo, K.; Mu, S.; Xie, Y.; Li, Z.; He, T. Isolation of Bioactive Compounds, Antibacterial Activity, and Action Mechanism of Spore Powder from Aspergillus niger Xj. Front. Microbiol. 2022, 13, 934857. [Google Scholar] [CrossRef]
- Zhao, J.-C.; Wang, Y.-L.; Zhang, T.-Y.; Chen, Z.-J.; Yang, T.-M.; Wu, Y.-Y.; Sun, C.-P.; Ma, X.-C.; Zhang, Y.-X. Indole Diterpenoids from the Endophytic Fungus Drechmeria sp. as Natural Antimicrobial Agents. Phytochemistry 2018, 148, 21–28. [Google Scholar] [CrossRef]
- Liang, J.-H.; Huo, X.-K.; Cheng, Z.-B.; Sun, C.-P.; Zhao, J.-C.; Kang, X.-H.; Zhang, T.-Y.; Chen, Z.-J.; Yang, T.-M.; Wu, Y.-Y.; et al. An Indole Diterpenoid Isolated from the Fungus Drechmeria sp. and Its Antimicrobial Activity. Nat. Prod. Res. 2019, 33, 2770–2776. [Google Scholar] [CrossRef] [PubMed]
- Ismaiel, A.A.; Rabie, G.H.; Abd El-Aal, M.A. Antimicrobial and Morphogenic Effects of Emodin Produced by Aspergillus awamori WAIR120. Biologia 2016, 71, 464–474. [Google Scholar] [CrossRef]
- George, T.K.; Joy, A.; Divya, K.; Jisha, M.S. In Vitro and in Silico Docking Studies of Antibacterial Compounds Derived from Endophytic Penicillium Setosum. Microb. Pathog. 2019, 131, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Trenado-Uribe, M.; Silva-Miranda, M.; Rivero-Cruz, J.F.; Rodríguez-Peña, K.; Espitia-Pinzón, C.I.; Rodríguez-Sanoja, R.; Sánchez, S. Antimycobacterial Activity of an Anthracycline Produced by an Endophyte Isolated from Amphipterygium Adstringens. Mol. Biol. Rep. 2018, 45, 2563–2570. [Google Scholar] [CrossRef] [PubMed]
- Nalli, Y.; Singh, S.; Gajjar, A.; Mahizhaveni, B.; Dusthackeer, V.N.A.; Shinde, P.B. Bacillibactin Class Siderophores Produced by the Endophyte Bacillus Subtilis NPROOT3 as Antimycobacterial Agents. Lett. Appl. Microbiol. 2023, 76, 1–8. [Google Scholar] [CrossRef]
- Adeleke, B.; Babalola, O. Pharmacological Potential of Fungal Endophytes Associated with Medicinal Plants: A Review. J. Fungi 2021, 7, 147. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Zhang, J.; Xiao, W.; Liu, S. Efficacy and Mechanism of Actions of Natural Antimicrobial Drugs. Pharmacol. Ther. 2020, 216, 107671. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Li, S.; Zhang, X.; Zhao, C. Biological Activities of Some New Secondary Metabolites Isolated from Endophytic Fungi: A Review Study. Int. J. Mol. Sci. 2021, 22, 959. [Google Scholar] [CrossRef]
- Ziyadullaev, M.; Karimov, R.; Abdurazakhov, A.; Parmanov, A.; Sasmakov, S.; Abdurakhmanov, J.; Eshboev, F.; Azimova, S. Synthesis of 6-Substituted 3(H)-Quinazolin-4-Ones and Their Antimicrobial Activity. Pharm. Chem. J. 2023, 57, 373–377. [Google Scholar] [CrossRef]
- Darby, E.M.; Trampari, E.; Siasat, P.; Gaya, M.S.; Alav, I.; Webber, M.A.; Blair, J.M.A. Molecular Mechanisms of Antibiotic Resistance Revisited. Nat. Rev. Microbiol. 2023, 21, 280–295. [Google Scholar] [CrossRef] [PubMed]
- Pasrija, P.; Girdhar, M.; Kumar, M.; Arora, S.; Katyal, A. Endophytes: An Unexplored Treasure to Combat Multidrug Resistance. Phytomed. Plus 2022, 2, 100249. [Google Scholar] [CrossRef]
- Silva, D.P.D.; Cardoso, M.S.; Macedo, A.J. Endophytic Fungi as a Source of Antibacterial Compounds—A Focus on Gram-Negative Bacteria. Antibiotics 2022, 11, 1509. [Google Scholar] [CrossRef] [PubMed]
- Munita, J.M.; Arias, C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 4, 1–30. [Google Scholar] [CrossRef]
- Irfan, M.; Almotiri, A.; AlZeyadi, Z.A. Antimicrobial Resistance and Its Drivers—A Review. Antibiotics 2022, 11, 1362. [Google Scholar] [CrossRef]
- Shimels Tikuye, Y. Review on Antibiotic Resistance: Resistance Mechanisms, Methods of Detection and Its Controlling Strategies. Biomed J. Sci. Tech. Res. 2020, 24, 18651–18657. [Google Scholar] [CrossRef]
- Urban-Chmiel, R.; Marek, A.; Stępień-Pyśniak, D.; Wieczorek, K.; Dec, M.; Nowaczek, A.; Osek, J. Antibiotic Resistance in Bacteria—A Review. Antibiotics 2022, 11, 1079. [Google Scholar] [CrossRef]
- Schaenzer, A.J.; Wright, G.D. Antibiotic Resistance by Enzymatic Modification of Antibiotic Targets. Trends Mol. Med. 2020, 26, 768–782. [Google Scholar] [CrossRef]
- Liu, Y.; Li, R.; Xiao, X.; Wang, Z. Molecules That Inhibit Bacterial Resistance Enzymes. Molecules 2018, 24, 43. [Google Scholar] [CrossRef]
- Saha, M.; Sarkar, A. Review on Multiple Facets of Drug Resistance: A Rising Challenge in the 21st Century. J. Xenobiot. 2021, 11, 197–214. [Google Scholar] [CrossRef]
- Egorov, A.M.; Ulyashova, M.M.; Rubtsova, M.Y. Bacterial Enzymes and Antibiotic Resistance. Acta Naturae 2018, 10, 33–48. [Google Scholar] [CrossRef]
- Whittle, E.E.; McNeil, H.E.; Trampari, E.; Webber, M.; Overton, T.W.; Blair, J.M.A. Efflux Impacts Intracellular Accumulation Only in Actively Growing Bacterial Cells. MBio 2021, 12, e0260821. [Google Scholar] [CrossRef]
- Davin-Regli, A.; Pages, J.-M.; Ferrand, A. Clinical Status of Efflux Resistance Mechanisms in Gram-Negative Bacteria. Antibiotics 2021, 10, 1117. [Google Scholar] [CrossRef]
- Lv, F.; Cai, J.; He, Q.; Wang, W.; Luo, Y.; Wang, X.; Mi, N.; Zhao, Z.; Li, G.; Luo, W. Overexpression of Efflux Pumps Mediate Pan Resistance of Klebsiella pneumoniae Sequence Type 11. Microb. Drug Resist. 2021, 27, 1405–1411. [Google Scholar] [CrossRef]
- Zhang, F.; Cheng, W. The Mechanism of Bacterial Resistance and Potential Bacteriostatic Strategies. Antibiotics 2022, 11, 1215. [Google Scholar] [CrossRef]
- Abebe, G.M. The Role of Bacterial Biofilm in Antibiotic Resistance and Food Contamination. Int. J. Microbiol. 2020, 2020, 1–10. [Google Scholar] [CrossRef]
- Dincer, S.; Masume Uslu, F.; Delik, A. Antibiotic Resistance in Biofilm. In Bacterial Biofilms; Dincer, S., Özdenefe, M.S., Arkut, A., Eds.; IntechOpen: London, UK, 2020; pp. 1–14. [Google Scholar] [CrossRef]
- Palmer, A.C.; Kishony, R. Opposing Effects of Target Overexpression Reveal Drug Mechanisms. Nat. Commun. 2014, 5, 4296. [Google Scholar] [CrossRef] [PubMed]
- Cafiso, V.; Bertuccio, T.; Purrello, S.; Campanile, F.; Mammina, C.; Sartor, A.; Raglio, A.; Stefani, S. dltA Overexpression: A Strain-Independent Keystone of Daptomycin Resistance in Methicillin-Resistant Staphylococcus aureus. Int. J. Antimicrob. Agents 2014, 43, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Huseby, D.L.; Brandis, G.; Hughes, D. Alternative Evolutionary Pathways for Drug-Resistant Small Colony Variant Mutants in Staphylococcus aureus. MBio 2017, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Tantapakul, C.; Promgool, T.; Kanokmedhakul, K.; Soytong, K.; Song, J.; Hadsadee, S.; Jungsuttiwong, S.; Kanokmedhakul, S. Bioactive Xanthoquinodins and Epipolythiodioxopiperazines from Chaetomium globosum 7s-1, an Endophytic Fungus Isolated from Rhapis cochinchinensis (Lour.) Mart. Nat. Prod. Res. 2020, 34, 494–502. [Google Scholar] [CrossRef]
- El-Zawawy, N.A.; Ali, S.S.; Nouh, H.S. Exploring the Potential of Rhizopus oryzae AUMC14899 as a Novel Endophytic Fungus for the Production of L-Tyrosine and Its Biomedical Applications. Microb. Cell Fact. 2023, 22, 31. [Google Scholar] [CrossRef] [PubMed]
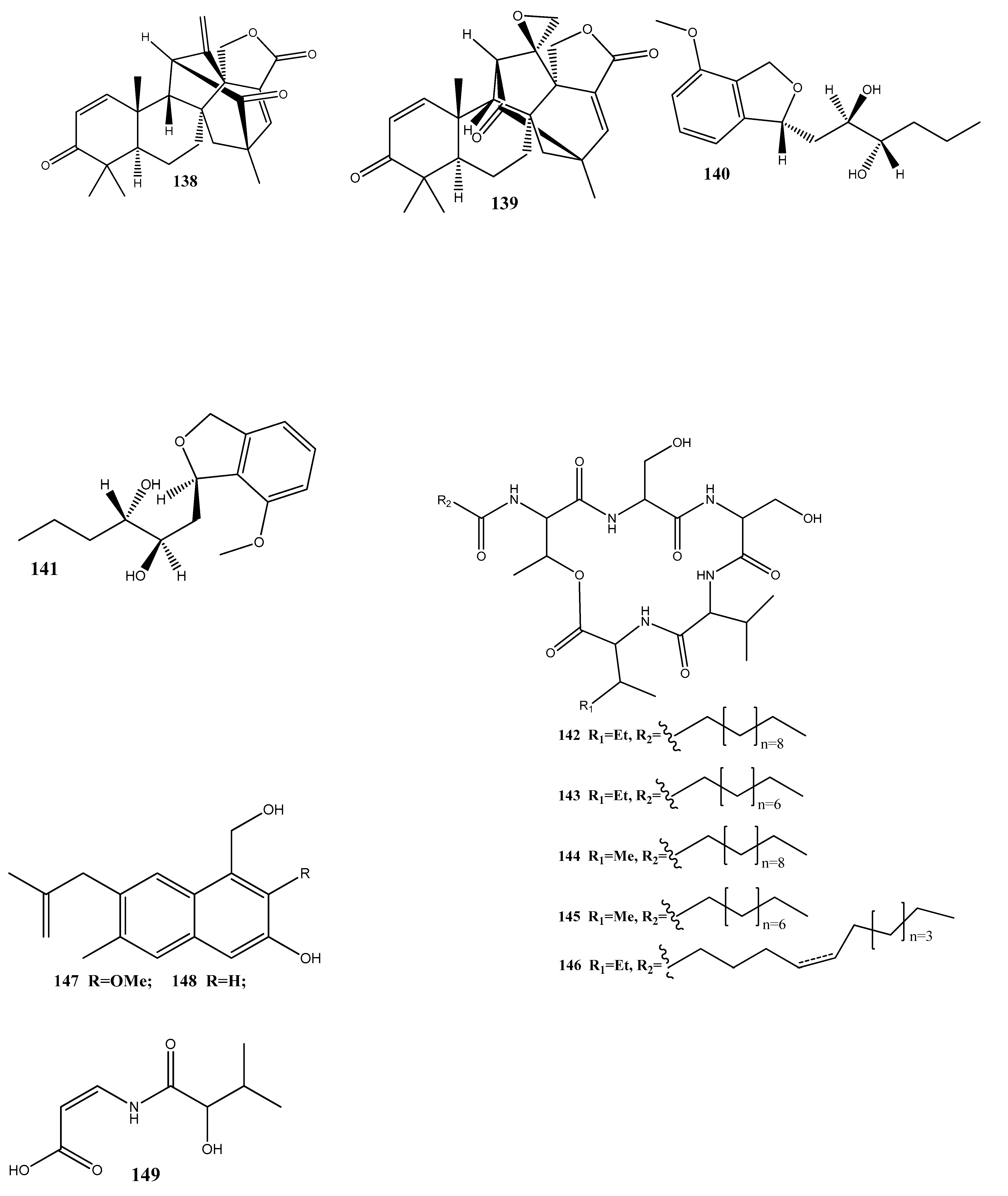
- Wu, Z.; Zhang, X.; Anbari, W.H.A.; Zhou, Q.; Zhou, P.; Zhang, M.; Zeng, F.; Chen, C.; Tong, Q.; Wang, J.; et al. Cysteine Residue Containing Merocytochalasans and 17,18-seco-Aspochalasins from Aspergillus micronesiensis. J. Nat. Prod. 2019, 82, 2653–2658. [Google Scholar] [CrossRef]
- Dai, J.-M.; Zhu, L.-C.; Xiao, D.; Xie, J.; Wang, X.; Mi, Q.-L.; Shi, J.-Q.; Yin, G.-Y.; Yang, Y.-K.; Yang, G.-Y.; et al. Two New Anthraquinones from the Cigar Tobacco-Derived Fungus Aspergillus versicolor and Their Bioactivities. Chem. Nat. Compd. 2022, 58, 1001–1005. [Google Scholar] [CrossRef]
- Zhang, W.; Lu, X.; Huo, L.; Zhang, S.; Chen, Y.; Zou, Z.; Tan, H. Sesquiterpenes and Steroids from an Endophytic Eutypella Scoparia. J. Nat. Prod. 2021, 84, 1715–1724. [Google Scholar] [CrossRef]
- Li, X.-M.; Zeng, Y.-C.; Chen, J.-H.; Yang, Y.-K.; Li, J.; Ye, L.; Du, G.; Zhou, M.; Hu, Q.-F.; Yang, G.; et al. Two New Naphthalene Derivatives from the Fermentation Products of an Endophytic Fungus Phomopsis sp. Chem. Nat. Compd. 2019, 55, 618–621. [Google Scholar] [CrossRef]
- Nurunnabi, T.R.; Nahar, L.; Al-Majmaie, S.; Rahman, S.M.M.; Sohrab, M.H.; Billah, M.M.; Ismail, F.M.D.; Rahman, M.M.; Sharples, G.P.; Sarker, S.D. Anti-MRSA Activity of Oxysporone and Xylitol from the Endophytic Fungus Pestalotia sp. Growing on the Sundarbans Mangrove Plant Heritiera fomes. Phytother. Res. 2018, 32, 348–354. [Google Scholar] [CrossRef]
- Deng, Q.; Li, G.; Sun, M.; Yang, X.; Xu, J. A New Antimicrobial Sesquiterpene Isolated from Endophytic Fungus Cytospora sp. from the Chinese mangrove plant Ceriops tagal. Nat. Prod. Res. 2020, 34, 1404–1408. [Google Scholar] [CrossRef]
- Wu, X.; Fang, L.-Z.; Liu, F.-L.; Pang, X.-J.; Qin, H.-L.; Zhao, T.; Xu, L.-L.; Yang, D.-F.; Yang, X.-L. New Prenylxanthones, Polyketide Hemiterpenoid Pigments from the Endophytic Fungus Emericella sp. XL029 and Their Anti-Agricultural Pathogenic Fungal and Antibacterial Activities. RSC Adv. 2017, 7, 31115–31122. [Google Scholar] [CrossRef]
- Shi, S.; Li, Y.; Ming, Y.; Li, C.; Li, Z.; Chen, I.; Luo, M. Biological Activity and Chemical Composition of the Endophytic Fungus Fusarium sp. TP-G1 Obtained from the Root of Dendrobium Officinale Kimura et Migo. Rec. Nat. Prod. 2018, 12, 549–556. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, L.; Kalscheuer, R.; Liu, Z.; Proksch, P. Antifungal Polyketide Derivatives from the Endophytic Fungus Aplosporella javeedii. Bioorg. Med. Chem. 2020, 28, 115456. [Google Scholar] [CrossRef]
- Wu, F.; Zhu, Y.-N.; Hou, Y.-T.; Mi, Q.-L.; Chen, J.-H.; Zhang, C.-M.; Miao, D.; Zhou, M.; Wang, W.-G.; Hu, Q.-F.; et al. Two New Antibacterial Anthraquinones from Cultures of an Endophytic Fungus Phomopsis sp. Chem. Nat. Compd. 2021, 57, 823–827. [Google Scholar] [CrossRef]
- Chen, H.-L.; Zhao, W.-T.; Liu, Q.-P.; Chen, H.-Y.; Zhao, W.; Yang, D.-F.; Yang, X.-L. (±)-Preisomide: A New Alkaloid Featuring a Rare Naturally Occurring Tetrahydro-2H-1,2-Oxazin Skeleton from an Endophytic Fungus Preussia Isomera by Using OSMAC Strategy. Fitoterapia 2020, 141, 104475. [Google Scholar] [CrossRef]
- Qi, X.; Li, X.; Zhao, J.; He, N.; Li, Y.; Zhang, T.; Wang, S.; Yu, L.; Xie, Y. GKK1032C, a New Alkaloid Compound from the Endophytic Fungus Penicillium sp. CPCC 400817 with Activity against Methicillin-Resistant S. aureus. J. Antibiot. 2019, 72, 237–240. [Google Scholar] [CrossRef]
- Yang, S.; Zhao, W.; Chen, H.; Zhang, L.; Liu, T.; Chen, H.; Yang, X. Aureonitols A and B, Two New C13-Polyketides from Chaetomium Globosum, an Endophytic Fungus in Salvia miltiorrhiza. Chem. Biodivers. 2019, 16, e1900364. [Google Scholar] [CrossRef] [PubMed]
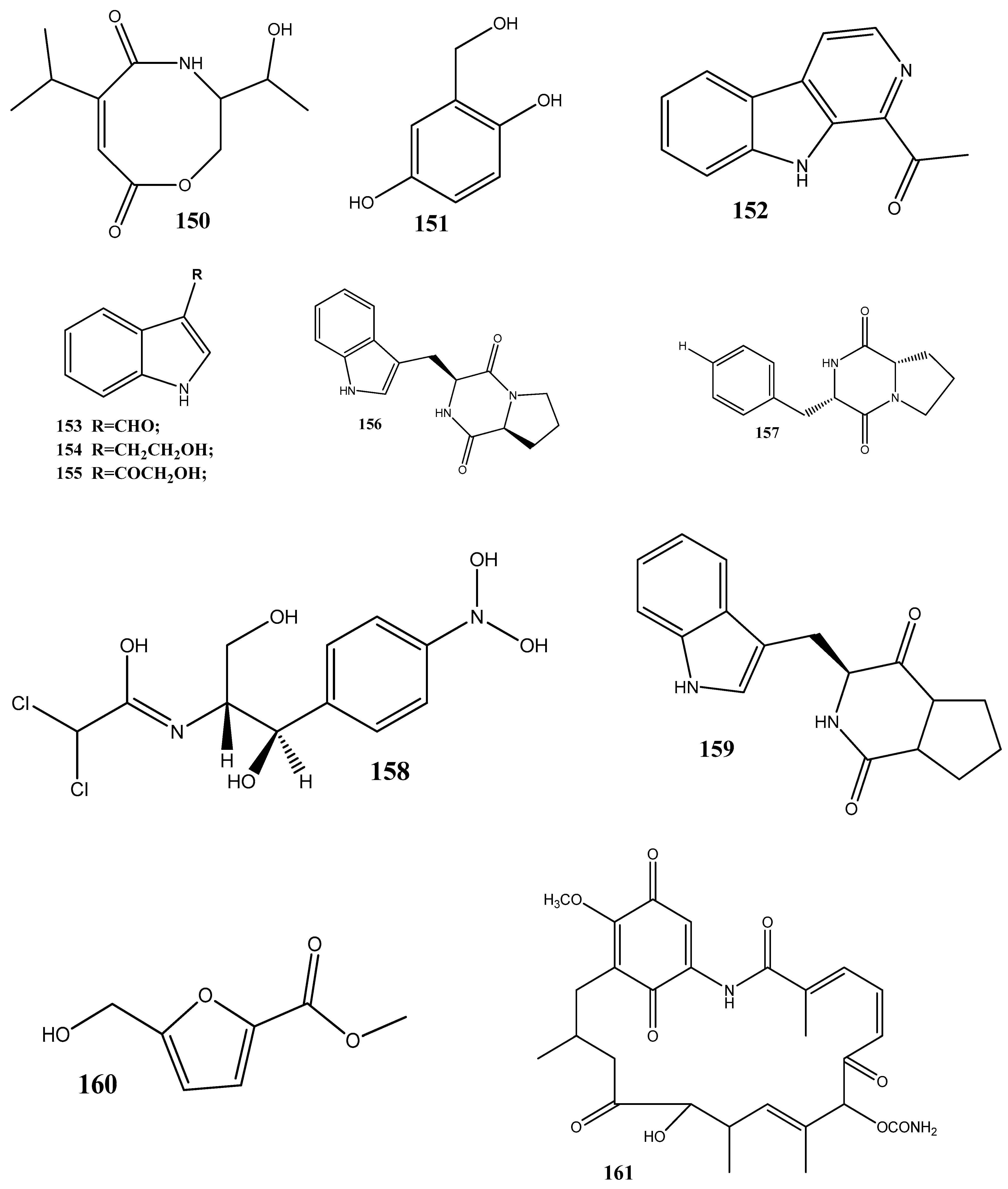
- He, Y.; Hu, Z.; Li, Q.; Huang, J.; Li, X.-N.; Zhu, H.; Liu, J.; Wang, J.; Xue, Y.; Zhang, Y. Bioassay-Guided Isolation of Antibacterial Metabolites from Emericella sp. TJ29. J. Nat. Prod. 2017, 80, 2399–2405. [Google Scholar] [CrossRef] [PubMed]
- Mai, P.-Y.; Levasseur, M.; Buisson, D.; Touboul, D.; Eparvier, V. Identification of Antimicrobial Compounds from Sandwithia Guyanensis-Associated Endophyte Using Molecular Network Approach. Plants 2019, 9, 47. [Google Scholar] [CrossRef]
- Li, X.-M.; Mi, Q.-L.; Gao, Q.; Li, J.; Song, C.-M.; Zeng, W.-L.; Xiang, H.-Y.; Liu, X.; Chen, J.-H.; Zhang, C.-M.; et al. Antibacterial Naphthalene Derivatives from the Fermentation Products of the Endophytic Fungus Phomopsis fukushii. Chem. Nat. Compd. 2021, 57, 293–296. [Google Scholar] [CrossRef]
- Marmouzi, I.; El Abbes Faouzi, M.; Saidi, N.; Cherrah, Y.; Rehberg, N.; Ebada, S.S.; Ebrahim, W.; Kalscheuer, R.; Proksch, P. Bioactive Secondary Metabolites from Chaetomium globosum, an Endophyte From the Moroccan Plant Avena sativa. Chem. Nat. Compd. 2017, 53, 1208–1211. [Google Scholar] [CrossRef]
- Noriler, S.A.; Savi, D.C.; Ponomareva, L.V.; Rodrigues, R.; Rohr, J.; Thorson, J.S.; Glienke, C.; Shaaban, K.A. Vochysiamides A and B: Two New Bioactive Carboxamides Produced by the New Species Diaporthe vochysiae. Fitoterapia 2019, 138, 104273. [Google Scholar] [CrossRef]
- Gos, F.M.W.R.; Savi, D.C.; Shaaban, K.A.; Thorson, J.S.; Aluizio, R.; Possiede, Y.M.; Rohr, J.; Glienke, C. Antibacterial Activity of Endophytic Actinomycetes Isolated from the Medicinal Plant Vochysia divergens (Pantanal, Brazil). Front. Microbiol. 2017, 8, 1642. [Google Scholar] [CrossRef]
- Zin, N.M.; Alshaibani, M.; Jalil, J.; Sidik, N.; Edrada-Ebel, R. Isolation and Characterization of Cyclo-(Tryptophanyl-Prolyl) and Chloramphenicol from Streptomyces sp. SUK 25 with Antimethicillin-Resistant Staphylococcus Aureus Activity. Drug Des. Dev. Ther. 2016, 10, 1817–1827. [Google Scholar] [CrossRef]
- Taechowisan, T.; Puckdee, W.; Phutdhawong, W.S. Streptomyces zerumbet, a Novel Species from Zingiber zerumbet (L.) Smith and Isolation of Its Bioactive Compounds. Adv. Microbiol. 2019, 9, 194–219. [Google Scholar] [CrossRef]
- Lee, C.; Kim, S.; Li, W.; Bang, S.; Lee, H.; Lee, H.-J.; Noh, E.-Y.; Park, J.-E.; Bang, W.Y.; Shim, S.H. Bioactive Secondary Metabolites Produced by an Endophytic Fungus Gaeumannomyces sp. JS0464 from a Maritime Halophyte Phragmites communis. J. Antibiot. 2017, 70, 737–742. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Eshboev, F.; Shukurov, O.; Alaylar, B.; Arora, N.K. Bacterial Bioprotectants: Biocontrol Traits and Induced Resistance to Phytopathogens. Microbiol. Res. (Pavia) 2023, 14, 689–703. [Google Scholar] [CrossRef]
- Ming, Q.; Huang, X.; He, Y.; Qin, L.; Tang, Y.; Liu, Y.; Huang, Y.; Zhang, H.; Li, P. Genome Mining and Screening for Secondary Metabolite Production in the Endophytic Fungus Dactylonectria alcacerensis CT-6. Microorganisms 2023, 11, 968. [Google Scholar] [CrossRef] [PubMed]
- Aghdam, S.A.; Brown, A.M.V. Deep Learning Approaches for Natural Product Discovery from Plant Endophytic Microbiomes. Environ. Microbiome 2021, 16, 6. [Google Scholar] [CrossRef]
- Blin, K.; Kim, H.U.; Medema, M.H.; Weber, T. Recent Development of antiSMASH and Other Computational Approaches to Mine Secondary Metabolite Biosynthetic Gene Clusters. Brief. Bioinform. 2019, 20, 1103–1113. [Google Scholar] [CrossRef]
- Deepika, V.B.; Murali, T.S.; Satyamoorthy, K. Modulation of Genetic Clusters for Synthesis of Bioactive Molecules in Fungal Endophytes: A Review. Microbiol. Res. 2016, 182, 125–140. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, M.; Tang, Y.; Shao, Y.; Wang, H.; Zhang, H. Genome Features and AntiSMASH Analysis of an Endophytic Strain Fusarium sp. R1. Metabolites 2022, 12, 521. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, M.-F.; Zhang, Q.; Yan, B.; Jiang, Y.-L. Screening for Broad-Spectrum Antimicrobial Endophytes from Rosa roxburghii and Multi-Omic Analyses of Biosynthetic Capacity. Front. Plant Sci. 2022, 13, 1060478. [Google Scholar] [CrossRef]
- Cheng, J.-T.; Cao, F.; Chen, X.-A.; Li, Y.-Q.; Mao, X.-M. Genomic and Transcriptomic Survey of an Endophytic Fungus Calcarisporium arbuscula NRRL 3705 and Potential Overview of Its Secondary Metabolites. BMC Genom. 2020, 21, 424. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.P.; Chunduri, J.R. Genome-Wide Analysis and in Silico Screening of Secondary Metabolite Potential of Endophytic Fungi Fusarium Multiceps BPAL1 Obtained in Mumbai, India. Egypt. J. Basic Appl. Sci. 2023, 10, 812–823. [Google Scholar] [CrossRef]
- Tao, J.; Bai, X.; Zeng, M.; Li, M.; Hu, Z.; Hua, Y.; Zhang, H. Whole-Genome Sequence Analysis of an Endophytic Fungus Alternaria sp. SPS-2 and Its Biosynthetic Potential of Bioactive Secondary Metabolites. Microorganisms 2022, 10, 1789. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, X.; Liu, L.; Xiang, M.; Wang, W.; Sun, X.; Che, Y.; Guo, L.; Liu, G.; Guo, L.; et al. Genomic and Transcriptomic Analysis of the Endophytic Fungus Pestalotiopsis Fici Reveals Its Lifestyle and High Potential for Synthesis of Natural Products. BMC Genom. 2015, 16, 28. [Google Scholar] [CrossRef] [PubMed]
- Ehsan, T.; Reza, R.N.; Das, A.; Ahmed, O.; Baten, A.K.M.A.; Ferdous, A.S.; Islam, M.R.; Khan, H. Genome and Secretome Analysis of Jute Endophyte Grammothele lineata Strain SDL-CO-2015-1: Insights into Its Lignocellulolytic Structure and Secondary Metabolite Profile. Genomics 2020, 112, 2794–2803. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.; Pucker, B.; Srivastava, S. Genomic and Transcriptomic Analysis of Camptothecin Producing Novel Fungal Endophyte: Alternaria Burnsii NCIM 1409. Sci. Rep. 2023, 13, 14614. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Li, X.; Iacovelli, R.; Hackl, T.; Haslinger, K. Genomic and Metabolomic Analysis of the Endophytic Fungus Fusarium sp. VM-40 Isolated from the Medicinal Plant Vinca minor. J. Fungi 2023, 9, 704. [Google Scholar] [CrossRef] [PubMed]
- Oberhofer, M.; Malfent, F.; Zehl, M.; Urban, E.; Wackerlig, J.; Reznicek, G.; Vignolle, G.A.; Rückert, C.; Busche, T.; Wibberg, D.; et al. Biosynthetic Potential of the Endophytic Fungus Helotiales sp. BL73 Revealed via Compound Identification and Genome Mining. Appl. Environ. Microbiol. 2022, 88, e0251021. [Google Scholar] [CrossRef]
- Cain, J.W.; Miller, K.I.; Kalaitzis, J.A.; Chau, R.; Neilan, B.A. Genome Mining of a Fungal Endophyte of Taxus Yunnanensis (Chinese Yew) Leads to the Discovery of a Novel Azaphilone Polyketide, Lijiquinone. Microb. Biotechnol. 2020, 13, 1415–1427. [Google Scholar] [CrossRef]
- Nazarov, P.A.; Baleev, D.N.; Ivanova, M.I.; Sokolova, L.M.; Karakozova, M.V. Infectious Plant Diseases: Etiology, Current Status, Problems and Prospects in Plant Protection. Acta Naturae 2020, 12, 46–59. [Google Scholar] [CrossRef] [PubMed]
- Chesnokov, Y.V. Plant Resistance to the Patogens. Agric. Biol. 2007, 1, 16–35. [Google Scholar]


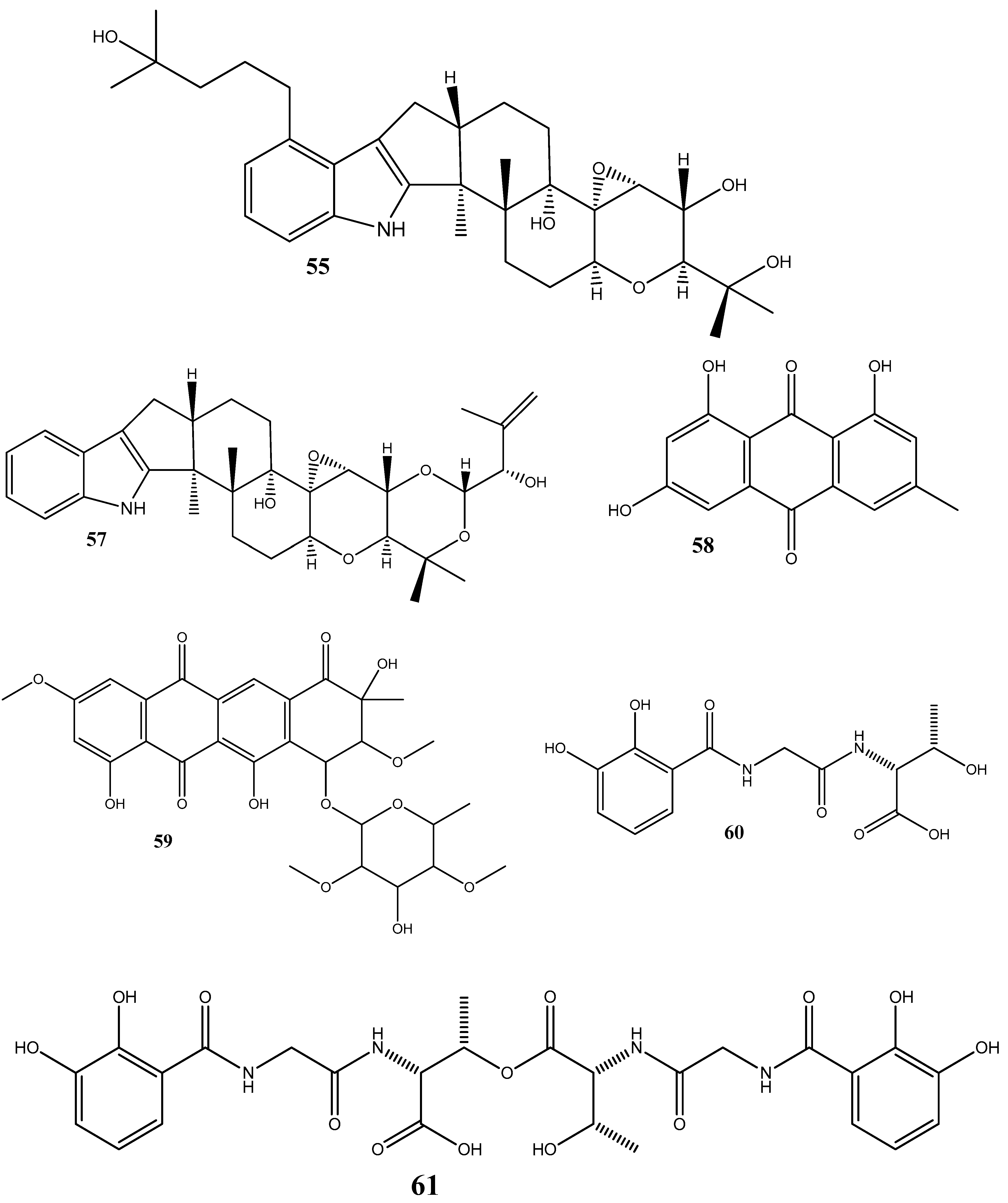

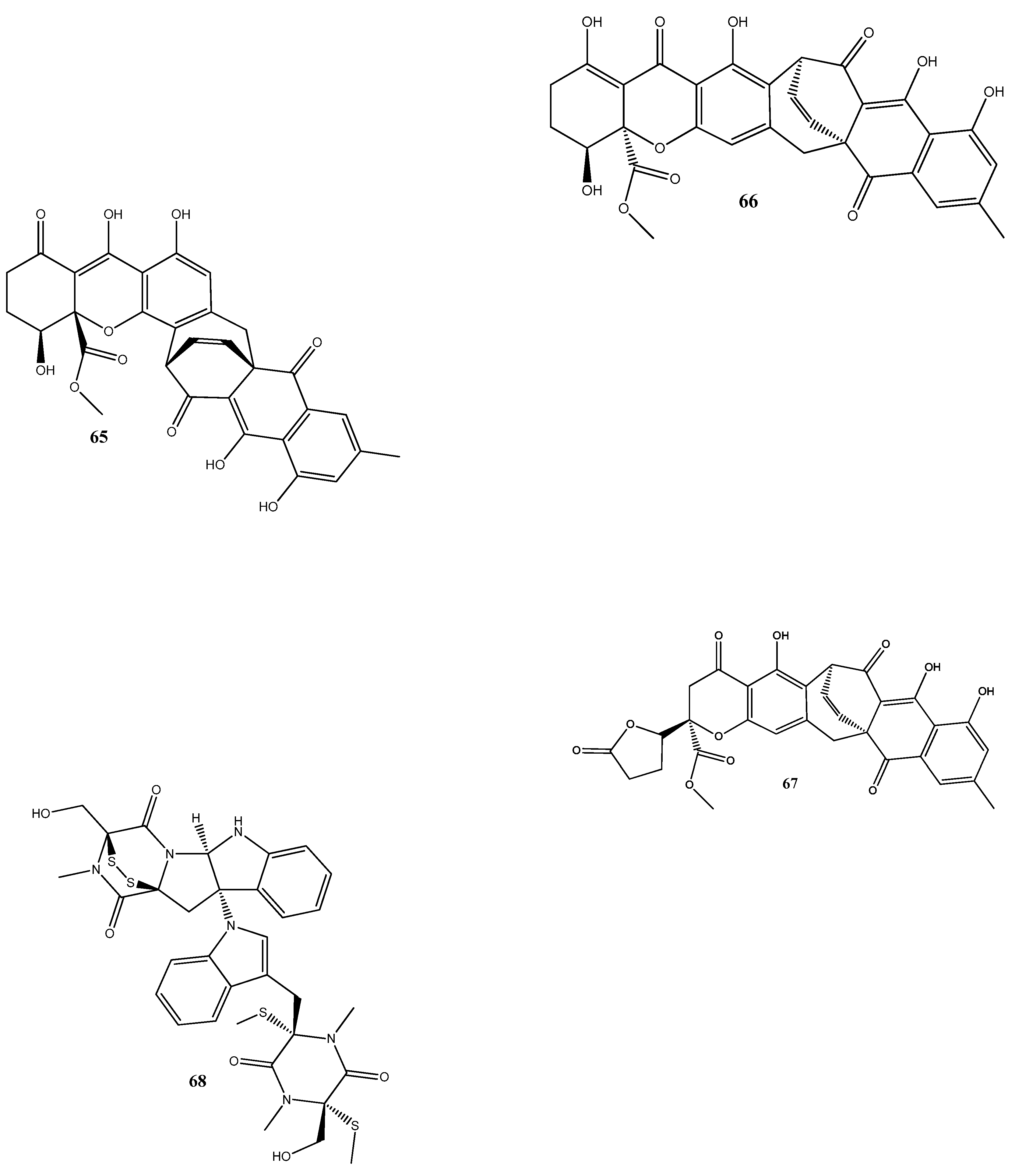
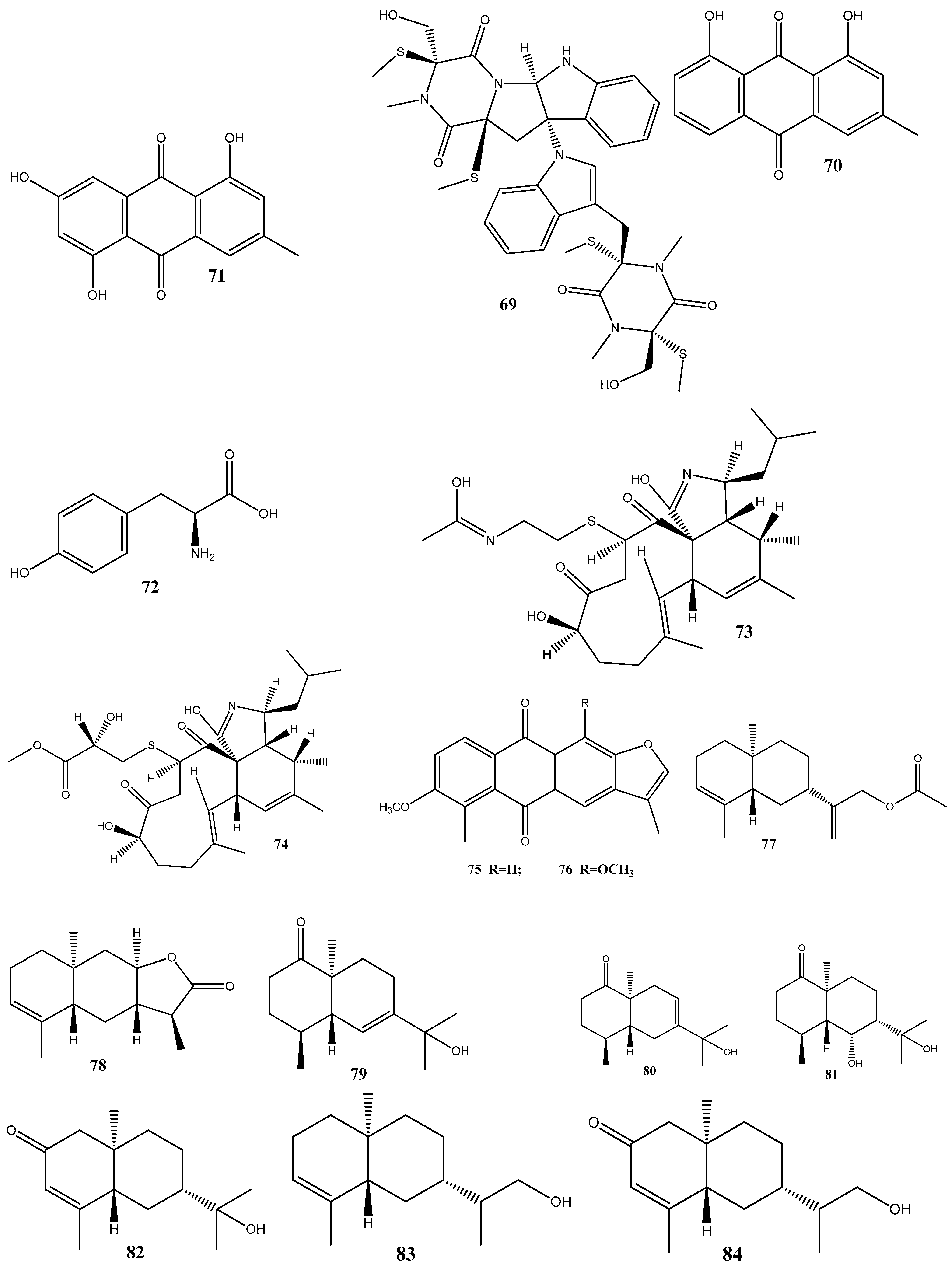
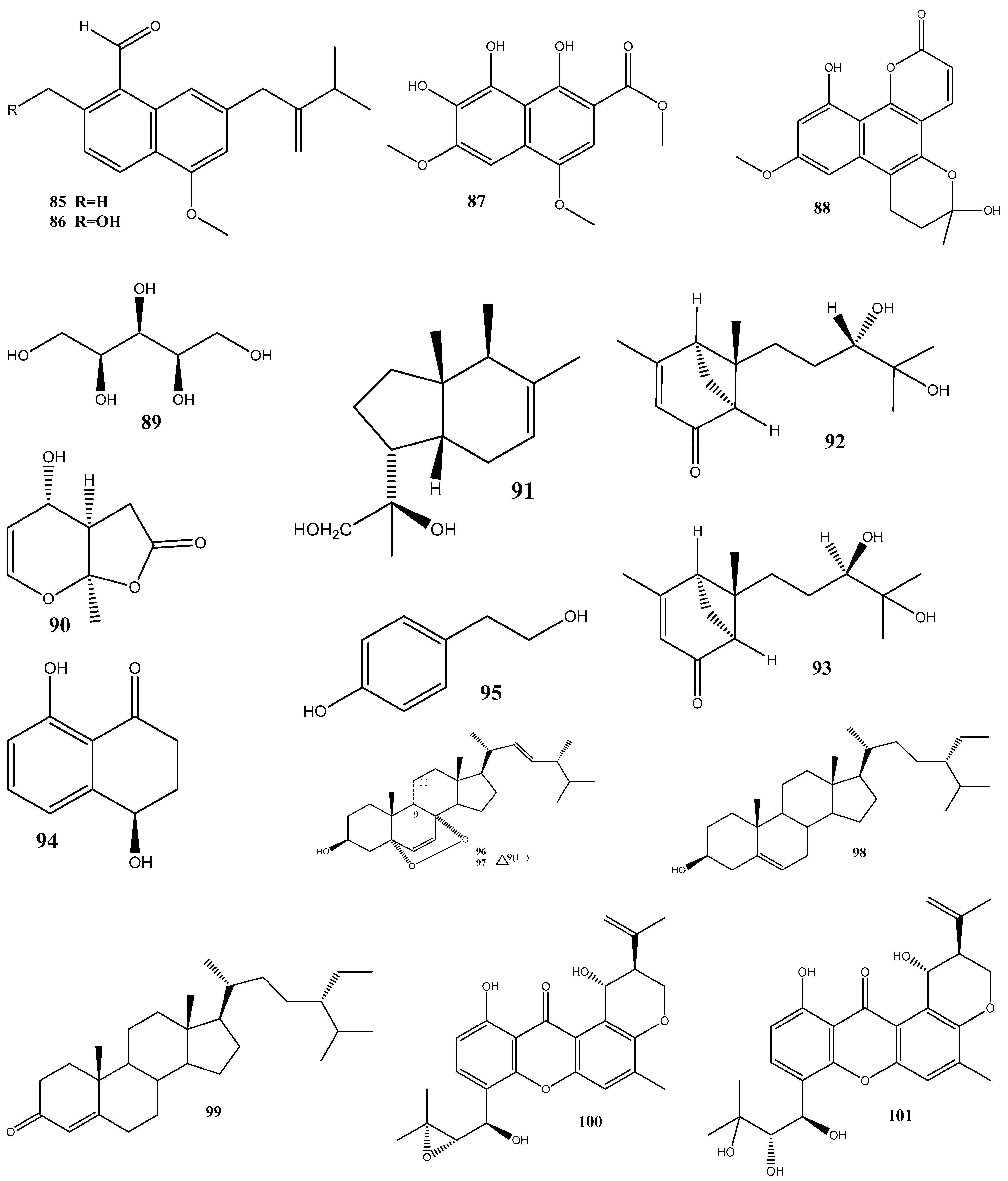


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eshboev, F.; Mamadalieva, N.; Nazarov, P.A.; Hussain, H.; Katanaev, V.; Egamberdieva, D.; Azimova, S. Antimicrobial Action Mechanisms of Natural Compounds Isolated from Endophytic Microorganisms. Antibiotics 2024, 13, 271. https://doi.org/10.3390/antibiotics13030271
Eshboev F, Mamadalieva N, Nazarov PA, Hussain H, Katanaev V, Egamberdieva D, Azimova S. Antimicrobial Action Mechanisms of Natural Compounds Isolated from Endophytic Microorganisms. Antibiotics. 2024; 13(3):271. https://doi.org/10.3390/antibiotics13030271
Chicago/Turabian StyleEshboev, Farkhod, Nilufar Mamadalieva, Pavel A. Nazarov, Hidayat Hussain, Vladimir Katanaev, Dilfuza Egamberdieva, and Shakhnoz Azimova. 2024. "Antimicrobial Action Mechanisms of Natural Compounds Isolated from Endophytic Microorganisms" Antibiotics 13, no. 3: 271. https://doi.org/10.3390/antibiotics13030271
APA StyleEshboev, F., Mamadalieva, N., Nazarov, P. A., Hussain, H., Katanaev, V., Egamberdieva, D., & Azimova, S. (2024). Antimicrobial Action Mechanisms of Natural Compounds Isolated from Endophytic Microorganisms. Antibiotics, 13(3), 271. https://doi.org/10.3390/antibiotics13030271










