Bacillus amyloliquefaciens—Derived Lipopeptide Biosurfactants Inhibit Biofilm Formation and Expression of Biofilm-Related Genes of Staphylococcus aureus
Abstract
:1. Introduction
2. Results
2.1. Genotypic Identification of the Isolate
2.2. Presence of Surfactin, Fengycin and Iturin A Genes
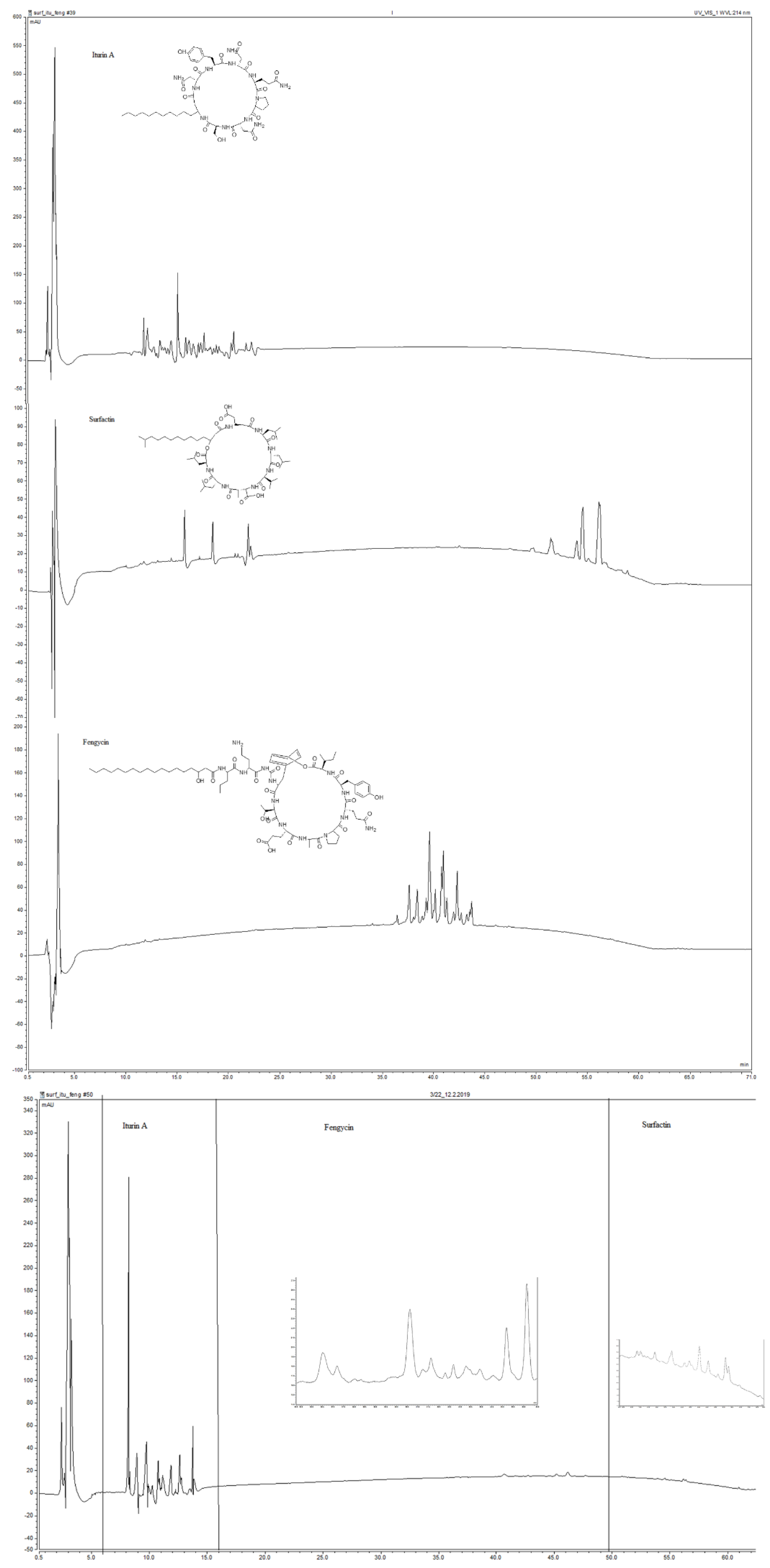
2.3. Ultrahigh-Performance Liquid Chromatography
2.4. MALDI-TOF/MS Analysis
2.5. The Effect of LPs 3/22 on S. aureus CCM 4223 Biofilm Formation
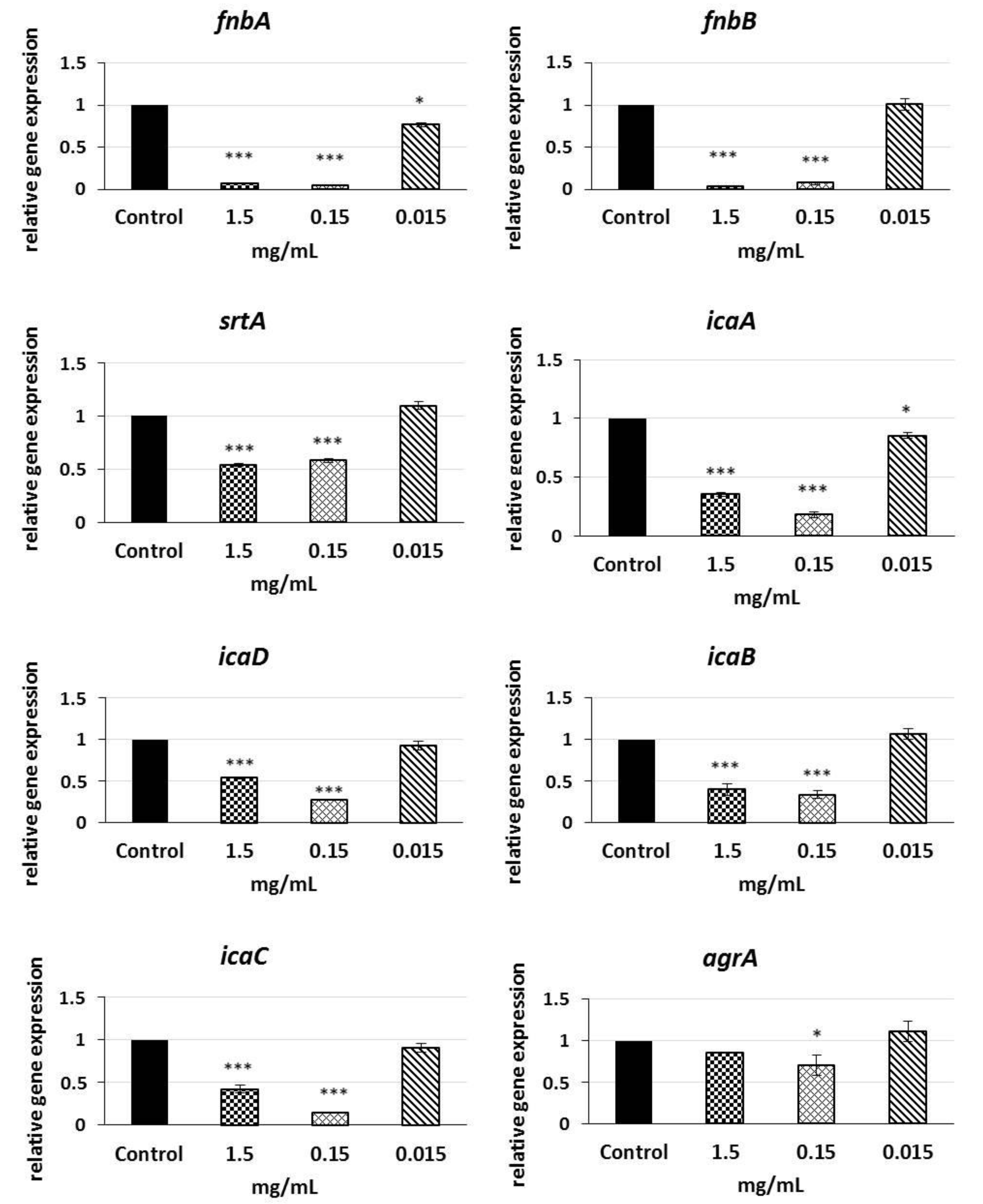
2.6. Analysis of qRT-PCR Results
3. Discussion
4. Materials and Methods
4.1. Microorganisms
4.2. Isolation and Screening of Isolates for BSs Production
4.3. Genotypic Identification
4.4. Detection of the LP Genes
4.5. Isolation of Biosurfactants
4.6. Ultrahigh-Performance Liquid Chromatography
4.7. MALDI-TOF/MS Analysis
4.8. Determination of Growth Inhibition Activity of LPs 3/22 against S. aureus in Planktonic Cells
4.9. Effect of LPs on Biofilm Formation
4.10. Quantification of Gene Expression Using qRT-PCR
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sharma, D.; Misba, L.; Khan, A.U. Antibiotics versus biofilm: An emerging battleground in microbial communities. Antimicrob. Resist. Infect. Control 2019, 8, 76. [Google Scholar] [CrossRef]
- Khan, J.; Tarar, S.M.; Gul, I.; Nawaz, U.; Arshad, M. Challenges of antibiotic resistance biofilms and potential combating strategies: A review. 3 Biotech 2021, 11, 169. [Google Scholar] [CrossRef]
- Tong, S.Y.C.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef] [Green Version]
- Sakr, A.; Brégeon, F.; Mège, J.L.; Rolain, J.M.; Blin, O. Staphylococcus aureus nasal colonization: An update on mechanisms, epidemiology, risk factors, and subsequent infections. Front. Microbiol. 2018, 9, 2419. [Google Scholar] [CrossRef]
- Lister, J.L.; Horswill, A.R. Staphylococcus aureus biofilms: Recent developments in biofilm dispersal. Front. Cell. Infect. Microbiol. 2014, 4, 178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galié, S.; García-Gutiérrez, C.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Biofilms in the food industry: Health aspects and control methods. Front. Microbiol. 2018, 9, 898. [Google Scholar] [CrossRef] [PubMed]
- Czaplewski, L.; Bax, R.; Clokie, M.; Dawson, M.; Fairhead, H.; Fischetti, V.A.; Foster, S.; Gilmore, B.F.; Hancock, R.E.W.; Harper, D.; et al. Alternatives to antibiotics-a pipeline portfolio review. Lancet Infect. Dis. 2016, 16, 239–251. [Google Scholar] [CrossRef] [Green Version]
- Cattior, V.; Felden, B. Future antibacterial strategies: From basic concepts to clinical challenges Vincent. J. Infect. Dis. 2019, 220, 350–360. [Google Scholar] [CrossRef]
- Marchant, R.; Banat, I.M. Microbial biosurfactants: Challenges and opportunities for future exploitation. Trends Biotechnol. 2012, 30, 558–565. [Google Scholar] [CrossRef]
- Hancock, R.E.W.; Alford, M.A.; Haney, E.F. Antibiofilm activity of host defence peptides: Complexity provides opportunities. Nat. Rev. Microbiol. 2021, 1–12. [Google Scholar] [CrossRef]
- Li, W.; Separovic, F.; O′Brien-Simpson, N.M.; Wade, J.D. Chemically modified and conjugated antimicrobial peptides against superbugs. Chem. Soc. Rev. 2021, 50, 4932–4973. [Google Scholar] [CrossRef]
- Cappello, S.; Genovese, M.; Della Torre, C.; Crisari, A.; Hassanshahian, M.; Santisi, S.; Calogero, R.; Yakimov, M.M. Effect of bioemulsificant exopolysaccharide (EPS2003) on microbial community dynamics during assays of oil spill bioremediation: A microcosm study. Mar. Pollut. Bull. 2012, 64, 2820–2828. [Google Scholar] [CrossRef]
- Balan, S.S.; Mani, P.; Kumar, C.G.; Jayalakshmi, S. Structural characterization and biological evaluation of Staphylosan (dimannooleate), a new glycolipid surfactant produced by a marine Staphylococcus saprophyticus SBPS-15. Enzyme Microb. Technol. 2019, 120, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Hentati, D.; Chebbi, A.; Hadrich, F.; Frikha, I.; Rabanal, F.; Sayadi, S.; Manresa, A.; Chamkha, M. Production, characterization and biotechnological potential of lipopeptide biosurfactants from a novel marine Bacillus stratosphericus strain FLU5. Ecotoxicol. Environ. Saf. 2019, 167, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Roy, A. A review on the biosurfactants: Properties, types and its applications. J. Fundam. Renew. Energy Appl. 2017, 8, 1–5. [Google Scholar] [CrossRef]
- Vecino, X.; Cruz, J.M.; Moldes, A.B.; Rodrigues, L.R. Biosurfactants in cosmetic formulations: Trends and challenges. Crit. Rev. Biotechnol. 2017, 37, 911–923. [Google Scholar] [CrossRef]
- Sachdev, D.P.; Cameotra, S.S. Biosurfactants in agriculture. Appl. Microbiol. Biotechnol. 2013, 97, 1005–1016. [Google Scholar] [CrossRef] [Green Version]
- Nitschke, M.; Silva, S.S.E. Recent food applications of microbial surfactants. Crit. Rev. Food Sci. Nutr. 2018, 58, 631–638. [Google Scholar] [CrossRef]
- Naughton, P.J.; Marchant, R.; Naughton, V.; Banat, I.M. Microbial biosurfactants: Current trends and applications in agricultural and biomedical industries. J. Appl. Microbiol. 2019, 127, 12–28. [Google Scholar] [CrossRef] [Green Version]
- Narendrakumar, L.; Das, B.; Paramasivan, B.; Rasu, J.; Thomas, S. Quorum quenching and biofilm inhibition: Alternative imminent strategies to control the disease cholera. In Biotechnological Applications of Quorum Sensing Inhibitors; Springer: Berlin, Germany, 2018; pp. 63–85. [Google Scholar] [CrossRef]
- Sarwar, A.; Brader, G.; Corretto, E.; Aleti, G.; Abaidullah, M.; Sessitsch, A.; Hafeez, F.Y. Qualitative analysis of biosurfactants from Bacillus species exhibiting antifungal activity. PLoS ONE 2018, 13, e0198107. [Google Scholar] [CrossRef] [Green Version]
- Jacques, P. Surfactin and other lipopeptides from Bacillus spp. In Biosurfactants; Springer: Berlin, Germany, 2011; pp. 57–91. [Google Scholar]
- Ongena, M.; Jacques, P. Bacillus lipopeptides: Versatile weapons for plant disease biocontrol. Trends Microbiol. 2008, 16, 115–125. [Google Scholar] [CrossRef]
- Arima, K.; Kakinuma, A.; Tamura, G. Surfactin, a crystalline peptidelipid surfactant produced by Bacillus subtilis: Isolation, characterization and its inhibition of fibrin clot formation. Biochem. Biophys. Res. Commun. 1968, 31, 488–494. [Google Scholar] [CrossRef]
- Kakinuma, A.; Sugino, H.; Isono, M.; Tamura, G.; Arima, K. Determination of fatty acid in surfactin and elucidation of the total structure of surfactin. Agric. Biol. Chem. 1969, 33, 973–976. [Google Scholar] [CrossRef]
- Huszcza, E.; Burczyk, B. Surfactin isoforms from Bacillus coagulans. Z. Naturforsch. Sect. C J. Biosci. 2006, 61, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Y.; Chen, C.L.; Lee, Y.H.; Cheng, Y.C.; Wu, Y.C.; Shu, H.Y.; Gotz, F.; Liu, S.T. Nonribosomal synthesis of fengycin on an enzyme complex formed by fengycin synthetases. J. Biol. Chem. 2007, 282, 5608–5616. [Google Scholar] [CrossRef] [Green Version]
- Meena, K.R.; Kanwar, S.S. Lipopeptides as the antifungal and antibacterial agents: Applications in food safety and therapeutics. Biomed Res. Int. 2015, 2015, 473050. [Google Scholar] [CrossRef] [Green Version]
- Bedlovičová, Z.; Englerová, K.; Nemcová, R.; Kačániová, M. Identification of lipopeptides produced by Bacillus amyloliquefaciens strains. J. Microbiol. Biotechnol. Food Sci. 2021, 10, e3690. [Google Scholar] [CrossRef]
- Plaza, G.; Chojniak, J.; Rudnicka, K.; Paraszkiewicz, K.; Bernat, P. Detection of biosurfactants in Bacillus species: Genes and products identification. J. Appl. Microbiol. 2015, 119, 1023–1034. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.X.; Zhang, Y.; Shan, H.H.; Tong, Y.H.; Chen, X.J.; Liu, F.Q. Isolation and identification of antifungal peptides from Bacillus amyloliquefaciens W10. Environ. Sci. Pollut. Res. 2017, 24, 25000–25009. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.H.; Ye, Z.W.; Zheng, Q.W.; Wei, T.; Lin, J.F.; Guo, L.Q. Isolation and characterization of cyclic lipopeptides with broad-spectrum antimicrobial activity from Bacillus siamensis JFL15. 3 Biotech 2018, 8, 444. [Google Scholar] [CrossRef] [PubMed]
- He, C.P.; Fan, L.Y.; Wu, W.H.; Liang, Y.Q.; Li, R.; Tang, W.; Zheng, X.L.; Xiao, Y.N.; Liu, Z.X.; Zheng, F.C. Identification of lipopeptides produced by Bacillus subtilis Czk1 isolated from the aerial roots of rubber trees. Genet. Mol. Res. 2017, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Habe, H.; Taira, T.; Imura, T. Screening of a Bacillus subtilis strain producing multiple types of cyclic lipopeptides and evaluation of their surface-tension-lowering activities. J. Oleo Sci. 2017, 66, 785–790. [Google Scholar] [CrossRef] [Green Version]
- Mulligan, C.N.; Sharma, S.K.; Mudhoo, A. Biosurfactants: Research Trends and Applications; CRC Press: Boca Raton, FL, USA, 2019; ISBN 1466518243. [Google Scholar]
- Ramyabharathi, S.; Sankari Meena, K.; Rajendran, L.; Karthikeyan, G.; Jonathan, E.I.; Raguchander, T. Biocontrol of wilt-nematode complex infecting gerbera by Bacillus subtilis under protected cultivation. Egypt. J. Biol. Pest Control 2018, 28, 21. [Google Scholar] [CrossRef] [Green Version]
- Amruta, N.; Prasanna Kumar, M.K.; Puneeth, M.E.; Sarika, G.; Kandikattu, H.K.; Vishwanath, K.; Narayanaswamy, S. Exploring the potentiality of novel rhizospheric bacterial strains against the rice blast fungus Magnaporthe oryzae. Plant Pathol. J. 2018, 34, 126–138. [Google Scholar] [CrossRef] [PubMed]
- Perez, K.J.; Viana, J.d.S.; Lopes, F.C.; Pereira, J.Q.; dos Santos, D.M.; Oliveira, J.S.; Velho, R.V.; Crispim, S.M.; Nicoli, J.R.; Brandelli, A.; et al. Bacillus spp. isolated from puba as a source of biosurfactants and antimicrobial lipopeptides. Front. Microbiol. 2017, 8, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerayeli, N.; Baghaee-Ravari, S.; Tarighi, S. Evaluation of the antagonistic potential of Bacillus strains against Pectobacterium carotovorum subsp. carotovorum and their role in the induction of resistance to potato soft rot infection. Eur. J. Plant Pathol. 2018, 150, 1049–1063. [Google Scholar] [CrossRef]
- Yang, H.; Li, X.; Li, X.; Yu, H.; Shen, Z. Identification of lipopeptide isoforms by MALDI-TOF-MS/MS based on the simultaneous purification of iturin, fengycin, and surfactin by RP-HPLC. Anal. Bioanal. Chem. 2015, 407, 2529–2542. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, Y.; Fu, X.; Li, Y.; Wang, Q. Isolation and characterization of Bacillus amyloliquefaciens PG12 for the biological control of apple ring rot. Postharvest Biol. Technol. 2016, 115, 113–121. [Google Scholar] [CrossRef]
- Biniarz, P.; Łukaszewicz, M. Direct quantification of lipopeptide biosurfactants in biological samples via HPLC and UPLC-MS requires sample modification with an organic solvent. Appl. Microbiol. Biotechnol. 2017, 101, 4747–4759. [Google Scholar] [CrossRef]
- Mubarak, M.Q.E.; Hassan, A.R.; Hamid, A.A.; Khalil, S.; Isa, M.H.M. A simple and effective isocratic HPLC method for fast identification and quantification of surfactin. Sains Malays. 2015, 44, 115–120. [Google Scholar] [CrossRef]
- Kim, P.I.; Ryu, J.; Kim, Y.H.; Chi, Y.T. Production of biosurfactant lipopeptides iturin A, fengycin, and surfactin A from Bacillus subtilis CMB32 for control of Colletotrichum gloeosporioides. J. Microbiol. Biotechnol. 2010, 20, 138–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernat, P.; Paraszkiewicz, K.; Siewiera, P.; Moryl, M.; Płaza, G.; Chojniak, J. Lipid composition in a strain of Bacillus subtilis, a producer of iturin A lipopeptides that are active against uropathogenic bacteria. World J. Microbiol. Biotechnol. 2016, 32, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banat, I.M.; Satpute, S.K.; Cameotra, S.S.; Patil, R.; Nyayanit, N.V. Cost effective technologies and renewable substrates for biosurfactants′ production. Front. Microbiol. 2014, 5, 697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Horn, J.N.; Sengillo, J.D.; Lin, D.; Romo, T.D.; Grossfield, A. Characterization of a potent antimicrobial lipopeptide via coarse-grained molecular dynamics. Biochim. Biophys. Acta BBA Biomembr. 2012, 1818, 212–218. [Google Scholar] [CrossRef] [Green Version]
- De Jesús Cortés-Sánchez, A.; Hernández-Sánchez, H.; Jaramillo-Flores, M.E. Biological activity of glycolipids produced by microorganisms: New trends and possible therapeutic alternatives. Microbiol. Res. 2013, 168, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.R.; Banat, I.M.; Van Der Mei, H.C.; Teixeira, J.A.; Oliveira, R. Interference in adhesion of bacteria and yeasts isolated from explanted voice prostheses to silicone rubber by rhamnolipid biosurfactants. J. Appl. Microbiol. 2006, 100, 470–480. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tahmourespour, A.; Salehi, R.; Kermanshahi, R.K. Lactobacillus acidophilus-derived biosurfactant effect on gtfb and gtfc expression level in Streptococcus mutans biofilm cells. Braz. J. Microbiol. 2011, 42, 330–339. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.; Gu, S.; Cui, X.; Shi, Y.; Wen, S.; Chen, H.; Ge, J. Antimicrobial, anti-adhesive and anti-biofilm potential of biosurfactants isolated from Pediococcus acidilactici and Lactobacillus plantarum against Staphylococcus aureus CMCC26003. Microb. Pathog. 2019, 127, 12–20. [Google Scholar] [CrossRef]
- Idrees, M.; Sawant, S.; Karodia, N.; Rahman, A. Staphylococcus aureus biofilm: Morphology, genetics, pathogenesis and treatment strategies. Int. J. Environ. Res. Public Health 2021, 18, 7602. [Google Scholar] [CrossRef]
- Giri, S.S.; Ryu, E.C.; Sukumaran, V.; Park, S.C. Antioxidant, antibacterial, and anti-adhesive activities of biosurfactants isolated from Bacillus strains. Microb. Pathog. 2019, 132, 66–72. [Google Scholar] [CrossRef]
- Janek, T.; Łukaszewicz, M.; Krasowska, A. Antiadhesive activity of the biosurfactant pseudofactin II secreted by the Arctic bacterium Pseudomonas fluorescens BD5. BMC Microbiol. 2012, 12, 24. [Google Scholar] [CrossRef] [Green Version]
- De Araujo, L.V.; Guimarães, C.R.; da Silva Marquita, R.L.; Santiago, V.M.J.; de Souza, M.P.; Nitschke, M.; Freire, D.M.G. Rhamnolipid and surfactin: Anti-adhesion/antibiofilm and antimicrobial effects. Food Control 2016, 63, 171–178. [Google Scholar] [CrossRef]
- Rivardo, F.; Turner, R.J.; Allegrone, G.; Ceri, H.; Martinotti, M.G. Anti-adhesion activity of two biosurfactants produced by Bacillus spp. prevents biofilm formation of human bacterial pathogens. Appl. Microbiol. Biotechnol. 2009, 83, 541–553. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, R.d.A.; Weslley Caracas Cedro, E.; Raquel Colares Andrade, A.; Serpa, R.; José de Jesus Evangelista, A.; Sales de Oliveira, J.; Santos Pereira, V.; Pereira Alencar, L.; Bruna Leite Mendes, P.; Cibelle Soares Farias, B.; et al. Inhibitory effect of a lipopeptide biosurfactant produced by Bacillus subtilis on planktonic and sessile cells of Trichosporon spp. Biofouling 2018, 34, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Quinn, G.A.; Maloy, A.P.; Banat, M.M.; Banat, I.M. A comparison of effects of broad-spectrum antibiotics and biosurfactants on established bacterial biofilms. Curr. Microbiol. 2013, 67, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, S.; Tofighi, Z.; Babaee, T.; Shamsi, M.; Rahimzadeh, G.; Rezvanifar, H.; Saeidi, E.; Amiri, M.M.; Ashtiani, Y.S.; Samadi, N. Evaluation of anti-oxidant and anti-biofilm activities of biogenic surfactants derived from Bacillus amyloliquefaciens and Pseudomonas aeruginosa. Iran. J. Pharm. Res. 2020, 19, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Meena, K.R.; Sharma, A.; Kanwar, S.S. Antitumoral and antimicrobial activity of surfactin extracted from Bacillus subtilis KLP2015. Int. J. Pept. Res. Ther. 2020, 26, 423–433. [Google Scholar] [CrossRef]
- Englerová, K.; Nemcová, R.; Bedlovičová, Z.; Styková, E. Antiadhesive, antibiofilm and dispersion activity of biosurfactants isolated from Bacillus amyloliquefaciens 3/22. Česká Slov. Farm. 2021, 70, 172–178, in press. [Google Scholar]
- Heilmann, C. Adhesion mechanisms of staphylococci. In Bacterial Adhesion; Springer: Berlin, Geramny, 2011; pp. 105–123. [Google Scholar]
- Soltani, E.; Farrokhi, E.; Zamanzad, B.; Abadi, M.S.S.; Deris, F.; Soltani, A.; Gholipour, A. Prevalence and distribution of adhesins and the expression of fibronectin-binding protein (FnbA and FnbB) among Staphylococcus aureus isolates from Shahrekord Hospitals. BMC Res. Notes 2019, 12, 49. [Google Scholar] [CrossRef] [Green Version]
- Tsompanidou, E.; Denham, E.L.; Sibbald, M.J.J.B.; Yang, X.-M.; Seinen, J.; Friedrich, A.W.; Buist, G.; van Dijl, J.M. The sortase A substrates FnbpA, FnbpB, ClfA and ClfB antagonize colony spreading of Staphylococcus aureus. PLoS ONE 2012, 7, e44646. [Google Scholar] [CrossRef] [Green Version]
- Ouyang, P.; He, X.; Yuan, Z.-W.; Yin, Z.-Q.; Fu, H.; Lin, J.; He, C.; Liang, X.; Lv, C.; Shu, G.; et al. Erianin against Staphylococcus aureus infection via inhibiting sortase, A. Toxins 2018, 10, 385. [Google Scholar] [CrossRef] [Green Version]
- Novais, J.S.; Carvalho, M.F.; Ramundo, M.S.; Beltrame, C.O.; Geraldo, R.B.; Jordão, A.K.; Ferreira, V.F.; Castro, H.C.; Figueiredo, A.M.S. Antibiofilm effects of N,O-acetals derived from 2-amino-1,4-naphthoquinone are associated with downregulation of important global virulence regulators in methicillin-resistant Staphylococcus aureus. Sci. Rep. 2020, 10, 19631. [Google Scholar] [CrossRef]
- Thappeta, K.R.V.; Zhao, L.N.; Nge, C.E.; Crasta, S.; Leong, C.Y.; Ng, V.; Kanagasundaram, Y.; Fan, H.; Ng, S.B. In-silico identified new natural sortase a inhibitors disrupt S. aureus biofilm formation. Int. J. Mol. Sci. 2020, 21, 8601. [Google Scholar] [CrossRef]
- Mirzaee, M.; Najar Peerayeh, S.; Ghasemian, A.-M. Detection of icaABCD genes and biofilm formation in clinical isolates of methicillin resistant Staphylococcus aureus. Iran. J. Pathol. 2014, 9, 257–262. [Google Scholar]
- Melo, T.A.; Dos Santos, T.F.; De Almeida, M.E.; Junior, L.A.G.F.; Andrade, E.F.; Rezende, R.P.; Marques, L.M.; Romano, C.C. Inhibition of Staphylococcus aureus biofilm by Lactobacillus isolated from fine cocoa. BMC Microbiol. 2016, 16, 250. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Xu, Y.; Yestrepsky, B.D.; Sorenson, R.J.; Chen, M.; Larsen, S.D.; Sun, H. Novel inhibitors of Staphylococcus aureus virulence gene expression and biofilm formation. PLoS ONE 2012, 7, e47255. [Google Scholar] [CrossRef] [Green Version]
- Rajasree, K.; Fasim, A.; Gopal, B. Conformational features of the Staphylococcus aureus AgrA-promoter interactions rationalize quorum-sensing triggered gene expression. Biochem. Biophys. Rep. 2016, 6, 124–134. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Li, W.; Zhu, X.; Zhao, H.; Lu, Y.; Zhang, C.; Lu, Z. Surfactin effectively inhibits Staphylococcus aureus adhesion and biofilm formation on surfaces. Appl. Microbiol. Biotechnol. 2019, 103, 4565–4574. [Google Scholar] [CrossRef]
- Cramton, S.E.; Gerke, C.; Schnell, N.F.; Nichols, W.W.; Götz, F. The intercellular adhesion (ICA) locus is present in Staphylococcus aureus and is required for biofilm formation. Infect. Immun. 1999, 67, 5427–5433. [Google Scholar] [CrossRef] [Green Version]
- Techaoei, S.; Leelapornpisid, P.; Santiarwarn, D.; Lumyong, S.; Mai, C. Preliminary screening of biosurfactant-producing microorganisms isolated from hot. Development 2007, 7, 38–43. [Google Scholar]
- Morikawa, M.; Yoshihiko, H.; Tadayuki, I. A study on the structure–function relationship of lipopeptide biosurfactants. Biochim. Biophys. Acta BBA Mol. Cell Biol. Lipids 2000, 1488, 211–218. [Google Scholar] [CrossRef]
- Frank, J.A.; Reich, C.I.; Sharma, S.; Weisbaum, J.S.; Wilson, B.A.; Olsen, G.J. Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl. Environ. Microbiol. 2008, 74, 2461–2470. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, F.C.; Li, M.C.; Lin, T.C.; Kao, S.S. Rapid detection and characterization of surfactin-producing Bacillus subtilis and closely related species based on PCR. Curr. Microbiol. 2004, 49, 186–191. [Google Scholar] [CrossRef]
- Mora, I.; Cabrefiga, J.; Montesinos, E. Antimicrobial peptide genes in Bacillus strains from plant environments. Int. Microbiol. 2011, 14, 213–223. [Google Scholar] [CrossRef] [Green Version]
- Caprioli, R.M.; Farmer, T.B.; Gile, J. Molecular imaging of biological samples: Localization of peptides and proteins using MALDI-TOF MS. Anal. Chem. 1997, 69, 4751–4760. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Pratt, L.A.; Watnick, P.I.; Newman, D.K.; Weaver, V.B.; Kolter, R. Genetic approaches to study of biofilms. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1999; Volume 310, pp. 91–109. [Google Scholar]
- Jadhav, S.; Shah, R.; Bhave, M.; Palombo, E.A. Inhibitory activity of yarrow essential oil on Listeria planktonic cells and biofilms. Food Control 2013, 29, 125–130. [Google Scholar] [CrossRef]
- Iqbal, Z.; Seleem, M.N.; Hussain, H.I.; Huang, L.; Hao, H.; Yuan, Z. Comparative virulence studies and transcriptome analysis of Staphylococcus aureus strains isolated from animals. Sci. Rep. 2016, 6, 35442. [Google Scholar] [CrossRef]
- Nourbakhsh, F.; Namvar, A. Detection of genes involved in biofilm formation in Staphylococcus aureus isolates. GMS Hyg. Infect. Control 2016, 11, Doc07. [Google Scholar] [CrossRef]
- Pereyra, E.A.L.; Picech, F.; Renna, M.S.; Baravalle, C.; Andreotti, C.S.; Russi, R.; Calvinho, L.F.; Diez, C.; Dallard, B.E. Detection of Staphylococcus aureus adhesion and biofilm-producing genes and their expression during internalization in bovine mammary epithelial cells. Vet. Microbiol. 2016, 183, 69–77. [Google Scholar] [CrossRef]


| Gene | GenBank Sequence Identity/Accession Number |
|---|---|
| 16S rRNA | B. amyloliquefaciens (100%) |
| KF811045.1 | |
| srfAA | B. subtilis (97.95%) |
| KC454625.1 | |
| sfp | B. amyloliquefaciens (99.56%) |
| KX346253.1 | |
| fenB | NA |
| fenD | B. amyloliquefaciens (96.63%) |
| KP453873.1 | |
| ituD | B. amyloliquefaciens (99.36%) |
| FJ815155.1 |
| Activity [%] | Concentration of LPs 3/22 [mg/mL] | |||
|---|---|---|---|---|
| 15 | 1.5 | 0.15 | 0.015 | |
| Inhibition of biofilm formation | 100.19 ± 4.14 | 84.46 ± 4.21 | 60.09 ± 2.06 | 38.84 ± 13.76 |
| Biosurfactants | Gene | Sequence | PCR Product Size [bp] | Annealing Temperature [°C] |
|---|---|---|---|---|
| Surfactin | sfp | F-5′ATGAAGATTTACGGAATTTA 3′ R-5′TTATAAAAGCTCTTCGTACG 3′ | 675 | 50 |
| srfAA | F-5′TCGGGACAGGAAGACATCAT 3′ R-5′CCACTCAAACGGATAATCCTGA 3′ | 201 | 60 | |
| Fengycin | fenB | F-5′CCTGGAGAAAGAATATACCGTACCY 3′ R-5′GCTGGTTCAGTT KGATCACAT 3′ | 670 | 57 |
| fenD | F-5′GGCCCGTTCTCTAAATCCAT 3′ F-5′GTCATGCTGACGAGAGCAAA 3′ | 269 | 60 | |
| Iturin A | ituD | F-5′ TTGAAYGTCAGYGCSCCTTT 3′ R-5′ TGCGMAAATAATGGSGTCGT 3′ | 482 | 57 |
| Gene | Primer | Primer Sequence (5′-3′) | Reference |
|---|---|---|---|
| fnbA | fnbA-F | GAAGTGGCACAGCCAAGAAC | This study |
| fnbA-R | ACGTTGACCAGCATGTGG | ||
| fnbB | fnbB-F | CAATGATCCTATCATTGAGAAGAGTG | This study |
| fnbB-R | CCTTCTACACCTTCAACAGCTGTA | ||
| srtA | srtA-F | GTGGTACTTATCCTAGTGGCAGC | This study |
| srtA-R | GCCTGCCACTTTCGATTTATC | ||
| icaA | icaA-F | CTTGCTGGCGCAGTCAATAC | [82] |
| icaA-R | GTAGCCAACGTCGACAACTG | ||
| icaD | icaD-F | ACCCAACGCTAAAATCATCG | [83] |
| icaD-R | GCGAAAATGCCCATAGTTTC | ||
| icaB | icaB-F | ATACCGGCGACTGGGTTTAT | [83] |
| icaB-R | ATGCAAATCGTGGGTATGTGT | ||
| icaC | icaC-F | CTTGGGTATTTGCACGCATT | [83] |
| icaC-R | GCAATATCATGCCGACACCT | ||
| agrA | agrA-F | TCGTAAGCATGACCCAGTTG | This study |
| agrA-R | AAATCCATCGCTGCAACTTT | ||
| gyrB | gyrB-F | CCAGGTAAATTAGCCGATTGC | [84] |
| gyrB-R | ATCGCCTGCGTTCTAGAGTC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Englerová, K.; Bedlovičová, Z.; Nemcová, R.; Király, J.; Maďar, M.; Hajdučková, V.; Styková, E.; Mucha, R.; Reiffová, K. Bacillus amyloliquefaciens—Derived Lipopeptide Biosurfactants Inhibit Biofilm Formation and Expression of Biofilm-Related Genes of Staphylococcus aureus. Antibiotics 2021, 10, 1252. https://doi.org/10.3390/antibiotics10101252
Englerová K, Bedlovičová Z, Nemcová R, Király J, Maďar M, Hajdučková V, Styková E, Mucha R, Reiffová K. Bacillus amyloliquefaciens—Derived Lipopeptide Biosurfactants Inhibit Biofilm Formation and Expression of Biofilm-Related Genes of Staphylococcus aureus. Antibiotics. 2021; 10(10):1252. https://doi.org/10.3390/antibiotics10101252
Chicago/Turabian StyleEnglerová, Karolína, Zdenka Bedlovičová, Radomíra Nemcová, Ján Király, Marián Maďar, Vanda Hajdučková, Eva Styková, Rastislav Mucha, and Katarína Reiffová. 2021. "Bacillus amyloliquefaciens—Derived Lipopeptide Biosurfactants Inhibit Biofilm Formation and Expression of Biofilm-Related Genes of Staphylococcus aureus" Antibiotics 10, no. 10: 1252. https://doi.org/10.3390/antibiotics10101252
APA StyleEnglerová, K., Bedlovičová, Z., Nemcová, R., Király, J., Maďar, M., Hajdučková, V., Styková, E., Mucha, R., & Reiffová, K. (2021). Bacillus amyloliquefaciens—Derived Lipopeptide Biosurfactants Inhibit Biofilm Formation and Expression of Biofilm-Related Genes of Staphylococcus aureus. Antibiotics, 10(10), 1252. https://doi.org/10.3390/antibiotics10101252







