Characterization and Evaluation of Layered Bi2WO6 Nanosheets as a New Antibacterial Agent
Abstract
:1. Introduction
2. Results and Discussion
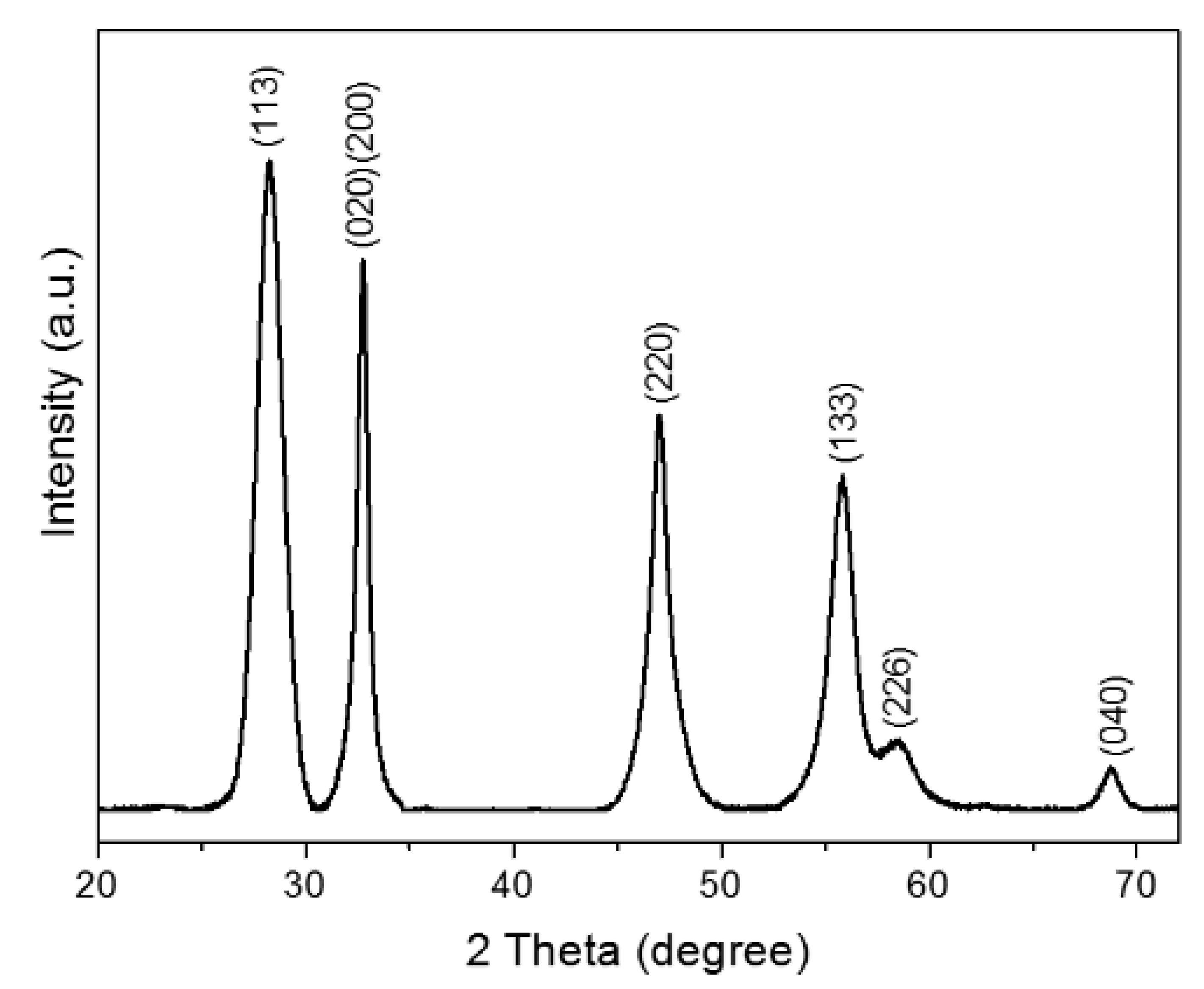
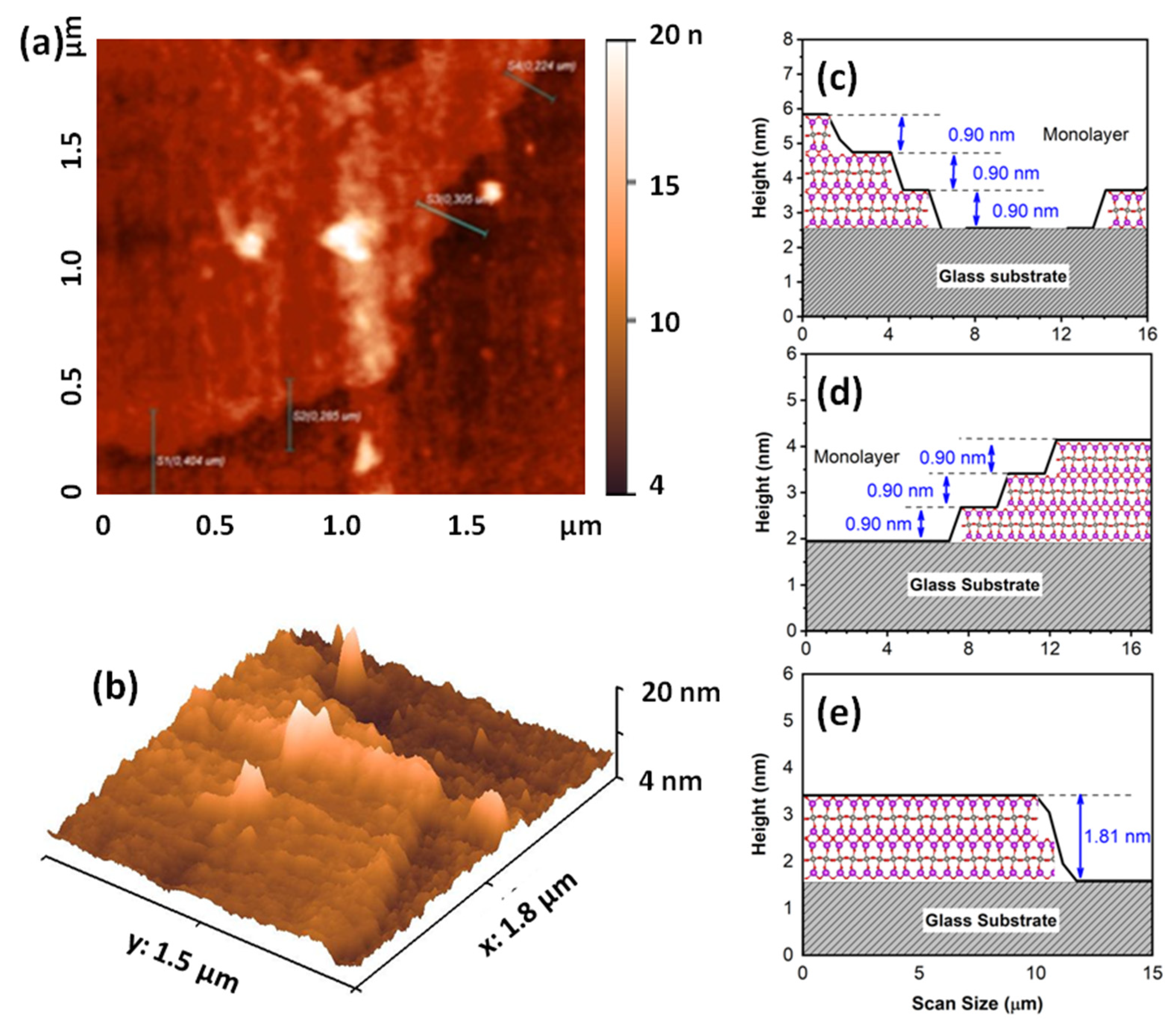
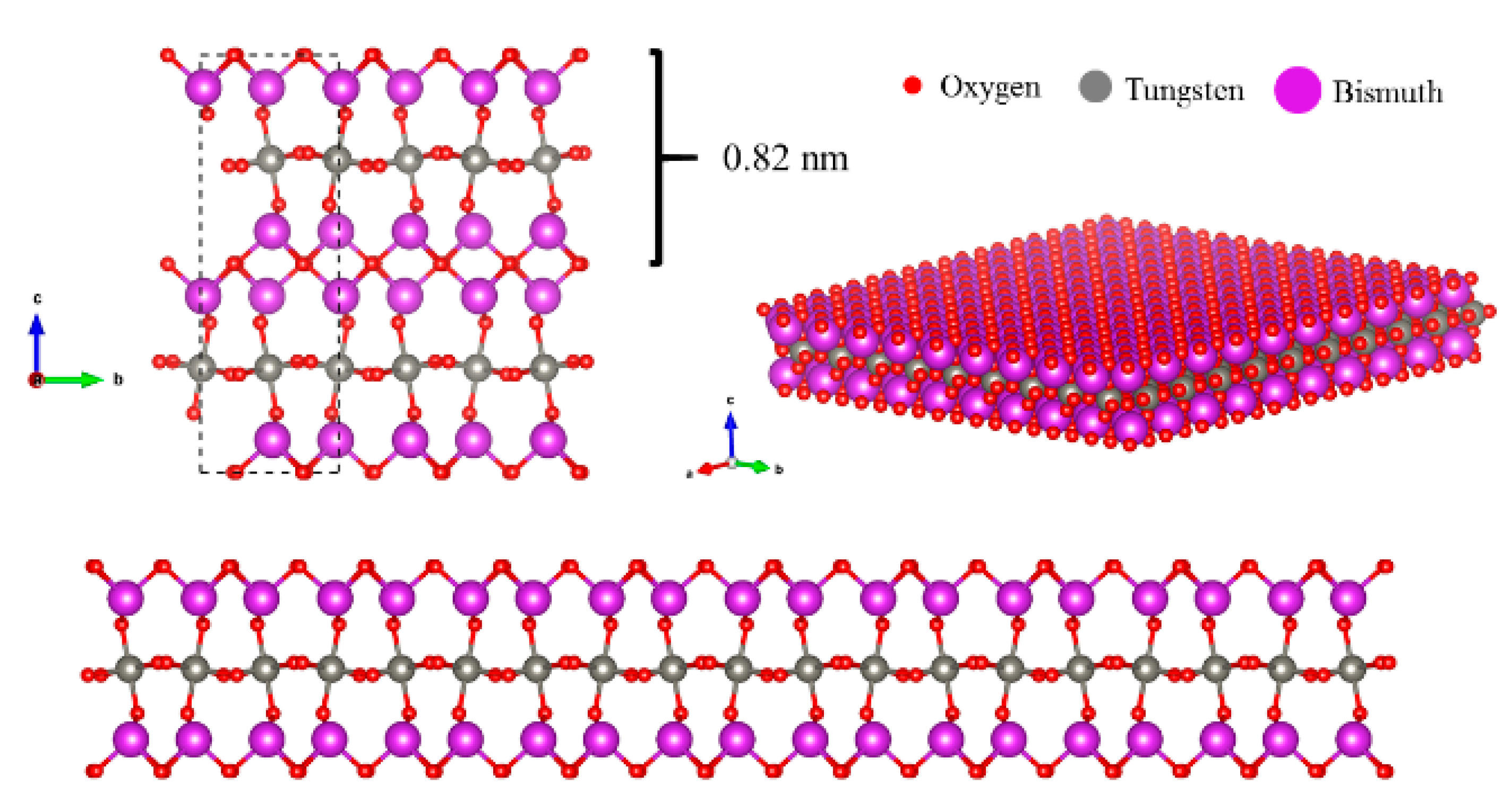
2.1. Characterization of Layered Bi2WO6 Nanosheets
2.2. Antibacterial Activity of Layered Bi2WO6 Nanosheets
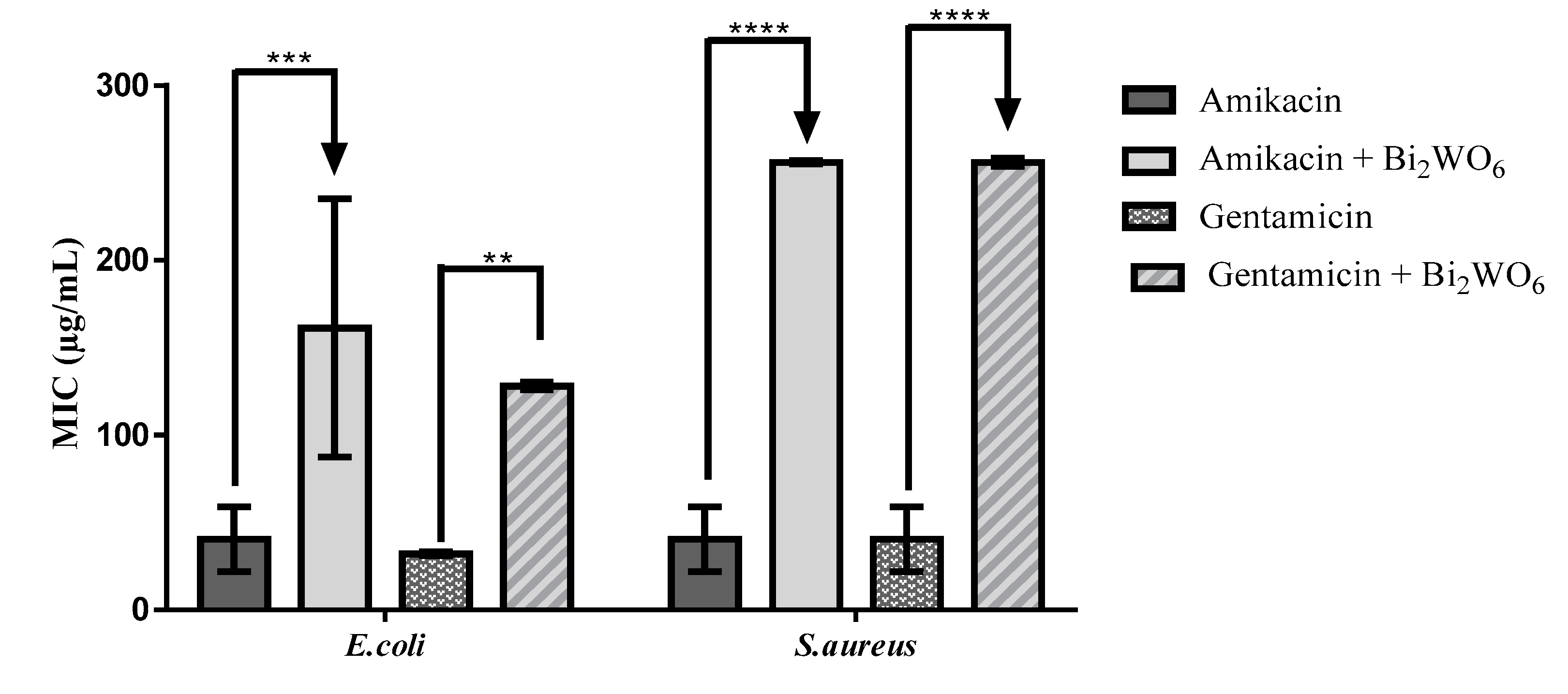
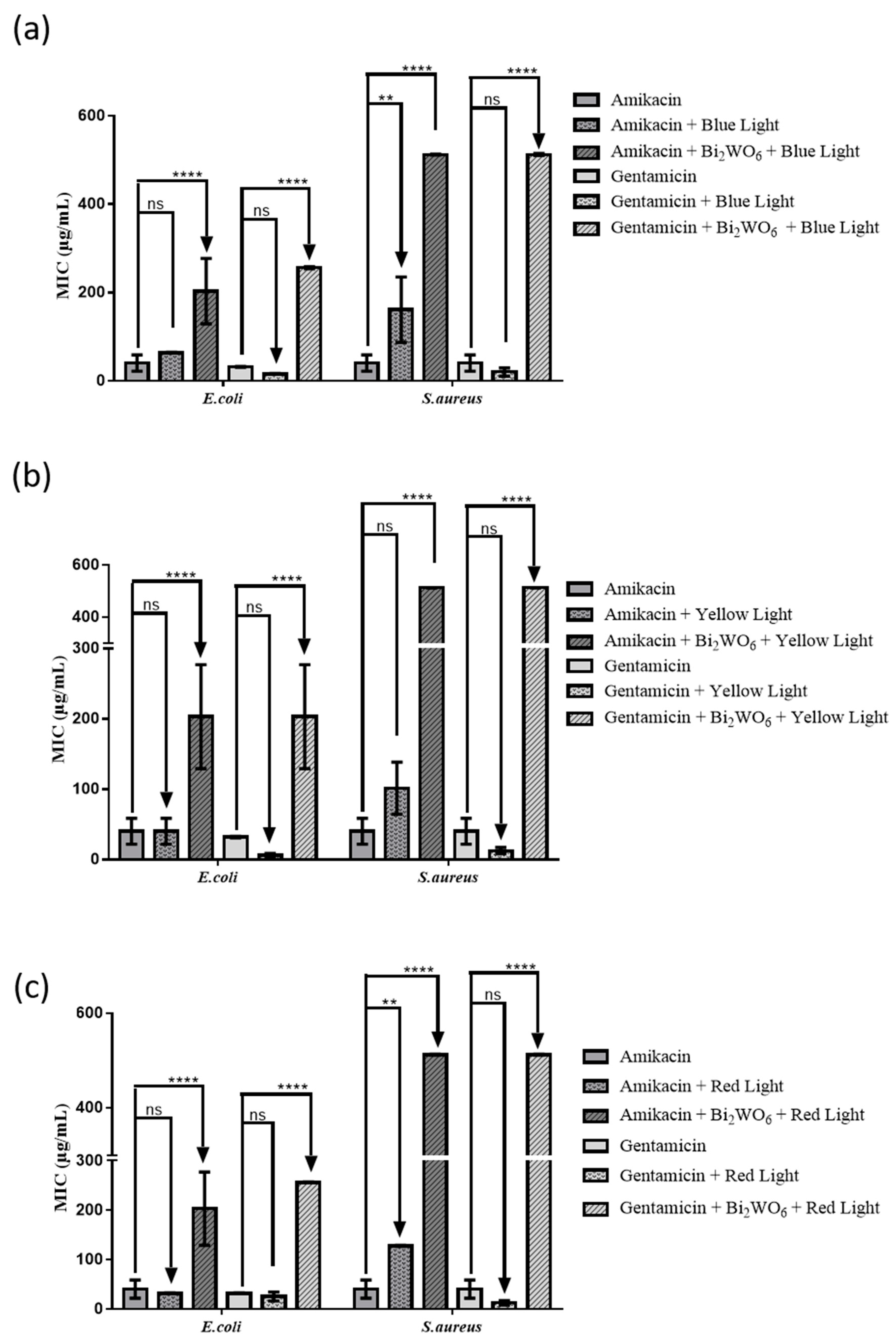
2.3. Modulation of Antibiotic Activity by Layered Bi2WO6 Nanosheets
3. Materials and Methods
3.1. Synthesis
3.2. Structural Characterization
3.3. Analysis of Antibacterial Activity and Antibiotic Resistance Modulation
3.4. Experiments with LED Light Exposure
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Oliveira, D.R.; Tintino, S.R.; Morais-Braga, M.F.B.; Boligon, A.A.; Athayde, M.L.; Coutinho, H.D.M.; De Menezes, I.R.A.; Fachinetto, R. In Vitro Antimicrobial and Modulatory Activity of the Natural Products Silymarin and Silibinin. BioMed Res. Int. 2015, 2015, 292797. [Google Scholar] [CrossRef] [Green Version]
- Lombardo, D.; Kiselev, M.A.; Caccamo, M.T. Smart Nanoparticles for Drug Delivery Application: Development of Versatile Nanocarrier Platforms in Biotechnology and Nanomedicine. J. Nanomater. 2019, 2019, 3702518. [Google Scholar] [CrossRef]
- Momeni, K.; Ji, Y.; Wang, Y.; Paul, S.; Neshani, S.; Yilmaz, D.E.; Shin, Y.K.; Zhang, D.; Jiang, J.-W.; Park, H.; et al. Multiscale computational understanding and growth of 2D materials: A review. npj Comput. Mater. 2020, 6, 22. [Google Scholar] [CrossRef] [Green Version]
- Pan, B.; Zhang, K.; Ding, C.-C.; Wu, Z.; Fan, Q.; Luo, T.; Zhang, L.; Zou, C.; Huang, S. Universal Precise Growth of 2D Transition-Metal Dichalcogenides in Vertical Direction. ACS Appl. Mater. Interfaces 2020, 12, 35337–35344. [Google Scholar] [CrossRef]
- Puthirath Balan, A.; Radhakrishnan, S.; Woellner, C.F.; Sinha, S.K.; Deng, L.; Reyes, C.D.L.; Rao, B.M.; Paulose, M.; Neupane, R.; Apte, A.; et al. Exfoliation of a non-van der Waals material from iron ore hematite. Nat. Nanotechnol. 2018, 13, 602–609. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, Y.; Lin, M.; Long, J.; Zhang, Z.; Lin, H.; Wu, J.C.-S.; Wang, X. Monolayered Bi2WO6 nanosheets mimicking heterojunction interface with open surfaces for photocatalysis. Nat. Commun. 2015, 6, 8340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, J.; Zou, Z.; Ye, J. Photocatalytic Decomposition of Organic Contaminants by Bi2WO6Under Visible Light Irradiation. Catal. Lett. 2004, 92, 53–56. [Google Scholar] [CrossRef]
- Lai, M.T.L.; Lai, C.W.; Lee, K.M.; Chook, S.W.; Yang, T.C.K.; Chong, S.; Juan, J.C. Facile one-pot solvothermal method to synthesize solar active Bi2WO6 for photocatalytic degradation of organic dye. J. Alloys Compd. 2019, 801, 502–510. [Google Scholar] [CrossRef]
- Huang, C.; Chen, L.; Li, H.; Mu, Y.; Yang, Z. Synthesis and application of Bi2WO6 for the photocatalytic degradation of two typical fluoroquinolones under visible light irradiation. RSC Adv. 2019, 9, 27768–27779. [Google Scholar] [CrossRef] [Green Version]
- Ren, J.; Wang, W.; Zhang, L.; Chang, J.; Hu, S. Photocatalytic inactivation of bacteria by photocatalyst Bi2WO6 under visible light. Catal. Commun. 2009, 10, 1940–1943. [Google Scholar] [CrossRef]
- Zhou, Y.; Lv, P.; Meng, X.; Tang, Y.; Huang, P.; Chen, X.; Shen, X.; Zeng, X. CTAB-Assisted Fabrication of Bi2WO6 Thin Nanoplates with High Adsorption and Enhanced Visible Light-Driven Photocatalytic Performance. Molecules 2017, 22, 859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma̧czka, M.; Macalik, L.; Hermanowicz, K.; Kȩpiński, L.; Tomaszewski, P. Phonon properties of nanosized bismuth layered ferroelectric material-Bi2 WO6. J. Raman Spectrosc. 2009, 41, 1059–1066. [Google Scholar] [CrossRef]
- Su, G.; Yang, C.; Zhu, J.-J. Fabrication of Gold Nanorods with Tunable Longitudinal Surface Plasmon Resonance Peaks by Reductive Dopamine. Langmuir 2015, 31, 817–823. [Google Scholar] [CrossRef] [PubMed]
- Gupta, H.; Archana, H.C.; Luthra, V. Lattice dynamical investigations for Raman and infrared frequencies of Bi2WO6. J. Mol. Struct. 2011, 1005, 53–58. [Google Scholar] [CrossRef]
- Ma̧czka, M.; Macalik, L.; Kojima, S. Temperature-dependent Raman scattering study of cation-deficient Aurivillius phases: Bi2WO6and Bi2W2O9. J. Phys. Condens. Matter 2011, 23, 405902. [Google Scholar] [CrossRef]
- Li, Z.; Meng, X.; Zhang, Z. Hexagonal SnS nanoplates assembled onto hierarchical Bi2WO6 with enhanced photocatalytic activity in detoxification and disinfection. J. Colloid Interface Sci. 2019, 537, 345–357. [Google Scholar] [CrossRef]
- Houghton, P.; Howes, M.-J.; Lee, C.; Steventon, G. Uses and abuses of in vitro tests in ethnopharmacology: Visualizing an elephant. J. Ethnopharmacol. 2007, 110, 391–400. [Google Scholar] [CrossRef]
- Paitan, Y. Current Trends in Antimicrobial Resistance of Escherichia coli. Curr. Top. Microbiol. Immunol. 2018, 416, 181–211. [Google Scholar] [CrossRef]
- Alós, J.-I. Resistencia bacteriana a los antibióticos: Una crisis global. Enferm. Infecc. Microbiol. Clin. 2015, 33, 692–699. [Google Scholar] [CrossRef]
- Mardare, C.C.; Tanasic, D.; Rathner, A.; Muller, N.; Hassel, A.W. Growth inhibition of Escherichia coli by zinc molybdate with different crystalline structures. Phys. Status Solidi A 2016, 213, 1471–1478. [Google Scholar] [CrossRef]
- Cai, Q.; Hu, J. Decomposition of sulfamethoxazole and trimethoprim by continuous UVA/LED/TiO2 photocatalysis: Decomposition pathways, residual antibacterial activity and toxicity. J. Hazard. Mater. 2017, 323, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, H.D.; Costa, J.G.; Lima, E.O.; Falcão-Silva, V.S.; Siqueira-Júnior, J.P. Enhancement of the Antibiotic Activity against a Multiresistant Escherichia coli by Mentha arvensis L. and Chlorpromazine. Chemotherapy 2008, 54, 328–330. [Google Scholar] [CrossRef]

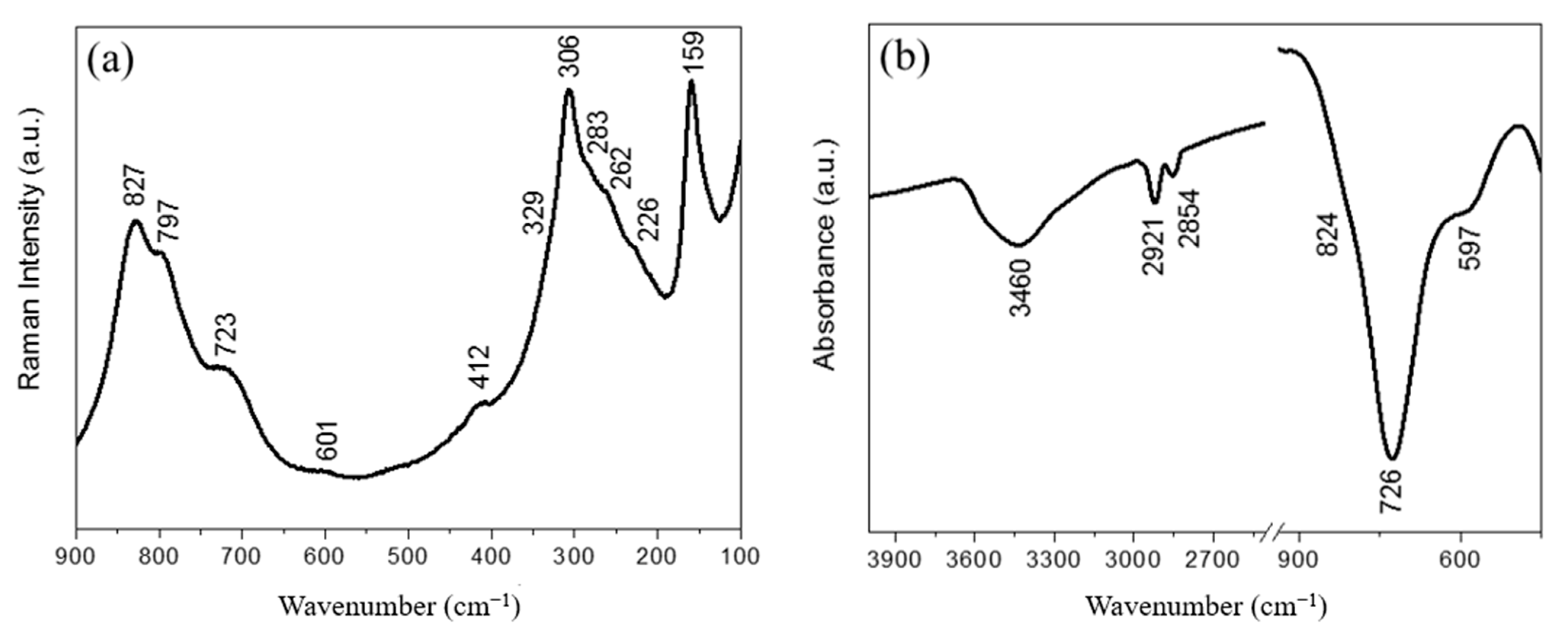
| Observed Modes (cm−1) | Assignment | |
|---|---|---|
| Raman | Infrared | |
| 827 | 824 | Asymmetric stretching of WO6 |
| 797 | - | Symmetric stretching of WO6 |
| 723 | 726 | Asymmetric stretching of WO6 |
| 601 | 597 | Bending of WO6 |
| 412 | - | Bending of WO6 |
| 329 | - | Bending of Bi-O bonds |
| 306 | - | Bending of WO6 |
| 283 | - | Bending of WO6 |
| 262 | - | Bending of WO6 |
| 226 | - | Bending of WO6 |
| 159 | - | Translational mode (Bi) |
| Treatment | E.C. ATCC 25922 | S.A. ATCC 25923 | E.C. 06 | S.A. 10 |
|---|---|---|---|---|
| Bi2WO6 monolayers | 256 | 256 | 256 | 32 |
| Bi2WO6 + Blue Light | 341.3 | 256 | 256 | 32 |
| Bi2WO6 + Yellow Light | 256 | 256 | 512 | 64 |
| Bi2WO6 + Red Light | 256 | 256 | 256 | 32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leandro, M.K.d.N.S.; Moura, J.V.B.; Freire, P.d.T.C.; Vega, M.L.; Lima, C.d.L.; Hidalgo, Á.A.; Araújo, A.C.J.d.; Freitas, P.R.; Paulo, C.L.R.; Sousa, A.K.d.; et al. Characterization and Evaluation of Layered Bi2WO6 Nanosheets as a New Antibacterial Agent. Antibiotics 2021, 10, 1068. https://doi.org/10.3390/antibiotics10091068
Leandro MKdNS, Moura JVB, Freire PdTC, Vega ML, Lima CdL, Hidalgo ÁA, Araújo ACJd, Freitas PR, Paulo CLR, Sousa AKd, et al. Characterization and Evaluation of Layered Bi2WO6 Nanosheets as a New Antibacterial Agent. Antibiotics. 2021; 10(9):1068. https://doi.org/10.3390/antibiotics10091068
Chicago/Turabian StyleLeandro, Maria Karollyna do Nascimento Silva, João Victor Barbosa Moura, Paulo de Tarso Cavalcante Freire, Maria Leticia Vega, Cleânio da Luz Lima, Ángel Alberto Hidalgo, Ana Carolina Justino de Araújo, Priscilla Ramos Freitas, Cicera Laura Roque Paulo, Amanda Karine de Sousa, and et al. 2021. "Characterization and Evaluation of Layered Bi2WO6 Nanosheets as a New Antibacterial Agent" Antibiotics 10, no. 9: 1068. https://doi.org/10.3390/antibiotics10091068
APA StyleLeandro, M. K. d. N. S., Moura, J. V. B., Freire, P. d. T. C., Vega, M. L., Lima, C. d. L., Hidalgo, Á. A., Araújo, A. C. J. d., Freitas, P. R., Paulo, C. L. R., Sousa, A. K. d., Rocha, J. E., Leandro, L. M. G., Silva, R. O. M. d., Cruz-Martins, N., & Coutinho, H. D. M. (2021). Characterization and Evaluation of Layered Bi2WO6 Nanosheets as a New Antibacterial Agent. Antibiotics, 10(9), 1068. https://doi.org/10.3390/antibiotics10091068







