Co-Existence of Certain ESBLs, MBLs and Plasmid Mediated Quinolone Resistance Genes among MDR E. coli Isolated from Different Clinical Specimens in Egypt
Abstract
1. Introduction
2. Results
2.1. Prevalence of E. coli Among Samples
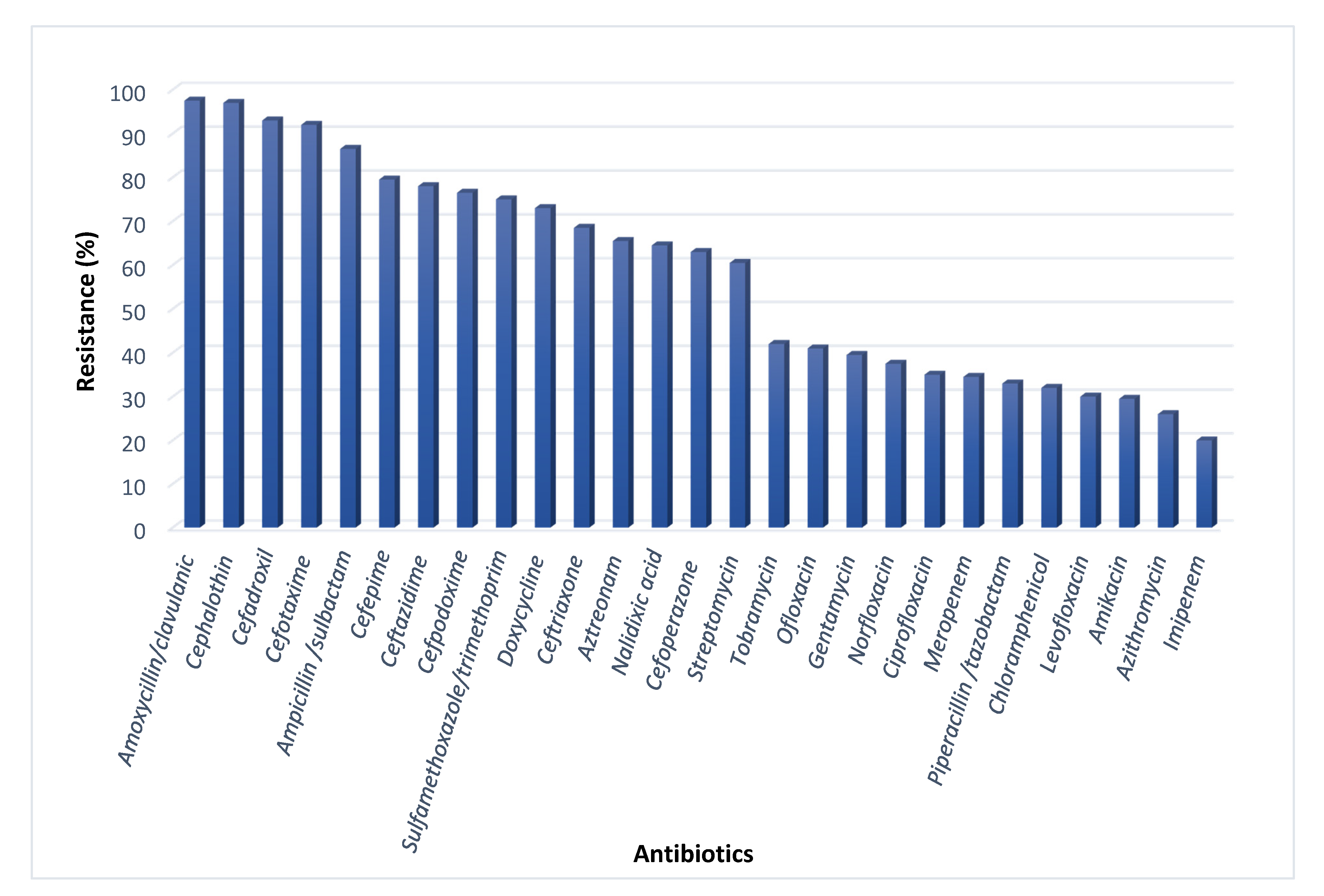
2.2. Antibiotic Resistance of the E. coli Pathogens
2.3. Serotyping of the Intestinal E. coli
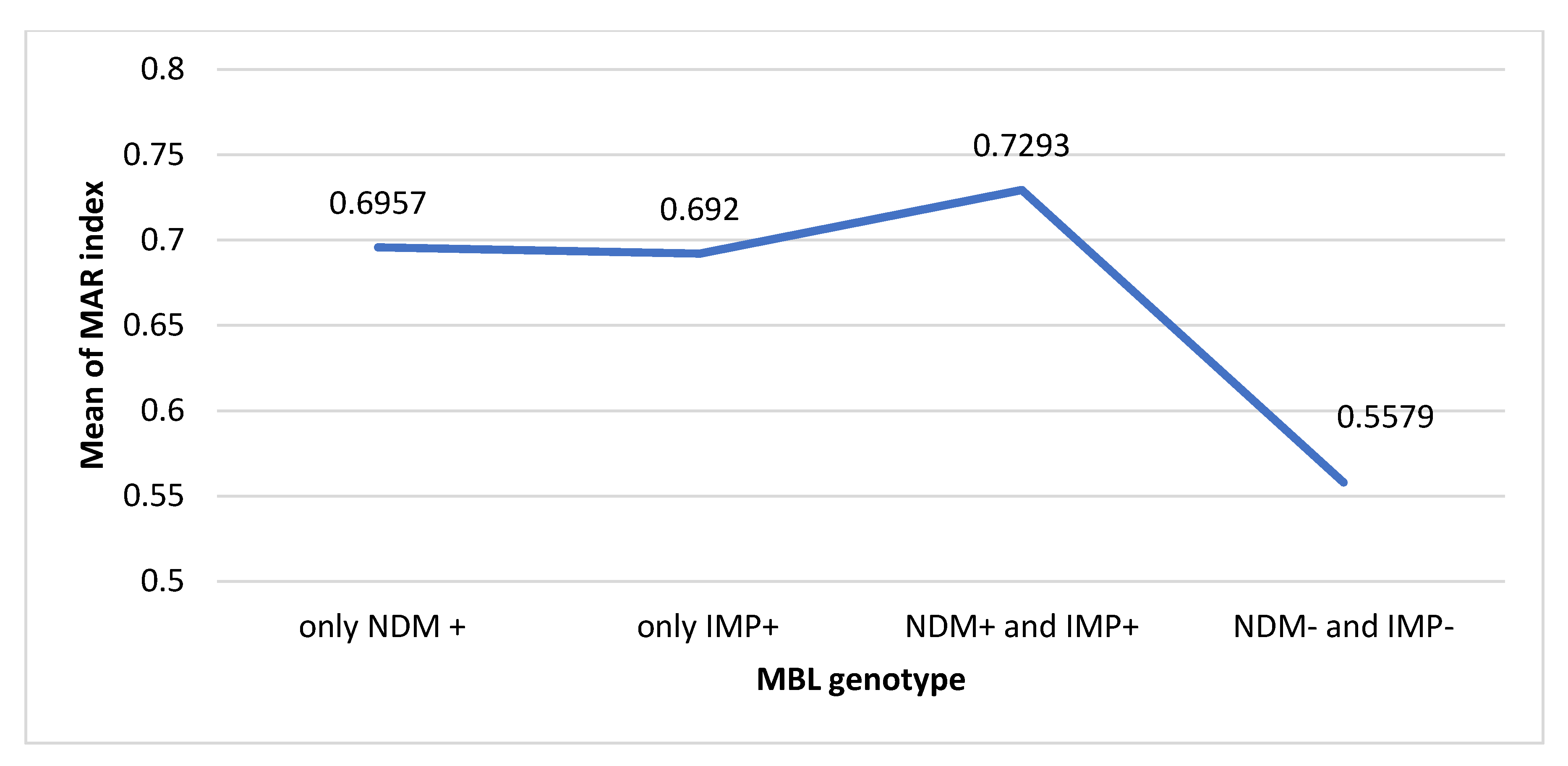
2.4. Multiple Antibiotic Resistance Index MDR E. coli Pathogens
2.5. Phenotypic Characteristics
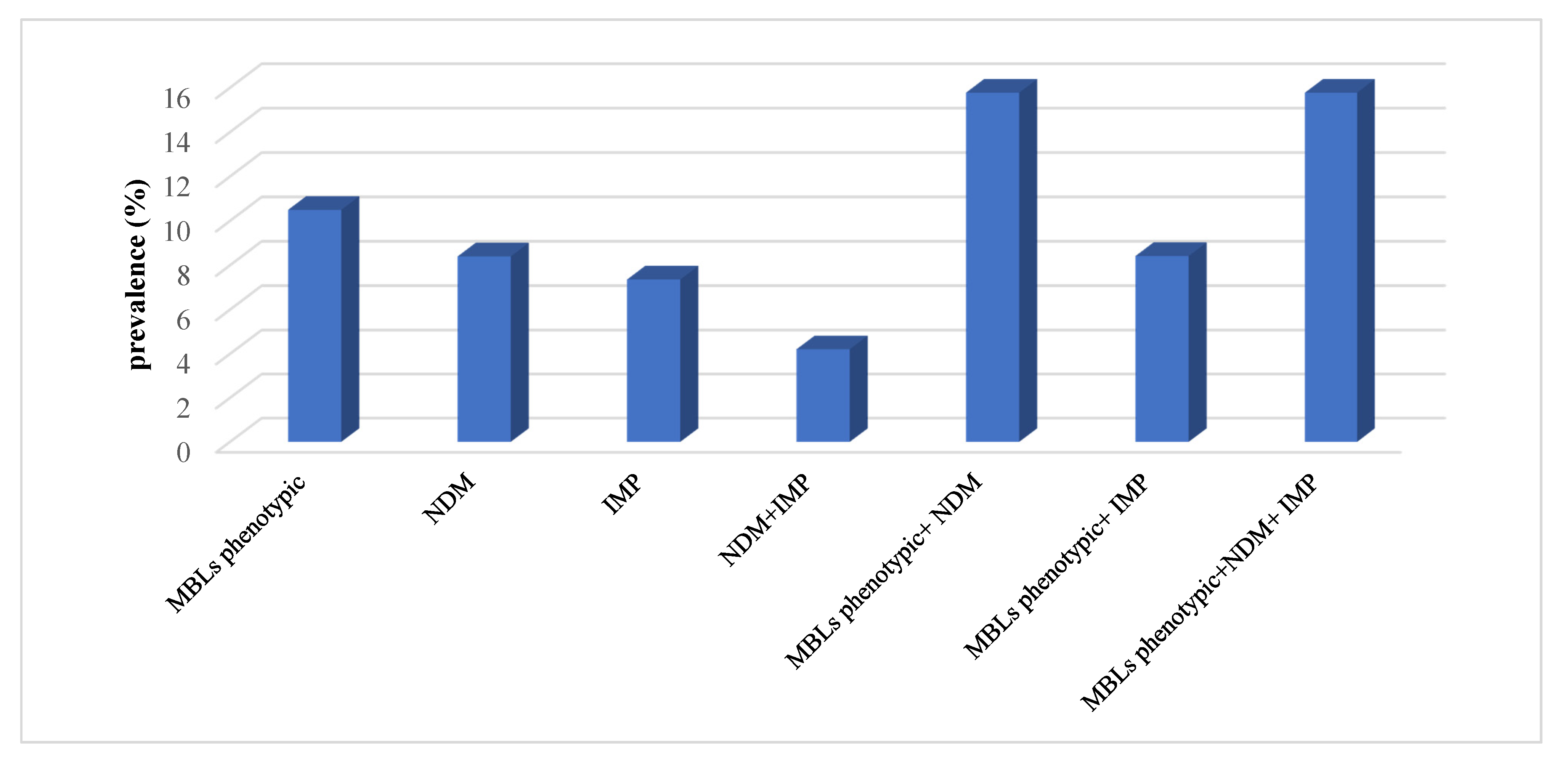
2.6. Antimicrobial Resistance of ESBLs and MBLs Producers
2.7. Prevalence of Resistance Genotypes Among the Tested Isolates
2.8. Genotypic-Phenotypic Agreement of the Tested Genes
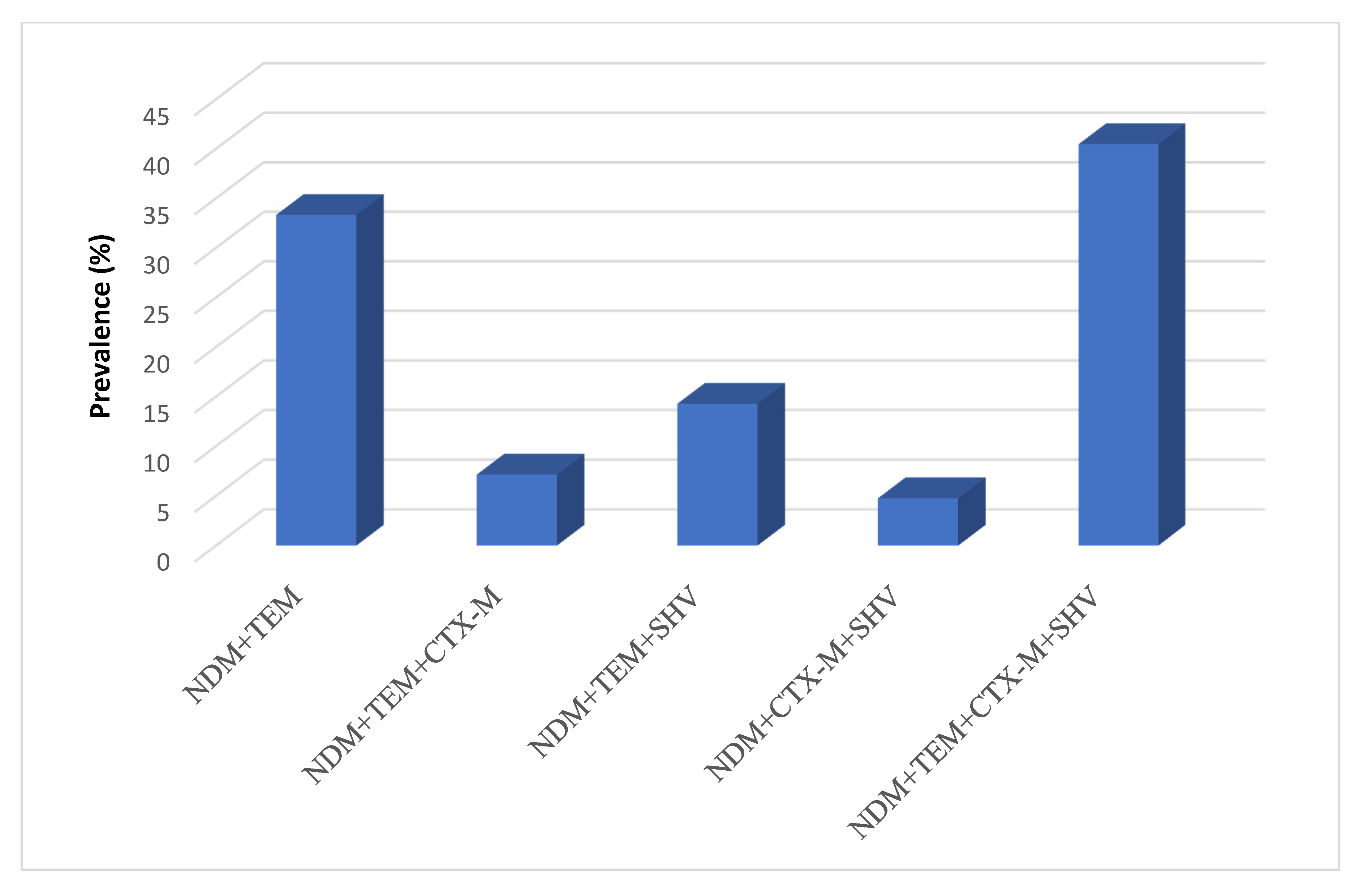
2.9. Association of Different Resistance Genotypes
3. Discussion
4. Material and Methods
4.1. Bacterial Isolates
4.2. Antimicrobial-Susceptibility Testing
4.3. Serotyping of Intestinal E. coli
4.4. Phenotypic Detection of ESBLs and MBLs Production
4.5. Amplification of Resistance Genes
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abdelwahab, R.; Yasir, M.; Godfrey, R.E.; Christie, G.S.; Element, S.J.; Saville, F.; Hassan, E.A.; Ahmed, E.H.; Abu-Faddan, N.H.; Daef, E.A.J.V. Antimicrobial Resistance and Gene Regulation in Enteroaggregative Escherichia Coli from Egyptian Children with Diarrhoea: Similarities and Differences. Virulence 2021, 12, 57–74. [Google Scholar] [CrossRef] [PubMed]
- Banin, E.; Hughes, D.; Kuipers, O.P. Bacterial Pathogens, Antibiotics and Antibiotic Resistance. FEMS Microbiol. Rev. 2017, 41, 450–452. [Google Scholar] [CrossRef]
- Sultan, I.; Rahman, S.; Jan, A.T.; Siddiqui, M.T.; Mondal, A.H.; Haq, Q.M.R. Antibiotics, Resistome and Resistance Mechanisms: A Bacterial Perspective. Front. Microbiol. 2018, 9, 2066. [Google Scholar] [CrossRef]
- Christaki, E.; Marcou, M.; Tofarides, A. Antimicrobial Resistance in Bacteria: Mechanisms, Evolution, and Persistence. J. Mol. Evol. 2020, 88, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Matlashewski, G.; Berghuis, A.; Sheppard, D.; Wainberg, M.A.; Gotte, M. Handbook of Antimicrobial Resistance; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Tooke, C.L.; Hinchliffe, P.; Bragginton, E.C.; Colenso, C.K.; Hirvonen, V.H.; Takebayashi, Y.; Spencer, J. β-Lactamases and β-Lactamase Inhibitors in the 21st Century. J. Mol. Biol. 2019, 431, 3472–3500. [Google Scholar] [CrossRef]
- Reck, F.; Bermingham, A.; Blais, J.; Capka, V.; Cariaga, T.; Casarez, A.; Colvin, R.; Dean, C.R.; Fekete, A.; Gong, W.; et al. Optimization of Novel Monobactams with Activity against Carbapenem-Resistant Enterobacteriaceae—Identification of LYS228. Bioorg. Med. Chem. Lett. 2018, 28, 748–755. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. Interplay between β-Lactamases and New β-lactamase Inhibitors. Nat. Rev. Genet. 2019, 17, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Castillo, F.Y.; Moreno-Flores, A.C.; Avelar-González, F.J.; Márquez-Díaz, F.; Harel, J.; Guerrero-Barrera, A.L. An Evaluation of Multidrug-Resistant Escherichia Coli Isolates in Urinary Tract Infections from Aguascalientes, Mexico: Cross-Sectional Study. Ann. Clin. Microbiol. Antimicrob. 2018, 17, 34. [Google Scholar] [CrossRef]
- Park, C.H.; Robicsek, A.; Jacoby, G.A.; Sahm, D.; Hooper, D.C. Prevalence in the United States of aac(6′)-Ib-cr Encoding a Ciprofloxacin-Modifying Enzyme. Antimicrob. Agents Chemother. 2006, 50, 3953–3955. [Google Scholar] [CrossRef]
- Peralta, G.; Sanchez, M.B.; Garrido, J.C.; De Benito, I.; Cano, M.E.; Martínez-Martínez, L.; Roiz, M.P. Impact of Antibiotic Resistance and of Adequate Empirical Antibiotic Treatment in the Prognosis of Patients with Escherichia coli bacteraemia. J. Antimicrob. Chemother. 2007, 60, 855–863. [Google Scholar] [CrossRef]
- Amer, R.; El-Baghdady, K.; Kamel, I.; El-Shishtawy, H. Prevalence of Extended Spectrum Beta- Lactamase Genes among Escherichia Coli and Klebsiella Pneumoniae Clinical Isolates. Egypt. J. Microbiol. 2019. [Google Scholar] [CrossRef]
- Fam, N.; Leflon-Guibout, V.; Fouad, S.; Aboul-Fadl, L.; Marcon, E.; Desouky, D.; El-Defrawy, I.; Abou-Aitta, A.; Klena, J.; Nicolas-Chanoine, M.-H. CTX-M-15-Producing Escherichia Coli Clinical Isolates in Cairo (Egypt), Including Isolates of Clonal Complex ST10 and Clones ST131, ST73, and ST405 in Both Community and Hospital Settings. Microb. Drug Resist. 2011, 17, 67–73. [Google Scholar] [CrossRef]
- Alanazi, M.Q.; Alqahtani, F.Y.; Aleanizy, F.S. An Evaluation of E. Coli in Urinary Tract Infection in Emergency Department at KAMC in Riyadh, Saudi Arabia: Retrospective Study. Ann. Clin. Microbiol. Antimicrob. 2018, 17, 3. [Google Scholar] [CrossRef]
- Koupetori, M.; Retsas, T.; Antonakos, N.; Vlachogiannis, G.; Perdios, I.; Nathanail, C.; Makaritsis, K.; Papadopoulos, A.; Sinapidis, D.; Giamarellos-Bourboulis, E.J.; et al. Bloodstream Infections and Sepsis in Greece: Over-Time Change of Epidemiology and Impact of de-escalation on final outcome. BMC Infect. Dis. 2014, 14, 272. [Google Scholar] [CrossRef]
- Mohammed, M.A.; Alnour, T.M.; Shakurfo, O.M.; Aburass, M.M. Prevalence and antimicrobial resistance pat-tern of bacterial strains isolated from patients with urinary tract infection in Messalata Central Hospital, Libya. Asian Pac. J. Trop. Med. 2016, 9, 771–776. [Google Scholar] [CrossRef]
- Musicha, P.; Cornick, J.E.; Bar-Zeev, N.; French, N.; Masesa, C.; Denis, B.; Kennedy, N.; Mallewa, J.; Gordon, M.A.; Msefula, C.L.; et al. Trends in Antimicrobial Resistance in Bloodstream Infection Isolates at a Large Urban Hospital in Malawi (1998–2016): A Surveillance Study. Lancet Infect. Dis. 2017, 17, 1042–1052. [Google Scholar] [CrossRef]
- Miller, D. Update on the Epidemiology and Antibiotic Resistance of Ocular Infections. Middle E. Afr. J. Ophthalmol. 2017, 24, 30. [Google Scholar]
- Zhou, Y.; Zhu, X.; Hou, H.; Lü, Y.; Yu, J.; Mao, L.; Mao, L.; Sun, Z. Characteristics of Diarrheagenic Escherichia Coli among Children under 5 Years of Age with Acute Diarrhea: A Hospital Based Study. BMC Infect. Dis. 2018, 18, 63. [Google Scholar] [CrossRef] [PubMed]
- El Gany, S.A.; Gad, G.M.; Mousa, S.; Ibrahem, R. Characterization of Verotoxigenic E.Coli and Enteropathogenic E.Coli Isolated from Infants with Diarrhea in Combination with Antimicrobial Resistance Pattern in Minia, Egypt. J. Adv. Biomed. Pharm. Sci. 2020, 3, 101–109. [Google Scholar] [CrossRef]
- Jit, M.; Ng, D.H.L.; Luangasanatip, N.; Sandmann, F.; Atkins, K.E.; Robotham, J.V.; Pouwels, K.B. Quantifying the Economic cost of Antibiotic Resistance and the Impact of Related Interventions: Rapid Methodological Review, Conceptual Framework and Recommendations for Future Studies. BMC Med. 2020, 18, 38. [Google Scholar] [CrossRef]
- Siwakoti, S.; Subedi, A.; Sharma, A.; Baral, R.; Bhattarai, N.R.; Khanal, B. Incidence and Outcomes of Multidrug-Resistant Gram-negative Bacteria Infections in Intensive Care Unit from Nepal- a Prospective Cohort Study. Antimicrob. Resist. Infect. Control. 2018, 7, 114. [Google Scholar] [CrossRef] [PubMed]
- Abdelaziz, S.M.; Aboshanab, K.M.; Yahia, I.S.; Yassien, M.A.; Hassouna, N.A.J.A. Correlation between the Antibiotic Resistance Genes and Susceptibility to Antibiotics among the Carbapenem-Resistant Gram-Negative Pathogens. Antibiotics 2021, 10, 255. [Google Scholar] [CrossRef]
- Abd El-Baky, R.M.; Ibrahim, R.A.; Mohamed, D.S.; Ahmed, E.F.; Hashem, Z.S.J.I.; Resistance, D. Prevalence of Virulence Genes and Their Association with Antimicrobial Resistance Among Pathogenic E. Coli Isolated from Egyptian Patients with Different Clinical Infections. Infect. Drug Resist. 2020, 13, 1221. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Y.; Sani, Y.; Saleh, Q.; Saleh, A.; Hakeem, G. Phenotypic Detection of Extended Spectrum Beta Lactamase and Carbapenemase Co-Producing Clinical Isolates from Two Tertiary Hospitals in Kano, North West Nigeria. Ethiop. J. Health Sci. 2017, 27, 3–10. [Google Scholar] [CrossRef]
- Farhan, S.M.; A Ibrahim, R.; Mahran, K.M.; Hetta, H.F.; El-Baky, R.M.A. Antimicrobial Resistance Pattern and Molecular Genetic Distribution of Metallo-β-Lactamases Producing Pseudomonas aeruginosa Isolated from Hospitals in Minia, Egypt. Infect. Drug Resist. 2019, 12, 2125–2133. [Google Scholar] [CrossRef]
- Fazlul, M.; Deepthi, S.; Farzana, Y.; Najnin, A.; Rashid, M.; Munira, B.; Srikumar, S. Detection of Metal-lo-Β-Lactamases-Encoding Genes among Clinical Isolates of Escherichia Coli in a Tertiary Care Hospital, Malaysia. BMC Res. Notes 2019, 11, 291–298. [Google Scholar]
- Mohamed, E.S.; Khairy, R.M.M.; Abdelrahim, S.S. Prevalence and Molecular Characteristics of ESBL and AmpC β -Lactamase Producing Enterobacteriaceae Strains Isolated from UTIs in Egypt. Antimicrob. Resist. Infect. Control. 2020, 9, 198. [Google Scholar] [CrossRef]
- El Sherif, R.H.; Maamoun, H.A.H. Molecular Characteristics of Extended-Spectrum Beta-Lactamases Among Gram-Negative Isolates Collected in Cairo University Hospital. Comp. Haematol. Int. 2012, 22, 733–739. [Google Scholar] [CrossRef]
- Zaki, M.; El-Halaby, H.; Elmansoury, E.; Zeid, M.; Khaled, K.; Nomir, M. Genetic Study of Extended Spectrum Beta-Lactamase and Carbapenemase Producing Escherichia Coli Causing Sepsis among Egyptian Children. Open Microbiol. J. 2019, 13, 128–137. [Google Scholar] [CrossRef]
- Myat, T.O.; Hannaway, R.F.; Zin, K.N.; Htike, W.W.; Win, K.K.; Crump, J.A.; Murdoch, D.R.; Ussher, J.E. ESBL-and Carbapenemase-Producing Enterobacteriaceae in Patients with Bacteremia, Yangon, Myanmar. Emerg. Infect. Dis. 2017, 23, 857. [Google Scholar] [CrossRef] [PubMed]
- Abrar, S.; Ain, N.U.; Liaqat, H.; Hussain, S.; Rasheed, F.; Riaz, S. Distribution of blaCTX—M, blaTEM, blaSHV and blaOXA Genes in Extended-Spectrum-β-Lactamase-Producing Clinical Isolates: A Three-Year Multi-Center Study from Lahore, Pakistan. Antimicrob. Resist. Infect. Control. 2019, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Eltai, N.O.; Al Thani, A.A.; Al-Ansari, K.; Deshmukh, A.S.; Wehedy, E.; Al-Hadidi, S.H.; Yassine, H.M. Molecular Characterization of Extended Spectrum β -Lactamases Enterobacteriaceae Causing Lower Urinary Tract Infection among Pediatric Population. Antimicrob. Resist. Infect. Control. 2018, 7, 90. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.S.; Bansal, D.; Acharya, A.; Elmi, A.A.; Hamid, J.M.; Ahmed, A.M.S.; Chandra, P.; Ibrahim, E.; Sultan, A.A.; Doiphode, S.; et al. Antimicrobial Susceptibility and Molecular Epidemiology of Extended-Spectrum Beta-Lactamase-Producing Enterobacteriaceae from Intensive Care Units at Hamad Medical Corporation, Qatar. Clin. Microbiol. Rev. 2016, 5. [Google Scholar] [CrossRef]
- Kumarasamy, K.K.; Toleman, M.A.; Walsh, T.R.; Bagaria, J.; Butt, F.; Balakrishnan, R.; Chaudhary, U.; Doumith, M.; Giske, C.G.; Irfan, S.; et al. Emergence of A New Antibiotic Resistance Mechanism in India, Pakistan, and the UK: A Molecular, Biological, and Epidemiological Study. Lancet Infect. Dis. 2010, 10, 597–602. [Google Scholar] [CrossRef]
- Amer, W.H.; Khalil, H.S.; Abd EL Wahab, M.A. Risk Factors, Phenotypic and Genotypic Characterization of Carbapenem Resistant Enterobacteriaceae in Tanta University Hospitals, Egypt. Int. J. Infect. Control. 2016, 12. [Google Scholar] [CrossRef]
- Khalifa, H.; Soliman, A.M.; Ahmed, A.M.; Shimamoto, T.; Hara, T.; Ikeda, M.; Kuroo, Y.; Kayama, S.; Sugai, M.; Shimamoto, T. High Carbapenem Resistance in Clinical Gram-Negative Pathogens Isolated in Egypt. Microb. Drug Resist. 2017, 23, 838–844. [Google Scholar] [CrossRef]
- Begum, N.; Shamsuzzaman, S. Emergence of Carbapenemase-Producing Urinary Isolates at A Tertiary Care Hospital in Dhaka, Bangladesh. Tzu Chi Med. J. 2016, 28, 94–98. [Google Scholar] [CrossRef]
- Memish, Z.A.; Assiri, A.; Almasri, M.; Roshdy, H.; Hathout, H.; Kaase, M.; Gatermann, S.G.; Yezli, S. Molecular Characterization of Carbapenemase Production Among Gram-Negative Bacteria in Saudi Arabia. Microb. Drug Resist. 2015, 21, 307–314. [Google Scholar] [CrossRef]
- Sonnevend, A.; Ghazawi, A.A.; Hashmey, R.; Jamal, W.; Rotimi, V.O.; Shibl, A.M.; Al-Jardani, A.; Al-Abri, S.S.; Tariq, W.U.Z.; Weber, S.; et al. Characterization of Carbapenem-Resistant Enterobacteriaceae with High Rate of Autochthonous Transmission in the Arabian Peninsula. PLoS ONE 2015, 10, e0131372. [Google Scholar] [CrossRef]
- Sekyere, J.O.; Govinden, U.; Essack, S. The Molecular Epidemiology and Genetic Environment of Carbapenemases Detected in Africa. Microb. Drug Resist. 2016, 22, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Anderson, D.; Paterson, D. Overview of the Epidemiology and the Threat of Klebsiella Pneumoniae Carbapenemases (KPC) Resistance. Infect. Drug Resist. 2012, 5, 133–141. [Google Scholar] [CrossRef]
- Al-Agamy, M.H.; Aljallal, A.; Radwan, H.H.; Shibl, A.M. Characterization of Carbapenemases, ESBLs, and Plasmid-Mediated Quinolone Determinants in Carbapenem-Insensitive Escherichia coli and Klebsiella pneumoniae in Riyadh hospitals. J. Infect. Public Health 2018, 11, 64–68. [Google Scholar] [CrossRef]
- Hassuna, N.A.; Khairalla, A.S.; Farahat, E.M.; Hammad, A.M.; Abdel-Fattah, M. Molecular Characterization of Extended-Spectrum β Lactamase- Producing E. coli Recovered from Community-Acquired Urinary Tract Infections in Upper Egypt. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Ouedraogo, A.-S.; Sanou, M.; Kissou, A.; Sanou, S.; Solaré, H.; Kaboré, N.F.; Poda, A.; Aberkane, S.; Bouzinbi, N.; Sano, I.; et al. High Prevalence of Extended-Spectrum ß-Lactamase producing Enterobacteriaceae among Clinical Isolates in Burkina Faso. BMC Infect. Dis. 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Faghri, J.; Maleki, N.; Tahanasab, Z.; Mobasherizadeh, S.; Rezaei, A. Prevalence of CTX-M and TEM β-lactamases in Klebsiella pneumoniae Isolates from Patients with Urinary Tract Infection, Al-Zahra Hospital, Isfahan, Iran. Adv. Biomed. Res. 2018, 7, 10. [Google Scholar] [CrossRef]
- Kamel, N.A.; El-Tayeb, W.N.; El-Ansary, M.R.; Mansour, M.T.; Aboshanab, K.M. Phenotypic Screening and Molecular Characterization of Carbapenemase-Producing Gram-Negative Bacilli Recovered from Febrile Neutropenic Pediatric Cancer Patients in Egypt. PLoS ONE 2018, 13, e0202119. [Google Scholar] [CrossRef]
- Bado, I.; Gutiérrez, C.; Garcia-Fulgueiras, V.; Cordeiro, N.F.; Pirez, L.A.; Seija, V.; Bazet, C.; Rieppi, G.; Vignoli, R. CTX-M-15 in Combination with aac(6′)-Ib-cr is the Most Prevalent Mechanism of Resistance both in Escherichia coli and Klebsiella pneumoniae, including K. pneumoniae ST258, in an ICU in Uruguay. J. Glob. Antimicrob. Resist. 2016, 6, 5–9. [Google Scholar] [CrossRef]
- Vignoli, R.; García-Fulgueiras, V.; Cordeiro, N.F.; Bado, I.; Seija, V.; Aguerrebere, P.; Laguna, G.; Araújo, L.; Bazet, C.; Gutkind, G.; et al. Extended-Spectrum β-lactamases, Transferable Quinolone Resistance, and Virulotyping in Extra-Intestinal E. coli in Uruguay. J. Infect. Dev. Ctries. 2016, 10, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Azargun, R.; Sadeghi, M.R.; Barhaghi, M.H.S.; Kafil, H.S.; Yeganeh, F.; Oskouee, M.A.; Ghotaslou, R. The Prevalence of Plasmid-mediated Quinolone Resistance and ESBL-Production in Enterobacteriaceae Isolated from Urinary tract Infections. Infect. Drug Resist. 2018, ume 11, 1007–1014. [Google Scholar] [CrossRef]
- Xue, G.; Li, J.; Feng, Y.; Xu, W.; Li, S.; Yan, C.; Zhao, H.; Sun, H. High Prevalence of Plasmid-Mediated Quinolone Resistance Determinants in Escherichia coli and Klebsiella pneumoniae Isolates from Pediatric Patients in China. Microb. Drug Resist. 2017, 23, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Mahato, S.; Mahato, A.; Yadav, J. Prevalence and identification of uropathogens in eastern Nepal and under-standing their antibiogram due to multidrug resistance and Esbl. Asian Pac. J. Microbiol. Res. 2018, 2, 09–17. [Google Scholar]
- Alfola, M.M.H.R.; Kamel, Z.; Nada, M.G.E.D.; Rashed, L.A.; El-Awady, B.A. Phenotypic and Genotypic Characterization of ESBL-Producing Escherichia coli and Klebsiella pneumonia Isolates from Patient’s Urine Specimens. Int. Arab. J. Antimicrob. Agents 2016, 6, 6. [Google Scholar] [CrossRef][Green Version]
- Abdel-Moaty, M.M.; Mohamed, W.S.; Abdel-All, S.M.; El-Hendawy, H.H. Prevalence and Molecular Epidemiology of Extended Spectrum β-Lactamase Producing Escherichia coli from Hospital and Community Settings in Egypt. J. Appl. Pharm. Sci. 2016, 6, 042–047. [Google Scholar] [CrossRef]
- Nepal, K.; Pant, N.D.; Neupane, B.; Belbase, A.; Baidhya, R.; Shrestha, R.K.; Lekhak, B.; Bhatta, D.R.; Jha, B. Extended Spectrum Beta-Lactamase and Metallo Beta-Lactamase Production among Escherichia coli and Klebsiella pneumoniae Isolated from Different Clinical Samples in A Tertiary Care Hospital in Kathmandu, Nepal. Ann. Clin. Microbiol. Antimicrob. 2017, 16, 62. [Google Scholar] [CrossRef]
- Javed, H.; Ejaz, H.; Zafar, A.; Rathore, A.W.; Haq, I.U. Metallo-Beta-Lactamase Producing Escherichia coli and Klebsiella pneumoniae: A Rising Threat for Hospitalized Children. J. Pak. Med. Assoc. 2016, 66, 1068–1072. [Google Scholar] [PubMed]
- Wu, T.-L.; Siu, L.; Su, L.-H.; Lauderdale, T.; Lin, F.; Leu, H.-S.; Lin, T.-Y.; Ho, M. Outer membrane protein change combined with co-existing TEM-1 and SHV-1 β-lactamases lead to false identification of ESBL-producing Klebsiella pneu-moniae. J. Antimicrob. Chemother. 2001, 47, 755–761. [Google Scholar] [CrossRef] [PubMed][Green Version]
- WHO. Egypt: Pharmaceutical Country Profile; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- El-Shazly, D.A.; Nasef, S.; Mahmoud, F.F.; Jonas, D. Expanded Spectrum β–Lactamase Producing Escherichia coli Isolated from Chickens with Colibacillosis in Egypt. Poult. Sci. 2017, 96, 2375–2384. [Google Scholar] [CrossRef] [PubMed]
- Tille, P. Bailey & Scott’s Diagnostic Microbiology-E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Test Protocol. Am. Soc. Microbiol. 2009, 3189, 1–23. [Google Scholar]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 28th ed.; CLSI supplement M100; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018. [Google Scholar]
- Lee, K.; Chong, Y.; Shin, H.; Kim, Y.; Yong, D.; Yum, J. Modified Hodge and EDTA-Disk Synergy Tests to Screen Metallo-β-Lactamase-Producing Strains of Pseudomonas and Acinetobactet Species. Clin. Microbiol. Infect. 2001, 7, 88–91. [Google Scholar] [CrossRef]
- Nordmann, P.; Poirel, L.; Carrër, A.; Toleman, M.A.; Walsh, T.R. How to detect NDM-1 producers. J. Clin. Microbiol. 2011, 49, 718–721. [Google Scholar] [CrossRef] [PubMed]
- Stürenburg, E.; Kühn, A.; Mack, D.; Laufs, R. A novel extended-spectrum β-lactamase CTX-M-23 with a P167T substitution in the active-site omega loop associated with ceftazidime resistance. J. Antimicrob. Chemother. 2004, 54, 406–409. [Google Scholar] [CrossRef][Green Version]
- Ghorbani-Dalini, S.; Kargar, M.; Doosti, A.; Abbasi, P.; Sarshar, M. Molecular epidemiology of ESBL genes and multi-drug resistance in diarrheagenic Escherichia coli strains isolated from adults in Iran. Iran. J. Pharm. Res. 2015, 14, 1257. [Google Scholar] [PubMed]
- Ellington, M.J.; Kistler, J.; Livermore, D.M.; Woodford, N. Multiplex PCR for rapid detection of genes encoding acquired metallo-β-lactamases. J. Antimicrob. Chemother. 2007, 59, 321–322. [Google Scholar] [CrossRef]
- Gröbner, S.; Linke, D.; Schütz, W.; Fladerer, C.; Madlung, J.; Autenrieth, I.B.; Witte, W.; Pfeifer, Y.J. Emergence of carbapenem-non-susceptible extended-spectrum β-lactamase-producing Klebsiella pneumoniae isolates at the university hospital of Tübingen, Germany. J. Med. Microbiol. 2009, 58, 912–922. [Google Scholar] [CrossRef] [PubMed]
- Poirel, L.; Héritier, C.; Tolün, V.; Nordmann, P. Emergence of oxacillinase-mediated resistance to imipenem in Klebsiella pneumoniae. J. Antimicrob. Agents. Chemother. 2004, 48, 15–22. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhou, Z.; Qian, Y.; Wei, Z.; Yu, Y.; Hu, S.; Li, L. Plasmid-mediated quinolone resistance determinants qnr and aac (6′)-Ib-cr in extended-spectrum β-lactamase-producing Escherichia coli and Klebsiella pneumoniae in China. J. Antimicrob. Chemother. 2008, 61, 1003–1006. [Google Scholar] [CrossRef] [PubMed]


| Infection | No. of Samples | No. of E. coli Isolates | E. coli (%) * |
|---|---|---|---|
| Wound infections (burns, diabetic foot, surgery wound, cuts) | 150 | 66 | 44% |
| Urinary tract infection | 100 | 62 | 62% |
| Gastro-enteritis | 50 | 24 | 48% |
| Blood | 75 | 39 | 52% |
| Chest infection | 20 | 4 | 20% |
| Ear infection | 20 | 4 | 20% |
| Eye | 10 | 1 | 10% |
| Total | 425 | 200 | 47% |
| Hospitals | No. of Samples | E. coli | |
|---|---|---|---|
| No. | % * | ||
| Minia University Hospitals | 290 | 149 | 51.37 |
| Minia Chest Hospital | 20 | 4 | 20 |
| Minia General Hospital | 115 | 47 | 40.8 |
| Total | 425 | 200 | |
| Type of Infection | β-Lactam Resistant Isolates | ESBLs | MBL | ||
|---|---|---|---|---|---|
| No. | % * | No. | % * | ||
| Wound infections | 25 | 25 | 100% | 12 | 48% |
| Urinary tract infection | 28 | 28 | 100% | 20 | 71.4% |
| Gastro-enteritis | 15 | 5 | 33.3% | 0 | 0% |
| Blood | 20 | 20 | 100% | 12 | 46.67% |
| Respiratory infection | 4 | 4 | 100% | 2 | 50% |
| Ear infection | 2 | 2 | 100% | 1 | 50% |
| Eye | 1 | 1 | 100% | 1 | 100% |
| Total | 95 | 85 | 89.4% ** | 48 | 50.5% ** |
| Type of Infection | β-Lactam Resistant Isolates N | blaNDM | blaTEM | blaCTX-M | blaSHV | blaIMP | aac-(6′)-Ib-cr |
|---|---|---|---|---|---|---|---|
| N (%) * | N (%) * | N (%) * | N (%) * | N (%) * | N (%) * | ||
| Wound infections | 25 | 14 (56%) | 23 (92%) | 14 (56%) | 15 (60%) | 12 (48%) | 9 (36%) |
| UTI | 28 | 17 (60.7%) | 27 (96.4%) | 11 (39.2%) | 12 (42.8%) | 16 (57%) | 6 (21%) |
| Gastro-enteritis | 15 | 1 (7.6%) | 7 (46.6%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Blood | 20 | 7 (35%) | 20 (100%) | 13 (65%) | 20 (100%) | 7 (35%) | 10 (50%) |
| Chest infection | 4 | 1 (25%) | 3 (75%) | 0 (0%) | 4 (100%) | 0 (0%) | 0 (0%) |
| Ear infection | 2 | 1 (50%) | 2 (100%) | 1 (50%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Eye | 1 | 1 (100%) | 1 (100%) | 1 (100%) | 1 (100%) | 0 (0%) | 0 (0%) |
| Total | 95 | 42 (44.2%) | 76 (80%) | 40 (42%) | 52 (54.7%) | 35 (36.8%) | 25 (26.3%) |
| p value ** | 0.11 | <0.001 | 0.011 | <0.001 | 0.011 | 0.042 |
| MBLs Phenotype | blaNDM | blaIMP | Imipenem Resistance | Meropenem Resistance | |
|---|---|---|---|---|---|
| MBLs phenotype | 1 | 0.372 ** | 0.232 * | 0.237 * | 0.465 ** |
| blaNDM | 1 | 0.155 | 0.054 | 0.212 * | |
| blaIMP | 1 | 0.122 | 0.275 ** | ||
| Imipenem resistance | 1 | 0.292 ** | |||
| Meropenem resistance | 1 |
| ESBLs Production | MBLs Production | blaNDM | blaIMP | blaTEM | blaCTX-M | blaSHV | aac(6′)1b-cr | |
|---|---|---|---|---|---|---|---|---|
| MAR index | 0.611 ** | 0.342 ** | 0.330 ** | 0.289 ** | 0.366 ** | 0.365 ** | 0.251 * | 0.360 ** |
| ESBLs phenotype | 1 | 0.347 ** | 0.305 ** | 0.262 * | 0.696 ** | 0.293 ** | 0.377 ** | 0.205 * |
| Antibiotics | Number of Resistant Isolates | Number of aac(6′)Ib-cr Positive Isolates (%) * | Person Correlation (r2) | p Value |
|---|---|---|---|---|
| Streptomycin | 61 | 19 (31) | 0.135 | 0.193 |
| Tobramycin | 43 | 17 (39.5) | 0.301 | 0.003 ** |
| Gentamycin | 43 | 13 (30.2) | 0.044 | 0.670 |
| Amikacin | 36 | 17 (47.2) | 0.374 | <0.01 *** |
| Ofloxacin | 39 | 9 (23) | 0.018 | 0.866 |
| Norfloxacin | 42 | 14 (33.3) | 0.1450 | 0.162 |
| Ciprofloxacin | 37 | 12 (32.4) | 0.152 | 0.142 |
| Number of Isolates (%) | |
|---|---|
| One gene | 9 (9.5) |
| blaTEM blaSHV | 8 (8.5) 1 (1) |
| Two genes | 20 (21.1) |
| blaTEM, aac(6′)Ib-cr blaIMP, blaTEM blaTEM, blaSHV blaNDM, blaTEM blaTEM, blaCTX-M | 1 (1) 5 (5.2) 5 (5.2) 7 (7.3) 2 (2.1) |
| Three genes | 21 (22.1) |
| blaIMP, blaTEM, blaSHV blaIMP, blaTEM, blaCTX-M blaNDM, blaTEM, aac(6′)Ib-cr blaTEM, blaCTX-M, blaSHV blaTEM, blaSHV, aac(6′)Ib-cr blaNDM, blaCTX-M, blaSHV blaNDM, blaIMP, blaTEM blaNDM, blaTEM, blaSHV | 4 (4.2) 1 (1) 2 (2.1) 5 (5.2) 1 (1) 1 (1) 2 (2.1) 5 (5.2) |
| Four genes | 21(22.1) |
| blaTEM, blaCTX-M, blaSHV, aac(6′)Ib-cr blaNDM, blaIMP, blaTEM, aac(6′)Ib-cr blaNDM, blaTEM, blaCTX-M, blaSHV blaIMP, blaTEM, blaCTX-M, blaSHV blaIMP, blaTEM, blaSHV, aac(6′)Ib-cr blaNDM, blaIMP, blaTEM, blaCTX-M blaNDM, blaTEM, blaSHV, aac(6′)Ib-cr blaNDM, blaCTX-M, blaSHV, aac(6′)Ib-cr blaNDM, blaTEM, blaCTX-M, aac(6′)Ib-cr | 5 (5.2) 3 (3.1) 3 (3.1) 3 (3.1) 1 (1) 1 (1) 1 (1) 1 (1) 3 (3.1) |
| Five genes | 12 (12.6) |
| blaNDM, blaIMP,, blaTEM, blaCTX-M, blaSHV blaNDM, blaIMP, blaTEM, blaCTX-M, aac(6′)Ib-cr blaIMP, blaTEM, blaCTX-M, blaSHV, aac(6′)Ib-cr | 8 (8.5) 2 (2.1) 2 (2.1) |
| Six genes | 3 (3.2) |
| Total | 95 |
| blaNDM | blaIMP | blaTEM | blaCTX-M | blaSHV | aac(6′)1b-cr | |
|---|---|---|---|---|---|---|
| blaNDM | 1 | 0.155 | 0.211 * | 0.185 | 0.086 | 0.190 |
| blaIMP | 1 | 0.290 ** | 0.233 * | 0.081 | 0.089 | |
| blaTEM | 1 | 0.196 | 0.227 * | 0.155 | ||
| blaCTX-M | 1 | 0.519 ** | 0.265 ** | |||
| blaSHV | 1 | 0.159 | ||||
| aac(6′)Ib-cr | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Masoud, S.M.; Abd El-Baky, R.M.; Aly, S.A.; Ibrahem, R.A. Co-Existence of Certain ESBLs, MBLs and Plasmid Mediated Quinolone Resistance Genes among MDR E. coli Isolated from Different Clinical Specimens in Egypt. Antibiotics 2021, 10, 835. https://doi.org/10.3390/antibiotics10070835
Masoud SM, Abd El-Baky RM, Aly SA, Ibrahem RA. Co-Existence of Certain ESBLs, MBLs and Plasmid Mediated Quinolone Resistance Genes among MDR E. coli Isolated from Different Clinical Specimens in Egypt. Antibiotics. 2021; 10(7):835. https://doi.org/10.3390/antibiotics10070835
Chicago/Turabian StyleMasoud, Salwa Mahmoud, Rehab Mahmoud Abd El-Baky, Sherine A. Aly, and Reham Ali Ibrahem. 2021. "Co-Existence of Certain ESBLs, MBLs and Plasmid Mediated Quinolone Resistance Genes among MDR E. coli Isolated from Different Clinical Specimens in Egypt" Antibiotics 10, no. 7: 835. https://doi.org/10.3390/antibiotics10070835
APA StyleMasoud, S. M., Abd El-Baky, R. M., Aly, S. A., & Ibrahem, R. A. (2021). Co-Existence of Certain ESBLs, MBLs and Plasmid Mediated Quinolone Resistance Genes among MDR E. coli Isolated from Different Clinical Specimens in Egypt. Antibiotics, 10(7), 835. https://doi.org/10.3390/antibiotics10070835






