Applications of Lysozyme, an Innate Immune Defense Factor, as an Alternative Antibiotic
Abstract
1. Introduction
2. Sources
2.1. Lysozyme in Eggs
2.2. Lysozyme in Milk
2.3. Saliva, Tears, Various Organs, and Tissues of Mammals
2.4. Aquatic Organisms
2.4.1. Fish
2.4.2. Marine Invertebrates
2.5. Insects
2.6. Plants
2.7. Microorganisms
2.8. Recombinant Human Lysozyme (rhLys)
3. Applications of Lysozyme
3.1. Medical Applications
3.1.1. Skin Diseases
3.1.2. Medical Devices
3.1.3. Biofilms
3.1.4. Oral Care
3.1.5. Respiratory Disorders
3.1.6. Gastrointestinal Tract Diseases
3.1.7. Ophthalmic Applications
3.1.8. Otitis and Sinusitis
3.1.9. Anti-Inflammatory Effects
3.2. Lysozyme as Food Preservative
3.3. Feed Uses of Lysozyme
3.3.1. Poultry
3.3.2. Pigs
3.3.3. Cows
3.3.4. Fish
3.3.5. Rabbits
3.4. Crop Protection
4. Synergistic Activity of Lysozyme
4.1. Antimicrobial Peptides (AMPs)
4.2. Lactoferrin
4.3. EDTA
4.4. Antibiotics
L-Form Bacteria
4.5. Phages
4.6. Selenium Nanoparticles
4.7. Plant Flavones
4.8. Galleria Mellonella Apolipophorin III
5. Lysozyme Modifications
5.1. Immobilization
5.2. Chemical Modifications
5.3. Physical Modifications
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grishin, A.V.; Karyagina, A.S.; Vasina, D.V.; Vasina, I.V.; Gushchin, V.A.; Lunin, V.G. Resistance to peptidoglycan-degrading enzymes. Crit. Rev. Microbiol. 2020, 46, 703–726. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A. On a remarkable bacteriolytic element found in tissues and secretions. Proc. R. Soc. Lond. B 1922, 93, 6–317. [Google Scholar] [CrossRef]
- Callewaert, L.; Michiels, C.W. Lysozymes in the animal kingdom. J. Biosci. 2010, 35, 127–160. [Google Scholar] [CrossRef] [PubMed]
- Jollès, P.; Jollès, J. What’s new in lysozyme research? Always a model system, today as yesterday. Mol. Cell. Biochem. 1984, 63, 165–189. [Google Scholar] [CrossRef]
- Beintema, J.J.; van Scheltinga, A.C.T. Plant lysozymes. EXS 1996, 75, 75–86. [Google Scholar] [CrossRef]
- Fastrez, J. Phage lysozymes. In Lysozymes: Model Enzymes in Biochemistry and Biology; Jollès, P., Ed.; Birkhäuser Basel: Basel, Switzerland, 1996; pp. 35–64. ISBN 978-3-0348-9952-9. [Google Scholar]
- Barbara, L.; Pellegrini, R. Fleming’s Lysozyme: Biological Significance and Therapeutic Applications; Edizioni Minerva Medica: Torino, Italy, 1976. [Google Scholar]
- Wilcox, F.H.; Cole, R.K. The inheritance of differences in the lysozyme level of Hens’ egg white. Genetics 1957, 42, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Sauter, E.A.; Montoure, J.E. The relationship of lysozyme content of egg white to volume and stability of foams. J. Food Sci. 1972, 37, 918–920. [Google Scholar] [CrossRef]
- Shahmohammadi, A. Lysozyme separation from chicken egg white: A review. Eur. Food Res. Technol. 2018, 244, 577–593. [Google Scholar] [CrossRef]
- Lesnierowski, G.; Kijowski, J.; Cegielska-Radziejewska, R. Ultrafiltration-modified chicken egg white lysozyme and its antibacterial action. Int. J. Food Sci. 2009, 44, 305–311. [Google Scholar] [CrossRef]
- Ahlborn, G.; Sheldon, B.W. Enzymatic and microbiological inhibitory activity in eggshell membranes as influenced by layer strains and age and storage variables. Poult. Sci. 2005, 84, 1935–1941. [Google Scholar] [CrossRef]
- Kozuka, M.; Murao, S.; Yamane, T.; Inoue, T.; Ohkubo, I.; Ariga, H. Rapid and simple purification of lysozyme from the egg shell membrane. J. Nutr. Sci. Vitaminol. 2015, 61, 101–103. [Google Scholar] [CrossRef][Green Version]
- Gomathi, S.; Vijitha, M.; Rathnasamy, S. Affinity separation of lysozyme from quail egg (Coturnix ypsilophora) and its antimicrobial characterization. Int. J. PharmTech Res. 2014, 6, 1286. [Google Scholar]
- Myint, S.L.; Kinoshita, K.; Shimogiri, T.; Ibrahim, H.R.; Tsusaki, T.; Tanoue, T.; Kawabe, K.; Maeda, Y.; Okamoto, S. Effect of polymorphism in egg white lysozyme on muramidase and antibacterial activities as well as hatchability in the Japanese quail (Coturnix japonica). J. Anim. Sci. 2012, 90, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Thammasirirak, S.; Pukcothanung, Y.; Preecharram, S.; Daduang, S.; Patramanon, R.; Fukamizo, T.; Araki, T. Antimicrobial peptides derived from goose egg white lysozyme. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 151, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Benkerroum, N. Antimicrobial activity of lysozyme with special relevance to milk. Afr. J. Biotechnol. 2008, 7, 4856–4867. [Google Scholar]
- Cosentino, C.; Labella, C.; Elshafie, H.S.; Camele, I.; Musto, M.; Paolino, R.; D’Adamo, C.; Freschi, P. Effects of different heat treatments on lysozyme quantity and antimicrobial activity of jenny milk. J. Dairy Sci. 2016, 99, 5173–5179. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshini, S.; Kansal, V.K. Purification, characterization, antibacterial activity and N-terminal sequencing of buffalo-milk lysozyme. J. Dairy Res. 2002, 69, 419–431. [Google Scholar] [CrossRef]
- Ibrahim, H.R.; Imazato, K.; Ono, H. Human lysozyme possesses novel antimicrobial peptides within its N-terminal domain that target bacterial respiration. J. Agric. Food Chem. 2011, 59, 10336–10345. [Google Scholar] [CrossRef] [PubMed]
- Fleming, A.; Allison, V.D. Observations on a Bacteriolytic Substance (“Lysozyme”) Found in Secretions and Tissues. Br. J. Exp. Pathol. 1922, 3, 252–260. [Google Scholar]
- Fleming, A.; Allison, V.D. Further observations on a bacteriolytic element found in tissues and secretions. Proc. R. Soc. Lond. B 1922, 94, 142–151. [Google Scholar] [CrossRef]
- Jolles, P. Chimie et Biochimie des Lysozymes: (1) Le Lysozyme de Blanc D’oeuf de Poule: Structure Chimique, Ponts Disulfures, Relations Entre Structure et Activité. (2) Etude Comparée de Quelques Lysozymes D’origines Diverses, 27th ed.; Masson: Paris, France, 1966. [Google Scholar]
- Fábián, T.K.; Hermann, P.; Beck, A.; Fejérdy, P.; Fábián, G. Salivary defense proteins: Their network and role in innate and acquired oral immunity. Int. J. Mol. Sci. 2012, 13, 4295–4320. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Yi, C.; Ming, W.; Tang, M.; Tang, X.; Luo, C.; Lei, B.; Chen, M.; Xu, H. Retinal Pigment Epithelial Cells Express Antimicrobial Peptide Lysozyme—A Novel Mechanism of Innate Immune Defense of the Blood-Retina Barrier. Investig. Ophthalmol. Vis. Sci. 2021, 62, 21. [Google Scholar] [CrossRef] [PubMed]
- Ben Bacha, A.; Abid, I. Secretory phospholipase A2 in dromedary tears: A host defense against staphylococci and other gram-positive bacteria. Appl. Biochem. Biotechnol. 2013, 169, 1858–1869. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Li, S.-J.; Shi, H.; Wang, H.-Y.; Rong, C.-T.; Zhu, P.; Jin, S.-H.; Liu, J.; Li, J.-Y. Characterisation of Lyzls in mice and antibacterial properties of human LYZL6. Asian J. Androl. 2013, 15, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Irwin, D.M. Evolution of cow nonstomach lysozyme genes. Genome 2004, 47, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Chen, Y.; Wang, Y.; Loor, J.J.; Ye, Y.; Wen, Y.; Zi, X.; Cai, Y.; Drackley, J.K. Yak (Bos grunniens) stomach lysozyme: Molecular cloning, expression and its antibacterial activities. Anim. Biotechnol. 2010, 21, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wang, Y.; Jiang, M.; Zhu, L.; Li, J.; Luo, M.; Ren, H.; Liu, L. Molecular cloning, recombinant protein expression, tissue distribution and functional analysis of a new c-type lysozyme from Lezhi black goat rumen. Can. J. Anim. Sci. 2014, 94, 27–34. [Google Scholar] [CrossRef][Green Version]
- Magnadottir, B.; Lange, S.; Gudmundsdottir, S.; Bøgwald, J.; Dalmo, R.A. Ontogeny of humoral immune parameters in fish. Fish Shellfish Immunol. 2005, 19, 429–439. [Google Scholar] [CrossRef]
- Guardiola, F.A.; Cuesta, A.; Abellán, E.; Meseguer, J.; Esteban, M.A. Comparative analysis of the humoral immunity of skin mucus from several marine teleost fish. Fish Shellfish Immunol. 2014, 40, 24–31. [Google Scholar] [CrossRef]
- Sridhar, A.; Krishnasamy Sekar, R.; Manikandan, D.B.; Arumugam, M.; Veeran, S.; Ramasamy, T. Activity profile of innate immune-related enzymes and bactericidal of freshwater fish epidermal mucus extract at different pH. Environ. Sci. Pollut. Res. Int. 2021, 28, 33914–33926. [Google Scholar] [CrossRef]
- Whang, I.; Lee, Y.; Lee, S.; Oh, M.-J.; Jung, S.-J.; Choi, C.Y.; Lee, W.S.; Kim, H.S.; Kim, S.-J.; Lee, J. Characterization and expression analysis of a goose-type lysozyme from the rock bream Oplegnathus fasciatus, and antimicrobial activity of its recombinant protein. Fish Shellfish Immunol. 2011, 30, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Harikrishnan, R.; Kim, J.-S.; Kim, M.-C.; Balasundaram, C.; Heo, M.-S. Molecular characterization, phylogeny, and expression pattern of c-type lysozyme in kelp grouper, Epinephelus bruneus. Fish Shellfish Immunol. 2011, 31, 588–594. [Google Scholar] [CrossRef]
- Zhao, L.; Sun, J.-S.; Sun, L. The g-type lysozyme of Scophthalmus maximus has a broad substrate spectrum and is involved in the immune response against bacterial infection. Fish Shellfish Immunol. 2011, 30, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Hinds Vaughan, N.; Smith, S.L. Isolation and characterization of a c-type lysozyme from the nurse shark. Fish Shellfish Immunol. 2013, 35, 1824–1828. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.H.; Bai, Z.Y.; Xia, J.H.; Liu, F.; Liu, P.; Yue, G.H. Analysis of two lysozyme genes and antimicrobial functions of their recombinant proteins in Asian seabass. PLoS ONE 2013, 8, e79743. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.; Wan, Q.; Bathige, S.D.N.K.; Lee, J. Molecular characterization, transcriptional profiling, and antibacterial potential of g-type lysozyme from seahorse (Hippocampus abdominalis). Fish Shellfish Immunol. 2016, 58, 622–630. [Google Scholar] [CrossRef]
- Lee, J.M.; Kim, S.M. Biochemical and antibacterial properties of lysozyme purified from the viscera of scallops (Patinopecten yessoensis). J. Food Biochem. 2008, 32, 474–489. [Google Scholar] [CrossRef]
- Korobov, V.P.; Lemkina, L.M.; Karnaukhova, I.V.; Poludova, T.V.; Solovykh, G.N.; Pavlova, M.M. Lysozyme of the bivalve mollusk Unio pictorum. Doklady. Biochem. Biophys. 2003, 391, 192–194. [Google Scholar] [CrossRef]
- Yue, X.; Liu, B.; Xue, Q. An i-type lysozyme from the Asiatic hard clam Meretrix meretrix potentially functioning in host immunity. Fish Shellfish Immunol. 2011, 30, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Wu, D.; Zhang, M.; Wen, C.; Xie, Y.; Hu, B.; Jian, S.; Zeng, M.; Tao, Z. Molecular cloning and functional characterization of a novel i-type lysozyme in the freshwater mussel Cristaria plicata. Microbiol. Immunol. 2015, 59, 744–755. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Wang, Q.; Chen, L.; Liu, Y.; Cao, R.; Wu, H.; Li, F.; Ji, C.; Cong, M.; Zhao, J. Molecular characterization and antibacterial activity of a phage-type lysozyme from the Manila clam, Ruditapes philippinarum. Fish Shellfish Immunol. 2017, 65, 17–24. [Google Scholar] [CrossRef]
- Ding, J.; Li, J.; Bao, Y.; Li, L.; Wu, F.; Zhang, G. Molecular characterization of a mollusk chicken-type lysozyme gene from Haliotis discus hannai Ino, and the antimicrobial activity of its recombinant protein. Fish Shellfish Immunol. 2011, 30, 163–172. [Google Scholar] [CrossRef] [PubMed]
- De-la-Re-Vega, E.; García-Galaz, A.; Díaz-Cinco, M.E.; Sotelo-Mundo, R.R. White shrimp (Litopenaeus vannamei) recombinant lysozyme has antibacterial activity against Gram negative bacteria: Vibrio alginolyticus, Vibrio parahemolyticus and Vibrio cholerae. Fish Shellfish Immunol. 2006, 20, 405–408. [Google Scholar] [CrossRef]
- Chen, T.; Ren, C.; Wang, Y.; Luo, P.; Jiang, X.; Huang, W.; Chen, C.; Hu, C. Molecular cloning, inducible expression and antibacterial analysis of a novel i-type lysozyme (lyz-i2) in Pacific white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol. 2016, 54, 197–203. [Google Scholar] [CrossRef]
- Tyagi, A.; Khushiramani, R.; Karunasagar, I.; Karunasagar, I. Antivibrio activity of recombinant lysozyme expressed from black tiger shrimp, Penaeus monodon. Aquaculture 2007, 272, 246–253. [Google Scholar] [CrossRef]
- Mai, W.; Hu, C. cDNA cloning, expression and antibacterial activity of lysozyme C in the blue shrimp (Litopenaeus stylirostris). Prog. Nat. Sci. 2009, 19, 837–844. [Google Scholar] [CrossRef]
- Kaizu, A.; Fagutao, F.F.; Kondo, H.; Aoki, T.; Hirono, I. Functional analysis of C-type lysozyme in penaeid shrimp. J. Biol. Chem. 2011, 286, 44344–44349. [Google Scholar] [CrossRef] [PubMed]
- Mai, W.; Hu, C. Molecular cloning, characterization, expression and antibacterial analysis of a lysozyme homologue from Fenneropenaeus merguiensis. Mol. Biol. Rep. 2009, 36, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Yang, X.; Wang, X.; Tada, M.; Lu, M.; Liu, H.; Zhu, B. Characterization of an i-type lysozyme gene from the sea cucumber Stichopus japonicus, and enzymatic and nonenzymatic antimicrobial activities of its recombinant protein. J. Biosci. Bioeng. 2009, 107, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Kurdyumov, A.S.; Manuvera, V.A.; Baskova, I.P.; Lazarev, V.N. A comparison of the enzymatic properties of three recombinant isoforms of thrombolytic and antibacterial protein–Destabilase-Lysozyme from medicinal leech. BMC Biochem. 2015, 16, 27. [Google Scholar] [CrossRef] [PubMed]
- Yudina, T.G.; Guo, D.; Piskunkova, N.F.; Pavlova, I.B.; Zavalova, L.L.; Baskova, I.P. Antifungal and antibacterial functions of medicinal leech recombinant destabilase-lysozyme and its heated-up derivative. Front. Chem. Sci. Eng. 2012, 6, 203–209. [Google Scholar] [CrossRef]
- Fiołka, M.J.; Ptaszyńska, A.A.; Czarniawski, W. Antibacterial and antifungal lysozyme-type activity in Cameraria ohridella pupae. J. Invertebr. Pathol. 2005, 90, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kajla, M.K.; Andreeva, O.; Gilbreath, T.M.; Paskewitz, S.M. Characterization of expression, activity and role in antibacterial immunity of Anopheles gambiae lysozyme c-1. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2010, 155, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-W.; Park, S.-I.; Yoe, J.; Yoe, S.M. Cloning and overexpression of lysozyme from Spodoptera litura in prokaryotic system. Anim. Cells Syst. 2011, 15, 29–36. [Google Scholar] [CrossRef]
- Kim, J.-W.; Yoe, J.; Lee, G.H.; Yoe, S.M. Recombinant expression and refolding of the c-type lysozyme from Spodoptera litura in E. coli. Electron. J. Biotechnol. 2011, 14, 6. [Google Scholar] [CrossRef]
- Park, S.-I.; Yoe, S.M. Overexpression and purification of recombinant lysozyme from Agrius convolvuli expressed as inclusion body in Escherichia coli. Anim. Cells Syst. 2012, 16, 455–461. [Google Scholar] [CrossRef]
- Sowa-Jasiłek, A.; Zdybicka-Barabas, A.; Stączek, S.; Wydrych, J.; Skrzypiec, K.; Mak, P.; Deryło, K.; Tchórzewski, M.; Cytryńska, M. Galleria mellonella lysozyme induces apoptotic changes in Candida albicans cells. Microbiol. Res. 2016, 193, 121–131. [Google Scholar] [CrossRef]
- Mohrig, W.; Messner, B. Lysozym als antibakterielles Agens im Bienenhoig und Bienengift. Acta Biol. Med. Ger. 1968, 21, 85–95. [Google Scholar]
- Israili, Z.H. Antimicrobial properties of honey. Am. J. Ther. 2014, 21, 304–323. [Google Scholar] [CrossRef]
- Al-Jabri, A.A. Honey, milk and antibiotics. Afr. J. Biotechnol. 2005, 4. [Google Scholar] [CrossRef]
- Wen, S.; Mao, T.-X.; Yao, D.-M.; Li, T.; Wang, F.-H. Yeast Surface Display of Antheraea pernyi Lysozyme Revealed α-Helical Antibacterial Peptides in Its N-Terminal Domain. J. Agric. Food Chem. 2018, 66, 9138–9146. [Google Scholar] [CrossRef]
- Wang, W.-X.; Wang, Y.-P.; Deng, X.-J.; Dang, X.-L.; Tian, J.-H.; Yi, H.-Y.; Li, Y.-F.; He, X.-F.; Cao, Y.; Xia, Q.-Y.; et al. Molecular and functional characterization of a c-type lysozyme from the Asian corn borer, Ostrinia furnacalis. J. Insect Sci. 2009, 9, 17. [Google Scholar] [CrossRef]
- Huang, H.; Du, J.; Li, S.-W.; Gong, T. Identification and Functional Analysis of a Lysozyme Gene from Coridius chinensis (Hemiptera: Dinidoridae). Biology 2021, 10, 330. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ng, T.B.; Chen, T.; Lin, D.; Wu, J.; Rao, P.; Ye, X. First report of a novel plant lysozyme with both antifungal and antibacterial activities. Biochem. Biophys. Res. Commun. 2005, 327, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ye, X.; Rao, P. Isolation of a novel leguminous lysozyme and study on the antifungal activity. Food Res. Int. 2012, 47, 341–347. [Google Scholar] [CrossRef]
- Khémiri, I.; Essghaier, B.; Sadfi-Zouaoui, N.; Bitri, L. Antioxidant and Antimicrobial Potentials of Seed Oil from Carthamus tinctorius L. in the Management of Skin Injuries. Oxid. Med. Cell. Longev. 2020, 2020, 4103418. [Google Scholar] [CrossRef] [PubMed]
- Pendzhiev, A.M. Proteolytic Enzymes of Papaya: Medicinal Applications. Pharm. Chem. J. 2002, 36, 315–317. [Google Scholar] [CrossRef]
- Wang, S.; Shao, B.; Chang, J.; Rao, P. Isolation and identification of a plant lysozyme from Momordica charantia L. Eur. Food Res. Technol. 2011, 232, 613–619. [Google Scholar] [CrossRef]
- Jin, M.; Zhang, T.; Zeng, R. DNA and Protein Sequences of Bacteriophage Lysozyme and Its Use for Preventing and Treating Bacterial Infection. Patent No. CN112662650, 16 April 2021. [Google Scholar]
- Liu, S.-T.; Saito, A.; Azakami, H.; Kato, A. Expression, purification, and characterization of an unstable lysozyme mutant in Pichia pastoris. Protein Expr. Purif. 2003, 27, 304–312. [Google Scholar] [CrossRef]
- Hao, W.-J.; Li, G.-Q.; Xu, M.-Y.; Wei, Z.-R.; Chen, H.-M.; Liu, D.-H.; Ai, T.M. Induction and expression of T4 lysozyme gene in Pichia pastoris. J. Chin. Pharm. Sci. 2007, 16, 33. [Google Scholar]
- Guan, C.; Wang, G.; Ji, J.; Wang, J.; Wang, H.; Tan, M. Bioencapsulation of living yeast (Pichia pastoris) with silica after transformation with lysozyme gene. J. Sol-Gel Sci. Technol. 2008, 48, 369–377. [Google Scholar] [CrossRef]
- Zhang, H.; Fu, G.; Zhang, D. Cloning, characterization, and production of a novel lysozyme by different expression hosts. J. Microbiol. Biotechnol. 2014, 24, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- van Kimmenade, A.; van Solingen, P.; Yao, J. Isolation of Lysozyme A from Trichoderma reesei for Use as Antimicrobial Agent in Detergents and Food Preservation. Patent No. WO2009102755, 20 August 2009. [Google Scholar]
- Wang, Z.; Carter, D. Chalaropsis Lysozyme Protein and Its Use in Antibacterial Applications. Patent No. WO2005011587, 2 October 2005. [Google Scholar]
- Ghasemi, S.; Ahmadian, G.; Sadeghi, M.; Zeigler, D.R.; Rahimian, H.; Ghandili, S.; Naghibzadeh, N.; Dehestani, A. First report of a bifunctional chitinase/lysozyme produced by Bacillus pumilus SG2. Enzyme Microb. Technol. 2011, 48, 225–231. [Google Scholar] [CrossRef]
- Mandal, A.; Herr, J.C. Preparation of Active Human Lysozyme and Transgenic Organisms. Patent No. WO2006028497, 16 March 2006. [Google Scholar]
- Ercan, D.; Demirci, A. Production of human lysozyme in biofilm reactor and optimization of growth parameters of Kluyveromyces lactis K7. Appl. Microbiol. Biotechnol. 2013, 97, 6211–6221. [Google Scholar] [CrossRef]
- Benecky, M.J.; Frew, J.E.; Scowen, N.; Jones, P.; Hoffman, B.M. EPR and ENDOR detection of compound I from Micrococcus lysodeikticus catalase. Biochemistry 1993, 32, 11929–11933. [Google Scholar] [CrossRef]
- Zhou, X.; Yu, Y.; Tao, J.; Yu, L. Production of LYZL6, a novel human c-type lysozyme, in recombinant Pichia pastoris employing high cell density fed-batch fermentation. J. Biosci. Bioeng. 2014, 118, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhao, H.L.; Feng, X.; Zhai, R.D.; Zhu, S.; DU, C.T.; Sun, C.J.; Lei, L.C. Expression of recombinant human lysozyme-tachyplesin I (hLYZ-TP I) in Pichia pastoris and analysis of antibacterial activity. Biomed. Environ. Sci. 2013, 26, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Wang, J.; Tang, B.; Liu, Y.; Guo, C.; Yang, P.; Yu, T.; Li, R.; Zhao, J.; Zhang, L.; et al. Characterization of bioactive recombinant human lysozyme expressed in milk of cloned transgenic cattle. PLoS ONE 2011, 6, e17593. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, G.G.; Mucci, N.C.; González, V.; Sánchez, L.; Parrón, J.A.; Pérez, M.D.; Calvo, M.; Aller, J.F.; Hozbor, F.A.; Mutto, A.A. Detection of recombinant human lactoferrin and lysozyme produced in a bitransgenic cow. J. Dairy Sci. 2017, 100, 1605–1617. [Google Scholar] [CrossRef]
- Liu, R.-Y.; Tian, T.T.; Liu, S.-H.; Huang, B.; Zhang, Y.-Y.; Hui, Y.-T. Study on Bovine Mammary Specific Expression Vector of Expressing Human Lysozyme Gene. J. Anim. Vet. Adv. 2012, 11, 1362–1367. [Google Scholar] [CrossRef]
- Cheng, G. Targeting the human lysozyme gene on bovine αs1-casein gene locus in fibroblasts. Afr. J. Biotechnol. 2011, 10, 17323–17331. [Google Scholar] [CrossRef]
- Wu, X.; Lin, Y.; Xi, Y.; Shao, Z.; Zhou, Y.; Liu, F.; Chen, H. The development of transgenic mice for the expression of large amounts of human lysozyme in milk. Biotechnol. Lett. 2014, 36, 1197–1202. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Li, X.; Lu, D.; Shang, S.; Wang, M.; Zheng, M.; Zhang, R.; Tang, B.; Li, Q.; Dai, Y.; et al. High-level expression of bioactive recombinant human lysozyme in the milk of transgenic mice using a modified human lactoferrin BAC. Transgenic Res. 2012, 21, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z. Expression of recombinant human lysozyme in the milk of transgenic mice. Chin. Sci. Bull. 2003, 48, 2331. [Google Scholar] [CrossRef]
- Yu, Z.; Meng, Q.; Yu, H.; Fan, B.; Yu, S.; Fei, J.; Wang, L.; Dai, Y.; Li, N. Expression and Bioactivity of Recombinant Human Lysozyme in the Milk of Transgenic Mice. J. Dairy Sci. 2006, 89, 2911–2918. [Google Scholar] [CrossRef]
- Tong, J.; Wei, H.; Liu, X.; Hu, W.; Bi, M.; Wang, Y.; Li, Q.; Li, N. Production of recombinant human lysozyme in the milk of transgenic pigs. Transgenic Res. 2011, 20, 417–419. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, I.D.S.; de Menezes, J.N.R.; Maia, J.A.; Miranda, A.M.; de Oliveira, V.B.S.; Murray, J.D.; Maga, E.A.; Bertolini, M.; Bertolini, L.R. Milk from transgenic goat expressing human lysozyme for recovery and treatment of gastrointestinal pathogens. Eur. J. Pharm. Sci. 2018, 112, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Maga, E.A.; Desai, P.T.; Weimer, B.C.; Dao, N.; Kültz, D.; Murray, J.D. Consumption of lysozyme-rich milk can alter microbial fecal populations. Appl. Environ. Microbiol. 2012, 78, 6153–6160. [Google Scholar] [CrossRef]
- Wu, H.; Cao, D.; Liu, T.; Zhao, J.; Hu, X.; Li, N. Purification and Characterization of Recombinant Human Lysozyme from Eggs of Transgenic Chickens. PLoS ONE 2015, 10, e0146032. [Google Scholar] [CrossRef] [PubMed]
- Saarinen, K.M.; Juntunen-Backman, K.; Järvenpää, A.L.; Klemetti, P.; Kuitunen, P.; Lope, L.; Renlund, M.; Siivola, M.; Vaarala, O.; Savilahti, E. Breast-feeding and the development of cows’ milk protein allergy. Adv. Exp. Med. Biol. 2000, 478, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wu, L.; Yalda, D.; Adkins, Y.; Kelleher, S.L.; Crane, M.; Lonnerdal, B.; Rodriguez, R.L.; Huang, N. Expression of functional recombinant human lysozyme in transgenic rice cell culture. Transgenic Res. 2002, 11, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, B.D.; Huang, N.; Klasing, K.C. Rice expressing lactoferrin and lysozyme has antibiotic-like properties when fed to chicks. J. Nutr. 2002, 132, 1214–1218. [Google Scholar] [CrossRef]
- Ibrahim, H.R.; Matsuzaki, T.; Aoki, T. Genetic evidence that antibacterial activity of lysozyme is independent of its catalytic function. FEBS Lett. 2001, 506, 27–32. [Google Scholar] [CrossRef]
- Masschalck, B.; van Houdt, R.; van Haver, E.G.; Michiels, C.W. Inactivation of gram-negative bacteria by lysozyme, denatured lysozyme, and lysozyme-derived peptides under high hydrostatic pressure. Appl. Environ. Microbiol. 2001, 67, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Ginsburg, I. Bactericidal cationic peptides can also function as bacteriolysis-inducing agents mimicking beta-lactam antibiotics? It is enigmatic why this concept is consistently disregarded. Med. Hypotheses 2004, 62, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Nash, J.A.; Ballard, T.N.S.; Weaver, T.E.; Akinbi, H.T. The peptidoglycan-degrading property of lysozyme is not required for bactericidal activity in vivo. J. Immunol. 2006, 177, 519–526. [Google Scholar] [CrossRef]
- Woods, C.M.; Hooper, D.N.; Ooi, E.H.; Tan, L.-W.; Carney, A.S. Human lysozyme has fungicidal activity against nasal fungi. Am. J. Rhinol. Allergy 2011, 25, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Lee-Huang, S.; Huang, P.L.; Sun, Y.; Kung, H.F.; Blithe, D.L.; Chen, H.C. Lysozyme and RNases as anti-HIV components in beta-core preparations of human chorionic gonadotropin. Proc. Natl. Acad. Sci. USA 1999, 96, 2678–2681. [Google Scholar] [CrossRef]
- Karachi, A.; Rajaian, H.; Aminlari, M.M.; Tabatabaee, A. Application of lysozyme and dextran conjugated lysozyme as natural antimicrobial agents in the treatment of experimental skin wound in mice. Int. J. Pharm. Sci. Res. 2013, 4, 4236. [Google Scholar] [CrossRef]
- Öhlknecht, C.; Tegl, G.; Beer, B.; Sygmund, C.; Ludwig, R.; Guebitz, G.M. Cellobiose dehydrogenase and chitosan-based lysozyme responsive materials for antimicrobial wound treatment. Biotechnol. Bioeng. 2017, 114, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Abouhmad, A.; Mamo, G.; Dishisha, T.; Amin, M.A.; Hatti-Kaul, R. T4 lysozyme fused with cellulose-binding module for antimicrobial cellulosic wound dressing materials. J. Appl. Microbiol. 2016, 121, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Ravensdale, J.; Wood, F.; O’Brien, F.; Gregg, K. Investigations into methods to improve the antibacterial activity of Acticoat. J. Med. Microbiol. 2016, 65, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Tavakolian, M.; Okshevsky, M.; van de Ven, T.G.M.; Tufenkji, N. Developing Antibacterial Nanocrystalline Cellulose Using Natural Antibacterial Agents. ACS Appl. Mater. Interfaces 2018, 10, 33827–33838. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Ni, W.; Zhao, X.; Guo, Y.; Li, X.; Wang, F.; Luo, G.; Zhan, R.; Xu, X. A moisture balanced antibacterial dressing loaded with lysozyme possesses antibacterial activity and promotes wound healing. Soft Matter 2021, 17, 3162–3173. [Google Scholar] [CrossRef]
- Liao, A.-H.; Hung, C.-R.; Lin, C.-F.; Lin, Y.-C.; Chen, H.-K. Treatment effects of lysozyme-shelled microbubbles and ultrasound in inflammatory skin disease. Sci. Rep. 2017, 7, 41325. [Google Scholar] [CrossRef]
- Cebrián, R.; Arévalo, S.; Rubiño, S.; Arias-Santiago, S.; Rojo, M.D.; Montalbán-López, M.; Martínez-Bueno, M.; Valdivia, E.; Maqueda, M. Control of Propionibacterium acnes by natural antimicrobial substances: Role of the bacteriocin AS-48 and lysozyme. Sci. Rep. 2018, 8, 11766. [Google Scholar] [CrossRef] [PubMed]
- Di Schiena, M.G.; Ferrari, S.; Rongen, R. Lysozyme Gel Formulations for Use as Disinfectants. U.S. Patent US20130259852, 3 October 2013. [Google Scholar]
- Tan, H.; Jin, D.; Qu, X.; Liu, H.; Chen, X.; Yin, M.; Liu, C. A PEG-Lysozyme hydrogel harvests multiple functions as a fit-to-shape tissue sealant for internal-use of body. Biomaterials 2019, 192, 392–404. [Google Scholar] [CrossRef]
- Ramiro-Gutiérrez, M.L.; Will, J.; Boccaccini, A.R.; Díaz-Cuenca, A. Reticulated bioactive scaffolds with improved textural properties for bone tissue engineering: Nanostructured surfaces and porosity. J. Biomed. Mater. Res. A 2014, 102, 2982–2992. [Google Scholar] [CrossRef] [PubMed]
- Pino-Ramos, V.H.; Flores-Rojas, G.G.; Alvarez-Lorenzo, C.; Concheiro, A.; Bucio, E. Graft copolymerization by ionization radiation, characterization, and enzymatic activity of temperature-responsive SR-g-PNVCL loaded with lysozyme. React. Funct. Polym. 2018, 126, 74–82. [Google Scholar] [CrossRef]
- Yuan, M.; Dai, F.; Li, D.; Fan, Y.; Xiang, W.; Tao, F.; Cheng, Y.; Deng, H. Lysozyme/collagen multilayers layer-by-layer deposited nanofibers with enhanced biocompatibility and antibacterial activity. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 112, 110868. [Google Scholar] [CrossRef] [PubMed]
- Al Meslmani, B.M.; Mahmoud, G.F.; Leichtweiß, T.; Strehlow, B.; Sommer, F.O.; Lohoff, M.D.; Bakowsky, U. Covalent immobilization of lysozyme onto woven and knitted crimped polyethylene terephthalate grafts to minimize the adhesion of broad spectrum pathogens. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 58, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Nezu, T.; Masuyama, T.; Sasaki, K.; Saitoh, S.; Taira, M.; Araki, Y. Effect of pH and addition of salt on the adsorption behavior of lysozyme on gold, silica, and titania surfaces observed by quartz crystal microbalance with dissipation monitoring. Dent. Mater. J. 2008, 27, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Gao, C.; Tang, L.; Wang, C.; Chen, Q.; Zheng, Q.; Yang, S.; Sheng, S.; Zan, X. Lysozyme (Lys), Tannic Acid (TA), and Graphene Oxide (GO) Thin Coating for Antibacterial and Enhanced Osteogenesis. ACS Appl. Bio Mater. 2020, 3, 673–684. [Google Scholar] [CrossRef]
- He, L.; Cui, Y.; Zhang, C. The corrosion resistance, cytotoxicity, and antibacterial properties of lysozyme coatings on orthodontic composite arch wires. RSC Adv. 2020, 10, 18131–18137. [Google Scholar] [CrossRef]
- Budny, J.A.; Budny, M.J. Compositions for Treating Biofilm. U.S. Patent US6830745, 14 December 2004. [Google Scholar]
- Frank, K.L.; Rouchon, C.; Harris, J.A. Antibacterial Methods and Related Kits of Treating a Bacterial Infection Using Lysozyme. Patent No. WO2019018368, 24 January 2019. [Google Scholar]
- Caro, A.; Humblot, V.; Méthivier, C.; Minier, M.; Salmain, M.; Pradier, C.-M. Grafting of lysozyme and/or poly(ethylene glycol) to prevent biofilm growth on stainless steel surfaces. J. Phys. Chem. B 2009, 113, 2101–2109. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Yin, J.; Jiang, W.; Liang, B.; Pehkonen, S.O.; Choong, C. Enhancing antibacterial activity of surface-grafted chitosan with immobilized lysozyme on bioinspired stainless steel substrates. Colloids Surf. B Biointerfaces 2013, 106, 11–21. [Google Scholar] [CrossRef]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Díez-Martínez, R.; de Paz, H.D.; García-Fernández, E.; Bustamante, N.; Euler, C.W.; Fischetti, V.A.; Menendez, M.; García, P. A novel chimeric phage lysin with high in vitro and in vivo bactericidal activity against Streptococcus pneumoniae. J. Antimicrob. Chemother. 2015, 70, 1763–1773. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.; Wang, S.; Lü, M.; Wang, X.; Fang, Y.; Jiao, Y.; Hu, J. Optimization of four types of antimicrobial agents to increase the inhibitory ability of marine Arthrobacter oxydans KQ11 dextranase mouthwash. Chin. J. Ocean. Limnol. 2016, 34, 354–366. [Google Scholar] [CrossRef]
- Okamoto, I.; Miyaji, H.; Miyata, S.; Shitomi, K.; Sugaya, T.; Ushijima, N.; Akasaka, T.; Enya, S.; Saita, S.; Kawasaki, H. Antibacterial and Antibiofilm Photodynamic Activities of Lysozyme-Au Nanoclusters/Rose Bengal Conjugates. ACS Omega 2021, 6, 9279–9290. [Google Scholar] [CrossRef]
- Zhong, X.; Song, Y.; Yang, P.; Wang, Y.; Jiang, S.; Zhang, X.; Li, C. Titanium Surface Priming with Phase-Transited Lysozyme to Establish a Silver Nanoparticle-Loaded Chitosan/Hyaluronic Acid Antibacterial Multilayer via Layer-by-Layer Self-Assembly. PLoS ONE 2016, 11, e0146957. [Google Scholar] [CrossRef] [PubMed]
- Guan, B.; Wang, H.; Xu, R.; Zheng, G.; Yang, J.; Liu, Z.; Cao, M.; Wu, M.; Song, J.; Li, N.; et al. Establishing Antibacterial Multilayer Films on the Surface of Direct Metal Laser Sintered Titanium Primed with Phase-Transited Lysozyme. Sci. Rep. 2016, 6, 36408. [Google Scholar] [CrossRef]
- Sebaa, S.; Hizette, N.; Boucherit-Otmani, Z.; Courtois, P. Dose-dependent effect of lysozyme upon Candida albicans biofilm. Mol. Med. Rep. 2017, 15, 1135–1142. [Google Scholar] [CrossRef] [PubMed]
- Kehail, A.A.; Brigham, C.J. Anti-biofilm Activity of Solvent-Cast and Electrospun Polyhydroxyalkanoate Membranes Treated with Lysozyme. J. Polym. Environ. 2018, 26, 66–72. [Google Scholar] [CrossRef]
- Hou, Y.; Wang, Z.; Zhang, P.; Bai, H.; Sun, Y.; Duan, J.; Mu, H. Lysozyme Associated Liposomal Gentamicin Inhibits Bacterial Biofilm. Int. J. Mol. Sci. 2017, 18, 784. [Google Scholar] [CrossRef] [PubMed]
- Eladawy, M.; El-Mowafy, M.; El-Sokkary, M.M.A.; Barwa, R. Effects of Lysozyme, Proteinase K, and Cephalosporins on Biofilm Formation by Clinical Isolates of Pseudomonas aeruginosa. Interdiscip. Perspect. Infect. Dis. 2020, 2020, 6156720. [Google Scholar] [CrossRef]
- Komal, S.; Abhishek, P.; Mansi, S.; Shilpa, G. Synergistic Effect of Antibiotics and Enzymes as Strategies for Combating Biofilm Formation by Pseudomonas aeruginosa Pao1. Int. J. Pharma. Bio Sci. 2020, 11, 168–178. [Google Scholar] [CrossRef]
- Thellin, O.; Zorzi, W.; Zorzi, D.; Delvenne, P.; Heinen, E.; ElMoualij, B.; Quatresooz, P. Lysozyme as a cotreatment during antibiotics use against vaginal infections: An in vitro study on Gardnerella vaginalis biofilm models. Int. Microbiol. 2016, 19, 101–107. [Google Scholar] [CrossRef]
- Singh, A.; Verma, A.; Singh, R.; Sahoo, A.K.; Samanta, S.K. Combination therapy of biogenic C-dots and lysozyme for enhanced antibacterial and antibiofilm activity. Nanotechnology 2021, 32, 85104. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. Oral Care Solution Containing Lysozyme and Preparation Method Thereof. Patent No. CN112933217, 11 June 2021. [Google Scholar]
- Tong, H.; Xu, Z. Preparation Method of Stain-Removing Whitening Biological Lysozyme Toothpaste Composition and Application. Patent No. CN112773727, 11 May 2021. [Google Scholar]
- Zhang, J. Composite Oral Spray Containing Lysozyme and Its Preparation Method. Patent No. CN112741900, 4 May 2021. [Google Scholar]
- Zhang, X.; Que, K.; Lu, D.; Wu, J.; Zhang, X. Anti-Biomembrane-Remineralization Material for Root Canal Irrigation and Blocking Treatment of Dentin Caries and Preparation Method Thereof. Patent No. CN112716812, 30 April 2021. [Google Scholar]
- Hu, W.; Liu, B.; Hu, H.; Tian, Q.; Mao, E.; Wu, X. Oral Cavity Care Spray Containing Lysozyme for Dog and Cat without Toxic and Side Effects and Preparation Method Thereof. Patent No. CN112315842, 5 February 2021. [Google Scholar]
- Hong, J.-Y.; Lee, J.-S.; Choi, S.-H.; Shin, H.-S.; Park, J.-C.; Shin, S.-I.; Chung, J.-H. A randomized, double-blind, placebo-controlled multicenter study for evaluating the effects of fixed-dose combinations of vitamin C, vitamin E, lysozyme, and carbazochrome on gingival inflammation in chronic periodontitis patients. BMC Oral Health 2019, 19, 40. [Google Scholar] [CrossRef] [PubMed]
- de Andrade, F.B.; de Oliveira, J.C.; Yoshie, M.T.; Guimarães, B.M.; Gonçalves, R.B.; Schwarcz, W.D. Antimicrobial activity and synergism of lactoferrin and lysozyme against cariogenic microorganisms. Braz. Dent. J. 2014, 25, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, S.L.; Azenha, G.R.; de Milito, F.; Democh, Y.M. Antimicrobial Capacity of Casein Phosphopeptide/Amorphous Calcium Phosphate and Enzymes in Glass Ionomer Cement in Dentin Carious Lesions. Acta Stomatol. Croat. 2015, 49, 104–111. [Google Scholar] [CrossRef]
- van Nieuw Amerongen, A.; Bolscher, J.G.M.; Veerman, E.C.I. Salivary proteins: Protective and diagnostic value in cariology? Caries Res. 2004, 38, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Cantor, J.O.; Shteyngart, B. Intratracheal Administration of Lysozyme. U.S. Patent US20010036443, 1 November 2001. [Google Scholar]
- Shteyngart, B. Intratracheal Administration of Lysozyme with Other Therapeutic Agents in the Prevention and Treatment of Respiratory Disorders. U.S. Patent US20050271645, 8 December 2005. [Google Scholar]
- Bhavsar, T.; Liu, M.; Hardej, D.; Liu, X.; Cantor, J. Aerosolized recombinant human lysozyme ameliorates Pseudomonas aeruginosa-induced pneumonia in hamsters. Exp. Lung Res. 2010, 36, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Weaver, T.E.; Akinbi, H.T. Lysozyme and Surfactant Protein B Fusion Protein for Treatment of Bacterial Infections. Patent No. WO2000029588, 25 May 2000. [Google Scholar]
- Wong, G.C.L.; Xian, W.; Sanders, L.K.; Purdy, K.R. Charge-Modified Lysozyme Antimicrobial Compositions, Surfactants, and Methods for Treating Infections in Cystic Fibrosis. Patent No. WO2007053800, 10 May 2007. [Google Scholar]
- Guáqueta, C.; Sanders, L.K.; Wong, G.C.L.; Luijten, E. The effect of salt on self-assembled actin-lysozyme complexes. Biophys. J. 2006, 90, 4630–4638. [Google Scholar] [CrossRef]
- Mohammadinejad, S.; Ghamkhari, B.; Abdolmaleki, S. Stability of actin-lysozyme complexes formed in cystic fibrosis disease. Soft Matter 2016, 12, 6557–6565. [Google Scholar] [CrossRef] [PubMed]
- Mogna, G. Composition Comprising N-Acetylcysteine and/or Microencapsulated Gastroprotected Lysozyme in Association with Probiotic Bacteria Capable of Restoring the Stomach’s Own Barrier Effect Which Is Lost during the Pharmacological Treatment of Gastric Hyperacidity. U.S. Patent US20140328932, 6 November 2014. [Google Scholar]
- Kjaerulff, S.; Cohn, M.T.; Kristensen, N.N. Microbial Lysozyme for Use in the Treatment of Irritable Bowel Syndrome or Inflammatory Bowel Disease. Patent No. WO2018127532, 12 July 2018. [Google Scholar]
- Abimuldina, S.T.; Koftanyuk, N.V.; Kapshakbayeva, Z.V.; Temerbayeva, M.V.; Zhumankulova, E.N. Developing Domestic Food Additive. Indian J. Sci. Technol. 2016, 9, 97656. [Google Scholar] [CrossRef][Green Version]
- Bugla-Płoskońska, G.; Kiersnowski, A.; Futoma-Kołoch, B.; Doroszkiewicz, W. Killing of Gram-negative bacteria with normal human serum and normal bovine serum: Use of lysozyme and complement proteins in the death of Salmonella strains O48. Microb. Ecol. 2009, 58, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, S. Novel Ophthalmic Compositions Containing Human Recombinant Lysozyme and Use Thereof for Treating Eye Conditions and as Contact Lens Solutions. U.S. Patent US20080213188, 4 September 2008. [Google Scholar]
- Wang, B.; Lin, Q.; Jin, T.; Shen, C.; Tang, J.; Han, Y.; Chen, H. Surface modification of intraocular lenses with hyaluronic acid and lysozyme for the prevention of endophthalmitis and posterior capsule opacification. RSC Adv. 2015, 5, 3597–3604. [Google Scholar] [CrossRef]
- Shimada, J.; Moon, S.K.; Lee, H.-Y.; Takeshita, T.; Pan, H.; Woo, J.-I.; Gellibolian, R.; Yamanaka, N.; Lim, D.J. Lysozyme M deficiency leads to an increased susceptibility to Streptococcus pneumoniae-induced otitis media. BMC Infect. Dis. 2008, 8, 134. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.J.; Lee, H.-Y.; Webster, P.; Andalibi, A.; Li, J.-D.; Ganz, T.; Cha, K. Use of Antimicrobial Proteins and Peptides for the Treatment of Otitis Media and Paranasal Sinusitis. U.S. Patent US20060034820, 16 February 2006. [Google Scholar]
- Lee, M.; Kovacs-Nolan, J.; Yang, C.; Archbold, T.; Fan, M.Z.; Mine, Y. Hen egg lysozyme attenuates inflammation and modulates local gene expression in a porcine model of dextran sodium sulfate (DSS)-induced colitis. J. Agric. Food Chem. 2009, 57, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Ku, S.-K.; Na, D.H.; Bae, J.-S. Anti-Inflammatory Effects of Lysozyme against HMGB1 in Human Endothelial Cells and in Mice. Inflammation 2015, 38, 1911–1924. [Google Scholar] [CrossRef] [PubMed]
- Bergamo, A.; Gerdol, M.; Pallavicini, A.; Greco, S.; Schepens, I.; Hamelin, R.; Armand, F.; Dyson, P.J.; Sava, G. Lysozyme-Induced Transcriptional Regulation of TNF-α Pathway Genes in Cells of the Monocyte Lineage. Int. J. Mol. Sci. 2019, 20, 5502. [Google Scholar] [CrossRef]
- D’Incecco, P.; Gatti, M.; Hogenboom, J.A.; Bottari, B.; Rosi, V.; Neviani, E.; Pellegrino, L. Lysozyme affects the microbial catabolism of free arginine in raw-milk hard cheeses. Food Microbiol. 2016, 57, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, W.; García-Ruiz, A.; Recio, I.; Moreno-Arribas, M.V. Antibacterial activity of hen egg white lysozyme modified by heat and enzymatic treatments against oenological lactic acid bacteria and acetic acid bacteria. J. Food Prot. 2014, 77, 1732–1739. [Google Scholar] [CrossRef]
- van Landschoot, A.; Villa, A. Antibacterial properties of hen egg white lysozyme against beer spoilage bacteria and effect of lysozyme on yeast fermentation. Cerevisia 2008, 32, 219–224. [Google Scholar]
- Li, Q.; Xu, J.; Zhang, D.; Zhong, K.; Sun, T.; Li, X.; Li, J. Preparation of a bilayer edible film incorporated with lysozyme and its effect on fish spoilage bacteria. J. Food Saf. 2020, 40, e12832. [Google Scholar] [CrossRef]
- Park, S.-I.; Daeschel, M.A.; Zhao, Y. Functional Properties of Antimicrobial Lysozyme-Chitosan Composite Films. J. Food Sci. 2004, 69, M215–M221. [Google Scholar] [CrossRef]
- Amiri, S.; Ramezani, R.; Aminlari, M. Antibacterial activity of dextran-conjugated lysozyme against Escherichia coli and Staphylococcus aureus in cheese curd. J. Food Prot. 2008, 71, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Amara, C.B.; Eghbal, N.; Oulahal, N.; Degraeve, P.; Gharsallaoui, A. Properties of lysozyme/sodium alginate complexes for the development of antimicrobial films. Food Res. Int. 2016, 89, 272–280. [Google Scholar] [CrossRef]
- Wu, T.; Wu, C.; Fu, S.; Wang, L.; Yuan, C.; Chen, S.; Hu, Y. Integration of lysozyme into chitosan nanoparticles for improving antibacterial activity. Carbohydr. Polym. 2017, 155, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Tu, H.; Huang, M.; Chen, J.; Shi, X.; Deng, H.; Wang, S.; Du, Y. Incorporation of lysozyme-rectorite composites into chitosan films for antibacterial properties enhancement. Int. J. Biol. Macromol. 2017, 102, 789–795. [Google Scholar] [CrossRef]
- Hashemi, M.; Aminlari, M.; Forouzan, M.; Moghimi, E.; Tavana, M.; Shekarforoush, S.; Mohammadifar, M. Production and Application of Lysozyme-Gum Arabic Conjugate in Mayonnaise as a Natural Preservative and Emulsifier. Pol. J. Food Nutr. Sci. 2018, 68, 33–43. [Google Scholar] [CrossRef]
- Xu, Y.; Yin, Y.; Li, T.; Zhao, H.; Li, X.; Li, J.; Sun, T. Effects of lysozyme combined with cinnamaldehyde on storage quality of olive flounder (Paralichthys olivaceus) fillets. J. Food Sci. 2020, 85, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Palotás, P.; Jónás, G.; Lehel, J.; Friedrich, L. Preservative Effect of Novel Combined Treatment with Electrolyzed Active Water and Lysozyme Enzyme to Increase the Storage Life of Vacuum-Packaged Carp. J. Food Qual. 2020, 2020, 4861471. [Google Scholar] [CrossRef]
- Cegielska-Radziejewska, R.; Szablewski, T.; Radziejewska-Kubzdela, E.; Tomczyk, Ł.; Biadała, A.; Leśnierowski, G. The Effect of Modified Lysozyme Treatment on the Microflora, Physicochemical and Sensory Characteristics of Pork Packaged in Preservative Gas Atmospheres. Coatings 2021, 11, 488. [Google Scholar] [CrossRef]
- López-Pedemonte, T.J.; Roig-Sagués, A.X.; Trujillo, A.J.; Capellas, M.; Guamis, B. Inactivation of Spores of Bacillus cereus in Cheese by High Hydrostatic Pressure with the Addition of Nisin or Lysozyme. J. Dairy Sci. 2003, 86, 3075–3081. [Google Scholar] [CrossRef]
- Cannarsi, M.; Baiano, A.; Sinigaglia, M.; Ferrara, L.; Baculo, R.; Del Nobile, M.A. Use of nisin, lysozyme and EDTA for inhibiting microbial growth in chilled buffalo meat. Int. J. Food Sci. 2008, 43, 573–578. [Google Scholar] [CrossRef]
- Apriliyani, M.W.; Rosyidi, D.; Purwadi, P.; Purnomo, H.; Manab, A. The Release of Egg White Lysozyme Containing EDTA from Composite Edible Film Based on Whey Protein, Konjac Flour and Lipid. Adv. J. Food Sci. Technol. 2014, 6, 48–55. [Google Scholar] [CrossRef]
- Rao, M.S.; Chander, R.; Sharma, A. Synergistic effect of chitooligosaccharides and lysozyme for meat preservation. LWT-Food Sci. Technol. 2008, 41, 1995–2001. [Google Scholar] [CrossRef]
- Huang, W.; Xu, H.; Xue, Y.; Huang, R.; Deng, H.; Pan, S. Layer-by-layer immobilization of lysozyme–chitosan–organic rectorite composites on electrospun nanofibrous mats for pork preservation. Food Res. Int. 2012, 48, 784–791. [Google Scholar] [CrossRef]
- Bhatia, S.; Bharti, A. Evaluating the antimicrobial activity of Nisin, Lysozyme and Ethylenediaminetetraacetate incorporated in starch based active food packaging film. J. Food Sci. Technol. 2015, 52, 3504–3512. [Google Scholar] [CrossRef]
- Ananou, S.; Rivera, S.; Madrid, M.I.; Maqueda, M.; Martínez-Bueno, M.; Valdivia, E. Application of enterocin AS-48 as biopreservative in eggs and egg fractions: Synergism through lysozyme. LWT 2018, 89, 409–417. [Google Scholar] [CrossRef]
- Khodanazary, A. Quality characteristics of refrigerated mackerel Scomberomorus commerson using gelatin-polycaprolactone composite film incorporated with lysozyme and pomegranate peel extract. Int. J. Food Prop. 2019, 22, 2057–2071. [Google Scholar] [CrossRef]
- Lucero Estrada, C.S.; Del Carmen Velázquez, L.; de Guzmán, A.M.S. Effects of organic acids, nisin, lysozyme and EDTA on the survival of Yersinia enterocolitica population in inoculated orange beverages. J. Food Saf. 2010, 30, 24–39. [Google Scholar] [CrossRef]
- Landers, T.F.; Cohen, B.; Wittum, T.E.; Larson, E.L. A review of antibiotic use in food animals: Perspective, policy, and potential. Public Health Rep. 2012, 127, 4–22. [Google Scholar] [CrossRef] [PubMed]
- Casewell, M.; Friis, C.; Marco, E.; McMullin, P.; Phillips, I. The European ban on growth-promoting antibiotics and emerging consequences for human and animal health. J. Antimicrob. Chemother. 2003, 52, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Abouelezz, K.F.M.; Li, L.; Gou, Z.; Wang, Y.; Lin, X.; Ye, J.; Jiang, S. Influence of Mushroom Polysaccharide, Nano-Copper, Copper Loaded Chitosan, and Lysozyme on Intestinal Barrier and Immunity of LPS-mediated Yellow-Feathered Chickens. Animals 2020, 10, 594. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Kong, J.; Zhang, G.; Zhang, X.; Seviour, R.; Kong, Y. Effects of dietary supplementation with lysozyme on the structure and function of the cecal microbiota in broiler chickens. PLoS ONE 2019, 14, e0216748. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Guo, Y.; Wang, Z.; Yuan, J. Exogenous lysozyme influences Clostridium perfringens colonization and intestinal barrier function in broiler chickens. Avian Pathol. 2010, 39, 17–24. [Google Scholar] [CrossRef]
- Gong, M.; Anderson, D.; Rathgeber, B.; MacIsaac, J. The effect of dietary lysozyme with EDTA on growth performance and intestinal microbiota of broiler chickens in each period of the growth cycle. J. Appl. Poult. Res. 2017, 26, 1–8. [Google Scholar] [CrossRef]
- Smith, S.R.; Ritchie, S.J.; Zhang, G. Antimicrobial Compositions Containing Lysozyme and Method for. Use. Patent No. WO2004026334, 1 April 2004. [Google Scholar]
- Zhao, P.; Cao, D.; Xiao, H.; Li, X.; Huang, Y.; Jiang, L.; Gan, X.; Deng, J.; Chen, Q.; Wen, X.; et al. Bacteriostatic Animal Growth Promoter Containing Recombinant Human Lysozyme and Application Thereof. Patent No. CN112515050, 19 March 2021. [Google Scholar]
- Oliver, W.T.; Wells, J.E. Lysozyme as an alternative to growth promoting antibiotics in swine production. J. Anim. Sci. Biotechnol. 2015, 6, 35. [Google Scholar] [CrossRef]
- Vanrolleghem, W.; Tanghe, S.; Verstringe, S.; Bruggeman, G.; Papadopoulos, D.; Trevisi, P.; Zentek, J.; Sarrazin, S.; Dewulf, J. Potential dietary feed additives with antibacterial effects and their impact on performance of weaned piglets: A meta-analysis. Vet. J. 2019, 249, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Fu, J.; Zhu, L.; Li, Z.; Jin, M.; Wang, Y. Overall assessment of antibiotic substitutes for pigs: A set of meta-analyses. J. Anim. Sci. Biotechnol. 2021, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Nyachoti, C.M.; Kiarie, E.; Bhandari, S.K.; Zhang, G.; Krause, D.O. Weaned pig responses to Escherichia coli K88 oral challenge when receiving a lysozyme supplement. J. Anim. Sci. 2012, 90, 252–260. [Google Scholar] [CrossRef]
- May, K.D.; Wells, J.E.; Maxwell, C.V.; Oliver, W.T. Granulated lysozyme as an alternative to antibiotics improves growth performance and small intestinal morphology of 10-day-old pigs. J. Anim. Sci. 2012, 90, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Oliver, W.T.; Wells, J.E. Lysozyme as an alternative to antibiotics improves growth performance and small intestinal morphology in nursery pigs. J. Anim. Sci. 2013, 91, 3129–3136. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.E.; Berry, E.D.; Kalchayanand, N.; Rempel, L.A.; Kim, M.; Oliver, W.T. Effect of lysozyme or antibiotics on faecal zoonotic pathogens in nursery pigs. J. Appl. Microbiol. 2015, 118, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Long, Y.; Lin, S.; Zhu, J.; Pang, X.; Fang, Z.; Lin, Y.; Che, L.; Xu, S.; Li, J.; Huang, Y.; et al. Effects of dietary lysozyme levels on growth performance, intestinal morphology, non-specific immunity and mRNA expression in weanling piglets. Anim. Sci. J. 2016, 87, 411–418. [Google Scholar] [CrossRef]
- Zou, L.; Xiong, X.; Liu, H.; Zhou, J.; Liu, Y.; Yin, Y. Effects of dietary lysozyme levels on growth performance, intestinal morphology, immunity response and microbiota community of growing pigs. J. Sci. Food Agric. 2019, 99, 1643–1650. [Google Scholar] [CrossRef] [PubMed]
- Oliver, W.T.; Wells, J.E.; Maxwell, C.V. Lysozyme as an alternative to antibiotics improves performance in nursery pigs during an indirect immune challenge. J. Anim. Sci. 2014, 92, 4927–4934. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Xiong, X.; Zou, L.; Yin, J.; Wang, K.; Shao, Y.; Yin, Y. Dietary lysozyme supplement alters serum biochemical makers and milk metabolite profile of sows via gut microbiota. bioRxiv 2018, 444778. [Google Scholar] [CrossRef]
- Park, J.H.; Sureshkumar, S.; Kim, I.H. Effects of dietary lysozyme supplementation on growth performance, nutrient digestibility, intestinal microbiota, and blood profiles of weanling pigs challenged with Escherichia coli. J. Anim. Sci. Technol. 2021, 63, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Li, X.; Lu, D.; Liu, S.; Suo, X.; Li, Q.; Li, N. Lysozyme improves gut performance and protects against enterotoxigenic Escherichia coli infection in neonatal piglets. Vet. Res. 2018, 49, 20. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.; Li, J.; Zhang, L.; Liu, S.; Wen, X.; Li, Q.; Zhao, Y.; Hu, X.; Zhang, R.; Li, N. Generation of bi-transgenic pigs overexpressing human lactoferrin and lysozyme in milk. Transgenic Res. 2015, 24, 365–373. [Google Scholar] [CrossRef]
- Rathi, C.L.; Risbud, S.P.; Manohar, B.M. Novel Compositions Comprising Lysozyme and Serratiopeptidase for Prevention and Treatment of Mastitis and Metritis. Patent No. WO2008035370, 27 March 2008. [Google Scholar]
- Ye, G.; Huang, Q. Biological Enzyme Disinfectant Preparation and Preparation Method Thereof. Patent No. CN112999337, 22 June 2021. [Google Scholar]
- Smulski, S.; Gehrke, M.; Libera, K.; Cieslak, A.; Huang, H.; Patra, A.K.; Szumacher-Strabel, M. Effects of various mastitis treatments on the reproductive performance of cows. BMC Vet. Res. 2020, 16, 99. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Xue, F.; Qian, K.; Fang, H.; Qiu, H.; Zhang, X.; Yin, Z. Intramammary expression and therapeutic effect of a human lysozyme-expressing vector for treating bovine mastitis. J. Zhejiang Univ. Sci. B 2006, 7, 324–330. [Google Scholar] [CrossRef]
- Adduci, F.; Elshafie, H.S.; Labella, C.; Musto, M.; Freschi, P.; Paolino, R.; Ragni, M.; Cosentino, C. Abatement of the clostridial load in the teats of lactating cows with lysozyme derived from donkey milk. J. Dairy Sci. 2019, 102, 6750–6755. [Google Scholar] [CrossRef] [PubMed]
- Małaczewska, J.; Kaczorek-Łukowska, E.; Wójcik, R.; Siwicki, A.K. Antiviral effects of nisin, lysozyme, lactoferrin and their mixtures against bovine viral diarrhoea virus. BMC Vet. Res. 2019, 15, 318. [Google Scholar] [CrossRef]
- Kumaresan, V.; Bhatt, P.; Ganesh, M.-R.; Harikrishnan, R.; Arasu, M.; Al-Dhabi, N.A.; Pasupuleti, M.; Marimuthu, K.; Arockiaraj, J. A novel antimicrobial peptide derived from fish goose type lysozyme disrupts the membrane of Salmonella enterica. Mol. Immunol. 2015, 68, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Verlhac, V. Feed Additives for Aquatic Animals Comprising Essential Oils and Lysozyme. Patent No. WO2017064092, 20 April 2017. [Google Scholar]
- Yousefi, M.; Vatnikov, Y.A.; Kulikov, E.V.; Plushikov, V.G.; Drukovsky, S.G.; Hoseinifar, S.H.; van Doan, H. The protective effects of dietary garlic on common carp (Cyprinus carpio) exposed to ambient ammonia toxicity. Aquaculture 2020, 526, 735400. [Google Scholar] [CrossRef]
- Elayaraja, S.; Mabrok, M.; Algammal, A.; Sabitha, E.; Rajeswari, M.V.; Zágoršek, K.; Ye, Z.; Zhu, S.; Rodkhum, C. Potential influence of jaggery-based biofloc technology at different C:N ratios on water quality, growth performance, innate immunity, immune-related genes expression profiles, and disease resistance against Aeromonas hydrophila in Nile tilapia (Oreochromis niloticus). Fish Shellfish Immunol. 2020, 107, 118–128. [Google Scholar] [CrossRef] [PubMed]
- El-Deep, M.H.; Amber, K.A.; Eid, Y.Z.; Aboelenin, S.M.; Soliman, M.M.; Sakr, M.S.; Dawood, M.A.O. The Influence of Chicken Egg Lysozyme or Zinc-Bacitracin Antibiotic on the Growth Performance, Antibacterial Capacity, Blood Profiles, and Antioxidative Status of Rabbits: A Comparative Study. Animals 2021, 11, 1731. [Google Scholar] [CrossRef] [PubMed]
- Wackernagel, W.; Duering, K. Transgenic potato plants expressing antimicrobial activity: Establishment of pest control and environmental safety. Forum Nutr. 2003, 56, 317–318. [Google Scholar]
- Chen, D.M.; Yang, H.J.; Huang, J.G.; Yuan, L. Lysobacter enzymogenes LE16 autolysates have potential as biocontrol agents—Lysobacter sp. autolysates as biofungicide. J. Appl. Microbiol. 2020, 129, 1684–1692. [Google Scholar] [CrossRef]
- Rasche, F.; Velvis, H.; Zachow, C.; Berg, G.; van Elsas, J.D.; Sessitsch, A. Impact of transgenic potatoes expressing anti-bacterial agents on bacterial endophytes is comparable with the effects of plant genotype, soil type and pathogen infection. J. Appl. Ecol. 2006, 43, 555–566. [Google Scholar] [CrossRef]
- Salm, H.; Kim, W.S.; Geider, K.; Schneider, B. Control of fire blight with a lysozyme from the Erwinia amylovora phage EA1H. Acta Hortic. 2006, 371–374. [Google Scholar] [CrossRef]
- Cabrefiga, J.; Montesinos, E. Lysozyme enhances the bactericidal effect of BP100 peptide against Erwinia amylovora, the causal agent of fire blight of rosaceous plants. BMC Microbiol. 2017, 17, 39. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuji, T.; Gallo, R.L. Antimicrobial peptides: Old molecules with new ideas. J. Investig. Dermatol. 2012, 132, 887–895. [Google Scholar] [CrossRef]
- Hancock, R.E.; Scott, M.G. The role of antimicrobial peptides in animal defenses. Proc. Natl. Acad. Sci. USA 2000, 97, 8856–8861. [Google Scholar] [CrossRef]
- Scott, M.G.; Hancock, R.E. Cationic antimicrobial peptides and their multifunctional role in the immune system. Crit. Rev. Immunol. 2000, 20, 407–431. [Google Scholar] [CrossRef]
- Yan, H.; Hancock, R.E. Synergistic interactions between mammalian antimicrobial defense peptides. Antimicrob. Agents Chemother. 2001, 45, 1558–1560. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, I.; Davis, L.R.; Berman, C.M.; Griswold, K.E. Nonclassical antagonism between human lysozyme and AMPs against Pseudomonas aeruginosa. FEBS Open Bio 2021, 11, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Patrzykat, A.; Zhang, L.; Mendoza, V.; Iwama, G.K.; Hancock, R.E. Synergy of histone-derived peptides of coho salmon with lysozyme and flounder pleurocidin. Antimicrob. Agents Chemother. 2001, 45, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Pérez, C.; Gracia, B.; Rodrigues, L.; Vitoria, A.; Cebrián, R.; Deboosère, N.; Song, O.-R.; Brodin, P.; Maqueda, M.; Aínsa, J.A. Synergy between Circular Bacteriocin AS-48 and Ethambutol against Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2018, 62, e00359-18. [Google Scholar] [CrossRef]
- Ellison, R.T.; Giehl, T.J. Killing of gram-negative bacteria by lactoferrin and lysozyme. J. Clin. Investig. 1991, 88, 1080–1091. [Google Scholar] [CrossRef]
- André, G.O.; Politano, W.R.; Mirza, S.; Converso, T.R.; Ferraz, L.F.C.; Leite, L.C.C.; Darrieux, M. Combined effects of lactoferrin and lysozyme on Streptococcus pneumoniae killing. Microb. Pathog. 2015, 89, 7–17. [Google Scholar] [CrossRef]
- Singh, P.K.; Tack, B.F.; McCray, P.B.; Welsh, M.J. Synergistic and additive killing by antimicrobial factors found in human airway surface liquid. Am. J. Physiol. Lung Cell. Mol. Physiol. 2000, 279, L799–L805. [Google Scholar] [CrossRef] [PubMed]
- Boland, J.S.; Davidson, P.M.; Bruce, B.; Weiss, J. Cations reduce antimicrobial efficacy of lysozyme-chelator combinations. J. Food Prot. 2004, 67, 285–294. [Google Scholar] [CrossRef]
- Boland, J.S.; Davidson, P.M.; Weiss, J. Enhanced inhibition of Escherichia coli O157:H7 by lysozyme and chelators. J. Food Prot. 2003, 66, 1783–1789. [Google Scholar] [CrossRef]
- Branen, J.K.; Davidson, P. Enhancement of nisin, lysozyme, and monolaurin antimicrobial activities by ethylenediaminetetraacetic acid and lactoferrin. Int. J. Food Microbiol. 2004, 90, 63–74. [Google Scholar] [CrossRef]
- Penta, J.; Jannu, K.K.; Musthyala, R. Antimicrobial studies of selected antibiotics and their combination with enzymes. Int. J. Pharm. Pharm. Sci. 2010, 2, 43–44. [Google Scholar]
- Perez, H.A.; Bustos, A.Y.; Taranto, M.P.; Frías, M.D.L.A.; Ledesma, A.E. Effects of Lysozyme on the Activity of Ionic of Fluoroquinolone Species. Molecules 2018, 23, 741. [Google Scholar] [CrossRef] [PubMed]
- Das, I.; Halder, M. Counterpointing Scenarios on the Fate of Different Prototropic Forms of Norfloxacin Housed in the Pocket of Lysozyme: The Nonelectrostatic Interactions in the Protein Interior Are in the Controlling Role on the Prototropic Equilibria of the Guest. ACS Omega 2017, 2, 5504–5517. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.S.; Waseem, M.; Subbarao, N.; Al-Lohedan, H.A. Dynamic interaction between lysozyme and ceftazidime: Experimental and molecular simulation approaches. J. Mol. Liq. 2021, 328, 115412. [Google Scholar] [CrossRef]
- Bhavsar, T.; Liu, M.; Liu, X.; Cantor, J. Aerosolized recombinant human lysozyme enhances the bactericidal effect of tobramycin in a hamster model of Pseudomonas aeruginosa-induced pneumonia. Exp. Lung Res. 2011, 37, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Torrens, G.; Pérez-Gallego, M.; Moya, B.; Munar-Bestard, M.; Zamorano, L.; Cabot, G.; Blázquez, J.; Ayala, J.A.; Oliver, A.; Juan, C. Targeting the permeability barrier and peptidoglycan recycling pathways to disarm Pseudomonas aeruginosa against the innate immune system. PLoS ONE 2017, 12, e0181932. [Google Scholar] [CrossRef]
- Torrens, G.; Barceló, I.M.; Pérez-Gallego, M.; Escobar-Salom, M.; Tur-Gracia, S.; Munar-Bestard, M.; Del González-Nicolau, M.M.; Cabrera-Venegas, Y.J.; Rigo-Rumbos, E.N.; Cabot, G.; et al. Profiling the susceptibility of Pseudomonas aeruginosa strains from acute and chronic infections to cell-wall-targeting immune proteins. Sci. Rep. 2019, 9, 3575. [Google Scholar] [CrossRef]
- Vouillamoz, J.; Entenza, J.M.; Giddey, M.; Fischetti, V.A.; Moreillon, P.; Resch, G. Bactericidal synergism between daptomycin and the phage lysin Cpl-1 in a mouse model of pneumococcal bacteraemia. Int. J. Antimicrob. Agents 2013, 42, 416–421. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, A.; Yu, H.; Xiong, Y.; Zhou, G.; Qin, M.; Dou, J.; Wang, J. Human Lysozyme Synergistically Enhances Bactericidal Dynamics and Lowers the Resistant Mutant Prevention Concentration for Metronidazole to Helicobacter pylori by Increasing Cell Permeability. Molecules 2016, 21, 1435. [Google Scholar] [CrossRef] [PubMed]
- Zharkova, M.S.; Orlov, D.S.; Golubeva, O.Y.; Chakchir, O.B.; Eliseev, I.E.; Grinchuk, T.M.; Shamova, O.V. Application of Antimicrobial Peptides of the Innate Immune System in Combination With Conventional Antibiotics-A Novel Way to Combat Antibiotic Resistance? Front. Cell. Infect. Microbiol. 2019, 9, 128. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.; Zhang, Y.; Chen, P.; Xue, J.; Jia, X.; Chen, J. Enhanced Synergistic Antibacterial Activity through a Smart Platform Based on UiO-66 Combined with Photodynamic Therapy and Chemotherapy. Langmuir 2020, 36, 4025–4032. [Google Scholar] [CrossRef] [PubMed]
- Kawai, Y.; Mickiewicz, K.; Errington, J. Lysozyme Counteracts β-Lactam Antibiotics by Promoting the Emergence of L-Form Bacteria. Cell 2018, 172, 1038–1049.e10. [Google Scholar] [CrossRef] [PubMed]
- Burke, T.P. The Unexpected Effects of the Combination of Antibiotics and Immunity. Cell 2018, 172, 891–893. [Google Scholar] [CrossRef]
- Mateus, L.; Costa, L.; Silva, Y.J.; Pereira, C.; Almeida, A. Effect of lysozyme addition on the activity of phages against Vibrio parahaemolyticus. Aquaculture 2014, 432, 125–129. [Google Scholar] [CrossRef]
- Vahdati, M.; Tohidi Moghadam, T. Synthesis and Characterization of Selenium Nanoparticles-Lysozyme Nanohybrid System with Synergistic Antibacterial Properties. Sci. Rep. 2020, 10, 510. [Google Scholar] [CrossRef]
- Hariri, B.M.; McMahon, D.B.; Chen, B.; Adappa, N.D.; Palmer, J.N.; Kennedy, D.W.; Lee, R.J. Plant flavones enhance antimicrobial activity of respiratory epithelial cell secretions against Pseudomonas aeruginosa. PLoS ONE 2017, 12, e0185203. [Google Scholar] [CrossRef] [PubMed]
- Zdybicka-Barabas, A.; Stączek, S.; Mak, P.; Skrzypiec, K.; Mendyk, E.; Cytryńska, M. Synergistic action of Galleria mellonella apolipophorin III and lysozyme against Gram-negative bacteria. Biochim. Biophys. Acta 2013, 1828, 1449–1456. [Google Scholar] [CrossRef]
- Homaei, A.A.; Sariri, R.; Vianello, F.; Stevanato, R. Enzyme immobilization: An update. J. Chem. Biol. 2013, 6, 185–205. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Schoevaart, R.; van Langen, L.M. Cross-linked enzyme aggregates (CLEAs): A novel and versatile method for enzyme immobilization (a review). Biocatal. Biotransform. 2005, 23, 141–147. [Google Scholar] [CrossRef]
- Alves, C.R.; Pimenta, M.G.R.; Vieira, R.H.; Furtado, R.F.; Guedes, M.I.F.; Silva, R.C.; Assis, O.B. Practical use of immobilized lysozyme for the remediation process of Escherichia coli in aqueous solution. Electron. J. Biotechnol. 2007, 10, 160–165. [Google Scholar] [CrossRef]
- Wang, Q.; Fan, X.; Hu, Y.; Yuan, J.; Cui, L.; Wang, P. Antibacterial functionalization of wool fabric via immobilizing lysozymes. Bioprocess Biosyst. Eng. 2009, 32, 633–639. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, N.; Wang, Q.; Wang, P.; Yu, Y. Development of an eco-friendly antibacterial textile: Lysozyme immobilization on wool fabric. Bioprocess Biosyst. Eng. 2020, 43, 1639–1648. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.V.; Prevost, N.; Condon, B.; Sethumadhavan, K.; Ullah, J.; Bopp, A. Immobilization of Lysozyme on Cotton Fabrics: Synthesis, Characterization, and Activity. AATCC Rev. 2011, 11, 73–79. [Google Scholar]
- Dickerson, M.B.; Knight, C.L.; Gupta, M.K.; Luckarift, H.R.; Drummy, L.F.; Jespersen, M.L.; Johnson, G.R.; Naik, R.R. Hybrid fibers containing protein-templated nanomaterials and biologically active components as antibacterial materials. Mater. Sci. Eng. C 2011, 31, 1748–1758. [Google Scholar] [CrossRef]
- Liu, W.; Cai, M.; He, Y.; Wang, S.; Zheng, J.; Xu, X. Development of antibacterial polyacrylonitrile membrane modified with a covalently immobilized lysozyme. RSC Adv. 2015, 5, 84432–84438. [Google Scholar] [CrossRef]
- Fang, J.; Liu, G.; Chen, C.; Lin, C.; Zhang, B.; Jin, H.; Chen, Y.; Lu, J.; Zhu, L. Intrinsically antibacterial thin film composite membranes with supramolecularly assembled lysozyme nanofilm as selective layer for molecular separation. Sep. Pur. Technol. 2021, 254, 117585. [Google Scholar] [CrossRef]
- Kim, M.; Park, J.-M.; Yoon, J.; Min, J.; Kim, Y.-H. Synthesis and characterization of CLEA-lysozyme immobilized PS/PSMA nanofiber. J. Nanosci. Nanotechnol. 2011, 11, 7894–7900. [Google Scholar] [CrossRef]
- Teixeira-Dias, B.; Del Valle, L.J.; Aradilla, D.; Estrany, F.; Alemán, C. A Conducting Polymer/Protein Composite with Bactericidal and Electroactive Properties. Macromol. Mater. Eng. 2012, 297, 427–436. [Google Scholar] [CrossRef]
- Li, W.; Li, X.; Wang, Q.; Pan, Y.; Wang, T.; Wang, H.; Song, R.; Deng, H. Antibacterial activity of nanofibrous mats coated with lysozyme-layered silicate composites via electrospraying. Carbohydr. Polym. 2014, 99, 218–225. [Google Scholar] [CrossRef]
- Dekina, S.; Romanovska, I.; Ovsepyan, A.; Tkach, V.; Muratov, E. Gelatin/carboxymethyl cellulose mucoadhesive films with lysozyme: Development and characterization. Carbohydr. Polym. 2016, 147, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Harvey, A.M.; Mailloux, S.; Halámek, J.; Bocharova, V.; Twiss, M.R.; Katz, E. Electrochemically stimulated release of lysozyme from an alginate matrix cross-linked with iron cations. J. Mater. Chem. 2012, 22, 19523. [Google Scholar] [CrossRef]
- Park, J.-M.; Kim, M.; Park, H.-S.; Jang, A.; Min, J.; Kim, Y.-H. Immobilization of lysozyme-CLEA onto electrospun chitosan nanofiber for effective antibacterial applications. Int. J. Biol. Macromol. 2013, 54, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Huang, J.; Jiang, Y.; Hu, Y.; Ye, X.; Liu, D.; Chen, J. Formation of hydrogels based on chitosan/alginate for the delivery of lysozyme and their antibacterial activity. Food Chem. 2018, 240, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.M.; Pereira, R.C.; Leonor, I.B.; Azevedo, H.S.; Reis, R.L. Chitosan scaffolds incorporating lysozyme into CaP coatings produced by a biomimetic route: A novel concept for tissue engineering combining a self-regulated degradation system with in situ pore formation. Acta Biomater. 2009, 5, 3328–3336. [Google Scholar] [CrossRef][Green Version]
- Bueno, V.B.; Petri, D.F.S. Xanthan hydrogel films: Molecular conformation, charge density and protein carriers. Carbohydr. Polym. 2014, 101, 897–904. [Google Scholar] [CrossRef]
- Levashov, P.A.; Matolygina, D.A.; Ovchinnikova, E.D.; Adamova, I.Y.; Gasanova, D.A.; Smirnov, S.A.; Nelyub, V.A.; Belogurova, N.G.; Tishkov, V.I.; Eremeev, N.L.; et al. The bacteriolytic activity of native and covalently immobilized lysozyme against Gram-positive and Gram-negative bacteria is differentially affected by charged amino acids and glycine. FEBS Open Bio 2019, 9, 510–518. [Google Scholar] [CrossRef]
- Yang, M.; Wang, Y.; Tao, G.; Cai, R.; Wang, P.; Liu, L.; Ai, L.; Zuo, H.; Zhao, P.; Umar, A.; et al. Fabrication of Sericin/Agrose Gel Loaded Lysozyme and Its Potential in Wound Dressing Application. Nanomaterials 2018, 8, 235. [Google Scholar] [CrossRef] [PubMed]
- Matouskova, P.; Marova, I.; Bokrova, J.; Benesova, P. Effect of Encapsulation on Antimicrobial Activity of Herbal Extracts with Lysozyme. Food Technol. Biotechnol. 2016, 54, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Kandemir, N.; Yemenicioğlu, A.; Mecitoğlu, Ç.; Elmacı, Z.S.; Baysal, T. Production of Antimicrobial Films by Incorporation of Partially Purified Lysozyme into Biodegradable Films of Crude Exopolysaccharides Obtained from Aureobasidium pullulans Fermentation. Food Technol. Biotechnol. 2005, 43, 343–350. [Google Scholar]
- Duan, L.; Wang, Y.; Zhang, Y.; Liu, J. Graphene immobilized enzyme/polyethersulfone mixed matrix membrane: Enhanced antibacterial, permeable and mechanical properties. Appl. Surf. Sci. 2015, 355, 436–445. [Google Scholar] [CrossRef]
- Hao, X.; Chen, S.; Zhu, H.; Wang, L.; Zhang, Y.; Yin, Y. The Synergy of Graphene Oxide and Polydopamine Assisted Immobilization of Lysozyme to Improve Antibacterial Properties. ChemistrySelect 2017, 2, 2174–2182. [Google Scholar] [CrossRef]
- Schoonen, L.; Maassen, S.; Nolte, R.J.M.; van Hest, J.C.M. Stabilization of a Virus-Like Particle and Its Application as a Nanoreactor at Physiological Conditions. Biomacromolecules 2017, 18, 3492–3497. [Google Scholar] [CrossRef]
- Zhou, D.; Yang, T.; Qian, W.; Xing, M.; Luo, G. Study of the mechanism of environmentally friendly translucent balsa-modified lysozyme dressing for facilitating wound healing. Int. J. Nanomed. 2018, 13, 4171–4187. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Yang, T.; Xing, M.; Luo, G. Preparation of a balsa-lysozyme eco-friendly dressing and its effect on wound healing. RSC Adv. 2018, 8, 13493–13502. [Google Scholar] [CrossRef]
- Shi, P.; Luo, S.; Voit, B.; Appelhans, D.; Zan, X. A facile and efficient strategy to encapsulate the model basic protein lysozyme into porous CaCO3. J. Mater. Chem. B 2018, 6, 4205–4215. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yu, H.; Ma, K.; Chen, Y.; Zhang, X.; Wang, T.; Li, S.; Zhu, X.; Wang, X. Flower-like Surface of Three-Metal-Component Layered Double Hydroxide Composites for Improved Antibacterial Activity of Lysozyme. Bioconjug. Chem. 2018, 29, 2090–2099. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, L.F.; de Almeida Gonçalves, K.; Boreli, F.H.; Kobarg, J.; Cardoso, M.B. Mechanism of interaction between colloids and bacteria as evidenced by tailored silica–lysozyme composites. J. Mater. Chem. 2012, 22, 22851. [Google Scholar] [CrossRef]
- Brunaugh, A.D.; Seo, H.; Warnken, Z.; Ding, L.; Seo, S.H.; Smyth, H.D.C. Development and evaluation of inhalable composite niclosamide-lysozyme particles: A broad-spectrum, patient-adaptable treatment for coronavirus infections and sequalae. PLoS ONE 2021, 16, e0246803. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-L.; Weng, Y.-M.; Huang, J.-J.; Lin, K.-J. Physicochemical characteristics and bacteriostatic ability of modified lysozyme from lactic acid-induced gelled egg white powder. J. Food Process. Preserv. 2012, 36, 232–241. [Google Scholar] [CrossRef]
- Cavalieri, F.; Micheli, L.; Kaliappan, S.; Teo, B.M.; Zhou, M.; Palleschi, G.; Ashokkumar, M. Antimicrobial and biosensing ultrasound-responsive lysozyme-shelled microbubbles. ACS Appl. Mater. Interfaces 2013, 5, 464–471. [Google Scholar] [CrossRef]
- Mahalingam, S.; Xu, Z.; Edirisinghe, M. Antibacterial Activity and Biosensing of PVA-Lysozyme Microbubbles Formed by Pressurized Gyration. Langmuir 2015, 31, 9771–9780. [Google Scholar] [CrossRef]
- Sarkar, S.; Gulati, K.; Mishra, A.; Poluri, K.M. Protein nanocomposites: Special inferences to lysozyme based nanomaterials. Int. J. Biol. Macromol. 2020, 151, 467–482. [Google Scholar] [CrossRef] [PubMed]
- Abouhmad, A.; Dishisha, T.; Amin, M.A.; Hatti-Kaul, R. Immobilization to Positively Charged Cellulose Nanocrystals Enhances the Antibacterial Activity and Stability of Hen Egg White and T4 Lysozyme. Biomacromolecules 2017, 18, 1600–1608. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zhang, C.; Zou, Y.; Li, Y.; Zhang, H. Immobilization of lysozyme on layer-by-layer self-assembled electrospun films: Characterization and antibacterial activity in milk. Food Hydrocoll. 2021, 113, 106468. [Google Scholar] [CrossRef]
- Huang, W.; Li, X.; Xue, Y.; Huang, R.; Deng, H.; Ma, Z. Antibacterial multilayer films fabricated by LBL immobilizing lysozyme and HTCC on nanofibrous mats. Int. J. Biol. Macromol. 2013, 53, 26–31. [Google Scholar] [CrossRef]
- Zhang, H.; Feng, M.; Chen, S.; Shi, W.; Wang, X. Incorporation of lysozyme into cellulose nanocrystals stabilized β-chitosan nanoparticles with enhanced antibacterial activity. Carbohydr. Polym. 2020, 236, 115974. [Google Scholar] [CrossRef] [PubMed]
- Uddin, K.M.A.; Orelma, H.; Mohammadi, P.; Borghei, M.; Laine, J.; Linder, M.; Rojas, O.J. Retention of lysozyme activity by physical immobilization in nanocellulose aerogels and antibacterial effects. Cellulose 2017, 24, 2837–2848. [Google Scholar] [CrossRef]
- Jiang, S.; Qin, Y.; Yang, J.; Li, M.; Xiong, L.; Sun, Q. Enhanced antibacterial activity of lysozyme immobilized on chitin nanowhiskers. Food Chem. 2017, 221, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Jia, P.; Zhe, T.; Bu, T.; Liu, Y.; Wang, Q.; Wang, L. Construction and multifunctionalization of chitosan-based three-phase nano-delivery system. Food Hydrocoll. 2019, 96, 402–411. [Google Scholar] [CrossRef]
- Piras, A.M.; Maisetta, G.; Sandreschi, S.; Esin, S.; Gazzarri, M.; Batoni, G.; Chiellini, F. Preparation, physical-chemical and biological characterization of chitosan nanoparticles loaded with lysozyme. Int. J. Biol. Macromol. 2014, 67, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sun, Y.; Xu, Y.; Feng, H.; Fu, S.; Tang, J.; Liu, W.; Sun, D.; Jiang, H.; Xu, S. Preparation and evaluation of lysozyme-loaded nanoparticles coated with poly-γ-glutamic acid and chitosan. Int. J. Biol. Macromol. 2013, 59, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, J.; Wang, D.; Sha, Y.; Chen, W.; Zhou, D.; Wang, X.; Sun, Q.; Xue, G.; Li, L. Low-temperature processing of polymer nanoparticles for bioactive composites. J. Polym. Sci. Part B Polym. Phys. 2016, 54, 2514–2520. [Google Scholar] [CrossRef]
- Yu, Q.; Ista, L.K.; López, G.P. Nanopatterned antimicrobial enzymatic surfaces combining biocidal and fouling release properties. Nanoscale 2014, 6, 4750–4757. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xue, Y.; Bi, Q.; Qin, D.; Du, Q.; Jin, P. Enhanced antibacterial activity of eugenol-entrapped casein nanoparticles amended with lysozyme against gram-positive pathogens. Food Chem. 2021, 360, 130036. [Google Scholar] [CrossRef]
- Li, J.; Liu, X.; Zhou, Z.; Tan, L.; Wang, X.; Zheng, Y.; Han, Y.; Chen, D.-F.; Yeung, K.W.K.; Cui, Z.; et al. Lysozyme-Assisted Photothermal Eradication of Methicillin-Resistant Staphylococcus aureus Infection and Accelerated Tissue Repair with Natural Melanosome Nanostructures. ACS Nano 2019, 13, 11153–11167. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Lu, M.; Liu, Y.; Chen, Q.; Taccardi, N.; Hüser, N.; Boccaccini, A.R. Monodispersed lysozyme-functionalized bioactive glass nanoparticles with antibacterial and anticancer activities. Biomed. Mater. 2016, 11, 35012. [Google Scholar] [CrossRef]
- Noor, M.M.; Goswami, J.; Davis, V.A. Comparison of Attachment and Antibacterial Activity of Covalent and Noncovalent Lysozyme-Functionalized Single-Walled Carbon Nanotubes. ACS Omega 2020, 5, 2254–2259. [Google Scholar] [CrossRef]
- Horn, D.W.; Tracy, K.; Easley, C.J.; Davis, V.A. Lysozyme Dispersed Single-Walled Carbon Nanotubes: Interaction and Activity. J. Phys. Chem. C 2012, 116, 10341–10348. [Google Scholar] [CrossRef]
- Nepal, D.; Minus, M.L.; Kumar, S. Lysozyme coated DNA and DNA/SWNT fibers by solution spinning. Macromol. Biosci. 2011, 11, 875–881. [Google Scholar] [CrossRef]
- Perevedentseva, E.; Cheng, C.-Y.; Chung, P.-H.; Tu, J.-S.; Hsieh, Y.-H.; Cheng, C.-L. The interaction of the protein lysozyme with bacteria E. coli observed using nanodiamond labelling. Nanotechnology 2007, 18, 315102. [Google Scholar] [CrossRef]
- Li, L.-L.; Wang, H. Enzyme-coated mesoporous silica nanoparticles as efficient antibacterial agents in vivo. Adv. Healthc. Mater. 2013, 2, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Yu, Q.; Zhan, W.; Chen, H. A Smart Antibacterial Surface for the On-Demand Killing and Releasing of Bacteria. Adv. Healthc. Mater. 2016, 5, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Liu, C.; Liu, J.; Zhang, Y. Development of a novel polyethersulfone ultrafiltration membrane with antibacterial activity and high flux containing halloysite nanotubes loaded with lysozyme. RSC Adv. 2015, 5, 38646–38653. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Shamova, O.V.; Yakovlev, A.V.; Zharkova, M.S. Synthesis and study of the biologically active lysozyme–silver nanoparticles–montmorillonite K10 complexes. Glass Phys. Chem. 2016, 42, 87–94. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Brazovskaya, E.Y.; Shamova, O.V. Biological activity and sorption ability of synthetic montmorillonite modified by silver/lysozyme nanoparticles. Appl. Clay Sci. 2018, 163, 56–62. [Google Scholar] [CrossRef]
- Zhan, Y.; Zeng, W.; Jiang, G.; Wang, Q.; Shi, X.; Zhou, Z.; Deng, H.; Du, Y. Construction of lysozyme exfoliated rectorite-based electrospun nanofibrous membranes for bacterial inhibition. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Ma, D.; Xie, C.; Wang, T.; Mei, L.; Zhang, X.; Guo, Z.; Yin, W. Liquid-Phase Exfoliation and Functionalization of MoS2 Nanosheets for Effective Antibacterial Application. Chembiochem 2020, 21, 2373–2380. [Google Scholar] [CrossRef]
- Remanan, S.; Samantaray, P.K.; Bose, S.; Das, N.C. Phase transited lysozyme particles and MoS2 nanosheets modified elastomer-like antibacterial and antifouling microfiltration membrane derived from poly(ethylene-co-methyl acrylate)/poly(vinylidene fluoride) (EMA/PVDF) blend for water purification application. Microporous Mesoporous Mater. 2021, 316, 110945. [Google Scholar] [CrossRef]
- Tripathy, N.; Ahmad, R.; Bang, S.H.; Min, J.; Hahn, Y.-B. Tailored lysozyme-ZnO nanoparticle conjugates as nanoantibiotics. Chem. Commun. 2014, 50, 9298–9301. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, D. Hetero-nanostructured film of titania nanosheets and lysozyme: Fabrication and synergistic antibacterial properties. Surf. Coat. Technol. 2012, 210, 71–77. [Google Scholar] [CrossRef]
- Yang, Q.-Z.; Chang, Y.-Y.; Zhao, H.-Z. Preparation and antibacterial activity of lysozyme and layered double hydroxide nanocomposites. Water Res. 2013, 47, 6712–6718. [Google Scholar] [CrossRef]
- Kalita, S.; Kandimalla, R.; Bhowal, A.C.; Kotoky, J.; Kundu, S. Functionalization of β-Lactam antibiotic on lysozyme capped gold nanoclusters retrogress MRSA and its persisters following awakening. Sci. Rep. 2018, 8, 5778. [Google Scholar] [CrossRef]
- Kumar, U.; Ranjan, A.K.; Sharan, C.; Hardikar, A.A.; Pundle, A.; Poddar, P. Green Approach Towards Size Controlled Synthesis of Biocompatible Antibacterial Metal Nanoparticles in Aqueous Phase Using Lysozyme. Curr. Nanosci. 2012, 8, 130–140. [Google Scholar] [CrossRef]
- Su, Y.; Li, C. Stable multilayer thin films composed of gold nanoparticles and lysozyme. Appl. Surf. Sci. 2008, 254, 2003–2008. [Google Scholar] [CrossRef]
- Chen, W.-Y.; Lin, J.-Y.; Chen, W.-J.; Luo, L.; Wei-Guang Diau, E.; Chen, Y.-C. Functional gold nanoclusters as antimicrobial agents for antibiotic-resistant bacteria. Nanomedicine 2010, 5, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Wei, T.; Zhao, J.; Jiang, S.; Yang, P.; Yu, Q.; Chen, H. Regenerable smart antibacterial surfaces: Full removal of killed bacteria via a sequential degradable layer. J. Mater. Chem. B 2018, 6, 3946–3955. [Google Scholar] [CrossRef] [PubMed]
- Eby, D.M.; Schaeublin, N.M.; Farrington, K.E.; Hussain, S.M.; Johnson, G.R. Lysozyme catalyzes the formation of antimicrobial silver nanoparticles. ACS Nano 2009, 3, 984–994. [Google Scholar] [CrossRef] [PubMed]
- Coll Ferrer, M.C.; Dastgheyb, S.; Hickok, N.J.; Eckmann, D.M.; Composto, R.J. Designing nanogel carriers for antibacterial applications. Acta Biomater. 2014, 10, 2105–2111. [Google Scholar] [CrossRef]
- Pellegrini, A.; Thomas, U.; Ibrahim, H.R. A Bactericidal Domain of Lysozyme with Helix-Loop-Helix Structure Present a Strong Antimicrobial Activity. In Proceedings of the Peptides 2002 27th European Peptide Symposium, Sorrento, Italy, 31 August–6 September 2002; Benedetti, E., Pedone, C., Eds.; pp. 582–583. [Google Scholar]
- Pellegrini, A. Antimicrobial peptides from food proteins. Curr. Pharm. Des. 2003, 9, 1225–1238. [Google Scholar] [CrossRef]
- Lee-Huang, S.; Huang, P.L.; Huang, P. Anti-HIV and Anti-Tumor Peptides and Fragments of Lysozyme. Patent No. WO2002004011, 17 January 2002. [Google Scholar]
- Ibrahim, H.R.; Inazaki, D.; Abdou, A.; Aoki, T.; Kim, M. Processing of lysozyme at distinct loops by pepsin: A novel action for generating multiple antimicrobial peptide motifs in the newborn stomach. Biochim. Biophys. Acta 2005, 1726, 102–114. [Google Scholar] [CrossRef]
- Carrillo, W.; Lucio, A.; Gaibor, J.; Morales, D.; Vásquez, G. Isolation of Antibacterial Hydrolysates from Hen Egg White Lysozyme and Identification of Antibacterial Peptides. J. Med. Food 2018, 21, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, A.; Thomas, U.; Wild, P.; Schraner, E.; von Fellenberg, R. Effect of lysozyme or modified lysozyme fragments on DNA and RNA synthesis and membrane permeability of Escherichia coli. Microbiol. Res. 2000, 155, 69–77. [Google Scholar] [CrossRef]
- Li, Q.; Zhou, Y.; Dong, K.; Guo, X. Potential therapeutic efficacy of a bactericidal-immunomodulatory fusion peptide against methicillin-resistant Staphylococcus aureus skin infection. Appl. Microbiol. Biotechnol. 2010, 86, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Lukacik, P.; Barnard, T.J.; Keller, P.W.; Chaturvedi, K.S.; Seddiki, N.; Fairman, J.W.; Noinaj, N.; Kirby, T.L.; Henderson, J.P.; Steven, A.C.; et al. Structural engineering of a phage lysin that targets gram-negative pathogens. Proc. Natl. Acad. Sci. USA 2012, 109, 9857–9862. [Google Scholar] [CrossRef]
- Appaiah, C.B.; Saravanan, R.S.; Sriram, B. Chimeric Antibacterial Polypeptides Constructed from Muralytic Domains and Membrane-Traversing Domains. Patent No. WO2012140676, 18 October 2012. [Google Scholar]
- Zhu, D.; Cai, G.; Li, X.; Lu, J.; Zhang, L. Enhancing the antimicrobial activity of Sus scrofa lysozyme by N-terminal fusion of a sextuple unique homologous peptide. J. Biotechnol. 2017, 243, 61–68. [Google Scholar] [CrossRef]
- Saito, H.; Sakakibara, Y.; Sakata, A.; Kurashige, R.; Murakami, D.; Kageshima, H.; Saito, A.; Miyazaki, Y. Antibacterial activity of lysozyme-chitosan oligosaccharide conjugates (LYZOX) against Pseudomonas aeruginosa, Acinetobacter baumannii and Methicillin-resistant Staphylococcus aureus. PLoS ONE 2019, 14, e0217504. [Google Scholar] [CrossRef]
- Tan, M.; Wang, H.; Wang, Y.; Chen, G.; Yuan, L.; Chen, H. Recyclable antibacterial material: Silicon grafted with 3,6-O-sulfated chitosan and specifically bound by lysozyme. J. Mater. Chem. B 2014, 2, 569–576. [Google Scholar] [CrossRef]
- Kim, S.; Fan, J.; Lee, C.-S.; Lee, M. Dual Functional Lysozyme-Chitosan Conjugate for Tunable Degradation and Antibacterial Activity. ACS Appl. Bio Mater. 2020, 3, 2334–2343. [Google Scholar] [CrossRef]
- Filatova, L.Y.; Balabushevich, N.G.; Klyachko, N.L. A physicochemical, structural, microbiological and kinetic study of hen egg white lysozyme in complexes with alginate and chitosan. Biocatal. Biotransform. 2021, 1–14. [Google Scholar] [CrossRef]
- Alahdad, Z.; Ramezani, R.; Aminlari, M.; Majzoobi, M. Preparation and properties of dextran sulfate-lysozyme conjugate. J. Agric. Food Chem. 2009, 57, 6449–6454. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.M.; Aminlari, M.; Moosavinasab, M. Preparation of and studies on the functional properties and bactericidal activity of the lysozyme–xanthan gum conjugate. LWT-Food Sci. Technol. 2014, 57, 594–602. [Google Scholar] [CrossRef]
- Tziveleka, L.-A.; Pippa, N.; Georgantea, P.; Ioannou, E.; Demetzos, C.; Roussis, V. Marine sulfated polysaccharides as versatile polyelectrolytes for the development of drug delivery nanoplatforms: Complexation of ulvan with lysozyme. Int. J. Biol. Macromol. 2018, 118, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Evran, S.; Yasa, I.; Telefoncu, A. Modification of lysozyme with oleoyl chloride for broadening the antimicrobial specificity. Prep. Biochem. Biotechnol. 2010, 40, 316–325. [Google Scholar] [CrossRef]
- Clementi, E.A.; Wilhelm, K.R.; Schleucher, J.; Morozova-Roche, L.A.; Hakansson, A.P. A complex of equine lysozyme and oleic acid with bactericidal activity against Streptococcus pneumoniae. PLoS ONE 2013, 8, e80649. [Google Scholar] [CrossRef]
- Nguyen, S.N.; Bobst, C.E.; Kaltashov, I.A. Compositions and Methods for Delivering Agents to the Central Nervous System. Patent No. WO2014152678, 25 September 2014. [Google Scholar]
- Nodake, Y.; Iwasaki, K.; Yamasaki, N. Interactions of a lysozyme-monomethoxypolyethylene glycol conjugate with lipopolysaccharides and lipid bilayers and effects of conjugate on Gram-negative bacteria. Biosci. Biotechnol. Biochem. 2002, 66, 1848–1852. [Google Scholar] [CrossRef] [PubMed]
- Steiert, E.; Radi, L.; Fach, M.; Wich, P.R. Protein-Based Nanoparticles for the Delivery of Enzymes with Antibacterial Activity. Macromol. Rapid Commun. 2018, 39, e1800186. [Google Scholar] [CrossRef] [PubMed]
- Diken Gür, S.; Bakhshpour, M.; Bereli, N.; Denizli, A. Antibacterial effect against both Gram-positive and Gram-negative bacteria via lysozyme imprinted cryogel membranes. J. Biomater. Sci. Polym. Ed. 2021, 32, 1024–1039. [Google Scholar] [CrossRef] [PubMed]
- Novaković, I.; Anđelković, U.; Zlatović, M.; Gašić, M.J.; Sladić, D. Bioconjugate of lysozyme and the antibacterial marine sesquiterpene quinone avarone and its derivatives. Bioconjug. Chem. 2012, 23, 57–65. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mansour, A.M. Antifungal activity, DNA and lysozyme binding affinity of Pd(II) and Pt(II) complexes bearing N, N-pyridylbenzimidazole ligand. J. Coord. Chem. 2018, 71, 3381–3391. [Google Scholar] [CrossRef]
- Mansour, A.M.; Shehab, O.R. Pyridylbenzimidazole-Based Gold(III) Complexes: Lysozyme Metalation, DNA Binding Studies, and Biological Activity. Eur. J. Inorg. Chem. 2019, 2019, 2830–2838. [Google Scholar] [CrossRef]
- Singh, G.; Kaur, M.; Kaur, H.; Kang, T.S. Synthesis and complexation of a new caffeine based surface active ionic liquid with lysozyme in aqueous medium: Physicochemical, computational and antimicrobial studies. J. Mol. Liq. 2021, 325, 115156. [Google Scholar] [CrossRef]
- Hoq, M.I.; Mitsuno, K.; Tsujino, Y.; Aoki, T.; Ibrahim, H.R. Triclosan-lysozyme complex as novel antimicrobial macromolecule: A new potential of lysozyme as phenolic drug-targeting molecule. Int. J. Biol. Macromol. 2008, 42, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Imranul Hoq, M.; Aoki, T.; Ibrahim, H.R. Triclosan–lysozyme complex: A promising antimicrobial macromolecule stable against photooxidative damage. Food Res. Int. 2009, 42, 298–306. [Google Scholar] [CrossRef]
- Ito, Y.; Kwon, O.H.; Ueda, M.; Tanaka, A.; Imanishi, Y. Gene-Engineered Hydrophobilization to Alter the Bactericidal Activity of Lysozyme. J. Bioact. Compat. Polym. 2000, 15, 376–395. [Google Scholar] [CrossRef]
- Muszanska, A.K.; Busscher, H.J.; Herrmann, A.; van der Mei, H.C.; Norde, W. Pluronic-lysozyme conjugates as anti-adhesive and antibacterial bifunctional polymers for surface coating. Biomaterials 2011, 32, 6333–6341. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, L.; Zhang, L.; Dong, Y. Human Lysozyme Modified by Cinnamaldehyde Coupling, and Modification Method and Application as Antibacterial Agent in Inhibiting Gram-Negative Bacteria. Patent No. CN112159802, 1 January 2021. [Google Scholar]
- Liu, J.; Wang, N.; Liu, Y.; Jin, Y.; Ma, M. The antimicrobial spectrum of lysozyme broadened by reductive modification. Poult. Sci. 2018, 97, 3992–3999. [Google Scholar] [CrossRef] [PubMed]
- Bogaerts, A.; Pankaj, A. Oxidized Lysozyme. Patent No. WO2020099556, 22 May 2020. [Google Scholar]
- Cegielska-Radziejewska, R.; Lesnierowski, G.; Szablewski, T.; Kijowski, J. Physico-chemical properties and antibacterial activity of modified egg white—lysozyme. Eur. Food Res. Technol. 2010, 231, 959–964. [Google Scholar] [CrossRef]
- González, R.; Mendive-Tapia, L.; Pastrian, M.B.; Albericio, F.; Lavilla, R.; Cascone, O.; Iannucci, N.B. Enhanced antimicrobial activity of a peptide derived from human lysozyme by arylation of its tryptophan residues. J. Pept. Sci. 2016, 22, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Leśnierowski, G. Changes in selected physicochemical properties of lysozyme modified with a new method using microwave field and oxidation. PLoS ONE 2019, 14, e0213021. [Google Scholar] [CrossRef]
- Yang, T.; Leśnierowski, G. Microwave Modification as an Excellent Way to Produce Unique Lysozyme with Potential for Food and Human Health. Foods 2021, 10, 1319. [Google Scholar] [CrossRef]
- Wen, S.; Yao, D.; Liu, X.; Wang, F. A Novel Fluorescence Resonance Energy Transfer-Based High-Throughput Screening Method for Generation of Lysozyme with Improved Antimicrobial Activity against Escherichia coli Strains. J. Agric. Food Chem. 2019, 67, 12584–12589. [Google Scholar] [CrossRef]
- Sharma, T.; Dohare, N.; Kumari, M.; Singh, U.K.; Khan, A.B.; Borse, M.S.; Patel, R. Comparative effect of cationic gemini surfactant and its monomeric counterpart on the conformational stability and activity of lysozyme. RSC Adv. 2017, 7, 16763–16776. [Google Scholar] [CrossRef]
- Diels, A.M.J.; de Taeye, J.; Michiels, C.W. Sensitisation of Escherichia coli to antibacterial peptides and enzymes by high-pressure homogenisation. Int. J. Food Microbiol. 2005, 105, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Nakimbugwe, D.; Masschalck, B.; Atanassova, M.; Zewdie-Bosüner, A.; Michiels, C.W. Comparison of bactericidal activity of six lysozymes at atmospheric pressure and under high hydrostatic pressure. Int. J. Food Microbiol. 2006, 108, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Lesnierowski, G.; Cegielska-Radziejewska, R.; Kijowski, J. Antibacterial activity of thermally modified lysozyme. Electron. J. Pol. Agric. Univ. 2001, 4, 17. [Google Scholar]
- Cegielska-Radziejewska, R.; Lesnierowski, G.; Kijowski, J. Antibacterial activity of hen egg white lysozyme modified by thermochemical technique. Eur. Food Res. Technol. 2009, 228, 841–845. [Google Scholar] [CrossRef]
- Vilcacundo, R.; Méndez, P.; Reyes, W.; Romero, H.; Pinto, A.; Carrillo, W. Antibacterial Activity of Hen Egg White Lysozyme Denatured by Thermal and Chemical Treatments. Sci. Pharm. 2018, 86, 48. [Google Scholar] [CrossRef] [PubMed]
- Derde, M.; Nau, F.; Guérin-Dubiard, C.; Lechevalier, V.; Paboeuf, G.; Jan, S.; Baron, F.; Gautier, M.; Vié, V. Native and dry-heated lysozyme interactions with membrane lipid monolayers: Lipid packing modifications of a phospholipid mixture, model of the Escherichia coli cytoplasmic membrane. Biochim. Biophys. Acta 2015, 1848, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Díez-Martínez, R.; de Paz, H.D.; de Paz, H.; Bustamante, N.; García, E.; Menéndez, M.; García, P. Improving the lethal effect of cpl-7, a pneumococcal phage lysozyme with broad bactericidal activity, by inverting the net charge of its cell wall-binding module. Antimicrob. Agents Chemother. 2013, 57, 5355–5365. [Google Scholar] [CrossRef]
- Teneback, C.C.; Scanlon, T.C.; Wargo, M.J.; Bement, J.L.; Griswold, K.E.; Leclair, L.W. Bioengineered lysozyme reduces bacterial burden and inflammation in a murine model of mucoid Pseudomonas aeruginosa lung infection. Antimicrob. Agents Chemother. 2013, 57, 5559–5564. [Google Scholar] [CrossRef] [PubMed]
- Griswold, K.E.; Bement, J.L.; Teneback, C.C.; Scanlon, T.C.; Wargo, M.J.; Leclair, L.W. Bioengineered lysozyme in combination therapies for Pseudomonas aeruginosa lung infections. Bioengineered 2014, 5, 143–147. [Google Scholar] [CrossRef][Green Version]
- Gill, A.; Scanlon, T.C.; Osipovitch, D.C.; Madden, D.R.; Griswold, K.E. Crystal structure of a charge engineered human lysozyme having enhanced bactericidal activity. PLoS ONE 2011, 6, e16788. [Google Scholar] [CrossRef]
- Osserman, E.F. Editorial: Lysozyme. N. Engl. J. Med. 1975, 292, 424–425. [Google Scholar] [CrossRef] [PubMed]



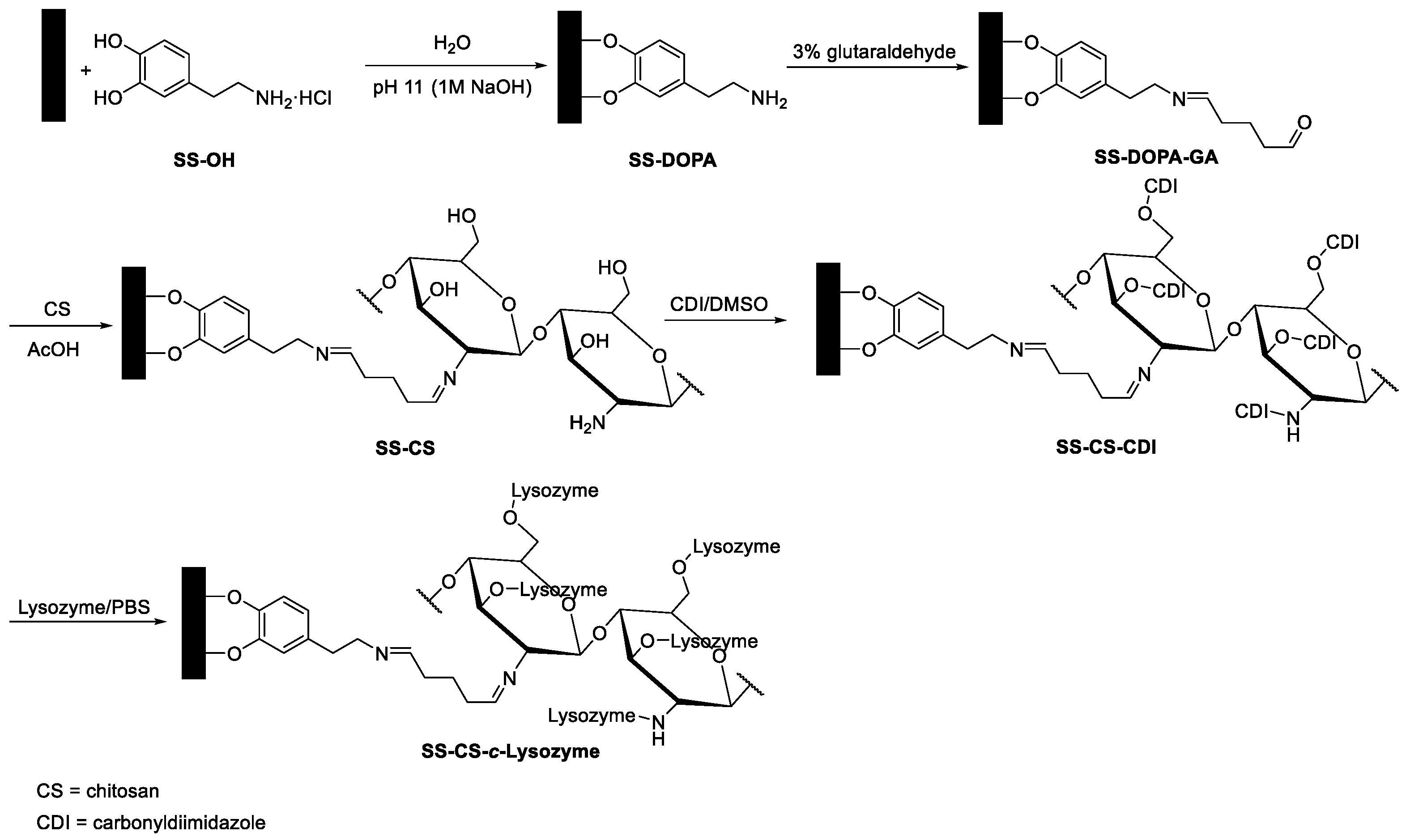

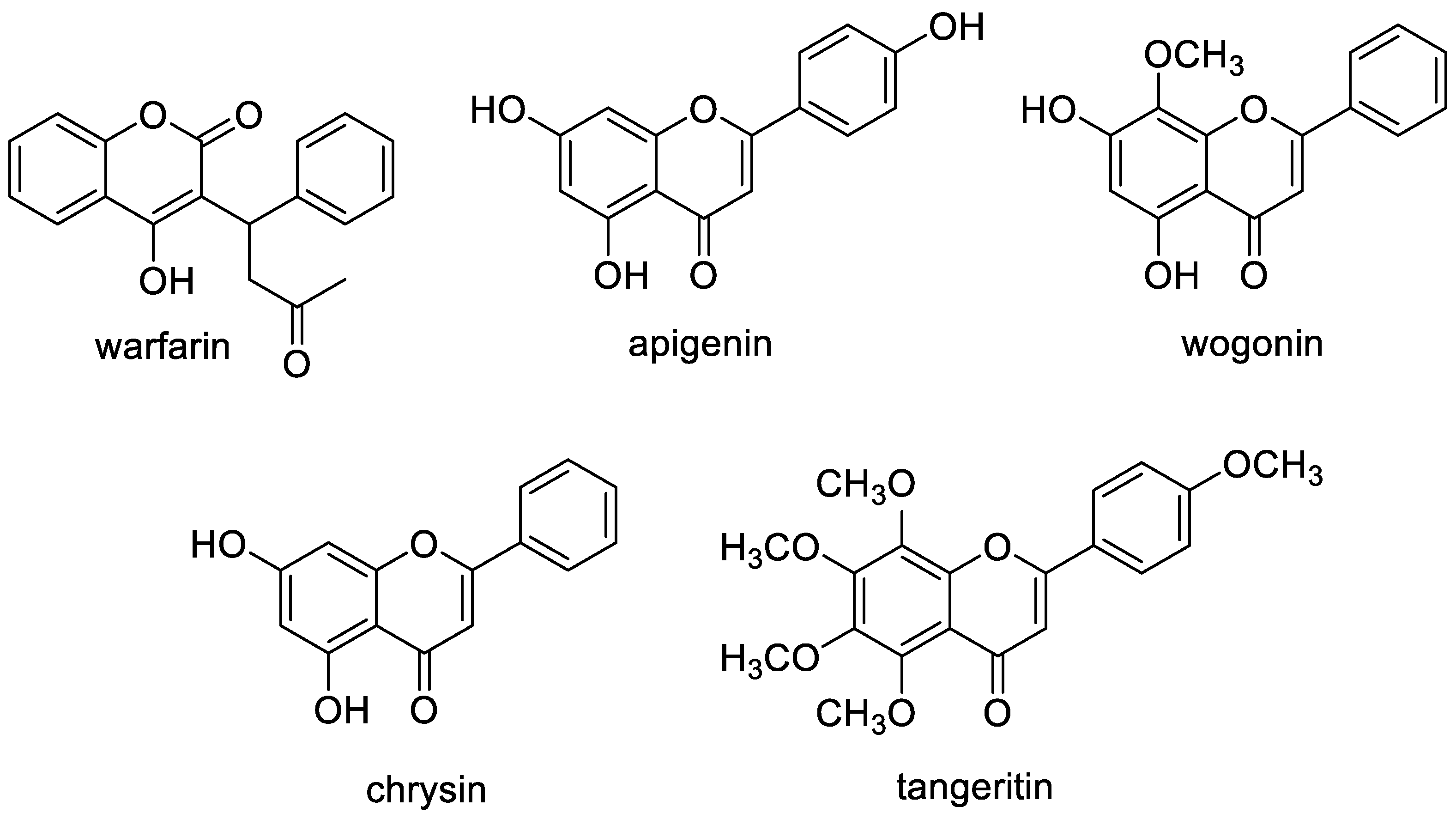
| Combined Use with | Application/Claimed Use | Antibiotic Activity Evaluated Against/On | Refs. |
|---|---|---|---|
| Cinnamaldehyde | Storage of olive flounder (Paralichthys olivaceus) fillets/lowered total viable count | S. putrefaciens and P. fluorescens | [177] |
| Acidic electrolyzed oxidizing water | To prolong the shelf life of carp fillets from a microbiological point of view | Total viable count, Enterobacteriaceae count, and anaerobic mesophilic count | [178] |
| Heat treatment and dilution with hydrogen peroxide associated with packaging in a controlled atmosphere | To extend the shelf life of pork meat by more than 20%, compared with the control sample without lysozyme | Aerobic plate count, Enterobacteriaceae, Pseudomonas spp., and lactic acid bacteria | [179] |
| High hydrostatic pressure | Not significant enough to differentiate lethality of lysozyme without additives in cheeses made from raw milk | B. cereus | [180] |
| Disodium ethylenediaminetetraacetate salt | Buffalo meat in refrigerated conditions | Total viable mesophilic count, total viable psychrotrophic count, lactic acid bacteria, Pseudomonas spp., and B. thermosphacta | [181] |
| Chitooligosaccharides | The microbiological quality of minced meat stored in refrigerated conditions was improved | Escherichia coli, Pseudomonas fluorescens, Bacillus cereus, and Staphylococcus aureus | [183] |
| Ethylenediaminetetraacetic acid disodium salt | To reduce the growth of Y. enterocolitica in orange beverages | Y. enterocolitica | [188] |
| Layer-by-layer chitosan-organic rectorite composites and negative charged sodium alginate. | The sensory analysis and physicochemical analysis applied to assess the effects of layer-by-layer film coating confirmed a higher score for the packaged pork (4 °C for 21 days) | E. coli and S. aureus | [184] |
| EDTA in anti-microbial starch-based active food packaging films | Cooked rice with pulses/effective to inhibit the growth of spoilage microorganisms | Contaminated from the open environmental sources (mainly Gram-negative cocci) | [185] |
| EDTA | A different lysozyme activity against the tested microorganisms was observed, increasing the ratio of lysozyme/EDTA from 11.14:8.14 mg/mL to 11.14:14.14 mg/mL. | Micrococcus lysodeikticus and Escherichia coli | [182] |
| Enterocin AS-48 | Synergy confirmed in liquid whole eggs, egg whites, and egg yolks, at 4 °C and 28 °C | B. cereus and S. aureus | [186] |
| Pomegranate peel extract | To maintain the quality of mackerel fillets wrapped with gelatin/polycaprolactone composite film and to prolong the shelf life of the product | Total mesophilic counts and psychrotrophic bacteria counts | [187] |
| Strain | Colistin MIC (μg/mL) | Percentage of Bacterial Survival after Each Treatment a | ||
|---|---|---|---|---|
| Colistin (0.1 μg/mL) | Lysozyme (25 μg/mL) | Lysozyme + Colistin (25 + 0.1 μg/mL) | ||
| PAO1 | 0.75 | 45.3 ± 11.2 | 35.9 ± 8.0 | 4.1 ± 1.3 |
| PA14 | 0.25 | 19.1 ± 3.9 | 52.3 ± 10.2 | 3.0 ± 0.62 |
| MIC a (μM) toward Bacterial Strains | ||||
|---|---|---|---|---|
| Gram-Negative | Gram-Positive | |||
| Sample | E. coli ML-35po | A. baumannii | MRSA ATCC 33591 | M. luteus CIP A270 |
| Lysozyme | >250 (500) | 1.2 | >250 (500) | 0.08 |
| Oxacillin | >250 (500) | 25 | 7.5 | 3.1 |
| Polymyxin B | 0.4 | 1.25 | 12.5 | 0.6 |
| Gentamicin | 0.625 | 0.06 | 0.16 | 0.16 |
| Rifampicin | 10 | 0.125 | 0.003 | 0.003 |
| Ofloxacin | 0.125 | 0.3 | 0.625 | 5 |
| Minimal FICIs a of the Lysozyme\Antibiotic Combination (AB) against Bacteria | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Gram-Negative | Gram-Positive | ||||||||||
| AB | RIF | PMB | GEN | OFL | OX | AB | RIF | PMB | GEN | OFL | OX |
| E. coli ML-35p | MRSA ATCC 33591 | ||||||||||
| Lysozyme | 0.62 | 0.25 | 0.53 | 1.12 | - | Lysozyme | 0.62 | 0.56 | 0.53 | 1.12 | 0.53 |
| A. baumannii | M. luteus CIP A270 | ||||||||||
| Lysozyme | 1 | 0.5 | 0.75 | 1 | 1 | Lysozyme | 0.75 | 0.62 | 0.62 | 0.62 | 0.75 |
| Support | Tested Microorganisms | Reagent or Immobilization Technique | Applications/Claimed Uses | Refs. |
|---|---|---|---|---|
| Vitreous surface | E. coli | Water decontamination | [259] | |
| Wool fabric | S. aureus | Glutaraldehyde | Antibacterial functionalization for textile goods | [260] |
| Wool fabric | E. coli | Tris(hydroxymethyl)phosphine | Antibacterial functionalization for textile goods | [261] |
| Cotton fabric | M. lysodeikticus | Glycine esterified | Medical hygiene | [262] |
| Silk textiles | S. aureus E. coli | Physically adsorbed | Functional wound dressing | [263] |
| Polyacrylonitrile membranes | S. aureus | Glutaraldehyde | Water treatment and food manufacturing | [264] |
| Polyacrylonitrile membrane hydrolyzed | E. coli S. aureus | Layer-by-layer self-assembly | Antibacterial thin film composite membranes for aqueous molecular separation | [265] |
| Polystyrene/poly (styrene-10-maleic anhydride) | S. aureus | CLEA | Antimicrobial process in biomedical and engineering industries | [266] |
| Polyethylene glycol | L. ivanovii M. luteus | Reductive amination on pretreated stainless steel surface | Antifouling agents | [125] |
| Poly(3,4-ethylenedioxythiophene) (PEDOT) | S. epidermidis | Incorporations in films | Regeneration of tissues | [267] |
| Cellulose acetate | S. aureus | Electrostatic adsorption, electrospraying | Food packaging and antimicrobial wound dressing | [268] |
| Gelatin/sodium carboxymethylcellulose | S. aureus P. aeruginosa E. coli | Inclusion in a polymeric matrix | Mucoadhesive form of lysozyme | [269] |
| Alginate, iron cations | M. luteus | Entrapment | Food packaging materials | [270] |
| Chitosan | S. aureus B. subtilis S. flexneri P. aeruginosa | CLEA | Repeated uses as antimicrobial material | [271] |
| Chitosan/alginate | E. coli S. aureus | Hydrogel | Food industry | [272] |
| Calcium phosphate on chitosan | M. lysodeikticus | Incorporation | Bone tissue engineering | [273] |
| Xanthan | M. luteus | Hydrogel | Wound dressing | [274] |
| Agarose, free amino acids | Gram-positive Gram-negative | Reductive amination | Blood plasma and whole blood purification by extracorporeal therapy procedures | [275] |
| Agarose, sericin | E. coli S. aureus | Gel | Wound dressing | [276] |
| Polysaccharides particles or liposome | B. subtilis M. luteus E. coli S. marcescens | Encapsulation with herbal extracts | Food preservation and wound healing | [277] |
| Exopolysaccharides | E. coli | Incorporation into films | Biodegradable coatings for fruits and vegetables | [278] |
| Graphene oxide | E. coli | Electrostatic interactions | Antibacterial membranes | [279] |
| Graphene oxide, polydopamine | E. coli | Electrostatic and hydrogen bond interactions | Medical treatments and food safety fields | [280] |
| Capsid | M. luteus | Encapsulation | Nanoreactor at physiological conditions | [281] |
| Balsa | E. coli S. aureus | Encapsulation | Wound healing | [282,283] |
| Calcium carbonate | M. lysodeikticus | Encapsulation | Catalysis, disease treatment, and tissue engineering | [284] |
| Layered double hydroxide | E. coli S. aureus | Van der Waals forces | Wound healing | [285] |
| Silica | E. coli S. aureus | Entrapment | Coating containers used to store chirurgical devices, catheters, implants, artificial prosthetics, and other materials to reduce hospital infections | [286] |
| Laser sintered titanium | S. gordonii S. sanguis | Layer-by-layer self-assembly | Biofilm inhibition | [132] |
| Niclosamide | MRSA MERS-COV SARS-CoV-2 | Embedded in h-Lys for inhalation route | Delivery to the upper and lower respiratory tracts | [287] |
| Lactic acid | B. cereus E. coli S. typhimurium | Gelled egg white powder | Food industry | [288] |
| Microbubbles, immobilized gold NPs | M. lysodeikticus | Pressurized gyration | Biosensor for the detection of analytes in aqueous solutions | [289] |
| Microbubbles, immobilized gold NPs, and polyvinyl alcohol | E. coli | Pressurized gyration | Diagnostic tools and environmental bioassays | [290] |
| NANOMATERIAL | ||||
|---|---|---|---|---|
| Tested Microorganisms | Reagent or Method | Applications/Claimed Uses | Refs. | |
| Cellulose nanocrystals | M. lysodeikticus Corynebacterium sp. E. coli P. mendocina | Glutaraldehyde | Improved antibacterial action also against Gram-negative bacteria | [292] |
| Cellulose nanocrystals | B. subtilis | Cellulose-CHO | Inhibition of biofilm | [110] |
| Cellulose acetate nanofibers, sodium alginate | S. aureus | Electrostatic interactions | Milk and dairy products | [293] |
| Cellulose acetate nanofibrous LBL | E. coli S. aureus | Electrostatic interactions | Food packaging, adhesive wound dressing, and tissue engineering | [294] |
| Cellulose NPs/b-chitosan | L. innocua E. coli | Encapsulation | Packaging material for shelf life extension | [295] |
| Nanocellulose aerogel | S. aureus E. coli | Electrostatic interactions | Wound dressings | [296] |
| Chitin nanowhiskers | E. coli S. aureus B. subtilis | Adsorption | Enhancement of the antibacterial efficiency for food preservation | [297] |
| Chitosan NPs/tannin | S. aureus S. enteriditis L. monocytogenes | Encapsulation | Food industry | [298] |
| Chitosan NPs | S. epidermidis | Encapsulation of antimicrobial peptides | [299] | |
| Poly-γ-glutamic acid, chitosan NPs | E. coli B. subtilis | Loading | Controlled delivery system | [300] |
| Polystyrene nanospheres | E. coli | Electrospinning | Food processing and medical equipment | [301] |
| Nanopatterned poly(isopropylacrylamide) | E. coli S. epidermidis | Adsorption | Mitigation of short-term bacterial biofouling | [302] |
| Eugenol-casein NPs | S. aureus Bacillus sp. | Encapsulation | Food preservation | [303] |
| Melanosome nanostructures | MRSA | Electrostatic interactions | Tissue repair | [304] |
| Glass NPs | B. subtilis, (human hepatocellular carcinoma) | Electrostatic interactions | Treatment of bone defects caused by tumors | [305] |
| Carbon NTs single-wallet carbon nanotubes | M. lysodeikticus | Noncovalent or covalent interactions | Covalent functionalization led to improved dispersion stability and longer duration of bacterial lysis relative to noncovalent lysozyme single-walled carbon nanotubes | [306,307] |
| Single walled carbon NTs, DNA fibers | M. lysodeikticus | Spinning, electrostatic interactions | Drug delivery, tissue engineering, and biocompatible composites | [308] |
| Nanodiamonds | E. coli | Electrostatic interactions | Biolabel to observe the interaction of Lys with bacteria | [309] |
| Silica NPs | E. coli | Electrostatic interactions | Highly efficient antibacterial agent in vitro and in vivo with low cytotoxicity and negligible hemolytic activity | [310] |
| Silicon nanowires, poly (methacrylic acid) | E. coli | Adsorption | Engineering of surfaces with switchable functionalities | [311] |
| Halloysite NTs | E. coli | 1,6-hexan-ethylene diisocyanate | Reduced fouling in water treatment | [312] |
| Montmorillonite K10-silver NPs | E. coli P. aeruginosa MRSA L. monocytogenes | Complexes | Activity against antibiotic resistant bacterial strains | [313] |
| Montmorillonite-silver NPs | E. coli S. aureus | Complexes | Production of sorbents with antibiotic properties | [314] |
| Rectorite nanofibrous membrane | S. aureus E. coli | Electrospinning | Food engineering and biomedical materials | [315] |
| Molybdenum disulfide nanosheets | Ampicillin-resistant E. coli B. subtilis | Electrostatic interactions | Design and synthesis of novel nanozyme antibacterial agents | [316] |
| Molybdenum disulfide nanosheets | E. coli S. aureus | Coating | Microfiltration membrane for water purification | [317] |
| Zinc oxide NPs | E. coli S. aureus | Glutaraldehyde + aminated ZnO NPs | Biomedical | [318] |
| Titania nanosheets | M. lysodeikticus | Layer-by-layer technique, electrostatic interactions | Antibacterial coatings | [319] |
| Layered double hydroxide nanocomposites | E. coli B. subtilis | Loading | Water purification processes | [320] |
| Gold capped nanoclusters with ampicillin | MRSA | Wound healing | [321] | |
| Gold NPs | B. subtilis | Tryptophan residues of Lys + N-bromo succinimide | Drug delivery and bioimaging | [322] |
| Silver NPs | Tyrosine residues of Lys + N-acetyl imidazole | |||
| Gold NPs | S. epidermidis E. coli | Layer by layer | Long-term antibacterial coating, biocatalysis, and biosensor | [323] |
| Gold NPs | Acinetobacter baumanii Enterococcus faecalis | Bacterial labeling and antimicrobial agents against antibiotic resistant bacteria | [324] | |
| Gold NPs | S. aureus E. coli | Hybrid film | Killing and removal of adherent bacteria on the surfaces of medical devices | [325] |
| Silver NPs | E. coli S. aureus B. anthracis C. albicans resistant P. mirabilis | Nanoparticles from stable colloid (silver acetate and lys in methanol) | Aseptic and therapeutic use | [326] |
| Silver NPs in nanogel (dextran + lys) | E. coli S. aureus | Maillard reaction (nanogel), AgNPs embedded in nanogel | Inhibition of biofilm formation | [327] |
| FRAGMENTATION | ||||
| Microorganisms | Method | Applications/Claimed Uses | Refs. | |
| Nine amino acids | Viral infection | Clostripain | HIV infection and inhibition of tumor growth | [330] |
| Bactericidal domain from human milk Lys | E. coli S. aureus C. albicans | Pepsin, pH 4 | Treatment of microbial infections | [20] |
| From c-type Lys | Gram-positive Gram-negative | Pepsin, pH 4 | Treatment of microbial infections | [331] |
| From goose egg white Lys | E. coli B. bronchiseptica S. enteridis H. pylori S. aureus S. epidermidis B. subtilis M. luteus | Pepsin, pH 2 | Treatment of microbial infections | [16] |
| From hen egg white Lys | E. coli S. carnosus | Pepsin, pH 2 | Treatment of microbial infections | [332] |
| Pentadecapeptide from chicken egg white Lys | E. coli | Clostripain | Treatment of microbial infections and nontoxic against erythrocytes | [333] |
| FUSION PEPTIDES and PROTEINS | ||||
| Fusion Product | Microorganisms | Applications/Claimed Uses | Refs. | |
| Human β-defensin-3-lysozyme | MRSA S. aureus | Therapy of MRSA infection | [334] | |
| Pesticin-N-terminus of T4 lysozyme | E. coli Yersinia | Therapeutic application to a wide variety of Yersiniae and pathogenic E. coli strains | [335] | |
| Propeptide of surfactant protein B | Streptococci P. aeruginosa | Prophylaxis or therapeutic treatment of respiratory and gastrointestinal bacterial infection | [152] | |
| Chimeric polypeptides | P. aeruginosa, K. pneumoniae E. coli A. baumanii S. typhimurium S. infantis Shigella P. mirabilis B. thailandensis | Preparation of new enzymes active against Gram-negative bacteria | [336] | |
| Chimeric phage lysin | S. pneumoniae | Treatment of multiresistant pneumococcal infections | [128] | |
| N-terminal hexapeptide-Sus scrofa lysozyme | B. licheniformis B. subtilis M. lysodeikticus S. aureus E. coli K. pneumoniae P. aeruginosa S. enteritidis | Antibacterial lysozyme derivatives as components of food additives. | [337] | |
| CONJUGATES and COMPLEXES | ||||
| Starting Material | Microorganism | Method or Reagent | Applications/Claimed Uses | Refs. |
| Chitosan | P.aeruginosa A. baumannii MRSA | Maillard reaction | Control of refractory infections | [338] |
| Chitosan film | S. faecalis E. coli | Surface coating on perishable fruits and vegetables to enhance microbial safety and extend shelf life of the products | [171] | |
| Chitosan-silicon | E. coli | New recyclable antibacterial materials | [339] | |
| Chitosan hydrogel | S. aureus E. coli | Methacrylate | Tissue engineering and wound healing | [340] |
| Chitosan, alginate complexes | E. coli | Medical preparations characterized by a sustained release and resistance to aggressive environmental conditions | [341] | |
| Dextran sulfate | S. aureus E. coli | Maillard reaction | Ingredient in formulated food systems or as therapeutic agent | [342] |
| Xanthan | M. lysodeikticus S. aureus E. coli | Maillard reaction | Functional ingredient with high quality emulsifier, foam producer, or natural antibacterial agent in food | [343] |
| Ulvan | S. aureus | Polyelectrolyte complexes | Nanocarrier for positively charged bioactive molecules | [344] |
| Oleyl chloride | E. coli | Covalent bond (amide) | Increase in hydrophobicity | [345] |
| Oleic acid | S. pneumoniae | Complex | Bactericidal activity against bacterial species with a respiratory tropism | [346] |
| Transferrin (human) | Gram-positive | Homofunctional linker | Delivery of the composition in the CNS by crossing the blood–brain-barrier | [347] |
| Monomethoxy polyethylene glycol | E. coli P. aeruginosa | Succinyl ester of mPEG | Increase in hydrophobicity | [348] |
| Monomethoxy polyethylene glycol | M. luteus | Tetrafluorophenyl 4-(mPEG)-4-oxobutanoate | Sustained release of the hydrophilic lysozyme by passive diffusion | [349] |
| N-methacryloyl-(L)-histidine methylester | S. aureus E. coli | Complex | Antibacterial coatings and tissue engineering studies | [350] |
| Avarone | Gram-positive Gram-negative | Addition to the quinone moiety | Targeting the cell wall vehicle for delivering the quinone | [351] |
| Cisplatin analogues | C. albicans Cryptococcus neoformans | Complexes | Antifungal activity | [352] |
| Pyridylbenzimidazole Au (III) | C. albicans Cryptococcus neoformans | Complexes | Antifungal activity | [353] |
| Caffeine, dioctyl sulfosuccinate | E. coli P. aeruginosa B. thuringensis | Colloidal complex | Development of antimicrobial colloidal systems | [354] |
| Triclosan | E. coli P. aeruginosa K. pneumoniae S. typhimurium | Complex | Delivery of phenolic drugs to microbial cells in food and drug systems | [355,356] |
| Polyproline | E. coli P. aeruginosa | Recombinant DNA technique | Increase in hydrophobicity | [357] |
| Pluronic F-127 | B. subtilis | Reductive amination | Biological systems, including the coating of biomaterials implant surfaces | [358] |
| Cinnamic aldehyde | E. coli | Covalent modification | Increased antibiotic spectrum | [359] |
| REACTIONS of LYSOZYME CHAIN | ||||
| Reaction | Microorganism | Reagent | Applications/Claimed Uses | Refs. |
| Reduction | S. aureus E. coli S. enteritidis | Sodium sulfite | Increased hydrophobicity, antibacterial agent in food industry | [360] |
| Oxidation of tyrosine and tryptophan | M. lysodeikticus | Reactive oxygen and nitrogen species | Treatment of cancer and infectious diseases, antibacterial, and disinfecting agent | [361] |
| Oxidation | P. fluorescens | Hydrogen peroxide | Increased hydrophobicity | [362] |
| Arylation | S. aureus S. epidermidis | Arylation of tryptophan residues with iodobenzene | Increased hydrophobicity | [363] |
| Physical Process | Microorganisms | Effect | Refs. |
|---|---|---|---|
| Microwave held followed by oxidation | M. lysodeikticus | Dimer and trimer formation | [364] |
| Microwave | M. lysodeikticus | Dimer and trimer formation and changes in surface hydrophobicity | [365] |
| Fluorescens resonance energy transfer | E. coli | Variants formation | [366] |
| Cationic surfactant (gemini) | M. luteus | Micelles formation | [367] |
| High hydrostatic pressure | Gram-negative: E. coli, P. fluorescens, S. entericas, S. sonnei, and S. flexneri | Sensitization of bacteria | [101] |
| High hydrostatic pressure | E. coli | Increased outer membrane permeability | [368] |
| Atmospheric and high hydrostatic pressure | Gram-positive Gram-negative | Sensitization of bacteria | [369] |
| Thermal treatment (t 80 °C) | M. luteus E. coli | Formation of dimer depending on pH and concentration | [370] |
| Thermochemical treatment (60–70 °C + 10–20% H2O2) | M. lysodeikticus S. epidermidis | Formation of dimer | [371] |
| Thermochemical treatment (denaturation with heat or with dithiothreitol) | E. coli S. carnosus | Oligomers formation | [372] |
| Dry heating (80 °C, 7 days) | E. coli | Increased insertion capacity and ability to induce lipid packing modifications | [373] |
| Heating of jenny milk | B. megaterium Clavibacter michiganensis Clostridium tyrobutyricum Xanthomonas campestris E. coli | Antimicrobial activity like synthetic antibiotics against some Gram-positive and Gram-negative strains | [18] |
| Bioengineered modifications: | |||
| Net charge inversion of a phage lysozyme | S. pneumoniae | Mutation of a Cpl-7 | [374] |
| Charge engineered variant of hLys | P. aeruginosa | Redesigned electrostatic potential field | [375,376,377] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferraboschi, P.; Ciceri, S.; Grisenti, P. Applications of Lysozyme, an Innate Immune Defense Factor, as an Alternative Antibiotic. Antibiotics 2021, 10, 1534. https://doi.org/10.3390/antibiotics10121534
Ferraboschi P, Ciceri S, Grisenti P. Applications of Lysozyme, an Innate Immune Defense Factor, as an Alternative Antibiotic. Antibiotics. 2021; 10(12):1534. https://doi.org/10.3390/antibiotics10121534
Chicago/Turabian StyleFerraboschi, Patrizia, Samuele Ciceri, and Paride Grisenti. 2021. "Applications of Lysozyme, an Innate Immune Defense Factor, as an Alternative Antibiotic" Antibiotics 10, no. 12: 1534. https://doi.org/10.3390/antibiotics10121534
APA StyleFerraboschi, P., Ciceri, S., & Grisenti, P. (2021). Applications of Lysozyme, an Innate Immune Defense Factor, as an Alternative Antibiotic. Antibiotics, 10(12), 1534. https://doi.org/10.3390/antibiotics10121534







