Structural characterizations. NMR analyses were conducted on a Bruker DPX 500 NMR spectrometer, operating at 500 MHz and with tetramethylsilane as reference. High-resolution electrospray ionization mass spectra (HR ESI-MS) were performed by the ICOA/CBM (FR2708) at the University of Orléans with a Bruker Q-TOF maXis mass spectrometer coupled to an Ultimate 3000 RSLC chain (Dionex).
Photophysical characterizations. The photophysical characterizations were conducted in water (HPLC grade, supplier = Merck). Both UV-Vis absorption spectra and fluorescence emission spectra were recorded at 21 °C. UV–Vis absorption spectra were measured in quartz cuvettes (1 cm path-length, Hellma) on a Shimadzu UV2700 spectrophotometer. Fluorescence emission spectra were recorded on a SPEX Fluorolog 3 fluorometer. In this fluorometer, double grating monochromators are used in the excitation and emission channels. A Xenon arc lamp (450 W, Osram) is the excitation light source, and a Peltier cooled photomultiplier tube (R636-10, Hamamatsu) is the detector for the fluorescence. For the singlet oxygen emission at 1275 nm, a highly sensitive liquid nitrogen-cooled InGaAs detector (Electro-Optical Systems DSS series cryogenic receiver, 2 mm InGaAs photodiode) was coupled to a Horiba Jobin Yvon Spex Fluorolog 3 spectrofluorometer. The fluorescence signal from the fluorophores in solution is collected in a right-angle geometry, and the fluorescence spectra are corrected for fluctuations of the excitation source flux and for the wavelength dependence of the detection sensitivity. The optical density for photoluminescence spectra was approximately 0.1.
Both quantum yields were determined using the SPEX fluorolog 3 fluorometer relative to a fluorescent standard of known quantum yield. From here, the integrated emission spectra of the unknown sample are compared with that of the standard under the same absorbance conditions (optical density = 0.1) at the same excitation wavelength of 370 nm.
3.1. Synthesis of the Triazolium Salts
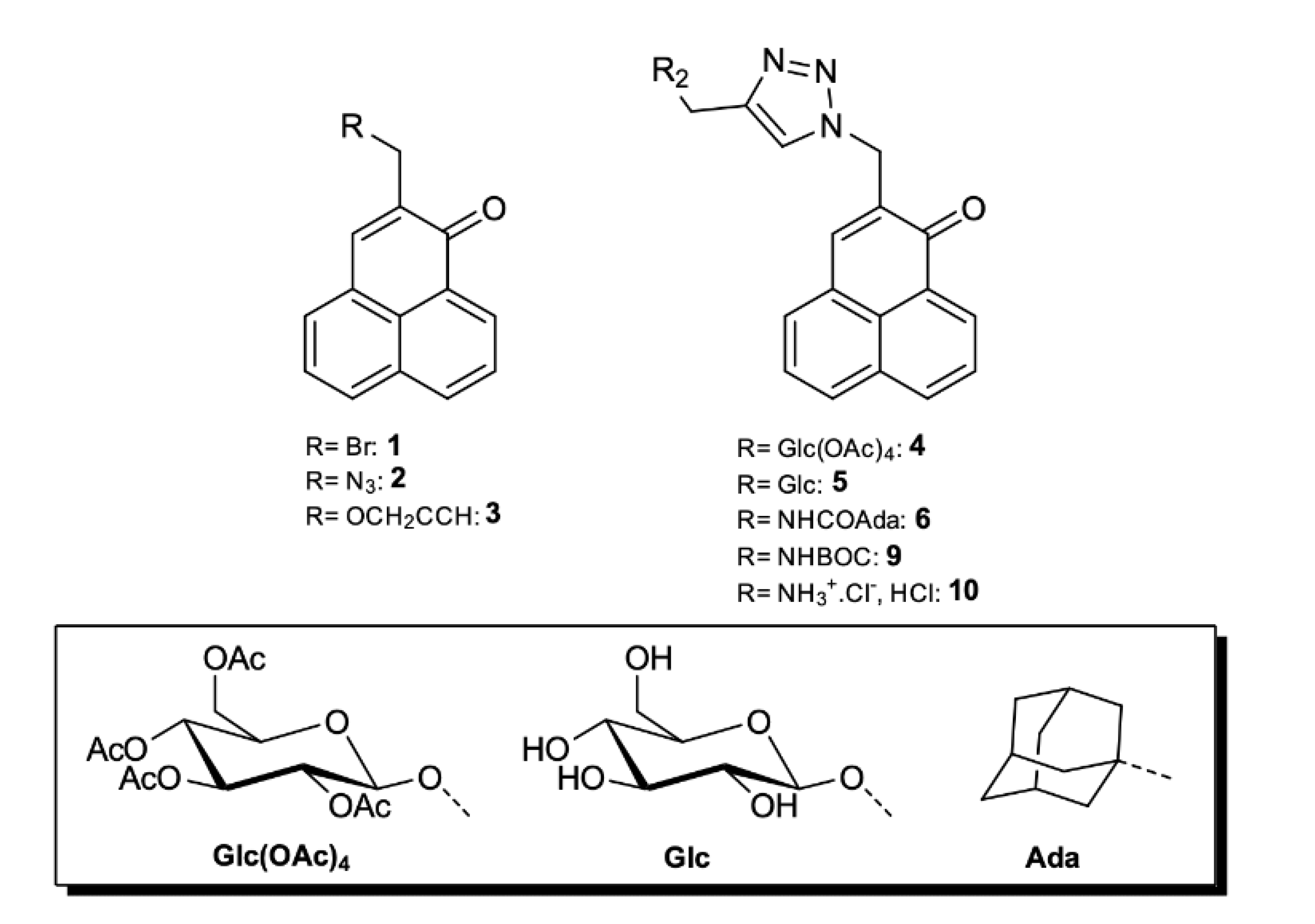
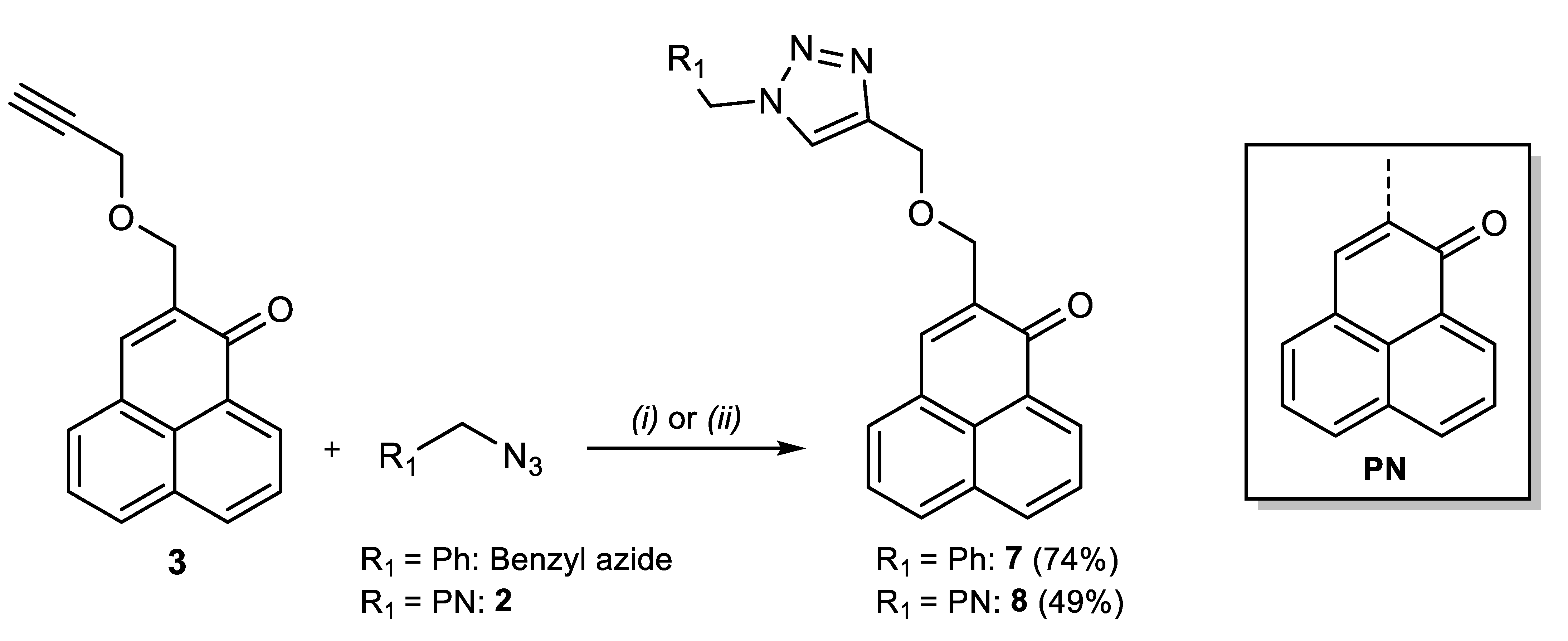
Synthesis of the triazole derivatives.2-(((1-benzyl-1H-1,2,3-triazol-4-yl)methoxy)methyl)-1H-phenalen-1-one (7). In total, 91 mg (0.37 mmol) of 3 and 100 µL (0.74 mmol) of benzyl azide were dissolved in 10 mL of CH2Cl2. 1.3 mg (7 µmol) of CuI, 3 µL (15 µmol) of DIPEA and 1 µL (7 µmol) of acetic acid was added, after which the reaction is stirred at room temperature for 55 h. The solvent was then evaporated, and the crude substance was purified by column chromatography (CH2Cl2 then CHCl3) to give 104 mg (0.27 mmol, 74%) of yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 8.64 (dd, J = 1.0, 7.4 Hz, 1H), 8.21 (dd, J = 0.6, 8.0 Hz, 1H), 8.01 (d, J = 8.2 Hz, 1H), 7.86 (m, 1H), 7.79 (t, J = 8 Hz, 1H), 7.76 (s, 1H), 7.59 (dd, J = 7.1, 8.5 Hz, 1H), 7.56 (s, 1H), 7.38–7.27 (m, 5H), 5.54 (s, 2H), 4.82 (s, 2H), 4.66 (d, J = 1.2, 2H)—13C-NMR (125 MHz, CDCl3): δ (ppm) = 184.42, 145.44, 138.08, 136.25, 134.95, 134.56, 132.03, 131.53, 131.51, 131.49, 130.42, 129.20, 129.13, 128.91, 128.77, 128.17, 127.89, 127.61, 127.08, 126.80, 122.55, 67.35, 64.62, 54.21.—MS: HRMS (ESI+), calcd for C24H20N3O2 [M + H]+: 382.155003, found 382.154573.—MW: 381.44 g/mol.—MP: 122 °C.
2-((4-(((1-oxo-1H-phenalen-2-yl)methoxy)methyl)-1H-1,2,3-triazol-1-yl)methyl)-1H-phenalen-1-one (8). In total, 235 mg (1 mmol) of 2 and 248 mg (1 mmol) of 3, 250 mg (0.2 mmol) of CuSO4.5H2O, and 198 mg (0.4 mmol) of sodium ascorbate were dissolved in 10 mL of tBuOH/H2O 1:1. A precipitate rapidly appears. The reaction was stirred 72 h at room temperature. The solution was poured in 300 mL of water, filtered, and the solid was extracted several times with CH2Cl2. The organic phase was dried and evaporated. The crude substance was purified on column chromatography (CHCl3/MeOH 98:2) to yield 236 mg (0.49 mmol, 49%) of bright yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 8.67 (dd, J = 0.9, 7.4 Hz, 1H), 8.62 (dd, J = 0.9, 7.5 Hz, 1H), 8.24 (d, J = 7.7 Hz, 1H), 8.19 (d, J = 7.8 Hz, 1H), 8.05 (d, J = 8.2 Hz, 1H), 7.99 (d, J = 8.3 Hz, 1H), 7.94 (s, 1H), 7.87 (t, J = 1.0 Hz, 1H), 7.80 (t, J = 7.7 Hz, 1H), 7.78–7.74 (m, 3H), 7.74 (s, 1H), 7.61 (dd, J = 7.2, 8.1 Hz, 1H), 7.58 (dd, J = 7.2, 8.1 Hz, 1H), 5.60 (s, 2H), 4.85 (s, 2H), 4.69 (d, J = 1.1 Hz, 2H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 184.40, 183.89, 145.18, 141.21, 137.93, 136.34, 135.53, 134.90, 133.53, 132.71, 132.65, 132.04, 132.01, 131.51, 131.43, 131.06, 130.38, 129.20, 128.85, 127.65, 127.35, 127.34, 127.07, 127.04, 126.91, 126.84, 126.78, 124.00, 67.30, 64.61, 48.89.—MS: HRMS (ESI+), calcd for C31H22N3O3 [M + H]+: 484.165568, found 484.165628.—MW: 483.53 g/mol.—MP: 125 °C (deg.).
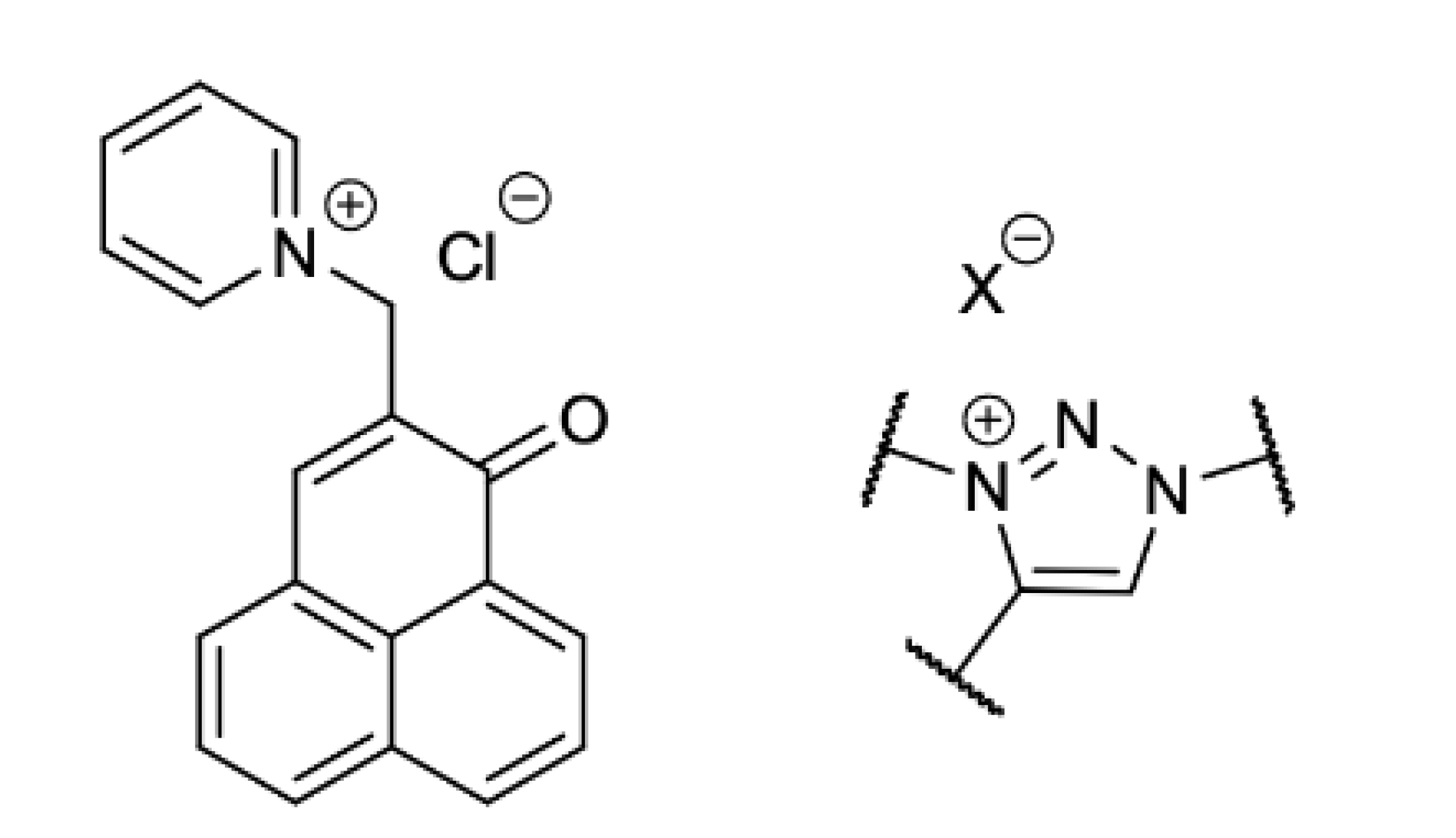
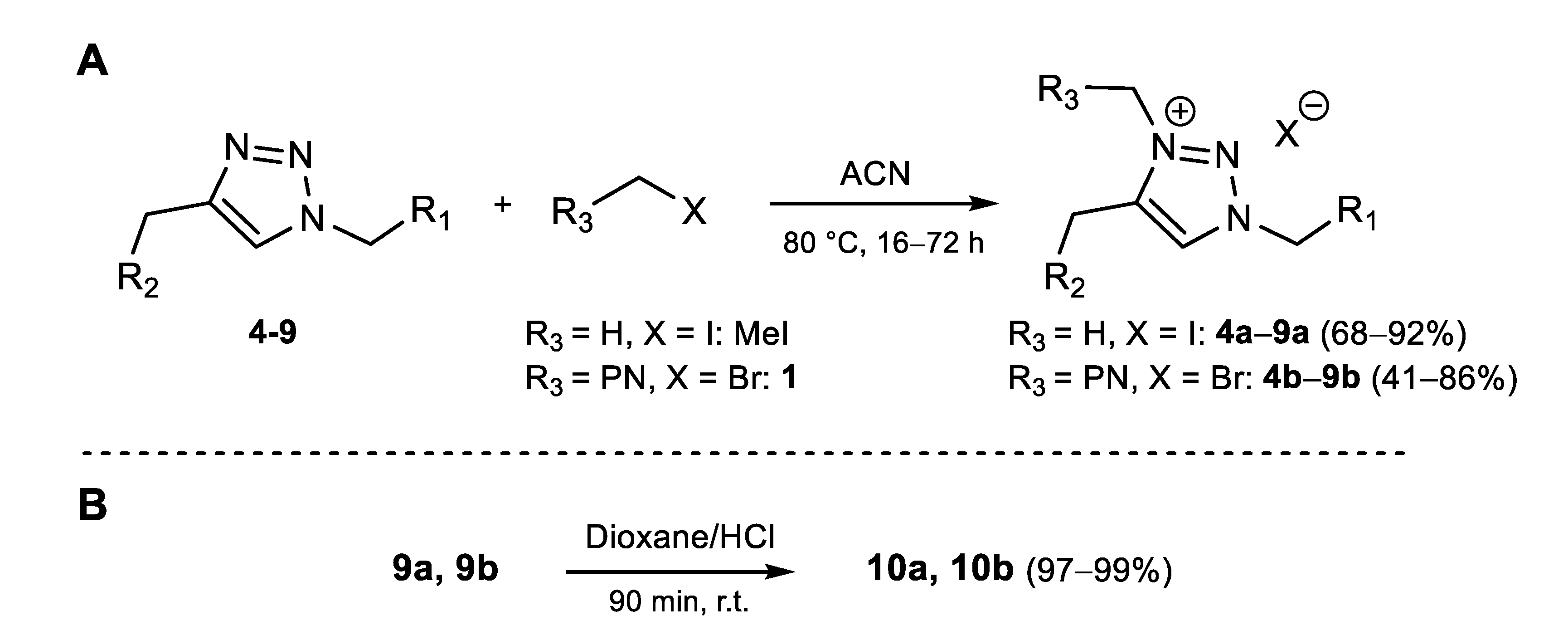
General procedure for the methylation of the triazoles. A total of 1 eq. of triazole compound and 20 eq. of methyl iodide were dissolved in the minimum amount of acetonitrile. The reaction was then left at 80 °C for 16 to 24 h. The solvent was evaporated, and the crude substance was purified by column chromatography (CHCl3/MeOH 98:2 to 95:5), except for 5a, and then crystallized in diethyl ether.
3-methyl-1-((1-oxo-1H-phenalen-2-yl)methyl)-4-((((2R,3R,4S,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)oxy)methyl)-1H-1,2,3-triazol-3-ium iodide (4a). A total of 350 mg (0.56 mmol) of 4 gave 362 mg (0.47 mmol, 85%) of orange yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 9.20 (s, 1H), 8.60 (d, J = 7.3 Hz, 1H), 8.49 (s, 1H), 8.26 (d, J = 8.0 Hz, 1H), 8.17 (d, J = 7.0 Hz, 1H), 8.11 (d, J = 8.3 Hz, 1H), 7.80 (t, J = 7.7 Hz, 1H), 7.68 (t, J = 7.7 Hz, 1H), 5.87 (d, J = 14.5 Hz, 1H), 5.83 (d, J = 14.5 Hz, 1H), 5.40 (d, J = 14.4 Hz, 1H), 5.23 (d, J = 14.3 Hz, 1H), 5.22 (t, J = 9.3 Hz, 1H), 5.07 (m, 2H), 4.98 (m, 1H), 4.34 (m, 4H), 4.07 (m, 2H), 2.04 (s, 3H), 2.01 (s, 6H), 1.98 (s, 3H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.45, 170.48, 169.89, 169.63, 169.52, 144.64, 140.52, 136.08, 134.62, 133.68, 132.05, 131.26, 131.24, 129.28, 128.54, 127.58, 127.45, 127.29, 126.25, 100.66, 72.70, 72.13, 71.04, 67.78, 61.07, 60.11, 53.31, 39.39, 20.86, 20.80, 20.60, 20.54.—MS: HRMS (ESI+), calcd for C32H34N3O11 [M]+: 636.218785, found 636.218796.—MW: 763.54 g/mol.
3-methyl-1-((1-oxo-1H-phenalen-2-yl)methyl)-4-((((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)methyl)-1H-1,2,3-triazol-3-ium iodide (5a). A total of 90 mg (0.20 mmol) of 5 gave 95 mg (0.16 mmol, 80%) of very hygroscopic yellow powder. The product is stocked in 10 mM aqueous solution at 4 °C until using. 1H-NMR (500 MHz, D2O): δ (ppm) = 8.76 (s, 1H), 8.22 (m, 2H), 8.10 (d, J = 8.3 Hz, 1H), 8.00 (s, 1H), 7.84 (d, J = 7.1 Hz, 1H), 7.69 (t, J = 7.7 Hz, 1H), 7.64 (t, J = 7.7 Hz, 1H), 5.63 (s, 2H), 5.21 (d, J = 14.5 Hz, 1H), 5.15 (d, J = 14.5 Hz, 1H), 4.61 (d, J = 7.9 Hz, 1H), 4.36 (s, 3H), 3.76 (dd, J = 1.7, 12.3 Hz, 1H), 3.60 (dd, J = 5.2, 12.3 Hz, 1H), 3.52 (t, J = 9.0 Hz, 1H), 3.40 (m, 2H), 3.36 (dd, J = 8.0, 9.3 Hz, 1H).—13C-NMR (125 MHz, D2O): δ (ppm) = 184.51, 145.76, 140.01, 137.21, 135.23, 134.59, 131.41, 131.32, 130.14, 128.71, 127.41, 127.26, 127.15, 126.01, 125.26, 102.23, 76.03, 75.56, 72.82, 69.33, 60.43, 58.80, 52.78, 38.20.—MS: HRMS (ESI+), calcd for C24H26N3O7 [M + H]+: 468.176527, found 468.176445.—MW: 595.39 g/mol.
4-(((1r,3r)-adamantane-1-carboxamido)methyl)-3-methyl-1-((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium iodide (6a). A total of 41 mg (90 µmol) of 6 gave 38 mg (63 µmol, 70%) of bright yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 8.97 (s, 1H), 8.59 (d, J = 7.3 Hz, 1H), 8.26 (s, 1H), 8.25 (d, J = 8 Hz, 1H), 8.11 (m, 3H), 7.80 (t, J = 7.7 Hz, 1H), 7.67 (t, J = 7.6 Hz, 1H), 5.72 (s, 2H), 4.75 (d, J = 5.3 Hz, 2H), 4.43 (s, 3H), 1.99 (s, 3H), 1.87 (s, 6H), 1.68 (s, 6H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.24, 179.80, 144.13, 142.90, 136.06, 134.32, 133.73, 132.02, 131.40, 131.09, 129.37, 128.47, 127.52, 127.51, 127.16, 126.17, 53.23, 40.79, 39.23, 39.06 (3C), 36.37 (3C), 32.28, 28.03 (3C).—MS: HRMS (ESI+), calcd for C29H31N4O2 [M]+: 467.244153, found 467.243968.—MW: 594.50 g/mol.
1-benzyl-3-methyl-4-(((1-oxo-1H-phenalen-2-yl)methoxy)methyl)-1H-1,2,3-triazol-3-ium iodide (7a). A total of 270 mg (0.71 mmol) of 7 gave 339 mg (0.65 mmol, 92%) of yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 9.55 (s, 1H), 8.56 (d, J = 7.2 Hz, 1H), 8.20 (d, J = 8.0 Hz, 1H), 8.03 (d, J = 7.2 Hz, 1H), 8.01 (s, 1H), 7.97 (d, J = 7.0 Hz, 1H), 7.77 (t, J = 7.7 Hz, 1H), 7.63 (t, J = 7.7 Hz, 1H), 7.59 (m, 2H), 7.40 (m, 3H), 5.92 (s, 2H), 5.08 (s, 2H), 4.64 (s, 2H), 4.39 (s, 3H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 184.41, 141.69, 140.60, 135.28, 134.00, 132.91, 132.33, 132.01, 131.05, 130.72, 130.56, 130.06, 129.61 (2C), 129.53 (2C), 129.06, 127.39, 127.14, 127.04, 126.99, 69.11, 60.69, 57.60, 39.41.—MS: HRMS (ESI+), calcd for C25H22N3O2 [M]+: 396.170653, found 396.170530.—MW: 523.37 g/mol.
3-methyl-4-(((1-oxo-1H-phenalen-2-yl)methoxy)methyl)-1-((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium iodide (8a). A total of 70 mg (0.14 mmol) of 8 gave 82 mg (0.13 mmol, 91%) of yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 9.36 (s, 1H), 8.62 (s, 1H), 8.50 (d, J = 7.5 Hz, 1H), 8.46 (d, J = 7.5 Hz, 1H), 8.16 (m, 3H), 8.03 (d, J = 8.2 Hz, 1H), 7.97 (d, J = 7.0 Hz, 1H), 7.96 (s, 1H), 7.91 (d, J = 7.0 Hz, 1H), 7.71 (t, J = 7.8 Hz, 1H), 7.69 (t, J = 7.8 Hz, 1H), 7.62 (t, J = 7.6 Hz, 1H), 7.58 (t, J = 7.6 Hz, 1H), 5.91 (s, 2H), 5.10 (s, 2H), 4.65 (s, 2H), 4.41 (s, 3H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 184.36, 183.54, 145.14, 141.43, 140.56, 135.92, 135.20, 134.70, 134.07, 133.51, 132.76, 132.26, 131.97, 131.96, 131.11, 130.91, 130.56, 129.16, 129.00, 128.54, 127.54, 127.32, 127.30, 127.26, 127.11, 126.99, 126.97, 126.29, 69.04, 60.76, 53.08, 39.41.—MS: HRMS (ESI+), calcd for C32H24N3O3 [M]+: 498.181218, found 498.180972.—MW: 625.47 g/mol.
4-(((tert-butoxycarbonyl)amino)methyl)-3-methyl-1-((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium iodide (9a). A total of 539 mg (1.38 mmol) of 9 gave 501 mg (0.94 mmol, 68%) of orange yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 8.94 (s, 1H), 8.60 (d, J = 7.3 Hz, 1H), 8.39 (s, 1H), 8.25 (d, J = 7.8 Hz, 1H), 8.14 (d, J = 6.9 Hz, 1H), 8.11 (d, J = 8.2 Hz, 1H), 7.80 (t, J = 7.7 Hz, 1H), 7.67 (dd, J = 7.4, 8.1 Hz, 1H), 6.40 (t, J = 5.4 Hz, 1H), 5.78 (s, 2H), 4.73 (d, J = 6.1 Hz, 2H), 4.41 (s, 3H), 1.40 (s, 9H). —13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.35, 156.06, 144.37, 143.07, 136.05, 134.54, 133.67, 132.02, 131.32, 130.87, 129.27, 128.50, 127.55, 127.44, 127.23, 126.21, 80.78, 53.15, 39.29, 34.16, 28.28 (3C).—MS: HRMS (ESI+), calcd for C23H25N4O3 [M]+: 405.192117, found 405.192461.—MW: 532.38 g/mol.
General procedure for the “phenalenomethylation” of the triazoles. A total of 1 eq. of the PN-triazole and 2 eq. of 1 are dissolved in the minimum amount of acetonitrile. The reaction remained at 80 °C for 48 to 72 h. The solvent was evaporated, and the crude substance was purified by column chromatography (CHCl3/MeOH 98:2 to 95:5), except for 5b, then crystallized in diethyl ether.
1,3-bis((1-oxo-1H-phenalen-2-yl)methyl)-4-((((2R,3R,4S,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)oxy)methyl)-1H-1,2,3-triazol-3-ium bromide (4b). A total of 350 mg (0.56 mmol) of 4 gave 335 mg (0.37 mmol, 67%) of yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 9.22 (s, 1H), 8.58 (dd, J = 1.0, 7.3 Hz, 1H), 8.57 (dd, J = 1.0, 7.3 Hz, 1H), 8.51 (s, 1H), 8.31 (s, 1H), 8.28 (d, J = 8.0 Hz, 1H), 8.25 (d, J = 8.0 Hz, 1H), 8.15 (m, 2H), 8.10 (m, 2H), 7.82 (t, J = 7.6 Hz, 1H), 7.79 (t, J = 7.6 Hz, 1H), 7.70 (dd, J = 7.3, 8.0 Hz, 1H), 7.67 (dd, J = 7.3, 8.0 Hz, 1H), 5.88 (s, 2H), 5.83 (s, 2H), 5.50 (d, J = 14.4 Hz, 1H), 5.41 (d, J = 14.4 Hz, 1H), 5.23 (t, J = 9.4 Hz, 1H), 5.05 (m, 3H), 4.21 (m, 1H), 4.01 (m, 2H), 2.02 (s, 3H), 2.00 (s, 3H), 1.97 (s, 3H), 1.92 (s, 3H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.42, 183.33, 170.45, 169.92, 169.64, 169.51, 144.55, 144.13, 141.19, 135.98, 135.90, 134.57, 134.24, 133.50, 133.44, 132.08, 132.02, 131.22, 131.13, 130.92, 129.64, 129.63, 128.60, 128.57, 127.54 (2C), 127.46, 127.33, 127.26, 127.22, 126.42 (2C), 100.76, 72.77, 72.11, 71.08, 67.90, 61.14, 60.40, 53.20, 20.74, 20.69, 20.59, 20.56.—MS: HRMS (ESI+), calcd for C45H40N3O12 [M]+: 814.260650, found 814.260689. —MW: 894.73 g/mol.
1,3-bis((1-oxo-1H-phenalen-2-yl)methyl)-4-((((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)methyl)-1H-1,2,3-triazol-3-ium bromide (5b). A total of 138 mg (0.30 mmol) of 5 gave 104 mg (0.14 mmol, 48%) of very hygroscopic orange yellow powder. The product is stocked in 10 mM aqueous solution at 4 °C until using. 1H-NMR (500 MHz, D2O): δ (ppm) = 9.09 (s, 1H), 8.52 (d, J = 8.2 Hz, 2H), 8.44 (d, J = 7.4 Hz, 2H), 8.34 (d, J = 8.2 Hz, 2H), 8.23 (s, 2H), 8.09 (m, 2H), 7.91 (t, J = 7.7 Hz, 2H), 7.80 (t, J = 7.7 Hz, 1H), 7.79 (t, J = 7.7 Hz, 1H), 5.80 (s, 2H), 5.79 (s, 2H), 5.28 (d, J = 14.1 Hz, 1H), 5.22 (d, J = 14.1 Hz, 1H), 4.44 (d, J = 7.8 Hz, 1H), 3.66 (d, J = 1.4, 11.7 Hz, 1H), 3.42 (dd, J = 6.2, 11.7 Hz, 1H), 3.17 (m, 2H), 3.06 (m, 2H).—13C-NMR (125 MHz, D2O): δ (ppm) = 182.50, 182.48, 143.48, 143.00, 140.87, 136.06 (2C), 133.69, 133.62, 133.33, 133.25, 131.60 (2C), 130.52, 130.40 (2C), 130.35, 130.20, 127.86, 127.85, 127.54, 127.53, 127.30, 127.26, 126.46, 126.40, 125.89, 125.84, 102.78, 77.02, 76.39, 73.09, 69.76, 60.89, 58.55, 52.63, 50.71.—MS: HRMS (ESI+), calcd for C37H32N3O8 [M]+: 646.218391, found 646.217763.—MW: 726.58 g/mol.
4-(((3r,5r,7r)-adamantane-1-carboxamido)methyl)-1,3-bis((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium bromide (6b). A total of 172 mg (0.38 mmol) of 6 gave 113 mg (0.16 mmol, 41%) of bright yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 8.93 (s, 1H), 8.54 (t, J = 7.0 Hz, 1H), 8.54 (t, J = 7.0 Hz, 1H), 8.45 (t, J = 5.7 Hz, 1H), 8.31 (s, 1H), 8.23 (t, J = 8.0 Hz, 1H), 8.23 (t, J = 8.0 Hz, 1H), 8.18 (s, 1H), 8.08 (dd, J = 7.1, 8.0 Hz, 1H), 8.08 (dd, J = 7.1, 8.0 Hz, 1H), 8.04 (d, J = 7.0 Hz, 1H), 8.04 (d, J = 7.0 Hz, 1H), 7.77 (t, J = 7.7 Hz, 1H), 7.76 (t, J = 7.7 Hz, 1H), 7.65 (dd, J = 7.4, 8.3 Hz, 1H), 7.63 (dd, J = 7.4, 8.3 Hz, 1H), 6.03 (s, 2H), 5.72 (s, 2H), 4.96 (d, J = 5.8 Hz, 2H), 1.93 (s, 3H), 1.84 (s, 6H), 1.64 (m, 6H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.33, 183.08, 179.70, 143.72, 143.58, 143.53, 135.89, 135.77, 134.20, 134.02, 133.49, 133.28, 131.99, 131.95, 131.24, 131.09, 131.04, 130.13, 129.66, 128.61, 128.43, 127.44, 127.41, 127.38, 127.37, 127.12, 127.10, 126.43, 126.24, 53.06, 51.90, 40.75, 38.90 (3C), 36.38 (3C), 32.37, 28.03 (3C).—MS: HRMS (ESI+), calcd for C42H37N4O3 [M]+: 645.286017, found 645.286100.—MW: 725.69 g/mol.
1-benzyl-4-(((1-oxo-1H-phenalen-2-yl)methoxy)methyl)-3-((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium bromide (7b). A total of 49 mg (0.13 mmol) of 7 gave 72 mg (0.11 mmol, 86%) of yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 9.79 (s, 1H), 8.51 (t, J = 8.0 Hz, 2H), 8.17 (d, J = 7.0 Hz, 1H), 8.16 (s, 1H), 8.11 (d, J = 7.9 Hz, 1H), 8.00 (m, 3H), 7.84 (s, 1H), 7.77 (d, J = 7.0 Hz, 1H), 7.73 (t, J = 7.7 Hz, 1H), 7.69 (t, J = 7.7 Hz, 1H), 7.62 (m, 2H), 7.58 (t, J = 7.7 Hz, 1H), 7.58 (t, J = 7.7 Hz, 1H), 7.39 (m, 3H), 6.04 (s, 2H), 5.89 (s, 2H), 5.16 (s, 2H), 4.63 (s, 2H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 184.21, 183.08, 143.18, 141.03, 140.74, 135.71, 135.06, 134.14, 133.94, 133.28, 132.57, 132.03, 131.91, 131.89, 131.40, 131.14, 130.97, 130.47, 129.92, 129.81, 129.61 (2C), 129.47 (2C), 128.98, 128.44, 127.31, 127.30, 127.22, 127.05, 127.02, 127.02, 126.92, 126.25, 68.90, 60.61, 57.70, 51.83.—MS: HRMS (ESI+), calcd for C38H28N3O3 [M]+: 574.212518, found 574.212562.—MW: 654.56 g/mol.
4-(((1-oxo-1H-phenalen-2-yl)methoxy)methyl)-1,3-bis((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium bromide (8b). A total of 202 mg (0.42 mmol) of 8 gave 256 mg (0.34 mmol, 81%) of bright yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 9.45 (s, 1H), 8.73 (s, 1H), 8.54 (dd, J = 0.8, 7.5 Hz, 1H), 8.41 (dd, J = 0.8, 7.5 Hz, 1H), 8.38 (dd, J = 0.8, 7.5 Hz, 1H), 8.20 (d, J = 8.1 Hz, 1H), 8.18 (s, 1H), 8.12 (m, 2H), 8.05 (d, J = 8.2 Hz, 1H), 8.03 (d, J = 7.2 Hz, 1H), 7.98 (d, J = 8.1 Hz, 1H), 7.92 (d, J = 8.2 Hz, 1H), 7.87 (d, J = 8.2 Hz, 1H), 7.74 (t, J = 7.7 Hz, 1H), 7.70 (s, 1H), 7.66 (t, J = 7.6 Hz, 1H), 7.63 (dd, J = 7.4, 8.2 Hz, 1H), 7.62 (m, 1H), 7.57 (t, J = 7.7 Hz, 1H), 7.54 (dd, J = 7.4, 8.1 Hz, 1H), 7.50 (dd, J = 7.4, 8.1 Hz, 1H), 6.06 (s, 2H), 5.92 (s, 2H), 5.18 (s, 2H), 4.61 (s, 2H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.94, 183.65, 183.05, 145.53, 142.86, 140.77, 139.89, 135.77, 135.48, 134.88, 134.64, 134.33, 134.10, 133.33, 133.01, 132.23, 131.97, 131.78, 131.74, 130.99, 130.97, 130.93, 130.34, 130.06, 129.50, 128.80, 128.65, 128.34, 127.55, 127.22 (2C), 127.16, 127.03 (3C), 126.98, 126.97, 126.91, 126.78, 126.48, 126.33, 68.64, 60.84, 53.33, 51.44.—MS: HRMS (ESI+), calcd for C45H30N3O4 [M]+: 676.223083, found 676.222808.—MW: 756.66 g/mol.
4-(((tert-butoxycarbonyl)amino)methyl)-1,3-bis((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium bromide (9b). A total of 460 mg (1.18 mmol) of 9 gave 658 mg (0.99 mmol, 84%) of yellow powder. 1H-NMR (500 MHz, CDCl3): δ (ppm) = 8.92 (s, 1H), 8.55 (dd, J = 1.1, 7.3 Hz, 1H), 8.54 (d, J = 7.3 Hz, 1H), 8.33 (s, 1H), 8.30(s, 1H), 8.24 (dd, J = 0.9, 8.1 Hz, 1H), 8.21 (d, J = 8.4 Hz, 1H), 8.09 (m, 4H), 7.78 (t, J = 7.8 Hz, 1H), 7.76 (t, J = 7.8 Hz, 1H), 7.66 (dd, J = 7.2, 8.1 Hz, 1H), 7.64 (dd, J = 7.2, 8.1 Hz, 1H), 6.84 (s, 1H), 5.97 (s, 2H), 5.79 (s, 2H), 4.89 (d, J = 6.0 Hz, 2H), 1.32 (s, 9H).—13C-NMR (125 MHz, CDCl3): δ (ppm) = 183.37, 183.28, 156.17, 144.02, 143.77, 135.88, 135.81, 134.45, 134.16, 133.44, 133.29, 131.98, 131.93, 131.16, 131.13, 130.65, 129.94, 129.56, 128.58, 128.46, 127.43, 127.34, 127.31, 127.17 (2C), 127.16 (2C), 126.44, 126.30, 80.39, 53.08, 51.65, 34.19, 28.22 (3C).—MS: HRMS (ESI+), calcd for C36H31N4O4 [M]+: 583.233982, found 583.233983.—MW: 663.57 g/mol.
Deprotection of BOC derivatives. Hydrolysis of BOC moiety was carried out by dissolving BOC derivatives in dioxane-HCl 37% mixture (10:1 v/v) and stirring the resulting solution for 90 min at room temperature. The solvents were evaporated, and the crude substance was repeatedly dissolved in a minimum amount of methanol, crystallized in diethyl ether and evaporated until obtaining a dry powder.
4-(ammoniomethyl)-3-methyl-1-((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium chloride (10a). A total of 100 mg (0.19 mmol) of 9a gave 70 mg (0.19 mmol, 99%) of orange yellow powder. 1H-NMR (500 MHz, DMSO-d6): δ (ppm) = 9.13 (s, 1H), 9.06 (s, 2H), 8.56 (d, J = 8.4 Hz, 1H), 8,55 (d, J = 7.3 Hz, 1H), 8.37 (d, J = 8.3 Hz, 1H), 8.35 (s, 1H), 8.22 (d, J = 7.0 Hz, 1H), 7.97 (t, J = 7.7 Hz, 1H), 7.83 (t, J = 7.7 Hz, 1H), 5.91 (s, 2H), 4.42 (s, 2H), 4,41 (s, 3H).—13C-NMR (125 MHz, DMSO-d6): δ (ppm) = 182.46, 143.13, 137.47, 136.25, 133.96, 133.45, 131.72 (2C), 130.56 (2C), 127.93, 127.69, 127.44, 126.52, 125.95, 52.41, 38.72, 30.60.—MS: HRMS (ESI+), calcd for C18H17N4O [M − H]+: 305.139688, found 305.139596.—MW: 377.27 g/mol.
4-(ammoniomethyl)-1,3-bis((1-oxo-1H-phenalen-2-yl)methyl)-1H-1,2,3-triazol-3-ium chloride (10b). A total of 144 mg (0.22 mmol) of 9b gave 117 mg (0.21 mmol, 97%) of orange yellow powder. 1H-NMR (500 MHz, D2O): δ (ppm) = 9.11 (s, 1H), 7.88 (d, J = 4.7 Hz, 1H), 7.73 (m, 1H), 7.63 (m, 1H), 7.59 (m, 1H), 7.57 (m, 1H), 7.53 (m, 1H), 7.52 (m, 1H), 7.46 (m, 1H), 7.37 (m, 1H), 7.20 (m, 1H), 7.13 (m, 2H), 7.06 (m, 2H), 5.63 (s, 2H), 5.54 (s, 2H), 4.96 (s, 2H).—13C-NMR (125 MHz, D2O): δ (ppm) = 186.79, 186.49, 148.63, 147.91, 139.46, 139.20, 139.12, 137.42, 137.09, 136.81, 136.56, 134.15, 133.50, 133.12 (2C), 133.02, 131.42, 131.05, 129.40, 129.30, 129.23, 129.07, 129.01, 128.83, 127.91, 127.65, 127.20, 127.00, 56.29, 54.22, 34.11.—MS: HRMS (ESI+), calcd for C31H23N4O2 [M − H]+: 483.181552, found 483.181547.—MW: 555.13 g/mol.