Donnan Potential across the Outer Membrane of Gram-Negative Bacteria and Its Effect on the Permeability of Antibiotics
Abstract
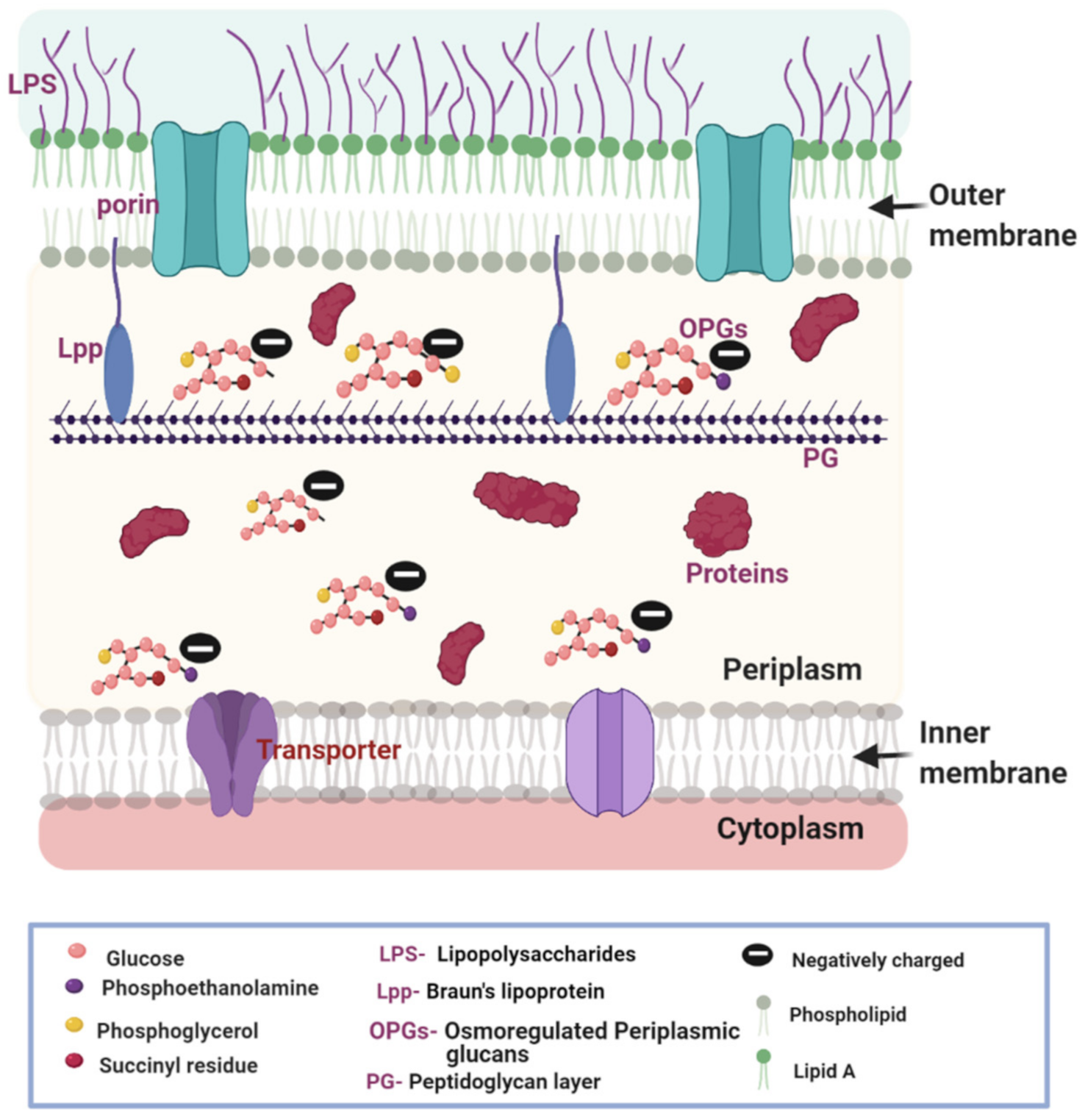
1. The Composition of the Gram-Negative Bacterial Cell Envelope
2. Membrane Potential (MP) Regulates Antibiotic Transport across the OM of Gram-Negative Bacteria
2.1. OPGs Contribute to the Generation of DP
2.2. Variation of pH of the External Media Alters DP
2.3. Synergy between Porins and DP in Cation Selection and Cellular Accumulation
3. Impact of DP on Susceptibility to Antibiotics in Bacterial Infections
3.1. Alteration of Bacterial Virulence
3.2. Impact on Susceptibility of Bacteria to Antibiotics
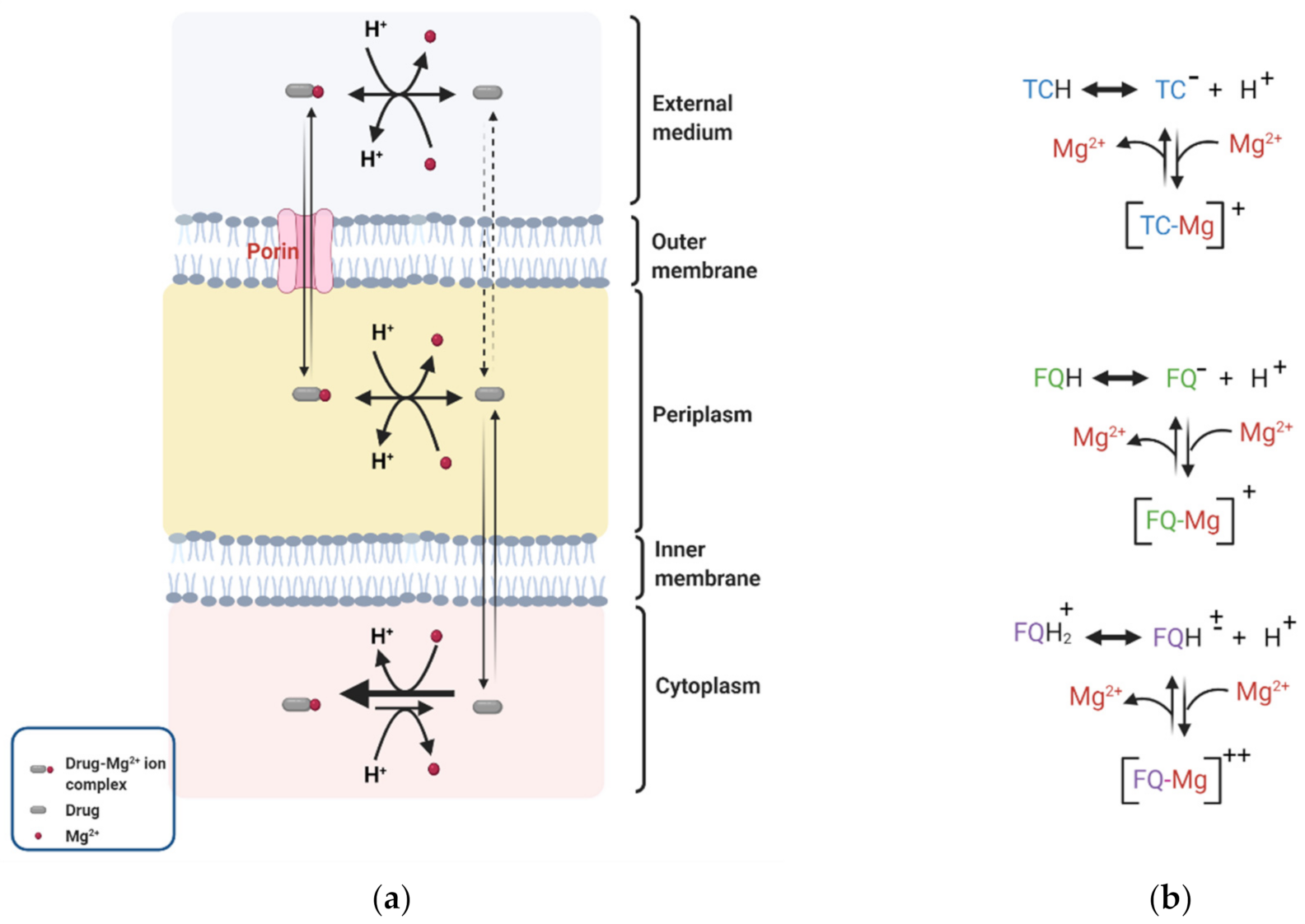
4. Effect of DP on Membrane Permeability to Selected Antibiotics
4.1. Fluoroquinolones
4.2. Tetracyclines
4.3. β-. Lactams
4.4. Trimethoprims
5. Measurement of DP across the OM of Gram-Negative Bacteria
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Beveridge, T. Ultrastructure, chemistry, and function of the bacterial wall. In International Review of Cytology; Elsevier: Amsterdam, The Netherlands, 1981; pp. 229–317. [Google Scholar]
- Shockman, G.; Kawamura, T.; Barrett, J.; Dolinger, D. The autolytic system of Streptococcus faecium. In Proceedings of the International FEMS Symposium, Berlin, Germany, 13–18 March 1983; pp. 165–172. [Google Scholar]
- Nikaido, H. Molecular Basis of Bacterial Outer Membrane Permeability Revisited. Microbiol. Mol. Biol. Rev. 2003, 67, 593–656. [Google Scholar] [CrossRef] [PubMed]
- Koebnik, R.; Locher, K.P.; Van Gelder, P. Structure and function of bacterial outer membrane proteins: Barrels in a nutshell. Mol. Microbiol. 2000, 37, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Schulz, G.E.J.B. The structure of bacterial outer membrane proteins. Biochim. Biophys. Acta (BBA)-Biomembr. 2002, 1565, 308–317. [Google Scholar] [CrossRef]
- Bos, M.P.; Robert, V.; Tommassen, J. Biogenesis of the Gram-Negative Bacterial Outer Membrane. Annu. Rev. Microbiol. 2007, 61, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Kadner, R.J.E. Cytoplasmic membrane. Cell. Mol. Biol. 1996, 1, 58–87. [Google Scholar]
- Hancock, R. Role of porins in outer membrane permeability. J. Bacteriol. 1987, 169, 929–933. [Google Scholar] [CrossRef]
- Benz, R.; Bauer, K.J. Permeation of hydrophilic molecules through the outer membrane of gram-negativ bacteria: Review of becterial porins. Eur. J. Biochem. 1988, 176, 1–19. [Google Scholar] [CrossRef]
- Schulz, G.E.; Schirmer, R.H. Principles of Protein Structure; Springer: New York, NY, USA, 1979; pp. 17–26. [Google Scholar]
- Zimmermann, W.; Rosselet, A. Function of the Outer Membrane of Escherichia coli as a Permeability Barrier to Beta-Lactam Antibiotics. Antimicrob. Agents Chemother. 1977, 12, 368–372. [Google Scholar] [CrossRef]
- Choi, U.; Lee, C.-R. Distinct Roles of Outer Membrane Porins in Antibiotic Resistance and Membrane Integrity in Escherichia coli. Front. Microbiol. 2019, 10, 953. [Google Scholar] [CrossRef]
- Buchanan, S.K. β-Barrel proteins from bacterial outer membranes: Structure, function and refolding. Curr. Opin. Struct. Biol. 1999, 9, 455–461. [Google Scholar] [CrossRef]
- Hirai, K.; Aoyama, H.; Irikura, T.; Iyobe, S.; Mitsuhashi, S. Differences in susceptibility to quinolones of outer membrane mutants of Salmonella typhimurium and Escherichia coli. Antimicrob. Agents Chemother. 1986, 29, 535–538. [Google Scholar] [CrossRef]
- Montero, M.T.; Hernàndez-Borrell, A.J.; Keough, K.M.W. Fluoroquinolone−Biomembrane Interactions: Monolayer and Calorimetric Studies. Langmuir 1998, 14, 2451–2454. [Google Scholar] [CrossRef]
- Bohin, J.-P. Osmoregulated periplasmic glucans in Proteobacteria. FEMS Microbiol. Lett. 2000, 186, 11–19. [Google Scholar] [CrossRef]
- Pandeya, A.; Ojo, I.; Alegun, O.; Wei, Y. Periplasmic Targets for the Development of Effective Antimicrobials against Gram-Negative Bacteria. ACS Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Asmar, A.T.; Ferreira, J.; Cohen, E.J.; Cho, S.-H.; Beeby, M.; Hughes, K.T.; Collet, J.-F. Communication across the bacterial cell envelope depends on the size of the periplasm. PLoS Biol. 2017, 15, e2004303. [Google Scholar] [CrossRef]
- Beveridge, T. The periplasmic space and the periplasm in gram-positive and gram-negative bacteria. ASM News 1995, 61, 125–130. [Google Scholar]
- Mai-Prochnow, A.; Clauson, M.; Hong, J.; Murphy, A. Gram positive and Gram negative bacteria differ in their sensitivity to cold plasma. Sci. Rep. 2016, 6, 38610. [Google Scholar] [CrossRef]
- Kennedy, E.P. Osmotic regulation and the biosynthesis of membrane-derived oligosaccharides in Escherichia coli. Proc. Natl. Acad. Sci. USA 1982, 79, 1092–1095. [Google Scholar] [CrossRef]
- Stock, J.; Rauch, B.; Roseman, S. Periplasmic space in Salmonella typhimurium and Escherichia coli. J. Biol. Chem. 1977, 252, 7850–7861. [Google Scholar] [CrossRef]
- Zgurskaya, H.I.; Rybenkov, V.V.; Krishnamoorthy, G.; Leus, I.V. Trans-envelope multidrug efflux pumps of Gram-negative bacteria and their synergism with the outer membrane barrier. Res. Microbiol. 2018, 169, 351–356. [Google Scholar] [CrossRef]
- Blair, J.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J.V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Genet. 2015, 13, 42–51. [Google Scholar] [CrossRef]
- Nikaido, H. Multidrug efflux pumps of gram-negative bacteria. J. Bacteriol. 1996, 178, 5853. [Google Scholar] [CrossRef]
- Poole, K.J. Efflux-me23diated antimicrobial resistance. J. Antimicrob. Chemother. 2005, 56, 20–51. [Google Scholar] [CrossRef]
- Li, X.-Z.; Plésiat, P.; Nikaido, H. The Challenge of Efflux-Mediated Antibiotic Resistance in Gram-Negative Bacteria. Clin. Microbiol. Rev. 2015, 28, 337–418. [Google Scholar] [CrossRef]
- Nikaido, H.; Pagès, J.-M. Broad-specificity efflux pumps and their role in multidrug resistance of Gram-negative bacteria. FEMS Microbiol. Rev. 2012, 36, 340–363. [Google Scholar] [CrossRef]
- Maloney, P.C.; Kashket, E.R.; Wilson, T.H. A Protonmotive Force Drives ATP Synthesis in Bacteria. Proc. Natl. Acad. Sci. USA 1974, 71, 3896–3900. [Google Scholar] [CrossRef]
- Padan, E.; Zilberstein, D.; Schuldiner, S. pH homesstasis in bacteria. Biochim. Biophys. Acta Rev. Biomembr. 1981, 650, 151–166. [Google Scholar] [CrossRef]
- Strahl, H.; Hamoen, L.W. Membrane potential is important for bacterial cell division. Proc. Natl. Acad. Sci. USA 2010, 107, 12281–12286. [Google Scholar] [CrossRef]
- Sirec, T.; Benarroch, J.; Buffard, P.; Garcia-Ojalvo, J.; Asally, M. Electrical Polarization Enables Integrative Quality Control during Bacterial Differentiation into Spores. iScience 2019, 16, 378–389. [Google Scholar] [CrossRef]
- Lee, D.-Y.D.; Prindle, A.; Liu, J.; Süel, G.M. SnapShot: Electrochemical Communication in Biofilms. Cell 2017, 170, 214–214.e1. [Google Scholar] [CrossRef]
- Novo, D.; Perlmutter, N.G.; Hunt, R.H.; Shapiro, H.M. Accurate flow cytometric membrane potential measurement in bacteria using diethyloxacarbocyanine and a ratiometric technique. Cytometry 1999, 35, 55–63. [Google Scholar] [CrossRef]
- Benarroch, J.M.; Asally, M. The Microbiologist’s Guide to Membrane Potential Dynamics. Trends Microbiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Teorell, T. Transport processes in ionic membranes. Prog. Biophys. Biophys. Chem. 1953, 3, 305. [Google Scholar] [CrossRef]
- Kennedy, E.P.; Rumley, M.K.; Schulman, H.; Van Golde, L.M. Identification of sn-glycero-1-phosphate and phosphoethanolamine residues linked to the membrane-derived Oligosaccharides of Escherichia coli. J. Biol. Chem. 1976, 251, 4208–4213. [Google Scholar] [CrossRef]
- Schulman, H.; Kennedy, E.P. Relation of turnover of membrane phospholipids to synthesis of membrane-derived oligosaccharides of Escherichia coli. J. Biol. Chem. 1977, 252, 4250–4255. [Google Scholar] [CrossRef]
- Schulman, H.; Kennedy, E.P. Identification of UDP-glucose as an intermediate in the biosynthesis of the membrane-derived oligosaccharides of Escherichia coli. J. Biol. Chem. 1977, 252, 6299–6303. [Google Scholar] [CrossRef]
- Golde, L.M.G.V.; Schulman, H.; Kennedy, E.P. Metabolism of Membrane Phospholipids and Its Relation to a Novel Class of Oligosaccharides in Escherichia coli. Proc. Natl. Acad. Sci. USA 1973, 70, 1368–1372. [Google Scholar] [CrossRef]
- Todt, J.C.; Rocque, W.J.; McGroarty, E.J. Effects of pH on bacterial porin function. Biochemistry 1992, 31, 10471–10478. [Google Scholar] [CrossRef]
- Sen, K.; Hellman, J.; Nikaido, H. Porin channels in intact cells of Escherichia coli are not affected by Donnan potentials across the outer membrane. J. Biol. Chem. 1988, 263, 1182–1187. [Google Scholar] [CrossRef]
- Miller, K.J.; Kennedy, E.P.; Reinhold, V.N. Osmotic adaptation by gram-negative bacteria: Possible role for periplasmic oligosaccharides. Science 1986, 231, 48–51. [Google Scholar] [CrossRef]
- Bakker, E.P. Membrane potential in a potassium transport-negative mutant of Escherichia coli K-12: The distribution of rubidium in the presence of valinomycin indicates a higher potential than that of the tetraphenylphosphonium cation. Biochim. Biophys. Acta (BBA)-Bioenerg. 1982, 681, 474–483. [Google Scholar] [CrossRef]
- Schulman, H.; Kennedy, E.P. Localization of membrane-derived oligosaccharides in the outer envelope of Escherichia coli and their occurrence in other Gram-negative bacteria. J. Bacteriol. 1979, 137, 686–688. [Google Scholar] [CrossRef]
- McLaggan, D.; Belkin, S.; Packer, L.; Matin, A. Electron spin resonance measurements of the effect of lonophores on the transmembrane pH gradient of an acidophilic bacterium. Arch. Biochem. Biophys. 1989, 273, 206–214. [Google Scholar] [CrossRef]
- Kobayashi, H. A proton-translocating ATPase regulates pH of the bacterial cytoplasm. J. Biol. Chem. 1985, 260, 72–76. [Google Scholar] [CrossRef]
- Card, G.L.; Trautman, J.K. Role of anionic lipid in bacterial membranes. Biochim. Biophys. Acta (BBA)-Lipids Lipid Metab. 1990, 1047, 77–82. [Google Scholar] [CrossRef]
- Koyama, N.; Niimura, Y.; Kozaki, M. Bioenergetic properties of a facultatively anaerobic alkalophile. FEMS Microbiol. Lett. 1988, 49, 123–126. [Google Scholar] [CrossRef]
- Goulbourne, E.; Matin, M.; Zychlinsky, E.; Matin, A. Mechanism of delta pH maintenance in active and inactive cells of an obligately acidophilic bacterium. J. Bacteriol. 1986, 166, 59–65. [Google Scholar] [CrossRef]
- Morisaki, H.; Nagai, S.; Kogure, K.; Ikemoto, E.; Ohshima, H. The effect of motility and cell-surface polymers on bacterial attachment. Microbiology 1999, 145, 2797–2802. [Google Scholar] [CrossRef]
- Plette, A.C.; van Riemsdijk, W.H.; Benedetti, M.F.; van der Wal, A. pH dependent charging behavior of isolated cell walls of a gram-positive soil bacterium. J. Colloid Interface Sci. 1995, 173, 354–363. [Google Scholar] [CrossRef]
- Kiers, P.J.M.; Bos, R.; Van Der Mei, H.C.; Busscher, H.J. The electrophoretic softness of the surface of Staphylococcus epidermidis cells grown in a liquid medium and on a solid agar. Microbiology 2001, 147, 757–762. [Google Scholar] [CrossRef]
- Burnett, P.-G.; Heinrich, H.; Peak, D.; Bremer, P.J.; McQuillan, A.J.; Daughney, C.J. The effect of pH and ionic strength on proton adsorption by the thermophilic bacterium Anoxybacillus flavithermus. Geochim. Cosmochim. Acta 2006, 70, 1914–1927. [Google Scholar] [CrossRef]
- Tsuneda, S.; Jung, J.; Hayashi, H.; Aikawa, H.; Hirata, A.; Sasaki, H. Influence of extracellular polymers on electrokinetic properties of heterotrophic bacterial cells examined by soft particle electrophoresis theory. Colloids Surf. B Biointerfaces 2003, 29, 181–188. [Google Scholar] [CrossRef]
- Jones, H.; Holland, I.; Campbell, A. Direct measurement of free Ca2+ shows different regulation of Ca2+ between the periplasm and the cytosol of Escherichia coli. Cell Calcium 2002, 32, 183–192. [Google Scholar] [CrossRef]
- Martinez, R.E.; Smith, S.; Kulczycki, E.; Ferris, F. Determination of Intrinsic Bacterial Surface Acidity Constants using a Donnan Shell Model and a Continuous pKa Distribution Method. J. Colloid Interface Sci. 2002, 253, 130–139. [Google Scholar] [CrossRef]
- Goto, T.; Hirabayashi, T.; Morimoto, H.; Yamazaki, K.; Inoue, N.; Matsuyama, H.; Yumoto, I. Contribution of intracellular negative ion capacity to Donnan effect across the membrane in alkaliphilic Bacillus spp. J. Bioenerg. Biomembr. 2016, 48, 87–96. [Google Scholar] [CrossRef]
- Yee, N.; Fowle, D.A.; Ferris, F. A Donnan potential model for metal sorption onto Bacillus subtilis. Geochim. Cosmochim. Acta 2004, 68, 3657–3664. [Google Scholar] [CrossRef]
- Barbosa, N.S.; Lima, E.R.; Tavares, F.W. The electrostatic behavior of the bacterial cell wall using a smoothing function to describe the charge-regulated volume charge density profile. Colloids Surf. B Biointerfaces 2015, 134, 447–452. [Google Scholar] [CrossRef]
- Pandeya, A.; Alegun, O.; Cai, Y.; Wei, Y. Distribution of fluoroquinolones in the two aqueous compartments of Escherichia coli. Biochem. Biophys. Rep. 2020, 24, 100849. [Google Scholar] [CrossRef] [PubMed]
- Prochnow, H.; Fetz, V.; Hotop, S.-K.; García-Rivera, M.A.; Heumann, A.; Brönstrup, M. Subcellular Quantification of Uptake in Gram-Negative Bacteria. Anal. Chem. 2019, 91, 1863–1872. [Google Scholar] [CrossRef] [PubMed]
- Ropponen, H.-K.; Diamanti, E.; Siemens, A.; Illarionov, B.; Haupenthal, J.; Fischer, M.; Rottmann, M.; Witschel, M.; Hirsch, A.K.H. Assessment of the rules related to gaining activity against Gram-negative bacteria. RSC Med. Chem. 2021, 12, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Perlmutter, S.J.; Geddes, E.J.; Drown, B.S.; Motika, S.E.; Lee, M.R.; Hergenrother, P.J. Compound Uptake into E. coli Can Be Facilitated by N-Alkyl Guanidiniums and Pyridiniums. ACS Infect. Dis. 2021, 7, 162–173. [Google Scholar] [CrossRef]
- Nikaido, H.; Thanassi, D.G. Penetration of lipophilic agents with multiple protonation sites into bacterial cells: Tetracyclines and fluoroquinolones as examples. Antimicrob. Agents Chemother. 1993, 37, 1393–1399. [Google Scholar] [CrossRef]
- Talaga, P.; Fournet, B.; Bohin, J.P. Periplasmic glucans of Pseudomonas syringae pv. syringae. J. Bacteriol. 1994, 176, 6538–6544. [Google Scholar] [CrossRef][Green Version]
- Schneider, J.; Reinhold, V.; Rumley, M.; Kennedy, E. Structural studies of the membrane-derived oligosaccharides of Escherichia coli. J. Biol. Chem. 1979, 254, 10135–10138. [Google Scholar] [CrossRef]
- Bontemps-Gallo, S.; Bohin, J.-P.; Lacroix, J.-M. Osmoregulated periplasmic glucans. EcoSal Plus 2017, 7, 0001–2017. [Google Scholar] [CrossRef]
- Kennedy, E. Membrane-derived oligosaccharides (periplasmic beta-D-glucans) of Escherichia coli. Cell. Mol. Biol. 1996, 1064–1071. [Google Scholar]
- Kennedy, E. Membrane-Derived Oligosaccharides; American Society for Microbiology: Washington, WA, USA, 1987; pp. 672–679. [Google Scholar]
- Pedebos, C.; Smith, I.P.S.; Boags, A.; Khalid, S.J.S. The hitchhiker’s guide to the periplasm: Unexpected molecular interactions of polymyxin B1 in E. coli. Structure 2021, 29, 444–456. [Google Scholar] [CrossRef]
- Delcour, A.H.; Adler, J.; Kung, C.; Martinac, B. Membrane-derived oligosaccharides (MDO’s) promote closing of an E. coli porin channel. FEBS Lett. 1992, 304, 216–220. [Google Scholar] [CrossRef]
- Bhagwat, A.A.; Jun, W.; Liu, L.; Kannan, P.; Dharne, M.; Pheh, B.; Tall, B.D.; Kothary, M.H.; Gross, K.C.; Angle, S. Osmoregulated periplasmic glucans of Salmonella enterica serovar Typhimurium are required for optimal virulence in mice. Microbiology 2009, 155, 229–237. [Google Scholar] [CrossRef]
- Bontemps-Gallo, S.; Cogez, V.; Robbe-Masselot, C.; Quintard, K.; Dondeyne, J.; Madec, E.; Lacroix, J.-M. Biosynthesis of osmoregulated periplasmic glucans in Escherichia coli: The phosphoethanolamine transferase is encoded by opgE. BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Lacroix, J.-M. Etude Génétique et Physiologique de la Régulation Osmotique de la Biosynthèse du MDO chez Escherichia coli. Ph.D. Thesis, Université de Paris-Sud, Centre d’Orsay, France, 1989. [Google Scholar]
- Cama, J.; Bajaj, H.; Pagliara, S.; Maier, T.; Braun, Y.; Winterhalter, M.; Keyser, U. Quantification of Fluoroquinolone Uptake through the Outer Membrane Channel OmpF of Escherichia coli. J. Am. Chem. Soc. 2015, 137, 13836–13843. [Google Scholar] [CrossRef]
- O’Neil, M.J.; Heckelman, P.; Koch, C.; Roman, K.J.M.; Co, W.S. An Encyclopedia of Chemicals, Drugs and Biologicals; Merck Research Laboratories: Kenilworth, NJ, USA, 2006. [Google Scholar]
- Nikaido, H.; Rosenberg, E.Y.; Foulds, J. Porin channels in Escherichia coli: Studies with beta-lactams in intact cells. J. Bacteriol. 1983, 153, 232–240. [Google Scholar] [CrossRef]
- Kojima, S.; Nikaido, H. High salt concentrations increase permeability through OmpC channels of Escherichia coli. J. Biol. Chem. 2014, 289, 26464–26473. [Google Scholar] [CrossRef]
- Nikaido, H.; Vaara, M. Molecular basis of bacterial outer membrane permeability. Microbiol. Rev. 1985, 49, 1–32. [Google Scholar] [CrossRef]
- Hasegawa, Y.; Yamada, H.; Mizushima, S. Interactions of Outer Membrane Proteins O-8 and O-9 with Peptidoglycan Sacculus of Escherichia coli K-121. J. Biochem. 1976, 80, 1401–1409. [Google Scholar] [CrossRef]
- Pratt, L.A.; Hsing, W.; Gibson, K.E.; Silhavy, T.J. From acids to osmZ: Multiple factors influence synthesis of the OmpF and OmpC porins in Escherichia coli. Mol. Microbiol. 1996, 20, 911–917. [Google Scholar] [CrossRef]
- Mulani, M.S.; Kamble, E.; Kumkar, S.N.; Tawre, M.S.; Pardesi, K.R. Emerging Strategies to Combat ESKAPE Pathogens in the Era of Antimicrobial Resistance: A Review. Front. Microbiol. 2019, 10, 539. [Google Scholar] [CrossRef]
- Tommasi, R.; Brown, D.G.; Walkup, G.K.; Manchester, J.I.; Miller, A.A. ESKAPEing the labyrinth of antibacterial discovery. Nat. Rev. Drug Discov. 2015, 14, 529–542. [Google Scholar] [CrossRef] [PubMed]
- O’Shea, R.; Moser, H.E. Physicochemical properties of antibacterial compounds: Implications for drug discovery. J. Med. Chem. 2008, 51, 2871–2878. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.G.; May-Dracka, T.L.; Gagnon, M.M.; Tommasi, R. Trends and Exceptions of Physical Properties on Antibacterial Activity for Gram-Positive and Gram-Negative Pathogens. J. Med. Chem. 2014, 57, 10144–10161. [Google Scholar] [CrossRef] [PubMed]
- Thanassi, D.G.; Suh, G.S.; Nikaido, H. Role of outer membrane barrier in efflux-mediated tetracycline resistance of Escherichia coli. J. Bacteriol. 1995, 177, 998–1007. [Google Scholar] [CrossRef]
- Silver, L.L. A Gestalt approach to Gram-negative entry. Bioorg. Med. Chem. 2016, 24, 6379–6389. [Google Scholar] [CrossRef]
- Franco, M.P.; Mulder, M.; Gilman, R.H.; Smits, H.L. Human brucellosis. Lancet Infect. Dis. 2007, 7, 775–786. [Google Scholar] [CrossRef]
- Galinska, E.M.; Zagórski, J. Brucellosis in humans-etiology, diagnostics, clinical forms. Ann. Agric. Environ. Med. 2013, 20, 233–238. [Google Scholar]
- Ryan, E.T. The Cholera Pandemic, Still with Us after Half a Century: Time to Rethink. PLoS Negl. Trop. Dis. 2011, 5, e1003. [Google Scholar] [CrossRef]
- Thomas, S.; Lekshmi, N.; Joseph, I.; Ramamurthy, T. Changing facades of Vibrio cholerae: An enigma in the epidemiology of cholera. Indian J. Med. Res. 2018, 147, 133. [Google Scholar] [CrossRef]
- Ma, A.; Glassman, H.; Chui, L. Characterization of Escherichia coli possessing the locus of heat resistance isolated from human cases of acute gastroenteritis. Food Microbiol. 2020, 88, 103400. [Google Scholar] [CrossRef]
- Gastañaduy, A.S.; Begue, R. Acute gastroentertis. Clin. Pediatr. 1999, 38, 1–12. [Google Scholar] [CrossRef]
- Crowley, D.S.; Ryan, M.J.; Wall, P.G. Gastroenteritis in children under 5 years of age in England and Wales. Commun. Dis. Rep. CDR Rev. 1997, 7, 82–86. [Google Scholar]
- Tapsall, J. Antibiotic resistance in Neisseria gonorrhoeae is diminishing available treatment options for gonorrhea: Some possible remedies. Expert Rev. Anti-Infect. Ther. 2006, 4, 619–628. [Google Scholar] [CrossRef]
- Osnes, M.N.; van Dorp, L.; Brynildsrud, O.B.; Alfsnes, K.; Schneiders, T.; Templeton, K.; Yahara, K.; Balloux, F.; Caugant, D.; Eldholm, V. Antibiotic Treatment Regimes as a Driver of the Global Population Dynamics of a Major Gonorrhea Lineage. Mol. Biol. Evol. 2021, 38, 1249–1261. [Google Scholar] [CrossRef]
- Cunha, B.; Burillo, A.; Bouza, E. Legionnaires’ disease. Lancet 2016, 387, 376–385. [Google Scholar] [CrossRef]
- Phin, N.; Parry-Ford, F.; Harrison, T.; Stagg, H.; Zhang, N.; Kumar, K.; Lortholary, O.; Zumla, A.; Abubakar, I. Epidemiology and clinical management of Legionnaires’ disease. Lancet Infect. Dis. 2014, 14, 1011–1021. [Google Scholar] [CrossRef]
- Rumbo, M.; Hozbor, D. Development of improved pertussis vaccine. Hum. Vaccines Immunother. 2014, 10, 2450–2453. [Google Scholar] [CrossRef]
- Heininger, U. Update on pertussis in children. Expert Rev. Anti-Infect. Ther. 2010, 8, 163–173. [Google Scholar] [CrossRef]
- Raoult, D.; Mouffok, N.; Bitam, I.; Piarroux, R.; Drancourt, M. Plague: History and contemporary analysis. J. Infect. 2013, 66, 18–26. [Google Scholar] [CrossRef]
- Poland, J.D.; Quan, T.; Barnes, A.M. Plague. In Handbook of Zoonoses; CRC Press: London, UK, 2019; pp. 93–112. [Google Scholar]
- Okesola, A.; Ige, O.M. Trends in bacterial pathogens of lower respiratory tract infections. Indian J. Chest Dis. Allied Sci. 2008, 50, 269–272. [Google Scholar]
- Jiancong, L. Clinical feature and drug resistance of lower respiratory nosocomial infection caused by Acinetobacter baumannii. Lab. Med. Clin. 2011, 1, 1. [Google Scholar]
- Jain, N.; Lodha, R.; Kabra, S.K. Upper respiratory tract infections. Indian J. Pediatr. 2001, 68, 1135–1138. [Google Scholar] [CrossRef]
- Scorza, F.B.; Colucci, A.M.; Maggiore, L.; Sanzone, S.; Rossi, O.; Ferlenghi, I.; Pesce, I.; Caboni, M.; Norais, N.; Di Cioccio, V.; et al. High Yield Production Process for Shigella Outer Membrane Particles. PLoS ONE 2012, 7, e35616. [Google Scholar]
- Kotloff, K.L.; Riddle, M.S.; Platts-Mills, J.A.; Pavlinac, P.; Zaidi, A.K. Shigellosis. Lancet 2018, 391, 801–812. [Google Scholar] [CrossRef]
- MacLennan, C.; Riddle, M.S.; Chen, W.H.; Talaat, K.R.; Jain, V.; Bourgeois, A.L.; Frenck, R.; Kotloff, K.; Porter, C.K. Consensus Report on Shigella Controlled Human Infection Model: Clinical Endpoints. Clin. Infect. Dis. 2019, 69, S591–S595. [Google Scholar] [CrossRef] [PubMed]
- Turtiainen, J.; Hakala, T.; Hakkarainen, T.; Karhukorpi, J. The Impact of Surgical Wound Bacterial Colonization on the Incidence of Surgical Site Infection After Lower Limb Vascular Surgery: A Prospective Observational Study. Eur. J. Vasc. Endovasc. Surg. 2014, 47, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Agwunglefah, F.; Nwabunike, C.; Nwaju, P. Antibiotic susceptibility pattern of bacteria isolated from surgical wounds of patients attending federal medical center and Christiana Specialist Hospital, Owerri. J. Nat. Sci. Res. 2014, 4, 2224–3186. [Google Scholar]
- Adegoke, A.A.; Tom, M.; Okoh, A.I.; Jacob, S. Studies on multiple antibiotic resistant bacterial isolated from surgical site infection. Sci. Res. Essays 2010, 5, 3876–3881. [Google Scholar]
- Control, C. Tularemia—United States, 1990–2000. MMWR. Morb. Mortal. Wkly. Rep. 2002, 51, 181–184. [Google Scholar]
- Gyuranecz, M. Tularemia. In Manual of Diagnostic tests and Vaccines for Terrestrial Animals; Office Interntional des Epizooties: Párizs, France, 2018; pp. 675–682. [Google Scholar]
- Kidgell, C.; Reichard, U.; Wain, J.; Linz, B.; Torpdahl, M.; Dougan, G.; Achtman, M. Salmonella typhi, the causative agent of typhoid fever, is approximately 50,000 years old. Infect. Genet. Evol. 2002, 2, 39–45. [Google Scholar] [CrossRef]
- Kadhiravan, T.; Wig, N.; Kapil, A.; Kabra, S.K.; Renuka, K.; Misra, A. Clinical outcomes in typhoid fever: Adverse impact of infection with nalidixic acid-resistant Salmonella typhi. BMC Infect. Dis. 2005, 5, 37. [Google Scholar] [CrossRef]
- Medeiros, A.A.; O’Brien, T.F.; Wacker, W.E.C.; Yulug, N.F. Effect of Salt Concentration on the Apparent In-Vitro Susceptibility of Pseudomonas and Other Gram-Negative Bacilli to Gentamicin. J. Infect. Dis. 1971, 124, S59–S64. [Google Scholar] [CrossRef]
- Thapa, P.; Parajuli, K.; Poudel, A.; Thapa, A.K.; Manandhar, B.; Laudari, D.; Malla, H.B.; Katiwada, R. Causative Agents and Susceptibility of Antimicrobials among Suspected Females with Urinary Tract Infection in Tertiary Care Hospitals of Western Nepal. J. Chitwan Med. Coll. 2013, 3, 16–19. [Google Scholar] [CrossRef]
- Khleifat, K.M.; Abboud, M.; Omar, S.; Al-Kurishy, J. Urinary tract infection in South Jordanian population. J. Med. Sci. 2006, 6, 5–11. [Google Scholar]
- Sabih, A.; Leslie, S.W. Complicated Urinary Tract Infections; StatPearls Publishing: Treasure Island, FL, USA, 2017. [Google Scholar]
- McLellan, L.K.; Hunstad, D.A. Urinary tract infection: Pathogenesis and outlook. Trends Mol. Med. 2016, 22, 946–957. [Google Scholar] [CrossRef]
- Arellano-Reynoso, B.; Lapaque, N.; Salcedo, S.; Briones, G.; Ciocchini, A.E.; Ugalde, R.; Moreno, E.; Moriyon, I.; Gorvel, J.-P. Cyclic β-1,2-glucan is a brucella virulence factor required for intracellular survival. Nat. Immunol. 2005, 6, 618–625. [Google Scholar] [CrossRef]
- Bhagwat, A.; Gross, K.C.; Tully, R.; Keister, D.L. Beta-glucan synthesis in Bradyrhizobium japonicum: Characterization of a new locus (ndvC) influencing beta-(1→6) linkages. J. Bacteriol. 1996, 178, 4635–4642. [Google Scholar] [CrossRef]
- Loubens, I.; Debarbieux, L.; Bohin, A.; Lacroix, J.M.; Bohin, J. Homology between a genetic locus (mdoA) involved in the osmoregulated biosynthesis of periplasmic glucans in Escherichia coli and a genetic locus (hrpM) controlling pathogenicity of Pseudomonas syringae. Mol. Microbiol. 1993, 10, 329–340. [Google Scholar] [CrossRef]
- Mahajan-Miklos, S.; Tan, M.-W.; Rahme, L.G.; Ausubel, F.M. Molecular Mechanisms of Bacterial Virulence Elucidated Using a Pseudomonas aeruginosa–Caenorhabditis elegans Pathogenesis Model. Cell 1999, 96, 47–56. [Google Scholar] [CrossRef]
- Page, F.; Altabe, S.; Hugouvieux-Cotte-Pattat, N.; Lacroix, J.-M.; Robert-Baudouy, J.; Bohin, J.-P. Osmoregulated Periplasmic Glucan Synthesis is Required for Erwinia chrysanthemi Pathogenicity. J. Bacteriol. 2001, 183, 3134–3141. [Google Scholar] [CrossRef][Green Version]
- Bowe, F.; Lipps, C.J.; Tsolis, R.M.; Groisman, E.; Heffron, F.; Kusters, J.G. At Least Four Percent of the Salmonella typhimurium Genome is Required for Fatal Infection of Mice. Infect. Immun. 1998, 66, 3372–3377. [Google Scholar] [CrossRef]
- Burland, V.; Plunkett, G.; Sofia, H.J.; Daniels, D.L.; Blattner, F.R. Analysis of the Escherichia coli genome VI: DNA sequence of the region from 92.8 through 100 minutes. Nucleic Acids Res. 1995, 23, 2105–2119. [Google Scholar] [CrossRef]
- Chopra, I. Molecular mechanisms involved in the transport of antibiotics into bacteria. Parasitology 1988, 96, S25–S44. [Google Scholar] [CrossRef]
- James, C.E.; Mahendran, K.R.; Molitor, A.; Bolla, J.-M.; Bessonov, A.N.; Winterhalter, M.; Pagès, J.-M. How β-Lactam Antibiotics Enter Bacteria: A Dialogue with the Porins. PLoS ONE 2009, 4, e5453. [Google Scholar] [CrossRef] [PubMed]
- Norris, S.; Mandell, G.L. The Quinolones: History and Overview. In The Quinolones; Elsevier: Amsterdam, The Netherlands, 1988; pp. 1–22. [Google Scholar]
- Smith, A.; Pennefather, P.M.; Kaye, S.B.; Hart, C. Fluoroquinolones. Drugs 2001, 61, 747–761. [Google Scholar] [CrossRef] [PubMed]
- Lesher, G.Y.; Froelich, E.J.; Gruett, M.D.; Bailey, J.H.; Brundage, R.P. 1,8-Naphthyridine Derivatives. A New Class of Chemotherapeutic Agents. J. Med. Chem. 1962, 5, 1063–1065. [Google Scholar] [CrossRef] [PubMed]
- Ball, P. Quinolone generations: Natural history or natural selection? J. Antimicrob. Chemother. 2000, 46, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Vila, J.; Ruiz, J.; Goñi, P.; De Anta, M.T. Detection of mutations in parC in quinolone-resistant clinical isolates of Escherichia coli. Antimicrob. Agents Chemother. 1996, 40, 491–493. [Google Scholar] [CrossRef]
- Pan, X.S.; Fisher, L.M. Targeting of DNA gyrase in Streptococcus pneumoniae by sparfloxacin: Selective targeting of gyrase or topoisomerase IV by quinolones. Antimicrob. Agents Chemother. 1997, 41, 471–474. [Google Scholar] [CrossRef]
- Takenouchi, T.; Tabata, F.; Iwata, Y.; Hanzawa, H.; Sugawara, M.; Ohya, S. Hydrophilicity of quinolones is not an exclusive factor for decreased activity in efflux-mediated resistant mutants of Staphylococcus aureus. Antimicrob. Agents Chemother. 1996, 40, 1835–1842. [Google Scholar] [CrossRef]
- Pan, X.S.; Ambler, J.; Mehtar, S.; Fisher, L.M. Involvement of topoisomerase IV and DNA gyrase as ciprofloxacin targets in Streptococcus pneumoniae. Antimicrob. Agents Chemother. 1996, 40, 2321–2326. [Google Scholar] [CrossRef]
- Timmers, K.; Sternglanz, R. Ionization and divalent cation dissociation constants of nalidixic and oxolinic acids. Bioinorg. Chem. 1978, 9, 145–155. [Google Scholar] [CrossRef]
- Ramón, M.S.; Cantón, E.; Pemán, J.; Pastor, A.; Martínez, J.P. Mechanisms of Action of Quinolones against Staphylococci and Relationship with Their in vitro Bactericidal Activity. Chemotherapy 1999, 45, 175–182. [Google Scholar] [CrossRef]
- Denis, A.; Moreau, N.J. Mechanisms of quinolone resistance in clinical isolates: Accumulation of sparfloxacin and of fluoroquinolones of various hydrophobicity, and analysis of membrane composition. J. Antimicrob. Chemother. 1993, 32, 379–392. [Google Scholar] [CrossRef]
- Chen, Z.-F.; Xiong, R.-G.; Zuo, J.-L.; Guo, Z.; You, X.-Z.; Fun, H.-K. X-Ray crystal structures of Mg2+ and Ca2+ dimers of the antibacterial drug norfloxacin. J. Chem. Soc. Dalton Trans. 2000, 4013–4014. [Google Scholar] [CrossRef]
- Fresta, M.; Guccione, S.; Beccari, A.R.; Furneri, P.M.; Puglisi, G. Combining molecular modeling with experimental methodologies: Mechanism of membrane permeation and accumulation of ofloxacin. Bioorganic Med. Chem. 2002, 10, 3871–3889. [Google Scholar] [CrossRef]
- Fedorowicz, J.; Sączewski, J. Modifications of quinolones and fluoroquinolones: Hybrid compounds and dual-action molecules. Mon. Chem. Chem. Mon. 2018, 149, 1199–1245. [Google Scholar] [CrossRef]
- Mach, T.; Neves, P.; Spiga, E.; Weingart, H.; Winterhalter, M.; Ruggerone, P.; Ceccarelli, M.; Gameiro, P. Facilitated Permeation of Antibiotics across Membrane Channels—Interaction of the Quinolone Moxifloxacin with the OmpF Channel. J. Am. Chem. Soc. 2008, 130, 13301–13309. [Google Scholar] [CrossRef]
- Lecomte, S.; Baron, M.H.; Chenon, M.T.; Coupry, C.; Moreau, N.J. Effect of magnesium complexation by fluoroquinolones on their antibacterial properties. Antimicrob. Agents Chemother. 1994, 38, 2810–2816. [Google Scholar] [CrossRef]
- Chopra, I.; Hawkey, P.M.; Hinton, M. Tetracyclines, molecular and clinical aspects. J. Antimicrob. Chemother. 1992, 29, 245–277. [Google Scholar] [CrossRef]
- Schnappinger, D.; Hillen, W. Tetracyclines: Antibiotic action, uptake, and resistance mechanisms. Arch. Microbiol. 1996, 165, 359–369. [Google Scholar] [CrossRef]
- Garrod, L.P.; O’grady, F. Antibiotic and Chemotherapy; Churchill Livingstone: Edinburgh, Scotland, UK, 1971; p. 377. [Google Scholar]
- Chopra, I.; Roberts, M. Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef]
- Barden, T.C.; Buckwalter, B.L.; Testa, R.T.; Petersen, P.J.; Lee, V. “Glycylcyclines” 3. 9-Aminodoxycyclinecarboxamides. J. Med. Chem. 1994, 37, 3205–3211. [Google Scholar] [CrossRef]
- Bergeron, J.; Ammirati, M.; Danley, D.; James, L.; Norcia, M.; Retsema, J.; Strick, C.; Su, W.G.; Sutcliffe, J.; Wondrack, L. Glycylcyclines bind to the high-affinity tetracycline ribosomal binding site and evade Tet(M)- and Tet(O)-mediated ribosomal protection. Antimicrob. Agents Chemother. 1996, 40, 2226–2228. [Google Scholar] [CrossRef] [PubMed]
- Testa, R.T.; Petersen, P.J.; Jacobus, N.V.; Sum, P.; Lee, V.J.; Tally, F.P. In vitro and in vivo antibacterial activities of the glycylcyclines, a new class of semisynthetic tetracyclines. Antimicrob. Agents Chemother. 1993, 37, 2270–2277. [Google Scholar] [CrossRef] [PubMed]
- Rogalski, W. Chemical Modification of the Tetracyclines. In The Tetracyclines; Springer: Berlin/Heidelberg, Germany, 1985; pp. 179–316. [Google Scholar]
- Leive, L.; Telesetsky, S.; Coleman, W.G.; Carr, D. Tetracyclines of various hydrophobicities as a probe for permeability of Escherichia coli outer membranes. Antimicrob. Agents Chemother. 1984, 25, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, P.G.S.; Piddock, L.J.; Piddok, L.J.V. The accumulation of five antibacterial agents in porin-deficient mutants of Escherichia coli. J. Antimicrob. Chemother. 1993, 32, 195–213. [Google Scholar] [CrossRef]
- Nikaido, H. Outer membrane of Salmonella typhimurium: Transmembrane diffusion of some hydrophobic substances. Biochim. Biophys. Acta (BBA)-Biomembr. 1976, 433, 118–132. [Google Scholar] [CrossRef]
- Nikaido, H.; Rosenberg, E.Y. Effect on solute size on diffusion rates through the transmembrane pores of the outer membrane of Escherichia coli. J. Gen. Physiol. 1981, 77, 121–135. [Google Scholar] [CrossRef]
- Fleming, A. On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of B. influenzae. Br. J. Exp. Pathol. 1929, 10, 226. [Google Scholar] [CrossRef]
- Chain, E.; Florey, H.; Gardner, A.; Heatley, N.; Jennings, M.; Orr-Ewing, J.; Sanders, A. Penicillin as a chemotherapeutic agent. Lancet 1940, 236, 226–228. [Google Scholar] [CrossRef]
- Mandell, G.; Sande, M.J. Antimicrobial agents: Penicillins, cephalosporins, and other beta-lactam antibiotics. In Goodman & Gilman’s The Pharmacologic Basis of Therapeutics; McGraw-Hill: New York, NY, USA, 1996; pp. 1073–1101. [Google Scholar]
- Shahid, M.; Sobia, F.; Singh, A.; Malik, A.; Khan, H.M.; Jonas, D.; Hawkey, P.M. Beta-lactams and Beta-lactamase-inhibitors in current- or potential-clinical practice: A comprehensive update. Crit. Rev. Microbiol. 2009, 35, 81–108. [Google Scholar] [CrossRef]
- Ghuysen, J.-M. Serine β-lactamases and penicillin-binding proteins. Annu. Rev. Microbiol. 1991, 45, 37–67. [Google Scholar] [CrossRef]
- Du Bois, S.K.; Marriott, M.S.; Amyes, S.G.B. TEM- and SHV-derived extended-spectrum β-lactamases: Relationship between selection, structure and function. J. Antimicrob. Chemother. 1995, 35, 7–22. [Google Scholar] [CrossRef]
- Bayles, K. The bactericidal action of penicillin: New clues to an unsolved mystery. Trends Microbiol. 2000, 8, 274–278. [Google Scholar] [CrossRef]
- Delcour, A.H. Outer membrane permeability and antibiotic resistance. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2009, 1794, 808–816. [Google Scholar] [CrossRef]
- Nikaido, H.; Liu, W.; Rosenberg, E.Y. Outer membrane permeability and beta-lactamase stability of dipolar ionic cephalosporins containing methoxyimino substituents. Antimicrob. Agents Chemother. 1990, 34, 337–342. [Google Scholar] [CrossRef]
- Bellido, F.; Pechere, J.C.; E Hancock, R. Novel method for measurement of outer membrane permeability to new beta-lactams in intact Enterobacter cloacae cells. Antimicrob. Agents Chemother. 1991, 35, 68–72. [Google Scholar] [CrossRef][Green Version]
- Bushby, S.R.M.; Hitchings, G.H. Trimethoprim, a sulphonamide potentiator. Br. J. Pharmacol. Chemother. 1968, 33, 72–90. [Google Scholar] [CrossRef]
- Eliopoulos, G.M.; Huovinen, P. Resistance to Trimethoprim-Sulfamethoxazole. Clin. Infect. Dis. 2001, 32, 1608–1614. [Google Scholar] [CrossRef]
- Phetsang, W.; Pelingon, R.; Butler, M.; Kc, S.; Pitt, M.; Kaeslin, G.; Cooper, M.A.; Blaskovich, M.A.T. Fluorescent Trimethoprim Conjugate Probes to Assess Drug Accumulation in Wild Type and Mutant Escherichia coli. ACS Infect. Dis. 2016, 2, 688–701. [Google Scholar] [CrossRef]
- Morrison, D.C.; Jacobs, D.M. Binding of polymyxin B to the lipid a portion of bacterial lipopolysaccharides. Immunochemistry 1976, 13, 813–818. [Google Scholar] [CrossRef]
| Diseases/Conditions | Associated Gram-Negative Bacteria |
|---|---|
| Brucellosis [89,90] | Brucella spp. |
| Cholera [91,92] | Vibrio cholerae |
| Gastroenteritis [93,94,95] | E. coli, Salmonella spp., Shigella spp., Campylobacter jejuni |
| Gonorrhea [96,97] | Neisseria gonorrhoeae |
| Legionnaires’ Disease [98,99] | Legionella pneumophila |
| Pertussis [100,101] | Bordetella pertussis |
| Plague [102,103] | Yersinia pestis |
| Respiratory tract infection, Pneumonia [104,105,106] | E. coli, Klebsiella pneumonia, Acinetobacter baumanii, Pseudomonas aeruginosa |
| Shigellosis [107,108,109] | Shigella spp. |
| Surgical wound infection [110,111,112] | E. coli, Klebsiella pneumonia, Acinetobacter baumanii, Pseudomonas aeruginosa |
| Tularemia [113,114] | Francisella tularensis |
| Typhoid fever [115,116] | Salmonella typhimurium |
| Urinary tract and urinary-catheter infections [117,118,119,120,121] | E. coli, Klebsiella pneumonia |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alegun, O.; Pandeya, A.; Cui, J.; Ojo, I.; Wei, Y. Donnan Potential across the Outer Membrane of Gram-Negative Bacteria and Its Effect on the Permeability of Antibiotics. Antibiotics 2021, 10, 701. https://doi.org/10.3390/antibiotics10060701
Alegun O, Pandeya A, Cui J, Ojo I, Wei Y. Donnan Potential across the Outer Membrane of Gram-Negative Bacteria and Its Effect on the Permeability of Antibiotics. Antibiotics. 2021; 10(6):701. https://doi.org/10.3390/antibiotics10060701
Chicago/Turabian StyleAlegun, Olaniyi, Ankit Pandeya, Jian Cui, Isoiza Ojo, and Yinan Wei. 2021. "Donnan Potential across the Outer Membrane of Gram-Negative Bacteria and Its Effect on the Permeability of Antibiotics" Antibiotics 10, no. 6: 701. https://doi.org/10.3390/antibiotics10060701
APA StyleAlegun, O., Pandeya, A., Cui, J., Ojo, I., & Wei, Y. (2021). Donnan Potential across the Outer Membrane of Gram-Negative Bacteria and Its Effect on the Permeability of Antibiotics. Antibiotics, 10(6), 701. https://doi.org/10.3390/antibiotics10060701






