Recent Improvement of Medical Optical Fibre Pressure and Temperature Sensors
Abstract
:1. Introduction
2. Theoretical Background
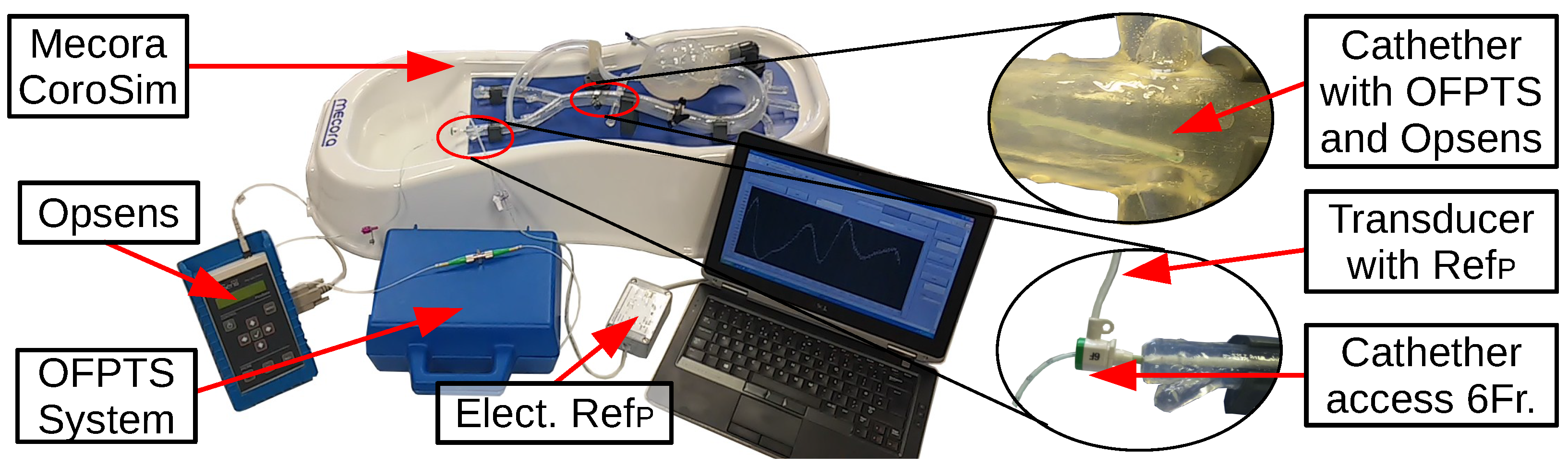
2.1. The Medical Evaluation System
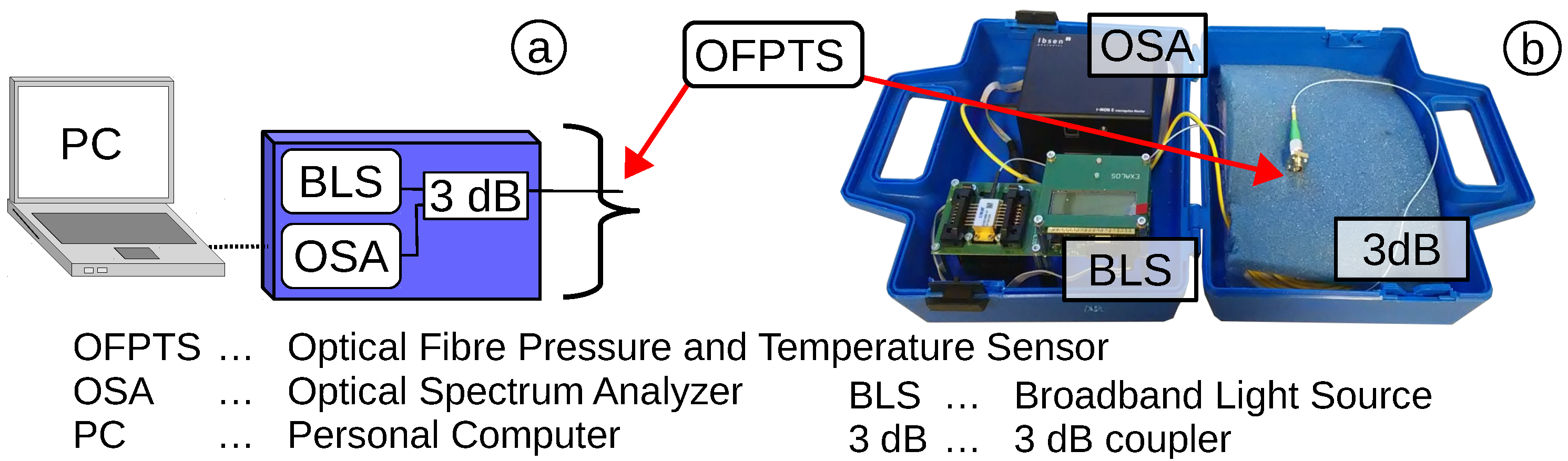
2.2. Optical Fibre Pressure and Temperature Sensor
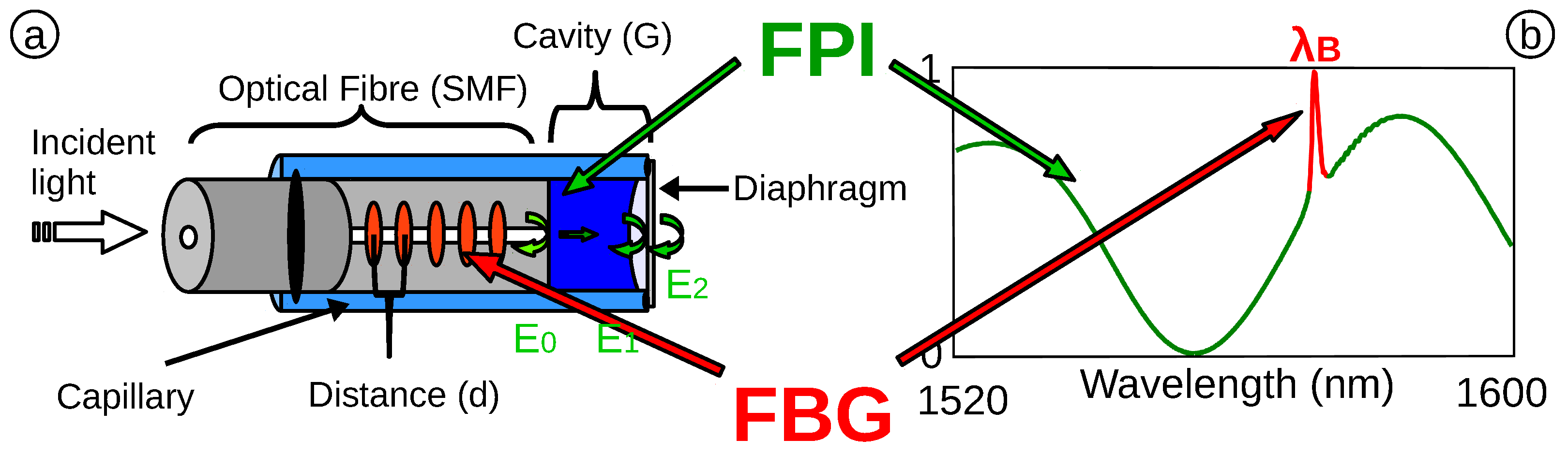
2.2.1. Fabry Perot Interferometer (FPI)
2.2.2. Integrated Fibre Bragg Grating
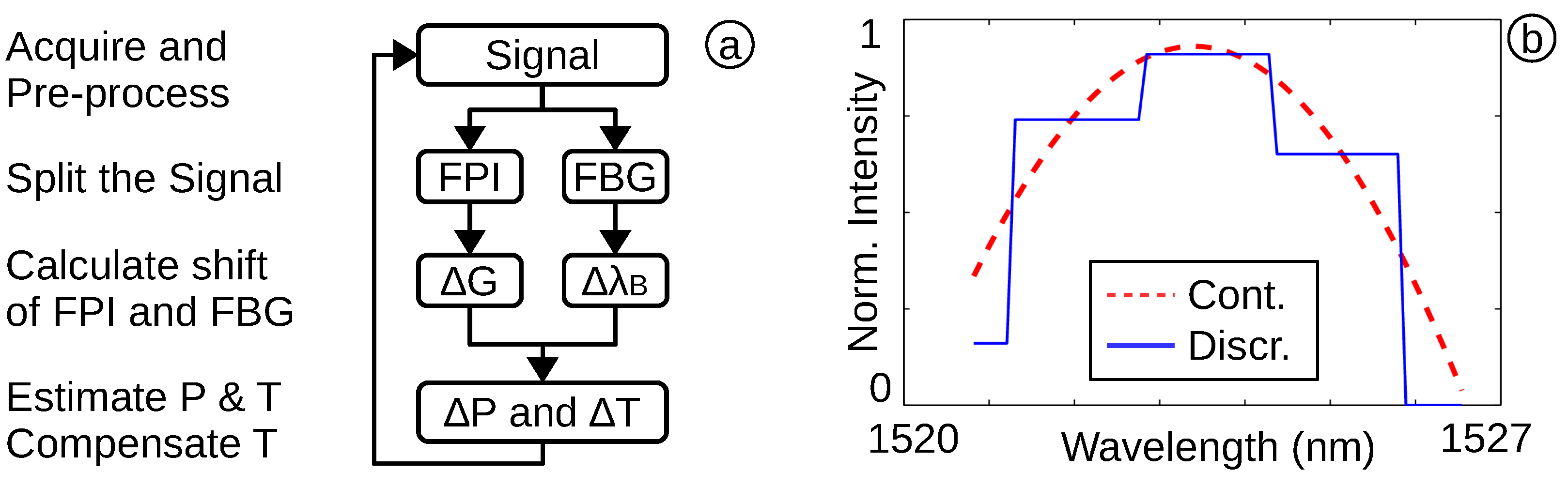
2.3. Software for Medical Evaluation System
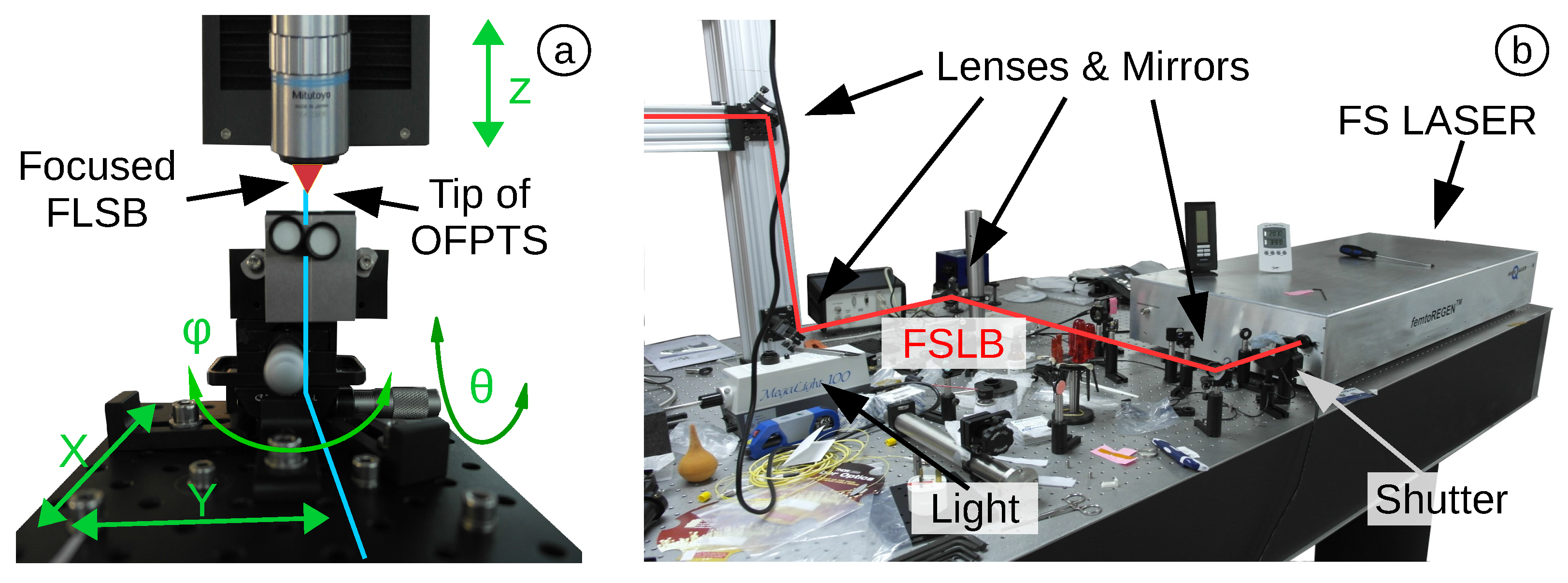
3. Fabrication of an Optical Fibre Pressure and Temperature Sensor (OFPTS)
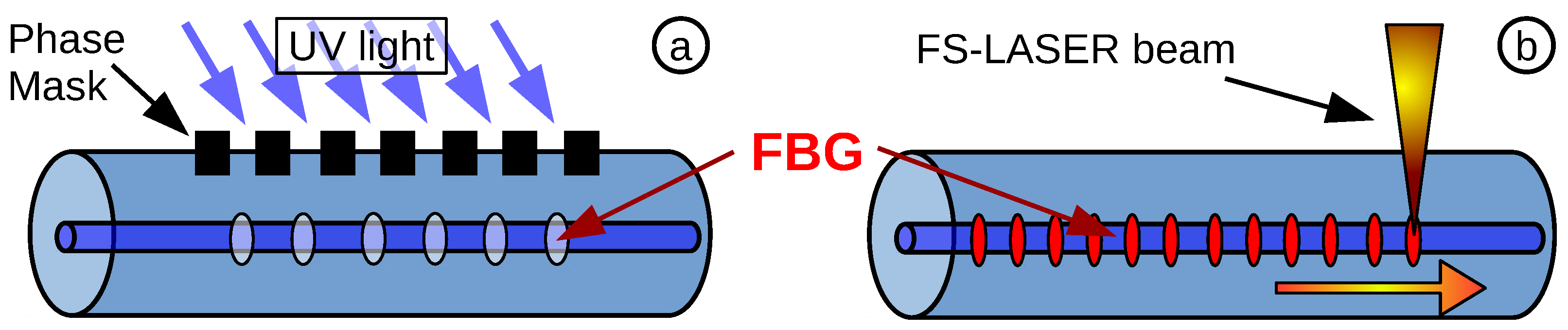
3.1. Fibre Bragg Grating Temperature Sensor
3.2. Fabry–Perot Interferometer Pressure Sensor

3.3. Etching the Diaphragm High-Sensitivity OFPTS
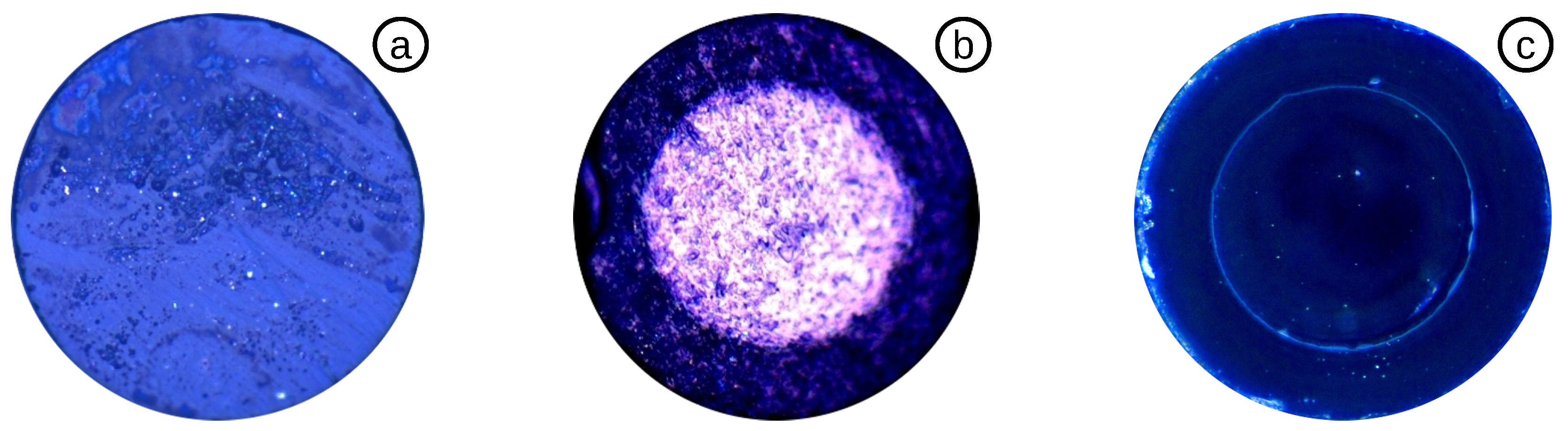
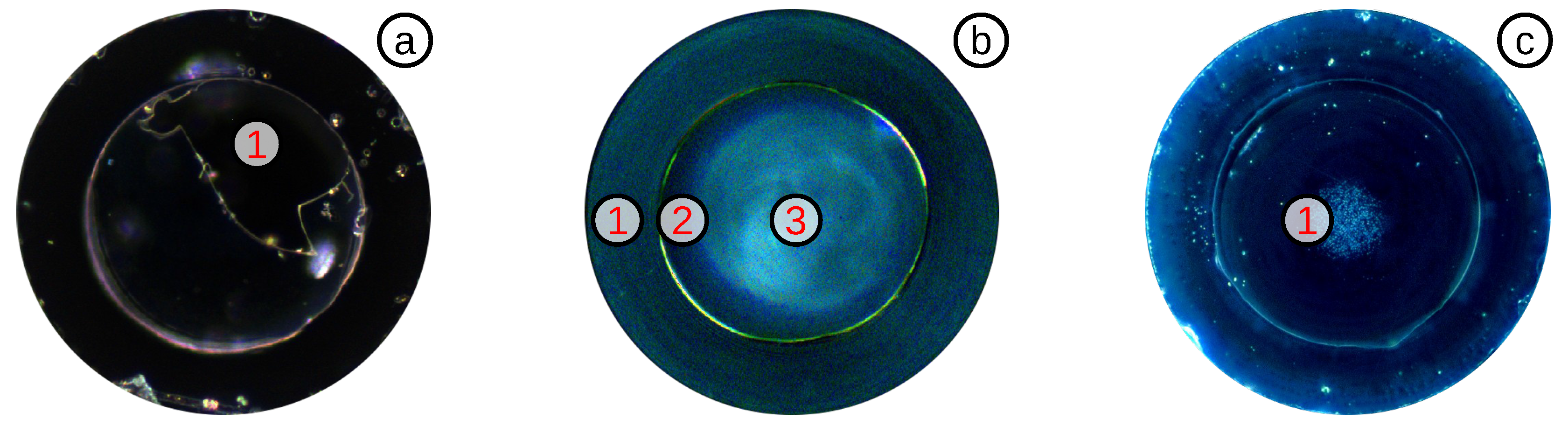
3.3.1. Diaphragm Reduction by HF Acid
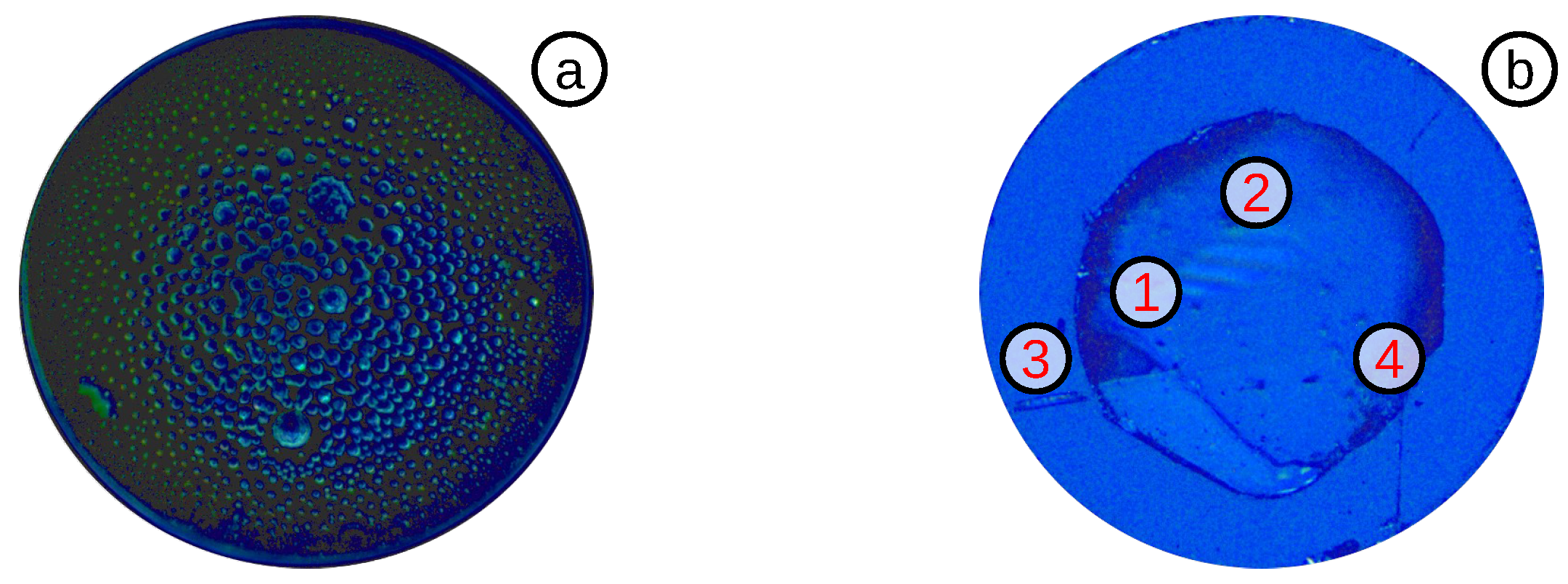
3.3.2. Diaphragm Reduction by a FSL
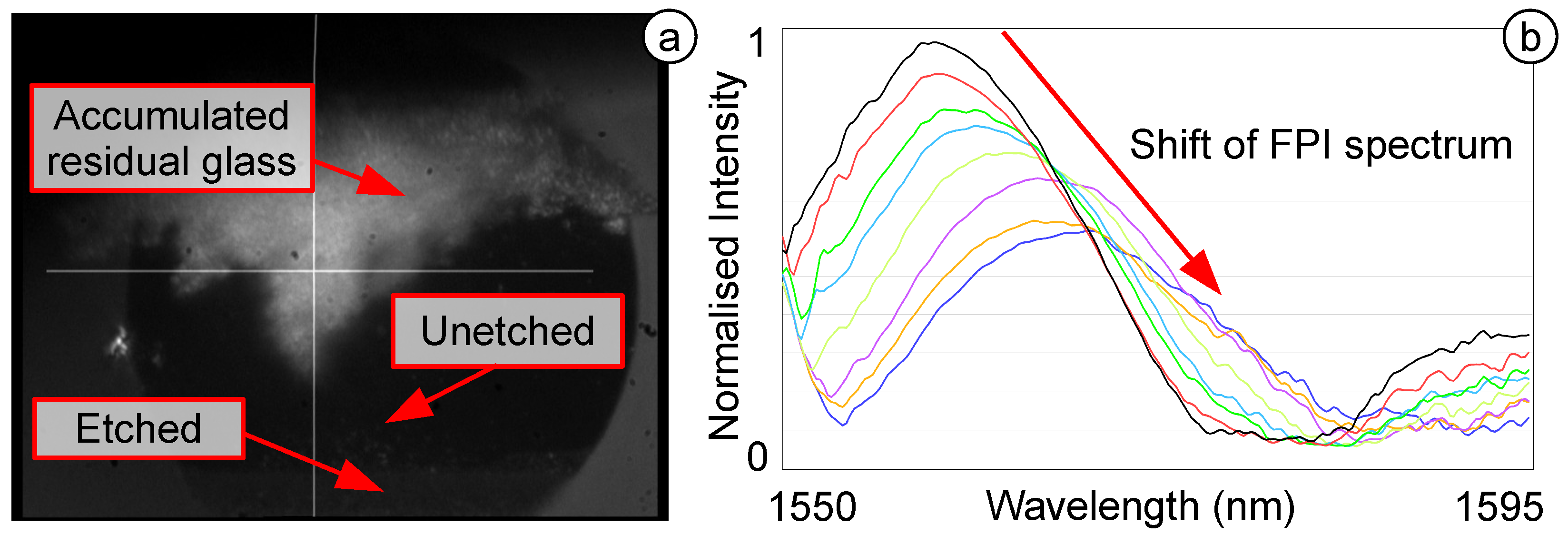
3.3.3. Highly Sensitive OFPTS
4. Testing and Results of the OFPTS System
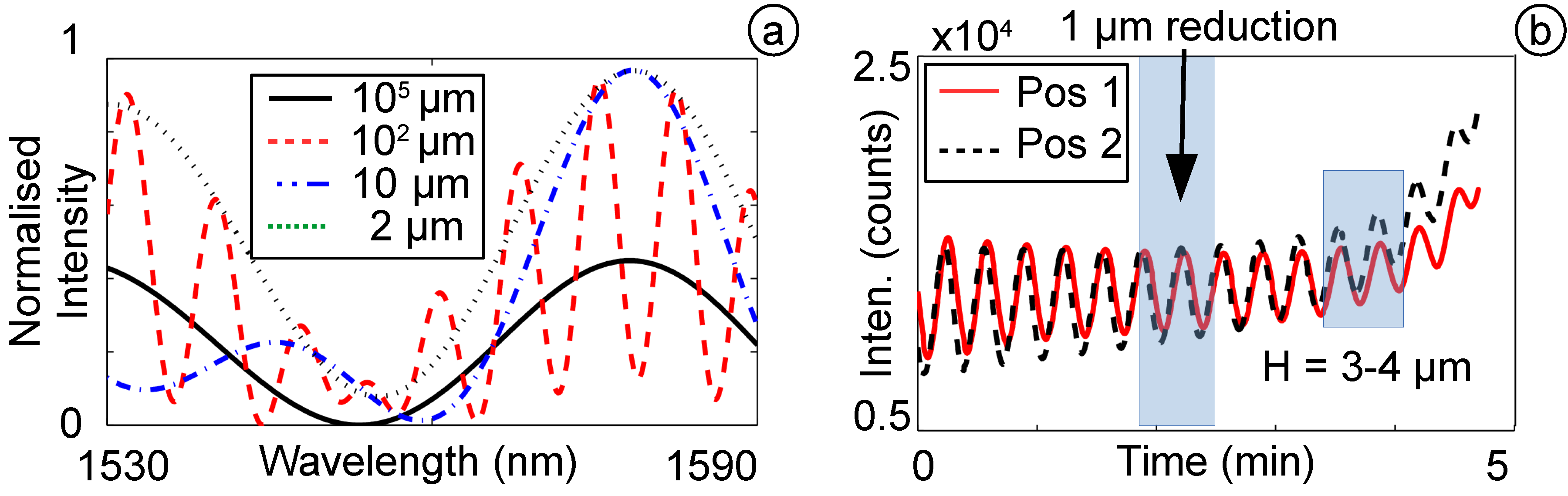
4.1. Pressure Sensitivity and Resolution
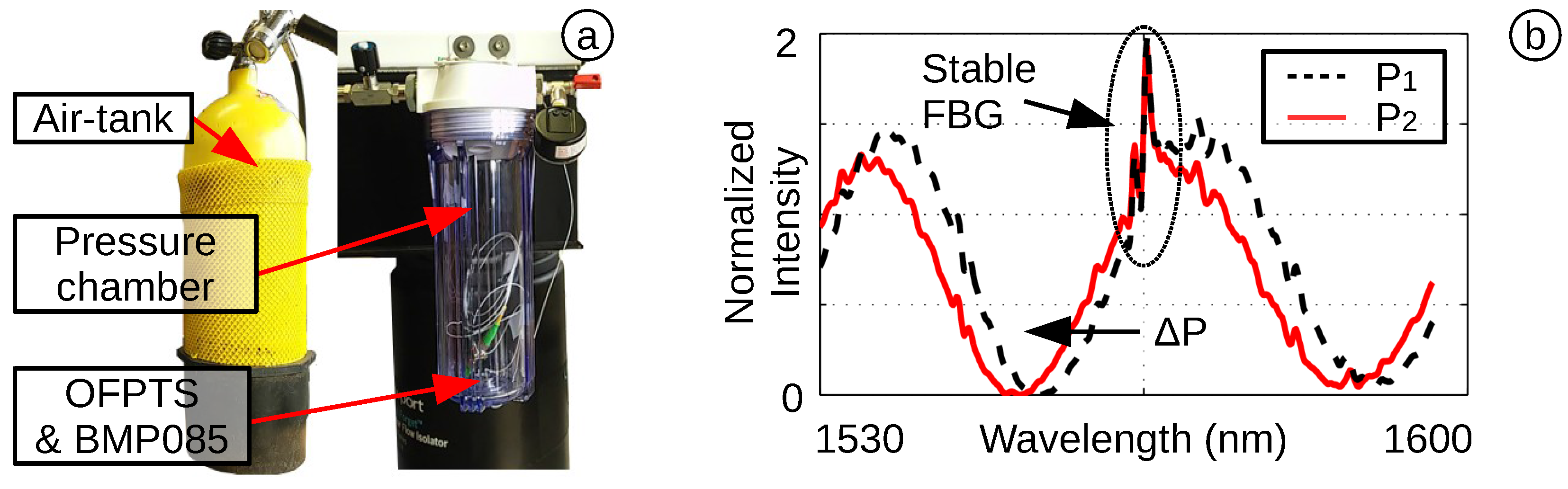
4.2. Temperature Sensitivity and Resolution

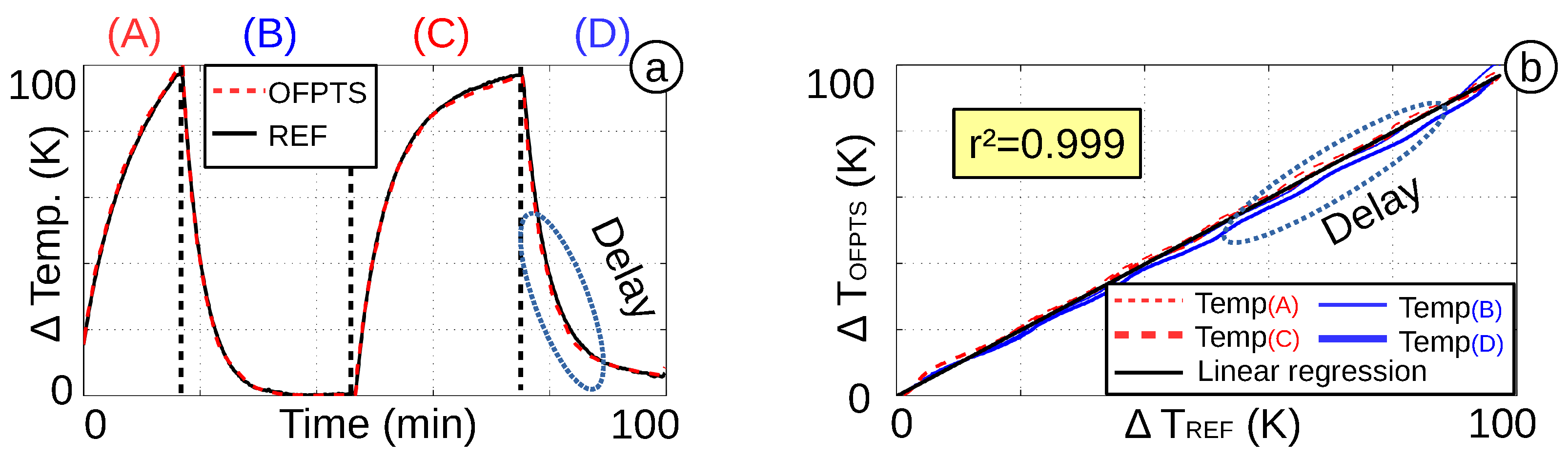
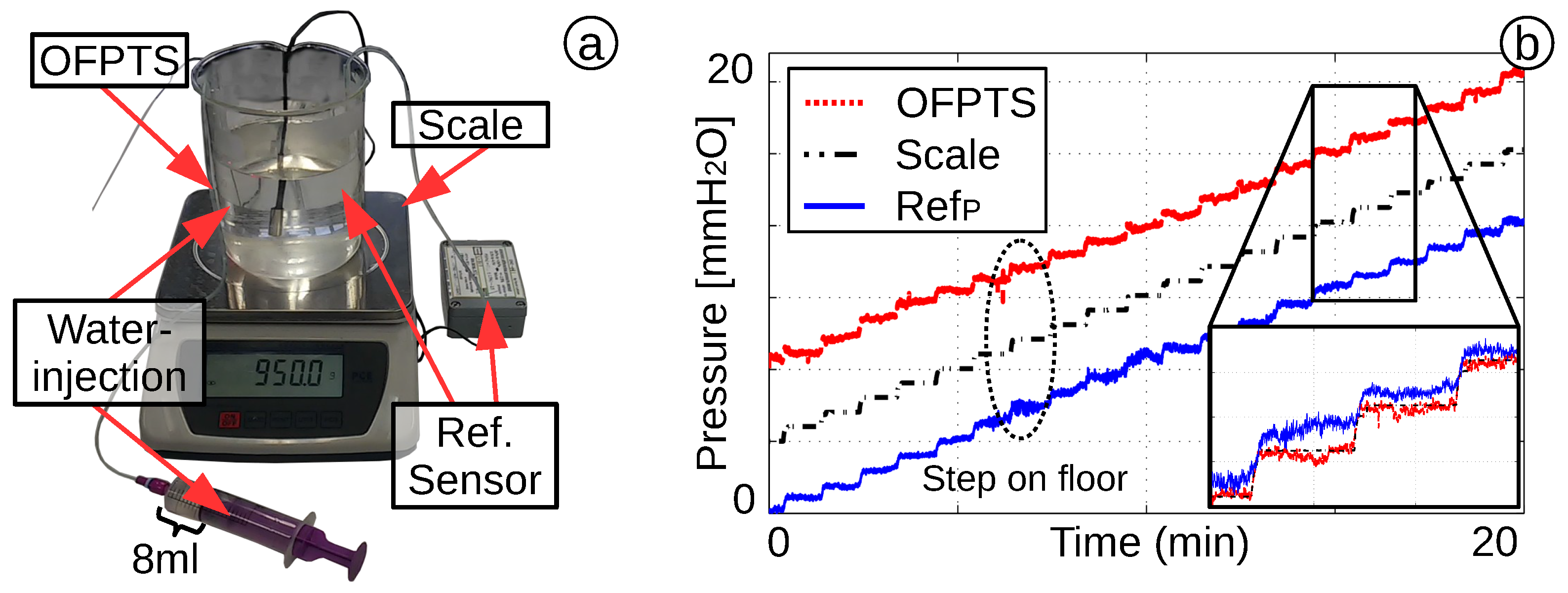
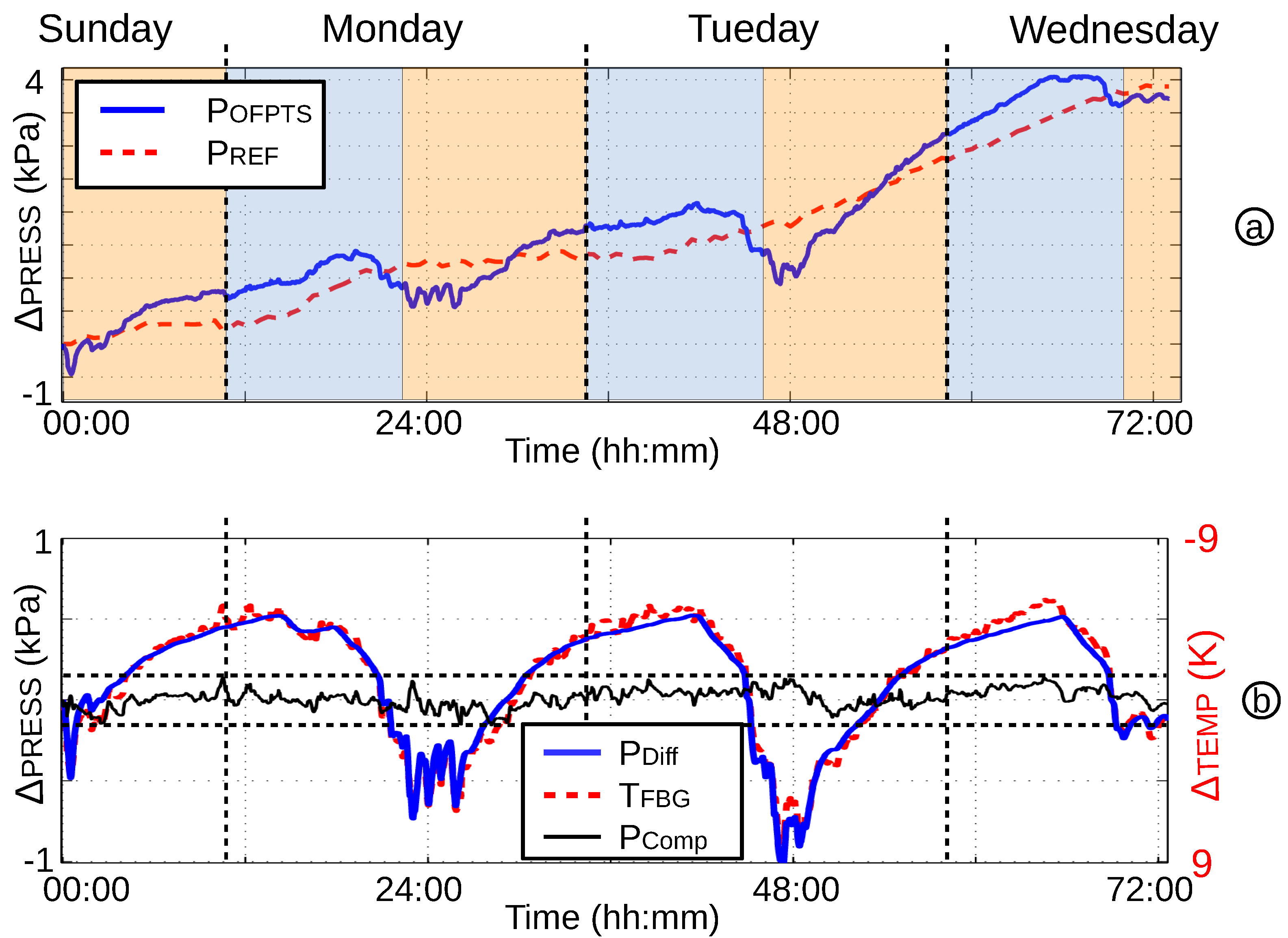
4.3. Short and Long Time Pressure Stability
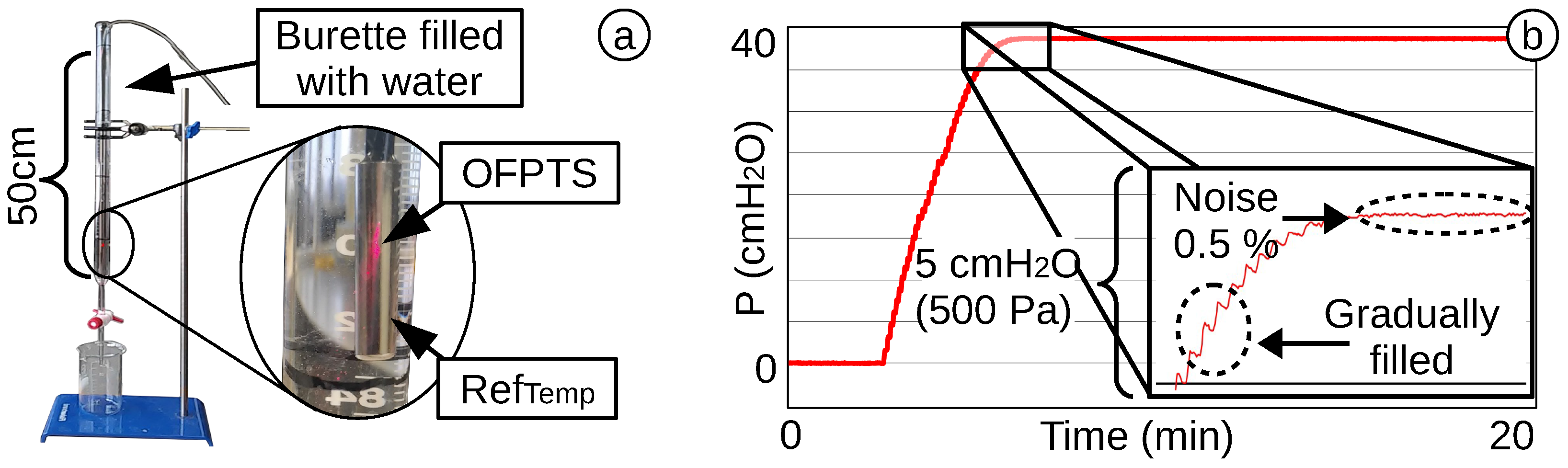
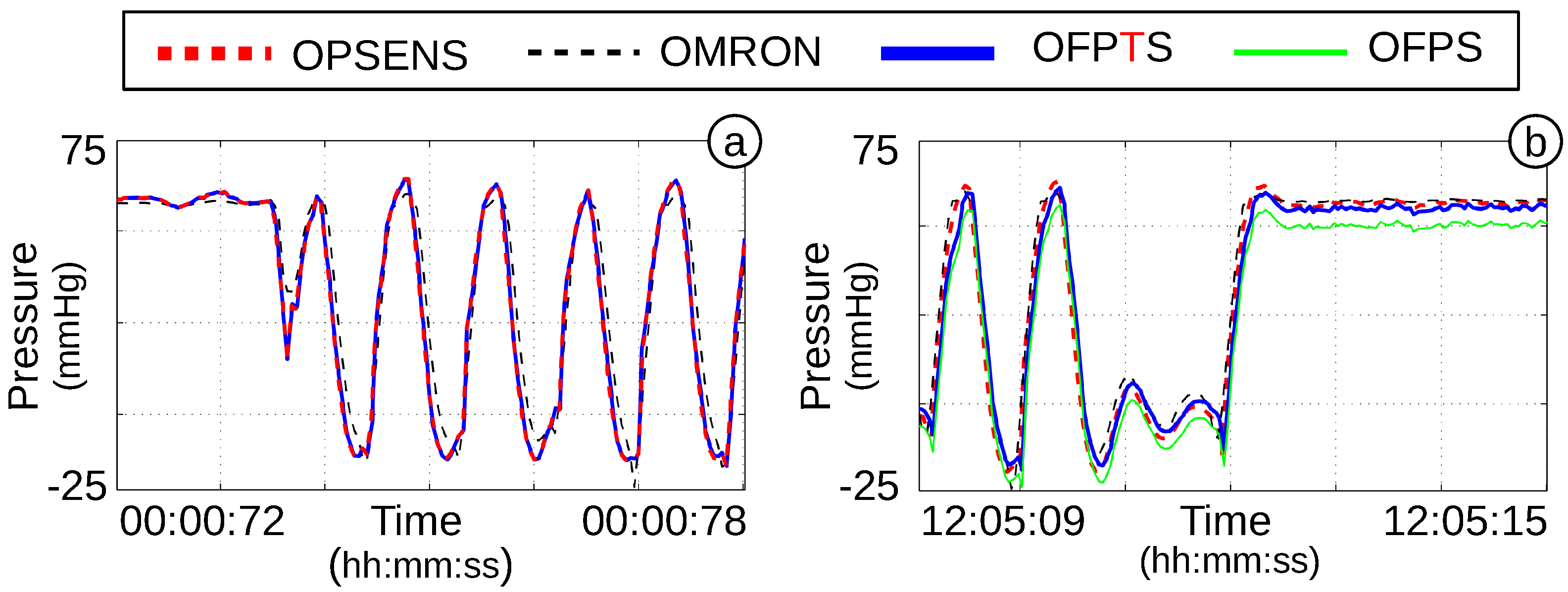
4.4. Dynamic Pressure Response and Stability of the OFPTS

5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roriz, P.; Frazao, O.; Lobo-Ribeiro, A.B.; Santos, J.L.; Simoes, J.A. Review of fiber-optic pressure sensors for biomedical and biomechanical applications. J. Biomed. Opt. 2013, 18, 050903-1–050903-18. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Zhou, X. Pressure sensor based on the fiber-optic extrinsic Fabry-Perot interferometer. Photonic Sens. 2011, 1, 72–83. [Google Scholar] [CrossRef]
- Liu, X.; Iordachita, I.I.; He, X.; Taylor, R.H.; Kang, J.U. Miniature fiber-optic force sensor based on low-coherence Fabry Perot interferometry for vitreoretinal microsurgery. Biomed. Opt. Express 2012, 3, 1062–1076. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Rao, Y.; Wen, W.; Yao, J.; Wu, D.; Xu, L.; Zhu, T. In-line all-fibre Fabry-Perot interferometer high temperature sensor formed by large lateral offset splicing. Electron. Lett. 2011, 47, 401–403. [Google Scholar] [CrossRef]
- Gao, R.; Jiang, Y.; Ding, W.; Wang, Z.; Liu, D. Filmed extrinsic Fabry-Perot interferometric sensors for the measurement of arbitrary refractive index of liquid. Sens. Actuators B Chem. 2013, 177, 924–928. [Google Scholar] [CrossRef]
- Chen, L.; Chan, C.; Yuan, W.; Goh, S.; Sun, J. High performance chitosan diaphragm-based fiber-optic acoustic sensor. Sens. Actuators A Phys. 2010, 163, 42–47. [Google Scholar] [CrossRef]
- Aref, S.; Latifi, H.; Zibaii, M.; Afshari, M. Fiber optic Fabry-Perot pressure sensor with low sensitivity to temperature changes for downhole application. Opt. Commun. 2007, 269, 322–330. [Google Scholar] [CrossRef]
- Leng, J.; Asundi, A. Structural health monitoring of smart composite materials by using EFPI and FBG sensors. Sens. Actuators A Phys. 2003, 103, 330–340. [Google Scholar] [CrossRef]
- Chin, K.; Sun, Y.; Feng, G.; Georgiou, G.; Guo, K.; Niver, E.; Roman, H.; Noe, K. Fabry-Perot diaphragm fiber-optic sensor. Appl. Opt. 2007, 46, 7614–7619. [Google Scholar] [CrossRef] [PubMed]
- Cibula, E.; Pevec, S.; Lenardic, B.; Pinet, E.; Donlagic, D. Miniature all-glass robust pressure sensor. Opt. Express 2009, 17, 5098–5106. [Google Scholar] [CrossRef] [PubMed]
- FDA. Use of International Standard ISO 10993, “Biological Evaluation of Medical Devices Part 1: Evaluation and Testing”U.S. Department of Health and Human Services, Ed.; Draft Guidance Ed.; FDA: Silver Spring, MD, USA, 2013. [Google Scholar]
- Poeggel, S.; Tosi, D.; Duraibabu, D.; Kelly, J.; Munroe, M.; Leen, G.; Lewis, E. Novel diaphragm microfabrication techniques for high-sensitivity biomedical fiber optic Fabry-Perot interferometric sensors. Proc. SPIE 2014, 9098, 909813-1–909813-4. [Google Scholar]
- Rao, Y.J. Recent progress in fiber-optic extrinsic Fabry-Perot interferometric sensors. Opt. Fiber Technol. 2006, 12, 227–237. [Google Scholar] [CrossRef]
- Bremer, K.; Lewis, E.; Leen, G.; Moss, B.; Lochmann, S.; Mueller, I. Feedback Stabilized Interrogation Technique for EFPI/FBG Hybrid Fiber-Optic Pressure and Temperature Sensors. Sens. J. IEEE 2012, 12, 133–138. [Google Scholar] [CrossRef]
- Poeggel, S.; Leen, G.; Bremer, K.; Lewis, E. Miniature optical fiber combined pressure and temperature sensor for medical applications. In Proceedings of the 2012 IEEE Sensors, Taipei, Taiwan, 28–31 October 2012; pp. 1–4.
- Poeggel, S.; Duraibabu, D.; Tosi, D.; Leen, G.; Lewis, E.; McGrath, D.; Fusco, F.; Sannino, S.; Lupoli, L.; Ippolito, J.; et al. Differential in vivo urodynamic measurement in a single thin catheter based on two optical fiber pressure sensors. J. Biomed. Opt. 2015, 20, 037005-1–037005-9. [Google Scholar] [CrossRef] [PubMed]
- Rao, Y.J. In-fibre Bragg grating sensors. Meas. Sci. Technol. 1997, 8, 355–375. [Google Scholar] [CrossRef]
- Othonos, A.; Kalli, K. Fiber Bragg Gratings: Fundamentals and Applications in Telecommunications and Sensing; Artech House: Boston, MA, USA, 1999. [Google Scholar]
- Bremer, K.; Lewis, E.; Moss, B.; Leen, G.; Lochmann, S.; Mueller, I. Conception and preliminary evaluation of an optical fibre sensor for simultaneous measurement of pressure and temperature. J. Phys. Conf. Ser. 2009, 178, 1–4. [Google Scholar] [CrossRef]
- Bae, H.; Yu, M. Miniature Fabry-Perot pressure sensor created by using UV-molding process with an optical fiber based mold. Opt. Express 2012, 20, 14573–14583. [Google Scholar] [CrossRef] [PubMed]
- Poeggel, S.; Duraibabu, D.; Tosi, D.; Leen, G.; Lewis, E.; Lacraz, A.; Hambalis, M.; Koutsides, C.; Kalli, K. Novel FBG femtosecond laser inscription method for improved FPI sensors for medical applications. In Proceedings of the 2014 IEEE Sensors, Valencia, Spain, 2–5 November 2014; pp. 25–28.
- Ranganathan, A. The Levenberg-Marquardt Algorithm. Avialiable online: http://users-phys.au.dk/ jensjh/numeric/project/10.1.1.135.865.pdf (accessed on 1 March 2015).
- Hill, K.O.; Meltz, G. Fiber Bragg grating technology fundamentals and overview. Lightwave Technol. J. 1997, 15, 1263–1276. [Google Scholar] [CrossRef]
- FBGS. FBGS-Draw Tower Gratings. Available online: http://www.fbgs.com/contactform/be-en/1/ detail/item/3/ (accessed on 22 October 2014).
- Koutsides, C.; Davies, E.; Kalli, K.; Komodromos, M.; Allsop, T.; Webb, D.J.; Zhang, L. Superstructure Fiber Gratings Via Single Step Femtosecond Laser Inscription. J. Lightwave Technol. 2012, 30, 1229–1236. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poeggel, S.; Duraibabu, D.; Kalli, K.; Leen, G.; Dooly, G.; Lewis, E.; Kelly, J.; Munroe, M. Recent Improvement of Medical Optical Fibre Pressure and Temperature Sensors. Biosensors 2015, 5, 432-449. https://doi.org/10.3390/bios5030432
Poeggel S, Duraibabu D, Kalli K, Leen G, Dooly G, Lewis E, Kelly J, Munroe M. Recent Improvement of Medical Optical Fibre Pressure and Temperature Sensors. Biosensors. 2015; 5(3):432-449. https://doi.org/10.3390/bios5030432
Chicago/Turabian StylePoeggel, Sven, Dineshbabu Duraibabu, Kyriacos Kalli, Gabriel Leen, Gerard Dooly, Elfed Lewis, Jimmy Kelly, and Maria Munroe. 2015. "Recent Improvement of Medical Optical Fibre Pressure and Temperature Sensors" Biosensors 5, no. 3: 432-449. https://doi.org/10.3390/bios5030432
APA StylePoeggel, S., Duraibabu, D., Kalli, K., Leen, G., Dooly, G., Lewis, E., Kelly, J., & Munroe, M. (2015). Recent Improvement of Medical Optical Fibre Pressure and Temperature Sensors. Biosensors, 5(3), 432-449. https://doi.org/10.3390/bios5030432







