Bee Bread as a Promising Source of Bioactive Molecules and Functional Properties: An Up-To-Date Review
Abstract
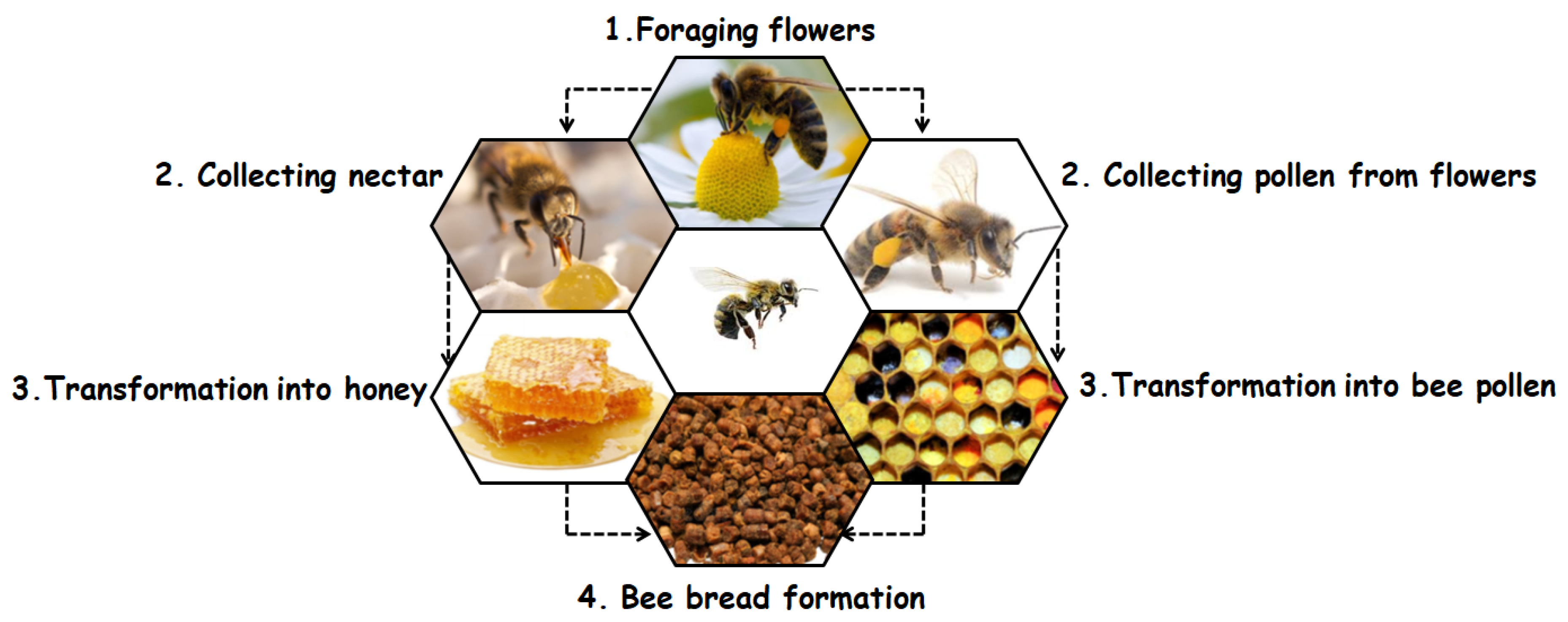
:1. Introduction
2. Methodology
3. Bee Bread Composition
3.1. Free Amino Acids
3.2. Sugars
3.3. Fatty Acids
3.4. Minerals
3.5. Organic Acids
3.6. Vitamins
3.7. Polyphenolic Composition
3.8. Microorganisms
3.9. Enzymes
| Component | Method Used | Country | Botanical Origin | References | |
|---|---|---|---|---|---|
| Free amino acids | Tryptophan, Taurine, l-Tyrosine, l-Phenylalanine, l-isoleucine, l-Leucine, Gamma-aminobutyric acid, 3-Amino isobutyric acid, l-Methionine, l-2-Aminoadipic acid, Beta-Alanine, l-Aspartic acid, l-Glutamic acid, l-Valine, l-2-aminobutyric acid, Ethanolamine, l-Alanine, l-Threonine, l-Serine, l-Glycine, l-Asparagine, l-Glutamine, l-Proline, Sarcosine, l-Arginine, l-Cystathionine, l-Cystine, Histidine, l-ornithine, l-Carnosine, l-Lysine, and l-Anserine | LC-MS/MS | Turkey | Five samples (not determined) | [15] |
| Phenylalanine, Valine, Histidine, Methionine, Isoleucine, Leucine, Threonine, Alanine, Arginine, Tyrosine, Glycine, Proline, Hydroxyproline, Serine, Glutamic acid, Aspartic acid, andLysine | Chromatographic separation | Malaysia | Four multifloral samples (Mimosa pudica, Sphagneticolatrilobata, Bidens ilosa, Cassia sp, Areca catechu, Peltophorumpterocarpum, Phaleria capitata, Cassia siamea, Citrus aurantifolia, and Ageratum conyzoides) | [11] | |
| Aspartate, Glutamate, Asparagine, Serine, Glutamine, Histidine, Glycine, Threonine, Arginine, Alanine, γ-aminobutyric acid, Tyrosine, Cysteine, Valine, Methionine, Tryptophan, Phenylalanine, Isoleucine, Leucine, Lysine, and Proline | UHPLC | England | Fifty-one samples of bee bread (Trifolium, Impatiens, Rubus, Acer, Cirsium, Euscaphis, Cryptotaenia, Glycine, Coriandrum, Rosa, Prunus, Taraxacum, Camelina, Ranunculus, Salix, and Andira) | [13] | |
| Alanine, Aspartic acid, Glutamine, Serine, Leucine, Iso-leucine, Methionine, Threonine, Valine, Tryptophan, Cysteine, Phenylalanine, and Proline. | (GC/MS) | USA | Two samples (not determined) | [14] | |
| Alanine, Arginine, Aspartic acid, Glutamic acid, Glycine, Histidine, Hydroxyproline, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Proline, Serine, Threonine, Tyrosine, and Valine | Waters AccQ Tag method | Malaysia | Three samples (not determined). | [12] | |
| Sugars | Trehalose, Glucose, and Fructose | HPLC-RID | Morocco | One multifloral sample (Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia) | [20] |
| Fructose, Glucose, Melezitose, andRaffinose | HPLC-RID | Romania | One sample (not determined). | [21] | |
| Fructose, Glucose, Turanose, and Maltose | HPLC-DAD | Romania and India | Five samples (Brassicaceae, Poaceae, Myrtaceae, Rutaceae, Asteraceae, Fabaceae, Tiliaceae, Fabaceae, Rosaceae, Plantaginaceae, Fabaceae, Asteraceae, Lamiaceae, Salicaceae, Rosaceae, and Fagaceae) | [22] | |
| Fructose, Glucose, Sucrose, and Maltose | HPLC coupled with an ELSD | Malaysia | Four multifloral samples (Mimosa pudica, Sphagneticolatrilobata, Bidens ilosa, Cassia sp, Areca catechu, Peltophorumpterocarpum, Phaleria capitata, Cassia siamea, Citrus aurantifolia, and Ageratum conyzoides) | [11] | |
| Microorganisms | Lactobacillus kunkeei | RAPD-PCR | Turkey | Four samples (not identified). | [60] |
| Cladosporium, Penicillium, Alternaria, Monilinia, Sclerotina, Ascosphaera, Toxicocladosporium, Botrytis, Pseudopithomyces, Camerosporium, Paraconiothyrium, Podosphaera, Golovinomyces, Paraphaeosphaeria, Periconia, and Septoriella | NGS-sequencing | Bulgaria | Four samples (not identified). | [54] | |
| Apilactobacilluskunkeei, Lactiplantibacillus plantarum, Fructobacillusfructosus, Levilactobacillus brevis, and Lactobacillus delbrueckii | PCR-DGGE analysis RAPD-PCR analysis | Italy | Twelve samples (not identified) | [55] | |
| Organic acids | Oxalic acid | UFLC-PDA | Morocco | One multifloral sample (Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia) | [20] |
| Gluconic acid, Formic acid, Lactic acid, Acetic acid, Succinic acid, Propionic acid, Butyric acid | HPLC-DAD | Romania | One sample (not determined) | [21] | |
| Vitamins | Vitamin A, Vitamin E, Thiamine, andRiboflavin | HPLC | Malaysia | One sample (not determined) | [30] |
| Vitamin C | Method AOAC 967.21 | Malaysia | Four multifloral samples (Mimosa pudica, Sphagneticolatrilobata, Bidens pilosa , Cassia sp, Areca catechu, Peltophorumpterocarpum, Phaleria capitata, Cassia siamea, Citrus aurantifolia, and Ageratum conyzoides) | [11] | |
| α-Tocopheroland δ-Tocopherol. | HPLC | Morocco | One multifloral sample (Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia) | [20] | |
| Octanoic, Decanoic, Undecanoic, Dodecanoic, Tridecanoic, Tetradecanoic, Pentadecanoic, Hexadecanoic, Palmitoleic, Heptadecanoic, Stearic, Oleic, Linoleic, α-Linolenic, Arachidic, Gadoleic, Eicosadienoic, Heneicosanoic, Eicosatrienoic, Arachidonic, Eicosatrienoic, Behenic, Eicosapentaenoic, Lignocerin, andNervonicacid. | GC-FID | Morocco | [20] | ||
| Fatty acids | Butanoic, Hexanoic, Octanoic, Decanoic, Undecanoic, Dodecanoic, Tridecanoic, Tetradecanoic, Pentadecanoic, Hexadecanoic, Heptadecanoic, Octadecanoic, Icosanoic, Heneicosanoic, Docosanoic, Tricosanoic, Tetracosanoic, (Z)-tetradec-9-enoic, (Z)-pentadec-10-enoic, (9Z)-hexadec-9-enoic, cis-10-heptadecenoic, (E)-octadec-9-enoic, (Z)-octadec-9-enoic, (Z)-icos-11-enoic, (Z)-docos-13-enoic, (Z)-tetracos-15-enoic, octadeca-9,12-dienoic, (9Z,12Z)-octadeca-9,12-dienoic, Octadeca-6,9,12-trienoic, Icosa-11,14-dienoic, (11Z,14Z,17Z)-icosa-11,14,17-trienoic, (5Z,8Z,11Z,14Z)-icosa-5,8,11,14-tetraenoic, Docosa-13,16-dienoic, (9Z,12Z,15Z)-octadeca-9,12,15-trienoic, (11Z,14Z,17Z)-icosa-11,14,17-trienoic, (5Z,8Z,11Z,14Z,17Z)-icosa-5,8,11,14,17-pentaenoic, and Docosa-4,7,10,13,16,19-hexaenoic | GC-MS | Turkey | Eight monofloral samples (Trifolium pratense, Gossypium hirsutum, Castanea sativa, Citrus spp., Helianthus annuus, and Trifolium repens) | [25] |
| Butanoic acid, Hexanoic acid, Octanoic acid, Decanoic acid, Undecanoic acid, Dodecanoic acid, Tridecanoic acid, Tetradecanoic acid, (cis-9) (Z)-tetradec-9-enoic acid, Pentadecanoic acid, (cis-10) (Z)-pentadec-10-enoic, Hexadecanoic acid, (9Z)-hexadec-9-enoic acid, Heptadecanoic acid, cis-10-heptadecenoic, Octadecanoic acid, (E)-octadec-9-enoic, (Z)-octadec-9-enoic, (all-trans-9,12) Octadeca-9,12-dienoic acid, (all-cis-9,12) (9Z,12Z)-octadeca-9,12-dienoic acid, (all-cis-6,9,12) Octadeca-6,9,12-trienoic acid, (all-cis-9,12,15) Octadeca-6,9,15-trienoic acid, Icosanoic acid, (cis-11) (Z)-icos-11-enoic acid, (all-cis-11,14,) Icosa-11,14-dienoic acid, (all-cis-8,11,14) Icosa-8,11,14-trienoic acid, (all-cis-11,14,17) Icosa-11,14,17-trienoic acid, (all-cis-5,8,11,14) Icosa-5,8,11,14-tetraenoic acid, (all-cis-5,8,11,14,17) Icosa-5,8,11,14,17-pentaenoic, Heneicosanoic acid, Docosanoic acid, (cis-13) (Z)-docos-13-enoic, (all-cis-13,16) Docosa-13,16-dienoic, (all-cis-4,7,10,13,16,19) Docosa-4,7,10,13,16,19-hexaenoic, Tricosanoic acid, Tetracosanoic acid, and (cis-15) (Z)-tetracos-15-enoic | GC-MS | Romania | One sample (not determined) | [21] | |
| Butyric acid, Caproic acid, Caprylic acid, Capric acid, Undecanoic acid, Lauric acid, Tridecanoic acid, Myristic acid, Pentadecanoic, Palmitic acid, Heptadecanoic acid, Stearic acid, Arachidic acid, Heneicosanoic, Behenic acid, Tricosanoic acid, Lignoceric acid, Myristoleic acid, cis-Pentadecanoic acid, Palmitoleic acid, cis-Heptadecanoic acid, Elaidic acid, Oleic acid, cis-Eicosenoic, Erucic acid, Nervonic acid, Linolelaidic acid, Linoleic acid, g-Linolenic acid, cis-11,14-Eicosadienoic, cis-8,11,14-Eicosatrienoic acid, Arachidonic acid, cis-13,16 Docosadienoic, a-Linolenic acid, cis-11,14,17-Eicosatrienoic acid, cis-5,8,11,14,17-Eicosapentaenoic acid, and Docosahexaenoic acid | GC-FID | Turkey | In five monofloral samples, the majority of pollen is Citrus spp. Secondary pollens belonging to Asteraceae, Fabaceae, and Brassicaceae families | [24] | |
| Minerals | Ca, Fe, K, Mg, Na, Zn, P, Pb, Ni, Se, Mn, Co, Cu, and Cd | ICP-AES | Morocco | One multifloral sample (Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia) | [20] |
| Ca, Fe, K, Mg, Mn, Na, Zn, P, and Se. | ICP-MS | Malaysia | Four multifloral samples (Mimosa pudica, Sphagneticolatrilobata, Bidens ilosa, Cassia sp, Areca catechu, Peltophorumpterocarpum, Phaleria capitata, Cassia siamea, Citrus aurantifolia, and Ageratum conyzoides | [11] | |
| Na, Mg, K, Ca, Mn, Se, Fe, Zn, and Cu. | ICP-MS | Serbia | Twelve bee bread samples (not determined) | [31] | |
| Ca, P, K, Mg, Fe, Zn, and Mn | AAS | Malaysia | One sample (not determined) | [30] | |
| Polyphenols composition | Kaempferol-3-O-glycoside, Hydroxycinnamic acid derivative, Quercetin-3-O-sophoroside, Kaempferol-3-O-derivative, Hydroxycinnamic acid derivative, Myricetin, Trycetin, Luteolin, Herbatin-3-O-glycoside, Quercetin-3-O-glycoside, and Isoramnethin-3-O-glycoside | HPLC-DAD | Romania and India | Five samples (Brassicaceae, Poaceae, Myrtaceae, Rutaceae, Asteraceae, Fabaceae, Tiliaceae, Fabaceae, Rosaceae, Plantaginaceae, Fabaceae, Asteraceae, Lamiaceae, Salicaceae, Rosaceae, and Fagaceae) | [22] |
| p-Coumaric acid, Kaempferol, Chrysin, and Apigenin | HPLC | Lithuania | Nine simples (not determined) | [48] | |
| Caffeic acid, p-Coumaric acid, Rosmarinic acid, Myricetin, Luteolin, Quercetin, and Kaempferol | HPLC-DAD | Romania | One sample (not determined) | [21] | |
| Naringin, Rutin, and Quercetin. | HPLC | Georgia | Two samples | [49] | |
| Kaempferol and Apigenin | GC/MS | Poland | Three samples. | [50] | |
| Kaempferol-O-hexosyl-O-rutinoside, Quercetin-O-hexosyl-O-hexoside, Quercetin-O-hexosyl-O-hexoside, Methylherbacetrin-O-dihexoside, Isorhamnetin-O-hexosyl-O-rutinoside, Quercetin-O-pentosyl-hexoside, Quercetin 3-O-rutinoside, Methylherbacetrin-3-O-rutinoside, Isorhamnetin-O-pentosyl-hexoside, Isorhamnetin-O-pentosyl-hexoside, Kaempferol-3-O-rutinoside, Isorhamnetin-O-rhamnoside-hexoside, and Isorhamnetin-3-O-rutinoside | LC-DAD–ESI/MSn | Morocco | One multifloral sample (Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia) | [20] | |
| Hesperetin, Quercetin-O-hexosyl-O-rutinosid, Quercetin-diglucoside, Methyl herbacetin-O-dihexoside, Kaempferol-O-dihexoside, Methyl herbacetin-O-rutinoside, Isorhamnetin-O-pentosyl-hexosid, Kaempferol-3-O-rutinoside, Quercetin-3-O-glucoside, Quercetin-O-malonyl hexoside, Kaempferol-O-malonyl hexoside, Di-p-coumaroylspermidine, Kaempferol-3-O-rhamnoside, and Isorhamnetin-O-deoxyhexoside | LC/DAD/ESI-MS | Portugal | Three samples. Sample 1: Plantago spp. (47%) Sample 2: Crepiscapillaris (60%) Sample 3: Cytisus striatus (48%) | [7] | |
| 2,5-Dihydroxybenzoic acid, 2-Hydroxycinnamic acid, Caffeic acid, Catechin, Epicatechin, Chlorogenic acid, Ethyl gallate, Gallic acid, Isorhamnetin, Kaempferol, Luteolin, Myricetin, Naringin, p-Coumaric acid, Phlorizin, Propyl gallate, Protocatechuic acid, Quercetin, Resveratrol, Rutin, Salicylic acid, Sinapic acid, Syringic acid, Trans ferulic acid | LC-MS/MS | Turkey | Five samples (not identified). | [15] | |
| Myricetin-3-O-rutinoside, Quercetin-O-hexosyl-O-rutinoside, Kaempfrol-O-hexosyl-O-rutinoside,Quercetin-O-hexosyl-O-hexoside, Isorhamnetin-O-hexosyl-O-rutinoside, Methyl herbacetrin-O-dihexoside, Myricetin-3-O-glucoside, Quercetin-O-pentosyl-hexoside, Quercetin-O-hexosyl-rutinoside, Quercetin 3-O-rutinoside, Methyl herbacetrin-O-hexosyl-rutinoside, Kaempferol-O-dihexoside, Methyl herbacetrin-3-O-rutinoside, Methyl herbacetrin-O-dihexoside, Kaempferol-O-hexosyl-rutinoside, Isorhamnetin-O-pentosyl-hexoside, Isorhamnetin-O-hexosyl-rutinoside, Kaempferol-3-O-rutinoside, Quercetin-3-O-glucoside, Isorhamnetin-o-pentosyl-hexoside, Isorhamnetin-O-pentosyl-hexoside, Acetyl kaempferol-O-deoxyhexosyl-hexoside, Methyl herbacetrin-3-O-glucoside, Laricitrin-3-O-rhamnoside, Isorhamnetin-3-O-rutinoside, Quercetin-3-O-rhamnoside, Kaempferol-o-pentosyl-deoxyhexoside, Isorhamnetin-3-O-glucoside, Acetyl kaempferol-O-hexoside, Kaempferol-3-O-rhamnoside, Isorhamnetin-3-O-rhamnoside, Acetyl isorhamnetin-O-hexoside | HPLC-DAD-ESI/MS | Portugal | Six samples (not identified) | [46] | |
4. The Bioactive Effect of Bee Bread: In Vitro Investigations
4.1. Antioxidant Capacity of Bee Bread
4.2. The Antitumoral Effect of Bee Bread
4.3. Hypotensive Effect
4.4. The Inhibition Effect of Carbohydrate-Hydrolyzing Enzymes
| Country | Functional Effect | The Majority of Pollen Grains Identified in Bee Bread (BB) | Extraction Used | Concentration | Extraction Time | Results Obtained | References |
|---|---|---|---|---|---|---|---|
| Malaysia | Antioxidant | One sample, not identified | Hexane and 70% ethanol using Soxhlet apparatus | Sixty gramsof sample extracted with 300 mL of solvent | 2 h | DPPH value of ethanolic extract (%): 93.60 ± 0.03 DPPH value of hexanic extract (%): 83.81 ± 0.05 ABTS value of ethanolic extract (%): 97.95 ± 0.01 ABTS value of hexanicextract (%): 71.23 ± 0.01 FRAP value of ethanolic extract (mM FE/g): 0.85 ± 0.01 FRAP value of hexanic extract (mM FE/g): 2.41 ± 0.02 | [61] |
| Portugal | Antioxidant | Three samples Sample 1: Plantago sp. (47%) Sample 2: Crepis capillaris (60%) Sample 3: Cytisus striatus (48%) | EtOH/H2O (80:20, v/v) | Two grams of sample was extracted with 40 mL of EtOH/H2O (80:20, v/v) | Total phenolic content value ranging between 3.2 and 3.8 ± 0.1 mg GAE/g Total flavonoid content value ranging between 0.6 and 2.7 mg QE/g | [47] | |
| Ukraine | Antioxidant | Five samples | Methanol/water solution (70%, v/v) | A total of 0.1 g of sample in 5 mL of methanol/water solution (70%, v/v) | Total phenolic content value ranging between 12.36 and 25.44 mg GAE/g Total flavonoid content ranging between 13.56 and 18.24 µg QE/g | [72] | |
| Lithuania | Antioxidant | Four samples | Not mentioned | Not mentioned | Not mentioned | Total phenolic content value ranging between 306 and 394 mg GAE/100g DPPH radical scavenging abilities ranged between 85 and 93% | [73] |
| Colombia | Antioxidant | 15 samples, not identified | Ethanol (96% v/v) | One gram of sample extracted with 30 mL of solvent | 24 h | FRAP value ranging between 35.0 and 70.1 μmoltrolox/g TEAC value ranging between 46.1 and 76.3 μmoltrolox/g Total flavonoid content value ranging between 1.9 and 4.5 mg eq-quercetine/g Total phenolic content value ranging between 2.5 and 13.7 mg eq-gallic acid/g | [62] |
| Lithuania | Antioxidant | One sample (not identified) | Three types of extraction: Hot distilled water Distilled water Ethanol | Extract 1: 3 g of BB was extracted by boiling with 10 volumes of distilled water. Extract 2: 3 g of BB was extracted by shaking with 10 volumes of distilled water. Extract 3: 3 g of BB with 10 volumes of ethanol | Extract 1: 1h Extract 2: 1 day Extract 3: 1 day | Superoxide anion radical scavenging abilities ranged between 9.92% and 100% Hydroxyl radical scavenging abilities ranged between 9.75 and 100% DPPH radical scavenging abilities ranged between 10.5 l ± 0.2% and 98.7 ± l 0.2% Antioxidant abilities ranged between 0.018 l ± 0.002 and 1.352 ± l 0.036% | [68] |
| Morocco | Antioxidant | One sample (not identified) | Ethanolic extraction (70%) | One gram of BB macerated in 20 mL of ethanol (70%) | 1 week | Polyphenol content was 14.88 ± 0.98 mg GAE/g, flavonoid content was 1.67 ± 0.12 mg QE/g, total antioxidant capacity was 143.78 ± 11.38 mg AAE/g, IC50 of DPPH was 0.05 ± 0.01 mg/mL, IC50 of ABTS was 0.08 ± 0.05 mg/mL, and reducing power was 0.05 ± 0.04 mg/mL | [63] |
| Morocco | Antioxidant | One multifloral sample (35% Anethum graveolens, 24% Quercus ilex, 16% Eucalyptus, and 25% other pollens) | Hydromethanolic extract | One gram of BB extracted twice with 30 mL of a mixture of methanol/water (80:20 v/v) | 60 min | Total antioxidant capacity: 143 ± 22 DPPH: 0.98 ± 0.06 ABTS: 0.50 ± 0.04 Reducing power: 0.19 ± 0.03 | [20] |
| Ukraine | Antioxidant | Five samples (not determined) | BB was extracted with ethanol | A total of 0.1 g of BB was extracted with 20 mL of ethanol (80%) | 2 h | Total polyphenol content ranged between 12.36 ± 0.34 mg GAE/g and 25.44 ± 0.22 mg GAE/g Total flavonoid content ranged between 13.56 ± 0.04 μg QE/g and 15.35 ± 0.09 μg QE/g The best value of the DPPH test was 15.78 mg TEAC/g The value for reducing power was 250 mg TEAC/g | [66] |
| Lithuania | Antioxidant | Nine samples (not determined) | Extraction of phenolic compounds using distilled water, methanol, and diethyl ether | Fifty grams of BB was extracted with 250 mL of distilled water and 250 mL of methanol; the residue obtained was dissolved in 5 mL of distilled water and extracted with 5 mL of diethyl ether (three times) | Not determined | DPPH values ranged between 64.2 ± 1.8% and 93.9 ± 0.6% ABTS values ranged between 76.5 ± 0.2% and 94.8 ± 0.5% | [48] |
| Morocco | Antioxidant | One sample (not identified) | BB was extracted with ethyl acetate | - | Not determined | Polyphenols: 27.27 ± 0.38 mg EqGA/g Flavonoids: 5.29 ± 0.27 mg EqQ/g TAC: 65.44 ± 6.34 mg EqAA/g IC50 of ABTS:1.52 ± 0.021 mg/mL IC50 of DPPH:0.43 ± 0.02 mg/mL EC50 of RP:0.71 ± 0.05 mg/ml | [64] |
| Poland | Antioxidant | Three samples (not identified) | Ethanolic Extract | Twenty grams were extracted with 80 g of 95% (v/v) ethanol and re-extracted with 40.0 g of 95%(v/v) ethanol | 12 h | Polyphenolic content: ranged between 33.43 ± 0.7 and 36.52 ± 0.6 mg GAE/g Antioxidant activity: ranged between 0.56 ± 0.06 and 1.11 ± 0.09 mmol/L | [50] |
| Antitumoral | The cytotoxicity of BB was studied using a glioblastoma cell line (U87MG), the results indicated a time-dependent inhibitory effect on the viability of U87MG cells treated with BB. The cell viability was decreased to 49–66% after 72 h. | ||||||
| Turkey | α-amylase inhibition | One sample; predominant pollen was Trifolium pretense (70.39%), important minor pollen (3–15%) presented by Cistaceae, Asteraceae, Rosaceae, and others (<3%) | BB was frozen, powdered, and then extracted with methanol | Two grams was mixed with 10 mL of methanol | 48h under stirring | IC50 of α-amylase inhibition was 3.57 ± 0.01, better than acarbose (IC50 = 5.93 ± 0.01) | [67] |
| Lithuania | Angiotensin I-converting enzyme inhibition | One sample (not determined) | Preparation of enzymatic hydrolysates from bee bread by digestion with three kinds of enzymes (pepsin, trypsin, and papain) | 10% (Pepsin hydrolysate) 10% (Trypsin hydrolysate), 4% (Papain hydrolysate) | Not determined | IC50 of angiotensin I-converting enzyme inhibition of pepsin hydrolysate: 1.48 mg protein/mL IC50 of angiotensin I-converting enzyme inhibition of trypsin hydrolysate: 2.16 mg protein/mL IC50 of angiotensin I-converting enzyme inhibition of papain hydrolysate: 5.41 mg protein/mL | [69] |
| Portugal | Antitumor activity | Six samples, not determined | BB was lyophilized and extracted twice with methanol:water (80:20, v/v) | One gram of BB in 30 mL of solvent | 1h | Three samples of BB hadcytotoxic activity against MCF-7 (breast adenocarcinoma), IG50 values were: 186 ± 6 µg/mL; 84 ± 3 µg/mL; and 164 ± 4 µg/mL. Three samples of BB had cytotoxic activity against NCI-H460 (non-small cell lung cancer), IG50 values were:253 ± 10 µg/mL; 85 ± 5 µg/mL; and 68 ± 8 µg/mL. Four samples of BB hadcytotoxic activity against HeLa (cervical carcinoma), IG50 values were:345 ± 13 µg/mL; 225 ± 12 µg/mL; 209 ± 21 µg/mL; and 366 ± 7 µg/mL. One sample of BB had cytotoxic activity against HepG2 (hepatocellular carcinoma), IG50 value was 67 ± 1 µg/mL. None of the BB samples had toxicity against non-tumor liver cells (porcine liver cells, PLP2). | [46] |
| Turkey | Antioxidant | One sample; predominant pollen was Trifolium pretense (70.39%), important minor pollen (3–15%) presented by Cistaceae, Asteraceae, Rosaceae, and others (<3%) | BB was frozen, powdered, and then extracted with methanol | Two grams was mixed with 10 mL of methanol | 48 h under stirring | Total phenolics:6.93 ± 0.09 mg GAE/g; Total flavonoids:2.27 ± 0.05 mg QE/g; Antioxidant capacity:83.62 ± 0.33 μmol FeSO4 7 H2O/g sample | [67] |
| Malaysia | Antioxidant | Three samples, not identified | Distilled water or ethanol 70% | Fifty grams of bee bread extracted with 10 volumes of distilled water or 70% of ethanol | 72 h | DPPH value of aqueous extract ranging between 2.86% and 8.95% DPPH value of ethanolic extract ranging between 72.04% and 79.34% FRAP value of aqueous extract ranging between 0.94 mmol Fe2/L and 1 mmol Fe2/L FRAP value of ethanolic extract ranging between 1.07 mmol Fe2/L and 1.08 mmol Fe2/L Total flavonoid content of aqueous extract ranging between 2.88 mg QE/g and 3.92 mg QE/g Total flavonoid content of ethanolic extract ranging between 16.48 mg QE/g and 26.57 mg QE/g Total phenolic content of aqueous extract ranging between 14.19 mg GAE/g and 15.38 mg GAE/g Total phenolic content of ethanolic extract ranging between 21.32 mg GAE/g and 22.54 mg GAE/g | [12] |
4.5. Probiotic Properties of Bee Bread
4.6. Antimicrobial Activity of Bee Bread
4.7. Proposed Mechanisms of Antimicrobial Action
| Effect | Country | Type of the Study | Bacteria/Fungi/Virus Strains | Extract Used | Key Results | References |
|---|---|---|---|---|---|---|
| Antibacterial | Morocco | In vitro | Gram-positive bacteria: Bacillus cereus (food isolate), Staphylococcus aureus (ATCC 6538), and Listeria monocytogenes (NCTC 7973) Gram-negative bacteria: Escherichia coli (ATCC 35210), Enterobacter cloacae (human isolate), and Salmonella typhimurium (ATCC13311) | Hydromethanolic extract One gramof BB extracted twice with 30 mL of a mixture of methanol/water (80:20 v/v) for 60 min | Gram-positive bacteria: MIC values ranging between 0.4 and 0.175 mg/mL MBC values ranging between 0.08 and 0.35 mg/mL Gram-negative bacteria: MIC values ranging between 0.175 and 0.25 mg/mL MBC values were 0.35 mg/mL for all strains | [20] |
| Antifungal | Aspergillus fumigatus (ATCC 1022), Aspergillus ochraceus (ATCC 12066), Aspergillus niger (ATCC 6275), Penicillium funiculosum (ATCC 36839), Penicillium ochrochloron (ATCC 9112), and Penicillium verrucosum var. cyclopium (food isolate) | MIC values ranged between 0.35 mg/mL and 1 mg/mL, and MFC values ranged between 0.70 mg/mL and 1.40 mg/mL | ||||
| Antibacterial | Morocco | In vitro | Gram-positive bacteria: Staphylococcus aureus, three strains of Streptococcus, Bacillus cereus Gram-negative bacteria: Three strains of Escherichia coli, Salmonella enteritidis, three strains of Pseudomonas aeruginosa. | DMSO (10%) 50%(v/v) | Gram-positive bacteria: The inhibition diameters ranged between 9 and 27 mm Gram-negative bacteria: The inhibition diameters ranged between 9 and 29 mm | [83] |
| Antibacterial | Lithuania | In vitro | Gram-positive bacteria: B. cereus 1801, methicillin-resistant Staphylococcus aureus (MRSA) M87fox, E. faecalis 86, S. epidermidis, S. haemolyticus, and Streptococcus mutans Gram-negative bacteria: A. baumannii17-380, C. freundii, E. cloacae, Enterococcus faecium 103, Klebsiella pneumoniae,P. multocida, P. mirabilis, Pseudomonas aeruginosa 17-331, and Salmonella enterica 24 SPn06. | One gram of bee products was dissolved in 20 mL of aqueous ethanol (500 mL/L) for 6 h | The inhibition diameters ranged between 9 and 22.8 mm | [73] |
| Greek | In vitro | Gram-positive bacteria: Methicillin-resistant Staphylococcus aureus strain 1552 Gram-negative bacteria: Carbapenem-resistant Pseudomonas aeruginosa strain 1773, Salmonella typhimurium, and Klebsiella pneumonia | Different amounts of bee bread were extracted with 10 mL of water at room temperature for 24 | Gram-positive bacteria: MIC values ranging between 3.9 mg/mL and 48 mg/mL MBC values ranging between 3.9 mg/mL and 48 mg/mL Gram-negative bacteria: MIC values ranging between 7.8 mg/mL and 90.4 mg/mL MBC values ranging between 9.9 mg/mL and >93.2 mg/mL | [84] | |
| Poland | In vitro | Gram-positive bacteria: S. aureusATCC 25923, S. aureus ATCC 29213, S. epidermidisATCC 12228, six MSSA (methicillin-susceptible Staphylococcus aureus), and three MRSA (methicillin resistant Staphylococcus aureus) Gram-negative bacteria: P. aeruginosa ATCC 27853 and E. coli ATCC 25922 | BB extracted in 70% ethanol at v/w ratio of 7:1 at ambient temperature for 2 h | Gram-positive bacteria: MIC values ranging between 2.5 and 10% (v/w). MBC values ranging between 2.5 and 20% Gram-negative bacteria: MIC values ranging between 5 and 20% (v/w) MBC values ranging between 10 and >20% (v/w) | [85] | |
| Antibacterial | Ukraine | In vitro | Gram-negative bacteria: Escherichia coli CCM 3988 andSalmonella enterica subs. Enterica CCM 3807 Gram-positive bacteria: Bacillus thuringiensis CCM 19 and Staphylococcus aureus subs. aureus CCM 4223. | BB was extracted with ethanol; 0.1 g of BB was extracted with 20 mL of ethanol (80%) for 2 h | Gram-positive bacteria: MICvalues ranging between 12.81 and 27.20 μg/mL Gram-negative bacteria: MIC values ranging between 6.40 and 13.64 μg/mL | [66] |
| Antiviral | Greece | Cell culture method | Enterovirus D68 | BB was dissolved in Dulbecco’s Modified Eagle cell culture medium for one hour at room temperature | IC50 ranged between 0.048 and 5.45 mg/mL CC50 ranged between 0.17 and 8.60 mg/mL | [90] |
| Antibacterial | Malaysia | In vitro | Gram-positive bacteria: L.monocytogene, S. aureus,and B. cereus Gram-negative bacteria: E.coli, Salmonella, and P. aeruginosa | Twenty grams of BB were extracted twice with 80 g of 95% (v/v) ethanol for 12 h and then with 40g of 95% (v/v) ethanol. | Gram-positive bacteria: The inhibition diameters ranged between 0 and 284.44 mm Gram-negative bacteria: The inhibition diameters ranged between 0 and 312.22 mm | [86] |
| Antibacterial | Malaysia | In vitro | Gram-negative bacteria: Klebsilla pneumonia, Escherichia coli, Shigella, and Salmonella typhi | Fifty grams of BB were extracted with 500 mL of 70% ethanol and with water and hot water for 72 h at room temperature | Shigella (MIC: 1.617 µg/mL), followed by Salmonella typhi (MIC: 1.813 µg/mL), E. coli (MIC: 1.914 µg/mL), and Klebsilla pneumonia (MIC: 1.923 µg/mL) | [87] |
| Antibacterial | Egypt | In vitro | Gram-positive bacteria: Staphylococcus aureus (ATCC 6538) and Bacillus Subtilis(ATCC 6633) Gram-negative bacteria: Escherichia coli (ATCC 8739) and K. pneumonia (ATCC 13883) | One hundred grams of BB were soaked in 200 mL of ethanol 80% for 24 h and then homogenized for 30 min | Gram-positive bacteria: The inhibition diameters ranged between 24 and 26 mm Gram-negative bacteria: The inhibition diameters ranged between 12 and 18 mm | [52] |
| Antifungal | Unicellular fungi Candida albicans (ATCC 10221) and filamentous fungi Asp. niger | The inhibition diameter for Candida albicans was 15 ± 0.73 mm No effect against Aspergillus niger | ||||
| Antibacterial | Ukraine | In vitro | Gram-positive bacteria: Bacillus cereus CCM 2010, Clostridium perfringens CCM 4435, and Staphylococcus aureus subsp. aureus CCM 4223 Gram-negative bacteria: Haemophilus influenza CCM 4456, Klebsiella pneumoniae CCM 2318, and Salmonella enterica subsp. enterica CCM 3807 | BB was extracted by maceration using 50% ethanol | Gram-positive bacteria: The inhibition diameters ranged between 9 and 16 mm Gram-negative bacteria: The inhibition diameters ranged between 1.7 and 5 mm | [88] |
| Antifungal | Candida albicans CCM 8186, Candida glabrata CCM 8270, Candida tropicalis CCM 8223, Aspergillus clavatus, Aspergillus flavus, Aspergillus versicolor, Penicillium expansum, Penicillium chrysogenum, and Penicillium griseofulvum | Candia species: The inhibition diameters ranged between 4 and 8 mm Fungi species: The inhibition diameters ranged between 1 and 3.7 mm |
5. The Pharmacological Effect of Bee Bread: In Vivo Investigations
| Country | Functional Effect | Protocol Used | Palynological Analysis of Bee Bread (BB) | The Majority of Compounds Identified | Extraction Used | Concentration/ Treatment Duration | Administration Routes | Model Used | Results Obtained | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Morocco | Hypoglycemic, hypolipidemic, and hepatoprotective effects | Streptozotocin-induced diabetic rats | Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia | Isorhamnetin-O-hexosyl-O-rutinoside was the major phenolic compound present, in addition to tocopherols | Ethyl acetate extract | 100 mg/kg For 15 days | Oral | Wistar rats | ↓ Blood glucose levels Protect against weightloss ↓ TC, ↑ HDL, ↓ TG, ↓ LDL, ↓ VLDL Coronary risk index ↓ Atherogenic index ↓ Cardiovascular index | [64] |
| Morocco | Protective effects against anemia, inflammation, and hepato-renal toxicity | Toxicity of aluminum | One sample (not determined) | Polyphenol content: 14.88 ± 0.98 mg GAE/g Flavonoid content: 1.67 ± 0.12mg QE/g | Maceration for one week in 70% ethanol under agitation | 500 and 750 mg/kg | Oral | Male Wistar rats | ↑ Hematocrit, hemoglobin, red blood cells, MCH, MCV, and MCHC ↓ Platelets, monocytes, lymphocytes, and leukocytes. ↓ ALT, AST, and CRP ↓ Blood urea ↑ Urine sodium and creatinine clearance | [63] |
| Morocco | Protective effects against the toxicity induced by titanium dioxide in biochemical parameters of the brain, liver, and kidney tissues | Titanium dioxide nanoparticles induced toxicity in rats | Bupleurum spinosum, Anethum graveolens, Calendula officinalis, Anacyclus, Quercus ilex, Eucalyptus, Punica granatum, and Acacia | Isorhamnetin-O-hexosyl-O-rutinoside was the major phenolic compound present, in addition to tocopherols | Ethanolic extract | 100 mg/kg For 30 days | Oral | Wistar rats | ↓ AST, ↓ ALT, ↓ LDH ↓ TC, ↑ HDL, ↓ LDL ↓ Blood glucose levels ↓ Urea, ↑ Albumin, ↑ Total protein ↓ Sodium, ↓ potassium, ↓ chloride Protection against histopathological changes in the brain, kidneys, and liver | [51] |
| Slovakia | Alleviates lipid abnormalities and impaired bone morphology in obese Zucker diabetic rats | Obese Zucker diabetic rats | Monofloral (Brassica napus L.) | Vitamin A, vitamin E vitamin C, vitamin B2, vitamin B3, and Beta carotene | Mixed with distilled water | 500 and 700 mg/kg | Oral | Zucker diabetic Fatty rats | ↓ Blood glucose level ↓ TC, ↓ triglycerides ↑ the relative volume of trabecular bone ↑ trabecular thickness, enhanced density of secondary osteons, accelerated periosteal bone apposition, and improved blood flow | [109] |
| Malaysia | Protective effect against testicular oxidative stress, inflammation, apoptosis, and lactate transport in the testes of obese rats | High-fat diet (HFD) | One sample (not determined) | Riboflavin, thiamine, vitamin A, vitamin E, Fe, Cu, Zn, apigenin, caffeic acid, ferulic acid, isorhamnetin, andkaempferol | Blended and used as a fine powder | 0.5 g/kg/day) once daily for 12 weeks. | Oral | Male Sprague–Dawley rats | ↓ Final body weight and weight gain ↓ Lee obesity index, BMI, and energy intake. ↓ TC, ↓ TG, ↓ LDL, ↑ HDL ↓ The absolute and relative weights of the epididymal fat. Histological improvements of the testis. ↑ the activities of SOD, CAT, GPx, GST, GR, TAC ↓ the level of TBARS. ↑ The mRNA expression of Nrf2, SOD, CAT, and GPx. ↓ The mRNA transcript levels of pro-inflammatory Nf-κb, Tnf-α, iNos, and Il-1β genes. ↓ the activities and mRNA expression pro-apoptotic (p53, Bax, Bax/Bcl2, Caspase-8, Caspase-9, and Caspase-3) genes in the testes ↑ the mRNA levels of glucose transporters (Glut1 and Glut3), monocarboxylate transporters (Mct2 and Mct4), and lactate dehydrogenase type C (Ldhc) ↑ lactate utilization. ↑ PCNA immunoexpression | [107] |
| Turkey | The effects of bee bread on leptin and ghrelin expression in obese rats | Female rats became obese with a high-fat diet | One sample (not determined) | Protein: 13.56g/100g Carbohydrate: 30.60 g/100g Dietary fiber: 18.18g/100g Fat: 21.69g/100g | Distilled water | Concentration1: 100 mg/kg/day Concentration2: 200 mg/kg/day | Oral | Sprague–Dawley adult female rats | ↓ Ghrelin immunoreactivity in obese rats that received BB (100 mg/kg/day and 200 mg/kg/day). ↑ Leptin immunoreactivity in obese rats that received BB (100mg/kg/day and 200 mg/kg/day). ↓ Apoptotic cell numbers in hypothalamus tissue in obese rats that received BB (100 mg/kg/day and 200 mg/kg/day) ↑ SOD activity in obese rats that received BB (200 mg/kg/day). ↓ Ghrelin amounts in obese rats that received BB (200 mg/kg/day). ↑ Leptin amounts in obese rats that received BB (200 mg/kg/day). ↓ MDA levels in obese rats that received BB (200 mg/kg/day) | [112] |
| Slovakia | The effects of dietary bee bread powder on the chemical composition of quail meat. | Dietary supplementation of bee bread powder in quails | One sample (not determined) | (not determined) | Bee bread powder | 2 g or 4 g or 6 g per 1 kg of feed mixture | Oral | Japanese quails | The effect of BB supplementation on the chemical composition of breast muscle in quails: ↓ Water content ↑ Crude protein ↓ Fat ↓ Cholesterol The effect of BB supplementation on the chemical composition of thigh muscle in quails ↑ Water content ↑ Fat ↑ cholesterol | [110] |
| China | Regulation of lipid metabolism | SPF rats | Not mentioned | - | 80, 400, and 800 mg/kg during 20 days | Oral | Rats | ↓ Fatty acid Synthase ↓ Acetyl CoA carboxylase ↓ lipoprotein lipase ↓ Total cholesterol level, ↓ triglycerides, ↑ HDL, ↓ LDL | [111] | |
| Malaysia | Bee bread attenuates high-fat diet (HFD) induced renal pathology in obese rats via The modulation of oxidative stress, down regulation of NF-kB mediated inflammation, and Bax signaling | High-fat diet | One sample (not determined) | Protein: 3.37 ± 0.30% Lipid: 4.32 ± 0.17% Carbohydrate: 82.45 ± 0.36% Iron:0.02 ppm Copper: 0.01 ppm Zinc: 0.002ppm Vitamin A: 146.8 ± 6.21 (mg/100 g) Vitamin E: 46.27 ± 0.67 (mg/100 g) Thiamine: 6.20 ± 0.06 (mg/100 g) Riboflavin: 0.50 ± 0.00 (mg/100 g) | Distilled water | 0.5g/kg | Oral | Male Sprague–Dawley rats | ↓ % change in body weight, BMI index in rats that received HFD treated or protected with BB. ↓ Kidney weight in rats that received HFD treated or protected with BB. ↑ SOD, GPx, GST, TAA in rats that received HFD treated or protected with BB ↓ MDA concentrations in rats that received HFD treated or protected with BB ↓ Inflammatory cells in kidney tissues of HFD groups treated or protected with BB. ↓ Bowman’s capsule space in the urinary chamber of kidneys of HFD groups treated or protected with BB. ↓ NFkB, TNF-α, interleukin-1-beta, and Bax in rats that received HFD treated or protected with BB | [30] |
| Malaysia | Bee bread ameliorates the impaired vasorelaxation response to ACh by improving the eNOS/NO/cGMP-signaling pathway in obese rats | High-fat diet | One sample (not determined) | Potassium (7323.04 mg/kg), magnesium (1530.87 mg/kg), calcium (1108.48 mg/kg), sodium (252.73 mg/kg) iron (56.58 mg/kg), zinc (42.36 mg/kg), copper (11.05 mg/kg), and selenium (0.13 mg/kg) | Distilled water | 0.5 g/kg | Oral | Male Sprague–Dawley rats | BB improves the lipid profile, aortic inflammatory markers, and impaired vasorelaxation activity. BB enhances nitric oxide release, promotes endothelial nitric oxide synthase (eNOS) and cyclic guanosine monophosphate (cGMP) immunoexpression. | [106] |
| Slovakia | Reduced femoral bone structure and improved glucose and lipid metabolism in Zucker diabetic fatty (ZDF) rats | Obese Zucker diabetic rats | Not mentioned | - | The sample was crushed and mixed with distilled water | 500 mg/kg | Oral | Diabetic fatty rats | ↓ Fasting blood glucose level ↓ Total cholesterol level, ↓ triglycerides, protection against body weight loss, ↓ ALP activity, ↓ cortical bone surface, relative bone volume, ↑ trabecular bone, ↑ trabecular thickness, and trabecular bone surface | [113] |
6. The Use of Bee Bread in Clinical Studies
6.1. Hepatoprotective Effect
6.2. Anti-Atherogenic Dyslipidemic Effect
6.3. Strengthening Visual Acuity
6.4. Ergogenic Effect and Improvement of Athletic Performance
7. Bee Bread Adulteration
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Premratanachai, P.; Chanchao, C. Review of the Anticancer Activities of Bee Products. Asian Pac. J. Trop. Biomed. 2014, 4, 337–344. [Google Scholar] [CrossRef] [Green Version]
- Kolayli, S.; Keskin, M. Chapter 7—Natural bee products and their apitherapeutic applications. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2020; Volume 66, pp. 175–196. ISBN 1572-5995. [Google Scholar]
- Urcan, A.; Mărghitaș, L.A.; Dezmirean, D.S.; Bobiș, O.; Bonta, V.; Mureșan, C.I.; Mărgăoan, R. Chemical composition and biological activities of beebread-review. In Bulletin of the University of Agricultural Sciences & Veterinary Medicine Cluj-Napoca; Animal Science & Biotechnologies; Academic Press: Cambridge, MA, USA, 2017; Volume 74. [Google Scholar]
- Dolezal, A.G.; Toth, A.L. Feedbacks between Nutrition and Disease in Honey Bee Health. Curr. Opin. Insect Sci. 2018, 26, 114–119. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Tooker, J.F.; Grozinger, C.M.; Patch, H.M. Bee Nutrition and Floral Resource Restoration. Curr. Opin. Insect Sci. 2015, 10, 133–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohammad, S.M.; Mahmud-Ab-Rashid, N.-K.; Zawawi, N. Stingless Bee-Collected Pollen (Bee Bread): Chemical and Microbiology Properties and Health Benefits. Molecules 2021, 26, 957. [Google Scholar] [CrossRef]
- Aylanc, V.; Falcão, S.I.; Ertosun, S.; Vilas-Boas, M. From the Hive to the Table: Nutrition Value, Digestibility and Bioavailability of the Dietary Phytochemicals Present in the Bee Pollen and Bee Bread. Trends Food Sci. Technol. 2021, 109, 464–481. [Google Scholar] [CrossRef]
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and Bee Bread as New Health-Oriented Products: A Review. Trends Food Sci. Technol. 2018, 71, 170–180. [Google Scholar] [CrossRef]
- Barene, I.; Daberte, I.; Siksna, S. Investigation of Bee Bread and Development of Its Dosage Forms. Med. Teor. Prakt. 2014, 21, 16–22. [Google Scholar] [CrossRef]
- Andjelkovic, B.; Jevtic, G.; Markovic, J.; Mladenovic, M.; Pseva, V. Quality of Honey Bee Bread Collected in Spring. J. Hyg. Eng. Des. 2012, 1, 275–277. [Google Scholar]
- Mohammad, S.M.; Mahmud-Ab-Rashid, N.-K.; Zawawi, N. Botanical Origin and Nutritional Values of Bee Bread of Stingless Bee (Heterotrigona itama) from Malaysia. J. Food Qual. 2020, 2020, 2845757. [Google Scholar] [CrossRef] [Green Version]
- Othman, Z.A.; Noordin, L.; Wan Ghazali, W.S.; Omar, N.; Mohamed, M. Nutritional, Phytochemical and Antioxidant Analysis of Bee Bread from Different Regions of Malaysia. Indian J. Pharm. Sci. 2019, 81, 955–960. [Google Scholar] [CrossRef] [Green Version]
- Donkersley, P.; Rhodes, G.; Pickup, R.W.; Jones, K.C.; Power, E.F.; Wright, G.A.; Wilson, K. Nutritional Composition of Honey Bee Food Stores Vary with Floral Composition. Oecologia 2017, 185, 749–761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeGrandi-Hoffman, G.; Eckholm, B.J.; Huang, M.H. A Comparison of Bee Bread Made by Africanized and European Honey Bees (Apis mellifera) and Its Effects on Hemolymph Protein Titers. Apidologie 2013, 44, 52–63. [Google Scholar] [CrossRef]
- Bayram, N.E.; Gercek, Y.C.; Çelik, S.; Mayda, N.; Kostić, A.Ž.; Dramićanin, A.M.; Özkök, A. Phenolic and Free Amino Acid Profiles of Bee Bread and Bee Pollen with the Same Botanical Origin-Similarities and Differences. Arab. J. Chem. 2021, 14, 103004. [Google Scholar] [CrossRef]
- Pirini, A.; Conte, L.S.; Francioso, O.; Lercker, G. Capillary Gas Chromatographic Determination of Free Amino Acids in Honey as a Means of Discrimination between Different Botanical Sources. J. High Resolut. Chromatogr. 1992, 15, 165–170. [Google Scholar] [CrossRef]
- Elnesr, S.; Elwan, H.; Xu, Q.; Xie, C.; Dong, X.; Zou, X. Effects of in Ovo Injection of Sulfur-Containing Amino Acids on Heat Shock Protein 70, Corticosterone Hormone, Antioxidant Indices, and Lipid Profile of Newly Hatched Broiler Chicks Exposed to Heat Stress during Incubation. Poult. Sci. 2019, 98, 2290–2298. [Google Scholar] [CrossRef] [PubMed]
- Birmani, M.W.; Raza, A.; Nawab, A.; Tang, S.; Ghani, M.W.; Li, G.; Xiao, M.; An, L. Importance of Arginine as Immune Regulator in Animal Nutrition. Int. J. Vet. Sci. Res. 2019, 5, 1–10. [Google Scholar] [CrossRef]
- Haydak, M.H. Honey Bee Nutrition. Annu. Rev. Entomol. 1970, 15, 143–156. [Google Scholar] [CrossRef]
- Bakour, M.; Fernandes, Â.; Barros, L.; Sokovic, M.; Ferreira, I.C.F.R. Badiaa lyoussi Bee Bread as a Functional Product: Chemical Composition and Bioactive Properties. LWT Food Sci. Technol. 2019, 109, 276–282. [Google Scholar] [CrossRef] [Green Version]
- Dranca, F.; Ursachi, F.; Oroian, M. Bee Bread: Physicochemical Characterization and Phenolic Content Extraction Optimization. Foods 2020, 9, 1358. [Google Scholar] [CrossRef]
- Urcan, A.; Criste, A.; Dezmirean, D.; Mărgăoan, R.; Caeiro, A.; Graça Campos, M. Similarity of Data from Bee Bread with the Same Taxa Collected in India and Romania. Molecules 2018, 23, 2491. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, G.R.; da Natividade, T.B.; Camara, C.A.; da Silva, E.M.S.; dos Santos, F.d.A.R.; Silva, T.M.S. Identification of Sugar, Amino Acids and Minerals from the Pollen of Jandaíra Stingless Bees (Melipona subnitida). Food Nutr. Sci. 2014, 2014, 46901. [Google Scholar]
- Kaplan, M.; Karaoğlu, Ö.; Silici, S. An Evaluation on Bee Bread: Chemical and Palynological Analysis. Mellifera 2019, 19, 21–29. [Google Scholar]
- Kaplan, M.; Karaoglu, Ö.; Eroglu, N. Sibel Silici Fatty Acids and Proximate Composition of Beebread. Food Technol. Biotechnol. 2016, 54, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Thakur, M.; Nanda, V. Exploring the Physical, Functional, Thermal, and Textural Properties of Bee Pollen from Different Botanical Origins of India. J. Food Process Eng. 2020, 43, e12935. [Google Scholar] [CrossRef]
- Yang, K.; Wu, D.; Ye, X.; Liu, D.; Chen, J.; Sun, P. Characterization of Chemical Composition of Bee Pollen in China. J. Agric. Food Chem. 2013, 61, 708–718. [Google Scholar] [CrossRef]
- AL-Kahtani, S.N. Fatty Acids and B Vitamins Contents in Honey Bee Collected Pollen in Relation to Botanical Origin. Sci. J. King Faisal Univ. 2017, 18, 41–48. [Google Scholar]
- Dong, J.; Yang, Y.; Wang, X.; Zhang, H. Fatty Acid Profiles of 20 Species of Monofloral Bee Pollen from China. J. Apic. Res. 2015, 54, 503–511. [Google Scholar] [CrossRef]
- Eleazu, C.; Suleiman, J.B.; Othman, Z.A.; Zakaria, Z.; Nna, V.U.; Hussain, N.H.N.; Mohamed, M. Bee Bread Attenuates High Fat Diet Induced Renal Pathology in Obese Rats via Modulation of Oxidative Stress, Downregulation of NF-KB Mediated Inflammation and Bax Signalling. Arch. Physiol. Biochem. 2020, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Ciric, J.; Spiric, D.; Baltic, T.; Janjic, J.; Petronijevic, R.; Simunovic, S.; Djordjevic, V. Element Concentration and Fatty Acid Composition of Serbian Bee Bread; IOP Publishing: Bristol, UK, 2019; Volume 333, p. 01 2050.
- Greer, R.C.; Marklund, M.; Anderson, C.A.; Cobb, L.K.; Dalcin, A.T.; Henry, M.; Appel, L.J. Potassium-Enriched Salt Substitutes as a Means to Lower Blood Pressure: Benefits and Risks. Hypertension 2020, 75, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.A.; Gallardo, M.; Cespedes, C.; Vio, C.P. Potassium Intake Prevents the Induction of the Renin-Angiotensin System and Increases Medullary ACE2 and COX-2 in the Kidneys of Angiotensin II-Dependent Hypertensive Rats. Front. Pharmacol. 2019, 10, 1212. [Google Scholar] [CrossRef] [Green Version]
- Ji, F.; Zhang, S.; An, Y.; Wang, Z.; Shao, Y.; Du, S.; Li, X.; Sun, X. Influence of Dietary Phosphorus Concentrations on the Performance of Rearing Pigeons (Columba Livia), and Bone Properties of Squabs. Poult. Sci. 2022, 101744. [Google Scholar] [CrossRef]
- Anwar, H.; Hussain, G.; Mustafa, I. Antioxidants from Natural Sources. Antioxid. Foods Appl. 2018, 1–27. [Google Scholar]
- Kalaycıoğlu, Z.; Kaygusuz, H.; Döker, S.; Kolaylı, S.; Erim, F.B. Characterization of Turkish Honeybee Pollens by Principal Component Analysis Based on Their Individual Organic Acids, Sugars, Minerals, and Antioxidant Activities. LWT 2017, 84, 402–408. [Google Scholar] [CrossRef]
- Wu, S.; Xu, C.; Zhu, Y.; Zheng, L.; Zhang, L.; Hu, Y.; Yu, B.; Wang, Y.; Xu, F. Biofilm-Sensitive Photodynamic Nanoparticles for Enhanced Penetration and Antibacterial Efficiency. Adv. Funct. Mater. 2021, 31, 2103591. [Google Scholar] [CrossRef]
- Peláez, A.L.; Cataño, C.S.; Yepes, E.Q.; Villarroel, R.G.; de Antoni, G.; Giannuzzi, L. Inhibitory Activity of Lactic and Acetic Acid on Aspergillus Flavus Growth for Food Preservation. Food Control 2012, 24, 177–183. [Google Scholar] [CrossRef]
- Ares, A.M.; Valverde, S.; Bernal, J.L.; Nozal, M.J.; Bernal, J. Extraction and Determination of Bioactive Compounds from Bee Pollen. J. Pharm. Biomed. Anal. 2018, 147, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Loper, G.M.; Standifer, L.N.; Thompson, M.J.; Gilliam, M. Biochemistry and Microbiology of Bee-Collected Almond (Prunus dulcis) Pollen and Bee Bread. I-Fatty Acids, Sterols, Vitamins and Minerals. Apidologie 1980, 11, 63–73. [Google Scholar] [CrossRef] [Green Version]
- Thakur, M.; Nanda, V. Composition and Functionality of Bee Pollen: A Review. Trends Food Sci. Technol. 2020, 98, 82–106. [Google Scholar] [CrossRef]
- Laaroussi, H.; Ferreira-Santos, P.; Genisheva, Z.; Bakour, M.; Ousaaid, D.; Teixeira, J.A.; Lyoussi, B. Unraveling the Chemical Composition, Antioxidant, α-Amylase and α-Glucosidase Inhibition of Moroccan Propolis. Food Biosci. 2021, 42, 101160. [Google Scholar] [CrossRef]
- Hudz, N.; Ivanova, R.; Brindza, J.; Grygorieva, O.; Schubertová, Z.; Ivanišová, E. Approaches to the Determination of Antioxidant Activity of Extracts from Bee Bread and Safflower Leaves and Flowers. Potravinarstvo 2017, 11, 480–488. [Google Scholar] [CrossRef] [Green Version]
- Oltica, S.; Mărghitaş, L.A.; Dezmirean, D. Examination of antioxidant capacity of beebread extracts by different complementary assays. In Bulletin of the University of Agricultural Sciences & Veterinary Medicine Cluj-Napoca; Animal Science & Biotechnologies; Academic Press: Cambridge, MA, USA, 2007; Volume 63, p. 64. [Google Scholar]
- Silva, B.N.; Cadavez, V.; Ferreira-Santos, P.; Alves, M.J.; Ferreira, I.C.; Barros, L.; Teixeira, J.A.; Gonzales-Barron, U. Chemical Profile and Bioactivities of Extracts from Edible Plants Readily Available in Portugal. Foods 2021, 10, 673. [Google Scholar] [CrossRef] [PubMed]
- Sobral, F.; Calhelha, R.; Barros, L.; Dueñas, M.; Tomás, A.; Santos-Buelga, C.; Vilas-Boas, M.; Ferreira, I. Flavonoid Composition and Antitumor Activity of Bee Bread Collected in Northeast Portugal. Molecules 2017, 22, 248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aylanc, V.; Tomás, A.; Russo-Almeida, P.; Falcão, S.I.; Vilas-Boas, M. Assessment of Bioactive Compounds under Simulated Gastrointestinal Digestion of Bee Pollen and Bee Bread: Bioaccessibility and Antioxidant Activity. Antioxidants 2021, 10, 651. [Google Scholar] [CrossRef]
- Baltrušaitytė, V.; Venskutonis, P.R.; Čeksterytė, V. Radical Scavenging Activity of Different Floral Origin Honey and Beebread Phenolic Extracts. Food Chem. 2007, 101, 502–514. [Google Scholar] [CrossRef]
- Tavdidishvili, D.; Khutsidze, T.; Pkhakadze, M.; Vanidze, M.; Kalandia, A. Flavonoids in Georgian Bee Bread and Bee Pollen. J. Chem. Chem. Eng. 2014, 8, 676–681. [Google Scholar]
- Markiewicz-Żukowska, R.; Naliwajko, S.K.; Bartosiuk, E.; Moskwa, J.; Isidorov, V.; Soroczyńska, J.; Borawska, M.H. Chemical Composition and Antioxidant Activity of Beebread, and Its Influence on the Glioblastoma Cell Line (U87MG). J. Apic. Sci. 2013, 57, 147–157. [Google Scholar] [CrossRef] [Green Version]
- Bakour, M.; Hammas, N.; Laaroussi, H.; Ousaaid, D.; Fatemi, H.E.; Aboulghazi, A.; Soulo, N.; Lyoussi, B. Moroccan Bee Bread Improves Biochemical and Histological Changes of the Brain, Liver, and Kidneys Induced by Titanium Dioxide Nanoparticles. BioMed Res. Int. 2021, 2021, 6632128. [Google Scholar] [CrossRef]
- Elsayed, N.; El-Din, H.S.; Altemimi, A.B.; Ahmed, H.Y.; Pratap-Singh, A.; Abedelmaksoud, T.G. In Vitro Antimicrobial, Antioxidant and Anticancer Activities of Egyptian Citrus Beebread. Molecules 2021, 26, 2433. [Google Scholar] [CrossRef]
- Hsu, C.-K.; Wang, D.-Y.; Wu, M.-C. A Potential Fungal Probiotic Aureobasidium Melanogenum CK-CsC for the Western Honey Bee, Apis mellifera. J. Fungi 2021, 7, 508. [Google Scholar] [CrossRef]
- Dimov, S.G.; Zagorchev, L.; Iliev, M.; Dekova, T.; Ilieva, R.; Kitanova, M.; Georgieva-Miteva, D.; Dimitrov, M.; Peykov, S. A Snapshot Picture of the Fungal Composition of Bee Bread in Four Locations in Bulgaria, Differing in Anthropogenic Influence. J. Fungi 2021, 7, 845. [Google Scholar] [CrossRef] [PubMed]
- Iorizzo, M.; Pannella, G.; Lombardi, S.J.; Ganassi, S.; Testa, B.; Succi, M.; Sorrentino, E.; Petrarca, S.; de Cristofaro, A.; Coppola, R. Inter-and Intra-Species Diversity of Lactic Acid Bacteria in Apis mellifera Ligustica Colonies. Microorganisms 2020, 8, 1578. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, S.M.; Mahmud-Ab-Rashid, N.-K.; Zawawi, N. Probiotic Properties of Bacteria Isolated from Bee Bread of Stingless Bee Heterotrigona itama. J. Apic. Res. 2020, 60, 172–187. [Google Scholar] [CrossRef]
- Machado De-Melo, A.A.; de Almeida-Muradian, L.B.; Sancho, M.T.; Pascual-Maté, A. Composition and Properties of Apis mellifera Honey: A Review. J. Apic. Res. 2018, 57, 5–37. [Google Scholar] [CrossRef]
- Gilliam, M.; Prest, D.B.; Lorenz, B.J. Microbiology of Pollen and Bee Bread: Taxonomy and Enzymology of Molds. Apidologie 1989, 20, 53–68. [Google Scholar] [CrossRef] [Green Version]
- Salazar-González, C.; Díaz-Moreno, C. The Nutritional and Bioactive Aptitude of Bee Pollen for a Solid-State Fermentation Process. J. Apic. Res. 2016, 55, 161–175. [Google Scholar] [CrossRef]
- Ispirli, H.; Dertli, E. Detection of Fructophilic Lactic Acid Bacteria (FLAB) in Bee Bread and Bee Pollen Samples and Determination of Their Functional Roles. J. Food Process. Preserv. 2021, 45, e15414. [Google Scholar] [CrossRef]
- Akhir, R.A.M.; Bakar, M.F.A.; Sanusi, S.B. Antioxidant and Antimicrobial Activity of Stingless Bee Bread and Propolis Extracts; AIP Publishing LLC: Melville, NY, USA, 2017; Volume 1891, p. 020090. [Google Scholar]
- Zuluaga, C.M.; Serratob, J.C.; Quicazana, M.C. Chemical, Nutritional and Bioactive Characterization of Colombian Bee-Bread. Chem. Eng. 2015, 43, 175–180. [Google Scholar]
- Bakour, M.; Al-Waili, N.S.; El Menyiy, N.; Imtara, H.; Figuira, A.C.; Al-Waili, T.; Lyoussi, B. Antioxidant Activity and Protective Effect of Bee Bread (Honey and Pollen) in Aluminum-Induced Anemia, Elevation of Inflammatory Makers and Hepato-Renal Toxicity. J. Food Sci. Technol. 2017, 54, 4205–4212. [Google Scholar] [CrossRef]
- Bakour, M.; Menyiy, N.E.; Ghouizi, A.E.; Lyoussi, B. Hypoglycemic, Hypolipidemic and Hepato-Protective Effect of Bee Bread in Streptozotocin-Induced Diabetic Rats. Avicenna J. Phytomed. 2021, 11, 10. [Google Scholar]
- Baltrušaitytė, V.; Venskutonis, P.R.; Čeksterylė, V. Antibacterial Activity of Honey and Beebread of Different Origin Against S. Aureus and S. Epidermidis. Food Technol. Biotechnol. 2007, 45, 201–208. [Google Scholar]
- Ivanišová, E.; Kačániová, M.; Frančáková, H.; Petrová, J.; Hutková, J.; Brovarskyi, V.; Velychko, S.; Adamchuk, L.; Schubertová, Z.; Musilová, J. Bee Bread—Perspective Source of Bioactive Compounds for Future. Potravinarstvo 2015, 9, 592–598. [Google Scholar] [CrossRef] [Green Version]
- Keskin, M.; Özkök, A. α-Amylase Inhibition Properties of Bee Pollen and Bee Bread (Perga). Hacet. J. Biol. Chem. 2020, 48, 389–393. [Google Scholar] [CrossRef]
- Nagai, T.; Nagashima, T.; Myoda, T.; Inoue, R. Preparation and Functional Properties of Extracts from Bee Bread. Food Nahr. 2004, 48, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Nagashima, T.; Suzuki, N.; Inoue, R. Antioxidant Activity and Angiotensin I-Converting Enzyme Inhibition by Enzymatic Hydrolysates from Bee Bread. Z. Naturforsch. C 2005, 60, 133–138. [Google Scholar] [CrossRef]
- Fallah, M.; Najafi, F.; Kavoosi, G. Bee Propolis, Bee Bread, and Royal Jelly: Proximate Analysis, Fatty Acid Composition, Nutritional Quality, and Anti-Amylase Activity; Research Gate: Berlin, Germany, 2021. [Google Scholar]
- Jo, Y.H.; Lee, S.; Yeon, S.W.; Ryu, S.H.; Turk, A.; Hwang, B.Y.; Han, Y.K.; Lee, K.Y.; Lee, M.K. Anti-α-Glucosidase and Anti-Oxidative Isoflavonoids from the Immature Fruits of Maclura Tricuspidata. Phytochemistry 2022, 194, 113016. [Google Scholar] [CrossRef]
- Węglińska, M.; Szostak, R.; Kita, A.; Nemś, A.; Mazurek, S. Determination of Nutritional Parameters of Bee Pollen by Raman and Infrared Spectroscopy. Talanta 2020, 212, 120790. [Google Scholar] [CrossRef]
- Bartkiene, E.; Lele, V.; Sakiene, V.; Zavistanaviciute, P.; Zokaityte, E.; Dauksiene, A.; Jagminas, P.; Klupsaite, D.; Bliznikas, S.; Ruzauskas, M. Variations of the Antimicrobial, Antioxidant, Sensory Attributes and Biogenic Amines Content in Lithuania-Derived Bee Products. LWT 2020, 118, 108793. [Google Scholar] [CrossRef]
- Hotel, A.C.P.; Cordoba, A. Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria. Prevention 2001, 5, 1–10. [Google Scholar]
- Isolauri, E.; Salminen, S.; Ouwehand, A.C. Probiotics. Best Pract. Res. Clin. Gastroenterol. 2004, 18, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Cerdó, T.; Ruíz, A.; Suárez, A.; Campoy, C. Probiotic, Prebiotic, and Brain Development. Nutrients 2017, 9, 1247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, G.; Scagnolari, C.; Pugliese, F.; Mastroianni, C.M.; d’Ettorre, G. Probiotics and COVID-19. Lancet Gastroenterol. Hepatol. 2020, 5, 721–722. [Google Scholar] [CrossRef]
- Terpou, A.; Papadaki, A.; Lappa, I.K.; Kachrimanidou, V.; Bosnea, L.A.; Kopsahelis, N. Probiotics in Food Systems: Significance and Emerging Strategies towards Improved Viability and Delivery of Enhanced Beneficial Value. Nutrients 2019, 11, 1591. [Google Scholar] [CrossRef] [Green Version]
- Mărgăoan, R.; Cornea-Cipcigan, M.; Topal, E.; Kösoğlu, M. Impact of Fermentation Processes on the Bioactive Profile and Health-Promoting Properties of Bee Bread, Mead and Honey Vinegar. Processes 2020, 8, 1081. [Google Scholar] [CrossRef]
- Toutiaee, S.; Mojgani, N.; Harzandi, N.; Moharrami, M.; Mokhberosafa, L. In-vitro Probiotic and Safety Attributes of Bacillus Spp. Isolated from Beebread, Honey Samples and Digestive Tract of Honeybees Apis mellifera. Lett. Appl. Microbiol. 2022. ahead of print. [Google Scholar] [CrossRef]
- Khalifa, S.A.; Elashal, M.; Kieliszek, M.; Ghazala, N.E.; Farag, M.A.; Saeed, A.; Xiao, J.; Zou, X.; Khatib, A.; Göransson, U. Recent Insights into Chemical and Pharmacological Studies of Bee Bread. Trends Food Sci. Technol. 2020, 97, 300–316. [Google Scholar] [CrossRef]
- Abouda, I.Z.; Zerdani, I.; Kalalou, M.; Faid, M.T. Ahami The Antibacterial Activity of Moroccan Bee Bread and Bee-Pollen (Fresh and Dried) against Pathogenic Bacteria. Res. J. Microbiol. 2011, 6, 376–384. [Google Scholar]
- Didaras, N.A.; Kafantaris, I.; Dimitriou, T.G.; Mitsagga, C.; Karatasou, K.; Giavasis, I.; Stagos, D.; Amoutzias, G.D.; Hatjina, F.; Mossialos, D. Biological Properties of Bee Bread Collected from Apiaries Located across Greece. Antibiotics 2021, 10, 555. [Google Scholar] [CrossRef]
- Pełka, K.; Otłowska, O.; Worobo, R.W.; Szweda, P. Bee Bread Exhibits Higher Antimicrobial Potential Compared to Bee Pollen. Antibiotics 2021, 10, 125. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F.; Lani, M.; Nazari, S.; Hajar, N.; Hassan, K.; Razak, S.; Hassan, Z. Antioxidant and Antimicrobial Properties of Honey, Propolis and Bee Bread of Stingless Bee (Geniotrigona thoracica). Asian J. Agric. Biol. 2019, 1, 1–10. [Google Scholar]
- Suleiman, J.B.; Mohamed, M.; Abu Bakar, A.B.; Nna, V.U.; Zakaria, Z.; Othman, Z.A.; Aroyehun, A.B. Chemical Profile, Antioxidant Properties and Antimicrobial Activities of Malaysian Heterotrigona itama Bee Bread. Molecules 2021, 26, 4943. [Google Scholar] [CrossRef]
- Hudz, N.; Yezerska, O.; Grygorieva, O.; Brindza, J.; Felsöciová, S.; Kačániová, M.; Wieczorek, P.P. Analytical Procedure Elaboration of Total Flavonoid Content Determination and Antimicrobial Activity of Bee Bread Extracts. Acta Pol. Pharm.-Drug Res. 2019, 76, 439–452. [Google Scholar] [CrossRef]
- Bakour, M.; Laaroussi, H.; Ousaaid, D.; Oumokhtar, B.; Lyoussi, B. Antioxidant and Antibacterial Effects of Pollen Extracts on Human Multidrug-Resistant Pathogenic Bacteria. J. Food Qual. 2021, 2021, 5560182. [Google Scholar] [CrossRef]
- Didaras, N.A.; Dimitriou, T.; Daskou, M.; Karatasou, K.; Mossialos, D. In vitro assessment of the antiviral activity of Greek bee bread and bee collected pollen against enterovirus D68. J. Microbiol. Biotechnol. Food Sci. 2021, 20, e4859. [Google Scholar]
- Al-Hatamleh, M.A.I.; Hatmal, M.M.; Sattar, K.; Ahmad, S.; Mustafa, M.Z.; Bittencourt, M.D.C.; Mohamud, R. Antiviral and Immunomodulatory Effects of Phytochemicals from Honey against COVID-19: Potential Mechanisms of Action and Future Directions. Molecules 2020, 25, 5017. [Google Scholar] [CrossRef]
- Lima, W.G.; Brito, J.C.M.; da Cruz Nizer, W.S. Bee Products as a Source of Promising Therapeutic and Chemoprophylaxis Strategies against COVID-19 (SARS-CoV-2). Phytother. Res. 2020, 35, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.S.; Sushma, P.; Dharmashekar, C.; Beelagi, M.S.; Prasad, S.K.; Shivamallu, C.; Prasad, A.; Syed, A.; Marraiki, N.; Prasad, K.S. In Silico Evaluation of Flavonoids as Effective Antiviral Agents on the Spike Glycoprotein of SARS-CoV-2. Saudi J. Biol. Sci. 2021, 28, 1040–1051. [Google Scholar] [CrossRef] [PubMed]
- Hashem, H. In Silico Approach of Some Selected Honey Constituents as SARS-CoV-2 Main Protease (COVID-19) Inhibitors. Eur. J. Med. Oncol. 2020, 4, 196–200. [Google Scholar] [CrossRef]
- Bouarab Chibane, L.; Degraeve, P.; Ferhout, H.; Bouajila, J.; Oulahal, N. Plant Antimicrobial Polyphenols as Potential Natural Food Preservatives. J. Sci. Food Agric. 2019, 99, 1457–1474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ultee, A.; Bennik, M.; Moezelaar, R. The Phenolic Hydroxyl Group of Carvacrol Is Essential for Action against the Food-Borne Pathogen Bacillus Cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Yao, J.; Zhou, B.; Yang, J.; Chaudry, M.T.; Wang, M.; Xiao, F.; Li, Y.; Yin, W. Bacteriostatic Effect of Quercetin as an Antibiotic Alternative in Vivo and Its Antibacterial Mechanism in Vitro. J. Food Prot. 2018, 81, 68–78. [Google Scholar] [CrossRef]
- Yu, L.; Shang, F.; Chen, X.; Ni, J.; Yu, L.; Zhang, M.; Sun, D.; Xue, T. The Anti-Biofilm Effect of Silver-Nanoparticle-Decorated Quercetin Nanoparticles on a Multi-Drug Resistant Escherichia Coli Strain Isolated from a Dairy Cow with Mastitis. PeerJ 2018, 6, e5711. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.-H.; Huang, C.-C.; Chen, C.-C.; Yang, K.-J.; Huang, C.-Y. Inhibition of Staphylococcus aureus PriA Helicase by Flavonol Kaempferol. Protein J. 2015, 34, 169–172. [Google Scholar] [CrossRef]
- Yu, Y.; Yi, Z.; Liang, Y.-Z. Validate Antibacterial Mode and Find Main Bioactive Components of Traditional Chinese Medicine Aquilegia Oxysepala. Bioorganic Med. Chem. Lett. 2007, 17, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Pham, V.H.; Abbas, W.; Huang, J.; He, Q.; Zhen, W.; Guo, Y.; Wang, Z. Effect of Blending Encapsulated Essential Oils and Organic Acids as an Antibiotic Growth Promoter Alternative on Growth Performance and Intestinal Health in Broilers with Necrotic Enteritis. Poult. Sci. 2022, 101, 101563. [Google Scholar] [CrossRef] [PubMed]
- Brul, S.; Coote, P. Preservative Agents in Foods: Mode of Action and Microbial Resistance Mechanisms. Int. J. Food Microbiol. 1999, 50, 1–17. [Google Scholar] [CrossRef]
- Masoura, M.; Passaretti, P.; Overton, T.W.; Lund, P.A.; Gkatzionis, K. Use of a Model to Understand the Synergies Underlying the Antibacterial Mechanism of H 2 O 2-Producing Honeys. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yagnik, D.; Ward, M.; Shah, A.J. Antibacterial Apple Cider Vinegar Eradicates Methicillin Resistant Staphylococcus aureus and Resistant Escherichia Coli. Sci. Rep. 2021, 11, 1–7. [Google Scholar] [CrossRef]
- Pothimon, R.; Krusong, W.; Daetae, P.; Tantratian, S.; Gullo, M. Determination of Antifungal Volatile Organic Compounds of Upland Rice Vinegar and Their Inhibition Effects on Aspergillus Flavus in Dried Chili Pepper. Food Biosci. 2022, 46, 101543. [Google Scholar] [CrossRef]
- Othman, Z.A.; Zakaria, Z.; Suleiman, J.B.; Nna, V.U.; Che Romli, A.; Wan Ghazali, W.S.; Mohamed, M. Bee Bread Ameliorates Vascular Inflammation and Impaired Vasorelaxation in Obesity-Induced Vascular Damage Rat Model: The Role of ENOS/NO/CGMP-Signaling Pathway. Int. J. Mol. Sci. 2021, 22, 4225. [Google Scholar] [CrossRef]
- Suleiman, J.B.; Nna, V.U.; Zakaria, Z.; Othman, Z.A.; Eleazu, C.O.; Abu Bakar, A.B.; Ahmad, A.; Usman, U.Z.; Abdul Rahman, W.F.W.; Mohamed, M. Protective Effects of Bee Bread on Testicular Oxidative Stress, NF-ΚB-Mediated Inflammation, Apoptosis and Lactate Transport Decline in Obese Male Rats. Biomed. Pharmacother. 2020, 131, 110781. [Google Scholar] [CrossRef] [PubMed]
- Othman, Z.A.; Wan Ghazali, W.S.; Noordin, L.; Mohd Yusof, N.A.; Mohamed, M. Phenolic Compounds and the Anti-Atherogenic Effect of Bee Bread in High-Fat Diet-Induced Obese Rats. Antioxidants 2019, 9, 33. [Google Scholar] [CrossRef] [Green Version]
- Martiniakova, M.; Blahova, J.; Kovacova, V.; Babikova, M.; Mondockova, V.; Kalafova, A.; Capcarova, M.; Omelka, R. Bee Bread Can Alleviate Lipid Abnormalities and Impaired Bone Morphology in Obese Zucker Diabetic Rats. Molecules 2021, 26, 2616. [Google Scholar] [CrossRef]
- Haščík, P. Chemical composition of muscle after bee bread application in the nutrition of Japanese quails. J. Microbiol. Biotechnol. Food Sci. 2020, 9, 831–835. [Google Scholar] [CrossRef]
- Li, Z.; Huang, Q.; Liu, Y.; Peng, C.; Zeng, Z. Natural Bee Bread Positively Regulates Lipid Metabolism in Rats. Int. J. Agric. Sci. Food Technol. 2021, 7, 266–271. [Google Scholar]
- Doğanyiğit, Z.; YAKAN, B.; Soylu, M.; Kaymak, E.; Silici, S. The Effects of Feeding Obese Rats with Bee Bread on Leptin and Ghrelin Expression. Turk. J. Zool. 2020, 44, 114–125. [Google Scholar] [CrossRef]
- Martiniakova, M.; Blahova, J.; Kovacova, V.; Mondockova, V.; Babosova, R.; Kalafova, A.; Capcarova, M.; Omelka, R. Effects of Bee Bread, Cornelian Cherries Treatment on the Femoral Bone Structure Using Zucker Diabetic Fatty Rats as an Animal Model. Veter. Med. 2021, 66, 342–349. [Google Scholar] [CrossRef]
- Čeksterytė, V.; Balžekas, J.; Baltuškevičius, A.; Jurgevičius, E. The Use of Beebread-Honey Mixture in the Treatment of Liver Diseases in Alcohol-Dependent Patients. Chem. Technol. 2012, 60, 62–66. [Google Scholar] [CrossRef] [Green Version]
- Kas’ianenko, V.I.; Komisarenko, I.A.; Dubtsova, E.A. Correction of atherogenic dyslipidemia with honey, pollen and bee bread in patients with different body mass. Ter. Arkh. 2011, 83, 58–62. [Google Scholar] [PubMed]
- Jarušaitienė, D.; Jankauskienė, J.; Čeksterytė, V. Changes of Ocular Signs in Children with Thyroid Diseases after Intake of Bee Bread. Chem. Technol. 2012, 61, 7–10. [Google Scholar] [CrossRef]
- Chen, C.K.; Ping, F.W.C.; Ooi, F.K.; Mohamed, M. Effects of Bee Bread Supplementation during Recovery on Time Trial Performance and Selected Physiological Parameters. Int. J. Pharma Bio Sci. 2018, 9, 281–288. [Google Scholar] [CrossRef]
- Fadzel, W.C.P.; Chen, C.K.; Ooi, F.K.; Mohamed, M. Effects of Bee Bread Supplementation on Endurance Running Performance and Total Antioxidant Status in Recreational Athletes. Int. J. Appl. Res. Nat. Prod. 2018, 11, 17–23. [Google Scholar]
- Semkiw, P.; Skubida, P. Bee Bread Production—A New Source of Income for Beekeeping Farms? Agriculture 2021, 11, 468. [Google Scholar] [CrossRef]
- Bakour, M.; Laaroussi, H.; El Menyiy, N.; Elaraj, T.; El Ghouizi, A.; Lyoussi, B. The Beekeeping State and Inventory of Mellifero-Medicinal Plants in the North-Central of Morocco. Sci. World J. 2021, 2021, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Reybroeck, W.; van Veen, J.W.; Gupta, A. Beekeeping for Poverty Alleviation and Livelihood Security; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- AbdulRahaman, A.A.; Liadi, M.; Musa, A.; Kolawole, O.; Oladele, F. Pollens in Bee-Breads as an Indicator of Honey Sources. Bangladesh J. Sci. Ind. Res. 2013, 48, 247–252. [Google Scholar] [CrossRef] [Green Version]
- Chica, M. Authentication of Bee Pollen Grains in Bright-field Microscopy by Combining One-class Classification Techniques and Image Processing. Microsc. Res. Tech. 2012, 75, 1475–1485. [Google Scholar] [CrossRef]
- Wang, Z.; Ren, P.; Wu, Y.; He, Q. Recent Advances in Analytical Techniques for the Detection of Adulteration and Authenticity of Bee Products—A Review. Food Addit. Contam. Part A 2021, 38, 533–549. [Google Scholar] [CrossRef]
- Araújo, J.S.; Chambó, E.D.; de Costa, M.A.P.C.; Cavalcante da Silva, S.M.P.; Lopes de Carvalho, C.A.; Estevinho, L.M. Chemical Composition and Biological Activities of Mono- and Heterofloral Bee Pollen of Different Geographical Origins. Int. J. Mol. Sci. 2017, 18, 921. [Google Scholar] [CrossRef] [Green Version]
- Kafantaris, I.; Amoutzias, G.D.; Mossialos, D. Foodomics in Bee Product Research: A Systematic Literature Review. Eur. Food Res. Technol. 2021, 247, 309–331. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakour, M.; Laaroussi, H.; Ousaaid, D.; El Ghouizi, A.; Es-Safi, I.; Mechchate, H.; Lyoussi, B. Bee Bread as a Promising Source of Bioactive Molecules and Functional Properties: An Up-To-Date Review. Antibiotics 2022, 11, 203. https://doi.org/10.3390/antibiotics11020203
Bakour M, Laaroussi H, Ousaaid D, El Ghouizi A, Es-Safi I, Mechchate H, Lyoussi B. Bee Bread as a Promising Source of Bioactive Molecules and Functional Properties: An Up-To-Date Review. Antibiotics. 2022; 11(2):203. https://doi.org/10.3390/antibiotics11020203
Chicago/Turabian StyleBakour, Meryem, Hassan Laaroussi, Driss Ousaaid, Asmae El Ghouizi, Imane Es-Safi, Hamza Mechchate, and Badiaa Lyoussi. 2022. "Bee Bread as a Promising Source of Bioactive Molecules and Functional Properties: An Up-To-Date Review" Antibiotics 11, no. 2: 203. https://doi.org/10.3390/antibiotics11020203
APA StyleBakour, M., Laaroussi, H., Ousaaid, D., El Ghouizi, A., Es-Safi, I., Mechchate, H., & Lyoussi, B. (2022). Bee Bread as a Promising Source of Bioactive Molecules and Functional Properties: An Up-To-Date Review. Antibiotics, 11(2), 203. https://doi.org/10.3390/antibiotics11020203








