Aptamer-Based Gold Nanoparticles–PDMS Composite Stamps as a Platform for Micro-Contact Printing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Glass Slides Cleaning and Surface Modification with DMOAP
2.3. Preparation of PDMS Stamps
2.4. U.V. Treatment of PDMS Stamp and DMOAP-Coated Slide
2.5. Preparation of AuNPs–PDMS Composite Stamp
2.6. Immobilisation of B40t77 Aptamer on AuNPs–PDMS Stamp
2.7. Characterisation of Prepared Solid Stamp Materials
2.8. Patterning of Target Protein through Micro-Contact Printing
2.9. Fabrication of LCs Optical Cell
2.10. Stability Check of the Prepared AuNPs–PDMS Stamp
3. Results and Discussion
3.1. Proposed Scheme of μCP of Gp-120 Protein Based on AuNPs–PDMS Composite Stamp
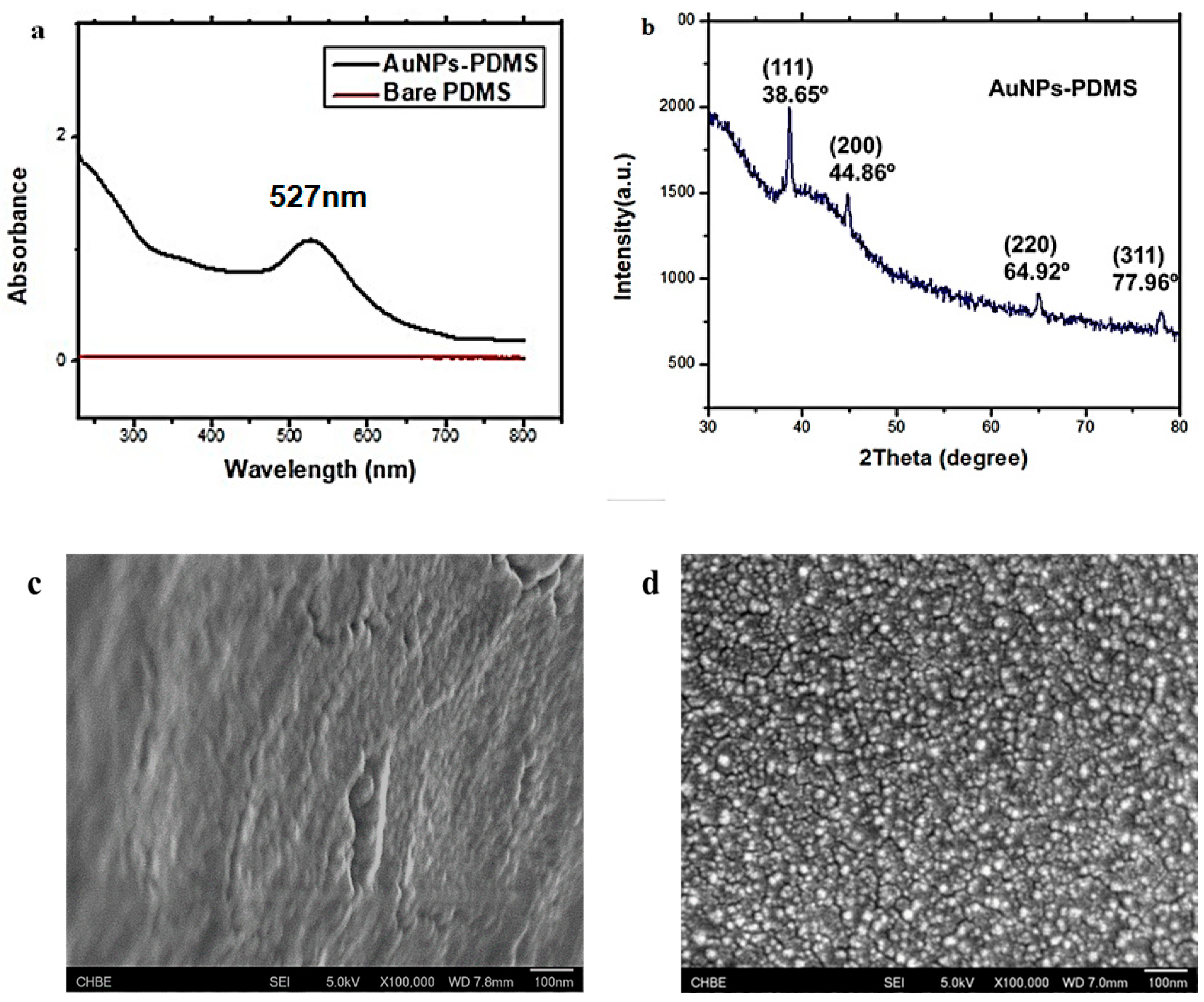
3.2. Characterisation of AuNPs–PDMS Composite Stamp
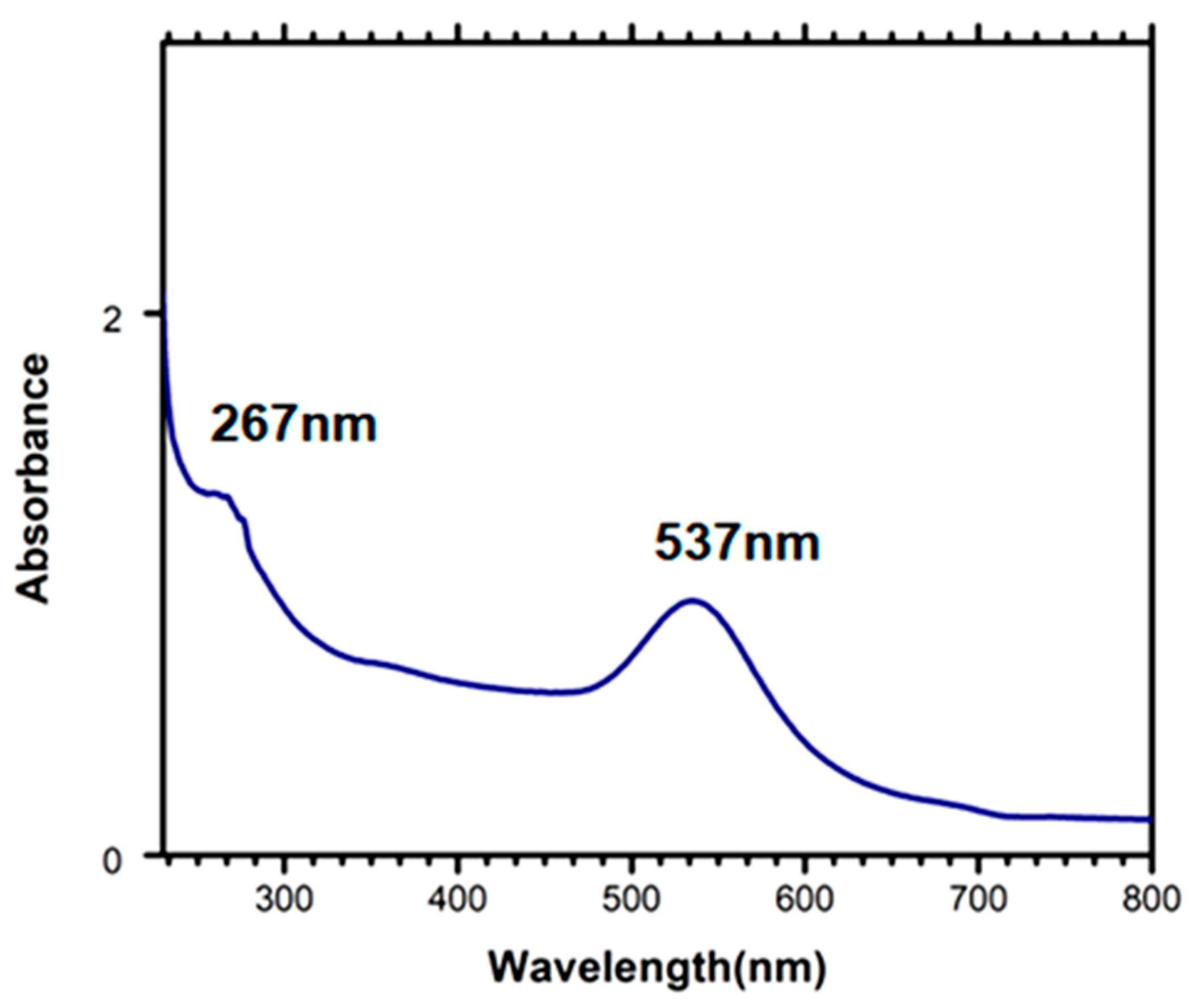
3.3. Characterisation of Aptamer Immobilised on PDMS–AuNPs Composite Stamps
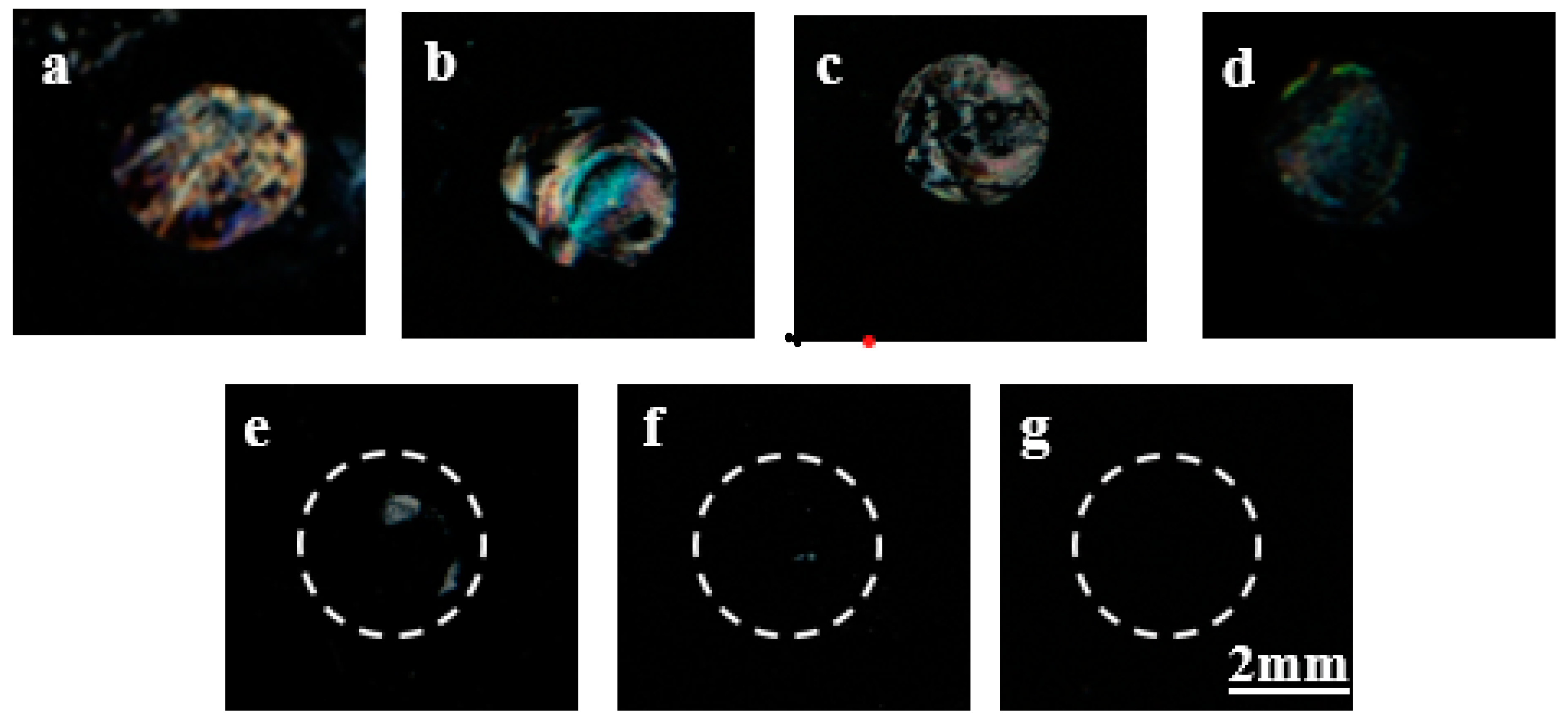
3.4. Detection of Gp-120 Target Protein through µCP by Using LCs
3.5. Capturing Desired Target Protein (Gp-120) in a Protein Mixture Solution
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sun, H.; Chen, G.Y.J.; Yao, S.Q. Review Recent Advances in Microarray Technologies for Proteomics. Chem. Biol. 2013, 20, 685–699. [Google Scholar] [CrossRef]
- Hölz, K.; Schaudy, E.; Lietard, J.; Somoza, M.M. Multi-level patterning nucleic acid photolithography. Nat. Commun. 2019, 10, 1–8. [Google Scholar] [CrossRef]
- Ogaki, R.; Alexander, M.; Kingshott, P. Chemical patterning in biointerface science Patterning of surfaces with different chemistries provides novel insights of new chemically patterned surfaces is highlighted. Mater. Today 2010, 13, 22–35. [Google Scholar] [CrossRef]
- Delamarche, E.; Pereiro, I.; Kashyap, A.; Kaigala, G.V. Biopatterning: The Art of Patterning Biomolecules on Surfaces. Langmuir 2021, 37, 9637–9651. [Google Scholar] [CrossRef]
- Juste-Dolz, A.; Avella-Oliver, M.; Puchades, R.; Maquieira, A. Indirect microcontact printing to create functional patterns of physisorbed antibodies. Sensors 2018, 18, 3163. [Google Scholar] [CrossRef]
- Bernard, A.; Renault, J.P.; Michel, B.; Bosshard, H.R.; Delamarche, E. Microcontact printing of proteins. Adv. Mater. 2000, 12, 1067–1070. [Google Scholar] [CrossRef]
- Chen, C.H.; Yang, K.L. Improving protein transfer efficiency and selectivity in affinity contact printing by using UV-modified surfaces. Langmuir 2011, 27, 5427–5432. [Google Scholar] [CrossRef]
- Miranda, I.; Souza, A.; Sousa, P.; Ribeiro, J.; Castanheira, E.M.S.; Lima, R.; Minas, G. Properties and applications of PDMS for biomedical engineering: A review. J. Funct. Biomater. 2022, 13, 2. [Google Scholar] [CrossRef]
- Gökaltun, A.; Kang, Y.B.; Yarmush, M.L.; Usta, O.B.; Asatekin, A. Simple Surface Modification of Poly(dimethylsiloxane) via Surface Segregating Smart Polymers for Biomicrofluidics. Sci. Rep. 2019, 9, 7377. [Google Scholar] [CrossRef]
- Tu, Q.; Wang, J.C.; Zhang, Y.; Liu, R.; Liu, W.; Ren, L.; Shen, S.; Xu, J.; Zhao, L.; Wang, J. Surface modification of poly(dimethylsiloxane) and its applications in microfluidics-based biological analysis. Rev. Anal. Chem. 2012, 31, 177–192. [Google Scholar] [CrossRef]
- Wang, W.; Wu, W.Y.; Zhong, X.; Wang, W.; Miao, Q.; Zhu, J.J. Aptamer-based PDMS-gold nanoparticle composite as a platform for visual detection of biomolecules with silver enhancement. Biosens. Bioelectron. 2011, 26, 3110–3114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Xu, J.J.; Liu, Y.; Chen, H.Y. In-situ synthesis of poly(dimethylsiloxane)-gold nanoparticles composite films and its application in microfluidic systems. Lab Chip 2008, 8, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhou, J.; Wu, X.; Riaud, A. Optimization of synthesis conditions of gold nanoparticlespolydimethylsiloxane composite for ultrasound generation. Nanotechnology 2021, 3, 2850–2857. [Google Scholar]
- Zhu, A.; Ali, S.; Xu, Y.; Ouyang, Q.; Chen, Q. A SERS aptasensor based on AuNPs functionalized PDMS film for selective and sensitive detection of Staphylococcus aureus. Biosens. Bioelectron. 2021, 172, 112806. [Google Scholar] [CrossRef]
- Yao, J.Y.; Fostier, A.H.; Santos, E.B. In situ formation of gold and silver nanoparticles on uniform PDMS films and colorimetric analysis of their plasmonic color. Colloids Surfaces A Physicochem. Eng. Asp. 2020, 607, 125463. [Google Scholar] [CrossRef]
- Pusty, M.; Shirage, P.M. Gold nanoparticle-cellulose/PDMS nanocomposite: A flexible dielectric material for harvesting mechanical energy. RSC Adv. 2020, 10, 10097–10112. [Google Scholar] [CrossRef]
- SadAbadi, H.; Badilescu, S.; Packirisamy, M.; Wüthrich, R. Integration of gold nanoparticles in PDMS microfluidics for lab-on-a-chip plasmonic biosensing of growth hormones. Biosens. Bioelectron. 2013, 44, 77–84. [Google Scholar] [CrossRef]
- Xue, Y.; Li, X.; Li, H.; Zhang, W. Quantifying thiol-gold interactions towards the efficient strength control. Nat. Commun. 2014, 5, 4348. [Google Scholar] [CrossRef]
- Wu, W.Y.; Bian, Z.P.; Wang, W.; Wang, W.; Zhu, J.J. PDMS gold nanoparticle composite film-based silver enhanced colorimetric detection of cardiac troponin I. Sens. Actuators B Chem. 2010, 147, 298–303. [Google Scholar] [CrossRef]
- Li, Y.; Liu, S.; Ling, L.; Xue, C.Y.; Chin, S.Y.; Khan, S.A.; Yang, K.L.; Tu, Q.; Wang, J.C.J.; Zhang, Y.; et al. Recent developments in aptasensors for diagnostic applications. Langmuir 2018, 18, 77–84. [Google Scholar] [CrossRef]
- Abbasi, A.D.; Hussain, Z.; Yang, K.L. Aptamer laden liquid crystals biosensing platform for the detection of HIV-1 glycoprotein-120. Molecules 2021, 26, 2893. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, Q.; Wu, W. Graphene-Based Steganographic Aptasensor for Information Computing and Monitoring Toxins of Biofilm in Food. Front. Microbiol. 2020, 10, 3139. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, A.D.; Hussain, Z.; Liaqat, U.; Arif, D.; Yang, K.-L. Liquid Crystal Based Binding Assay for Detecting HIV-1 Surface Glycoprotein. Front. Chem. 2021, 9, 257. [Google Scholar] [CrossRef] [PubMed]
- Dey, A.K.; Griffiths, C.; Lea, S.M.; James, W. Structural characterization of an anti-gp120 RNA aptamer that neutralizes R5 strains of HIV-1. RNA 2005, 11, 873–884. [Google Scholar] [CrossRef]
- Chen, J.; Liu, Z.; Yang, R.; Liu, M.; Yao, J.; Zhang, M.; Li, N.; Yuan, Z.; Jin, M.; Shui, L. A label-free optical immunoassay based on birefringence of liquid crystal for insulin-like growth factor-I sensing. Sens. Actuators B Chem. 2022, 352, 131028. [Google Scholar] [CrossRef]
- Lin, C.T.; Hsu, W.T.; Hwang, S.J. Real-time liquid crystal-based creatinine sensor using a micro-patterned flexible substrate. Liq. Cryst. 2021, 48, 1660–1670. [Google Scholar] [CrossRef]
- Khan, M.; Liu, S.; Qi, L.; Ma, C.; Munir, S.; Yu, L.; Hu, Q. Liquid crystal-based sensors for the detection of biomarkers at the aqueous/LC interface. TrAC Trends Anal. Chem. 2021, 144, 116434. [Google Scholar] [CrossRef]
- Hong, P.T.K.; Jang, C.H. Simple, sensitive technique for α-amylase detection facilitated by liquid crystal-based microcapillary sensors. Microchem. J. 2021, 162, 105864. [Google Scholar] [CrossRef]
- Yin, F.; Cheng, S.; Liu, S.; Ma, C.; Wang, L.; Zhao, R.; Lin, J.M.; Hu, Q. A portable digital optical kanamycin sensor developed by surface-anchored liquid crystal droplets. J. Hazard. Mater. 2021, 420, 126601. [Google Scholar] [CrossRef]
- SadAbadi, H.; Badilescu, S.; Packirisamy, M.; Wuẗhrich, R. PDMS-gold nanocomposite platforms with enhanced sensing properties. J. Biomed. Nanotechnol. 2012, 8, 539–549. [Google Scholar] [CrossRef]
- John, S.V.; Khati, M.; Mamba, B.B.; Arotiba, O.; Rotherham, L.S. Towards HIV detection: Novel Poly (propylene) dendrimer-streptavidin platform for electrochemical DNA and gp-120 aptamer biosensors. Int. J. Electrochem. Sci. 2014, 9, 5425–5437. [Google Scholar]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbasi, A.D.; Hussain, Z.; Yang, K.-L. Aptamer-Based Gold Nanoparticles–PDMS Composite Stamps as a Platform for Micro-Contact Printing. Biosensors 2022, 12, 1067. https://doi.org/10.3390/bios12121067
Abbasi AD, Hussain Z, Yang K-L. Aptamer-Based Gold Nanoparticles–PDMS Composite Stamps as a Platform for Micro-Contact Printing. Biosensors. 2022; 12(12):1067. https://doi.org/10.3390/bios12121067
Chicago/Turabian StyleAbbasi, Amna Didar, Zakir Hussain, and Kun-Lin Yang. 2022. "Aptamer-Based Gold Nanoparticles–PDMS Composite Stamps as a Platform for Micro-Contact Printing" Biosensors 12, no. 12: 1067. https://doi.org/10.3390/bios12121067
APA StyleAbbasi, A. D., Hussain, Z., & Yang, K.-L. (2022). Aptamer-Based Gold Nanoparticles–PDMS Composite Stamps as a Platform for Micro-Contact Printing. Biosensors, 12(12), 1067. https://doi.org/10.3390/bios12121067





