Oral Vancomycin Prophylaxis for Primary and Secondary Prevention of Clostridioides difficile Infection in Patients Treated with Systemic Antibiotic Therapy: A Systematic Review, Meta-Analysis and Trial Sequential Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Screening and Eligibility
- Population: Patients undergoing SAT;
- Intervention: Administration of OVP;
- Comparators/controls: Standard of care, placebo, or any kind of intervention alternative to vancomycin;
- Outcomes: The main outcome of interest was the occurrence of a CDI episode, defined as first or recurrent; secondary outcome pertained to safety profile of intervention and was represented by the incidence of vancomycin-resistant Enterococcus species (VRE) infection;
- Study design: Randomized, non-randomized and observational (cohort, case-control, observational, cross-sectional) studies were included as long as they were peer-reviewed and published in full; case reports and series (fewer than 10 patients per group) were ineligible.
2.3. Data Extraction
- Study-related variables (authors, year of publication, study country and design, setting);
- Patient-related variables (age, sex, underlying comorbidities);
- Infection-related variables (severity—severe versus mild-moderate forms, as defined by each study; epidemiological origin—community-onset versus healthcare-associated);
- Treatment variables (timing, sequence, dosage and duration of OVP and comparators);
- Concurrent SAT (type of drug, duration);
- Outcome variables (raw numbers of events and adjusted estimates if present).
2.4. Definitions
2.5. Data Synthesis and Analysis
2.6. Publication Bias and Quality Appraisal
3. Results
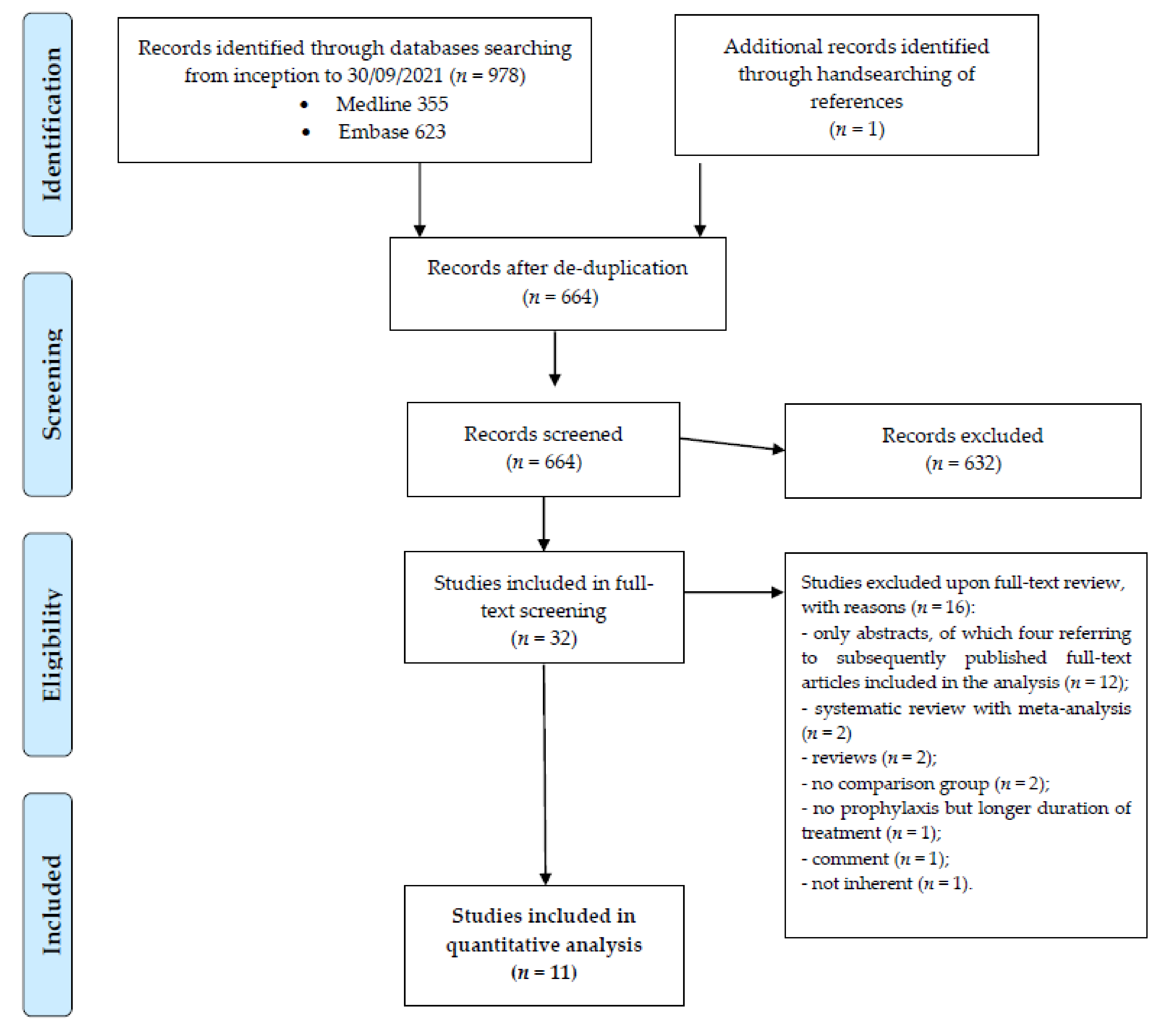
3.1. Study Selection
3.2. Study Characteristics
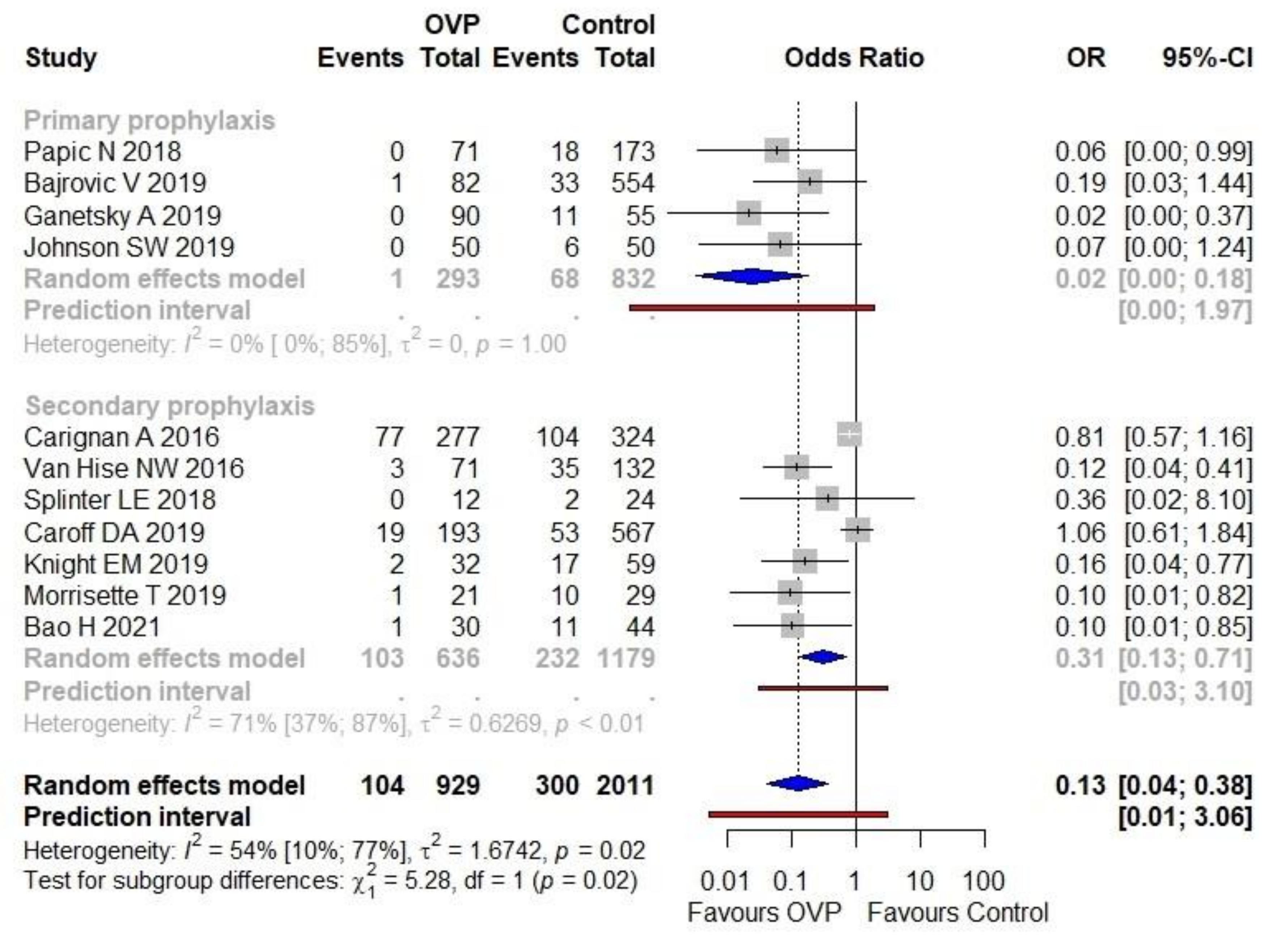
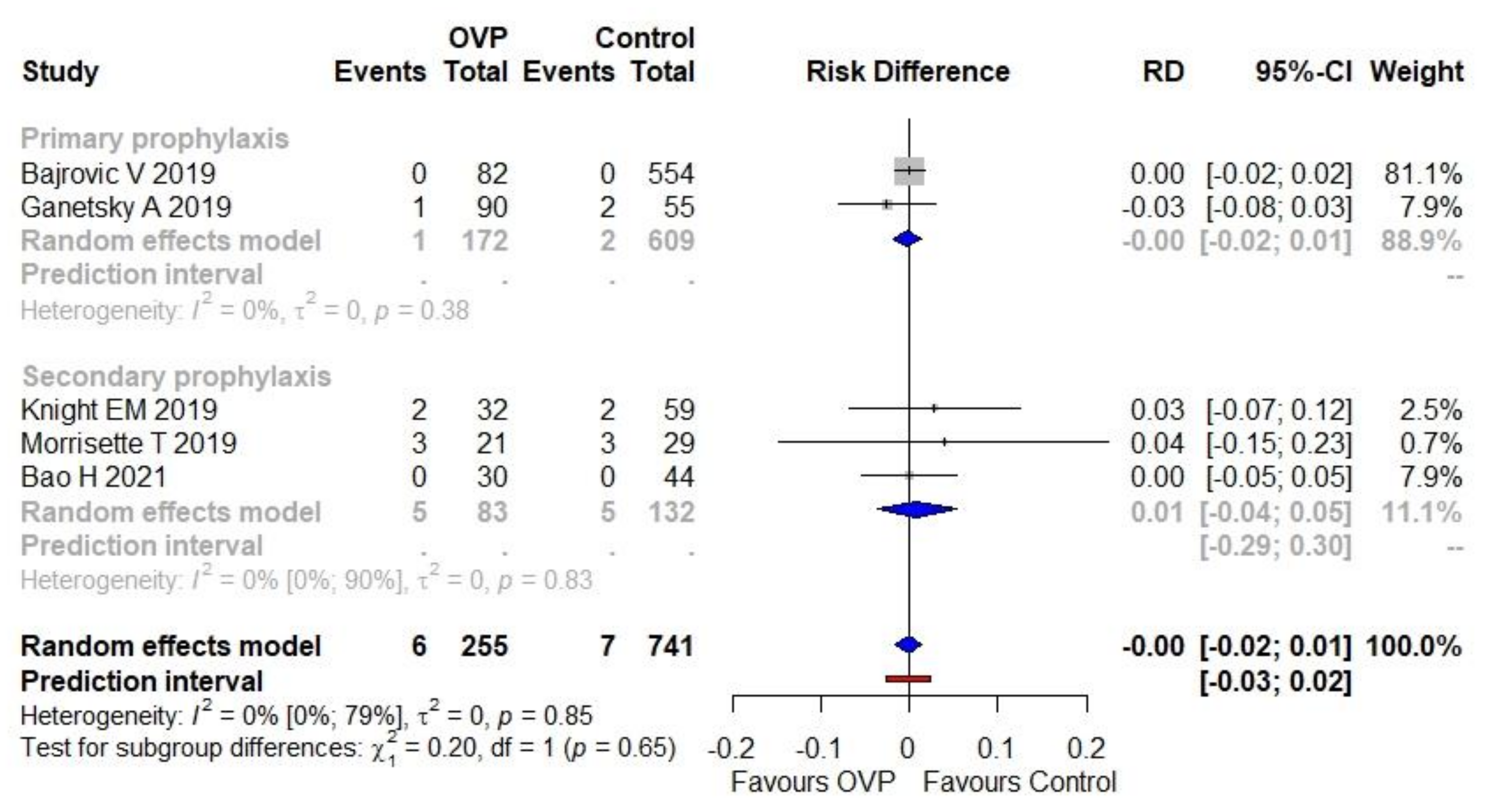
3.3. Primary Outcome
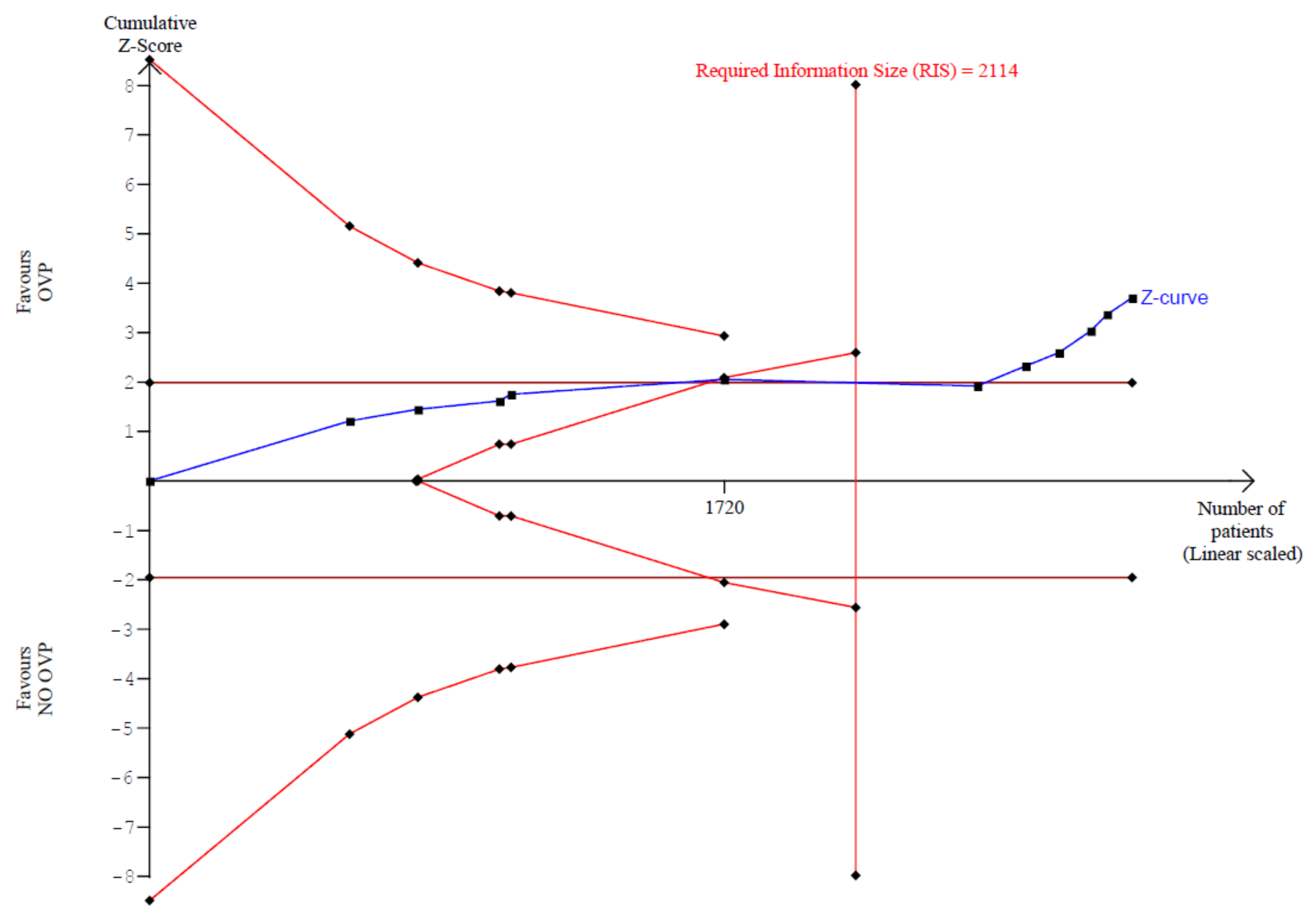
3.3.1. Primary Outcome: Trial Sequential Analysis
3.3.2. Primary Outcome: Sensitivity Analyses
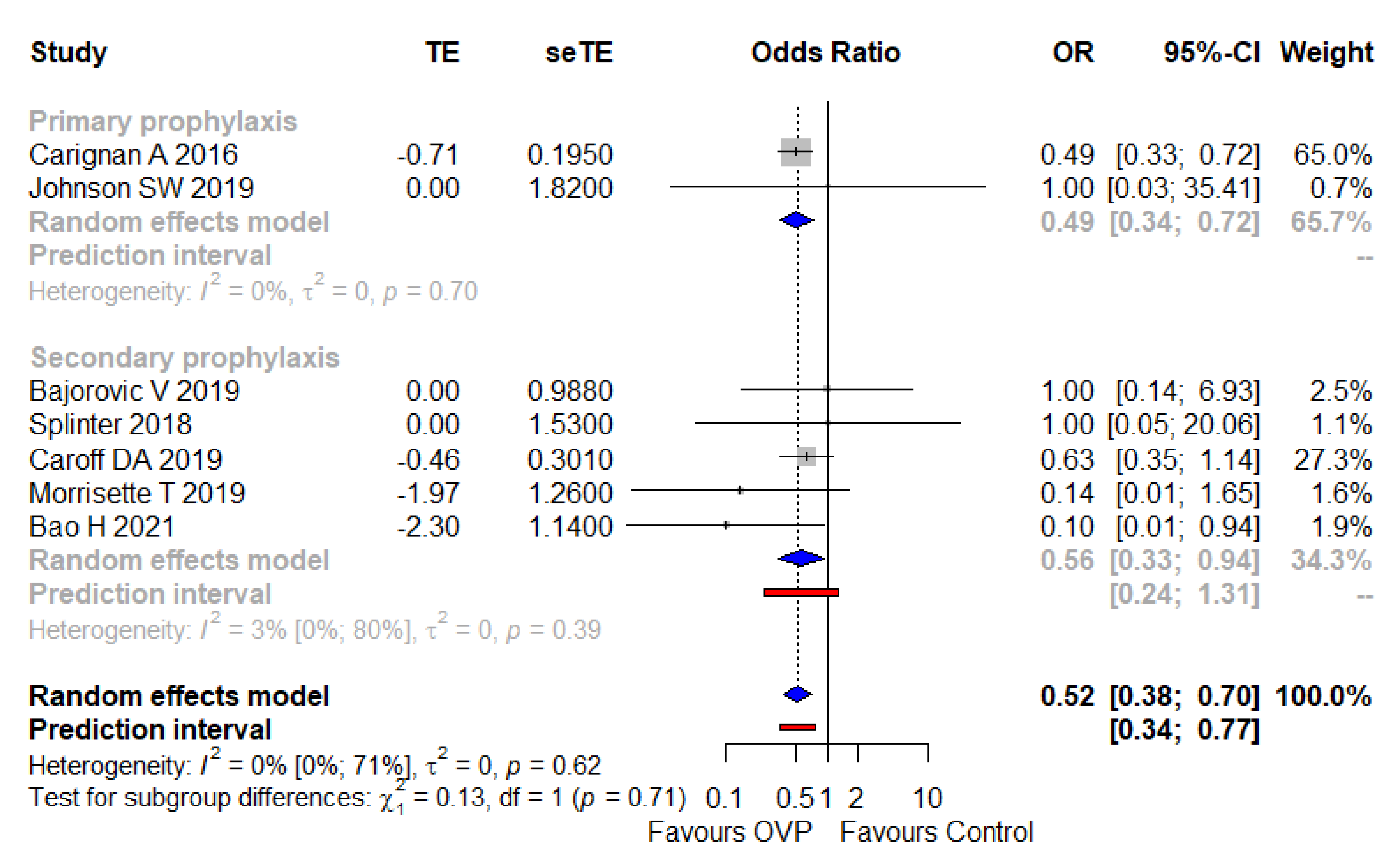
3.4. Secondary Outcome
3.5. Publication Bias and Quality Appraisal
4. Discussion
5. Conclusions
6. Ethics
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Note
References
- Guery, B.; Galperine, T.; Barbut, F. Clostridioides difficile: Diagnosis and treatments. BMJ 2019, 366, l4609. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guh, A.Y.; Kutty., P.K. Clostridioides difficile Infection. Ann. Intern. Med. 2018, 169, ITC49–ITC64. [Google Scholar] [CrossRef] [PubMed]
- Smits, W.K.; Lyras, D.; Lacy, D.B.; Wilcox, M.H.; Kuijper, E.J. Clostridium difficile infection. Nat. Rev. Dis. Primers 2016, 2, 16020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antonelli, M.; Martin-Loeches, I.; Dimopoulos, G.; Gasbarrini, A.; Vallecoccia, M.S. Clostridioides difficile (formerly Clostridium difficile) infection in the critically ill: An expert statement. Intensive Care Med. 2020, 46, 215–224. [Google Scholar] [CrossRef]
- Balsells, E.; Shi, T.; Leese, C.; Lyell, I.; Burrows, J.; Wiuff, C.; Campbell, H.; Kyaw, M.H.; Nair, H. Global burden of Clostridium difficile infections: A systematic review and meta-analysis. J. Glob. Health 2019, 9, 010407. [Google Scholar] [CrossRef]
- Furuya-Kanamori, L.; Stone, J.C.; Clark., J.; McKenzie, S.J.; Yakob, L.; Paterson, D.L.; Riley, T.V.; Doi, S.A.; Clements, A.C. Comorbidities, Exposure to Medications, and the Risk of Community-Acquired Clostridium difficile Infection: A systematic review and meta-analysis. Infect. Control Hosp. Epidemiol. 2015, 36, 132–141. [Google Scholar] [CrossRef] [Green Version]
- Deshpande, A.; Pasupuleti, V.; Thota, P.; Pant, C.; Rolston, D.D.; Hernandez, A.V.; Donskey, C.J.; Fraser, T.G. Risk factors for recurrent Clostridium difficile infection: A systematic review and meta-analysis. Infect. Control Hosp. Epidemiol. 2015, 36, 452–460. [Google Scholar] [CrossRef]
- Reigadas, E.; van Prehn, J.; Falcone, M.; Fitzpatrick, F.; Vehreschild, M.J.G.T.; Kuijper, E.J.; Bouza, E.; European Society of Clinical Microbiology and Infectious Diseases Study Group on Clostridioides difficile (ESGCD); Study Group for Host and Microbiota Interaction (ESGHAMI). How to: Prophylactic interventions for prevention of Clostridioides difficile infection. Clin. Microbiol. Infect. 2021, 27, 1777–1783. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Bramer, W.M.; Milic, J.; Mast, F. Reviewing retrieved references for inclusion in systematic reviews using EndNote. J. Med. Libr. Assoc. 2017, 105, 84–87. [Google Scholar] [CrossRef] [Green Version]
- Muka, T.; Glisic, M.; Milic, J.; Verhoog, S.; Bohlius, J.; Bramer, W.; Chowdhury, R.; Franco, O.H. A 24-step guide on how to design, conduct, and successfully publish a systematic review and meta-analysis in medical research. Eur. J. Epidemiol. 2020, 35, 49–60. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. European Surveillance of Clostridioides (Clostridium) difficile Infections: Surveillance Protocol Version 2.4; ECDC: Stockholm, Sweden, 2019; Available online: https://www.ecdc.europa.eu/sites/default/files/documents/clostridium-difficile-infections-EU-surveillance-protocol-vers2.4.pdf (accessed on 9 October 2021).
- Xu, C.; Furuya-Kanamori, L.; Zorzela, L.; Lin, L.; Vohra, S. A proposed framework to guide evidence synthesis practice for meta-analysis with zero-events studies. J. Clin. Epidemiol. 2021, 135, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Bakbergenuly, I.; Kulinskaya, E. Meta-analysis of binary outcomes via generalized linear mixed models: A simulation study. BMC Med. Res. Methodol. 2018, 18, 70. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IntHout, J.; Ioannidis, J.P.A.; Rovers, M.M.; Goeman, J.J. Plea for routinely presenting prediction intervals in meta-analysis. BMJ Open 2016, 6, e010247. [Google Scholar] [CrossRef] [Green Version]
- Schünemann, H.J.; Vist, G.E.; Higgins, J.P.T.; Santesso, N.; Deeks, J.J.; Glasziou, P.; Akl, E.A.; Guyatt, G.H. Chapter 15: Interpreting results and drawing conclusions. In Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (Updated February 2021); Higgins, J.P.T., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M.J., Welch, V.A., Eds.; Cochrane: London, UK, 2021; Available online: https://training.cochrane.org/handbook (accessed on 10 October 2021).
- Maraolo, A.E.; Giaccone, A.; Gentile, I.; Saracino, A.; Bavaro, D.F. Daptomycin versus Vancomycin for the Treatment of Methicillin-Resistant Staphylococcus aureus Bloodstream Infection with or without Endocarditis: A Systematic Review and Meta-Analysis. Antibiotics 2021, 10, 1014. [Google Scholar] [CrossRef]
- Geissbühler, M.; Hincapié, C.A.; Aghlmandi, S.; Zwahlen, M.; Jüni, P.; da Costa, B.R. Most published meta-regression analyses based on aggregate data sufer from methodological pitfalls: A meta-epidemiological study. BMC Med. Res. Methodol. 2021, 21, 123. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Liu., J.; Tong., T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef] [Green Version]
- Shah, A.; Smith, A.F. Trial sequential analysis: Adding a new dimension to meta-analysis. Anaesthesia 2019, 74, 793–800. [Google Scholar] [CrossRef] [Green Version]
- Kang, H. Trial sequential analysis: Novel approach for meta-analysis. Anesth. Pain Med. 2021, 16, 138–150. [Google Scholar] [CrossRef]
- VanderWeele, T.J.; Ding, P. Sensitivity Analysis in Observational Research: Introducing the E-Value. Ann. Intern. Med. 2017, 167, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Baujat, B.; Mahé, C.; Pignon, J.P.; Hill, C. A graphical method for exploring heterogeneity in meta-analyses: Application to a meta-analysis of 65 trials. Stat. Med. 2002, 21, 2641–2652. [Google Scholar] [CrossRef]
- Olkin, I.; Dahabreh, I.J.; Trikalinos, T.A. GOSH—A graphical display of study heterogeneity. Res. Synth. Methods 2012, 3, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Peters, J.L.; Sutton, A.J.; Jones, D.R.; Abrams, K.R.; Rushton, L. Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J. Clin. Epidemiol. 2008, 61, 991–996. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slim, K.; Nini, E.; Forestier, D.; Kwiatkowski, F.; Panis, Y.; Chipponi, J. Methodological index for non-randomized studies (minors): Development and validation of a new instrument. ANZ J. Surg. 2003, 73, 712–716. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef] [Green Version]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-bias VISualization (robvis): An R package and Shiny web app for visualizing risk-of-bias assessments. Res. Syn. Meth. 2021, 1, 55–61. [Google Scholar] [CrossRef]
- Granholm, A.; Alhazzani, W.; Møller, M.H. Use of the GRADE approach in systematic reviews and guidelines. Br. J. Anaesth. 2019, 123, 554–559. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Oxman, A.D.; Sultan, S.; Glasziou, P.; Akl, E.A.; Alonso-Coello, P.; Atkins, D.; Kunz, R.; Brozek, J.; Montori, V.; et al. GRADE Working Group. GRADE guidelines: 9. Rating up the quality of evidence. J. Clin. Epidemiol. 2011, 64, 1311–1316. [Google Scholar] [CrossRef]
- Carignan, A.; Poulin, S.; Martin, P.; Labbé, A.C.; Valiquette, L.; Al-Bachari, H.; Montpetit, L.P.; Pépin, J. Efficacy of secondary prophylaxis with vancomycin for preventing recurrent Clostridium difficile infections. Am. J. Gastroenterol. 2016, 111, 1834–1840. [Google Scholar] [CrossRef]
- Van Hise, N.W.; Bryant, A.M.; Hennessey, E.K.; Crannage, A.J.; Khoury, J.A.; Manian, F.A. Efficacy of oral vancomycin in preventing recurrent Clostridium difficile infection in patients treated with systemic antimicrobial agents. Clin. Infect. Dis. 2016, 63, 651–653. [Google Scholar] [CrossRef] [Green Version]
- Papic, N.; Maric, L.S.; Vince, A. Efficacy of oral vancomycin in primary prevention of Clostridium difficile infection in elderly patients treated with systemic antibiotic therapy. Infect. Dis. 2018, 50, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Splinter, L.E.; Kerstenetzky, L.; Jorgenson, M.R.; Descourouez, J.L.; Leverson, G.E.; Saddler, C.M.; Smith, J.A.; Safdar, N.; Redfield, R.R. Vancomycin prophylaxis for prevention of Clostridium difficile infection recurrence in renal transplant patients. Ann. Pharmacother. 2018, 52, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Bajrovic, V.; Budev, M.; McCurry, K.R.; Brizendine, K.D. Vancomycin prophylaxis for Clostridium difficile infection among lung transplant recipients. J. Heart Lung Transplant. 2019, 38, 874–876. [Google Scholar] [CrossRef]
- Caroff, D.A.; Menchaca, J.T.; Zhang, Z.; Rhee, C.; Calderwood, M.S.; Kubiak, D.W.; Yokoe, D.S.; Klompas, M. Oral vancomycin prophylaxis during systemic antibiotic exposure to prevent Clostridiodes difficile infection relapses. Infect. Control Hosp. Epidemiol. 2019, 40, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Ganetsky, A.; Han, J.H.; Hughes, M.E.; Babushok, D.V.; Frey, N.V.; Gill, S.I.; Hexner, E.O.; Loren, A.W.; Luger, S.M.; Mangan, J.K.; et al. Oral vancomycin prophylaxis is highly effective in preventing Clostridium difficile infection in allogeneic hematopoietic cell transplant recipients. Clin. Infect. Dis. 2019, 68, 2003–2009. [Google Scholar] [CrossRef] [PubMed]
- Knight, E.M.; Schiller, D.S.; Fulman, M.K.; Rastogi, R. Long-Term Efficacy of Oral Vancomycin Prophylaxis for the Prevention of Clostridium difficile Recurrence. J. Pharm. Pract. 2020, 33, 633–639. [Google Scholar] [CrossRef]
- Morrisette, T.; Van Matre, A.G.; Miller, M.A.; Mueller, S.W.; Bajrovic, V.; Abidi, M.Z.; Benamu, E.; Kaiser, J.N.; Barber, G.R.; Chase, S.; et al. Oral vancomycin prophylaxis as secondary prevention against Clostridioides difficile infection in the hematopoietic stem cell transplantation and hematologic malignancy population. Biol. Blood Marrow Transplant. 2019, 25, 2091–2209. [Google Scholar] [CrossRef]
- Johnson, S.W.; Brown, S.V.; Priest, D.H. Effectiveness of oral vancomycin for prevention of healthcare facility-onset Clostridioides difficile infection in targeted patients during systemic antibiotic exposure. Clin. Infect. Dis. 2020, 71, 1133–1139. [Google Scholar] [CrossRef]
- Bao, H.; Lighter, J.; Dubrovskaya, Y.; Merchan, C.; Siegfried, J.; Papadopoulos, J.; Jen, S.P. Oral Vancomycin as Secondary Prophylaxis for Clostridioides difficile Infection. Pediatrics 2021, 148, e2020031807. [Google Scholar] [CrossRef] [PubMed]
- Babar, S.; El Kurdi, B.; El Iskandarani, M.; Haddad, I.; Imam, Z.; Alomari, M.; Myers, J.; Moorman, J. Oral vancomycin prophylaxis for the prevention of Clostridium difficile infection: A systematic review and meta-analysis. Infect. Control Hosp. Epidemiol. 2020, 41, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Tariq, R.; Laguio-Vila, M.; Tahir, M.W.; Orenstein, R.; Pardi, D.S.; Khanna, S. Efficacy of oral vancomycin prophylaxis for prevention of Clostridioides difficile infection: A systematic review and meta-analysis. Therap. Adv. Gastroenterol. 2021, 14, 1756284821994046. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.R.; Fischer, M.; Allegretti, J.R.; LaPlante, K.; Stewart, D.B.; Limketkai, B.N.; Stollman, N.H. ACG Clinical Guidelines: Prevention, Diagnosis, and Treatment of Clostridioides difficile Infections. Am. J. Gastroenterol. 2021, 116, 1124–1147. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.; Lavergne, V.; Skinner, A.M.; Gonzales-Luna, A.J.; Garey, K.W.; Kelly, C.P.; Wilcox, M.H. Clinical Practice Guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused Update Guidelines on Management of Clostridioides difficile Infection in Adults. Clin. Infect. Dis. 2021, 73, 755–757. [Google Scholar] [CrossRef]
- Van Prehn, J.; Reigadas, E.; Vogelzang, E.H.; Bouza, E.; Hristea, A.; Guery, B.; Krutova, M.; Norén, T.; Allerberger, F.; Coia, J.; et al. European Society of Clinical Microbiology and Infectious Diseases: 2021 update on the treatment guidance document for Clostridioides difficile infection in adults. Clin. Microbiol. Infect. 2021, 27, S1–S21. [Google Scholar] [CrossRef]
- Garey, K.W. Perils, Pitfalls, and Promise of Primary Prophylaxis for Clostridioides difficile Infection. Clin. Infect. Dis. 2020, 71, 1140–1141. [Google Scholar] [CrossRef]
- Tobar-Marcillo, M.; Guerrero-Duran, M.; Avecillas-Segovia, A.; Pacchiano-Aleman, L.; Basante-Díaz, R.; Vela-Vizcaino, H.; Espinosa-Aznar, E.; Castorena García, P.; Santiago-Ramírez, R.; Rivas-Bucio, I. Metronidazole in the prevention of antibiotic-associated diarrhoea and Clostridium difficile infection in high-risk hospitalised patients. Gastroenterol. Hepatol. 2018, 41, 362–368. [Google Scholar] [CrossRef]
- Mullane, K.M.; Winston, D.J.; Nooka, A.; Morris, M.I.; Stiff, P.; Dugan, M.J.; Holland, H.; Gregg, K.; Adachi, J.A.; Pergam, S.A.; et al. A randomized, placebo-controlled trial of fidaxomicin for prophylaxis of Clostridium difficile-associated diarrhea in adults undergoing hematopoietic stem cell transplantation. Clin. Infect. Dis. 2019, 68, 196–203. [Google Scholar] [CrossRef] [Green Version]
- Johnson, S. Potential Risks and Rewards with Prophylaxis for Clostridium difficile Infection. Clin. Infect. Dis. 2016, 63, 654–655. [Google Scholar] [CrossRef] [Green Version]
- Johnson, S.; Homann, S.R.; Bettin, K.M.; Quick, J.N.; Clabots, C.R.; Peterson, L.R.; Gerding, D.N. Treatment of asymptomatic Clostridium difficile carriers (fecal excretors) with vancomycin or metronidazole. A randomized, placebo-controlled trial. Ann. Intern. Med. 1992, 117, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Isaac, S.; Scher, J.U.; Djukovic, A.; Jiménez, N.; Littman, D.R.; Abramson, S.B.; Pamer, E.G.; Ubeda, C. Short- and long-term effects of oral vancomycin on the human intestinal microbiota. J. Antimicrob. Chemother. 2017, 72, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Falcone, M.; Russo, A.; Iraci, F.; Carfagna, P.; Goldoni, P.; Vullo, V.; Venditti, M. Risk Factors and Outcomes for Bloodstream Infections Secondary to Clostridium difficile Infection. Antimicrob. Agents Chemother. 2015, 60, 252–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saha, S.; Kapoor, S.; Tariq, R.; Schuetz, A.N.; Tosh, P.K.; Pardi, D.S.; Khanna, S. Increasing antibiotic resistance in Clostridioides difficile: A systematic review and meta-analysis. Anaerobe 2019, 58, 35–46. [Google Scholar] [CrossRef]
- Chan, J.D.; Bryson-Cahn, C.; Jain, R.; Lynch, J.B.; Liu, C. Does oral vancomycin prophylaxis during systemic antibiotic exposure prevent Clostridioides difficile infection relapses? Still in search of an answer. Infect. Control Hosp. Epidemiol. 2019, 40, 1084–1086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borgia, G.; Maraolo, A.E.; Foggia, M.; Buonomo, A.R.; Gentile, I. Fecal microbiota transplantation for Clostridium difficile infection: Back to the future. Expert Opin. Biol. Ther. 2015, 15, 1001–1014. [Google Scholar] [CrossRef] [PubMed]
- Giacobbe, D.R.; Dettori, S.; Di Bella, S.; Vena, A.; Granata, G.; Luzzati, R.; Petrosillo, N.; Bassetti, M. Bezlotoxumab for Preventing Recurrent Clostridioides difficile Infection: A Narrative Review from Pathophysiology to Clinical Studies. Infect. Dis. Ther. 2020, 9, 481–494. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.C.; Manis, M.M.; Bohm, N.M.; Curry, S.R. Oral Vancomycin for Secondary Prophylaxis of Clostridium difficile Infection. Ann. Pharmacother. 2019, 53, 396–401. [Google Scholar] [CrossRef]
- Cataldo, M.A.; Granata, G.; D’Arezzo, S.; Tonziello, G.; Vulcano, A.; De Giuli, C.; Meledandri, M.; Di Caro, A.; Petrosillo, N. Hospitalized patients with diarrhea: Rate of Clostridioides difficile infection underdiagnosis and drivers of clinical suspicion. Anaerobe 2021, 70, 102380. [Google Scholar] [CrossRef]
- Granata, G.; Petrosillo, N.; Adamoli, L.; Bartoletti, M.; Bartoloni, A.; Basile, G.; Bassetti, M.; Bonfanti, P.; Borromeo, R.; Ceccarelli, G.; et al. Prospective Study on Incidence, Risk Factors and Outcome of Recurrent Clostridioides difficile Infections. J. Clin. Med. 2021, 10, 1127. [Google Scholar] [CrossRef]
- Castro, I.; Tasias, M.; Calabuig, E.; Salavert, M. Doctor, my patient has CDI and should continue to receive antibiotics. The (unresolved) risk of recurrent CDI. Rev. Esp. Quimioter. 2019, 32 (Suppl. S2), 47–54. [Google Scholar] [PubMed]
- Dubberke, E.R.; Yan, Y.; Reske, K.A.; Butler, A.M.; Doherty, J.; Pham, V.; Fraser, V.J. Development and Validation of a Clostridium difficile Infection Risk Prediction Model. Infect. Control Hosp. Epidemiol. 2011, 32, 360–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, J.; Makar, M.; Fusco, C.; McCaffrey, R.; Rao, K.; Ryan, E.E.; Washer, L.; West, L.R.; Young, V.B.; Guttag, J.; et al. A Generalizable, Data-Driven Approach to Predict Daily Risk of Clostridium difficile Infection at Two Large Academic Health Centers. Infect. Control Hosp. Epidemiol. 2018, 39, 425–433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Cassai, A.; Pasin, L.; Boscolo, A.; Salvagno, M.; Navalesi, P. Trial sequential analysis: Plain and simple. Korean J. Anesthesiol. 2021, 74, 363–365. [Google Scholar] [CrossRef]
- Slimings, C.; Riley, T.V. Antibiotics and healthcare facility-associated Clostridioides difficile infection: Systematic review and meta-analysis 2020 update. J. Antimicrob. Chemother. 2021, 76, 1676–1688. [Google Scholar] [CrossRef] [PubMed]
- D’Silva, K.M.; Mehta, R.; Mitchell, M.; Lee, T.C.; Singhal, V.; Wilson, M.G.; McDonald, E.G. Proton pump inhibitor use and risk for recurrent Clostridioides difficile infection: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Bunnell, K.L.; Danziger, L.H.; Johnson, S. Economic Barriers in the Treatment of Clostridium difficile Infection with Oral Vancomycin. Open Forum Infect. Dis. 2017, 4, ofx078. [Google Scholar] [CrossRef] [Green Version]
| Author [Ref.] | Year of Publication | Study Design and Type of Prophylaxis | Study Period | N. Patients (OVP vs. Controls) | Characteristics of the Population | OVP Dose | OVP Duration | Event Definition | Time of Follow-Up | Incidence of CDI (OVP vs. Controls) | More Frequent Class of Antibiotic Used as SAT (OVP vs. Controls) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Carignan et al. [33] | 2016 | Retrospective cohort—Secondary | 2003–2011 | 227 vs. 324 | Adults receiving antibiotics within 90 days of initial/recurrent CDI/secondary prophylaxis | 125 mg qid (84%) | 7 days | diarrhea + toxin evidence or typical colitis | 6 months | 28% vs. 32% | NA (however, patients receiving second-generation cephalosporins more likely to experience rCDI) |
| Van Hise et al. [34] | 2016 | Retrospective cohort—Secondary | 2010–2014 | 71 vs. 132 | Adults with history of CDI, subsequently hospitalized and treated with systemic antimicrobial therapy/secondary prophylaxis | 125 mg or 250 bid (59%) | 14 days | diarrhea + NAAT | 4 weeks | 4.2% vs. 26.6% (p < 0.001) | Aminopenicillin, 49.3% vs. 47.7% (p = 0.88) |
| Papic et al. [35] | 2018 | Retrospective cohort—Primary | 2015–2017 | 71 vs. 173 | Elderly patients hospitalized for more than 72 h who received parenteral antibiotics for more than 24 h/primary prophylaxis | 125 mg once daily | 9 days | diarrhea + two-stage algorithm (GDH for screening and NAAT) | During index hospitalization | 0% vs. 10.4% (p = 0.0022) | Piperacillin-tazobactam (53.5%) vs. any cephalosporin (43.4%) |
| Splinter et al. [36] | 2018 | Retrospective cohort—Secondary | 2012–2015 | 11 vs.18 | Adults renal transplanted patients with history of CDI/secondary prophylaxis | 125 mg bid | 19 days | NAAT | 30 days | 0% vs. 8 % (p = 0.54) | NA |
| Bajrovic et al. [37] | 2019 | Retrospective cohort—Primary | 2007–2013 | 82 vs. 554 | Adults receiving lung transplantation | 125 mg bid (median) | 14 days | diarrhea + NAAT | During index hospitalization | 1.2% vs. 5.9% | Intravenous vancomycin, 100% vs. 69% (p < 0.01) |
| Caroff et al. [38] | 2019 | Retrospective cohort—Secondary | 2009–2015 | 193 vs. 597 | Adults given at least 1 dose of systemic antibiotic with history of CDI in previous 30–150 days/secondary prophylaxis | NA | 2 days | toxin evidence or NAAT | 90 days | 9.8% vs. 9.4% | High-risk antibiotics according to study’s definition, 66% vs. 85% (p < 0.01) |
| Ganetsky et al. [39] | 2019 | Retrospective cohort—Primary * | 2015–2016 | 90 vs. 55 | Adults receiving allogenic hematopoietic cell transplantation | 125 mg bid for the duration of stay | 29 days | 2/3-stage algorithm (GDH for screening, toxin detection or NAAT) | 90 days | 0% vs. 20% (p < 0.001) | Anti-Gram-negative antibiotics according to study’s definition, 76% vs. 71% (p = 0.54) |
| Knight et al. [40] | 2019 | Retrospective cohort—Secondary | 2013–2015 | 32 vs. 59 | Adults with history of CDI, subsequently hospitalized within 12 months and treated with systemic antimicrobials/secondary prophylaxis | 125 or 250 (69%) mg qid | 8.5 days | diarrhea + NAAT | 12 months | 6.3% vs. 27.8% (p = 0.011) | Penicillins vs. cephalosporins (in terms of sums of daily doses received) |
| Morrisette et al. [41] | 2019 | Retrospective cohort—Secondary | 2014–2018 | 21 vs. 29 | Hematological adults with and without HSCT treated for the initial episode of CDI first with planned oral vancomycin monotherapy and must have been receiving a BSA at time of CDI diagnosis and/or during the course of CDI treatment/prophylaxis | 125 mg bid | 7 days | diarrhea + NAAT | 60 days | 10% vs. 35% (p = 0.051) | Third/fourth generation cephalosporins, 95% vs. 93% (p > 0.99) |
| Johnson et al. [42] | 2019 | Randomized, prospective, open label—Primary | 2018–2019 | 50 vs. 50 | Adults admitted for more than 72 h, aged ≥60 years, hospitalized ≤30 days prior to the index hospitalization, and received systemic antibiotics during that prior hospitalization | 125 mg once daily | 12 days | diarrhea + NAAT | 3 months post-discharge | 0 vs. 12% (p = 0.03) | Cephalosporins (in terms of days of therapy, 173 vs. 171) |
| Bao et al. [43] | 2021 | Retrospective cohort—Secondary | 2013–2019 | 30 vs. 44 | Pediatric population, 50% affected by a malignancy | 10 mg/kg (up to 125 mg per dose for non-severe CDI and 500 mg per dose for severe or fulminant CDI) | 12 days | diarrhea + NAAT or typical colitis | 8 weeks | 3% vs. 25% (p = 0.02) | Third/fourth generation cephalosporins, 64% vs. 57% (p = 0.55) |
| Variable | Included Studies, n | Sample Size, n | OR (95% CI) | I2 | Test for Subgroup Difference, p Value |
|---|---|---|---|---|---|
| Study place | 0.51 | ||||
| US | 9 | 2095 | 0.15 (0.06–0.36) | 59% | |
| Not US | 2 | 845 | 0.31 (0.04–2.26) | 0% | |
| Study design | 0.99 | ||||
| Retrospective | 10 | 2840 | 0.19 (0.09–0.44) | 59.4% | |
| Prospective | 1 | 100 | 0.07 (0.00–1.24) | - | |
| Study population | 0.30 | ||||
| Adult hospitalized | 6 | 1999 | 0.27 (0.10–0.78) | 64.1% | |
| SOT | 2 | 672 | 0.16 (0.02–1.21) | 0% | |
| Hematological | 2 | 195 | 0.03 (0.00–0.23) | 0% | |
| Pediatric | 1 | 74 | 0.10 (0.01–0.85) | - | |
| OVP dose * | 0.11 | ||||
| 125 mg od | 2 | 344 | 0.06 (0.01–0.48) | 0% | |
| 125 bid | 5 | 951 | 0.11 (0.04–0.32) | 0% | |
| Other (variable/mixed dosages) | 4 | 1645 | 0.43 (0.15–1.23) | 78.5% | |
| Timing of follow-up | <0.01 | ||||
| 28/30-day | 2 | 239 | 0.12 (0.03–0.39) | 0% | |
| 90-day | 3 | 1461 | 0.82 (0.61–1.11) | 0% | |
| In-hospital | 3 | 1025 | 0.03 (0.00–0.22) | 0% | |
| Other | 3 | 207 | 0.12 (0.04–0.36) | 0% | |
| Mean duration of OVP | 0.01 | ||||
| (Compared with SAT) | |||||
| Longer | 7 | 1244 | 0.08 (0.03–0.18) | 0% | |
| Shorter | 4 | 1696 | 0.44 (0.16–1.23) | 40% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maraolo, A.E.; Mazzitelli, M.; Zappulo, E.; Scotto, R.; Granata, G.; Andini, R.; Durante-Mangoni, E.; Petrosillo, N.; Gentile, I. Oral Vancomycin Prophylaxis for Primary and Secondary Prevention of Clostridioides difficile Infection in Patients Treated with Systemic Antibiotic Therapy: A Systematic Review, Meta-Analysis and Trial Sequential Analysis. Antibiotics 2022, 11, 183. https://doi.org/10.3390/antibiotics11020183
Maraolo AE, Mazzitelli M, Zappulo E, Scotto R, Granata G, Andini R, Durante-Mangoni E, Petrosillo N, Gentile I. Oral Vancomycin Prophylaxis for Primary and Secondary Prevention of Clostridioides difficile Infection in Patients Treated with Systemic Antibiotic Therapy: A Systematic Review, Meta-Analysis and Trial Sequential Analysis. Antibiotics. 2022; 11(2):183. https://doi.org/10.3390/antibiotics11020183
Chicago/Turabian StyleMaraolo, Alberto Enrico, Maria Mazzitelli, Emanuela Zappulo, Riccardo Scotto, Guido Granata, Roberto Andini, Emanuele Durante-Mangoni, Nicola Petrosillo, and Ivan Gentile. 2022. "Oral Vancomycin Prophylaxis for Primary and Secondary Prevention of Clostridioides difficile Infection in Patients Treated with Systemic Antibiotic Therapy: A Systematic Review, Meta-Analysis and Trial Sequential Analysis" Antibiotics 11, no. 2: 183. https://doi.org/10.3390/antibiotics11020183
APA StyleMaraolo, A. E., Mazzitelli, M., Zappulo, E., Scotto, R., Granata, G., Andini, R., Durante-Mangoni, E., Petrosillo, N., & Gentile, I. (2022). Oral Vancomycin Prophylaxis for Primary and Secondary Prevention of Clostridioides difficile Infection in Patients Treated with Systemic Antibiotic Therapy: A Systematic Review, Meta-Analysis and Trial Sequential Analysis. Antibiotics, 11(2), 183. https://doi.org/10.3390/antibiotics11020183










