Temocillin: Applications in Antimicrobial Stewardship as a Potential Carbapenem-Sparing Antibiotic
Abstract
1. Introduction
2. Results
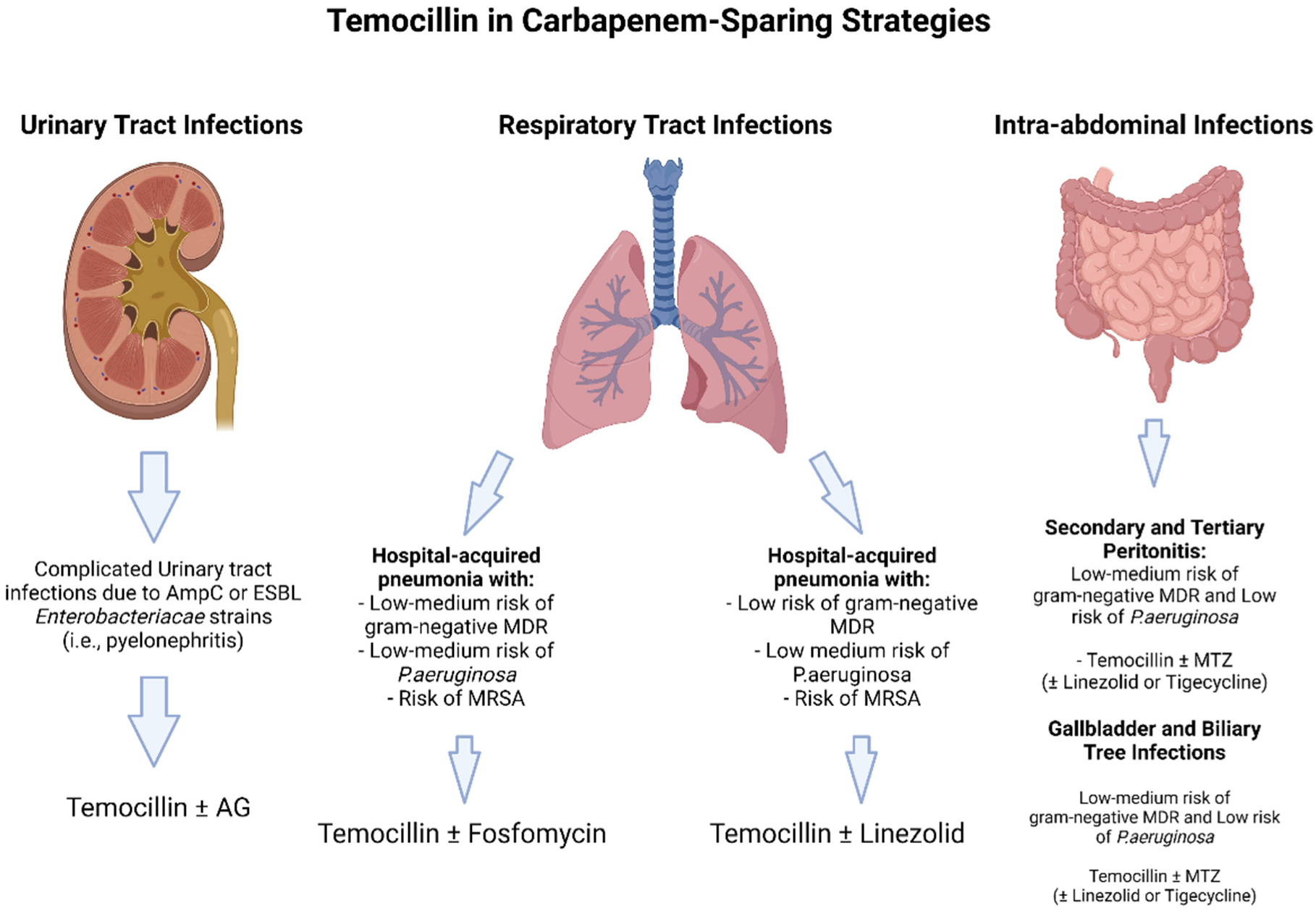
2.1. Temocillin in Urinary Tract Infections
2.2. Temocillin in Bloodstream Infections (BSI)
2.3. Temocillin in Pneumonia
2.4. Temocillin in Abdominal Infections
2.5. Temocillin in Central Nervous System (CNS) Infections
2.6. Temocillin in Skin, Soft Tissues, Surgical Sites, and Osteoarticular Infections
2.7. Temocillin in Venereal and Sexual Transmitted Diseases
2.8. Outpatient Antibiotic Treatment (OPAT)
2.9. Impact on Microbiome
3. Discussion
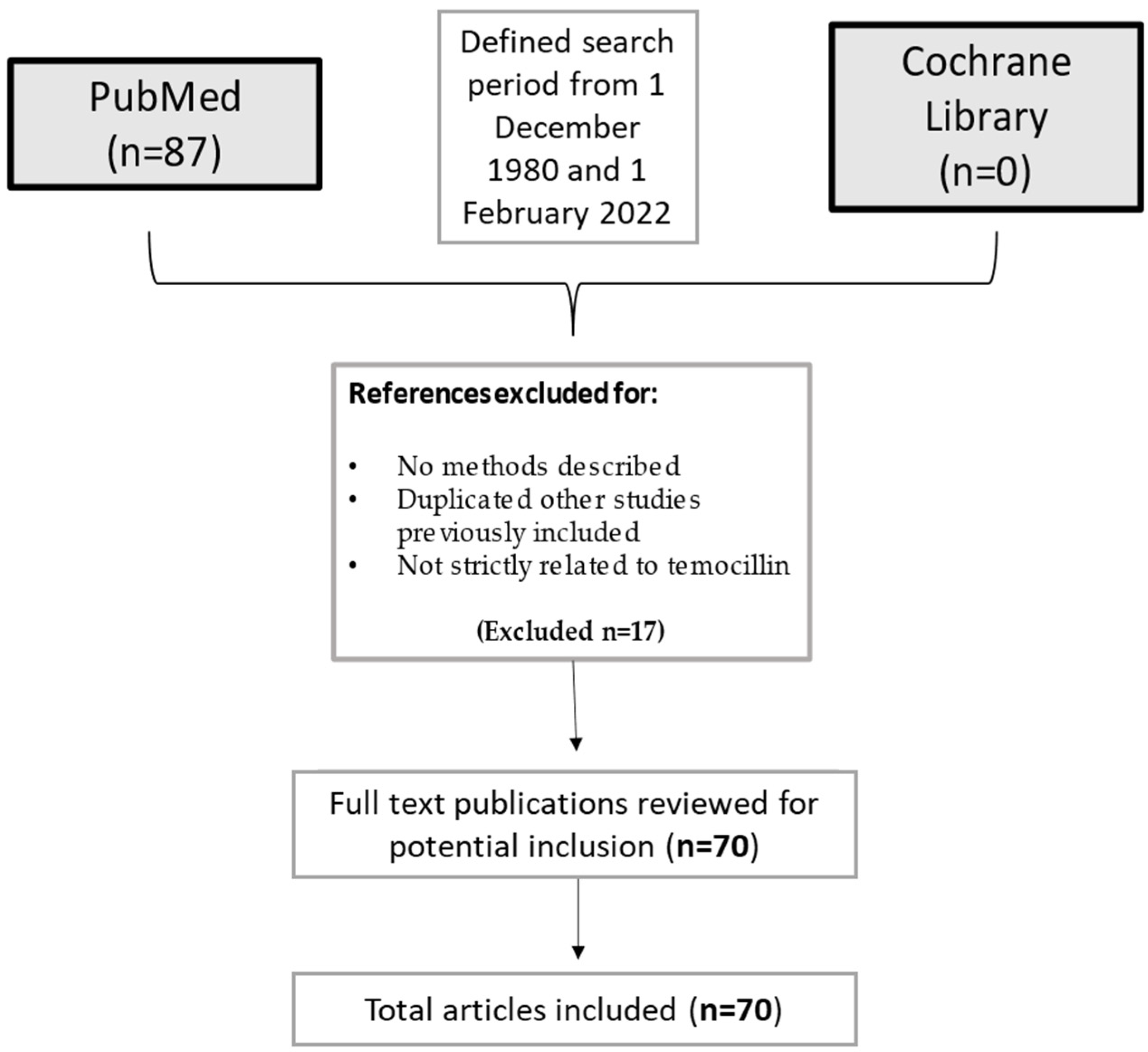
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peirano, G.; Pitout, J.D.D. Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae: Update on Molecular Epidemiology and Treatment Options. Drugs 2019, 79, 1529–1541. [Google Scholar] [CrossRef] [PubMed]
- Cantón, R.; Novais, Â.; Valverde, A.; Machado, E.; Peixe, L.; Baquero, F.; Coque, T. Prevalence and spread of extended-spectrum β-lactamase-producing Enterobacteriaceae in Europe. Clin. Microbiol. Infect. 2008, 14, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Van Duin, D.; Doi, Y. The global epidemiology of carbapenemase-producing Enterobacteriaceae. Virulence 2017, 8, 460–469. [Google Scholar] [CrossRef]
- Martin, A.; Fahrbach, K.; Zhao, Q.; Lodise, T. Association Between Carbapenem Resistance and Mortality Among Adult, Hospitalized Patients with Serious Infections Due to Enterobacteriaceae: Results of a Systematic Literature Review and Meta-analysis. Open Forum Infect. Dis. 2018, 5, ofy150. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Kennedy, S.; Perry, M.; Wilson, J.; Chase-Topping, M.; Anderson, E.; Woolhouse, M.; Lockhart, M. Epidemiology of and risk factors for mortality due to carbapenemase-producing organisms (CPO) in healthcare facilities. J. Hosp. Infect. 2021, 110, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Corcione, S.; Lupia, T.; Maraolo, A.E.; Pinna, S.M.; Gentile, I.; De Rosa, F.G. Carbapenem-sparing strategy: Carbapenemase, treatment, and stewardship. Curr. Opin. Infect. Dis. 2019, 32, 663–673. [Google Scholar] [CrossRef]
- Chen, H.; Williams, J.D.; Chen, H.Y. The Activity of Temocillin Against Enterobacteriaceae, Pseudomonas and Haemophilus influenzae. Drugs 1985, 29, 85–90. [Google Scholar] [CrossRef]
- Just, H.-M.; Becker, C.; Gieringer, J.; Wenz, A.; Bassler, M.; Daschner, F. In Vitro Combination-Effect of Temocillin with Ticarcillin and Aminoglycosides on Gram-Negative and Gram-Positive Bacteria1. Drugs 1985, 29, 74–77. [Google Scholar] [CrossRef]
- Martinez-Beltran, J.; Loza, E.; Gomez-Alferez, A.; Romero-Vivas, J.; Bouza, E. Temocillin. In vitro activity compared with other antibiotics. Drugs 1985, 29 (Suppl. 5), 91–97. [Google Scholar] [CrossRef]
- Yang, Y.; Livermore, D.M. Activity of temocillin and other penicillins against β-lactamase-inducible and -stably derepressed enterobacteria. J. Antimicrob. Chemother. 1988, 22, 299–306. [Google Scholar] [CrossRef]
- Kresken, M.; Körber-Irrgang, B.; Pfeifer, Y.; Werner, G. Activity of temocillin against CTX-M-producing Escherichia coli and Klebsiella pneumoniae from Germany. Int. J. Antimicrob. Agents 2018, 51, 159–160. [Google Scholar] [CrossRef] [PubMed]
- Chalhoub, H.; Pletzer, D.; Weingart, H.; Braun, Y.; Tunney, M.M.; Elborn, J.S.; Rodriguez-Villalobos, H.; Plésiat, P.; Kahl, B.C.; Denis, O.; et al. Mechanisms of intrinsic resistance and acquired susceptibility of Pseudomonas aeruginosa isolated from cystic fibrosis patients to temocillin, a revived antibiotic. Sci. Rep. 2017, 7, 40208. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, K.; Fantin, B. Pharmacokinetics and Pharmacodynamics of Temocillin. Clin. Pharmacokinet. 2017, 57, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Soubirou, J.F.; Rossi, B.; Couffignal, C.; Ruppé, E.; Chau, F.; Massias, L.; Lepeule, R.; Mentre, F.; Fantin, B. Activity of temocillin in a murine model of urinary tract infection due to Escherichia coli producing or not producing the ESBL CTX-M-15. J. Antimicrob. Chemother. 2015, 70, 1466–1472. [Google Scholar] [CrossRef]
- Alexandre, K.; Chau, F.; Guérin, F.; Massias, L.; Lefort, A.; Cattoir, V.; Fantin, B. Activity of temocillin in a lethal murine model of infection of intra-abdominal origin due to KPC-producingEscherichia coli. J. Antimicrob. Chemother. 2016, 71, 1899–1904. [Google Scholar] [CrossRef]
- Vandecasteele, S.J.; Bastos, A.C.M.; Capron, A.; Spinewine, A.; Tulkens, P.M.; Van Bambeke, F. Thrice-weekly temocillin administered after each dialysis session is appropriate for the treatment of serious Gram-negative infections in haemodialysis patients. Int. J. Antimicrob. Agents 2015, 46, 660–665. [Google Scholar] [CrossRef]
- De Jongh, R.; Hens, R.; Basma, V.; Mouton, J.W.; Tulkens, P.M.; Carryn, S. Continuous versus intermittent infusion of temocillin, a directed spectrum penicillin for intensive care patients with nosocomial pneumonia: Stability, compatibility, population pharmacokinetic studies and breakpoint selection. J. Antimicrob. Chemother. 2008, 61 (Suppl. 5), 382–388. [Google Scholar] [CrossRef]
- Cockburn, A.; Mellows, G.; Jackson, D.; White, D.J. Temocillin. Summary of safety studies. Drugs 1985, 29 (Suppl. 5), 103–105. [Google Scholar] [CrossRef]
- Deshayes, S.; Coquerel, A.; Verdon, R. Neurological Adverse Effects Attributable to β-Lactam Antibiotics: A Literature Review. Drug Saf. 2017, 40, 1171–1198. [Google Scholar] [CrossRef]
- Stapleton, P.; Shannon, K.; Phillips, I. The ability of beta-lactam antibiotics to select mutants with derepressed beta-lactamase synthesis from Citrobacter freundii. J. Antimicrob. Chemother. 1995, 36, 483–496. [Google Scholar] [CrossRef]
- Tsakris, A.; Koumaki, V.; Politi, L.; Balakrishnan, I. Activity of temocillin against KPC-producing Enterobacteriaceae clinical isolates. Int. J. Antimicrob. Agents 2020, 55, 105843. [Google Scholar] [CrossRef] [PubMed]
- Woodford, N.; Pike, R.; Meunier, D.; Loy, R.; Hill, R.; Hopkins, K.L. In vitro activity of temocillin against multidrug-resistant clinical isolates of Escherichia coli, Klebsiella spp. and Enterobacter spp., and evaluation of high-level temocillin resistance as a diagnostic marker for OXA-48 carbapenemase. J. Antimicrob. Chemother. 2013, 69, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Clinical Breakpoints—Breakpoints and Guidance. Available online: https://www.eucast.org/clinical_breakpoints/ (accessed on 27 February 2022).
- Susceptibility Testing. Available online: https://bsac.org.uk/susceptibility/ (accessed on 27 February 2022).
- Lockley, M.; Brown, R.; Wise, R. Pharmacokinetics and Tissue Penetration of Temocillin. Drugs 1985, 29, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Overbosch, D.; van Gulpen, C.; Mattie, H. Renal Clearance of Temocillin in Volunteers1. Drugs 1985, 29, 128–134. [Google Scholar] [CrossRef]
- Slocombe, B.; Basker, M.J.; Bentley, P.H.; Clayton, J.P.; Cole, M.; Comber, K.R.; Dixon, R.A.; Edmondson, R.A.; Jackson, D.; Merrikin, D.J.; et al. BRL 17421, a novel beta-lactam antibiotic, highly resistant to beta-lactamases, giving high and prolonged serum levels in humans. Antimicrob. Agents Chemother. 1981, 20, 38–46. [Google Scholar] [CrossRef]
- Verbist, L. In vitro activity of temocillin (BRL 17421), a novel beta-lactamase-stable penicillin. Antimicrob. Agents Chemother. 1982, 22, 157–161. [Google Scholar] [CrossRef]
- Bolivar, R.; Weaver, S.S.; Bodey, G.P. Comparative in vitro study of temocillin (BRL 17421), a new penicillin. Antimicrob. Agents Chemother. 1982, 21, 641–645. [Google Scholar] [CrossRef]
- Gobernado, M.; Canton, E. Comparative In Vitro Activity of Temocillin. Drugs 1985, 29, 24–31. [Google Scholar] [CrossRef]
- Vanhoof, R.; Berin, C.; Carpentier, M.; Fagnart, O.; Glupczynski, Y.; Mans, I.; Nyssen, H.-J.; Surmont, I.; Van Nimmen, L. Comparativein vitroactivity of temocillin and other antimicrobial agents against enterobacteriaceae isolated from patients admitted to five belgian hospitals. Acta Clin. Belg. 2001, 56, 354–359. [Google Scholar] [CrossRef]
- Glupczynski, Y.; Huang, T.-D.; Berhin, C.; Claeys, G.; Delmée, M.; Ide, L.; Ieven, G.; Pierard, D.; Rodriguez-Villalobos, H.; Struelens, M.; et al. In vitro activity of temocillin against prevalent extended-spectrum beta-lactamases producing Enterobacteriaceae from Belgian intensive care units. Eur. J. Clin. Microbiol. 2007, 26, 777–783. [Google Scholar] [CrossRef]
- Asbach, H.; Becker-Boost, E.; Melekos, M.D.; Asbach, P.-D.D.H.W. Clinical Evaluation of Temocillin in Urinary Tract Infections. Drugs 1985, 29 (Suppl. 5), 175–177. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, G.; Beattie, A.D.; Taylor, E.W. Temocillin in the Treatment of Serious Gram-Negative Infections. Drugs 1985, 29 (Suppl. 5), 191–193. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.D.; Smith, R.E.; Balakrishnan, I. Clinical efficacy of temocillin. J. Antimicrob. Chemother. 2009, 64, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Tärnberg, M.; Östholm-Balkhed, Å.; Monstein, H.-J.; Hallgren, A.; Hanberger, H.; Nilsson, L.E. In vitro activity of beta-lactam antibiotics against CTX-M-producing Escherichia coli. Eur. J. Clin. Microbiol. 2011, 30, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Ip, M.; Lai, C.; Fung, K.S.C.; Wong, K.-T.; Zhu, C.; Van De Velde, S.; Tsang, D.N.; Hawkey, P. Activity of temocillin and 15 other agents, including fosfomycin and colistin, against Enterobacteriaceae in Hong Kong. Eur. J. Clin. Microbiol. 2017, 36, 2491–2494. [Google Scholar] [CrossRef]
- Kuch, A.; Zieniuk, B.; Żabicka, D.; Van de Velde, S.; Literacka, E.; Skoczyńska, A.; Hryniewicz, W. Activity of temocillin against ESBL-, AmpC-, and/or KPC-producing Enterobacterales isolated in Poland. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1185–1191. [Google Scholar] [CrossRef] [PubMed]
- Kitzis, M.D.; Billot-Klein, D.; Goldstein, F.W.; Williamson, R.; Tran Van Nhieu, G.; Carlet, J.; Acar, J.F.; Gutmann, L. Dissemination of the Novel Plasmid-Mediated P-Lactamase CTX-1, Which Confers Resistance to Broad-Spectrum Cephalosporins, and Its Inhibition by 1-Lactamase Inhibitors. Antimicrob. Agents Chemother. 1988, 32, 9–14. [Google Scholar] [CrossRef]
- Williams, J.; Chen, H.Y.; Williams, J.D. The Place of Temocillin in the Treatment of Hospital Infections. Drugs 1985, 29, 234–239. [Google Scholar] [CrossRef]
- Temocillin Versus a Carbapenem as Initial Intravenous Treatment for ESBL Related Urinary Tract Infections (TEMO-CARB). Available online: https://clinicaltrials.gov/ct2/show/NCT03543436 (accessed on 1 March 2022).
- Temocillin vs Meropenem for the Targeted Treatment of Bacteraemia Resistant to Third Gen Cephalosporins (ASTARTÉ). Available online: https://clinicaltrials.gov/ct2/show/NCT04478721 (accessed on 1 March 2022).
- Jacoby, G.A.; Carreras, I. Activities of beta-lactam antibiotics against Escherichia coli strains producing extended-spectrum beta-lactamases. Antimicrob. Agents Chemother. 1990, 34, 858–862. [Google Scholar] [CrossRef]
- Kosmidis, J. The Treatment of Complicated and Uncomplicated Urinary Tract Infections with Temocillin. Drugs 1985, 29, 172–174. [Google Scholar] [CrossRef]
- Schulze, B.; Heilmann, H.-D.; Heilmann, H.-D. Treatment of Severe Infections with Temocillin Clinical and Bacteriological Evaluation. Drugs 1985, 29, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Offenstadt, G.; Lesage, D.; Hericord, P.; Pinta, P.; Leaute, J.B.; Amstutz, P. Temocillin Treatment of Serious Infections due to Gram-Negative Bacilli in an Intensive Care Unit1. Drugs 1985, 29, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Verboven, M.; Lauwers, S.; Pintens, H. Temocillin in the treatment of pyelonephritis in children. Drugs Under Exp. Clin. Res. 1987, 13, 171–173. [Google Scholar]
- Balakrishnan, I.; Awad-El-Kariem, F.M.; Aali, A.; Kumari, P.; Mulla, R.; Tan, B.; Brudney, D.; Ladenheim, D.; Ghazy, A.; Khan, I.; et al. Temocillin use in England: Clinical and microbiological efficacies in infections caused by extended-spectrum and/or derepressed AmpC β-lactamase-producing Enterobacteriaceae. J. Antimicrob. Chemother. 2011, 66, 2628–2631. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, K.; Réveillon-Istin, M.; Fabre, R.; Delbos, V.; Etienne, M.; Pestel-Caron, M.; Dahyot, S.; Caron, F. Temocillin against Enterobacteriaceae isolates from community-acquired urinary tract infections: Low rate of resistance and good accuracy of routine susceptibility testing methods. J. Antimicrob. Chemother. 2018, 73, 1848–1853. [Google Scholar] [CrossRef]
- Livermore, D.M.; Hope, R.; Fagan, E.J.; Warner, M.; Woodford, N.; Potz, N. Activity of temocillin against prevalent ESBL- and AmpC- producing Enterobacteriaceae from south-east England. J. Antimicrob. Chemother. 2006, 57, 1012–1014. [Google Scholar] [CrossRef]
- Heard, K.L.; Killington, K.; Mughal, N.; Moore, L.S.P.; Hughes, S. Clinical outcomes of temocillin use for invasive Enterobacterales infections: A single-centre retrospective analysis. JAC-Antimicrob. Resist. 2021, 3, dlab005. [Google Scholar] [CrossRef]
- Delory, T.; Gravier, S.; Le Pluart, D.; Gaube, G.; Simeon, S.; Davido, B.; Piet, E.; Lepeule, R.; Lesprit, P.; Lafaurie, M. Temocillin versus carbapenems for urinary tract infection due to ESBL-producing Enterobacteriaceae: A multicenter matched case-control study. Int. J. Antimicrob. Agents 2021, 58, 106361. [Google Scholar] [CrossRef]
- Ciesielczuk, H.; Doumith, M.; Hope, R.; Woodford, N.; Wareham, D.W. Characterization of the extra-intestinal pathogenic Escherichia coli ST131 clone among isolates recovered from urinary and bloodstream infections in the United Kingdom. J. Med Microbiol. 2015, 64, 1496–1503. [Google Scholar] [CrossRef][Green Version]
- Alexandre, K.; de Rohello, F.L.; Dahyot, S.; Etienne, M.; Tiret, I.; Gillibert, A.; Pestel-Caron, M.; Caron, F. Efficacy of temocillin against MDR Enterobacterales: A retrospective cohort study. J. Antimicrob. Chemother. 2021, 76, 784–788. [Google Scholar] [CrossRef]
- Legge, J.S.; Reid, T.M.S.; Palmer, J.B.D. Clinical Efficacy, Tolerance and Pharmacokinetics of Temocillin in Patients with Respiratory Tract Infections. Drugs 1985, 29 (Suppl. 5), 118–121. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.M.; Leiper, J.M.; Lawson, D.H.; Cowan, W.; Baird, A.; Sleigh, J.D.; Gray, J.; Gray, J.M.B. Temocillin in the Treatment of Chest Infections. Drugs 1985, 29 (Suppl. 5), 197–200. [Google Scholar] [CrossRef] [PubMed]
- Verbist, L.; Verhaegen, J. Effect of temocillin in combination with other beta-lactam antibiotics. Antimicrob. Agents Chemother. 1984, 25, 142–144. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lekkas, A.; Gyi, K.M.; Hodson, M.E. Temocillin in the treatment of Burkholderia cepacia infection in cystic fibrosis. J. Cyst. Fibros. 2006, 5, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Habayeb, H.; Sajin, B.; Patel, K.; Grundy, C.; Al-Dujaili, A.; Van De Velde, S. Amoxicillin plus temocillin as an alternative empiric therapy for the treatment of severe hospital-acquired pneumonia: Results from a retrospective audit. Eur. J. Clin. Microbiol. 2015, 34, 1693–1699. [Google Scholar] [CrossRef]
- Layios, N.; Visée, C.; Mistretta, V.; Denooz, R.; Maes, N.; Descy, J.; Frippiat, F.; Marchand, S.; Grégoire, N. Modelled Target Attainment after Temocillin Treatment in Severe Pneumonia: Systemic and Epithelial Lining Fluid Pharmacokinetics of Continuous versus Intermittent Infusions. Antimicrob. Agents Chemother. 2022, 66, e02052-21. [Google Scholar] [CrossRef]
- Pfeiffer, M.; Fock, R.R.E. Therapeutic Experience with Temocillin in Peritonitis. Drugs 1985, 29 (Suppl. 5), 194–196. [Google Scholar] [CrossRef]
- Wise, R.; Donovan, I.A.; Drumm, J.; Dyas, A.; Cross, C. The intraperitoneal penetration of temocillin. J. Antimicrob. Chemother. 1983, 12, 93–96. [Google Scholar] [CrossRef]
- Wittke, M.R.R.; Adam, D.; Klein, H.E. Therapeutic Results and Tissue Concentrations of Temocillin in Surgical Patients1. Drugs 1985, 29 (Suppl. 5), 221–226. [Google Scholar] [CrossRef]
- Berleur, M.; Guerin, F.; Massias, L.; Chau, F.; Poujade, J.; Cattoir, V.; Fantin, B.; De Lastours, V. Activity of fosfomycin alone or combined with temocillin in vitro and in a murine model of peritonitis due to KPC-3- or OXA-48-producing Escherichia coli. J. Antimicrob. Chemother. 2018, 73, 3074–3080. [Google Scholar] [CrossRef]
- Woodnutt, G.; Catherall, E.J.; Kernutt, I.; Mizen, L. Influence of simulated human pharmacokinetics on the efficacy of temocillin against a Klebsiella pneumoniae meningitis infection in the rabbit. J. Chemother. 1989, 1 (Suppl. 4), 475–476. [Google Scholar]
- Brückner, O.; Trautmann, M.; Borner, K. A Study of the Penetration of Temocillin in the Cerebrospinal Fluid1. Drugs 1985, 29 (Suppl. 5), 162–166. [Google Scholar] [CrossRef] [PubMed]
- Woodnutt, G.; Catherall, E.J.; Kernutt, I.; Mizen, L. Temocillin efficacy in experimental Klebsiella pneumoniae meningitis after infusion into rabbit plasma to simulate antibiotic concentrations in human serum. Antimicrob. Agents Chemother. 1988, 32, 1705–1709. [Google Scholar] [CrossRef] [PubMed]
- Barton, E.; Flanagan, P.; Hill, S. Spinal infection caused by ESBL-producing Klebsiella pneumoniae treated with Temocillin. J. Infect. 2008, 57, 347–349. [Google Scholar] [CrossRef] [PubMed]
- Saylam, K.; Anaf, V.; Kirkpatrick, C. Successful medical management of multifocal psoas abscess following cesarean section: Report of a case and review of the literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2002, 102, 211–214. [Google Scholar] [CrossRef]
- Rodriguez, M.; Nelson, M.; Kelly, J.E.; Elward, A.; Morley, S.C. Successful Use of Temocillin as Salvage Therapy for Cervical Osteomyelitis Secondary to Multidrug-Resistant Burkholderia cepacia. J. Pediatr. Infect. Dis. Soc. 2014, 3, 77–80. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Duerinckx, J.F.H. Subacute Synovitis of the Knee After a Rose Thorn Injury: Unusual Clinical Picture. Clin. Orthop. Relat. Res. 2008, 466, 3138–3142. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barker, S.; Nichol, T.; Harrison, P.L.; Stockley, I.; Townsend, R.; Smith, T.J. Temocillin: A new candidate antibiotic for local antimicrobial delivery in orthopaedic surgery? J. Antimicrob. Chemother. 2014, 70, 780–783. [Google Scholar] [CrossRef][Green Version]
- Tjiam, K.H.; Van Heijst, B.Y.M.; Van Joost, T.; Stolz, E.; Michel, M.F. In vitro activity of temocillin (BRL 17421) against Chlamydia trachomatis. Eur. J. Clin. Microbiol. 1984, 3, 39–40. [Google Scholar] [CrossRef]
- Piot, P.; Van Dyck, E. In vitro activity of BRL 17421 against Haemophilus influenzae, Neisseria gonorrhoeae, and Branhamella catarrhalis. Antimicrob. Agents Chemother. 1982, 21, 166–167. [Google Scholar] [CrossRef]
- Reimer, G.; Milbradt, R.; Hulla, F.; Kammacher, D.; Hulla, F.W. Single Dose Therapy with Temocillin in Acute Gonorrhoea1. Drugs 1985, 29 (Suppl. 5), 210–212. [Google Scholar] [CrossRef] [PubMed]
- Coovadia, Y.M.; Ramsaroop, U. In vitro antimicrobial susceptibilities of penicillinase-producing and non-penicillinase-producing strains of Neisseria gonorrhoeae isolated in Durban, South Africa. Antimicrob. Agents Chemother. 1984, 26, 770–772. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ghathian, K.; Calum, H.; Gyssens, I.C.; Frimodt-Møller, N. Temocillinin vitroactivity against recent clinical isolates ofNeisseria gonorrhoeaecompared with penicillin, ceftriaxone and ciprofloxacin. J. Antimicrob. Chemother. 2016, 71, 1122–1123. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Raby, E.; Naicker, S.; Sime, F.B.; Manning, L.; Wallis, S.C.; Pandey, S.; Roberts, J.A. Ceftolozane-tazobactam in an elastomeric infusion device for ambulatory care: An in vitro stability study. Eur. J. Hosp. Pharm. 2020, 27, e84–e86. [Google Scholar] [CrossRef]
- Jones, B.M.; Huelfer, K.; Bland, C.M. Clinical and Safety Evaluation of Continuously Infused Ceftolozane/Tazobactam in the Outpatient Setting. Open Forum Infect. Dis. 2020, 7, ofaa014. [Google Scholar] [CrossRef]
- Carryn, S.; Couwenbergh, N.; Tulkens, P.M. Long-term stability of temocillin in elastomeric pumps for outpatient antibiotic therapy in cystic fibrosis patients. J. Antimicrob. Chemother. 2010, 65, 2045–2046. [Google Scholar] [CrossRef]
- Di Bella, S.; Beović, B.; Fabbiani, M.; Valentini, M.; Luzzati, R. Antimicrobial Stewardship: From Bedside to Theory. Thirteen Examples of Old and More Recent Strategies from Everyday Clinical Practice. Antibiotics 2020, 9, 398. [Google Scholar] [CrossRef]
- Laterre, P.-F.; Wittebole, X.; Van de Velde, S.; Muller, A.; Mouton, J.W.; Carryn, S.; Tulkens, P.M.; Dugernier, T. Temocillin (6 g daily) in critically ill patients: Continuous infusion versus three times daily administration. J. Antimicrob. Chemother. 2014, 70, 891–898. [Google Scholar] [CrossRef]
- Boon, R.J.; Beale, A.S. Studies with temocillin in a hamster model of antibiotic-associated colitis. Antimicrob. Agents Chemother. 1985, 27, 980–981. [Google Scholar] [CrossRef][Green Version]
- De Vries-Hospers, H.G.; Hofstra, W.; Welling, G.W.; Van der Waaij, D. Influence of temocillin on colonisation resistance and consequences for therapy. Drugs 1985, 29 (Suppl. 5), 227–233. [Google Scholar] [CrossRef]
- Mittermayer, H.; Mittermayer, H.W. Influence of Temocillin on Human Bowel Flora. Drugs 1985, 29 (Suppl. 5), 43–48. [Google Scholar] [CrossRef] [PubMed]
- Edlund, C.; Ternhag, A.; Ståhlgren, G.S.; Edquist, P.; Balkhed, Å.Ö.; Athlin, S.; Månsson, E.; Tempé, M.; Bergström, J.; Giske, C.G.; et al. The clinical and microbiological efficacy of temocillin versus cefotaxime in adults with febrile urinary tract infection, and its effects on the intestinal microbiota: A randomised multicentre clinical trial in Sweden. Lancet Infect. Dis. 2021, 28, 390–400. [Google Scholar] [CrossRef]
- Shafiq, N.; Pandey, A.K.; Malhotra, S.; Holmes, A.; Mendelson, M.; Malpani, R.; Balasegaram, M.; Charani, E. Shortage of essential antimicrobials: A major challenge to global health security. BMJ Glob. Health 2021, 6, e006961. [Google Scholar] [CrossRef] [PubMed]
- Tamma, P.D.; Aitken, S.L.; Bonomo, R.A.; Mathers, A.J.; van Duin, D.; Clancy, C.J. Infectious Diseases Society of America Guidance on the Treatment of Extended-Spectrum β-lactamase Producing Enterobacterales (ESBL-E), Carbapenem-Resistant Enterobacterales (CRE), and Pseudomonas aeruginosa with Difficult-to-Treat Resistance (DTR-P. aeruginosa). Clin. Infect. Dis. 2021, 72, e169–e183. [Google Scholar] [CrossRef] [PubMed]
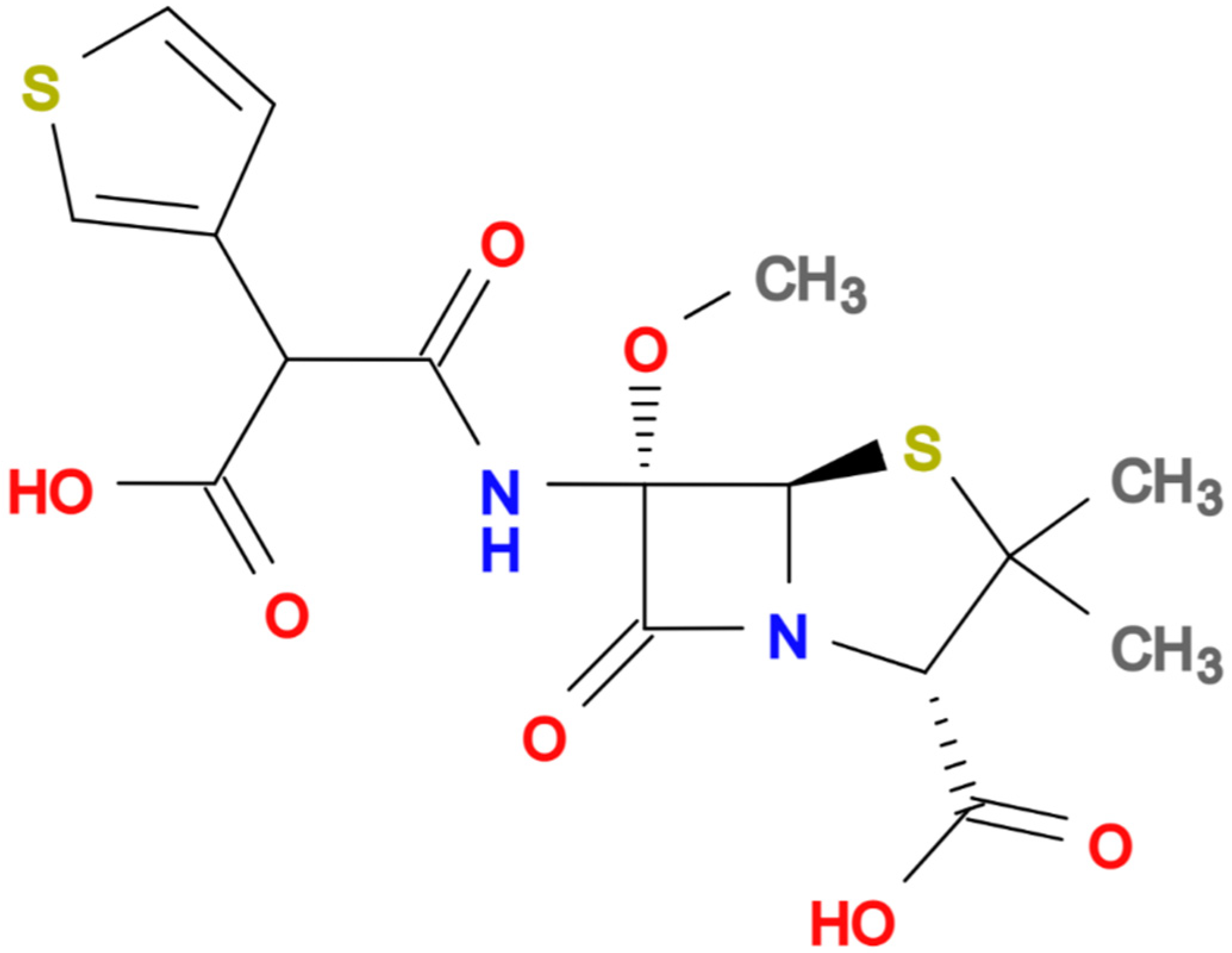
| Author, Year, and Reference | Study Design | Number of Patients | Antibiotic and Dosing | Source of Infection | Isolates | Clinical Outcomes |
|---|---|---|---|---|---|---|
| Kosmidis J, 1985 | Interventional Study | 33 | Temocillin, 500 mg q24h (IM) or 1 g q24h (IV), for 7 to 10 days | UTI and cUTI | E. coli (24), P. mirabilis (7), and E. cloacae (2) | Temocillin IM (Clinical cure 83% in UTI, Not effective in cUTI); IV (Clinical Cure 100% in UTI, 70% in cUTI) |
| Asbach HW et al., 1985 | Interventional Study | 29 | Temocillin, 500 mg q12h (IV), for 5 to 7 days | UTI and cUTI | E. coli (20), Proteus spp. (9), Klebsiella spp. (4), E. faecalis (2), S. epidermidis (1), and P. stuartii (1) | Clinical and microbiological cure 93% |
| Schulze B et al., 1985 | Open Clinical Study | 20 | Temocillin, 500 mg q12h (IV) for 7 to 10 days or 1 g q12h (IV), for 7 to 15 days | UTI, cUTI, LRTIs, and BSI | E. coli (14), M. catarrhalis (3), P. vulgaris (2), K. oxytoca (1), H. influenzae (1), H. haemolyticus (1), and E. aerogenes (1) | Clinical cure 100% in both groups |
| Lindsay G et al., 1985 | Interventional Study | 32 | Temocillin, 1 g q12h (IV or IM) for 7 to 14 days | UTI, cUTI, and LRTIs | E. coli (6), Klebsiella spp. (9), Enterobacter spp. (4), Proteus mirabilis (2), C. freundii (1), and H.alveii (1) | Clinical cure 78% |
| Pfeiffer et al., 1985 | Retrospective Study | 30 | Temocillin, 1 g q12h (IV) | IAIs, SSTIs, and LRTIs | E. coli (17), Proteus spp. (5), Enterococci (3), Pseudomonas spp. (3), K. pneumoniae (3), Citrobacter spp. (2), Bacteroides spp. (2), Streptococci (1), and Peptococci (1) | Clinical cure 77% |
| Legge et al., 1985 | Interventional Study | 13 | Temocillin, 500 mg q12h (IV) or 1 g q12h (IV) or 2 g q12h (IV) for 7 to 10 days | LRTIs | E. coli, Klebsiella spp., Acinetobacter species, P. mirabilis, H. influenzae, and H. haemoglobinophilus | Clinical cure 84.6% |
| Gray et al., 1985 | Interventional Study | 16 | Temocillina, 2–3 g day for 5–10 days | LRTIs | H. influenzae (8) and S. pneumoniae (5) | Clinical cure 81.25% |
| Saylam et al., 2002 | Case Report | 1 | NA | Vertebral Osteomyelitis, Pyomiositis, and CRBSI | K. pneumoniae, S. aureus | Complete clinical cure |
| Lekkas et al. 2005 | Interventional Study | 23 | Temocillin, 2–6 g day, 14 (range 1–40) | CF | B. cepacia | Clinical Improvement 56.25% |
| Duerinckx, 2008 | Case Report | 1 | Temocillin, 1 g q12h (IV) for 6 days | Synovitis | Pantoea agglomerans | Complete clinical cure |
| Barton et al., 2008 | Case Report | 1 | Temocillin, 2 g q12h (IV) for 12 weeks | Epidural abscess | ESBL K. pneumoniae | Complete clinical cure |
| Gupta et al., 2009 | Retrospective Study | 6 | Temocillin, 1 g q24h (IV), from 4 days to 24 months | UTI, cUTI, LRTIs, IAIs, and BSI | Klebsiella spp. (4), E. coli (1), and E. aerogenes (1) | Clinical cure 66% |
| Balakrishnan et al., 2011 | Retrospective Study | 92 | Temocillin 1 g q12h (IV) or 2 g q12h (IV) | UTI, cUTI, LRTIs, IAIs, and BSI | ESBL and/or dAmpC Enterobacterales (53) | Clinical Cure 86%; Microbiological Cure 84% |
| Rodriguez et al., 2013 | Case Report | 1 | NA | Osteomyelitis | B. cepacia | Complete clinical cure |
| Habayeb et al., 2015 | Interventional Study | 188 | Temocillin, 2 g q12h (IV) for 5–7 days vs. PTZ 4.5 g q8h for 5–7 days | LRTIs | NA | Clinical cure 82% |
| Laterre et al., 2015 | Randomized controlled Trial | 32 | Temocillin, 2 g q8h (IV) or 6 g (continous infusion) or CVVH | IAIs and LRTIs | E. coli (13), Klebsiella spp. (7) or Enterobacter spp. (5) | Clinical cure 79% (8 h), 93% (continuous infusion), and 75% (CVVH) |
| Alexandre et al., 2021 | Retrospective Study | 153 | Temocillin 2 g q8h (IV) or 2 g q12h (IV) | UTI, cUTI, LRTIs, IAIs, bone infections, and BSI | Enterobacterales (67.5% ESBL-producers) | Early Clinical Failure (UTI: 4.9%; non-UTI: 13.8%), Late Clinical Failure (UTI: 26.7%; non-UTI: 33.3%) |
| Heard et al., 2021 | Retrospective Study | 205 | Temocillin, 2 g q12h (IV) | UTI, cUTI, LRTIs, IAIs, bone infections, and BSI | E. coli (81.1% ESBL), Non-E. coli Enterobacterales (41.4% AmpC, 41.4% ESBL, 2.9% KPC) | Treatment Failure at 30 days: 20.5% |
| Delory et al., 2021 | Multicenter retrospective case-control study | 144 | Temocillin, 2 g q12h (IV) vs. Carbapenems (Ertapenem, Meropenem or Imipenem) (IV) | UTI and cUTI | ESBL Enterobacterales [K. pneumoniae (59), E. coli (57), and Enterobacter spp. (24)] | Clinical cure 94% (Temocillin Groups) and 99% (Carbapenem Comparators) |
| Edlund et al., 2022 | Randomised, multicentre, superiority, open-label, phase 4 trial | 152 | Temocillin 2 g q8h (IV) or Cefotaxime 1–2 g q8h, for 7 to 10 days | UTI e cUTI | Citrobacter spp. (5), Enterobacter spp. (2), Proteus spp. (5), Pseudomonas spp. (2), S. aureus (1), Aerococcus spp. (5), E. faecalis (5) | Clinical Cure 98% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupia, T.; De Benedetto, I.; Stroffolini, G.; Di Bella, S.; Mornese Pinna, S.; Zerbato, V.; Rizzello, B.; Bosio, R.; Shbaklo, N.; Corcione, S.; et al. Temocillin: Applications in Antimicrobial Stewardship as a Potential Carbapenem-Sparing Antibiotic. Antibiotics 2022, 11, 493. https://doi.org/10.3390/antibiotics11040493
Lupia T, De Benedetto I, Stroffolini G, Di Bella S, Mornese Pinna S, Zerbato V, Rizzello B, Bosio R, Shbaklo N, Corcione S, et al. Temocillin: Applications in Antimicrobial Stewardship as a Potential Carbapenem-Sparing Antibiotic. Antibiotics. 2022; 11(4):493. https://doi.org/10.3390/antibiotics11040493
Chicago/Turabian StyleLupia, Tommaso, Ilaria De Benedetto, Giacomo Stroffolini, Stefano Di Bella, Simone Mornese Pinna, Verena Zerbato, Barbara Rizzello, Roberta Bosio, Nour Shbaklo, Silvia Corcione, and et al. 2022. "Temocillin: Applications in Antimicrobial Stewardship as a Potential Carbapenem-Sparing Antibiotic" Antibiotics 11, no. 4: 493. https://doi.org/10.3390/antibiotics11040493
APA StyleLupia, T., De Benedetto, I., Stroffolini, G., Di Bella, S., Mornese Pinna, S., Zerbato, V., Rizzello, B., Bosio, R., Shbaklo, N., Corcione, S., & De Rosa, F. G. (2022). Temocillin: Applications in Antimicrobial Stewardship as a Potential Carbapenem-Sparing Antibiotic. Antibiotics, 11(4), 493. https://doi.org/10.3390/antibiotics11040493









