Sensing a Sensor: Identifying the Mechanosensory Function of Primary Cilia
Abstract
:1. Introduction
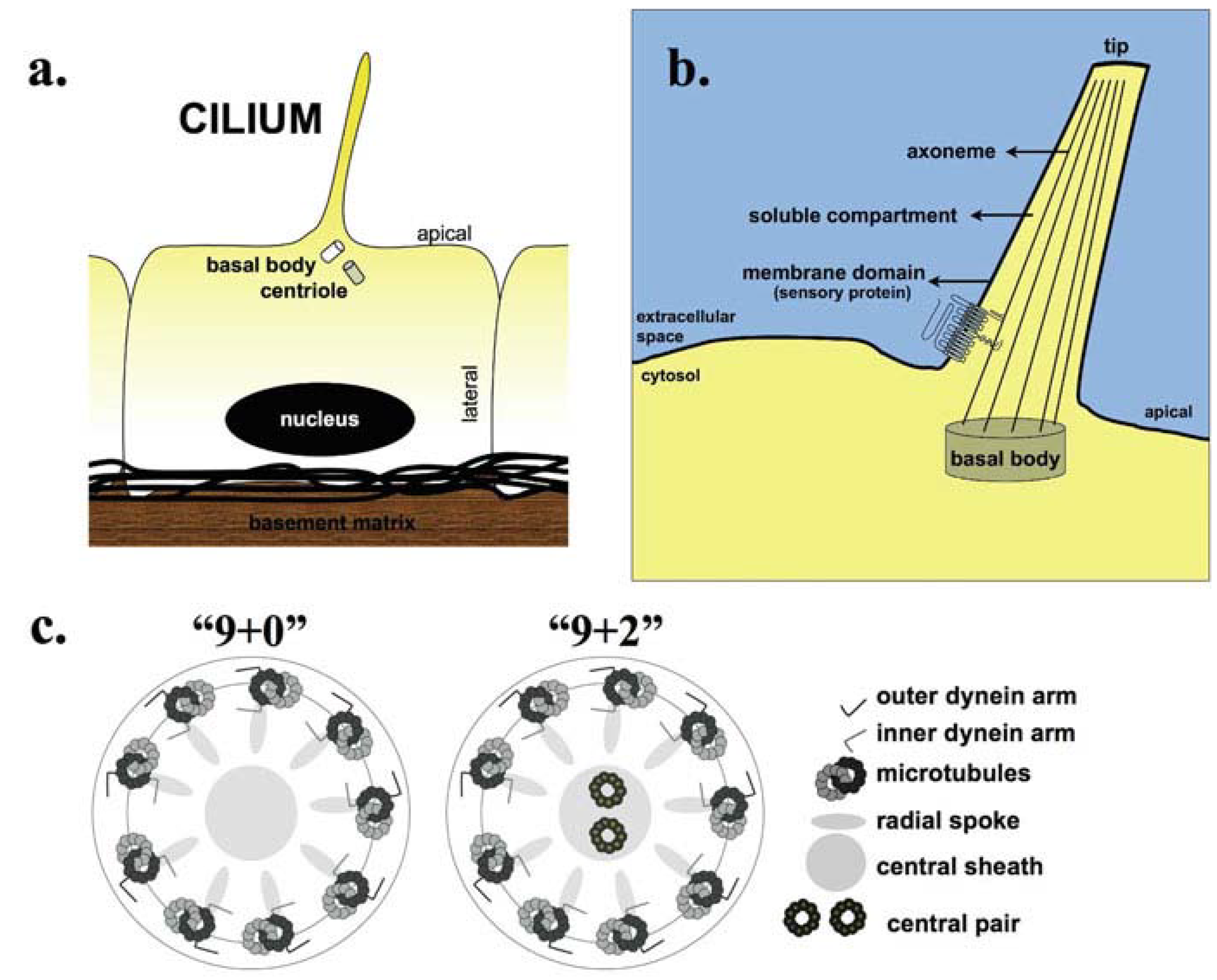
| Axoneme | Motility | Function | Disease Relevance | Reference |
|---|---|---|---|---|
| “9+0” | Motile | Generation of nodal flow | Situs inversus; Situs ambiguous; Situs isomerism | [12,13,14] |
| Non-motile | Sensation of nodal flow | Situs inversus; Situs ambiguous; Situs isomerism | [14,15] | |
| Mechanosensor | Polycystic Kidney, Liver, and Pancreas Diseases | [5,16,17] | ||
| Shear stress sensor | Hypertension; Atherosclerosis; Aneurysm formation | [6,7,18,19] | ||
| Osmolarity sensor | Respiratory diseases; Infertility | [20,21] | ||
| Gravitational sensor | Osteoporosis; Chondroporosis | [22,23,24] | ||
| Olfaction sensor | Anosmia; Hyposmia | [25,26] | ||
| Light sensor | Retinitis pigmentosa; Blindness | [27,28,29] | ||
| Chemosensor | Nephrocystin; Diabetes; Obesity | [30,31,32] | ||
| Neurotransmitter sensor | Impaired brain plasticity | [33] | ||
| Developmental regulator | Developmental defects; Cancer | [34,35,36] | ||
| Pressure sensor | Bone maintenance, development | [22,37,38] | ||
| “9+2” | Motile | Chemosensor | Chronic obstructive pulmonary disease (COPD) | [39] |
| Airway remodeling | Bronchiectasis; Hyperreactive airways | [40] | ||
| Fluid Clearance | Chronic obstructive pulmonary disease (COPD) | [41,42,43] | ||
| Non-motile | Oocyte Transport | Infertility | [44,45] | |
| Sperm Motility | Infertility | [46,47,48] | ||
| Fluid Transport | Hydrocephalus; Cell Migration | [49,50,51] |
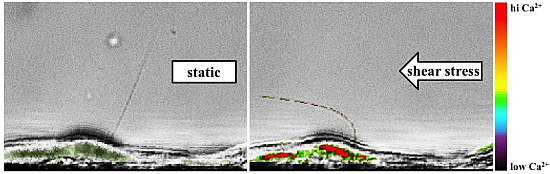
2. Cell Culture Assay
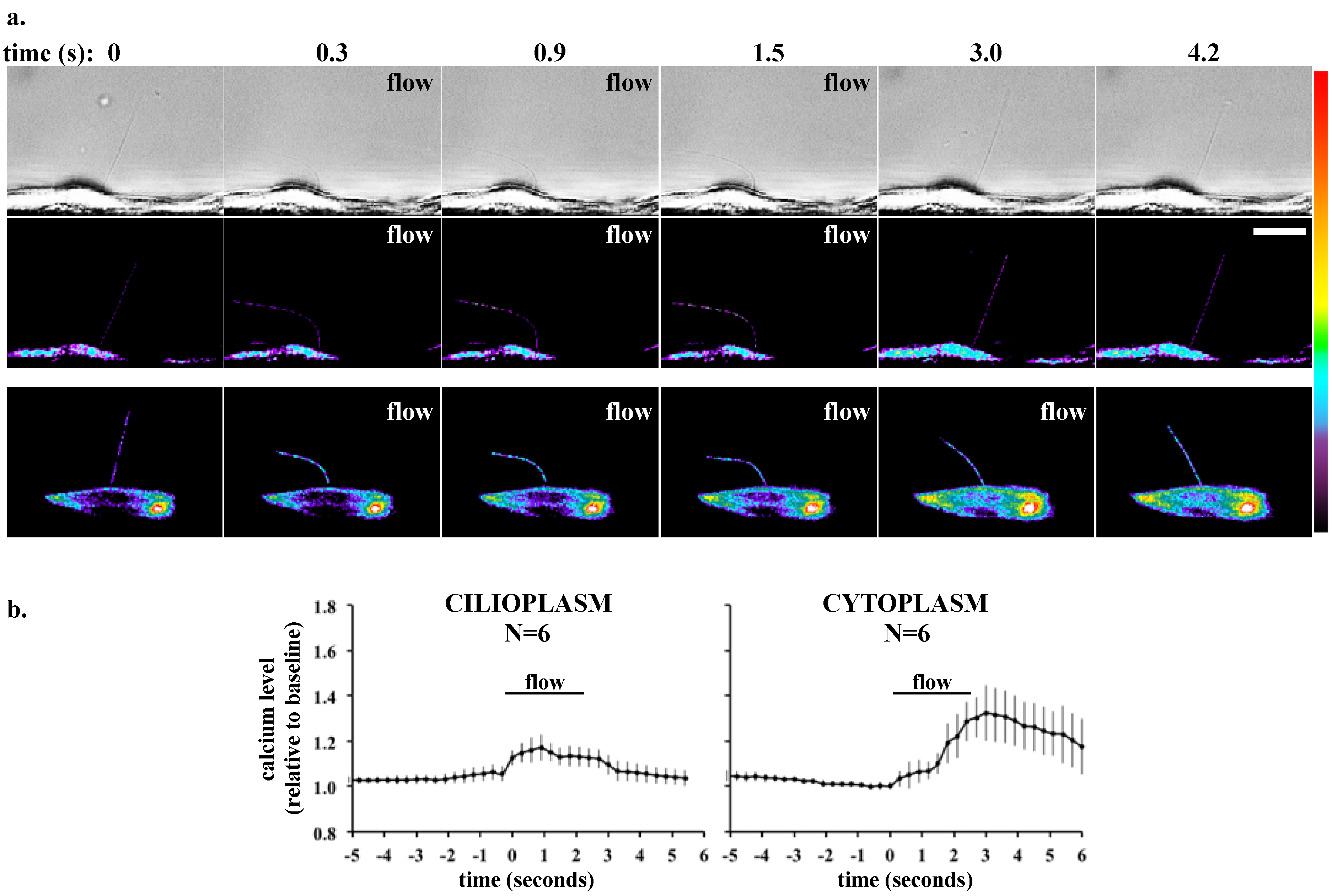
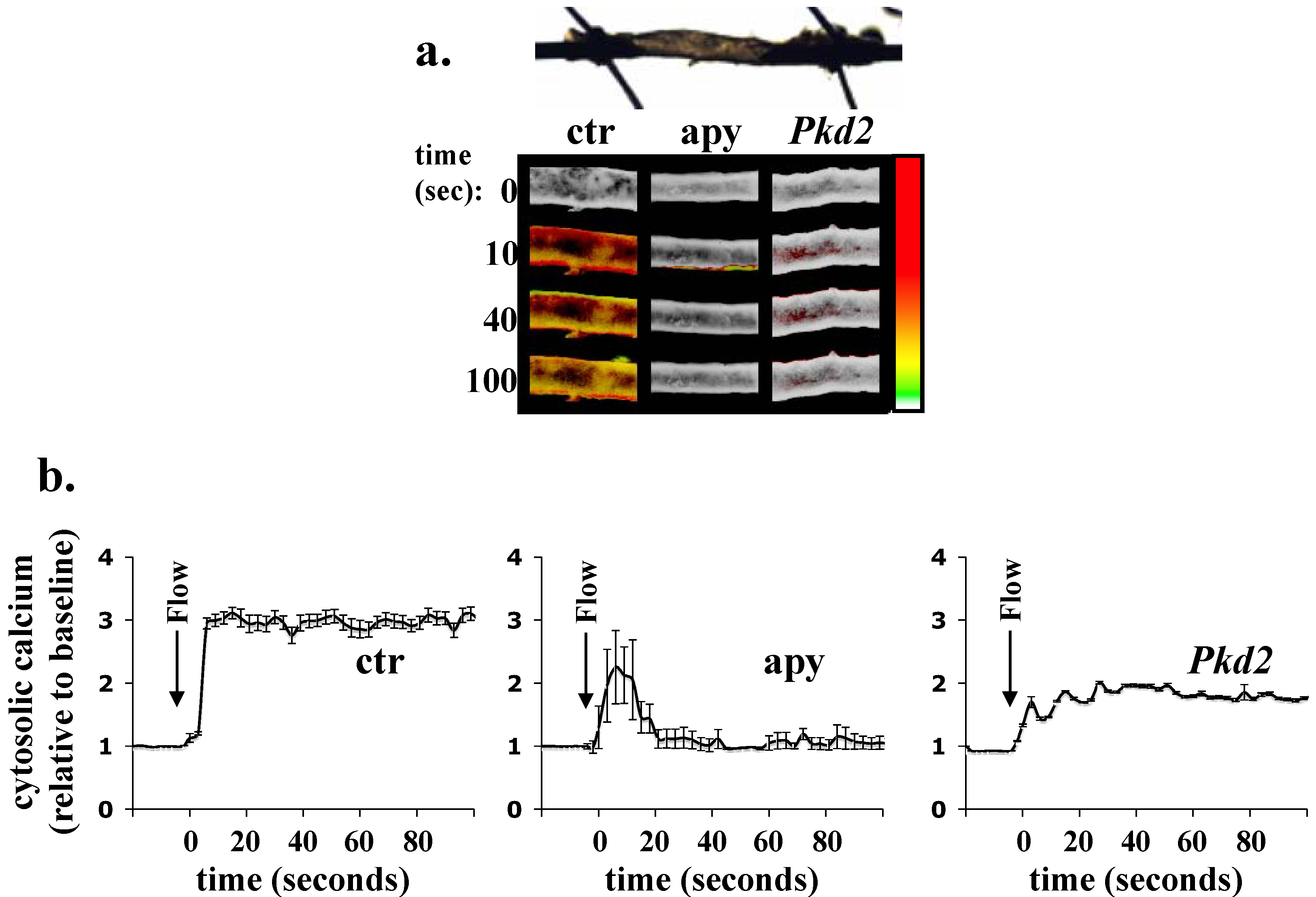
3. Single Cell Assays
| Fluorescent Indicator | Targeting Efficiency | Cytosolic Signal Dynamic Range (Maximum Percent Change in Fluorescence or FRET [SEM]) | Ciliary Signal Dynamic Range (Maximum Percent Change in Fluorescence or FRET [SEM]) |
|---|---|---|---|
| GFP | 0% | - | - |
| 5HT6-GFP | 90% | - | - |
| 5HT6-GCaMP5G | 0% | 65.1% [7.9%] | |
| 5HT6-YC3.60 | 80% | 58.2% [11.9%] | 74.6% [8.8%] |
| 5HT6-G-GECO1.0 | 75% | 135.1% [42.4%] | 360.0% [62.1%] |
| IA-GECO1.0 | 65% | 175.6% [42.3%] | 443.1 [39.5%] |
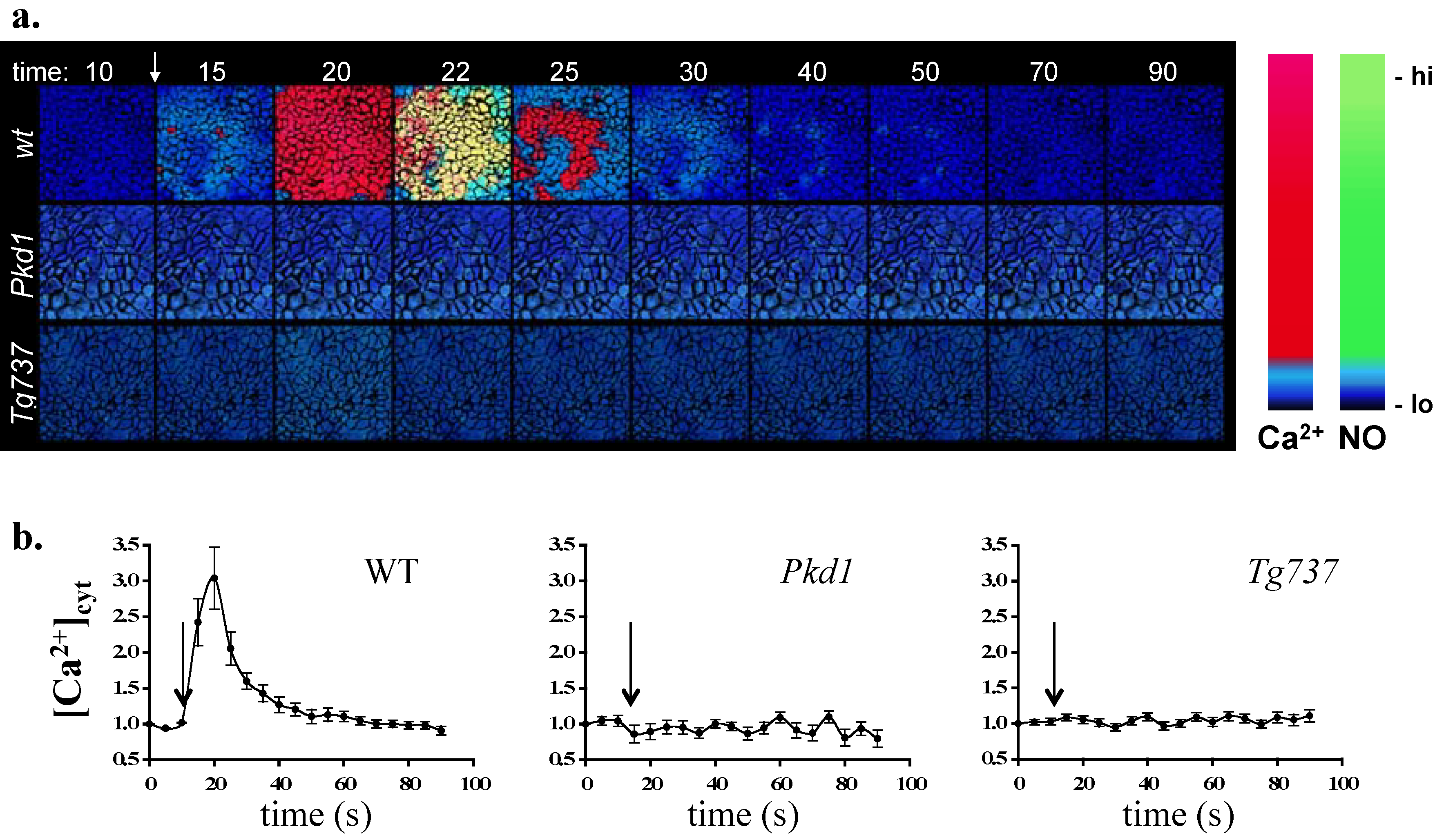
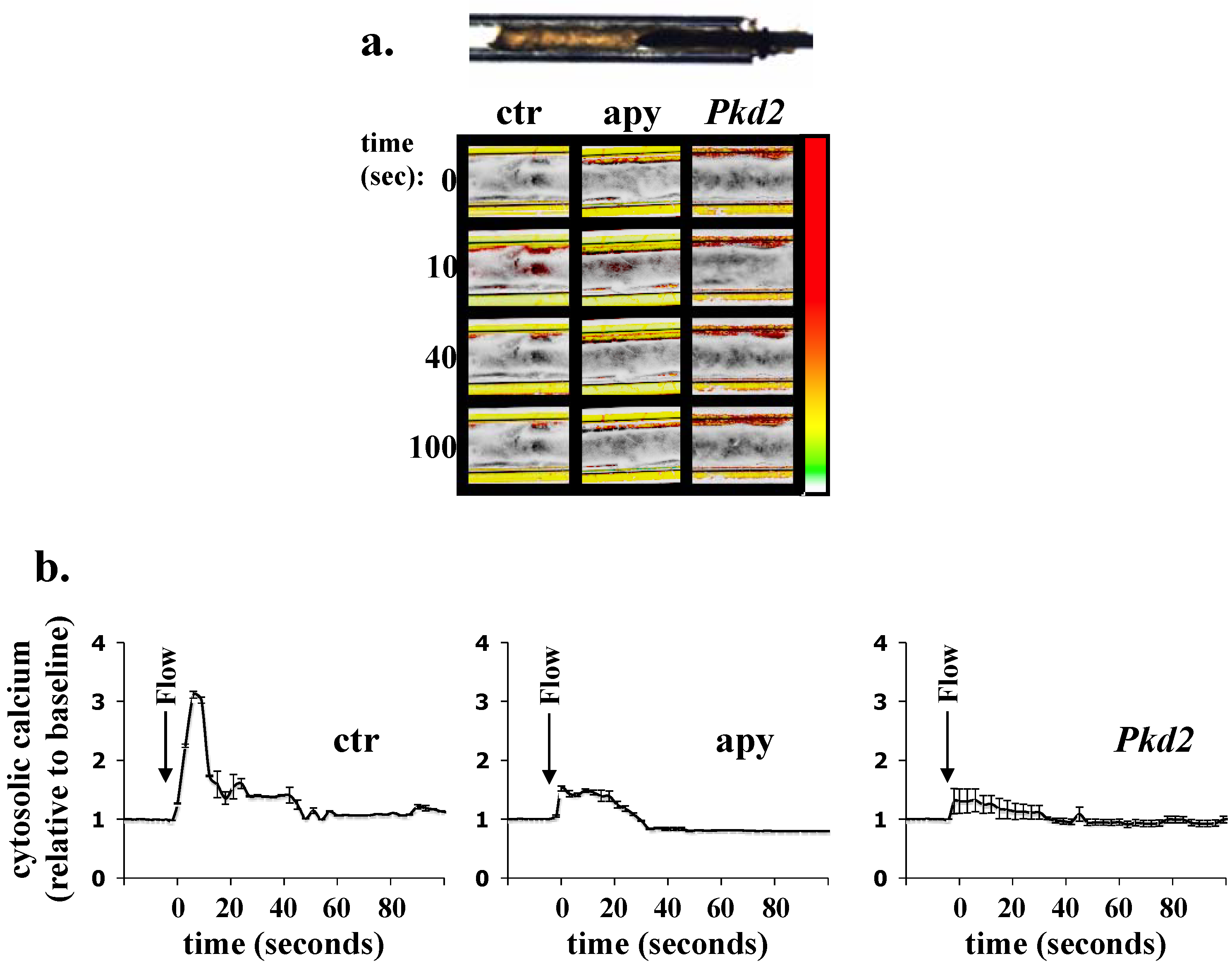
4. Ex Vivo Assays
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Nauli, S.M.; Haymour, H.S.; Aboualaiwi, W.A.; Lo, S.T.; Nauli, A.M. Primary cilia are mechanosensory organelles in vestibular tissues. In Mechanosensitivity and Mechanotransduction; Kamkin, A., Kiseleva, I., Eds.; Springer Science+Buisness Media: New York, NY, USA, 2011; pp. 317–350. [Google Scholar]
- Wheatley, D.N. Landmarks in the first hundred years of primary (9+0) cilium research. Cell Biol. Int. 2005, 29, 333–339. [Google Scholar]
- Muhammad, H.; Rais, Y.; Miosge, N.; Ornan, E.M. The primary cilium as a dual sensor of mechanochemical signals in chondrocytes. Cell. Mol. Life Sci. 2012, 69, 2101–2107. [Google Scholar] [CrossRef]
- McGrath, J.; Somlo, S.; Makova, S.; Tian, X.; Brueckner, M. Two populations of node monocilia initiate left-right asymmetry in the mouse. Cell 2003, 114, 61–73. [Google Scholar] [CrossRef]
- Masyuk, A.I.; Masyuk, T.V.; Splinter, P.L.; Huang, B.Q.; Stroope, A.J.; LaRusso, N.F. Cholangiocyte cilia detect changes in luminal fluid flow and transmit them into intracellular Ca2+ and camp signaling. Gastroenterology 2006, 131, 911–920. [Google Scholar] [CrossRef]
- AbouAlaiwi, W.A.; Takahashi, M.; Mell, B.R.; Jones, T.J.; Ratnam, S.; Kolb, R.J.; Nauli, S.M. Ciliary polycystin-2 is a mechanosensitive calcium channel involved in nitric oxide signaling cascades. Circ. Res. 2009, 104, 860–869. [Google Scholar] [CrossRef]
- Nauli, S.M.; Kawanabe, Y.; Kaminski, J.J.; Pearce, W.J.; Ingber, D.E.; Zhou, J. Endothelial cilia are fluid shear sensors that regulate calcium signaling and nitric oxide production through polycystin-1. Circulation 2008, 117, 1161–1171. [Google Scholar]
- Lim, K.T.; Kim, J.; Seonwoo, H.; Chang, J.U.; Choi, H.; Hexiu, J.; Cho, W.J.; Choung, P.H.; Chung, J.H. Enhanced osteogenesis of human alveolar bone-derived mesenchymal stem cells for tooth tissue engineering using fluid shear stress in a rocking culture method. Tissue Eng. C Meth. 2013, 19, 128–145. [Google Scholar]
- Nauli, S.M.; Rossetti, S.; Kolb, R.J.; Alenghat, F.J.; Consugar, M.B.; Harris, P.C.; Ingber, D.E.; Loghman-Adham, M.; Zhou, J. Loss of polycystin-1 in human cyst-lining epithelia leads to ciliary dysfunction. J. Am. Soc. Nephrol. 2006, 17, 1015–1025. [Google Scholar] [CrossRef]
- Riddle, R.C.; Taylor, A.F.; Genetos, D.C.; Donahue, H.J. Map kinase and calcium signaling mediate fluid flow-induced human mesenchymal stem cell proliferation. Am. J. Physiol. Cell Physiol. 2006, 290, C776–C784. [Google Scholar]
- Sankar, K.S.; Green, B.J.; Crocker, A.R.; Verity, J.E.; Altamentova, S.M.; Rocheleau, J.V. Culturing pancreatic islets in microfluidic flow enhances morphology of the associated endothelial cells. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Essner, J.J.; Vogan, K.J.; Wagner, M.K.; Tabin, C.J.; Yost, H.J.; Brueckner, M. Conserved function for embryonic nodal cilia. Nature 2002, 418, 37–38. [Google Scholar] [CrossRef]
- Nonaka, S.; Shiratori, H.; Saijoh, Y.; Hamada, H. Determination of left-right patterning of the mouse embryo by artificial nodal flow. Nature 2002, 418, 96–99. [Google Scholar]
- Nonaka, S.; Tanaka, Y.; Okada, Y.; Takeda, S.; Harada, A.; Kanai, Y.; Kido, M.; Hirokawa, N. Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking kif3b motor protein. Cell 1998, 95, 829–837. [Google Scholar] [CrossRef]
- Karcher, C.; Fischer, A.; Schweickert, A.; Bitzer, E.; Horie, S.; Witzgall, R.; Blum, M. Lack of a laterality phenotype in pkd1 knock-out embryos correlates with absence of polycystin-1 in nodal cilia. Differentiation 2005, 73, 425–432. [Google Scholar] [CrossRef]
- Nauli, S.M.; Alenghat, F.J.; Luo, Y.; Williams, E.; Vassilev, P.; Li, X.; Elia, A.E.; Lu, W.; Brown, E.M.; Quinn, S.J.; et al. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat. Genet. 2003, 33, 129–137. [Google Scholar]
- Cano, D.A.; Sekine, S.; Hebrok, M. Primary cilia deletion in pancreatic epithelial cells results in cyst formation and pancreatitis. Gastroenterology 2006, 131, 1856–1869. [Google Scholar]
- Van der Heiden, K.; Hierck, B.P.; Krams, R.; de Crom, R.; Cheng, C.; Baiker, M.; Pourquie, M.J.; Alkemade, F.E.; DeRuiter, M.C.; Gittenberger-de Groot, A.C.; et al. Endothelial primary cilia in areas of disturbed flow are at the base of atherosclerosis. Atherosclerosis 2008, 196, 542–550. [Google Scholar] [CrossRef]
- Aboualaiwi, W.A.; Muntean, B.S.; Ratnam, S.; Joe, B.; Liu, L.; Booth, R.L.; Rodriguez, I.; Herbert, B.S.; Bacallao, R.L.; Fruttiger, M.; et al. Survivin-induced abnormal ploidy contributes to cystic kidney and aneurysm formation. Circulation 2014, 129, 660–672. [Google Scholar] [CrossRef]
- Andrade, Y.N.; Fernandes, J.; Vazquez, E.; Fernandez-Fernandez, J.M.; Arniges, M.; Sanchez, T.M.; Villalon, M.; Valverde, M.A. Trpv4 channel is involved in the coupling of fluid viscosity changes to epithelial ciliary activity. J. Cell Biol. 2005, 168, 869–874. [Google Scholar] [CrossRef]
- Teilmann, S.C.; Christensen, S.T. Localization of the angiopoietin receptors tie-1 and tie-2 on the primary cilia in the female reproductive organs. Cell Biol. Int. 2005, 29, 340–346. [Google Scholar] [CrossRef]
- Malone, A.M.; Anderson, C.T.; Tummala, P.; Kwon, R.Y.; Johnston, T.R.; Stearns, T.; Jacobs, C.R. Primary cilia mediate mechanosensing in bone cells by a calcium-independent mechanism. Proc. Nat. Acad. Sci. USA 2007, 104, 13325–13330. [Google Scholar]
- McGlashan, S.R.; Haycraft, C.J.; Jensen, C.G.; Yoder, B.K.; Poole, C.A. Articular cartilage and growth plate defects are associated with chondrocyte cytoskeletal abnormalities in tg737orpk mice lacking the primary cilia protein polaris. Matrix Biol. 2007, 26, 234–246. [Google Scholar] [CrossRef]
- Moorman, S.J.; Shorr, A.Z. The primary cilium as a gravitational force transducer and a regulator of transcriptional noise. Dev. Dynam. 2008, 237, 1955–1959. [Google Scholar] [CrossRef]
- Kulaga, H.M.; Leitch, C.C.; Eichers, E.R.; Badano, J.L.; Lesemann, A.; Hoskins, B.E.; Lupski, J.R.; Beales, P.L.; Reed, R.R.; Katsanis, N. Loss of bbs proteins causes anosmia in humans and defects in olfactory cilia structure and function in the mouse. Nat. Genet. 2004, 36, 994–998. [Google Scholar]
- Layman, W.S.; McEwen, D.P.; Beyer, L.A.; Lalani, S.R.; Fernbach, S.D.; Oh, E.; Swaroop, A.; Hegg, C.C.; Raphael, Y.; Martens, J.R.; et al. Defects in neural stem cell proliferation and olfaction in chd7 deficient mice indicate a mechanism for hyposmia in human charge syndrome. Hum. Mol. Genet. 2009, 18, 1909–1923. [Google Scholar]
- Ghosh, A.K.; Murga-Zamalloa, C.A.; Chan, L.; Hitchcock, P.F.; Swaroop, A.; Khanna, H. Human retinopathy-associated ciliary protein retinitis pigmentosa gtpase regulator mediates cilia-dependent vertebrate development. Hum. Mol. Genet. 2010, 19, 90–98. [Google Scholar]
- Moore, A.; Escudier, E.; Roger, G.; Tamalet, A.; Pelosse, B.; Marlin, S.; Clement, A.; Geremek, M.; Delaisi, B.; Bridoux, A.M.; et al. Rpgr is mutated in patients with a complex x linked phenotype combining primary ciliary dyskinesia and retinitis pigmentosa. J. Med. Genet. 2006, 43, 326–333. [Google Scholar]
- Nishimura, D.Y.; Fath, M.; Mullins, R.F.; Searby, C.; Andrews, M.; Davis, R.; Andorf, J.L.; Mykytyn, K.; Swiderski, R.E.; Yang, B.; et al. Bbs2-null mice have neurosensory deficits, a defect in social dominance, and retinopathy associated with mislocalization of rhodopsin. Proc. Nat. Acad. Sci. USA 2004, 101, 16588–16593. [Google Scholar] [CrossRef]
- Davenport, J.R.; Watts, A.J.; Roper, V.C.; Croyle, M.J.; van Groen, T.; Wyss, J.M.; Nagy, T.R.; Kesterson, R.A.; Yoder, B.K. Disruption of intraflagellar transport in adult mice leads to obesity and slow-onset cystic kidney disease. Curr. Biol. 2007, 17, 1586–1594. [Google Scholar]
- Hearn, T.; Spalluto, C.; Phillips, V.J.; Renforth, G.L.; Copin, N.; Hanley, N.A.; Wilson, D.I. Subcellular localization of alms1 supports involvement of centrosome and basal body dysfunction in the pathogenesis of obesity, insulin resistance, and type 2 diabetes. Diabetes 2005, 54, 1581–1587. [Google Scholar] [CrossRef]
- Winkelbauer, M.E.; Schafer, J.C.; Haycraft, C.J.; Swoboda, P.; Yoder, B.K. The C. elegans homologs of nephrocystin-1 and nephrocystin-4 are cilia transition zone proteins involved in chemosensory perception. J. Cell Sci. 2005, 118, 5575–5587. [Google Scholar] [CrossRef]
- Einstein, E.B.; Patterson, C.A.; Hon, B.J.; Regan, K.A.; Reddi, J.; Melnikoff, D.E.; Mateer, M.J.; Schulz, S.; Johnson, B.N.; Tallent, M.K. Somatostatin signaling in neuronal cilia is critical for object recognition memory. J. Neurosci. 2010, 30, 4306–4314. [Google Scholar]
- Christensen, S.T.; Pedersen, S.F.; Satir, P.; Veland, I.R.; Schneider, L. The primary cilium coordinates signaling pathways in cell cycle control and migration during development and tissue repair. Curr. Top. Dev. Biol. 2008, 85, 261–301. [Google Scholar] [CrossRef]
- Han, Y.G.; Kim, H.J.; Dlugosz, A.A.; Ellison, D.W.; Gilbertson, R.J.; Alvarez-Buylla, A. Dual and opposing roles of primary cilia in medulloblastoma development. Nat. Med. 2009, 15, 1062–1065. [Google Scholar]
- Wong, S.Y.; Seol, A.D.; So, P.L.; Ermilov, A.N.; Bichakjian, C.K.; Epstein, E.H., Jr.; Dlugosz, A.A.; Reiter, J.F. Primary cilia can both mediate and suppress hedgehog pathway-dependent tumorigenesis. Nat. Med. 2009, 15, 1055–1061. [Google Scholar]
- Xiao, Z.; Zhang, S.; Mahlios, J.; Zhou, G.; Magenheimer, B.S.; Guo, D.; Dallas, S.L.; Maser, R.; Calvet, J.P.; Bonewald, L.; et al. Cilia-like structures and polycystin-1 in osteoblasts/osteocytes and associated abnormalities in skeletogenesis and runx2 expression. J. Biol. Chem. 2006, 281, 30884–30895. [Google Scholar] [CrossRef]
- Hoey, D.A.; Tormey, S.; Ramcharan, S.; O’Brien, F.J.; Jacobs, C.R. Primary cilia-mediated mechanotransduction in human mesenchymal stem cells. Stem Cells 2012, 30, 2561–2570. [Google Scholar] [CrossRef]
- Shah, A.S.; Ben-Shahar, Y.; Moninger, T.O.; Kline, J.N.; Welsh, M.J. Motile cilia of human airway epithelia are chemosensory. Science 2009, 325, 1131–1134. [Google Scholar]
- Gilley, S.K.; Stenbit, A.E.; Pasek, R.C.; Sas, K.M.; Steele, S.L.; Amria, M.; Bunni, M.A.; Estell, K.P.; Schwiebert, L.M.; Flume, P.; et al. Deletion of airway cilia results in non-inflammatory bronchiectasis and hyperreactive airways. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013. [Google Scholar] [CrossRef]
- Mall, M.A. Role of cilia, mucus, and airway surface liquid in mucociliary dysfunction: Lessons from mouse models. J. Aerosol. Med. Pulm. Drug Deliv. 2008, 21, 13–24. [Google Scholar] [CrossRef]
- Salathe, M. Regulation of mammalian ciliary beating. Annu. Rev. Physiol. 2007, 69, 401–422. [Google Scholar] [CrossRef]
- Zariwala, M.A.; Knowles, M.R.; Omran, H. Genetic defects in ciliary structure and function. Annu. Rev. Physiol. 2007, 69, 423–450. [Google Scholar] [CrossRef]
- Eddy, C.A.; Pauerstein, C.J. Anatomy and physiology of the fallopian tube. Clin. Obstet. Gynecol. 1980, 23, 1177–1193. [Google Scholar] [CrossRef]
- Lyons, R.A.; Saridogan, E.; Djahanbakhch, O. The reproductive significance of human fallopian tube cilia. Hum. Reprod. Update 2006, 12, 363–372. [Google Scholar] [CrossRef]
- Brunner, S.; Colman, D.; Travis, A.J.; Luhmann, U.F.; Shi, W.; Feil, S.; Imsand, C.; Nelson, J.; Grimm, C.; Rulicke, T.; et al. Overexpression of RPGR leads to male infertility in mice due to defects in flagellar assembly. Biol. Reprod. 2008, 79, 608–617. [Google Scholar] [CrossRef]
- Imai, H.; Hakkaku, N.; Iwamoto, R.; Suzuki, J.; Suzuki, T.; Tajima, Y.; Konishi, K.; Minami, S.; Ichinose, S.; Ishizaka, K.; et al. Depletion of selenoprotein gpx4 in spermatocytes causes male infertility in mice. J. Biol. Chem. 2009, 284, 32522–32532. [Google Scholar] [CrossRef]
- Lee, L.; Campagna, D.R.; Pinkus, J.L.; Mulhern, H.; Wyatt, T.A.; Sisson, J.H.; Pavlik, J.A.; Pinkus, G.S.; Fleming, M.D. Primary ciliary dyskinesia in mice lacking the novel ciliary protein pcdp1. Mole. Cell. Biol. 2008, 28, 949–957. [Google Scholar] [CrossRef]
- Ibanez-Tallon, I.; Pagenstecher, A.; Fliegauf, M.; Olbrich, H.; Kispert, A.; Ketelsen, U.P.; North, A.; Heintz, N.; Omran, H. Dysfunction of axonemal dynein heavy chain mdnah5 inhibits ependymal flow and reveals a novel mechanism for hydrocephalus formation. Hum. Mol. Genet. 2004, 13, 2133–2141. [Google Scholar] [CrossRef]
- Sawamoto, K.; Wichterle, H.; Gonzalez-Perez, O.; Cholfin, J.A.; Yamada, M.; Spassky, N.; Murcia, N.S.; Garcia-Verdugo, J.M.; Marin, O.; Rubenstein, J.L.; et al. New neurons follow the flow of cerebrospinal fluid in the adult brain. Science 2006, 311, 629–632. [Google Scholar] [CrossRef]
- Wodarczyk, C.; Rowe, I.; Chiaravalli, M.; Pema, M.; Qian, F.; Boletta, A. A novel mouse model reveals that polycystin-1 deficiency in ependyma and choroid plexus results in dysfunctional cilia and hydrocephalus. PLoS One 2009, 4. [Google Scholar] [CrossRef]
- Nauli, S.M.; Jin, X.; AbouAlaiwi, W.A.; El-Jouni, W.; Su, X.; Zhou, J. Non-motile primary cilia as fluid shear stress mechanosensors. Meth. Enzymol. 2013, 525, 1–20. [Google Scholar] [CrossRef]
- Delling, M.; DeCaen, P.G.; Doerner, J.F.; Febvay, S.; Clapham, D.E. Primary cilia are specialized calcium signalling organelles. Nature 2013, 504, 311–314. [Google Scholar] [CrossRef]
- Abdul-Majeed, S.; Nauli, S.M. Dopamine receptor type 5 in the primary cilia has dual chemo- and mechano-sensory roles. Hypertension 2011, 58, 325–331. [Google Scholar] [CrossRef]
- Liu, W.; Xu, S.; Woda, C.; Kim, P.; Weinbaum, S.; Satlin, L.M. Effect of flow and stretch on the [Ca2+]i response of principal and intercalated cells in cortical collecting duct. Am. J. Physiol. Ren. Physiol. 2003, 285, F998–F1012. [Google Scholar]
- Kotsis, F.; Nitschke, R.; Boehlke, C.; Bashkurov, M.; Walz, G.; Kuehn, E.W. Ciliary calcium signaling is modulated by kidney injury molecule-1 (Kim1). Pflugers Arch. 2007, 453, 819–829. [Google Scholar] [CrossRef]
- Siroky, B.J.; Ferguson, W.B.; Fuson, A.L.; Xie, Y.; Fintha, A.; Komlosi, P.; Yoder, B.K.; Schwiebert, E.M.; Guay-Woodford, L.M.; Bell, P.D. Loss of primary cilia results in deregulated and unabated apical calcium entry in ARPKD collecting duct cells. Am. J. Physiol. Ren. Physiol. 2006, 290, F1320–F1328. [Google Scholar] [CrossRef]
- Su, S.; Phua, S.C.; DeRose, R.; Chiba, S.; Narita, K.; Kalugin, P.N.; Katada, T.; Kontani, K.; Takeda, S.; Inoue, T. Genetically encoded calcium indicator illuminates calcium dynamics in primary cilia. Nat. Meth. 2013, 10, 1105–1107. [Google Scholar] [CrossRef]
- DeCaen, P.G.; Delling, M.; Vien, T.N.; Clapham, D.E. Direct recording and molecular identification of the calcium channel of primary cilia. Nature 2013, 504, 315–318. [Google Scholar] [CrossRef]
- Jin, X.; Mohieldin, A.M.; Muntean, B.S.; Green, J.A.; Shah, J.V.; Mykytyn, K.; Nauli, S.M. Cilioplasm is a cellular compartment for calcium signaling in response to mechanical and chemical stimuli. Cell. Mol. Life Sci. 2013. [Google Scholar] [CrossRef]
- Praetorius, H.A.; Spring, K.R. Bending the MDCK cell primary cilium increases intracellular calcium. J. Membr. Biol. 2001, 184, 71–79. [Google Scholar] [CrossRef]
- Ishihara, Y.; Sugawara, Y.; Kamioka, H.; Kawanabe, N.; Kurosaka, H.; Naruse, K.; Yamashiro, T. In situ imaging of the autonomous intracellular Ca(2+) oscillations of osteoblasts and osteocytes in bone. Bone 2012, 50, 842–852. [Google Scholar] [CrossRef]
- Ishihara, Y.; Sugawara, Y.; Kamioka, H.; Kawanabe, N.; Hayano, S.; Balam, T.A.; Naruse, K.; Yamashiro, T. Ex vivo real-time observation of Ca(2+) signaling in living bone in response to shear stress applied on the bone surface. Bone 2013, 53, 204–215. [Google Scholar] [CrossRef]
- Lee, D.J.; Hsu, Y.H. Fluid flow in capillary suction apparatus. Ind. Eng. Chem. Res. 1992, 31, 2379–2385. [Google Scholar] [CrossRef]
- Kamioka, H.; Honjo, T.; Takano-Yamamoto, T. A three-dimensional distribution of osteocyte processes revealed by the combination of confocal laser scanning microscopy and differential interference contrast microscopy. Bone 2001, 28, 145–149. [Google Scholar] [CrossRef]
- Woda, C.B.; Leite, M., Jr.; Rohatgi, R.; Satlin, L.M. Effects of luminal flow and nucleotides on [Ca2+]i in rabbit cortical collecting duct. Am. J. Physiol. Ren. Physiol. 2002, 283, F437–F446. [Google Scholar]
- Olteanu, D.; Liu, X.; Liu, W.; Roper, V.C.; Sharma, N.; Yoder, B.K.; Satlin, L.M.; Schwiebert, E.M.; Bevensee, M.O. Increased Na+/H+ exchanger activity on the apical surface of a cilium-deficient cortical collecting duct principal cell model of polycystic kidney disease. Am. J. Physiol. Cell Physiol. 2012, 302, C1436–C1451. [Google Scholar] [CrossRef]
- Sipos, A.; Vargas, S.; Peti-Peterdi, J. Direct demonstration of tubular fluid flow sensing by macula densa cells. Am. J. Physiol. Ren. Physiol. 2010, 299, F1087–F1093. [Google Scholar] [CrossRef]
- Tanaka, Y.; Okada, Y.; Hirokawa, N. FGF-induced vesicular release of Sonic hedgehog and retinoic acid in leftward nodal flow is critical for left-right determination. Nature 2005, 435, 172–177. [Google Scholar] [CrossRef]
- Song, H.; Hu, J.; Chen, W.; Elliott, G.; Andre, P.; Gao, B.; Yang, Y. Planar cell polarity breaks bilateral symmetry by controlling ciliary positioning. Nature 2010, 466, 378–382. [Google Scholar] [CrossRef]
- Rodat-Despoix, L.; Hao, J.; Dandonneau, M.; Delmas, P. Shear stress-induced Ca²+mobilization in MDCK cells is ATP dependent, no matter the primary cilium. Cell Calcium. 2013, 53, 327–337. [Google Scholar] [CrossRef]
- Boehlke, C.; Kotsis, F.; Patel, V.; Braeg, S.; Voelker, H.; Bredt, S.; Beyer, T.; Janusch, H.; Hamann, C.; Gödel, M.; et al. Primary cilia regulate mTORC1 activity and cell size through Lkb1. Nat. Cell Biol. 2010, 12, 1115–1122. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Prasad, R.M.; Jin, X.; Nauli, S.M. Sensing a Sensor: Identifying the Mechanosensory Function of Primary Cilia. Biosensors 2014, 4, 47-62. https://doi.org/10.3390/bios4010047
Prasad RM, Jin X, Nauli SM. Sensing a Sensor: Identifying the Mechanosensory Function of Primary Cilia. Biosensors. 2014; 4(1):47-62. https://doi.org/10.3390/bios4010047
Chicago/Turabian StylePrasad, Rahul M., Xingjian Jin, and Surya M. Nauli. 2014. "Sensing a Sensor: Identifying the Mechanosensory Function of Primary Cilia" Biosensors 4, no. 1: 47-62. https://doi.org/10.3390/bios4010047
APA StylePrasad, R. M., Jin, X., & Nauli, S. M. (2014). Sensing a Sensor: Identifying the Mechanosensory Function of Primary Cilia. Biosensors, 4(1), 47-62. https://doi.org/10.3390/bios4010047