Natural Agents against Bovine Mastitis Pathogens
Abstract
1. Introduction
2. Results
2.1. Bacteriological Testing of Milk Samples
2.2. Antibiotic Susceptibility Testing of Mastitis-Associated Bacteria
2.3. EOs’ Chemical Composition Analysis
2.4. EOs’ Antioxidant Potential Evaluation
2.5. EOs Effectiveness against Mastitis-Associated Bacteria
2.6. Interpretations of MBC, MIC, Thymus vulgaris, and Thymus serpyllum EOs in Relation to the Chemical Composition of the EOs
3. Discussion
4. Material and Methods
4.1. Essential Oils
4.2. Sampling Procedure
4.3. Antibiotic Susceptibility Testing of Mastitis-Associated Bacteria
4.4. EOs Chemical Composition Analysis
4.5. EOs’ Antioxidant Potential Evaluation
4.6. EOs’ Effectiveness Determination against Mastitis-Associated Bacteria
4.7. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gruet, P.; Maincent, P.; Berthelot, X.; Kaltsatos, V. Bovine mastitis and intramammary drug delivery: Review and perspectives. Adv. Drug Deliv. Rev. 2001, 50, 245–259. [Google Scholar] [CrossRef]
- Garcia, S.N.; Osburn, B.I.; Cullor, J.S. A one health perspective on dairy production and dairy food safety. One Health 2019, 7, 100086. [Google Scholar] [CrossRef]
- Halasa, T.; Huijps, K.; Osteras, O.; Hogeveen, H. Economic effects of bovine mastitis and mastitis management: A review. Vet. Q. 2007, 29, 18–31. [Google Scholar] [CrossRef] [PubMed]
- Drašković, B.; Stošić, I.; Rajković, Z. Tržišna struktura i nestašice mleka u Srbiji. In Agrarna i ruralna politika u Srbiji: Nužnost ubrzanja reformi: Tematski zbornik; DAES Društvo agrarnih ekonomista Srbije: Beograd, Serbia, 2011; pp. 65–79. [Google Scholar]
- Hristov, S.; Stanković, B.M.; Relić, R. Klinički i subklinički mastitis u krava. Biotechnol. Anim. Husb. 2005, 21, 29–39. [Google Scholar] [CrossRef]
- Kim, T.; Heald, C.W. Inducing inference rules for the classification of bovine mastitis. Comput. Electron. Agric. 1999, 23, 27–42. [Google Scholar] [CrossRef]
- Zadoks, R.; Fitzpatrick, J. Changing trends in mastitis. Ir. Vet. J. 2009, 62 (Suppl. 4), S59–S70. [Google Scholar] [CrossRef] [PubMed]
- Boboš, S.; Vidić, B. Mlečna žlezda preživara, morfologija, patologija, terapija; Univeryitet u Novom Sadu, Poljoprivredni fakultet: Novi Sad, Serbia, 2005; pp. 57–91. [Google Scholar]
- Krömker, V.; Leimbach, S. Mastitis treatment— Reduction in antibiotic usage in dairy cows. Reprod Dom Anim 2017, 52, 21–29. [Google Scholar] [CrossRef]
- Erskine, R.J.; Wagner, S.; DeGraves, F.J. Mastitis therapy and pharmacology. Vet. Clin. North Am. Food Anim. 2003, 19, 109–138. [Google Scholar] [CrossRef]
- Gupta, R.; Kumar, S.; Khurana, R. Essential oils and mastitis in dairy animals: A review. Haryana Vet. 2020, 59, 1–9. [Google Scholar]
- Sandegren, L. Selection of antibiotic resistance at very low antibiotic concentrations. Upsala J. Med. Sci. 2014, 119, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Dingwell, R.T.; Kelton, D.F.; Leslie, K.E. Management of the dry cow in control of peripartum disease and mastitis. Vet. Clin. N. Am. Food Anim. 2003, 19, 235–265. [Google Scholar] [CrossRef]
- Oliver, S.P.; Jayarao, B.M.; Almeida, R.A. Foodborne pathogens in milk and the dairy farm environment: Food safety and public health implications. Foodborne Pathog. Dis. 2005, 2, 115–129. [Google Scholar] [CrossRef]
- Fusco, V.; Chieffi, D.; Fanelli, F.; Logrieco, A.F.; Cho, G.S.; Kabisch, J.; Böhnlein, C.; Franz, C.M.A.P. Microbial quality and safety of milk and milk products in the 21st century. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2013–2049. [Google Scholar] [CrossRef]
- WHO. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2015; pp. 1–28. [Google Scholar]
- Kokoska, L.; Kloucek, P.; Leuner, O.; Novy, P. Plant-Derived Products as Antibacterial and Antifungal Agents in Human Health Care. Curr. Med. Chem. 2019, 26, 5501–5541. [Google Scholar] [CrossRef] [PubMed]
- O’Bryan, C.A.; Pendleton, S.J.; Crandall, P.G.; Ricke, S.C. Potential of Plant Essential Oils and Their Components in Animal Agriculture – in vitro Studies on Antibacterial Mode of Action. Front. Vet. Sci. 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Ananda Baskaran, S.; Kazmer, G.W.; Hinckley, L.; Andrew, S.M.; Venkitanarayanan, K. Antibacterial effect of plant-derived antimicrobials on major bacterial mastitis pathogens in vitro. Int. J. Dairy Sci. 2009, 92, 1423–1429. [Google Scholar] [CrossRef] [PubMed]
- Bozin, B.; Mimica-Dukic, N.; Simin, N.; Anackov, G. Characterization of the Volatile Composition of Essential Oils of Some Lamiaceae Spices and the Antimicrobial and Antioxidant Activities of the Entire Oils. J. Agric. Food Chem. 2006, 54, 1822–1828. [Google Scholar] [CrossRef]
- Gavaric, N.; Mozina, S.S.; Kladar, N.; Bozin, B. Chemical Profile, Antioxidant and Antibacterial Activity of Thyme and Oregano Essential Oils, Thymol and Carvacrol and Their Possible Synergism. J. Essent. Oil-Bear. Plants. 2015, 18, 1013–1021. [Google Scholar] [CrossRef]
- Lemos, M.F.; Lemos, M.F.; Pacheco, H.P.; Guimarães, A.C.; Fronza, M.; Endringer, D.C.; Scherer, R. Seasonal variation affects the composition and antibacterial and antioxidant activities of Thymus vulgaris. Ind. Crops Prod. 2017, 95, 543–548. [Google Scholar] [CrossRef]
- Nikolić, M.; Glamočlija, J.; Ferreira, I.C.F.R.; Calhelha, R.C.; Fernandes, Â.; Marković, T.; Marković, D.; Giweli, A.; Soković, M. Chemical composition, antimicrobial, antioxidant and antitumor activity of Thymus serpyllum L., Thymus algeriensis Boiss. and Reut and Thymus vulgaris L. essential oils. Ind. Crops Prod. 2014, 52, 183–190. [Google Scholar] [CrossRef]
- Langeveld, W.T.; Veldhuizen, E.J.; Burt, S.A. Synergy between essential oil components and antibiotics: A review. Crit. Rev. Microbiol. 2014, 40, 76–94. [Google Scholar] [CrossRef] [PubMed]
- Hemaiswarya, S.; Kruthiventi, A.K.; Doble, M. Synergism between natural products and antibiotics against infectious diseases. Phytomedicine 2008, 15, 639–652. [Google Scholar] [CrossRef]
- Malankina, E.L.; Kozlovskaya, L.N.; Kuzmenko, A.N.; Evgrafov, A.A. Determination of the Component Composition of Essential Oil of Thyme Species by the Method of Gas Chromatography. Mosc. Univ. Chem. Bull. 2020, 74, 310–314. [Google Scholar] [CrossRef]
- Ćavar-Zeljković, S.; Maksimović, M. Chemical composition and bioactivity of essential oil from Thymus species in Balkan Peninsula. Phytochem. Rev. 2015, 14, 335–352. [Google Scholar] [CrossRef]
- Tohidi, B.; Rahimmalek, M.; Arzani, A. Essential oil composition, total phenolic, flavonoid contents, and antioxidant activity of Thymus species collected from different regions of Iran. Food Chem. 2017, 220, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Helal, I.M.; El-Bessoumy, A.; Al-Bataineh, E.; Joseph, M.R.P.; Rajagopalan, P.; Chandramoorthy, H.C.; Ben Hadj Ahmed, S. Antimicrobial Efficiency of Essential Oils from Traditional Medicinal Plants of Asir Region, Saudi Arabia, over Drug Resistant Isolates. Biomed. Res. Int. 2019, 2019, 8928306. [Google Scholar] [CrossRef] [PubMed]
- Acimovic, M.; Zoric, M.; Zheljazkov, V.D.; Pezo, L.; Cabarkapa, I.; Stankovic Jeremic, J.; Cvetkovic, M. Chemical Characterization and Antibacterial Activity of Essential Oil of Medicinal Plants from Eastern Serbia. Molecules 2020, 25, 5482. [Google Scholar] [CrossRef]
- Radinović, M.; Davidov, I.; Kovačević, Z.; Stojanović, D.; Galfi, A.; Erdeljan, M. Basic Principles of Mastitis Therapy. Vet. J. Republic Srpska. 2019, 19. [Google Scholar] [CrossRef]
- Bozin, B.; Kladar, N.; Grujic, N.; Anackov, G.; Samojlik, I.; Gavaric, N.; Conic, B.S. Impact of origin and biological source on chemical composition, anticholinesterase and antioxidant properties of some St. John’s wort species (Hypericum spp., Hypericaceae) from the Central Balkans. Molecules 2013, 18, 11733–11750. [Google Scholar] [CrossRef]
- Mushtaq, S.; Shah, A.M.; Shah, A.; Lone, S.A.; Hussain, A.; Hassan, Q.P.; Ali, M.N. Bovine mastitis: An appraisal of its alternative herbal cure. Microb. Pathog. 2018, 114, 357–361. [Google Scholar] [CrossRef]
- Yang, W.-T.; Ke, C.-Y.; Wu, W.-T.; Lee, R.-P.; Tseng, Y.-H. Effective Treatment of Bovine Mastitis with Intramammary Infusion of Angelica dahurica and Rheum officinale Extracts. Evid. Based Complementary Altern. Med. 2019, 2019, 7242705. [Google Scholar] [CrossRef] [PubMed]
- Lopes, T.S.; Fontoura, P.S.; Oliveira, A.; Rizzo, F.A.; Silveira, S.; Streck, A.F. Use of plant extracts and essential oils in the control of bovine mastitis. Res. Vet. Sci. 2020, 131, 186–193. [Google Scholar] [CrossRef]
- McGaw, L. Use of Plant-Derived Extracts and Essential Oils against Multidrug-Resistant Bacteria Affecting Animal Health and Production. In Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components; Rai, M.K., Kon, K.V., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 191–203. Available online: https://doi.org/10.1016/B978-0-12-398539-2.00013-6 (accessed on 12 December 2020).
- Doehring, C.; Sundrum, A. The informative value of an overview on antibiotic consumption, treatment efficacy and cost of clinical mastitis at farm level. Prev. Vet. Med. 2019, 165, 63–70. [Google Scholar] [CrossRef] [PubMed]
- EDQM. European Pharmacopoeia 10.3; The European Directorate for the Quality of Medicines & HealthCare, Council of Europe: Brusselles, Belgium, 2020; pp. 1648–1650. [Google Scholar]
- Bogavac, M.; Karaman, M.; Janjusevic, L.; Sudji, J.; Radovanovic, B.; Novakovic, Z.; Simeunovic, J.; Bozin, B. Alternative treatment of vaginal infections-in vitro antimicrobial and toxic effects of Coriandrum sativum L. and Thymus vulgaris L. essential oils. J. Appl. Microbiol. 2015, 119, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.; Cefola, M.; Bonifacio, M.A.; Cometa, S.; Bocchino, C.; Pace, B.; De Giglio, E.; Palumbo, M.; Sada, A.; Logrieco, A.F.; et al. Effect of red thyme oil (Thymus vulgaris L.) vapours on fungal decay, quality parameters and shelf-life of oranges during cold storage. Food Chem. 2021, 336, 127590. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.; Bonifacio, M.A.; De Giglio, E.; Cometa, S.; Logrieco, A.F.; Baruzzi, F. Unravelling the Antifungal Effect of Red Thyme Oil (Thymus vulgaris L.) Compounds in Vapor Phase. Molecules 2020, 25, 4761. [Google Scholar] [CrossRef]
- Mimica-Dukic, N.; Bozin, B. Mentha L. species (Lamiaceae) as promising sources of bioactive secondary metabolites. Curr. Pharm. Des. 2008, 14, 3141–3150. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Antonio, A.L.; Oliveira, M.B.; Ferreira, I.C. A comparative study between natural and synthetic antioxidants: Evaluation of their performance after incorporation into biscuits. Food Chem. 2017, 216, 342–346. [Google Scholar] [CrossRef]
- Lanigan, R.S.; Yamarik, T.A. Final report on the safety assessment of BHT(1). Int. J. Toxicol. 2002, 21, 19–94. [Google Scholar] [CrossRef]
- Lesjak, M.M.; Beara, I.N.; Orčić, D.Z.; Anačkov, G.T.; Balog, K.J.; Francišković, M.M.; Mimica-Dukić, N.M. Juniperus sibirica Burgsdorf. as a novel source of antioxidant and anti-inflammatory agents. Food Chem. 2011, 124, 850–856. [Google Scholar] [CrossRef]
- Molinari, P.C.C.; Blagitz, M.G.; Libera, A.M.M.P.D.; Batista, C.F.; Souza, F.N. Intracellular reactive oxygen species production and phagocytosis of Staphylococcus aureus by milk neutrophils as tool to diagnose mastitis and identify susceptible dairy cows. Pesqui. Vet. Bras. 2018, 38, 659–664. [Google Scholar] [CrossRef]
- Dal Pozzo, M.; Santurio, D.F.; Rossatto, L.; Vargas, A.C.; Alves, S.H.; Loreto, E.S.; Viegas, J. Activity of essential oils from spices against Staphylococcus spp. isolated from bovine mastitis. Arq. Bras. Med. Vet. Zootec. 2011, 63, 1229–1232. [Google Scholar] [CrossRef]
- Fratini, F.; Casella, S.; Leonardi, M.; Pisseri, F.; Ebani, V.V.; Pistelli, L.; Pistelli, L. Antibacterial activity of essential oils, their blends and mixtures of their main constituents against some strains supporting livestock mastitis. Fitoterapia. 2014, 96, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods-a review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Burt, S.A.; Vlielander, R.; Haagsman, H.P.; Veldhuizen, E.J.A. Increase in Activity of Essential Oil Components Carvacrol and Thymol against Escherichia coli O157:H7 by Addition of Food Stabilizers. J. Food Prot. 2005, 68, 919–926. [Google Scholar] [CrossRef]
- Veldhuizen, E.J.; Tjeerdsma-van Bokhoven, J.L.; Zweijtzer, C.; Burt, S.A.; Haagsman, H.P. Structural requirements for the antimicrobial activity of carvacrol. J. Agric. Food. Chem. 2006, 54, 1874–1879. [Google Scholar] [CrossRef]
- Du, E.; Gan, L.; Li, Z.; Wang, W.; Liu, D.; Guo, Y. In vitro antibacterial activity of thymol and carvacrol and their effects on broiler chickens challenged with Clostridium perfringens. J. Anim. Sci. Biotechnol. 2015, 6, 58. [Google Scholar] [CrossRef]
- Hayashi, M.A.; Bizerra, F.C.; Da Silva, P.I., Jr. Antimicrobial compounds from natural sources. Front. Microbiol. 2013, 4, 195. [Google Scholar] [CrossRef]
- Xu, J.; Zhou, F.; Ji, B.P.; Pei, R.S.; Xu, N. The antibacterial mechanism of carvacrol and thymol against Escherichia coli. Lett. Appl. Microbiol. 2008, 47, 174–179. [Google Scholar] [CrossRef]
- Sikkema, J.; De Bont, J.A.M.; Poolman, B. Mechanisms of membrane toxicity of hydrocarbons. Microbiol. Rev. 1995, 59, 201–222. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; De Martino, L.; Coppola, R.; De Feo, V. Effect of Essential Oils on Pathogenic Bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef] [PubMed]
- Marinelli, L.; Di Stefano, A.; Cacciatore, I. Carvacrol and its derivatives as antibacterial agents. Phytochem. Rev. 2018, 17, 903–921. [Google Scholar] [CrossRef]
- Ultee, A.; Slump, R.A.; Steging, G.; Smid, E.J. Antimicrobial activity of carvacrol toward Bacillus cereus on rice. J. Food Prot. 2000, 63, 620–624. [Google Scholar] [CrossRef]
- Chouhan, S.; Sharma, K.; Guleria, S. Antimicrobial Activity of Some Essential Oils-Present Status and Future Perspectives. Medicines 2017, 4, 58. [Google Scholar] [CrossRef]
- Lambert, R.J.W.; Skandamis, P.N.; Coote, P.J.; Nychas, G.J.E. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Ultee, A.; Bennik, M.H.; Moezelaar, R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl. Environ. Microbiol. 2002, 68, 1561–1568. [Google Scholar] [CrossRef]
- Helander, I.K.; Alakomi, H.L.; Latva-Kala, K.; Mattila-Sandholm, T.; Pol, I.; Smid, E.J.; von Wright, A. Characterization of the action of selected essential oil components on Gram-negative bacteria. J. Agric. Food Chem. 1998, 46, 3590–3595. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef]
- Cristani, M.; D’Arrigo, M.; Mandalari, G.; Castelli, F.; Sarpietro, M.G.; Micieli, D.; Venuti, V.; Bisignano, G.; Saija, A.; Trombetta, D. Interaction of four monoterpenes contained in essential oils with model membranes: Implications for their antibacterial activity. J. Agric. Food Chem. 2007, 55, 6300–6308. [Google Scholar] [CrossRef]
- Marchese, A.; Arciola, C.R.; Barbieri, R.; Silva, A.S.; Nabavi, S.F.; Tsetegho Sokeng, A.J.; Izadi, M.; Jafari, N.J.; Suntar, I.; Daglia, M.; et al. Update on Monoterpenes as Antimicrobial Agents: A Particular Focus on p-Cymene. Materials 2017, 10, 947. [Google Scholar] [CrossRef]
- Belmar-Liberato, R.; Gonzalez-Canga, A.; Tamame-Martin, P.; Escribano-Salazar, M. Amoxicillin and amoxicillin-clavulanic acid resistance in veterinary medicine—the situation in Europe: A review. Vet. Med. (Praha) 2011, 56, 473–485. [Google Scholar] [CrossRef]
- Supre, K.; Lommelen, K.; De Meulemeester, L. Antimicrobial susceptibility and distribution of inhibition zone diameters of bovine mastitis pathogens in Flanders, Belgium. Vet. Microbiol. 2014, 171, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Yap, P.S.; Yiap, B.C.; Ping, H.C.; Lim, S.H. Essential oils, a new horizon in combating bacterial antibiotic resistance. Open Microbiol. J. 2014, 8, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Cuaron, J.A.; Dulal, S.; Song, Y.; Singh, A.K.; Montelongo, C.E.; Yu, W.; Nagarajan, V.; Jayaswal, R.K.; Wilkinson, B.J.; Gustafson, J.E. Tea tree oil-induced transcriptional alterations in Staphylococcus aureus. Phytother. Res. 2013, 27, 390–396. [Google Scholar] [CrossRef]
- Hudzicki, J. Kirby-Bauer Disk Diffusion Susceptibility Protocol; American Society for Microbiology (ASM): Washington, DC, USA, 2009; Available online: https://www.asmscience.org/docserver/fulltext/education/protocol/protocol.3189.pdf?expires=1613515898&id=id&accname=guest&checksum=835F5125F08BB2AB46FF21C23963C9B7 (accessed on 1 December 2020).
- CLSI. Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals, 3rd ed.; CLSI: Wayne, PA, USA, 2008. [Google Scholar]
- CLSI. Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved standard M02-A12; CLSI: Wayne, PA, USA, 2012. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Nuutila, A.M.; Puupponen-Pimiä, R.; Aarni, M.; Oksman-Caldentey, K.-M. Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity. Food Chem. 2003, 81, 485–493. [Google Scholar] [CrossRef]
- MacDonald-Wicks, L.K.; Wood, L.G.; Garg, M.L. Methodology for the determination of biological antioxidant capacity in vitro: A review. J. Sci. Food Agric. 2006, 86, 2046–2056. [Google Scholar] [CrossRef]
- CLSI. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 11th ed.; Approved Standard document M07; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018; Available online: https://clsi.org/media/1928/m07ed11_sample.pdf (accessed on 1 December 2020).
- Elshikh, M.; Ahmed, S.; Funston, S.; Dunlop, P.; McGaw, M.; Marchant, R.; Banat, I.M. Resazurin-based 96-well plate microdilution method for the determination of minimum inhibitory concentration of biosurfactants. Biotechnol. Lett. 2016, 38, 1015–1019. [Google Scholar] [CrossRef] [PubMed]
- Čabarkapa, I.; Čolović, R.; Đuragić, O.; Popović, S.; Kokić, B.; Milanov, D.; Pezo, L. Anti-biofilm activities of essential oils rich in carvacrol and thymol against Salmonella Enteritidis. Biofouling 2019, 35, 361–375. [Google Scholar] [CrossRef] [PubMed]
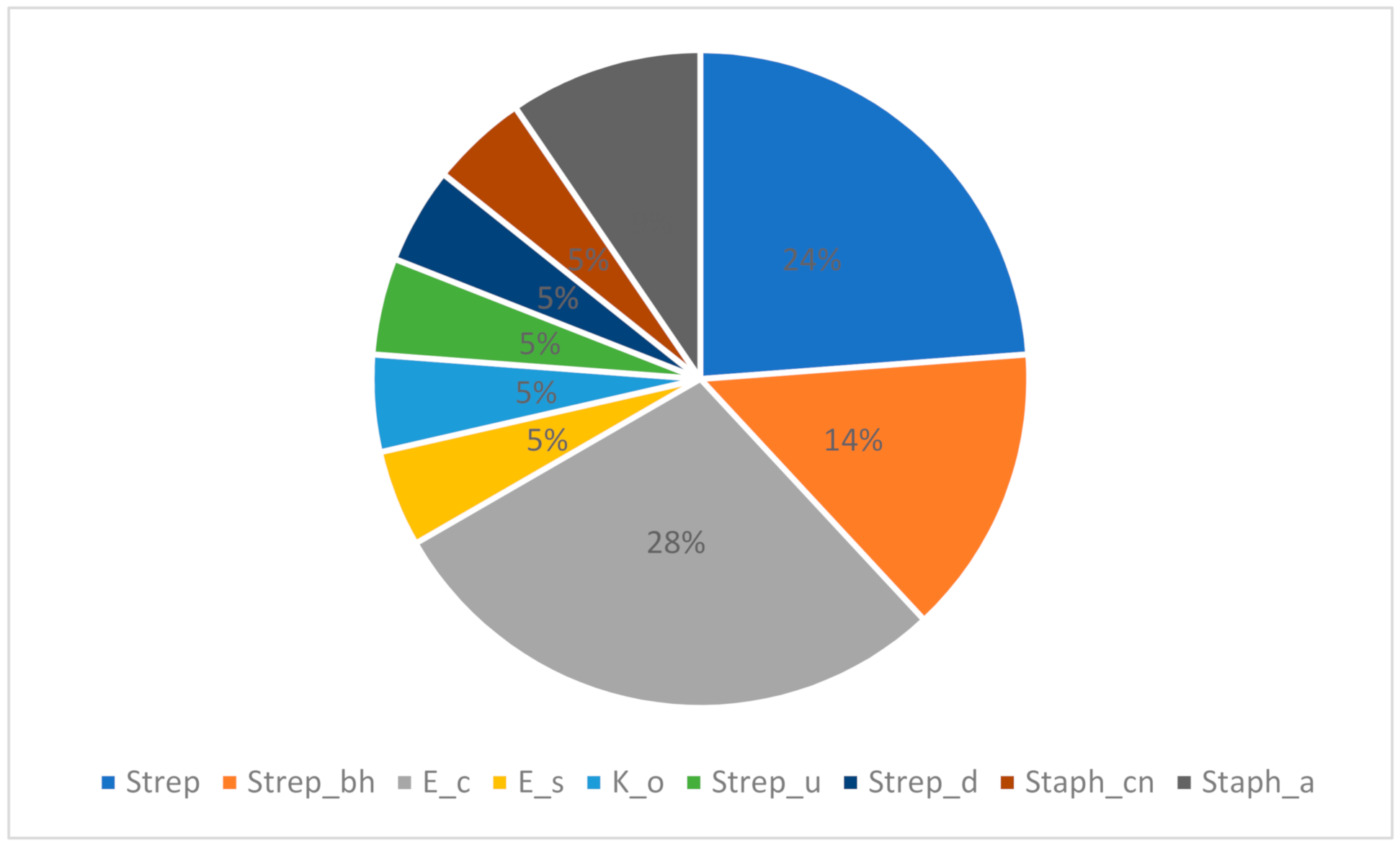
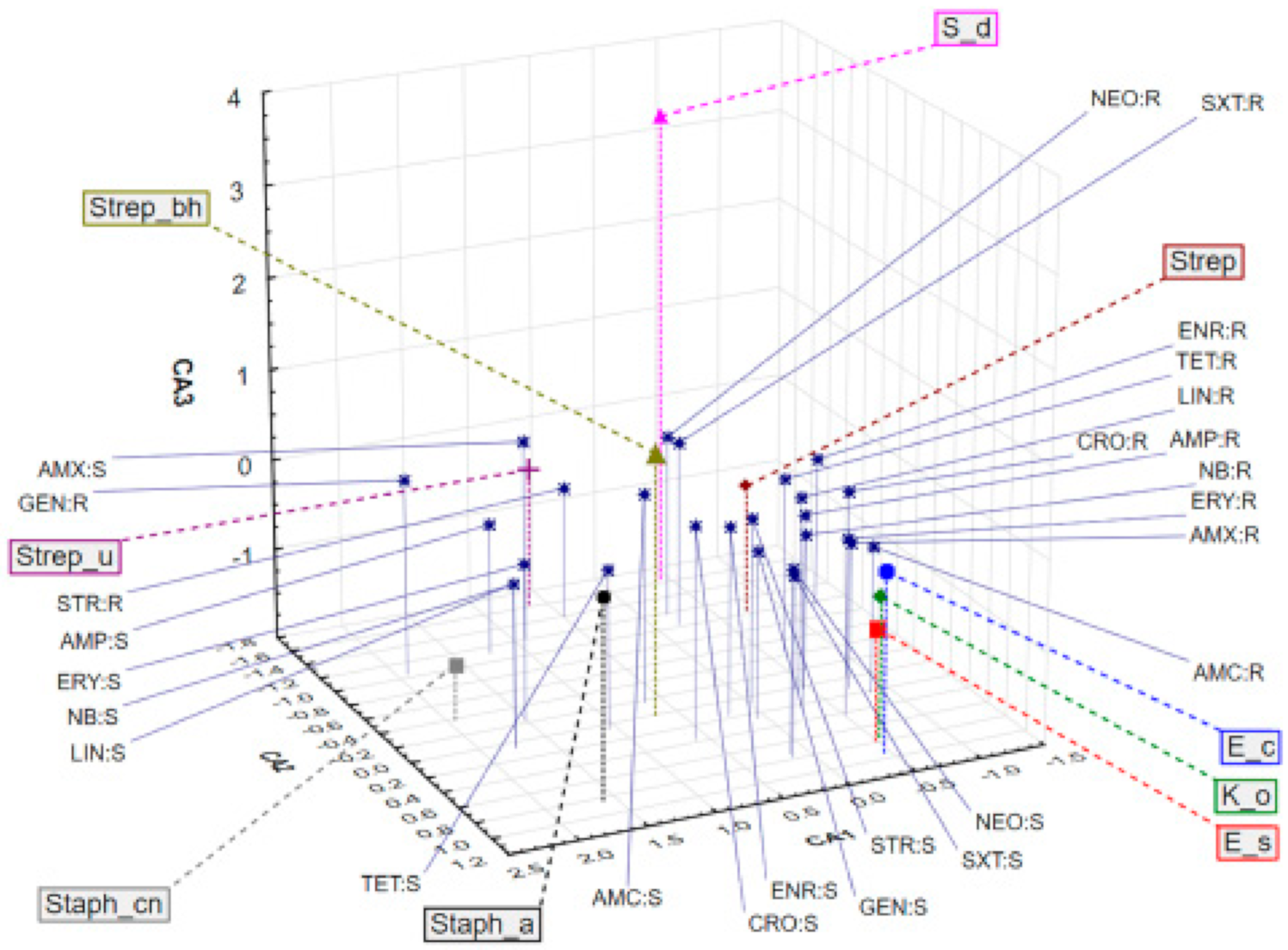
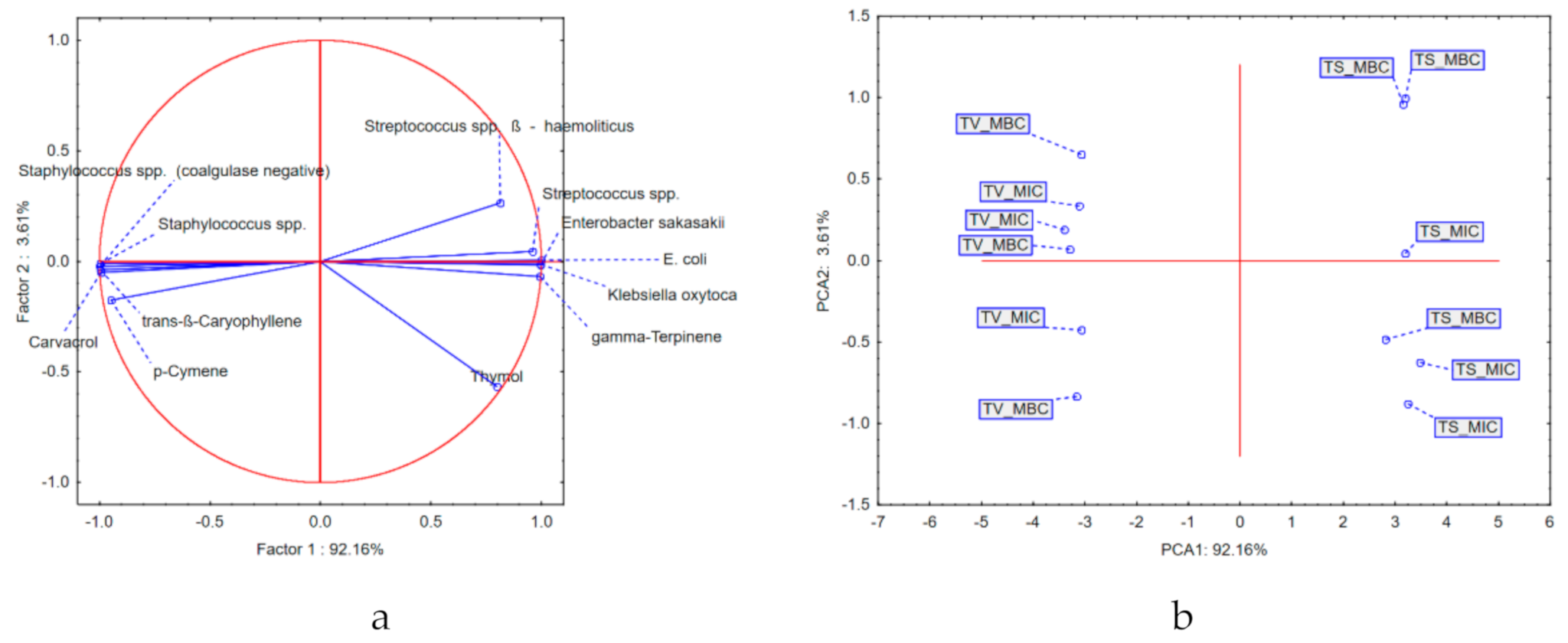
| Bacterial Strains Culture | AMX | AMP | CRO | ENR | ERY | GEN | LIN | NEO | PEN | STR | TET | AMC | NB | SXT | CLO |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Streptococcus spp. β heamoliticus | S | S | S | S | S | R | S | R | R | S | S | S | S | R | R |
| Staphylococcus spp. | R | R | R | R | R | I | R | R | R | S | R | R | R | R | R |
| Staphylococcus spp. | R | R | R | R | R | I | R | R | R | S | R | R | R | R | R |
| Staphylococcus spp. coagulase negative | S | S | I | S | S | S | S | S | R | S | S | S | S | S | R |
| Staphylococcus spp. | R | R | R | R | I | S | R | R | R | S | R | R | R | R | R |
| Streptococcus spp. β heamoliticus | I | R | S | S | R | S | R | S | R | S | I | S | I | S | R |
| E. coli | R | R | R | S | R | S | R | S | R | S | R | R | R | S | R |
| E. coli | R | R | R | S | R | S | R | S | R | S | I | R | R | S | R |
| Streptococcus spp. β heamoliticus | R | R | S | S | R | S | R | S | R | S | R | S | R | R | R |
| Klebsiella oxytoca | R | R | S | S | R | S | R | S | R | S | R | R | R | S | R |
| E. coli | R | R | R | S | R | S | R | S | R | S | I | R | R | S | R |
| Staphylococcus spp. | R | R | I | S | R | S | I | R | R | S | R | S | R | R | R |
| E. coli | R | R | R | S | R | S | R | S | R | S | R | R | R | S | R |
| Enterobacter sakazakii | R | R | R | S | R | S | R | S | R | S | S | R | R | S | R |
| Staphylococcus aureus | I | R | S | S | S | S | S | S | R | S | S | S | S | S | R |
| E. coli | I | R | S | S | R | S | R | S | R | S | I | S | R | S | R |
| Streptococcus uberis | S | S | I | S | I | S | R | R | R | S | S | S | R | R | R |
| E. coli | I | R | S | S | R | S | R | S | R | S | S | S | R | S | R |
| Staphylococcus aureus | I | R | S | S | S | S | S | S | R | S | S | S | S | S | R |
| Streptococcus dysgalactiae | S | R | R | S | I | I | R | R | R | S | R | S | I | R | R |
| Staphylococcus spp. | S | S | S | S | S | S | R | R | R | R | R | S | R | R | R |
| Peak No. | Compounds | RI a | T. vulgaris | T. serpyllum |
|---|---|---|---|---|
| Monoterpene Hydrocarbons | 10.84 | 25.4 | ||
| 1. | α-Pinene | 937 | 1.51 | 0.18 |
| 2. | Camphene | 952 | 1.67 | 0.19 |
| 3. | β-Pinene | 978 | 0.21 | 2.15 |
| 4. | β-Myrcene | 991 | 1.64 | 0.28 |
| 5. | α-Phellandrene | 1005 | 0.11 | 0.08 |
| 6. | α-Terpinene | 1017 | 0.87 | 0.13 |
| 8. | Limonene | 1030 | 1.71 | 0.21 |
| 12. | γ-Terpinene | 1060 | 3.12 | 22.18 |
| Aromatic Monoterpene Hydrocarbons | 23.83 | 16.66 | ||
| 7. | p-Cymene | 1025 | 23.83 | 16.66 |
| Oxygenated Monoterpenes | 7.19 | 2.05 | ||
| 9. | 1,8-Cineole | 1032 | 0.93 | 0.16 |
| 10. | Linalool | 1099 | 2.55 | 0 |
| 11. | Camphor | 1145 | 0.33 | 0.77 |
| 13. | endo-Borneol | 1167 | 1.68 | 0 |
| 14. | Terpinen-4-ol | 1177 | 1.42 | 0.07 |
| 15. | Isomenthol | 1183 | 0 | 0.84 |
| 16. | α-Terpineol | 1189 | 0.23 | 0.03 |
| 19. | Bornyl acetate | 1285 | 0.05 | 0.07 |
| 26. | trans-β-Ionone | 1486 | 0 | 0.11 |
| Aromatic Oxygenated Monoterpenes | 51.49 | 54.98 | ||
| 17. | Isothymol methyl ether | 1230 | 0.92 | 0 |
| 18. | Thymol methyl ether | 1235 | 1.49 | 0 |
| 20. | Thymol | 1291 | 45.22 | 54.17 |
| 21. | Carvacrol | 1299 | 3.86 | 0.81 |
| Sesquiterpene Hydrocarbons | 4.91 | 0.2 | ||
| 22. | α-Cubebene | 1351 | 0.08 | 0 |
| 23. | β-Cubenene | 1388 | 0.03 | 0 |
| 24. | trans-β-Caryophyllene | 1419 | 4.04 | 0.12 |
| 25. | Humulene | 1454 | 0.41 | 0.08 |
| 27. | δ-Cadinene | 1524 | 0.35 | 0 |
| Oxygenated Sesquiterpenes | 0.94 | 0 | ||
| 28. | Caryophyllene oxide | 1581 | 0.94 | 0 |
| Total of identified compounds (%) | 99.2 | 99.29 | ||
| Samples | Assay | |||
|---|---|---|---|---|
| DPPH IC50 | OH IC50 (µg / mL) | LP IC50 | FRAP (mg AAE a /mL EO) | |
| X̅ b ± SD c | X̅ ± SD | X̅ ± SD | X̅ ± SD | |
| T. vulgaris | 14 ± 0.85 | 230 ± 1.19 | 19 ± 1.02 | 34.95 ± 3.50 |
| T. serpyllum | 16 ± 0.93 | 170 ± 2.02 | 17 ± 0.92 | 29.00 ± 2.90 |
| AA | / | 2003 ± 0.39 | / | / |
| PG | 0.71 ± 0.04 | 9.07 ± 0.59 | / | / |
| BHT | / | 0.03 ± 0.01 | 7.29 ± 0.56 | / |
| Sample | TV * (MIC) (mg/mL) | TV * (MBC) (mg/mL) | TS ** (MIC) (mg/mL) | TS ** (MBC) (mg/mL) |
|---|---|---|---|---|
| 4 E. coli | 3.125 | 6.25 | 6.25 | 12.5 |
| Enterobacter sakazakii | 3.125 | 6.25 | 6.25 | 12.5 |
| Streptococcus spp. β heamoliticus | 0.39 | 0.78 | 1.56 | 3.125 |
| Streptococcus spp. β heamoliticus | 0.78 | 1.56 | 1.56 | 3.125 |
| Streptococcus spp. β heamoliticus | 0.39 | 0.78 | 0.78 | 1.56 |
| Streptococcus spp. | 1.56 | 3.125 | 3.125 | 6.25 |
| Streptococcus spp. | 0.78 | 1.56 | 3.125 | 6.25 |
| Streptococcus spp. | 1.56 | 6.25 | 3.125 | 6.25 |
| Staphylococcus spp. | 6.25 | 12.5 | 3.125 | 6.25 |
| Staphylococcus spp. coagulase negative | 6.25 | 12.5 | 3.125 | 6.25 |
| Klebsiella oxytoca | 1.56 | 6.25 | 3.125 | 6.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovačević, Z.; Radinović, M.; Čabarkapa, I.; Kladar, N.; Božin, B. Natural Agents against Bovine Mastitis Pathogens. Antibiotics 2021, 10, 205. https://doi.org/10.3390/antibiotics10020205
Kovačević Z, Radinović M, Čabarkapa I, Kladar N, Božin B. Natural Agents against Bovine Mastitis Pathogens. Antibiotics. 2021; 10(2):205. https://doi.org/10.3390/antibiotics10020205
Chicago/Turabian StyleKovačević, Zorana, Miodrag Radinović, Ivana Čabarkapa, Nebojša Kladar, and Biljana Božin. 2021. "Natural Agents against Bovine Mastitis Pathogens" Antibiotics 10, no. 2: 205. https://doi.org/10.3390/antibiotics10020205
APA StyleKovačević, Z., Radinović, M., Čabarkapa, I., Kladar, N., & Božin, B. (2021). Natural Agents against Bovine Mastitis Pathogens. Antibiotics, 10(2), 205. https://doi.org/10.3390/antibiotics10020205







