Understanding Antibiotic Usage on Small-Scale Dairy Farms in the Indian States of Assam and Haryana Using a Mixed-Methods Approach—Outcomes and Challenges
Abstract
:1. Introduction
2. Results
2.1. Farmer Training
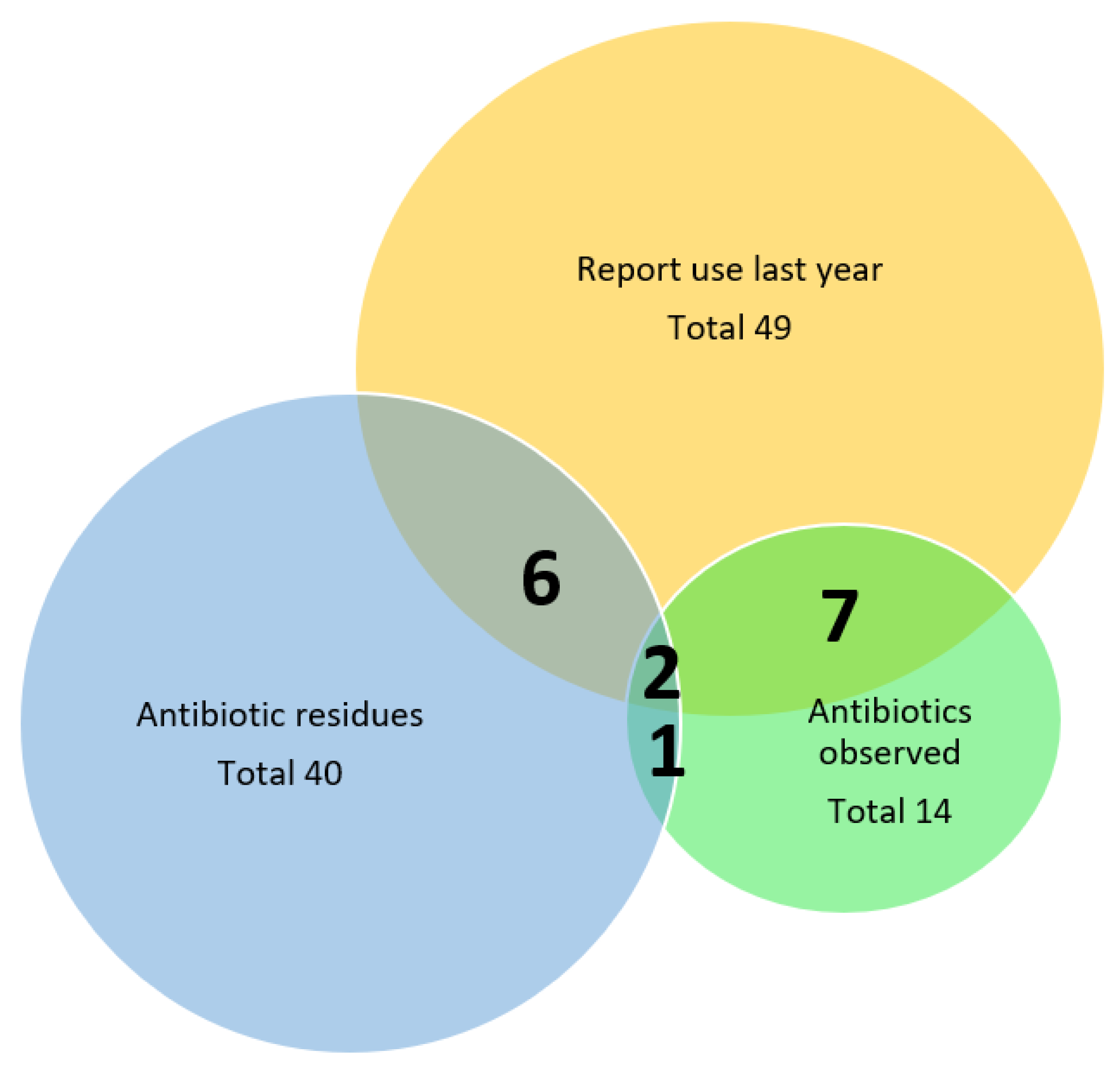
2.2. Knowledge and Use of Antibiotics
2.3. Visual Recognition of Antibiotics
2.4. On-Farm Inspection
2.5. Milk Samples
2.6. Veterinary Services
2.7. Multivariable Analysis
3. Discussion
4. Conclusions
5. Materials and Methods
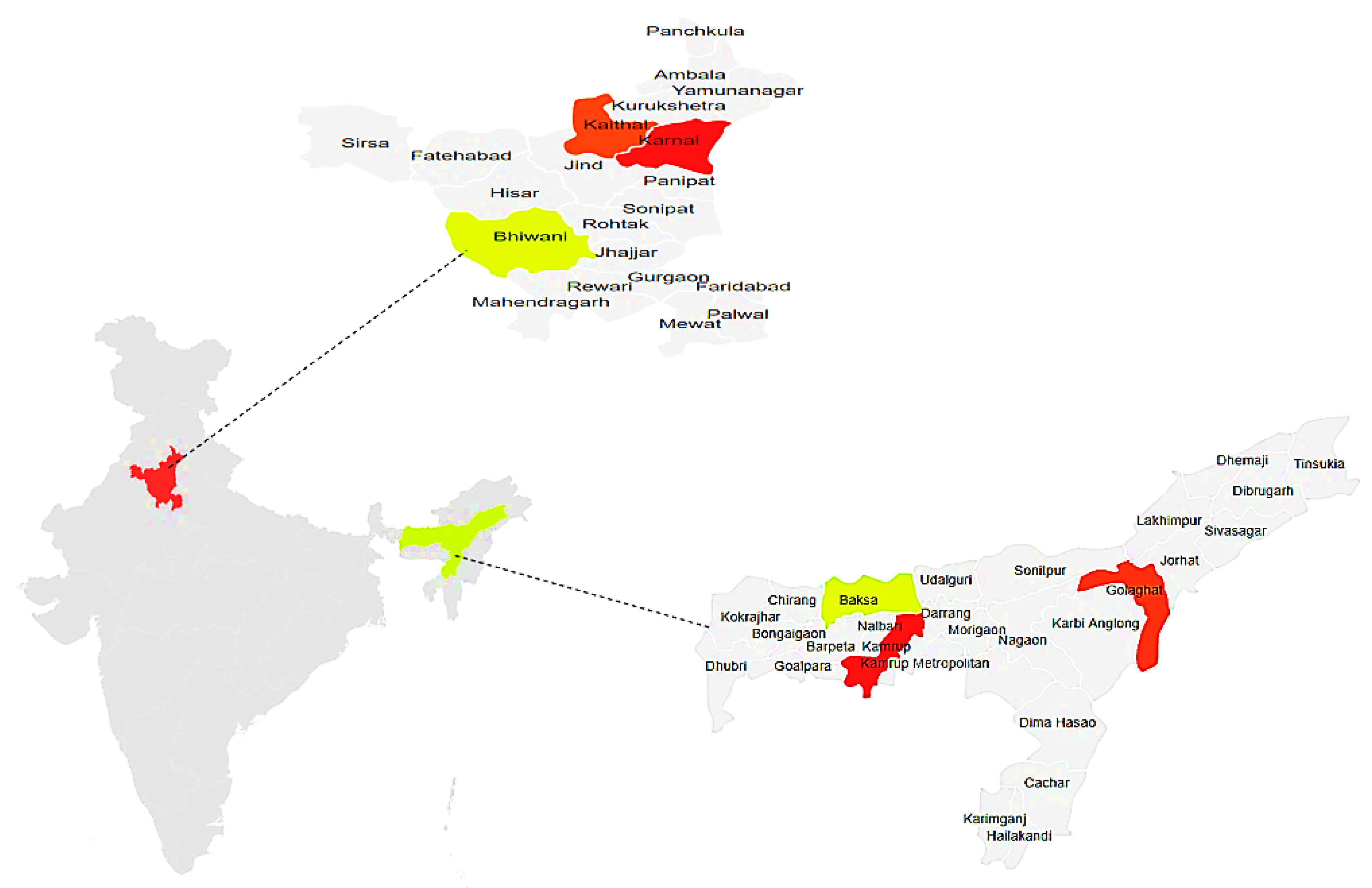
5.1. Sampling Design
5.2. Ethics Statement
5.3. Data Collection
5.4. Analysis of Antibiotic Residues
5.5. Antibiotic Residue Analysis by Dipicolinic Acid (DPA) and Strip Test
5.6. Quantitative Charm ROSA (Rapid One Step Assay) Test
5.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vijay, D.; Bedi, J.S.; Dhaka, P.; Singh, R.; Singh, J.; Arora, A.K.; Gill, J.P.S. Knowledge, attitude, and practices (KAP) survey among veterinarians, and risk factors relating to antimicrobial use and treatment failure in dairy herds of India. Antibiotics 2021, 10, 216. [Google Scholar] [CrossRef] [PubMed]
- Mutua, F.; Sharma, G.; Grace, D.; Bandyopadhyay, S.; Shome, B.; Lindahl, J. A review of animal health and drug use practices in India, and their possible link to antimicrobial resistance. Antimicrob. Resist. Infect. Control 2020, 9, 103. [Google Scholar] [CrossRef]
- Kumar, A.; Wright, I.A.; Singh, D.K. Adoption of food safety practices in milk production: Implications for dairy farmers in India. J. Int. Food Agribus. Mark. 2011, 23, 330–344. [Google Scholar] [CrossRef]
- Sachi, S.; Ferdous, J.; Sikder, M.H.; Hussani, S.M.A.K. Antibiotic residues in milk: Past, present, and future. J. Adv. Vet. Anim. Res. 2019, 6, 315. [Google Scholar] [CrossRef] [PubMed]
- Cuong, N.V.; Padungtod, P.; Thwaites, G.; Carrique-Mas, J.J. Antimicrobial usage in animal production: A review of the literature with a focus on low- and middle-income countries. Antibiotics 2018, 7, 75. [Google Scholar] [CrossRef] [Green Version]
- Ben, Y.; Fu, C.; Hu, M.; Liu, L.; Wong, M.H.; Zheng, C. Human health risk assessment of antibiotic resistance associated with antibiotic residues in the environment: A review. Environ. Res. 2019, 169, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Jindal, P.; Bedi, J.; Singh, R.; Aulakh, R.; Gill, J.P. Epidemiological assessment of antibiotic residues in dairy farm milk and farm waste and water in northern India. Environ. Sci. Pollut. Res. 2021, 28, 29455–29466. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Gupta, J. Prevailing practices in the use of antibiotics by dairy farmers in Eastern Haryana region of India. Vet. World 2018, 11, 274–280. [Google Scholar] [CrossRef] [Green Version]
- Douphrate, D.I.; Hagevoort, G.R.; Nonnenmann, M.W.; Lunner Kolstrup, C.; Reynolds, S.J.; Jakob, M.; Kinsel, M. The dairy industry: A brief description of production practices, trends, and farm characteristics around the world. J. Agromedicine 2013, 18, 187–197. [Google Scholar] [CrossRef]
- Ministry of Finance. Prices, agriculture and food management. In Economic Survey Volume II, 1st ed.; Ministry of Finance: New Delhi, India, 2015. [Google Scholar]
- Chauhan, A.S.; George, M.S.; Chatterjee, P.; Lindahl, J.; Grace, D.; Kakkar, M. The social biography of antibiotic use in smallholder dairy farms in India. Antimicrob. Resist. Infect. Control 2018, 7, 60. [Google Scholar] [CrossRef] [Green Version]
- Sharma, G.; Mutua, F.; Deka, R.P.; Shome, R.; Bandyopadhyay, S.; Shome, B.; Goyal Kumar, N.; Grace, D.; Dey, T.K.; Venugopal, N.; et al. A qualitative study on antibiotic use and animal health management in smallholder dairy farms of four regions of India. Infect. Ecol. Epidemiol. 2020, 10, 1792033. [Google Scholar] [CrossRef]
- Barker, A.K.; Brown, K.; Ahsan, M.; Sengupta, S.; Safdar, N. What drives inappropriate antibiotic dispensing? A mixed-methods study of pharmacy employee perspectives in Haryana, India. BMJ Open 2017, 7, e013190. [Google Scholar] [CrossRef]
- Lindahl, J.F.; Deka, R.P.; Asse, R.; Lapar, L.; Grace, D. Hygiene knowledge, attitudes and practices among dairy value chain actors in Assam, north-east India and the impact of a training intervention. Infect. Ecol. Epidemiol. 2018, 8, 1555444. [Google Scholar] [CrossRef]
- Ranjalkar, J.; Chandy, S.J. India’s national action plan for antimicrobial resistance—An overview of the context, status, and way ahead. J. Fam. Med. Prim. Care 2019, 8, 1828. [Google Scholar] [CrossRef] [PubMed]
- FSSAI. Food Safety and Standards (Contaminants, Toxins And Residues) Regulations, 2011; FSSAI: New Delhi, India, 2011.
- FSSAI. Gazette Notification on Food Safety and Standards (Contaminants, Toxins and Residues) Amendment Regulation Related to Tolerance Limit of Antibiotics and Pharmacology Active Substances; FSSAI: New Delhi, India, 2018.
- Deka, R.P.; Magnusson, U.; Grace, D.; Shome, R.; Lindahl, J.F. Knowledge and practices of dairy farmers relating to brucellosis in urban, peri-urban and rural areas of Assam and Bihar, India. Infect. Ecol. Epidemiol. 2020, 10, 1769531. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, J.F.; Goyal Kumar, N.; Deka, R.P.; Shome, R.; Grace, D. Serological evidence of Brucella infections in dairy cattle in Haryana, India. Infect. Ecol. Epidemiol. 2018, 8, 1555445. [Google Scholar] [CrossRef]
- EU. Commission Regulation (EU) No 37/2010. Off. J. Eur. Union 2010, 1–72. Available online: https://ec.europa.eu/health/sites/default/files/files/eudralex/vol-5/reg_2010_37/reg_2010_37_en.pdf (accessed on 1 August 2021).
- Moudgil, P.; Bedi, J.S.; Aulakh, R.S.; Gill, J.P.S. Analysis of antibiotic residues in raw and commercial milk in Punjab, India vis-à-vis human health risk assessment. J. Food Saf. 2019, 39, e12643. [Google Scholar] [CrossRef]
- Moudgil, P.; Bedi, J.S.; Moudgil, A.D.; Gill, J.P.S.; Aulakh, R.S. Emerging issue of antibiotic resistance from food producing animals in India: Perspective and legal framework. Food Rev. Int. 2018, 34, 447–462. [Google Scholar] [CrossRef]
- Parkunan, T.; Ashutosh, M.; Sukumar, B.; Chera, J.S.; Ramadas, S.; Chandrasekhar, B.; Kumar, S.A.; Sharma, R.; Kumar, M.S.; De, S. Antibiotic resistance: A cross-sectional study on knowledge, attitude, and practices among veterinarians of Haryana state in India. Vet. World 2019, 12, 258–265. [Google Scholar] [CrossRef] [Green Version]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277. [Google Scholar]
- Cambaco, O.; Alonso Menendez, Y.; Kinsman, J.; Sigaúque, B.; Wertheim, H.; Do, N.; Gyapong, M.; John-Langba, J.; Sevene, E.; Munguambe, K. Community knowledge and practices regarding antibiotic use in rural Mozambique: Where is the starting point for prevention of antibiotic resistance? BMC Public Health 2020, 20, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ngoh, L.N.; Shepherd, M.D. Design, development, and evaluation of visual aids for communicating prescription drug instructions to nonliterate patients in rural Cameroon. Patient Educ. Couns. 1997, 31, 245–261. [Google Scholar] [CrossRef]
- Alonso, S.; Dominguez-Salas, P.; Grace, D. The role of livestock products for nutrition in the first 1000 days of life. Anim. Front. 2019, 9, 24–31. [Google Scholar] [CrossRef] [Green Version]
- Lindahl, J.F.; Deka, R.P.; Melin, D.; Berg, A.; Lundén, H.; Lapar, M.L.; Asse, R.; Grace, D. An inclusive and participatory approach to changing policies and practices for improved milk safety in Assam, northeast India. Glob. Food Sec. 2018, 17, 9–13. [Google Scholar] [CrossRef]
- Setlow, P. Spore germination. Curr. Opin. Microbiol. 2003, 6, 550–556. [Google Scholar] [CrossRef]
- Kumar, N.; Raghu, H.V.; Kumar, A.; Haldar, L.; Khan, A.; Rane, S.; Malik, R.K. Spore germination based assay for monitoring antibiotic residues in milk at dairy farm. World J. Microbiol. Biotechnol. 2012, 28, 2559–2566. [Google Scholar] [CrossRef] [PubMed]
- Rajesh, G.; Arora, S. Bacterial spore based biosensor for detection of contaminants in milk. J. Food Process. Technol. 2013, 4, 277. [Google Scholar]
- Salter, R.S.; Markovsky, R.J.; Douglas, D.W.; Saul, S.J.; Tran, A.C.; Legg, D.R.; Schwartz, J.A.; Conaway, D.M.; McRobbie, L.W.; Kalinowski, E.; et al. TRIO method for detection of beta-lactams, sulfonamides, and tetracyclines in raw commingled cows’ milk: AOAC performance tested MethodSM 121601. J. AOAC Int. 2021, 103, 1366–1377. [Google Scholar] [CrossRef] [Green Version]
- The R Core Team. R: A Language and Environment for Statistical Computing; The R Core Team: Vienna, Austria, 2013. [Google Scholar]
| Total Farmers | Total % (CI $) | Small-Scale Farm% (CI) | Medium-Scale Farm% (CI) | Large-Scale Farm% (CI) | p-Value # | |
|---|---|---|---|---|---|---|
| Completed training in animal management | 29/491 | 6% (4–8%) | 4.5% (3–7%) * | 14% (7–27%) | 20% (8–42%) * | 0.002 |
| Completed training in animal disease | 17/491 | 4% (2–6%) | 3% (2–5%) | 2% (0.2–12%) | 10% (3–30%) | 0.195 |
| Farmers who recognise antibiotics on picture cards | 55/491 | 11% (9–14%) | 10% (7–13%) * | 19% (10–32%) | 30% (15–52%) * | 0.006 |
| Farmers who say they have heard of antibiotics | 193/491 | 39% (35–44%) | 39% (34–43%) | 47% (33–61%) | 40% (22–61%) | 0.591 |
| Farmers who say they have heard of a withdrawal period | 8/491 | 2% (0.8–3%) | 1.4% (0.6–3) | 2.3% (0.1–12) | 5% (0.3–24) | 0.180 |
| Farmers who say they used antibiotics in last year on their farm | 49/491 | 10% (8–13%) | 9% (6–12%) * | 16% (8–30%) | 25% (11–47%) * | 0.021 |
| Farms where antibiotic residues were found in milk | 40/491 | 8% (6–11%) | 7% (5–10) | 12% (5–25%) | 20% (8–42%) | 0.076 |
| Farms where antibiotics were found on inspection | 14/491 | 3% (2–5%) | 2% (1–4%) | 5% (1–16%) | 10% (3–30%) | 0.068 |
| Farms who report veterinary visits yearly or less | 292/491 | 61% (55–64%) | 87% (83–90%) | 9% (6–13%) | 4% (2–6%) | 0.827 |
| Farms who report never having a veterinary visit | 58/491 | 12% (9–15%) | 97% (88–99%) | 3% (1–12%) | 0% | 0.07 |
| Antibiotic/Antibiotic Group | MRL in Milk (European Commission) | Number of Samples Exceeding MRL |
|---|---|---|
| Beta-lactam antibiotics (penicillin) | 4 µg/kg | 3 (0.6%) |
| Tetracycline/chlortetracycline/oxytetracycline | 100 µg/kg | 4 (0.8%) |
| Sulphonamides | 100 µg/kg | 5 (1.0%) |
| Streptomycin/Dihydrostreptomycin | 200 µg/kg | 0 |
| Gentamicin | 100 µg/kg | 0 |
| Novobiocin | 50 µg/kg | 17 (3.5%) |
| Macrolides (Erythromycin) | 40 µg/kg | 12 (2.4%) |
| Quinolones (Enrofloxacin) | 100 µg/kg | 0 |
| Chloramphenicol | Prohibited | 0 |
| Coefficients | Standard Error | p-Value | 95% CI | |
|---|---|---|---|---|
| Farm Size | ||||
| Small farms | Reference category | |||
| Medium farms | 0.07 | 0.03 | 0.054 | 0.001–0.14 |
| Large farms | 0.08 | 0.05 | 0.050 | 0.01–0.18 |
| State | ||||
| Assam | Reference category | |||
| Haryana | 0.03 | 0.02 | 0.19 | 0.01–0.07 |
| Gender | ||||
| Male | Reference category | |||
| Female | 0.11 | 0.03 | <0.001 | 0.17–0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, N.; Sharma, G.; Leahy, E.; Shome, B.R.; Bandyopadhyay, S.; Deka, R.P.; Shome, R.; Dey, T.K.; Lindahl, J.F. Understanding Antibiotic Usage on Small-Scale Dairy Farms in the Indian States of Assam and Haryana Using a Mixed-Methods Approach—Outcomes and Challenges. Antibiotics 2021, 10, 1124. https://doi.org/10.3390/antibiotics10091124
Kumar N, Sharma G, Leahy E, Shome BR, Bandyopadhyay S, Deka RP, Shome R, Dey TK, Lindahl JF. Understanding Antibiotic Usage on Small-Scale Dairy Farms in the Indian States of Assam and Haryana Using a Mixed-Methods Approach—Outcomes and Challenges. Antibiotics. 2021; 10(9):1124. https://doi.org/10.3390/antibiotics10091124
Chicago/Turabian StyleKumar, Naresh, Garima Sharma, Eithne Leahy, Bibek R. Shome, Samiran Bandyopadhyay, Ram Pratim Deka, Rajeswari Shome, Tushar Kumar Dey, and Johanna Frida Lindahl. 2021. "Understanding Antibiotic Usage on Small-Scale Dairy Farms in the Indian States of Assam and Haryana Using a Mixed-Methods Approach—Outcomes and Challenges" Antibiotics 10, no. 9: 1124. https://doi.org/10.3390/antibiotics10091124
APA StyleKumar, N., Sharma, G., Leahy, E., Shome, B. R., Bandyopadhyay, S., Deka, R. P., Shome, R., Dey, T. K., & Lindahl, J. F. (2021). Understanding Antibiotic Usage on Small-Scale Dairy Farms in the Indian States of Assam and Haryana Using a Mixed-Methods Approach—Outcomes and Challenges. Antibiotics, 10(9), 1124. https://doi.org/10.3390/antibiotics10091124







