A Novel Urine Test Biosensor Platform for Early Lung Cancer Detection
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Characteristics
2.2. Animal Habitat, Nutrition, and Welfare
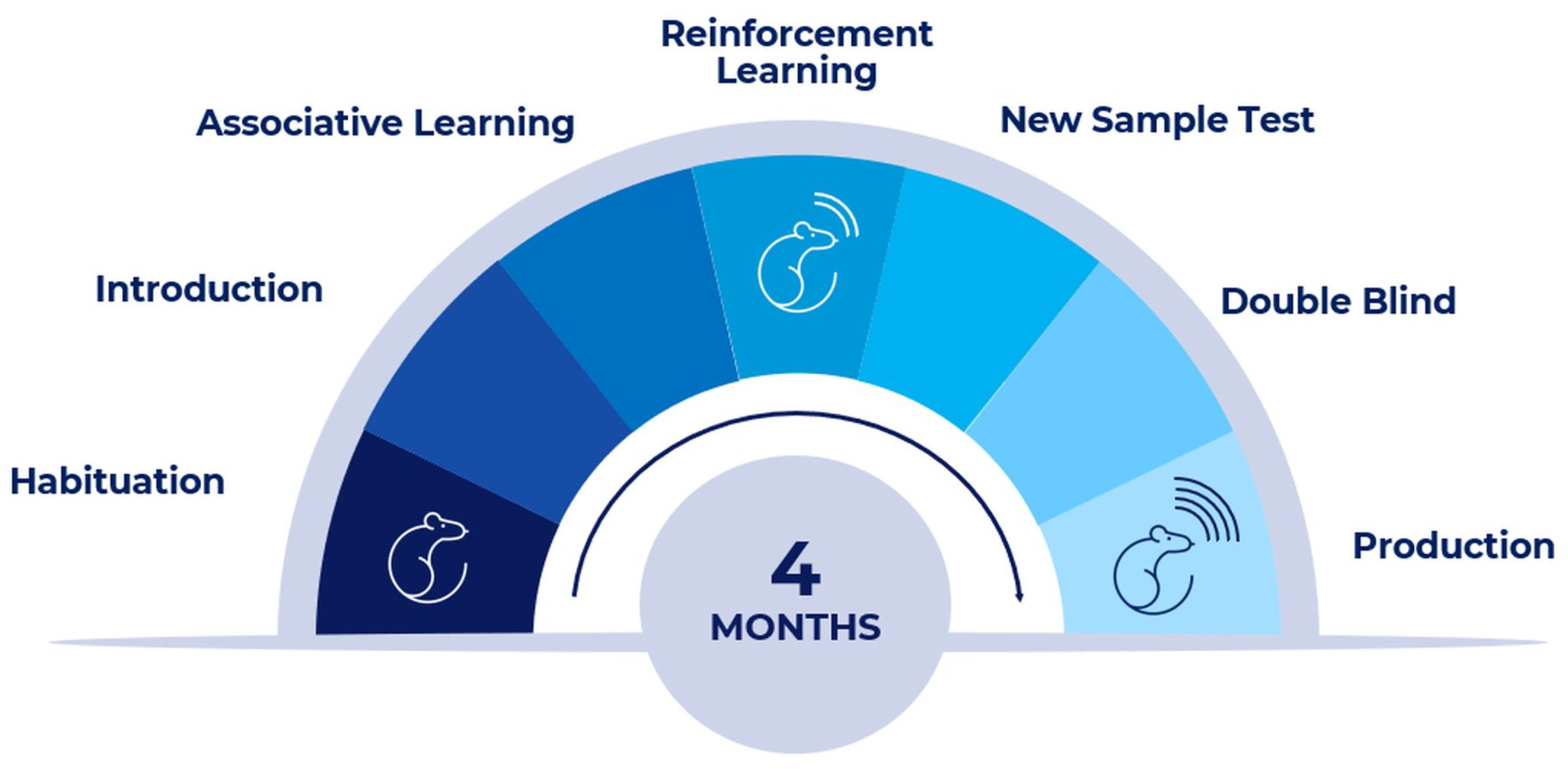
2.3. The BS’s Training, Validation, and Qualification Process
2.4. The BSP as a Binary Test for Lung Cancer Detection
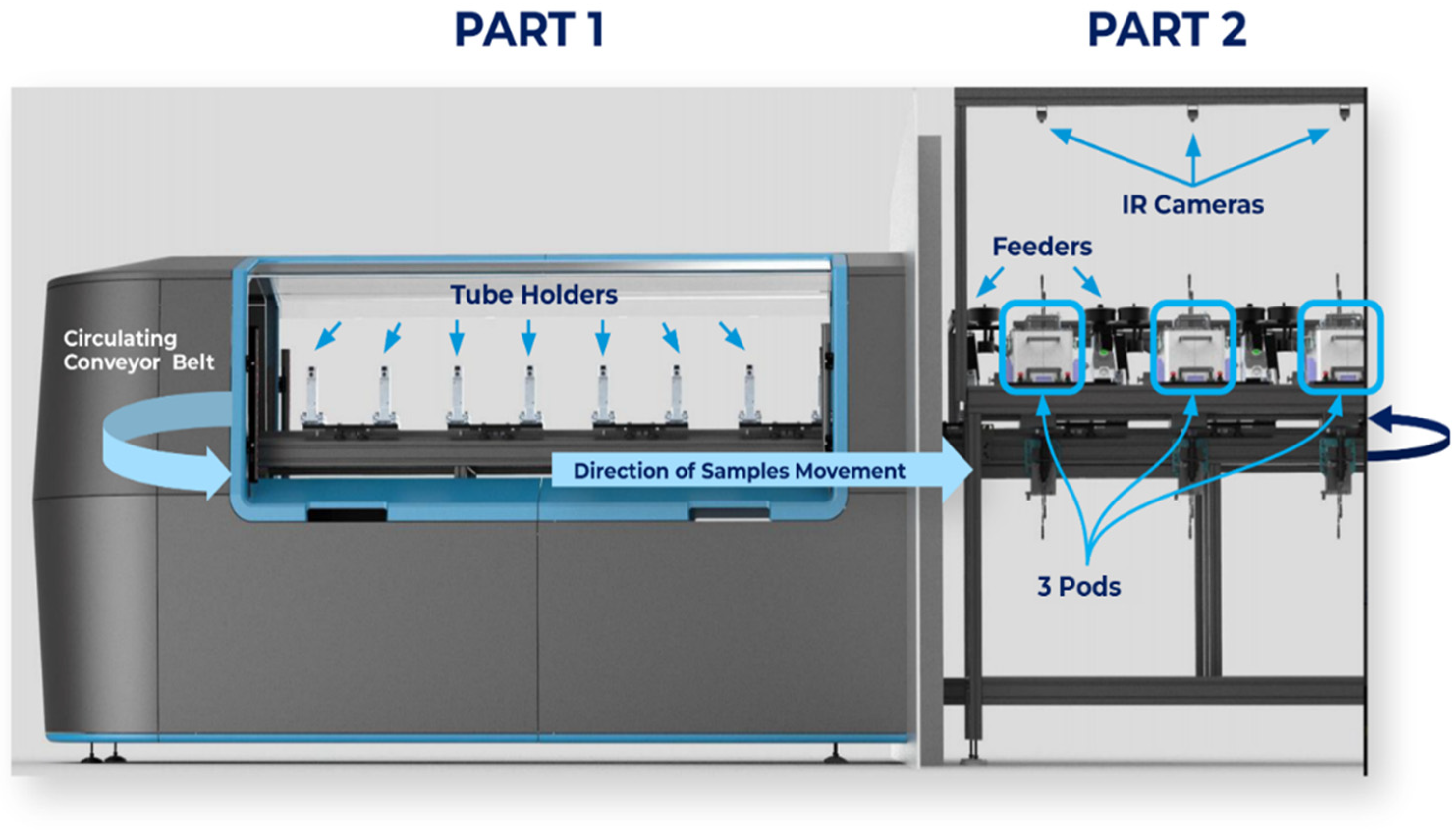
2.4.1. The System General Overview
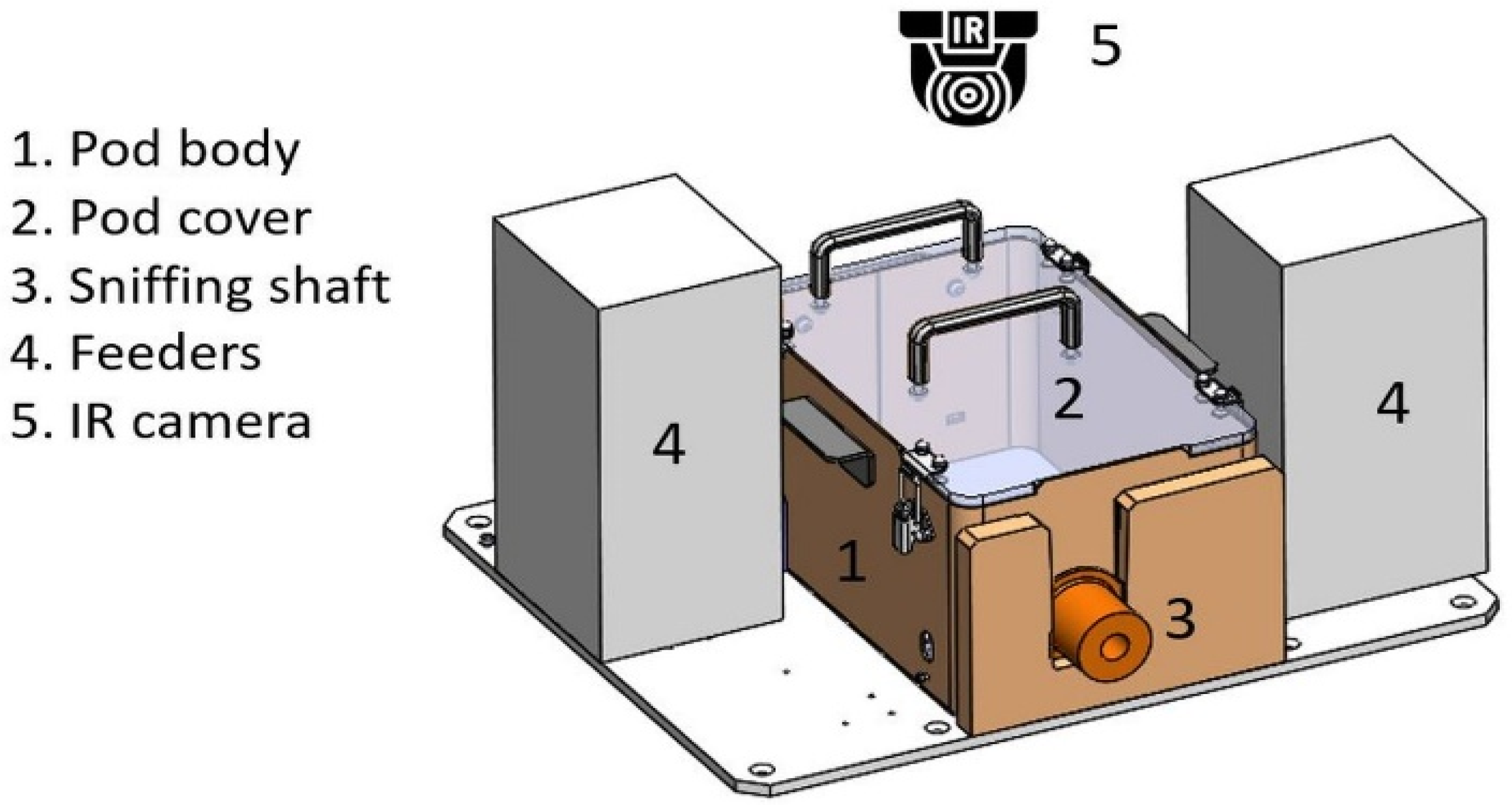
2.4.2. Pods or “Sniffing Positions”
2.5. Sample Preparation and Processing
2.6. Training Learning Curve
2.7. System Software Final Sample Result Calculation
3. Results
3.1. A Double-Blind Study for the Assessment of BSP Final Performance
Study Demographic and Patient Characteristics
3.2. Validation Study Results
3.3. Urine Sample Assessment Repeatability Testing after Sample Thawing and Refreezing
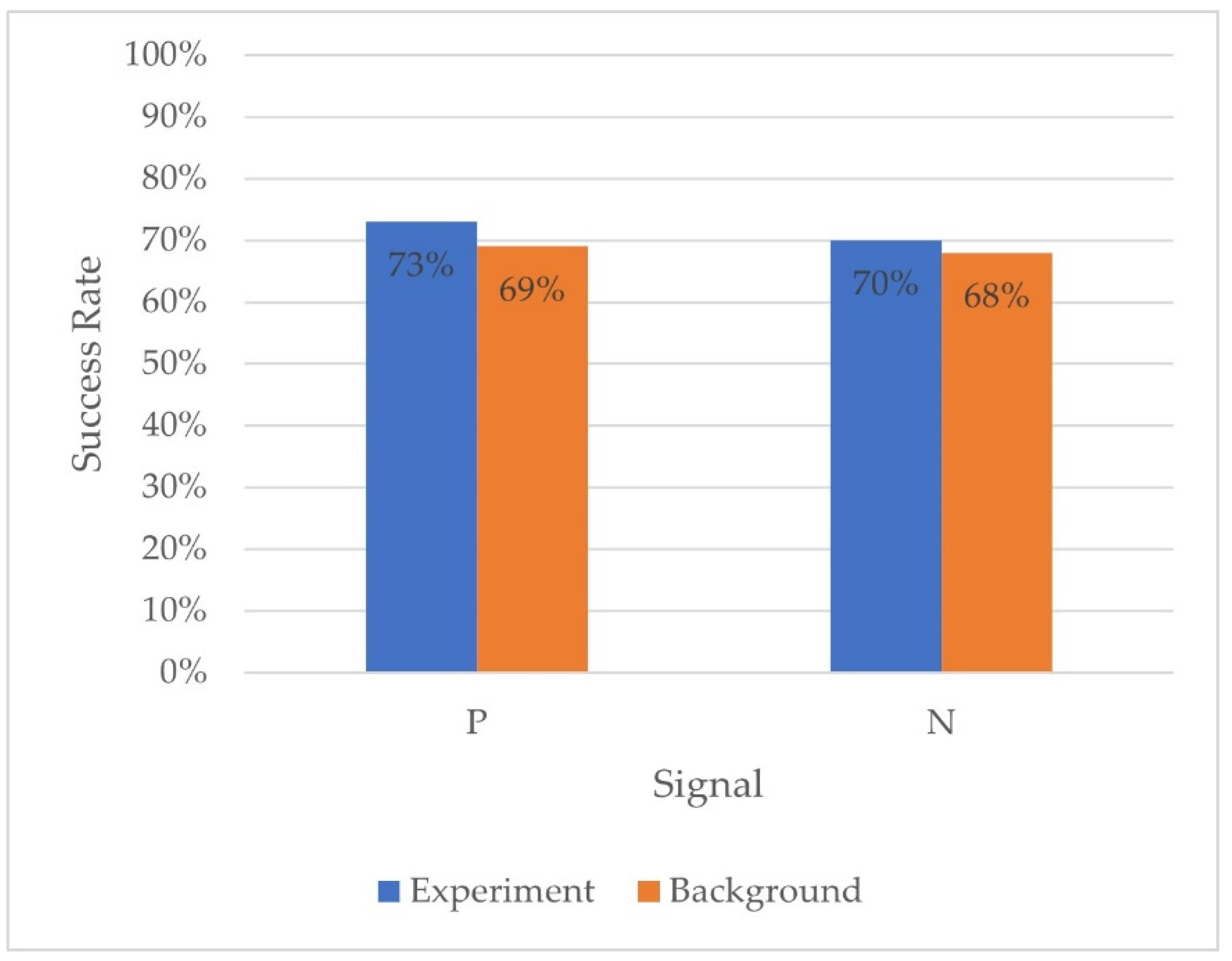
3.4. The Utility of the BSP for Pooled Sample Diagnosis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends—An Update. Cancer Epidemiol. Biomark. Prev. 2016, 25, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Office of the Surgeon General. Report of the Surgeon General: Health Consequences of Smoking; United States Government Printing Office: Washington, DC, USA, 2004. [Google Scholar]
- Thun, M.J.; Henley, S.J.; Burns, D.; Jemal, A.; Shanks, T.G.; Calle, E.E. Lung Cancer Death Rates in Lifelong Nonsmokers. JNCI J. Natl. Cancer Inst. 2006, 98, 691–699. [Google Scholar] [CrossRef]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- American Cancer Society. About Lung Cancer. 2023. Available online: http://cancer.org/1.800.227.2345 (accessed on 4 February 2023).
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef]
- Wood, D.E.; Kazerooni, E.; Baum, S.L.; Dransfield, M.T.; Eapen, G.A.; Ettinger, D.S.; Hou, L.; Jackman, D.M.; Klippenstein, D.; Kumar, R.; et al. Lung Cancer Screening, Version 1.2015. J. Natl. Compr. Cancer Netw. 2015, 13, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Heymach, J.V.; Lippman, S.M. Molecular Origins of Cancer, Lung Cancer. N. Engl. J. Med. 2008, 359, 1367–1380. [Google Scholar] [CrossRef]
- Nesbitt, J.C.; Putnam, J.B.; Walsh, G.L.; Roth, J.A.; Mountain, C.F. Survival in Early-Stage Non-Small Cell Lung Cancer. Ann. Thorac. Surg. 1995, 60, 466–472. [Google Scholar] [CrossRef]
- Goldstraw, P.; Chansky, K.; Crowley, J.; Rami-Porta, R.; Asamura, H.; Eberhardt, W.E.; Nicholson, A.G.; Groome, P.; Mitchell, A.; Bolejack, V.; et al. The IASLC Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Lung Cancer. J. Thorac. Oncol. 2016, 11, 39–51. [Google Scholar] [CrossRef]
- Detterbeck, F.C.; Mazzone, P.J.; Naidich, D.P.; Bach, P.B. Screening for Lung Cancer. Chest 2013, 143, e78S–e92S. [Google Scholar] [CrossRef]
- Klein, E.; Richards, D.; Cohn, A.; Tummala, M.; Lapham, R.; Cosgrove, D.; Chung, G.; Clement, J.; Gao, J.; Hunkapiller, N.; et al. Clinical validation of a targeted methylation-based multi-cancer early detection test using an independent validation set. Ann. Oncol. 2021, 32, 1167–1177. [Google Scholar] [CrossRef]
- World Health Organization. Promoting Cancer Early Diagnosis. World Health Organization. Available online: https://www.who.int/activities/promoting-cancer-early-diagnosis (accessed on 2 April 2023).
- De Koning, H.J.; van der Aalst, C.M.; de Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.W.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N. Engl. J. Med. 2020, 382, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Forum of International Respiratory Societies. 2021. Available online: https://www.thoracic.org/about/global-public-health/firs/resources/world-lung-cancer-day-fact-sheet-2021.pdf (accessed on 4 February 2023).
- Nooreldeen, R.; Bach, H. Current and Future Development in Lung Cancer Diagnosis. Int. J. Mol. Sci. 2021, 22, 8661. [Google Scholar] [CrossRef] [PubMed]
- Mulshine, J.L.; Scott, F. Molecular Markers in Early Cancer Detection. Chest 1995, 107, 280S–286S. [Google Scholar] [CrossRef]
- Smith, R.A.; Cokkinides, V.; Eyre, H.J. American Cancer Society Guidelines for the Early Detection of Cancer. CA Cancer J. Clin. 2003, 53, 27–43. [Google Scholar] [CrossRef] [PubMed]
- Marrugo-Ramírez, J.; Mir, M.; Samitier, J. Blood-Based Cancer Biomarkers in Liquid Biopsy: A Promising Non-Invasive Alternative to Tissue Biopsy. Int. J. Mol. Sci. 2018, 19, 2877. [Google Scholar] [CrossRef]
- Schmidt, K.; Podmore, I. Current Challenges in Volatile Organic Compounds Analysis as Potential Biomarkers of Cancer. J. Biomark. 2015, 2015, 981458. [Google Scholar] [CrossRef] [PubMed]
- Jalal, A.H.; Alam, F.; Roychoudhury, S.; Umasankar, Y.; Pala, N.; Bhansali, S. Prospects and Challenges of Volatile Organic Compound Sensors in Human Healthcare. ACS Sens. 2018, 3, 1246–1263. [Google Scholar] [CrossRef]
- Thriumani, R.; Zakaria, A.; Hashim, Y.Z.H.-Y.; Jeffree, A.I.; Helmy, K.M.; Kamarudin, L.M.; Omar, M.I.; Shakaff, A.Y.M.; Adom, A.H.; Persaud, K.C. A study on volatile organic compounds emitted by in-vitro lung cancer cultured cells using gas sensor array and SPME-GCMS. BMC Cancer 2018, 18, 362. [Google Scholar] [CrossRef]
- Matsumura, K.; Opiekun, M.; Oka, H.; Vachani, A.; Albelda, S.M.; Yamazaki, K.; Beauchamp, G.K. Urinary Volatile Compounds as Biomarkers for Lung Cancer: A Proof of Principle Study Using Odor Signatures in Mouse Models of Lung Cancer. PLoS ONE 2010, 5, e8819. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Tao, J.; Li, J.; Tao, S. Volatile organic compounds analysis as a potential novel screening tool for colorectal cancer: A systematic review and meta-analysis. Medicine 2020, 99, e20937. [Google Scholar] [CrossRef] [PubMed]
- Janssens, E.; van Meerbeeck, J.P.; Lamote, K. Volatile organic compounds in human matrices as lung cancer biomarkers: A systematic review. Crit. Rev. Oncol. Hematol. 2020, 153, 103037. [Google Scholar] [CrossRef]
- Worthy, S.E.; Rojas, G.L.; Taylor, C.J.; Glater, E.E. Identification of Odor Blend Used by Caenorhabditis elegans for Pathogen Recognition. Chem. Senses 2018, 43, 169–180. [Google Scholar] [CrossRef]
- Di Luccio, E.; Morishita, M.; Hirotsu, T.C. elegans as a Powerful Tool for Cancer Screening. Biomedicines 2022, 10, 2371. [Google Scholar] [CrossRef] [PubMed]
- Edwards, T.L.; Browne, C.M.; Schoon, A.; Cox, C.; Poling, A. Animal olfactory detection of human diseases: Guidelines and systematic review. J. Vet. Behav. 2017, 20, 59–73. [Google Scholar] [CrossRef]
- Saetun, P.; Semangoen, T.; Thongboonkerd, V. Characterizations of urinary sediments precipitated after freezing and their effects on urinary protein and chemical analyses. Am. J. Physiol.-Ren. Physiol. 2009, 296, F1346–F1354. [Google Scholar] [CrossRef]
- Muppidi, S.S.; Katragadda, R.; Lega, J.; Alford, T.; Aidman, C.B.; Moore, C. A review of the efficacy of a low-cost cancer screening test using cancer sniffing canines. J. Breath Res. 2021, 15, 024001. [Google Scholar] [CrossRef]
- Jia, Z.; Patra, A.; Kutty, V.; Venkatesan, T. Critical Review of Volatile Organic Compound Analysis in Breath and In Vitro Cell Culture for Detection of Lung Cancer. Metabolites 2019, 9, 52. [Google Scholar] [CrossRef]
- Uchida, N.; Mainen, Z.F. Speed and accuracy of olfactory discrimination in the rat. Nat. Neurosci. 2003, 6, 1224–1229. [Google Scholar] [CrossRef]
- Slotnick, B. Animal cognition and the rat olfactory system. Trends Cogn. Sci. 2001, 5, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Arakawa, H.; Blanchard, D.C.; Arakawa, K.; Dunlap, C.; Blanchard, R.J. Scent marking behavior as an odorant communication in mice. Neurosci. Biobehav. Rev. 2008, 32, 1236–1248. [Google Scholar] [CrossRef] [PubMed]
- Becker, R. Non-invasive cancer detection using volatile biomarkers: Is urine superior to breath? Med. Hypothese 2020, 143, 110060. [Google Scholar] [CrossRef] [PubMed]
- Bekker, L.-G.; Alleyne, G.; Baral, S.; Cepeda, J.; Daskalakis, D.; Dowdy, D.; Dybul, M.; Eholie, S.; Esom, K.; Garnett, G.; et al. Advancing global health and strengthening the HIV response in the era of the Sustainable Development Goals: The International AIDS Society—Lancet Commission. Lancet 2018, 392, 312–358. [Google Scholar] [CrossRef] [PubMed]
- United Kingdom Lung Cancer Coalition. COVID-19 Matters. A Review of the Impact of COVID-19 on the Lung Cancer Pathway and Opportunities for Innovation Emerging from the Health System Response to the Pandemic. GB-NON-03328. 2020. Available online: https://www.uklcc.org.uk/ (accessed on 1 October 2020).
- Park, H.J.; Lee, S.H.; Chang, Y.S. Recent advances in diagnostic technologies in lung cancer. Korean J. Intern. Med. 2020, 35, 257–268. [Google Scholar] [CrossRef]
- Sciarra, A.; Di Lascio, G.; Del Giudice, F.; Leoncini, P.P.; Salciccia, S.; Gentilucci, A.; Porreca, A.; Chung, B.I.; Di Pierro, G.; Busetto, G.M.; et al. Comparison of the clinical usefulness of different urinary tests for the initial detection of bladder cancer: A systematic review. Curr. Urol. 2021, 15, 22–32. [Google Scholar] [CrossRef]
- Sabeena, S.; Kuriakose, S.; Binesh, D.; Abdulmajeed, J.; Dsouza, G.; Ramachandran, A.; Vijaykumar, B.; Aswathyraj, S.; Devadiga, S.; Ravishankar, N.; et al. The Utility of Urine-Based Sampling for Cervical Cancer Screening in Low-Resource Settings. Asian Pac. J. Cancer Prev. 2019, 20, 2409–2413. [Google Scholar] [CrossRef]
- Saman, H.; Raza, A.; Patil, K.; Uddin, S.; Crnogorac-Jurcevic, T. Non-Invasive Biomarkers for Early Lung Cancer Detection. Cancers 2022, 14, 5782. [Google Scholar] [CrossRef]

| Lung Cancer-Diagnosed Group = Positive Samples (N = 165) | ||
|---|---|---|
| Gender | F | 79 |
| M | 86 | |
| Mean Age | 68.19 | |
| Stage 0 (1) | 1 | |
| Adenocarcinoma | ||
| Stage 1 (92) | ||
| Adenocarcinoma | 77 | |
| Large-cell | 1 | |
| Squamous cell | 13 | |
| Other non-small-cell | 1 | |
| Stage 2 (14) | ||
| Adenocarcinoma | 5 | |
| Squamous cell | 4 | |
| Large-cell | 3 | |
| Small-cell | 2 | |
| Stage 3 (16) | ||
| Adenocarcinoma | 15 | |
| Other non-small-cell | 1 | |
| Stage 4 (40) | ||
| Adenocarcinoma | 29 | |
| Large-cell | 2 | |
| Squamous cell | 6 | |
| Small-cell | 3 | |
| Healthy Group = Negative Samples (N = 150) | ||
| Gender | F | 51 |
| M | 99 | |
| Mean Age | 51.22 | |
| LDCT Main Radiologic Findings (one or more) | 55 (37%) | |
| Soft tissue | 20 (36%) | |
| Granuloma | 12 (22%) | |
| Fibrosis | 8 (14.5%) | |
| Nodules | 42 (76.3%) | |
| Cancer Type | Number of Patients |
|---|---|
| Appendix neoplasm | 1 |
| Biliary tract cancer | 1 |
| Skin cancer | 1 |
| Breast cancer | 8 |
| Esophageal cancer | 2 |
| Rib neoplasm | 1 |
| Pancreatic carcinoma | 5 |
| Peritoneal mesothelioma | 1 |
| Solitary fibrous tumor of the pleura | 1 |
| Thymoma | 3 |
| Total | 24 |
| Smoking Status | Healthy Controls | LC Patients | Total |
|---|---|---|---|
| No | 32 (21%) | 50 (30%) | 82 |
| Yes | 28 (19%) | 34 (21%) | 62 |
| Unknown | 90 | 81 | 171 |
| Total | 150 | 165 | 315 |
| Medications | |||
| No | 34 (23%) | 15 (9%) | 49 |
| Yes | 26 (17%) | 62 (38%) | 88 |
| Unknown | 90 | 88 | 178 |
| Total | 150 | 165 | 315 |
| Positive | Samples | Negative | Samples |
|---|---|---|---|
| True positive | 154 | False positive | 13 2 |
| False negative | 11 1 | True negative | 137 |
| Total | 165 | Total | 150 |
| Sensitivity | Specificity | PPV | NPV |
| 93% | 91% | 92% | 93% |
| Lung Cancer | Healthy | ||
|---|---|---|---|
| Gender | 2 males, 8 females | 11 males, 2 females | |
| Age | 65.6 ± 10.7 | 50.6 ± 9.4 | |
| Cancer stage | I-IIIA, I-IIB | N/A | |
| Cancer subtypes | Adenocarcinoma | 8 | N/A |
| Squamous cell carcinoma | 1 | N/A | |
| Small-cell carcinoma | 1 | N/A | |
| Sample Name | Avg. 1 SR ± STD | ||
|---|---|---|---|
| Lung cancer patients | LC1 | 97% ± 2% | |
| LC2 | 73% ± 11% | ||
| LC3 | 98% ± 1% | ||
| LC4 | 80% ± 20% | ||
| LC5 | 87% ± 13% | ||
| LC6 | 80% ± 14% | ||
| LC7 | 71% ± 8% | ||
| LC8 | 81% ± 15% | ||
| LC9 | 97% ± 2% | ||
| LC10 | 80% ± 14% | ||
| Healthy patients | H1 | 95% ± 4% | |
| H2 | 93% ± 5% | ||
| H3 | 100% ± 0% | ||
| H4 | 93% ± 7% | ||
| H5 | 98% ± 2% | ||
| H6 | 88% ± 10% | ||
| H7 | 84% ± 5% | ||
| H8 | 87% ± 7% | ||
| H9 | 77% ± 16% | ||
| H10 | 84% ± 17% | ||
| H11 | 72% ± 10% | ||
| H12 | 94% ± 5% | ||
| H13 | 74% ± 13% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiesel, O.; Sung, S.-W.; Katz, A.; Leibowitz, R.; Bar, J.; Kamer, I.; Berger, I.; Nir-Ziv, I.; Mark Danieli, M. A Novel Urine Test Biosensor Platform for Early Lung Cancer Detection. Biosensors 2023, 13, 627. https://doi.org/10.3390/bios13060627
Wiesel O, Sung S-W, Katz A, Leibowitz R, Bar J, Kamer I, Berger I, Nir-Ziv I, Mark Danieli M. A Novel Urine Test Biosensor Platform for Early Lung Cancer Detection. Biosensors. 2023; 13(6):627. https://doi.org/10.3390/bios13060627
Chicago/Turabian StyleWiesel, Ory, Sook-Whan Sung, Amit Katz, Raya Leibowitz, Jair Bar, Iris Kamer, Itay Berger, Inbal Nir-Ziv, and Michal Mark Danieli. 2023. "A Novel Urine Test Biosensor Platform for Early Lung Cancer Detection" Biosensors 13, no. 6: 627. https://doi.org/10.3390/bios13060627
APA StyleWiesel, O., Sung, S.-W., Katz, A., Leibowitz, R., Bar, J., Kamer, I., Berger, I., Nir-Ziv, I., & Mark Danieli, M. (2023). A Novel Urine Test Biosensor Platform for Early Lung Cancer Detection. Biosensors, 13(6), 627. https://doi.org/10.3390/bios13060627





