Detection of Salmonella Typhimurium in Romaine Lettuce Using a Surface Plasmon Resonance Biosensor
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instrument
2.2. Preparation of SPR Sensor Surface
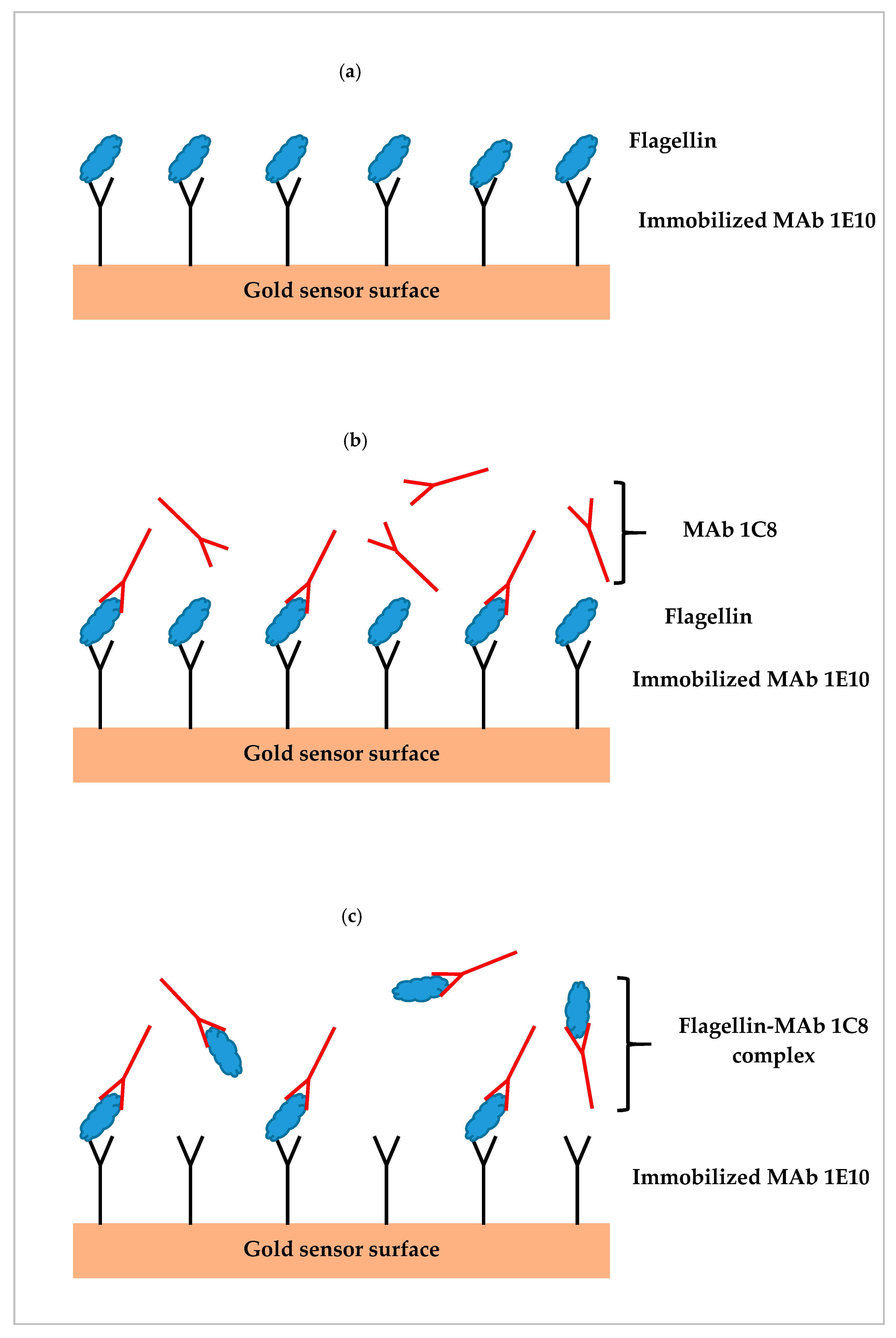
2.3. SPR Assay Formats
2.4. Preparation of S. Typhimurium Flagellin
2.5. Preparation of S. Typhimurium Contaminated Romaine Lettuce
2.6. Analytical Procedures of SPR Assay
2.7. Flagellin Purification Using Immunomagnetic Separation
2.8. Data Analysis
3. Results
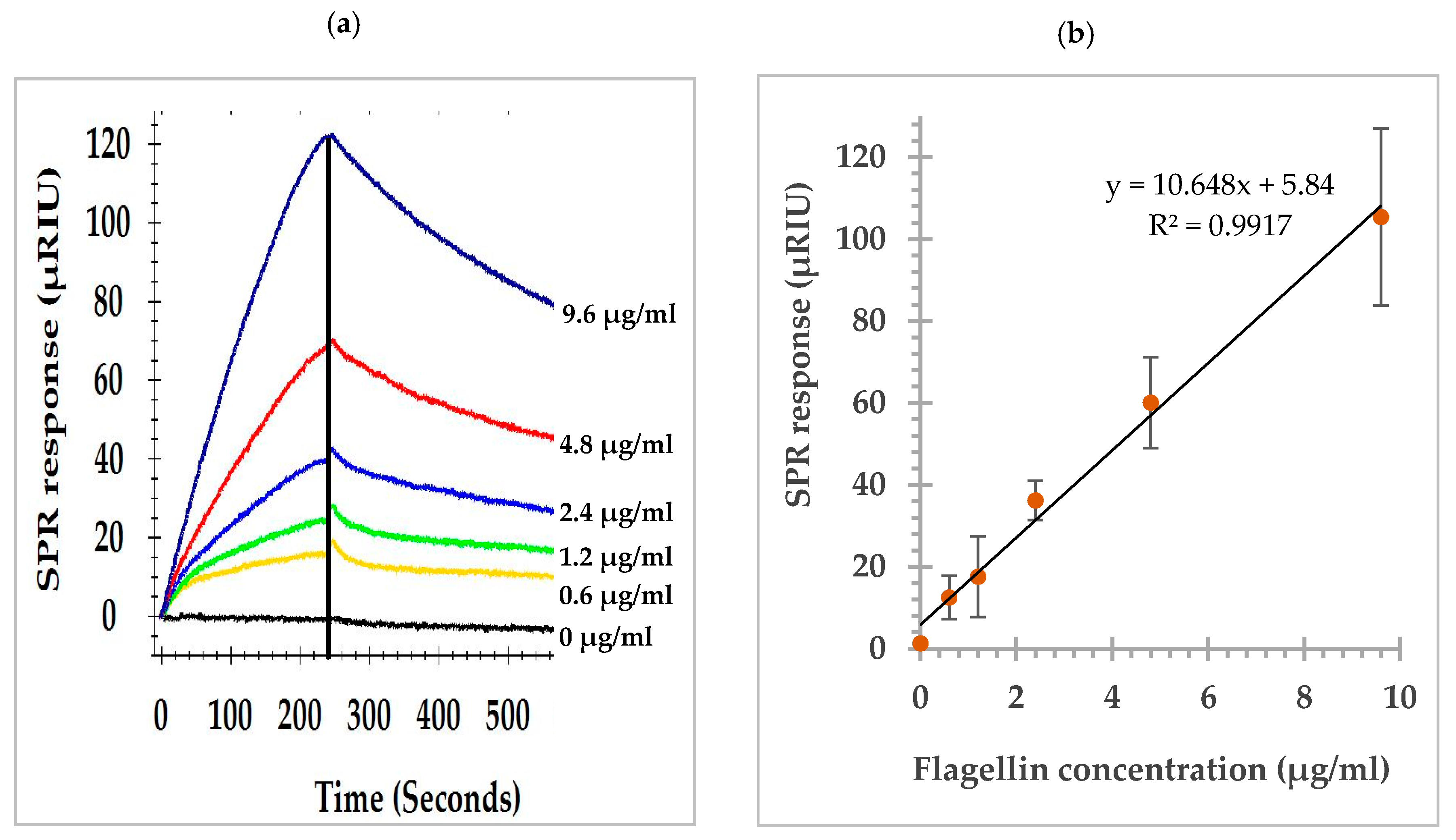
3.1. SPR Sensorgram and Dose Response Curve
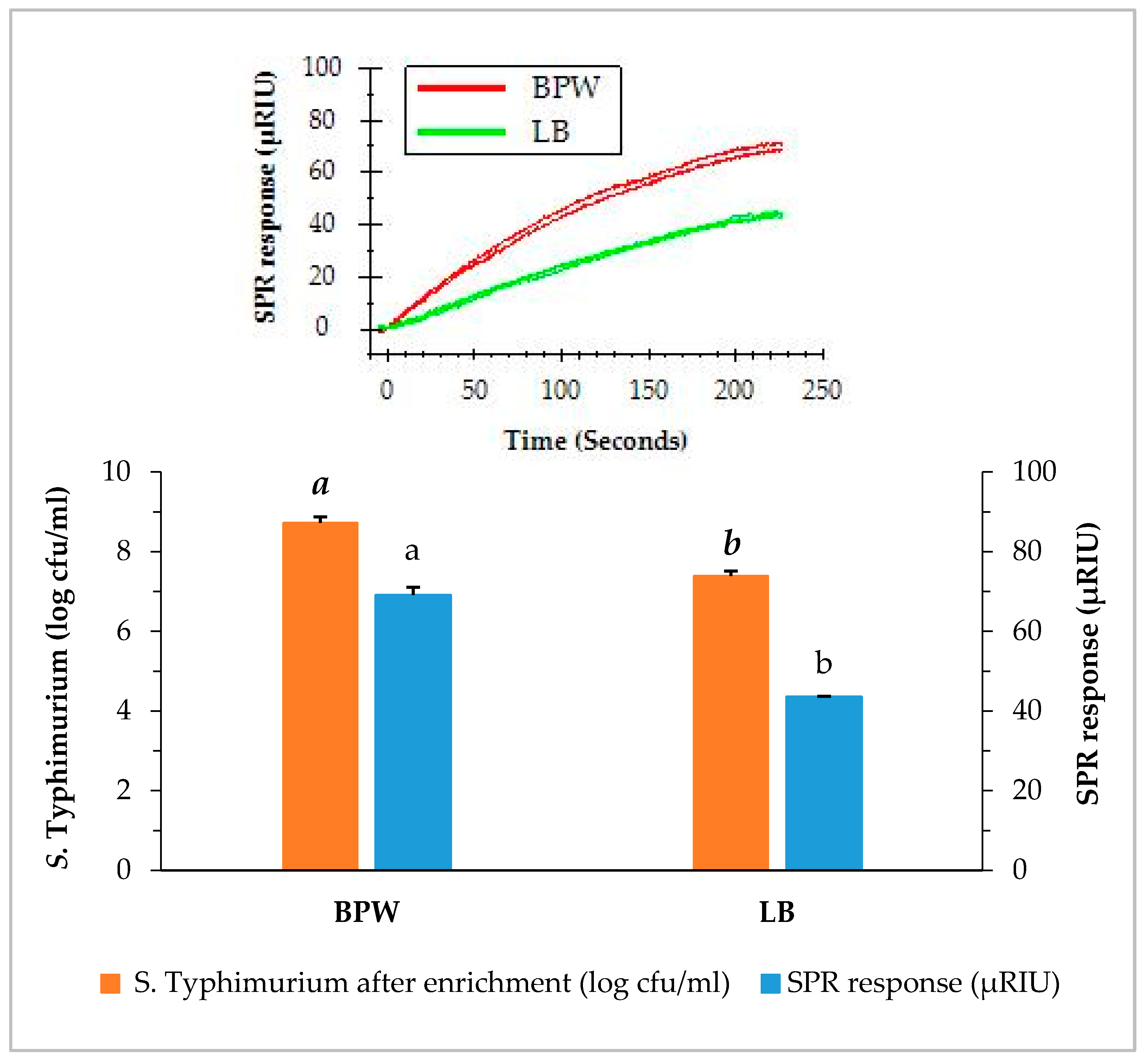
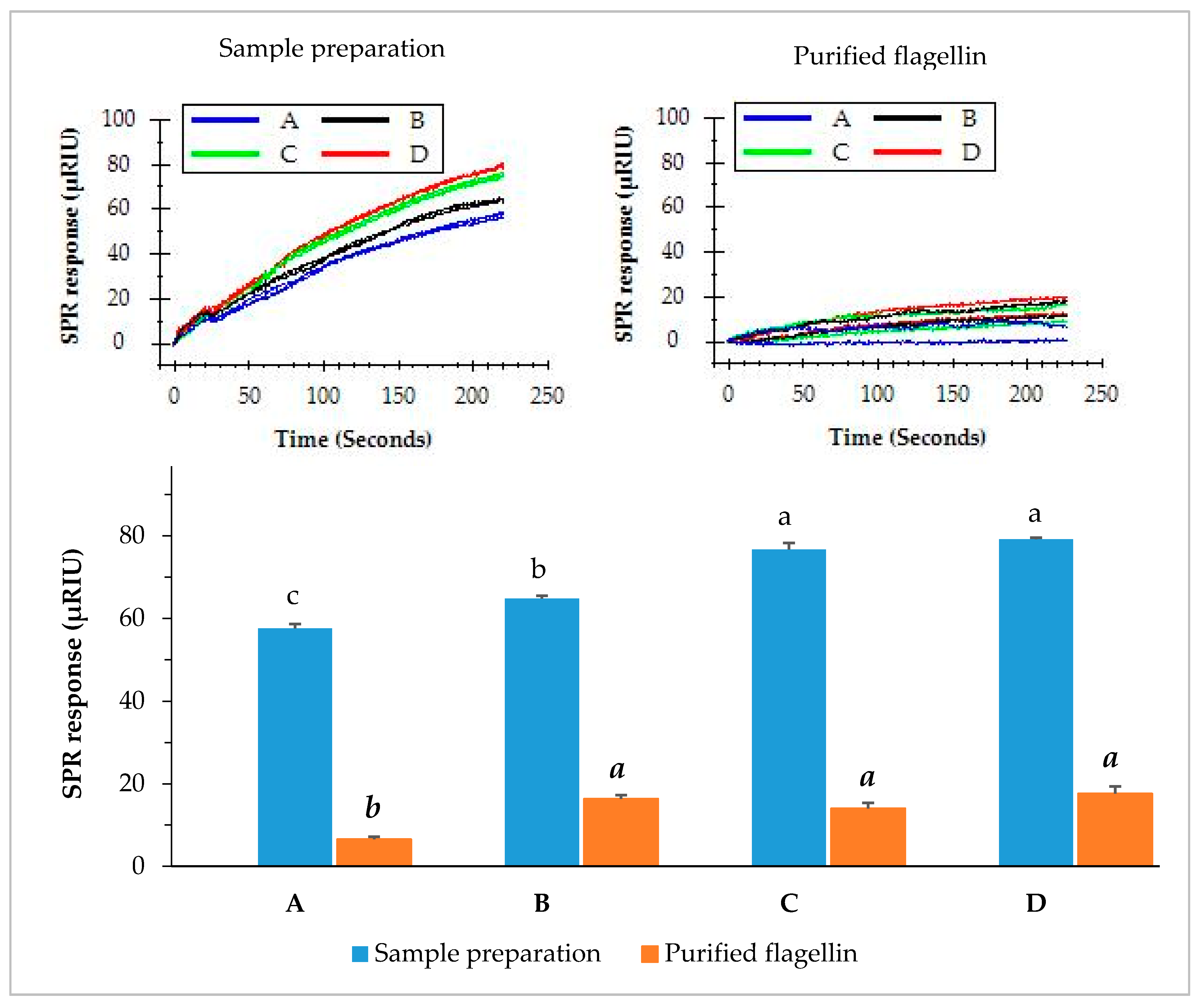
3.2. SPR Assay of Contaminated Romaine Lettuce Using Two Enrichment Methods
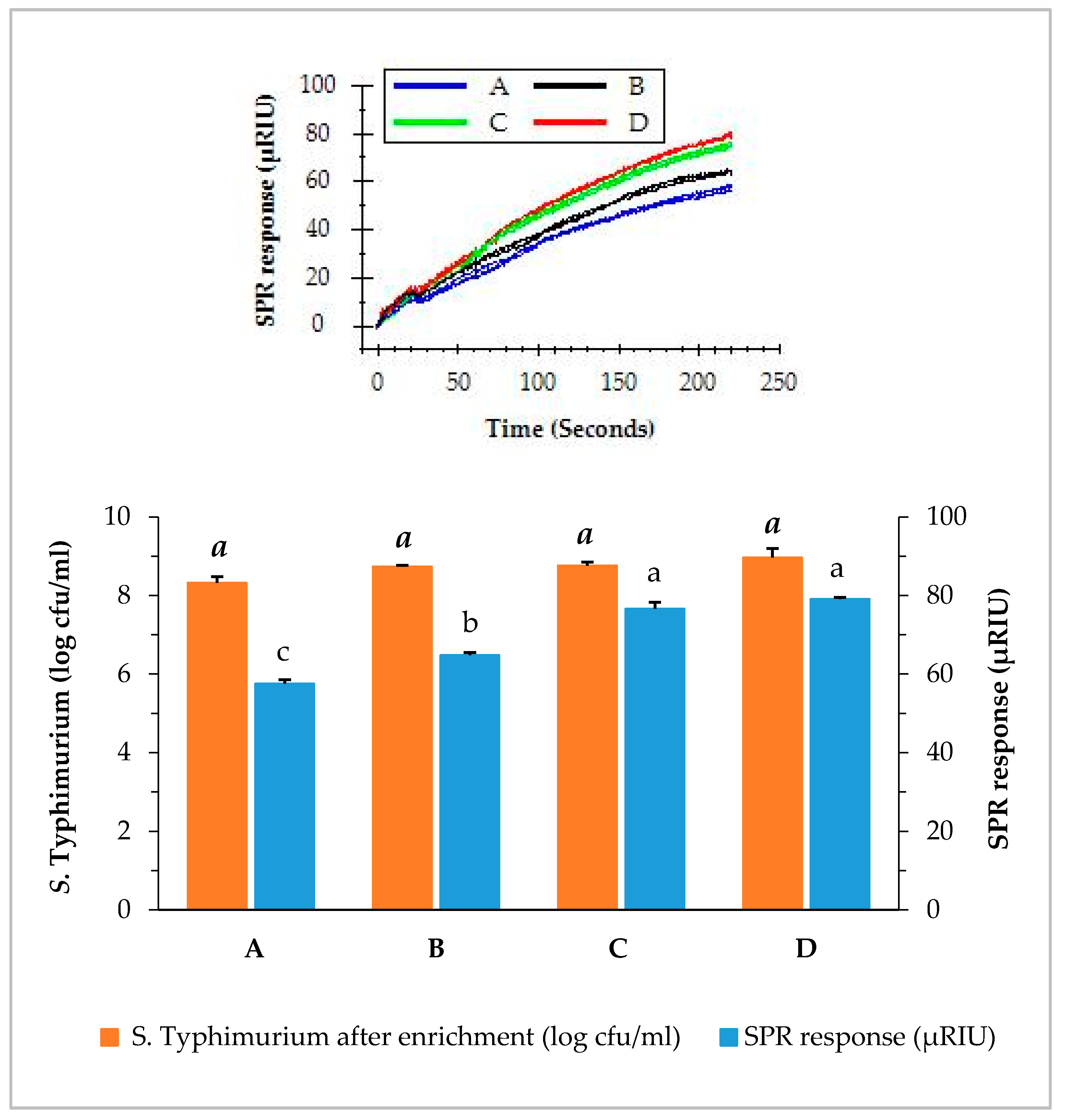
3.3. SPR Assay of Romaine Lettuce with Different Contamination Levels
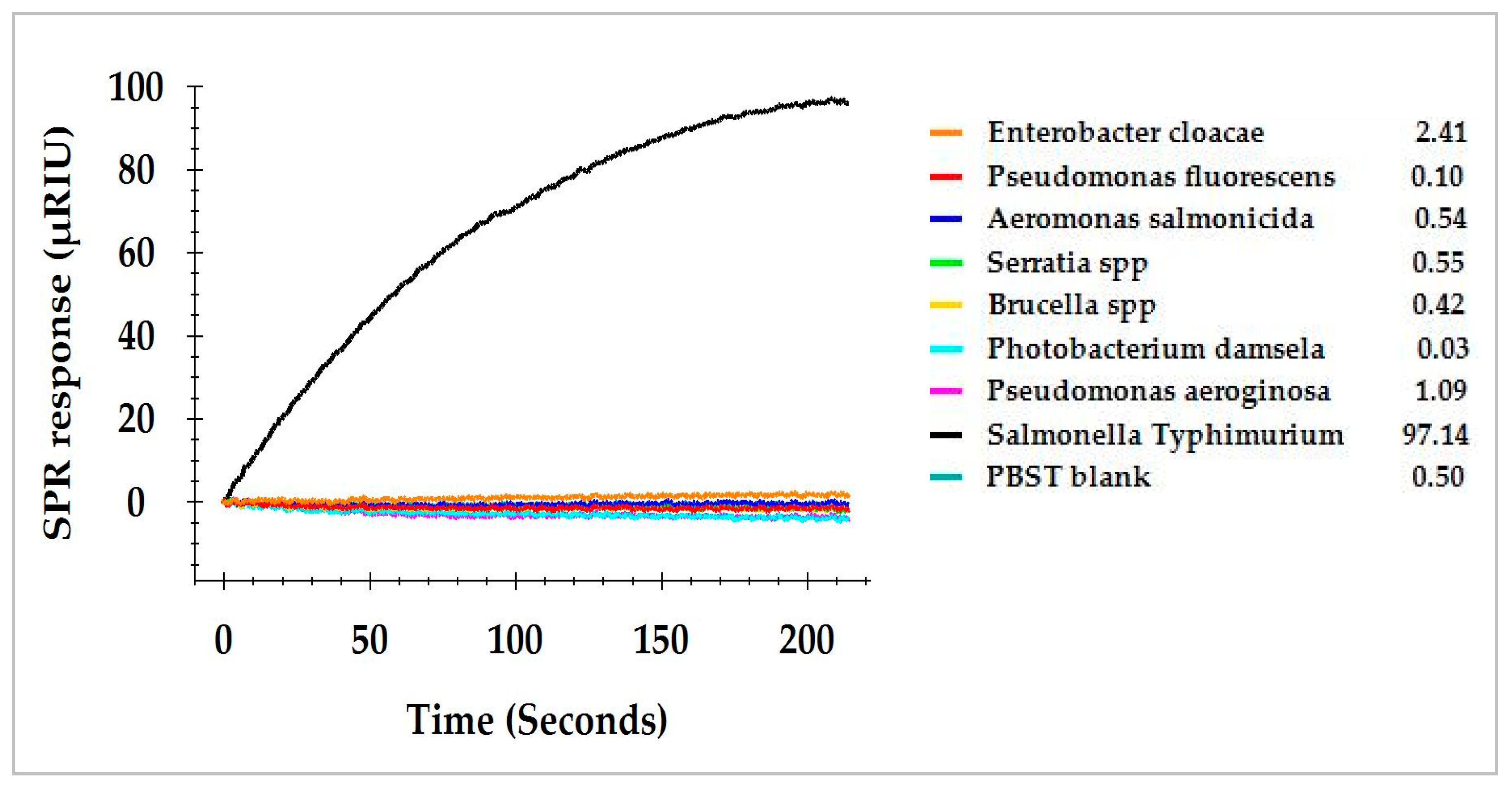
3.4. SPR Assay of Commensal Bacteria Isolated from Romaine Lettuce
3.5. SPR Assay of Contaminated Romaine Lettuce Using Immunomagnetic Separation
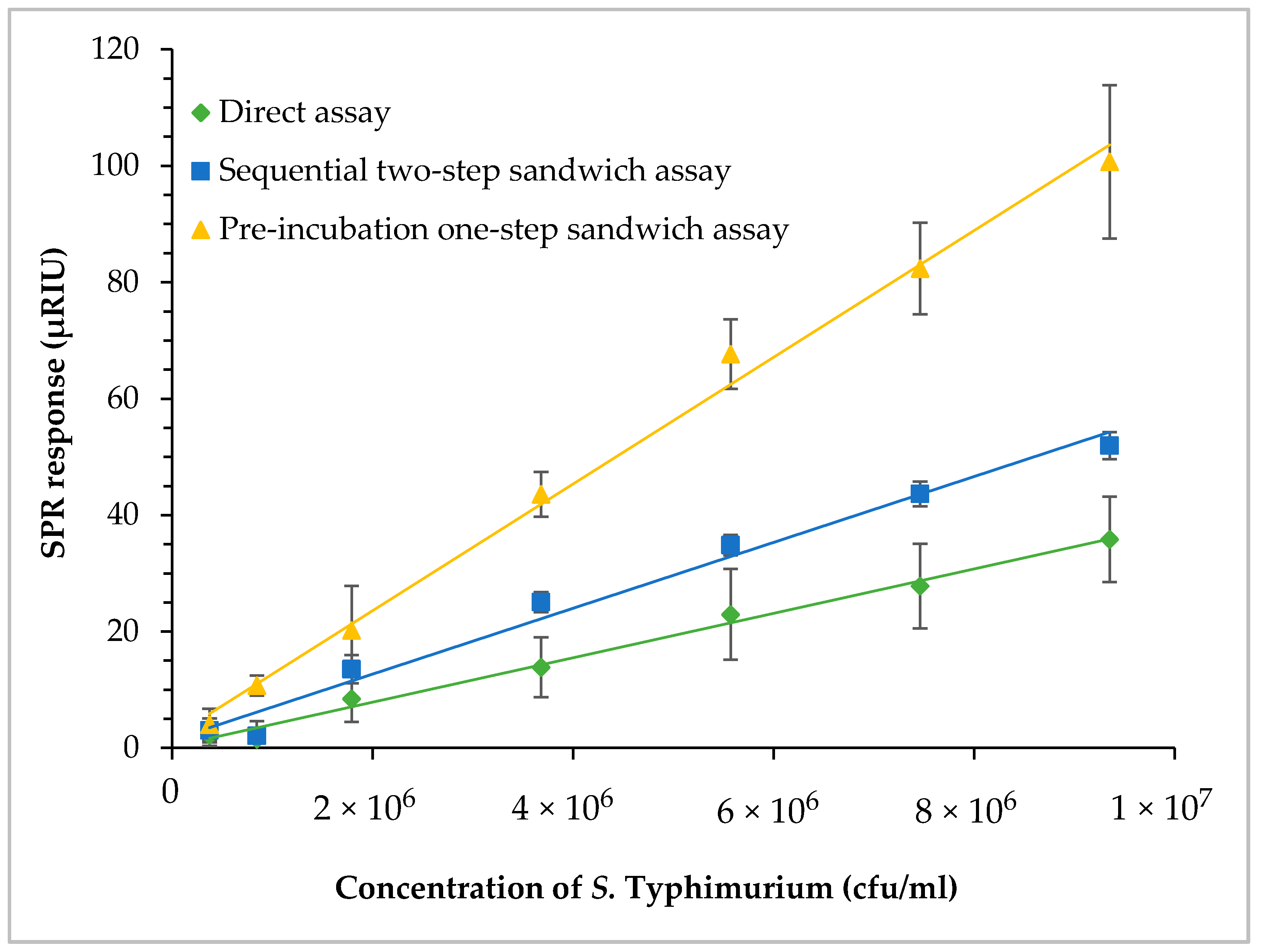
3.6. SPR Assay of Contaminated Romaine Lettuce Using Different Assay Formats
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hendriksen, R.S.; Vieira, A.R.; Karlsmose, S.; Lo Fo Wong, D.M.A.; Jensen, A.B.; Wegener, H.C.; Aarestrup, F.M. Global monitoring of Salmonella serovar distribution from the world health organization global foodborne infections network country data Bank: Results of quality assured laboratories from 2001 to 2007. Foodborne Pathog. Dis. 2011, 8, 887–900. [Google Scholar] [CrossRef] [PubMed]
- Scallan, E.; Hoekstra, R.M.; Angulo, F.J.; Tauxe, R.V.; Widdowson, M.A.; Roy, S.L.; Jones, J.L.; Griffin, P.M. Foodborne illness acquired in the United States-Major pathogens. Emerg. Infect. Dis. 2011, 17, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, S.; Batz, M.B.; Morris, J.G. Annual cost of illness and quality-adjusted life year losses in the United States due to 14 foodborne pathogens. J. Food Prot. 2012, 75, 1292–1302. [Google Scholar] [CrossRef] [PubMed]
- Galanis, E.; Lo Fo Wong, D.M.A.; Patrick, M.E.; Binsztein, N.; Cieslik, A.; Chalermchikit, T.; Aidara-Kane, A.; Ellis, A.; Angulo, F.J.; Wegener, H.C.; et al. Web-based surveillance and global Salmonella distribution, 2000–2002. Emerg. Infect. Dis. 2006, 12, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Andrew, W.H.; Wang, H.; Jacobson, A.; Hammack, T. Bacteriological analytical manual (BAM): Salmonella. Available online: https://www.fda.gov/food/laboratory-methods-food/bacteriological-analytical-manual-bam-chapter-5-salmonella (accessed on 26 July 2019).
- Lee, K.-M.; Runyon, M.; Herrman, T.J.; Phillips, R.; Hsieh, J. Review of Salmonella detection and identification methods: Aspects of rapid emergency response and food safety. Food Control. 2015, 47, 264–276. [Google Scholar] [CrossRef]
- Beumer, R.R.; Brinkman, E.; Rombouts, F.M. Enzyme-linked immunoassays for the detection of Salmonella spp.: A comparison with other methods. Int. J. Food Microbiol. 1991, 12, 363–374. [Google Scholar] [CrossRef]
- Lee, H.A.; Wyatt, G.M.; Bramham, S.; Morgan, M.R. Enzyme-linked immunosorbent assay for Salmonella Typhimurium in food: Feasibility of 1-day Salmonella detection. Appl. Environ. Microbiol. 1990, 56, 1541–1546. [Google Scholar] [PubMed]
- Minnich, S.A.; Hartman, P.A.; Heimmunomagnetic Separationch, R.C. Enzyme immunoassay for detection of Salmonellae in foods. Appl. Environ. Microbiol. 1982, 43, 877–893. [Google Scholar] [PubMed]
- Valdivieso-Garcia, A.; Riche, E.; Abubakar, O.; Waddell, T.E.; Brooks, B.W. A Double antibody sandwich enzyme-linked immunosorbent assay for the detection of Salmonella using biotinylated monoclonal antibodies. J. Food Prot. 2001, 64, 1166–1171. [Google Scholar] [CrossRef]
- Kumar, R.; Surendran, P.K.; Thampuran, N. Evaluation of culture, ELISA and PCR assays for the detection of Salmonella in seafood. Lett. Appl. Microbiol. 2007, 46, 221–226. [Google Scholar] [CrossRef]
- Riyaz-Ul-Hassan, S.; Verma, V.; Qazi, G.N. Rapid detection of Salmonella by polymerase chain reaction. Mol. Cell. Probes 2004, 18, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Bennasar, A.; De Luna, G.; Cabrer, B.; Lalucat, J. Rapid identification of Salmonella Typhimurium, S. Enteritidis and S. virchow isolates by polymerase chain reaction based fingerprinting methods. Int. Microbiol. 2000, 3, 31–38. [Google Scholar] [PubMed]
- Lin, J.-S.; Tsen, H.-Y. Development and use of polymerase chain reaction for the specific detection of Salmonella Typhimurium in stool and food samples. J. Food Prot. 1999, 62, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, S.; Fratamico, P.M.; Horikoshi, N.; Okada, Y.; Takeshita, K.; Sameshima, T.; Kawamoto, S. Multiplex real-time polymerase chain reaction assay for simultaneous detection and quantification of Salmonella species, Listeria monocytogenes, and Escherichia coli O157:H7 in Ground Pork Samples. Foodborne Pathog. Dis. 2010, 7, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Cocolin, L.; Manzano, M.; Cantoni, C.; Comi, G. Use of polymerase chain reaction and restriction enzyme analysis to directly detect and identify Salmonella Typhimurium in food. J. Appl. Microbiol. 1998, 85, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Malorny, B.; Paccassoni, E.; Fach, P.; Bunge, C.; Martin, A.; Helmuth, R. Diagnostic real-time PCR for detection of Salmonella in food. Appl. Environ. Microbiol. 2004, 70, 7046–7052. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.-F.; Cao, W.-W.; Cerniglia, C.E. A universal protocol for PCR detection of 13 species of foodborne pathogens in foods. J. Appl. Microbiol. 1997, 83, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Rossen, L.; Nørskov, P.; Holmstrøm, K.; Rasmussen, O.F. Inhibition of PCR by components of food samples, microbial diagnostic assays and DNA-extraction solutions. Int. J. Food Microbiol. 1992, 17, 37–45. [Google Scholar] [CrossRef]
- Abu Al-Soud, W.; Rådström, P. Effects of amplification facilitators on diagnostic PCR in the presence of blood, feces, and meat. J. Clin. Microbiol. 2000, 38, 4463–4470. [Google Scholar] [PubMed]
- Pathmanathan, S.G.; Cardona-Castro, N.; Sánchez-Jiménez, M.M.; Correa-Ochoa, M.M.; Puthucheary, S.D.; Thong, K.L. Simple and rapid detection of Salmonella strains by direct PCR amplification of the hilA gene. J. Med. Microbiol. 2003, 52, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Homola, J. Present and future of surface plasmon resonance biosensors. Anal. Bioanal. Chem. 2003, 377, 528–539. [Google Scholar] [CrossRef] [PubMed]
- Bokken, G.C.A.M.; Corbee, R.J.; Van Knapen, F.; Bergwerff, A.A. Immunochemical detection of Salmonella group B, D and E using an optical surface plasmon resonance biosensor. FEMS Microbiol. Lett. 2003, 222, 75–82. [Google Scholar] [CrossRef]
- Lan, Y.; Wang, S.; Yin, Y.; Hoffmann, W.C.; Zheng, X. Using a surface plasmon resonance biosensor for rapid detection of Salmonella Typhimurium in chicken carcass. J. Bionic Eng. 2008, 5, 239–246. [Google Scholar] [CrossRef]
- Oh, B.-K.; Kim, Y.-K.; Park, K.W.; Lee, W.H.; Choi, J.-W. Surface plasmon resonance immunosensor for the detection of Salmonella Typhimurium. Biosens. Bioelectron. 2004, 19, 1497–1504. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.D.; Ladd, J.; Yu, Q.; Chen, S.; Homola, J.; Jiang, S. Quantitative and simultaneous detection of four foodborne bacterial pathogens with a multi-channel SPR sensor. Biosens. Bioelectron. 2006, 22, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Mazumdar, S.D.; Hartmann, M.; Kämpfer, P.; Keusgen, M. Rapid method for detection of Salmonella in milk by surface plasmon resonance (SPR). Biosens. Bioelectron. 2007, 22, 2040–2046. [Google Scholar] [CrossRef] [PubMed]
- Rementeria, A.; Vivanco, A.B.; Ramirez, A.; Hernando, F.L.; Bikandi, J.; Herrera-León, S.; Echeita, A.; Garaizar, J. Characterization of a monoclonal antibody directed against Salmonella enterica serovar Typhimurium and serovar. Appl. Environ. Microbiol. 2009, 75, 1345–1354. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, D.; Chen, F.C.; Hamal, S.; Bridgeman, R.C. Kinetic analysis and epitope mapping of monoclonal antibodies to Salmonella Typhimurium flagellin using a surface plasmon resonance biosensor. Antibodies 2019, 8, 22. [Google Scholar] [CrossRef]
- ISO 6579-1:2017. Microbiology of food and animal feeding stuffs-horizontal method for detection of Salmonella spp. Available online: https://www.iso.org/standard/56712.html (accessed on 26 July 2019).
- Löfström, C.; Knutsson, R.; Axelsson, C.E.; Rådström, P. Rapid and specific detection of Salmonella spp. in animal feed samples by PCR after culture enrichment. Appl. Environ. Microbiol. 2004, 70, 69–75. [Google Scholar] [CrossRef]
- Ohtsuka, K.; Yanagawa, K.; Takatori, K.; Hara-Kudo, Y. Detection of Salmonella Enterica in naturally contaminated liquid eggs by loop-mediated isothermal amplification, and characterization of Salmonella isolates. Appl. Environ. Microbiol. 2005, 71, 6730–6735. [Google Scholar] [CrossRef]
- Bej, A.K.; Mahbubani, M.H.; Boyce, M.J.; Atlas, R.M. Detection of Salmonella spp. in oysters by PCR. Appl. Environ. Microbiol. 1994, 60, 368–373. [Google Scholar]
- Salam, F.; Tothill, I.E. Detection of Salmonella Typhimurium using an electrochemical immunosensor. Biosens. Bioelectron. 2009, 24, 2630–2636. [Google Scholar] [CrossRef]
- Saroj, S.D.; Shashidhar, R.; Karani, M.; Bandekar, J.R. Rapid, sensitive, and validated method for detection of Salmonella in food by an enrichment broth culture–Nested PCR combination assay. Mol. Cell. Probes 2008, 22, 201–206. [Google Scholar] [CrossRef]
- Upadhyay, B.P.; Utrarachkij, F.; Thongshoob, J.; Mahakunkijcharoen, Y.; Wongchinda, N.; Suthienkul, O.; Khusmith, S. Detection of Salmonella invA gene in shrimp enrichment culture by polymerase chain reaction. Southeast. Asian J. Trop. Med. Public Health 2010, 41, 426–435. [Google Scholar]
- Maciorowski, K.G.; Pillai, S.D.; Ricke, S.C. Efficacy of a commercial polymerase chain reaction-based assay for detection of Salmonella spp. in animal feeds. J. Appl. Microbiol. 2000, 89, 710–718. [Google Scholar] [CrossRef][Green Version]
- Baylis, C.L.; MacPhee, S.; Betts, R.P. Comparison of two commercial preparations of buffered peptone water for the recovery and growth of Salmonella bacteria from foods. J. Appl. Microbiol. 2000, 89, 501–510. [Google Scholar] [CrossRef]
- Smith, C. Striving for purity: Advances in protein purification. Nat. Methods 2005, 2, 71–77. [Google Scholar] [CrossRef]
- Trkov, M.; Majeríková, I.; Jerašek, B.; Štefanovičová, A.; Rijpens, N.; Kuchta, T. Detection of Salmonella in food over 30 h using enrichment and polymerase chain reaction. Food Microbiol. 1999, 16, 393–399. [Google Scholar] [CrossRef]
- Rijpens, N.; Herman, L.; Vereecken, F.; Jannes, G.; De Smedt, J.; De Zutter, L. Rapid detection of stressed Salmonella spp. in dairy and egg products using immunomagnetic separation and PCR. Int. J. Food Microbiol. 1999, 46, 37–44. [Google Scholar] [CrossRef]
- Cudjoe, K.S.; Hagtvedt, T.; Dainty, R. Immunomagnetic separation of Salmonella from foods and their detection using immunomagnetic particle (IMP)-ELISA. Int. J. Food Microbiol. 1995, 27, 11–25. [Google Scholar] [CrossRef]
- Cudjoe, K.S.; Krona, R. Detection of Salmonella from raw food samples using Dynabeads® anti-Salmonella and a conventional reference method. Int. J. Food Microbiol. 1997, 37, 55–62. [Google Scholar] [CrossRef]
- Edelman, G.M.; Cunningham, B.A.; Gall, W.E.; Gottlieb, P.D.; Rutishauser, U.; Waxdal, M.J. The covalent structure of an entire gammaG immunoglobulin molecule. Proc. Natl. Acad. Sci. USA 1969, 63, 78–85. [Google Scholar] [CrossRef]
- Kondoh, H.; Hotani, H. Flagellin from Escherichia coli K12: Polymerization and molecular weight in comparison with Salmonella flagellins. BBA-Protein Struct. 1974, 336, 117–139. [Google Scholar] [CrossRef]
- Homola, J. Surface plasmon resonance sensors for detection of chemical and biological species. Chem. Rev. 2008, 108, 462–493. [Google Scholar] [CrossRef]
- Soelberg, S.D.; Stevens, R.C.; Limaye, A.P.; Furlong, C.E. Surface plasmon resonance detection using antibody-linked magnetic nanoparticles for analyte capture, purification, concentration, and signal amplification. Anal. Chem. 2009, 81, 2357–2363. [Google Scholar] [CrossRef]
- Wolffs, P.F.G.; Glencross, K.; Thibaudeau, R.; Griffiths, M.W. Direct quantitation and detection of Salmonellae in biological samples without enrichment, using two-step filtration and real-time PCR. Appl. Environ. Microbiol. 2006, 72, 3896–3900. [Google Scholar] [CrossRef]
- De Vries, N.; Zwaagstra, K.A.; Huis In’t Veld, J.H.J.; Van Knapen, F.; Van Zijderveld, F.G.; Kusters, J.G. Production of monoclonal antibodies specific for the i and 1,2 flagellar antigens of Salmonella Typhimurium and characterization of their respective epitopes. Appl. Environ. Microbiol. 1998, 64, 5033–5038. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhandari, D.; Chen, F.-C.; Bridgman, R.C. Detection of Salmonella Typhimurium in Romaine Lettuce Using a Surface Plasmon Resonance Biosensor. Biosensors 2019, 9, 94. https://doi.org/10.3390/bios9030094
Bhandari D, Chen F-C, Bridgman RC. Detection of Salmonella Typhimurium in Romaine Lettuce Using a Surface Plasmon Resonance Biosensor. Biosensors. 2019; 9(3):94. https://doi.org/10.3390/bios9030094
Chicago/Turabian StyleBhandari, Devendra, Fur-Chi Chen, and Roger C. Bridgman. 2019. "Detection of Salmonella Typhimurium in Romaine Lettuce Using a Surface Plasmon Resonance Biosensor" Biosensors 9, no. 3: 94. https://doi.org/10.3390/bios9030094
APA StyleBhandari, D., Chen, F.-C., & Bridgman, R. C. (2019). Detection of Salmonella Typhimurium in Romaine Lettuce Using a Surface Plasmon Resonance Biosensor. Biosensors, 9(3), 94. https://doi.org/10.3390/bios9030094




