Early Diagnosis and Antibiotic Treatment Combined with Multicomponent Hemodynamic Support for Addressing a Severe Case of Lemierre’s Syndrome
Abstract
:1. Introduction
2. Case Presentation
2.1. History and Hospital Admission
2.2. Intensive Care Unit Management
3. Discussion
- Without resolution in less than a week;
- With unusual lateral cervical pain, dysphagia, and swelling as well as trismus, unilateral (or bilateral) anterior cervical lymphadenopathy, and induration at the angle of the mandible;
- With diffuse abscesses (imaging);
- Followed by systemic (e.g., fever, malaise, confusion, stupor) and/or respiratory findings such as dyspnea or tachypnea;
- Succeeded by a progressive worsening of the clinical picture with hemodynamic and laboratory problems suggestive for sepsis.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Walkty, A.; Embil, J. Lemierre’s Syndrome. N. Engl. J. Med. 2019, 380, e16. [Google Scholar] [CrossRef]
- Srivali, N.; Ungprasert, P.; Kittanamongkolchai, W.; Ammannagari, N. Lemierr’s syndrome: An often missed life-threatening infection. Indian J. Crit. Care Med. 2014, 18, 170–172. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Jean, S.S.; Chen, F.L.; Hsieh, S.M.; Hsueh, P.R. Lemierre’s syndrome: A forgotten and re-emerging infection. J. Microbiol. Immunol. Infect. 2020, 53, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Hagelskjaer Kristensen, L.; Prag, J. Lemierre’s syndrome and other disseminated Fusobacterium necrophorum infections in Denmark: A prospective epidemiological and clinical survey. Eur. J. Clin. Microbiol. Infect. Dis. 2008, 27, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Riordan, T.; Wilson, M. Lemierre’s syndrome: More than a historical curiosa. Postgrad. Med. J. 2004, 80, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Osowicki, J.; Kapur, S.; Phuong, L.K.; Dobson, S. The long shadow of lemierre’s syndrome. J. Infect. 2017, 74 (Suppl. S1), S47–S53. [Google Scholar] [CrossRef]
- Eilbert, W.; Singla, N. Lemierre’s syndrome. Int. J. Emerg. Med. 2013, 6, 40. [Google Scholar] [CrossRef] [Green Version]
- Karkos, P.D.; Asrani, S.; Karkos, C.D.; Leong, S.C.; Theochari, E.G.; Alexopoulou, T.D.; Assimakopoulos, A.D. Lemierre’s syndrome: A systematic review. Laryngoscope 2009, 6, 1552–1559. [Google Scholar] [CrossRef]
- Raith, E.P.; Udy, A.A.; Bailey, M.; McGloughlin, S.; MacIsaac, C.; Bellomo, R.; Pilcher, D.V. Australian and New Zealand Intensive Care Society (ANZICS) Centre for Outcomes and Resource Evaluation (CORE). Prognostic Accuracy of the SOFA Score, SIRS Criteria, and qSOFA Score for In-Hospital Mortality Among Adults With Suspected Infection Admitted to the Intensive Care Unit. JAMA 2017, 317, 290–300. [Google Scholar] [PubMed]
- Croft, D.P.; Philippo, S.M.; Prasad, P. A case of Lemierre’s syndrome with septic shock and complicated parapneumonic effusions requiring intrapleural fibrinolysis. Respir. Med. Case Rep. 2015, 16, 86–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nemakayala, D.R.; P Rai, M.; Kavuturu, S.; Rayamajhi, S. Atypical Presentation of Lemierre’s Syndrome Causing Septic Shock and Acute Respiratory Distress Syndrome. Case Rep. Infect. Dis. 2018, 2018, 5469053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wright, W.F.; Shiner, C.N.; Ribes, J.A. Lemierre syndrome. South Med. J. 2012, 6, 283–288. [Google Scholar] [CrossRef]
- Kuppalli, K.; Livorsi, D.; Talati, N.J.; Osborn, M. Lemierre’s syndrome due to Fusobacterium necrophorum. Lancet Infect. Dis. 2012, 12, 808–815. [Google Scholar] [CrossRef]
- De Waele, J.J. Early source control in sepsis. Langenbecks Arch. Surg. 2010, 395, 489–494. [Google Scholar] [CrossRef]
- Cramer, J.D.; Purkey, M.R.; Smith, S.S.; Schroeder, J.W., Jr. The impact of delayed surgical drainage of deep neck abscesses in adult and pediatric populations. Laryngoscope 2016, 126, 1753–1760. [Google Scholar] [CrossRef]
- Huang, T.T.; Liu, T.C.; Chen, P.R.; Tseng, F.Y.; Yeh, T.H.; Chen, Y.S. Deep neck infection: Analysis of 185 cases. Head Neck. 2004, 26, 854–860. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.A.; Thamboo, A.; Burton, M.J.; Diamond, C.; Nunez, D.A. Needle aspiration versus incision and drainage for the treatment of peritonsillar abscess. Cochrane Database Syst Rev. 2016, 12, CD006287. [Google Scholar] [CrossRef] [PubMed]
- Gehrke, T.; Scherzad, A.; Hagen, R.; Hackenberg, S. Deep neck infections with and without mediastinal involvement: Treatment and outcome in 218 patients. In European Archives of Oto-Rhino-Laryngology; Springer: Berlin/Heidelberg, Germany, 2021. [Google Scholar] [CrossRef]
- Potter, J.K.; Herford, A.S.; Ellis, E., 3rd. Tracheotomy versus endotracheal intubation for airway management in deep neck space infections. J. Oral. Maxillofac. Surg. 2002, 60, 349–354; discussion 354–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Griffiths, J.; Barber, V.S.; Morgan, L.; Young, J.D. Systematic review and meta-analysis of studies of the timing of tracheostomy in adult patients undergoing artificial ventilation. BMJ 2005, 330, 1243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrer, R.; Martin-Loeches, I.; Phillips, G.; Osborn, T.M.; Townsend, S.; Dellinger, R.P.; Artigas, A.; Schorr, C.; Levy, M.M. Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: Results from a guideline-based performance improvement program. Crit. Care Med. 2014, 42, 1749–1755. [Google Scholar] [CrossRef] [PubMed]
- Brazier, J.S.; Hall, V.; Yusuf, E.; Duerden, B.I. Fusobacterium necrophorum infections in England and Wales 1990–2000. J. Med. Microbiol. 2002, 51, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Klasinc, R.; Lupyr, K.; Zeller, I.; Paula, H.; Makristathis, A.; Tuchmann, F.; Wrba, T.; Assadian, O.; Presterl, E. Clinical characteristics of a large cohort of patients with positive culture of Fusobacterium necrophorum. GMS Infect. Dis. 2018, 6, Doc03. [Google Scholar] [CrossRef]
- Jensen, A.; Hagelskjaer Kristensen, L.; Nielsen, H.; Prag, J. Minimum requirements for a rapid and reliable routine identification and antibiogram of Fusobacterium necrophorum. Eur. J. Clin. Microbiol. Infect. Dis 2008, 27, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Hagelskjaer Kristensen, L.; Prag, J. Human Necrobacillosis, with Emphasis on Lemierre’s Syndrome. Clin. Infect. Dis. 2000, 31, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Gulliford, M.C.; Moore, M.V.; Little, P.; Hay, A.D.; Fox, R.; Prevost, A.T.; Juszczyk, D.; Charlton, J.; Ashworth, M. Safety of reduced antibiotic prescribing for self limiting respiratory tract infections in primary care: Cohort study using electronic health records. BMJ 2016, 354, i3410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akata, K.; Muratani, T.; Yatera, K.; Naito, K.; Noguchi, S.; Yamasaki, K.; Kawanami, T.; Kido, T.; Mukae, H. Induction of plasmid-mediated AmpC β-lactamase DHA-1 by piperacillin/tazobactam and other β-lactams in Enterobacteriaceae. PLoS ONE 2019, 14, e0218589. [Google Scholar] [CrossRef]
- Leone, S.; Damiani, G.; Pezone, I.; Kelly, M.E.; Cascella, M.; Alfieri, A.; Pace, M.C.; Fiore, M. New antimicrobial options for the management of complicated intra-abdominal infections. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 819–827. [Google Scholar] [CrossRef]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med. 2017, 43, 304–377. [Google Scholar] [CrossRef] [PubMed]
- Landry, D.W.; Oliver, J.A. The pathogenesis of vasodilatatory shock. N. Eng. J. Med. 2001, 345, 588–595. [Google Scholar] [CrossRef] [PubMed]
- Buckley, J.F.; Singer, M.; Clapp, L.H. Role of KATP channels in sepsis. Cardiovasc. Res. 2006, 72, 220–230. [Google Scholar] [CrossRef]
- Ukor, I.F.; Walley, K.R. Vasopressin in Vasodilatory Shock. Crit. Care Clin. 2019, 35, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Annane, D.; Pastores, S.M.; Arlt, W.; Balk, R.A.; Beishuizen, A.; Briegel, J.; Carcillo, J.; Christ-Crain, M.; Cooper, M.S.; Marik, P.E.; et al. Critical illness-related corticosteroid insufficiency (CIRCI): A narrative review from a Multispecialty Task Force of the Society of Critical Care Medicine (SCCM) and the European Society of Intensive Care Medicine (ESICM). Intensive Care Med. 2017, 43, 1781–1792. [Google Scholar] [CrossRef] [PubMed]
- Bichet, D.G. Regulation of Thirst and Vasopressin Release. Annu. Rev. Physiol. 2019, 81, 359–373. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.A. Vasopressin in septic shock. Crit. Care Med. 2007, 35 (Suppl. S9), S609–S615. [Google Scholar] [CrossRef]
- Holmes, C.L.; Patel, B.M.; Russell, J.A.; Walley, K.R. Physiology of vasopressin relevant to management of septic shock. Chest 2001, 120, 989–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russell, J.A.; Walley, K.R.; Singer, J.; Gordon, A.C.; Hébert, P.C.; Cooper, D.J.; Holmes, C.L.; Mehta, S.; Granton, J.T.; Storms, M.M.; et al. Vasopressin versus norepinephrine infusion in patients with septic shock. N. Eng. J. Med. 2008, 358, 877–887. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russell, J.A.; Walley, K.R.; Gordon, A.C.; Cooper, D.J.; Hébert, P.C.; Singer, J.; Holmes, C.L.; Mehta, S.; Granton, J.T.; Storms, M.M.; et al. Interaction of vasopressin infusion, corticosteroid treatment, and mortality of septic shock. Crit. Care Med. 2009, 37, 811–818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Annane, D.; Sébille, V.; Charpentier, C.; Bollaert, P.E.; François, B.; Korach, J.M.; Capellier, G.; Cohen, Y.; Azoulay, E.; Troché, G.; et al. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 2002, 288, 862–871. [Google Scholar] [CrossRef] [Green Version]
- Moreno, R.; Sprung, C.L.; Annane, D.; Chevret, S.; Briegel, J.; Keh, D.; Singer, M.; Weiss, Y.G.; Payen, D.; Cuthbertson, B.H.; et al. Time course of organ failure in patients with septic shock treated with hydrocortisone: Results of the Corticus study. Intensive Care Med. 2011, 37, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.C.; Mason, A.J.; Thirunavukkarasu, N.; Perkins, G.D.; Cecconi, M.; Cepkova, M.; Pogson, D.G.; Aya, H.D.; Anjum, A.; Frazier, G.J.; et al. Effect of Early Vasopressin vs. Norepinephrine on Kidney Failure in Patients With Septic Shock: The VANISH Randomized Clinical Trial. JAMA 2016, 316, 509–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schurr, J.W.; Szumita, P.M.; De Grado, J.R. Neuroendocrine Derangements in Early Septic Shock: Pharmacotherapy for Relative Adrenal and Vasopressin Insufficiency. Shock 2017, 48, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.C.; Mason, A.J.; Perkins, G.D.; Stotz, M.; Terblanche, M.; Ashby, D.; Brett, S.J. The interaction of vasopressin and corticosteroids in septic shock: A pilot randomized controlled trial. Crit. Care Med. 2014, 42, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
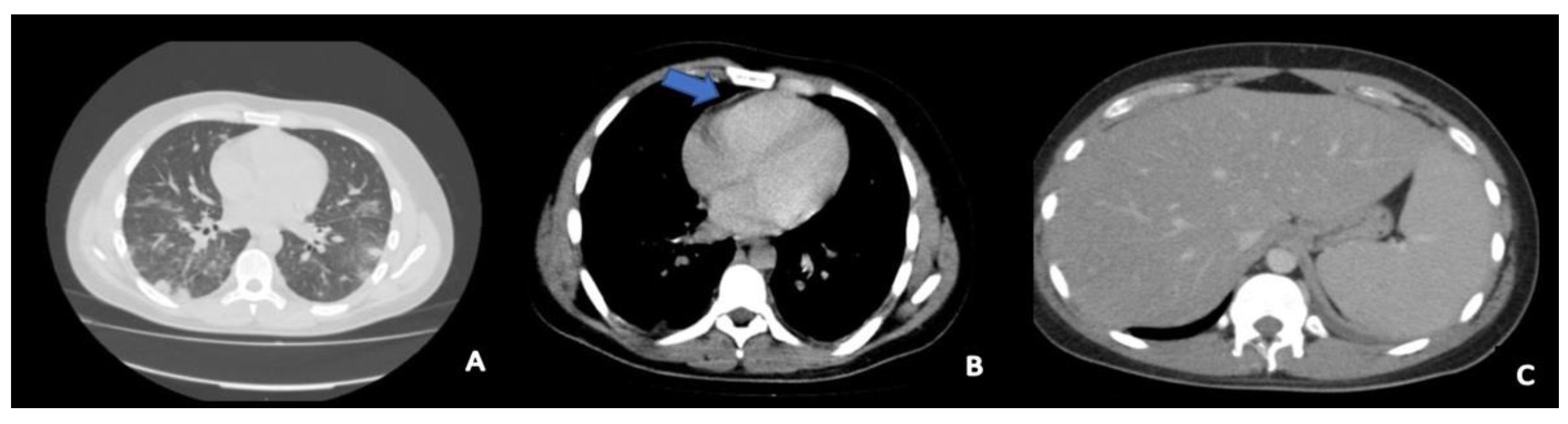
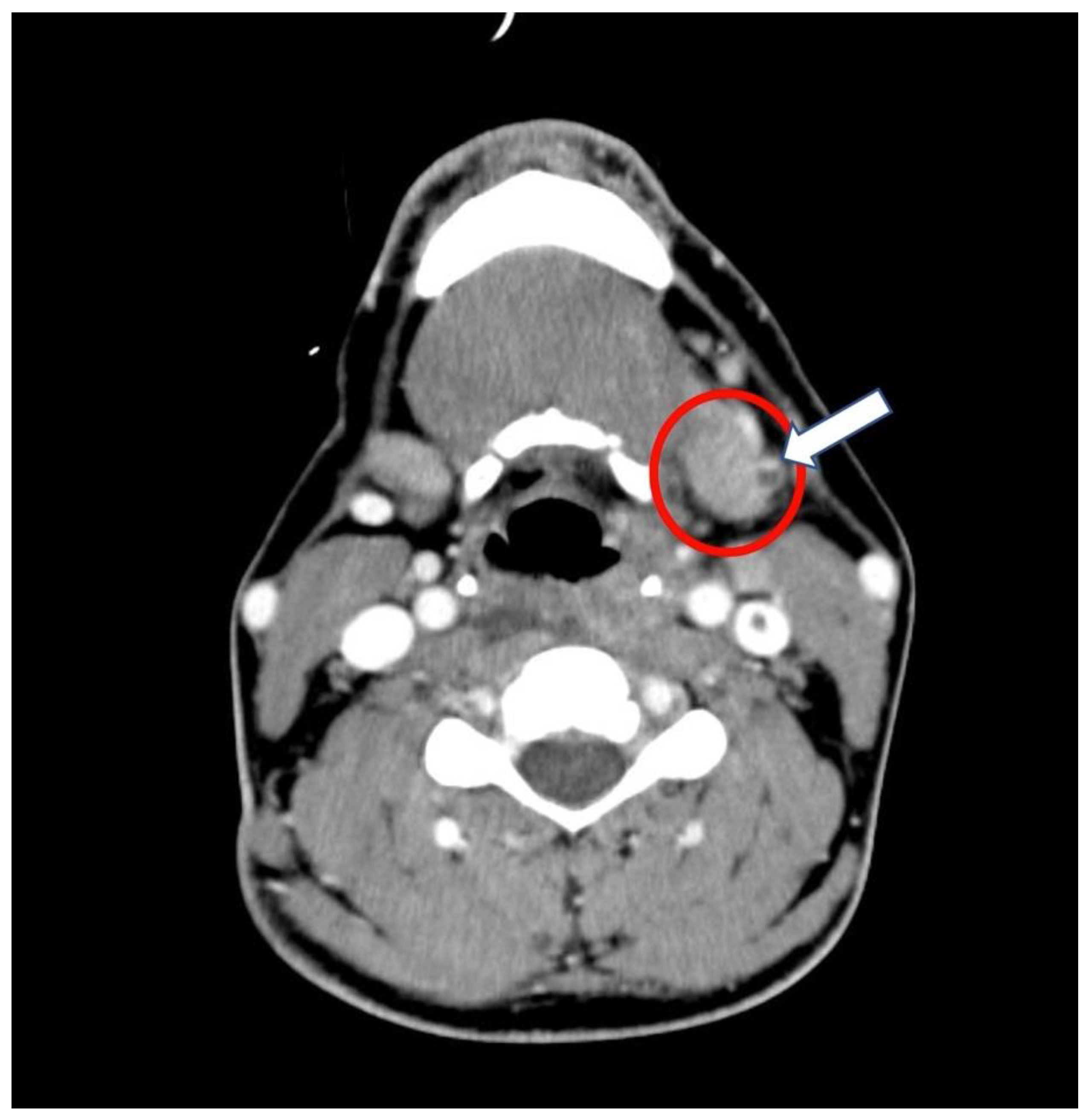
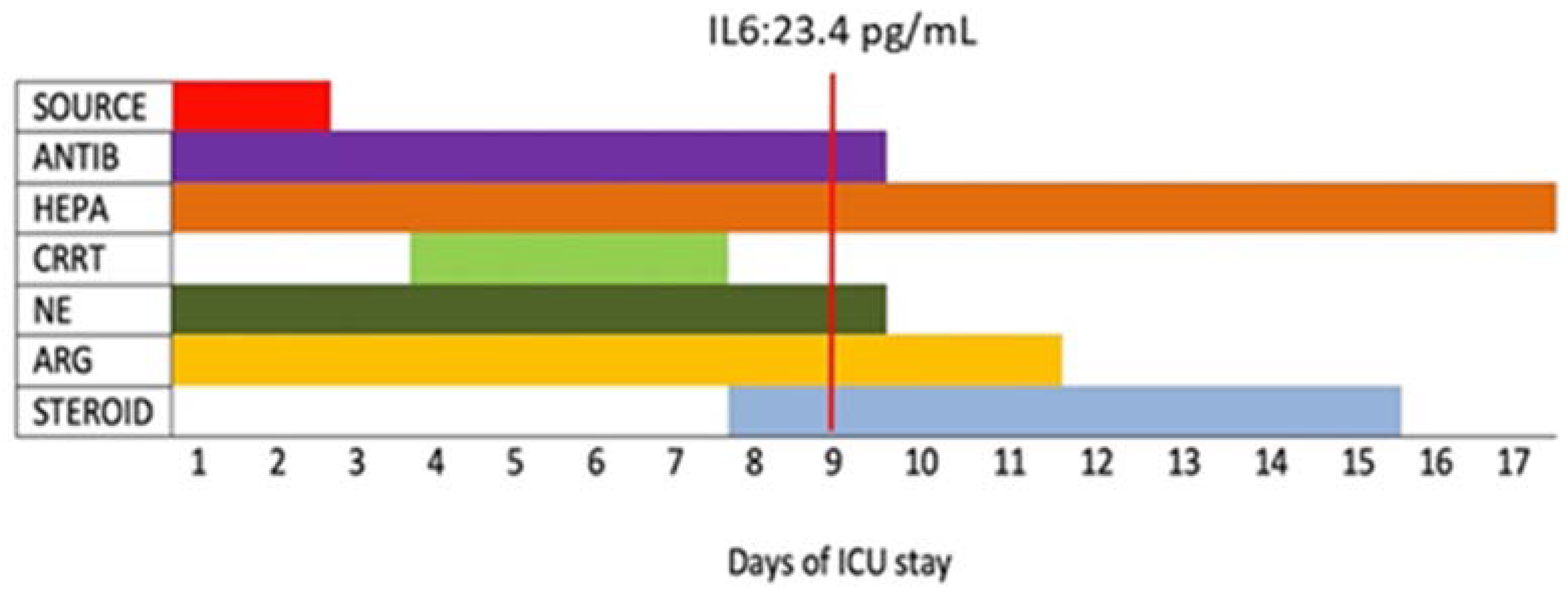
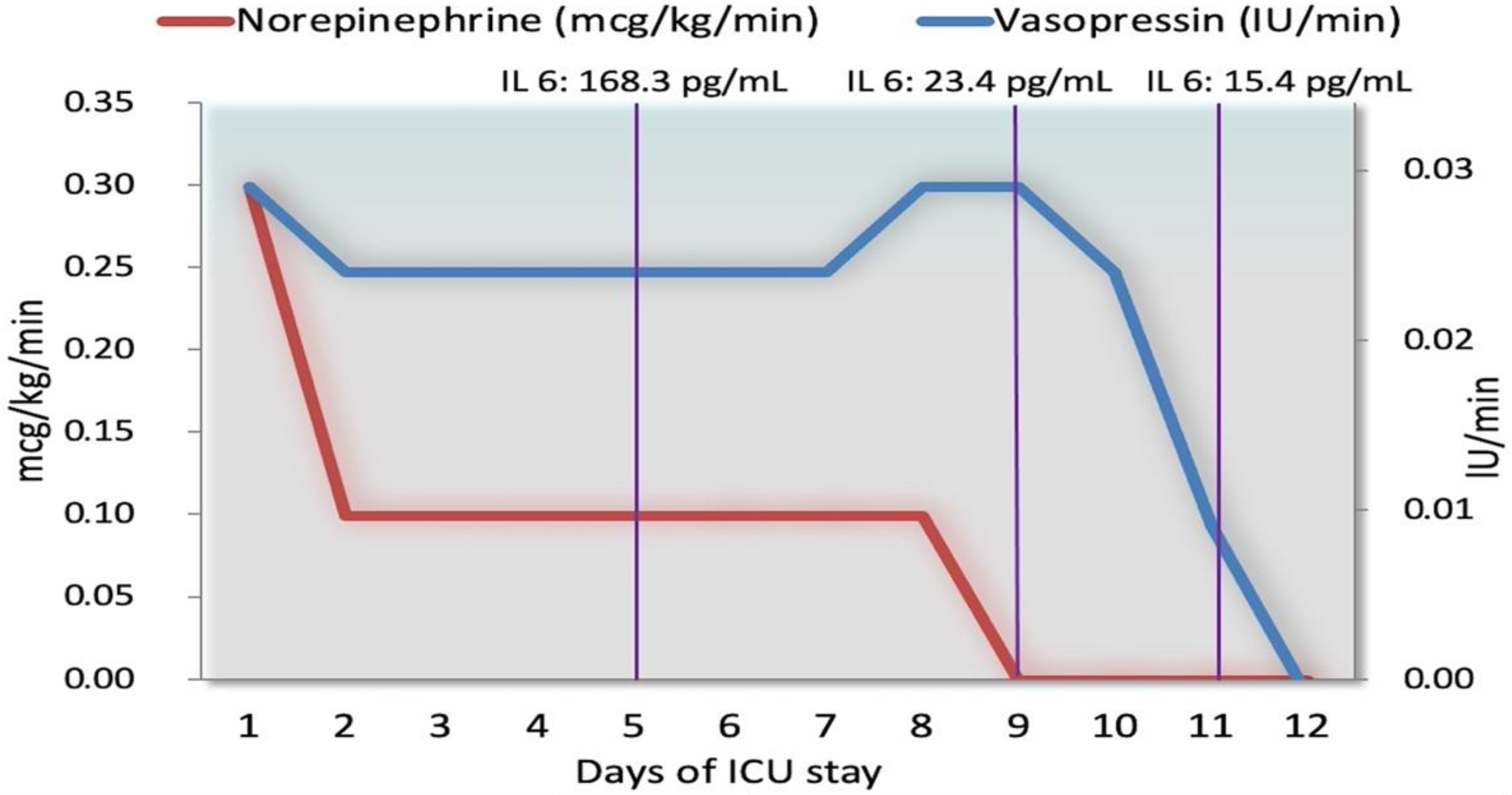
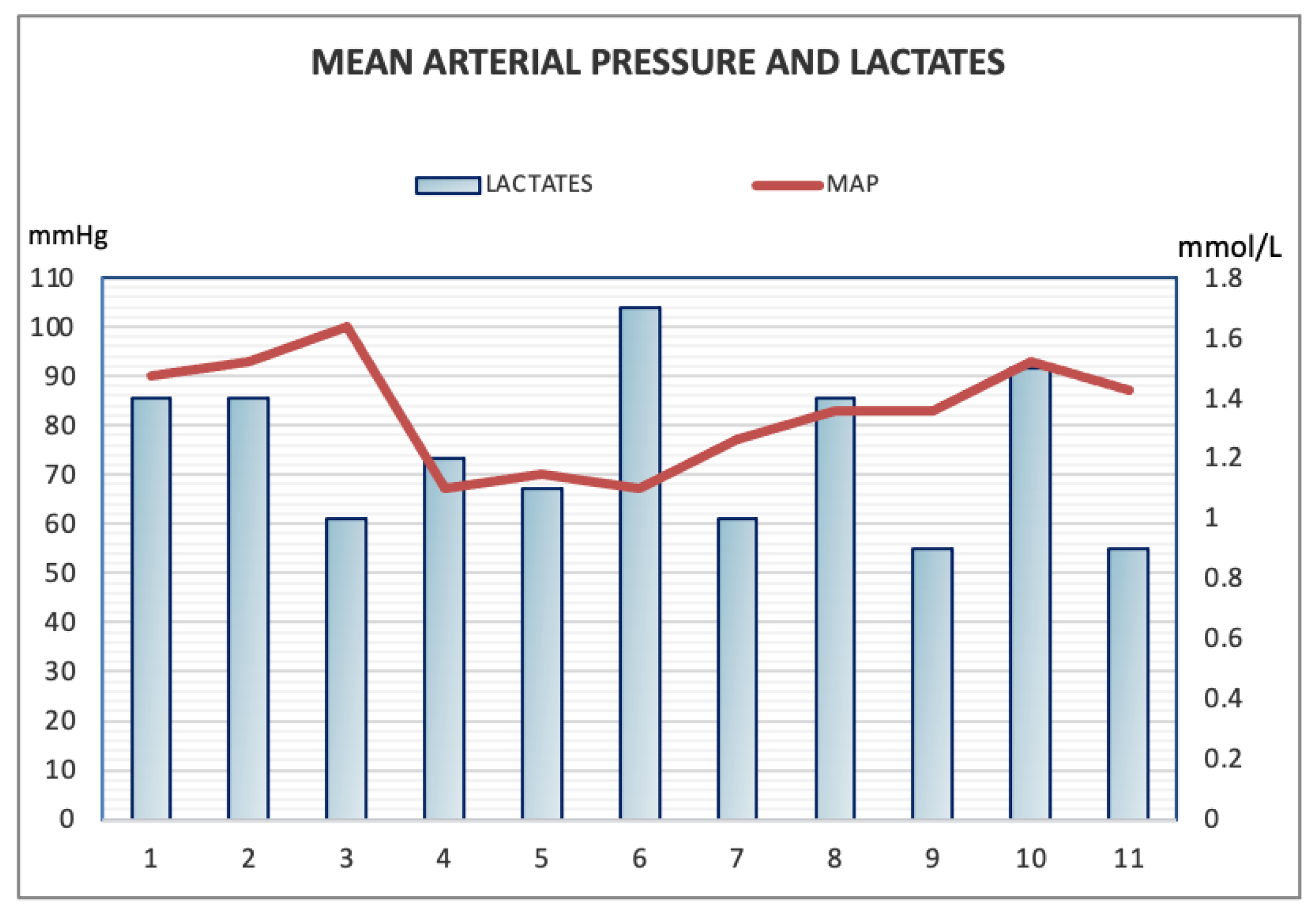
| Day 1 | Day 2 | Day 4 to 6 1 | Day 9 | Day 11 | Day 17 | |
|---|---|---|---|---|---|---|
| Laboratory | ||||||
| WBC (103/mcL) | 12.8 | 12.45 | 16.49 | 11.86 | 6.06 | 6.66 |
| Platelets (103/mcL) | 62 | 94 | 86 | 312 | 318 | 266 |
| Hemoglobin (g/dL) | 12.3 | 10.6 | 6.8 | 8.2 | 8.1 | 9.1 |
| Procalcitonin (ng/mL) | 100.7 | 56.6 | 13.3 | 2.09 | 1.22 | 0.2 |
| IL-6 (pg/mL) | No data | No data | 168.3 | 23.4 | 15.4 | No data |
| Fibrinogen (mg/dL) | 811 | 636 | 519 | 613 | 504 | 422 |
| D-dimer (ng/mL) | 2514 | No data | 4177 | 2266 | 5574 | No data |
| Total bilirubin (mg/dL) | 7.5 | 5.5 | 6.9 | 12.5 | 14.5 | 3.9 |
| Creatinine (mg/dL) | 0.8 | 0.8 | 0.8 | 0,7 | 0.6 | 0.5 |
| Clinical data | ||||||
| Temperature (°C) | 38.3 | 38.8 | 39.3 | 39.7 | 38 | 37.2 |
| PaO2/FiO2 (FiO2%) | 298 (80%) | 259 (55%) | 77 (80%) | 134 (0.65) | 248 (0.5) | 355 (0.3) 2 |
| Lactic acid (mmol/L) | 1.4 | 1.4 | 1.8 | 0.9 | 1.4 | 1 |
| Blood Pressure (mean) (mmHg) | 130/70 (90) | 140/70 (93) | 100/50 (95) | 130/60 (83) | 120/70 (87) | 120/60 (80) |
| Heart rate (rpm) | 100 | 100 | 95 | 100 | 60 | 80 |
| Cardiac Output (L/min) | No data | No data | 15 | No data | 8.7 | No data |
| SVR 1 (dynes/seconds/cm) SOFA SCORE | No data 12 | No data 10 | 300 13 | No data 7 | 917 6 | No data 3 |
| Vasoconstrictors doses | ||||||
| Argipressin (IU/min) | 0.03 | 0.025 | 0.025 | 0.03 | 0.01 | No drug |
| Norepinephrine (mcg/kg/min) | 0.3 | 0.1 | 0.1 | No drug | No drug | No drug |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Recchia, A.; Cascella, M.; Altamura, S.; Borrelli, F.; De Nittis, N.; Dibenedetto, E.; Labonia, M.; Pavone, G.; Del Gaudio, A. Early Diagnosis and Antibiotic Treatment Combined with Multicomponent Hemodynamic Support for Addressing a Severe Case of Lemierre’s Syndrome. Antibiotics 2021, 10, 1526. https://doi.org/10.3390/antibiotics10121526
Recchia A, Cascella M, Altamura S, Borrelli F, De Nittis N, Dibenedetto E, Labonia M, Pavone G, Del Gaudio A. Early Diagnosis and Antibiotic Treatment Combined with Multicomponent Hemodynamic Support for Addressing a Severe Case of Lemierre’s Syndrome. Antibiotics. 2021; 10(12):1526. https://doi.org/10.3390/antibiotics10121526
Chicago/Turabian StyleRecchia, Andreaserena, Marco Cascella, Sabrina Altamura, Felice Borrelli, Nazario De Nittis, Elisabetta Dibenedetto, Maria Labonia, Giovanna Pavone, and Alfredo Del Gaudio. 2021. "Early Diagnosis and Antibiotic Treatment Combined with Multicomponent Hemodynamic Support for Addressing a Severe Case of Lemierre’s Syndrome" Antibiotics 10, no. 12: 1526. https://doi.org/10.3390/antibiotics10121526
APA StyleRecchia, A., Cascella, M., Altamura, S., Borrelli, F., De Nittis, N., Dibenedetto, E., Labonia, M., Pavone, G., & Del Gaudio, A. (2021). Early Diagnosis and Antibiotic Treatment Combined with Multicomponent Hemodynamic Support for Addressing a Severe Case of Lemierre’s Syndrome. Antibiotics, 10(12), 1526. https://doi.org/10.3390/antibiotics10121526






