Systematic Screening of Trigger Moieties for Designing Formaldehyde Fluorescent Probes and Application in Live Cell Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.1.1. Apparatus
2.1.2. Reagents
2.2. Synthesis
2.3. General Procedure for Absorption and Fluorescent Measurement
2.4. Cells Imaging
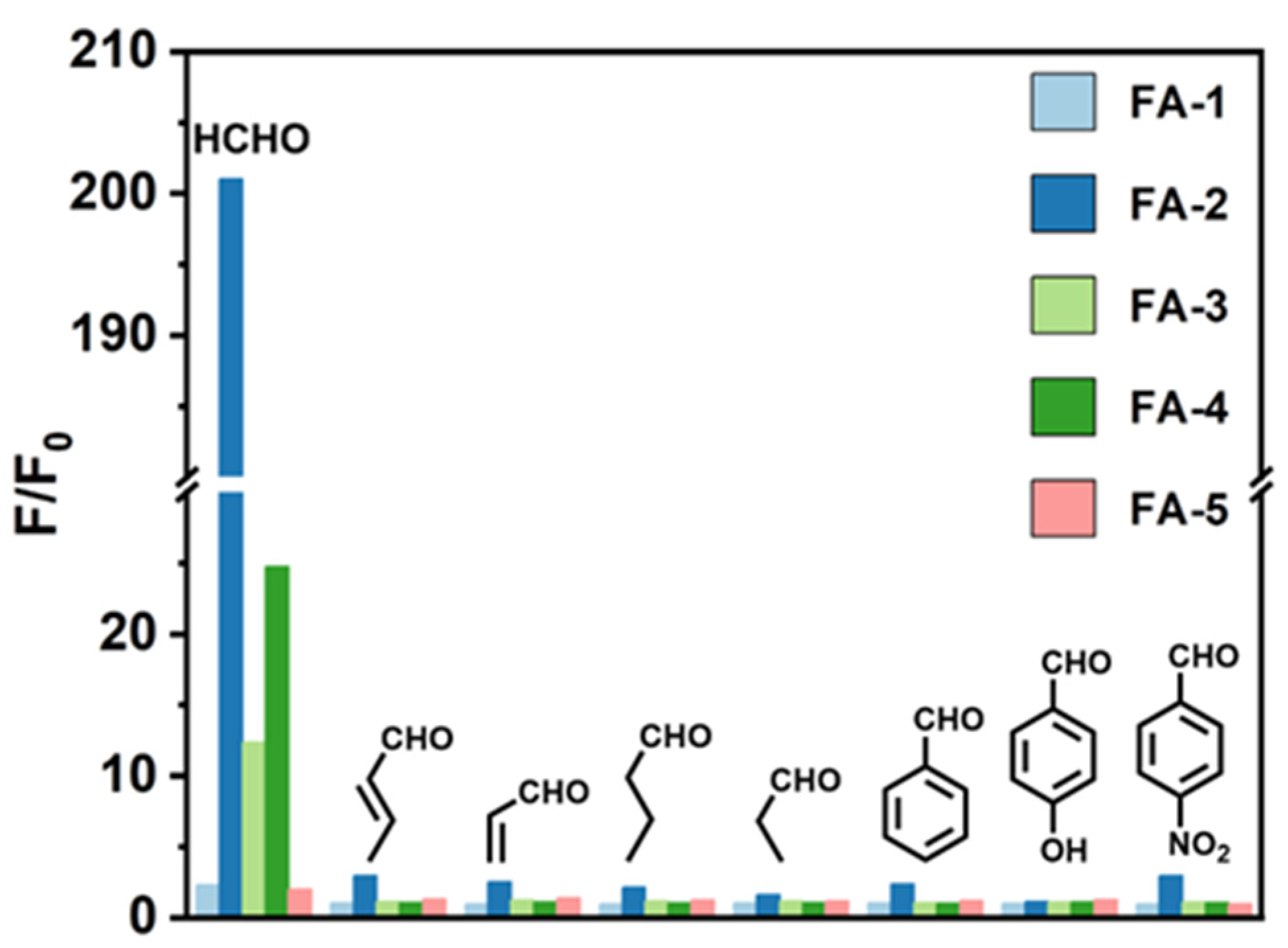
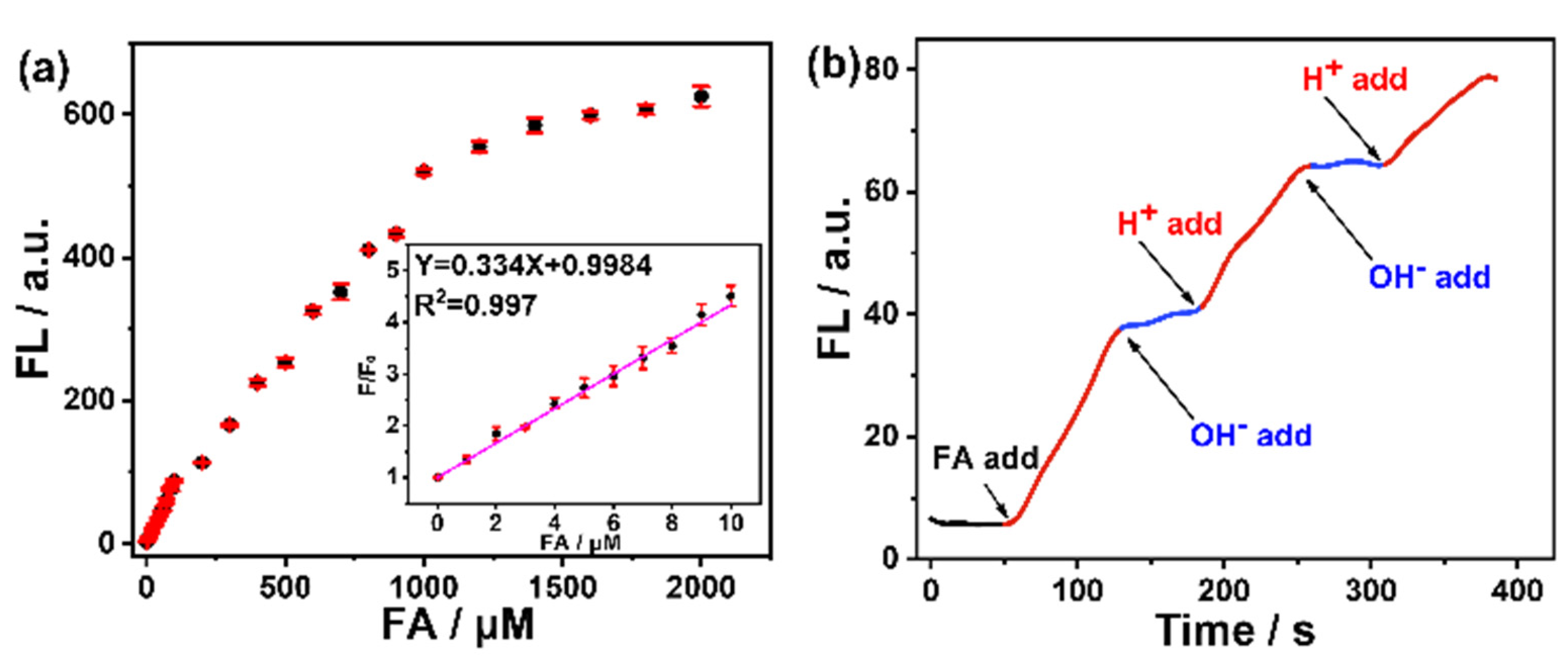
3. Result and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Heim, L.E.; Konnerth, H.; Prechtl, M.H.G. Future perspectives for formaldehyde: Pathways for reductive synthesis and energy storage. Green Chem. 2017, 19, 2347–2355. [Google Scholar] [CrossRef]
- Nadalutti, C.A.; Prasad, R.; Wilson, S.H. Perspectives on formaldehyde dysregulation: Mitochondrial DNA damage and repair in mammalian cells. DNA Repair 2021, 105, 103134. [Google Scholar] [CrossRef] [PubMed]
- Tong, Z.; Han, C.; Luo, W.; Wang, X.; Li, H.; Luo, H.; Zhou, J.; Qi, J.; He, R. Accumulated hippocampal formaldehyde induces age−dependent memory decline. Age 2013, 35, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Fei, X.; Zhang, Y.; Mei, Y.; Yue, X.; Jiang, W.; Ai, L.; Yu, Y.; Luo, H.; Li, H.; Luo, W.; et al. Degradation of FA reduces Aβ neurotoxicity and Alzheimer−related phenotypes. Mol. Psychiatry 2021, 26, 5578–5591. [Google Scholar] [CrossRef]
- Tan, T.; Zhang, Y.; Luo, W.; Lv, J.; Han, C.; Hamlin, J.N.R.; Luo, H.; Li, H.; Wan, Y.; Yang, X.; et al. Formaldehyde induces diabetes−associated cognitive impairments. FASEB J. 2018, 32, 3669–3679. [Google Scholar] [CrossRef] [PubMed]
- McGwin, G.; Lienert, J.; Kennedy, J.I. Formaldehyde exposure and asthma in children: A systematic review. Environ. Health Perspect. 2010, 118, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Pontel, L.B.; Rosado, I.V.; Burgos−Barragan, G.; Garaycoechea, J.I.; Yu, R.; Arends, M.J.; Chandrasekaran, G.; Broecker, V.; Wei, W.; Liu, L.; et al. Endogenous Formaldehyde Is a Hematopoietic Stem Cell Genotoxin and Metabolic Carcinogen. Mol. Cell 2015, 60, 177–188. [Google Scholar] [CrossRef]
- Jin, T.X.; Cui, M.Y.; Wu, D.; Zhu, W.P.; Xu, Y.F.; Qian, X.H. NCL−based mitochondrial−targeting fluorescent probe for the detection of Glutathione in living cells. Chin. Chem. Lett. 2021, 32, 3899–3902. [Google Scholar] [CrossRef]
- Fang, Z.; Su, Z.; Qin, W.; Li, H.; Fang, B.; Du, W.; Wu, Q.; Peng, B.; Li, P.; Yu, H.; et al. Two−photon dual−channel fluorogenic probe for in situ imaging the mitochondrial H2S/viscosity in the brain of drosophila Parkinson’s disease model. Chin. Chem. Lett. 2020, 31, 2903–2908. [Google Scholar] [CrossRef]
- Jin, Q.; Ma, H.; Feng, L.; Wang, P.; He, R.; Ning, J.; Yang, L.; Ge, G. Sensing cytochrome P450 1A1 activity by a resorufin−based isoform−specific fluorescent probe. Chin. Chem. Lett. 2020, 31, 2945–2949. [Google Scholar] [CrossRef]
- Zhou, N.; Huo, F.; Yue, Y.; Ma, K.; Yin, C. Rearrangement regulated cysteine fluorescent probe for cellular oxidative stress evaluation induced by copper(II). Chin. Chem. Lett. 2020, 31, 2970–2974. [Google Scholar] [CrossRef]
- Fang, Q.; Yang, L.; Xiong, H.; Han, S.; Zhang, Y.; Wang, J.; Chen, W.; Song, X. Coumarinocoumarin−based fluorescent probe for the sensitive and selective detection of hydrazine in living cells and zebra fish. Chin. Chem. Lett. 2020, 31, 129–132. [Google Scholar] [CrossRef]
- Feng, L.; Wang, P.; Feng, Y.; Zhang, J.; Chen, Q.; Xie, Y.; Luo, J.; Xia, J.; Yao, S.; Sun, H. A reversible microarray immobilization strategy based on thiol−quinone reaction. Chin. Chem. Lett. 2022, 33, 213–216. [Google Scholar] [CrossRef]
- Feng, L.; Chhabra, M.; So, W.H.; Zhu, Q.; Xia, J.; Sun, H. A proximity−induced covalent fluorescent probe for selective detection of bromodomain 4. Chin. Chem. Lett. 2018, 29, 1147–1150. [Google Scholar] [CrossRef]
- Noor Aini, B.; Siddiquee, S.; Ampon, K. Development of formaldehyde biosensor for determination of formalin in fish samples; malabar red snapper (Lutjanus malabaricus) and longtail tuna (Thunnus tonggol). Biosensors 2016, 6, 32. [Google Scholar] [CrossRef] [PubMed]
- Lyu, J.; Wang, C.; Zhang, X. Rational Construction of a mitochondria−targeted reversible fluorescent probe with intramolecular FRET for ratiometric monitoring sulfur dioxide and formaldehyde. Biosensors 2022, 12, 715. [Google Scholar] [CrossRef]
- Brewer, T.F.; Chang, C.J. An aza−Cope reactivity−based fluorescent probe for imaging formaldehyde in living cells. J. Am. Chem. Soc. 2015, 137, 10886–10889. [Google Scholar] [CrossRef]
- Roth, A.; Li, H.; Anorma, C.; Chan, J. A reaction−based fluorescent probe for imaging of formaldehyde in living cells. J. Am. Chem. Soc. 2015, 137, 10890–10893. [Google Scholar] [CrossRef]
- Song, H.; Rajendiran, S.; Kim, N.; Jeong, S.K.; Koo, E.; Park, G.; Thangadurai, T.D.; Yoon, S. A tailor designed fluorescent ‘turn−on’ sensor of formaldehyde based on the BODIPY motif. Tetrahedron Lett. 2012, 53, 4913–4916. [Google Scholar] [CrossRef]
- Tang, Y.; Kong, X.; Xu, A.; Dong, B.; Lin, W. Development of a two−photon fluorescent probe for imaging of endogenous formaldehyde in living tissues. Angew. Chem. Int. Ed. 2016, 55, 3356–3359. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Shao, C.; Wang, X.; Gu, J.; Zhu, H.; Qian, Y. Imaging of formaldehyde fluxes in epileptic brains with a two−photon fluorescence probe. Chem. Commun. 2020, 56, 3871–3874. [Google Scholar] [CrossRef]
- He, L.; Yang, X.; Liu, Y.; Kong, X.; Lin, W. A ratiometric fluorescent formaldehyde probe for bioimaging applications. Chem. Commun. 2016, 52, 4029–4032. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Zhang, Y.; Huang, M.; Wang, S.; Wang, J.; Liao, K.; Wu, X.; Zhou, Q.; Zhang, X.; Wu, Y.D.; et al. Systematic investigation of the aza−Cope reaction for fluorescence imaging of formaldehyde in vitro and in vivo. Chem. Sci. 2021, 12, 13857–13869. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Du, Y.; Li, M.; Zhang, D.; Xiang, Z.; Peng, T. Activity−based genetically encoded fluorescent and luminescent probes for detecting formaldehyde in living cells. Angew. Chem. Int. Ed. 2020, 59, 16352–16356. [Google Scholar] [CrossRef]
- Huang, S.; Li, Z.; Liu, M.; Zhou, M.; Weng, J.; He, Y.; Jiang, Y.; Zhang, H.; Sun, H. Reaction−based fluorescent and chemiluminescent probes for formaldehyde detection and imaging. Chem. Commun. 2022, 58, 1442–1453. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, J.; Hu, L.; Tan, Y.; Liu, S.; Yin, J. Recent advances in formaldehyde−responsive fluorescent probes. Chin. Chem. Lett. 2017, 28, 1935–1942. [Google Scholar] [CrossRef]
- Wang, H.; Wei, J.; Zhang, C.; Zhang, Y.; Zhang, Y.; Li, L.; Yu, C.; Zhang, P.; Chen, J. Red carbon dots as label−free two−photon fluorescent nanoprobes for imaging of formaldehyde in living cells and zebrafishes. Chin. Chem. Lett. 2020, 31, 759–763. [Google Scholar] [CrossRef]
- Singha, S.; Jun, Y.W.; Bae, J.; Ahn, K.H. Ratiometric imaging of tissue by two−photon microscopy: Observation of a high level of formaldehyde around mouse intestinal crypts. Anal. Chem. 2017, 89, 3724–3731. [Google Scholar] [CrossRef]
- Kuznetsov, N.Y.; Tikhov, R.M.; Strelkova, T.V.; Bubnov, Y.N. Adducts of triallylborane with ammonia and aliphatic amines as stoichiometric allylating agents for aminoallylation reaction of carbonyl compounds, Org. Lett. 2018, 20, 3549–3552. [Google Scholar]
- Li, Z.; Huang, S.; He, Y.; Duan, Q.; Zheng, G.; Jiang, Y.; Cai, L.; Jia, Y.; Zhang, H.; Ho, D. AND logic gate based fluorescence probe for simultaneous detection of peroxynitrite and hypochlorous acid, Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 230, 118073. [Google Scholar] [CrossRef]
- Abo, M.; Urano, Y.; Hanaoka, K.; Terai, T.; Komatsu, T.; Nagano, T. Development of a highly sensitive fluorescence probe for hydrogen peroxide, J. Am. Chem. Soc. 2011, 133, 10629–10637. [Google Scholar] [CrossRef]
- Zheng, G.; Li, Z.; Duan, Q.; Cheng, K.; He, Y.; Huang, S.; Zhang, H.; Jiang, Y.; Jia, Y.; Sun, H. Two quenching groups are better than one: A robust strategy for constructing HOCl fluorescent probe with minimized background fluorescence and ultra−high sensitivity and its application of HOCl imaging in living cells and tissues, Sens. Actuators B Chem. 2020, 310, 127890. [Google Scholar] [CrossRef]
- Li, J.; Wang, Q.; Yuan, L.; Wu, Y.; Hu, X.; Zhang, X.; Tan, W. A two−photon fluorescent probe for bio−imaging of formaldehyde in living cells and tissues. Analyst 2016, 141, 3395–3402. [Google Scholar] [CrossRef]
- Dou, K.; Chen, G.; Yu, F.; Liu, Y.; Chen, L.; Cao, Z.; Chen, T.; Li, Y.; You, J. Bright and sensitive ratiometric fluorescent probe enabling endogenous FA imaging and mechanistic exploration of indirect oxidative damage due to FA in various living systems. Chem. Sci. 2017, 8, 7851–7861. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Jia, Y.; Zou, G.; Yu, Y.; Wang, J. A ratiometric fluorescent nanoprobe based on naphthalimide derivative−functionalized carbon dots for imaging lysosomal formaldehyde in HeLa cells. Nanoscale 2019, 11, 6377–6383. [Google Scholar] [CrossRef]
- Zhu, R.; Zhang, G.; Jing, M.; Han, Y.; Li, J.; Zhao, J.; Li, Y.; Chen, P.R. Genetically encoded formaldehyde sensors inspired by a protein intra−helical crosslinking reaction. Nat. Commun. 2021, 12, 581. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Y.; Huang, S.; Liu, M.; Li, Z.; Xiao, W.; Zhang, H.; Yang, L.; Sun, H. Systematic Screening of Trigger Moieties for Designing Formaldehyde Fluorescent Probes and Application in Live Cell Imaging. Biosensors 2022, 12, 855. https://doi.org/10.3390/bios12100855
Jiang Y, Huang S, Liu M, Li Z, Xiao W, Zhang H, Yang L, Sun H. Systematic Screening of Trigger Moieties for Designing Formaldehyde Fluorescent Probes and Application in Live Cell Imaging. Biosensors. 2022; 12(10):855. https://doi.org/10.3390/bios12100855
Chicago/Turabian StyleJiang, Yin, Shumei Huang, Minghui Liu, Zejun Li, Weimin Xiao, Huatang Zhang, Liu Yang, and Hongyan Sun. 2022. "Systematic Screening of Trigger Moieties for Designing Formaldehyde Fluorescent Probes and Application in Live Cell Imaging" Biosensors 12, no. 10: 855. https://doi.org/10.3390/bios12100855
APA StyleJiang, Y., Huang, S., Liu, M., Li, Z., Xiao, W., Zhang, H., Yang, L., & Sun, H. (2022). Systematic Screening of Trigger Moieties for Designing Formaldehyde Fluorescent Probes and Application in Live Cell Imaging. Biosensors, 12(10), 855. https://doi.org/10.3390/bios12100855



