Emergence of Hybrid Resistance and Virulence Plasmids Harboring New Delhi Metallo-β-Lactamase in Klebsiella pneumoniae in Russia
Abstract
1. Introduction
2. Results
2.1. Clinical Manifestations and Antimicrobial Susceptibility of Recovered Isolates
2.2. General Sequencing Results
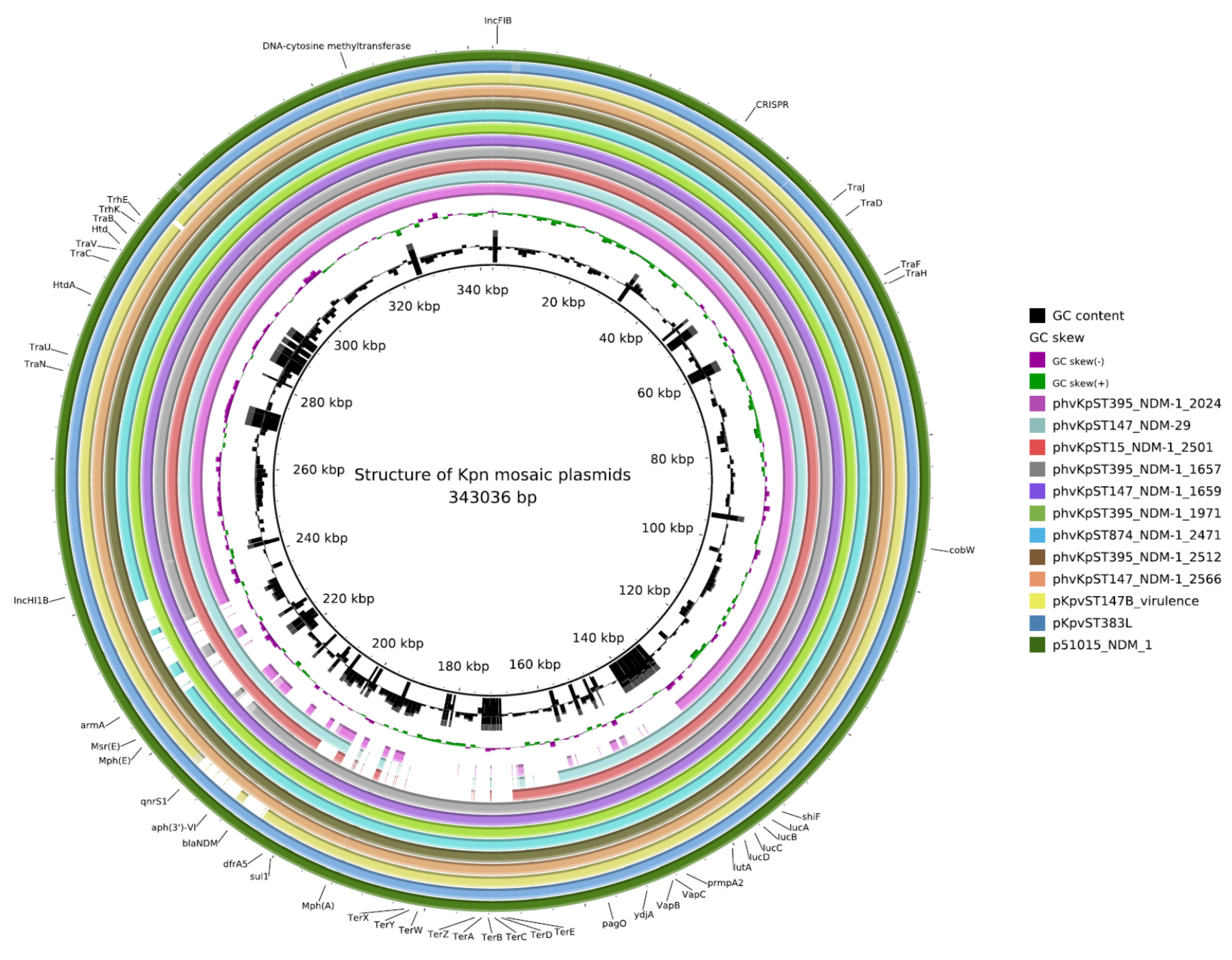
2.3. Plasmids
2.4. AMR Determinants
2.5. blaNDM
2.6. Functional Analysis of blandm-29
2.7. Virulence
2.8. Restriction Modification and TA Systems
2.9. CRISPR-Cas System
3. Discussion
4. Materials and Methods
4.1. Identification of Bacterial Strains and Antimicrobial Susceptibility Testing
4.2. Cloning
4.3. Genomic Sequencing
4.4. Sequence Analysis
4.5. MURINE Model of Sepsis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gu, D.; Dong, N.; Zheng, Z.; Lin, D.; Huang, M.; Wang, L.; Chan, E.W.; Shu, L.; Yu, J.; Zhang, R.; et al. A fatal outbreak of ST11 carbapenem-resistant hypervirulent Klebsiella pneumoniae in a Chinese hospital: A molecular epidemiological study. Lancet Infect Dis. 2018, 18, 37–46. [Google Scholar] [CrossRef]
- Boyd, S.E.; Livermore, D.M.; Hooper, D.C.; Hope, W.W. Metallo-beta-Lactamases: Structure, Function, Epidemiology, Treatment Options, and the Development Pipeline. Antimicrob. Agents Chemother. 2020, 64. [Google Scholar] [CrossRef]
- Wyres, K.L.; Wick, R.R.; Judd, L.M.; Froumine, R.; Tokolyi, A.; Gorrie, C.L.; Lam, M.M.C.; Duchêne, S.; Jenney, A.; Holt, K.E. Distinct evolutionary dynamics of horizontal gene transfer in drug resistant and virulent clones of Klebsiella pneumoniae. PLoS Genet. 2019, 15, e1008114. [Google Scholar] [CrossRef] [PubMed]
- Lan, P.; Jiang, Y.; Zhou, J.; Yu, Y. A Global Perspective on the Convergence of Hypervirulence and Carbapenem-Resistance in Klebsiella pneumoniae. J. Glob. Antimicrob. Resist. 2021. [Google Scholar] [CrossRef] [PubMed]
- Yong, D.; Toleman, M.A.; Giske, C.G.; Cho, H.S.; Sundman, K.; Lee, K.; Walsh, T.R. Characterization of a new metallo-beta-lactamase gene, bla(NDM-1), and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob. Agents Chemother. 2009, 53, 5046–5054. [Google Scholar] [CrossRef]
- Khan, A.U.; Maryam, L.; Zarrilli, R. Structure, Genetics and Worldwide Spread of New Delhi Metallo-beta-lactamase (NDM): A threat to public health. BMC Microbiol. 2017, 17, 101. [Google Scholar] [CrossRef]
- Bahr, G.; Vitor-Horen, L.; Bethel, C.R.; Bonomo, R.A.; Gonzalez, L.J.; Vila, A.J. Clinical Evolution of New Delhi Metallo-beta-Lactamase (NDM) Optimizes Resistance under Zn(II) Deprivation. Antimicrob. Agents Chemother. 2018, 62. [Google Scholar] [CrossRef]
- Cheng, Z.; Thomas, P.W.; Ju, L.; Bergstrom, A.; Mason, K.; Clayton, D.; Miller, C.; Bethel, C.R.; VanPelt, J.; Tierney, D.L.; et al. Evolution of New Delhi metallo-beta-lactamase (NDM) in the clinic: Effects of NDM mutations on stability, zinc affinity, and mono-zinc activity. J. Biol. Chem. 2018, 293, 12606–12618. [Google Scholar] [CrossRef] [PubMed]
- Lazareva, I.; Ageevets, V.; Sopova, J.; Lebedeva, M.; Starkova, P.; Likholetova, D.; Lebedeva, M.; Gostev, V.; Moiseenko, V.; Egorenkov, V.; et al. The emergence of hypervirulent blaNDM-1-positive Klebsiella pneumoniae sequence type 395 in an oncology hospital. Infect. Genet. Evol. 2020, 85, 104527. [Google Scholar] [CrossRef]
- Vasilyev, I.Y.; Nikolaeva, I.V.; Siniagina, M.N.; Kharchenko, A.M.; Shaikhieva, G.S. Multidrug-Resistant Hypervirulent Klebsiella pneumoniae Found Persisting Silently in Infant Gut Microbiota. Int. J. Microbiol. 2020, 2020, 4054393. [Google Scholar] [CrossRef] [PubMed]
- Shaidullina, E.; Shelenkov, A.; Yanushevich, Y.; Mikhaylova, Y.; Shagin, D.; Alexandrova, I.; Ershova, O.; Akimkin, V.; Kozlov, R.; Edelstein, M. Antimicrobial Resistance and Genomic Characterization of OXA-48- and CTX-M-15-Co-Producing Hypervirulent Klebsiella pneumoniae ST23 Recovered from Nosocomial Outbreak. Antibiotics 2020, 9, 862. [Google Scholar] [CrossRef] [PubMed]
- Turton, J.; Davies, F.; Turton, J.; Perry, C.; Payne, Z.; Pike, R. Hybrid Resistance and Virulence Plasmids in “High-Risk” Clones of Klebsiella pneumoniae, Including Those Carrying bla(NDM-5). Microorganisms 2019, 7, 326. [Google Scholar] [CrossRef] [PubMed]
- Turner, M.S.; Andersson, P.; Bell, J.M.; Turnidge, J.D.; Harris, T.; Giffard, P.M. Plasmid-borne blaSHV genes in Klebsiella pneumoniae are associated with strong promoters. J. Antimicrob. Chemother. 2009, 64, 960–964. [Google Scholar] [CrossRef] [PubMed]
- Weber, R.E.; Pietsch, M.; Fruhauf, A.; Pfeifer, Y.; Martin, M.; Luft, D.; Gatermann, S.; Pfennigwerth, N.; Kaase, M.; Werner, G.; et al. IS26-Mediated Transfer of bla NDM-1 as the Main Route of Resistance Transmission During a Polyclonal, Multispecies Outbreak in a German Hospital. Front. Microbiol. 2019, 10, 2817. [Google Scholar] [CrossRef]
- Poirel, L.; Bonnin, R.A.; Nordmann, P. Analysis of the resistome of a multidrug-resistant NDM-1-producing Escherichia coli strain by high-throughput genome sequencing. Antimicrob. Agents Chemother. 2011, 55, 4224–4229. [Google Scholar] [CrossRef]
- Poirel, L.; Dortet, L.; Bernabeu, S.; Nordmann, P. Genetic features of blaNDM-1-positive Enterobacteriaceae. Antimicrob. Agents Chemother. 2011, 55, 5403–5407. [Google Scholar] [CrossRef]
- Toleman, M.A.; Spencer, J.; Jones, L.; Walsh, T.R. blaNDM-1 is a chimera likely constructed in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2012, 56, 2773–2776. [Google Scholar] [CrossRef]
- Wu, W.; Feng, Y.; Tang, G.; Qiao, F.; McNally, A.; Zong, Z. NDM Metallo-beta-Lactamases and Their Bacterial Producers in Health Care Settings. Clin. Microbiol. Rev. 2019, 32. [Google Scholar] [CrossRef]
- Zhou, S.; Chen, X.; Meng, X.; Zhang, G.; Wang, J.; Zhou, D.; Guo, X. “Roar” of blaNDM-1 and “silence” of blaOXA-58 co-exist in Acinetobacter pittii. Sci Rep. 2015, 5, 8976. [Google Scholar] [CrossRef]
- Acman, M.; Wang, R.; van Dorp, L.; Shaw, L.P.; Wang, Q.; Luhmann, N.; Yuyao, Y.; Sun, S.; Chen, H.; Wang, H.; et al. Role of the mobilome in the global dissemination of the carbapenem resistance gene blaNDM. bioRxiv 2021. [Google Scholar] [CrossRef]
- Zou, D.; Huang, Y.; Zhao, X.; Liu, W.; Dong, D.; Li, H.; Wang, X.; Huang, S.; Wei, X.; Yan, X.; et al. A novel New Delhi metallo-β-lactamase variant, NDM-14, isolated in a Chinese Hospital possesses increased enzymatic activity against carbapenems. Antimicrob. Agents Chemother. 2015, 59, 2450–2453. [Google Scholar] [CrossRef]
- Nordmann, P.; Boulanger, A.E.; Poirel, L. NDM-4 metallo-beta-lactamase with increased carbapenemase activity from Escherichia coli. Antimicrob. Agents Chemother. 2012, 56, 2184–2186. [Google Scholar] [CrossRef] [PubMed]
- Cuzon, G.; Bonnin, R.A.; Nordmann, P. First identification of novel NDM carbapenemase, NDM-7, in Escherichia coli in France. PLoS ONE 2013, 8, e61322. [Google Scholar] [CrossRef]
- Mancini, S.; Keller, P.M.; Greiner, M.; Bruderer, V.; Imkamp, F. Detection of NDM-19, a novel variant of the New Delhi metallo-beta-lactamase with increased carbapenemase activity under zinc-limited conditions, in Switzerland. Diagn. Microbiol. Infect. Dis. 2019, 95, 114851. [Google Scholar] [CrossRef]
- Tang, Y.; Fu, P.; Zhou, Y.; Xie, Y.; Jin, J.; Wang, B.; Yu, L.; Huang, Y.; Li, G.; Li, M.; et al. Absence of the type I-E CRISPR-Cas system in Klebsiella pneumoniae clonal complex 258 is associated with dissemination of IncF epidemic resistance plasmids in this clonal complex. J. Antimicrob. Chemother. 2020, 75, 890–895. [Google Scholar] [CrossRef]
- Ogura, T.; Hiraga, S. Mini-F plasmid genes that couple host cell division to plasmid proliferation. Proc. Natl. Acad. Sci. USA 1983, 80, 4784–4788. [Google Scholar] [CrossRef]
- Pilla, G.; Tang, C.M. Going around in circles: Virulence plasmids in enteric pathogens. Nat. Rev. Microbiol. 2018, 16, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.M.; Jin, C.; Kim, D.H.; Lee, Y.; Lee, B.J. Structural and Functional Study of the Klebsiella pneumoniae VapBC Toxin-Antitoxin System, Including the Development of an Inhibitor That Activates VapC. J. Med. Chem. 2020, 63, 13669–13679. [Google Scholar] [CrossRef] [PubMed]
- Kamruzzaman, M.; Iredell, J.R. CRISPR-Cas System in Antibiotic Resistance Plasmids in Klebsiella pneumoniae. Front. Microbiol. 2019, 10, 2934. [Google Scholar] [CrossRef] [PubMed]
- Shmakov, S.A.; Utkina, I.; Wolf, Y.I.; Makarova, K.S.; Severinov, K.V.; Koonin, E.V. CRISPR Arrays Away from cas Genes. CRISPR J. 2020, 3, 535–549. [Google Scholar] [CrossRef]
- Green, V.L.; Verma, A.; Owens, R.J.; Phillips, S.E.; Carr, S.B. Structure of New Delhi metallo-beta-lactamase 1 (NDM-1). Acta Cryst. Sect. Struct. Biol. Cryst. Commun. 2011, 67, 1160–1164. [Google Scholar] [CrossRef] [PubMed]
- Hadano, Y. String test. BMJ Case Rep. 2013, 2013. [Google Scholar] [CrossRef]
- Lee, T.D.; Adie, K.; McNabb, A.; Purych, D.; Mannan, K.; Azana, R.; Ng, C.; Tang, P.; Hoang, L.M. Rapid Detection of KPC, NDM, and OXA-48-Like Carbapenemases by Real-Time PCR from Rectal Swab Surveillance Samples. J. Clin. Microbiol. 2015, 53, 2731–2733. [Google Scholar] [CrossRef]
- Wyres, K.L.; Wick, R.R.; Gorrie, C.; Jenney, A.; Follador, R.; Thomson, N.R.; Holt, K.E. Identification of Klebsiella capsule synthesis loci from whole genome data. Microb. Genom. 2016, 2, e000102. [Google Scholar] [CrossRef]
- Russo, T.A.; Marr, C.M. Hypervirulent Klebsiella pneumoniae. Clin. Microbiol. Rev. 2019, 32. [Google Scholar] [CrossRef]
- Horesh, G.; Harms, A.; Fino, C.; Parts, L.; Gerdes, K.; Heinz, E.; Thomson, N.R. SLING: A tool to search for linked genes in bacterial datasets. Nucleic Acids Res. 2018, 46, e128. [Google Scholar] [CrossRef]
- Siguier, P.; Perochon, J.; Lestrade, L.; Mahillon, J.; Chandler, M. ISfinder: The reference centre for bacterial insertion sequences. Nucleic Acids Res. 2006, 34, D32–D36. [Google Scholar] [CrossRef] [PubMed]
- Alikhan, N.F.; Petty, N.K.; Ben Zakour, N.L.; Beatson, S.A. BLAST Ring Image Generator (BRIG): Simple prokaryote genome comparisons. BMC Genom. 2011, 12, 402. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.J.; Petty, N.K.; Beatson, S.A. Easyfig: A genome comparison visualizer. Bioinformatics 2011, 27, 1009–1010. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.M.C.; Wick, R.R.; Wyres, K.L.; Holt, K.E. Genomic surveillance framework and global population structure for Klebsiella pneumoniae. bioRxiv 2020. [Google Scholar] [CrossRef]
- Heffernan, R.; Paliwal, K.; Lyons, J.; Singh, J.; Yang, Y.; Zhou, Y. Single-sequence-based prediction of protein secondary structures and solvent accessibility by deep whole-sequence learning. J. Comput. Chem. 2018, 39, 2210–2216. [Google Scholar] [CrossRef] [PubMed]
| Isolate | Year of Isolation | Location | ST | Capsule Type | Mouse Lethality (LD50, CFU) | blaNDM | Virulence Scores | Resistance Score | Predominant Diagnosis | Outcomes |
|---|---|---|---|---|---|---|---|---|---|---|
| 1657_kpn | 2017 | SPb_1 1 | 395 | KL2 | 102 | blaNDM-1 | 4 | 2 | Adenocarcinoma of the sigmoid colon Breast cancer | death |
| 1659_kpn | 2017 | SPb_1 | 147 | KL20 | 104 | blaNDM-1 | 3 | 2 | ||
| 1971_kpn | 2018 | SPb_1 | 395 | KL2 | 105 | blaNDM-1 | 4 | 2 | Pancreatic cancer | no change |
| 1970_kpn | 2018 | SPb_1 | 147 | KL20 | 104 | blaNDM-29 | 3 | 2 | Prostate cancer | improvement |
| 2024_kpn | 2018 | SPb_1 | 395 | KL2 | 105 | - | 1 | 2 | B cell lymphoma | no change |
| 2471_kpn | 2019 | SPb_1 | 874 | KL45 | 104 | blaNDM-1 | 3 | 2 | Hysterocarcinoma, IA st., pT1aN0M0 | no change |
| 2501_kpn | 2019 | SPb_1 | 15 | KL19 | 104 | blaNDM-1 | 3 | 2 | Rectal cancer cT3N1M0, IV Stomach body cancer T2N1M0 | improvement |
| 2512_kpn | 2019 | SPb_2 2 | 395 | KL2 | 103 | blaNDM-1 | 4 | 2 | Cornea injury | left eye vision loss |
| 2566_kpn | 2019 | SPb_1 | 147 | KL20 | 104 | blaNDM-1 | 3 | 2 | Follicular lymphoma, grade 1–2, IV | improvement |
| Enzymes | Amino Acids | ||
|---|---|---|---|
| blaNDM-1 | - | - | - |
| blaNDM-29 | N130 | - | - |
| blaNDM-14 | G130 | - | - |
| blaNDM-4 | - | L154 | - |
| blaNDM-6 | - | - | V233 |
| blaNDM-8 | G130 | L154 | - |
| blaNDM-7 | N130 | L154 | - |
| blaNDM-15 | - | L154 | V233 |
| blaNDM-19 | N130 | L154 | V233 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Starkova, P.; Lazareva, I.; Avdeeva, A.; Sulian, O.; Likholetova, D.; Ageevets, V.; Lebedeva, M.; Gostev, V.; Sopova, J.; Sidorenko, S. Emergence of Hybrid Resistance and Virulence Plasmids Harboring New Delhi Metallo-β-Lactamase in Klebsiella pneumoniae in Russia. Antibiotics 2021, 10, 691. https://doi.org/10.3390/antibiotics10060691
Starkova P, Lazareva I, Avdeeva A, Sulian O, Likholetova D, Ageevets V, Lebedeva M, Gostev V, Sopova J, Sidorenko S. Emergence of Hybrid Resistance and Virulence Plasmids Harboring New Delhi Metallo-β-Lactamase in Klebsiella pneumoniae in Russia. Antibiotics. 2021; 10(6):691. https://doi.org/10.3390/antibiotics10060691
Chicago/Turabian StyleStarkova, Polina, Irina Lazareva, Alisa Avdeeva, Ofeliia Sulian, Darya Likholetova, Vladimir Ageevets, Marina Lebedeva, Vladimir Gostev, Julia Sopova, and Sergey Sidorenko. 2021. "Emergence of Hybrid Resistance and Virulence Plasmids Harboring New Delhi Metallo-β-Lactamase in Klebsiella pneumoniae in Russia" Antibiotics 10, no. 6: 691. https://doi.org/10.3390/antibiotics10060691
APA StyleStarkova, P., Lazareva, I., Avdeeva, A., Sulian, O., Likholetova, D., Ageevets, V., Lebedeva, M., Gostev, V., Sopova, J., & Sidorenko, S. (2021). Emergence of Hybrid Resistance and Virulence Plasmids Harboring New Delhi Metallo-β-Lactamase in Klebsiella pneumoniae in Russia. Antibiotics, 10(6), 691. https://doi.org/10.3390/antibiotics10060691








