Enzyme-Responsive Nanoparticles and Coatings Made from Alginate/Peptide Ciprofloxacin Conjugates as Drug Release System
Abstract
1. Introduction
2. Results
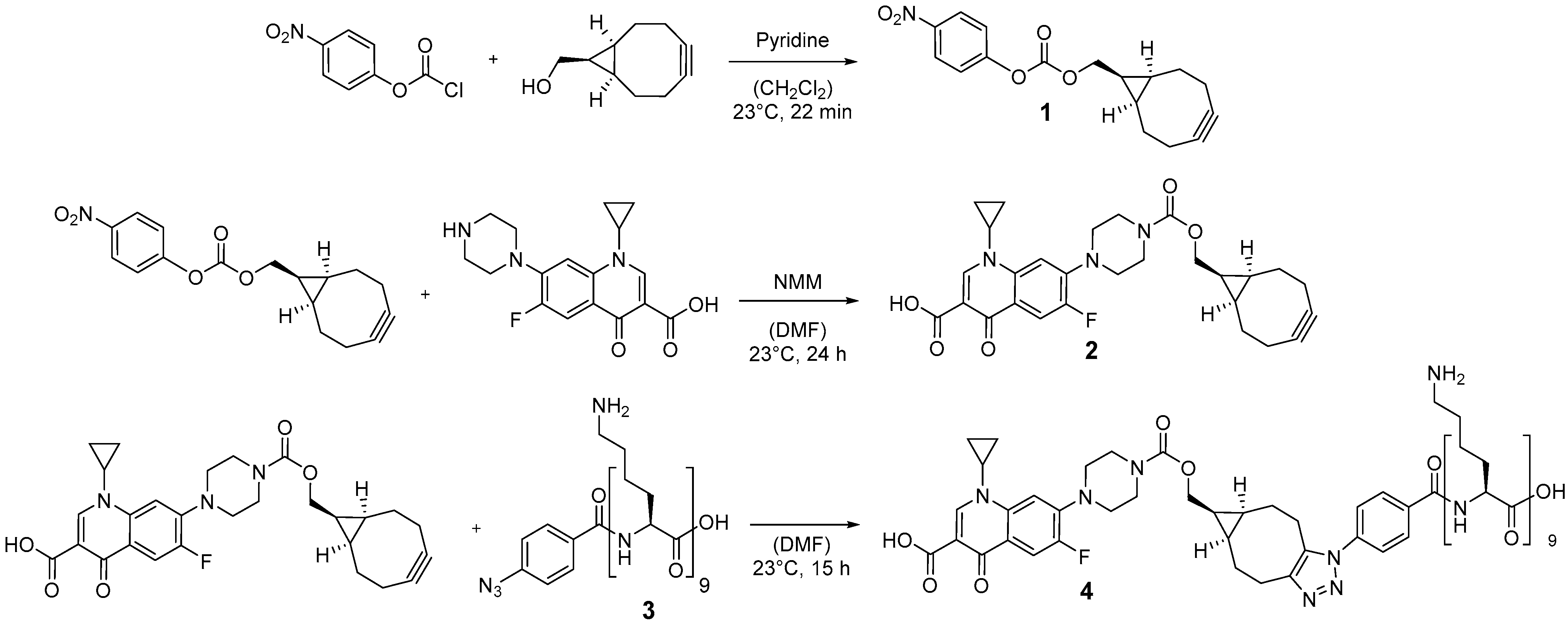
2.1. Conjugation of the Ciprofloxacin with Poly-l-Lysine
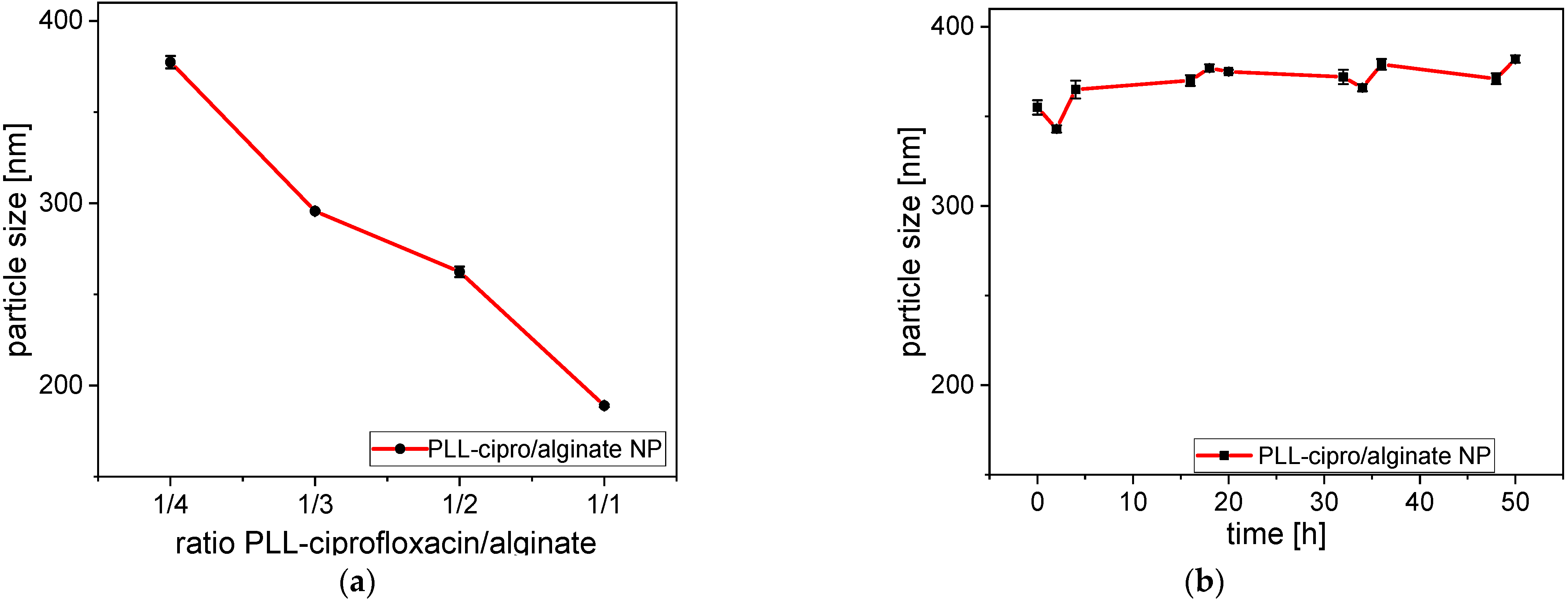
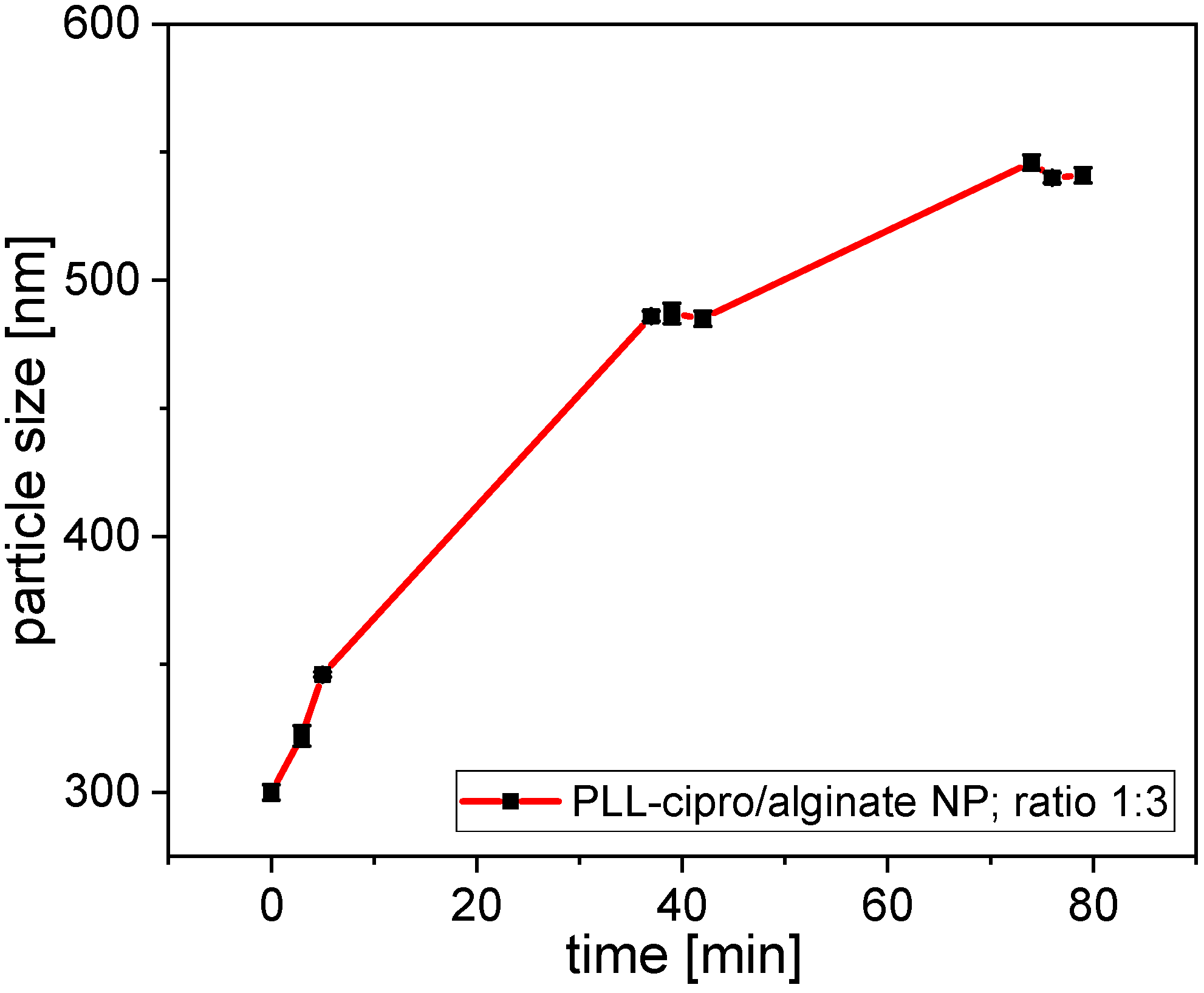
2.2. Nanoparticle Formation and Characterization
2.3. Coating of Titanium Samples
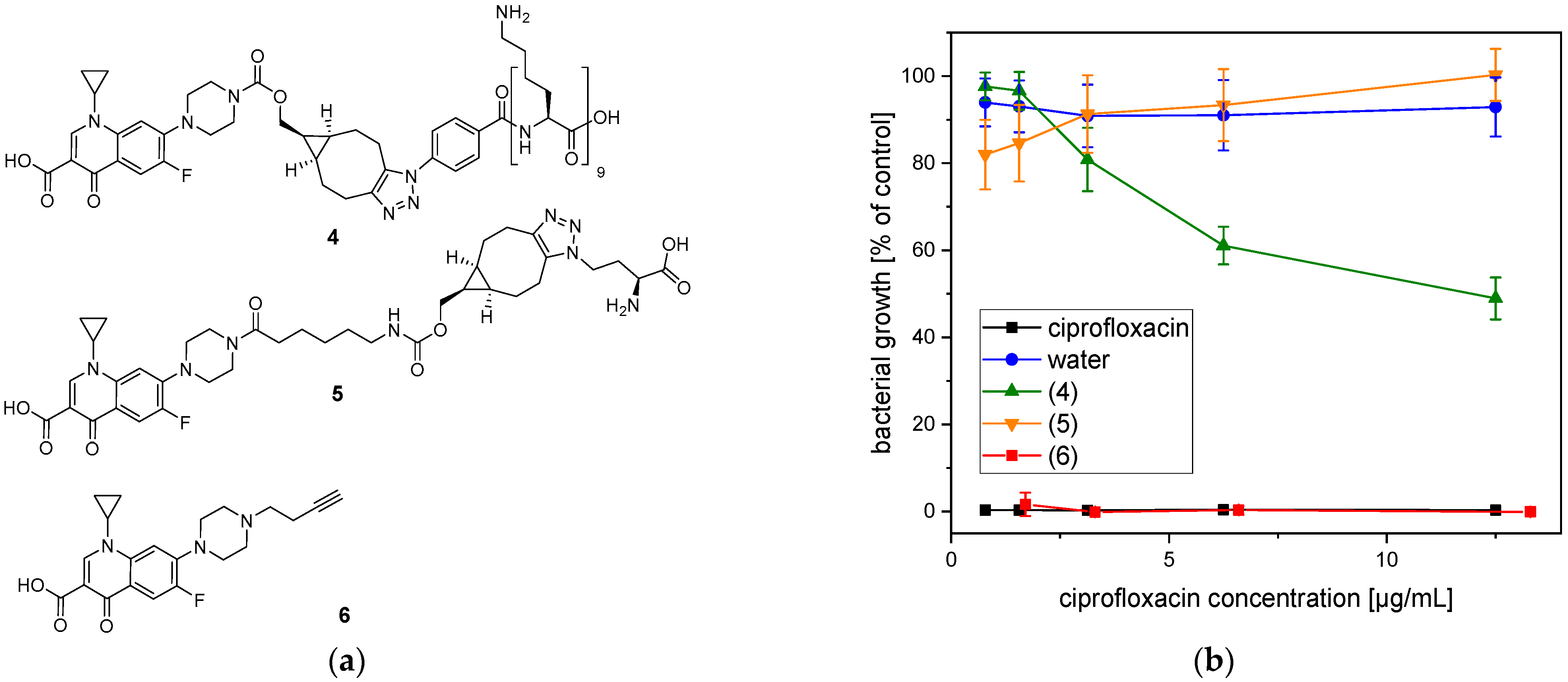
2.4. Antimicrobial Efficacy of the Conjugated Ciprofloxacin
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Materials
5.2. Synthesis of BCN-O(CO)O(4-NO2-Ph) 1
5.3. Synthesis of BCN-O(CO)HN-Ciprofloxacin 2
5.4. Synthesis of Poly-l-lysine-Ciprofloxacin (4)
5.5. Synthesis of 5
5.6. Synthesis of 6
5.7. Purification of Alginate
5.8. Preparation of Alginate/α-PLL-BCN-O(CO)HN-Ciprofloxacin Nanoparticles
5.9. Coating of Alginate/PLL-Ciprofloxacin Nanoparticles on Ti Plates
5.10. Stability of Nanoparticles in Aqueous Dispersion
5.11. Stability of Coatings on Titanium
5.12. Enzymatic Degradation of Nanoparticles and Coatings
5.13. Enzyme-Triggered Release
5.14. Antibacterial Efficacy of the Conjugated Ciprofloxacins
5.15. Spectroscopic Characterization
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jiskoot, W.; Randolph, T.W.; Volkin, D.B.; Middaugh, C.R.; Schöneich, C.; Winter, G.; Friess, W.; Crommelin, D.J.A.; Carpenter, J.F. Protein instability and immunogenicity: Roadblocks to clinical application of injectable protein delivery systems for sustained release. J. Pharm. Sci. 2012, 101, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 16071. [Google Scholar] [CrossRef]
- Varma, V.N.S.K.; Shivakumar, H.G.; Balamuralidhara, V.; Navya, M.; Hani, U. Development of pH Sensitive Nanoparticles for Intestinal Drug Delivery Using Chemically Modified Guar Gum Co-Polymer. Iran. J. Pharm. Res. 2016, 15, 83–94. [Google Scholar] [PubMed]
- Zhang, G.; Jiang, X. Temperature Responsive Nanoparticles Based on PEGylated Polyaspartamide Derivatives for Drug Delivery. Polymers 2019, 11, 316. [Google Scholar] [CrossRef]
- Xu, X.; Wu, J.; Liu, S.; Saw, P.E.; Tao, W.; Li, Y.; Krygsman, L.; Yegnasubramanian, S.; Marzo, A.M.d.; Shi, J.; et al. Redox-Responsive Nanoparticle-Mediated Systemic RNAi for Effective Cancer Therapy. Small 2018, 14, 1802565. [Google Scholar] [CrossRef]
- Ge, J.; Neofytou, E.; Cahill, T.J.; Beygui, R.E.; Zare, R.N. Drug release from electric-field-responsive nanoparticles. ACS Nano 2012, 6, 227–233. [Google Scholar] [CrossRef]
- McGill, S.L.; Cuylear, C.L.; Adolphi, N.L.; Osiński, M.; Smyth, H.D.C. Magnetically responsive nanoparticles for drug delivery applications using low magnetic field strengths. IEEE Trans. Nanobioscience 2009, 8, 33–42. [Google Scholar] [CrossRef]
- Hu, Q.; Katti, P.S.; Gu, Z. Enzyme-Responsive Nanomaterials for Controlled Drug Delivery. Nanoscale 2014, 6, 12273–12286. [Google Scholar] [CrossRef]
- Pye, A.D.; Lockhart, D.E.A.; Dawson, M.P.; Murray, C.A.; Smith, A.J. A review of dental implants and infection. J. Hosp. Infect. 2009, 72, 104–110. [Google Scholar] [CrossRef]
- Annibali, S.; Ripari, M.; La Monaca, G.; Tonoli, F.; Cristalli, M.P. Local complications in dental implant surgery: Prevention and treatment. Oral Implantol. 2008, 1, 21–33. [Google Scholar]
- Vickery, K.; Hu, H.; Jacombs, A.S.; Bradshaw, D.A.; Deva, A.K. A review of bacterial biofilms and their role in device-associated infection. Healthc. Infect. 2013, 18, 61–66. [Google Scholar] [CrossRef]
- Veerachamy, S.; Yarlagadda, T.; Manivasagam, G.; Yarlagadda, P. Bacterial adherence and biofilm formation on medical implants: A review. Proc. Inst. Mech. Eng. Part H 2014, 228, 1083–1099. [Google Scholar] [CrossRef]
- Garrett, T.R.; Bhakoo, M.; Zhang, Z. Bacterial adhesion and biofilms on surfaces. Prog. Nat. Sci. 2008, 18, 1049–1056. [Google Scholar] [CrossRef]
- Drenkard, E.; Ausubel, F.M. Pseudomonas biofilm formation and antibiotic resistance are linked to phenotypic variation. Nature 2002, 416, 740–743. [Google Scholar] [CrossRef]
- Yamamoto, H.; Amaike, M. Biodegradation of Cross-Linked Chitosan Gels by a Microorganism. Macromolecules 1997, 30, 3936–3937. [Google Scholar] [CrossRef]
- Zimmerli, W. Orthopaedic implant-associated infections: Update of antimicrobial therapy. Der Orthopade 2015, 44, 961–966. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Montanaro, L. Implant infections: Adhesion, biofilm formation and immune evasion. Nat. Rev. Microbiol. 2018, 16, 397–409. [Google Scholar] [CrossRef]
- Buchholz, H.W.; Elson, R.A.; Engelbrecht, E.; Lodenkamper, H.; Rottger, J.; Siegel, A. Management of deep infection of total hip replacement. J. Bone Jt. Surg. Br. 1981, 63-B, 342–353. [Google Scholar] [CrossRef]
- Simchi, A.; Tamjid, E.; Pishbin, F.; Boccaccini, A.R. Recent progress in inorganic and composite coatings with bactericidal capability for orthopaedic applications. Nanomedicine 2011, 7, 22–39. [Google Scholar] [CrossRef]
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as drug delivery systems. Pharmacol. Rep. 2012, 64, 1020–1037. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Yadav, S.K.; Yadav, S.C. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B 2010, 75, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Kabanov, A.V.; Vinogradov, S.V. Nanogels as pharmaceutical carriers: Finite networks of infinite capabilities. Angew. Chem. Int. Ed. Engl. 2009, 48, 5418–5429. [Google Scholar] [CrossRef]
- Kamaly, N.; Yameen, B.; Wu, J.; Farokhzad, O.C. Degradable Controlled-Release Polymers and Polymeric Nanoparticles: Mechanisms of Controlling Drug Release. Chem. Rev. 2016, 116, 2602–2663. [Google Scholar] [CrossRef]
- Magenheim, B.; Levy, M.Y.; Benita, S. A new in vitro technique for the evaluation of drug release profile from colloidal carriers—Ultrafiltration technique at low pressure. Int. J. Pharm. 1993, 94, 115–123. [Google Scholar] [CrossRef]
- Ganta, S.; Devalapally, H.; Shahiwala, A.; Amiji, M. A review of stimuli-responsive nanocarriers for drug and gene delivery. J. Control Release 2008, 126, 187–204. [Google Scholar] [CrossRef]
- Shahriari, M.; Zahiri, M.; Abnous, K.; Taghdisi, S.M.; Ramezani, M.; Alibolandi, M. Enzyme responsive drug delivery systems in cancer treatment. J. Control Release 2019, 308, 172–189. [Google Scholar] [CrossRef]
- Allen, T.M. Ligand-targeted therapeutics in anticancer therapy. Nat. Rev. Cancer 2002, 2, 750–763. [Google Scholar] [CrossRef] [PubMed]
- De la Rica, R.; Aili, D.; Stevens, M.M. Enzyme-responsive nanoparticles for drug release and diagnostics. Adv. Drug Deliv. Rev. 2012, 64, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Ungerleider, J.L.; Kammeyer, J.K.; Braden, R.L.; Christman, K.L.; Gianneschi, N.C. Enzyme-targeted nanoparticles for delivery to ischemic skeletal muscle. Polym. Chem. 2017, 8, 5212–5219. [Google Scholar] [CrossRef] [PubMed]
- Sarker, B. Advanced Hydrogels Concepts Based on Combinations of Alginate, Gelatin and Bioactive Glasses for Tissue Engineering. Ph.D. Thesis, Washington University, St. Louis, St. Louis, MI, USA, 2015. [Google Scholar]
- Tolle, C.; Riedel, J.; Mikolai, C.; Winkel, A.; Stiesch, M.; Wirth, D.; Menzel, H. Biocompatible Coatings from Smart Biopolymer Nanoparticles for Enzymatically Induced Drug Release. Biomolecules 2018, 8, 103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.-F.; Liu, X.; Zhang, S.; Pan, B.; Liu, M.-L. Ciprofloxacin derivatives and their antibacterial activities. Eur. J. Med. Chem. 2018, 146, 599–612. [Google Scholar] [CrossRef]
- Drlica, K.; Zhao, X. DNA gyrase, topoisomerase IV, and the 4-quinolones. Microbiol. Mol. Biol. Rev. 1997, 61, 377–392. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.B.; Patel, S.D.; Patel, J.N.; Patel, J.C.; Gorgamwala, Y.S. Synthesis and Antibacterial Activity of Thioureido Amide of Fluoroquinolone. Int. J. Biol. Chem. 2010, 5, 37–45. [Google Scholar] [CrossRef]
- Cormier, R.; Burda, W.N.; Harrington, L.; Edlinger, J.; Kodigepalli, K.M.; Thomas, J.; Kapolka, R.; Roma, G.; Anderson, B.E.; Turos, E.; et al. Studies on the antimicrobial properties of N-acylated ciprofloxacins. Bioorg. Med. Chem. Lett. 2012, 22, 6513–6520. [Google Scholar] [CrossRef]
- Vázquez, J.L.; Merino, S.; Doměnech, Ò.; Berlanga, M.; Viñas, M.; Montero, M.T.; Hernández-Borrell, J. Determination of the partition coefficients of a homologous series of ciprofloxacin: Influence of the N-4 piperazinyl alkylation on the antimicrobial activity. Int. J. Pharm. 2001, 220, 53–62. [Google Scholar] [CrossRef]
- Akeroyd, N. Click Chemistry for the Preparation of Advanced Macromolecular Architectures. Ph.D. Thesis, University of Stellenbosch, Stellenbosch, South Africa, 2010. [Google Scholar]
- Klahn, P.; Fetz, V.; Ritter, A.; Collisi, W.; Hinkelmann, B.; Arnold, T.; Tegge, W.; Rox, K.; Hüttel, S.; Mohr, K.I.; et al. The nuclear export inhibitor aminoratjadone is a potent effector in extracellular-targeted drug conjugates. Chem. Sci. 2019, 10, 5197–5210. [Google Scholar] [CrossRef] [PubMed]
- Vaezifar, S.; Razavi, S.; Golozar, M.A.; Karbasi, S.; Morshed, M.; Kamali, M. Effects of Some Parameters on Particle Size Distribution of Chitosan Nanoparticles Prepared by Ionic Gelation Method. J. Clust. Sci. 2013, 24, 891–903. [Google Scholar] [CrossRef]
- Giri, T.K.; Thakur, A.; Alexander, A.; Badwaik, H.; Tripathi, D.K. Modified chitosan hydrogels as drug delivery and tissue engineering systems: Present status and applications. Acta Pharm. Sin. B 2012, 2, 439–449. [Google Scholar] [CrossRef]
- Pistone, S.; Qoragllu, D.; Smistad, G.; Hiorth, M. Formulation and preparation of stable cross-linked alginate–zinc nanoparticles in the presence of a monovalent salt. Soft Matter 2015, 11, 5765–5774. [Google Scholar] [CrossRef]
- Poth, N.; Seiffart, V.; Gross, G.; Menzel, H.; Dempwolf, W. Biodegradable chitosan nanoparticle coatings on titanium for the delivery of BMP-2. Biomolecules 2015, 5, 3–19. [Google Scholar] [CrossRef]
- Sydow, S.; de Cassan, D.; Hänsch, R.; Gengenbach, T.R.; Easton, C.D.; Thissen, H.; Menzel, H. Layer-by-layer deposition of chitosan nanoparticles as drug-release coatings for PCL nanofibers. Biomater. Sci. 2018, 7, 233–246. [Google Scholar] [CrossRef]
- Rossi, J.-C.; Maret, B.; Vidot, K.; Francoia, J.-P.; Cangiotti, M.; Lucchi, S.; Coppola, C.; Ottaviani, M.F. Multi-technique characterization of poly-l-lysine dendrigrafts-Cu(II) complexes for biocatalysis. Macromol. Biosci. 2015, 15, 275–290. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.C.; McKay, C.S.; Legault, M.C.B.; Danielson, D.C.; Blake, J.A.; Pegoraro, A.F.; Stolow, A.; Mester, Z.; Pezacki, J.P. Cellular consequences of copper complexes used to catalyze bioorthogonal click reactions. J. Am. Chem. Soc. 2011, 133, 17993–18001. [Google Scholar] [CrossRef] [PubMed]
- Lallana, E.; Riguera, R.; Fernandez-Megia, E. Reliable and efficient procedures for the conjugation of biomolecules through Huisgen azide-alkyne cycloadditions. Angew. Chem. Int. Ed. 2011, 50, 8794–8804. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Miller, P.A.; Miller, M.J. Syntheses and Antibacterial Activity of N-Acylated Ciprofloxacin Derivatives Based on the Trimethyl Lock. ACS Med. Chem. Lett. 2015, 6, 707–710. [Google Scholar] [CrossRef]
- Ojkic, N.; Lilja, E.; Direito, S.; Dawson, A.; Allen, R.J.; Waclaw, B. A Roadblock-and-Kill Mechanism of Action Model for the DNA-Targeting Antibiotic Ciprofloxacin. Antimicrob. Agents Chemother. 2020, 64, e02487-19. [Google Scholar] [CrossRef]
- Zheng, M.; Pan, M.; Zhang, W.; Lin, H.; Wu, S.; Lu, C.; Tang, S.; Liu, D.; Cai, J. Poly(α-l-lysine)-based nanomaterials for versatile biomedical applications: Current advances and perspectives. Bioact. Mater. 2021, 6, 1878–1909. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bourgat, Y.; Mikolai, C.; Stiesch, M.; Klahn, P.; Menzel, H. Enzyme-Responsive Nanoparticles and Coatings Made from Alginate/Peptide Ciprofloxacin Conjugates as Drug Release System. Antibiotics 2021, 10, 653. https://doi.org/10.3390/antibiotics10060653
Bourgat Y, Mikolai C, Stiesch M, Klahn P, Menzel H. Enzyme-Responsive Nanoparticles and Coatings Made from Alginate/Peptide Ciprofloxacin Conjugates as Drug Release System. Antibiotics. 2021; 10(6):653. https://doi.org/10.3390/antibiotics10060653
Chicago/Turabian StyleBourgat, Yannick, Carina Mikolai, Meike Stiesch, Philipp Klahn, and Henning Menzel. 2021. "Enzyme-Responsive Nanoparticles and Coatings Made from Alginate/Peptide Ciprofloxacin Conjugates as Drug Release System" Antibiotics 10, no. 6: 653. https://doi.org/10.3390/antibiotics10060653
APA StyleBourgat, Y., Mikolai, C., Stiesch, M., Klahn, P., & Menzel, H. (2021). Enzyme-Responsive Nanoparticles and Coatings Made from Alginate/Peptide Ciprofloxacin Conjugates as Drug Release System. Antibiotics, 10(6), 653. https://doi.org/10.3390/antibiotics10060653






