Antimicrobial, Antioxidant, and Cytotoxic Activities of Juglans regia L. Pellicle Extract
Abstract
1. Introduction
2. Results
2.1. Cytotoxicity on HEp-2 and Vero Cell Lines
2.2. Antiviral Activity
2.3. Antibacterial Activity
2.4. Antifungal Activity
2.5. Antioxidant Activity and Total Phenolic and Flavonoid Contents
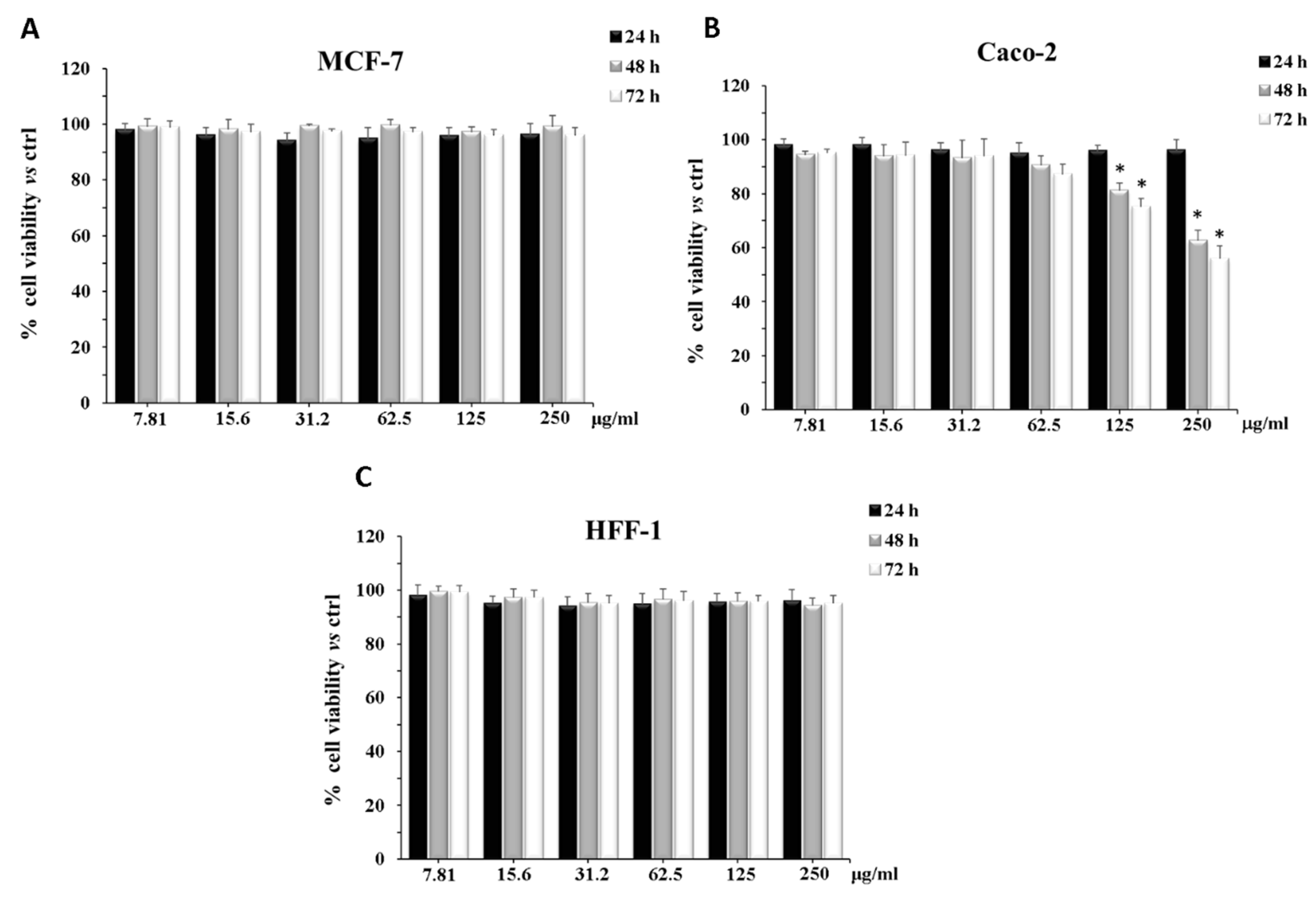
2.6. Cytotoxicity on CaCo-2, MCF-7, and HFF1 Cell Lines
3. Discussion
4. Materials and Methods
4.1. Chemical Reagents
4.2. Plant Material and Preparation of the Extract
4.3. Cytotoxicity Assay on HEp-2 and Vero Cell Lines
4.4. Antiviral Activity
4.5. Antibacterial Activity
4.6. Antifungal Activity
4.7. Antioxidant Activity and Determination of Total Phenolic and Flavonoid Contents
4.8. Cell Culture
4.9. Cytotoxicity Assay on CaCo-2, MCF-7, and HFF1 Cell Lines
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hay, S.I.; Rao, P.C.; Dolecek, C.; Day, N.P.J.; Stergachis, A.; Lopez, A.D.; Murray, C.J.L. Measuring and mapping the global burden of antimicrobial resistance. BMC Med. 2018, 16, 1–3. [Google Scholar] [CrossRef]
- Raymond, D.P.; Kuehnert, M.J.; Sawyer, R.G. Preventing Antimicrobial-Resistant Bacterial Infections in Surgical Patients. Surg. Infect. 2002, 3, 375–385. [Google Scholar] [CrossRef]
- Zhou, S.; Fan, L.; Wang, Z.; Wang, Q.; Xiong, Z.; Xu, Y.; Li, D. Increasing rates of Acinetobacter baumannii infection and resistance in an oncology department. J. Cancer Res. Ther. 2018, 14, 68–71. [Google Scholar] [CrossRef]
- Chng, K.R.; MetaSUB Consortium; Li, C.; Bertrand, D.; Ng, A.H.Q.; Kwah, J.S.; Low, H.M.; Tong, C.; Natrajan, M.; Zhang, M.H.; et al. Cartography of opportunistic pathogens and antibiotic resistance genes in a tertiary hospital environment. Nat. Med. 2020, 26, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.A.; Baig, F.K.; Mehboob, R. Nosocomial infections: Epidemiology, prevention, control and surveillance. Asian Pac. J. Trop. Biomed. 2017, 7, 478–482. [Google Scholar] [CrossRef]
- Weiner-Lastinger, L.M.; Abner, S.; Benin, A.L.; Edwards, J.R.; Kallen, A.J.; Karlsson, M.; Magill, S.S.; Pollock, D.; See, I.; Soe, M.M.; et al. Antimicrobial-resistant pathogens associated with pediatric healthcare-associated infections: Summary of data reported to the National Healthcare Safety Network, 2015–2017. Infect. Control. Hosp. Epidemiol. 2019, 41, 19–30. [Google Scholar] [CrossRef]
- Cabrera-Cancio, M.R. Infections and the Compromised Immune Status in the Chronically Critically Ill Patient: Prevention Strategies. Respir. Care 2012, 57, 979–992. [Google Scholar] [CrossRef] [PubMed]
- Genovese, C.; Acquaviva, R.; Ronsisvalle, S.; Tempera, G.; Malfa, G.A.; D’Angeli, F.; Ragusa, S.; Nicolosi, D. In vitro evaluation of biological activities of Orobanche crenata Forssk. leaves extract. Nat. Prod. Res. 2020, 34, 3234–3238. [Google Scholar] [CrossRef]
- Papoutsi, Z.; Kassi, E.; Chinou, I.; Halabalaki, M.; Skaltsounis, L.A.; Moutsatsou, P. Walnut extract (Juglans regia L.) and its component ellagic acid exhibit anti-inflammatory activity in human aorta endothelial cells and osteoblastic activity in the cell line KS483. Br. J. Nutr. 2008, 99, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Jahanban-Esfahlan, A.; Ostadrahimi, A.; Tabibiazar, M.; Amarowicz, R. A Comparative Review on the Extraction, Antioxidant Content and Antioxidant Potential of Different Parts of Walnut (Juglans regia L.) Fruit and Tree. Molecules 2019, 24, 2133. [Google Scholar] [CrossRef]
- Salimi, M.; Ardestaniyan, M.H.; Kandelous, H.M.; Saeidnia, S.; Gohari, A.R.; Amanzadeh, A.; Sanati, H.; Sepahdar, Z.; Ghorbani, S. Anti-proliferative and apoptotic activities of constituents of chloroform extract of Juglans regialeaves. Cell Prolif. 2014, 47, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Amirou, A.; Bnouham, M.; Legssyer, A.; Ziyyat, A.; Aziz, M.; Berrabah, M.; Mekhfi, H. Effects of Juglans regia Root Bark Extract on Platelet Aggregation, Bleeding Time, and Plasmatic Coagulation: In Vitro and Ex Vivo Experiments. Evid. Based Complement. Altern. Med. 2018, 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nasiry, D.; Khalatbary, A.R.; Ahmadvand, H.; Amiri, F.B.T.; Akbari, E. Protective effects of methanolic extract of Juglans regia L. leaf on streptozotocin-induced diabetic peripheral neuropathy in rats. BMC Complement. Altern. Med. 2017, 17, 476. [Google Scholar] [CrossRef] [PubMed]
- Bakht, J.; Khan, S.; Shafi, M.; Iqbal, A. Fractionation of crude extracts from controlled dried and commercially available stem bark of Juglans regia and their antimicrobial effects. Pak. J. Pharm. Sci. 2017, 30, 1581–1588. [Google Scholar]
- Ahmad, S.; Wahid, M.A.; Bukhari, A.Q.S. Fungistatic Action of Juglans. Antimicrob. Agents Chemother. 1973, 3, 436–438. [Google Scholar] [CrossRef] [PubMed]
- Genovese, C.; Cambria, M.T.; D’Angeli, F.; Addamo, A.P.; Malfa, G.A.; Siracusa, L.; Pulvirenti, L.; Anfuso, C.D.; Lupo, G.; Salmeri, M. The double effect of walnut septum extract (Juglans regia L.) counteracts A172 glioblastoma cell survival and bacterial growth. Int. J. Oncol. 2020, 57, 1129–1144. [Google Scholar] [CrossRef]
- Wu, S.; Ni, Z.; Wang, R.; Zhao, B.; Han, Y.; Zheng, Y.; Liu, F.; Gong, Y.; Tang, F.; Liu, Y. The effects of cultivar and climate zone on phytochemical components of walnut (Juglans regia L.). Food Energy Secur. 2020, 9. [Google Scholar] [CrossRef]
- Acquaviva, R.; D’Angeli, F.; Malfa, G.A.; Ronsisvalle, S.; Garozzo, A.; Stivala, A.; Ragusa, S.; Nicolosi, D.; Salmeri, M.; Genovese, C. Antibacterial and anti-biofilm activities of walnut pellicle extract (Juglans regia L.) against coagulase-negative staphylococci. Nat. Prod. Res. 2019, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xu, C.; Hao, C.; Zhang, Y.; Wang, Z.; Wang, S.; Wang, W. Inhibition of herpes simplex virus by myricetin through targeting viral gD protein and cellular EGFR/PI3K/Akt pathway. Antivir. Res. 2020, 177, 104714. [Google Scholar] [CrossRef]
- Stojković, D.; Živković, J.; Sokovic, M.; Glamočlija, J.; Ferreira, I.C.F.R.; Jankovic, T.; Maksimović, Z. Antibacterial activity of Veronica montana L. extract and of protocatechuic acid incorporated in a food system. Food Chem. Toxicol. 2013, 55, 209–213. [Google Scholar] [CrossRef]
- Zhang, Z.; Liao, L.; Moore, J.; Wu, T.; Wang, Z. Antioxidant phenolic compounds from walnut kernels (Juglans regia L.). Food Chem. 2009, 113, 160–165. [Google Scholar] [CrossRef]
- El-Toumy, S.A.; Salib, J.Y.; El-Kashak, W.A.; Marty, C.; Bedoux, G.; Bourgougnon, N. Antiviral effect of polyphenol rich plant extracts on herpes simplex virus type 1. Food Sci. Hum. Wellness 2018, 7, 91–101. [Google Scholar] [CrossRef]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial Activity and Mode of Action of Ferulic and Gallic Acids Against Pathogenic Bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef]
- Ho, K.-V.; Roy, A.; Foote, S.; Vo, P.H.; Lall, N.; Lin, C.-H. Profiling Anticancer and Antioxidant Activities of Phenolic Compounds Present in Black Walnuts (Juglans nigra) Using a High-Throughput Screening Approach. Molecules 2020, 25, 4516. [Google Scholar] [CrossRef]
- Chen, C. Sinapic Acid and Its Derivatives as Medicine in Oxidative Stress-Induced Diseases and Aging. Oxidative Med. Cell. Longev. 2016, 2016, 1–10. [Google Scholar] [CrossRef]
- Li, L.; Tsao, R.; Yang, R.; Kramer, A.J.K.G.; Hernandez, M. Fatty Acid Profiles, Tocopherol Contents, and Antioxidant Activities of Heartnut (Juglans ailanthifolia Var. cordiformis) and Persian Walnut (Juglans regia L.). J. Agric. Food Chem. 2007, 55, 1164–1169. [Google Scholar] [CrossRef] [PubMed]
- Cotticelli, M.G.; Forestieri, R.; Xia, S.; Joyasawal, S.; Lee, T.; Xu, K.; Smith, I.A.B.; Huryn, D.M.; Wilson, R.B. Identification of a Novel Oleic Acid Analog with Protective Effects in Multiple Cellular Models of Friedreich Ataxia. ACS Chem. Neurosci. 2020, 11, 2535–2542. [Google Scholar] [CrossRef]
- Batirel, S.; Yilmaz, A.M.; Sahin, A.; Perakakis, N.; Ozer, N.K.; Mantzoros, C.S. Antitumor and antimetastatic effects of walnut oil in esophageal adenocarcinoma cells. Clin. Nutr. 2018, 37, 2166–2171. [Google Scholar] [CrossRef] [PubMed]
- Negi, A.S.; Luqman, S.; Srivastava, S.; Krishna, V.; Gupta, N.; Darokar, M.P. Antiproliferative and antioxidant activities of Juglans regiafruit extracts. Pharm. Biol. 2011, 49, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Johnston, K.; Sharp, P.; Clifford, M.; Morgan, L. Dietary polyphenols decrease glucose uptake by human intestinal Caco-2 cells. FEBS Lett. 2005, 579, 1653–1657. [Google Scholar] [CrossRef] [PubMed]
- Lyu, S.-Y.; Rhim, J.-Y.; Park, W.-B. Antiherpetic activities of flavonoids against herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) in vitro. Arch. Pharmacal Res. 2005, 28, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Jakub, T.; Gazdová, M.; Smejkal, K.; Šudomová, M.; Kubatka, P.; Hassan, S.T. Natural Products-Derived Chemicals: Breaking Barriers to Novel Anti-HSV Drug Development. Viruses 2020, 12, 154. [Google Scholar] [CrossRef]
- Pereira, J.A.; Oliveira, I.; Sousa, A.; Valentão, P.; Andrade, P.B.; Ferreira, I.C.F.R.; Ferreres, F.; Bento, A.; Seabra, R.M.; Estevinho, L. Walnut (Juglans regia L.) leaves: Phenolic compounds, antibacterial activity and antioxidant potential of different cultivars. Food Chem. Toxicol. 2007, 45, 2287–2295. [Google Scholar] [CrossRef] [PubMed]
- Al-Aboody, M.S.; Mickymaray, S. Anti-Fungal Efficacy and Mechanisms of Flavonoids. Antibiotics 2020, 9, 45. [Google Scholar] [CrossRef] [PubMed]
- Nakatani, N.; Kayano, S.-I.; Kikuzaki, H.; Sumino, K.; Katagiri, A.K.; Mitani, T. Identification, Quantitative Determination, and Antioxidative Activities of Chlorogenic Acid Isomers in Prune (Prunusdomestica L.). J. Agric. Food Chem. 2000, 48, 5512–5516. [Google Scholar] [CrossRef]
- Parvez, M.K.; Alam, P.; Arbab, A.H.; Al-Dosari, M.S.; Alhowiriny, T.A.; Alqasoumi, S.I. Analysis of antioxidative and antiviral biomarkers β-amyrin, β-sitosterol, lupeol, ursolic acid in Guiera senegalensis leaves extract by validated HPTLC methods. Saudi Pharm. J. 2018, 26, 685–693. [Google Scholar] [CrossRef]
- Sa, F.A.D.S.; de Paula, J.A.M.; dos Santos, P.A.; Oliveira, L.D.A.R.; Oliveira, G.D.A.R.; Lião, L.M.; Paula, J.R.; Silva, M. Phytochemical Analysis and Antimicrobial Activity of Myrcia tomentosa (Aubl.) DC. Leaves. Molecules 2017, 22, 1100. [Google Scholar] [CrossRef]
- Müller, U.; Stübl, F.; Schwarzinger, B.; Sandner, G.; Iken, M.; Himmelsbach, M.; Schwarzinger, C.; Ollinger, N.; Stadlbauer, V.; Höglinger, O.; et al. In Vitro and In Vivo Inhibition of Intestinal Glucose Transport by Guava (Psidium Guajava) Extracts. Mol. Nutr. Food Res. 2018, 62, e1701012. [Google Scholar] [CrossRef]
- Vivek-Ananth, R.P.; Rana, A.; Rajan, N.; Biswal, H.S.; Samal, A. In Silico Identification of Potential Natural Product Inhibitors of Human Proteases Key to SARS-CoV-2 Infection. Molecules 2020, 25, 3822. [Google Scholar] [CrossRef]
- Luca, S.V.; Bujor, A.; Miron, A.; Aprotosoaie, A.C.; Skalicka-Woźniak, K.; Trifan, A. Preparative separation and bioactivity of oligomeric proanthocyanidins. Phytochem. Rev. 2020, 19, 1093–1140. [Google Scholar] [CrossRef]
- Han, K.-I.; Jung, E.-G.; Patnaik, B.B.; Hong, C.-I.; Kim, Y.-J.; Jung, S.; Han, M.-D. Antibacterial and Antioxidant Activities of Leaf Extracts from Juglans sinensis, and its Phenolic Compositions. Nat. Prod. Commun. 2017, 12, 1797–1800. [Google Scholar] [CrossRef]
- Hung, P.-Y.; Ho, B.-C.; Lee, S.-Y.; Chang, S.-Y.; Kao, C.-L.; Lee, S.-S.; Lee, C.-N. Houttuynia cordata Targets the Beginning Stage of Herpes Simplex Virus Infection. PLoS ONE 2015, 10, e0115475. [Google Scholar] [CrossRef]
- Lobdell, K.W.; Stamou, S.; Sanchez, J.A. Hospital-Acquired Infections. Surg. Clin. North. Am. 2012, 92, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Boev, C.; Kiss, E. Hospital-Acquired Infections. Crit. Care Nurs. Clin. N. Am. 2017, 29, 51–65. [Google Scholar] [CrossRef]
- Kaur, K.; Michael, H.; Arora, S.; Härkönen, P.L.; Kumar, S. Studies on Correlation of Antimutagenic and Antiproliferative Activities of Juglans regia L. J. Environ. Pathol. Toxicol. Oncol. 2003, 22, 57–66. [Google Scholar] [CrossRef]
- Jagtap, A.G.; Karkera, S.G. Extract ofJuglandaceae regiaInhibits Growth, In-vitro Adherence, Acid Production and Aggregation ofStreptococcus mutans. J. Pharm. Pharmacol. 2000, 52, 235–242. [Google Scholar] [CrossRef]
- Moghaddam, P.Z.; Mohammadi, A.; Feyzi, P.; Alesheikh, P. In vitro antioxidant and antibacterial activity of various extracts from exocarps and endocarps of walnut. Pak. J. Pharm. Sci. 2017, 30, 1725–1731. [Google Scholar]
- Bisignano, C.; Mandalari, G.; Smeriglio, A.; Trombetta, D.; Pizzo, M.M.; Pennisi, R.; Sciortino, M.T. Almond Skin Extracts Abrogate HSV-1 Replication by Blocking Virus Binding to the Cell. Viruses 2017, 9, 178. [Google Scholar] [CrossRef]
- Astani, A.; Reichling, J.; Schnitzler, P. Melissa officinalis Extract Inhibits Attachment of Herpes Simplex Virus in vitro. Chemotherapy 2012, 58, 70–77. [Google Scholar] [CrossRef]
- Yang, C.-M.; Cheng, H.-Y.; Lin, T.-C.; Chiang, L.-C.; Lin, C.-C. The in vitro activity of geraniin and 1,3,4,6-tetra-O-galloyl-β-d-glucose isolated from Phyllanthus urinaria against herpes simplex virus type 1 and type 2 infection. J. Ethnopharmacol. 2007, 110, 555–558. [Google Scholar] [CrossRef]
- Lee, S.; Lee, H.H.; Shin, Y.-S.; Kang, H.; Cho, H. The anti-HSV-1 effect of quercetin is dependent on the suppression of TLR-3 in Raw 264.7 cells. Arch. Pharmacal Res. 2017, 40, 623–630. [Google Scholar] [CrossRef]
- Hopkins, J.; Yadavalli, T.; Agelidis, A.M.; Shukla, D. Host Enzymes Heparanase and Cathepsin L Promote Herpes Simplex Virus 2 Release from Cells. J. Virol. 2018, 92. [Google Scholar] [CrossRef]
- Auer, G.K.; Weibel, D.B. Bacterial Cell Mechanics. Biochemistry 2017, 56, 3710–3724. [Google Scholar] [CrossRef]
- Exner, M.; Bhattacharya, S.; Christiansen, B.; Gebel, J.; Goroncy-Bermes, P.; Hartemann, P.; Heeg, P.; Ilschner, C.; Kramer, A.; Larson, E.; et al. Antibiotic resistance: What is so special about multidrug-resistant Gram-negative bacteria? GMS Hyg. Infect. Control. 2017, 12. [Google Scholar] [CrossRef]
- Caporarello, N.; Olivieri, M.; Cristaldi, M.; Scalia, M.; Toscano, M.A.; Genovese, C.; Addamo, A.; Salmeri, M.; Lupo, G.; Anfuso, C.D. Blood–Brain Barrier in a Haemophilus influenzae Type a In Vitro Infection: Role of Adenosine Receptors A2A and A2B. Mol. Neurobiol. 2017, 55, 5321–5336. [Google Scholar] [CrossRef]
- Saraiva, M.; Castro, R.H.A.; Cordeiro, R.P.; Sobrinho, T.J.S.P.; Amorim, E.L.C.; Xavier, H.S.; Pisciottano, M.N.C. In vitro evaluation of antioxidant, antimicrobial and toxicity properties of extracts of Schinopsis brasiliensis Engl. (Anacardiaceae). Afr. J. Pharm. Pharmacol. 2011, 5, 1724–1731. [Google Scholar] [CrossRef]
- Rudramurthy, S.M.; Singh, S. Candida Infections in Immunocompetent Hosts: Pathogenesis and Diagnosis. Curr. Fungal Infect. Rep. 2020, 14, 233–245. [Google Scholar] [CrossRef]
- Genovese, C.; Corsello, S.; Nicolosi, D.; Aidala, V.; Falcidia, E.; Tempera, G. Alterations of the vaginal microbiota in the third trimester of pregnancy and pPROM. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 3336–3343. [Google Scholar]
- Gintjee, T.J.; Donnelley, M.A.; Thompson, G.R. Aspiring Antifungals: Review of Current Antifungal Pipeline Developments. J. Fungi 2020, 6, 28. [Google Scholar] [CrossRef]
- Genovese, C.; Cianci, A.; Corsello, S.; Ettore, G.; Mattana, P.; Tempera, G. Combined systemic (fluconazole) and topical (metronidazole + clotrimazole) therapy for a new approach to the treatment and prophylaxis of recurrent candidiasis. Minerva Ginecol. 2019, 71, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Genovese, C.; Pulvirenti, L.; Cardullo, N.; Muccilli, V.; Tempera, G.; Nicolosi, D.; Tringali, C. Bioinspired benzoxanthene lignans as a new class of antimycotic agents: Synthesis and Candida spp. growth inhibition. Nat. Prod. Res. 2018, 34, 1653–1662. [Google Scholar] [CrossRef]
- Sytykiewicz, H.; Chrzanowski, G.; Czerniewicz, P.; Leszczyński, B.; Sprawka, I.; Krzyżanowski, R.; Matok, H. Antifungal Activity of Juglans regia (L.) Leaf Extracts Against Candida albicans Isolates. Pol. J. Environ. Stud. 2015, 24, 1339–1348. [Google Scholar] [CrossRef]
- Noumi, E.; Snoussi, M.; Hajlaoui, H.; Valentin, E.; Bakhrouf, A. Antifungal properties of Salvadora persica and Juglans regia L. extracts against oral Candida strains. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 29, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Raja, V.; Ahmad, S.; Irshad, M.; Wani, W.; Siddiqi, W.; Shreaz, S. Anticandidal activity of ethanolic root extract of Juglans regia (L.): Effect on growth, cell morphology, and key virulence factors. J. Med. Mycol. 2017, 27, 476–486. [Google Scholar] [CrossRef]
- Jafer, F.N.; A Naser, L. The Biological Activity of Aqueous and Methanolic Extracts of Juglans regia on Yeasts and Pathologic Bacteria. Arch. Clin. Microbiol. 2020, 11, 2405–2410. [Google Scholar] [CrossRef]
- An, R.-B.; Kim, H.-C.; Tian, Y.-H.; Kim, Y.-C. Free Radical Scavenging and Hepatoprotective Constituents from the Leaves of Juglans sinensis. Arch. Pharmacal. Res. 2005, 28, 529–533. [Google Scholar] [CrossRef]
- Thevissen, K.; Ghazi, A.; de Samblanx, G.W.; Brownlee, C.; Osborn, R.W.; Broekaert, W.F. Fungal Membrane Responses Induced by Plant Defensins and Thionins. J. Biol. Chem. 1996, 271, 15018–15025. [Google Scholar] [CrossRef] [PubMed]
- Tay, L.Y.; Jorge, J.H.; Herrera, D.R.; Campanha, N.H.; Gomes, B.P.F.A.; dos Santos, F.A. Evaluation of different treatment methods against denture stomatitis: A randomized clinical study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2014, 118, 72–77. [Google Scholar] [CrossRef]
- Taplitz, R.A.; Kennedy, E.B.; Bow, E.J.; Crews, J.; Gleason, C.; Hawley, D.K.; Langston, A.A.; Nastoupil, L.J.; Rajotte, M.; Rolston, K.V.; et al. Antimicrobial Prophylaxis for Adult Patients with Cancer-Related Immunosuppression: ASCO and IDSA Clinical Practice Guideline Update. J. Clin. Oncol. 2018, 36, 3043–3054. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Milbury, P.E.; Lapsley, K.; Blumberg, J.B. Flavonoids from Almond Skins Are Bioavailable and Act Synergistically with Vitamins C and E to Enhance Hamster and Human LDL Resistance to Oxidation. J. Nutr. 2005, 135, 1366–1373. [Google Scholar] [CrossRef]
- Schreck, K.; Melzig, M. Intestinal Saturated Long-Chain Fatty Acid, Glucose and Fructose Transporters and Their Inhibition by Natural Plant Extracts in Caco-2 Cells. Molecules 2018, 23, 2544. [Google Scholar] [CrossRef]
- Marbaniang, C.; Kma, L. Dysregulation of Glucose Metabolism by Oncogenes and Tumor Suppressors in Cancer Cells. Asian Pac. J. Cancer Prev. 2018, 19, 2377–2390. [Google Scholar]
- Reckzeh, E.S.; Karageorgis, G.; Schwalfenberg, M.; Ceballos, J.; Nowacki, J.; Stroet, M.C.; Binici, A.; Knauer, L.; Brand, S.; Choidas, A.; et al. Inhibition of Glucose Transporters and Glutaminase Synergistically Impairs Tumor Cell Growth. Cell Chem. Biol. 2019, 26, 1214–1228.e25. [Google Scholar] [CrossRef]
- Taviano, M.F.; Miceli, N.; Acquaviva, R.; Malfa, G.A.; Ragusa, S.; Giordano, D.; Cásedas, G.; Les, F.; López, V. Cytotoxic, Antioxidant, and Enzyme Inhibitory Properties of the Traditional Medicinal Plant Matthiola incana (L.) R. Br. Biol. 2020, 9, 163. [Google Scholar] [CrossRef]
- Trandafir, I.; Cosmulescu, S.; Botu, M.; Nour, V. Antioxidant activity, and phenolic and mineral contents of the walnut kernel (Juglans regia L.) as a function of the pellicle color. Fruits 2016, 71, 177–184. [Google Scholar] [CrossRef]
- Akbari, V.; Jamei, R.; Heidari, R.; Esfahlan, A.J. Antiradical activity of different parts of Walnut (Juglans regia L.) fruit as a function of genotype. Food Chem. 2012, 135, 2404–2410. [Google Scholar] [CrossRef] [PubMed]
- Cutrí, C.C.; Garozzo, A.; Siracusa, M.A.; Sarvá, M.C.; Tempera, G.; Geremia, E.; Pinizzotto, M.R.; Guerrera, F. Synthesis and antiviral activity of a new series of 4-isothiazolecarbonitriles. Bioorganic Med. Chem. 1998, 6, 2271–2280. [Google Scholar] [CrossRef]
- Performance Standards for Antimicrobial Susceptibility: M100—Performance Standards for Antimicrobial Susceptibility Testing, 29th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2019.
- Clinical and Laboratory Standards Institute. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, Approved Standard; CLSI document M27-A3 and Supplement S; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2008; Volume 3, pp. 6–12. [Google Scholar]
- Tenuta, M.C.; Deguin, B.; Loizzo, M.R.; Dugay, A.; Acquaviva, R.; Malfa, G.A.; Bonesi, M.; Bouzidi, C.; Tundis, R. Contribution of Flavonoids and Iridoids to the Hypoglycaemic, Antioxidant, and Nitric Oxide (NO) Inhibitory Activities of Arbutus unedo L. Antioxidants 2020, 9, 184. [Google Scholar] [CrossRef] [PubMed]
- Malfa, G.A.; Tomasello, B.; Acquaviva, R.; la Mantia, A.; Pappalardo, F.; Ragusa, M.; Renis, M.; di Giacomo, C. The Antioxidant Activities of Betula etnensis Rafin. Ethanolic Extract Exert Protective and Anti-diabetic Effects on Streptozotocin-Induced Diabetes in Rats. Antioxidants 2020, 9, 847. [Google Scholar] [CrossRef] [PubMed]

| CD50 a | ID50 b | |||||||
|---|---|---|---|---|---|---|---|---|
| HEp-2 | VERO | HSV-1 | HSV-2 | ECHO 9 | Polio 1 | Coxsackie B1 | Adeno 2 | |
| WPE (µg/mL) c | 25.00 | 28.00 | 10.00 | 8.00 | ORC d | ORC | ORC | ORC |
| Acyclovir (µg/mL) | >600.00 | >600.00 | 0.12 | 0.36 | ORC | ORC | ORC | ORC |
| Bacterial Strains b | Source | MIC a (µg/mL) | I.C. e | |
|---|---|---|---|---|
| WPE c | Cip d | |||
| Range [0.53–275.00] | [0.06–32.00] | |||
| Gram-positive | ||||
| E. faecalis 012/040 | Abscess | 8.59 | 0.50 | S |
| E. faecium 019/040 | Catheter cystitis | 8.59 | 1.00 | S |
| S. epidermidis 007/040 | Osteomyelitis | 8.59 | 0.031 | S |
| S. epidermidis ATCC 14990 | Standard | 8.59 | 0.125 | S |
| E. faecalis 015/040 | Abscess | 17.18 | 0.25 | S |
| S. aureus 002/040 | Endophtalmitis | 17.18 | 0.25 | S |
| S. aureus 004/040 | Pneumonia | 17.18 | 0.25 | S |
| E. faecalis 013/040 | Septicemia | 34.37 | 0.50 | S |
| S. aureus 005/040 | Endophtalmitis | 34.37 | 0.50 | S |
| E. faecalis ATCC 29212 | Standard | 68.75 | 0.50 | S |
| E. faecalis 014/040 | Pneumonia | 68.75 | 1.00 | S |
| E. faecium 018/040 | Catheter cystitis | 275.00 | 8.00 | R |
| S. aureus ATCC 29213 | Standard | 275.00 | 0.50 | S |
| S. epidermidis 009/040 | Endophtalmitis | 275.00 | 0.015 | S |
| E. faecium 017/040 | Cholecystitis | >275.00 | >32.00 | R |
| E. faecium 020/040 | Cholecystitis | >275.00 | 16.00 | R |
| E. faecium ATCC 700221 | Standard | >275.00 | >32.00 | R |
| S. aureus 003/040 | Pneumonia | >275.00 | 4.00 | R |
| S. epidermidis 008/040 | Septicemia | >275.00 | 8.00 | R |
| S. epidermidis 010/040 | Septicemia | >275.00 | 8.00 | R |
| Gram-negative | ||||
| E. coli 024/040 | Cystitis | 275.00 | 4.00 | R |
| E. coli 025/040 | Cystitis | 275.00 | 8.00 | R |
| K. pneumoniae 035/040 | Nephritis | 275.00 | 4.00 | R |
| P. aeruginosa 028/040 | Septicemia | 275.00 | 4.00 | R |
| P. aeruginosa 029/040 | Pneumonia | 275.00 | 0.125 | S |
| P. mirabilis 038/040 | Cystitis | 275.00 | 0.015 | S |
| P. mirabilis ATCC 7002 | Standard | 275.00 | 0.25 | S |
| E. coli 022/040 | Septicemia | >275.00 | 0.015 | S |
| E. coli 023/040 | Septicemia | >275.00 | >32.00 | R |
| E. coli ATCC 35218 | Standard | >275.00 | 0.015 | S |
| K. pneumoniae 032/040 | Nephritis | >275.00 | 4.00 | R |
| K. pneumoniae 033/040 | Pneumonia | >275.00 | 32.00 | R |
| K. pneumoniae 034/040 | Pneumonia | >275.00 | 8.00 | R |
| K. pneumoniae ATCC 700630 | Standard | >275.00 | 0.25 | S |
| P. aeruginosa 027/040 | Septicemia | >275.00 | 0.06 | S |
| P. aeruginosa 030/040 | Pneumonia | >275.00 | 16.00 | R |
| P. aeruginosa ATCC 27853 | Standard | >275.00 | 0.25 | S |
| P. mirabilis 037/040 | Cystitis | >275.00 | 1.00 | S |
| P. mirabilis 039/040 | Cystitis | >275.00 | 0.015 | S |
| P. mirabilis 040/040 | Cystitis | >275.00 | 8.00 | R |
| Fungal Strains c | Source | WPE a (µg/mL) | FLU b (µg/mL) | |||||
|---|---|---|---|---|---|---|---|---|
| MFC d | MIC e | MFC/MIC f | MFC | MIC | MFC/MIC | I.C. g | ||
| C. albicans 003/040 | V.S. h | >275.00 | 17.18 | N.C. i | >64.00 | 0.12 | N.C. | S |
| C. albicans 006/040 | V.S. | >275.00 | 17.18 | N.C. | >64.00 | 0.12 | N.C. | S |
| C. albicans 002/040 | V.S. | >275.00 | 34.37 | N.C. | >64.00 | 0.12 | N.C. | S |
| C. albicans 001/040 | V.S. | >275.00 | 137.50 | N.C. | >64.00 | 8.00 | N.C. | S |
| C. parapsilosis ATCC 90018 | V.S. | >275.00 | 137.50 | N.C. | 2.00 | 0.25 | 8 | S |
| C. albicans 005/040 | V.S. | >275.00 | 275.00 | N.C. | >64.00 | 0.12 | N.C. | S |
| C. glabrata 005/020 | V.S. | >275.00 | 275.00 | N.C. | 64.00 | 8.00 | 8.00 | S |
| C. parapsilosis 018/020 | V.S. | >275.00 | 275.00 | N.C. | >64.00 | 16.00 | N.C. | S-DD |
| C. tropicalis ATCC 750 | V.S. | >275.00 | 275.00 | N.C. | >64.00 | 2.00 | N.C. | S |
| C. albicans 004/040 | V.S. | >275.00 | >275.00 | N.C. | >64.00 | 0.50 | N.C. | S |
| C. albicans 007/040 | V.S. | >275.00 | >275.00 | N.C. | 32.00 | 2.00 | 16 | S |
| C. albicans ATCC 90028 | V.S. | >275.00 | >275.00 | N.C. | 16.00 | 0.50 | 32 | S |
| C. glabrata ATCC 2001 | V.S. | >275.00 | >275.00 | N.C. | >64.00 | >64.00 | N.C. | R |
| C. krusei 002/020 | V.S. | >275.00 | >275.00 | N.C. | >64.00 | >64.00 | N.C. | R |
| C. krusei ATCC 6258 | V.S. | >275.00 | >275.00 | N.C. | >64.00 | 64.00 | N.C. | R |
| C. tropicalis 011/020 | V.S. | >275.00 | >275.00 | N.C. | >64.00 | 32.00 | N.C. | S-DD |
| Sample | SOD-like Activity | DPPH Test | Total Phenolic | Total Flavonoids |
|---|---|---|---|---|
| IC50 (μg/mL) | Gallic Acid (mg/g) | Catechin (mg/g) | ||
| WPE a | 80 ± 0.51 | 48.35 ± 1.7 | 0.377 ± 0.01 | 0.292 ± 0.08 |
| SOD b | 50 mU ± 0.85 | - | - | - |
| Trolox | - | 15 mM ± 0.62 | - | - |
| Chemical Name | Chemical Class | Chemical Structure | RT * (min) | m/z (g/mol) | Pharmacological Activities |
|---|---|---|---|---|---|
| Protocatechuic acid | Benzoic acid and Phenol derivatives | 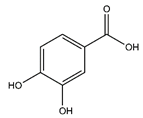 | 35.09 | 154.027 | Antiviral [19] Antibacterial [20] Antioxidant [21] |
| Gallic acid | Benzoic acid and Phenol derivatives |  | 18.60 | 170.021 | Antiviral [22] Antibacterial [23] Antioxidant [21] |
| Ferulic acid | Cinnamic acids and derivatives | 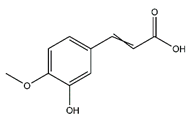 | 36.20 | 194.058 | Antibacterial [23] Antioxidant [24] |
| Sinapate | Cinnamic acids and derivatives | 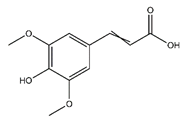 | n.r. | 224.210 | Antibacterial, Antifungal, Antitumor [25] |
| Palmitic acid | Fatty acids | 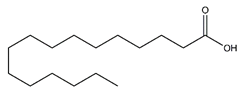 | 12.43 | 256.240 | Antioxidant [26,27] Antitumor [28] |
| Oleic acid | Fatty acids |  | 37.24 | 282.256 | Antioxidant [26,27] Antitumor [28] |
| Ellagic acid | Tannins | 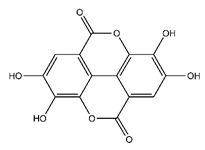 | 33.20 | 302.006 | Antiviral [22] Antioxidant [29] Antitumor [30] |
| Quercetin | Flavonoids | 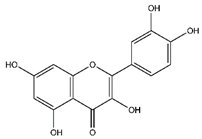 | 33.97 | 302.043 | Antiviral [31,32] Antibacterial [33] Antifungal [34] Antioxidant [24] Antitumor [30] |
| Myricetin | Flavonoids | 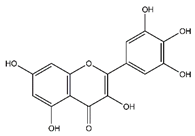 | 33.78 | 318.038 | Antiviral [19] Antifungal [34] Antitumor [30] |
| Chlorogenic acid | Cinnamate ester derivatives | 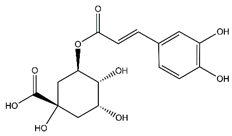 | 39.49 | 354.095 | Antioxidant [35] |
| Beta-sitosterol | Steroids and steroid derivatives |  | 33.89 | 411.386 | Antiviral [36] |
| Kaempferol-arabinoside | Flavonoids | 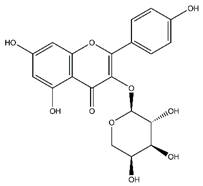 | n.r. | 418.080 | Antiviral [22] |
| Tocopherol | Prenol lipids | 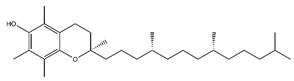 | 36.07 | 430.381 | Antioxidant [26] |
| Avicularin | Flavonoids | 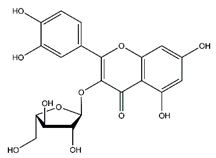 | 20.08 | 434.085 | Antifungal [37] Antitumor [38] |
| Tris-juglone | Phenanthrenes and derivatives | 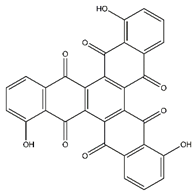 | 36.94 | 516.048 | Anti-SARS-CoV-2 (in-silico analysis) [39] |
| (+)-Procyanidin B2 | Flavonoids |  | 38.25 | 578.520 | Antibacterial, Antioxidant, Antitumor [40] |
| Rutin | Flavonoids | 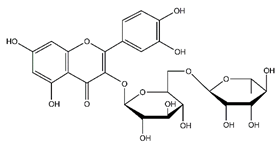 | 45.11 | 610.153 | Antibacterial [41] Antiviral [31,32,42] Antifungal [34] Antioxidant [24] Antitumor [30] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Angeli, F.; Malfa, G.A.; Garozzo, A.; Li Volti, G.; Genovese, C.; Stivala, A.; Nicolosi, D.; Attanasio, F.; Bellia, F.; Ronsisvalle, S.; et al. Antimicrobial, Antioxidant, and Cytotoxic Activities of Juglans regia L. Pellicle Extract. Antibiotics 2021, 10, 159. https://doi.org/10.3390/antibiotics10020159
D’Angeli F, Malfa GA, Garozzo A, Li Volti G, Genovese C, Stivala A, Nicolosi D, Attanasio F, Bellia F, Ronsisvalle S, et al. Antimicrobial, Antioxidant, and Cytotoxic Activities of Juglans regia L. Pellicle Extract. Antibiotics. 2021; 10(2):159. https://doi.org/10.3390/antibiotics10020159
Chicago/Turabian StyleD’Angeli, Floriana, Giuseppe Antonio Malfa, Adriana Garozzo, Giovanni Li Volti, Carlo Genovese, Aldo Stivala, Daria Nicolosi, Francesco Attanasio, Francesco Bellia, Simone Ronsisvalle, and et al. 2021. "Antimicrobial, Antioxidant, and Cytotoxic Activities of Juglans regia L. Pellicle Extract" Antibiotics 10, no. 2: 159. https://doi.org/10.3390/antibiotics10020159
APA StyleD’Angeli, F., Malfa, G. A., Garozzo, A., Li Volti, G., Genovese, C., Stivala, A., Nicolosi, D., Attanasio, F., Bellia, F., Ronsisvalle, S., & Acquaviva, R. (2021). Antimicrobial, Antioxidant, and Cytotoxic Activities of Juglans regia L. Pellicle Extract. Antibiotics, 10(2), 159. https://doi.org/10.3390/antibiotics10020159











