Design and Engineering of a Palm-Sized Optical Immunosensing Device for the Detection of a Kidney Dysfunction Biomarker
Abstract
1. Introduction
2. Experimental Section
2.1. Materials and Reagents
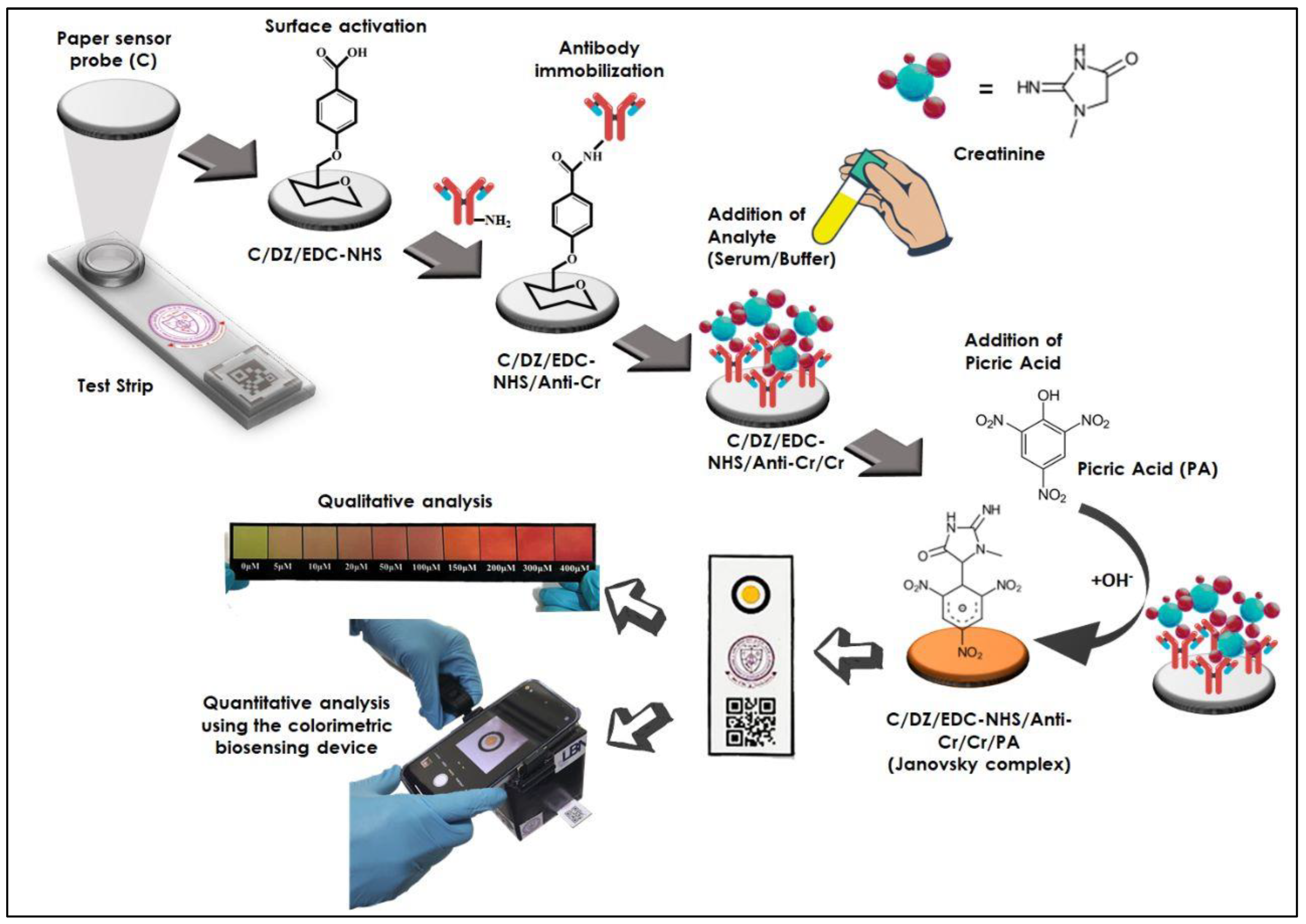
2.2. Designing of Sensing Probe
2.3. C/DZ/EDC-NHS/Anti-CR Biosensor Probe Testing
3. Experimental Results
3.1. Color Channel Selection
3.2. Characterization of C/DZ/EDC-NHS/Anti-CR/Cr Probe
3.3. Analytical Performance of C/DZ/EDC-NHS/Anti-CR Biosensor
3.4. Selectivity Assay
3.5. Analysis of Spiked Samples
3.6. Device Design
3.7. Storage, Stability and Reproducibility Test
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kovesdy, C.P. Epidemiology of Chronic Kidney Disease: An Update 2022. Kidney Int. Suppl. 2022, 12, 7–11. [Google Scholar] [CrossRef]
- Cockwell, P.; Fisher, L.A. The Global Burden of Chronic Kidney Disease. Lancet 2020, 395, 662–664. [Google Scholar] [CrossRef]
- Lv, J.C.; Zhang, L.X. Prevalence and Disease Burden of Chronic Kidney Disease. Adv. Exp. Med. Biol. 2019, 1165, 3–15. [Google Scholar] [CrossRef]
- Jankowski, J.; Floege, J.; Fliser, D.; Böhm, M.; Marx, N. Cardiovascular Disease in Chronic Kidney Disease: Pathophysiological Insights and Therapeutic Options. Circulation 2021, 143, 1157. [Google Scholar] [CrossRef]
- Bates, C.M.; Charlton, J.R.; Ferris, M.E.; Hildebrandt, F.; Hoshizaki, D.K.; Warady, B.A.; Moxey-Mims, M.M. Pediatric Kidney Disease: Tracking Onset and Improving Clinical Outcomes. Clin. J. Am. Soc. Nephrol. 2014, 9, 1141–1143. [Google Scholar] [CrossRef]
- Haffner, D. Strategies for Optimizing Growth in Children With Chronic Kidney Disease. Front. Pediatr. 2020, 8, 399. [Google Scholar] [CrossRef]
- Alshelleh, S.; Alhouri, A.; Taifour, A.; Abu-Hussein, B.; Alwreikat, F.; Abdelghani, M.; Badran, M.; Al-Asa’d, Y.; Alhawari, H.; Oweis, A.O. Prevelance of Depression and Anxiety with Their Effect on Quality of Life in Chronic Kidney Disease Patients. Sci. Rep. 2022, 12, 17627. [Google Scholar] [CrossRef]
- Lousa, I.; Reis, F.; Beirão, I.; Alves, R.; Belo, L.; Santos-Silva, A. New Potential Biomarkers for Chronic Kidney Disease Management-A Review of the Literature. Int. J. Mol. Sci. 2020, 22, 43. [Google Scholar] [CrossRef]
- Ali, A.; Asif, N.; Rais, Z. Estimation of GFR by MDRD Formula and Its Correlation to Cockcroft-Gault Equation in Five Stages of Chronic Kidney Disease. Undefined 2013, 3, 37–40. [Google Scholar] [CrossRef]
- Nashar, K.; Egan, B.M. Relationship between Chronic Kidney Disease and Metabolic Syndrome: Current Perspectives. Diabetes Metab. Syndr. Obes. Targets Ther. 2014, 7, 421–435. [Google Scholar] [CrossRef]
- Fassett, R.G.; Venuthurupalli, S.K.; Gobe, G.C.; Coombes, J.S.; Cooper, M.A.; Hoy, W.E. Biomarkers in Chronic Kidney Disease: A Review. Kidney Int. 2011, 80, 806–821. [Google Scholar] [CrossRef]
- Zhang, W.R.; Parikh, C.R. Biomarkers of Acute and Chronic Kidney Disease. Annu. Rev. Physiol. 2019, 81, 309–333. [Google Scholar] [CrossRef]
- Levy, J.; Brown, E.; Lawrence, A. Oxford Handbook of Dialysis; Oxford University Press: Oxford, UK, 2016. [Google Scholar]
- Washington, I.M.; Van Hoosier, G. Clinical Biochemistry and Hematology. In The Laboratory Rabbit, Guinea Pig, Hamster, and Other Rodents; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 9780123809209. [Google Scholar]
- Vupputuri, S.; Sandler, D.P. Lifestyle Risk Factors and Chronic Kidney Disease. Ann. Epidemiol. 2003, 13, 712–720. [Google Scholar] [CrossRef]
- Himmelfarb, J.; Ikizler, T.A. Chronic Kidney Disease, Dialysis, and Transplantation: A Companion to Brenner and Rector’s The Kidney; Elsevier Health Sciences: Amsterdam, The Netherlands, 2018; ISBN 9780323529785. [Google Scholar]
- Lopez-Giacoman, S. Biomarkers in Chronic Kidney Disease, from Kidney Function to Kidney Damage. World J. Nephrol. 2015, 4, 57. [Google Scholar] [CrossRef]
- Kreider, R.B.; Stout, J.R. Creatine in Health and Disease. Nutrients 2021, 13, 447. [Google Scholar] [CrossRef]
- Biomarkers in Acute and Chronic Kidney Diseases—ClinicalKey. Available online: https://www.clinicalkey.com/#!/content/book/3-s2.0-B9780323532655000277 (accessed on 14 October 2022).
- Pundir, C.S.; Yadav, S.; Kumar, A. Creatinine Sensors. TrAC Trends Anal. Chem. 2013, 50, 42–52. [Google Scholar] [CrossRef]
- Laboratory Exams For Chronic Kidney Disease—Renal Diet HQ. Available online: https://www.renaldiethq.com/laboratory-exams-chronic-kidney-disease/ (accessed on 14 October 2022).
- Chronic Kidney Disease Tests & Diagnosis|NIDDK. Available online: https://www.niddk.nih.gov/health-information/kidney-disease/chronic-kidney-disease-ckd/tests-diagnosis (accessed on 14 October 2022).
- Tajarrod, N.; Rofouei, M.K.; Masteri-Farahani, M.; Zadmard, R. A Quantum Dot-Based Fluorescence Sensor for Sensitive and Enzymeless Detection of Creatinine. Anal. Methods 2016, 8, 5911–5920. [Google Scholar] [CrossRef]
- Aly, A.H.; Mohamed, D.; Mohaseb, M.A.; El-Gawaad, N.S.A.; Trabelsi, Y. Biophotonic Sensor for the Detection of Creatinine Concentration in Blood Serum Based on 1D Photonic Crystal. RSC Adv. 2020, 10, 31765–31772. [Google Scholar] [CrossRef]
- Kalasin, S.; Sangnuang, P.; Khownarumit, P.; Tang, I.M.; Surareungchai, W. Evidence of Cu(I) Coupling with Creatinine Using Cuprous Nanoparticles Encapsulated with Polyacrylic Acid Gel-Cu(II) in Facilitating the Determination of Advanced Kidney Dysfunctions. ACS Biomater. Sci. Eng. 2020, 6, 1247–1258. [Google Scholar] [CrossRef]
- Lad, U.; Khokhar, S.; Kale, G.M. Electrochemical Creatinine Biosensors. Anal. Chem. 2008, 80, 7910–7917. [Google Scholar] [CrossRef]
- Ekelund, S.; Paby, P. High-Performance Liquid Chromatographic Determination of Creatinine. Scand. J. Clin. Lab. Investig. 1991, 51, 67–71. [Google Scholar] [CrossRef]
- Fernández-Fernández, M.; Rodríguez-González, P.; Añón Álvarez, M.E.; Rodríguez, F.; Álvarez Menéndez, F.V.; García Alonso, J.I. Simultaneous Determination of Creatinine and Creatine in Human Serum by Double-Spike Isotope Dilution Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) and Gas Chromatography-Mass Spectrometry (GC-MS). Anal. Chem. 2015, 87, 3755–3763. [Google Scholar] [CrossRef]
- Purohit, B.; Vernekar, P.R.; Shetti, N.P.; Chandra, P. Biosensor Nanoengineering: Design, Operation, and Implementation for Biomolecular Analysis. Sens. Int. 2020, 1, 100040. [Google Scholar] [CrossRef]
- Chandra, P.; Prakash, R. Nanobiomaterial Engineering: Concepts and Their Applications in Biomedicine and Diagnostics; Springer: Berlin, Germany, 2020; pp. 1–294. [Google Scholar] [CrossRef]
- Bonsnes, R.W.; Taussky, H.H. ON the colorimetric determination of creatinine by the jaffe reaction. J. Biol. Chem. 1945, 158, 581–591. [Google Scholar] [CrossRef]
- Siedel, J.; Deeg, R.; Seidel, H.; Mollering, H.; Staepels, J.; Gauhl, H.; Ziegenhorn, J. Fully Enzymatic Colorimetric Assay of Serum and Urine Creatinine Which Obviates the Need for Sample Blank Measurements. Anal. Lett. 2006, 21, 1009–1017. [Google Scholar] [CrossRef]
- Sin, M.L.; Mach, K.E.; Wong, P.K.; Liao, J.C. Advances and Challenges in Biosensor-Based Diagnosis of Infectious Diseases. Expert Rev. Mol. Diagn. 2014, 14, 225–244. [Google Scholar] [CrossRef]
- Mahato, K.; Srivastava, A.; Chandra, P. Paper Based Diagnostics for Personalized Health Care: Emerging Technologies and Commercial Aspects. Biosens. Bioelectron. 2017, 96, 246–259. [Google Scholar] [CrossRef]
- Mahato, K.; Chandra, P. Paper-Based Miniaturized Immunosensor for Naked Eye ALP Detection Based on Digital Image Colorimetry Integrated with Smartphone. Biosens. Bioelectron. 2019, 128, 9–16. [Google Scholar] [CrossRef]
- Mahato, K.; Purohit, B.; Kumar, A.; Chandra, P. Paper-Based Biosensors for Clinical and Biomedical Applications: Emerging Engineering Concepts and Challenges. In Comprehensive Analytical Chemistry; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Natarajan, S.; Derosa, M.C.; Shah, M.I.; Jayaraj, J. Development and Evaluation of a Quantitative Fluorescent Lateral Flow Immunoassay for Cystatin-C, a Renal Dysfunction Biomarker. Sensors 2021, 21, 3178. [Google Scholar] [CrossRef]
- Gao, B.; Liu, H.; Gu, Z. Patterned Photonic Nitrocellulose for Pseudo-Paper Microfluidics. Anal. Chem. 2016, 88, 5424–5429. [Google Scholar] [CrossRef]
- De Carvalho Oliveira, G.; Machado, C.C.S.; Inácio, D.K.; Petruci, J.F.S.; da Silva, S.G. RGB Color Sensor for Colorimetric Determinations: Evaluation and Quantitative Analysis of Colored Liquid Samples. Talanta 2022, 241, 123244. [Google Scholar] [CrossRef] [PubMed]
- Chun, H.J.; Han, Y.D.; Park, Y.M.; Kim, K.R.; Lee, S.J.; Yoon, H.C. An Optical Biosensing Strategy Based on Selective Light Absorption and Wavelength Filtering from Chromogenic Reaction. Materials 2018, 11, 388. [Google Scholar] [CrossRef]
- Saha, J.; Wang, S.Y.; Davis, A.J. Examining DNA Double-Strand Break Repair in a Cell Cycle-Dependent Manner. Methods Enzymol. 2017, 591, 97–118. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Yang, X.; Ji, Z.; Zhu, L.; Ma, N.; Chen, D.; Jia, X.; Tang, J.; Cao, Y. DFT-Calculated IR Spectrum Amide I, II, and III Band Contributions of N-Methylacetamide Fine Components. ACS Omega 2020, 5, 8572–8578. [Google Scholar] [CrossRef] [PubMed]
- Talalak, K.; Noiphung, J.; Songjaroen, T.; Chailapakul, O.; Laiwattanapaisal, W. A Facile Low-Cost Enzymatic Paper-Based Assay for the Determination of Urine Creatinine. Talanta 2015, 144, 915–921. [Google Scholar] [CrossRef]
- Mohammadi, S.; Khayatian, G. Highly Selective and Sensitive Photometric Creatinine Assay Using Silver Nanoparticles. Microchim. Acta 2015, 182, 1379–1386. [Google Scholar] [CrossRef]
- Sivasankaran, U.; Jos, T.C.; Kumar, K.G. Selective Recognition of Creatinine—Development of a Colorimetric Sensor. Anal. Biochem. 2018, 544, 1–6. [Google Scholar] [CrossRef]
- Ciou, D.S.; Wu, P.H.; Huang, Y.C.; Yang, M.C.; Lee, S.Y.; Lin, C.Y. Colorimetric and Amperometric Detection of Urine Creatinine Based on the ABTS Radical Cation Modified Electrode. Sensors Actuators B Chem. 2020, 314, 128034. [Google Scholar] [CrossRef]
- Lewińska, I.; Speichert, M.; Granica, M.; Tymecki, Ł. Colorimetric Point-of-Care Paper-Based Sensors for Urinary Creatinine with Smartphone Readout. Sens. Actuators B Chem. 2021, 340, 129915. [Google Scholar] [CrossRef]
- Thammajinno, S.; Buranachai, C.; Kanatharana, P.; Thavarungkul, P.; Thammakhet-Buranachai, C. A Copper Nanoclusters Probe for Dual Detection of Microalbumin and Creatinine. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 270, 120816. [Google Scholar] [CrossRef]
- Babu, P.J.; Raichur, A.M.; Doble, M. Synthesis and Characterization of Biocompatible Carbon-Gold (C-Au) Nanocomposites and Their Biomedical Applications as an Optical Sensor for Creatinine Detection and Cellular Imaging. Sens. Actuators B Chem. 2018, 258, 1267–1278. [Google Scholar] [CrossRef]
- Mathew, M.S.; Joseph, K. Green Synthesis of Gluten-Stabilized Fluorescent Gold Quantum Clusters: Application As Turn-On Sensing of Human Blood Creatinine. ACS Sustain. Chem. Eng. 2017, 5, 4837–4845. [Google Scholar] [CrossRef]
- Du, J.; Zhu, B.; Leow, W.R.; Chen, S.; Sum, T.C.; Peng, X.; Chen, X. Colorimetric Detection of Creatinine Based on Plasmonic Nanoparticles via Synergistic Coordination Chemistry. Small 2015, 11, 4104–4110. [Google Scholar] [CrossRef] [PubMed]
- Alula, M.T.; Karamchand, L.; Hendricks, N.R.; Blackburn, J.M. Citrate-Capped Silver Nanoparticles as a Probe for Sensitive and Selective Colorimetric and Spectrophotometric Sensing of Creatinine in Human Urine. Anal. Chim. Acta 2018, 1007, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Lewińska, I.; Tymecki, Ł.; Michalec, M. An Alternative, Single-Point Method for Creatinine Determination in Urine Samples with Optoelectronic Detector. Critical Comparison to Jaffé Method. Talanta 2019, 195, 865–869. [Google Scholar] [CrossRef]
- Krishnegowda, A.; Padmarajaiah, N.; Anantharaman, S.; Honnur, K. Spectrophotometric Assay of Creatinine in Human Serum Sample. Arab. J. Chem. 2017, 10, S2018–S2024. [Google Scholar] [CrossRef]
- He, Y.; Zhang, X.; Yu, H. Gold Nanoparticles-Based Colorimetric and Visual Creatinine Assay. Microchim. Acta 2015, 182, 2037–2043. [Google Scholar] [CrossRef]
- Forbes, A.; Gallagher, H. Chronic Kidney Disease in Adults: Assessment and Management. Clin. Med. 2020, 20, 128–132. [Google Scholar] [CrossRef]






| Sr. No. | Analyte | Detection Method | Sensor Configuration | Time | Real Sample | Detection Range | Clinical Range | Point-of-Care Device Prototype | Limit of Detection | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. | Creatinine | Optical (Colorimetric) | CuNPs have been integrated with L-cysteine for selective and sensitive interaction with Creatinine | 20 min | Serum and Urine matrix | 0.33–5.33 μM | NO | NO | 0.454 nM | [45] |
| 2. | Creatinine | Optical (Colorimetric) | ABTS was introduced and entrapped in fluorine-doped tin oxide-modified chitosan film | 12.5 min | Urine matrix | 0–21300 μM | YES | NO | 400 μM | [46] |
| 3. | Creatinine | Colorimetric | 3D-printed element was integrated with smartphone for detection using Hue channel intensity in Jaffe method | 6 min | Urine matrix | 1000–2000 μM | NO | NO | 350 μM | [47] |
| 4. | Creatinine | Colorimetric | 3,5-dinitrobenzoate was used for quantification by targeting green channel intensity | 6 min | Urine matrix | 820–105 μM | NO | NO | 270 μM | [47] |
| 5. | Creatinine | Fluorescence | Glutathione based copper nanoclusters were designed using ascorbic acid for turn-on mode | 15 min | Urine matrix | 30–1000 μM | YES | NO | 13.0 μM | [48] |
| 6. | Creatinine | Optical | C-gold nanocomposite was designed using carbon nanodots derived from Citrullus lanatus for photoluminescent imaging | 10 min | Not Performed | 17–1700 μM | YES | NO | 6.19 μM | [49] |
| 7. | Creatinine | Optical | H2O2 released through creatinine conversion was detected by reacting it with 4-aminophenazone and hydroxybenzoic acid on a μPAD | 15 min | Urine matrix | 221–2210 μM | NO | NO | 176 μM | [43] |
| 8. | Creatinine | Optical (fluorescence) | Gluten stabilized gold quantum cluster was developed to form on/off system for creatinine | 3 hr | Serum matrix | 20–520 μM | YES | NO | 2 nM | [50] |
| 9. | Creatinine | Optical | Silver nanoparticles were capped with 2,2-thiodiacetic acid to react with creatinine | 5 min | Serum matrix | 0.01–1 μM | NO | NO | 3 nM | [44] |
| 10. | Creatinine | Optical (Colorimetric) | Gold nanoparticles were modified with mercury and detection was performed on synergistic coordination | 5 min | Urine matrix | 15–35 µM | NO | NO | 19.8 nM | [51] |
| 11. | Creatinine | Optical (Colorimetric) | Silver nanoparticles capped with citrate for creatinine detection | 1 min | Urine matrix | 0–4.2 µM | NO | NO | 53.4 nM | [52] |
| 12. | Creatinine | Optical | 3,5 dinitrobenzoic acid was used as chromophore for creatinine detection | 2 min | Urine matrix | 10–30 µM | NO | NO | Not reported | [53] |
| 13. | Creatinine | Optical | Methylamino phenol sulfate was used for oxidation using copper sulfate in presence of creatinine | 30 min | Serum matrix | 4.4–620 µM | YES | NO | 145 nM | [54] |
| 14. | Creatinine | Optical | Gold nanoparticles were capped with citrate for the detection of creatinine | 24 min | Urine matrix | 0.1–20 mM | NO | NO | 80 µM | [55] |
| 15. | Creatinine | Optical | C/DZ/EDC-NHS/Anti-CR/Cr | 7 min | Serum matrix | 5–400 μM | YES | YES | 15.37 nM | This work |
| S.No | Spiked (μM) | Recovered (μM) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| 1 | 5 | 4.77 (±0.05) | 95.40 | 1.10 |
| 2 | 7.5 | 6.75 (±0.18) | 90.09 | 2.80 |
| 3 | 10 | 9.54 (±0.25) | 95.46 | 2.66 |
| 4 | 15 | 13.45 (±0.26) | 89.71 | 1.96 |
| 5 | 20 | 19.46 (±0.39) | 97.30 | 2.02 |
| 6 | 35 | 32.99 (±0.99) | 94.27 | 3.02 |
| 7 | 50 | 47.31 (±1.41) | 94.62 | 2.98 |
| 8 | 100 | 92.55 (±2.92) | 92.55 | 3.15 |
| 9 | 150 | 139.99 (±4.61) | 93.32 | 3.29 |
| 10 | 200 | 189.29 (±5.39) | 94.64 | 2.84 |
| 11 | 300 | 288.73 (±3.62) | 96.24 | 1.25 |
| 12 | 400 | 384.50 (±4.01) | 96.12 | 1.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Divya; Mahapatra, S.; Chandra, P. Design and Engineering of a Palm-Sized Optical Immunosensing Device for the Detection of a Kidney Dysfunction Biomarker. Biosensors 2022, 12, 1118. https://doi.org/10.3390/bios12121118
Divya, Mahapatra S, Chandra P. Design and Engineering of a Palm-Sized Optical Immunosensing Device for the Detection of a Kidney Dysfunction Biomarker. Biosensors. 2022; 12(12):1118. https://doi.org/10.3390/bios12121118
Chicago/Turabian StyleDivya, Supratim Mahapatra, and Pranjal Chandra. 2022. "Design and Engineering of a Palm-Sized Optical Immunosensing Device for the Detection of a Kidney Dysfunction Biomarker" Biosensors 12, no. 12: 1118. https://doi.org/10.3390/bios12121118
APA StyleDivya, Mahapatra, S., & Chandra, P. (2022). Design and Engineering of a Palm-Sized Optical Immunosensing Device for the Detection of a Kidney Dysfunction Biomarker. Biosensors, 12(12), 1118. https://doi.org/10.3390/bios12121118






