Novel Seleno- and Thio-Urea Containing Dihydropyrrol-2-One Analogues as Antibacterial Agents
Abstract
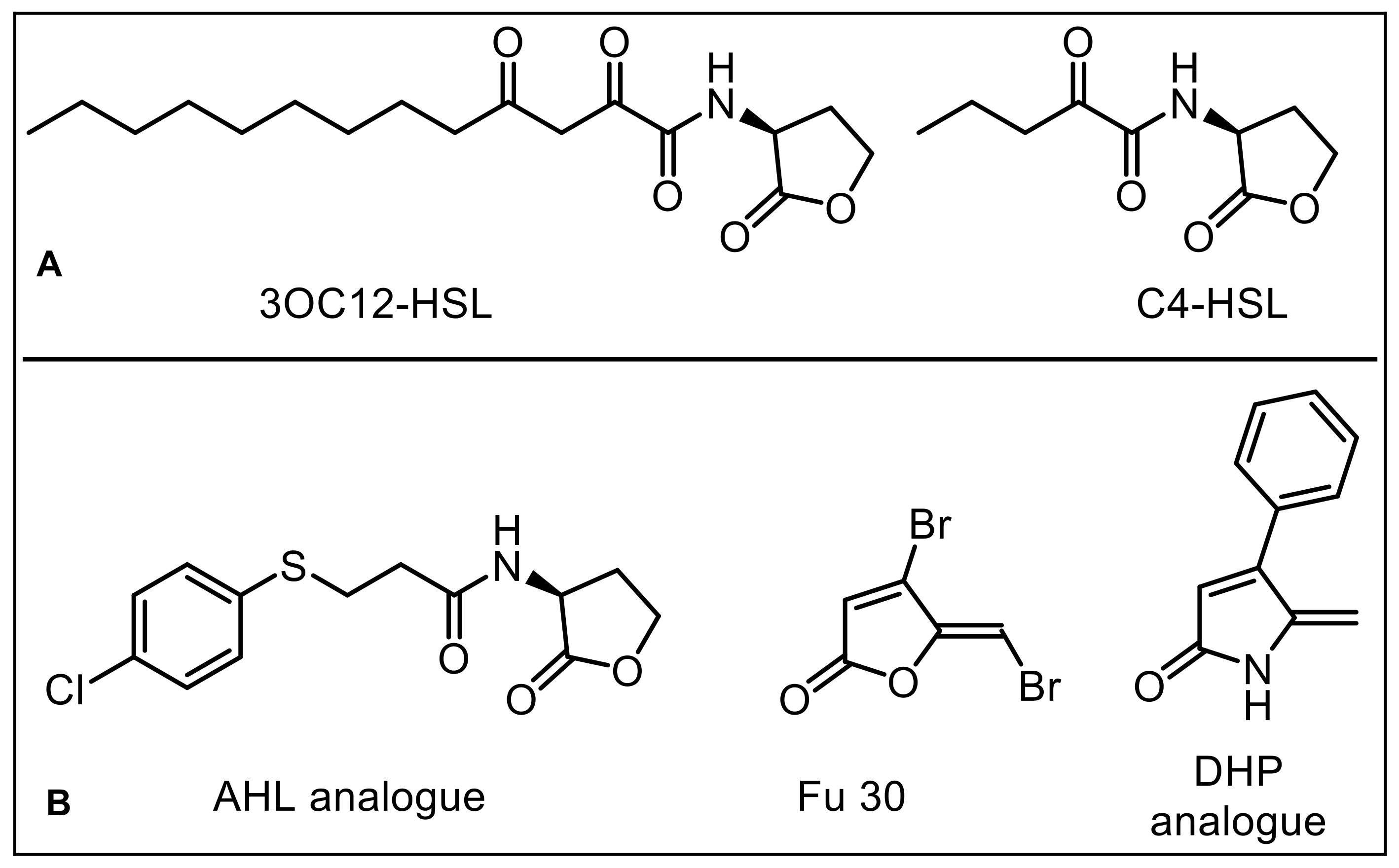
1. Introduction
2. Results
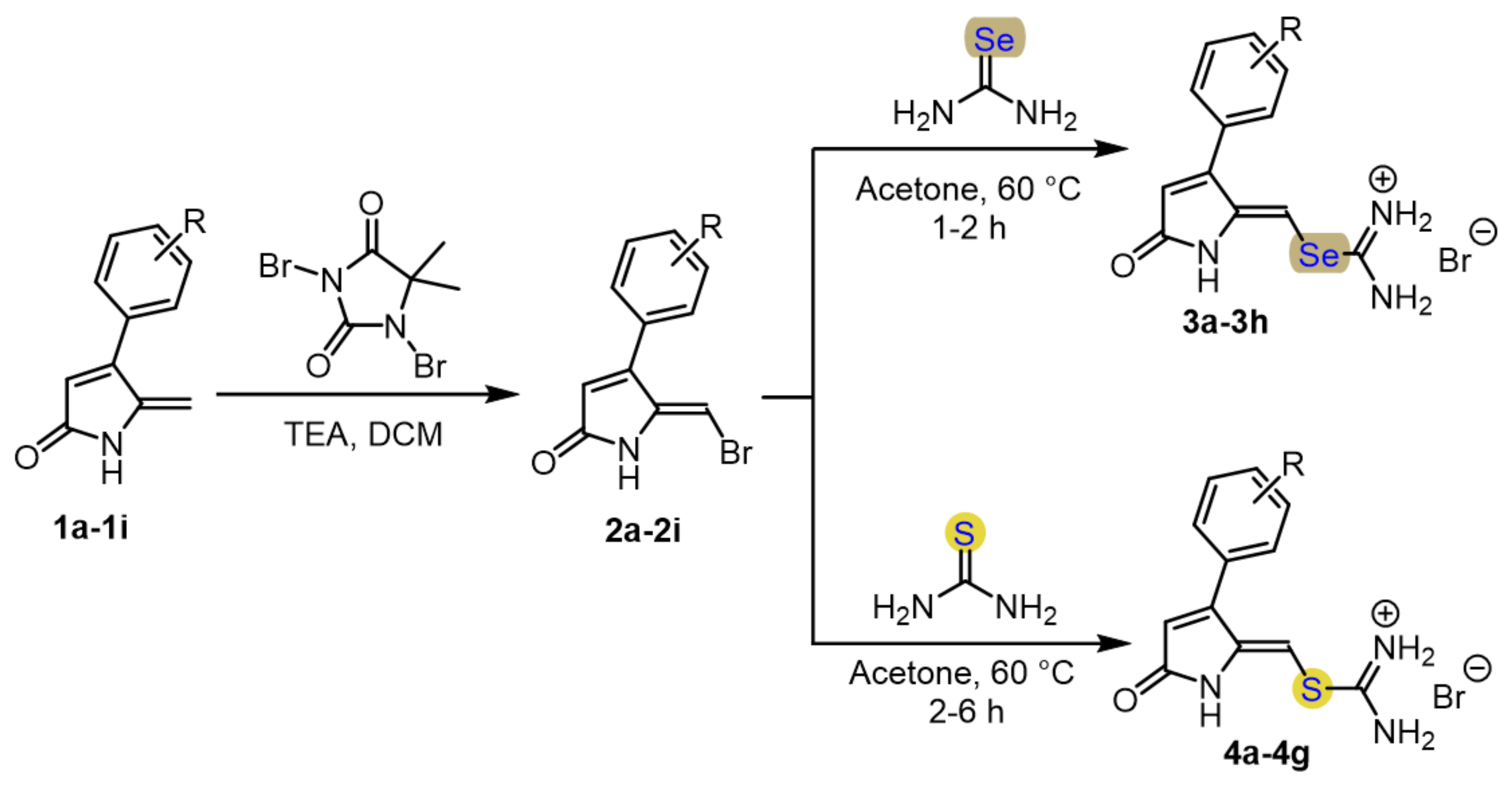
2.1. Chemistry
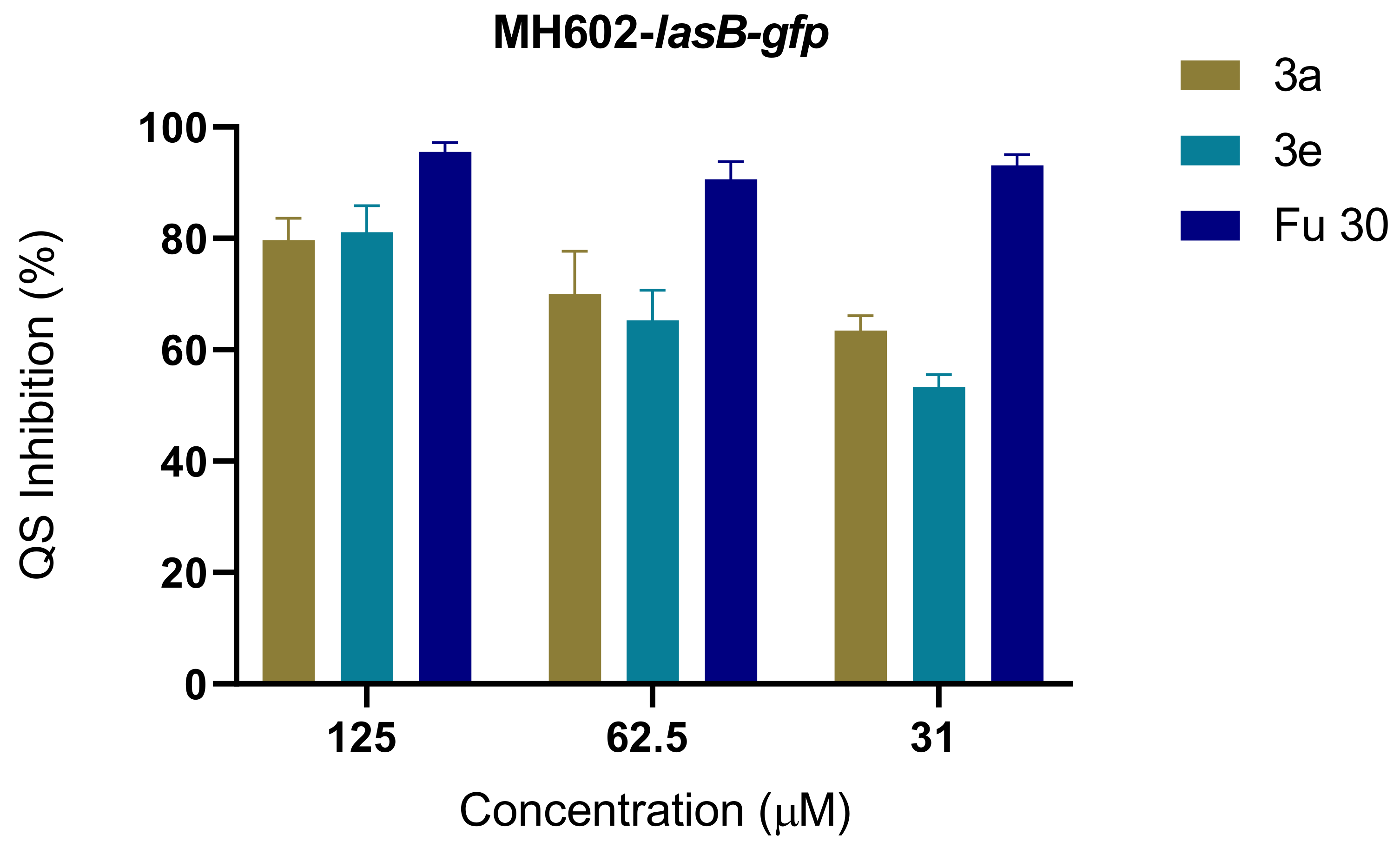
2.2. Quorum-Sensing Inhibition Assay
2.3. Antibacterial Activity
3. Conclusions
4. Materials and Methods
4.1. General Procedure for Synthesis of Selenourea and Thiourea DHP Compounds
4.2. Quorum-Sensing Inhibition Assay
4.3. Analysis of the Minimum Inhibitory and Minimum Bactericidal Concentration of Compounds
4.4. Lysis of Horse Red Blood Cells
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kadri, S.S. Key Takeaways From the U.S. CDC’s 2019 Antibiotic Resistance Threats Report for Frontline Providers. Crit. Care Med. 2020, 48, 939–945. [Google Scholar] [CrossRef]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J. How antibiotics kill bacteria: From targets to networks. Nat. Rev. Microbiol. 2010, 8, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C. Antibiotics: Actions, Origins, Resistance; American Society for Microbiology (ASM): Washington, DC, USA, 2003. [Google Scholar]
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef]
- Livermore, D.M. Multiple Mechanisms of Antimicrobial Resistance in Pseudomonas aeruginosa: Our Worst Nightmare? Clin. Infect. Dis. 2002, 34, 634–640. [Google Scholar] [CrossRef]
- Tenover, F.C. Mechanisms of Antimicrobial Resistance in Bacteria. Am. J. Med. 2006, 119, S3–S10. [Google Scholar] [CrossRef]
- Blair, J.M.A.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J.V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015, 13, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Årdal, C.; Balasegaram, M.; Laxminarayan, R.; McAdams, D.; Outterson, K.; Rex, J.H.; Sumpradit, N. Antibiotic development —Economic, regulatory and societal challenges. Nat. Rev. Microbiol. 2020, 18, 267–274. [Google Scholar] [CrossRef]
- Papenfort, K.; Bassler, B.L. Quorum sensing signal–response systems in Gram-negative bacteria. Nat. Rev. Microbiol. 2016, 14, 576–588. [Google Scholar] [CrossRef]
- Waters, C.M.; Bassler, B.L. QUORUM SENSING: Cell-to-Cell Communication in Bacteria. Annu. Rev. Cell Dev. Biol. 2005, 21, 319–346. [Google Scholar] [CrossRef] [PubMed]
- Schuster, M.; Greenberg, E.P. A network of networks: Quorum-sensing gene regulation in Pseudomonas aeruginosa. Int. J. Med. Microbiol. 2006, 296, 73–81. [Google Scholar] [CrossRef]
- Pesci, E.C.; Pearson, J.P.; Seed, P.C.; Iglewski, B.H. Regulation of las and rhl quorum sensing in Pseudomonas aeruginosa. J. Bacteriol. 1997, 179, 3127–3132. [Google Scholar] [CrossRef]
- Gilbert, K.B.; Kim, T.H.; Gupta, R.; Greenberg, E.P.; Schuster, M. Global position analysis of the Pseudomonas aeruginosa quorum-sensing transcription factor LasR. Mol. Microbiol. 2009, 73, 1072–1085. [Google Scholar] [CrossRef]
- Heurlier, K.; Dénervaud, V.; Haenni, M.; Guy, L.; Krishnapillai, V.; Haas, D. Quorum-Sensing-Negative (lasR) Mutants of Pseudomonas aeruginosa Avoid Cell Lysis and Death. J. Bacteriol. 2005, 187, 4875–4883. [Google Scholar] [CrossRef]
- Chang, Y.; Wang, P.-C.; Ma, H.-M.; Chen, S.-Y.; Fu, Y.-H.; Liu, Y.-Y.; Wang, X.; Yu, G.-C.; Huang, T.; Hibbs, D.E.; et al. Design, synthesis and evaluation of halogenated furanone derivatives as quorum sensing inhibitors in Pseudomonas aeruginosa. Eur. J. Pharm. Sci. 2019, 140, 105058. [Google Scholar] [CrossRef]
- Ho, K.K.; Kutty, S.K.; Chan, D.; Chen, R.; Willcox, M.D.; Kumar, N. Development of fimbrolides, halogenated furanones and their derivatives as antimicrobial agents. In Antibacterial Surfaces; Springer: Heidelberg, Germany, 2015; pp. 149–170. [Google Scholar]
- De Nys, R.; Givskov, M.; Kumar, N.; Kjelleberg, S.; Steinberg, P. Furanones. In Antifouling Compounds; Springer: New York, NY, USA, 2006; pp. 55–86. [Google Scholar]
- Grandclément, C.; Tannières, M.; Moréra, S.; Dessaux, Y.; Faure, D.D. Quorum quenching: Role in nature and applied developments. FEMS Microbiol. Rev. 2016, 40, 86–116. [Google Scholar] [CrossRef] [PubMed]
- Almohaywi, B.; Yu, T.T.; Iskander, G.; Chan, D.S.; Ho, K.K.; Rice, S.; Black, D.S.; Griffith, R.; Kumar, N. Dihydropyrrolones as bacterial quorum sensing inhibitors. Bioorg. Med. Chem. Lett. 2019, 29, 1054–1059. [Google Scholar] [CrossRef]
- Sabir, S.; Suresh, D.; Subramoni, S.; Das, T.; Bhadbhade, M.; Black, D.S.; Rice, S.A.; Kumar, N. Thioether-linked dihydropyrrol-2-one analogues as PqsR antagonists against antibiotic resistant Pseudomonas aeruginosa. Bioorg. Med. Chem. 2021, 31, 115967. [Google Scholar] [CrossRef] [PubMed]
- Mugesh, G.; Du Mont, W.-W.; Sies, H. Chemistry of Biologically Important Synthetic Organoselenium Compounds. Chem. Rev. 2001, 101, 2125–2180. [Google Scholar] [CrossRef] [PubMed]
- Reich, H.J.; Hondal, R.J. Why Nature Chose Selenium. ACS Chem. Biol. 2016, 11, 821–841. [Google Scholar] [CrossRef]
- Jain, V.K.; Priyadarsini, K.I. Organoselenium Compounds in Biology and Medicine: Synthesis, Biological and Therapeutic Treatments; Royal Society of Chemistry: London, UK, 2017. [Google Scholar]
- Mugesh, G.; Singh, H.B. Synthetic organoselenium compounds as antioxidants: Glutathione peroxidase activity. Chem. Soc. Rev. 2000, 29, 347–357. [Google Scholar] [CrossRef]
- Alcolea, V.; Plano, D.; Karelia, D.N.; Palop, J.A.; Amin, S.; Sanmartín, C.; Sharma, A.K. Novel seleno-and thio-urea derivatives with potent in vitro activities against several cancer cell lines. Eur. J. Med. Chem. 2016, 113, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Bartolini, D.; Sancineto, L.; de Bem, A.F.; Tew, K.D.; Santi, C.; Radi, R.; Toquato, P.; Galli, F. Selenocompounds in cancer therapy: An overview. In Advances in Cancer Research; Elsevier: Amsterdam, The Netherlands, 2017; Volume 136, pp. 259–302. [Google Scholar]
- Sancineto, L.; Piccioni, M.; De Marco, S.; Pagiotti, R.; Nascimento, V.; Braga, A.L.; Santi, C.; Pietrella, D. Diphenyl diselenide derivatives inhibit microbial biofilm formation involved in wound infection. BMC Microbiol. 2016, 16, 220. [Google Scholar] [CrossRef] [PubMed]
- Pietrella, D. Antimicrobial activity of organoselenium compounds. In Organoselenium Chemistry: Between Synthesis and Biochemistry; Bentham Books: Sharjah, United Arab Emirates, 2014; pp. 328–344. [Google Scholar]
- Pietka-Ottlik, M.; Burda-Grabowska, M.; Woźna, M.; Waleńska, J.; Kaleta, R.; Zaczyńska, E.; Piasecki, E.; Giurg, M. Synthesis of new alkylated and methoxylated analogues of ebselen with antiviral and antimicrobial properties. ARKIVOC 2017, 2017, 546–556. [Google Scholar] [CrossRef]
- Sharma, N.; Kumar, S.; Maurya, I.K.; Bhasin, K.K.; Verma, A.; Wangoo, N.; Bhasin, A.K.K.; Mehta, S.K.; Kumar, S.; Sharma, R.K. Synthesis, structural analysis, antimicrobial evaluation and synergistic studies of imidazo[1,2-a]pyrimidine chalcogenides. RSC Adv. 2016, 6, 114224–114234. [Google Scholar] [CrossRef]
- Sancineto, L.; Mariotti, A.; Bagnoli, L.; Marini, F.; DeSantis, J.; Iraci, N.; Santi, C.; Pannecouque, C.; Tabarrini, O. Design and Synthesis of DiselenoBisBenzamides (DISeBAs) as Nucleocapsid Protein 7 (NCp7) Inhibitors with anti-HIV Activity. J. Med. Chem. 2015, 58, 9601–9614. [Google Scholar] [CrossRef]
- Sartori, G.; Jardim, N.S.; Marcondes Sari, M.H.; Dobrachinski, F.; Pesarico, A.P.; Rodrigues, L.C., Jr.; Cargnelutti, J.; Flores, E.F.; Prigol, M.; Nogueira, C.W. Antiviral action of diphenyl diselenide on herpes simplex virus 2 infection in female BALB/c mice. J. Cell. Biochem. 2016, 117, 1638–1648. [Google Scholar] [CrossRef] [PubMed]
- Amosova, S.V.; Novokshonova, I.A.; Penzik, M.V.; Filippov, A.S.; Albanov, A.I.; Potapov, V.A. Reaction of 2-bromomethyl-1,3-thiaselenole with thiourea: En route to the first representatives of 2-(organylsulfanyl)-2,3-dihydro-1,4-thiaselenines. Tetrahedron Lett. 2017, 58, 4381–4383. [Google Scholar] [CrossRef]
- Hentzer, M.; Riedel, K.; Rasmussen, T.B.; Heydorn, A.; Andersen, J.B.; Parsek, M.R.; Rice, S.A.; Eberl, L.; Molin, S.; Høiby, N.; et al. Inhibition of quorum sensing in Pseudomonas aeruginosa biofilm bacteria by a halogenated furanone compound. Microbiology 2002, 148, 87–102. [Google Scholar] [CrossRef]
- Manefield, M.; Rasmussen, T.B.; Henzter, M.; Andersen, J.B.; Steinberg, P.; Kjelleberg, S.; Givskov, M. Halogenated furanones inhibit quorum sensing through accelerated LuxR turnover. Microbiology 2002, 148, 1119–1127. [Google Scholar] [CrossRef]
- Yasir, M.; Dutta, D.; Willcox, M.D.P. Mode of action of the antimicrobial peptide Mel4 is independent of Staphylococcus aureus cell membrane permeability. PLoS ONE 2019, 14, e0215703. [Google Scholar] [CrossRef] [PubMed]
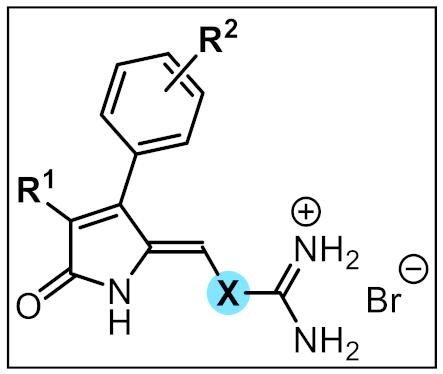 | ||||
| Compound | R1 | R2 | X | Yield (%) |
| 3a | H | H | Se | 83 |
| 3b | H | 4-F | Se | 82 |
| 3c | H | 4-Cl | Se | 85 |
| 3d | H | 4-Br | Se | 70 |
| 3e | H | 2-Cl | Se | 84 |
| 3f | CH3 | 4-Cl | Se | 63 |
| 3g | H | 2-F | Se | 82 |
| 3h | H | 4-OCH3 | Se | 80 |
| 4a | H | H | S | 69 |
| 4b | H | 4-F | S | 94 |
| 4c | H | 4-Cl | S | 71 |
| 4d | H | 4-Br | S | 96 |
| 4e | H | 2-F | S | 66 |
| 4f | H | 2-Cl | S | 48 |
| 4g | H | 3-CF3 | S | 76 |
| Compound | P. aeruginosa MH602 | ||
|---|---|---|---|
| 125 µM | 61.5 µM | 31 µM | |
| 3a | 80 ± 4 | 70 ± 8 | 63 ± 3 |
| 3b | 67 ± 2 | 43 ± 6 | 38 ± 6 |
| 3c | 74 ± 2 | 52 ± 3 | 42 ± 4 |
| 3d | 76 ± 1 | 58 ± 6 | 42 ± 4 |
| 3e | 81 ± 5 | 65 ± 5 | 53 ± 2 |
| 3f | 69 ± 6 | 49 ± 6 | 37 ± 3 |
| 3g | 66 ± 7 | 49 ± 4 | 38 ± 4 |
| 3h | 80 ± 6 | 64 ± 7 | 53 ± 4 |
| 4a | 50 ± 1 | 44 ± 0 | 37 ± 1 |
| 4b | 54 ± 3 | 38 ± 13 | 34 ± 5 |
| 4c | 70 ± 3 | 48 ± 5 | 35 ± 3 |
| 4d | 74 ± 2 | 54 ± 5 | 41 ± 5 |
| 4e | 40 ± 4 | 29 ± 7 | 23 ± 9 |
| 4f | 63 ± 1 | 55 ± 6 | 49 ± 11 |
| 4g | 56 ± 2 | 42 ± 2 | 34 ± 3 |
| Fu 30 | 96 ± 2 | 91 ± 3 | 93 ± 2 |
| Entry | Compounds | MIC/MBC (µM) | |
|---|---|---|---|
| S. aureus 38 | E. coli K12 | ||
| 1 | 3a | 31.2 | >250 |
| 2 | 3b | 125 | NT |
| 3 | 3c | 15.6/31.2 | >250 |
| 4 | 3d | >250 | NT |
| 5 | 3e | 15.6/62.4 | >250 |
| 6 | 3f | 62.5 | >250 |
| 7 | 3g | 62.5 | >250 |
| 8 | 3h | >250 | NT |
| 9 | 4a | >250 | NT |
| 10 | 4b | >250 | NT |
| 11 | 4c | >250 | NT |
| 12 | 4d | >250 | NT |
| 13 | 4e | >250 | NT |
| 14 | 4g | >250 | NT |
| 15 | 4f | >250 | NT |
| 16 | Gentamicin | <0.4 | NT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabir, S.; Yu, T.T.; Kuppusamy, R.; Almohaywi, B.; Iskander, G.; Das, T.; Willcox, M.D.P.; Black, D.S.; Kumar, N. Novel Seleno- and Thio-Urea Containing Dihydropyrrol-2-One Analogues as Antibacterial Agents. Antibiotics 2021, 10, 321. https://doi.org/10.3390/antibiotics10030321
Sabir S, Yu TT, Kuppusamy R, Almohaywi B, Iskander G, Das T, Willcox MDP, Black DS, Kumar N. Novel Seleno- and Thio-Urea Containing Dihydropyrrol-2-One Analogues as Antibacterial Agents. Antibiotics. 2021; 10(3):321. https://doi.org/10.3390/antibiotics10030321
Chicago/Turabian StyleSabir, Shekh, Tsz Tin Yu, Rajesh Kuppusamy, Basmah Almohaywi, George Iskander, Theerthankar Das, Mark D. P. Willcox, David StClair Black, and Naresh Kumar. 2021. "Novel Seleno- and Thio-Urea Containing Dihydropyrrol-2-One Analogues as Antibacterial Agents" Antibiotics 10, no. 3: 321. https://doi.org/10.3390/antibiotics10030321
APA StyleSabir, S., Yu, T. T., Kuppusamy, R., Almohaywi, B., Iskander, G., Das, T., Willcox, M. D. P., Black, D. S., & Kumar, N. (2021). Novel Seleno- and Thio-Urea Containing Dihydropyrrol-2-One Analogues as Antibacterial Agents. Antibiotics, 10(3), 321. https://doi.org/10.3390/antibiotics10030321










